Abstract
Multiple myeloma (MM) is a tumor of terminally differentiated B cells that arises in the bone marrow. Immune interactions appear as key determinants of MM progression. While myeloid cells foster myeloma-promoting inflammation, Natural Killer cells and T lymphocytes mediate protective anti-myeloma responses. The profound immune deregulation occurring in MM patients may be involved in the transition from a premalignant to a malignant stage of the disease. In the last decades, the advent of stem cell transplantation and new therapeutic agents including proteasome inhibitors and immunoregulatory drugs has dramatically improved patient outcomes, suggesting potentially key roles for innate and adaptive immunity in disease control. Nevertheless, MM remains largely incurable for the vast majority of patients. A better understanding of the complex interplay between myeloma cells and their immune environment should pave the way for designing better immunotherapies with the potential of very long term disease control. Here, we review the immunological microenvironment in myeloma. We discuss the role of naturally arising anti-myeloma immune responses and their potential corruption in MM patients. Finally, we detail the numerous promising immune-targeting strategies approved or in clinical trials for the treatment of MM.
Keywords: Multiple myeloma, Tumor microenvironment, Immune responses, Immune escape, Immunotherapy
Introduction
Multiple myeloma (MM) is a tumor localized at various sites within the bone marrow (BM) [1]. With over 20,000 new cases diagnosed per year in the United States, MM represents 1 % of all cancers and approximately 10 % of all hematological malignancies. The median age at diagnosis is 65 years [2]. MM is clinically defined by the CRAB symptoms: hyperCalcemia, Renal insufficiency, Anemia and/or Bone lesions [3]. Autologous stem cell transplantation (SCT) in eligible patients, proteasome inhibitors and immunoregulatory drugs have substantially increased response rates and overall survival during the past two decades [4]. In spite of these tremendous improvements, MM remains a largely incurable disease with a median survival of 6 years.
Myeloma cells are the malignant counterpart of plasma cells, which are terminally differentiated B cells. Antibody-secreting plasma cells differentiate from naïve B cells that have recognized a foreign antigen [5]. This takes place in germinal centers of secondary lymphoid organs, where B cells undergo proliferation and somatic hypermutations followed by the selection of B cells with high antigen affinity. Plasmablasts exiting the germinal center migrate to the BM where they find an appropriate environment allowing them to differentiate into mature long-lived plasma cells [5, 6]. Similarly, myeloma cells depend on the BM microenvironment for their survival, growth and differentiation [7]. The primary function of long-lived plasma cells is the secretion of antibodies (immunoglobulin, Ig) that mediate humoral immunity against infections. In contrast to normal plasma cells, myeloma cells secrete monoclonal Ig (M-proteins) which are central to disease pathogenesis and serve as diagnostic marker detectable in the blood and urine of MM patients.
MM is a multistep progressing disease that starts with an asymptomatic premalignant lesion called monoclonal gammopathy of undetermined significance (MGUS). MGUS is present in 1 % of the adult population and progresses to malignant MM at the rate of 1 % per year [7]. Although MM develops in the BM, late stages may involve a loss of BM-dependency and the development of extramedullary tumors in the blood, liver, spleen, lymph nodes, pleural fluid and skin [8]. When a high percentage (>20 %) of malignant plasma cells is detected in the blood, the disease is then called plasma-cell leukemia. Malignant plasma cells arise from successive genetic lesions [9]. Early immortalizing events likely occur in germinal centers and involve translocations between Ig enhancers and oncogenes. Subsequently, secondary translocations activating proliferation and survival pathways contribute to increased tumor growth and extramedullary spread. Yet, the factors determining the progression from a premalignant MGUS stage to active myeloma are not well understood. Microarray expression analysis has revealed a large number of genes differentially expressed between plasma cells of healthy donors and those of MGUS/MM patients; but very few genes could distinguish MGUS from MM plasma cells [10]. Along with genetic changes in plasma cells, the BM microenvironment is believed to play a crucial role in disease progression to symptomatic myeloma. Immune cells are important components of this microenvironment. Here, we review the importance of the immune network in promoting or controlling myeloma growth. We describe the interactions between the different members of the immune system, the BM stroma and the myeloma cells. Finally, we discuss various strategies implemented to trigger the immune elimination of myeloma cells.
Role of the BM microenvironment in MM pathology
Immune composition of the BM microenvironment
MM develops in the BM, a well organized tissue residing in the cavities of bones. In adults, the BM is the primary site of hematopoiesis, the process by which hematopoietic stem cells give rise to the different types of blood cells including erythrocytes, megakaryocytes, platelets and immune cells. Besides providing hematopoietic stem cells with the specific microenvironmental niches required for their maintenance, proliferation and differentiation [11], the BM is also the primary residential site of plasma cells [5, 6]. Factors provided by highly specialized niches within the BM allow plasma cells to survive for years, even for decades. It is postulated that the same factors support the growth of myeloma cells.
The BM microenvironment consists of a cellular compartment, the extracellular matrix and soluble factors such as cytokines, chemokines and growth factors [12, 13]. BM residing cells can be subdivided into hematopoietic cells including immune cells, and non-hematopoietic cells such as stromal cells, adipocytes, osteoclasts, osteoblasts and components of the vasculature. Complex interactions between immune, non-immune and malignant myeloma cells influence MM progression (Fig. 1). The crucial role of immunity in the development and pathology of MM are the main focus of this review.
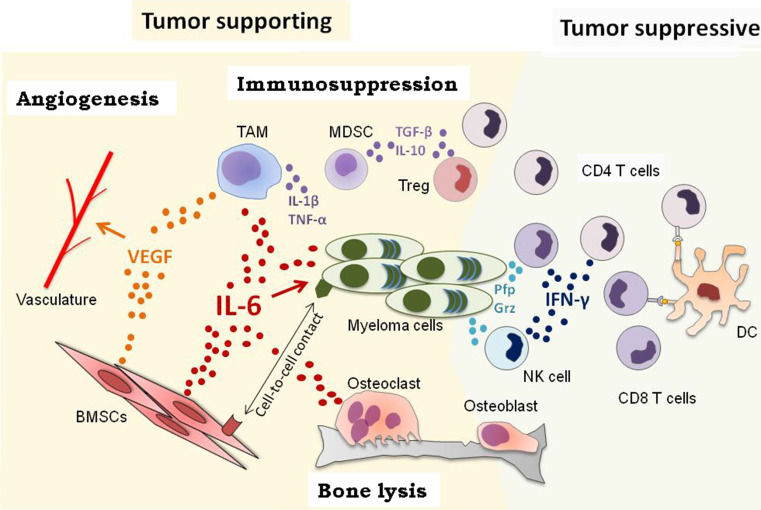
Fig. 1.
The MM BM microenvironment. Myeloma microenvironment include bone marrow stromal cells (BMSCs), bone forming (osteoblasts) and resorbing (osteoclasts) cells, the vasculature and immune cells. Reciprocal interactions between BMSCs and MM cells involve cell-to-cell contacts and soluble factors. BMSCs are the main source of IL-6, which is also produced by osteoclasts, tumor associated macrophages (TAMs) or MM cells. IL-6 promotes myeloma cell proliferation, survival and drug resistance. In addition, BMSCs and TAMs secrete vascular endothelial growth factor (VEGF) and thereby favor angiogenesis. Direct and indirect interactions involving MM cells, osteoblasts and osteoclasts imbalance the bone remodeling process and result in bone lysis. Myeloid derived suppressor cells (MDSCs) or regulatory T cells (Tregs) secrete immunosuppressive factors contributing to MM escape from the immune system. Dendritic cells (DCs) prime CD4 and CD8 T cells. Depending on their polarization, CD4 T cells either promote or inhibit tumor growth. IFN-γ secretion by T cells and NK cells may limit MM progression. Furthermore, CD8 T cells and NK cells directly kill tumor cells by releasing cytotoxic granules containing perforin (pfp) and granzymes (Grz)
The BM is a primary organ of hematopoiesis and therefore contains hematopoietic stem cells and progenitors of the myeloid and lymphoid lineages [11]. Myeloid cells such as monocytes, macrophages, dendritic cells (DCs) and granulocytes develop in the BM, are rapidly recruited to damaged or infected tissues and play major roles in early immune responses [14]. Their immature precursors may participate to MM pathology by favoring the proliferation of malignant plasma cells [15]. Monocytes differentiate into inflammatory macrophages or monocyte-derived DCs. Macrophages are phagocytes that contribute to inflammatory and healing responses through the secretion of diverse cytokines. Abundant in the BM of MM patients [16], macrophages have been shown to support the proliferation and the survival of myeloma cells [17]. DCs have major functions in the initiation and orientation of adaptive immune responses. Indeed, to acquire effector function, naïve T cells need to be ‘educated’ by antigen presenting cells (APCs). DCs are professional APCs that display antigens on major histocompatibility complex (MHC) molecules and deliver the appropriate signals (co-stimulation and cytokines) necessary for T cell activation. Distinct subsets of DCs harbor specific antigen presenting and immunoregulatory capacities [18]. The BM contains progenitors of conventional DCs as well as developing and mature plasmacytoid DCs (pDCs) [19]. In addition, circulating conventional DCs can migrate back to the BM where they may stimulate T cell proliferation [20]. Such local activation of T cell responses may have considerable impact on the T cell mediated control of MM. Granulocytes are subdivided into neutrophils, eosinophils and basophils [14]. Neutrophils are functionally impaired in MM patients [21], while eosinophils promote human and mouse myeloma cell growth [22, 23].
The lymphoid lineage comprises innate lymphoid cells (ILCs) and T and B lymphocytes. ILC progenitors, including the common helper ILC progenitor [24], group 2 ILC-restricted [25] and natural killer (NK) cell-restricted progenitors [26] are present in the BM. So far, NK cells have been the most studied members of the ILC family. NK cells play an important role in cancer immunology due to their capacity to directly recognize and kill tumor cells [27] and have received particular interest in MM [28]. B and T lymphocytes mediate adaptive immunity. Adaptive immune responses are antigen-specific but develop slower than innate responses carried on by myeloid cells or ILCs. An important feature of adaptive immune responses is the memory that allows faster and more potent responses following a subsequent encounter with the same antigen. Memory T cells certainly play a crucial role in controlling dormant tumor cells and preventing relapse [29]. B cells complete their development in the BM whereas early T cell progenitors leave the BM to achieve their development in the thymus. In addition, the BM cellular compartment typically contains less than 5 % plasma cells [6, 30] and 1–5 % of re-circulating mature T cells [31, 32]. There are different subsets of T cells. CD8 T cells are cytotoxic lymphocytes that eliminate tumor cells in an antigen-specific manner. Memory CD8 T cells preferentially home to the BM where they undergo basal proliferation allowing the maintenance of a cytotoxic memory [31]. CD4 T cells, also called helper T cells (Th), secrete cytokines that regulate immune responses [33]. They also help CD8 T cells functions as well as B cell differentiation into long-lived antibody-secreting plasma cells. Depending on the signals they receive, naive CD4 T cells differentiate into different helper lineages with distinct cytokine secretion profiles. For instance, Th1 cells mainly produce interferon (IFN)-γ whereas Th2 cells secrete IL-4, IL-5, IL-10 and IL-13. Regulatory T cells (Treg) are another CD4 T cell subset characterized by the expression of the Foxp3 transcription factor. These cells down-regulate immune responses and are often, albeit not always, associated with poor outcome in cancer patients [34]. Interestingly, the BM is particularly rich in Tregs, which represent 25 % of CD4 T cells in this organ [35]. High numbers of immature myeloid cells and Tregs in the BM indicate a tolerance-prone microenvironment that may hamper the development of protective immune responses against MM.
Dependency of myeloma cells on the BM microenvironment
Even though extramedullary disease is detected in 7–18 % of newly diagnosed MM patients [36], myeloma cells are believed to be strictly confined to the BM in the early stages of the disease [7]. The observation that MM cells do not proliferate when cultured alone highlights the strong dependency of these cells on microenvironmental factors [30].
The chemokine stromal derived factor-1 (SDF-1 or CXCL12) is a key regulator of myeloma cell homing to the BM [37]. CXCL12 is produced by BM stromal cells (BMSCs) and interacts with CXCR4 on myeloma cells. In addition, the retention of myeloma cells within the BM is ensured by a range of interactions between myeloma cells, the BMSCs and the extracellular matrix. For instance, syndican-1 (CD138), CD44, CD38 and integrins expressed by myeloma cells bind to various components of the extracellular matrix and serve as major anchors mediating physical interactions between malignant plasma cells and the solid textures of the BM [37]. Importantly, these receptors do not only mediate adhesion but also initiate signaling cascades within myeloma cells that contribute to their proliferation and survival.
The cytokine IL-6 is probably the most important factor sustaining MM growth in the BM [38]. In fact, the loss of IL-6 dependency observed in advanced disease stages may facilitate the colonization of extramedullary sites by myeloma cells [39]. It is interesting to note that normal and malignant plasma cells respond quite differently to IL-6 stimulation: IL-6 increases Ig production by normal plasma cells but stimulates proliferation and resistance to apoptosis in MM cells [40]. BMSCs as well as T cells, B cells, monocytes or myeloma cells themselves produce IL-6 [41]. BMSCs are considered as the predominant source of IL-6 in MM [42]. Still, the role of immune cells in IL-6-driven myeloma pathology should not be neglected. For instance, macrophages promote the proliferation of human MM cells in an IL-6-dependent manner [17]. Interestingly, mouse eosinophil-derived IL-6 contributes to the maintenance of long-lived plasma cells in the BM [43]. Similarly, human eosinophils have been shown to enhance the proliferation of MM cell lines, even though the IL-6-dependency of this phenomena was questioned [22]. Despite being a key myeloma growth factor, IL-6 blockade with monoclonal antibodies has shown disappointing results in MM patients when administered with conventional chemotherapeutics [44]. This suggests that IL-6 inhibition is likely to be redundant for disease control during currently standard therapies.
B-cell activating factor (BAFF) and a proliferation-inducing ligand (APRIL) are related members of the TNF superfamily whose receptors are expressed on B cells at different stages of differentiation [5]. BAFF is necessary for the early stages of human plasmablast differentiation whereas long-term survival of plasma cells is APRIL-dependent [6]. Human primary myeloma cells express receptors for BAFF and APRIL [45]. Addition of these growth factors to an IL-6-deprived milieu rescues IL-6-dependent myeloma cell lines from apoptosis [46]. In the myeloma-infiltrated BM microenvironment, monocytes and neutrophils are the mains source of BAFF, while APRIL is produced by monocytes and osteoclasts [47]. Moreover, APRIL is expressed by mouse BM eosinophils that support normal plasma cell survival [43], but the role of APRIL production by eosinophils in MM requires further investigation.
Although IL-6, BAFF and APRIL are probably the key proliferation and survival factors for myeloma cells, other factors contribute to the MM pathology. In late MM stages, insulin-like growth factor (IGF)-1 may drive proliferation and survival of IL-6-independent myeloma cells [40]. Additional growth factors and cytokines promoting MM growth include G-CSF, GM-CSF, SCF, TNF-α, HGF, IL-3, IL-10, IL-15, IL-17, IL-21, vascular endothelial growth factor (VEGF) and osteopontin [8, 12, 48].
Impact of myeloma on the BM microenvironment
The BM provides survival niches for both normal and malignant plasma cells. In MM, malignant plasma cells hijack the diverse components of this microenvironment to further sustain MM growth and development. For instance, myeloma cells induce BMSCs, osteoblasts and immature myeloid cells to produce IL-6, thereby promoting their own proliferation [8, 15, 49]. Importantly, myeloma perturbs normal bone remodeling, promotes angiogenesis and causes immune deficiencies.
Bone destruction is a key pathological feature of MM. Development of focal lytic bone lesions or diffuse osteopenia leads to spontaneous fractures and increased calcium release that largely contribute to MM morbidity and mortality [40]. A study performed in a humanized severe combined immunodeficient (SCID) mouse model suggested that bone remodeling might also contribute to MM progression [50]. This process involves multiple interactions between myeloma cells, BMSCs, and bone forming and resorbing cells and their progenitors. In physiological conditions, osteoclasts clear away old bone tissue while osteoblasts create new bone. In MM, the balance between bone resorption and bone formation is disturbed. Myeloma cells inhibit osteoblast differentiation and promote osteoclast differentiation and activity [13]. Several factors have been involved in myeloma-induced osteolysis. Interactions between the receptor activator of NF-κB (RANK) and its ligand (RANKL) are thought to play a crucial role in this process [40]. Even if immune cells have been poorly investigated in the context of myeloma bone disease, few studies indicated a pivotal role of T cells. In MM patients, T cells are the main source of IL-3, a cytokine that triggers osteoclast formation while blocking osteoblast formation [51]. In addition, human MM cell lines induce RANKL expression on T cells, thus favoring osteoclastogenesis [52]. Interestingly, IL-17-producing T cells were shown to induce osteoclast activation and the levels of IL-17 were found to directly correlate with lytic bone disease in MM patients [53]. Thus, T cells significantly contribute to myeloma-induced osteolysis. Further work should determine whether other members of the immune system are also involved.
