Abstract
Formation of myelin sheaths by Schwann cells (SCs) enables rapid and efficient transmission of action potentials in peripheral axons, and disruption of myelination results in disorders that involve decreased sensory and motor functions. Given that construction of SC myelin requires high levels of lipid and protein synthesis, mitochondria, which are pivotal in cellular metabolism, may be potential regulators of the formation and maintenance of SC myelin. Supporting this notion, abnormal mitochondria are found in SCs of neuropathic peripheral nerves in both human patients and the relevant animal models. However, evidence for the importance of SC mitochondria in myelination has been limited, until recently. Several studies have recently used genetic approaches that allow SC-specific ablation of mitochondrial metabolic activity in living animals to show the critical roles of SC mitochondria in the development and maintenance of peripheral nerve axons. Here, we review current knowledge about the involvement of SC mitochondria in the formation and dysfunction of myelinated axons in the peripheral nervous system.
Keywords: Schwann cell, Myelin sheath, Axon, Mitochondria, Peripheral neuropathy
Introduction
Schwann cells (SCs), the principal glial cell of the peripheral nervous system (PNS), form spiral membrane insulators known as myelin sheaths around axons, which enables rapid and efficient transmission of sensory and motor information by saltatory conduction [1]. A differentiated form of SCs, called myelinating SCs, extend their plasma membranes to construct multilamellar wrappings around axons during development, and the structure of this myelin sheath is maintained after development in healthy nerves. If the maintenance of myelin sheaths is disturbed under various conditions, such as nerve damage, myelinating SCs lose their myelin sheaths and assume a dedifferentiated state. The dedifferentiated SCs show characteristics similar to those of immature SCs during development and have a capacity to initiate remyelination [2, 3]. Therefore, the remyelination process can be viewed as a recapitulation of the developmental myelination. Because dedifferentiated SCs are observed in nerves damaged by demyelinating diseases [2], understanding the mechanisms of SC differentiation and maintenance will provide insight into how demyelinating diseases progress, and will potentially contribute to the development of new therapeutics.
Mitochondria play diverse roles (e.g., energy production, lipid and amino acid metabolism, and regulation of cell death) in both physiological and pathological states in eukaryotic cells. Defects in mitochondrial metabolism are one of the common features of neurodegenerative diseases and are thought to be the key regulators of peripheral neuropathic symptoms [4–6]. Structurally or functionally abnormal mitochondria are often found in SCs of demyelinating peripheral nerves injured in disorders such as mitochondrial diseases, Charcot–Marie–Tooth disease, and diabetes [7–9]. Therefore, mitochondria may play key roles in the differentiation and maintenance of myelinating SCs, and mitochondrial abnormalities may contribute to disease progression in peripheral nerves. Supporting this idea, recent studies using animal models with decreased mitochondrial metabolism in myelinating SCs have provided evidence for a critical role of SC mitochondrial metabolism in the development and maintenance of myelinated axons [10–12].
In this topical review, we first briefly introduce the current knowledge on the diseases that involve abnormal mitochondria in SCs. We then summarize the recent findings from animal models that show the involvement of SC mitochondria in peripheral nerve disorders. Finally, we describe more recent findings that have revealed the critical mechanisms regulating mitochondrial metabolism during the development and maintenance of myelinating SCs [13–15], defects of which may be key pathological contributors to disease progression in peripheral nerves. We offer apologies to the many authors whose work could not be discussed here due to the space limitation.
Abnormal SC mitochondria in peripheral neuropathies
Mitochondrial dysfunction is a likely cause of peripheral neuropathy [5, 6, 16]. Although researchers have focused on the role of axonal mitochondria in the progression of peripheral neuropathy in light of the high energy demand of neurons, findings also support the idea that abnormal SC mitochondria participate in disease progression. Before discussing this further, however, we will first summarize the peripheral neuropathies in which abnormal mitochondria are found in SCs.
Mitochondrial diseases
Mitochondrial diseases result from structurally, numerically, or functionally abnormal mitochondria [5, 6]. In patients with mitochondrial diseases, mitochondrial abnormalities are caused primarily by mutations in mitochondrial DNA (mtDNA) [6], which encodes 13 proteins of the electron transport system, 22 transfer RNAs, and 2 ribosomal DNAs. Mutations in nuclear DNA encoding essential proteins for mitochondrial function can also be the cause of mitochondrial defects. In patients with mitochondrial diseases, abnormalities are frequently found in the nervous system, striated muscles, or both, in which a high density of mitochondria exist, and approximately one-third of these patients develop peripheral neuropathic phenotypes involving sensory and motor dysfunctions in the context of systemic disorders [16]. In particular, defects in PNS myelinated axons are often found in patients with diseases associated with mutations of mtDNA, such as chronic progressive external ophthalmoplegia, mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes, and myoclonus epilepsy with ragged-red fibers [16]. Peripheral nerves affected by such diseases show reduced conduction velocity [17, 18] and decreased myelinated axon density [7, 8, 17–19]. Interestingly, electron microscopic analyses of tissue samples obtained from those neuropathic patients showed that structurally abnormal mitochondria (e.g., swollen mitochondria, mitochondria with distorted cristae, and mitochondria with paracrystalline inclusions) are found primarily in SCs but not in axons [7, 8, 17–19]. Given that mitochondrial structure is a key determinant of mitochondrial function [20], these findings suggest a potential involvement of abnormal mitochondria in SCs in the development of the neuropathic phenotype in mitochondrial diseases. Although how defects in mitochondrial diseases are caused by abnormal mitochondrial functions remains largely unknown, reduction of energy and metabolic supply or excess generation of toxic agents, such as reactive oxygen species (ROS) and lactic acid, are considered to be causally linked. Indeed, compounds that support mitochondrial metabolism (coenzyme Q10, riboflavin, thiamine, carnitine, and creatine), that work as antioxidants (coenzyme Q10, vitamin C, and vitamin E), or that improve lactic acidosis (dichloroacetate) are used for the treatment of mitochondrial diseases [21].
