Abstract
Cell-permeable phosphorescent probes enable the study of cell and tissue oxygenation, bioenergetics, metabolism, and pathological states such as stroke and hypoxia. A number of such probes have been described in recent years, the majority consisting of cationic small molecule and nanoparticle structures. While these probes continue to advance, adequate staining for the study of certain cell types using live imaging techniques remains elusive; this is particularly true for neural cells. Here we introduce novel probes for the analysis of neural cells and tissues: negatively charged poly(methyl methacrylate-co-methacrylic acid)-based nanoparticles impregnated with a phosphorescent Pt(II)-tetrakis(pentafluorophenyl)porphyrin (PtPFPP) dye (this form is referred to as PA1), and with an additional reference/antennae dye poly(9,9-diheptylfluorene-alt-9,9-di-p-tolyl-9H-fluorene) (this form is referred to as PA2). PA1 and PA2 are internalised by endocytosis, result in efficient staining in primary neurons, astrocytes, and PC12 cells and multi-cellular aggregates, and allow for the monitoring of local O2 levels on a time-resolved fluorescence plate reader and PLIM microscope. PA2 also efficiently stains rat brain slices and permits detailed O2 imaging experiments using both one and two-photon intensity-based modes and PLIM modes. Multiplexed analysis of embryonic rat brain slices reveals age-dependent staining patterns for PA2 and a highly heterogeneous distribution of O2 in tissues, which we relate to the localisation of specific progenitor cell populations. Overall, these anionic probes are useful for sensing O2 levels in various cells and tissues, particularly in neural cells, and facilitate high-resolution imaging of O2 in 3D tissue models.
Electronic supplementary material
The online version of this article (doi:10.1007/s00018-014-1673-5) contains supplementary material, which is available to authorized users.
Keywords: Biomaterials, Cell and tissue oxygen, Intracellular oxygen probe, Multiplexed detection, Nanosensors, Phosphorescence quenching microscopy, PLIM
Introduction
Optical bio-imaging has evolved as a powerful tool for studying biological processes on a cellular level, providing single cell and even single molecule resolution [1, 2]. However, targeted delivery, intracellular distribution and operational performance of many bio-imaging probes are still far from optimal, especially for live-cell imaging applications. Nanoparticle (NP) probes offer significant advantages including increased brightness, photostability and shielding of the indicator dye from unwanted interferences, but their chemical modification and successful intracellular penetration remain difficult to achieve [3]. NP probes often use complex endocytic delivery mechanisms that limit their specificity to a narrow range of cell and tissue types [4], have limited stability in biological media (e.g. resulting in aggregation or leaking of cargo [5]) and display batch to batch variability. In order to increase the cell permeability of NP probes they are generally small in size (<100 nm), positively charged and contain cell targeting groups located on their surface [4, 6–8].
Recently, the ability to sense and image molecular oxygen (O2) by phosphorescence quenching has shown significant advances. A number of NP probes based on Pt(II)-tetrakis(pentafluorophenyl)porphyrin (PtPFPP) and Ru(dpp)2+3 dyes have been described; these probes possess increased brightness, photostability and compatibility with different detection modalities including ratiometric intensity and lifetime imaging under one and two-photon excitation modes [9–14]. Successful results using such probes have been demonstrated with 2D and 3D cell and tissue models (multi-cellular aggregates, spheroids [15]), in vivo imaging [8, 16], ex vivo cultures [17–19], analysis of cellular O2 gradients [20], as well as hypoxia and cellular responses to drug treatment [15, 21, 22]. For applications involving gross anatomical features it is sometimes sufficient to stain the surface of cells and tissue samples, rather than deliver the probe into the cytosol or mitochondria [15, 23]. Alternatively, intracellular localisation of bio-imaging probes is beneficial for high-resolution O2 mapping of meso- and microscopic objects (i.e. individual cells, multi-cellular aggregates, tissue explants) and allows for advanced investigations into detailed cellular processes. For instance, there is currently a debate regarding whether significant intracellular O2 gradients exist in respiring cells and tissues, and the extent to which these molecular gradients may play regulatory roles [20, 24–28]. The dynamics of changing O2 levels are also important for a comprehensive understanding of the physiology, metabolism and bioenergetics of neural tissue [29–32]. Oxygenation of neural tissue models has traditionally been analysed with micro-electrodes or extracellular probes [33, 34], which provide limited spatial information regarding O2 levels. In current research, many model systems (e.g. models of stroke, neurodegenerative disorders, nervous system development, microcirculation, tumorigenesis, and drug delivery) are now shifting from 2D cell culture [35] towards 3D cell and tissue models (e.g. spheroids, organotypic slices, ganglions, organoids) [36–38] and in vivo models [14, 39, 40]; therefore, novel, high-resolution O2 imaging probes are becoming increasingly necessary. To date, cell-penetrating O2 probes have been largely used in “proof-of-principle” studies within specific model systems [7, 41–46], and their narrow range of cell specificity as well as general incompatibility with other models greatly limits their use [47–49]. Ultimately, very few O2 imaging probes are suitable for detailed physiological studies with neural cell and tissue models [16, 19, 50].
Here we describe two new cell-penetrating probes based on anionic NPs of acrylic co-polymer, poly(methyl methacrylate-co-methacrylic acid); these polyacrylate NP (PA) probes are highly suitable for imaging of neural cell and tissue models. We evaluated their cell specificity, transport mechanisms, and cytotoxicity, and found them to be superior in many aspects when compared with the cationic NP probes currently in use. Further, we demonstrated their analytical performance in O2 sensing and imaging experiments using various model systems, including 2D cultures of primary neural cells, multi-cellular aggregates (3D spheroids) exposed to metabolic stimulation and cultured organotypic brain slices. The relation between local O2 levels and development of neural progenitor cells in developing tissue slices was also examined, using multi-parametric imaging combining live and subsequently fixed samples.
Results and discussion
Design and characterisation of PA probes
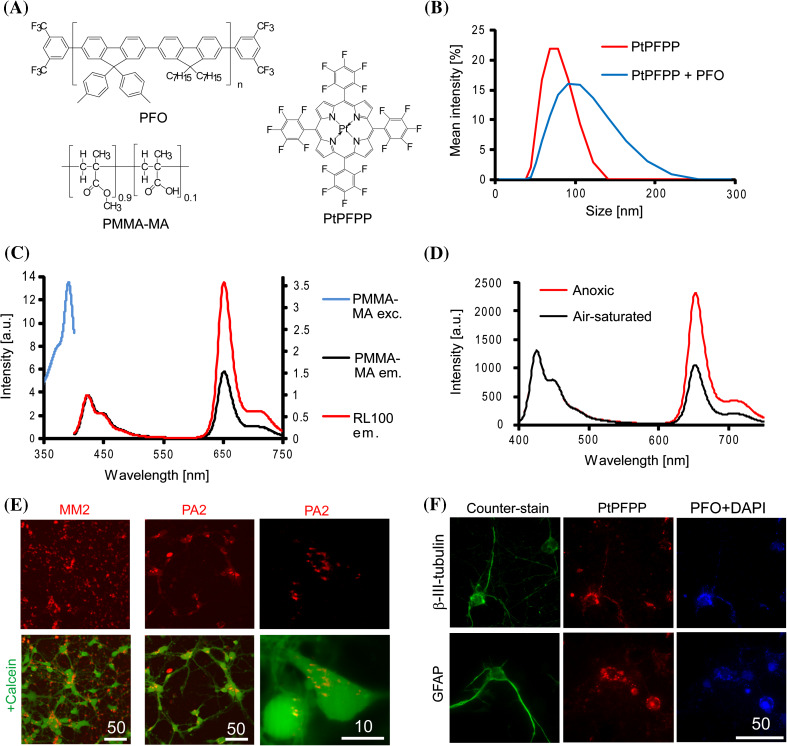
In previous work with the positively charged probes NanO2 and MM2, we observed poor intracellular staining of neuron-like PC12 and SH-SY5Y cell lines as well as neurospheres from primary cells [17, 51], with ‘patchy’ patterns that were not ideal for high-resolution O2 imaging such as confocal or light-sheet microscopy. We therefore searched for alternative polymers harbouring charged groups and allowing for easy fabrication of O2 sensors by nanoprecipitation techniques [52]. Although better cell penetration is usually achieved with cationic nanoparticle structures [4, 53], we found that anionic poly(methyl methacrylate-co-methacrylic acid) (PMMA-MA) had moderate oxygen permeability [54] and formed stable NPs in aqueous solution, leading to very promising results. Two NP structures (PA probes) were prepared from this polymer: PA1 containing a single phosphorescent dye PtPFPP, and PA2 with PtPFPP and a fluorescent antenna and reference conjugated polymer dye PFO (Fig. 1a). PA1 displayed a smaller particle size and narrower distribution than PA2, with Z AV = 72 nm, PDI = 0.041 and Z AV = 95 nm, PDI = 0.096 (Fig. 1b). For all probes and pure polymer the Z-potential was in the range of −18 to −35 mV (depending on the particle size, see “Methods” section). Spectral characteristics of the PA probes were similar to their cationic analogues NanO2 and MM2 [42, 55]. At O2 levels of 21 and 0 %, the emission lifetimes were 39 and 68 µs for PA1 and 31 and 70 µs for PA2, respectively (PBS, 37 °C); these levels are well suited for O2 sensing in the physiological range (0–200 µM). The PA2 probe showed efficient FRET and more intense emission of PtPFPP than PFO (Fig. 1c). It produced a ratiometric response to O2 comparable to MM2 (~2.5 times for I 0 %/I 21 %, Fig. 1d), and displayed similar imaging properties under two-photon excitation (see below).
