Abstract
Bone loss and the resulting skeletal fragility is induced by several pathological or natural conditions, the most prominent of which being aging as well as the decreased levels of circulating estrogens in post-menopause females. To date, most treatments against bone loss aim at preventing excess bone resorption. We here summarize data indicating that the estrogen-related receptors (ERRs) α and γ prevent bone formation. Inhibiting these receptors may thus constitute an anabolic approach by increasing bone formation.
Keywords: Nuclear receptors, Bone, ERR, Osteoblasts, Menopause
Introduction
Bone is a highly dynamic tissue that is under constant remodeling, a phenomenon that comprises two complementary processes: bone formation and bone resorption (reviewed in Frenkel et al. [1]). Two main cell types participate to these features. Osteoclasts are cells of the hematopoietic lineage that resorb bone, whereas osteoblasts are cells of mesenchymal origin that mineralize the bone matrix. The equilibrium between these two processes is tightly controlled under “normal” conditions. However, this equilibrium can be disrupted under pathological conditions but also under naturally occurring ones. Indeed aging, affecting both males and females, reduces bone formation by decreasing the capacities of pre-osteoblasts to differentiate into mature cells (reviewed in Khosla [2]). In addition, the cessation of the ovarian functions at menopause, leading to reduced circulating levels of estrogens, results in increased osteoclast differentiation and thus enhanced bone resorption (reviewed in Manolagas et al. [3]). This leads to osteoporosis, a bone fragility syndrome that includes an increased fracture risk particularly in aging females, due to the combination of both processes. To date most treatments against osteoporosis are anti-catabolic, i.e, aim at reducing excess bone resorption by osteoclasts. However, anabolic treatments (aiming at enhancing bone formation) are starting to emerge (reviewed in Marie and Kassem [4]). Here we review data that indicate that negatively targeting the estrogen-related receptors (ERR) α and/or γ may constitute a promising approach to design anabolic treatments.
The estrogen-related receptors: ligand-independent nuclear receptors
The nuclear receptor (NR) superfamily comprises 48 members in the human that are generally defined as ligand-dependent transcription factors [5, 6]. With few exceptions, these factors all share a similar protein organization. A centrally located DNA-binding domain (DBD) composed of two zinc finger modules mediates a direct interaction with cognate response elements on the promoters of their target genes. A hinge region links the DBD to the C-terminally located ligand-binding domain (LBD). The latter is a globular structure comprising several alpha-helices and undergoes a conformational change upon interaction with a specific ligand. This results in the production of an interaction surface that allows the recruitment of transcriptional co-activators. In turn these co-activators, directly or indirectly, induce chromatin modifications and the recruitment of the transcriptional machinery at the promoters, leading to increased expression of target genes. In addition, some but not all, receptors possess an N-terminally located domain that can mediate ligand-independent transcriptional activation.
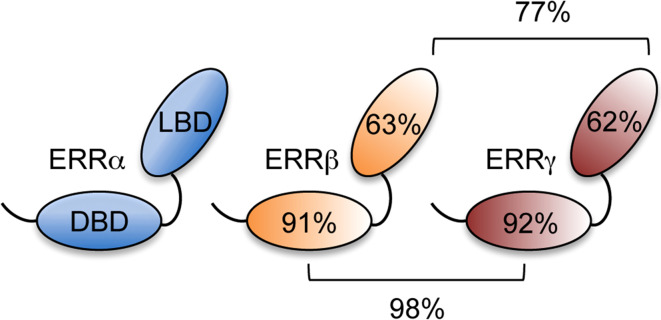
Ligands (such as 17β-estradiol or the thyroid hormone) had been described well before the first characterization of their cognate receptors (estrogen receptor [ER] and thyroid hormone receptor [TR]) in the mid-eighties [6]. Using newly cloned receptor sequences as probes, several other nuclear receptors have been isolated starting in the late eighties and have been referred to as “orphans” in the initial absence of an identified natural ligand. Although specific ligands have been identified for some of these receptors, a number of receptors remain orphan. This is for instance the case of the estrogen-related receptors (ERR) α and β, the first orphan receptors identified in 1988 [7]. Together with the more recently isolated ERRγ [8], they form a distinct sub-family, and display a strong level of sequence identity with each other, in particular within their DBD and LBD (Fig. 1).
Fig. 1.
Organization of the estrogen-related receptors. The ERRs comprise two conserved domains, the centrally located DNA-binding domain (DBD) and the C-terminally located ligand-binding domain (LBD). The percentage of sequence identity within these domains is indicated relative to ERRα on the receptors, and between ERRβ and ERRγ, above and below the brackets
Determination of the 3D structure of the LBDs of ERRα and γ has shown that these receptors display an “active” conformation, allowing to contact co-activators in the absence of any ligand in their putative ligand-binding pocket [9, 10]. Although the crystal structure of ERRβ has not been published, it seems thus likely that all ERRs act as ligand-independent transcription factors, although clearly belonging to the NR superfamily (reviewed in Horard and Vanacker [11]). Several publications indicate that the transcriptional activities of the ERRs can be regulated by various processes, such as sub-cellular localization or post-translational modifications (see examples in [12–17]. One key point is, however, the capacity to interact with specific co-modulators that can be viewed as protein ligands and may be available or not in a given cellular context (see examples in [18–20]).
