Abstract
Beta cell connectivity describes the phenomenon whereby the islet context improves insulin secretion by providing a three-dimensional platform for intercellular signaling processes. Thus, the precise flow of information through homotypically interconnected beta cells leads to the large-scale organization of hormone release activities, influencing cell responses to glucose and other secretagogues. Although a phenomenon whose importance has arguably been underappreciated in islet biology until recently, a growing number of studies suggest that such cell–cell communication is a fundamental property of this micro-organ. Hence, connectivity may plausibly be targeted by both environmental and genetic factors in type 2 diabetes mellitus (T2DM) to perturb normal beta cell function and insulin release. Here, we review the mechanisms that contribute to beta cell connectivity, discuss how these may fail during T2DM, and examine approaches to restore insulin secretion by boosting cell communication.
Keywords: Mouse, Human, Signaling, Insulin, Diabetes, Imaging, Network
Introduction
Type 2 diabetes mellitus (T2DM) is a global epidemic that currently consumes ~10 % of the healthcare budget in the developed world [1]. This syndrome has a complex etiology but can be summarized as a failure of the beta cell mass to adequately compensate for insulin resistance, or alternatively a primary beta cell defect that leads to insulin resistance. The resulting glucose intolerance, coupled with dyslipidemia, drives a range of costly secondary complications including retinopathy, vasculopathy, renal failure, cancer, and cardiovascular disease [2, 3]. Consequently, elucidation of the mechanisms underlying the control of insulin secretion from individual beta cells has been the focus of intense research efforts. Thus, in response to an elevation of blood glucose, equilibration of the sugar across the plasma membrane occurs rapidly and is achieved via either the low affinity glucose transporter Glut2/slc2a2 (rodents) or the higher affinity transporter Glut1/slc2a1 (man) [4]. The low affinity hexokinase, glucokinase is then chiefly responsible for determining glycolytic flux toward pyruvate [5]. Conversion of the latter to acetyl-CoA in the mitochondrial matrix, and its oxidation via the tricarboxylate cycle, then ensues [6, 7]. The resultant increases in the ratio of free adenosine triphosphate (ATP) to adenosine diphosphate (ADP) (ATP:ADP) in the cytosol [8] and subplasma membrane domain [9] then leads to closure of ATP-sensitive K+ channels (KATP), membrane depolarization and the influx of calcium (Ca2+) through voltage-dependent Ca2+-channels (VDCC) [6, 7, 10, 11]. Together with the activation of a less well-defined “amplifying” pathway [12, 13], localized increases in the intracellular free Ca2+ concentration [14], including at the surface of the secretory granule [15], then provoke insulin release through interactions with the exocytotic machinery [16, 17].
By comparison, the population-level regulation of insulin release is less well understood, although the idea that it may contribute to T2DM risk has been suggested [18–22]. Providing evidence that cell–cell interactions are a prerequisite for proper hormone secretion is the observation that beta cells incommunicado (i.e., as isolated cells) release less insulin per capita than their properly connected counterparts within the intact islet [19, 23, 24]. Indeed, a feature of the endocrine pancreas is the three-dimensional encapsulation of beta, and other cell types, into islets of Langerhans, a biological scaffold for cell–cell communications. Since these micro-organs are conserved throughout the mammalian kingdom and beyond [25], albeit with important differences in the numbers of each cell type and their arrangement within the islet (see below), the intraislet mechanisms governing insulin secretion may represent an underappreciated target through which T2DM insults provoke hyperglycemia. Building upon recent findings from our own [26–28] and others’ [20–22, 29–31] laboratories, the aim of the present review is to describe our current understanding as to how beta cell–beta cell communication (hereafter referred to as “connectivity”) contributes to the normal regulation of insulin secretion in healthy subjects. We also discuss how changes in this property may contribute to T2DM risk in genetically susceptible individuals.
Islets as discrete secretory units
The term “endocrine pancreas” describes the thousands (millions in man) of islets of Langerhans scattered throughout the exocrine tissue. Each islet can range in size from 20 to 400 µM and comprises alpha- (glucagon), beta- (insulin), delta- (somatostatin), epsilon- (ghrelin), and pancreatic polypeptide (PP) cells. Strikingly, islets are evolutionarily stable structures and are present in most mammals studied to date, including the Beluga whale, with a similar range of sizes reported in each species [25]. With the exception of bats, horses, hyenas, primates, and humans, the arrangement of endocrine cells within islets is similar [25]. Thus, in rodent islets, the most-studied model, beta cells form a central core, with alpha cells occupying the mantle [25, 32, 33]. Suggesting that this may be a consequence of the vasculature, blood flow has been shown to follow an inner–outer flow pattern, irrigating beta before alpha cells in this species [34], and the vasculature appears to be instructive for pancreas development [35]. By contrast, beta cells in human islets are interspersed with alpha cells, in part the consequence of the tertiary folding of an initial trilaminar alpha–beta–alpha sheet, which promotes heterologous contacts [33, 36–38]. As well as differences in islet architecture, alterations to cell proportion are also apparent between species. For example, the ratio of beta:alpha cells in rodent islets is ~4:1, whereas in humans it is ~1.25:1. Such divergence in islet architecture likely influences cell–cell communication by altering the extent and nature of cell–cell signaling processes, and may be an important source of species differences in islet function. Regardless, the islet structure is permissive for insulin secretion, and beta cells in two dimensions display blunted responses to input, both in terms of Ca2+ signaling and magnitude hormone release [27, 39–41].
High-speed imaging of beta cell connectivity
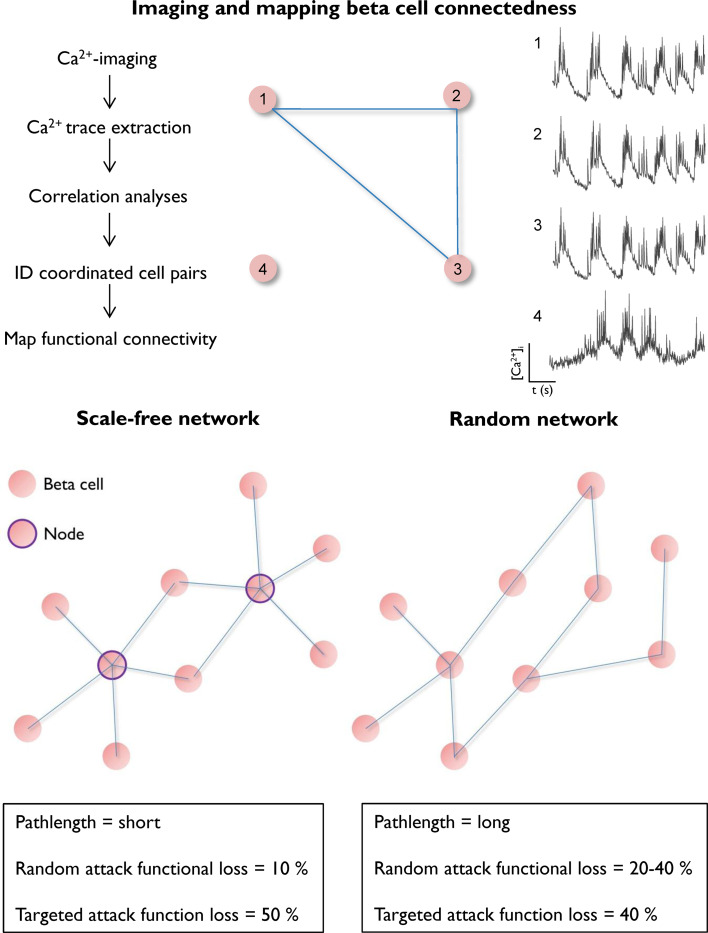
Over the last decade, advances in microscopy have allowed cell dynamics to be monitored in situ within the intact tissue setting [42]. Key to this is the use of high-speed imaging, which when combined with highly sensitive detectors, allows a large area to be rapidly traversed at cellular resolution. In terms of endocrine organ function, the physiologically relevant output is hormone release. However, large-scale imaging of exocytosis in individual cells is only just becoming possible, although the currently available dyes possess signal-to-noise ratios incompatible with high-speed acquisition at visible light wavelengths [43–46]. To circumvent these issues, membrane voltage or intracellular Ca2+ concentrations can instead be used as a proxy for Ca2+-dependent hormone release [47–50]. To this end, functional multicellular Ca2+ imaging (fMCI), originally used to map activity in cortical circuits [51–53], has recently been adapted for use in beta cells [27, 28]. By coupling a laser bank to a Nipkow spinning disk, the millisecond organization of beta cell population Ca2+-spiking activity can be captured in near real time with reduced phototoxicity and photobleaching. Following acquisition, the 1datasets are subjected to nondeterministic Monte Carlo-based models to identify the cells with similar behavioral profiles, i.e., those with correlated activity and which are assumed to contribute to the same secretory process [42, 54]. Statistical significance is determined by shuffling the experimental dataset and calculating the likelihood of detecting the same correlation pattern due to chance. A functional connectivity map can then be constructed based on the location of significantly correlated cell pairs, allowing perturbations to beta cell connectivity to be evaluated (see Fig. 1, top panel, for an example). In a refinement of this method, beta cell metabolic interconnectivity has recently been mapped in intact islets by monitoring intracellular free ATP:ADP dynamics, as for Ca2+ [55]. When using these techniques, it is important to note that the territories of communicating beta cells within intact islets are larger than those that can be recorded, limiting the physiological inferences that can be drawn.
Fig. 1.
Imaging and mapping beta cell network topology. (Above) Functional multicellular Ca2+ imaging is used to monitor the large-scale organization of glucose-induced population dynamics (above, left). By subjecting the resulting traces (from ~ 50–100 individual cells per islet) to correlation analyses, cells with coordinated activity can be identified and a functional connectivity map plotted based upon position within the imaged field (x–y) (above, right). Scale-free connection distributions are typified by a minority of cells that host the majority of connections (nodes), while maintaining streamlined information flow due to a short pathlength. Although robust in the face of random attack, they are prone to collapse following a targeted attack (below, left). By contrast, nonscale-free networks (e.g., random or lattice) may not efficiently propagate signals due to a long pathlength, and random attacks significantly reduce capacity (below, right)
Islet wiring patterns
Network science principally relies on the use of graph theory to identify the interactions that govern behavior in complex systems (see [42] for a review of network science in Endocrinology). Using these approaches, it has become increasingly clear that network topology tends to be conserved (e.g., scale-free and random) irrespective of the components examined (e.g., cells vs. people) [42, 56, 57]. Recent research has shown that graph theory is also applicable to the description of complex dynamics in the endocrine pancreas. Thus, analysis reveals that beta cells comprise glucose-responsive scale-free networks in which cells can communicate over long distances, through presently undefined mechanisms [29]. Such network topologies are defined by a power-law distributed link probability in which a minority of cells (termed highly connected nodes) host the majority of connections and are said to possess small-world properties if there is a tendency toward formation of cliques (6° of separation concept) (Fig. 1, bottom panel). Price was the first to describe scale-free networks, noting that journal citations follow a power-law distribution, sharing features in keeping with Pareto’s law (the ‘rich-get-richer’ hypothesis) [58]. Subsequently, Barabasi and Albert showed that preferential attachment is responsible for the emergence of scale-free properties [59]. Notably, scale-free distributions are ubiquitous and have been described in social networks, computer networks, neural networks, and anterior pituitary networks [54, 60–63]. An important feature of scale-free networks is robustness at low wiring cost: the chances of a random attack disabling communication are low and the use of hubs to route information reduces signal transmission length [42]. However, should the highly connected nodes be specifically targeted, the network is vulnerable to collapse, since a high proportion of links will be lost (Fig. 1, bottom panel). Therefore, an interesting but untested possibility is that highly connected beta cell nodes may represent a subpopulation which is particularly susceptible to T2DM insults. Conversely, these highly connected nodes may serve as a functional reserve to maintain islet function in the face of gross perturbation by allowing the redistribution of information, again, a hypothesis that requires experimental validation.