Charcot–Marie–Tooth diseases
Charcot–Marie–Tooth disease (CMT) is the most common hereditary peripheral neuropathy, with a prevalence of about 40 in 100,000 people [9]. CMT is organized into two groups: (1) the demyelinating form (CMT1, CMT3, and CMT4), associated with decreased conduction velocity, and segmental demyelination and remyelination of SCs; (2) the axonal form (CMT2), characterized by preserved or slightly reduced conduction velocity, and axonal loss without demyelination. More than 40 loci and about 30 genes, which are associated with CMT, have been identified to date, and several genes encode mitochondrial proteins. The ganglioside-induced differentiation associated protein 1 gene (GDAP1), mutations of which are found in CMT4A (recessive, demyelinating form) and CMT2K (dominant, axonal form), encodes a mitochondrial outer membrane protein that has a key role in mitochondrial fission [22]. Indeed, overexpression of GDAP1 promotes fragmentation of mitochondria, whereas either knockdown of GDAP1 or overexpression of a disease-related mutant of GDAP1 causes mitochondria to assume an elongated structure in vitro [22]. Although most genes associated with mitochondria-related CMT are mainly expressed in neurons, GDAP1 is expressed both in myelinating SCs and neurons [22]. Niemann and colleagues have recently shown that myelinating SC-specific Gdap1 deletion in mice results in decreased conduction velocity and hypomyelination in the sciatic nerve, whereas motor neuron-specific Gdap1 deletion does not affect conduction velocity and the structure of myelin [23], suggesting that SC mitochondria without normal GDAP1 activity are likely to be involved in the progression of peripheral neuropathy. However, how mutant GDAP1 causes the neuropathy is not well understood. Given that mitochondrial structure has a key role in the regulation of cellular metabolism [24, 25], defects in GDAP1-mediated mitochondrial fission may result in the abnormal metabolic state in SCs.
Diabetes
Diabetes is a huge health problem worldwide, and, according to a report from the World Health Organization, about 9 % of adults (18 years and older) were diagnosed with diabetes in 2014 (http://www.who.int/mediacentre/factsheets/fs312/en/). One of the major complications of diabetes is diabetic peripheral neuropathy, which involves axonal degeneration, demyelination, and loss of both myelinated and unmyelinated fibers [26]. A potential key player in the progression of diabetic neuropathy is abnormal mitochondria [26, 27]. Interestingly, an anatomical analysis has shown that structurally abnormal mitochondria (swollen mitochondria with effaced cristae) are preferentially found in SCs in the peripheral nerves of both human patients with diabetes and animal models of diabetes [28, 29]. Furthermore, chronic exposure of cells to high levels of glucose causes dramatic changes in the mitochondrial proteome, which lead to decreased mitochondrial metabolic activity, in various cell types [27], including SCs [30]. These results support the notion that SC mitochondria are involved in the development of diabetic peripheral neuropathy. Although the detailed mechanism of diabetic neuropathy progression remains to be further explored, generation of ROS by abnormal mitochondria may be a key process for the progression of diabetic neuropathy, because administration of α-lipoic acid, an antioxidant, significantly improves neuropathic symptoms and deficits in diabetic patients [31].
Mouse models of SC-specific mitochondrial metabolic defects
As described above, the presence of abnormal mitochondria in SCs in diseased peripheral nerves implicates the potential involvement of SC mitochondria in the development and maintenance of myelinated axons. However, it is difficult to know whether mitochondrial abnormalities are the direct cause of disease progression in human patients. Thus, to gain the insight into the roles of SC mitochondria, research using animal models is important. Recent studies using mutant mice in which mitochondrial molecules indispensable for mitochondrial oxidative respiration were disrupted selectively in myelinating SCs have provided evidence for the critical roles of SC mitochondria in the development and maintenance of myelinated axons [10–12].
Cox10 knockout mice
COX10 encodes heme A:farnesyl transferase, an enzyme for the biosynthesis of heme A, which is essential for cytochrome c oxidase integrity, and mutations of COX10 have been found in patients with mitochondrial diseases who experience systemic disorders accompanying demyelinating peripheral neuropathy [32]. Conditional deletion of the Cox10 gene in various cell types such as skeletal muscle cells [33], hepatocytes [34], and neuronal cells [35] in mice results in the loss of mitochondrial respiration, and the phenotype of these mutant mice recapitulates several symptoms of human diseases. SC-specific Cox10 deletion decreases the number of functional mitochondria and increases swollen mitochondria in SCs [11]. SC-specific Cox10-knockout (KO) mice (Cox10-SCKO mice) show hypomyelination and reduced conduction velocity as early as postnatal day 21 (P21) [11]. Cox10 elimination in SCs does not cause SC death and inflammatory response in the peripheral nerves. These results suggest the importance of mitochondrial oxidative phosphorylation in the development of myelin.
Note that deletion of the Cox10 gene in oligodendrocytes, myelinating glia in the central nervous system, does not cause detectable defects. The differential effects could be explained as follows: (1) elimination of Cox10 results in gradual loss of mitochondrial respiration in myelinating oligodendrocytes because of slow mitochondrial turnover (t 1/2 ≈ 3 weeks) in the brain, whereas intact mitochondria in proliferating Schwann cells are more rapidly lost by dilution in the progeny, (2) increased glycolysis likely compensates for the mitochondrial metabolic defects in mature oligodendrocytes [11]. Thus, whether mitochondrial metabolic activity in oligodendrocytes is involved in myelin development and maintenance requires further clarification.
Tfam knockout mice
Mitochondrial transcription factor A (Tfam) is an essential protein for the transcription and replication of mtDNA, and deletion of the Tfam gene results in the absence of mtDNA, which leads to the loss of mitochondrial respiration. Thus, Tfam-KO mice have been used as a model of mitochondrial diseases [36–39]. Viader and colleagues reported that SC-specific Tfam-KO mice (Tfam-SCKO mice) show abnormalities in peripheral nerves [10, 12], but the phenotype is different from that of Cox10-SCKO mice. Tfam-SCKO mice show reduced nerve conduction velocity and defects in sensory and motor functions. As is also observed in the nerves of humans with mitochondrial diseases, abnormal mitochondria are found in Tfam-null SCs. Interestingly, the peripheral nerves of Tfam-SCKO mice show not only demyelination but also axonal degeneration both in myelinated and unmyelinated axons. This suggests that Tfam-null SCs have deleterious effects on axons in a non-cell-autonomous manner. Deletion of Tfam in SCs results in the activation of the integrated stress response (ISR) [12], a signaling mechanism that is commonly activated when eukaryotic cells are exposed to a stressful environment [40]. The induction of ISR in SCs switches lipid metabolism from lipid synthesis to lipid oxidation, which results in depletion of glycosphingolipids, such as cerebrosides and sulfatides, which are important for myelin maintenance. Thus, this is a likely cause of demyelination. Furthermore, accumulation of acylcarnitines has also been observed in SCs as a consequence of abnormal lipid metabolism [12]. Because chronic application of palmitoylcarnitine, an acylcarnitine species that is markedly increased in the nerves of Tfam-SCKO mice, results in degeneration of axons of dorsal root ganglion neurons in vitro, release of acylcarnitines from SCs is likely to cause axonal damage in Tfam-SCKO mice [12].