Angiogenesis is increased in patients with active MM, in comparison with MGUS or smoldering MM patients [54] and BM microvessel density has emerged as an independent prognosis factor in myeloma [55]. Inflammatory cells recruited and activated within the tumor microenvironment trigger the angiogenic switch [56]. In particular, MM-associated macrophages were shown to promote neovascularization [57]. Intriguingly, in solid tumors, vessels derived from neoangiogenesis show impaired structure and function, thus influencing leukocytes recruitment from the blood [58]. In these settings, angiogenesis blockade could reverse immunosuppression. The role of angiogenesis on the immune composition of the BM microenvironment remains to be investigated in MM.
It is widely recognized that MM patients have greater susceptibility to infections and secondary malignancies [59]. Immune dysfunctions are the consequences of both niche-occupancy and direct immunosuppression by malignant plasma cells [60]. Specific immune deregulations and their impact on anti-myeloma responses will be discussed later.
The BM niche protects myeloma cells from chemotherapy
Reciprocal interactions involving myeloma cells and the BM milieu contribute to the resistance to conventional chemotherapeutic agents. Therefore, novel therapeutic approaches aim to target not only the malignant cells, but also myeloma cell-stromal cell interactions and the BM microenvironment [51].
Response of myeloma cells to conventional therapies, such as glucocorticoids or cytotoxic chemotherapeutics, is attenuated by the presence of BMSCs [40]. The concept of cell-adhesion mediated drug resistance (CAM-DR) was first introduced in 1999 to describe the role of fibronectin-adhesion in protecting myeloma cells against apoptosis when exposed to cytotoxic agents. Fibronectin or BMSCs induce CAM-DR to a variety of drugs (e.g. bortezomib, vincristine, doxorubicin and dexamethasone) and integrins expressed by malignant plasma cells play a key role in this process [37]. In addition to cell-to-cell contacts, soluble factors such as IL-6 contribute to myeloma cell resistance to chemotherapy [61]. The role of immune cells in drug resistance has been poorly investigated. Still, macrophages were found to protect myeloma cells from dexamethasone-, melphalan-, bortezomib- and doxorubicin- induced apoptosis [16, 62].
Furthermore, hematopoietic stem cell niches in the BM might promote the survival of MM stem cells. Even if the concept of cancer stem cell in MM remains controversial, these cells have been proposed to be the root cause of drug resistance [63].
Spontaneous immune responses to MM
Myeloid cells promote inflammation in the MM microenvironment
Inflammation is one of the hallmarks of cancer [64]. It has been well established that the inflammatory microenvironment facilitates proliferation, invasion, and metastasis of malignant cells in solid tumors [65]. In this context, myeloid cells are key inflammatory mediators that produce proinflammatory cytokines through recognition of diverse pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs) by their pattern-recognition receptors [66, 67]. Recently, subpopulations of tumor-associated myeloid cells have gained prominence due to their immunosuppressive functions. These cells include myeloid-derived suppressor cells (MDSC) and tumor-associated macrophages (TAM) [68]. Although these myeloid cells have been intensively studied in solid tumors, there is emerging evidence that they are key players in BM milieu of hematological malignancies including MM [57].
MDSC
MDSC are heterogeneous immature myeloid cells which are characterized by a potent ability to suppress anti-tumor immune responses mediated by T cells and NK cells [68, 69]. Under pathological conditions including cancer, perturbation of normal differentiation of myeloid cells leads to generation of MDSC, which is triggered by persistent exposure to tumor microenvironment-derived soluble factors such as stem-cell factor, GM-CSF, prostaglandins, IL-6, and VEGF [70]. MDSC are subsequently recruited into tumor site or lymphoid tissues in response to CCL2 [71], CXCL5 [72], and S100 proteins [73]. Initially, MDSC were identified in tumor-bearing mice as Gr-1+CD11b+ cells [74, 75]. Phenotypically, MDSC can be divided into granulocytic subset (CD11b+Gr-1highLy6G+Ly6Clow G-MDSC) or monocytic subset (CD11b+Gr-1midLy6G−Ly6Chigh MO-MDSC) [68]. Accordingly, two subsets of MDSC possess different suppressive mechanisms: G-MDSC chiefly use reactive oxygen-species (ROS) such as hydrogen-peroxidase, whereas MO-MDSC use inducible nitric oxide synthase (iNOS) and arginase. Hydrogen-peroxide and iNOS-derived peroxynitrite inhibit T-cell receptor signal transduction [68], whereas arginase sequestrates l-arginine that is required for T cell proliferation [76], both of which dampen T cell-mediated anti-tumour immune responses in a cell-to-cell contact dependent manner. In addition to their direct immunosuppressive activities, MDSC are capable of inducing Tregs. Though the exact mechanism has not been fully understood, diverse molecules are reported to be implicated in the cross-talk between MDSC and Treg cells including arginase [77], CD40 [78] or cytokines (TGF-β, IFN-γ and IL-10) [79]. Moreover, MDSC stimulate tumor angiogenesis through secretion of MMP-9 or direct differentiation into CD31+ endothelial cells [80]. Thus, MDSC have multifaceted pro-tumor functions in tumor microenvironment. Recently, several studies have shown that MDSC are important players in myeloma-infiltrated immune microenvironments. In ATLN and DP42 murine myeloma models, both the proportion and the absolute number of G-MDSC and MO-MDSC in BM were significantly increased as early as 1 week after inoculation, and thereafter gradually decreased due to progressive expansion of myeloma cells [81]. Similar results were reported by another group in 5TMM models [82, 83], suggesting that the expansion of MDSC is an early event in MM. Proinflammatory S100 proteins play a pivotal role in the accumulation of MDSC in solid tumors [73]. Notably, S100A9-deficient mice showed prolonged survival compared to wild type mice after inoculation of OVA-expressing DP42 cells, which was associated with a reduction of MDSC in BM and an increase in OVA-specific CD8+ T cells. Furthermore, the survival benefit in S100A9-deficient mice was abrogated by antibody depletion of CD8+ T cells or adoptive transfer of MDSC [81], demonstrating that MDSCs dampen CD8+ T cell-dependent anti-myeloma immune responses, leading to myeloma progression.
In humans, Brimnes et al. [84] firstly reported that newly diagnosed MM patients have increased frequencies of CD14+ MO-MDSC in peripheral blood compared to healthy donors. On the contrary, recent studies showed that CD14−CD15+ G-MDSC, but not CD14+ MO-MDSC are increased in myeloma patients [81, 85, 86], while both subsets show the same level of suppressive activity against autologous T cells [86, 87]. In addition to their immunosuppressive activities, MDSC can directly stimulate proliferation of myeloma cells. Görgün et al. [85] showed that co-culture with MDSC marked enhanced proliferation of myeloma cells in vitro. Importantly, in this study, co-culture of myeloma cells and peripheral blood mononuclear cells (PBMC) from healthy donors was able to induce generation of MDSC, providing evidence for bidirectional interaction between myeloma cells and MDSC. Moreover, Favaloro et al. [86] reported that MDSC from MM patients can markedly induce Treg after co-culture with PBMC. Thus, MDSC contribute to immunosuppressive, tumor-favoring environment, providing a potential target in myeloma therapy.
Myeloma-associated macrophages
It is now established that tumor tissues are abundantly infiltrated by TAMs, which support tumor progression by angiogenesis, matrix remodeling and potent immunosuppression [88, 89]. In general, higher level of TAMs correlates with poor prognosis in many types of solid tumors [90] as well as hematological malignancies including MM [91]. In terms of ontogeny of TAMs, both tissue-resident macrophages and recruited macrophages coexist in tumor microenvironment [92]; however, recent studies have clarified that TAMs are phenotypically distinct from residential macrophages, and originate from circulating Ly6C+ inflammatory monocytes [71, 93, 94]. Colony-stimulating factor-1 receptor (CSF1R) signaling and CCL2-CCR2 interaction are implicated in the recruitment of monocytes to tumor microenvironment [71, 95] where differentiation and functional maturation of TAMs are regulated by Notch signaling and environmental factors such as hypoxia [94, 96, 97]. It remains unknown whether myeloma-associated macrophages originate from circulating monocytes or BM residential precursors; however, massive BM infiltration by CD68+ macrophages are observed in active MM patients, but not in MGUS patients or healthy donors [16, 98], indicating that macrophages represent a pivotal cellular component of the myeloma microenvironment. Myeloma-associated macrophages contribute to myeloma pathology by at least three different ways.
Firstly, myeloma-associated macrophages support myeloma growth through cytokine production. Importantly, myeloma-associated macrophages highly express IL-1 and TNF-α [99, 100], both of which stimulate production of IL-6 from mesenchymal stem cells (MSCs). Additionally, myeloma-associated macrophages secrete the anti-inflammatory cytokine IL-10, another growth factor for myeloma cells [100, 101]. Until recently, it remained unclear whether or not myeloma-associated macrophages produce cytokines in response to specific PAMPs and/or endogenous DAMPs in the myeloma microenvironment. Hope et al. firstly showed that myeloma-associated macrophages contribute to the inflammatory milieu through toll-like receptor (TLR)-2/6-mediated recognition of its proteoglycan agonist, versican. Furthermore, they also found that genetic ablation of tpl2 (Cot/MAP3K8), a downstream effector of TLRs, delays myeloma progression in Vk*myc transgenic mice [100], highlighting the importance of this pathway.
Another important function of myeloma-associated macrophages is angiogenesis and vasculogenesis. Angiogenesis within the myeloma microenvironment is amplified by a positive feedback loop of proangiogenic factors including VEGF, basic fibroblast growth factor (bFGF), TNF-α, and IL-6 [102]. In addition to secretion of these proangiogenic factors, myeloma-associated macrophages contribute to angiogenesis through a commitment toward an endothelial phenotype. Scavelli et al. [98] reported that exposure to VEGF and bFGF convert myeloma-associated macrophages into cells which are functionally and phenotypically similar to endothelial cells, leading to formation of capillary-like structures.
Lastly, myeloma-associated macrophages support myeloma cells in a cell-to-cell contact dependent manner. Zheng et al. [16] reported that myeloma-associated macrophages protect myeloma cells from caspase-dependent apoptosis, which confers resistance to chemotherapy. Notably, IL-6 was dispensable in this mechanism. Instead, macrophage-mediated myeloma survival was depend on interaction between P-selectin glycoprotein ligand-1 (PSGL-1)/selectins and ICAM-1/CD18 which transmit survival signaling including Src, Erk1/2 and c-myc [62].
NK cells mediate early responses to MM
NK cells are ILCs which play a key role in tumor immunosurveillance [27]. They express a wide range of germline-encoded receptors that allow them to recognize stressed or unhealthy cells such as tumor cells. NK cells directly kill the target cells by releasing lytic granules containing granzymes and perforin or through the membrane death receptors TNF-related apoptosis inducing ligand (TRAIL) and Fas ligand (FasL). NK cells also secrete a large array of cytokines and chemokines, among which IFN-γ is known for its potent anti-tumor properties. In humans, NK cells are often characterized as CD3−CD56+ lymphocytes, which are further divided into two populations: CD56dimCD16+ and CD56brightCD16− cells [103]. Noteworthy, CD56 expression by malignant plasma cells represents an obstacle to NK cell analysis in MM patients, even if size parameters should allow the distinction between NK cells and myeloma cells [104].
NK cells recognize and kill myeloma cells
The importance of NK cells for the control of myeloma progression has been demonstrated using NK cell-depleting antibodies in various mouse MM models [105, 106]. Furthermore, several groups established the ability of human NK cells to kill MM cell lines [107–109]; and cytotoxic activity of autologous NK cells against patient-derived myeloma targets has also been reported [108, 110].
A particularity of NK cells is their ability to sense cells that have down-regulated MHC class I molecules. Human NK cells express various combinations of killer cell immunoglobulin–like receptors (KIR) that deliver negative signals upon binding to MHC class I molecules, thus preventing reactivity against normal healthy cells [111]. MHC class I down-regulation is frequently observed in cancer cells. Accordingly, early stages myeloma cells express low levels of MHC I, and are readily recognized by NK cells [108].
In addition, myeloma cell recognition by NK cells involves various activating receptors including NKG2D, DNAX accessory molecule (DNAM-1 or CD226) and the natural cytotoxicity receptors (NCRs) NKp46, NKp30, NKp44 [107, 108]. NKp46 is certainly a key receptor for NK cell recognition of malignant plasma cells because its inhibition strongly reduced NK cell-mediated killing of all the myeloma cell lines so far tested [107]. Human NKG2D binds to MHC class I related chain A and B (MICA/MICB) and to UL16 binding proteins (ULBP1-6). ULPB1-3 has been detected on some myeloma cell lines while high levels of MICA were observed on BM-derived MM cells [108]. The NK cell-mediated killing of MICA-expressing myeloma cells was found to be NKG2D-dependent [107]. Moreover, nectin-2 (CD112) and the poliovirus receptor (PVR, CD155), the two known DNAM-1 ligands, are heterogeneously expressed on malignant plasma cells. Indeed, a study including 12 MM patients revealed CD155 and/or CD112 expression on all but two samples [107]. Blocking DNAM-1 inhibited the in vitro killing of CD155-expressing myeloma cell lines [107]. Importantly, the role of DNAM-1 in controlling myeloma progression has been investigated in vivo, in Vk*myc transgenic mice that spontaneously develop MM [105]. In this study, DNAM-1+/+, DNAM-1+/− and DNAM-1−/− Vk*myc mice were monitored for disease development and survival over 800 days. Mice lacking DNAM-1 exhibited higher levels of serum monoclonal protein and succumbed earlier to MM. Although this work highlights the importance of DNAM-1 in MM immunosurveillance, the role of DNAM-1 for NK cell-mediated control of MM growth is still to be demonstrated because, akin to NKG2D, DNAM-1 is not a NK cell-specific receptor but is also expressed on T cells.
Most studies have focused on the cytolytic activity of NK cells against MM cells. However, little is known about NK cell-derived IFN-γ in this context. Interestingly, IFN-γ-deficient mice injected with MM cell lines show shorter survival associated with higher tumor burden, when compared with WT mice [105]. IFN-γ not only stimulates innate and adaptive immune responses [112], but it also inhibits the in vitro proliferation of myeloma cells [113] and interferes with the RANKL signaling pathway to decrease osteoclastogenesis [114]. Thus, IFN-γ production by NK cells may significantly reduce MM pathology and this pathway would require further investigation.
Impaired NK cell activity in MM
An early report described increased numbers of CD56+CD3− NK cells in the BM and blood of newly diagnosed myeloma patients [104]. Subsequent studies confirmed that patients with MGUS or active myeloma present elevated numbers of circulating NK cells [115]. Surprisingly, patients with higher numbers of NK cells at diagnosis were found to have worse prognoses [104]. In fact, increased NK cell numbers may be seen as an unsuccessful attempt of the immune system to control myeloma cell expansion. It is now well established that NK cell activity is largely compromised in MM patients since various mechanisms contribute to impair NK cell recognition and killing of myeloma cells.
Immune escape of cancer cells involves two mechanisms: the immunoediting of tumor cells and the suppression of immune functions [116]. Both phenomena have been observed in MM. Interestingly, NK cell receptor ligands on myeloma cells are progressively edited during myeloma progression, outlining the role of NK cell control in the early stages of the disease and suggesting that impairment of NK cells responses may constitute a major event in promoting MGUS progression to MM. Indeed, malignant plasma cells or myeloma cell lines derived from early-stage MM/MGUS patients exhibit higher levels of MICA or Fas than plasma cells obtained from patients with active disease or cell lines derived from late-stage pleural effusions [108, 117, 118]. In addition, two studies observed a down-regulation of MHC class I molecules on the surface of plasma cells from early stages but not late stages MM patients [108, 117]. Of note, these results contrast with a third study that reported opposite observations i.e. an up-regulation of MHC class I molecules on BM plasma cells from MGUS patients compared with healthy donors and MM patients [119]. Still, myeloma cell-lines established from the BM are sensitive to NK cell-mediated lysis whereas cell-lines generated from pleural effusions from the same donor are resistant [108]. Likewise, increased degranulation of NK cells in the BM of MM-bearing mice could only be observed at early time points of disease development [106]. MICA shedding from the surface of malignant plasma cells generates soluble MICA that may contribute to altered NKG2D expression and defective NK cell functions [118]. While decreased NKG2D expression on NK cells from MM patients has been confirmed by another study, the role of soluble MICA in this process has been questioned [120]. In addition to NKG2D, other activating receptors showed reduced expression on NK cells from active myeloma patients. These include DNAM-1, 2B4/CD244 as well as the low affinity Fc receptor CD16 [107, 121]. Therefore, altered expression of activating receptors is likely to contribute to myeloma cell escape from cancer immunosurveillance. A recent study indicated that skewed chemokine levels hinder NK cell trafficking to the BM during the early asymptomatic stages of the disease [106]. This may represent another mechanism that contributes to myeloma cell escape from NK cell control.