Mechanisms underlying beta cell–beta cell connectivity
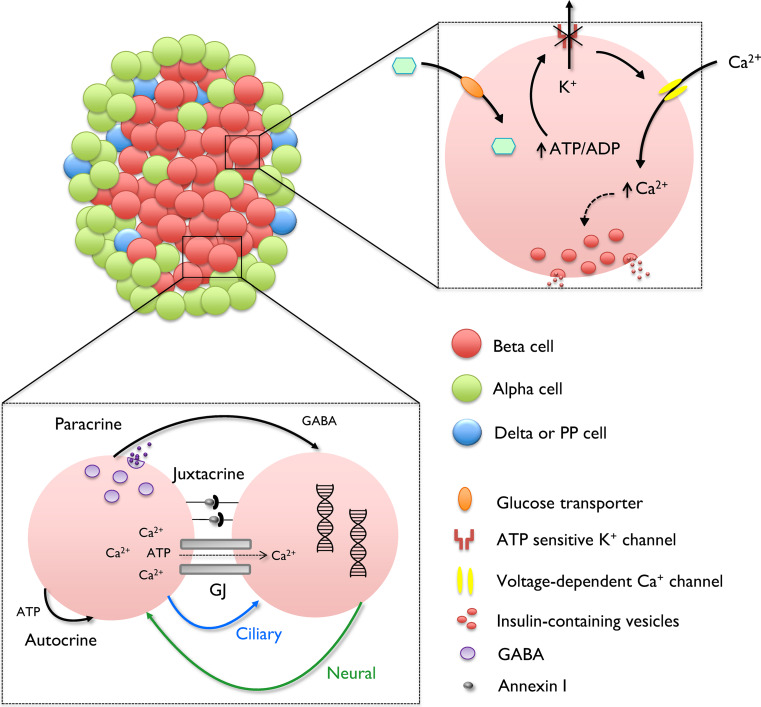
Neural circuits have a clear basis for long-range connectivity, since neurons send out axonal projections that can form synapses located millimeters apart. By contrast, it is less easy to conceptualize how beta cells within the islet can communicate over long distances to organize their activities. Might this involve, for example, “physical connections” (e.g., through islet interneurons) between remote cells, or alternatively linearly connected “trains” of beta or other cells along which signals are transmitted to a distant cell(s) from a controller (“pacemaker”) at a coordination hub? In any case, the islet possesses a formidable signaling toolbox (see Fig. 2). This is reviewed in depth elsewhere [20, 21, 28, 64], so here we limit our discussion to the pathways which may conceivably underlie connectivity between beta cells.
Fig. 2.
Schematic showing single cell and population-level beta cell signaling. At the molecular level, glucose is transported into the beta cell before undergoing glycolysis to increase the ratio of free cytosolic ATP:ADP. This closes KATP channels, leading to opening of VDCC, Ca2+ influx, and Ca2+-dependent exocytosis. At the population-level, beta cell dynamics are further dictated by signaling circuits involving paracrine, juxtacrine, autocrine, electrotonic (GJ), neural and ciliary communications
Gap junctions
The best characterized cell–cell coupling mechanism in the pancreas is provided by gap junctions (GJs). Beta cells within rodent and human islets are homotypically connected by connexin 36 (Cx36 or GJD2) [65, 66]. GJs comprising Cx36 are charge- and size-selective channels that allow the intercellular passage of ions (e.g., Ca2+, Na+, and Zn2+) and nucleotides (e.g., ATP) [19, 20, 67]. Providing evidence that Cx36 is critical for coordinating islet activity are the observations that dispersed beta cells fail to synchronize their responses to glucose, and islets lacking Cx36 display more stochastic activity patterns due to increases in beta cell functional heterogeneity [31, 68–70]. GJ linkages are essential for the regulation of normal hormone release, since mice deleted for Cx36 are glucose intolerant and display impaired pulsatility, as well as elevated basal insulin secretion [22, 68, 71]. It is unclear how GJs could account for the long-range functional connections that project between distant cells, as practically all beta cells express Cx36 protein, meaning that communication should encompass even close neighbors [65, 72]. However, heterogeneity exists in fluorescence recovery after photobleaching (FRAP) within islets [73], suggesting that connectivity patterns between individual beta cells may at least reflect differences in functional GJ coupling. As proposed above, this may lead to the formation of linear groups of cells, tightly interconnected in three dimensions between one another, but (relatively) isolated from neighboring cells outside the train, thus forming a conduit for the passage of ionic (Ca2+) or other (e.g., paracrine, see below) signals.
Neural
Islets receive rich innervation from the autonomic nervous system, and neural regulation of insulin secretion is critical for normal glucose homeostasis in vivo. The existence of a physical network of neurons to couple remote beta cells within the islet thus provides a conceptually straightforward model to explain recent experimental observations [26, 27, 29]. Indeed, insulin release is strongly stimulated by postganglionic cholinergic fibers that signal via acetylcholine (ACh)-mediated activation of muscarinic receptors to phase set and synchronize beta cell activity within and, potentially, between islets [74–76]. Such activation underpins the cephalic phase of insulin secretion in anticipation of food [77]. In addition, other neuropeptides including pituitary adenylate cyclase activating peptide (PACAP) and vasoactive intestinal peptide (VIP) may contribute to the parasympathetic control of beta cell function [74, 78]. By contrast, insulin release is suppressed by noradrenergic sympathetic neurons that signal via α2-adrenoreceptors to open KATP channels [74, 79, 80], although a stimulatory effect of noradrenaline has also been observed, probably through effects on cyclic adenosine monophosphate (cAMP) accumulation and β-adrenoreceptor activation [81, 82]. Marked differences exist in the neural regulation of insulin secretion between rodents and man. Thus, human islets are relatively devoid of parasympathetic nerve fibers [83], and glucose-sensitization of beta cell activity instead relies upon ACh release from vesicular acetylcholine transporter-expressing alpha cells [84, 85]. This lack of direct innervation may partly explain why beta cell glucose responses in human islets are largely stochastic, with synchrony detected only between small cell clusters [27, 33, 86]. Conversely, the assessment of whether neurons contribute to long-range connectivity in mouse islets firstly requires confirmation of cholinergic fiber survival in isolated islets, followed by their specific manipulation (e.g., using patch clamp).
Primary cilia
Cilia can be regarded as cell extensions that act as signaling hubs due to expression of G protein-coupled receptors (GPCRs), ion channels and transcription factors [87]. Primary cilia are immotile and are formed from a ring of nine microtubule doublets wrapped in a membrane sheath [88]. While studies of Kif3a, Lkb1, and Rfx3 knockout mice have all invoked a role for cilia in pancreatic development (i.e., ductal and endocrine cell specification) [87, 89–91], little is known about their involvement in cell–cell signaling processes within the islet. Given the role of cilia in signal transmission in in other tissues [92], and potentially in exosome-mediated intercellular communications [93], we believe this warrants further investigation.
Paracrine signaling
Intercellular communication may also be possible via the production and secretion of messengers which act on neighboring cells [20, 21, 28]. Over 230 secreted factors have been identified in rodent islets [94], and a number of signaling loops with roles in the regulation of beta cell function and insulin release are now well characterized (see references [21, 28, 64]). Despite this, it is unclear how paracrine factors could contribute to the complex functional islet wiring patterns described using graph theory [29, 30], since all beta cells within the molecule diffusion path would be expected to be affected. Although it is plausible that active transport mechanisms and cognate receptor expression levels/patterns may allow more precise communication between beta cells, this needs further study.
Despite the plethora of signaling mechanisms available within the islet, we suggest that a combination of modalities is required for producing the complex activity patterns that underlie beta cell–beta cell communication and connectivity. Notably, differences in signaling input, together with alterations to islet architecture, may play an important role in determining species-specific responses to secretagogues such as glucose and incretins.
Glucose and GLP-1-regulated connectivity: metabolic signals
It is generally acknowledged that metabolic activity within individual beta cells is oscillatory, and that this generates the membrane bursting activity required for Ca2+ influx and exocytosis [95]. Whether metabolic oscillations are driven by Ca2+ oscillations, or vice versa, is still the source of debate [95, 96], but the islet context seems to be critical, since dispersed beta cells display reduced periodicity in mitochondrial potential [97]. Moreover, total internal reflection fluorescence (TIRF) microscopy of mouse islets has shown that near-membrane glucose-induced oscillations in ATP:ADP are coordinated between small beta cell clusters [98], confirming earlier observations that employed lower resolution autofluorescence imaging of NAD(P)H [99–101]. The mechanisms underlying the synchronous propagation of energy status between beta cells remain unknown, but may reflect Ca2+ feedback and intrinsic metabolic behavior [96], or alternatively, metabolic coupling via GJs [102, 103].
In addition to glucose, secretory potentiators, including members of the incretin family, are able to influence beta cell energetics. The incretin, glucagon-like peptide-1 (GLP-1), is released from the gut in response to bile transit and glucose-dependently augments insulin secretion [104–106]. While its effects on cAMP-Epac2, MAPK, and beta-arrestin signaling pathways are well characterized [107–109], little is known about whether GLP-1 alters the beta cell metabolic set point to influence ATP:ADP. Whereas luciferase-based studies by us have demonstrated a role for GLP-1 in mitochondrial ATP synthesis in clonal MIN6 beta cells [110], others have observed no effect of the incretin in rodent islets using biochemical detection methods [111]. Since ATP dynamics and/or cell heterogeneity may mask actions of incretin on metabolism, the effects of GLP-1 on intracellular free ATP:ADP were monitored with cellular resolution by expressing the recombinant probe Perceval throughout the first few layers of rodent and human islets [8, 55, 112]. Using these methods, we found that GLP-1 engages a metabolically coupled subnetwork of beta cells to amplify insulin secretion, an action that is dependent upon Ca2+ influx and elevations in cAMP [55]. Of note, in these studies, beta cells within mouse islets responded coordinately to GLP-1 with synchronous ATP:ADP oscillations, whereas human islets exhibited more random dynamics. Thus, the regulation of beta cell–beta cell metabolic connectivity may potentially contribute to the disparate actions of incretin in rodents and man, although confirmation of this will require simultaneous measures of Ca2+ and ATP:ADP in islets from both species.
Glucose- and GLP-1-regulated connectivity: Ca2+ signals
Ca2+-imaging of pancreatic islet slices has revealed that glucose likely drives large-scale increases in population synchrony by coaxing activity in a scale-free and small-world network of beta cells [29, 30, 49]. Notably, propagation of Ca2+ waves via GJs is hypothesized to underlie islet dynamics in response to glucose, since the length of individual correlated links depends on Euclidean distance, although long-range communications are still evident [29]. Confirming these findings, we have recently shown that the rapid (ms) oscillations in electrical activity are similarly dictated by scale-free and small-world beta cell wiring patterns [113]. Thus, under conditions of high glucose, beta cells work together as defined subpopulations to orchestrate and drive insulin release from the islet.
As well as glucose, insulin secretion is also reliant upon the amplifying or potentiating actions of incretins. Indeed, in humans, almost 70 % of the insulin-raising effects of oral glucose challenge can be attributed to the incretin effect [114]. Notably, the insulinotropic activity of exogenously administered glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 is diminished in T2DM [115, 116], suggesting that altered beta cell incretin responsiveness may contribute to the disease state, although causality is not well defined [117]. Since the single biggest T2DM risk factor remains obesity, and high body mass index (BMI) individuals present with reduced GLP-1-stimulated insulin secretion [118, 119], excess lipid may target incretin action to impair beta cell function. To investigate this, we subjected human islets to fMCI to map population dynamics, and found that both GIP and GLP-1 recruit a highly coordinated subnetwork of GJ-coupled beta cells to augment insulin secretion [27, 28]. This process of incretin-regulated beta cell connectivity may be a target for the insulin-lowering effects of free fatty acid (FFA), since it could be disrupted in a GJ-dependent manner following exposure to a lipotoxic milieu, and was inversely correlated with donor BMI [27]. Mechanistically, this may involve FFA-induced overexpression of inducible cAMP early repressor gamma (ICER-γ), a protein that binds a cAMP-response element in the Cx36 promoter [120, 121]. By contrast, a similar effect of incretin on beta cell interactivity was not present in mouse islets, but could be revealed by placing mice on a high fat diet (HFD) to disrupt normal glucose responses [27, 28]. We, therefore, speculate that such divergent regulation of the incretin axis, potentially stemming from structural and functional differences in islet architecture, may represent a novel target for pro-diabetogenic insults in man.