Critical signals for mitochondrial metabolism in myelinating SCs
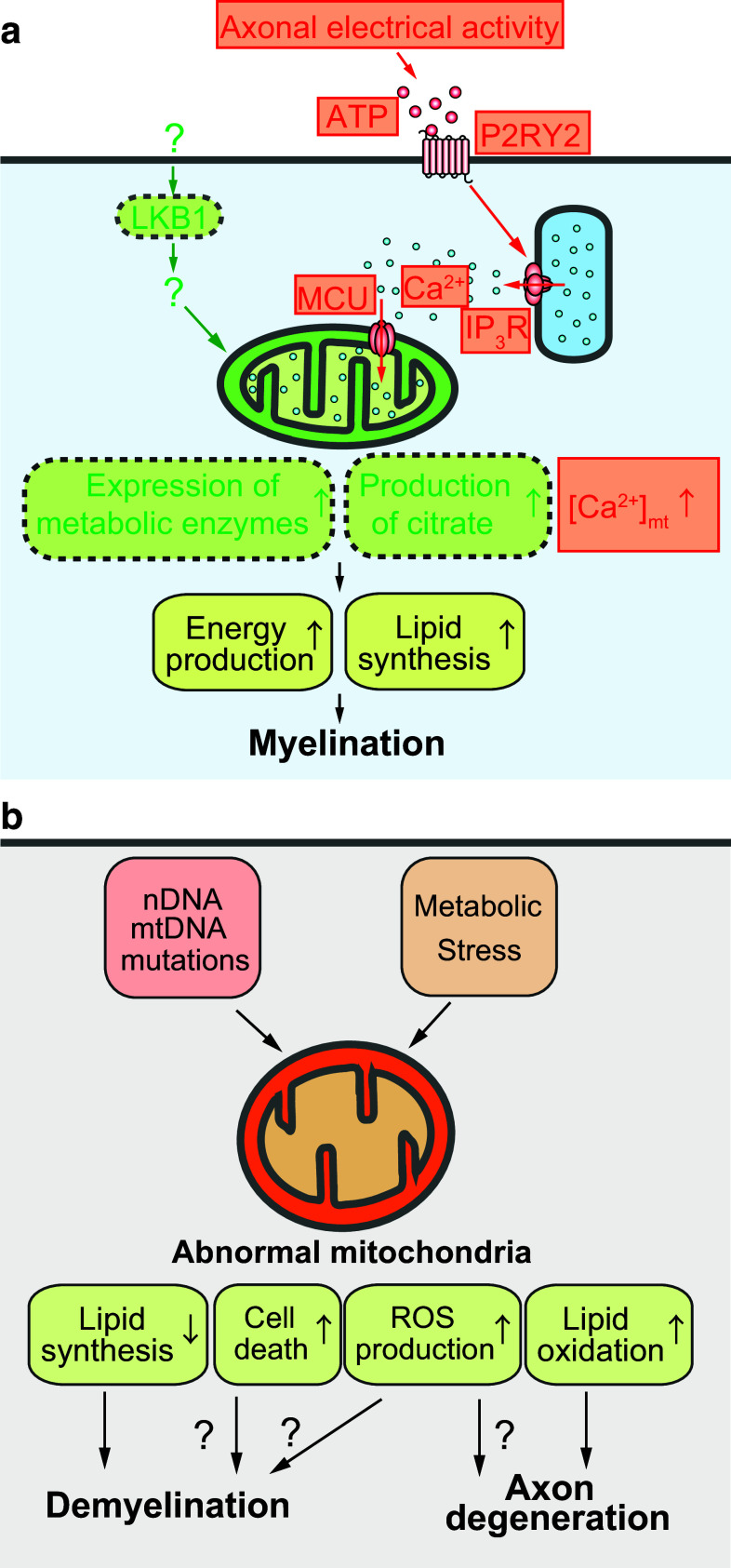
Mitochondrial metabolism can be controlled by a variety of intracellular signaling mechanisms, depending on the cellular metabolic demand [41]. Considering that the development and maintenance of the myelin sheath are metabolically demanding, those signaling mechanisms may also control mitochondrial functions in SCs. Recently, two key signals, Lkb1 [13, 14] and mitochondrial Ca2+ signals [15], have been identified as critical regulators for mitochondrial metabolism in myelinating SCs (Fig. 1a).
Fig. 1.
Schematic diagram depicting how SC mitochondria are involved in the myelination (a) and pathogenesis of peripheral axons (b). a Signaling that regulates mitochondrial metabolism in SCs during myelination. Lkb1 plays a key role in the upregulation of mitochondrial metabolic enzymes and production of citrate, an important metabolite for mitochondrial energy production and lipid synthesis. Upstream and downstream regulators of Lkb1 in SC have not been well characterized. However, stimulation of P2RY2, a Gq-coupled receptor, by ATP released from firing axons increases the cytoplasmic Ca2+ concentration via IP3 receptor-mediated Ca2+ release from intracellular Ca2+ stores. Then, Ca2+ is delivered to the mitochondrial matrix via the MCU. An increase in the Ca2+ concentration within the mitochondrial matrix ([Ca2+]mt) stimulates mitochondrial metabolic activity. These signaling inputs enhance energy production and lipid synthesis in myelinating SCs. b Potential pathogenic mechanisms of abnormal SC mitochondria in demyelination and axon degeneration. Abnormalities in mitochondrial metabolism are caused by various factors, such as mutations in nuclear DNA or mtDNA and metabolic stress (e.g., hyperglycemia). Abnormalities of mitochondrial metabolism can cause adverse effects in SCs, including remodeling of lipid metabolism (decreased lipid synthesis and increased lipid oxidation), production of excessive ROS, and sensitization to cell death. Under these conditions, SCs may undergo demyelination, cause axonal degeneration, or both
LKB1
The serine-threonine liver kinase B1 (LKB1) was originally discovered as a tumor suppressor gene, and is now recognized to be a master kinase for metabolic regulation [42]. One of the major substrates of LKB1 is AMP-activated protein kinase (AMPK), an evolutionarily conserved metabolic sensor kinase that is activated in response to intracellular ATP deprivation (i.e., enhanced AMP levels) [43]. AMPK is a heterotrimeric complex composed of a catalytic α subunit and regulatory β and γ subunits. Binding of AMP to the γ subunit induces a conformational change that makes the α subunit more sensitive to LKB1-mediated phosphorylation. The phosphorylated AMPK controls the activity and expression of the enzymes for glucose and lipid metabolism. LKB1 also phosphorylates multiple kinases, which regulate cell growth, metabolism, and cell polarity in an AMPK-independent manner. Because the activity of AMPK is suppressed under hyperglycemic conditions [42], defects in LKB1-mediated signaling may be related to diabetic peripheral neuropathy.