Fig. 1.
Initial evaluation of PMMA-MA-based nanoprobes with neural cells. a Chemical structures of nanoparticle-forming components; b size distribution of the two probe samples obtained on a Zetasizer analyzer; c luminescence excitation and emission spectra of PA2 compared with MM2, measured at 0 % O2 at 37 °C. d Corrected emission spectra of PA2 at air saturation and anoxic conditions (λ exc 390 nm, 25 °C). e Confocal images of neural cells (E18, 6 DIV) stained with MM2 and PA2 (1 µg ml−1, 16 h, red) and co-localised with cell bodies by Calcein Green staining (1 µM, 0.5 h, green). Right: localisation of PA2 in mouse primary neurons (E16, 8 DIV) at higher magnification. Optical sections are 0.4 µm thick. f Wide-field images of neural cells stained with PA2 (1 µg ml−1, 16 h): PtPFPP (red), PFO and DAPI (blue) signals and counter-staining (green) with antibodies against β-III-tubulin (mature neurons) and GFAP (astrocytes). Scale bar unit is µm
Cell staining with PA probes
PA1 and PA2 were first evaluated with cultures of primary neural cells from rodent brain (rat and mouse cortical neurons of E16–18, 3–9 DIV), which represent mixed populations of developing neurons and astrocytes (Fig. 1e). When used at concentrations of 1–10 µg ml−1 in the culture medium, the PA probes stained cell bodies and neurites, co-localising with the cytosolic tracer Calcein Green and producing punctuated patterns with a small number of extracellular aggregates. The PA2 probe co-localised with immunohistochemically stained mature neurons (β-III-tubulin) and astrocytes (GFAP) (Fig. 1f), thus reflecting efficient staining efficiency of two dominant but diverse neural cell types. In contrast, the previously described MM2 and NanO2 showed negligible internalisation by neural cells and prominent formation of extracellular aggregates (Fig. 1e, S1).
We also tested fibroblast (MEF, Fig. S2), neuron-like PC12 cells and cortical neurons from mouse brain (E18) (not shown) and observed efficient intracellular accumulation of PA1 and PA2, with endosomal and lysosomal patterns of localisation, different from staining of mitochondria (TMRM). PC12 cells showed linear concentration dependence of staining up to 25 µg ml−1 (3, 20 h), while in MEF cells saturation was observed at 2.5 and 10 µg ml−1 (Fig. S2b–c). This suggests a slightly lower staining degree of fibroblast cells compared with NanO2 [55] and a different internalisation mechanisms for PA probes in different cell types. Nonetheless, all tested cells produced sufficient phosphorescent signals for O2 monitoring experiments (exceeded 30,000 cps threshold on the TR-F reader [55]). Total ATP assay revealed no significant toxicity of the PA2 probe in PC12, MEF cells, primary cortical or cerebellar granule neurons at concentrations 0–25 µg ml−1 and staining times ≤20 h (Fig. S3a–c). This is in agreement with reported low or no toxicity of PMMA-MA nanoparticles [56, 57].
By modifying the synthesis procedure (see “Methods” section), we also produced 135 nm NPs and tested the size dependence of NP internalisation on primary rat neural cells (Fig. 2a). The larger NPs showed less internalisation and a high number of aggregates outside the cells (indicated by blue arrows in Fig. 2a), while NPs <100 nm were localised largely intracellularly (as indicated by Calcein Green counter-staining). Staining patterns for PC12, MEF and mixed population of primary neurons and astrocytes suggested that internalisation occurred via complex mechanisms involving endocytosis. Indeed, under ATP depletion conditions, we observed a significant decrease of PA2 uptake by neural cells (Fig. 2b). Inhibitors of endocytic pathways (CPZ for clathrin-mediated endocytosis, EIPA for macropinocytosis and MβCD for lipid-raft/caveolae-dependent endocytosis) [58] also decreased probe uptake markedly, but not completely (Fig. 2c). We conclude that PA NPs utilise mixed endocytic, energy-dependent mechanisms of cell entry, similar to the cationic RL100-based probes [55].
Fig. 2.
Evaluation of mechanisms of cell entry for PA2 with cultured rat primary neural cells (E18, 6 DIV). a Effect of NP size on cellular uptake. Confocal images of PA NPs (2 µg ml−1, 16 h) and Calcein Green (1 µM, 0.5 h) probes, yellow arrows indicate intracellular localisation, blue-aggregates. b Effect of ATP depletion on cellular uptake of PA2. Cells were incubated for 1.5 h in glucose-free medium, then treated with oligomycin (10 µM, 0.5 h) and PA2 (10 µg ml−1, 3 h) and analysed. c Effect of inhibitors of endocytosis on cellular uptake of PA2. Cells were treated for 2 h with CPZ (50 µM), EIPA (50 µM) or MβCD (5 mM), then stained with PA2 (10 µg ml−1, 3 h) and analysed. Average intensity signals for multiple ROIs are shown. Scale bar in µm
Lifetime-based sensing of cellular O2
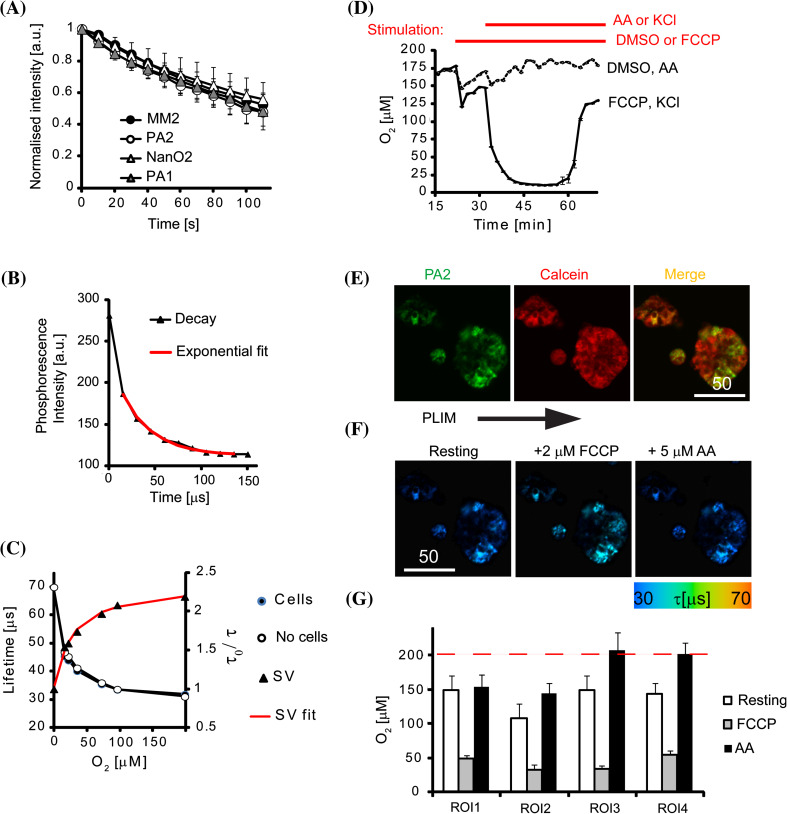
We found that the PA NPs demonstrated efficient cell penetration and photostability when compared with previously described cationic NP O2 probes (Fig. 3a); therefore, these PA probes are suitable for measurements of intracellular O2 in neuron-like cells on a TR-F plate reader (high throughput analysis of cell populations) and by phosphorescence lifetime imaging microscopy (PLIM). Using the TR-F plate reader, we calibrated the PA2 probe in solution and in stained mouse cortical neurons in which cellular respiration was blocked by treatment with antimycin A (AA), and exposed the cells to different levels of atmospheric O2 (Fig. 3b–c). The PA probes displayed non-linear Stern–Volmer relationships, similar to other reported O2 probes [10], which reflects the heterogeneous environment within the nanoparticle space [59, 60] (Fig. 3c). However, the calibrations were not affected by the local environment, and no drift of the lifetime signal was observed in stained cells cultured over periods of up to 36 h (not shown). This is important since such drifts would indicate leakage of the dye(s) or degradation of the NPs due to normal cellular processes (e.g. in lysosomes). Notably, both aqueous solution and “in cell” calibrations were identical, indicating that there was a minimal effect of the local environment on the O2 sensing (PA2 inside or outside the cells). Additionally, the calibration data fitted well with the “two-site” modified Stern–Volmer model [11, 59].