Genes and connectivity
Type 2 diabetes has a strong hereditary component [122–124]. Consequently, genome-wide association studies (GWAS) have identified a number of gene variants linked with an increased odds ratio (OR) of developing elevated fasting glucose and T2DM. Although the effects of these variants are usually quite small, their very existence indicates that genes in the associated loci are highly likely to play a role in disease etiology [125, 126]. While gene variants and glucose homeostasis are well studied in man, relatively less is known about their precise mechanisms of action at the islet level [125], and in particular upon beta cell connectivity. Several dozen (>90) risk-associated polymorphisms have been identified to date, and those with the strongest OR for development of T2DM, or with known effects on beta cell–cell communication, are discussed below (see Fig. 3).
Fig. 3.
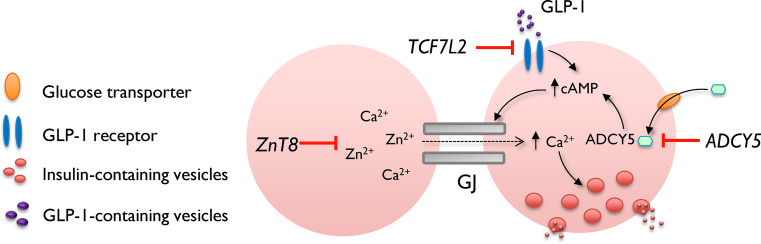
Potential mechanisms by which T2D-associated genes may alter beta cell connectivity. ZnT8 gene variants disrupt cytosolic Ca2+ and Zn2+ handling, and both of these ions are required for normal GJ activity. ADCY5 gene variants decrease glucose-stimulated cAMP rises, a second messenger shown to increase GJ communications between beta cells. By contrast, TCF7L2 gene variants may disrupt normal GJ function through effects upon glucose-stimulated Ca2+ increases, as well as GLP-1-stimulated cAMP generation
TCF7L2
TCF7L2 is a member of the canonical Wnt-signaling pathway and a transcriptional partner for beta-catenin. Individuals who possess a single nucleotide polymorphism (SNP), rs7903146, in intron 3 of the TCFL72 gene on chromosome 10, have an increased risk of developing T2DM, with an OR of 1.45 for the T allele [127–130]. This is believed largely to be due to defects in insulin secretion (insulin sensitivity is slightly impaired in T allele carriers), as well as a markedly (~50 %) attenuated incretin effect [127, 131, 132] (though see [125] for a discussion of a role for hepatic glucose handling). Although the subject of debate, these results have subsequently been confirmed in conditional rodent models and human islets. Thus, TCF7L2 silencing leads to impaired insulin secretion from isolated mouse and human islets [133, 134], and deletion of Tcf7l2 throughout the pancreas or selectively in the beta cell causes glucose intolerance [135, 136], particularly after oral glucose administration, with the observed effects increasing with age or exposure to an HFD. Of note, a further study failed to detect any effects on glycemia of deleting Tcf7l2 in the adult beta cell, although this report was restricted to examination of intraperitoneal glucose tolerance in young (<12 weeks) animals [137]. GLP-1-stimulated insulin secretion is strongly inhibited by Tcf7l2 elimination in vitro [134, 135], the latter due largely to reduced GLP-1R expression and defects in the exocytotic apparatus [133, 135, 138, 139]. Interestingly, when investigated in dissociated islets, TF7L2 knockdown leads to a slight potentiation of glucose-induced Ca2+ increases [133, 140], although only single (or clusters) of beta cells were studied, precluding analysis of synchrony or coordination. By contrast, ablation of the Tcf7l2 gene selectively in the beta cell through Ins1Cre-directed recombination of flox’d alleles impairs these increases when assessed in the intact islet setting [136]. The reasons for these differences remain obscure but suggest that either silencing in nonbeta cells in the former case, or altered beta cell–beta cell interactions in the latter, are at play. Of note, Tcf7l2 silencing in INS1 beta cells lowers the expression of Ca2+ channel subunits [141], suggesting that TCF7L2 may exert control, either directly or indirectly, over the Ca2+-signaling machinery. Of relevance, when studied in islets from mice maintained on an HFD, glucose-stimulated beta cell connectivity in Tcf7l2 null animals was significantly reduced versus that of control animals [136] (manuscript submitted). Of note, this alteration was not associated with any changes in GJ mRNA expression, though may conceivably involve changes in Cx36 protein abundance.
ADCY5
ADCY5 gene products encode isoform V of the adenylate cylase family, a type III Ca2+-inhibited enzyme tasked with generation of cAMP [142, 143], a second messenger involved in glucoregulation as part of the “amplifying” pathway [144]. Whereas other isoforms predominate in the rodent islet, ADCY5 is among the most abundant members of this family in human beta cells [26, 145]. The T2DM-associated SNP rs11708067 on chromosome 3 lies within intron 3 of the ADCY5 gene and is associated with increased fasting glucose and 2-h glucose, but not oral glucose responses [146], with an OR of 1.23 for the major A-allele [147]. Using lentiviral shRNA approaches to silence gene and protein expression in human islets, we have recently shown that ADCY5 is required for the coupling of glucose but not incretin to insulin secretion [148]. Although the former is partly due to impaired insulin processing (i.e., proinsulin → insulin conversion) [149], islets depleted for ADCY5 also displayed impaired glucose- but not GLP-1-induced increases in cAMP, and consequent impairments in glucose-induced metabolism (ATP:ADP ratios). Moreover, ADCY5-silenced islets showed more stochastic long-term evolutions in coordinated beta cell activity following glucose exposure [148]. By contrast, GLP-1-regulated connectivity was normal, suggesting that ADCY5 is unlikely to link incretin signaling to cAMP generation and beta cell communication. Thus, ADCY5 preferentially affects glucose-induced human islet dynamics, possibly through cAMP, which has been shown to increase GJ conductance and trafficking [22, 73, 150], although this has only been so far demonstrated in rodent tissues.
ZnT8
The R325W variant of SLC30A8, the gene encoding zinc transporter 8 (ZnT8), is associated with reduced insulin secretion. ZnT8 is highly expressed in beta cells where its activation leads to Zn2+ accumulation in secretory granules, promoting normal insulin crystallization, storage, and processing [151–154]. While global ZnT8 deletion results in mild insulin secretory deficits, which are only observed in vivo and are undetectable at the dispersed islet level [151, 152], beta cell-specific deletion of the same gene has been reported either to inhibit [153] or stimulate [155] insulin release from isolated islets. Indeed, it has been suggested that defects in glycemia resulting from either global or beta cell specific ZnT8 elimination [152, 153, 155] are due to enhanced insulin clearance by the liver [155]. In any case, and complicating the picture further, rare loss-of-function mutations in SLC30A8 protect against T2DM in man [156]. Nonetheless, alterations in ZnT8 expression lead to altered Ca2+/Zn2+-handling [133, 152, 157], and GJ gating is dependent on fine-regulation of both ions in the vicinity of the plasma membrane [158, 159]; whether this also applies to islets is unknown. Thus, while an effect of SLC30A8/ZnT8 risk alleles on beta cell–beta cell connectivity is not entirely implausible, further studies are required to assess effects of the gene on coordinated activity and the mechanisms underlying this (e.g., changes in Cx36 expression or GJ function).
It should be noted that the studies concerning ADCY5, TCF7L2 and beta cell connectivity were conducted on models in which expression has essentially been eliminated (through gene silencing or genomic deletion). It is likely that any phenotype observed in vivo in man is a consequence of more subtle cellular changes coupled with exposure to a permissive environment. It remains to be seen whether similar effects can be recapitulated in tissue obtained from normoglycemic donors harboring specific risk alleles. Lastly, even the strongest GWAS hits contribute only marginally (though in a statistically significant manner) to T2DM risk, and effects of gene variants on beta cell coordination are presently of uncertain importance in the absence of defined mechanisms/targets.
Rescuing beta cell connectivity during T2DM
Since the intraislet regulation of insulin release may be altered by both genes and the environment to reduce insulin secretion, beta cell connectivity may represent a novel target for the pharmaceutical restoration of functional beta cell mass. While upregulated GJ-signaling provides a logical starting point for the enhancement of beta cell connectivity, investigation of Cx36-modulating compounds has so far been complicated by their off-target effects. Notwithstanding, a recent study has described a panel of seventeen molecules that increase beta cell–beta cell communication, and further screening is warranted to validate their activity profiles and specificity [160]. In addition, atlases of both GPCR and paracrine factor expression/secretion have been reported for human and rodent islets [94, 161], potentially accelerating the elucidation and development of putative candidates for manipulation of beta cell connectivity. Alternatively, personalized medicine/deep-phenotyping approaches [162] could be used to identify individuals where the beneficial effects of GLP-1 and GIP to enhance beta cell connectivity may be exploited [27, 28]. For example, carriers of ADCY5 risk alleles are predicted to respond well to the insulin-raising actions of the incretins, as this gene preferentially impacts glucose action [148]. By contrast, obese subjects would potentially benefit more from the pro-communicatory effects of the sulfonylureas due to altered GLP-1- and GIP-signaling inputs [27, 163, 164].
Future perspectives
The network description of beta cells is still in its infancy and more refined methods are required to better delineate connection topology. Without statistical methods, such as Granger causality, it is impossible to say whether coordinated behavior in an individual cell is the origin or consequence of the connections it shares with its neighbors [42, 165]. Likewise, our understanding of the structural basis for functional connectivity is presently lacking and imaging approaches are required that allow the large-scale interrogation of any underlying physical cell–cell linkages. This is particularly applicable to human islets, where differences in architecture may lead to divergent regulation of insulin secretion and susceptibility to T2DM insults [28, 37, 64]. Lastly, it remains unknown how beta cell population dynamics are influenced by episodes of functional/pathological plasticity in the pancreas, and whether a wiring footprint persists during T2DM that can be exploited to restore insulin secretion.
Summary
The three-dimensional organization of beta cells into islets produces a gain of function in insulin release by fine-tuning beta cell intercommunication. Each islet operates as a self-supported signaling unit in which the spatiotemporally precise propagation of information between neighboring and distant cell ensembles is facilitated by GJ, neural, and paracrine communications. Using imaging approaches together with statistical methods borne from graph theory, the flow of information throughout the beta cell population can be monitored online and mapped. Pertinently, coordinated activity in rodent islets appears to be driven and orchestrated by a subpopulation of beta cells, and wiring density can be increased by both glucose and incretin to stimulate hormone release. We, therefore, propose that, alongside “cell autonomous” effects, environmental and genetic insults may target the intraislet regulation of insulin secretion to precipitate beta cell dysfunction and glucose intolerance, contributing to the risk of developing T2DM.
Acknowledgments
The writing of this review article was supported by a Diabetes UK R.D. Lawrence Research Fellowship (12/0004431) to D.J.H. and Wellcome Trust Senior Investigator (WT098424AIA), MRC Programme (MR/J0003042/1), Diabetes UK Project Grant (11/0004210) and Royal Society Wolfson Research Merit Awards to G.A.R. The work leading to this publication has received support from the Innovative Medicines Initiative Joint Undertaking under grant agreement n° 155005 (IMIDIA), resources of which are composed of financial contribution from the European Union’s Seventh Framework Programme (FP7/2007-2013) and EFPIA companies’ in-kind contribution (G.A.R.). Lastly, we thank Miss Maria Paiva Pessoa for assistance with the figures.