Despite the capacity of the ERRs to act in a ligand-independent manner, several synthetic compounds have been identified that [positively (agonists) or negatively (inverse agonists)] modulate their transcriptional activities, more or less specifically. For instance, 4-hydroxy-tamoxifene [OHT; a selective estrogen receptor modulator (SERM) which is broadly used in breast cancer therapy] or its analog GSK5182 reduce the activities of both ERRβ and γ [21–23] whereas GSK4716 and DY131 act as agonists for both receptors [24, 25]. Published literature suggests that it is difficult to identify modulators that clearly discriminate between ERRβ and γ, likely because of the particularly high level of sequence identity in their LBDs. To date, one exception is bisphenol A (BPA), which counteracts the effects of inverse agonists on ERRγ in a seemingly specific manner both in vitro and in vivo [26–28]. In contrast, compounds have been isolated that specifically target ERRα, and not β or γ. This is the case of pyrido[1,2-α]pyrimidine-4-ones derivatives that act as agonists [29]. Conversely for instance XCT790 and C29 act as inverse agonists and, at least for the former, promote proteasome-dependent degradation of the receptor [30]. However, data are often lacking that could indicate whether these compounds indeed act in vivo, although this has been shown for instance for C29, GSK5182 and BPA (see [28, 31, 32]). Whether the effects of a given drug strictly depend on a given ERR species is also often an open question. Despite these restrictions, these compounds can be viewed as useful tools to study the functions of the ERRs, and may suggest promising approaches to modulate the activities of the ERRs in given pathological processes.
Physiopathological functions of the ERRs
In vitro and in vivo studies have contributed to identify several physiopathological functions played by the ERRs. ERRβ is mainly expressed in embryonic tissues in the mouse and regulates placental development [33] as well as the maintenance of self-renewal in both embryonic and trophoblast stem cells [34–38]. The role played by ERRβ in human embryonic tissues is unknown. It should be noted that the receptor is not expressed in human embryonic stem cells, in contrast to mouse ones, but an expression in other human embryonic tissues has not been documented to date [39]. ERRβ is also involved in the specification of epithelial cells in the mouse inner ear [40]. Consistently, mutations in the human ESRRB gene (encoding ERRβ) result in a form of hearing impairment [41]. It has also been shown that maintenance of the number of rod photoreceptor cells during mouse aging depends on ERRβ but no data are available concerning the impact of the receptor on human retina [42].
ERRα and γ are strongly expressed in tissues with high energy demand (for instance heart, muscle, liver and fat) where they control various metabolic processes such as mitochondrial biogenesis and function, lipid uptake and oxidation, tricarboxylic acid cycle and neoglucogenesis (reviewed in [43–46]. These activities of ERRα and γ are exerted not only in conventional metabolic tissues but also have a considerable impact on pathological processes such as cancer (reviewed in [47–49]). For instance both receptors are at the heart of the metabolic switch referred to as the Warburg effect in which cancer cells shift from oxidative to glycolytic metabolism [20, 50, 51]). Interestingly, these receptors display opposite functions in this process as well as in the establishment of other traits of cancer progression such as proliferation and epithelial–mesenchymal transition (EMT) [52–56]. All these features are promoted by ERRα while repressed by ERRγ. This is consistent with ERRα and γ being factors of unfavorable and favorable prognosis (respectively), as described in several cancer types (review in [47, 57]). In this line, additional works have also shown that ERRα promotes cell migration, invasion and the establishment of metastasis [58–60].
ERRα is also a critical component of innate immune response, regulating the production of mitochondrial reactive oxygen species in response to γ-interferon as well as attenuating toll-like receptor inflammatory response in macrophaghes [61, 62].
Functions of the ERRs in mineralized tissues
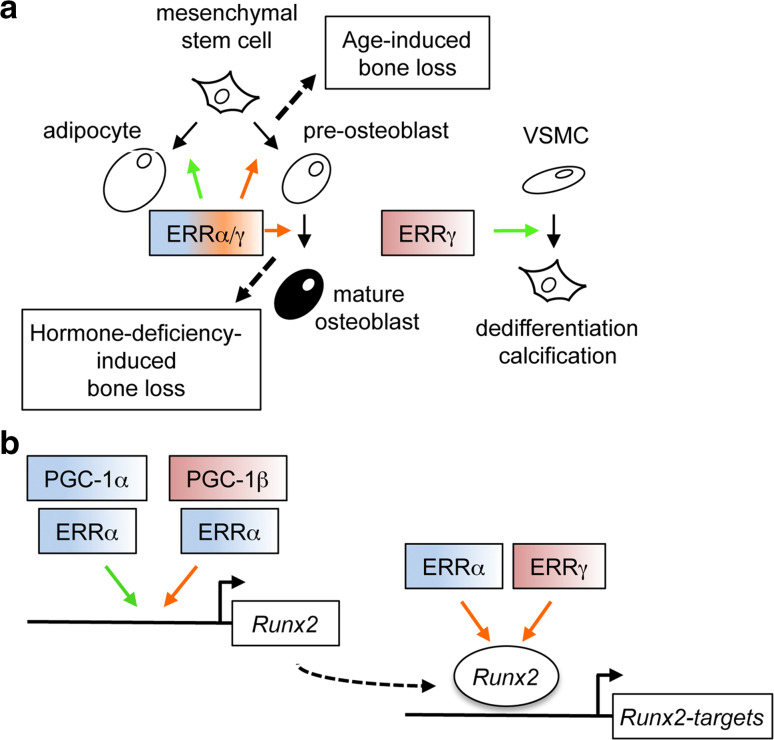
ERRα is highly expressed in the ossification zones (long as well as flat bones) during mouse embryonic development [63] suggesting a contribution to endochondral as well as intramembranous ossification. However, examination of ERRα knock-out (ERRαKO) animals has shown that the receptor is not required for bone morphogenesis or ossification in young animals, i.e. up to 14 weeks of age, at which peak bone mass is reached [64, 65]. This was estimated by measuring both trabecular and cortical bone parameters that do not significantly vary between mutant and wild type littermates. However, ERRαKO animals do not lose bone with aging (i.e, between 14 and 24 weeks of age), in contrast to wild type counterparts. The latter also dramatically lose bone upon ovariectomy (which mimicks menopause in mice). In striking opposition, ERRαKO animals are resistant to this bone loss. As evidenced by the analysis of dynamic bone