T cell responses are altered in MM
Compared with healthy donors, CD4/CD8 T cell ratios are decreased in the blood of MM patients [122]. Soluble factors present in the MM microenvironment (e.g. TGF-β, IL-10 and VEGF) along with defective antigen presentation by DCs may lead to deficient T cell responses in MM patients [123, 124].
Alterations of DC functions result in impaired T cell priming
Though BM is a primary lymphoid organ, BM also functions as a secondary lymphoid organ where T cells responses are initiated [125]. In this context, efficient uptake and processing of circulating tumor-associated antigens by BM CD11c+ DCs is critical for the priming of T cell-mediated anti-tumor immune responses. Many studies concluded that DCs from MM patients have impaired T-cell stimulation capacities, whereas contradictory results exist regarding the frequency and phenotype of DCs [126–129]. Several soluble factors including IL-6, TGF-β and IL-10 seem to be involved in the impairment of DC functions [127, 128]. Recently, Leone et al. reported that DCs accumulate in BM during the MGUS-to-MM progression. In this study, DCs purified from MGUS/MM patients were able to engulf apoptotic myeloma cells, cross-present them and activate tumor-specific CD8+ T cells whereas CD28–CD80/86 interaction between live myeloma cells and DCs down-regulated expression of proteasome subunits in myeloma cells [130]. This mechanism may enable myeloma cells to evade CD8+ T-cell killing in spite of efficient T cell priming.
pDCs, the other major subset of DCs, are also involved in myeloma pathology. pDCs play pivotal roles for generation of normal plasma cells and antibody responses through secretion of type I IFN and IL-6 [131]. Chauhan et al. [132] showed that numbers and frequency of BM pDCs are increased in MM patients and that pDCs confer growth, survival, chemotaxis, and drug resistance in myeloma cells.
Myeloma-specific T cell responses
Mouse models support an instrumental role of cytotoxic CD8 T cells in MM immunosurveillance [105]. Several pieces of evidence indicate that myeloma cells express tumor antigens able to trigger T cell responses. Analysis of the T cell receptor (TCR) variable gene repertoire revealed clonal expansions of CD8 T cells in MGUS and early stage MM patients that probably reflect chronic stimulations with myeloma-derived antigens [133]. Tumor-specific T cells able to lyse autologous myeloma cells can be generated from the blood or BM of myeloma patients using myeloma lysate-pulsed DCs [134, 135]. Nonetheless, T cells freshly isolated from MM patients fail to recognize autologous tumor cells and to secrete IFN-γ, suggesting that they probably do not exert a strong anti-myeloma activity in vivo [134]. Conversely, freshly isolated T cells from the BM of MGUS patients produce IFN-γ when stimulated in vitro with DCs loaded with autologous tumor cells [136]. These data suggest that the anti-myeloma activity of tumor antigen-specific T cells is lost during the progression from MGUS to active myeloma. Noteworthy, T cells reactive against the embryonal stem cell-associated antigen SOX2 have been detected in MGUS but not MM patients [137]. SOX2 was reported to be expressed in a progenitor fraction of myeloma cells and anti-SOX2 T cell immunity correlates with a favorable outcome. In addition, patients that survived more than 10 years present expanded cytolytic T cell clones that, unlike the majority of MM patients, respond to stimulation by proliferating and producing IFN-γ [138]. Interestingly, T cells isolated from MGUS or MM patients are activated by DCs loaded with autologous but not allogeneic tumor lysates [134–136]. This indicates that T cell responses against MM are specific of each myeloma clone and differ from one patient to another. The antigenic properties of the variable region of the secreted monoclonal protein (idiotope) have been extensively studied [139]. Unfortunately, idiotype-specific responses are usually hinder by several tolerance mechanisms, including the deletion of high avidity idiotype-specific T cells [140]. In addition to idiotopes, general tumor antigens are shared among MM cells from different patients. Those include NY-ESO-1, MAGE-A3, Muc-1, sperm protein 17, PRDI-BF1 and XBP-1 and CD138 [139]. Adoptive transfer of T cells engineered to express a high affinity TCR for a myeloma-specific antigen represents an attractive therapy. As a example, the infusion of NY-ESO-1-specific engineered T cells recently showed promising results in a phase I/II clinical trial [141].
Helper T cell subsets in MM
Helper T cells play pivotal roles in adaptive immune responses and imbalanced polarization of CD4 T cell responses could largely impact on MM growth. Several reports describe a deregulated cytokine network in MM but not all of them agree on the nature of the changes. An early study established that T cells from MGUS patients stimulated with autologous monoclonal IgG are more efficient producers of IL-2 and IFN-γ when compared with idiotype-reactive T cells from late MM patients [142]. In the same line, increased IL-4 production by T cells from MM patients indicated that a shift toward Th2 polarization emerges with disease progression. This hypothesis is supported by another study describing decreased levels of IFN-γ and increased levels of IL-10 and IL-4 in the serum of 62 myeloma patients compared with 50 healthy donors [143]. IL-6 production by T cells may contribute to decreased Th1 responses in MM patients [144]. However, elevated Th1/Th2 ratios in the blood of MM patients in initial diagnosis and refractory phase have also been reported [145, 146] and high percentages of IFN-γ producing T cells were observed in MM patients [147]. Further work is needed to determine whether these discrepancies could account for variations in Th1/Th2 polarization during the course of the disease or may be explained by differences between BM and peripheral blood. Likewise, the role of Treg responses in MM is still unclear. An initial study demonstrated decreased FoxP3-expressing Tregs in spite of elevated percentages of CD25+CD4+ cells in the blood of MGUS and MM patients [148]. This group suggested that CD25+ T cells from MGUS or MM patients failed to suppress the proliferation of PBMC stimulated with anti-CD3 and concluded that MM Tregs were dysfunctional. However, one caveat in this assay is the intrinsic defect in the proliferation of PBMC isolated from MM patients. It was later established that CD4+CD25hi Tregs from MM patients are as efficient as Tregs from healthy donors at suppressing allogeneic T cell proliferation [149]. Results are still conflicting regarding Treg frequencies that are alternatively described as increased [138, 149, 150] or reduced [147, 148] in MM patients. Interestingly, patients with high peripheral Treg frequencies show reduced survival [150]. Th17 cells are a pro-inflammatory subset of CD4 T cells that produces IL-17 and IL-22. IL-6 plays a pivotal role in dictating the balance between Tregs and Th17 cells [151]. Treg/Th17 ratios were reportedly increased in MM patients, albeit lower in patients with long-term survival [138]. Yet, increased proportions of IL-17-producing CD4 T cells and increased serum concentrations of the Th17-associated cytokines IL-1β, IL-6, IL-17, IL-21, IL-22 and IL-23 have been observed in MM patients, when compared with healthy controls [48, 147, 152]. IL-17 might contribute to MM pathology as it induces the proliferation of MM cell lines in vitro [48] and promotes MM-associated bone lesions [53]. Finally, increased frequency of IL-22 and IL-13 double-producing T cells has been detected in the blood and BM of relapsed and late stage MM patients [153]. These Th22 cells are likely to sustain MM pathology since IL-22 favors the proliferation and resistance to drug-induced cell death of some MM cell lines and IL-13 indirectly promotes MM cell survival through the activation of BMSCs.
NKT cells recognize and respond to MM cells
NKT cells are characterized by the expression of both T cell and NK cell receptors. NKT cells recognize glycolipid antigens presented by the MHC-class I-like molecule CD1d and exert strong anti-tumor responses through direct cytotoxicity or release of pro-inflammatory cytokines, including IFN-γ [154]. Despite its absence on MM cell lines, CD1d is expressed by primary myeloma cells [155]. Lisophosphatidylcholine has been identified as a NKT cell ligand expressed on plasma cells from MM patients [156]. Interestingly, frequencies of lisophosphatidylcholine-recognizing NKT cells are dramatically increased in MM patients. Lisophosphatidylcholine stimulates IL-13 production by NKT and thus probably favors angiogenesis and tumor-promoting inflammation. Actually, NKT cells from MM patients are dysfunctional and unable to produce IFN-γ when stimulated with the glycolipid α-galactosylceramide [157]. Of note, similarly to conventional T cells, NKT cells isolated from MM patients can be rescued in vitro; and APC-stimulated NKT cells efficiently lyse primary autologous myeloma targets as well as CD1d-transfected MM cell lines [155, 157].
MR1-restricted mucosal associated invariant T (MAIT) cells are another type of invariant T cells that, similarly to NKT cells, have simplified patterns of TCR expression and respond immediately to antigen stimulation [158]. Albeit abundant in humans (5 % of total blood T cells), MAIT cells have not been investigated in the context of MM.
Harnessing the immune system to cure MM
Immunosuppression is an important characteristic of MM pathology [159]. Reversing this immunesuppression could potentially restore myeloma immunosurveillance and improve disease control (Fig. 2).
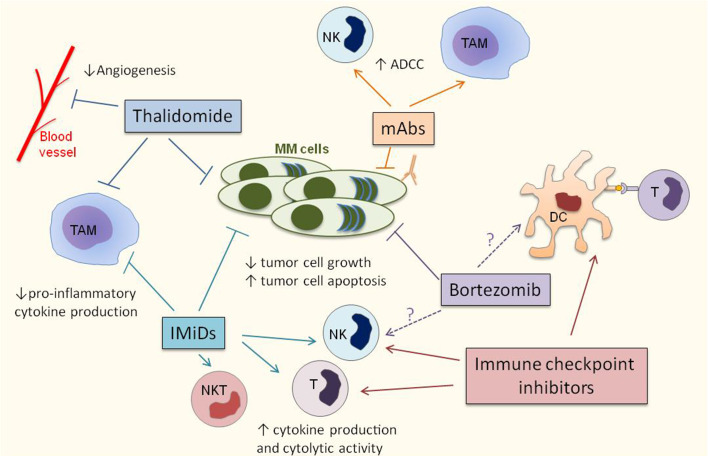
Fig. 2.
MM therapies modulate the immune microenvironment. Thalidomide, IMiDs, Bortezomib and monoclonal antibodies (mAbs) directly regulate tumor cell proliferation and survival. In addition, mAbs induce tumor cell death by triggering NK cell-mediated and tumor associated macrophage (TAM)-mediated antibody dependent cellular cytotoxicity (ADCC). Besides, thalidomide inhibits angiogenesis and both thalidomide and IMiDs decrease the production of pro-inflammatory cytokines by TAMs. IMiDs also promote the anti-MM activity of T cells, NK cells and NKT cells. The impact of bortezomib on anti-myeloma immune responses is unclear. Bortezomib may favor NK cell and dendritic cell (DC) functions by increasing the immunogenicity of MM cells; however toxic effects of bortezomib toward NK cells, DCs and T cells have also been described. Immune checkpoint inhibitors target the immune system to induce potent anti-tumor responses
Stem cell transplantation
Although MM remains an incurable malignancy, the introduction of autologous stem cell transplantation (SCT) following myeloablative treatment contributed significantly to the improved survival of MM patients observed in the last 15 years [160]. By introducing a new immune system and facilitating homeostatic lymphocytic proliferation in the setting of minimal residual disease, autologous SCT may overcome the acquired immune defects induced by myeloma. Absolute lymphocyte count recovery post-autologous SCT constitutes an independent prognostic factor for transplanted MM patients [161]. Intriguingly, Wolniak et al. [162] described a clonal population of CD8+CD57+ large granular lymphocytes in the BM and blood of MM patients post-autologous SCT. Although the specificity of these cells remains to be established, they might recognize tumor antigens and potentially drive graft-versus-myeloma (GvM) responses. Unfortunately, the GvM induced by autologous SCT, if it exists, is generally weak and most patients relapse. The transfer of marrow-infiltrating lymphocytes enriched in myeloma-specific T cells may enhance GvM effect [163]. An alternative is allogeneic SCT, which has the advantage of providing recipients with new T cell repertoire and triggers potent GvM effects against multiple minor histocomaptibility antigens [164]. The infusion of primed lymphocytes collected after donor immunization with a tumor-specific antigen may further enhance the GvM effect following SCT [165]. An interesting study used a pre-clinical humanized mouse model of MM to demonstrate the therapeutic potential of allogeneic T cell infusions [166]. Immunodeficient mice were used as a recipient for human MM cell lines and transferred or not with naïve allogeneic human T cells. In this model, a nonconventional population of double positive CD4+CD8+ T cells was induced in MM-bearing mice. These myeloma-induced alloreactive T cells produced IFN-γ and perforin and may mediate GvM responses. Nonetheless, because allogeneic SCT is associated with a high transplant-related mortality in the setting of myeloma, its place as a therapeutic strategy in this setting remains investigational [167].
Immunomodulatory drugs and proteasome inhibitors
Thalidomide is a glutamic acid derivative proved to be highly effective for the treatment of advanced MM patients [168]. The anti-angiogenic properties of thalidomide [169] together with its direct effect on MM cells [170] and its potent anti-inflammatory capacities [171] contribute to its anti-myeloma activity. Structural analogs of thalidomide have been selected based on their ability to inhibit TNF-α production. Among them, a class of compounds was found to significantly inhibit pro-inflammatory cytokine production by lipopolysaccharide (LPS)-stimulated PBMCs while increasing T cell responses to anti-CD3 stimulation [171]. These thalidomide analogs with unique immune regulatory properties are called immunomodulary drugs (IMiDs). Besides their direct anti-tumor effect [170], the real feature of IMiDs is their ability to promote host immunity while abrogating the protection conferred by the BM microenvironment [172]. Lenalidomide (Revlimid, CC-5013) and pomalidomide (Actimid, CC-4047) are the two most studied IMiDs. Both of them increase the cytotoxic activity of T cells [173, 174] and NK cells [175] against MM cells. In addition, lenalidomide further potentiates IFN-γ production by anti-CD3/anti-CD28 stimulated CD8 T cells from MM patients [174] and NK cells cultured for several days in the presence of lenalidomide produce higher levels of TNF-α and IFN-γ when stimulated with MM cell lines [176]. The lenalidomide-mediated up-regulation of TRAIL expression on NK cells could partially explain the enhanced cytotoxicity [176]. By contrast, pomalidomide-mediated enhancement of NK cell activity requires the presence of other cell populations [177]. IMiDs were found to stimulate IL-2 production by T cells and thereby indirectly trigger NK cell functions [178]. Furthermore, the ability of lenalidomide to boost NKT cell responses [155, 179] raises the possibility of its combination with NKT cell targeting approaches. Finally, lenalidomide may be particularly active in patients who have relapsed following allogeneic SCT [180–182]. These data are consistent with the immune stimulatory capacities of lenalidomide that likely boost endogenous GvM effects. In fact, an increase in activated T and NK cells has been observed in the blood of lenalidomide treated patients [182]. Surprisingly, two studies reported increased circulating Tregs during lenalidomide treatment of allogeneic SCT patients [180, 182]. However, these observations were made on very small cohorts comprising less than ten patients. Further studies should not only confirm the effect of IMiDs on immune cell frequencies/activation in the blood but also investigate how these new agents modulate immune responses in the BM of transplanted or non-transplanted MM patients.
The ubiquitin–proteasome pathway carries out protein turnover and its disruption induces the apoptosis of some cancer cells, including MM cells [183]. Bortezomib (Velcade), a proteasome inhibitor, has proven efficacy in MM [184]. In addition to sensitizing tumor cells to apotosis, bortezomib modulates host immune responses [185]. In vitro incubation of MM cells with bortezomib decreases their expression of MHC class I molecules while augmenting the display of activating NK cell receptor ligands [110, 186]. Such changes may facilitate NK cell recognition and killing of MM cells. Moreover, bortezomib induces immunogenic cell death of MM cells, thereby facilitating the DC-mediated elicitation of anti-myeloma T cell responses [187]. However, several reports indicated an immunosuppressive effect of bortezomib. Lymphopenia has been observed in about 10 % of bortezomib-treated patients [184] and is consistent with the in vitro toxicity of this drug toward lymphocytes [188]. Additional in vitro studies established the ability of bortezomib to inhibit DC [189] and NK cell [190] functions. Of note, bortezomib has been used at high concentrations in most experiments supporting an immunosuppressive effect [185]. Such effects may not occur in vivo, where immune cell exposure to this drug may be lower. Notably, bortezomib augments the anti-tumor effect of autologous NK cell infusions in mice [191]. Besides, the T and NK cell activating receptor DNAM-1 was found necessary for the therapeutic efficacy of bortezomib in MM-bearing mice [105]. Interestingly, early bortezomib treatment following allogeneic SCT protects mice from acute GvHD [192]. However, caution should be taken when combining bortezomib with allogeneic SCT because two subsequent studies in mice established that delayed bortezomib treatment post-transfer exacerbates GvHD-dependent mortality [193, 194]. While the mechanisms behind these observations remain unclear, bortezomib may differently regulate the distinct phases of GvHD.