Conflict of interest
The authors have nothing to disclose.
Abbreviations
- AC
Adenyl cyclase
- ACh
Acetylcholine
- ADP
Adenosine diphosphate
- ATP
Adenosine triphosphate
- cAMP
Cyclic adenosine monophosphate
- Cx36
Connexin 36
- Epac
Exchange protein activated by cAMP
- fMCI
Functional multicellular calcium imaging
- GABA
Gamma aminobutyric acid
- GIP
Glucose-dependent insulinotropic polypeptide
- GJ
Gap junction
- GLP-1
Glucagon-like peptide-1
- GWAS
Genome-wide association studies
- GPCR
G protein-coupled receptor
- KATP
ATP-sensitive K+ channel
- SST
Somatostatin
- SNP
Single nucleotide polymorphism
- T2DM
Type 2 diabetes mellitus
- VDCC
Voltage-dependent Ca2+-channel
Footnotes
A correction to this article is available online at https://doi.org/10.1007/s00018-018-2767-2.
Change history
2/15/2018
The original version of this article unfortunately contained a mistake. Legends of Figures 1 and 2 were interchanged. The correct versions are given below.
References
- 1.International Diabetes Federation (2013) IDF Diabetes Atlas, 6th edn. International Diabetes Federation, Brussels, Belgium
- 2.Currie CJ, Poole CD, Gale EA. The influence of glucose-lowering therapies on cancer risk in type 2 diabetes. Diabetologia. 2009;52(9):1766–1777. doi: 10.1007/s00125-009-1440-6. [DOI] [PubMed] [Google Scholar]
- 3.Stitt AW. AGEs and diabetic retinopathy. Invest Ophthalmol Vis Sci. 2010;51(10):4867–4874. doi: 10.1167/iovs.10-5881. [DOI] [PubMed] [Google Scholar]
- 4.van de Bunt M, Gloyn AL. A tale of two glucose transporters: how GLUT2 re-emerged as a contender for glucose transport into the human beta cell. Diabetologia. 2012;55(9):2312–2315. doi: 10.1007/s00125-012-2612-3. [DOI] [PubMed] [Google Scholar]
- 5.Iynedjian PB. Mammalian glucokinase and its gene. Biochem J. 1993;293(Pt 1):1–13. doi: 10.1042/bj2930001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Prentki M, Matschinsky FM, Madiraju SR. Metabolic signaling in fuel-induced insulin secretion. Cell Metab. 2013;18(2):162–185. doi: 10.1016/j.cmet.2013.05.018. [DOI] [PubMed] [Google Scholar]
- 7.Rutter GA. Nutrient-secretion coupling in the pancreatic islet beta-cell: recent advances. Mol Aspects Med. 2001;22(6):247–284. doi: 10.1016/S0098-2997(01)00013-9. [DOI] [PubMed] [Google Scholar]
- 8.Tarasov AI, Semplici F, Ravier MA, Bellomo EA, Pullen TJ, Gilon P, Sekler I, Rizzuto R, Rutter GA. The mitochondrial Ca2+ uniporter MCU is essential for glucose-induced ATP increases in pancreatic beta-cells. PLoS ONE. 2012;7(7):e39722. doi: 10.1371/journal.pone.0039722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kennedy HJ, Pouli AE, Ainscow EK, Jouaville LS, Rizzuto R, Rutter GA. Glucose generates sub-plasma membrane ATP microdomains in single islet beta-cells. Potential role for strategically located mitochondria. J Biol Chem. 1999;274(19):13281–13291. doi: 10.1074/jbc.274.19.13281. [DOI] [PubMed] [Google Scholar]
- 10.Ashcroft FM, Harrison DE, Ashcroft SJ. Glucose induces closure of single potassium channels in isolated rat pancreatic beta-cells. Nature. 1984;312(5993):446–448. doi: 10.1038/312446a0. [DOI] [PubMed] [Google Scholar]
- 11.Ashcroft FM, Gribble FM. ATP-sensitive K+ channels and insulin secretion: their role in health and disease. Diabetologia. 1999;42(8):903–919. doi: 10.1007/s001250051247. [DOI] [PubMed] [Google Scholar]
- 12.Ammala C, Ashcroft FM, Rorsman P. Calcium-independent potentiation of insulin release by cyclic AMP in single beta-cells. Nature. 1993;363(6427):356–358. doi: 10.1038/363356a0. [DOI] [PubMed] [Google Scholar]
- 13.Henquin JC. Triggering and amplifying pathways of regulation of insulin secretion by glucose. Diabetes. 2000;49(11):1751–1760. doi: 10.2337/diabetes.49.11.1751. [DOI] [PubMed] [Google Scholar]
- 14.Rutter GA, Tsuboi T, Ravier MA. Ca2+ microdomains and the control of insulin secretion. Cell Calcium. 2006;40(5–6):539–551. doi: 10.1016/j.ceca.2006.08.015. [DOI] [PubMed] [Google Scholar]
- 15.Emmanouilidou E, Teschemacher AG, Pouli AE, Nicholls LI, Seward EP, Rutter GA. Imaging Ca2+ concentration changes at the secretory vesicle surface with a recombinant targeted cameleon. Curr Biol. 1999;9(16):915–918. doi: 10.1016/S0960-9822(99)80398-4. [DOI] [PubMed] [Google Scholar]
- 16.Tsuboi T, Rutter GA. Multiple forms of “kiss-and-run” exocytosis revealed by evanescent wave microscopy. Curr Biol. 2003;13(7):563–567. doi: 10.1016/S0960-9822(03)00176-3. [DOI] [PubMed] [Google Scholar]
- 17.Rutter GA, Varadi A, Tsuboi T, Parton L, Ravier M. Insulin secretion in health and disease: genomics, proteomics and single vesicle dynamics. Biochem Soc Trans. 2006;34(Pt 2):247–250. doi: 10.1042/BST0340247. [DOI] [PubMed] [Google Scholar]
- 18.Serre-Beinier V, Mas C, Calabrese A, Caton D, Bauquis J, Caille D, Charollais A, Cirulli V, Meda P. Connexins and secretion. Biol Cell. 2002;94(7–8):477–492. doi: 10.1016/S0248-4900(02)00024-2. [DOI] [PubMed] [Google Scholar]
- 19.Bavamian S, Klee P, Britan A, Populaire C, Caille D, Cancela J, Charollais A, Meda P. Islet-cell-to-cell communication as basis for normal insulin secretion. Diabetes Obes Metab. 2007;9(Suppl 2):118–132. doi: 10.1111/j.1463-1326.2007.00780.x. [DOI] [PubMed] [Google Scholar]
- 20.Bosco D, Haefliger JA, Meda P. Connexins: key mediators of endocrine function. Physiol Rev. 2011;91(4):1393–1445. doi: 10.1152/physrev.00027.2010. [DOI] [PubMed] [Google Scholar]
- 21.Meda P. Protein-mediated interactions of pancreatic islet cells. Scientifica (Cairo) 2013;2013:621249. doi: 10.1155/2013/621249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Farnsworth NL, Benninger RK. New insights into the role of connexins in pancreatic islet function and diabetes. FEBS Lett. 2014;588(8):1278–1287. doi: 10.1016/j.febslet.2014.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Salomon D, Meda P. Heterogeneity and contact-dependent regulation of hormone secretion by individual B cells. Exp Cell Res. 1986;162(2):507–520. doi: 10.1016/0014-4827(86)90354-X. [DOI] [PubMed] [Google Scholar]
- 24.Caton D, Calabrese A, Mas C, Serre-Beinier V, Wonkam A, Meda P. Beta-cell crosstalk: a further dimension in the stimulus-secretion coupling of glucose-induced insulin release. Diabetes Metab. 2002;28(6 Pt 2):3S45–3S53. [PubMed] [Google Scholar]
- 25.Steiner DJ, Kim A, Miller K, Hara M. Pancreatic islet plasticity: interspecies comparison of islet architecture and composition. Islets. 2010;2(3):135–145. doi: 10.4161/isl.2.3.11815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hodson DJ, Mitchell RK, Marselli L, Pullen TJ, Brias SG, Semplici F, Everett KL, Cooper DM, Bugliani M, Marchetti P, Lavallard V, Bosco D, Piemonti L, Johnson PR, Hughes SJ, Li D, Li WH, Shapiro AM, Rutter GA. ADCY5 couples glucose to insulin secretion in human islets. Diabetes. 2014 doi: 10.2337/db13-1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hodson DJ, Mitchell RK, Bellomo EA, Sun G, Vinet L, Meda P, Li D, Li WH, Bugliani M, Marchetti P, Bosco D, Piemonti L, Johnson P, Hughes SJ, Rutter GA. Lipotoxicity disrupts incretin-regulated human beta cell connectivity. J Clin Invest. 2013;123(10):4182–4194. doi: 10.1172/JCI68459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rutter GA, Hodson DJ. Minireview: intraislet regulation of insulin secretion in humans. Mol Endocrinol. 2013;27(12):1984–1995. doi: 10.1210/me.2013-1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stozer A, Gosak M, Dolensek J, Perc M, Marhl M, Rupnik MS, Korosak D. Functional connectivity in islets of Langerhans from mouse pancreas tissue slices. PLoS Comput Biol. 2013;9(2):e1002923. doi: 10.1371/journal.pcbi.1002923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stozer A, Dolensek J, Rupnik MS. Glucose-stimulated calcium dynamics in islets of Langerhans in acute mouse pancreas tissue slices. PLoS ONE. 2013;8(1):e54638. doi: 10.1371/journal.pone.0054638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Benninger RK, Piston DW. Cellular communication and heterogeneity in pancreatic islet insulin secretion dynamics. Trends Endocrinol Metab. 2014 doi: 10.1016/j.tem.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Orci L, Unger RH. Functional subdivision of islets of Langerhans and possible role of D cells. Lancet. 1975;2(7947):1243–1244. doi: 10.1016/S0140-6736(75)92078-4. [DOI] [PubMed] [Google Scholar]
- 33.Cabrera O, Berman DM, Kenyon NS, Ricordi C, Berggren PO, Caicedo A. The unique cytoarchitecture of human pancreatic islets has implications for islet cell function. Proc Natl Acad Sci USA. 2006;103(7):2334–2339. doi: 10.1073/pnas.0510790103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nyman LR, Wells KS, Head WS, McCaughey M, Ford E, Brissova M, Piston DW, Powers AC. Real-time, multidimensional in vivo imaging used to investigate blood flow in mouse pancreatic islets. J Clin Invest. 2008;118(11):3790–3797. doi: 10.1172/JCI36209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cleaver O, Dor Y. Vascular instruction of pancreas development. Development. 2012;139(16):2833–2843. doi: 10.1242/dev.065953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Brissova M, Fowler MJ, Nicholson WE, Chu A, Hirshberg B, Harlan DM, Powers AC. Assessment of human pancreatic islet architecture and composition by laser scanning confocal microscopy. J Histochem Cytochem. 2005;53(9):1087–1097. doi: 10.1369/jhc.5C6684.2005. [DOI] [PubMed] [Google Scholar]
- 37.Bosco D, Armanet M, Morel P, Niclauss N, Sgroi A, Muller YD, Giovannoni L, Parnaud G, Berney T. Unique arrangement of alpha- and beta-cells in human islets of Langerhans. Diabetes. 2010;59(5):1202–1210. doi: 10.2337/db09-1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kilimnik G, Zhao B, Jo J, Periwal V, Witkowski P, Misawa R, Hara M. Altered islet composition and disproportionate loss of large islets in patients with type 2 diabetes. PLoS ONE. 2011;6(11):e27445. doi: 10.1371/journal.pone.0027445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Halban PA, Wollheim CB, Blondel B, Meda P, Niesor EN, Mintz DH. The possible importance of contact between pancreatic islet cells for the control of insulin release. Endocrinology. 1982;111(1):86–94. doi: 10.1210/endo-111-1-86. [DOI] [PubMed] [Google Scholar]