Recent reports have suggested an important role of LKB1 in SC mitochondrial metabolism [13, 14]. Although mice lacking the Lkb1 gene in SCs (Lkb1-SCKO mice) show neuropathic phenotypes, the phenotypes of Lkb1-SCKO mice reported by two independent groups are slightly different. Pooya et al. generated Lkb1-SCKO mice using a Dhh-Cre driver [14], in which Cre expression begins around embryonic day (E) 12.5, which corresponds to the SC precursor stage [44]. These Lkb1-SCKO mice show hypomyelination and begin to exhibit a neuropathic phenotype as early as around P30, and the phenotype continues thereafter. Pooya et al. investigated how Lkb1 deletion affects SC myelination, and discovered that expression levels of proteins important for mitochondrial metabolism significantly increase in SCs after the initiation of myelination [14]. However, this upregulation of mitochondrial proteins is suppressed in the Lkb1-SCKO mice, while expression levels of proteins involved in myelin construction (protein zero, peripheral myelin protein 22, and myelin basic protein) and Krox20, an essential transcription factor for their expression, are unaltered. In line with this result, mitochondrial oxygen consumption of sciatic nerves in the Lkb1-SCKO mice is significantly decreased compared with that in wild-type animals. These findings suggest that LKB1 has a key role in the maturation of mitochondrial metabolic activity in myelinating SCs. How, then, does decreased mitochondrial metabolism in the Lkb1-mutant mice cause hypomyelination? Pooya et al. found that the Lkb1-null SCs show decreased production of citrate, a metabolite of the tricarboxylic acid (TCA) cycle important for mitochondrial energy production and lipid synthesis [14]. Overexpression of citrate synthase rescues the defects in differentiation and myelination in primary cultured Lkb1-null SCs. Therefore, a decreased amount of citrate, which leads to energy depletion and decreased lipid synthesis, is a likely cause of hypomyelination in the Lkb1-SCKO mice. In addition to its role in the control of mitochondrial metabolic activity, Lkb1 also has a key role in the regulation of radial (i.e. adaxonal-to-abaxonal) SC polarity [45], which is important for the initiation of myelination [46]. Whether a link exists between LKB1-mediated metabolic control and polarity regulation remains to be elucidated.
Conversely, Beirowski et al. generated Lkb1-SCKO mice using a P0-Cre driver [13], in which Cre starts to be expressed between E13.5 and 14.5, which corresponds to the immature SC stage [47]. These Lkb1-SCKO mice show hypomyelination at P14, which recovers by P30. This is likely to be due to the compensatory effects of elevated neuregulin-erbB signaling, a signaling axis critical for SC myelination [48]. At 3 months of age, the mutant mice start to show degeneration of myelinated and unmyelinated axons and also to exhibit the neuropathic phenotype, and most of the unmyelinated axons are lost by 12–18 months of age. The axonal loss in the Lkb1-SCKO mice is not due to SC developmental defects because similar axonal loss is observed when the Lkb1 gene in SCs is deleted using a tamoxifen-inducible conditional KO system after the completion of myelination. Although the mechanism of axonal loss in the Lkb1-SCKO mice requires further clarification, Lkb1-mediated metabolic regulation in SCs is likely to be important for axonal maintenance. Interestingly, preferential loss of small unmyelinated fibers in the Lkb1-SCKO mice is similar to the features of peripheral nerves injured in diabetic neuropathy. Thus, understanding the mechanism of axonal loss in the Lkb1-SCKO mice may inform therapies for diabetic neuropathic nerves.
Mitochondrial Ca2+ signaling
One of the classic signaling mechanisms in mitochondrial metabolism is an increase in the Ca2+ concentration of the mitochondrial matrix [49]. Ca2+ uptake by energized mitochondria was discovered about half century ago [50, 51]. Subsequently, it has been found that increases in the Ca2+ concentration within mitochondria stimulate mitochondrial metabolism [52, 53]. The main targets of Ca2+ are three enzymes in the TCA cycle: pyruvate dehydrogenase phosphatase, α-ketoglutarate dehydrogenase, and isocitrate dehydrogenase [49, 53]. Ca2+ binding to these enzymes enhances the production of TCA cycle metabolites and nicotinamide adenine dinucleotide, and subsequently drives oxidative phosphorylation in the electron transport system. Ca2+ uptake by mitochondria is mediated by a Ca2+ channel, the mitochondrial Ca2+ uniporter (MCU), which resides on the inner membrane of mitochondria. After extensive study on the basic properties of the MCU, a 40-kDa mitochondrial protein was finally identified in 2011 as a core component of the Ca2+ channel [54, 55].
Ino et al. found that MCU-mediated mitochondrial Ca2+ signaling plays a key role in myelination [15]. The in vitro search for potential ligands that affect mitochondrial metabolism in SCs has suggested that purinergic receptor signaling mediates MCU-dependent mitochondrial metabolism in SCs. In line with this idea, stimulation of the rat sciatic nerve with ATP induces increases in the Ca2+ concentration in both the cytosol and mitochondrial matrix in myelinating SCs. The ATP-induced cytoplasmic Ca2+ increase is mediated by the signaling cascade consisting of the activation of the P2Y2 purinergic receptor (P2RY2), production of inositol 1,4,5-trisphosphate (IP3) via hydrolysis of phosphatidylinositol 4,5-bisphosphate by phospholipase C, and Ca2+ release from intracellular Ca2+ stores via IP3 receptors. This P2Y2-IP3-mediated cytoplasmic Ca2+ mobilization subsequently induces mitochondrial Ca2+ signaling via the MCU.
The physiological significance of mitochondrial Ca2+ signaling in myelinating SCs has been examined in vivo by disrupting the P2RY2-IP3-MCU pathway using shRNA for P2RY2 (shP2RY2), or IP3-5 phosphatase (5ppase), an enzyme that suppress IP3 receptor-mediated Ca2+ release by rapidly hydrolyzing IP3 [56–62], or shRNA for MCU (shMCU). Expression of shP2RY2, 5ppase, or shMCU in myelinating SCs results in increased levels of phosphorylated AMPK, suggesting a critical role for P2RY2-IP3-MCU signaling in energy metabolism in myelinating SCs. Inhibition of P2RY2-IP3-MCU signaling does not cause SC death, excluding the involvement of this pathway in cell death. Silencing of P2RY2-IP3-MCU signaling for 4 weeks starting on P3, when myelination initiates in rodents, results in decreased longitudinal and radial development as well as hypomyelination in myelinating SCs. Similar results are observed when P2RY2-IP3-MCU is silenced during the period of active myelination (from P3 to P14); by contrast, no significant changes are observed when P2RY2-IP3-MCU is silenced after the period of active myelination (from P14 to P31). These findings suggest that mitochondrial Ca2+ signaling mediated by P2RY2-IP3-MCU pathway has a key role in the process of myelin development rather than in myelin maintenance [15].