Other proteasome inhibitors have generated interest for the treatment of MM. Carfilzomib is a second-generation proteasome inhibitor that demonstrated anti-myeloma efficacy in clinical studies [195].
Immunotherapy in MM
NK cell-based therapies
Several therapeutic agents exert their anti-MM efficacy at least in part through the recovery or the augmentation of NK cell responses [28]. As previously mentioned, new drugs such as thalidomide, IMiDs and proteasome inhibitors potentiate NK cell-mediated killing of MM cells. Furthermore, compared with T and B cells, NK cells reconstitute early following autologous SCT and may contribute to the success of this therapy [196]. NK cells are also important mediators of the GvM effects after T cell depleted allogeneic SCT, especially in the case of KIR-ligand incompatibility i.e. when donor NK cells do not express inhibitory KIRs recognizing host MHC class I molecules. Reduced relapse rates have been observed in MM patients receiving KIR-ligand mismatched allogeneic transplants [197]; and infusion of KIR-ligand mismatched NK cells followed by autologous SCT achieved 50 % of near complete remission in advanced MM patients [198]. Furthermore, IPH2101 (1-7F9), an anti-inhibitory KIR antibody could restore NK cell responses in relapsed/refractory MM patients [199]. Phase I clinical trials indicate that IPH2101 is well tolerated when given as a single agent or in combination with lenalidomide, but its efficacy against MM has yet to be proven [199, 200]. Alternatively, reprogramming NK cells with chimeric antigen receptors (CARs) specific for MM antigen could increase their reactivity toward myeloma cells. Myeloma cells express high levels of CD138 and a pre-clinical study demonstrated the ability of CD138-specific CAR NK cells to markedly delay MM growth and prolong survival in a xenograft model [201]. Finally, monoclonal antibody (mAb) therapy targeting myeloma cells have demonstrated clinical efficacy when combined with bortezomib or lenamidomide [202]. CD16 on NK cells binds to the constant region of Ig and certainly plays a key role in mAb therapies by triggering antibody-dependent cellular cytotoxicity (ADCC) of mAb-coated tumor cells. Among the different mAbs tested, daratumumab targets CD38, an ectoenzyme commonly used as a marker of myeloma cells. Daratumumab administered as a single agent has demonstrated anti-myeloma activity in clinical trials [203]. Elotuzumab is another successful mAb [204]. Elotuzumab recognizes CS1 (SLAMF7), a glycoprotein universally expressed on MM cells. Elotuzumab activity appears to be dependent on NK cell-mediated ADCC [205]. Interestingly, NK cells express low levels of CS1. The binding of elotuzumab to CS1 directly promotes NK cell activity and thus contributes to enhanced anti-tumor effects [206].
Immune checkpoint blockade
Immune checkpoints such as programmed-death 1 (PD-1) and cytotoxic T-lymphocyte associated protein 4 (CTLA-4) down-regulate T cell responses and thereby maintain self-tolerance. The use of mAbs to disrupt the receptor-ligand interactions involved in these pathways has shown remarkable results in melanoma [207]. Immune checkpoint modulation also holds promise for the treatment of hematological malignancies [208]. The high levels of PD-1 observed on NK and T cells from MM patients together with the expression of PD-1 ligand (PD-L1) on MM cells [209, 210] strongly encouraged the investigation of PD-1 blockade in MM patients. Yet, a phase 1 clinical trial using anti-PD1 mAb nivolumab in MM patients yielded disappointing initial results, with no objective responses [208]. Noteworthy, disease remained stable in 18 of 27 patients, indicating that PD-1 blockade might still have an effect in MM and could be efficient in combination with other therapeutics. In vitro studies suggested that PD-1 blockade by pidilizumab (CT-011) would synergically combine with lenalidomide [210] or with a DC/Myeloma fusion vaccine [209]. Consequently, a phase 2 clinical trial is currently ongoing to assess the efficacy of a DC/tumor vaccine in conjunction with pidilizumab following autologous SCT (NCT011067287) [164]. Additionally, a preclinical study indicated that blocking PD-L1 together with other immune checkpoints (CTLA-4, LAG-3 or TIM-3) promotes the survival of MM-bearing mice following low dose total body irradiation [211]. Investigation of immune checkpoint inhibitors is currently booming and should eventually lead to the advent of efficient combinations strategies in MM.
An alternative to immune checkpoint blockade is the use of agonist mAbs directed again co-stimulatory molecules. Approaches using anti-CD137 mAbs has been shown to elicit potent T and NK cell-mediated responses in murine MM models [105, 212]. Clinical trials are presently ongoing to evaluate the safety and beneficial effects of two agonist anti-CD137 mAbs in cancer patients [213].
Other therapies
Additional strategies able to promote the immune-mediated elimination of myeloma cells include DC-based therapies and vaccines as well as T cell infusions [164]. Vaccination can be directed against a specific tumor antigen such as the idiotype protein but can also use full tumor lysates, apoptotic bodies or fusion between DCs and MM cells [214]. This second option allows the development of T cell responses directed toward the whole spectrum of tumor antigens and thus prevents a possible escape caused by the down-regulation of a single targeted antigen. DC-based therapies aim to foster the expansion of tumor-specific lymphocytes in vivo; an alternative strategy is the infusion of ex vivo expanded T cells. Similarly to NK cells, T cells can be engineered to express CARs, thereby allowing the specific targeting of myeloma cells [215].
Concluding remarks
Early studies established the role of the BM microenvironment in MM pathology but the immune component of this microenvironment has not received full attention until recently. MM appears to represent a good disease model of the cancer editing process [216] where a premalignant equilibrium phase (MGUS) and an escape phase (active MM) can be observed. Several lines of evidence indicate that changes in immune responses may drive MGUS to MM progression [117, 119, 136]. Therapeutic options such as autologous SCT, thalidomide, IMiDs and proteasome inhibitors have the ability to restore and enhance anti-myeloma immune responses, properties that have likely contributed to their clinical success. Still, MM remains largely incurable and most patients succumb to relapsed disease. Research is currently ongoing to design new therapeutic strategies able to eradicate residual disease and to prevent relapse. In this regard, harnessing the immune system is an appealing solution and new approaches such as NK cell-based therapies or immune checkpoint modulation hold great promises for MM patients.
Acknowledgments
K.N. is supported by The Naito Foundation. M. J. S. is supported by a NH&MRC Australia Fellowship (628623) and Program Grant (1013667). C.G. is supported by a NH&MRC early career fellowship (1107417).
Abbreviations
- ADCC
Antibody-dependent cellular cytotoxicity
- APC
Antigen presenting cell
- APRIL
A proliferation-inducing ligand
- BAFF
B-cell activating factor
- BM
Bone marrow
- BMSC
BM stromal cell
- CAM-DR
Cell-adhesion mediated drug resistance
- CAR
Chimeric antigen receptor
- DAMPs
Damage-associated molecular patterns
- DC
Dendritic cell
- GvHD
Graft versus host disease
- GvM
Graft-versus-myeloma
- IFN
Interferon
- Ig
Immunoglobulin
- ILC
Innate lymphoid cell
- KIR
Killer cell immunoglobulin-like receptor
- LPS
Lipopolysaccharide
- mAb
Monoclonal antibody
- MDSC
Myeloid-derived suppressor cells
- MGUS
Monoclonal gammopathy of undetermined significance
- MHC
Major histocompatibility complex
- MM
Multiple myeloma
- MSC
Mesenchymal stem cell
- NK
Natural killer
- PAMPs
Pathogen-associated molecular patterns
- pDC
Plasmacytoid DC
- PBMC
Peripheral blood mononuclear cells
- RANK
Receptor activator of NF-κB
- SCID
Severe combined immunodeficient
- TAM
Tumor-associated macrophages
- TCR
T cell receptor
- TGF
Transforming growth factor
- TLR
Toll-like receptor
- TRAIL
TNF-related apoptosis inducing ligand
- Treg
Regulatory T cell
- VEGF
Vascular endothelial growth factor
References
- 1.Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011;364(11):1046–1060. doi: 10.1056/NEJMra1011442. [DOI] [PubMed] [Google Scholar]
- 2.Vincent Rajkumar S. Multiple myeloma: 2014 update on diagnosis, risk-stratification, and management. Am J Hematol. 2014;89(10):999–1009. doi: 10.1002/ajh.23810. [DOI] [PubMed] [Google Scholar]
- 3.International Myeloma Working G Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol. 2003;121(5):749–757. doi: 10.1046/j.1365-2141.2003.04355.x. [DOI] [PubMed] [Google Scholar]
- 4.Rollig C, Knop S, Bornhauser M. Multiple myeloma. Lancet. 2015;385(9983):2197–2208. doi: 10.1016/S0140-6736(14)60493-1. [DOI] [PubMed] [Google Scholar]
- 5.Tangye SG. Staying alive: regulation of plasma cell survival. Trends Immunol. 2011;32(12):595–602. doi: 10.1016/j.it.2011.09.001. [DOI] [PubMed] [Google Scholar]
- 6.Chu VT, Berek C. The establishment of the plasma cell survival niche in the bone marrow. Immunol Rev. 2013;251(1):177–188. doi: 10.1111/imr.12011. [DOI] [PubMed] [Google Scholar]
- 7.Kuehl WM, Bergsagel PL. Multiple myeloma: evolving genetic events and host interactions. Nat Rev Cancer. 2002;2(3):175–187. doi: 10.1038/nrc746. [DOI] [PubMed] [Google Scholar]
- 8.De Raeve HR, Vanderkerken K. The role of the bone marrow microenvironment in multiple myeloma. Histol Histopathol. 2005;20(4):1227–1250. doi: 10.14670/HH-20.1227. [DOI] [PubMed] [Google Scholar]
- 9.Bergsagel PL, Kuehl WM. Chromosome translocations in multiple myeloma. Oncogene. 2001;20(40):5611–5622. doi: 10.1038/sj.onc.1204641. [DOI] [PubMed] [Google Scholar]
- 10.Davies FE, Dring AM, Li C, Rawstron AC, Shammas MA, O’Connor SM, Fenton JA, Hideshima T, Chauhan D, Tai IT, Robinson E, Auclair D, Rees K, Gonzalez D, Ashcroft AJ, Dasgupta R, Mitsiades C, Mitsiades N, Chen LB, Wong WH, Munshi NC, Morgan GJ, Anderson KC. Insights into the multistep transformation of MGUS to myeloma using microarray expression analysis. Blood. 2003;102(13):4504–4511. doi: 10.1182/blood-2003-01-0016. [DOI] [PubMed] [Google Scholar]
- 11.Wilson A, Trumpp A. Bone-marrow haematopoietic-stem-cell niches. Nat Rev Immunol. 2006;6(2):93–106. doi: 10.1038/nri1779. [DOI] [PubMed] [Google Scholar]
- 12.Romano A, Conticello C, Cavalli M, Vetro C, La Fauci A, Parrinello NL, Di Raimondo F. Immunological dysregulation in multiple myeloma microenvironment. BioMed Res Int. 2014;2014:198539. doi: 10.1155/2014/198539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kawano Y, Moschetta M, Manier S, Glavey S, Gorgun GT, Roccaro AM, Anderson KC, Ghobrial IM. Targeting the bone marrow microenvironment in multiple myeloma. Immunol Rev. 2015;263(1):160–172. doi: 10.1111/imr.12233. [DOI] [PubMed] [Google Scholar]
- 14.De Kleer I, Willems F, Lambrecht B, Goriely S. Ontogeny of myeloid cells. Front Immunol. 2014;5:423. doi: 10.3389/fimmu.2014.00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Matthes T, Manfroi B, Zeller A, Dunand-Sauthier I, Bogen B, Huard B. Autocrine amplification of immature myeloid cells by IL-6 in multiple myeloma-infiltrated bone marrow. Leukemia. 2015;29(9):1882–1890. doi: 10.1038/leu.2015.145. [DOI] [PubMed] [Google Scholar]
- 16.Zheng Y, Cai Z, Wang S, Zhang X, Qian J, Hong S, Li H, Wang M, Yang J, Yi Q. Macrophages are an abundant component of myeloma microenvironment and protect myeloma cells from chemotherapy drug-induced apoptosis. Blood. 2009;114(17):3625–3628. doi: 10.1182/blood-2009-05-220285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim J, Denu RA, Dollar BA, Escalante LE, Kuether JP, Callander NS, Asimakopoulos F, Hematti P. Macrophages and mesenchymal stromal cells support survival and proliferation of multiple myeloma cells. Br J Haematol. 2012;158(3):336–346. doi: 10.1111/j.1365-2141.2012.09154.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Villadangos JA, Schnorrer P. Intrinsic and cooperative antigen-presenting functions of dendritic-cell subsets in vivo. Nat Rev Immunol. 2007;7(7):543–555. doi: 10.1038/nri2103. [DOI] [PubMed] [Google Scholar]
- 19.Shortman K, Sathe P, Vremec D, Naik S, O’Keeffe M. Plasmacytoid dendritic cell development. Adv Immunol. 2013;120:105–126. doi: 10.1016/B978-0-12-417028-5.00004-1. [DOI] [PubMed] [Google Scholar]
- 20.Cavanagh LL, Bonasio R, Mazo IB, Halin C, Cheng G, van der Velden AW, Cariappa A, Chase C, Russell P, Starnbach MN, Koni PA, Pillai S, Weninger W, von Andrian UH. Activation of bone marrow-resident memory T cells by circulating, antigen-bearing dendritic cells. Nat Immunol. 2005;6(10):1029–1037. doi: 10.1038/ni1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Karle H, Hansen NE, Plesner T. Neutrophil defect in multiple myeloma. Studies on intraneutrophilic lysozyme in multiple myeloma and malignant lymphoma. Scand J Haematol. 1976;17(1):62–70. doi: 10.1111/j.1600-0609.1976.tb02842.x. [DOI] [PubMed] [Google Scholar]
- 22.Wong TW, Kita H, Hanson CA, Walters DK, Arendt BK, Jelinek DF. Induction of malignant plasma cell proliferation by eosinophils. PLoS One. 2013;8(7):e70554. doi: 10.1371/journal.pone.0070554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wong D, Winter O, Hartig C, Siebels S, Szyska M, Tiburzy B, Meng L, Kulkarni U, Fahnrich A, Bommert K, Bargou R, Berek C, Chu VT, Bogen B, Jundt F, Manz RA. Eosinophils and megakaryocytes support the early growth of murine MOPC315 myeloma cells in their bone marrow niches. PLoS One. 2014;9(10):e109018. doi: 10.1371/journal.pone.0109018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Klose CS, Flach M, Mohle L, Rogell L, Hoyler T, Ebert K, Fabiunke C, Pfeifer D, Sexl V, Fonseca-Pereira D, Domingues RG, Veiga-Fernandes H, Arnold SJ, Busslinger M, Dunay IR, Tanriver Y, Diefenbach A. Differentiation of type 1 ILCs from a common progenitor to all helper-like innate lymphoid cell lineages. Cell. 2014;157(2):340–356. doi: 10.1016/j.cell.2014.03.030. [DOI] [PubMed] [Google Scholar]
- 25.Halim TY, MacLaren A, Romanish MT, Gold MJ, McNagny KM, Takei F. Retinoic-acid-receptor-related orphan nuclear receptor alpha is required for natural helper cell development and allergic inflammation. Immunity. 2012;37(3):463–474. doi: 10.1016/j.immuni.2012.06.012. [DOI] [PubMed] [Google Scholar]