- 40.Hauge-Evans AC, Squires PE, Persaud SJ, Jones PM. Pancreatic beta-cell-to-beta-cell interactions are required for integrated responses to nutrient stimuli: enhanced Ca2+ and insulin secretory responses of MIN6 pseudoislets. Diabetes. 1999;48(7):1402–1408. doi: 10.2337/diabetes.48.7.1402. [DOI] [PubMed] [Google Scholar]
- 41.Squires PE, Hauge-Evans AC, Persaud SJ, Jones PM. Synchronization of Ca(2+)-signals within insulin-secreting pseudoislets: effects of gap-junctional uncouplers. Cell Calcium. 2000;27(5):287–296. doi: 10.1054/ceca.2000.0117. [DOI] [PubMed] [Google Scholar]
- 42.Hodson DJ, Molino F, Fontanaud P, Bonnefont X, Mollard P. Investigating and Modelling Pituitary Endocrine Network Function. J Neuroendocrinol. 2010;22:1217–1225. doi: 10.1111/j.1365-2826.2010.02052.x. [DOI] [PubMed] [Google Scholar]
- 43.Takahashi N, Kishimoto T, Nemoto T, Kadowaki T, Kasai H. Fusion pore dynamics and insulin granule exocytosis in the pancreatic islet. Science. 2002;297(5585):1349–1352. doi: 10.1126/science.1073806. [DOI] [PubMed] [Google Scholar]
- 44.Li D, Chen S, Bellomo EA, Tarasov AI, Kaut C, Rutter GA, Li WH. Imaging dynamic insulin release using a fluorescent zinc indicator for monitoring induced exocytotic release (ZIMIR) Proc Natl Acad Sci USA. 2011;108(52):21063–21068. doi: 10.1073/pnas.1109773109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Low JT, Mitchell JM, Do OH, Bax J, Rawlings A, Zavortink M, Morgan G, Parton RG, Gaisano HY, Thorn P. Glucose principally regulates insulin secretion in mouse islets by controlling the numbers of granule fusion events per cell. Diabetologia. 2013;56(12):2629–2637. doi: 10.1007/s00125-013-3019-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pancholi J, Hodson DJ, Jobe K, Rutter GA, Goldup SM, Watkinson M. Biologically targeted probes for Zn2+: a diversity oriented modular “click-SNAr-click” approach. Chem Sci. 2014 doi: 10.1039/c4sc01249f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Akemann W, Mutoh H, Perron A, Rossier J, Knopfel T. Imaging brain electric signals with genetically targeted voltage-sensitive fluorescent proteins. Nat Methods. 2010;7(8):643–649. doi: 10.1038/nmeth.1479. [DOI] [PubMed] [Google Scholar]
- 48.Hodson DJ, Romano N, Schaeffer M, Fontanaud P, Lafont C, Fiordelisio T, Mollard P. Coordination of calcium signals by pituitary endocrine cells in situ. Cell Calcium. 2012;51(3–4):222–230. doi: 10.1016/j.ceca.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 49.Dolensek J, Stozer A, Skelin Klemen M, Miller EW, Slak Rupnik M. The relationship between membrane potential and calcium dynamics in glucose-stimulated beta cell syncytium in acute mouse pancreas tissue slices. PLoS ONE. 2013;8(12):e82374. doi: 10.1371/journal.pone.0082374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schlegel W, Winiger BP, Mollard P, Vacher P, Wuarin F, Zahnd GR, Wollheim CB, Dufy B. Oscillations of cytosolic Ca2+ in pituitary cells due to action potentials. Nature. 1987;329(6141):719–721. doi: 10.1038/329719a0. [DOI] [PubMed] [Google Scholar]
- 51.Peterlin ZA, Kozloski J, Mao BQ, Tsiola A, Yuste R. Optical probing of neuronal circuits with calcium indicators. Proc Natl Acad Sci USA. 2000;97(7):3619–3624. doi: 10.1073/pnas.97.7.3619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cossart R, Ikegaya Y, Yuste R. Calcium imaging of cortical networks dynamics. Cell Calcium. 2005;37(5):451–457. doi: 10.1016/j.ceca.2005.01.013. [DOI] [PubMed] [Google Scholar]
- 53.Ikegaya Y, Le Bon-Jego M, Yuste R. Large-scale imaging of cortical network activity with calcium indicators. Neurosci Res. 2005;52(2):132–138. doi: 10.1016/j.neures.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 54.Hodson DJ, Schaeffer M, Romano N, Fontanaud P, Lafont C, Birkenstock J, Molino F, Christian H, Lockey J, Carmignac D, Fernandez-Fuente M, Le Tissier P, Mollard P. Existence of long-lasting experience-dependent plasticity in endocrine cell networks. Nature Commun. 2012;3:605. doi: 10.1038/ncomms1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hodson DJ, Tarasov AI, Gimeno Brias S, Mitchell RK, Johnston NR, Haghollahi S, Cane MC, Bugliani M, Marchetti P, Bosco D, Johnson PR, Hughes SJ, Rutter GA. Incretin-modulated beta cell energetics in intact islets of Langerhans. Mol Endocrinol. 2014;28(6):860–871. doi: 10.1210/me.2014-1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Alon U. Network motifs: theory and experimental approaches. Nat Rev Genet. 2007;8(6):450–461. doi: 10.1038/nrg2102. [DOI] [PubMed] [Google Scholar]
- 57.Bullmore E, Sporns O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10(3):186–198. doi: 10.1038/nrn2575. [DOI] [PubMed] [Google Scholar]
- 58.Price DJ. Networks of Scientific Papers. Science. 1965;149:510–515. doi: 10.1126/science.149.3683.510. [DOI] [PubMed] [Google Scholar]
- 59.Barabasi AL, Albert R. Emergence of scaling in random networks. Science. 1999;286(5439):509–512. doi: 10.1126/science.286.5439.509. [DOI] [PubMed] [Google Scholar]
- 60.Barabasi AL. Scale-free networks: a decade and beyond. Science. 2009;325(5939):412–413. doi: 10.1126/science.1173299. [DOI] [PubMed] [Google Scholar]
- 61.Bonifazi P, Goldin M, Picardo MA, Jorquera I, Cattani A, Bianconi G, Represa A, Ben-Ari Y, Cossart R. GABAergic hub neurons orchestrate synchrony in developing hippocampal networks. Science. 2009;326(5958):1419–1424. doi: 10.1126/science.1175509. [DOI] [PubMed] [Google Scholar]
- 62.Mollard P, Hodson DJ, Lafont C, Rizzoti K, Drouin J. A tridimensional view of pituitary development and function. Trends Endocrinol Metab. 2012 doi: 10.1016/j.tem.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 63.Le Tissier PR, Hodson DJ, Lafont C, Fontanaud P, Schaeffer M, Mollard P. Anterior pituitary cell networks. Front Neuroendocrinol. 2012;33(3):252–266. doi: 10.1016/j.yfrne.2012.08.002. [DOI] [PubMed] [Google Scholar]
- 64.Caicedo A. Paracrine and autocrine interactions in the human islet: more than meets the eye. Semin Cell Dev Biol. 2013;24(1):11–21. doi: 10.1016/j.semcdb.2012.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Serre-Beinier V, Le Gurun S, Belluardo N, Trovato-Salinaro A, Charollais A, Haefliger JA, Condorelli DF, Meda P. Cx36 preferentially connects beta-cells within pancreatic islets. Diabetes. 2000;49(5):727–734. doi: 10.2337/diabetes.49.5.727. [DOI] [PubMed] [Google Scholar]
- 66.Condorelli DF, Belluardo N, Trovato-Salinaro A, Mudo G. Expression of Cx36 in mammalian neurons. Brain Res Brain Res Rev. 2000;32(1):72–85. doi: 10.1016/S0165-0173(99)00068-5. [DOI] [PubMed] [Google Scholar]
- 67.Charpantier E, Cancela J, Meda P. Beta cells preferentially exchange cationic molecules via connexin 36 gap junction channels. Diabetologia. 2007;50(11):2332–2341. doi: 10.1007/s00125-007-0807-9. [DOI] [PubMed] [Google Scholar]
- 68.Ravier MA, Guldenagel M, Charollais A, Gjinovci A, Caille D, Sohl G, Wollheim CB, Willecke K, Henquin JC, Meda P. Loss of connexin36 channels alters beta-cell coupling, islet synchronization of glucose-induced Ca2+ and insulin oscillations, and basal insulin release. Diabetes. 2005;54(6):1798–1807. doi: 10.2337/diabetes.54.6.1798. [DOI] [PubMed] [Google Scholar]
- 69.Speier S, Gjinovci A, Charollais A, Meda P, Rupnik M. Cx36-mediated coupling reduces beta-cell heterogeneity, confines the stimulating glucose concentration range, and affects insulin release kinetics. Diabetes. 2007;56(4):1078–1086. doi: 10.2337/db06-0232. [DOI] [PubMed] [Google Scholar]
- 70.Rocheleau JV, Remedi MS, Granada B, Head WS, Koster JC, Nichols CG, Piston DW. Critical role of gap junction coupled KATP channel activity for regulated insulin secretion. PLoS Biol. 2006;4(2):e26. doi: 10.1371/journal.pbio.0040026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Head WS, Orseth ML, Nunemaker CS, Satin LS, Piston DW, Benninger RK. Connexin-36 gap junctions regulate in vivo first- and second-phase insulin secretion dynamics and glucose tolerance in the conscious mouse. Diabetes. 2012;61(7):1700–1707. doi: 10.2337/db11-1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Serre-Beinier V, Bosco D, Zulianello L, Charollais A, Caille D, Charpantier E, Gauthier BR, Diaferia GR, Giepmans BN, Lupi R, Marchetti P, Deng S, Buhler L, Berney T, Cirulli V, Meda P. Cx36 makes channels coupling human pancreatic beta-cells, and correlates with insulin expression. Hum Mol Genet. 2009;18(3):428–439. doi: 10.1093/hmg/ddn370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Farnsworth NL, Hemmati A, Pozzoli M, Benninger RK. Fluorescence recovery after photobleaching reveals regulation and distribution of Cx36 gap junction coupling within mouse islets of langerhans. J Physiol. 2014 doi: 10.1113/jphysiol.2014.276733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ahren B. Autonomic regulation of islet hormone secretion–implications for health and disease. Diabetologia. 2000;43(4):393–410. doi: 10.1007/s001250051322. [DOI] [PubMed] [Google Scholar]
- 75.Zhang M, Fendler B, Peercy B, Goel P, Bertram R, Sherman A, Satin L. Long lasting synchronization of calcium oscillations by cholinergic stimulation in isolated pancreatic islets. Biophys J. 2008;95(10):4676–4688. doi: 10.1529/biophysj.107.125088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fendler B, Zhang M, Satin L, Bertram R. Synchronization of pancreatic islet oscillations by intrapancreatic ganglia: a modeling study. Biophys J. 2009;97(3):722–729. doi: 10.1016/j.bpj.2009.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ahren B, Holst JJ. The cephalic insulin response to meal ingestion in humans is dependent on both cholinergic and noncholinergic mechanisms and is important for postprandial glycemia. Diabetes. 2001;50(5):1030–1038. doi: 10.2337/diabetes.50.5.1030. [DOI] [PubMed] [Google Scholar]