parameters, osteoclasts number and activity is unchanged whereas bone formation rate (i.e., osteoblast activity) is increased in ERRαKO animals relative to wild types. Consistently, pre-osteoblasts originating from mutant animals are more prone to differentiate, express enhanced levels of osteoblast molecular markers (including those of Runx2, the master gene of osteoblast differentiation) and display increased mineralizing activity ex vivo. Taken together with the decreased capacity displayed by ERRαKO mesenchymal cells to differentiate, at least in vivo, into the adipocyte lineage [64, 66, 67], this suggests that ERRα affects the early determination of mesenchymal stem cells, promoting their commitment into the adipocyte lineage at the detriment of the osteoblast one (Fig. 2a). This is in contradiction with earlier results showing that overexpression of ERRα in pre-osteoblasts in vitro promotes rather than decreases osteoblast differentiation [68]. One possible explanation to reconcile these discrepant results comes from the data published by Kammerer et al. [69] who showed a complex effect of ERRα on Runx2 expression in cell culture. Indeed the receptor can stimulate or repress Runx2 expression in the presence of PGC-1α or PGC-1β, respectively. In contrast ERα activates Runx2 expression in the presence of PGC-1β (Fig. 2b). This suggests that ERα may also indirectly promote the positive activities of ERRα by competing for PGC-1β binding. It could be hypothesized that the expression of all these factors varies with age, hormonal status and/or pre-osteoblast differentiation state. For example, it has been shown that 17β-estradiol regulates ERα mRNA expression and protein stability (reviewed in [70]) as well as PGC-1α expression [71]. Taking these data together, it is thus possible that ERRα is a fine-tuning modulator of Runx2 expression and thus of osteoblast differentiation in vivo. The consequences of these subtle regulations may be cumulative and therefore would only be obvious in terms of bone mass after a given amount of time, i.e., after the occurrence of peak bone mass.
Fig. 2.
Functions of ERRα and γ in mineralizing cells. a Effects of ERRα and ERRγ on mineralizing cells. ERRα exerts early effects on mesenchymal cells inducing commitment to the adipocyte lineage (green arrow), while repressing commitment to the osteoblast one (red arrow). ERRα also acts later in this lineage, inhibiting osteoblast maturation, an activity shared by ERRγ. The two repressive effects of ERRα on osteoblast differentiation contribute to bone loss during aging and hormonal deficiency, respectively. Furthermore ERRγ decreases trabecular bone mass but promotes calcification in vascular smooth muscle cell (VSMC). Note that the effect of ERRγ on hormone deficiency-induced bone loss has not been investigated so far. b Effects of ERRα and γ on Runx2 expression and activity. ERRα activates (green arrow) or repress (red arrow) Runx2 expression in the presence of PGC-1α or β, respectively. Both ERRα and γ inhibit the transcriptional activities of the Runx2 protein. See text for details and references
On another hand, the increased capacity of ERRαKO pre-osteoblast to differentiate ex vivo can be rescued by reintroduction of the receptor after the onset of differentiation, suggesting a later effect of ERRα, i.e., rather on osteoblast maturation [65]. It is, thus, possible that the receptor exerts two independent effects on osteoblast differentiation (early at the commitment level, late at the maturation level). In this line, it is worth noting that when ERRα is inactivated during osteoblast maturation (using conditional knock-out mice), Runx2 expression is not modulated, in contrast to that of its target genes. This suggested that the receptor also impacts on Runx2 activity, a hypothesis which has been confirmed [72]. In addition ERRα positively and directly modulates the expression of osteopontin (opn), a late marker of osteoblast maturation which inhibits mineralization [64, 65, 73–76]. Interestingly ERRα conditional knock-out animals resist to ovariectomy-induced but not to age-induced bone loss.
Altogether this shows that ERRα exerts at least two independent effects on osteoblast differentiation, resulting in two independent phenotypes in vivo. In other terms, the repressive effects of ERRα on osteoblast commitment contribute to bone aging, whereas the negative action of the receptor on osteoblast maturation participates to bone loss induced by hormone withdrawal. Although additional research in needed to determine the precise molecular mechanisms through which ERRα exerts these effects, this suggests that deactivating the receptor could increase bone formation in vivo. Targeting ERRα could thus be a promising strategy to prevent bone loss during aging and after menopause. In support to this statement, inactivating the receptor in human pre-osteoblast also leads to increased differentiation in cell culture [64]. However, it should be noted that all the above data have been obtained using a genetic inactivation of ERRα. Obviously a pharmacological approach would be preferred but, to date, no report has been published concerning the effect of ERRα-deactivating compounds on osteoblast in vitro and bone in vivo.
