Abstract
Background
Primary Sjögren's syndrome (pSS) is recognized for its autoimmune origin. Its hallmark symptoms, dry eyes and mouth, result from glandular inflammation. Prior literature indicates that pSS not only affects the peripheral system but also involves the central nervous system (CNS), giving rise to various neuropsychiatric symptoms. However, there is limited published research on the psychiatric comorbidities in individuals with pSS.
Methods
A comprehensive search was conducted on PubMed and Google Scholar for this narrative review. The search spanned from inception until August 2023. Its aim was to locate studies focusing on the psychiatric manifestations of pSS and the potential underlying mechanisms.
Results
The most commonly reported psychiatric complications among these individuals are depression and cognitive dysfunction. Other psychiatric manifestations that have been reported in pSS individuals include anxiety, sleep disorders, psychosis, catatonia, bipolar disorder, and obsessive-compulsive disorder.
Conclusion
In conclusion, patients with pSS often display multiple psychiatric symptoms. These symptoms can significantly impair functioning and reduce quality of life. Hence, prompt diagnosis and management are crucial.
1. Introduction
Sjögren's syndrome (SS) is an autoimmune disorder characterized by the infiltration of mononuclear cells and subsequent damage to the salivary and lacrimal glands. This syndrome can be either secondary Sjögren's syndrome, occurring with another autoimmune disease, or primary Sjögren's syndrome (pSS) when it manifests independently [1]. The prevalence of pSS varies across different populations and regions, but it is estimated to affect approximately 0.1% to 3% of the general population [2, 3]. The female-to-male ratio is around 9 : 1 [4]. Emerging evidence suggests that pSS can also affect the nervous system, leading to various psychiatric manifestations [5]. However, there is limited published research on the epidemiological aspects of these comorbidities among individuals with pSS. The prevalence of psychiatric manifestations in pSS varies widely across prior studies due to discrepancies in their methods [6–9]. It appears that psychiatric disorders are the primary manifestations of the disease. They do not appear to result from complications of the disease, its treatment, or a concurrent non-pSS-related event [10]. These conditions include various psychiatric disorders [11]. The comorbid psychiatric disorders include cognitive dysfunction [12], depression [13], bipolar mood disorder [14], psychosis [15, 16], catatonia [15, 17], and obsessive-compulsive disorder [11, 18]. Cognitive dysfunction, often called “brain fog,” is the most common psychiatric manifestation reported by pSS patients. This can affect attention, memory, and information processing speed [19]. Brain fog is usually followed by depression, which significantly impact patients' quality of life [6].
The precise underlying mechanism contributing to psychiatric involvement in pSS remains unknown. However, several potential mechanisms have been suggested. Antineuronal antibodies, such as those targeting the NR2 subunit of the NMDA receptor (anti-NR2 antibodies), antiribosomal P proteins found on neuronal surfaces (anti-P antibodies), and anti-aquaporin-4 (AQP4) antibodies, have been identified as potentially affecting the brain [12, 20, 21]. These antibodies are thought to contribute to the development of these conditions [20–23]. Other possible mechanisms are inflammation-related disturbances in central nervous system (CNS) [24], neurotransmitter dysregulation [25, 26], and ischemic damage caused by CNS vasculitis [11, 27].
Diagnosing these manifestations is challenging due to the lack of specific biomarkers and overlapping symptoms with other rheumatic and psychiatric disorders. Collaboration among rheumatologists, neurologists, and psychiatrists is crucial for accurate diagnosis and appropriate management [28]. Treatment strategies are multidimensional with the goal to alleviate symptoms, improve quality of life, and manage the underlying autoimmune process. Symptomatic treatment options include cognitive rehabilitation, antidepressant and anxiolytic medications, and pain management. Immunomodulatory therapies, such as corticosteroids, disease-modifying antirheumatic drugs, and biologic agents, should also be considered for patients [29, 30].
These manifestations significantly impact patients' quality of life [6]. They can also be the primary presenting symptoms of the disease in 50% to 80% of patients [31]. This can further complicate the diagnosis of pSS. Therefore, it is crucial to begin screening for these complications at disease onset and treat them adequately with the most effective options available.
In this paper, our aim is to review existing literature to enhance understanding of psychiatric aspects in pSS. We aim to shed light on the underlying mechanisms, clinical manifestations, diagnostic challenges, and treatment approaches.
2. Materials and Methods
We conducted a thorough search of PubMed and Google Scholar for this narrative review from the beginning until August 2023. Our aim was to find studies addressing psychiatric symptoms in pSS and their potential underlying mechanisms, without limiting based on publication dates. Only English language articles were considered during the review process. The search terms used included “Sjögren's syndrome,” “Sjögren,” “psychiatric manifestations,” “Psychiatric conditions,” “bipolar”, “obsessive-compulsive,” “obsession,” “anxiety disorder,” “depressive disorder,” “sleep disorder,” “catatonia,” “psychosis,” and “cognitive impairment.” Upon identification of each pertinent article, the reference list was explored to find additional relevant sources. Gray literature was not included in the search process.
3. Results
We found 1500 records through a database search, along with screening titles and abstracts. Upon reading the full texts, we excluded 1395 papers. In total, 105 full-text articles were included in this study.
The most common reported psychiatric complications among these individuals were depression and cognitive dysfunction. Other psychiatric manifestations that have been reported in pSS individuals include anxiety, sleep disorders, psychosis, catatonia, bipolar disorder, and obsessive-compulsive disorder (Box 1).
Box 1.

Psychiatric comorbidities in primary Sjögren's syndrome (pSS).
We extensively examined each of these psychiatric manifestations and their potential underlying pathophysiology, as well as their diagnosis and treatment approaches.
3.1. Sleep Disorders
Sleep disturbances are increasingly found to be an essential clinical presentation of rheumatologic disorders [32]. Pronounced sleep issues were found in pSS, with one study revealing that 75% of pSS patients reported moderate to severe sleep disruptions [33].
A recent meta-analysis underscored heightened sleep disruptions in pSS patients compared with controls. These disruptions encompass drowsiness, self-reported sleep problems, and frequent nocturnal awakenings [34]. Managing these disturbances is crucial, as they can significantly impact the quality of life in pSS patients [6, 35]. Accurate diagnosis and proper treatment of these sleep problems could also relieve associated symptoms like pain, mood swings, and fatigue, which leads to improved physical and cognitive performance and overall well-being [34]. Using a standardized sleep questionnaire, Gudbjörnsson et al. found a significantly greater sleep deficit (defined as the difference between required and actual sleep time) in pSS patients compared to healthy controls and rheumatoid arthritis (RA) patients [36]. This study revealed significant difficulties in sleep initiation and maintenance in pSS patients.
The sleep disturbances in pSS are related to several factors. These factors may overlap with symptoms of fibromyalgia [33]. They also relate to mood disorders [37], muscle and joint pain, and sleep interruptions due to hydration needs from sicca symptoms [38]. Restless leg syndrome and coexisting obstructive sleep apnea (OSA) are also more prevalent in pSS patients and cause the sleep problems [39]. A recent case-control study in Italy evaluated the sleep quality in 29 female patients with pSS using Pittsburgh Sleep Quality Index (PSQI) [35]. They found reduced sleep quality in 83% of pSS patients, which was correlated with mood disturbances, as well as low levels of physical and mental health in these patients [35].