- 78.Filipsson K, Kvist-Reimer M, Ahren B. The neuropeptide pituitary adenylate cyclase-activating polypeptide and islet function. Diabetes. 2001;50(9):1959–1969. doi: 10.2337/diabetes.50.9.1959. [DOI] [PubMed] [Google Scholar]
- 79.Kurose T, Seino Y, Nishi S, Tsuji K, Taminato T, Tsuda K, Imura H. Mechanism of sympathetic neural regulation of insulin, somatostatin, and glucagon secretion. Am J Physiol. 1990;258(1 Pt 1):E220–E227. doi: 10.1152/ajpendo.1990.258.1.E220. [DOI] [PubMed] [Google Scholar]
- 80.Nilsson T, Arkhammar P, Rorsman P, Berggren PO. Inhibition of glucose-stimulated insulin release by alpha 2-adrenoceptor activation is parallelled by both a repolarization and a reduction in cytoplasmic free Ca2+ concentration. J Biol Chem. 1988;263(4):1855–1860. [PubMed] [Google Scholar]
- 81.Kuo WN, Hodgins DS, Kuo JF. Adenylate cyclase in islets of Langerhans. Isolation of islets and regulation of adenylate cyclase activity by various hormones and agents. J Biol Chem. 1973;248(8):2705–2711. [PubMed] [Google Scholar]
- 82.Asensio C, Jimenez M, Kuhne F, Rohner-Jeanrenaud F, Muzzin P. The lack of beta-adrenoceptors results in enhanced insulin sensitivity in mice exhibiting increased adiposity and glucose intolerance. Diabetes. 2005;54(12):3490–3495. doi: 10.2337/diabetes.54.12.3490. [DOI] [PubMed] [Google Scholar]
- 83.Rodriguez-Diaz R, Abdulreda MH, Formoso AL, Gans I, Ricordi C, Berggren PO, Caicedo A. Innervation patterns of autonomic axons in the human endocrine pancreas. Cell Metab. 2011;14(1):45–54. doi: 10.1016/j.cmet.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Rodriguez-Diaz R, Dando R, Jacques-Silva MC, Fachado A, Molina J, Abdulreda MH, Ricordi C, Roper SD, Berggren PO, Caicedo A. Alpha cells secrete acetylcholine as a non-neuronal paracrine signal priming beta cell function in humans. Nat Med. 2011;17(7):888–892. doi: 10.1038/nm.2371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Molina J, Rodriguez-Diaz R, Fachado A, Jacques-Silva MC, Berggren PO, Caicedo A. control of insulin secretion by cholinergic signaling in the human pancreatic islet. Diabetes. 2014 doi: 10.2337/db13-1371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Martin F, Soria B. Glucose-induced [Ca2+]i oscillations in single human pancreatic islets. Cell Calcium. 1996;20(5):409–414. doi: 10.1016/S0143-4160(96)90003-2. [DOI] [PubMed] [Google Scholar]
- 87.diIorio P, Rittenhouse AR, Bortell R, Jurczyk A. Role of cilia in normal pancreas function and in diseased states. Birth Defects Res C Embryo Today. 2014;102(2):126–138. doi: 10.1002/bdrc.21064. [DOI] [PubMed] [Google Scholar]
- 88.Oh EC, Katsanis N. Cilia in vertebrate development and disease. Development. 2012;139(3):443–448. doi: 10.1242/dev.050054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cano DA, Sekine S, Hebrok M. Primary cilia deletion in pancreatic epithelial cells results in cyst formation and pancreatitis. Gastroenterology. 2006;131(6):1856–1869. doi: 10.1053/j.gastro.2006.10.050. [DOI] [PubMed] [Google Scholar]
- 90.Ait-Lounis A, Baas D, Barras E, Benadiba C, Charollais A, Nlend Nlend R, Liegeois D, Meda P, Durand B, Reith W. Novel function of the ciliogenic transcription factor RFX3 in development of the endocrine pancreas. Diabetes. 2007;56(4):950–959. doi: 10.2337/db06-1187. [DOI] [PubMed] [Google Scholar]
- 91.Granot Z, Swisa A, Magenheim J, Stolovich-Rain M, Fujimoto W, Manduchi E, Miki T, Lennerz JK, Stoeckert CJ, Jr, Meyuhas O, Seino S, Permutt MA, Piwnica-Worms H, Bardeesy N, Dor Y. LKB1 regulates pancreatic beta cell size, polarity, and function. Cell Metab. 2009;10(4):296–308. doi: 10.1016/j.cmet.2009.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Green JA, Mykytyn K. Neuronal primary cilia: an underappreciated signaling and sensory organelle in the brain. Neuropsychopharmacology. 2014;39(1):244–245. doi: 10.1038/npp.2013.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Wang J, Silva M, Haas LA, Morsci NS, Nguyen KC, Hall DH, Barr MM. C. elegans ciliated sensory neurons release extracellular vesicles that function in animal communication. Curr Biol. 2014;24(5):519–525. doi: 10.1016/j.cub.2014.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yang YH, Szabat M, Bragagnini C, Kott K, Helgason CD, Hoffman BG, Johnson JD. Paracrine signalling loops in adult human and mouse pancreatic islets: netrins modulate beta cell apoptosis signalling via dependence receptors. Diabetologia. 2011;54(4):828–842. doi: 10.1007/s00125-010-2012-5. [DOI] [PubMed] [Google Scholar]
- 95.Ren J, Sherman A, Bertram R, Goforth PB, Nunemaker CS, Waters CD, Satin LS. Slow oscillations of KATP conductance in mouse pancreatic islets provide support for electrical bursting driven by metabolic oscillations. Am J Physiol Endocrinol Metab. 2013;305(7):E805–E817. doi: 10.1152/ajpendo.00046.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Merrins MJ, Fendler B, Zhang M, Sherman A, Bertram R, Satin LS. Metabolic oscillations in pancreatic islets depend on the intracellular Ca2+ level but not Ca2+ oscillations. Biophys J. 2010;99(1):76–84. doi: 10.1016/j.bpj.2010.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nunemaker CS, Satin LS. Comparison of metabolic oscillations from mouse pancreatic beta cells and islets. Endocrine. 2004;25(1):61–67. doi: 10.1385/ENDO:25:1:61. [DOI] [PubMed] [Google Scholar]
- 98.Li J, Shuai HY, Gylfe E, Tengholm A. Oscillations of sub-membrane ATP in glucose-stimulated beta cells depend on negative feedback from Ca(2+) Diabetologia. 2013;56(7):1577–1586. doi: 10.1007/s00125-013-2894-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Bennett BD, Jetton TL, Ying G, Magnuson MA, Piston DW. Quantitative subcellular imaging of glucose metabolism within intact pancreatic islets. J Biol Chem. 1996;271(7):3647–3651. doi: 10.1074/jbc.271.7.3647. [DOI] [PubMed] [Google Scholar]
- 100.Piston DW, Knobel SM. Quantitative imaging of metabolism by two-photon excitation microscopy. Methods Enzymol. 1999;307:351–368. doi: 10.1016/S0076-6879(99)07023-8. [DOI] [PubMed] [Google Scholar]
- 101.Nunemaker CS, Dishinger JF, Dula SB, Wu R, Merrins MJ, Reid KR, Sherman A, Kennedy RT, Satin LS. Glucose metabolism, islet architecture, and genetic homogeneity in imprinting of [Ca2+](i) and insulin rhythms in mouse islets. PLoS ONE. 2009;4(12):e8428. doi: 10.1371/journal.pone.0008428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kohen E, Kohen C, Thorell B, Mintz DH, Rabinovitch A. Intercellular communication in pancreatic islet monolayer cultures: a microfluorometric study. Science. 1979;204(4395):862–865. doi: 10.1126/science.35828. [DOI] [PubMed] [Google Scholar]
- 103.Meda P, Amherdt M, Perrelet A, Orci L. Metabolic coupling between cultured pancreatic b-cells. Exp Cell Res. 1981;133(2):421–430. doi: 10.1016/0014-4827(81)90335-9. [DOI] [PubMed] [Google Scholar]
- 104.Reimann F, Gribble FM. Glucose-sensing in glucagon-like peptide-1-secreting cells. Diabetes. 2002;51(9):2757–2763. doi: 10.2337/diabetes.51.9.2757. [DOI] [PubMed] [Google Scholar]
- 105.Tolhurst G, Reimann F, Gribble FM. Nutritional regulation of glucagon-like peptide-1 secretion. J Physiol. 2009;587(Pt 1):27–32. doi: 10.1113/jphysiol.2008.164012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Parker HE, Wallis K, le Roux CW, Wong KY, Reimann F, Gribble FM. Molecular mechanisms underlying bile acid-stimulated glucagon-like peptide-1 secretion. Br J Pharmacol. 2012;165(2):414–423. doi: 10.1111/j.1476-5381.2011.01561.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Gomez E, Pritchard C, Herbert TP. cAMP-dependent protein kinase and Ca2+ influx through L-type voltage-gated calcium channels mediate Raf-independent activation of extracellular regulated kinase in response to glucagon-like peptide-1 in pancreatic beta-cells. J Biol Chem. 2002;277(50):48146–48151. doi: 10.1074/jbc.M209165200. [DOI] [PubMed] [Google Scholar]
- 108.Leech CA, Dzhura I, Chepurny OG, Kang G, Schwede F, Genieser HG, Holz GG. Molecular physiology of glucagon-like peptide-1 insulin secretagogue action in pancreatic beta cells. Prog Biophys Mol Biol. 2011;107(2):236–247. doi: 10.1016/j.pbiomolbio.2011.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Ravier MA, Leduc M, Richard J, Linck N, Varrault A, Pirot N, Roussel MM, Bockaert J, Dalle S, Bertrand G. beta-Arrestin2 plays a key role in the modulation of the pancreatic beta cell mass in mice. Diabetologia. 2013 doi: 10.1007/s00125-013-3130-7. [DOI] [PubMed] [Google Scholar]
- 110.Tsuboi T, da Xavier Silva G, Holz GG, Jouaville LS, Thomas AP, Rutter GA. Glucagon-like peptide-1 mobilizes intracellular Ca2+ and stimulates mitochondrial ATP synthesis in pancreatic MIN6 beta-cells. Biochem J. 2003;369(Pt 2):287–299. doi: 10.1042/bj20021288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Peyot ML, Gray JP, Lamontagne J, Smith PJ, Holz GG, Madiraju SR, Prentki M, Heart E. Glucagon-like peptide-1 induced signaling and insulin secretion do not drive fuel and energy metabolism in primary rodent pancreatic beta-cells. PLoS ONE. 2009;4(7):e6221. doi: 10.1371/journal.pone.0006221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Berg J, Hung YP, Yellen G. A genetically encoded fluorescent reporter of ATP:ADP ratio. Nat Methods. 2009;6(2):161–166. doi: 10.1038/nmeth.1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Hodson DJ, Mitchell RK, Johnston N, Thorens B, Ferrer J, Rutter GA. Optical control of beta cell function. Diabet Med. 2014;31:6. doi: 10.1111/dme.12271. [DOI] [Google Scholar]
- 114.Nauck MA, Homberger E, Siegel EG, Allen RC, Eaton RP, Ebert R, Creutzfeldt W. Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J Clin Endocrinol Metab. 1986;63(2):492–498. doi: 10.1210/jcem-63-2-492. [DOI] [PubMed] [Google Scholar]