Fig. 3.
O2 monitoring experiments with neural cells and PA2. a Photobleaching under continuous illumination on a microscope in comparison with other NP probes (650 nm emission). b Example of phosphorescence intensity decay shown with single-exponential decay fitting curve (red). c O2 calibration and Stern–Volmer (SV) relationship produced with mouse cortical neurons (7 DIV, “cells”) and in solution (“no cells”) on a TR-F reader. Fitting of Stern–Volmer relationship accordingly to two-site model is shown in red (SV fit). d Oxygenation profiles of a confluent monolayer of mouse cortical neurons (7 DIV) stained with PA2 (2.5 µg ml−1; 16 h) and measured on a TR-F reader. After baseline stabilisation, cells were treated with FCCP (500 nM) or DMSO (mock), followed up KCl (50 mM) or AntimycinA (AA, 10 µM), as indicated by red lines (top of the graph). N = 3. e Single optical section of a PC12 cell aggregates stained with PA2 (green) and Calcein Green (red), and merged image. Scale bar in µm. f PLIM images of PC12 aggregates (shown above) at rest and after sequential treatment with FCCP and AA. g Corresponding O2 levels calculated for selected ROIs within the aggregates, N = 5. Dashed line shows air-saturated O2 level
We also monitored local oxygenation of a confluent monolayer of mouse cortical neurons stained with PA2 (Figs. 3c, S3d) and observed a basal O2 level of approximately 175 µM at rest. This level was markedly decreased upon mitochondrial uncoupling and subsequent depolarisation of the plasma membrane after application of FCCP and KCl, respectively (Fig. 3d), which are used in a two drug model with known mechanisms of action in this regard [24, 61]. In contrast, the mitochondrial inhibitor Antimycin A (AA) reverted cellular O2 to air-saturated levels (Fig. 3d). The magnitude and timescale of the responses demonstrate a moderate respiration rate for this cell type.
For demonstration of the PA2 probe performance in PLIM we used cultured PC12 cell aggregates, which possess active respiration and display local O2 gradients [19, 24]. Staining with PA2 overlapped with Calcein Green, thus confirming its intracellular accumulation (Fig. 3e). The aggregates located within cells showed rapid responses (2–4 min) to stimulation with FCCP and AA (Fig. 3f), similar to neurons cultured on microplates (Fig. 3d). Randomly selected ROIs (1–4) within the same aggregate showed different responses (Fig. 3f), with a stronger magnitude response corresponding to higher respiratory activity. Subsequently, AA brought back local deoxygenation to initial (resting) levels (Fig. 3g). These data demonstrate that O2 levels in neuron-like cells can be reliably measured with the PA2 probe in plate reader [24] and PLIM [19, 55] formats.
Analysis of cultured brain slices
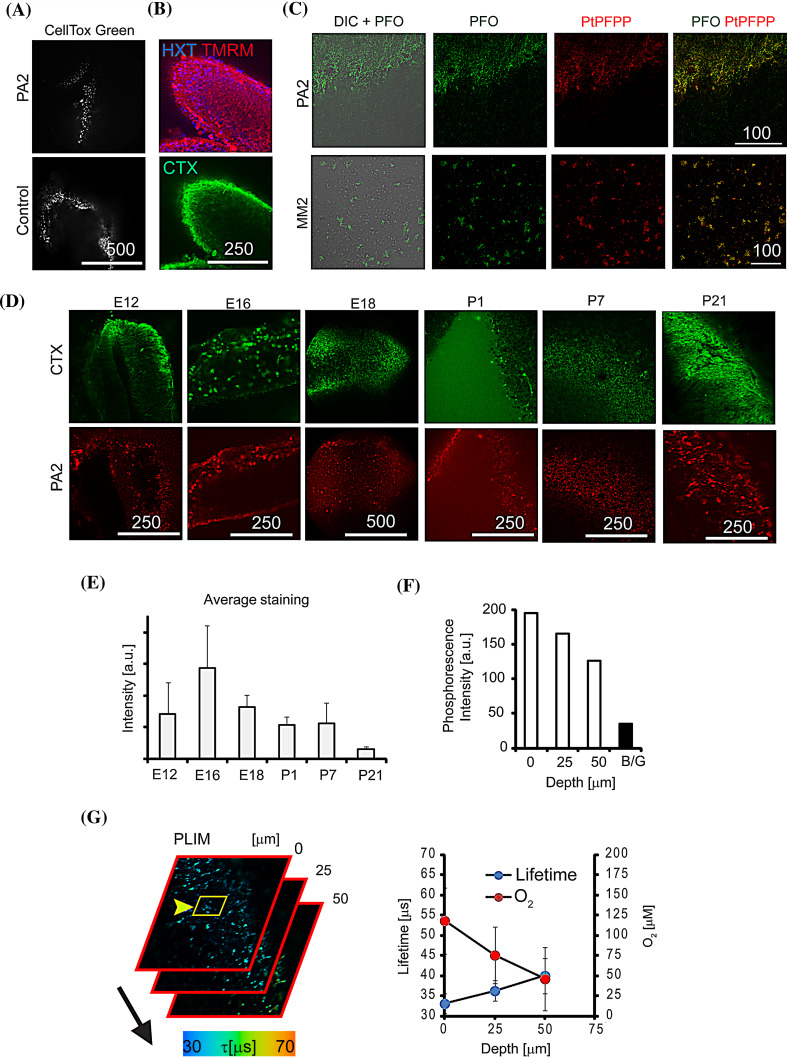
Two-dimensional neuronal cultures often lack the cyto-architectural context of 3D tissues and organs, and therefore have limited translational potential. We tested a common 3D tissue model of live organotypic rat brain slices (400 µm thick explants) maintained under ambient O2 [19, 62] with the PA2 probe. The presence of the PFO antenna in the PA2 probe allowed us to perform two-photon imaging at depths >100 µm. Both control and PA2-stained (25 µg ml−1 for 3–24 h) samples showed a comparable (or lower) degree of staining with the membrane integrity probe CellTox Green (Fig. 4a), indicating that the PA2 probe had no effect on cell viability. Slices cultured for 1–3 days also demonstrated positive staining with TMRM (functional mitochondria) and cholera toxin (neuronal cells), confirming the presence of a high number of viable cells in cultured slices (Fig. 4b). This was consistent with 2D culture data (Fig. S3a–c).
Fig. 4.
Staining of live rat cortical slices with PA2. a E16 slices cultured 3 DIV and stained with PA2 (25 µg ml−1, 3 h) and CellTox Green dye (0.05 %, 3 h). b Analysis of 3 DIV slices with live cell markers Hoechst 33342 (HXT, 2 µM), TMRM (20 nM) and CTX-Alexa 488 (1 ng ml−1). c Two-photon images of brain slices (P7) stained with MM2 and PA2 (100 µg ml−1, 24 h): 4 µm thick optical sections for PFO (420–460 nm) and PtPFPP (605–680 nm) dyes (excited at 780 nm). d One-photon confocal single optical sections showing staining of brain slices of different age with CTX and PA2 (25 µg ml−1, 3 h). e Average staining intensities calculated from (d). f, g One-photon O2 PLIM images of P21 slices stained with PA2, and imaged at different depths (0–50 µm). f Average phosphorescence intensities of PA2 deep in the tissue, depending on scanning depth. B/G: background counts. g Combined PLIM images and calculated profiles of lifetime and O2 concentration for selected ROI. Scale bar in µm
Next, we compared PA2 with the MM2 probe in imaging experiments in brain slices (P7 slices). Both probes were clearly visible but demonstrated different patterns of staining (Fig. 4c): MM2 probe showed marked aggregation and poor penetration into tissue, and the PA2 probe showed diffuse uniform staining across the tissue volume, which penetrated at least 50–100 µm into tissue (not shown). Staining for 3–4 h was sufficient for positive labelling with the PA2 probe and neuronal tracer Cholera toxin (CTX), while longer incubation gave no improvement (Fig. S4a). This short sample preparation time facilitates the analysis of ex vivo brain slices, particularly in conjunction with sensitive confocal PLIM systems [14, 17].