The complete inactivation of ERRγ in mice leads to perinatal death [77], preventing the study of the bone status of mutant animals. Cardelli and Aubin [78] recently reported that ERRγ+/− animals displayed increased trabecular bone as compared to wild type counterparts. Intriguingly this phenotype only affects males, but not females, suggesting an undocumented cross-talk of ERRγ with hormone signaling. ERRγ+/− bone phenotype can be observed as early as after 8 weeks after birth, aggravates with age and correlates with increased osteoblast number and activity in vivo. As for ERRα, inactivation of ERRγ leads enhanced pre-osteoblast differentiation ex vivo [78] as well as reduced adipocyte differentiation in vitro [79]. The former effect is thought to rely on unchecked Runx2 activity in the absence of ERRγ [80], as is again the case for ERRα [72]. A second level of ERRγ activity has been suggested with the receptor inducing the expression of miR-433, which targets Runx2 mRNA for degradation [81]. These studies suggest that ERRγ is anti-osteogenic and that its inhibition could lead to increased mineral density. Intriguingly, however, a recent report [82] shows that ERRγ promotes vascular calcification, a major component of morbidity and mortality in patients with such diseases as atherosclerosis. In cultures of vascular smooth muscle cells ERRγ expression is induced by calcification medium and in turn directly and indirectly induces the expression of BMP2. Importantly, in vivo treatment with an ERRγ specific inverse agonist reduces vascular calcification in the mouse. ERRγ is therefore anti-osteogenic in bone and pro-osteogenic in the vasculature. The mechanisms that accounts for these antagonistic activities is not clear but a recent report proposes that in liver cells, the transcriptional activation of Cyp2E1 by ERRγ can be switched off by interaction with the RORα nuclear receptor [83]. Whether such a type of interference mechanism is at work in mineralizing cells is presently unknown. Since ERRα and ERRγ display rather similar effects on bone cells (i.e., anti-osteogenic), it will be interesting to determine whether the former also displays anti-mineralizing activities in the vasculature.
The effects of ERRs on the other major cellular component of bone (i.e., osteoclasts) have also been addressed. Study of ERRγ+/− has shown that osteoclast number and activities do not vary comparing to wild type animals [78]. Analysis of ERRαKO mice in terms of osteoclast number and activities has raised contradictory results, ranging from no variation [64, 65] or decreased parameters [84]. Noteworthy it had previously been shown that ERRα promotes osteoclast spreading and migration in cell culture [85] raising the possibility that the absence of the receptor in knock-out animals may be compensated for by unidentified factor(s). In addition an indirect effect of ERRα on osteoclasts has also be reported. Overexpression of the receptor in xenografted breast cancer cells results in increased production of osteoprotegerin, an inhibitor of osteoclastogenesis, leading to decreased osteoclast differentiation by recipient immunodeficient mice and reduced capacity of tumor cells to metastasize in the bone [59].
Possible future directions
Both ERRα and γ exert anti-osteogenic effects in bone, through both overlapping and divergent mechanisms. This suggests that deactivating these receptors could be a promising approach to reduce bone loss, possibly whatever its cause, since inhibiting these receptors leads to increased bone formation. However it should be reminded that most of the results obtained to date originate from mouse models and care should be taken concerning translation to human. The activities of ERRα and ERRγ appear convergent on bone, yet the receptors strikingly differ concerning their impact on cancer. ERRα is a factor of poor prognosis and promotes traits of cancer aggressiveness whereas ERRγ is a factor of favorable prognosis and likely decreases cancer aggressiveness. It can therefore be hypothesized that deactivating ERRα, in contrast to ERRγ, may reduce the risk of cancer-related side effects. The recent discovery of a pro-osteogenic effect of ERRγ on vasculature is, however, intriguing in that it, in particular, also questions the effect of ERRα on this process. Whether both receptors there behave in a similar (as in bone) or divergent manner (as in cancer) is an important question to solve.
Although it is difficult to definitely exclude the possibility of a natural ligand in vivo, it is highly likely that the ERRs act as ligand-independent manner. However the activity of these receptors can be modulated by synthetic compound that impact on their protein stability and/or transcriptional activities. This renders them attractive targets to tackle pathophysiological processes in which they are involved. The ability of these specific compounds to modulate the activities of the ERRs in vivo is only starting to be studied and there is no doubt that efforts will be developed in this direction.
Acknowledgments
Work in JMV’s laboratory is funded by Ligue contre le Cancer (comité Drôme and Puy-de-Dôme), Agence Nationale pour la Recherche (ANR-14-CE12-0003-03) and Agence Nationale de sécurité sanitaire de l’alimentation, de l’environnement et du travail (ANSES).