How, then, is the P2RY2-IP3-MCU pathway activated in SCs in vivo? Most developmental processes in SCs, such as proliferation, survival, and differentiation, are regulated by signaling from axons [48]. Thus, it seems plausible that the P2RY2-IP3-MCU pathway in SC myelination is regulated by axons. Electrically active PNS axons release ATP through maxi-anion channels [63], which have a 1.3-nm-radius open pore through which ATP (which has a radius of 0.6 nm) can permeate [64]. Thus, Ino et al. examined whether purinergic receptor-mediated mitochondrial Ca2+ signaling is controlled by activity-dependent axonal signaling [15]. The results of microdialysis analysis in neonatal rat sciatic nerve have shown that electrical stimulation with a paradigm relevant to the firing pattern in response to sensory stimulation enhances the extracellular ATP level to approximately 0.5 μM [15], which is sufficient to activate most P2 purinergic receptors [65, 66]. Indeed, electrical stimulation with the same paradigm induces a reversible Ca2+ increase in both the cytosol and mitochondrial matrix in myelinating SCs. This suggests that Ca2+ levels within mitochondria, which control the activity of mitochondrial metabolism, in SCs juxtaposed to firing axons may be kept high. Considering that the firing rate of afferent sensory nerves markedly increases during the early postnatal period in rodents [67], during which SCs actively undergo myelination, axons may stimulate mitochondrial metabolism activity in SCs to meet the high metabolic demand of myelination. Whether this mechanism is functioning in living animals is an important issue to be examined in the future.
In addition to regulating cellular metabolism, mitochondria also control various cellular functions in a Ca2+-dependent manner [49]. In particular, excessive Ca2+ accumulation within the mitochondrial matrix, which is induced by Ca2+ overload in the cytosol, results in severe cellular damage. In the central nervous system, it has been proposed that Ca2+ overload via the N-methyl-D-aspartate glutamate receptor [68] or the TRPA1 [69] channel mediates myelin loss during ischemia. Therefore, Ca2+ overload within SCs may also underlie the damaging effects on PNS myelin during disease progression.
Conclusion and perspectives
Recent studies have indicated that SC mitochondria play critical roles in energy production and lipid synthesis during the development and maintenance of myelinated axons. So far, two key signaling mechanisms, Lkb1 and mitochondrial Ca2+, have been shown to be involved in these processes (Fig. 1a). The construction of the myelin sheath is considered to require high levels of lipid and protein synthesis [70]; it has been estimated that as much as 0.5 mol (i.e., about 250 grams) of ATP is required to synthesize 1 g of myelin [71]. Supporting this idea, ablation of signals for SC mitochondrial metabolism impairs myelin sheath formation [13–15]. Although further clarification is required, these mechanisms may contribute to pathological progression in demyelinating diseases.
In addition to their metabolic roles, mitochondria may also cause damaging effects and cell death by generating toxic agents [49], which may also be involved in disease progression. In mutant animals with mitochondrial abnormalities, such as Tfam-SCKO and Lkb1-SCKO mice, gradual onset of demyelination and axonal degeneration is observed [10, 12, 13]. These findings suggest the possibility that accumulation of abnormal mitochondria in SCs can cause damaging effects in both SCs and axons. How do abnormal mitochondria cause demyelination and axonal degeneration? Lipidomic analyses have shown that ablation of mitochondrial metabolism results in decreased synthesis of myelin lipids and cholesterol, which leads to demyelination [72]. These studies have also shown that ablation of mitochondrial metabolism results in accumulation of acylcarnitines, lipid oxidation metabolites that can cause toxic effects in axons. Therefore, altering lipid metabolism from lipid synthesis to lipid oxidation in abnormal SC mitochondria may underlie a key process in gradual-onset damage of peripheral nerves. In addition, increase in the production of ROS, a common risk factor in neurodegenerative diseases [4], has been observed in Lkb1-KO SCs. Because ROS can cause damage in both cell-autonomous and non-cell-autonomous manners [73], ROS may be a potential factor in demyelination and axonal damage. Consistent with this idea, administration of antioxidants can improve neuropathic symptoms in human patients [21, 31]. Furthermore, because abnormal mitochondria also play a critical role in cell death, a key feature of neurodegenerative diseases [74], abnormal mitochondria may also induce SC death and subsequent demyelination. Indeed, apoptotic SCs were found in human peripheral nerves injured in diabetic neuropathy [75], supporting the notion that SC death is a likely cause of demyelination. However, no signs of cell death are found in SCs in which mitochondrial metabolism has been suppressed [10, 11, 13–15]. Thus, additional mechanisms may be involved in the induction of SC death in human diseased nerves.
Taking all these results together, abnormal mitochondria are likely to trigger undesirable cellular responses in SCs, including remodeling of lipid metabolism, excessive production of ROS, and enhanced susceptibility to cell death, which can lead to demyelination, axonal degeneration, or both (Fig. 1b). Because some of the phenotypes of mutant animals with mitochondrial abnormalities are similar to those found in neuropathic peripheral nerves of human patients, the reactions triggered by abnormal mitochondria may underlie the pathogenesis of human peripheral neuropathy. Further accumulation of knowledge about the detailed roles of SC mitochondria may assist in the discovery of new therapeutic targets for peripheral neuropathy.