- 115.Nauck MA, Heimesaat MM, Orskov C, Holst JJ, Ebert R, Creutzfeldt W. Preserved incretin activity of glucagon-like peptide 1 [7-36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J Clin Invest. 1993;91(1):301–307. doi: 10.1172/JCI116186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kjems LL, Holst JJ, Volund A, Madsbad S. The influence of GLP-1 on glucose-stimulated insulin secretion: effects on beta-cell sensitivity in type 2 and nondiabetic subjects. Diabetes. 2003;52(2):380–386. doi: 10.2337/diabetes.52.2.380. [DOI] [PubMed] [Google Scholar]
- 117.Meier JJ, Nauck MA. Is the diminished incretin effect in type 2 diabetes just an epi-phenomenon of impaired beta-cell function? Diabetes. 2010;59(5):1117–1125. doi: 10.2337/db09-1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Muscelli E, Mari A, Casolaro A, Camastra S, Seghieri G, Gastaldelli A, Holst JJ, Ferrannini E. Separate impact of obesity and glucose tolerance on the incretin effect in normal subjects and type 2 diabetic patients. Diabetes. 2008;57(5):1340–1348. doi: 10.2337/db07-1315. [DOI] [PubMed] [Google Scholar]
- 119.Knop FK, Aaboe K, Vilsboll T, Volund A, Holst JJ, Krarup T, Madsbad S. Impaired incretin effect and fasting hyperglucagonaemia characterizing type 2 diabetic subjects are early signs of dysmetabolism in obesity. Diabetes Obes Metab. 2012;14(6):500–510. doi: 10.1111/j.1463-1326.2011.01549.x. [DOI] [PubMed] [Google Scholar]
- 120.Allagnat F, Alonso F, Martin D, Abderrahmani A, Waeber G, Haefliger JA. ICER-1gamma overexpression drives palmitate-mediated connexin36 down-regulation in insulin-secreting cells. J Biol Chem. 2008;283(9):5226–5234. doi: 10.1074/jbc.M708181200. [DOI] [PubMed] [Google Scholar]
- 121.Haefliger JA, Martin D, Favre D, Petremand Y, Mazzolai L, Abderrahmani A, Meda P, Waeber G, Allagnat F. Reduction of connexin36 content by ICER-1 contributes to insulin-secreting cells apoptosis induced by oxidized LDL particles. PLoS ONE. 2013;8(1):e55198. doi: 10.1371/journal.pone.0055198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Newman B, Selby JV, King MC, Slemenda C, Fabsitz R, Friedman GD. Concordance for type 2 (non-insulin-dependent) diabetes mellitus in male twins. Diabetologia. 1987;30(10):763–768. doi: 10.1007/BF00275741. [DOI] [PubMed] [Google Scholar]
- 123.Pierce M, Keen H, Bradley C. Risk of diabetes in offspring of parents with non-insulin-dependent diabetes. Diabet Med. 1995;12(1):6–13. doi: 10.1111/j.1464-5491.1995.tb02054.x. [DOI] [PubMed] [Google Scholar]
- 124.Medici F, Hawa M, Ianari A, Pyke DA, Leslie RD. Concordance rate for type II diabetes mellitus in monozygotic twins: actuarial analysis. Diabetologia. 1999;42(2):146–150. doi: 10.1007/s001250051132. [DOI] [PubMed] [Google Scholar]
- 125.Rutter GA. Understanding genes identified by genome-wide association studies for type 2 diabetes. Diabet Med. 2014 doi: 10.1111/dme.12579. [DOI] [PubMed] [Google Scholar]
- 126.McCarthy MI. Genomics, type 2 diabetes, and obesity. N Engl J Med. 2010;363(24):2339–2350. doi: 10.1056/NEJMra0906948. [DOI] [PubMed] [Google Scholar]
- 127.Lyssenko V, Lupi R, Marchetti P, Del Guerra S, Orho-Melander M, Almgren P, Sjogren M, Ling C, Eriksson KF, Lethagen AL, Mancarella R, Berglund G, Tuomi T, Nilsson P, Del Prato S, Groop L. Mechanisms by which common variants in the TCF7L2 gene increase risk of type 2 diabetes. J Clin Invest. 2007;117(8):2155–2163. doi: 10.1172/JCI30706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Salonen JT, Uimari P, Aalto JM, Pirskanen M, Kaikkonen J, Todorova B, Hypponen J, Korhonen VP, Asikainen J, Devine C, Tuomainen TP, Luedemann J, Nauck M, Kerner W, Stephens RH, New JP, Ollier WE, Gibson JM, Payton A, Horan MA, Pendleton N, Mahoney W, Meyre D, Delplanque J, Froguel P, Luzzatto O, Yakir B, Darvasi A. Type 2 diabetes whole-genome association study in four populations: the DiaGen consortium. Am J Hum Genet. 2007;81(2):338–345. doi: 10.1086/520599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Vaxillaire M, Veslot J, Dina C, Proenca C, Cauchi S, Charpentier G, Tichet J, Fumeron F, Marre M, Meyre D, Balkau B, Froguel P. Impact of common type 2 diabetes risk polymorphisms in the DESIR prospective study. Diabetes. 2008;57(1):244–254. doi: 10.2337/db07-0615. [DOI] [PubMed] [Google Scholar]
- 130.Palmer ND, Lehtinen AB, Langefeld CD, Campbell JK, Haffner SM, Norris JM, Bergman RN, Goodarzi MO, Rotter JI, Bowden DW. Association of TCF7L2 gene polymorphisms with reduced acute insulin response in Hispanic Americans. J Clin Endocrinol Metab. 2008;93(1):304–309. doi: 10.1210/jc.2007-1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Schafer SA, Tschritter O, Machicao F, Thamer C, Stefan N, Gallwitz B, Holst JJ, Dekker JM, t Hart LM, Nijpels G, van Haeften TW, Haring HU, Fritsche A. Impaired glucagon-like peptide-1-induced insulin secretion in carriers of transcription factor 7-like 2 (TCF7L2) gene polymorphisms. Diabetologia. 2007;50(12):2443–2450. doi: 10.1007/s00125-007-0753-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Pilgaard K, Jensen CB, Schou JH, Lyssenko V, Wegner L, Brons C, Vilsboll T, Hansen T, Madsbad S, Holst JJ, Volund A, Poulsen P, Groop L, Pedersen O, Vaag AA. The T allele of rs7903146 TCF7L2 is associated with impaired insulinotropic action of incretin hormones, reduced 24 h profiles of plasma insulin and glucagon, and increased hepatic glucose production in young healthy men. Diabetologia. 2009;52(7):1298–1307. doi: 10.1007/s00125-009-1307-x. [DOI] [PubMed] [Google Scholar]
- 133.da Silva Xavier G, Loder MK, McDonald A, Tarasov AI, Carzaniga R, Kronenberger K, Barg S, Rutter GA. TCF7L2 regulates late events in insulin secretion from pancreatic islet beta-cells. Diabetes. 2009;58(4):894–905. doi: 10.2337/db08-1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Shu L, Sauter NS, Schulthess FT, Matveyenko AV, Oberholzer J, Maedler K. Transcription factor 7-like 2 regulates beta-cell survival and function in human pancreatic islets. Diabetes. 2008;57(3):645–653. doi: 10.2337/db07-0847. [DOI] [PubMed] [Google Scholar]
- 135.da Silva Xavier G, Mondragon A, Sun G, Chen L, McGinty JA, French PM, Rutter GA. Abnormal glucose tolerance and insulin secretion in pancreas-specific Tcf7l2-null mice. Diabetologia. 2012;55(10):2667–2676. doi: 10.1007/s00125-012-2600-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.da Silva Xavier G, Mondragon A, Mitchell RK, Hodson DJ, Ferrer J, Thoren B, Chen L, McGinty JA, French PM, Rutter GA (2014) Defective glucose homeostasis in mice inactivated selectively for Tcf7l2 in the adult beta cell with an Ins1-controlled Cre. In: EASD, Vienna. Diabetologia. http://www.easdvirtualmeeting.org/resources/17180 (in press)
- 137.Boj SF, van Es JH, Huch M, Li VS, Jose A, Hatzis P, Mokry M, Haegebarth A, van den Born M, Chambon P, Voshol P, Dor Y, Cuppen E, Fillat C, Clevers H. Diabetes risk gene and Wnt effector Tcf7l2/TCF4 controls hepatic response to perinatal and adult metabolic demand. Cell. 2012;151(7):1595–1607. doi: 10.1016/j.cell.2012.10.053. [DOI] [PubMed] [Google Scholar]
- 138.Shu L, Matveyenko AV, Kerr-Conte J, Cho JH, McIntosh CH, Maedler K. Decreased TCF7L2 protein levels in type 2 diabetes mellitus correlate with downregulation of GIP- and GLP-1 receptors and impaired beta-cell function. Hum Mol Genet. 2009;18(13):2388–2399. doi: 10.1093/hmg/ddp178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Rosengren AH, Braun M, Mahdi T, Andersson SA, Travers ME, Shigeto M, Zhang E, Almgren P, Ladenvall C, Axelsson AS, Edlund A, Pedersen MG, Jonsson A, Ramracheya R, Tang Y, Walker JN, Barrett A, Johnson PR, Lyssenko V, McCarthy MI, Groop L, Salehi A, Gloyn AL, Renstrom E, Rorsman P, Eliasson L. Reduced insulin exocytosis in human pancreatic beta-cells with gene variants linked to type 2 diabetes. Diabetes. 2012;61(7):1726–1733. doi: 10.2337/db11-1516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Loder MK, da Silva Xavier G, McDonald A, Rutter GA. TCF7L2 controls insulin gene expression and insulin secretion in mature pancreatic beta-cells. Biochem Soc Trans. 2008;36(Pt 3):357–359. doi: 10.1042/BST0360357. [DOI] [PubMed] [Google Scholar]
- 141.Zhou Y, Park SY, Su J, Bailey K, Ottosson-Laakso E, Shcherbina L, Oskolkov N, Zhang E, Thevenin T, Fadista J, Bennet H, Vikman P, Wierup N, Fex M, Rung J, Wollheim C, Nobrega M, Renstrom E, Groop L, Hansson O. TCF7L2 is a master regulator of insulin production and processing. Hum Mol Genet. 2014 doi: 10.1093/hmg/ddu359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Yoshimura M, Cooper DM. Cloning and expression of a Ca(2+)-inhibitable adenylyl cyclase from NCB-20 cells. Proc Natl Acad Sci USA. 1992;89(15):6716–6720. doi: 10.1073/pnas.89.15.6716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Yan L, Vatner DE, O’Connor JP, Ivessa A, Ge H, Chen W, Hirotani S, Ishikawa Y, Sadoshima J, Vatner SF. Type 5 adenylyl cyclase disruption increases longevity and protects against stress. Cell. 2007;130(2):247–258. doi: 10.1016/j.cell.2007.05.038. [DOI] [PubMed] [Google Scholar]
- 144.Holz GG, Leech CA, Chepurny OG. New insights concerning the molecular basis for defective glucoregulation in soluble adenylyl cyclase knockout mice. Biochim Biophys Acta. 2014 doi: 10.1016/j.bbadis.2014.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Leech CA, Castonguay MA, Habener JF. Expression of adenylyl cyclase subtypes in pancreatic beta-cells. Biochem Biophys Res Commun. 1999;254(3):703–706. doi: 10.1006/bbrc.1998.9906. [DOI] [PubMed] [Google Scholar]