Effective management of sleep disruption needs a comprehensive approach. Treating the underlying autoimmune inflammation is necessary, often achieved through immunosuppressive medications [40]. Additionally, it is crucial to address dryness-related symptoms, which are a hallmark of pSS. Moisturizing therapy for the eyes and mouth can alleviate discomfort, thereby promoting more restful sleep [38]. Moreover, cognitive behavioral therapy for insomnia (CBT-I) has shown promise in improving sleep quality by modifying maladaptive sleep behaviors and thought patterns [41].
3.2. Psychosis and Catatonia
While peripheral nervous system (PNS) disease is a well-established complication of pSS, this disorder's CNS complications have received relatively little attention until recently [42]. Although CNS involvement may be infrequent, it remains a significant complication. CNS involvement can manifest with focal symptoms like motor, sensory, or speech disturbances. It can also present with nonfocal neurological symptoms such as cognitive dysfunction and various psychiatric disorders [7]. Spezialetti et al. evaluated a group of 77 patients with both secondary SS and pSS who also had CNS involvement and reported psychiatric and cognitive dysfunction in over 80% of these patients, with psychosis occurring in 8% [12]. While comorbid depression is commonly reported in patients with pSS, comorbid psychosis is a relatively rare condition [12, 16].
Catatonia manifests as a range of physical and behavioral symptoms that arise from various psychiatric, neurological, or medical sources. This condition, which affects motor functioning, behavior, and emotional responses, is associated with numerous underlying factors [43]. A recent meta-analysis revealed a mean prevalence of catatonia to be 9.0%. The highest prevalence was observed in the presence of medical or neurological illness without comorbid psychiatric conditions (20.6%), while the lowest prevalence was found in mixed psychiatric samples (5.7%) [44]. The prevalence of catatonia in rheumatologic patients has not been extensively studied and documented to date. However, there are a few case studies highlighting its manifestation in rheumatologic disorders. One of the most prevalent occurrences is in anti N-methyl-D-aspartate receptor (NMDAR) encephalitis, with an estimated prevalence of 64% [45]. A significant number of conditions that make individuals prone to catatonia demonstrate connections with neuroinflammation. Neuroinflammation, characterized by microglial activation and the release of proinflammatory cytokines, disrupts neurotransmitter balance and synaptic functioning. This disruption contributes to the motor abnormalities and cognitive deficits observed in catatonia [46]. Immune system dysregulation, including autoimmunity and elevated levels of inflammatory markers, has been noted in individuals with catatonic features. This further underscores the link between the immune response and neuropsychiatric manifestations [45].
Psychosis is a mental health condition characterized by a disconnection from reality, with symptoms including hallucinations, delusions, disorganized thinking, and impaired insight. It can manifest as a primary feature of psychotic disorders like schizophrenia or as a symptom of other mental health conditions, neurological and medical disorders, or substance abuse [47]. A recent meta-analysis found the median lifetime prevalence of psychosis to be 7.49 per 1000 [48]. The majority of epidemiological studies have established a broad association between autoimmunity and psychotic disorders [49, 50]. In a Danish nationwide study, the risk of psychosis following an autoimmune disease diagnosis was found to increase by 45% [51]. Additionally, a recent meta-analysis by Cullen et al. [49] revealed that a diagnosis of a non-neurological autoimmune disease raised the risk of subsequent psychotic disorder diagnosis by 43%.
There are limited case studies on the presence of psychosis and catatonia in pSS [16, 23, 52–55]. Raps et al. presented a case of a 47-year-old man admitted due to psychosis, previously diagnosed with schizophrenia 18 years ago. During admission, pSS was clinically and historically diagnosed, and an unexplained temporal relationship between psychotic flare-ups and highly elevated erythrocyte sedimentation rates (ESR) was noted [53]. Two case studies reported female adolescents with new onset psychosis which did not typically respond to antipsychotic medications. Further investigations confirmed a diagnosis of pSS [16, 55]. Interestingly, rituximab infusion resulted in an improvement of psychiatric symptoms in these patients. This suggests that early consideration of rituximab as a treatment option for pSS-associated psychiatric disturbances may be warranted [16, 55]. Interestingly, rituximab infusion led to an improvement of psychiatric symptoms in these patients, indicating that early consideration of rituximab as a treatment option for pSS-associated psychiatric disturbances may be warranted [55]. Moreover, Moll et al. managed similar psychotic symptoms by prescribing oral prednisone. Remarkably, within a week, the patient had a considerable reduction in visual hallucinations and paranoid ideation, alongside an increased IQ [23].
Only two case studies reported catatonia and psychosis in pSS patients [16, 54]. Rosado et al. documented the case of a 21-year-old African female with psychosis. She initially received risperidone but later experienced a catatonic state characterized by negative mood, mutism, immobility, and refusal to eat or drink, along with fever. Further investigations revealed lymphadenopathy, bicytopenia, and elevated inflammatory markers. A salivary gland biopsy confirmed the diagnosis of pSS. The patient's psychiatric symptoms improved upon discontinuation of antipsychotic medications and the initiation of immunosuppressive therapy with hydroxychloroquine and prednisolone [54]. Similarly, Sivakumar et al. reported a 19-year-old female with acute onset psychosis and catatonia whose symptoms worsened with antipsychotic medications. She was later diagnosed with pSS, and her psychiatric symptoms gradually improved on immunomodulators [16]. Previous literature also reported instances where immunosuppressants such as azathioprine, methotrexate, and cyclosporine yielded positive outcomes in the treatment of pSS-related CNS involvements [56]. It is important to note that psychotropic medications should be administered cautiously in patients with autoimmune diseases. Some of these drugs, such as chlorpromazine, carbamazepine, and lithium carbonate, are known to potentially trigger autoimmune-like conditions [57].
As emphasized in these studies, new-onset psychosis can be the initial manifestation of pSS. Therefore, it is crucial to consider pSS in the differential diagnosis when assessing patients with these emergent symptoms, especially if they do not respond typically to psychotropic medications.
3.3. Obsessive-Compulsive Disorder
Obsessive-compulsive disorder (OCD) is a mental health condition characterized by intrusive, distressing thoughts (obsessions) and repetitive behaviors or mental acts performed to reduce the anxiety associated with these obsessions (compulsions). These behaviors can significantly impair a person's daily functioning and quality of life [58]. OCD can manifest as an initial symptom before the more commonly recognized symptoms of pSS appear [59, 60]. Wang et al. conducted a cohort study on a nationwide sample of patients with systemic autoimmune diseases including pSS. They found an increased risk of OCD in these populations with a hazard ratio of 2.38 in pSS patients [61]. Moreover, in a study of 103 pSS patients by Karaiskos et al. [62], 27.2% displayed traits of obsessiveness in their personalities. This heightened prevalence of OCD can significantly affect overall well-being and contribute to increased distress among these individuals [61].
Only a few studies have explored the connection between pSS and OCD. In one case report, a 17-year-old female with treatment-resistant OCD and depression was later diagnosed with pSS during her hospitalization. Her OCD symptoms completely disappeared after initiating immunotherapy with plasmapheresis and intravenous methylprednisolone [59]. Similarly, De Carvalho and Ribeiro described a case of a 40-year-old woman who initially presented with dry mouth and eyes alongside behavioral changes. She was diagnosed with OCD and treated with fluoxetine, risperidone, and later aripiprazole. However, her laboratory tests revealed positive autoimmune markers for pSS, and diagnostic tests confirmed it. She was started on hydroxychloroquine which completely controlled her OCD symptoms without psychiatric medications [63].
