Abstract
Numerous studies attest to essential roles for Eph receptors and their ephrin ligands in controlling cell positioning and tissue patterning during normal and oncogenic development. These studies suggest multiple, sometimes contradictory, functions of Eph-ephrin signalling, which under different conditions can promote either spreading and cell-cell adhesion or cytoskeletal collapse, cell rounding, de-adhesion and cell-cell segregation. A principle determinant of the balance between these two opposing responses is the degree of receptor/ligand clustering and activation. This equilibrium is likely altered in cancers and modulated by somatic mutations of key Eph family members that have emerged as candidate cancer markers in recent profiling studies. In addition, cross-talk amongst Ephs and with other signalling pathways significantly modulates cell-cell adhesion, both between and within Eph- and ephrin-expressing cell populations. This review summarises our current understanding of how Eph receptors control cell adhesion and morphology, and presents examples demonstrating the importance of these events in normal development and cancer.
Keywords: Eph receptor, Ephrin, Receptor tyrosine kinase, Cell-cell adhesion, Cancer
Introduction to Eph receptors and their ligands
Eph receptors represent the largest family of receptor tyrosine kinases [1–3]. They play essential roles during the development of invertebrate and vertebrate embryos where they guide cell migration and positioning during tissue modelling programs such as gastrulation and patterning of vascular, skeletal and nervous systems [2–4]. Their diverse biological functions are achieved principally by modulating the adhesion or de-adhesion/segregation between Eph-expressing cells and cells expressing their membrane-bound ligands, the ephrins. Emerging evidence suggests that Eph activation can also affect cell proliferation and apoptosis, an area recently reviewed elsewhere [5, 6].
A distinguishing feature of Eph-ephrin interactions is the phenomenon of bidirectional signalling. Ephrin binding promotes receptor clustering and phosphorylation of tyrosine residues, initiating forward signalling in Eph-bearing cells while, in parallel, clustering of the ephrins on the cell surface of ligand-expressing cells triggers a reverse signal into these cells [1]. Thus, the instructions derived from Eph/ephrin interactions are relayed into both cell populations to facilitate their mutual adhesion or segregation, which within the context of a tissue layer governs cell positioning and cell-cell contacts underlying normal and oncogenic patterning [7].
Eph receptors (and ephrins) are highly conserved in vertebrates and have been found in invertebrates including Drosophila, C. elegans and, interestingly, also in sponges [8–10]. The fact that the invertebrates express only one Eph receptor, VAB-1 in C. elegans, which interacts with four ephrins [11, 12], might indicate that increasing body plan complexity correlates with the growing size of the Eph/ephrin family [8, 9]. While Ephs and ephrins are predominantly expressed and active during development, their roles in normal tissue maintenance and homeostasis as well as their aberrant expression in a wide range of cancers is increasingly recognised, wherein both tumourigenic and tumour-suppressive functions have been described [4, 5]. In addition, Eph mutations are emerging as prevalent in cancers, which, as will be discussed below, likely modulate Eph-mediated cell-cell adhesion and de-adhesion events promoting tumourigenesis.
Nomenclature of Eph receptors and ephrins
There are two subtypes of Eph receptors distinguished by structural features and ephrin-binding preferences [1]—the EphA subtype, which comprises nine members in mammals (EphA1–A8, EphA10), and the EphB group with five members (EphB1–EphB4, EphB6) [13, 14]. The first isolated family member (EphA1) was found in a cDNA sample from an Erythropoietin-Producing Hepatocellular carcinoma cell line [15], explaining the acronym EPH-like receptors. The ligands of Eph receptors, the ephrins (Eph receptor interacting proteins), are cell membrane-bound and in vivo usually present on the surface of opposing cells. Ephrins are also divided into two subclasses based on the type of their cell-membrane attachment: A-type ephrins (ephrin-A1–A6) are linked to the membrane via a GPI (glycosyl-phosphatidylinositol) anchor, while type B ephrins (ephrin-B1–B3) contain a single transmembrane domain and a short cytoplasmic tail [13, 14]. The A subclass receptors bind preferentially to type A ephrins, while EphBs favour type B ephrins [13, 14], although cross-subclass interactions have also been reported: EphA4 is able to bind A- and B-type ephrins with comparable affinities [16–18], and ephrin-A5 binds and activates EphB2 in addition to A-type Ephs [19]. In comparison, receptor-ligand interactions within the same subclass are very promiscuous, whereby within a subclass every Eph receptor can bind to multiple ephrins with very similar affinities [1].
Eph structure
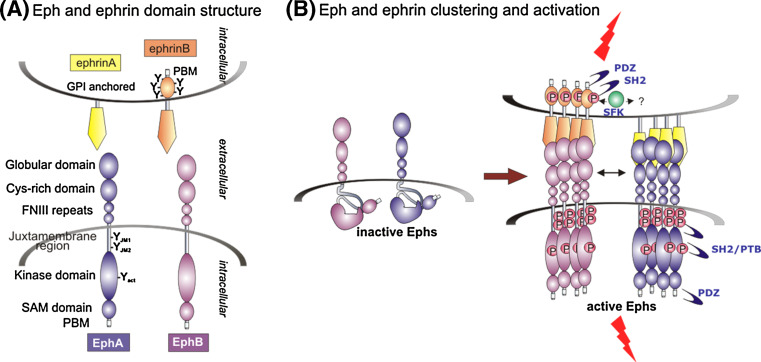
The general structure of Ephs is highly conserved throughout the animal kingdom [9] (see Fig. 1a). The extracellular domain consists of an N-terminal globular domain responsible for ephrin binding, a cysteine-rich EGF-like domain and two fibronectin III repeats [20, 21]. The globular, together with the cysteine-rich domain, is additionally involved in ephrin-independent receptor dimerisation and clustering [22–25]. Analogous to other RTKs, Eph receptors contain a single transmembrane spanning domain. The intracellular domain is composed of a juxtamembrane region containing two conserved regulatory tyrosine residues that control kinase activity, a single tyrosine kinase domain, a SAM (sterile alpha motif) protein-protein interaction domain and a PDZ protein-binding motif (PBM), which can bind PDZ domain-containing proteins serving as scaffolds for the assembly of multi-protein signalling complexes [3, 26]. Amongst RTKs the presence of a SAM domain is unique to the Eph receptor family and may, along with the ligand-binding and cysteine-rich domains within the extracellular region, play a role in receptor-receptor interactions aiding homo- or heterotypic oligomerisation [27], as well as in regulating receptor endocytosis [28, 29].
Fig. 1.
Models of the structure and activation of Eph receptors. a Eph receptors contain an extracellular region with a globular ligand-binding domain, followed by cysteine-rich and fibronectin type III domains. Ligand binding promotes clustering and phosphorylation of the tyrosine kinase-containing intracellular region, at conserved sites within the juxtamembrane and kinase domains. b Inactive Ephs display an auto-inhibited conformation (left), in which the kinase domain interacts with the juxtamembrane domain, which is released upon activation to enable robust phosphorylation and phosphorylation-dependent signalling (right). Receptor-receptor interactions in the ECD further promote clustering [22], including co-clustering of EphA and EphB receptors [57]. The transmembrane ephrin-Bs also become phosphorylated on tyrosine residues by Src family kinases, which likely also promote signalling by the GPI-linked ephrin-As via clustering of lipid raft microdomains and associated signalling molecules
Receptor clustering and activation
Unlike ligands of other RTK subfamilies, only membrane-bound or artificially clustered ephrins can trigger receptor signalling [30], while non-clustered soluble ephrins act as antagonists [23, 31–33]. More specifically, at least tetramerisation of Ephs is necessary to achieve full biological activation [34, 35]. Prior to ephrin contact, Ephs are loosely arranged within the plasma membrane displaying minimal kinase activity [1, 36], although this may be elevated by high receptor expression levels [37–40]. Eph/ephrin signalling is initiated by 1:1 high-affinity interactions between the Eph globular domain and a conserved Eph-binding domain of ephrins. Additional protein interfaces in these domains then tether dimers so that each ephrin binds two Ephs and each Eph binds two ephrins leading to the assembly of Eph/ephrin tetramers [20, 22, 23, 41, 42]. Further analysis of signalling cluster assembly demonstrated that Eph/Eph interactions resulting in expansion of these clusters progress independent of ephrin contact [24] through Eph protein interfaces located within the ligand-binding and cysteine-rich domains [22, 25]. This explains how, similar to ErbB receptors [43], Eph receptors spontaneously cluster at high cell surface densities, a phenomenon that could be further enhanced by transmembrane domain interactions [44], whereas a suspected role of cytoplasmic interactions promoting Eph clustering is yet to be confirmed.
Eph kinase domain activation relies on conformational changes that release inhibitory interactions between the kinase domain activation loop and the juxtamembrane region, and are triggered by transphosphorylation of three invariant tyrosines, one within the activation loop of the kinase domain and two within a highly conserved 10-amino acid motif in the juxtamembrane region (see Fig. 1b). While phosphorylation of the kinase domain tyrosine directly activates the enzyme, additional phosphorylation of the juxtamembrane tyrosine residues is required for full kinase activity [45–48], with recent work suggesting the second juxtamembrane tyrosine being most important for Eph activation [49]. The three invariant and potentially other tyrosines, for example in the SAM domain [50, 51], once phosphorylated, serve as docking sites for SH2 domain- and PTB-containing proteins, linking to signalling pathways that will be described below.