- 26.Fathman JW, Bhattacharya D, Inlay MA, Seita J, Karsunky H, Weissman IL. Identification of the earliest natural killer cell-committed progenitor in murine bone marrow. Blood. 2011;118(20):5439–5447. doi: 10.1182/blood-2011-04-348912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Guillerey C, Smyth MJ. NK cells and cancer immunoediting. Curr Top Microbiol Immunol. 2015 doi: 10.1007/82_2015_446. [DOI] [PubMed] [Google Scholar]
- 28.Godfrey J, Benson DM., Jr The role of natural killer cells in immunity against multiple myeloma. Leuk Lymphoma. 2012;53(9):1666–1676. doi: 10.3109/10428194.2012.676175. [DOI] [PubMed] [Google Scholar]
- 29.Koebel CM, Vermi W, Swann JB, Zerafa N, Rodig SJ, Old LJ, Smyth MJ, Schreiber RD. Adaptive immunity maintains occult cancer in an equilibrium state. Nature. 2007;450(7171):903–907. doi: 10.1038/nature06309. [DOI] [PubMed] [Google Scholar]
- 30.Hughes V. Microenvironment: neighbourhood watch. Nature. 2011;480(7377):S48–S49. doi: 10.1038/480S48a. [DOI] [PubMed] [Google Scholar]
- 31.Di Rosa F, Pabst R. The bone marrow: a nest for migratory memory T cells. Trends Immunol. 2005;26(7):360–366. doi: 10.1016/j.it.2005.04.011. [DOI] [PubMed] [Google Scholar]
- 32.Zhao E, Xu H, Wang L, Kryczek I, Wu K, Hu Y, Wang G, Zou W. Bone marrow and the control of immunity. Cell Mol Immunol. 2012;9(1):11–19. doi: 10.1038/cmi.2011.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhu J, Paul WE. Heterogeneity and plasticity of T helper cells. Cell Res. 2010;20(1):4–12. doi: 10.1038/cr.2009.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Whiteside TL. What are regulatory T cells (Treg) regulating in cancer and why? Semin Cancer Biol. 2012;22(4):327–334. doi: 10.1016/j.semcancer.2012.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zou L, Barnett B, Safah H, Larussa VF, Evdemon-Hogan M, Mottram P, Wei S, David O, Curiel TJ, Zou W. Bone marrow is a reservoir for CD4+CD25+ regulatory T cells that traffic through CXCL12/CXCR4 signals. Cancer Res. 2004;64(22):8451–8455. doi: 10.1158/0008-5472.CAN-04-1987. [DOI] [PubMed] [Google Scholar]
- 36.Blade J, Fernandez de Larrea C, Rosinol L, Cibeira MT, Jimenez R, Powles R. Soft-tissue plasmacytomas in multiple myeloma: incidence, mechanisms of extramedullary spread, and treatment approach. J Clin Oncol. 2011;29(28):3805–3812. doi: 10.1200/JCO.2011.34.9290. [DOI] [PubMed] [Google Scholar]
- 37.Katz BZ. Adhesion molecules—the lifelines of multiple myeloma cells. Semin Cancer Biol. 2010;20(3):186–195. doi: 10.1016/j.semcancer.2010.04.003. [DOI] [PubMed] [Google Scholar]
- 38.Klein B, Zhang XG, Lu ZY, Bataille R. Interleukin-6 in human multiple myeloma. Blood. 1995;85(4):863–872. [PubMed] [Google Scholar]
- 39.Asaoku H, Kawano M, Iwato K, Tanabe O, Tanaka H, Hirano T, Kishimoto T, Kuramoto A. Decrease in BSF-2/IL-6 response in advanced cases of multiple myeloma. Blood. 1988;72(2):429–432. [PubMed] [Google Scholar]
- 40.Mitsiades CS, McMillin DW, Klippel S, Hideshima T, Chauhan D, Richardson PG, Munshi NC, Anderson KC. The role of the bone marrow microenvironment in the pathophysiology of myeloma and its significance in the development of more effective therapies. Hematol/Oncol Clin N Am. 2007;21(6):1007–1034. doi: 10.1016/j.hoc.2007.08.007. [DOI] [PubMed] [Google Scholar]
- 41.Kishimoto T. The biology of interleukin-6. Blood. 1989;74(1):1–10. [PubMed] [Google Scholar]
- 42.Gunn WG, Conley A, Deininger L, Olson SD, Prockop DJ, Gregory CA. A crosstalk between myeloma cells and marrow stromal cells stimulates production of DKK1 and interleukin-6: a potential role in the development of lytic bone disease and tumor progression in multiple myeloma. Stem Cells. 2006;24(4):986–991. doi: 10.1634/stemcells.2005-0220. [DOI] [PubMed] [Google Scholar]
- 43.Chu VT, Frohlich A, Steinhauser G, Scheel T, Roch T, Fillatreau S, Lee JJ, Lohning M, Berek C. Eosinophils are required for the maintenance of plasma cells in the bone marrow. Nat Immunol. 2011;12(2):151–159. doi: 10.1038/ni.1981. [DOI] [PubMed] [Google Scholar]
- 44.San-Miguel J, Blade J, Shpilberg O, Grosicki S, Maloisel F, Min CK, Polo Zarzuela M, Robak T, Prasad SV, Tee Goh Y, Laubach J, Spencer A, Mateos MV, Palumbo A, Puchalski T, Reddy M, Uhlar C, Qin X, van de Velde H, Xie H, Orlowski RZ. Phase 2 randomized study of bortezomib-melphalan-prednisone with or without siltuximab (anti-IL-6) in multiple myeloma. Blood. 2014;123(26):4136–4142. doi: 10.1182/blood-2013-12-546374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Novak AJ, Darce JR, Arendt BK, Harder B, Henderson K, Kindsvogel W, Gross JA, Greipp PR, Jelinek DF. Expression of BCMA, TACI, and BAFF-R in multiple myeloma: a mechanism for growth and survival. Blood. 2004;103(2):689–694. doi: 10.1182/blood-2003-06-2043. [DOI] [PubMed] [Google Scholar]
- 46.Moreaux J, Legouffe E, Jourdan E, Quittet P, Reme T, Lugagne C, Moine P, Rossi JF, Klein B, Tarte K. BAFF and APRIL protect myeloma cells from apoptosis induced by interleukin 6 deprivation and dexamethasone. Blood. 2004;103(8):3148–3157. doi: 10.1182/blood-2003-06-1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Moreaux J, Cremer FW, Reme T, Raab M, Mahtouk K, Kaukel P, Pantesco V, De Vos J, Jourdan E, Jauch A, Legouffe E, Moos M, Fiol G, Goldschmidt H, Rossi JF, Hose D, Klein B. The level of TACI gene expression in myeloma cells is associated with a signature of microenvironment dependence versus a plasmablastic signature. Blood. 2005;106(3):1021–1030. doi: 10.1182/blood-2004-11-4512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Prabhala RH, Pelluru D, Fulciniti M, Prabhala HK, Nanjappa P, Song W, Pai C, Amin S, Tai YT, Richardson PG, Ghobrial IM, Treon SP, Daley JF, Anderson KC, Kutok JL, Munshi NC. Elevated IL-17 produced by TH17 cells promotes myeloma cell growth and inhibits immune function in multiple myeloma. Blood. 2010;115(26):5385–5392. doi: 10.1182/blood-2009-10-246660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Barille S, Collette M, Bataille R, Amiot M. Myeloma cells upregulate interleukin-6 secretion in osteoblastic cells through cell-to-cell contact but downregulate osteocalcin. Blood. 1995;86(8):3151–3159. [PubMed] [Google Scholar]
- 50.Yaccoby S, Wezeman MJ, Zangari M, Walker R, Cottler-Fox M, Gaddy D, Ling W, Saha R, Barlogie B, Tricot G, Epstein J. Inhibitory effects of osteoblasts and increased bone formation on myeloma in novel culture systems and a myelomatous mouse model. Haematologica. 2006;91(2):192–199. [PMC free article] [PubMed] [Google Scholar]
- 51.Podar K, Chauhan D, Anderson KC. Bone marrow microenvironment and the identification of new targets for myeloma therapy. Leukemia. 2009;23(1):10–24. doi: 10.1038/leu.2008.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Giuliani N, Colla S, Sala R, Moroni M, Lazzaretti M, La Monica S, Bonomini S, Hojden M, Sammarelli G, Barille S, Bataille R, Rizzoli V. Human myeloma cells stimulate the receptor activator of nuclear factor-kappa B ligand (RANKL) in T lymphocytes: a potential role in multiple myeloma bone disease. Blood. 2002;100(13):4615–4621. doi: 10.1182/blood-2002-04-1121. [DOI] [PubMed] [Google Scholar]
- 53.Noonan K, Marchionni L, Anderson J, Pardoll D, Roodman GD, Borrello I. A novel role of IL-17-producing lymphocytes in mediating lytic bone disease in multiple myeloma. Blood. 2010;116(18):3554–3563. doi: 10.1182/blood-2010-05-283895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vacca A, Ribatti D, Presta M, Minischetti M, Iurlaro M, Ria R, Albini A, Bussolino F, Dammacco F. Bone marrow neovascularization, plasma cell angiogenic potential, and matrix metalloproteinase-2 secretion parallel progression of human multiple myeloma. Blood. 1999;93(9):3064–3073. [PubMed] [Google Scholar]
- 55.Giuliani N, Storti P, Bolzoni M, Palma BD, Bonomini S. Angiogenesis and multiple myeloma. Cancer Microenviron. 2011;4(3):325–337. doi: 10.1007/s12307-011-0072-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ribatti D, Nico B, Vacca A. Multiple myeloma as a model for the role of bone marrow niches in the control of angiogenesis. Int Rev Cell Molec Biol. 2015;314:259–282. doi: 10.1016/bs.ircmb.2014.10.004. [DOI] [PubMed] [Google Scholar]
- 57.Berardi S, Ria R, Reale A, De Luisi A, Catacchio I, Moschetta M, Vacca A. Multiple myeloma macrophages: pivotal players in the tumor microenvironment. J Oncol. 2013;2013:183602. doi: 10.1155/2013/183602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tartour E, Pere H, Maillere B, Terme M, Merillon N, Taieb J, Sandoval F, Quintin-Colonna F, Lacerda K, Karadimou A, Badoual C, Tedgui A, Fridman WH, Oudard S. Angiogenesis and immunity: a bidirectional link potentially relevant for the monitoring of antiangiogenic therapy and the development of novel therapeutic combination with immunotherapy. Cancer Metastasis Rev. 2011;30(1):83–95. doi: 10.1007/s10555-011-9281-4. [DOI] [PubMed] [Google Scholar]
- 59.Rossi M, Botta C, Correale P, Tassone P, Tagliaferri P. Immunologic microenvironment and personalized treatment in multiple myeloma. Exp Opin Biol Ther. 2013;13(Suppl 1):S83–S93. doi: 10.1517/14712598.2013.799130. [DOI] [PubMed] [Google Scholar]
- 60.Tete SM, Bijl M, Sahota SS, Bos NA. Immune defects in the risk of infection and response to vaccination in monoclonal gammopathy of undetermined significance and multiple myeloma. Front Immunol. 2014;5:257. doi: 10.3389/fimmu.2014.00257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xu FH, Sharma S, Gardner A, Tu Y, Raitano A, Sawyers C, Lichtenstein A. Interleukin-6-induced inhibition of multiple myeloma cell apoptosis: support for the hypothesis that protection is mediated via inhibition of the JNK/SAPK pathway. Blood. 1998;92(1):241–251. [PubMed] [Google Scholar]
- 62.Zheng Y, Yang J, Qian J, Qiu P, Hanabuchi S, Lu Y, Wang Z, Liu Z, Li H, He J, Lin P, Weber D, Davis RE, Kwak L, Cai Z, Yi Q. PSGL-1/selectin and ICAM-1/CD18 interactions are involved in macrophage-induced drug resistance in myeloma. Leukemia. 2013;27(3):702–710. doi: 10.1038/leu.2012.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Franqui-Machin R, Wendlandt EB, Janz S, Zhan F, Tricot G. Cancer stem cells are the cause of drug resistance in multiple myeloma: fact or fiction? Oncotarget. 2015;6(38):40496–40506. doi: 10.18632/oncotarget.5800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A. Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinogenesis. 2009;30(7):1073–1081. doi: 10.1093/carcin/bgp127. [DOI] [PubMed] [Google Scholar]
- 65.Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140(6):883–899. doi: 10.1016/j.cell.2010.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chow MT, Moller A, Smyth MJ. Inflammation and immune surveillance in cancer. Semin Cancer Biol. 2012;22(1):23–32. doi: 10.1016/j.semcancer.2011.12.004. [DOI] [PubMed] [Google Scholar]
- 67.Ridnour LA, Cheng RY, Switzer CH, Heinecke JL, Ambs S, Glynn S, Young HA, Trinchieri G, Wink DA. Molecular pathways: toll-like receptors in the tumor microenvironment—poor prognosis or new therapeutic opportunity. Clin Cancer Res. 2013;19(6):1340–1346. doi: 10.1158/1078-0432.CCR-12-0408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gabrilovich DI, Ostrand-Rosenberg S, Bronte V. Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol. 2012;12(4):253–268. doi: 10.1038/nri3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Talmadge JE, Gabrilovich DI. History of myeloid-derived suppressor cells. Nat Rev Cancer. 2013;13(10):739–752. doi: 10.1038/nrc3581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Condamine T, Gabrilovich DI. Molecular mechanisms regulating myeloid-derived suppressor cell differentiation and function. Trends Immunol. 2011;32(1):19–25. doi: 10.1016/j.it.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Qian BZ, Li J, Zhang H, Kitamura T, Zhang J, Campion LR, Kaiser EA, Snyder LA, Pollard JW. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature. 2011;475(7355):222–225. doi: 10.1038/nature10138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yang L, Huang J, Ren X, Gorska AE, Chytil A, Aakre M, Carbone DP, Matrisian LM, Richmond A, Lin PC, Moses HL. Abrogation of TGF beta signaling in mammary carcinomas recruits Gr-1+CD11b+ myeloid cells that promote metastasis. Cancer Cell. 2008;13(1):23–35. doi: 10.1016/j.ccr.2007.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sinha P, Okoro C, Foell D, Freeze HH, Ostrand-Rosenberg S, Srikrishna G. Proinflammatory S100 proteins regulate the accumulation of myeloid-derived suppressor cells. J Immunol. 2008;181(7):4666–4675. doi: 10.4049/jimmunol.181.7.4666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Watson GA, Fu YX, Lopez DM. Splenic macrophages from tumor-bearing mice co-expressing MAC-1 and MAC-2 antigens exert immunoregulatory functions via two distinct mechanisms. J Leukoc Biol. 1991;49(2):126–138. doi: 10.1002/jlb.49.2.126. [DOI] [PubMed] [Google Scholar]
- 75.Kusmartsev SA, Li Y, Chen SH. Gr-1 + myeloid cells derived from tumor-bearing mice inhibit primary T cell activation induced through CD3/CD28 costimulation. J Immunol. 2000;165(2):779–785. doi: 10.4049/jimmunol.165.2.779. [DOI] [PubMed] [Google Scholar]
- 76.Rodriguez PC, Quiceno DG, Zabaleta J, Ortiz B, Zea AH, Piazuelo MB, Delgado A, Correa P, Brayer J, Sotomayor EM, Antonia S, Ochoa JB, Ochoa AC. Arginase I production in the tumor microenvironment by mature myeloid cells inhibits T-cell receptor expression and antigen-specific T-cell responses. Cancer Res. 2004;64(16):5839–5849. doi: 10.1158/0008-5472.CAN-04-0465. [DOI] [PubMed] [Google Scholar]
- 77.Serafini P, Mgebroff S, Noonan K, Borrello I. Myeloid-derived suppressor cells promote cross-tolerance in B-cell lymphoma by expanding regulatory T cells. Cancer Res. 2008;68(13):5439–5449. doi: 10.1158/0008-5472.CAN-07-6621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Pan PY, Ma G, Weber KJ, Ozao-Choy J, Wang G, Yin B, Divino CM, Chen SH. Immune stimulatory receptor CD40 is required for T-cell suppression and T regulatory cell activation mediated by myeloid-derived suppressor cells in cancer. Cancer Res. 2010;70(1):99–108. doi: 10.1158/0008-5472.CAN-09-1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J, Divino CM, Chen SH. Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res. 2006;66(2):1123–1131. doi: 10.1158/0008-5472.CAN-05-1299. [DOI] [PubMed] [Google Scholar]
- 80.Yang L, DeBusk LM, Fukuda K, Fingleton B, Green-Jarvis B, Shyr Y, Matrisian LM, Carbone DP, Lin PC. Expansion of myeloid immune suppressor Gr+CD11b+ cells in tumor-bearing host directly promotes tumor angiogenesis. Cancer Cell. 2004;6(4):409–421. doi: 10.1016/j.ccr.2004.08.031. [DOI] [PubMed] [Google Scholar]