There are several possible mechanisms regarding the link between OCD and pSS. Some studies suggest that cytokines found in pSS may cross the blood-brain barrier, potentially causing OCD through various processes such as causing abnormalities in serotonergic signaling [64, 65]. Additionally, pSS could exacerbate OCD symptoms. This may occur in individuals with subclinical OCD vulnerability. It could happen directly by affecting the biological basis of OCD. Alternatively, it could occur indirectly by worsening vulnerabilities to depression or other anxiety disorders [66]. Another hypothesis suggests a shared genetic or epigenetic basis between pSS and OCD. This is similar to observed features in other autoimmune diseases associated with OCD, like rheumatic fever [67]. Future research should aim at documenting more cases of individuals with both pSS and OCD diagnoses. Additionally, it should investigate the role of immunosuppressive drugs in managing OCD symptoms.
3.4. Bipolar Disorder
Bipolar disorder (BD) is a complex mental health condition characterized by extreme mood swings. These mood swings can fluctuate between periods of elevated and irritable mood (mania or hypomania) and depressive episodes [68]. Previous studies have shown that systemic autoimmune diseases including pSS are associated with an increased risk of BD [14, 69]. However, research on the connection between BD and pSS remains limited.
Chebli et al. [70] presented a 54-year-old female who was diagnosed with pSS and was started on corticosteroids. Despite treatment, she still experienced mood disturbances. As a result, she was referred to a psychiatric ward. There, she received a diagnosis of bipolar disorder (BD) and histrionic personality disorder. Treatment with mood stabilizers and prednisone resulted in significant improvement within four weeks. This resolved her mood instability and agitation. Another study reported a 43-year-old male presented with neuropsychiatric complaints, such as elevated mood, increased energy, and headaches. Examination indicated xerophthalmia and xerostomia, leading to a diagnosis of pSS. MRI evaluation showed periventricular white matter lesions on T2-weighted images. Additionally, a psychiatric evaluation diagnosed the patient with BD, which was effectively treated with carbamazepine and azathioprine [71]. It is important to note that while pSS-associated psychiatric symptoms may respond well to immunosuppressive therapy, caution is warranted when considering corticosteroids. This is because corticosteroids have the potential to trigger psychiatric symptoms such as manic episodes and psychosis [72].
The relationship between bipolar disorder and immune diseases is bidirectional, with prior research indicating a significant rise in the occurrence of immune diseases among individuals with BD [73]. However, the underlying pathophysiological mechanisms remain unclear. Patients with both BD and autoimmune diseases tend to have a lower average life expectancy, a higher risk of self-injury, increased readmission rates, and face more challenging treatment and management. This includes a higher risk of in-hospital death [70, 74]. Therefore, timely diagnosis and management of BD in pSS patients are crucial.
3.5. Anxiety and Depression
Depression manifests as persistent sadness, lack of interest or fatigue, low energy, and guilt. It can negatively affect individuals' function and decrease their quality of life [75]. It has been shown that depression is common among patients with chronic diseases, including cancer, cardiovascular disease, diabetes, arthritis, and autoimmune diseases [76]. Previous studies showed that the prevalence of depression among pSS patients could be as high as 32% to 46% [77–79]. According to a systematic review and meta-analysis by Wan et al., the prevalence of depression and anxiety is approximately threefold higher in dry eye disorder (DED) patients compared to healthy controls. The difference was irrespective of the etiology of DED. However, data showed that patients with DED due to pSS suffer from more severe depression and anxiety [80].
While the precise mechanisms underlying depression and anxiety in pSS patients remain unclear, there are several possible mechanisms that can be considered (Figure 1).
Figure 1.
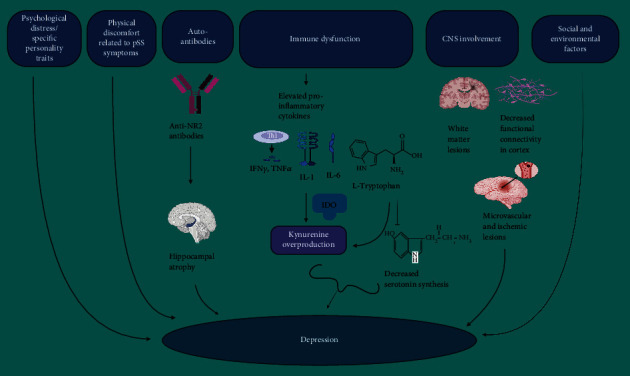
Possible mechanisms underlying depression in primary Sjögren's syndrome (pSS). (1) General psychological distress and specific personality traits like neuroticism, psychoticism, and obsessiveness might reduce stress tolerance and lead to depression. (2) Physical discomfort of pSS including fatigue, cognitive symptoms, sicca symptoms, and autonomic nervous system issues could contribute to depressive symptoms. (3) Elevated serum antibodies against N-methyl aspartate receptor (NMDAR) subtype NR2 found in pSS patients are associated with hippocampal atrophy, contributing to cognitive impairment and mood disorders in these patients. (4) Elevated levels of cytokines such as interferon-γ (IFN-γ), interleukin 1 (IL-1), interleukin 6 (IL-6), and tumor necrosis factor alpha (TNF-α) in pSS can trigger the overproduction of kynurenine and its metabolites from tryptophan through the indoleamine 2,3-dioxygenase (IDO) enzyme, diverting tryptophan away from serotonin production in the central nervous system (CNS). This imbalance in the hippocampus leads to depression, slower cognitive function, and other cognitive disorders. (5) Frequent central nervous system (CNS) white matter lesions (WML) were found in pSS, which can be linked to depression. Moreover, microstructural changes and decreased functional connectivity in the somatosensory cortex and corticospinal tract, as well as microvascular and ischemic lesions causing functional impairment in brain regions, have been observed in pSS. (6) Negative social and environmental factors, such as increased economic burden and lack of social support, may exacerbate depressive tendencies. Abbreviations: pSS: primary Sjögren's syndrome; TNF-α: tumor necrosis factor alpha; IL-6: interleukin 6; IL-1: interleukin 1; IFN- γ: interferon-γ; Anti-NR2: serum antibodies against N-methyl aspartate receptor (NMDAR) subtype NR2; IDO: indoleamine 2,3-dioxygenase enzyme; CNS: central nervous system. (This figure is created by Bioreneder.com).
General Psychological Distress and Personality Traits. Elevated levels of general psychological distress [81] and specific personality traits like neuroticism, psychoticism, and obsessiveness [82] might reduce stress tolerance.
Physical Discomfort of pSS. The physical discomfort associated with pSS, including fatigue, cognitive symptoms, sicca symptoms, and autonomic nervous system issues could contribute to depressive symptoms [83].
Social and Environmental Factors. Negative social and environmental factors, such as increased economic burden and lack of social support, may exacerbate depressive tendencies [76].
Common Immune Dysfunction/Inflammation. Shared immune dysfunction between pSS and depression has been observed. Inflammation has been identified as a mediating pathway for both the risk and neuroprogression of depression [84, 85]. Proinflammatory cytokines such as interleukin 1 (IL-1) and interleukin 6 (IL-6) have been implicated in both pSS and depression [40, 86, 87].