Co-clustering of Eph family members
Although RTK hetero-oligomerisation is a concept common, for instance, for receptors of the EGFR family [52], to date the evidence for cross-talk between Eph receptors is mostly limited to intra-subclass interactions. Given the promiscuous nature of ephrin-Eph interactions within subclasses, it is expected that ephrins will concurrently bind Ephs of the same subclass when co-expressed, and indeed EphB1 and EphB4 can form functional hetero-oligomeric complexes with kinase-dead EphB6, resulting in its transphosphorylation [53, 54]. However, there is also evidence for cross-class transphosphorylation: the EphA4 kinase domain is capable of phosphorylating the juxtamembrane tyrosines of EphB2 and EphA2 in vitro [51], and a phosphoproteomics approach of ephrin-B1-stimulated palate mesenchyme cells revealed phosphorylation of EphA2, EphA3, EphA7 and EphB4, in addition to EphB2 and EphB3, which are known to bind ephrin-B1 [55]. Similarly, phosphorylation of various EphAs was modulated in EphB2-expressing HEK293 cells when stimulated with ephrin-B1-expressing cells [56], although interestingly EphA2 phosphorylation decreased. Emerging evidence suggests that co-clustering occurs also between EphA and EphB receptors: selective activation of EphA3 with an agonistic monoclonal antibody [35] also co-clusters and activates co-expressed EphB2 in HEK293T cells, as well as other endogenous EphA and EphB receptors frequently co-expressed in tumour cells [57]. Overall, the notion of Eph co-clustering impacts profoundly on the understanding of Eph-mediated cell behaviour, since cellular responses of Eph-bearing cells in contact with ephrin-bearing cells will not be limited to those able to directly bind the activating ephrin, but by the overall abundance of EphA and EphB receptors on that cell.
Cross-talk with other receptors
It is highly likely that RTK signalling networks are necessarily interconnected as they share prominent cytoplasmic signalling cascades, such as the MAPK or PI3K pathways [58]. Thus, it is not surprising that co-clustering and interdependent signalling between Ephs and other RTKs has been suggested. For example, EphA4 and FGFR were reported to transphosphorylate each other and synergistically activate shared downstream signalling, such as MAPK activation [59–61]. By contrast, FGFR1 appears to inhibit ephrin-dependent EphB2 signalling by blocking a positive feedback loop via transcriptional down-regulation of the Ras-ERK pathway while indirectly increasing the phosphorylation of unstimulated EphB2 [62]. Interestingly, EGFR reportedly colocalises with EphA2 at cell-cell contacts [63] and adhesion-induced EphA2 expression is thought to be regulated by EGFR activation [64], while EphA3 silencing modulates EGFR downstream signalling in squamous cell carcinoma cells [65]. Accordingly, analysis of the EGFR interactome revealed amongst many other interaction partners ligand-independent association of EphA2 and EphB4 [66, 67]. Moreover, EphA2 co-operates with ErbB2 to amplify its tumourigenic effects, conferring resistance to anti-ErbB2 therapy [68]. Interactions between Ephs and VEGFR2 [69, 70] as well as the atypical RTK RYK [71–74], which contains an inactive kinase domain and shows overlapping biological functions with Eph RTKs, have also been proposed.
In addition, there is considerable interest in the cross-talk between Ephs and the receptor CXCR4 for the chemokine SDF-1 [75, 76], which plays important roles during embryonic and oncogenic development [77] and has been proposed to control endothelial cell movement in cooperation with EphB2 and EphB4 [75]. Furthermore, crosstalk of EphBs with NMDA-type [78–81] and AMPA-type [82] glutamate receptors is important for the regulation of synaptic plasticity. Further insight into the crosstalk between Ephs, other RTKs and other types of transmembrane receptors will be essential for a better understanding of Eph function and its potential application in the development of combination therapies, particularly for cancer treatment regimes.
Biological roles of Eph/ephrin-mediated adhesion and repulsion
Cell positioning as a central mechanism underlying Eph function
Eph receptors primarily function during embryogenesis to position motile cells and cell layers within surrounding tissues. They control the direction of cell movement and the choice of interacting cells or cell layers by initiating assembly or disassembly of cell-cell contacts. During embryonic development Ephs and ephrins are expressed in temporally and spatially restricted patterns in developing tissues and organs, where they participate in germ layer formation, gastrulation, organogenesis and tissue patterning [3, 83–86]. Originally identified as axon guidance molecules, their next best studied role lies within the developing vascular system. Eph and ephrin expression and function in healthy adult tissues are less prevalent, while re-emerging cell guidance functions during tumour invasion, neoangiogenesis and metastasis are now well established (see below).
Eph/ephrin-mediated cell positioning relies on cell-cell contact since both receptors and ligands are membrane-bound. Thus, migration of an Eph-expressing cell into an area of graded ephrin expression is directed by contact-dependent cell-cell segregation, instructing a change of direction to avoid the ephrin-rich area and a final destination that is determined by competition for ephrin interactions with other Eph expressing cells or axons [87, 88]. However, cell location is not only determined by graded expression and the overall abundance of interacting Ephs and ephrins defining the final position, but also by their crosstalk with other signalling systems [89–91]. Together, these multifaceted interactions facilitate accurately controlled segmentation and tissue boundary formation—processes that are vitally important during most developmental patterning programs [14, 92].
Cell sorting and boundary formation
One of the earliest studied developmental mechanisms of Eph/ephrin-mediated cell-cell segregation restricting cell movement and defining tissue compartments is boundary formation between zebrafish rhombomeres: during this developmental stage Ephs and ephrins are expressed in complementary segments, and their bi-directional signalling maintains boundaries between even and odd rhombomeres [16, 93]. This biological concept has been replicated in vitro in models of cell-cell segregation using zebrafish blastomeres [92] and more recently in mammalian cell co-cultures of Eph- and ephrin-expressing cells [56, 62, 94]. Also during mouse embryogenesis, EphB2 and EphB3 interactions with ephrin-B2 in developing colonic crypts mediate sorting and positioning of epithelial cells along the crypt-villum axis [95]. Furthermore, Eph/ephrin interactions were shown to restrict intermingling between ephrin-positive astrocytes and Eph-expressing fibroblastic meningeal cells [96] or Schwann cells [97]. Interestingly, during nerve wound repair EphB2-mediated segregation of Schwann cells and fibroblasts relies both on repulsion between the two cell types as well as tight adhesion of Schwann cells within their collective [98].
A number of pathways are considered to be vital for cell-cell segregation and maintenance of boundaries, including inhibition of gap junction communication [92, 99], MAPK activation downstream of EphB2 signalling [62] and Wnt pathway-dependent RhoA activation [37]. Indeed, an siRNA screen for proteins required for EphB2/ephrin-B1-mediated cell sorting yielded 200 targets, roughly a quarter of which displayed altered levels of tyrosine phosphorylation [56]. Amongst these were known regulators of the cytoskeleton, endocytosis, cell adhesion and migration and cell polarity, highlighting the importance of a range of cellular processes in Eph-mediated cell segregation [56], which will be discussed below. In addition to these roles in cell positioning and sorting Eph/ephrin signalling is also implicated in mesenchymal-to-epithelial transition (MET) and endothelial-to-mesenchymal transition (EMT), which are essential cell-cell communication mechanisms involved in establishing identity and well-defined boundaries between endothelial/epithelial and mesenchymal tissues during somitogenesis [100–102] and heart development [103, 104].
Axon guidance and neural patterning
Amongst the receptor/ligand families implicated in axonal guidance, Ephs and ephrins constitute the most prevalent system orchestrating topographic mapping and neural patterning during early embryonic development [83, 105]. Eph/ephrin function is essential in many functional domains within the developing central nervous system, such as the retinotectal [106] and the corticospinal tract [107]. Thus, in the vertebrate visual system, ephrin-A ligands allow outgrowth of EphA-expressing retinal axons when present in low doses, but block extension at higher concentrations [89, 90, 108, 109], a system regarded as a paradigm for the establishment of topographic maps [88, 91, 105]. In accord, gene knock-outs confirmed ephrin-A2 and ephrin-A5 as essential for guidance cues for retinal axons in mammals [106, 110], which also regulate axon branching of retinal ganglion cells [111]. Ephrin-B3-induced EphA4 forward signalling is vital for corticospinal tract axon guidance, connecting brain and spinal cord [107, 112, 113].
Eph/ephrin controlled cell-cell segregation is essential for neural crest cell migration [114–116] and normal skeletal morphogenesis, and deletion of ephrin-B1 or mutation in its PDZ binding domain results in defects in neural crest derived tissues [117] and in the corpus callosum, a major commissural axon tract [118], leading to perinatal lethality due to skeletal defects and incomplete body closure [119]. Importantly, repulsive Eph/ephrin interactions between motor and sensory projections during neuromuscular circuit assembly prevent their intermingling, thereby ensuring the establishment of tightly associated yet functionally discrete peripheral nerve pathways [120].
Eph and ephrin function are also essential for synaptogenesis and synapse maturation [121, 122]. EphBs and ephrin-Bs are implicated in pre- and post-synaptic specification during formation of synapses affecting the stabilisation of initial axon/dendrite contacts, formation of postsynaptic specialisations and promotion of filopodia motility [82, 123, 124] and function of NMDA receptors [78, 81, 125]. For example, excitatory synapse maturation involves dendritic spine morphogenesis that requires EphB forward [126–128] and ephrin-B reverse [129] signalling in hippocampal neurons, while EphA4 activation triggers dendritic spine retraction [130]. Transsynaptic EphB/ephrin-B signalling also regulates structural plasticity of presynaptic nerve terminals [131].
Vasculogenesis and angiogenesis
Not surprisingly, axonal growth cones and capillary tip cells use common repulsive and attractive signals that determine directional guidance of their position [132]. Significant evidence suggests that B-type Ephs and ephrins, especially interactions between EphB2/B4 and ephrin-B2, play pivotal roles in the development of the embryonic cardiovascular system [133–137]. The prominent expression of ephrin-B2 on embryonic arteries and its cognate receptor EphB4 on veins, together with prominent and lethal vascular defects in knock-out mice lead to the concept that reciprocal signalling at the boundaries between these two types of vessels prevents arterial and venous endothelial cell intermingling [132, 138]. In agreement, deletion of full-length ephrin-B2, its intracellular domain or EphB4, although displaying differential effects on neural crest cell migration, all cause similar defects in the remodelling of the primary capillary plexus, inhibit the formation of major embryonic vessels and cause embryonic lethality [133–136, 139, 140]. Recently, a different model challenged this view of vasculogenic assembly, proposing the formation of a precursor vessel that segregates to form the dorsal aorta and the cardinal vein—a process that again is critically dependent on signalling between EphB4 and ephrin-B2 [141]. There is evidence that EphB4 forward signalling directs these functions in early vascular development, while ephrin-B2 reverse signalling is required for later processes, such as cardiac valve formation [142]. While EphB4 seems to play a predominant role in vascular patterning, a combined knock-out of EphB2 and EphB3 has a similar phenotype, however, with only 30% penetrance, suggesting the integrated activity of additional Eph receptors [133]. Notably, EphB2 and EphB4 appear to co-operate with SDF-1 chemokine signalling during endothelial cell migration to form capillary-like structures [75].