- 81.Ramachandran IR, Martner A, Pisklakova A, Condamine T, Chase T, Vogl T, Roth J, Gabrilovich D, Nefedova Y. Myeloid-derived suppressor cells regulate growth of multiple myeloma by inhibiting T cells in bone marrow. J Immunol. 2013;190(7):3815–3823. doi: 10.4049/jimmunol.1203373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Van Valckenborgh E, Schouppe E, Movahedi K, De Bruyne E, Menu E, De Baetselier P, Vanderkerken K, Van Ginderachter JA. Multiple myeloma induces the immunosuppressive capacity of distinct myeloid-derived suppressor cell subpopulations in the bone marrow. Leukemia. 2012;26(11):2424–2428. doi: 10.1038/leu.2012.113. [DOI] [PubMed] [Google Scholar]
- 83.De Veirman K, Van Valckenborgh E, Lahmar Q, Geeraerts X, De Bruyne E, Menu E, Van Riet I, Vanderkerken K, Van Ginderachter JA. Myeloid-derived suppressor cells as therapeutic target in hematological malignancies. Front Oncol. 2014;4:349. doi: 10.3389/fonc.2014.00349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Brimnes MK, Vangsted AJ, Knudsen LM, Gimsing P, Gang AO, Johnsen HE, Svane IM. Increased level of both CD4+FOXP3+ regulatory T cells and CD14+HLA-DR(-)/low myeloid-derived suppressor cells and decreased level of dendritic cells in patients with multiple myeloma. Scand J Immunol. 2010;72(6):540–547. doi: 10.1111/j.1365-3083.2010.02463.x. [DOI] [PubMed] [Google Scholar]
- 85.Gorgun GT, Whitehill G, Anderson JL, Hideshima T, Maguire C, Laubach J, Raje N, Munshi NC, Richardson PG, Anderson KC. Tumor-promoting immune-suppressive myeloid-derived suppressor cells in the multiple myeloma microenvironment in humans. Blood. 2013;121(15):2975–2987. doi: 10.1182/blood-2012-08-448548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Favaloro J, Liyadipitiya T, Brown R, Yang S, Suen H, Woodland N, Nassif N, Hart D, Fromm P, Weatherburn C, Gibson J, Ho PJ, Joshua D. Myeloid derived suppressor cells are numerically, functionally and phenotypically different in patients with multiple myeloma. Leuk Lymphoma. 2014;55(12):2893–2900. doi: 10.3109/10428194.2014.904511. [DOI] [PubMed] [Google Scholar]
- 87.De Keersmaecker B, Fostier K, Corthals J, Wilgenhof S, Heirman C, Aerts JL, Thielemans K, Schots R. Immunomodulatory drugs improve the immune environment for dendritic cell-based immunotherapy in multiple myeloma patients after autologous stem cell transplantation. Cancer Immunol Immunother: CII. 2014;63(10):1023–1036. doi: 10.1007/s00262-014-1571-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer. 2004;4(1):71–78. doi: 10.1038/nrc1256. [DOI] [PubMed] [Google Scholar]
- 89.Noy R, Pollard JW. Tumor-associated macrophages: from mechanisms to therapy. Immunity. 2014;41(1):49–61. doi: 10.1016/j.immuni.2014.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Komohara Y, Jinushi M, Takeya M. Clinical significance of macrophage heterogeneity in human malignant tumors. Cancer Sci. 2014;105(1):1–8. doi: 10.1111/cas.12314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Suyani E, Sucak GT, Akyurek N, Sahin S, Baysal NA, Yagci M, Haznedar R. Tumor-associated macrophages as a prognostic parameter in multiple myeloma. Ann Hematol. 2013;92(5):669–677. doi: 10.1007/s00277-012-1652-6. [DOI] [PubMed] [Google Scholar]
- 92.Van Overmeire E, Laoui D, Keirsse J, Van Ginderachter JA, Sarukhan A. Mechanisms driving macrophage diversity and specialization in distinct tumor microenvironments and parallelisms with other tissues. Front Immunol. 2014;5:127. doi: 10.3389/fimmu.2014.00127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Movahedi K, Laoui D, Gysemans C, Baeten M, Stange G, Van den Bossche J, Mack M, Pipeleers D, In’t Veld P, De Baetselier P, Van Ginderachter JA. Different tumor microenvironments contain functionally distinct subsets of macrophages derived from Ly6C(high) monocytes. Cancer Res. 2010;70(14):5728–5739. doi: 10.1158/0008-5472.CAN-09-4672. [DOI] [PubMed] [Google Scholar]
- 94.Franklin RA, Liao W, Sarkar A, Kim MV, Bivona MR, Liu K, Pamer EG, Li MO. The cellular and molecular origin of tumor-associated macrophages. Science (New York, NY) 2014;344(6186):921–925. doi: 10.1126/science.1252510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hume DA, MacDonald KP. Therapeutic applications of macrophage colony-stimulating factor-1 (CSF-1) and antagonists of CSF-1 receptor (CSF-1R) signaling. Blood. 2012;119(8):1810–1820. doi: 10.1182/blood-2011-09-379214. [DOI] [PubMed] [Google Scholar]
- 96.Laoui D, Van Overmeire E, Di Conza G, Aldeni C, Keirsse J, Morias Y, Movahedi K, Houbracken I, Schouppe E, Elkrim Y, Karroum O, Jordan B, Carmeliet P, Gysemans C, De Baetselier P, Mazzone M, Van Ginderachter JA. Tumor hypoxia does not drive differentiation of tumor-associated macrophages but rather fine-tunes the M2-like macrophage population. Cancer Res. 2014;74(1):24–30. doi: 10.1158/0008-5472.CAN-13-1196. [DOI] [PubMed] [Google Scholar]
- 97.Corzo CA, Condamine T, Lu L, Cotter MJ, Youn JI, Cheng P, Cho HI, Celis E, Quiceno DG, Padhya T, McCaffrey TV, McCaffrey JC, Gabrilovich DI. HIF-1alpha regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. J Exp Med. 2010;207(11):2439–2453. doi: 10.1084/jem.20100587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Scavelli C, Nico B, Cirulli T, Ria R, Di Pietro G, Mangieri D, Bacigalupo A, Mangialardi G, Coluccia AM, Caravita T, Molica S, Ribatti D, Dammacco F, Vacca A. Vasculogenic mimicry by bone marrow macrophages in patients with multiple myeloma. Oncogene. 2008;27(5):663–674. doi: 10.1038/sj.onc.1210691. [DOI] [PubMed] [Google Scholar]
- 99.Costes V, Portier M, Lu Z-Y, Rossi J-F, Bataille R, Klein B. Interleukin-1 in multiple myeloma: producer cells and their role in the control of IL-6 production. Br J Haematol. 1998;103(4):1152–1160. doi: 10.1046/j.1365-2141.1998.01101.x. [DOI] [PubMed] [Google Scholar]
- 100.Hope C, Ollar SJ, Heninger E, Hebron E, Jensen JL, Kim J, Maroulakou I, Miyamoto S, Leith C, Yang DT, Callander N, Hematti P, Chesi M, Bergsagel PL, Asimakopoulos F. TPL2 kinase regulates the inflammatory milieu of the myeloma niche. Blood. 2014;123(21):3305–3315. doi: 10.1182/blood-2014-02-554071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Otsuki T, Yata K, Sakaguchi H, Uno M, Fujii T, Wada H, Sugihara T, Ueki A. IL-10 in myeloma cells. Leuk Lymphoma. 2002;43(5):969–974. doi: 10.1080/10428190290021579. [DOI] [PubMed] [Google Scholar]
- 102.Ribatti D, Nico B, Vacca A. Importance of the bone marrow microenvironment in inducing the angiogenic response in multiple myeloma. Oncogene. 2006;25(31):4257–4266. doi: 10.1038/sj.onc.1209456. [DOI] [PubMed] [Google Scholar]
- 103.Cooper MA, Fehniger TA, Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol. 2001;22(11):633–640. doi: 10.1016/S1471-4906(01)02060-9. [DOI] [PubMed] [Google Scholar]
- 104.Garcia-Sanz R, Gonzalez M, Orfao A, Moro MJ, Hernandez JM, Borrego D, Carnero M, Casanova F, Barez A, Jimenez R, Portero JA, San Miguel JF. Analysis of natural killer-associated antigens in peripheral blood and bone marrow of multiple myeloma patients and prognostic implications. Br J Haematol. 1996;93(1):81–88. doi: 10.1046/j.1365-2141.1996.4651006.x. [DOI] [PubMed] [Google Scholar]
- 105.Guillerey C, Ferrari de Andrade L, Vuckovic S, Miles K, Ngiow SF, Yong MC, Teng MW, Colonna M, Ritchie DS, Chesi M, Bergsagel PL, Hill GR, Smyth MJ, Martinet L. Immunosurveillance and therapy of multiple myeloma are CD226 dependent. J Clin Investig. 2015;125(5):2077–2089. doi: 10.1172/JCI77181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ponzetta A, Benigni G, Antonangeli F, Sciume G, Sanseviero E, Zingoni A, Ricciardi MR, Petrucci MT, Santoni A, Bernardini G. Multiple myeloma impairs bone marrow localization of effector natural killer cells by altering the chemokine microenvironment. Cancer Res. 2015 doi: 10.1158/0008-5472.CAN-15-1320. [DOI] [PubMed] [Google Scholar]
- 107.El-Sherbiny YM, Meade JL, Holmes TD, McGonagle D, Mackie SL, Morgan AW, Cook G, Feyler S, Richards SJ, Davies FE, Morgan GJ, Cook GP. The requirement for DNAM-1, NKG2D, and NKp46 in the natural killer cell-mediated killing of myeloma cells. Cancer Res. 2007;67(18):8444–8449. doi: 10.1158/0008-5472.CAN-06-4230. [DOI] [PubMed] [Google Scholar]
- 108.Carbone E, Neri P, Mesuraca M, Fulciniti MT, Otsuki T, Pende D, Groh V, Spies T, Pollio G, Cosman D, Catalano L, Tassone P, Rotoli B, Venuta S. HLA class I, NKG2D, and natural cytotoxicity receptors regulate multiple myeloma cell recognition by natural killer cells. Blood. 2005;105(1):251–258. doi: 10.1182/blood-2004-04-1422. [DOI] [PubMed] [Google Scholar]
- 109.Frohn C, Hoppner M, Schlenke P, Kirchner H, Koritke P, Luhm J. Anti-myeloma activity of natural killer lymphocytes. Br J Haematol. 2002;119(3):660–664. doi: 10.1046/j.1365-2141.2002.03879.x. [DOI] [PubMed] [Google Scholar]
- 110.Soriani A, Zingoni A, Cerboni C, Iannitto ML, Ricciardi MR, Di Gialleonardo V, Cippitelli M, Fionda C, Petrucci MT, Guarini A, Foa R, Santoni A. ATM-ATR-dependent up-regulation of DNAM-1 and NKG2D ligands on multiple myeloma cells by therapeutic agents results in enhanced NK-cell susceptibility and is associated with a senescent phenotype. Blood. 2009;113(15):3503–3511. doi: 10.1182/blood-2008-08-173914. [DOI] [PubMed] [Google Scholar]
- 111.Parham P. MHC class I molecules and KIRs in human history, health and survival. Nat Rev Immunol. 2005;5(3):201–214. doi: 10.1038/nri1570. [DOI] [PubMed] [Google Scholar]
- 112.Ikeda H, Old LJ, Schreiber RD. The roles of IFN gamma in protection against tumor development and cancer immunoediting. Cytokine Growth Factor Rev. 2002;13(2):95–109. doi: 10.1016/S1359-6101(01)00038-7. [DOI] [PubMed] [Google Scholar]
- 113.Portier M, Zhang XG, Caron E, Lu ZY, Bataille R, Klein B. gamma-Interferon in multiple myeloma: inhibition of interleukin-6 (IL-6)-dependent myeloma cell growth and downregulation of IL-6-receptor expression in vitro. Blood. 1993;81(11):3076–3082. [PubMed] [Google Scholar]
- 114.Takayanagi H, Ogasawara K, Hida S, Chiba T, Murata S, Sato K, Takaoka A, Yokochi T, Oda H, Tanaka K, Nakamura K, Taniguchi T. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature. 2000;408(6812):600–605. doi: 10.1038/35046102. [DOI] [PubMed] [Google Scholar]
- 115.Viel S, Charrier E, Marcais A, Rouzaire P, Bienvenu J, Karlin L, Salles G, Walzer T. Monitoring NK cell activity in patients with hematological malignancies. Oncoimmunology. 2013;2(9):e26011. doi: 10.4161/onci.26011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ. Natural innate and adaptive immunity to cancer. Annu Rev Immunol. 2011;29:235–271. doi: 10.1146/annurev-immunol-031210-101324. [DOI] [PubMed] [Google Scholar]
- 117.Bernal M, Garrido P, Jimenez P, Carretero R, Almagro M, Lopez P, Navarro P, Garrido F, Ruiz-Cabello F. Changes in activatory and inhibitory natural killer (NK) receptors may induce progression to multiple myeloma: implications for tumor evasion of T and NK cells. Hum Immunol. 2009;70(10):854–857. doi: 10.1016/j.humimm.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 118.Jinushi M, Vanneman M, Munshi NC, Tai YT, Prabhala RH, Ritz J, Neuberg D, Anderson KC, Carrasco DR, Dranoff G. MHC class I chain-related protein A antibodies and shedding are associated with the progression of multiple myeloma. Proc Natl Acad Sci USA. 2008;105(4):1285–1290. doi: 10.1073/pnas.0711293105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Perez-Andres M, Almeida J, Martin-Ayuso M, Moro MJ, Martin-Nunez G, Galende J, Borrego D, Rodriguez MJ, Ortega F, Hernandez J, Moreno I, Dominguez M, Mateo G, San Miguel JF, Orfao A, Spanish Network on multiple m. Spanish Network of Cancer Research C Clonal plasma cells from monoclonal gammopathy of undetermined significance, multiple myeloma and plasma cell leukemia show different expression profiles of molecules involved in the interaction with the immunological bone marrow microenvironment. Leukemia. 2005;19(3):449–455. doi: 10.1038/sj.leu.2403647. [DOI] [PubMed] [Google Scholar]
- 120.von Lilienfeld-Toal M, Frank S, Leyendecker C, Feyler S, Jarmin S, Morgan R, Glasmacher A, Marten A, Schmidt-Wolf IG, Brossart P, Cook G. Reduced immune effector cell NKG2D expression and increased levels of soluble NKG2D ligands in multiple myeloma may not be causally linked. Cancer Immunol Immunother: CII. 2010;59(6):829–839. doi: 10.1007/s00262-009-0807-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Fauriat C, Mallet F, Olive D, Costello RT. Impaired activating receptor expression pattern in natural killer cells from patients with multiple myeloma. Leukemia. 2006;20(4):732–733. doi: 10.1038/sj.leu.2404096. [DOI] [PubMed] [Google Scholar]
- 122.Mills KH, Cawley JC. Abnormal monoclonal antibody-defined helper/suppressor T-cell subpopulations in multiple myeloma: relationship to treatment and clinical stage. Br J Haematol. 1983;53(2):271–275. doi: 10.1111/j.1365-2141.1983.tb02021.x. [DOI] [PubMed] [Google Scholar]
- 123.Cook G, Campbell JD. Immune regulation in multiple myeloma: the host-tumour conflict. Blood Rev. 1999;13(3):151–162. doi: 10.1054/blre.1999.0111. [DOI] [PubMed] [Google Scholar]
- 124.Pinzon-Charry A, Maxwell T, Lopez JA. Dendritic cell dysfunction in cancer: a mechanism for immunosuppression. Immunol Cell Biol. 2005;83(5):451–461. doi: 10.1111/j.1440-1711.2005.01371.x. [DOI] [PubMed] [Google Scholar]
- 125.Feuerer M, Beckhove P, Garbi N, Mahnke Y, Limmer A, Hommel M, Hammerling GJ, Kyewski B, Hamann A, Umansky V, Schirrmacher V. Bone marrow as a priming site for T-cell responses to blood-borne antigen. Nat Med. 2003;9(9):1151–1157. doi: 10.1038/nm914. [DOI] [PubMed] [Google Scholar]
- 126.Raje N, Gong J, Chauhan D, Teoh G, Avigan D, Wu Z, Chen D, Treon SP, Webb IJ, Kufe DW, Anderson KC. Bone marrow and peripheral blood dendritic cells from patients with multiple myeloma are phenotypically and functionally normal despite the detection of Kaposi’s sarcoma herpesvirus gene sequences. Blood. 1999;93(5):1487–1495. [PubMed] [Google Scholar]
- 127.Brown RD, Pope B, Murray A, Esdale W, Sze DM, Gibson J, Ho PJ, Hart D, Joshua D. Dendritic cells from patients with myeloma are numerically normal but functionally defective as they fail to up-regulate CD80 (B7-1) expression after huCD40LT stimulation because of inhibition by transforming growth factor-beta1 and interleukin-10. Blood. 2001;98(10):2992–2998. doi: 10.1182/blood.V98.10.2992. [DOI] [PubMed] [Google Scholar]