Neurotransmitters Dysregulations. Elevated levels of cytokines such as interferon-γ (IFN-γ), IL-1, and tumor necrosis factor alpha (TNF-α) in pSS can trigger the overproduction of kynurenine and its metabolites from tryptophan through indoleamine 2,3-dioxygenase (IDO) enzyme. This diverts tryptophan away from serotonin production in the CNS [25, 26]. This imbalance in the hippocampus leads to depression, slower cognitive function, and other cognitive disorders [88]. The affected cells, including microglia and astrocytes, experience reduced glutamate reuptake and increased glutamatergic signaling. This results in decreased serotonin production and can induce nociceptive and depressive behaviors. This cascade of effects can also perpetuate inflammation [26, 64, 89].
CNS Involvement. Frequent central nervous system white matter lesions (WML) were found in pSS [90] which can be linked to depression. Additionally, microstructural changes and decreased functional connectivity have been noted in the somatosensory cortex and corticospinal tract [91]. Furthermore, microvascular and ischemic lesions causing functional impairment in brain regions have been observed in pSS [27].
Anti-NR2 Antibodies and Hippocampal Atrophy. Elevated serum antibodies against N-methyl aspartate receptor (NMDAR) subtype NR2 were found in more pSS patients with depression than nondepressed individuals [20]. Positive anti-NR2 antibodies are also associated with hippocampal atrophy in pSS, contributing to cognitive impairment and mood symptoms [92].
In summary, the connection between pSS and depression involves various complex factors, including psychological, physiological, immunological, and neurostructural aspects.
Different studies have conflicting views on the impact of certain anti-inflammatory drugs, like IL-1 receptor antagonists and rituximab, on depressive disorder and mental fatigue in pSS patients [93, 94]. To address depression and mental fatigue, supplementing the medication regimen with antianxiety and antidepressant medications like paroxetine is recommended [76]. However, it is important to note that antidepressants and anxiolytics might induce dry eye disease due to their anticholinergic effects [95]. While both selective serotonin reuptake inhibitors (SSRIs) and serotonin and norepinephrine reuptake inhibitors (SNRIs) carry a risk of dry eye disease, SSRIs tend to result in lower Schirmer scores [96]. One nonmedication treatment modality in this group could be exercise therapy, such as a walking program. This approach can reduce muscle tension, depression, and fatigue by increasing endorphins and providing microelectrical stimulation in the nervous system [97]. Psychotherapy and behavioral interventions are also suggested to improve the patient's understanding of their disease and mitigate negative emotions [98]. Moreover, traditional Chinese medicine (TCM) has also shown promise in alleviating anxiety and depressive symptoms in pSS patients in recent studies [99, 100].
3.6. Cognitive Dysfunction
Cognitive dysfunction, often called “brain fog,” is one of the most common neuropsychiatric manifestations reported by pSS patients. It can affect attention, memory, and information processing speed [101]. The clinical presentations of cognitive dysfunction in pSS include difficulty concentrating, memory issues, slowed thinking or cognitive processing, mental fatigue, word-finding problems, and decreased multitasking abilities [101]. The prevalence of cognitive dysfunction in pSS patients varies between 44% and 100% [6, 13, 79, 102].
Previous studies have found that the majority of cognitive deficits in pSS may result from fronto-subcortical dysfunction. These deficits tend to remain relatively stable over time, with only rare instances of progression to dementia [103, 104]. Additionally, cognitive dysfunction has been associated with pain, depression, fatigue, and lower quality of life in pSS patients [78, 79].
The exact cause of cognitive deficits in pSS is not fully understood. However, a strong association has been found between cognitive deficits in pSS and the presence of anti-SSA (anti-Sjögren's syndrome-related antigen A) autoantibodies and magnetic resonance spectroscopy (MRS) alterations. This association persists regardless of an individual's age or gender. These studies found that despite the late onset of pSS, cognitive issues appear to be more common than age-related cognitive decline. This could potentially be due to subclinical inflammatory damage rather than structural microvascular damage, such as white matter lesions seen in cerebrovascular disease [79, 103]. Diagnostic and treatment challenges arise due to the varied neurological manifestations in pSS. However, previous studies emphasize on using MRI scans to identify underlying causes and assess the potential benefits of immunomodulatory treatments [79, 105].
4. Conclusions
In conclusion, pSS patients frequently exhibit various psychiatric manifestations including anxiety, sleep disorders, psychosis, catatonia, bipolar disorder, cognitive dysfunction, and obsessive-compulsive disorder. These psychiatric manifestations can be the primary presenting symptoms of pSS causing significant functional impairment and reduced quality of life. Therefore, timely diagnosis and management of these conditions are crucial.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Vivino F. B. Sjogren's syndrome: clinical aspects. Clinical Immunology . 2017;182:48–54. doi: 10.1016/j.clim.2017.04.005. [DOI] [PubMed] [Google Scholar]
- 2.Mavragani C. P., Moutsopoulos H. M. The geoepidemiology of Sjögren's syndrome. Autoimmunity Reviews . 2010;9(5):A305–A310. doi: 10.1016/j.autrev.2009.11.004. [DOI] [PubMed] [Google Scholar]
- 3.Haugen A. J., Peen E., Hultén B., et al. Estimation of the prevalence of primary Sjögren's syndrome in two age-different community-based populations using two sets of classification criteria: the Hordaland Health Study. Scandinavian Journal of Rheumatology . 2008;37(1):30–34. doi: 10.1080/03009740701678712. [DOI] [PubMed] [Google Scholar]
- 4.Gabriel S. E., Michaud K. Epidemiological studies in incidence, prevalence, mortality, and comorbidity of the rheumatic diseases. Arthritis Research & Therapy . 2009;11(3):p. 229. doi: 10.1186/ar2669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tobón G. J., Pers J. O., Devauchelle-Pensec V., Youinou P. Neurological disorders in primary Sjögren's syndrome. Autoimmune Diseases . 2012;2012:11. doi: 10.1155/2012/645967.645967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Harboe E., Tjensvoll A. B., Maroni S., et al. Neuropsychiatric syndromes in patients with systemic lupus erythematosus and primary Sjögren syndrome: a comparative population-based study. Annals of the Rheumatic Diseases . 2009;68(10):1541–1546. doi: 10.1136/ard.2008.098301. [DOI] [PubMed] [Google Scholar]