References
- 1.Frenkel B, Hong A, Baniwal SK, et al. Regulation of adult bone turnover by sex steroids. J Cell Physiol. 2010;224:305–310. doi: 10.1002/jcp.22159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Khosla S. Pathogenesis of age-related bone loss in humans. J Gerontol A Biol Sci Med Sci. 2013;68:1226–1235. doi: 10.1093/gerona/gls163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Manolagas SC, O’Brien CA, Almeida M. The role of estrogen and androgen receptors in bone health and disease. Nat Rev Endocrinol. 2013;9:699–712. doi: 10.1038/nrendo.2013.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marie PJ, Kassem M. Osteoblasts in osteoporosis: past, emerging, and future anabolic targets. Eur J Endocrinol. 2011;165:1–10. doi: 10.1530/EJE-11-0132. [DOI] [PubMed] [Google Scholar]
- 5.Laudet V, Gronemeyer H. The nuclear receptor factbook. San Diego, CA: Academic Press; 2002. [Google Scholar]
- 6.Gustafsson J-A. Historical overview of nuclear receptors. J Steroid Biochem Mol Biol. 2016;157:3–6. doi: 10.1016/j.jsbmb.2015.03.004. [DOI] [PubMed] [Google Scholar]
- 7.Giguère V, Yang N, Segui P, Evans RM. Identification of a new class of steroid hormone receptors. Nature. 1988;331:91–94. doi: 10.1038/331091a0. [DOI] [PubMed] [Google Scholar]
- 8.Hong H, Yang L, Stallcup MR. Hormone-independent transcriptional activation and coactivator binding by novel orphan nuclear receptor ERR3. J Biol Chem. 1999;274:22618–22626. doi: 10.1074/jbc.274.32.22618. [DOI] [PubMed] [Google Scholar]
- 9.Greschik H, Wurtz J-M, Sanglier S, et al. Structural and functional evidence for ligand-independent transcriptional activation by the estrogen-related receptor 3. Mol Cell. 2002;9:303–313. doi: 10.1016/S1097-2765(02)00444-6. [DOI] [PubMed] [Google Scholar]
- 10.Kallen J, Schlaeppi J-M, Bitsch F, et al. Evidence for ligand-independent transcriptional activation of the human estrogen-related receptor alpha (ERRalpha): crystal structure of ERRalpha ligand binding domain in complex with peroxisome proliferator-activated receptor coactivator-1alpha. J Biol Chem. 2004;279:49330–49337. doi: 10.1074/jbc.M407999200. [DOI] [PubMed] [Google Scholar]
- 11.Horard B, Vanacker J-M. Estrogen receptor-related receptors: orphan receptors desperately seeking a ligand. J Mol Endocrinol. 2003;31:349–357. doi: 10.1677/jme.0.0310349. [DOI] [PubMed] [Google Scholar]
- 12.Barry JB, Giguère V. Epidermal growth factor-induced signaling in breast cancer cells results in selective target gene activation by orphan nuclear receptor estrogen-related receptor alpha. Cancer Res. 2005;65:6120–6129. doi: 10.1158/0008-5472.CAN-05-0922. [DOI] [PubMed] [Google Scholar]
- 13.Vu EH, Kraus RJ, Mertz JE. Phosphorylation-dependent sumoylation of estrogen-related receptor alpha1. Biochemistry. 2007;46:9795–9804. doi: 10.1021/bi700316g. [DOI] [PubMed] [Google Scholar]
- 14.Tremblay AM, Wilson BJ, Yang X-J, Giguère V. Phosphorylation-dependent sumoylation regulates estrogen-related receptor-alpha and -gamma transcriptional activity through a synergy control motif. Mol Endocrinol. 2008;22:570–584. doi: 10.1210/me.2007-0357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilson BJ, Tremblay AM, Deblois G, et al. An acetylation switch modulates the transcriptional activity of estrogen-related receptor alpha. Mol Endocrinol. 2010;24:1349–1358. doi: 10.1210/me.2009-0441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rossi M, Colecchia D, Iavarone C, et al. Extracellular signal-regulated kinase 8 (ERK8) controls estrogen-related receptor α (ERRα) cellular localization and inhibits its transcriptional activity. J Biol Chem. 2011;286:8507–8522. doi: 10.1074/jbc.M110.179523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim D-K, Kim Y-H, Hynx D, et al. PKB/Akt phosphorylation of ERRγ contributes to insulin-mediated inhibition of hepatic gluconeogenesis. Diabetologia. 2014;57:2576–2585. doi: 10.1007/s00125-014-3366-x. [DOI] [PubMed] [Google Scholar]
- 18.Kamei Y, Ohizumi H, Fujitani Y, et al. PPARgamma coactivator 1beta/ERR ligand 1 is an ERR protein ligand, whose expression induces a high-energy expenditure and antagonizes obesity. Proc Natl Acad Sci USA. 2003;100:12378–12383. doi: 10.1073/pnas.2135217100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rodríguez-Calvo R, Jové M, Coll T, et al. PGC-1beta down-regulation is associated with reduced ERRalpha activity and MCAD expression in skeletal muscle of senescence-accelerated mice. J Gerontol A Biol Sci Med Sci. 2006;61:773–780. doi: 10.1093/gerona/61.8.773. [DOI] [PubMed] [Google Scholar]
- 20.Eichner LJ, Perry M-C, Dufour CR, et al. miR-378(∗) mediates metabolic shift in breast cancer cells via the PGC-1β/ERRγ transcriptional pathway. Cell Metab. 2010;12:352–361. doi: 10.1016/j.cmet.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 21.Coward P, Lee D, Hull MV, Lehmann JM. 4-Hydroxytamoxifen binds to and deactivates the estrogen-related receptor gamma. Proc Natl Acad Sci USA. 2001;98:8880–8884. doi: 10.1073/pnas.151244398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tremblay GB, Bergeron D, Giguere V. 4-Hydroxytamoxifen is an isoform-specific inhibitor of orphan estrogen-receptor-related (ERR) nuclear receptors beta and gamma. Endocrinology. 2001;142:4572–4575. doi: 10.1210/endo.142.10.8528. [DOI] [PubMed] [Google Scholar]