- 128.Ratta M, Fagnoni F, Curti A, Vescovini R, Sansoni P, Oliviero B, Fogli M, Ferri E, Della Cuna GR, Tura S, Baccarani M, Lemoli RM. Dendritic cells are functionally defective in multiple myeloma: the role of interleukin-6. Blood. 2002;100(1):230–237. doi: 10.1182/blood.V100.1.230. [DOI] [PubMed] [Google Scholar]
- 129.Brimnes MK, Svane IM, Johnsen HE. Impaired functionality and phenotypic profile of dendritic cells from patients with multiple myeloma. Clin Exp Immunol. 2006;144(1):76–84. doi: 10.1111/j.1365-2249.2006.03037.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Leone P, Berardi S, Frassanito MA, Ria R, De Re V, Cicco S, Battaglia S, Ditonno P, Dammacco F, Vacca A, Racanelli V. Dendritic cells accumulate in the bone marrow of myeloma patients where they protect tumor plasma cells from CD8+ T-cell killing. Blood. 2015;126(12):1443–1451. doi: 10.1182/blood-2015-01-623975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Jego G, Palucka AK, Blanck JP, Chalouni C, Pascual V, Banchereau J. Plasmacytoid dendritic cells induce plasma cell differentiation through type I interferon and interleukin 6. Immunity. 2003;19(2):225–234. doi: 10.1016/S1074-7613(03)00208-5. [DOI] [PubMed] [Google Scholar]
- 132.Chauhan D, Singh AV, Brahmandam M, Carrasco R, Bandi M, Hideshima T, Bianchi G, Podar K, Tai YT, Mitsiades C, Raje N, Jaye DL, Kumar SK, Richardson P, Munshi N, Anderson KC. Functional interaction of plasmacytoid dendritic cells with multiple myeloma cells: a therapeutic target. Cancer Cell. 2009;16(4):309–323. doi: 10.1016/j.ccr.2009.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Halapi E, Werner A, Wahlstrom J, Osterborg A, Jeddi-Tehrani M, Yi Q, Janson CH, Wigzell H, Grunewald J, Mellstedt H. T cell repertoire in patients with multiple myeloma and monoclonal gammopathy of undetermined significance: clonal CD8+ T cell expansions are found preferentially in patients with a low tumor burden. Eur J Immunol. 1997;27(9):2245–2252. doi: 10.1002/eji.1830270919. [DOI] [PubMed] [Google Scholar]
- 134.Dhodapkar MV, Krasovsky J, Olson K. T cells from the tumor microenvironment of patients with progressive myeloma can generate strong, tumor-specific cytolytic responses to autologous, tumor-loaded dendritic cells. Proc Natl Acad Sci USA. 2002;99(20):13009–13013. doi: 10.1073/pnas.202491499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Wen YJ, Min R, Tricot G, Barlogie B, Yi Q. Tumor lysate-specific cytotoxic T lymphocytes in multiple myeloma: promising effector cells for immunotherapy. Blood. 2002;99(9):3280–3285. doi: 10.1182/blood.V99.9.3280. [DOI] [PubMed] [Google Scholar]
- 136.Dhodapkar MV, Krasovsky J, Osman K, Geller MD. Vigorous premalignancy-specific effector T cell response in the bone marrow of patients with monoclonal gammopathy. J Exp Med. 2003;198(11):1753–1757. doi: 10.1084/jem.20031030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Spisek R, Kukreja A, Chen LC, Matthews P, Mazumder A, Vesole D, Jagannath S, Zebroski HA, Simpson AJ, Ritter G, Durie B, Crowley J, Shaughnessy JD, Jr, Scanlan MJ, Gure AO, Barlogie B, Dhodapkar MV. Frequent and specific immunity to the embryonal stem cell-associated antigen SOX2 in patients with monoclonal gammopathy. J Exp Med. 2007;204(4):831–840. doi: 10.1084/jem.20062387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Bryant C, Suen H, Brown R, Yang S, Favaloro J, Aklilu E, Gibson J, Ho PJ, Iland H, Fromm P, Woodland N, Nassif N, Hart D, Joshua DE. Long-term survival in multiple myeloma is associated with a distinct immunological profile, which includes proliferative cytotoxic T-cell clones and a favourable Treg/Th17 balance. Blood Cancer J. 2013;3:e148. doi: 10.1038/bcj.2013.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Bogen B, Ruffini PA, Corthay A, Fredriksen AB, Froyland M, Lundin K, Rosjo E, Thompson K, Massaia M. Idiotype-specific immunotherapy in multiple myeloma: suggestions for future directions of research. Haematologica. 2006;91(7):941–948. [PubMed] [Google Scholar]
- 140.Bogen B, Schenck K, Munthe LA, Dembic Z. Deletion of idiotype (Id)-specific T cells in multiple myeloma. Acta Oncol. 2000;39(7):783–788. doi: 10.1080/028418600750063505. [DOI] [PubMed] [Google Scholar]
- 141.Rapoport AP, Stadtmauer EA, Binder-Scholl GK, Goloubeva O, Vogl DT, Lacey SF, Badros AZ, Garfall A, Weiss B, Finklestein J, Kulikovskaya I, Sinha SK, Kronsberg S, Gupta M, Bond S, Melchiori L, Brewer JE, Bennett AD, Gerry AB, Pumphrey NJ, Williams D, Tayton-Martin HK, Ribeiro L, Holdich T, Yanovich S, Hardy N, Yared J, Kerr N, Philip S, Westphal S, Siegel DL, Levine BL, Jakobsen BK, Kalos M, June CH. NY-ESO-1-specific TCR-engineered T cells mediate sustained antigen-specific antitumor effects in myeloma. Nat Med. 2015;21(8):914–921. doi: 10.1038/nm.3910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Yi Q, Osterborg A, Bergenbrant S, Mellstedt H, Holm G, Lefvert AK. Idiotype-reactive T-cell subsets and tumor load in monoclonal gammopathies. Blood. 1995;86(8):3043–3049. [PubMed] [Google Scholar]
- 143.Sharma A, Khan R, Joshi S, Kumar L, Sharma M. Dysregulation in T helper 1/T helper 2 cytokine ratios in patients with multiple myeloma. Leuk Lymphoma. 2010;51(5):920–927. doi: 10.3109/10428191003699563. [DOI] [PubMed] [Google Scholar]
- 144.Frassanito MA, Cusmai A, Dammacco F. Deregulated cytokine network and defective Th1 immune response in multiple myeloma. Clin Exp Immunol. 2001;125(2):190–197. doi: 10.1046/j.1365-2249.2001.01582.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Ogawara H, Handa H, Yamazaki T, Toda T, Yoshida K, Nishimoto N, Al-ma’Quol WH, Kaneko Y, Matsushima T, Tsukamoto N, Nojima Y, Matsumoto M, Sawamura M, Murakami H. High Th1/Th2 ratio in patients with multiple myeloma. Leuk Res. 2005;29(2):135–140. doi: 10.1016/j.leukres.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 146.Murakami H, Ogawara H, Hiroshi H. Th1/Th2 cells in patients with multiple myeloma. Hematology. 2004;9(1):41–45. doi: 10.1080/10245330310001652437. [DOI] [PubMed] [Google Scholar]
- 147.Feng P, Yan R, Dai X, Xie X, Wen H, Yang S. The alteration and clinical significance of Th1/Th2/Th17/Treg cells in patients with multiple myeloma. Inflammation. 2015;38(2):705–709. doi: 10.1007/s10753-014-9980-4. [DOI] [PubMed] [Google Scholar]
- 148.Prabhala RH, Neri P, Bae JE, Tassone P, Shammas MA, Allam CK, Daley JF, Chauhan D, Blanchard E, Thatte HS, Anderson KC, Munshi NC. Dysfunctional T regulatory cells in multiple myeloma. Blood. 2006;107(1):301–304. doi: 10.1182/blood-2005-08-3101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Beyer M, Kochanek M, Giese T, Endl E, Weihrauch MR, Knolle PA, Classen S, Schultze JL. In vivo peripheral expansion of naive CD4+CD25 high FoxP3+ regulatory T cells in patients with multiple myeloma. Blood. 2006;107(10):3940–3949. doi: 10.1182/blood-2005-09-3671. [DOI] [PubMed] [Google Scholar]
- 150.Giannopoulos K, Kaminska W, Hus I, Dmoszynska A. The frequency of T regulatory cells modulates the survival of multiple myeloma patients: detailed characterisation of immune status in multiple myeloma. Br J Cancer. 2012;106(3):546–552. doi: 10.1038/bjc.2011.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Braga WM, Atanackovic D, Colleoni GW. The role of regulatory T cells and TH17 cells in multiple myeloma. Clin Dev Immunol. 2012;2012:293479. doi: 10.1155/2012/293479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Shen CJ, Yuan ZH, Liu YX, Hu GY. Increased numbers of T helper 17 cells and the correlation with clinicopathological characteristics in multiple myeloma. J Int Med Res. 2012;40(2):556–564. doi: 10.1177/147323001204000217. [DOI] [PubMed] [Google Scholar]
- 153.Di Lullo G, Marcatti M, Heltai S, Brunetto E, Tresoldi C, Bondanza A, Bonini C, Ponzoni M, Tonon G, Ciceri F, Bordignon C, Protti MP. Th22 cells increase in poor prognosis multiple myeloma and promote tumor cell growth and survival. Oncoimmunology. 2015;4(5):e1005460. doi: 10.1080/2162402X.2015.1005460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.McEwen-Smith RM, Salio M, Cerundolo V. The regulatory role of invariant NKT cells in tumor immunity. Cancer Immunol Res. 2015;3(5):425–435. doi: 10.1158/2326-6066.CIR-15-0062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Song W, van der Vliet HJ, Tai YT, Prabhala R, Wang R, Podar K, Catley L, Shammas MA, Anderson KC, Balk SP, Exley MA, Munshi NC. Generation of antitumor invariant natural killer T cell lines in multiple myeloma and promotion of their functions via lenalidomide: a strategy for immunotherapy. Clin Cancer Res. 2008;14(21):6955–6962. doi: 10.1158/1078-0432.CCR-07-5290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Chang DH, Deng H, Matthews P, Krasovsky J, Ragupathi G, Spisek R, Mazumder A, Vesole DH, Jagannath S, Dhodapkar MV. Inflammation-associated lysophospholipids as ligands for CD1d-restricted T cells in human cancer. Blood. 2008;112(4):1308–1316. doi: 10.1182/blood-2008-04-149831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Dhodapkar MV, Geller MD, Chang DH, Shimizu K, Fujii S, Dhodapkar KM, Krasovsky J. A reversible defect in natural killer T cell function characterizes the progression of premalignant to malignant multiple myeloma. J Exp Med. 2003;197(12):1667–1676. doi: 10.1084/jem.20021650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Godfrey DI, Uldrich AP, McCluskey J, Rossjohn J, Moody DB. The burgeoning family of unconventional T cells. Nat Immunol. 2015;16(11):1114–1123. doi: 10.1038/ni.3298. [DOI] [PubMed] [Google Scholar]
- 159.Pratt G, Goodyear O, Moss P. Immunodeficiency and immunotherapy in multiple myeloma. Br J Haematol. 2007;138(5):563–579. doi: 10.1111/j.1365-2141.2007.06705.x. [DOI] [PubMed] [Google Scholar]
- 160.Kumar SK, Rajkumar SV, Dispenzieri A, Lacy MQ, Hayman SR, Buadi FK, Zeldenrust SR, Dingli D, Russell SJ, Lust JA, Greipp PR, Kyle RA, Gertz MA. Improved survival in multiple myeloma and the impact of novel therapies. Blood. 2008;111(5):2516–2520. doi: 10.1182/blood-2007-10-116129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Jimenez-Zepeda VH, Reece DE, Trudel S, Chen C, Franke N, Winter A, Tiedemann R, Kukreti V. Absolute lymphocyte count as predictor of overall survival for patients with multiple myeloma treated with single autologous stem cell transplant. Leuk Lymphoma. 2015 doi: 10.3109/10428194.2014.1003057. [DOI] [PubMed] [Google Scholar]
- 162.Wolniak KL, Goolsby CL, Chen YH, Chenn A, Singhal S, LA JayeshMehta Peterson. Expansion of a clonal CD8+CD57+ large granular lymphocyte population after autologous stem cell transplant in multiple myeloma. Am J Clin Pathol. 2013;139(2):231–241. doi: 10.1309/AJCP1T0JPBLSLAQF. [DOI] [PubMed] [Google Scholar]
- 163.Noonan KA, Huff CA, Davis J, Lemas MV, Fiorino S, Bitzan J, Ferguson A, Emerling A, Luznik L, Matsui W, Powell J, Fuchs E, Rosner GL, Epstein C, Rudraraju L, Ambinder RF, Jones RJ, Pardoll D, Borrello I. Adoptive transfer of activated marrow-infiltrating lymphocytes induces measurable antitumor immunity in the bone marrow in multiple myeloma. Sci Transl Med. 2015;7(288):288ra278. doi: 10.1126/scitranslmed.aaa7014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Karp Leaf R, Cho HJ, Avigan D. Immunotherapy for multiple myeloma, past, present, and future: monoclonal antibodies, vaccines, and cellular therapies. Curr Hematol Malig Rep. 2015 doi: 10.1007/s11899-015-0283-0. [DOI] [PubMed] [Google Scholar]
- 165.Wang L, Jin N, Schmitt A, Greiner J, Malcherek G, Hundemer M, Mani J, Hose D, Raab MS, Ho AD, Chen BA, Goldschmidt H, Schmitt M. T cell-based targeted immunotherapies for patients with multiple myeloma. Int J Cancer J Int Cancer. 2015;136(8):1751–1768. doi: 10.1002/ijc.29190. [DOI] [PubMed] [Google Scholar]
- 166.Freeman LM, Lam A, Petcu E, Smith R, Salajegheh A, Diamond P, Zannettino A, Evdokiou A, Luff J, Wong PF, Khalil D, Waterhouse N, Vari F, Rice AM, Catley L, Hart DN, Vuckovic S. Myeloma-induced alloreactive T cells arising in myeloma-infiltrated bones include double-positive CD8+CD4+ T cells: evidence from myeloma-bearing mouse model. J Immunol. 2011;187(8):3987–3996. doi: 10.4049/jimmunol.1101202. [DOI] [PubMed] [Google Scholar]
- 167.Bensinger W. Allogeneic stem cell transplantation for multiple myeloma. Hematol Oncol Clin N Am. 2014;28(5):891–902. doi: 10.1016/j.hoc.2014.06.001. [DOI] [PubMed] [Google Scholar]
- 168.Kumar S, Gertz MA, Dispenzieri A, Lacy MQ, Geyer SM, Iturria NL, Fonseca R, Hayman SR, Lust JA, Kyle RA, Greipp PR, Witzig TE, Rajkumar SV. Response rate, durability of response, and survival after thalidomide therapy for relapsed multiple myeloma. Mayo Clin Proc. 2003;78(1):34–39. doi: 10.4065/78.1.34. [DOI] [PubMed] [Google Scholar]
- 169.Kumar S, Witzig TE, Dispenzieri A, Lacy MQ, Wellik LE, Fonseca R, Lust JA, Gertz MA, Kyle RA, Greipp PR, Rajkumar SV. Effect of thalidomide therapy on bone marrow angiogenesis in multiple myeloma. Leukemia. 2004;18(3):624–627. doi: 10.1038/sj.leu.2403285. [DOI] [PubMed] [Google Scholar]
- 170.Hideshima T, Chauhan D, Shima Y, Raje N, Davies FE, Tai YT, Treon SP, Lin B, Schlossman RL, Richardson P, Muller G, Stirling DI, Anderson KC. Thalidomide and its analogs overcome drug resistance of human multiple myeloma cells to conventional therapy. Blood. 2000;96(9):2943–2950. [PubMed] [Google Scholar]
- 171.Corral LG, Haslett PA, Muller GW, Chen R, Wong LM, Ocampo CJ, Patterson RT, Stirling DI, Kaplan G. Differential cytokine modulation and T cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-alpha. J Immunol. 1999;163(1):380–386. [PubMed] [Google Scholar]
- 172.Quach H, Ritchie D, Stewart AK, Neeson P, Harrison S, Smyth MJ, Prince HM. Mechanism of action of immunomodulatory drugs (IMiDS) in multiple myeloma. Leukemia. 2010;24(1):22–32. doi: 10.1038/leu.2009.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Gorgun G, Calabrese E, Soydan E, Hideshima T, Perrone G, Bandi M, Cirstea D, Santo L, Hu Y, Tai YT, Nahar S, Mimura N, Fabre C, Raje N, Munshi N, Richardson P, Anderson KC. Immunomodulatory effects of lenalidomide and pomalidomide on interaction of tumor and bone marrow accessory cells in multiple myeloma. Blood. 2010;116(17):3227–3237. doi: 10.1182/blood-2010-04-279893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Luptakova K, Rosenblatt J, Glotzbecker B, Mills H, Stroopinsky D, Kufe T, Vasir B, Arnason J, Tzachanis D, Zwicker JI, Joyce RM, Levine JD, Anderson KC, Kufe D, Avigan D. Lenalidomide enhances anti-myeloma cellular immunity. Cancer Immunol Immunother: CII. 2013;62(1):39–49. doi: 10.1007/s00262-012-1308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Davies FE, Raje N, Hideshima T, Lentzsch S, Young G, Tai YT, Lin B, Podar K, Gupta D, Chauhan D, Treon SP, Richardson PG, Schlossman RL, Morgan GJ, Muller GW, Stirling DI, Anderson KC. Thalidomide and immunomodulatory derivatives augment natural killer cell cytotoxicity in multiple myeloma. Blood. 2001;98(1):210–216. doi: 10.1182/blood.V98.1.210. [DOI] [PubMed] [Google Scholar]