- 7.Soliotis F., Mavragani C., Moutsopoulos H. Central nervous system involvement in Sjogren's syndrome. Annals of the Rheumatic Diseases . 2004;63(6):616–620. doi: 10.1136/ard.2003.019497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Belin C., Moroni C., Caillat-Vigneron N., et al. Central nervous system involvement in Sjögren's syndrome: evidence from neuropsychological testing and HMPAO-SPECT. Annales de Medecine Interne . 1999;150(8):598–604. [PubMed] [Google Scholar]
- 9.Andonopoulos A. P., Lagos G., Drosos A. A., Moutsopoulos H. M. The spectrum of neurological involvement in Sjögren's syndrome. Rheumatology . 1990;29(1):21–24. doi: 10.1093/rheumatology/29.1.21. [DOI] [PubMed] [Google Scholar]
- 10.Tay S. H., Fairhurst A. M., Mak A. Clinical utility of circulating anti-N-methyl-d-aspartate receptor subunits NR2A/B antibody for the diagnosis of neuropsychiatric syndromes in systemic lupus erythematosus and Sjögren's syndrome: an updated meta-analysis. Autoimmunity Reviews . 2017;16(2):114–122. doi: 10.1016/j.autrev.2016.12.002. [DOI] [PubMed] [Google Scholar]
- 11.Alexander E. L. Neurologic disease in Sjogren’s syndrome: mononuclear inflammatory vasculopathy affecting central/peripheral nervous system and muscle: a clinical review and update of immunopathogenesis. Rheumatic Disease Clinics of North America . 1993;19(4):869–908. doi: 10.1016/S0889-857X(21)00211-8. [DOI] [PubMed] [Google Scholar]
- 12.Spezialetti R., Bluestein H. G., Peter J. B., Alexander E. L. Neuropsychiatric disease in Sjögren's syndrome: anti-ribosomal P and anti-neuronal antibodies. The American Journal of Medicine . 1993;95(2):153–160. doi: 10.1016/0002-9343(93)90255-N. [DOI] [PubMed] [Google Scholar]
- 13.Segal B. M., Pogatchnik B., Holker E., et al. Primary Sjogren’s syndrome: cognitive symptoms, mood, and cognitive performance. Acta Neurologica Scandinavica . 2012;125(4):272–278. doi: 10.1111/j.1600-0404.2011.01530.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen M., Jiang Q., Zhang L. The prevalence of bipolar disorder in autoimmune disease: a systematic review and meta-analysis. Annals of Palliative Medicine . 2021;10(1):350–361. doi: 10.21037/apm-20-2293. [DOI] [PubMed] [Google Scholar]
- 15.Inagaki T., Kudo K., Kurimoto N., Aoki T., Kuriyama K. A case of prolonged catatonia caused by Sjögren’s syndrome. Case Reports in Immunology . 2020;2020:4. doi: 10.1155/2020/8881503.8881503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sivakumar P. T., Jacob J. A., Modi U., George D. Primary Sjogren's syndrome presenting as acute psychosis with catatonic symptoms. Indian Journal of Psychiatry . 2022;64(9) Supplement 3:687–S688. doi: 10.4103/0019-5545.342028. [DOI] [Google Scholar]
- 17.Wong J. K., Nortley R., Andrews T., D'Cruz D. Psychiatric manifestations of primary Sjogren's syndrome: a case report and literature review. Case Reports . 2014;2014, article bcr2012008038 doi: 10.1136/bcr-2012-008038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chang Y. J., Tseng J. C., Leong P. Y., Wang Y. H., Wei J. C. C. Increased risk of Sjögren's syndrome in patients with obsessive-compulsive disorder: a nationwide population-based cohort study. International Journal of Environmental Research and Public Health . 2021;18(11):p. 5936. doi: 10.3390/ijerph18115936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Alunno A., Carubbi F., Bartoloni E., Cipriani P., Giacomelli R., Gerli R. The kaleidoscope of neurological manifestations in primary Sjögren’s syndrome. Clinical and Experimental Rheumatology . 2019;37(3) Supplement 118:192–198. [PubMed] [Google Scholar]
- 20.Lauvsnes M. B., Maroni S. S., Appenzeller S., et al. Memory dysfunction in primary Sjögren's syndrome is associated with anti-NR2 antibodies. Arthritis & Rheumatism . 2013;65(12):3209–3217. doi: 10.1002/art.38127. [DOI] [PubMed] [Google Scholar]
- 21.Estiasari R., Matsushita T., Masaki K., et al. Comparison of clinical, immunological and neuroimaging features between anti-aquaporin-4 antibody-positive and antibody-negative Sjögren’s syndrome patients with central nervous system manifestations. Multiple Sclerosis Journal . 2012;18(6):807–816. doi: 10.1177/1352458511431727. [DOI] [PubMed] [Google Scholar]
- 22.Kowal C., DeGiorgio L. A., Nakaoka T., et al. Cognition and immunity: antibody impairs memory. Immunity . 2004;21(2):179–188. doi: 10.1016/j.immuni.2004.07.011. [DOI] [PubMed] [Google Scholar]
- 23.Moll J., Markusse H. M., Pijnenburg J. J. J. M., Vecht C. J., Henzen-Logmans S. C. Antineuronal antibodies in patients with neurologic complicat'lons of primary Sjögren's syndrome. Neurology . 1993;43(12):2574–2581. doi: 10.1212/WNL.43.12.2574. [DOI] [PubMed] [Google Scholar]
- 24.Sanders M. E., Alexander E. L., Koski C. L., Frank M. M., Joiner K. A. Detection of activated terminal complement (C5b-9) in cerebrospinal fluid from patients with central nervous system involvement of primary Sjogren's syndrome or systemic lupus erythematosus. Journal of Immunology . 1987;138(7):2095–2099. doi: 10.4049/jimmunol.138.7.2095. [DOI] [PubMed] [Google Scholar]
- 25.Arnone D., Saraykar S., Salem H., Teixeira A. L., Dantzer R., Selvaraj S. Role of kynurenine pathway and its metabolites in mood disorders: a systematic review and meta-analysis of clinical studies. Neuroscience & Biobehavioral Reviews . 2018;92:477–485. doi: 10.1016/j.neubiorev.2018.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Müller N., Schwarz M. J. The immune-mediated alteration of serotonin and glutamate: towards an integrated view of depression. Molecular Psychiatry . 2007;12(11):988–1000. doi: 10.1038/sj.mp.4002006. [DOI] [PubMed] [Google Scholar]
- 27.Alexander E. L., Beall S. S., Gordon B., et al. Magnetic resonance imaging of cerebral lesions in patients with the Sjögren syndrome. Annals of Internal Medicine . 1988;108(6):815–823. doi: 10.7326/0003-4819-108-6-815. [DOI] [PubMed] [Google Scholar]
- 28.André F., Böckle B. C. Sjögren-Syndrom. JDDG: Journal der Deutschen Dermatologischen Gesellschaft . 2022;20(7):980–1003. doi: 10.1111/ddg.14823_g. [DOI] [PubMed] [Google Scholar]
- 29.Appenzeller S., Andrade S. O., Bombini M. F., Sepresse S. R., Reis F., França M. C., Jr. Neuropsychiatric manifestations in primary Sjogren syndrome. Expert Review of Clinical Immunology . 2022;18(10):1071–1081. doi: 10.1080/1744666X.2022.2117159. [DOI] [PubMed] [Google Scholar]
- 30.Govoni M., Padovan M., Rizzo N., Trotta F. CNS involvement in primary Sjögren’s syndrome. CNS Drugs . 2001;15(8):597–607. doi: 10.2165/00023210-200115080-00003. [DOI] [PubMed] [Google Scholar]