In contrast there is only limited evidence for A-type Ephs and ephrins during cardiovascular development, the exception being EphA3 and ephrin-A1, expressed during embryogenesis on major blood vessels and heart valves [103, 104]. Their interaction is essential for segregation between the endothelial lining and cardiac jelly, trans-differentiation of endothelial to mesenchymal cells and their directed migration to form the endocardial cushion critical for proper heart septation [103]. However, emerging evidence suggests important roles for EphA receptors in physiological and pathological neo-angiogenesis, with EphA2 functioning in endothelial cell migration during post-natal vascular assembly in mice, and being expressed in endothelial cells in tumours [143, 144] (below).
Since Eph/ephrin- and VEGFR-signalling both control blood vessel growth and arterial differentiation, it is perhaps not surprising that functional interactions have also been reported. Two recent reports demonstrated that ephrin-B2 regulates VEGFR internalisation and thus critically affects VEGFR function during angiogenesis [145, 146]. Altered expression of ephrin-B2 causes severe vascular defects during VEGF-induced angiogenesis [146]. Moreover, ephrin-B2 PDZ signalling-deficient mice lack regulation of VEGFR2 signalling, thus displaying reduced vascularisation in brain and retina as well as within tumours [145]. Intriguingly, ephrin-B2 appears to directly activate VEGFR2, even in the absence of VEGF-A [145].
Interaction of endothelial cells with supporting cells
In addition to orchestrating the interaction between endothelial cells during vessel assembly and angiogenesis, Eph/ephrin interactions also guide the communication with vascular smooth muscle cells and pericytes that are essential to stabilise arteries, and to a lesser extent veins, and contribute to the maintenance of mature, non-angiogenic vessels once angiogenesis is completed [132]. EphrinB2 is expressed on vascular smooth muscle cells and pericytes [147, 148], and directs the interactions between endothelial and mural cells that are essential for the stability of blood-perfused vessels [140, 149]. Conditional gene targeting of ephrinB2 on pericytes and vascular smooth muscle cells leads to defective mural cell coverage and in leaky vasculature, a defect that is typical also for chronic diseases, such as diabetes and cancer [150]. EphrinB2 and EphB2 are also expressed in the surrounding mesenchyme [133], and ephrin/Eph interactions between mesenchymal and endothelial cells [151] may help differentiate mesenchymal cells into perivascular supporting cells. Finally, EphB4/ephrinB2 interactions are also thought to mediate monocyte adhesion and transmigration through the vascular endothelium [152].
Lymphangiogenesis
Lymphangiogenesis occurs predominantly during embryonic development, but also has a documented role in tumour metastasis and, similar to blood vessel morphogenesis, involves EphB4/ephrinB2 and VEGF signalling [153]. Interestingly, whilst mice lacking the PDZ motif of ephrinB2 show normal blood vasculature development, they display severe postnatal lymphangiogenesis defects due to inhibition of endothelial cell sprouting from a primitive plexus [154], which could be explained by the ephrinB2-mediated inhibition of VEGFR3 internalisation [146].
Eph receptors in tumour progression and metastasis
Increasing interest in the Eph/ephrin signalling system stems from its documented importance in a wide range of epithelial and mesenchymal tumours [2, 6], indeed most—if not all—family members were initially identified in tumour cell lines [7]. Eph cell guidance functions normally active during embryogenesis re-emerge unscheduled and often de-regulated in tumours, modulating cell-cell and cell-matrix attachment and survival during invasion, neo-angiogenesis and metastasis [2, 7, 155]. Eph overexpression often correlates with more aggressive, invasive and metastatic phenotypes and poor prognosis. An example is melanoma, where expression of Ephs correlates with increased tumour progression and invasive potential [156–158], and primary melanomas express significantly less EphA2 than distant metastases [159]. Similarly in breast cancer, EphA3 reportedly is highly expressed in lymph node metastases but not in the primary tumour [160]. Also EphA2 is highly elevated in breast cancer [144, 161] and associated with poor prognosis [68, 162]. Over-expression of EphA2 in the non-transformed mammary epithelial cell line MCF10A induces anchorage-independent growth, increased invasiveness in matrigel and tumour formation in nude mice [161]. EphB4 is also overexpressed in breast cancer [163] and suggested to be causally involved in its progression [164]. Tumour-promoting roles of EphB4 and EphA2 have been confirmed in MMTV/Neu breast cancer mouse models, where EphB4 overexpression appears to enhance tumour initiation and lung metastasis [165], while loss of EphA2 has the opposite effect [68]. Furthermore, co-clustering of EphA2 with ErbB2 was reported to enhance Ras-MAPK and RhoGTPase signalling [68]. In addition to melanoma and breast cancer, Eph expression is elevated in a wide variety of cancers, including colon, lung, kidney, ovarian and prostate cancers, sarcoma and neuroblastoma [7].
Tumour neo-angiogenesis
As in embryonic development, Ephs and ephrins play important roles in neovascularisation during tumour development. Interacting ephrin-A1 and EphA2 [144, 166], as well as ephrin-B2 and EphB4 [147, 148] are overexpressed in tumour vasculature, where the ligand commonly marks endothelial cells. Thus, while EphA2 is not expressed on embryonic vasculature [167], and EphA2−/− mice do not show vascular defects during embryonic development [143, 168], ephrin-A1 and EphA2 are prevalent in the vasculature of tumour xenografts [144]. Administration of soluble EphA2-Fc or EphA3-Fc inhibits tumour growth and endothelial cell migration in different in vivo models [32, 166, 169], while similarly EphA2 or ephrin-A1 deficiency reduces tumour angiogenesis [170, 171]. In addition, EphA2 activation on vascular smooth muscle cells might also induce the retraction of perivascular supporting cells via the inhibition of Rac/PAK (see below), thus allowing endothelial cells to respond to angiogenic cues [172]. Moreover, EphA2 was identified as an important mediator of melanoma vasculogenic mimicry [158].
Ephrin-B2 and EphB4 are also prominent in tumour angiogenesis. Ephrin-B2 expression, which in cultured human microvascular and arterial endothelial cells is induced by VEGF, bFGF and HGF [173], is up-regulated at sites of neovascularisation, such as tumours and wounds [150]. In mouse models of ischemia, ephrin-B2-mediated activation of EphB4 enhances the pro-angiogenic activity of endothelial cells [174, 175], and EphB4 expression on tumour cells increases angiogenesis by recruiting ephrin-B2-positive endothelial cells [176]. By contrast, in a retinal model of postnatal angiogenesis, EphB4 overexpression resulted in larger blood vessels, disorganised branching and reduced vascular permeability via inhibition of the angiopoitin-1/Tie2 pathway due to increased ephrin-B2 reverse signalling [177]. De-regulated EphB4/ephrin-B2 function in endothelial cells has also been implicated in Kaposi sarcoma [178] and hepatocarcinoma [179]. Interestingly, manipulation of ephrin-B2-signalling using the EphB4 extracellular domain fused to human serum albumin led to reduced blood vessel density, reduced pericyte recruitment and increased hypoxia, similar to effects seen with anti-VEGF treatment, whereby concurrent ephrin-B2- and VEGF-signalling blockade indeed showed a synergistic effect [180].
Role in tumour suppression
Interestingly, Ephs are not only implicated in tumour promotion but, depending on the context, are considered as tumour suppressors. Thus, in contrast to roles of EphA2 in tumour promotion, EphA2 knock-out mice show increased susceptibility to carcinogen-induced skin tumours and their invasive malignant progression [181]. Similarly, EphB4 can elicit promotion or suppression of tumour growth [164, 176, 182–184]: while it is found up-regulated in colon and endometrial cancers [185, 186], its loss is correlated with more invasive cancers of both colon [182] and breast [187]. One possible explanation for this dichotomy is that an initial increase in Eph receptor expression and function is followed by epigenetic silencing or modulation of function as the tumour progresses [6]. Thus, expression of EphA7 in colorectal and prostate cancers is frequently suppressed by promoter hypermethylation [188, 189], and re-expression in DU145 prostate cancer cells inhibits colony formation [188]. In accord, hypermethylation of EphB6 in breast cancer [190], of EphB2 in colorectal cancer [191] and of EphA3 [192] and almost all other Eph and ephrin family members in acute lymphoblastic leukaemia [193] has been reported.
Such a two-step model of Eph expression is best exemplified in colorectal carcinoma (CRC) where EphB receptor expression is elevated in intestinal adenomas [95, 185], but is lost during progression to a carcinoma and the initiation of invasive growth [182]. Similarly, EphA1 is overexpressed in earlier stages of CRC, but down-regulated in further advanced carcinomas, a trend that is also associated with poor survival [194, 195]. Gain-of-function mutations in the Wnt pathway are strongly implicated in the initiation of CRC and at the same time responsible for up-regulated EphB expression [196]. Mechanistically, EphB signalling may suppress cancer progression at this stage by regulating adherens junction formation and promoting compartmentalisation of colon carcinoma cells [197], although in parallel contributing to adenoma growth via Abl-cyclin D1 proliferative signalling [198]. Loss of EphB receptor expression or activity during CRC progression leads to uncoupling of EphB function from cyclin D1 signalling and results in increased tumour invasion while maintaining a high proliferation rate [182, 183, 197, 198]. Recent evidence now also suggests that EphB2 marks tumour-initiating intestinal stem cells in aggressive CRCs potentially contributing to disease relapse [199].
Interestingly, EphA7 has recently been implicated as a tumour suppressor in follicular lymphoma [200]. Normal B lymphocytes express truncated, soluble EphA7, which can inhibit the activity of other expressed Eph receptors, notably EphA2, apparently by direct interaction, although competition for ligand binding may also occur. Frequent loss of EphA7 in this context due to methylation is suggested to promote EphA2 signalling and thus oncogenesis [200].