The developing brain is a highly heterogeneous structure with many different cell types and pronounced structural changes upon maturation. Therefore, to test the performance of the PA probe with diverse tissue cyto-architectures we examined rat brain cortical slices ranging in age from embryonic stages (E12) to adult (P21). We observed efficient staining with PA2 (3–4 h incubation) for all samples, however, postnatal tissue (especially P21) showed higher probe aggregation and reduced probe penetration (Fig. 4d). Quantitative analysis of ROIs free of aggregates confirmed improved staining of embryonic samples E12–E16 (Fig. 4e). In contrast, the MM2 probe produced mostly ‘patchy’ patterns in tissue of all ages (Fig. S4b). In-depth deoxygenation of stained tissue samples was analysed by confocal PLIM, by exciting the PA2 probe at 405 nm and collecting emission at 650 nm (Fig. 4f, g). We found that the PA2 staining was bright and photostable with a uniform tissue distribution, and produced high phosphorescence signals and accurate lifetime measurements from under 405 nm laser excitation (Fig. 4f). Postnatal (P21) samples showed more than twofold reduction in O2 across a depth of 50 µm, which reflects the diffusion limitations of O2 transport to actively respiring cells. Spatial heterogeneity across the 50 × 50 µm2 region was also high as evidenced from the calculated profiles: ±25 µΜ O2 (Fig. 4f, right).
Multiplexed analysis of development of neural progenitor cells
To further demonstrate potential applications of PA2, we carried out multiplexed analysis of cultured brain tissue using live cell imaging techniques combined with immunofluorescence. We transfected a specific population of progenitor cells in cultured embryonic brain slices (E12) using plasmid DNA where GFP expression is driven by a brain lipid-binding protein (BLBP) promoter (BLBP-GFP); this plasmid is used as a specific marker of developing radial glial cells [62, 63]. Slices were then cultured for 1–3 days in vitro (DIV). Subsequently, confocal microscopy confirmed that the distribution of GFP-positive cells co-localised with markers of neural progenitor cells (SOX2, nestin) and actively proliferating cells (Ki67) (Fig. 5a). This co-localisation gradually decreased with the age of slices in culture (Fig. 5b), indicating that active differentiation and developmental processes were ongoing in the tissue slices. Therefore, BLBP-GFP transfection provided visualisation of a population of developing neural progenitor cells in this 3D tissue model.
Fig. 5.
Analysis of O2 influence on development of BLBP-GFP-positive cells. a Visualisation of BLBP-GFP-positive, progenitor cells (SOX2) and proliferating cells (Ki67) in an organotypic brain slice (E12, 3 DIV) by immunofluorescence (IF). b Co-localisation of BLBP-GFP, SOX2 and Ki67 at 1 and 3 DIV showing that a large portion of the GFP expressing cells are actively dividing (Ki67+) and remain in an undifferentiated state (SOX2+). c Examples of images of regions with overlapping BLBP-GFP and PA2 distributions (intensity, lifetime and overlay) showing ROIs used for quantification. d Profiles of BLBP-GFP expression and corresponding O2 levels on 1 and 3 DIV, determined for such overlapping regions. Scale bar in µm
We then analysed the distribution of live neural progenitor cells in relation to local O2 levels. BLBP-GFP-transfected slices (1 or 3 DIV) were stained with the PA2 probe (3 h) and analysed by multi-parametric imaging and PLIM (Fig. 5c). We selected regions of tissue (20–50 µm2) with overlapping PA2 and BLBP-GFP staining (Fig. 5c, left panel) and compared 10 ROIs after 1 DIV (at this stage 25 % of GFP labelled cells were actively proliferating cells and 40 % of GFP labelled cells remained a progenitor population, i.e. SOX2-positive) and at 3 DIV (at this stage GFP labelled cells exhibit decreased proliferation and expression of progenitor cell markers). We found a large variation in O2 levels within slices, ranging 50–200 µM (Fig. 5b–d), however this range was similar at 1 DIV and 3 DIV. This suggests that the O2 requirements for development and differentiation of organotypic slices is heterogeneous, but is not necessarily dependent on an absolute change in O2 levels over this developmental time period; although the actively proliferating population of BLBP-GFP labelled cells was larger at 1 DIV, this did not correspond to an increased/decreased level of overall tissue oxygenation. Importantly, the range and average value of tissue oxygenation in the slices were unchanged after 3 DIV, indicating that the culture environment did not result in significant deterioration of tissue slices due to deoxygenation over this time period, further validating this 3D model system.
Although the range and average of O2 levels in slices did not differ over this time period, the high spatial heterogeneity of O2 within live tissue slices (Figs. 4f, 5d) could be due to the presence of micro-regions or cell types with different O2 demands. This is suggested by the correlation between O2 levels and the distribution of specific cell populations. For example, this can be seen in the two highlighted ROIs in Figure S5: ROI1 contains a region with GFP expressing cells that are Ki67-negative, indicating that they are of radial glial cell lineage but are no longer actively proliferating, and this specific region displayed high O2 levels (150–200 µM); in contrast, ROI2 contains no GFP labelled cells but does contain nestin-positive fibres and faintly positive Ki67 staining, indicating the presence of recently proliferating neural stem cells, and this region had twofold lower O2 values (Fig. S5). These findings suggest that meaningful relationships between specific cell populations and O2 levels can be determined using these PA probes, however further studies are needed to determine the full implications of changes in oxygenation in these, and other, cell types.
As with other fluorescence microscopy imaging methods, O2 imaging results can potentially be affected by measurement artefacts, such as: (1) non-ideal optical sectioning on one-photon confocal PLIM microscope due to limited tissue penetration of excitation light, scattering effects, limited photon “budget” available for precise calculation of phosphorescence lifetimes, and unwanted recording of out-of-focus phosphorescence [64–66]; (2) uneven distribution of the NP probes across the tissue volume can potentially affect the imaging precision [49] (3) artifacts related to photoinduced O2 consumption [67] and others [15]. However, PLIM method will be among the methods that are most minimally affected by these factors and its reliability in O2 measurements has been demonstrated in many physiological studies [26, 40, 68, 69].
The functional imaging of O2 levels in relation to specific cell populations in living brain tissue in this study represent intriguing “proof of concept” results and demonstrates the potential to investigate dynamic changes in O2 levels in relation to ongoing developmental processes occurring in on a cellular as well as a whole tissue level. Future studies optimizing both the distribution of O2 probes and live detection of specific cell types in organotypic tissue slices are required to allow for the specific correlation of O2 levels with neuronal and neuroglial activity in living and developing neural tissues.
Conclusions
The PA1 and PA2 nanoparticle probes (<100 nm) based on anionic PMMA-MA polymer demonstrate good staining efficiency and intracellular penetration across a range of cell types and neural tissues. We attribute this to their lower aggregation and non-specific adsorption [70] and more specific interaction with membranes of neural cells. Similar to other nanoparticles [4, 53], their internalisation was strongly inhibited by endocytosis inhibitors and ATP depletion, suggesting their utilization of energy-dependent cell entry (Figs. 1, 2, S2). These probes showed lysosomal patterns of localisation and photo-physical properties similar to cationic NP probes [42, 55] including high brightness and photostability provided by PtPFPP, two-photon antenna and reference PFO dye, and stable and reproducible O2 calibration (Fig. 3). Therefore, these PA probes offer significant advantages as they allow for similar applications but on a broader/different range of cell and tissue models which include primary neural cell culture, as well as 3D models such as cell aggregates and brain tissue slices. In contrast with recently developed red and infra-red molecular probes [19, 71] PA2 and related nanoparticles provide multi-modal imaging capability with efficient two-photon excitation, this can be very beneficial for ex vivo and in vivo models.
Monitoring of O2 in 2D cultures produced metabolic responses similar to those observed with existing probes; however, with 3D models (aggregates and tissue explants) the PA2 probe resulted in improved distribution across tissue volumes and ultimately better imaging performance. Probe penetration into brain tissue (Fig. 4) was comparable to small molecule conjugates (e.g. Pt-Glc) [19], with the added advantage of increased brightness and the option of two-photon and ratiometric detection. Using the PA2 probe, BLBP-GFP plasmid DNA transfection to identify specific cell populations, live-cell imaging, and immunofluorescence techniques (Fig. 5) we performed multi-parametric functional analysis, relating GFP labelled developing progenitor cell populations with local O2 levels in cultured embryonic brain tissue over a 3-day developmental period. Heterogeneity of tissue oxygenation was shown and its possible effects on various cell types discussed-highlighting the potential applications for future studies with these PA probes. The results of this study and subsequent advancement in the knowledge of cell-penetrating behaviour of anionic PMMA-MA nanoparticles can be utilised for designing future sensor probes and drug delivery systems for a variety of tissues, importantly including neural tissue [57].
Methods
Materials
Platinum(II) meso-tetra(pentafluorophenyl)porphine (PtPFPP) was from Frontier Scientific (Inochem Ltd, Lancashire UK), poly(methyl methacrylate-co-methacrylic acid), 9:1 (PMMA-MA) was from Polysciences (Warrington, PA, USA), poly(9,9-diheptylfluorene-alt-9,9-di-p-tolyl-9H-fluorene) was prepared as described previously [42]. MitoImage-NanO2 and MM2 probes were from Luxcel Biosciences (Cork, Ireland). Secondary antibodies labelled with Alexa Fluor 488, 555, 594, 633, recombinant cholera toxin, subunit B-Alexa Fluor 488 conjugate (CTX), B27 serum-free supplement, Calcein Green AM, tetramethylrhodamine methyl ester (TMRM) and Pro-long Gold anti-fade mounting medium were from Life Technologies (BioSciences, Dublin, Ireland). Goat anti-SOX2 antibody was from Abcam (Cambridge, UK). Rat anti-Ki67 antibody and Fluoromount-G mounting medium were from eBiosciences (San Diego, CA). Viability Assay kits CellTiter-Glo™ and CellTox™ Green were from Promega (MyBio, Ireland). Mouse anti-nestin antibody was from Millipore (Cork, Ireland). Anti-GFAP (G-A-5) antibody, bis-Benzimide Hoechst 33342 and all the other reagents were from Sigma-Aldrich (Dublin, Ireland).