- 31.Massara A., Bonazza S., Castellino G., et al. Central nervous system involvement in Sjögren’s syndrome: unusual, but not unremarkable—clinical, serological characteristics and outcomes in a large cohort of Italian patients. Rheumatology . 2010;49(8):1540–1549. doi: 10.1093/rheumatology/keq111. [DOI] [PubMed] [Google Scholar]
- 32.Abad V. C., Sarinas P. S. A., Guilleminault C. Sleep and rheumatologic disorders. Sleep Medicine Reviews . 2008;12(3):211–228. doi: 10.1016/j.smrv.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 33.Tishler M., Barak Y., Paran D., Yaron M. Sleep disturbances, fibromyalgia and primary Sjögren's syndrome. Clinical and Experimental Rheumatology . 1997;15(1):71–74. [PubMed] [Google Scholar]
- 34.Hackett K. L., Gotts Z. M., Ellis J., et al. An investigation into the prevalence of sleep disturbances in primary Sjögren’s syndrome: a systematic review of the literature. Rheumatology . 2017;56(4):570–580. doi: 10.1093/rheumatology/kew443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Priori R., Minniti A., Antonazzo B., Fusconi M., Valesini G., Curcio G. Sleep quality in patients with primary Sjögren's syndrome. Clinical and Experimental Rheumatology . 2016;34(3):373–379. [PubMed] [Google Scholar]
- 36.Gudbjörnsson B., Broman J. E., Hetta J., Hällgren R. Sleep disturbances in patients with primary Sjögren's syndrome. Rheumatology . 1993;32(12):1072–1076. doi: 10.1093/rheumatology/32.12.1072. [DOI] [PubMed] [Google Scholar]
- 37.Segal B., Bowman S. J., Fox P. C., et al. Primary Sjögren's syndrome: health experiences and predictors of health quality among patients in the United States. Health and Quality of Life Outcomes . 2009;7(1):p. 46. doi: 10.1186/1477-7525-7-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Goodchild C., Treharne G. J., Booth D. A., Bowman S. J. Daytime patterning of fatigue and its associations with the previous night's discomfort and poor sleep among women with primary Sjögren's syndrome or rheumatoid arthritis. Musculoskeletal Care . 2010;8(2):107–117. doi: 10.1002/msc.174. [DOI] [PubMed] [Google Scholar]
- 39.Usmani Z. A., Hlavac M., Rischmueller M., et al. Sleep disordered breathing in patients with primary Sjögren’s syndrome: a group controlled study. Sleep Medicine . 2012;13(8):1066–1070. doi: 10.1016/j.sleep.2012.06.010. [DOI] [PubMed] [Google Scholar]
- 40.Mavragani C. P., Moutsopoulos H. M. Sjögren's syndrome. Annual Review of Pathology . 2014;9(1):273–285. doi: 10.1146/annurev-pathol-012513-104728. [DOI] [PubMed] [Google Scholar]
- 41.Mitchell M. D., Gehrman P., Perlis M., Umscheid C. A. Comparative effectiveness of cognitive behavioral therapy for insomnia: a systematic review. BMC Family Practice . 2012;13(1):p. 40. doi: 10.1186/1471-2296-13-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Alexander E. Central nervous system disease in Sjögren’s syndrome. Rheumatic Disease Clinics of North America . 1992;18(3):637–672. doi: 10.1016/S0889-857X(21)00321-5. [DOI] [PubMed] [Google Scholar]
- 43.Gelenberg A. The catatonic syndrome. The Lancet . 1976;307(7973):1339–1341. doi: 10.1016/S0140-6736(76)92669-6. [DOI] [PubMed] [Google Scholar]
- 44.Solmi M., Pigato G. G., Roiter B., et al. Prevalence of catatonia and its moderators in clinical samples: results from a meta-analysis and meta-regression analysis. Schizophrenia Bulletin . 2018;44(5):1133–1150. doi: 10.1093/schbul/sbx157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rogers J. P., Pollak T. A., Blackman G., David A. S. Catatonia and the immune system: a review. The Lancet Psychiatry . 2019;6(7):620–630. doi: 10.1016/S2215-0366(19)30190-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Beach S. R., Luccarelli J., Praschan N., Fusunyan M., Fricchione G. L. Molecular and immunological origins of catatonia. Schizophrenia Research . 2024;263:169–177. doi: 10.1016/j.schres.2023.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Diagnostic A. P. A. Statistical Manual of Mental Disorders . Washington, DC: American Psychiatric Association; 1994. [Google Scholar]
- 48.Moreno-Küstner B., Martín C., Pastor L. Prevalence of psychotic disorders and its association with methodological issues. A systematic review and meta-analyses. PLoS One . 2018;13(4, article e0195687) doi: 10.1371/journal.pone.0195687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cullen A. E., Holmes S., Pollak T. A., et al. Associations between non-neurological autoimmune disorders and psychosis: a meta-analysis. Biological Psychiatry . 2019;85(1):35–48. doi: 10.1016/j.biopsych.2018.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Eaton W. W., Pedersen M. G., Nielsen P. R., Mortensen P. B. Autoimmune diseases, bipolar disorder, and non-affective psychosis. Bipolar Disorders . 2010;12(6):638–646. doi: 10.1111/j.1399-5618.2010.00853.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Benros M. E., Nielsen P. R., Nordentoft M., Eaton W. W., Dalton S. O., Mortensen P. B. Autoimmune diseases and severe infections as risk factors for schizophrenia: a 30-year population-based register study. American Journal of Psychiatry . 2011;168(12):1303–1310. doi: 10.1176/appi.ajp.2011.11030516. [DOI] [PubMed] [Google Scholar]
- 52.Cox P. D., Hales R. E. CNS Sjögren's syndrome. The Journal of Neuropsychiatry and Clinical Neurosciences . 1999;11(2):241–247. doi: 10.1176/jnp.11.2.241. [DOI] [PubMed] [Google Scholar]
- 53.Raps A., Abramovich Y., Assael M., Green L. Relation between schizophrenic-like psychosis and Sjogren syndrome (SS) Israel Journal of Psychiatry and Related Sciences . 1986;23(4):321–324. [PubMed] [Google Scholar]
- 54.Rosado S. N., Silveira V., Reis A. I., Gordinho A., Noronha C. Catatonia and psychosis as manifestations of primary Sjögren's syndrome. European Journal of Case Reports in Internal Medicine . 2018;5(6, article 000855) doi: 10.12890/2018_000855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hammett E. K., Fernandez-Carbonell C., Crayne C., Boneparth A., Cron R. Q., Radhakrishna S. M. Adolescent Sjogren’s syndrome presenting as psychosis: a case series. Pediatric Rheumatology. . 2020;18(1):p. 15. doi: 10.1186/s12969-020-0412-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vivino F. B., Carsons S. E., Foulks G., et al. New treatment guidelines for Sjögren's disease. Rheumatic Disease Clinics . 2016;42(3):531–551. doi: 10.1016/j.rdc.2016.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Denko J. D. Problems in diagnosis and treatment of lupus psychosis. American Journal of Psychotherapy . 1977;31(1):125–137. doi: 10.1176/appi.psychotherapy.1977.31.1.125. [DOI] [PubMed] [Google Scholar]
- 58.Veale D., Roberts A. Obsessive-compulsive disorder. BMJ . 2014;348, article g2183 doi: 10.1136/bmj.g2183. [DOI] [PubMed] [Google Scholar]