Kinase active versus inactive Eph oncogenic function
In addition to stage-dependent changes of expression it is likely that ephrin- or Eph kinase-independent functions may play a significant part in their tumour-promoting role; this notion reflects several reports suggesting that Eph activation by agonists attenuates malignant cell phenotypes and that effective bi-directional signalling may not be prevalent in a cancer setting [6, 161, 201, 202]. Consistent with this, Eph receptor overexpression can be associated with loss of cognate ligand expression, as exemplified by the expression pattern of EphA2 and ephrin-A1 in glioblastoma and breast cancer cells [190, 203]. Interestingly, in mouse xenografts, treatment with the recombinant EphB4 ligand ephrin-B2 inhibited tumour progression and breast cancer cell migration via the Abl-Crk pathway [176], suggesting a tumour promoter role of EphB4 in this model that is ligand-independent or requires low ligand levels while high EphB4 kinase activity inhibits tumour progression. Furthermore, overexpression of kinase-dead EphB4 in breast cancer cells promotes tumour vascularisation by inducing recruitment, survival and proliferation of ephrin-B2-positive endothelial cells [204]. The notion of both kinase-dependent and -independent Eph functions is of particular relevance given that somatic mutations likely to affect Eph signalling activity are emerging as prevalent in a wide variety of cancers.
Somatic mutations
Recent cDNA array screening approaches revealed that somatic mutations in Eph receptor RTKs occur frequently in a variety of cancers. For instance, frame shift mutations of EphB2 were found in 41% of colorectal and 39% of gastric tumours with microsatellite instability [205, 206], and somatic mutations were detected in a total of 11 Eph receptors in a set of breast, lung, ovarian and prostate cancer samples, where most of the mutations were in lung cancer [207]. Moreover, a genome-wide screen of diagnostic somatic mutations in breast and colon cancers emphasised Eph family members, particularly EphA3, among the most frequently mutated genes in colon and lung cancer [208, 209], and Ephs were also reported as a candidate cancer genes in glioblastoma, melanoma and pancreatic carcinoma [210, 211]. Furthermore, a recent study in breast cancer patient samples, investigating a potential link between susceptibility loci and somatic mutations of ‘driver kinases’, identified SNPs in EPHB1, EPHA3 and EPHA7 genes as cancer risk alleles [212].
Typically, the somatic Eph mutations identified in these screens do not tightly cluster in a specific region but are widely distributed in diverse domains. However, as exemplified by EphA3 mutations, many of these lie in regions regulating receptor clustering and activity, including kinase and extracellular domains, and may alter cellular responses, for example to promote adhesive, rather than repulsive, signalling. Accordingly, EphA3 and EphA5 somatic mutations in lung cancer samples were detected within the activation loop or in the transmembrane domain [213], and EphA3 was one of seven tyrosine kinases identified in CRC harbouring kinase domain mutations [214]. Interestingly for EphB6, a family member lacking a functional kinase, frequent somatic mutations [215] and a germline sequence variant [216] were also found in CRC samples. While the effects of these mutations on kinase activity/ligand binding and downstream responses remain to be elucidated, some indications exist for two EphB2 mutations: The G787R variant found in rare cases of hereditary colorectal cancer inhibits kinase activity [217] while the non-sense EphB2 Q722X mutation suggested to promote prostate cancer progression results in truncation and kinase inactivation [218, 219]. Thus Eph somatic mutations likely result in change of function, rather than loss of function mutants, as with classical tumour suppressors.
How does Eph signalling switch cell-cell adhesion to retraction?
The roles of Ephs and ephrins in the diverse developmental programs, and in cancer, described above rely primarily on their control of cell navigation and patterning, by modulating cell-cell adhesion and de-adhesion, or retraction. Whether Eph receptor signalling results in adhesion or retraction is determined by various parameters, including Eph and ephrin cell surface densities, composition of signalling clusters, disruption and internalisation of Eph/ephrin complexes, participation of various cytoskeletal- and membrane-associated regulatory molecules, and cross-talk with other cell signalling systems.
The importance of kinase activity
While ample evidence supports the notion that Eph-ephrin contact results in cell contraction, de-adhesion and segregation [3, 220], Eph/ephrin interactions can also promote increased cell adhesion. As a prominent example, co-expression of a cytoplasmic-truncated EphA7 splice variant suppresses the phosphorylation of the full-length EphA7 and shifts the response of migrating cells from ephrin-A5-mediated cell-cell segregation to adhesion, an essential event during embryogenesis that induces fusion of the neural tube [221]. Demonstration of this developmentally regulated expression and dominant negative function of the truncated kinase-dead receptor confirms the essential role of this Eph kinase-controlled switch between adhesive and de-adhesive cell-cell interactions. Other examples include the fusion events occurring during tabularisation of the urethra, and the partition between urinary and alimentary tracts that are implemented by EphB/ephrin-B-facilitated cell adhesion [222]. Further kinase-independent roles have been described based on the ability of kinase-dead receptors to functionally compensate for loss of wild-type receptors in knockout mice. For example, kinase dead EphA4 can rescue some but not all aspects of neural development [112], and similarly kinase dead EphB receptors can compensate for defective retinal axon pathfinding in EphB2/B3 null mice [223], or interact with NMDA receptors to promote synapse formation [78]. EphB2 kinase-dependent and -independent roles are also reported in the intestinal epithelium, where enhanced proliferation is communicated kinase-dependently through Abl-cyclin D1 signalling, while cell positioning is mediated kinase-independently via the phosphatidylinositol 3′ -kinase (PI3K) pathway [198]. PI3K was previously found to promote integrin-mediated adhesion downstream of EphA8 but autonomous of Eph kinase activity [224].
Consistent with the notion of this switch in function, Eph receptors can promote cell adhesion and de-adhesion within the same cell type depending on the density of the interacting ephrin. It has been demonstrated early on that a certain ephrin concentration threshold is required for HEK293 cells to respond with retraction, while in the same cells low ephrin density promotes integrin-mediated ECM attachment [225]. In agreement, ephrin-A2 stimulation enhances axon outgrowth at low concentrations but inhibits it at higher concentrations [226], while kinase-deficient EphB6 signals cell adhesion and migration at low ephrin-B2 concentrations, and retraction and inhibited migration at high concentrations, where it is phosphorylated by the associated Src family kinase Fyn [227]. Not only ligand concentration seems to be a determining factor but in addition also the total amount of displayed ligand, as demonstrated in a quantitative analysis of axon outgrowth [228].
The examples above all suggest a reoccurring theme where active kinase signalling, initiated by sufficient ligand levels, is required for Eph-controlled cell de-adhesion [229]. This concept is substantiated by the observation that high Protein Tyrosine Phosphatase (PTP) activity, controlling tyrosine kinase activation and phosphorylation, reverses retraction of an Eph-expressing cell from an ephrin neighbour and causes cell adhesion; in the case of EphA3-positive leukaemia cells PTP inhibition reverses adhesion to an ephrin-A5 surface [230]. Substantial further evidence attests to PTP-regulation of Eph receptor signalling: PTP-RO controls the sensitivity of retinal axons to ephrin stimulation by negatively regulating EphA4 receptor phosphorylation [231], while EphB2 activation is controlled by the leukocyte common antigen related receptor tyrosine phosphatase (LAR-1) [62]. Furthermore the prototypical phosphatase PTP1B regulates EphA3, and probably EphB2, activation and trafficking, and modulates EphA3-driven cell contraction and segregation [94].
Since Ephs are often co-expressed during normal development and in tumour cells, it is likely that the sum of adhesive and repulsive cues that are determined by the overall Eph receptor expression pattern on one cell relative to ephrin expression patterns on opposing cells relays the response resulting from their interaction. In prostate cancer cells, co-expression of EphA and EphB receptors can mediate either repulsion (inhibiting migration) or failure of repulsion (allowing migration), depending on the relative ligand density on the interacting fibroblasts [232]. Furthermore, co-clustering of EphA and EphB receptors results in signalling from both receptor types not requiring the presence of both ligands and with outcomes depending on the relative receptor expression, such that high expression of a kinase inactive EphA receptor can block EphB-mediated signalling, cell rounding and cell-cell segregation [57].
Disruption of Eph-ephrin complexes allows cell-cell repulsion to proceed
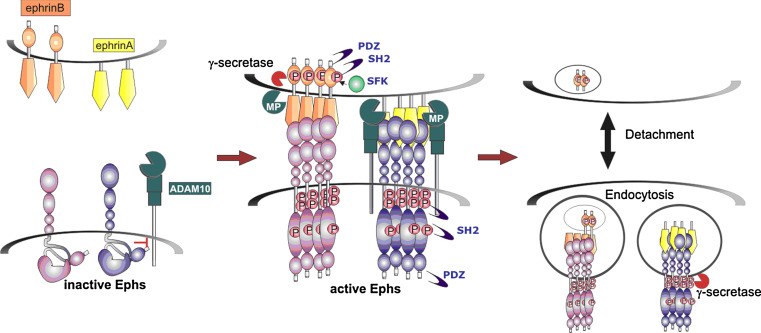
Eph/ephrin interactions form large clusters of high interaction avidity, which physically tether receptor- and ligand-expressing cells; this demands that cell repulsion can only proceed if this tether is disrupted. A number of studies now indicate a prominent role for the transmembrane metalloprotease ADAM (a disintegrin and a metalloprotease) 10, which sheds the extracellular domain of a number of ligands and/or receptors, and is also involved in TNF, Notch and EGF/ErbB signalling pathways, amongst others [233]. ADAM10 cleaves A-type ephrins following Eph receptor activation [234], controlled by association of ADAM10 with the Eph receptor, and specific binding and cleavage of the Eph-bound ephrin in trans (i.e. from the adjacent cell) [235]. Co-clustering of ADAM10 with EphAs relies on ephrin-induced Eph clustering [236], while ephrin cleavage is regulated by a conformational change that occurs upon Eph activation, where the inhibitory interaction between the juxtamembrane and kinase domains is released (Fig. 2, [48]): this promotes a productive association with ADAM10 by overcoming steric hindrance by the raised kinase domain in the inactive conformation. Thus, a receptor with an open, activated conformation promotes ADAM-mediated ephrin cleavage irrespective of kinase activity, as does a receptor lacking the entire intracellular domain [237]. Such a mechanism explains previous conflicting evidence for an intact function of ADAMs lacking their cytoplasmic domain [237, 238], despite the well-established dependence of ADAM-mediated shedding of a number of cell surface proteins on intracellular kinase activity [233, 239], and may indicate a broadly applicable mechanism in particular for shedding of RTK ligands.