Standard cell culture grade plasticware was from Sarstedt (Wexford, Ireland) and Corning (VWR, Ireland), glass bottom mini-dishes were from MatTek (Ashland, USA), glass bottom multiwell slides were from Ibidi (Martinsried, Germany). Polystyrene scaffold membranes Alvetex™ were from Reinnervate (Amsbio, UK). Plasmid DNA BLBP-GFP [63] was kindly provided by Dr. E.S. Anton (University of North Carolina).
Preparation of PMMA-MA nanoparticles
PA1: 200 mg of PMMA-MA and 3 mg of PtPFPP were dissolved in 45 mL of tetrahydrofuran and the solution was diluted to 100 mL with acetone. The solution was transferred into a 1 L glass beaker and 600 mL of deionised water was added within 3–5 s under vigorous stirring. Using a rotary evaporator, organic phase was removed and the nanoparticles were concentrated to 10 mg ml−1. The solution was cleared from aggregates by centrifugation and then stored at 4 °C.
PA2: nanoparticles were prepared similarly, by using 2.4 mg of PtPFPP and 20 mg of poly(9,9-diheptylfluorene-alt-9,9-di-p-tolyl-9H-fluorene). Particle size and charge were determined on a particle size analyzer Zetasizer Nano ZS (Malvern Instruments, Germany), UV–Vis and luminescence spectra as described before in [42, 52]. Phosphorescence lifetimes were measured in air-saturated and deoxygenated (5 mg ml−1 KH2PO4, 5 mg ml−1 Na2SO3) conditions. Large size NPs bearing PtPFPP and PFO dyes (135 nm, −24.5 mV) were produced when water was added with slower speed.
Cell and primary neural cultures
All the procedures with animals were performed under a licence issued by the Irish Government Department of Health and in accordance with the EU Directive 2010/63/EU. Embryonic tissue from time mated Sprague–Dawley rats (Biological Services Unit, University College Cork, Ireland) at embryonic day (E)12 to postnatal day (P)21 were used.
Mouse embryonic fibroblast (MEF) and rat pheochromocytoma (PC12) cells from ATCC (Manassas, VA, USA) were handled as described before [51, 72]. Mouse primary cortical (E16) and cerebellar granule neurons (P7) were obtained as described before [73]. The culture of rat primary cortical cells (E18) was prepared and handled under differentiation conditions (DMEM F12 Ham medium, supplemented with 2 % B27, 1 % FBS, 1 % penicillin–streptomycin solution) for 5–7 DIV, adhered onto poly-d-lysine-coated glass or plastic dishes as described before at 75–100 % confluence [17]. The staining with probes was achieved by incubation at indicated concentration (2.5–25 µg ml−1) in regular culture medium for 3–24 h, followed by 2–3 washes.
PC12 cell aggregates were grown in RPMI-1640 medium supplemented with 10 % horse serum (HS), 5 % FBS, 10 mM HEPES-Na, pH 7.2 and penicillin–streptomycin. For cell staining kinetics, leakage and ATP assays, MEF and PC12 cells were seeded on collagen IV-coated 96-well plates and cultured for 48 h to reach 75–100 % confluence. Metabolic stimulation was performed in 35 mm Cell+ mini-dishes (Sarstedt) in 2 mL of Phenol Red-free DMEM supplemented with NGF (100 ng ml−1) at 37 °C and 20 % O2, adding 10× stock solutions of drugs (1/10 of the volume) to the samples.
In the studies of cell entry mechanism, cells grown on multi-well coverslips (Ibidi) were pre-treated with inhibitors of endocytosis (as indicated), incubated with PA2 (10 µg ml−1, 3 h) and analysed on a wide-field microscope [17]. ATP depletion was performed as described before [72]. Intensity signals (collected under 390 nm LED excitation and 635–675 nm emission filter, background corrected) from 6 to 7 randomly selected ROI were used for quantification of drug effects.
Brain slice culture
Tissue slices were obtained and handled as described previously [19]. Pregnant rats (E12–E18) were anesthetized with isoflurane followed by decapitation, embryos were collected by laparotomy and placed into ice cold sterile HBSS. For postnatal ages (P1–P21), rat pups were anesthetized with isoflurane followed by decapitation and the brain was dissected out. Tissues were embedded in 4 % low melting agarose (prepared in PBS, supplemented with 27 mM glucose). Transverse sections (400 µm) were sliced in ice cold L15 dissecting media using a vibratome (VT1200, Leica Microsystems). Using the interface method [74] slices were positioned on Millicell inserts (0.4 µm, 30 mm diameter, Millipore) or pre-wet polystyrene scaffold membranes Alvetex™ (Reinnervate, Amsbio, UK) in phenol red-free DMEM supplemented with 25 % HBSS, 10 % FBS, 1 % penicillin–streptomycin, 10 mM Glc, 20 mM HEPES-Na, pH 7.2 (“Slice medium”) at 20 % O2, 5 % CO2, 37 °C in a humidified incubator. Electroporation with BLBP-GFP plasmid [62, 63] was carried out directly on culture inserts using a pair of gold-plated “genepaddle” electrodes on BTX ECM 830 Square Wave Electroporator (Harvard Apparatus, UK), using 2 µl of 1.0 µg µl−1 DNA under previously optimised settings (60 V, 5 pulses at 50 ms pulse duration, 950 ms interval) [62, 75]. 3–5 h after dissection slices were analysed or cultured for 1–3 DIV. Tissue staining was achieved by incubation with the O2 probe for 3–24 h at 25–100 µg ml−1, or with CTX (1 ng ml−1) in Slice medium, followed by one wash.
Immunofluorescence
Brain slices were fixed in 4 % paraformaldehyde solution (4 h, 4 °C), washed with PBS (0.5 h, 5 times), blocked in 10 % HS supplemented with 0.4 % Triton X100 in PBS (1 h, room temp.), incubated with primary antibodies diluted (1:500) in 2.5 % HS, 0.4 % Triton X100 in PBS (48 h, 4 °C), washed with PBS, incubated with appropriate Alexa Fluor-conjugated secondary antibodies (18 h, 4 °C). They were then washed in PBS, placed between two coverslips with a 400 µm thick slice of agarose to act as a spacer, mounted in Fluoromount-G medium (eBiosciences) and imaged on a confocal laser-scanning microscope Olympus FV1000 [42]. Maximum intensity projections of an imaged z-stack were processed in Olympus Fluoview (FV10-ASW 4.0 Viewer) and brightness and contrast were adjusted accordingly in Adobe Photoshop and Illustrator software.
Two-photon microscopy
This was performed on an upright microscope Olympus FV1000 MPE equipped with Mai Tai DeepSee Ti:Sapphire laser (Newport Corporation, CA), XPLN 25x NA:1.05 water immersion objective and temperature (temperature set at 37 °C)/CO2 (5 %) control, using excitation at 770 nm (2 % laser power) for PA2 and MM2 probes [17].
One-photon confocal PLIM
This was performed on a system based on an upright fluorescent microscope Axio Examiner Z1 (Zeiss UK) equipped with 20× NA:1.0 W-Plan-Apochromat and 40× NA:1.1 LD-C Apochromat W Korr. objectives, heated stage (temperature set at 37 °C) with motorized Z-axis control, DCS-120 confocal scanner (Becker and Hickl), R10467U-40 photon counting detector (Hamamatsu Photonics K·K.) and TCSPC hardware (Becker and Hickl) [17]. Phosphorescent probe-stained cells were excited with a 405 nm picosecond diode laser BDL-SMC (Becker and Hickl) and emission was collected at 635–675 nm. Fluorescent dyes Calcein Green, Alexa Fluor 488, BLBP-GFP, CellTox Green and CTX were excited with a picosecond supercontinuum laser SC400-4 (Fianium, UK) at 488 nm with emission collected at 512–536 nm; TMRM was excited at 540 nm and emission collected at 565–605 nm; LysoTracker Red and MitoTracker Red were excited at 590 nm and emission collected at 635–675 nm. DIC images were recorded with D5100 digital camera (Nikon) connected to the microscope. Phosphorescence lifetimes were calculated in SPCImage software (Becker and Hickl) using mono-exponential decay fits. 2D matrices of lifetime data for each measurement were then correlated with intensity and DIC images, and selected ROIs were converted in O2 concentration using the following calibration function produced on TR-F microplate reader Victor2: [O2] = 87,270.1 × exp(−τ/5.07006) (mono-exponential fitting) or [O2] = (0.57575/(τ/69.86 − 1 + 0.57575) − 1)/0.0861 (two-site model, r 2 = 0.997, Ksv = 0.0861 µM−1, τ 0 = 69.86 µs).