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
References
- 1.Sherman DL, Brophy PJ. Mechanisms of axon ensheathment and myelin growth. Nat Rev Neurosci. 2005;6(9):683–690. doi: 10.1038/nrn1743. [DOI] [PubMed] [Google Scholar]
- 2.Jessen KR, Mirsky R. Negative regulation of myelination: relevance for development, injury, and demyelinating disease. Glia. 2008;56(14):1552–1565. doi: 10.1002/glia.20761. [DOI] [PubMed] [Google Scholar]
- 3.Decker L, Desmarquet-Trin-Dinh C, Taillebourg E, Ghislain J, Vallat JM, Charnay P. Peripheral myelin maintenance is a dynamic process requiring constant Krox20 expression. J Neurosci. 2006;26(38):9771–9779. doi: 10.1523/JNEUROSCI.0716-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wallace DC. Mitochondrial diseases in man and mouse. Science. 1999;283(5407):1482–1488. doi: 10.1126/science.283.5407.1482. [DOI] [PubMed] [Google Scholar]
- 5.Schapira AH. Mitochondrial disease. Lancet. 2006;368(9529):70–82. doi: 10.1016/S0140-6736(06)68970-8. [DOI] [PubMed] [Google Scholar]
- 6.Schapira AH. Mitochondrial diseases. Lancet. 2012;379(9828):1825–1834. doi: 10.1016/S0140-6736(11)61305-6. [DOI] [PubMed] [Google Scholar]
- 7.Schroder JM, Sommer C. Mitochondrial abnormalities in human sural nerves: fine structural evaluation of cases with mitochondrial myopathy, hereditary and non-hereditary neuropathies, and review of the literature. Acta Neuropathol. 1991;82(6):471–482. doi: 10.1007/BF00293381. [DOI] [PubMed] [Google Scholar]
- 8.Schroder JM. Neuropathy associated with mitochondrial disorders. Brain Pathol. 1993;3(2):177–190. doi: 10.1111/j.1750-3639.1993.tb00742.x. [DOI] [PubMed] [Google Scholar]
- 9.Pareyson D, Marchesi C. Diagnosis, natural history, and management of Charcot–Marie–Tooth disease. Lancet Neurol. 2009;8(7):654–667. doi: 10.1016/S1474-4422(09)70110-3. [DOI] [PubMed] [Google Scholar]
- 10.Viader A, Golden JP, Baloh RH, Schmidt RE, Hunter DA, Milbrandt J. Schwann cell mitochondrial metabolism supports long-term axonal survival and peripheral nerve function. J Neurosci. 2011;31(28):10128–10140. doi: 10.1523/JNEUROSCI.0884-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fünfschilling U, Supplie LM, Mahad D, Boretius S, Saab AS, Edgar J, Brinkmann BG, Kassmann CM, Tzvetanova ID, Mobius W, Diaz F, Meijer D, Suter U, Hamprecht B, Sereda MW, Moraes CT, Frahm J, Goebbels S, Nave KA. Glycolytic oligodendrocytes maintain myelin and long-term axonal integrity. Nature. 2012;485(7399):517–521. doi: 10.1038/nature11007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Viader A, Sasaki Y, Kim S, Strickland A, Workman CS, Yang K, Gross RW, Milbrandt J. Aberrant Schwann cell lipid metabolism linked to mitochondrial deficits leads to axon degeneration and neuropathy. Neuron. 2013;77(5):886–898. doi: 10.1016/j.neuron.2013.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Beirowski B, Babetto E, Golden JP, Chen YJ, Yang K, Gross RW, Patti GJ, Milbrandt J. Metabolic regulator LKB1 is crucial for Schwann cell-mediated axon maintenance. Nat Neurosci. 2014;17(10):1351–1361. doi: 10.1038/nn.3809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pooya S, Liu X, Kumar VB, Anderson J, Imai F, Zhang W, Ciraolo G, Ratner N, Setchell KD, Yoshida Y, Jankowski MP, Dasgupta B. The tumour suppressor LKB1 regulates myelination through mitochondrial metabolism. Nat Commun. 2014;5:4993. doi: 10.1038/ncomms5993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ino D, Sagara H, Suzuki J, Kanemaru K, Okubo Y, Iino M. Neuronal regulation of Schwann cell mitochondrial Ca(2+) signaling during myelination. Cell Rep. 2015;12(12):1951–1959. doi: 10.1016/j.celrep.2015.08.039. [DOI] [PubMed] [Google Scholar]
- 16.Pareyson D, Piscosquito G, Moroni I, Salsano E, Zeviani M. Peripheral neuropathy in mitochondrial disorders. Lancet Neurol. 2013;12(10):1011–1024. doi: 10.1016/S1474-4422(13)70158-3. [DOI] [PubMed] [Google Scholar]
- 17.Chu CC, Huang CC, Fang W, Chu NS, Pang CY, Wei YH. Peripheral neuropathy in mitochondrial encephalomyopathies. Eur Neurol. 1997;37(2):110–115. doi: 10.1159/000117420. [DOI] [PubMed] [Google Scholar]
- 18.Yiannikas C, McLeod JG, Pollard JD, Baverstock J. Peripheral neuropathy associated with mitochondrial myopathy. Ann Neurol. 1986;20(2):249–257. doi: 10.1002/ana.410200211. [DOI] [PubMed] [Google Scholar]
- 19.Santoro L, Carrozzo R, Malandrini A, Piemonte F, Patrono C, Villanova M, Tessa A, Palmeri S, Bertini E, Santorelli FM. A novel SURF1 mutation results in Leigh syndrome with peripheral neuropathy caused by cytochrome c oxidase deficiency. Neuromuscul Disord. 2000;10(6):450–453. doi: 10.1016/S0960-8966(99)00122-4. [DOI] [PubMed] [Google Scholar]
- 20.Detmer SA, Chan DC. Functions and dysfunctions of mitochondrial dynamics. Nat Rev Mol Cell Biol. 2007;8(11):870–879. doi: 10.1038/nrm2275. [DOI] [PubMed] [Google Scholar]
- 21.Parikh S, Saneto R, Falk MJ, Anselm I, Cohen BH, Haas R, Medicine Society TM A modern approach to the treatment of mitochondrial disease. Curr Treat Options Neurol. 2009;11(6):414–430. doi: 10.1007/s11940-009-0046-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Niemann A, Ruegg M, La Padula V, Schenone A, Suter U. Ganglioside-induced differentiation associated protein 1 is a regulator of the mitochondrial network: new implications for Charcot–Marie–Tooth disease. J Cell Biol. 2005;170(7):1067–1078. doi: 10.1083/jcb.200507087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Niemann A, Huber N, Wagner KM, Somandin C, Horn M, Lebrun-Julien F, Angst B, Pereira JA, Halfter H, Welzl H, Feltri ML, Wrabetz L, Young P, Wessig C, Toyka KV, Suter U. The Gdap1 knockout mouse mechanistically links redox control to Charcot–Marie–Tooth disease. Brain. 2014;137(Pt 3):668–682. doi: 10.1093/brain/awt371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Picard M, Shirihai OS, Gentil BJ, Burelle Y. Mitochondrial morphology transitions and functions: implications for retrograde signaling? Am J Physiol Regul Integr Comp Physiol. 2013;304(6):R393–R406. doi: 10.1152/ajpregu.00584.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Westermann B. Mitochondrial fusion and fission in cell life and death. Nat Rev Mol Cell Biol. 2010;11(12):872–884. doi: 10.1038/nrm3013. [DOI] [PubMed] [Google Scholar]
- 26.Chowdhury SK, Smith DR, Fernyhough P. The role of aberrant mitochondrial bioenergetics in diabetic neuropathy. Neurobiol Dis. 2013;51:56–65. doi: 10.1016/j.nbd.2012.03.016. [DOI] [PubMed] [Google Scholar]