- 176.Jungkunz-Stier I, Zekl M, Stuhmer T, Einsele H, Seggewiss-Bernhardt R. Modulation of natural killer cell effector functions through lenalidomide/dasatinib and their combined effects against multiple myeloma cells. Leuk Lymphoma. 2014;55(1):168–176. doi: 10.3109/10428194.2013.794270. [DOI] [PubMed] [Google Scholar]
- 177.Zhu D, Corral LG, Fleming YW, Stein B. Immunomodulatory drugs revlimid (lenalidomide) and CC-4047 induce apoptosis of both hematological and solid tumor cells through NK cell activation. Cancer Immunol Immunother: CII. 2008;57(12):1849–1859. doi: 10.1007/s00262-008-0512-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Hayashi T, Hideshima T, Akiyama M, Podar K, Yasui H, Raje N, Kumar S, Chauhan D, Treon SP, Richardson P, Anderson KC. Molecular mechanisms whereby immunomodulatory drugs activate natural killer cells: clinical application. Br J Haematol. 2005;128(2):192–203. doi: 10.1111/j.1365-2141.2004.05286.x. [DOI] [PubMed] [Google Scholar]
- 179.Chang DH, Liu N, Klimek V, Hassoun H, Mazumder A, Nimer SD, Jagannath S, Dhodapkar MV. Enhancement of ligand-dependent activation of human natural killer T cells by lenalidomide: therapeutic implications. Blood. 2006;108(2):618–621. doi: 10.1182/blood-2005-10-4184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Minnema MC, van der Veer MS, Aarts T, Emmelot M, Mutis T, Lokhorst HM. Lenalidomide alone or in combination with dexamethasone is highly effective in patients with relapsed multiple myeloma following allogeneic stem cell transplantation and increases the frequency of CD4+Foxp3+ T cells. Leukemia. 2009;23(3):605–607. doi: 10.1038/leu.2008.247. [DOI] [PubMed] [Google Scholar]
- 181.Spina F, Montefusco V, Crippa C, Citro A, Sammassimo S, Olivero B, Gentili S, Galli M, Guglielmelli T, Rossi D, Falcone AP, Grasso M, Patriarca F, De Muro M, Corradini P. Lenalidomide can induce long-term responses in patients with multiple myeloma relapsing after multiple chemotherapy lines, in particular after allogeneic transplant. Leuk Lymphoma. 2011;52(7):1262–1270. doi: 10.3109/10428194.2011.564695. [DOI] [PubMed] [Google Scholar]
- 182.Lioznov M, El-Cheikh J, Jr, Hoffmann F, Hildebrandt Y, Ayuk F, Wolschke C, Atanackovic D, Schilling G, Badbaran A, Bacher U, Fehse B, Zander AR, Blaise D, Mohty M, Kroger N. Lenalidomide as salvage therapy after allo-SCT for multiple myeloma is effective and leads to an increase of activated NK (NKp44(+)) and T (HLA-DR(+)) cells. Bone Marrow Transpl. 2010;45(2):349–353. doi: 10.1038/bmt.2009.155. [DOI] [PubMed] [Google Scholar]
- 183.Nencioni A, Grunebach F, Patrone F, Ballestrero A, Brossart P. Proteasome inhibitors: antitumor effects and beyond. Leukemia. 2007;21(1):30–36. doi: 10.1038/sj.leu.2404444. [DOI] [PubMed] [Google Scholar]
- 184.Jagannath S, Barlogie B, Berenson J, Siegel D, Irwin D, Richardson PG, Niesvizky R, Alexanian R, Limentani SA, Alsina M, Adams J, Kauffman M, Esseltine DL, Schenkein DP, Anderson KC. A phase 2 study of two doses of bortezomib in relapsed or refractory myeloma. Br J Haematol. 2004;127(2):165–172. doi: 10.1111/j.1365-2141.2004.05188.x. [DOI] [PubMed] [Google Scholar]
- 185.Pellom ST, Jr, Dudimah DF, Thounaojam MC, Sayers TJ, Shanker A. Modulatory effects of bortezomib on host immune cell functions. Immunotherapy. 2015 doi: 10.2217/imt.15.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Shi J, Tricot GJ, Garg TK, Malaviarachchi PA, Szmania SM, Kellum RE, Storrie B, Mulder A, Shaughnessy JD, Jr, Barlogie B, van Rhee F. Bortezomib down-regulates the cell-surface expression of HLA class I and enhances natural killer cell-mediated lysis of myeloma. Blood. 2008;111(3):1309–1317. doi: 10.1182/blood-2007-03-078535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Spisek R, Charalambous A, Mazumder A, Vesole DH, Jagannath S, Dhodapkar MV. Bortezomib enhances dendritic cell (DC)-mediated induction of immunity to human myeloma via exposure of cell surface heat shock protein 90 on dying tumor cells: therapeutic implications. Blood. 2007;109(11):4839–4845. doi: 10.1182/blood-2006-10-054221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Chauhan D, Catley L, Li G, Podar K, Hideshima T, Velankar M, Mitsiades C, Mitsiades N, Yasui H, Letai A, Ovaa H, Berkers C, Nicholson B, Chao TH, Neuteboom ST, Richardson P, Palladino MA, Anderson KC. A novel orally active proteasome inhibitor induces apoptosis in multiple myeloma cells with mechanisms distinct from Bortezomib. Cancer Cell. 2005;8(5):407–419. doi: 10.1016/j.ccr.2005.10.013. [DOI] [PubMed] [Google Scholar]
- 189.Nencioni A, Schwarzenberg K, Brauer KM, Schmidt SM, Ballestrero A, Grunebach F, Brossart P. Proteasome inhibitor bortezomib modulates TLR4-induced dendritic cell activation. Blood. 2006;108(2):551–558. doi: 10.1182/blood-2005-08-3494. [DOI] [PubMed] [Google Scholar]
- 190.Feng X, Yan J, Wang Y, Zierath JR, Nordenskjold M, Henter JI, Fadeel B, Zheng C. The proteasome inhibitor bortezomib disrupts tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) expression and natural killer (NK) cell killing of TRAIL receptor-positive multiple myeloma cells. Mol Immunol. 2010;47(14):2388–2396. doi: 10.1016/j.molimm.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 191.Lundqvist A, Yokoyama H, Smith A, Berg M, Childs R. Bortezomib treatment and regulatory T-cell depletion enhance the antitumor effects of adoptively infused NK cells. Blood. 2009;113(24):6120–6127. doi: 10.1182/blood-2008-11-190421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Sun K, Welniak LA, Panoskaltsis-Mortari A, O’Shaughnessy MJ, Liu H, Barao I, Riordan W, Sitcheran R, Wysocki C, Serody JS, Blazar BR, Sayers TJ, Murphy WJ. Inhibition of acute graft-versus-host disease with retention of graft-versus-tumor effects by the proteasome inhibitor bortezomib. Proc Natl Acad Sci USA. 2004;101(21):8120–8125. doi: 10.1073/pnas.0401563101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Sun K, Wilkins DE, Anver MR, Sayers TJ, Panoskaltsis-Mortari A, Blazar BR, Welniak LA, Murphy WJ. Differential effects of proteasome inhibition by bortezomib on murine acute graft-versus-host disease (GVHD): delayed administration of bortezomib results in increased GVHD-dependent gastrointestinal toxicity. Blood. 2005;106(9):3293–3299. doi: 10.1182/blood-2004-11-4526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Vodanovic-Jankovic S, Hari P, Jacobs P, Komorowski R, Drobyski WR. NF-kappaB as a target for the prevention of graft-versus-host disease: comparative efficacy of bortezomib and PS-1145. Blood. 2006;107(2):827–834. doi: 10.1182/blood-2005-05-1820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.McBride A, Klaus JO, Stockerl-Goldstein K. Carfilzomib: a second-generation proteasome inhibitor for the treatment of multiple myeloma. Am J Health-Syst Pharm: AJHP. 2015;72(5):353–360. doi: 10.2146/ajhp130281. [DOI] [PubMed] [Google Scholar]
- 196.Porrata LF, Litzow MR, Markovic SN. Immune reconstitution after autologous hematopoietic stem cell transplantation. Mayo Clin Proc. 2001;76(4):407–412. doi: 10.1016/S0025-6196(11)62388-4. [DOI] [PubMed] [Google Scholar]
- 197.Kroger N, Shaw B, Iacobelli S, Zabelina T, Peggs K, Shimoni A, Nagler A, Binder T, Eiermann T, Madrigal A, Schwerdtfeger R, Kiehl M, Sayer HG, Beyer J, Bornhauser M, Ayuk F, Zander AR, Marks DI, Clinical Trial Committee of the British Society of B. Marrow T, the German Cooperative Transplant G Comparison between antithymocyte globulin and alemtuzumab and the possible impact of KIR-ligand mismatch after dose-reduced conditioning and unrelated stem cell transplantation in patients with multiple myeloma. Br J Haematol. 2005;129(5):631–643. doi: 10.1111/j.1365-2141.2005.05513.x. [DOI] [PubMed] [Google Scholar]
- 198.Shi J, Tricot G, Szmania S, Rosen N, Garg TK, Malaviarachchi PA, Moreno A, Dupont B, Hsu KC, Baxter-Lowe LA, Cottler-Fox M, Shaughnessy JD, Jr, Barlogie B, van Rhee F. Infusion of haplo-identical killer immunoglobulin-like receptor ligand mismatched NK cells for relapsed myeloma in the setting of autologous stem cell transplantation. Br J Haematol. 2008;143(5):641–653. doi: 10.1111/j.1365-2141.2008.07340.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Benson DM, Jr, Hofmeister CC, Padmanabhan S, Suvannasankha A, Jagannath S, Abonour R, Bakan C, Andre P, Efebera Y, Tiollier J, Caligiuri MA, Farag SS. A phase 1 trial of the anti-KIR antibody IPH2101 in patients with relapsed/refractory multiple myeloma. Blood. 2012;120(22):4324–4333. doi: 10.1182/blood-2012-06-438028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Benson DM, Jr, Cohen AD, Jagannath S, Munshi NC, Spitzer G, Hofmeister CC, Efebera YA, Andre P, Zerbib R, Caligiuri MA. A phase I trial of the anti-KIR antibody IPH2101 and lenalidomide in patients with relapsed/refractory multiple myeloma. Clin Cancer Res. 2015;21(18):4055–4061. doi: 10.1158/1078-0432.CCR-15-0304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Jiang H, Zhang W, Shang P, Zhang H, Fu W, Ye F, Zeng T, Huang H, Zhang X, Sun W, Man-Yuen Sze D, Yi Q, Hou J. Transfection of chimeric anti-CD138 gene enhances natural killer cell activation and killing of multiple myeloma cells. Mol Oncol. 2014;8(2):297–310. doi: 10.1016/j.molonc.2013.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Richardson PG, Lonial S, Jakubowiak AJ, Harousseau JL, Anderson KC. Monoclonal antibodies in the treatment of multiple myeloma. Br J Haematol. 2011;154(6):745–754. doi: 10.1111/j.1365-2141.2011.08790.x. [DOI] [PubMed] [Google Scholar]
- 203.Laubach JP, Tai YT, Richardson PG, Anderson KC. Daratumumab granted breakthrough drug status. Expert Opin Investig Drugs. 2014;23(4):445–452. doi: 10.1517/13543784.2014.889681. [DOI] [PubMed] [Google Scholar]
- 204.Starr P. Elotuzumab, first-in-class monoclonal antibody immunotherapy, improves outcomes in patients with multiple myeloma. Am Health Drug Benefits. 2015;8(Spec Issue):17. [PMC free article] [PubMed] [Google Scholar]
- 205.Hsi ED, Steinle R, Balasa B, Szmania S, Draksharapu A, Shum BP, Huseni M, Powers D, Nanisetti A, Zhang Y, Rice AG, van Abbema A, Wong M, Liu G, Zhan F, Dillon M, Chen S, Rhodes S, Fuh F, Tsurushita N, Kumar S, Vexler V, Shaughnessy JD, Jr, Barlogie B, van Rhee F, Hussein M, Afar DE, Williams MB. CS1, a potential new therapeutic antibody target for the treatment of multiple myeloma. Clin Cancer Res. 2008;14(9):2775–2784. doi: 10.1158/1078-0432.CCR-07-4246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Collins SM, Bakan CE, Swartzel GD, Hofmeister CC, Efebera YA, Kwon H, Starling GC, Ciarlariello D, Bhaskar S, Briercheck EL, Hughes T, Yu J, Rice A, Benson DM., Jr Elotuzumab directly enhances NK cell cytotoxicity against myeloma via CS1 ligation: evidence for augmented NK cell function complementing ADCC. Cancer Immunol Immunother: CII. 2013;62(12):1841–1849. doi: 10.1007/s00262-013-1493-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Wolchok JD, Kluger H, Callahan MK, Postow MA, Rizvi NA, Lesokhin AM, Segal NH, Ariyan CE, Gordon RA, Reed K, Burke MM, Caldwell A, Kronenberg SA, Agunwamba BU, Zhang X, Lowy I, Inzunza HD, Feely W, Horak CE, Hong Q, Korman AJ, Wigginton JM, Gupta A, Sznol M. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369(2):122–133. doi: 10.1056/NEJMoa1302369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Armand P. Immune checkpoint blockade in hematologic malignancies. Blood. 2015;125(22):3393–3400. doi: 10.1182/blood-2015-02-567453. [DOI] [PubMed] [Google Scholar]
- 209.Rosenblatt J, Glotzbecker B, Mills H, Vasir B, Tzachanis D, Levine JD, Joyce RM, Wellenstein K, Keefe W, Schickler M, Rotem-Yehudar R, Kufe D, Avigan D. PD-1 blockade by CT-011, anti-PD-1 antibody, enhances ex vivo T-cell responses to autologous dendritic cell/myeloma fusion vaccine. J Immunother. 2011;34(5):409–418. doi: 10.1097/CJI.0b013e31821ca6ce. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Benson DM, Jr, Bakan CE, Mishra A, Hofmeister CC, Efebera Y, Becknell B, Baiocchi RA, Zhang J, Yu J, Smith MK, Greenfield CN, Porcu P, Devine SM, Rotem-Yehudar R, Lozanski G, Byrd JC, Caligiuri MA. The PD-1/PD-L1 axis modulates the natural killer cell versus multiple myeloma effect: a therapeutic target for CT-011, a novel monoclonal anti-PD-1 antibody. Blood. 2010;116(13):2286–2294. doi: 10.1182/blood-2010-02-271874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Jing W, Gershan JA, Weber J, Tlomak D, McOlash L, Sabatos-Peyton C, Johnson BD. Combined immune checkpoint protein blockade and low dose whole body irradiation as immunotherapy for myeloma. J Immunother Cancer. 2015;3(1):2. doi: 10.1186/s40425-014-0043-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Murillo O, Arina A, Hervas-Stubbs S, Gupta A, McCluskey B, Dubrot J, Palazon A, Azpilikueta A, Ochoa MC, Alfaro C, Solano S, Perez-Gracia JL, Oyajobi BO, Melero I. Therapeutic antitumor efficacy of anti-CD137 agonistic monoclonal antibody in mouse models of myeloma. Clin Cancer Res. 2008;14(21):6895–6906. doi: 10.1158/1078-0432.CCR-08-0285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Lesokhin AM, Callahan MK, Postow MA, Wolchok JD. On being less tolerant: enhanced cancer immunosurveillance enabled by targeting checkpoints and agonists of T cell activation. Sci Transl Med. 2015;7(280):280sr281. doi: 10.1126/scitranslmed.3010274. [DOI] [PubMed] [Google Scholar]
- 214.Hoang MD, Jung SH, Lee HJ, Lee YK, Nguyen-Pham TN, Choi NR, Vo MC, Lee SS, Ahn JS, Yang DH, Kim YK, Kim HJ, Lee JJ. Dendritic cell-based cancer immunotherapy against multiple myeloma: from bench to clinic. Chonnam Med J. 2015;51(1):1–7. doi: 10.4068/cmj.2015.51.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Maus MV, June CH. CARTs on the road for myeloma. Clin Cancer Res. 2014;20(15):3899–3901. doi: 10.1158/1078-0432.CCR-14-0721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Dunn GP, Old LJ, Schreiber RD. The three Es of cancer immunoediting. Annu Rev Immunol. 2004;22:329–360. doi: 10.1146/annurev.immunol.22.012703.104803. [DOI] [PubMed] [Google Scholar]