Plate reader measurements
These were performed on Victor2 (Perkin Elmer) TR-F plate reader placed into hypoxia Glove Box (Coy lab products, Michigan, USA) as described before [42]. Briefly, cells stained with PA2 probe treated with antimycin A (10 µM) were exposed to different levels of atmospheric O2 at 37 °C, and measured lifetime values were used to produce the calibration function by fitting. Calibration without cells was performed in PBS.
Cell viability and toxicity assessment
This was performed by measuring changes in total cellular ATP in 2D cell culture, or by staining brain slices with live cell markers (TMRM, CTX, HXT). In the ATP assay, MEF, PC12 or primary neural cells grown on 96-well plates were stained with 0–25 µg ml−1 of PA2 for 3–24 h, washed with fresh medium, lysed with CellTiter-Glo reagent (Promega) and analysed for total luminescence. Membrane integrity was assessed by staining with CellTox Green (Promega) reagent [17]. Briefly, slices were incubated with reagent (0.05 %), washed with medium, imaged on the confocal microscope. Stained nuclei were counted and compared with control.
Data assessment
Plate reader data represent averaged values with error bars (standard deviation) for >6 replicates. Cell loading studies were performed on wide-field microscope in triplicate, averaging intensity values from 6 to 10 randomly selected ROI. Experiments with brain slices were performed in duplicate, with analysis of 3–5 independent ROI. Phosphorescence lifetime images (2D matrices) were generated in SPCImage software (Becker and Hickl) by single-exponential fitting. They were converted into ASCII format and processed in Microsoft Excel to produce O2 concentration values using calibration function. For BLBP-GFP and O2 correlation, photon counts per pixel for different spectral channels were compared in MS Excel software, then co-localising regions were selected and analysed. To ensure consistency, all the experiments were repeated at least for 2–3 times, as indicated in figure descriptions.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
This work was supported by the Science Foundation Ireland, Grant 12/RC/2276, the European Commission FP7 Program, grant FP7-HEALTH-2012-INNOVATION-304842-2, the Irish Research Council for Science, Engineering and Technology, the Health Research Board, the Programme for Research at Third Level Institutions Cycle 4 and Marie Curie IAPP Oxy-Sense (No. 230641). We thank T. Foley and Dr. Y. Nolan (Department of Anatomy and Neuroscience, UCC) for the help with primary neuronal cultures and Dr. Heiko Dussmann (Royal College of Surgeons in Ireland, Dublin) for help with microscopy imaging.
Abbreviations
- 3D
Three-dimensional
- BLBP
Brain lipid-binding protein
- CPZ
Chlorpromazine
- CTX
Cholera toxin, subunit B
- DIV
Days in vitro
- DMF
N,N-dimethylformamide
- DMSO
Dimethyl sulfoxide
- EIPA
5-(N-ethyl-N-isopropyl)amiloride
- GFP
Green fluorescent protein
- HBSS
Hanks balanced salt solution
- HXT
Hoechst 33342
- iO2
Intracellular O2
- MβCD
Methyl-β-cyclodextrin
- NP
Nanoparticles
- PA
Polyacrylate NP
- PBS
Phosphate buffered saline
- PDL
Poly-d-lysine
- PDT
Photodynamic therapy
- PLIM
Phosphorescence lifetime imaging microscopy
- PMMA-MA
Poly(methyl methacrylate-co-methacrylic acid
- PFO
Poly(9,9-diheptylfluorene-alt-9,9-di-p-tolyl-9H-fluorene)
- PtPFPP
Pt(II)-tetrakis(pentafluorophenyl)porphine
- ROI
Region of interest
- RT
Room temperature
- TBST
Tris-buffered saline, tween 20
- TCSPC
Time-correlated single photon counting
- TR-F
Time-resolved fluorescence
References
- 1.Kobayashi H, Ogawa M, Alford R, Choyke PL, Urano Y. New strategies for fluorescent probe design in medical diagnostic imaging. Chem Rev. 2009;110(5):2620–2640. doi: 10.1021/cr900263j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schäferling M. The art of fluorescence imaging with chemical sensors. Angew Chem Int Ed. 2012;51(15):3532–3554. doi: 10.1002/anie.201105459. [DOI] [PubMed] [Google Scholar]
- 3.Algar WR, Prasuhn DE, Stewart MH, Jennings TL, Blanco-Canosa JB, Dawson PE, Medintz IL. The controlled display of biomolecules on nanoparticles: a challenge suited to bioorthogonal chemistry. Bioconjug Chem. 2011;22(5):825–858. doi: 10.1021/bc200065z. [DOI] [PubMed] [Google Scholar]
- 4.Canton I, Battaglia G. Endocytosis at the nanoscale. Chem Soc Rev. 2012;41(7):2718–2739. doi: 10.1039/c2cs15309b. [DOI] [PubMed] [Google Scholar]
- 5.Soenen SJ, Himmelreich U, Nuytten N, Pisanic TR, Ferrari A, De Cuyper M. Intracellular nanoparticle coating stability determines nanoparticle diagnostics efficacy and cell functionality. Small. 2010;6(19):2136–2145. doi: 10.1002/smll.201000763. [DOI] [PubMed] [Google Scholar]
- 6.Karamchand L, Kim G, Wang S, Hah HJ, Ray A, Jiddou R, Koo Lee Y-E, Philbert MA, Kopelman R. Modulation of hydrogel nanoparticle intracellular trafficking by multivalent surface engineering with tumor targeting peptide. Nanoscale. 2013;5(21):10327–10344. doi: 10.1039/c3nr00908d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Koo Lee Y-E, Ulbrich EE, Kim G, Hah H, Strollo C, Fan W, Gurjar R, Koo S, Kopelman R. Near infrared luminescent oxygen nanosensors with nanoparticle matrix tailored sensitivity. Anal Chem. 2010;82(20):8446–8455. doi: 10.1021/ac1015358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Napp J, Behnke T, Fischer L, Würth C, Wottawa M, Katschinski DM, Alves F, Resch-Genger U, Schäferling M. Targeted luminescent near-infrared polymer-nanoprobes for in vivo imaging of tumor hypoxia. Anal Chem. 2011;83(23):9039–9046. doi: 10.1021/ac201870b. [DOI] [PubMed] [Google Scholar]
- 9.Xiang H, Cheng J, Ma X, Zhou X, Chruma JJ. Near-infrared phosphorescence: materials and applications. Chem Soc Rev. 2013 doi: 10.1039/c3cs60029g. [DOI] [PubMed] [Google Scholar]
- 10.Dmitriev RI, Papkovsky DB. Optical probes and techniques for O2 measurement in live cells and tissue. Cell Mol Life Sci. 2012;69(12):2025–2039. doi: 10.1007/s00018-011-0914-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Quaranta M, Borisov SM, Klimant I. Indicators for optical oxygen sensors. Bioanal Rev. 2012;4(2–4):115–157. doi: 10.1007/s12566-012-0032-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ruggi A, van Leeuwen FW, Velders AH. Interaction of dioxygen with the electronic excited state of Ir(III) and Ru (II) complexes: principles and biomedical applications. Coord Chem Rev. 2011;255(21):2542–2554. doi: 10.1016/j.ccr.2011.05.012. [DOI] [Google Scholar]
- 13.Lee Y-EK, Kopelman R, Smith R. Nanoparticle PEBBLE sensors in live cells and in vivo. Annu Rev Anal Chem (Palo Alto, Calif) 2009;2:57. doi: 10.1146/annurev.anchem.1.031207.112823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Devor A, Sakadžić S, Yaseen MA, Roussakis E, Tian P, Slovin H, Vanzetta I, Teng I, Saisan PA, Sinks LE. Optical imaging of neocortical dynamics. Berlin: Springer; 2014. Functional imaging of cerebral oxygenation with intrinsic optical contrast and phosphorescent probes; pp. 225–253. [Google Scholar]
- 15.Papkovsky DB, Dmitriev RI. Biological detection by optical oxygen sensing. Chem Soc Rev. 2013;42(22):8700–8732. doi: 10.1039/c3cs60131e. [DOI] [PubMed] [Google Scholar]
- 16.Tsytsarev V, Arakawa H, Borisov S, Pumbo E, Erzurumlu RS, Papkovsky DB. In vivo imaging of brain metabolism activity using a phosphorescent oxygen-sensitive probe. J Neurosci Methods. 2013;216(2):146–151. doi: 10.1016/j.jneumeth.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dmitriev RI, Zhdanov AV, Nolan YM, Papkovsky DB. Imaging of neurosphere oxygenation with phosphorescent probes. Biomaterials. 2013;34(37):9307–9317. doi: 10.1016/j.biomaterials.2013.08.065. [DOI] [PubMed] [Google Scholar]