Fig. 2.
Disruption of Eph/ephrin cell-cell interactions during cell-cell segregation. Engagement of Ephs and ephrins results in large, high affinity complexes that require disruption to allow cell-cell detachment. For ephrin-As this occurs through ephrin cleavage by the ADAM10 metalloprotease, which recognises the Eph/ephrin complex to release ephrin from the adjacent cell. ADAM10 association with EphAs is promoted by clustering and conformational change of the Eph receptor, to stimulate ephrin shedding. Metalloprotease (MP) cleavage of ephrin-Bs has also been described, as has γ-secretase-mediated release of the intracellular domain of both ephrin-Bs and Ephs. Alternatively, transendocytosis of the entire EphB/ephrin-B complex has also been described [248, 249]
Interestingly, as in Notch and ErbB signalling, ADAMs appear to shed both receptors and ligands in regulating Eph signalling, but under different conditions. Thus, whereas cell-bound ligand induces ADAM-mediated ligand cleavage and receptor endocytosis, stimulation of calcium influx in the absence of ligand is thought to induce ADAM-mediated EphB2 extracellular domain shedding, followed by γ-secretase cleavage of the intracellular domain to release a cytosolic fragment capable of regulating NMDA receptor signalling [240]. These sequential cleavage events, first established for Notch1 and cadherin [241], can also be applied to ephrin-B1 and ephrin-B2, where shedding of the extracellular domain, which at least in some cases is mediated by ADAM13 [242], is subsequently followed by γ-secretase cleavage of the short intracellular domain [243, 244]. The released intracellular domain might function in ephrin-B reverse signalling, in particular in Src-family kinase activation [243]. Also EphA4 is cleaved by γ-secretase with the resulting released intracellular domain being important for Rac activation in dendritic spine formation [245]. Roles for other proteases in Eph/ephrin signalling have also been described, including cleavage of ligand-activated EphB2 by matrix metalloproteases MMP-2/MMP-8 [246], and cleavage within the transmembrane domain of ephrin-B ligands by the rhomboid serine protease RHBDL2 [247].
Protease-independent disruption of Eph/ephrin-mediated cell contacts has also been reported, implying ‘trans-endocytosis’ of the entire complex. Interactions of ephrin-B- and EphB-expressing cells lead to endocytosis of phosphorylated EphB primarily into receptor-expressing cells, but to a lesser extent also into ligand-expressing cells: detection of the ephrin or Eph cytoplasmic domain in respective opposing cells was thus interpreted as trans-endocytosis of the whole complex into either cell [248, 249]. This internalisation of either ligand or receptor into the ‘parental’ cell was dependent on Rac and cytoskeletal signalling and on an intact cytoplasmic domain: intracellular truncation of either ligand or receptor enhanced its uptake into the adjacent cell, while truncation of both blocked endocytosis and cell retraction, promoting adhesion [248, 249]. Thus, while different mechanisms may exist to achieve disruption and endocytosis of Eph-ephrin complexes at cell-cell junctions, there is a common dependence on intracellular, kinase-dependent signalling controlling endocytosis, necessary for cytoskeletal re-organisation and cell retraction.
Eph-regulated adhesion dynamics
Eph-cadherin crosstalk in cell-cell adhesion
In addition to the direct consequences of Eph/ephrin interaction, cross-talk of Ephs with adherence molecules is critically implicated in Eph-modulated cell-cell adhesion. Cadherins are responsible for homotypic and heterotypic cell-cell adhesion and also involved in cell sorting, and it is likely that the combined action of cadherin-mediated adhesion and Eph/ephrin signalling directs the final outcome during boundary formation, as was demonstrated for segmental patterning of neural-crest-derived sympathetic ganglia [250]. Likewise, spreading of EphB-expressing colorectal tumour cells into ephrin-B1-positive surrounding epithelial tissues is restricted by a compartmentalisation mechanism that relies on both EphB and E-cadherin function [197]. Moreover, EphB2-mediated collective cell migration during early nerve regeneration involves Sox2-dependent N-cadherin redistribution to cell-cell contacts [98]. Loss of E-cadherin expression leads to weakened cell-cell contacts and coincides with an altered Eph/ephrin expression pattern in embryonic stem cells [251], and alters EphA2 membrane localisation in mammary epithelial cells, embryonic stem cells and aggressive melanoma cells during vasculogenic mimicry [201, 251, 252]. Correspondingly, inhibition of β-catenin/TCF-mediated transcription downstream of E-cadherin resulted in down-regulation of EphB2 and EphB3, while ephrin-B1 was up-regulated [95, 253]. Interestingly, at EphB/ephrinB epithelial boundaries ADAM10 sheds colocalised E-cadherin, allowing segregation of the two cell populations while relative adhesiveness within the populations is increased [254]. Thus, de-regulated Eph-cadherin crosstalk might contribute to the tumorigenic potential of Eph/ephrin-positive cancer cells through alterations of cell-cell adhesion [255, 256].
Ephs and integrin-mediated cell-matrix adhesion
A variety of studies show that Eph receptors can affect extracellular-matrix (ECM) adhesion and migration by modulating integrin signalling, although both positive and negative effects have been described (summarised in Table 1). This is possibly due to distinct effects depending on the strength of ephrin-Eph interactions, since increasing ephrin density results in a biphasic pattern of cell attachment on ECM proteins, enhancing attachment at lower concentrations and inhibiting at higher ligand density [34, 225]. This is consistent with changes from adhesion to de-adhesion at higher Eph receptor activation levels, promoting cytoskeletal collapse rather than cell spreading. In accord with this (kinase-dead) EphA8-enhanced adhesion is kinase-independent [224], while EphB3 inhibition of integrin-dependent adhesion is kinase dependent [257]. Eph-mediated inhibition of integrin activity involves changes in integrin conformation and clustering [258, 259], as well as modulation of focal adhesion signalling (below). Also, EphA2 was recently reported to promote cancer cell migration through ECM in a ligand-independent manner via Akt-mediated serine phosphorylation, which was opposed by ligand-induced tyrosine phosphorylation [260]. In addition, EphB4 expression was reported to inhibit cell-substrate adhesion and migration by down-regulating β1-integrin protein levels in breast cancer cells [40]. Lastly, a direct interaction between fibronectin and EphA1 was shown to promote cell-spreading and endothelial tube formation in an Eph-kinase-independent manner [261].
Table 1.
Eph signalling regulates integrin activity

FN fibronectin, VSMCs vascular smooth muscle cells
Ephs and modulation of focal adhesion components
An essential event in ECM detachment and cell migration is the initiation of focal adhesion turnover and the involvement of focal adhesion modulators such as FAK, paxillin, p130Cas and Src-family kinases (SFKs) during Eph signalling is widely acknowledged (see summary in Table 2). Although evidence is conflicting as to whether activation of these proteins is up- or down-regulated upon ephrin stimulation, in general their involvement correlates with adhesive or de-adhesive cell responses. For instance, FAK and p130Cas were found phosphorylated in stimulated EphA2-expressing murine fibroblasts and are required for ephrin-A1-induced cell spreading [31]. Likewise, FAK and paxillin phosphorylation was observed during adhesion of LK63 leukemic cells to ephrin-A5 [230], while in PC3 cells FAK phosphorylation and association are lost upon EphA2 stimulation, concurrent with cell contraction [259]. However, decreased FAK phosphorylation observed in colorectal cancer cells seems to be transient and is recovered within 10 min after stimulation [95], and a recent phospho-proteomics screen confirmed increased paxillin and FAK phosphorylation in EphB2-expressing cells interacting with ephrin-B1-expressing cells [56].
Table 2.
Change in focal adhesion (FA) modulators during ligand-stimulated Eph signalling

If not noted otherwise PY-FAK refers to Y397, PY-Src to Src Y418 (m)/Y416 (h) and PY-paxillin to paxillin PY31
FA modulators in bold apply to all references in groups
aEffect caused by receptor overexpression, ligand-independent
Eph-directed reorganisation of the actin cytoskeleton
Eph-mediated control of cell-cell adhesion and repulsion depends not only on interactions at cell-cell contacts but also on global control of their cytoskeletal function, via adaptor proteins and small GTPases that regulate actin reorganisation (for overview see Fig. 3). For example, the SH2/SH3 adaptor proteins of the Crk family, which have a well-documented function in cell migration, adhesion and actin reorganisation, are involved in the regulation of cell repulsion and membrane ruffling during Eph signalling [33, 262]. CrkII recruitment to ligand-activated EphA3 and ensuing RhoA signalling are required for cell repulsion in EphA3/HEK293T and melanoma cells [33], while Crk is recruited to nascent focal complexes by p130Cas and essential for Rap1-mediated stabilisation of these complexes and Rac1-induced membrane ruffling during Eph signalling in human aortic endothelial cells [262]. Similarly, in vascular smooth muscle cells the Crk-p130Cas pathway is important for the modulation of focal adhesions and directional cell migration during bi-directional ephrin-B2/EphB signalling [140]. In ephrin-A1-stimulated PC3 prostate cancer cells, Abl-CrkII-C3G signalling inhibits Rap1 activation and cooperates with the Rho-ROCK1 pathway induced by serum factors to facilitate cell detachment [263]. Interestingly, Abl-mediated Crk phosphorylation mediates the tumour suppressor function of ephrin-B2-activated EphB4 in breast cancer cells partly by attenuating cell motility and invasion, via inhibition of Crk/p130Cas complex formation and potentially Rac1 activation [176].