- 27.Chowdhury SK, Dobrowsky RT, Fernyhough P. Nutrient excess and altered mitochondrial proteome and function contribute to neurodegeneration in diabetes. Mitochondrion. 2011;11(6):845–854. doi: 10.1016/j.mito.2011.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kalichman MW, Powell HC, Mizisin AP. Reactive, degenerative, and proliferative Schwann cell responses in experimental galactose and human diabetic neuropathy. Acta Neuropathol. 1998;95(1):47–56. doi: 10.1007/s004010050764. [DOI] [PubMed] [Google Scholar]
- 29.Mizisin AP, Nelson RW, Sturges BK, Vernau KM, Lecouteur RA, Williams DC, Burgers ML, Shelton GD. Comparable myelinated nerve pathology in feline and human diabetes mellitus. Acta Neuropathol. 2007;113(4):431–442. doi: 10.1007/s00401-006-0163-8. [DOI] [PubMed] [Google Scholar]
- 30.Zhang L, Yu C, Vasquez FE, Galeva N, Onyango I, Swerdlow RH, Dobrowsky RT. Hyperglycemia alters the schwann cell mitochondrial proteome and decreases coupled respiration in the absence of superoxide production. J Proteome Res. 2010;9(1):458–471. doi: 10.1021/pr900818g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vallianou N, Evangelopoulos A, Koutalas P. Alpha-lipoic Acid and diabetic neuropathy. Rev Diabet Stud. 2009;6(4):230–236. doi: 10.1900/RDS.2009.6.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pitceathly RD, Taanman JW, Rahman S, Meunier B, Sadowski M, Cirak S, Hargreaves I, Land JM, Nanji T, Polke JM, Woodward CE, Sweeney MG, Solanki S, Foley AR, Hurles ME, Stalker J, Blake J, Holton JL, Phadke R, Muntoni F, Reilly MM, Hanna MG, Consortium UK COX10 mutations resulting in complex multisystem mitochondrial disease that remains stable into adulthood. JAMA Neurol. 2013;70(12):1556–1561. doi: 10.1001/jamaneurol.2013.3242. [DOI] [PubMed] [Google Scholar]
- 33.Diaz F, Thomas CK, Garcia S, Hernandez D, Moraes CT. Mice lacking COX10 in skeletal muscle recapitulate the phenotype of progressive mitochondrial myopathies associated with cytochrome c oxidase deficiency. Hum Mol Genet. 2005;14(18):2737–2748. doi: 10.1093/hmg/ddi307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Diaz F, Garcia S, Hernandez D, Regev A, Rebelo A, Oca-Cossio J, Moraes CT. Pathophysiology and fate of hepatocytes in a mouse model of mitochondrial hepatopathies. Gut. 2008;57(2):232–242. doi: 10.1136/gut.2006.119180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fukui H, Diaz F, Garcia S, Moraes CT. Cytochrome c oxidase deficiency in neurons decreases both oxidative stress and amyloid formation in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci USA. 2007;104(35):14163–14168. doi: 10.1073/pnas.0705738104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang J, Wilhelmsson H, Graff C, Li H, Oldfors A, Rustin P, Bruning JC, Kahn CR, Clayton DA, Barsh GS, Thoren P, Larsson NG. Dilated cardiomyopathy and atrioventricular conduction blocks induced by heart-specific inactivation of mitochondrial DNA gene expression. Nat Genet. 1999;21(1):133–137. doi: 10.1038/5089. [DOI] [PubMed] [Google Scholar]
- 37.Wredenberg A, Wibom R, Wilhelmsson H, Graff C, Wiener HH, Burden SJ, Oldfors A, Westerblad H, Larsson NG. Increased mitochondrial mass in mitochondrial myopathy mice. Proc Natl Acad Sci USA. 2002;99(23):15066–15071. doi: 10.1073/pnas.232591499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Silva JP, Kohler M, Graff C, Oldfors A, Magnuson MA, Berggren PO, Larsson NG. Impaired insulin secretion and beta-cell loss in tissue-specific knockout mice with mitochondrial diabetes. Nat Genet. 2000;26(3):336–340. doi: 10.1038/81649. [DOI] [PubMed] [Google Scholar]
- 39.Sorensen L, Ekstrand M, Silva JP, Lindqvist E, Xu B, Rustin P, Olson L, Larsson NG. Late-onset corticohippocampal neurodepletion attributable to catastrophic failure of oxidative phosphorylation in MILON mice. J Neurosci. 2001;21(20):8082–8090. doi: 10.1523/JNEUROSCI.21-20-08082.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Harding HP, Zhang Y, Zeng H, Novoa I, Lu PD, Calfon M, Sadri N, Yun C, Popko B, Paules R, Stojdl DF, Bell JC, Hettmann T, Leiden JM, Ron D. An integrated stress response regulates amino acid metabolism and resistance to oxidative stress. Mol Cell. 2003;11(3):619–633. doi: 10.1016/S1097-2765(03)00105-9. [DOI] [PubMed] [Google Scholar]
- 41.Boneh A. Regulation of mitochondrial oxidative phosphorylation by second messenger-mediated signal transduction mechanisms. Cell Mol Life Sci. 2006;63(11):1236–1248. doi: 10.1007/s00018-005-5585-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shackelford DB, Shaw RJ. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer. 2009;9(8):563–575. doi: 10.1038/nrc2676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012;13(4):251–262. doi: 10.1038/nrm3311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jaegle M, Ghazvini M, Mandemakers W, Piirsoo M, Driegen S, Levavasseur F, Raghoenath S, Grosveld F, Meijer D. The POU proteins Brn-2 and Oct-6 share important functions in Schwann cell development. Genes Dev. 2003;17(11):1380–1391. doi: 10.1101/gad.258203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shen YA, Chen Y, Dao DQ, Mayoral SR, Wu L, Meijer D, Ullian EM, Chan JR, Lu QR. Phosphorylation of LKB1/Par-4 establishes Schwann cell polarity to initiate and control myelin extent. Nat Commun. 2014;5:4991. doi: 10.1038/ncomms5991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chan JR, Jolicoeur C, Yamauchi J, Elliott J, Fawcett JP, Ng BK, Cayouette M. The polarity protein Par-3 directly interacts with p75NTR to regulate myelination. Science. 2006;314(5800):832–836. doi: 10.1126/science.1134069. [DOI] [PubMed] [Google Scholar]
- 47.Feltri ML, Graus Porta D, Previtali SC, Nodari A, Migliavacca B, Cassetti A, Littlewood-Evans A, Reichardt LF, Messing A, Quattrini A, Mueller U, Wrabetz L. Conditional disruption of beta 1 integrin in Schwann cells impedes interactions with axons. J Cell Biol. 2002;156(1):199–209. doi: 10.1083/jcb.200109021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Taveggia C, Feltri ML, Wrabetz L. Signals to promote myelin formation and repair. Nat Rev Neurol. 2010;6(5):276–287. doi: 10.1038/nrneurol.2010.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rizzuto R, De Stefani D, Raffaello A, Mammucari C. Mitochondria as sensors and regulators of calcium signalling. Nat Rev Mol Cell Biol. 2012;13(9):566–578. doi: 10.1038/nrm3412. [DOI] [PubMed] [Google Scholar]