- 18.Lambrechts D, Roeffaers M, Kerckhofs G, Roberts SJ, Hofkens J, Van de Putte T, Van Oosterwyck H, Schrooten J. Fluorescent oxygen sensitive microbead incorporation for measuring oxygen tension in cell aggregates. Biomaterials. 2013;34(4):922–929. doi: 10.1016/j.biomaterials.2012.10.019. [DOI] [PubMed] [Google Scholar]
- 19.Dmitriev RI, Kondrashina AV, Koren K, Klimant I, Zhdanov AV, Pakan JMP, McDermott KW, Papkovsky DB. Small molecule phosphorescent probes for O2 imaging in 3D tissue models. Biomater Sci. 2014;2:853–866. doi: 10.1039/c3bm60272a. [DOI] [PubMed] [Google Scholar]
- 20.Dmitriev RI, Zhdanov AV, Jasionek G, Papkovsky DB. Assessment of cellular oxygen gradients with a panel of phosphorescent oxygen-sensitive probes. Anal Chem. 2012;84(6):2930–2938. doi: 10.1021/ac3000144. [DOI] [PubMed] [Google Scholar]
- 21.Zhdanov AV, Dmitriev RI, Golubeva AV, Gavrilova SA, Papkovsky DB. Chronic hypoxia leads to a glycolytic phenotype and suppressed HIF-2 signaling in PC12 cells. Biochim Biophys Acta Gen Subj. 1830;6:3553–3569. doi: 10.1016/j.bbagen.2013.02.016. [DOI] [PubMed] [Google Scholar]
- 22.Zhdanov AV, Waters AH, Golubeva AV, Dmitriev RI, Papkovsky DB. Availability of the key metabolic substrates dictates the respiratory response of cancer cells to the mitochondrial uncoupling. Biochim Biophys Acta Bioenerg. 1837;1:51–62. doi: 10.1016/j.bbabio.2013.07.008. [DOI] [PubMed] [Google Scholar]
- 23.Apreleva SV, Wilson DF, Vinogradov SA. Tomographic imaging of oxygen by phosphorescence lifetime. Appl Opt. 2006;45(33):8547–8559. doi: 10.1364/AO.45.008547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhdanov AV, Ogurtsov VI, Taylor CT, Papkovsky DB. Monitoring of cell oxygenation and responses to metabolic stimulation by intracellular oxygen sensing technique. Integr Biol. 2010;2(9):443–451. doi: 10.1039/c0ib00021c. [DOI] [PubMed] [Google Scholar]
- 25.Takahashi E, Asano K. Mitochondrial respiratory control can compensate for intracellular O2 gradients in cardiomyocytes at low Po 2. Am J Physiol Heart Circ Physiol. 2002;283(3):H871–H878. doi: 10.1152/ajpheart.00162.2002. [DOI] [PubMed] [Google Scholar]
- 26.Finikova OS, Lebedev AY, Aprelev A, Troxler T, Gao F, Garnacho C, Muro S, Hochstrasser RM, Vinogradov SA. Oxygen microscopy by two-photon-excited phosphorescence. ChemPhysChem. 2008;9(12):1673–1679. doi: 10.1002/cphc.200800296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mik EG, Stap J, Sinaasappel M, Beek JF, Aten JA, van Leeuwen TG, Ince C. Mitochondrial PO2 measured by delayed fluorescence of endogenous protoporphyrin IX. Nat Methods. 2006;3(11):939–945. doi: 10.1038/nmeth940. [DOI] [PubMed] [Google Scholar]
- 28.Clanton T, Hogan M, Gladden L. Regulation of cellular gas exchange, oxygen sensing, and metabolic control. Compr Physiol. 2013 doi: 10.1002/cphy.c120030. [DOI] [PubMed] [Google Scholar]
- 29.Erecińska M, Silver IA. Tissue oxygen tension and brain sensitivity to hypoxia. Respir Physiol. 2001;128(3):263–276. doi: 10.1016/S0034-5687(01)00306-1. [DOI] [PubMed] [Google Scholar]
- 30.Mohyeldin A, Garzón-Muvdi T, Quiñones-Hinojosa A. Oxygen in stem cell biology: a critical component of the stem cell niche. Cell Stem Cell. 2010;7(2):150–161. doi: 10.1016/j.stem.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 31.Toussaint O, Weemaels G, Debacq-Chainiaux F, Scharffetter-Kochanek K, Wlaschek M. Artefactual effects of oxygen on cell culture models of cellular senescence and stem cell biology. J Cell Physiol. 2011;226(2):315–321. doi: 10.1002/jcp.22416. [DOI] [PubMed] [Google Scholar]
- 32.Semenza GL. Regulation of oxygen homeostasis by hypoxia-inducible factor 1. Physiology. 2009;24(2):97–106. doi: 10.1152/physiol.00045.2008. [DOI] [PubMed] [Google Scholar]
- 33.McHugh SB, Fillenz M, Lowry JP, Rawlins JNP, Bannerman DM. Brain tissue oxygen amperometry in behaving rats demonstrates functional dissociation of dorsal and ventral hippocampus during spatial processing and anxiety. Eur J Neurosci. 2011;33(2):322–337. doi: 10.1111/j.1460-9568.2010.07497.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Choi SW, Gerencser AA, Ng R, Flynn JM, Melov S, Danielson SR, Gibson BW, Nicholls DG, Bredesen DE, Brand MD. No consistent bioenergetic defects in presynaptic nerve terminals isolated from mouse models of Alzheimer’s disease. J Neurosci. 2012;32(47):16775–16784. doi: 10.1523/JNEUROSCI.2414-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nicholls DG, Johnson-Cadwell L, Vesce S, Jekabsons M, Yadava N. Bioenergetics of mitochondria in cultured neurons and their role in glutamate excitotoxicity. J Neurosci Res. 2007;85(15):3206–3212. doi: 10.1002/jnr.21290. [DOI] [PubMed] [Google Scholar]
- 36.Lancaster MA, Renner M, Martin C-A, Wenzel D, Bicknell LS, Hurles ME, Homfray T, Penninger JM, Jackson AP, Knoblich JA. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501:373–379. doi: 10.1038/nature12517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Noraberg J, Kristensen BW, Zimmer J. Markers for neuronal degeneration in organotypic slice cultures. Brain Res Protoc. 1999;3(3):278–290. doi: 10.1016/S1385-299X(98)00050-6. [DOI] [PubMed] [Google Scholar]
- 38.Zimmer J, Kristensen BW, Jakobsen B, Noraberg J. Excitatory amino acid neurotoxicity and modulation of glutamate receptor expression in organotypic brain slice cultures. Amino Acids. 2000;19(1):7–21. doi: 10.1007/s007260070029. [DOI] [PubMed] [Google Scholar]
- 39.Kazmi S, Salvaggio AJ, Estrada AD, Hemati MA, Shaydyuk NK, Roussakis E, Jones TA, Vinogradov SA, Dunn AK. Three-dimensional mapping of oxygen tension in cortical arterioles before and after occlusion. Biomed Optics Express. 2013;4(7):1061–1073. doi: 10.1364/BOE.4.001061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lecoq J, Parpaleix A, Roussakis E, Ducros M, Houssen YG, Vinogradov SA, Charpak S. Simultaneous two-photon imaging of oxygen and blood flow in deep cerebral vessels. Nat Med. 2011;17(7):893–898. doi: 10.1038/nm.2394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wolfbeis OS. Editorial: probes, sensors, and labels: Why is real progress slow? Angew Chem Int Ed. 2013;52(38):9864–9865. doi: 10.1002/anie.201305915. [DOI] [PubMed] [Google Scholar]
- 42.Kondrashina AV, Dmitriev RI, Borisov SM, Klimant I, O’Brien I, Nolan YM, Zhdanov AV, Papkovsky DB. A phosphorescent nanoparticle-based probe for sensing and imaging of (Intra) cellular oxygen in multiple detection modalities. Adv Funct Mater. 2012;22(23):4931–4939. doi: 10.1002/adfm.201201387. [DOI] [Google Scholar]
- 43.Liu H, Yang H, Hao X, Xu H, Lv Y, Xiao D, Wang H, Tian Z. Development of polymeric nanoprobes with improved lifetime dynamic range and stability for intracellular oxygen sensing. Small. 2013;9(15):2639–2648. doi: 10.1002/smll.201203127. [DOI] [PubMed] [Google Scholar]
- 44.X-d Wang, Gorris HH, Stolwijk JA, Meier RJ, Groegel DB, Wegener J, Wolfbeis OS. Self-referenced RGB colour imaging of intracellular oxygen. Chem Sci. 2011;2(5):901–906. doi: 10.1039/c0sc00610f. [DOI] [Google Scholar]
- 45.X-d Wang, Stolwijk JA, Lang T, Sperber M, Meier RJ, Wegener J, Wolfbeis OS. Ultra-small, highly stable, and sensitive dual nanosensors for imaging intracellular oxygen and pH in cytosol. J Am Chem Soc. 2012;134(41):17011–17014. doi: 10.1021/ja308830e. [DOI] [PubMed] [Google Scholar]