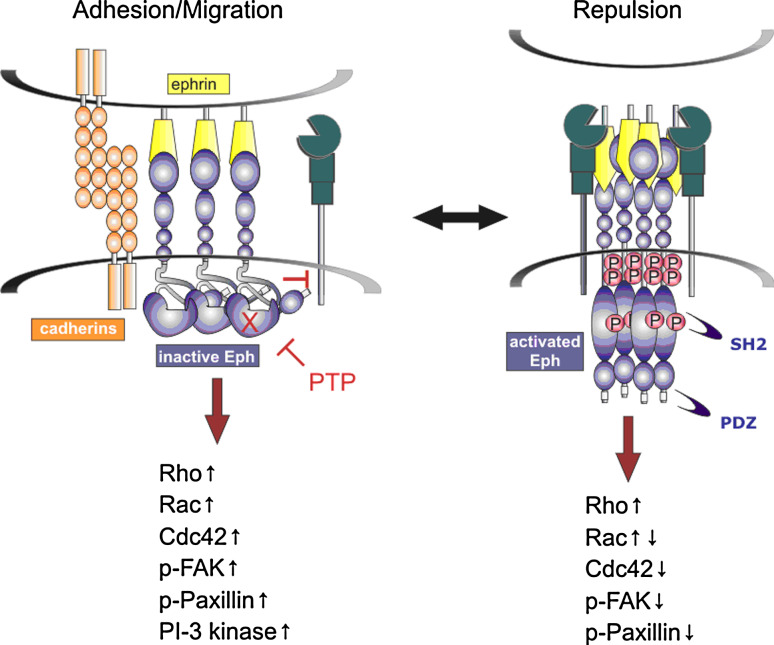
Fig. 3.
Eph-mediated cell-cell adhesion versus repulsion. Cell-cell adhesion occurs under conditions of low Eph kinase activity, such as at low Eph and/or ephrin density, high protein tyrosine phosphatase (PTP) activity or inactivating Eph mutations (X). Under such conditions kinase-independent Eph signalling promotes actin-cytoskeleton-mediated cell spreading and attachment, facilitated by focal adhesions and co-operation with other adhesive molecules including cadherins. In contrast robust Eph activation at high Eph/ephrin densities promotes disruption of Eph/ephrin complexes, cell detachment, and Rho-GTPase-mediated actin-cytoskeleton collapse and cell rounding. (↑activation, ↑↓ transient activation, ↓ inactivation). In epithelial cells, ADAM10-mediated cleavage of E-cadherin also contributes to ephrin-B/EphB-mediated cell segregation [254]
Nck SH2/SH3 adaptor proteins also link RTK and other molecules like p130Cas to modulators of the cytoskeleton [264]. Nck binds to the juxtamembrane tyrosines of activated EphB and EphA receptors [265, 266] and likely recruits paxillin [267]. Selective inactivation of Nck in the developing nervous system results in similar neuronal guidance defects as seen in EphA4−/− mice [268], and Nck has also been shown to contribute to the inhibitory effect of EphA3 on cell migration [269]. Signalling network modelling from mass spectrometric analysis of tyrosine phosphorylation events during EphB2/ephrin-B1-mediated cell segregation also identified Nck, as well as SHC1 and SHB, another adaptor protein not previously described in Eph signalling, which likely links EphB2 with Crk, RasGAP (RASA1) and the PI3K regulatory subunit p85α (PIK3R1) [56].
Dynamic changes in Rho GTPase activity during Eph signalling
As indicated above, the Rho family GTPases—including the most commonly known members RhoA, Rac1 and Cdc42—plays a major role in Eph kinase signalling [270]. Like other GTPases, Rho GTPases act as molecular switches that cycle between an active GTP-bound form and an inactive GDP-bound form regulated by guanine nucleotide exchange factors (GEFs) elevating GTP loading and GTPase-activating proteins (GAPs) that stimulate the intrinsic GTPase activity that hydrolyses GTP to GDP [271]. Transient RhoA activation is crucial for actin contractility and disassembly to facilitate repulsion downstream of activated Eph receptors (see Table 3), and can be mediated indirectly through FAK and/or Crk [31, 33, 262, 272] or directly through GEFs (see below). RhoA commonly signals via ROCK (Rho-associated kinase) during Eph activation, and is important for EphB2/ephrin-B1-mediated cell segregation in Xenopus animal cap explants and dendritic spine remodelling, via LIMK-1 [37, 273, 274]. Another possible link between EphA1 and RhoA/ROCK signalling could be integrin-linked kinase (ILK), which has been suggested to interact via its ankyrin region with the SAM domain of EphA1 and modulate ephrin-induced cell contraction [275]. The ILK/PINCH/Parvin complex negatively regulates actomyosin contractility directly downstream of β-integrins [276], by which it may also affect Eph receptor signalling.
Table 3.
Changes in activity of Rho GTPases in Eph downstream signalling

VSMCs vascular smooth muscle endothelial cells, HUVECs human umbilical cord venous endothelial cells, Dsh dishevelled, FA focal adhesion
RhoGTPases in bold apply to all references in groups
In some instances RhoA signalling alone appears insufficient for cytoskeletal contraction [172], and co-operation with Cdc42 signalling through myotonic dystrophy-related Cdc42 kinase (MRCK) may be required for full contraction [277]. In general, however, Cdc42 appears to function in opposition to RhoA, with its activity being more associated with cell adhesion and migration (Table 3): in PC3 prostate cancer cells it is thought to facilitate EphB3/4-mediated migration and lack of contact inhibition, as compared to Rho-mediated ephrin-A-stimulated retraction [232]. Previously, Cdc42 has been implicated in regulating actin polymerisation during EphB-mediated dendritic spine morphogenesis [278], while in Xenopus blastomere adhesion, in addition to down-regulated RhoA activity, Cdc42 function was reported to be indirectly reduced via sequestration by xPAK1 activated downstream of EphA4 [279].
In addition, Eph receptor signalling also modulates the activity of Rac1, which, after transient activation, is typically down-regulated during ephrin-stimulated cell retraction (Table 3), for example during RhoA activation in ephrin-A5-activated retinal ganglion cells [274]. Thus, ephrin-B1 stimulation of CRC and kidney epithelial cells resulted in Rac1/CDC42 inhibition and increased Rho signalling [257]. However, Rac is required for endocytosis and actin re-organisation during ligand-induced nerve growth cone collapse [280], suggesting that Rac1 needs to be activated, at least temporarily. Upon RTK phosphorylation, Rac1 activation stimulates NADPH-oxidase-mediated production of reactive oxygen species (ROS), which is thought to be responsible for allowing sustained phosphorylation and signalling by inactivating associated PTPs [281]. Rac-induced ROS generation is likely implicated in cross-talk between integrins and RTKs, potentiating downstream signalling and effects such as cell adhesion and spreading [282]. Thus, ephrin-A1 stimulation in PC3 cells reportedly decreases Rac activation, inhibiting integrin-induced ROS production, and indirectly leading to increased Rho activation and thus inhibited spreading [283]. On the other hand, prolonged Rac1 activation reportedly is associated with adhesion and migration, is promoting vascular assembly of endothelial cells [143] and migration and invasion of breast cancer cells [284]. The importance of Rac1 for Eph/ephrin-mediated cell segregation was confirmed by phosphoproteomics, which identified a significant number of proteins involved in regulating Rac1 activity, such as the GEF complex DOCK180/ELMO and GAPs like α- and β-chimerin [56].
GEFs and GAPs of Rho family GTPases involved in Eph signalling
As would be anticipated, GEFs and GAPs are critical in regulating Rho GTPase activities downstream of Eph receptor signalling. Ephexin-1, a member of the Dbl family of Rho family GEFs, is activated after EphA4 stimulation and leads to growth cone collapse in retinal ganglion cells [285, 286]. Tyrosine phosphorylated ephexin-1 has preferential activity towards RhoA that changes the overall balance between RhoA, Cdc42 and Rac1 activities, while in the absence of EphA4 activation, ephexin-1 activates all three Rho GTPases to promote neurite extension [285, 286]. Activation of ephexin downstream of EphA receptors might occur via SFKs during retinal axon repulsion [287] or via Cdk5 in dendritic spine retraction [130]. In addition to ephexin-1, other ephexin family members, such as the vascular smooth muscle cell-specific Vsm-RhoGEF [288] and ephexin-4 [284], have also been implicated in Eph signalling. Thus, in response to EGF stimulation, EphA2 interacts with ephexin-4 acting as a GEF for RhoG, which in turn activates Rac1 via ELMO/DOCK4 and promotes breast cancer cell migration and invasion [284]. Vav proteins, also members of the Dbl family acting downstream of many RTKs, are also implicated in ephrin-induced growth cone collapse [289] and thought to facilitate Rac1 activation during EphA2-induced angiogenesis [290]. Other GEFs connecting Eph activation and RhoGTPase/PAK1 downstream response during EphB-facilitated dendritic spine morphogenesis are the Cdc42-GEF intersectin-1 [278] and the Rac1 GEF kalirin [128], while the Rac1 GEF Tiam1 facilitates neurite outgrowth in ephrin-B1-mediated reverse and EphA2-mediated forward signalling [291].
Examples of negative Rho GTPase regulators implicated in Eph signalling include the Rac-specific RhoGAPs α- and β-chimerin: α2-chimerin binds to EphA4, is activated by ephrin-B3 stimulation and is essential for corticospinal axon guidance during the development of motor circuits [292–295]. Interestingly, loss of α-chimerin function in mice mimics the characteristic hopping gait phenotypes of EphA4 and ephrin-B3 knock-out animals [292, 293, 295]. However, α-chimerin does not seem to impact on EphA4-mediated axon guidance within the anterior commissure [296]. β2-Chimerin also binds to the kinase domains of EphA2 and EphA4, and has been proposed to act in a similar way as α2-chimerin [297]. In addition, LMW-PTP-mediated dephosphorylation of p190RhoGAP leads to RhoA activation, important in destabilisation of adherens junctions [298].