- 50.Deluca HF, Engstrom GW. Calcium uptake by rat kidney mitochondria. Proc Natl Acad Sci USA. 1961;47:1744–1750. doi: 10.1073/pnas.47.11.1744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vasington FD, Murphy JV. Ca ion uptake by rat kidney mitochondria and its dependence on respiration and phosphorylation. J Biol Chem. 1962;237:2670–2677. [PubMed] [Google Scholar]
- 52.McCormack JG, Denton RM. The effects of calcium ions and adenine nucleotides on the activity of pig heart 2-oxoglutarate dehydrogenase complex. Biochem J. 1979;180(3):533–544. doi: 10.1042/bj1800533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.McCormack JG, Halestrap AP, Denton RM. Role of calcium ions in regulation of mammalian intramitochondrial metabolism. Physiol Rev. 1990;70(2):391–425. doi: 10.1152/physrev.1990.70.2.391. [DOI] [PubMed] [Google Scholar]
- 54.Baughman JM, Perocchi F, Girgis HS, Plovanich M, Belcher-Timme CA, Sancak Y, Bao XR, Strittmatter L, Goldberger O, Bogorad RL, Koteliansky V, Mootha VK. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature. 2011;476(7360):341–345. doi: 10.1038/nature10234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.De Stefani D, Raffaello A, Teardo E, Szabo I, Rizzuto R. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature. 2011;476(7360):336–340. doi: 10.1038/nature10230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hirose K, Kadowaki S, Tanabe M, Takeshima H, Iino M. Spatiotemporal dynamics of inositol 1,4,5-trisphosphate that underlies complex Ca2+ mobilization patterns. Science. 1999;284(5419):1527–1530. doi: 10.1126/science.284.5419.1527. [DOI] [PubMed] [Google Scholar]
- 57.Okubo Y, Kakizawa S, Hirose K, Iino M. Visualization of IP(3) dynamics reveals a novel AMPA receptor-triggered IP(3) production pathway mediated by voltage-dependent Ca(2+) influx in Purkinje cells. Neuron. 2001;32(1):113–122. doi: 10.1016/S0896-6273(01)00464-0. [DOI] [PubMed] [Google Scholar]
- 58.Okubo Y, Kakizawa S, Hirose K, Iino M. Cross talk between metabotropic and ionotropic glutamate receptor-mediated signaling in parallel fiber-induced inositol 1,4,5-trisphosphate production in cerebellar Purkinje cells. J Neurosci. 2004;24(43):9513–9520. doi: 10.1523/JNEUROSCI.1829-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Furutani K, Okubo Y, Kakizawa S, Iino M. Postsynaptic inositol 1,4,5-trisphosphate signaling maintains presynaptic function of parallel fiber-Purkinje cell synapses via BDNF. Proc Natl Acad Sci USA. 2006;103(22):8528–8533. doi: 10.1073/pnas.0600497103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kanemaru K, Okubo Y, Hirose K, Iino M. Regulation of neurite growth by spontaneous Ca2+ oscillations in astrocytes. J Neurosci. 2007;27(33):8957–8966. doi: 10.1523/JNEUROSCI.2276-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mashimo M, Okubo Y, Yamazawa T, Yamasaki M, Watanabe M, Murayama T, Iino M. Inositol 1,4,5-trisphosphate signaling maintains the activity of glutamate uptake in Bergmann glia. Eur J Neurosci. 2010;32(10):1668–1677. doi: 10.1111/j.1460-9568.2010.07452.x. [DOI] [PubMed] [Google Scholar]
- 62.Kanemaru K, Kubota J, Sekiya H, Hirose K, Okubo Y, Iino M. Calcium-dependent N-cadherin up-regulation mediates reactive astrogliosis and neuroprotection after brain injury. Proc Natl Acad Sci USA. 2013;110(28):11612–11617. doi: 10.1073/pnas.1300378110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fields RD, Ni Y. Nonsynaptic communication through ATP release from volume-activated anion channels in axons. Sci Signal. 2010;3(142):ra73. doi: 10.1126/scisignal.2001128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sabirov RZ, Okada Y. The maxi-anion channel: a classical channel playing novel roles through an unidentified molecular entity. J Physiol Sci. 2009;59(1):3–21. doi: 10.1007/s12576-008-0008-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.von Kugelgen I. Pharmacological profiles of cloned mammalian P2Y-receptor subtypes. Pharmacol Ther. 2006;110(3):415–432. doi: 10.1016/j.pharmthera.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 66.Wildman SS, Unwin RJ, King BF. Extended pharmacological profiles of rat P2Y2 and rat P2Y4 receptors and their sensitivity to extracellular H+ and Zn2+ ions. Br J Pharmacol. 2003;140(7):1177–1186. doi: 10.1038/sj.bjp.0705544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Fitzgerald M. Cutaneous primary afferent properties in the hind limb of the neonatal rat. J Physiol. 1987;383:79–92. doi: 10.1113/jphysiol.1987.sp016397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Micu I, Jiang Q, Coderre E, Ridsdale A, Zhang L, Woulfe J, Yin X, Trapp BD, McRory JE, Rehak R, Zamponi GW, Wang W, Stys PK. NMDA receptors mediate calcium accumulation in myelin during chemical ischaemia. Nature. 2006;439(7079):988–992. doi: 10.1038/nature04474. [DOI] [PubMed] [Google Scholar]
- 69.Hamilton NB, Kolodziejczyk K, Kougioumtzidou E, Attwell D. Proton-gated Ca(2+)-permeable TRP channels damage myelin in conditions mimicking ischaemia. Nature. 2016;529(7587):523–527. doi: 10.1038/nature16519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Garbay B, Heape AM, Sargueil F, Cassagne C. Myelin synthesis in the peripheral nervous system. Prog Neurobiol. 2000;61(3):267–304. doi: 10.1016/S0301-0082(99)00049-0. [DOI] [PubMed] [Google Scholar]
- 71.Harris JJ, Attwell D. The energetics of CNS white matter. J Neurosci. 2012;32(1):356–371. doi: 10.1523/JNEUROSCI.3430-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chrast R, Saher G, Nave KA, Verheijen MH. Lipid metabolism in myelinating glial cells: lessons from human inherited disorders and mouse models. J Lipid Res. 2011;52(3):419–434. doi: 10.1194/jlr.R009761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107(9):1058–1070. doi: 10.1161/CIRCRESAHA.110.223545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lin MT, Beal MF. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 2006;443(7113):787–795. doi: 10.1038/nature05292. [DOI] [PubMed] [Google Scholar]
- 75.Vincent AM, Brownlee M, Russell JW. Oxidative stress and programmed cell death in diabetic neuropathy. Ann N Y Acad Sci. 2002;959:368–383. doi: 10.1111/j.1749-6632.2002.tb02108.x. [DOI] [PubMed] [Google Scholar]