- 23.Chao EYH, Collins JL, Gaillard S, et al. Structure-guided synthesis of tamoxifen analogs with improved selectivity for the orphan ERRgamma. Bioorg Med Chem Lett. 2006;16:821–824. doi: 10.1016/j.bmcl.2005.11.030. [DOI] [PubMed] [Google Scholar]
- 24.Yu DD, Forman BM. Identification of an agonist ligand for estrogen-related receptors ERRbeta/gamma. Bioorg Med Chem Lett. 2005;15:1311–1313. doi: 10.1016/j.bmcl.2005.01.025. [DOI] [PubMed] [Google Scholar]
- 25.Zuercher WJ, Gaillard S, Orband-Miller LA, et al. Identification and structure-activity relationship of phenolic acyl hydrazones as selective agonists for the estrogen-related orphan nuclear receptors ERRbeta and ERRgamma. J Med Chem. 2005;48:3107–3109. doi: 10.1021/jm050161j. [DOI] [PubMed] [Google Scholar]
- 26.Takayanagi S, Tokunaga T, Liu X, et al. Endocrine disruptor bisphenol A strongly binds to human estrogen-related receptor gamma (ERRgamma) with high constitutive activity. Toxicol Lett. 2006;167:95–105. doi: 10.1016/j.toxlet.2006.08.012. [DOI] [PubMed] [Google Scholar]
- 27.Matsushima A, Kakuta Y, Teramoto T, et al. Structural evidence for endocrine disruptor bisphenol A binding to human nuclear receptor ERR gamma. J Biochem. 2007;142:517–524. doi: 10.1093/jb/mvm158. [DOI] [PubMed] [Google Scholar]
- 28.Tohmé M, Prud’homme SM, Boulahtouf A, et al. Estrogen-related receptor γ is an in vivo receptor of bisphenol A. FASEB J. 2014;28:3124–3133. doi: 10.1096/fj.13-240465. [DOI] [PubMed] [Google Scholar]
- 29.Peng L, Gao X, Duan L, et al. Identification of pyrido[1,2-α]pyrimidine-4-ones as new molecules improving the transcriptional functions of estrogen-related receptor α. J Med Chem. 2011;54:7729–7733. doi: 10.1021/jm200976s. [DOI] [PubMed] [Google Scholar]
- 30.Lanvin O, Bianco S, Kersual N, et al. Potentiation of ICI182,780 (Fulvestrant)-induced estrogen receptor-alpha degradation by the estrogen receptor-related receptor-alpha inverse agonist XCT790. J Biol Chem. 2007;282:28328–28334. doi: 10.1074/jbc.M704295200. [DOI] [PubMed] [Google Scholar]
- 31.Chaveroux C, Eichner LJ, Dufour CR, et al. Molecular and genetic crosstalks between mTOR and ERRα are key determinants of rapamycin-induced nonalcoholic fatty liver. Cell Metab. 2013;17:586–598. doi: 10.1016/j.cmet.2013.03.003. [DOI] [PubMed] [Google Scholar]
- 32.Kim D-K, Gang G-T, Ryu D, et al. Inverse agonist of nuclear receptor ERRγ mediates antidiabetic effect through inhibition of hepatic gluconeogenesis. Diabetes. 2013;62:3093–3102. doi: 10.2337/db12-0946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Luo J, Sladek R, Bader JA, et al. Placental abnormalities in mouse embryos lacking the orphan nuclear receptor ERR-beta. Nature. 1997;388:778–782. doi: 10.1038/42022. [DOI] [PubMed] [Google Scholar]
- 34.Tremblay GB, Kunath T, Bergeron D, et al. Diethylstilbestrol regulates trophoblast stem cell differentiation as a ligand of orphan nuclear receptor ERR beta. Genes Dev. 2001;15:833–838. doi: 10.1101/gad.873401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Festuccia N, Osorno R, Halbritter F, et al. Esrrb is a direct Nanog target gene that can substitute for Nanog function in pluripotent cells. Cell Stem Cell. 2012;11:477–490. doi: 10.1016/j.stem.2012.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Martello G, Sugimoto T, Diamanti E, et al. Esrrb is a pivotal target of the Gsk3/Tcf3 axis regulating embryonic stem cell self-renewal. Cell Stem Cell. 2012;11:491–504. doi: 10.1016/j.stem.2012.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Percharde M, Lavial F, Ng J-H, et al. Ncoa3 functions as an essential Esrrb coactivator to sustain embryonic stem cell self-renewal and reprogramming. Genes Dev. 2012;26:2286–2298. doi: 10.1101/gad.195545.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Latos PA, Goncalves A, Oxley D, et al. Fgf and Esrrb integrate epigenetic and transcriptional networks that regulate self-renewal of trophoblast stem cells. Nat Commun. 2015;6:7776. doi: 10.1038/ncomms8776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xie C-Q, Jeong Y, Fu M, et al. Expression profiling of nuclear receptors in human and mouse embryonic stem cells. Mol Endocrinol. 2009;23:724–733. doi: 10.1210/me.2008-0465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chen J, Nathans J. Estrogen-related receptor beta/NR3B2 controls epithelial cell fate and endolymph production by the stria vascularis. Dev Cell. 2007;13:325–337. doi: 10.1016/j.devcel.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 41.Collin RWJ, Kalay E, Tariq M, et al. Mutations of ESRRB encoding estrogen-related receptor beta cause autosomal-recessive nonsyndromic hearing impairment DFNB35. Am J Hum Genet. 2008;82:125–138. doi: 10.1016/j.ajhg.2007.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Onishi A, Peng G-H, Poth EM, et al. The orphan nuclear hormone receptor ERRbeta controls rod photoreceptor survival. Proc Natl Acad Sci USA. 2010;107:11579–11584. doi: 10.1073/pnas.1000102107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Villena JA, Kralli A. ERRalpha: a metabolic function for the oldest orphan. Trends Endocrinol Metab. 2008;19:269–276. doi: 10.1016/j.tem.2008.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Giguère V. Transcriptional control of energy homeostasis by the estrogen-related receptors. Endocr Rev. 2008;29:677–696. doi: 10.1210/er.2008-0017. [DOI] [PubMed] [Google Scholar]