Ras family of small GTPases
Although the Ras family GTPases, including H-Ras, R-Ras and Rap amongst others, are best known as regulators of proliferation, they also have documented roles in regulating adhesion and migration, for example via integrin activation [299]. Indeed, during Eph signalling the Ras-Raf-MEK-MAPK cascade functions not only to modulate cell proliferation, but also to influence cell adhesion and axon guidance (see Table 4). For example, the down-regulation of Ras activity via p120RasGAP is required for retraction of NG108 neuronal cells [300, 301]. R-Ras phosphorylation was also suggested to be essential for EphB2 activation-induced loss of integrin-mediated adhesion [302]. Further, it was proposed that R-Ras function during EphB2 signalling is regulated through two parallel mechanisms: down-regulation of Ras activity via p120RasGAP, which is sufficient to trigger COS7 cell retraction, while both decreased Ras activation level and tyrosine phosphorylation are needed to mediate cell migration and growth cone collapse [303]. Interestingly, R-Ras was also phosphorylated and vital in promoting invasion in EphA2-positive glioma cells [304]. SHEP-1, which contains a SH2 domain interacting with activated Eph and ErbB receptors, and an inactive RasGEF domain that binds Ras and Cas proteins [305], was suggested to link Eph receptors and Ras and Rap1 [306]. Rap1 inactivation synergises with activated RhoA to signal cell repulsion of ephrinB1-stimulated EphB2-overexpressing colon carcinoma cells [307] and ephrinA-mediated growth cone collapse is facilitated by the GTPase-activating protein spine-associated RapGAP (SPAR) [308]. In some cases ephrin stimulation can also promote Rap1 activity, mediating adhesion rather than repulsion by stabilising nascent focal complexes in human aortic endothelial cells [262], and MAP kinase-dependent neurogenesis [309].
Table 4.
Eph-modulated cytoskeletal responses involve altered activity of Ras family GTPases

Ephrin-mediated reverse signalling and co-ordination with Eph forward signalling
Ephrin signalling is not as well understood as that of Eph receptors; however, recent studies identified several analogies between Eph- and ephrin-mediated cytoskeletal responses [3, 310, 311] (for a summary refer to Table 5). For example, ephrin-mediated cell retraction and adhesion have been described, and it is likely that mechanisms underlying the switch between co-ordinated cell-cell adhesion and segregation rely on intricately linked Eph forward and ephrin reverse signalling. Emerging evidence for this notion comes from quantitative mass spectrometry analysis of interacting EphB2- and ephrin-B1-expressing HEK293 cells, which revealed different networks being activated in EphB2-expressing cells, depending on the presence or absence of the ephrin-B1 intracellular signalling domain [56]. In agreement, also mutation of the C-terminal ephrin-B1 PDZ-binding motif also influences EphB2 signalling in neighbouring cells [56], implying that the response in each of the interacting Eph- and ephrin-expressing cells reflects the signalling capacity in both cells. Considering these findings it may be instructive to review the concept of ‘unidirectional’ Eph/ephrin signalling that has been used to describe functions of kinase-deficient Ephs, such as EphB6 lacking a functional kinase domain [53] and cytoplasmic-truncated splice variants of EphA7 [221]. Not only do these kinase-deficient Ephs continue to act as signalling hubs, typically conferring cell-cell adhesion rather than retraction, but are likely altering the signalling in interacting ephrin cells.
Table 5.
Ephrin-mediated cytoskeletal dynamics

p75NTR p75 neurotrophin receptor, Dsh-PCP dishevelled/planar cell polarity pathway, MET mesenchymal-to-epithelial transition
aEffect caused by ephrin overexpression, Eph stimulation-independent
Further evidence for this concept of co-regulation is implicit in the need for co-ordinated endocytosis of ephrin and Eph that is facilitated by the disruption of the Eph-ephrin complex. Just as endocytosis is an important step in Eph and other RTK signalling, endocytosis of ephrins is similarly critical for cell retraction and cytoskeletal signalling [248, 249]. Ephrin-induced Eph receptor phosphorylation, which signals endocytosis of Eph clusters, at the same time signals ADAM10-mediated ephrin cleavage to facilitate Eph/ephrin internalisation, so that indeed a kinase deficient Eph blocks ephrin cleavage [237]. This would suggest that Eph kinase-dependent ephrin cleavage simultaneously may co-ordinate endocytosis of ephrin signalling complexes within the ephrin-bearing cell, to ensure coordinated induction and progression of cell-cell segregation in both cell populations.
Since ephrin signalling induces cytoskeletal changes comparable to those seen upon Eph forward signalling, involved signalling pathways are likely to be similar. Interestingly, proteomics analysis showed asymmetric tyrosine phosphorylation profiles in EphB2 and ephrin-B1 interacting cells for some but not all signalling proteins, predominantly affecting those that were shown to be essential for cell sorting [56]. Such an asymmetry is perhaps not surprising given the distinct localisation of Ephs and ephrins to different domains within the plasma membrane, as shown for EphAs and ephrin-As, that are co-expressed in spinal cord motor neurons [312]. Indeed, unlike Ephs, ephrin-Bs and ephrin-As are both harboured in detergent resistant membrane microdomains or lipid rafts, although there is some evidence that their association with these domains differs to some degree [313, 314]. Since ephrin-As are membrane-tethered via a GPI anchor and do not contain an intracellular domain like ephrin-Bs, their localisation to these discrete membrane microdomains appears necessary for their signalling capacity to facilitate co-localisation with SFKs [314, 315]. It is interesting in this context to note that in medial spinal cord neurons EphA3 and ephrin-A5 seem to colocalise to the same membrane patches, while in lateral spinal cord neurons they occupy distinct membrane domains [316].
Ephrin-B signalling
The transmembrane ephrin-B ligands contain a cytosolic domain with five conserved tyrosine residues, a proline-rich- and a C-terminal PDZ-domain binding motif [3, 311]. In response to Eph-ligation, ephrin-Bs are clustered and phosphorylated on the conserved tyrosines [317, 318]. Src family kinases, such as Src and Fyn, are thought to be critical for this phosphorylation and thus the biological functions of B-type ephrins [317–319]. For example, Src-dependent ephrin-B2 phosphorylation is required for in vitro assembly of endothelial cells into vascular structures [149], whereby MMP/γ-secretase-mediated cleavage of ephrin-Bs appears to be required for Src activation [243]. In addition, crosstalk with RTKs, such as Tie-2 and PDGFR, shown in vitro [133, 317] and FGFR in Xenopus embryos in vivo [320], leads to ephrinB phosphorylation.
One of the important adaptors recruited to activated ephrin-B1 via SH2-domain-dependent interactions is Grb4, mediating FAK activation and cell rounding [321]. Several PDZ domain-containing proteins, including PICK1, GRIPs and syntenin, were reported to bind the C-terminal PDZ binding motif of ephrin-Bs [124, 322, 323]. Furthermore, the GAP PDZ-RGS3 associates with activated ephrin-B1 and negatively regulates SDF-1-induced G-protein coupled signalling [324]. Ephrin-B1 tyrosine phosphorylation is negatively regulated through PDZ-dependent recruitment of the phosphatase PTP-BL, which concurrently dephosphorylates and thus inactivates Src [319]. Interestingly, effects on cell contraction and (lymph) angiogenic sprouting [145, 146, 154, 325] seem to be independent of the five conserved ephrin-B tyrosines but rely on PDZ-domain interactions. Similarly, phosphotyrosine-independent ephrin-B2 signalling occurs during embryonic midline development and relies on PDZ domain function [326], and both tyrosine-dependent and -independent ephrin-B3 signalling contribute to post-synaptic neuron maturation [327].
In addition to Eph-ephrin-stimulated signalling, ephrins also bind other proteins and can therebymodulate diverse signalling pathways. Ephrin-B2 co-clustering with VEGFR2 [145] is essential for VEGFR internalisation and biological function in endothelial cells [145, 146], while in turn ephrin-B2 expression is regulated by VEGF and hypoxia [147, 328]. Ephrin-B2 and -B3 also act as receptors for the Hendra and Nipah viruses [329, 330], and similarly EphA2, together with the EGF receptor, can act an entry cofactor for the hepatitis C virus [331]. In addition, ephrin-B3, as well as ephrin-A3, binds sulphated proteoglycans [332, 333], including heparin sulphate, required for efficient ephrin-mediated cell-rounding and growth cone collapse.
Ephrin-A signalling
Despite lacking a cytoplasmic domain, there is little doubt that signalling by GPI-linked A-type ephrins modulates cell adhesion and morphology. However, the underlying signalling concepts are poorly understood. While recruitment of Fyn to clustered ephrin-A5 has been demonstrated, the mechanism leading to MAPK pathway activation and increased cell adhesion remains to be identified [315, 334]. Also in T cells, where EphA2-induced ephrin-A activation results in the phosphorylation of Lck, Fyn and Akt, and inhibits antigen receptor-induced apoptosis, signal transduction from the ephrin signalling cluster remains undefined [335]. While the aforementioned co-localisation of ephrins and SFKs to similar membrane microdomains likely facilitates signalling upon ephrin clustering, transmembrane proteins are also thought to be important. Thus, in retinal ganglion cells, Ephrin-A-mediated clustering of lipid rafts and associated SFKs likely involves co-clustering with the neurotrophin receptor p75 (p75NTR), required for EphA7 and Fyn-mediatedaxon retraction [336]. Accordingly, recruitment of the neurotrophin receptor TrkB into ephrin-A5 clusters seems to modulate BDNF-promoted retinal axon branching [337].
Ephrin-modulation of adhesion and repulsion
As with Eph receptors, ephrin clustering can modulate integrin-mediated adhesion. For example, activation of ephrin-A5 increases cell attachment and neurite outgrowth, which depends on β1-integrin and involves prolonged ERK activation [334]. In addition, ephrin-B2 is highly expressed in the invasive front of advanced malignant melanoma, and its overexpression in mouse melanoma cells was suggested to increase migration and β1 integrin-mediated adhesion [338]. In endothelial cells, ephrin-B1 signalling activates integrin αvβ3- and α5β1-mediated adhesion and migration via JNK [339], while in ephrin-B1-overexpressing HEK293 cells JNK activation results in cell rounding, an effect that is apparently independent of ephrin-B1 phosphorylation or PDZ interactions [340]. Moreover, integrin and ephrin-B2 reverse signalling act in co-operation during somitogenesis in developing zebrafish [102, 341].