- 46.Wu C, Bull B, Christensen K, McNeill J. Ratiometric single-nanoparticle oxygen sensors for biological imaging. Angew Chem Int Ed. 2009;48(15):2741–2745. doi: 10.1002/anie.200805894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yim H, S-j Park, Bae YH, Na K. Biodegradable cationic nanoparticles loaded with an anticancer drug for deep penetration of heterogeneous tumours. Biomaterials. 2013;34(31):7674–7682. doi: 10.1016/j.biomaterials.2013.06.058. [DOI] [PubMed] [Google Scholar]
- 48.Owens Iii DE, Peppas NA. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int J Pharm. 2006;307(1):93–102. doi: 10.1016/j.ijpharm.2005.10.010. [DOI] [PubMed] [Google Scholar]
- 49.Ingram J, Zhang C, Cressman JR, Hazra A, WEI Y, Koo Y-E, Ziburkus J, Kopelman R, Xu J, Schiff SJ. Oxygen and seizure dynamics: I. Experiments. J Neurophysiol. 2014;00540:02013. doi: 10.1152/jn.00540.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ingram JM, Zhang C, Xu J, Schiff SJ. FRET excited ratiometric oxygen sensing in living tissue. J Neurosci Methods. 2013;214(1):45–51. doi: 10.1016/j.jneumeth.2013.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Koren K, Dmitriev RI, Borisov SM, Papkovsky DB, Klimant I. Complexes of IrIII-Octaethylporphyrin with Peptides as Probes for Sensing Cellular O2. Chembiochem. 2012;13(8):1184–1190. doi: 10.1002/cbic.201200083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Borisov SM, Mayr T, Mistlberger G, Waich K, Koren K, Chojnacki P, Klimant I. Precipitation as a simple and versatile method for preparation of optical nanochemosensors. Talanta. 2009;79(5):1322–1330. doi: 10.1016/j.talanta.2009.05.041. [DOI] [PubMed] [Google Scholar]
- 53.Albanese A, Tang PS, Chan WC. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14:1–16. doi: 10.1146/annurev-bioeng-071811-150124. [DOI] [PubMed] [Google Scholar]
- 54.Yang W-H, Smolen VF, Peppas NA. Oxygen permeability coefficients of polymers for hard and soft contact lens applications. J Membr Sci. 1981;9(1):53–67. doi: 10.1016/S0376-7388(00)85117-0. [DOI] [Google Scholar]
- 55.Fercher A, Borisov SM, Zhdanov AV, Klimant I, Papkovsky DB. Intracellular O2 sensing probe based on cell-penetrating phosphorescent nanoparticles. ACS Nano. 2011;5(7):5499–5508. doi: 10.1021/nn200807g. [DOI] [PubMed] [Google Scholar]
- 56.Mendes AN, Hubber I, Siqueira M, Barbosa GM, de Lima Moreira D, Holandino C, Pinto JC, Nele M (2012) Preparation and Cytotoxicity of Poly (Methyl Methacrylate) Nanoparticles for Drug Encapsulation. In: Macromolecular Symposia, Wiley Online Library, pp 34–40
- 57.Araujo L, Sheppard M, Löbenberg R, Kreuter J. Uptake of PMMA nanoparticles from the gastrointestinal tract after oral administration to rats: modification of the body distribution after suspension in surfactant solutions and in oil vehicles. Int J Pharm. 1999;176(2):209–224. doi: 10.1016/S0378-5173(98)00314-7. [DOI] [Google Scholar]
- 58.Ivanov AI (2008) Pharmacological inhibition of endocytic pathways: is it specific enough to be useful? In: Exocytosis and Endocytosis. Springer, pp 15–33 [DOI] [PubMed]
- 59.Carraway E, Demas J, DeGraff B, Bacon J. Photophysics and photochemistry of oxygen sensors based on luminescent transition-metal complexes. Anal Chem. 1991;63(4):337–342. doi: 10.1021/ac00004a007. [DOI] [Google Scholar]
- 60.DeGraff B, Demas J. Luminescence-based oxygen sensors. Reviews in Fluorescence. 2005;2005:125–151. [Google Scholar]
- 61.Foster KA, Galeffi F, Gerich FJ, Turner DA, Müller M. Optical and pharmacological tools to investigate the role of mitochondria during oxidative stress and neurodegeneration. Prog Neurobiol. 2006;79(3):136–171. doi: 10.1016/j.pneurobio.2006.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pakan JM, McDermott KW. A method to investigate radial glia cell behavior using two-photon time-lapse microscopy in an ex vivo model of spinal cord development. Front Neuroanat. 2014;8:22. doi: 10.3389/fnana.2014.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schmid RS, Yokota Y, Anton E. Generation and characterization of brain lipid-binding protein promoter-based transgenic mouse models for the study of radial glia. Glia. 2006;53(4):345–351. doi: 10.1002/glia.20274. [DOI] [PubMed] [Google Scholar]
- 64.Becker W. Fluorescence lifetime imaging—techniques and applications. J Microsc. 2012;247(2):119–136. doi: 10.1111/j.1365-2818.2012.03618.x. [DOI] [PubMed] [Google Scholar]
- 65.Ingaramo M, York AG, Wawrzusin P, Milberg O, Hong A, Weigert R, Shroff H, Patterson GH. Two-photon excitation improves multifocal structured illumination microscopy in thick scattering tissue. Proc Natl Acad Sci. 2014;111(14):5254–5259. doi: 10.1073/pnas.1314447111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pampaloni F, Ansari N, Stelzer EH. High-resolution deep imaging of live cellular spheroids with light-sheet-based fluorescence microscopy. Cell Tissue Res. 2013;352:161–177. doi: 10.1007/s00441-013-1589-7. [DOI] [PubMed] [Google Scholar]
- 67.Golub AS, Pittman RN. PO2 measurements in the microcirculation using phosphorescence quenching microscopy at high magnification. Am J Physiol Heart Circ Physiol. 2008;63(6):H2905. doi: 10.1152/ajpheart.01347.2007. [DOI] [PubMed] [Google Scholar]
- 68.Devor A, Sakadzic S, Srinivasan VJ, Yaseen MA, Nizar K, Saisan PA, Tian P, Dale AM, Vinogradov SA, Franceschini MA, Boas DA. Frontiers in optical imaging of cerebral blood flow and metabolism. J Cereb Blood Flow Metab. 2012;32(7):1259–1276. doi: 10.1038/jcbfm.2011.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Spencer JA, Ferraro F, Roussakis E, Klein A, Wu J, Runnels JM, Zaher W, Mortensen LJ, Alt C, Turcotte R, Yusuf R, Cote D, Vinogradov SA, Scadden DT, Lin CP. Direct measurement of local oxygen concentration in the bone marrow of live animals. Nature advance online publication. 2014 doi: 10.1038/nature13034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Verma A, Stellacci F. Effect of surface properties on nanoparticle-cell interactions. Small. 2010;6(1):12–21. doi: 10.1002/smll.200901158. [DOI] [PubMed] [Google Scholar]
- 71.Nichols AJ, Roussakis E, Klein OJ, Evans CL. Click-Assembled, Oxygen-Sensing Nanoconjugates for Depth-Resolved, Near-Infrared Imaging in a 3 D Cancer Model. Angew Chem Int Ed n/a-n/a. 2014 doi: 10.1002/anie.201311303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Dmitriev RI, Ropiak HM, Ponomarev GV, Yashunsky DV, Papkovsky DB. Cell-penetrating conjugates of coproporphyrins with oligoarginine peptides: rational design and application for sensing intracellular O2. Bioconjugate Chem. 2011;22(12):2507–2518. doi: 10.1021/bc200324q. [DOI] [PubMed] [Google Scholar]
- 73.Anilkumar U, Weisová P, Düssmann H, Concannon CG, König H-G, Prehn JHM. AMP-activated protein kinase (AMPK)–induced preconditioning in primary cortical neurons involves activation of MCL-1. J Neurochem. 2013;124(5):721–734. doi: 10.1111/jnc.12108. [DOI] [PubMed] [Google Scholar]
- 74.Stoppini L, Buchs P-A, Muller D. A simple method for organotypic cultures of nervous tissue. J Neurosci Methods. 1991;37(2):173–182. doi: 10.1016/0165-0270(91)90128-M. [DOI] [PubMed] [Google Scholar]
- 75.Murphy RC, Messer A. Gene transfer methods for CNS organotypic cultures: a comparison of three nonviral methods. Mol Ther. 2001;3(1):113–121. doi: 10.1006/mthe.2000.0235. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.