- 45.Deblois G, Giguère V. Functional and physiological genomics of estrogen-related receptors (ERRs) in health and disease. Biochim Biophys Acta. 2011;1812:1032–1040. doi: 10.1016/j.bbadis.2010.12.009. [DOI] [PubMed] [Google Scholar]
- 46.Huss JM, Garbacz WG, Xie W. Constitutive activities of estrogen-related receptors: transcriptional regulation of metabolism by the ERR pathways in health and disease. Biochim Biophys Acta. 2015;1852:1912–1927. doi: 10.1016/j.bbadis.2015.06.016. [DOI] [PubMed] [Google Scholar]
- 47.Bianco S, Sailland J, Vanacker J-M. ERRs and cancers: effects on metabolism and on proliferation and migration capacities. J Steroid Biochem Mol Biol. 2012;130:180–185. doi: 10.1016/j.jsbmb.2011.03.014. [DOI] [PubMed] [Google Scholar]
- 48.Chang C, McDonnell DP. Molecular pathways: the metabolic regulator estrogen-related receptor α as a therapeutic target in cancer. Clin Cancer Res. 2012;18:6089–6095. doi: 10.1158/1078-0432.CCR-11-3221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Deblois G, Giguère V. Oestrogen-related receptors in breast cancer: control of cellular metabolism and beyond. Nat Rev Cancer. 2013;13:27–36. doi: 10.1038/nrc3396. [DOI] [PubMed] [Google Scholar]
- 50.Chang C, Kazmin D, Jasper JS, et al. The metabolic regulator ERRα, a downstream target of HER2/IGF-1R, as a therapeutic target in breast cancer. Cancer Cell. 2011;20:500–510. doi: 10.1016/j.ccr.2011.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cai Q, Lin T, Kamarajugadda S, Lu J. Regulation of glycolysis and the Warburg effect by estrogen-related receptors. Oncogene. 2013;32:2079–2086. doi: 10.1038/onc.2012.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yu S, Wang X, Ng C-F, et al. ERRgamma suppresses cell proliferation and tumor growth of androgen-sensitive and androgen-insensitive prostate cancer cells and its implication as a therapeutic target for prostate cancer. Cancer Res. 2007;67:4904–4914. doi: 10.1158/0008-5472.CAN-06-3855. [DOI] [PubMed] [Google Scholar]
- 53.Bianco S, Lanvin O, Tribollet V, et al. Modulating estrogen receptor-related receptor-alpha activity inhibits cell proliferation. J Biol Chem. 2009;284:23286–23292. doi: 10.1074/jbc.M109.028191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tiraby C, Hazen BC, Gantner ML, Kralli A. Estrogen-related receptor gamma promotes mesenchymal-to-epithelial transition and suppresses breast tumor growth. Cancer Res. 2011;71:2518–2528. doi: 10.1158/0008-5472.CAN-10-1315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lam SS, Mak AS, Yam JW, et al. Targeting estrogen-related receptor alpha inhibits epithelial-to-mesenchymal transition and stem cell properties of ovarian cancer cells. Mol Ther. 2014;22:743–751. doi: 10.1038/mt.2014.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu Y-M, Chen Z-J, Liu H, et al. Inhibition of ERRα suppresses epithelial mesenchymal transition of triple negative breast cancer cells by directly targeting fibronectin. Oncotarget. 2015;6:25588–25601. doi: 10.18632/oncotarget.4436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ariazi EA, Jordan VC. Estrogen-related receptors as emerging targets in cancer and metabolic disorders. Curr Top Med Chem. 2006;6:203–215. doi: 10.2174/1568026610606030203. [DOI] [PubMed] [Google Scholar]
- 58.Dwyer MA, Joseph JD, Wade HE, et al. WNT11 expression is induced by estrogen-related receptor alpha and beta-catenin and acts in an autocrine manner to increase cancer cell migration. Cancer Res. 2010;70:9298–9308. doi: 10.1158/0008-5472.CAN-10-0226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fradet A, Sorel H, Bouazza L, et al. Dual function of ERRα in breast cancer and bone metastasis formation: implication of VEGF and osteoprotegerin. Cancer Res. 2011;71:5728–5738. doi: 10.1158/0008-5472.CAN-11-1431. [DOI] [PubMed] [Google Scholar]
- 60.Sailland J, Tribollet V, Forcet C, et al. Estrogen-related receptor α decreases RHOA stability to induce orientated cell migration. Proc Natl Acad Sci USA. 2014;111:15108–15113. doi: 10.1073/pnas.1402094111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sonoda J, Laganière J, Mehl IR, et al. Nuclear receptor ERR alpha and coactivator PGC-1 beta are effectors of IFN-gamma-induced host defense. Genes Dev. 2007;21:1909–1920. doi: 10.1101/gad.1553007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yuk J-M, Kim TS, Kim SY, et al. Orphan nuclear receptor ERRα controls macrophage metabolic signaling and A20 expression to negatively regulate TLR-induced inflammation. Immunity. 2015;43:80–91. doi: 10.1016/j.immuni.2015.07.003. [DOI] [PubMed] [Google Scholar]
- 63.Bonnelye E, Vanacker JM, Dittmar T, et al. The ERR-1 orphan receptor is a transcriptional activator expressed during bone development. Mol Endocrinol. 1997;11:905–916. doi: 10.1210/mend.11.7.9948. [DOI] [PubMed] [Google Scholar]
- 64.Delhon I, Gutzwiller S, Morvan F, et al. Absence of estrogen receptor-related-alpha increases osteoblastic differentiation and cancellous bone mineral density. Endocrinology. 2009;150:4463–4472. doi: 10.1210/en.2009-0121. [DOI] [PubMed] [Google Scholar]