- 59.Ong L. T. C., Galambos G., Brown D. A. Primary Sjogren's syndrome associated with treatment-resistant obsessive-compulsive disorder. Frontiers in Psychiatry . 2017;8:p. 124. doi: 10.3389/fpsyt.2017.00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ampélas J. F., Wattiaux M. J., Van Amerongen A. P. Psychiatric manifestations of lupus erythematosus systemic and Sjogren's syndrome. L'encephale . 2001;27(6):588–599. [PubMed] [Google Scholar]
- 61.Wang L.-Y., Chen S.-F., Chiang J.-H., Hsu C.-Y., Shen Y.-C. Systemic autoimmune diseases are associated with an increased risk of obsessive–compulsive disorder: a nationwide population-based cohort study. Social Psychiatry and Psychiatric Epidemiology . 2019;54(4):507–516. doi: 10.1007/s00127-018-1622-y. [DOI] [PubMed] [Google Scholar]
- 62.Karaiskos D., Mavragani C. P., Sinno M. H., et al. Psychopathological and personality features in primary Sjogren's syndrome--associations with autoantibodies to neuropeptides. Rheumatology . 2010;49(9):1762–1769. doi: 10.1093/rheumatology/keq158. [DOI] [PubMed] [Google Scholar]
- 63.De Carvalho J. F., Ribeiro F. M. Sjögren syndrome associated with obsessive-compulsive disorder. European Review for Medical and Pharmacological Sciences . 2020;24(22):11801–11803. doi: 10.26355/eurrev_202011_23836. [DOI] [PubMed] [Google Scholar]
- 64.Miller A. H., Maletic V., Raison C. L. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biological Psychiatry . 2009;65(9):732–741. doi: 10.1016/j.biopsych.2008.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rao N. P., Venkatasubramanian G., Ravi V., Kalmady S., Cherian A., Yc J. R. Plasma cytokine abnormalities in drug-naïve, comorbidity-free obsessive-compulsive disorder. Psychiatry Research . 2015;229(3):949–952. doi: 10.1016/j.psychres.2015.07.009. [DOI] [PubMed] [Google Scholar]
- 66.Lougee L., Perlmutter S. J., Nicolson R., Garvey M. A., Swedo S. E. Psychiatric disorders in first-degree relatives of children with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections (PANDAS) Journal of the American Academy of Child & Adolescent Psychiatry . 2000;39(9):1120–1126. doi: 10.1097/00004583-200009000-00011. [DOI] [PubMed] [Google Scholar]
- 67.Murphy T. K., Storch E. A., Turner A., Reid J. M., Tan J., Lewin A. B. Maternal history of autoimmune disease in children presenting with tics and/or obsessive-compulsive disorder. Journal of Neuroimmunology . 2010;229(1-2):243–247. doi: 10.1016/j.jneuroim.2010.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Clemente A. S., Diniz B. S., Nicolato R., et al. Bipolar disorder prevalence: a systematic review and meta-analysis of the literature. Brazilian Journal of Psychiatry . 2015;37(2):155–161. doi: 10.1590/1516-4446-2012-1693. [DOI] [PubMed] [Google Scholar]
- 69.Wang L. Y., Chiang J. H., Chen S. F., Shen Y. C. Systemic autoimmune diseases are associated with an increased risk of bipolar disorder: a nationwide population-based cohort study. Journal of Affective Disorders . 2018;227:31–37. doi: 10.1016/j.jad.2017.10.027. [DOI] [PubMed] [Google Scholar]
- 70.Chebli S., Zgueb Y., Ouali U., Taleb S., Nacef F. Bipolar disorder as comorbidity with Sjögren’s syndrome: what can we do? Case Reports in Psychiatry . 2020;2020:3. doi: 10.1155/2020/8899615.8899615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Khalayli N., Kudsi M. Sjögren's syndrome with bipolar disorder, case report. Annals of Medicine and Surgery . 2022;80, article 104243 doi: 10.1016/j.amsu.2022.104243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lu Y., Ann L., McCarron R. Steroid-induced psychiatric symptoms: what you need to know. Current Psychiatry . 2021;20(4):33–38. doi: 10.12788/cp.0104. [DOI] [Google Scholar]
- 73.Cremaschi L., Kardell M., Johansson V., et al. Prevalences of autoimmune diseases in schizophrenia, bipolar I and II disorder, and controls. Psychiatry Research . 2017;258:9–14. doi: 10.1016/j.psychres.2017.09.071. [DOI] [PubMed] [Google Scholar]
- 74.Dickerson F., Origoni A., Schroeder J., et al. Mortality in schizophrenia and bipolar disorder: clinical and serological predictors. Schizophrenia Research . 2016;170(1):177–183. doi: 10.1016/j.schres.2015.11.010. [DOI] [PubMed] [Google Scholar]
- 75.Malhi G. S., Mann J. J. Depression. The Lancet . 2018;392(10161):2299–2312. doi: 10.1016/S0140-6736(18)31948-2. [DOI] [PubMed] [Google Scholar]
- 76.Huang T., Li Y., Luo Y., Zhou Y., Zhao Y., Liu Y. Research progress on the pathogenesis and quality of life of patients with primary Sjögren's syndrome complicated by depression. Clinical and Experimental Rheumatology . 2022;40(3):647–654. doi: 10.55563/clinexprheumatol/nspi47. [DOI] [PubMed] [Google Scholar]
- 77.Cui Y., Xia L., li L., Zhao Q., Chen S., Gu Z. Anxiety and depression in primary Sjögren's syndrome: a cross-sectional study. BMC Psychiatry . 2018;18(1):p. 131. doi: 10.1186/s12888-018-1715-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Koçer B., Tezcan M. E., Batur H. Z., et al. Cognition, depression, fatigue, and quality of life in primary Sjögren's syndrome: correlations. Brain and Behavior . 2016;6(12, article e00586) doi: 10.1002/brb3.586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Morreale M., Marchione P., Giacomini P., et al. Neurological involvement in primary Sjögren syndrome: a focus on central nervous system. PLoS One . 2014;9(1, article e84605) doi: 10.1371/journal.pone.0084605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wan K. H., Chen L. J., Young A. L. Depression and anxiety in dry eye disease: a systematic review and meta-analysis. Eye . 2016;30(12):1558–1567. doi: 10.1038/eye.2016.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hyphantis T., Mantis D., Voulgari P. V., Tsifetaki N., Drosos A. A. The psychological defensive profile of primary Sjögren's syndrome patients and its relationship to health-related quality of life. Clinical and Experimental Rheumatology . 2011;29(3):485–493. [PubMed] [Google Scholar]
- 82.Milic V., Grujic M., Barisic J., et al. Personality, depression and anxiety in primary Sjogren’s syndrome–Association with sociodemographic factors and comorbidity. PLoS One . 2019;14(1, article e0210466) doi: 10.1371/journal.pone.0210466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gandía M., Morales-Espinoza E. M., Martín-González R. M., et al. Factors influencing dry mouth in patients with primary Sjögren syndrome: usefulness of the ESSPRI index. Oral Health and Dental Management . 2014;13(2):402–407. [PubMed] [Google Scholar]