- 146.Dupuis J, Langenberg C, Prokopenko I, Saxena R, Soranzo N, Jackson AU, Wheeler E, Glazer NL, Bouatia-Naji N, Gloyn AL, Lindgren CM, Magi R, Morris AP, Randall J, Johnson T, Elliott P, Rybin D, Thorleifsson G, Steinthorsdottir V, Henneman P, Grallert H, Dehghan A, Hottenga JJ, Franklin CS, Navarro P, Song K, Goel A, Perry JR, Egan JM, Lajunen T, Grarup N, Sparso T, Doney A, Voight BF, Stringham HM, Li M, Kanoni S, Shrader P, Cavalcanti-Proenca C, Kumari M, Qi L, Timpson NJ, Gieger C, Zabena C, Rocheleau G, Ingelsson E, An P, O’Connell J, Luan J, Elliott A, McCarroll SA, Payne F, Roccasecca RM, Pattou F, Sethupathy P, Ardlie K, Ariyurek Y, Balkau B, Barter P, Beilby JP, Ben-Shlomo Y, Benediktsson R, Bennett AJ, Bergmann S, Bochud M, Boerwinkle E, Bonnefond A, Bonnycastle LL, Borch-Johnsen K, Bottcher Y, Brunner E, Bumpstead SJ, Charpentier G, Chen YD, Chines P, Clarke R, Coin LJ, Cooper MN, Cornelis M, Crawford G, Crisponi L, Day IN, de Geus EJ, Delplanque J, Dina C, Erdos MR, Fedson AC, Fischer-Rosinsky A, Forouhi NG, Fox CS, Frants R, Franzosi MG, Galan P, Goodarzi MO, Graessler J, Groves CJ, Grundy S, Gwilliam R, Gyllensten U, Hadjadj S, Hallmans G, Hammond N, Han X, Hartikainen AL, Hassanali N, Hayward C, Heath SC, Hercberg S, Herder C, Hicks AA, Hillman DR, Hingorani AD, Hofman A, Hui J, Hung J, Isomaa B, Johnson PR, Jorgensen T, Jula A, Kaakinen M, Kaprio J, Kesaniemi YA, Kivimaki M, Knight B, Koskinen S, Kovacs P, Kyvik KO, Lathrop GM, Lawlor DA, Le Bacquer O, Lecoeur C, Li Y, Lyssenko V, Mahley R, Mangino M, Manning AK, Martinez-Larrad MT, McAteer JB, McCulloch LJ, McPherson R, Meisinger C, Melzer D, Meyre D, Mitchell BD, Morken MA, Mukherjee S, Naitza S, Narisu N, Neville MJ, Oostra BA, Orru M, Pakyz R, Palmer CN, Paolisso G, Pattaro C, Pearson D, Peden JF, Pedersen NL, Perola M, Pfeiffer AF, Pichler I, Polasek O, Posthuma D, Potter SC, Pouta A, Province MA, Psaty BM, Rathmann W, Rayner NW, Rice K, Ripatti S, Rivadeneira F, Roden M, Rolandsson O, Sandbaek A, Sandhu M, Sanna S, Sayer AA, Scheet P, Scott LJ, Seedorf U, Sharp SJ, Shields B, Sigurethsson G, Sijbrands EJ, Silveira A, Simpson L, Singleton A, Smith NL, Sovio U, Swift A, Syddall H, Syvanen AC, Tanaka T, Thorand B, Tichet J, Tonjes A, Tuomi T, Uitterlinden AG, van Dijk KW, van Hoek M, Varma D, Visvikis-Siest S, Vitart V, Vogelzangs N, Waeber G, Wagner PJ, Walley A, Walters GB, Ward KL, Watkins H, Weedon MN, Wild SH, Willemsen G, Witteman JC, Yarnell JW, Zeggini E, Zelenika D, Zethelius B, Zhai G, Zhao JH, Zillikens MC, Borecki IB, Loos RJ, Meneton P, Magnusson PK, Nathan DM, Williams GH, Hattersley AT, Silander K, Salomaa V, Smith GD, Bornstein SR, Schwarz P, Spranger J, Karpe F, Shuldiner AR, Cooper C, Dedoussis GV, Serrano-Rios M, Morris AD, Lind L, Palmer LJ, Hu FB, Franks PW, Ebrahim S, Marmot M, Kao WH, Pankow JS, Sampson MJ, Kuusisto J, Laakso M, Hansen T, Pedersen O, Pramstaller PP, Wichmann HE, Illig T, Rudan I, Wright AF, Stumvoll M, Campbell H, Wilson JF, Bergman RN, Buchanan TA, Collins FS, Mohlke KL, Tuomilehto J, Valle TT, Altshuler D, Rotter JI, Siscovick DS, Penninx BW, Boomsma DI, Deloukas P, Spector TD, Frayling TM, Ferrucci L, Kong A, Thorsteinsdottir U, Stefansson K, van Duijn CM, Aulchenko YS, Cao A, Scuteri A, Schlessinger D, Uda M, Ruokonen A, Jarvelin MR, Waterworth DM, Vollenweider P, Peltonen L, Mooser V, Abecasis GR, Wareham NJ, Sladek R, Froguel P, Watanabe RM, Meigs JB, Groop L, Boehnke M, McCarthy MI, Florez JC, Barroso I. New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat Genet. 2010;42(2):105–116. doi: 10.1038/ng.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Rees SD, Hydrie MZ, O’Hare JP, Kumar S, Shera AS, Basit A, Barnett AH, Kelly MA. Effects of 16 genetic variants on fasting glucose and type 2 diabetes in South Asians: ADCY5 and GLIS3 variants may predispose to type 2 diabetes. PLoS ONE. 2011;6(9):e24710. doi: 10.1371/journal.pone.0024710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hodson DJ, Mitchell RK, Marselli L, Pullen TJ, Gimeno Brias S, Semplici F, Everett KL, Cooper DMF, Bugliani M, Marchetti P, Lavallard V, Bosco D, Piemonti L, Johnson PR, Hughes SJ, Li D, Li WH, Shapiro AMJ, Rutter GA (2014) ADCY5 couples glucose to insulin secretion in human islets Diabetes. (in press) [DOI] [PMC free article] [PubMed]
- 149.Wagner R, Dudziak K, Herzberg-Schafer SA, Machicao F, Stefan N, Staiger H, Haring HU, Fritsche A. Glucose-raising genetic variants in MADD and ADCY5 impair conversion of proinsulin to insulin. PLoS ONE. 2011;6(8):e23639. doi: 10.1371/journal.pone.0023639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Mears D, Sheppard NF, Jr, Atwater I, Rojas E. Magnitude and modulation of pancreatic beta-cell gap junction electrical conductance in situ. J Membr Biol. 1995;146(2):163–176. doi: 10.1007/BF00238006. [DOI] [PubMed] [Google Scholar]
- 151.Lemaire K, Ravier MA, Schraenen A, Creemers JW, Van de Plas R, Granvik M, Van Lommel L, Waelkens E, Chimienti F, Rutter GA, Gilon P, in’t Veld PA, Schuit FC. Insulin crystallization depends on zinc transporter ZnT8 expression, but is not required for normal glucose homeostasis in mice. Proc Natl Acad Sci USA. 2009;106(35):14872–14877. doi: 10.1073/pnas.0906587106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Nicolson TJ, Bellomo EA, Wijesekara N, Loder MK, Baldwin JM, Gyulkhandanyan AV, Koshkin V, Tarasov AI, Carzaniga R, Kronenberger K, Taneja TK, da Silva Xavier G, Libert S, Froguel P, Scharfmann R, Stetsyuk V, Ravassard P, Parker H, Gribble FM, Reimann F, Sladek R, Hughes SJ, Johnson PR, Masseboeuf M, Burcelin R, Baldwin SA, Liu M, Lara-Lemus R, Arvan P, Schuit FC, Wheeler MB, Chimienti F, Rutter GA. Insulin storage and glucose homeostasis in mice null for the granule zinc transporter ZnT8 and studies of the type 2 diabetes-associated variants. Diabetes. 2009;58(9):2070–2083. doi: 10.2337/db09-0551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Wijesekara N, Dai FF, Hardy AB, Giglou PR, Bhattacharjee A, Koshkin V, Chimienti F, Gaisano HY, Rutter GA, Wheeler MB. Beta cell-specific ZnT8 deletion in mice causes marked defects in insulin processing, crystallisation and secretion. Diabetologia. 2010;53(8):1656–1668. doi: 10.1007/s00125-010-1733-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Rutter GA. Think zinc: new roles for zinc in the control of insulin secretion. Islets. 2010;2(1):49–50. doi: 10.4161/isl.2.1.10259. [DOI] [PubMed] [Google Scholar]
- 155.Tamaki M, Fujitani Y, Hara A, Uchida T, Tamura Y, Takeno K, Kawaguchi M, Watanabe T, Ogihara T, Fukunaka A, Shimizu T, Mita T, Kanazawa A, Imaizumi MO, Abe T, Kiyonari H, Hojyo S, Fukada T, Kawauchi T, Nagamatsu S, Hirano T, Kawamori R, Watada H. The diabetes-susceptible gene SLC30A8/ZnT8 regulates hepatic insulin clearance. J Clin Invest. 2013;123(10):4513–4524. doi: 10.1172/JCI68807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Flannick J, Thorleifsson G, Beer NL, Jacobs SB, Grarup N, Burtt NP, Mahajan A, Fuchsberger C, Atzmon G, Benediktsson R, Blangero J, Bowden DW, Brandslund I, Brosnan J, Burslem F, Chambers J, Cho YS, Christensen C, Douglas DA, Duggirala R, Dymek Z, Farjoun Y, Fennell T, Fontanillas P, Forsen T, Gabriel S, Glaser B, Gudbjartsson DF, Hanis C, Hansen T, Hreidarsson AB, Hveem K, Ingelsson E, Isomaa B, Johansson S, Jorgensen T, Jorgensen ME, Kathiresan S, Kong A, Kooner J, Kravic J, Laakso M, Lee JY, Lind L, Lindgren CM, Linneberg A, Masson G, Meitinger T, Mohlke KL, Molven A, Morris AP, Potluri S, Rauramaa R, Ribel-Madsen R, Richard AM, Rolph T, Salomaa V, Segre AV, Skarstrand H, Steinthorsdottir V, Stringham HM, Sulem P, Tai ES, Teo YY, Teslovich T, Thorsteinsdottir U, Trimmer JK, Tuomi T, Tuomilehto J, Vaziri-Sani F, Voight BF, Wilson JG, Boehnke M, McCarthy MI, Njolstad PR, Pedersen O, Groop L, Cox DR, Stefansson K, Altshuler D. Loss-of-function mutations in SLC30A8 protect against type 2 diabetes. Nat Genet. 2014;46(4):357–363. doi: 10.1038/ng.2915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Gerber PA, Bellomo EA, Hodson DJ, Meur G, Solomou A, Mitchell RK, Hollinshead M, Chimienti F, Bosco D, Hughes SJ, Johnson PR, Rutter GA. Hypoxia lowers SLC30A8/ZnT8 expression and free cytosolic Zn2+ in pancreatic beta cells. Diabetologia. 2014 doi: 10.1007/s00125-014-3266-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Sun Z, Zhang DQ, McMahon DG. Zinc modulation of hemi-gap-junction channel currents in retinal horizontal cells. J Neurophysiol. 2009;101(4):1774–1780. doi: 10.1152/jn.90581.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lurtz MM, Louis CF. Intracellular calcium regulation of connexin43. Am J Physiol Cell Physiol. 2007;293(6):C1806–C1813. doi: 10.1152/ajpcell.00630.2006. [DOI] [PubMed] [Google Scholar]
- 160.Bavamian S, Pontes H, Cancela J, Charollais A, Startchik S, Van de Ville D, Meda P. The intercellular synchronization of Ca2+ oscillations evaluates Cx36-dependent coupling. PLoS ONE. 2012;7(7):e41535. doi: 10.1371/journal.pone.0041535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Amisten S, Salehi A, Rorsman P, Jones PM, Persaud SJ. An atlas and functional analysis of G-protein coupled receptors in human islets of Langerhans. Pharmacol Ther. 2013;139(3):359–391. doi: 10.1016/j.pharmthera.2013.05.004. [DOI] [PubMed] [Google Scholar]
- 162.Zhou K, Pearson ER. Insights from genome-wide association studies of drug response. Annu Rev Pharmacol Toxicol. 2013;53:299–310. doi: 10.1146/annurev-pharmtox-011112-140237. [DOI] [PubMed] [Google Scholar]
- 163.Meda P, Michaels RL, Halban PA, Orci L, Sheridan JD. In vivo modulation of gap junctions and dye coupling between B-cells of the intact pancreatic islet. Diabetes. 1983;32(9):858–868. doi: 10.2337/diab.32.9.858. [DOI] [PubMed] [Google Scholar]
- 164.Donnelly LA, Doney ASF, Hattersley AT, Morris AD, Pearson ER. The effect of obesity on glycaemic response to metformin or sulphonylureas in type 2 diabetes. Diabet Med. 2006;23(2):128–133. doi: 10.1111/j.1464-5491.2005.01755.x. [DOI] [PubMed] [Google Scholar]
- 165.Kim S, Putrino D, Ghosh S, Brown EN. A Granger causality measure for point process models of ensemble neural spiking activity. PLoS Comput Biol. 2011;7(3):e1001110. doi: 10.1371/journal.pcbi.1001110. [DOI] [PMC free article] [PubMed] [Google Scholar]