- 65.Teyssier C, Gallet M, Rabier B, et al. Absence of ERRalpha in female mice confers resistance to bone loss induced by age or estrogen-deficiency. PLoS One. 2009;4:e7942. doi: 10.1371/journal.pone.0007942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ijichi N, Ikeda K, Horie-Inoue K, et al. Estrogen-related receptor alpha modulates the expression of adipogenesis-related genes during adipocyte differentiation. Biochem Biophys Res Commun. 2007;358:813–818. doi: 10.1016/j.bbrc.2007.04.209. [DOI] [PubMed] [Google Scholar]
- 67.Rajalin A-M, Pollock H, Aarnisalo P. ERRalpha regulates osteoblastic and adipogenic differentiation of mouse bone marrow mesenchymal stem cells. Biochem Biophys Res Commun. 2010;396:477–482. doi: 10.1016/j.bbrc.2010.04.120. [DOI] [PubMed] [Google Scholar]
- 68.Bonnelye E, Merdad L, Kung V, Aubin JE. The orphan nuclear estrogen receptor-related receptor alpha (ERRalpha) is expressed throughout osteoblast differentiation and regulates bone formation in vitro. J Cell Biol. 2001;153:971–984. doi: 10.1083/jcb.153.5.971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kammerer M, Gutzwiller S, Stauffer D, et al. Estrogen receptor α (ERα) and estrogen related receptor α (ERRα) are both transcriptional regulators of the Runx2-I isoform. Mol Cell Endocrinol. 2013;369:150–160. doi: 10.1016/j.mce.2013.01.024. [DOI] [PubMed] [Google Scholar]
- 70.Calligé M, Richard-Foy H. Ligand-induced estrogen receptor alpha degradation by the proteasome: new actors? Nucl Recept Signal. 2006;4:e004. doi: 10.1621/nrs.04004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Macari C, Teyssier C, Tribollet V, et al. Estrogens repress PGC1-α expression in the uterus. Mol Cell Endocrinol. 2010;330:33–40. doi: 10.1016/j.mce.2010.08.003. [DOI] [PubMed] [Google Scholar]
- 72.Gallet M, Saïdi S, Haÿ E, et al. Repression of osteoblast maturation by ERRα accounts for bone loss induced by estrogen deficiency. PLoS One. 2013;8:e54837. doi: 10.1371/journal.pone.0054837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Vanacker JM, Delmarre C, Guo X, Laudet V. Activation of the osteopontin promoter by the orphan nuclear receptor estrogen receptor related alpha. Cell Growth Differ. 1998;9:1007–1014. [PubMed] [Google Scholar]
- 74.Zirngibl RA, Chan JSM, Aubin JE. Estrogen receptor-related receptor alpha (ERRalpha) regulates osteopontin expression through a non-canonical ERRalpha response element in a cell context-dependent manner. J Mol Endocrinol. 2008;40:61–73. doi: 10.1677/JME-07-0114. [DOI] [PubMed] [Google Scholar]
- 75.Zirngibl RA, Chan JSM, Aubin JE. Divergent regulation of the osteopontin promoter by the estrogen receptor-related receptors is isoform- and cell context dependent. J Cell Biochem. 2013;114:2356–2362. doi: 10.1002/jcb.24583. [DOI] [PubMed] [Google Scholar]
- 76.Boudjadi S, Bernatchez G, Beaulieu J-F, Carrier JC. Control of the human osteopontin promoter by ERRα in colorectal cancer. Am J Pathol. 2013;183:266–276. doi: 10.1016/j.ajpath.2013.03.021. [DOI] [PubMed] [Google Scholar]
- 77.Alaynick WA, Kondo RP, Xie W, et al. ERRgamma directs and maintains the transition to oxidative metabolism in the postnatal heart. Cell Metab. 2007;6:13–24. doi: 10.1016/j.cmet.2007.06.007. [DOI] [PubMed] [Google Scholar]
- 78.Cardelli M, Aubin JE. ERRγ is not required for skeletal development but is a RUNX2-dependent negative regulator of postnatal bone formation in male mice. PLoS One. 2014;9:e109592. doi: 10.1371/journal.pone.0109592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kubo M, Ijichi N, Ikeda K, et al. Modulation of adipogenesis-related gene expression by estrogen-related receptor gamma during adipocytic differentiation. Biochim Biophys Acta. 2009;1789:71–77. doi: 10.1016/j.bbagrm.2008.08.012. [DOI] [PubMed] [Google Scholar]
- 80.Jeong B-C, Lee Y-S, Park Y-Y, et al. The orphan nuclear receptor estrogen receptor-related receptor gamma negatively regulates BMP2-induced osteoblast differentiation and bone formation. J Biol Chem. 2009;284:14211–14218. doi: 10.1074/jbc.M808345200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kim E-J, Kang I-H, Lee JW, et al. MiR-433 mediates ERRγ-suppressed osteoblast differentiation via direct targeting to Runx2 mRNA in C3H10T1/2 cells. Life Sci. 2013;92:562–568. doi: 10.1016/j.lfs.2013.01.015. [DOI] [PubMed] [Google Scholar]
- 82.Kim J-H, Choi Y-K, Do J-Y, et al. Estrogen-related receptor γ plays a key role in vascular calcification through the upregulation of BMP2 expression. Arterioscler Thromb Vasc Biol. 2015;35:2384–2390. doi: 10.1161/ATVBAHA.115.306102. [DOI] [PubMed] [Google Scholar]
- 83.Han Y-H, Kim D-K, Na T-Y, et al. RORα switches transcriptional mode of ERRγ that results in transcriptional repression of CYP2E1 under ethanol-exposure. Nucleic Acids Res. 2016;44:1095–1104. doi: 10.1093/nar/gkv1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wei W, Wang X, Yang M, et al. PGC1beta mediates PPARgamma activation of osteoclastogenesis and rosiglitazone-induced bone loss. Cell Metab. 2010;11:503–516. doi: 10.1016/j.cmet.2010.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bonnelye E, Saltel F, Chabadel A, et al. Involvement of the orphan nuclear estrogen receptor-related receptor α in osteoclast adhesion and transmigration. J Mol Endocrinol. 2010;45:365–377. doi: 10.1677/JME-10-0024. [DOI] [PMC free article] [PubMed] [Google Scholar]