- 84.Berk M., Williams L. J., Jacka F. N., et al. So depression is an inflammatory disease, but where does the inflammation come from? BMC Medicine . 2013;11(1):p. 200. doi: 10.1186/1741-7015-11-200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Dantzer R., O'Connor J. C., Freund G. G., Johnson R. W., Kelley K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nature Reviews Neuroscience . 2008;9(1):46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Gentile A., Fresegna D., Musella A., et al. Interaction between interleukin-1β and type-1 cannabinoid receptor is involved in anxiety-like behavior in experimental autoimmune encephalomyelitis. Journal of Neuroinflammation . 2016;13(1):p. 231. doi: 10.1186/s12974-016-0682-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tiosano S., Yavne Y., Watad A., et al. The impact of tocilizumab on anxiety and depression in patients with rheumatoid arthritis. European Journal of Clinical Investigation . 2020;50(9, article e13268) doi: 10.1111/eci.13268. [DOI] [PubMed] [Google Scholar]
- 88.Fasick V., Spengler R. N., Samankan S., Nader N. D., Ignatowski T. A. The hippocampus and TNF: common links between chronic pain and depression. Neuroscience & Biobehavioral Reviews . 2015;53:139–159. doi: 10.1016/j.neubiorev.2015.03.014. [DOI] [PubMed] [Google Scholar]
- 89.Levite M. Glutamate receptor antibodies in neurological diseases: anti-AMPA-GluR3 antibodies, anti-NMDA-NR1 antibodies, anti-NMDA-NR2A/B antibodies, anti-mGluR1 antibodies or anti-mGluR5 antibodies are present in subpopulations of patients with either: epilepsy, encephalitis, cerebellar ataxia, systemic lupus erythematosus (SLE) and neuropsychiatric SLE, Sjogren’s syndrome, schizophrenia, mania or stroke. These autoimmune anti-glutamate receptor antibodies can bind neurons in few brain regions, activate glutamate receptors, decrease glutamate receptor’s expression, impair glutamate-induced signaling and function, activate blood brain barrier endothelial cells, kill neurons, damage the brain, induce behavioral/psychiatric/cognitive abnormalities and ataxia in animal models, and can be removed or silenced in some patients by immunotherapy. Journal of Neural Transmission . 2014;121(8):1029–1075. doi: 10.1007/s00702-014-1193-3. [DOI] [PubMed] [Google Scholar]
- 90.Akasbi M., Berenguer J., Saiz A., et al. White matter abnormalities in primary Sjögren syndrome. QJM . 2012;105(5):433–443. doi: 10.1093/qjmed/hcr218. [DOI] [PubMed] [Google Scholar]
- 91.Andrianopoulou A., Zikou A. K., Astrakas L. G., et al. Functional connectivity and microstructural changes of the brain in primary Sjögren syndrome: the relationship with depression. Acta Radiologica . 2020;61(12):1684–1694. doi: 10.1177/0284185120909982. [DOI] [PubMed] [Google Scholar]
- 92.Lauvsnes M. B., Beyer M. K., Kvaløy J. T., et al. Association of hippocampal atrophy with cerebrospinal fluid antibodies against the NR2 subtype of the N-methyl-d-aspartate receptor in patients with systemic lupus erythematosus and patients with primary Sjögren's syndrome. Arthritis and Rheumatology . 2014;66(12):3387–3394. doi: 10.1002/art.38852. [DOI] [PubMed] [Google Scholar]
- 93.Norheim K. B., Harboe E., Gøransson L. G., Omdal R. Interleukin-1 inhibition and fatigue in primary Sjögren's syndrome--a double blind, randomised clinical trial. PLoS One . 2012;7(1, article e30123) doi: 10.1371/journal.pone.0030123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mekinian A., Ravaud P., Larroche C., et al. Rituximab in central nervous system manifestations of patients with primary Sjögren's syndrome: results from the AIR registry. Clinical and Experimental Rheumatology . 2012;30(2):208–212. [PubMed] [Google Scholar]
- 95.Wong J., Lan W., Ong L. M., Tong L. Non-hormonal systemic medications and dry eye. The Ocular Surface . 2011;9(4):212–226. doi: 10.1016/S1542-0124(11)70034-9. [DOI] [PubMed] [Google Scholar]
- 96.Koçer E., Koçer A., Özsütçü M., Dursun A. E., Krpnar İ. Dry eye related to commonly used new antidepressants. Journal of Clinical Psychopharmacology . 2015;35(4):411–413. doi: 10.1097/JCP.0000000000000356. [DOI] [PubMed] [Google Scholar]
- 97.Strömbeck B. E., Theander E., Jacobsson L. T. H. Effects of exercise on aerobic capacity and fatigue in women with primary Sjogren's syndrome. Rheumatology . 2007;46(5):868–871. doi: 10.1093/rheumatology/kem004. [DOI] [PubMed] [Google Scholar]
- 98.Kotsis K., Voulgari P. V., Tsifetaki N., Drosos A. A., Carvalho A. F., Hyphantis T. Illness perceptions and psychological distress associated with physical health-related quality of life in primary Sjögren's syndrome compared to systemic lupus erythematosus and rheumatoid arthritis. Rheumatology International . 2014;34(12):1671–1681. doi: 10.1007/s00296-014-3008-0. [DOI] [PubMed] [Google Scholar]
- 99.Wu G. L., Li T. Y., Fan Y. S., Yu G. Y. Therapeutic effect of Chinese herbal medicine for strengthening qi, nourishing yin, and removing stasis on serum osteopontin and quality of life of patients with primary Sjogren's syndrome. Chinese Journal of Integrative Medicine . 2011;17(9):710–714. doi: 10.1007/s11655-011-0850-8. [DOI] [PubMed] [Google Scholar]
- 100.Chen H. H., Lai J. N., Yu M. C., et al. Traditional Chinese medicine in patients with primary Sjogren's syndrome: a randomized, double-blind, placebo-controlled clinical trial. Frontiers in Medicine . 2021;8, article 744194 doi: 10.3389/fmed.2021.744194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Riega-Torres J. C. L., Treviño-Castro M. A., Hernandez-Galarza I. D. J., et al. Cognitive dysfunction in Sjögren’s syndrome using the Montreal cognitive assessment questionnaire and the automated neuropsychological assessment metrics: a cross-sectional study. International Journal of Rheumatic Diseases . 2020;23(8):1019–1023. doi: 10.1111/1756-185X.13889. [DOI] [PubMed] [Google Scholar]
- 102.Indart S., Hugon J., Guillausseau P. J., et al. Impact of pain on cognitive functions in primary Sjögren syndrome with small fiber neuropathy: 10 cases and a literature review. Medicine . 2017;96(16, article e6384) doi: 10.1097/MD.0000000000006384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Martínez S., Cáceres C., Mataró M., Escudero D., Latorre P., Dávalos A. Is there progressive cognitive dysfunction in Sjögren syndrome? A preliminary study. Acta Neurologica Scandinavica . 2010;122(3):182–188. doi: 10.1111/j.1600-0404.2009.01293.x. [DOI] [PubMed] [Google Scholar]
- 104.Moreira I., Teixeira F., Martins Silva A., Vasconcelos C., Farinha F., Santos E. Frequent involvement of central nervous system in primary Sjögren syndrome. Rheumatology International . 2015;35(2):289–294. doi: 10.1007/s00296-014-3097-9. [DOI] [PubMed] [Google Scholar]
- 105.Blanc F., Longato N., Jung B., et al. Cognitive dysfunction and dementia in primary Sjögren’s syndrome. International Scholarly Research Notices . 2013;2013:7. doi: 10.1155/2013/501327.501327 [DOI] [PMC free article] [PubMed] [Google Scholar]