Not surprisingly, small GTPases are also involved in ephrin signalling, including Rap1, which together with PI3K and PLCβ facilitate platelet aggregation [342]. In Xenopus, ephrin-B1 stimulation leads to downstream activation of Rho and ROCK via dishevelled, a scaffold protein implicated in the Wnt signalling pathway [37, 343]. This mechanism appears to be critical for repulsion and segregation of EphB2- and ephrin-B1-positive cell populations [37]. Recruitment of Grb4 to phosphorylated ephrin-Bs is important to relay cytoskeletal effects, including regulating focal adhesions and actin stress fibres concurrent with cell rounding [321] and dendritic spine morphogenesis via G-protein-coupled receptor kinase-interacting protein (GIT) [129]. Activation of Rac1 and its effector PAK is also communicated by Grb4-mediated recruitment of the GEF DOCK180 resulting in axon retraction and pruning [344]. Rac1 further regulates EphB2-induced invasion of ephrin-B3-positive glioblastoma cells in vitro and in vivo [345] in a similar fashion to EphB2-mediated promotion of glioma cell invasion [304]. Parallels between Eph and ephrin cytoskeletal signalling via Src and PI3K pathways were similarly reported for ephrin-B2-controlled cell migration of retinal endothelial cells [346] and EphB4 response in microvascular endothelial cells [347]. Interestingly, ephrin-B2-mediated down-regulation of Cdc42 activity was suggested to induce mesenchymal-to-epithelial transition and gap induction during chick somitogenesis [348].
More recently, a cell-autonomous function of ephrin-B2 signalling was proposed in endothelial cells [325]. Ephrin-B2 overexpression in endothelial cells caused enhanced but less directional cell migration, and Eph-independent cell shape oscillations, which relied on ROCK and JNK activation and actomyosin contractility and seemed to be specific for B-class ephrins [325]. Similarly, ephrin-B2 was reported to control cell motility and adhesion in vSMCs independently of cell-cell contact [140]. Another cell-autonomous function reported for ephrin-B involves ephrin-B1 phosphorylation by FGFR, modulating the planar cell polarity pathway and thereby regulating the movement of retinal progenitors into the Xenopus eye field [349, 350].
Ephrin-B1 is additionally involved in manipulating gap junction communication by regulating connexin43 distribution [99] and the regulation of cell-cell adhesion and tight junctions, for instance via the interaction with claudins in epithelial and cancer cells [351] or through the Par polarity complex during early Xenopus development [352]. Interestingly, in the latter example, overexpressed ephrin-B1 acts by competing with Cdc42 for association with Par-6, a major scaffold of the Par polarity complex, causing loss of tight junctions while ephrin-B1 phosphorylation on Y310 disrupts this interaction. Par-6 is important for activation of atypical PKC indicating that maintenance of tight junction integrity during normal development requires ephrin-B1 phosphorylation [352]. The authors further propose a model where loss of Eph/ephrin interaction might contribute to increased tumour invasion [353]. However, ephrin reverse signalling has also been implicated in promoting cancer cell migration and invasion. Ephrin-A5 overexpression in NIH3T3 fibroblasts increases their invasive potential, branching morphogenesis in matrigel and anchorage-independent growth [354] while ephrin-B signalling modulates invasiveness of T-ALL [355], melanoma [338] and glioma cells [345, 356], which might additionally be fostered by ephrin-induced MMP-8 secretion, as has been described for pancreas carcinoma cell lines [357].
Concluding remarks
The Eph/ephrin signalling system has emerged as one of the principle regulators of cell-cell interactions that guide tissue patterning during development. Re-emerging in many cancer types, Ephs and ephrins are mainly implicated in tumour cell migration and invasion, neoangiogenesis and metastasis. The diametrically opposing outcomes of their activities in normal development, leading to cell-cell adhesion or de-adhesion and segregation, are matched during oncogenesis by their apparently opposing roles in promoting or suppressing tumour progression in different cancers. Clearly, Eph kinase activity is essential in determining the direction of the outcome: cell-cell repulsion depends on robust Eph kinase activation and cytoskeletal re-arrangement, while Eph kinase-independent signalling promotes adhesion and/or cell migration and invasion. Thus, somatic mutations of Ephs, which likely effect receptor clustering and activation, have been described in a range of highly metastatic cancers, suggesting that tumour progression favours change-of-function rather than loss-of-function mutations. It may be that increased levels of Eph receptors in some instances necessitate their inactivation for tumour cell survival or that the change of function actively promotes tumour progression. However, the correlation of Eph receptor expression and tumorigenicity in the absence of mutation in many cancers suggests that in some cases Eph activity is directly tumour promoting, advising caution to over-generalise. Despite the diversity of functions reported for Ephs in various cancers, their frequent overexpression and their implication in the progression of most metastatic cancers suggest Ephs as attractive prognostic markers and promising targets for cancer therapeutics. The effectiveness of such therapeutics, however, will depend on improving our emerging understanding of the distinct Eph functions in specific cancer types.
Abbreviations
- ADAM10
A disintegrin and metalloprotease 10
- ALL
Acute lymphoblastic leukaemia
- Cas
Crk-associated substrate
- CRC
Colorectal cancer
- Crk
CT10 regulator of kinase
- ECD
Extracellular domain
- ECM
Extra-cellular matrix
- EGFR
Epithelial growth factor receptor
- FAK
Focal adhesion kinase
- GAP
GTPase activating protein
- GEF
Guanine nucleotide exchange factor
- GPI
Glycosyl phosphatidylinositol
- JNK
c-Jun N-terminal kinase
- MMP
Matrix metalloprotease
- NMDA
N-methyl d-aspartate
- PBM
PDZ-binding motif
- PDZ
Postsynaptic density protein/discs large/zona occludens protein (domain)
- PH
Pleckstrin homology (domain)
- PI3K
Phosphatidylinositol 3′-kinase
- PLC
Phospholipase C
- PTB
Phospho-tyrosine binding (domain)
- PTP
Protein tyrosine phosphatase
- PY
Phospho-tyrosine
- ROCK
Rho-associated coiled-coil-containing protein kinase
- ROS
Reactive oxygen species
- RTK
Receptor tyrosine kinase
- SAM
Sterile alpha motif
- SH2
Src homology 2 (domain)
- TNFα
Tumour necrosis factor α
Contributor Information
Martin Lackmann, Email: martin.lackmann@monash.edu.
Peter W. Janes, Email: peter.janes@monash.edu
References
- 1.Himanen J-P, Saha N, Nikolov DB. Cell-cell signaling via Eph receptors and ephrins. Curr Opin Cell Biol. 2007;19:534–542. doi: 10.1016/j.ceb.2007.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lackmann M, Boyd AW (2008) Eph, a protein family coming of age: more confusion, insight, or complexity? Sci Signal 1: re2 [DOI] [PubMed]
- 3.Pasquale EB. Eph–ephrin bidirectional signaling in physiology and disease. Cell. 2008;133:38–52. doi: 10.1016/j.cell.2008.03.011. [DOI] [PubMed] [Google Scholar]
- 4.Merlos-Suárez A, Batlle E. Eph–ephrin signalling in adult tissues and cancer. Curr Opin Cell Biol. 2008;20:194–200. doi: 10.1016/j.ceb.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 5.Genander M, Frisén J. Ephrins and Eph receptors in stem cells and cancer. Curr Opin Cell Biol. 2010;22:611–616. doi: 10.1016/j.ceb.2010.08.005. [DOI] [PubMed] [Google Scholar]
- 6.Pasquale EB. Eph receptors and ephrins in cancer: bidirectional signalling and beyond. Nat Rev Cancer. 2010;10:165–180. doi: 10.1038/nrc2806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Janes PW, Adikari S, Lackmann M. Eph/ephrin signalling and function in oncogenesis: lessons from embryonic development. Curr Cancer Drug Targets. 2008;8:473–479. doi: 10.2174/156800908785699315. [DOI] [PubMed] [Google Scholar]
- 8.Amit I, Wides R, Yarden Y. Evolvable signaling networks of receptor tyrosine kinases: relevance of robustness to malignancy and to cancer therapy. Mol Syst Biol. 2007;3:151. doi: 10.1038/msb4100195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Drescher U. Eph family functions from an evolutionary perspective. Curr Opin Genet Dev. 2002;12:397–402. doi: 10.1016/S0959-437X(02)00316-7. [DOI] [PubMed] [Google Scholar]
- 10.Suga H, Hoshiyama D, Kuraku S, Katoh K, Kubokawa K, Miyata T. Protein tyrosine kinase cDNAs from amphioxus, hagfish, and lamprey: isoform duplications around the divergence of cyclostomes and gnathostomes. J Mol Evol. 1999;49:601–608. doi: 10.1007/PL00006581. [DOI] [PubMed] [Google Scholar]
- 11.George SE, Simokat K, Hardin J, Chisholm AD. The VAB-1 Eph receptor tyrosine kinase functions in neural and epithelial morphogenesis in C. elegans . Cell. 1998;92:633–643. doi: 10.1016/S0092-8674(00)81131-9. [DOI] [PubMed] [Google Scholar]
- 12.Wang X, Roy PJ, Holland SJ, Zhang LW, Culotti JG, Pawson T. Multiple ephrins control cell organization in C. elegans using kinase-dependent and -independent functions of the VAB-1 Eph receptor. Mol Cell. 1999;4:903–913. doi: 10.1016/S1097-2765(00)80220-8. [DOI] [PubMed] [Google Scholar]
- 13.Eph Nomenclature Committee Unified nomenclature for Eph family receptors and their ligands, the ephrins. Cell. 1997;90:403–404. doi: 10.1016/S0092-8674(00)80500-0. [DOI] [PubMed] [Google Scholar]
- 14.Gale NW, Holland SJ, Valenzuela DM, Flenniken A, Pan L, Ryan TE, Henkemeyer M, Strebhardt K, Hirai H, Wilkinson DG, Pawson T, Davis S, Yancopoulos GD. Eph receptors and ligands comprise two major specificity subclasses and are reciprocally compartmentalized during embryogenesis. Neuron. 1996;17:9–19. doi: 10.1016/S0896-6273(00)80276-7. [DOI] [PubMed] [Google Scholar]
- 15.Hirai H, Maru Y, Hagiwara K, Nishida J, Takaku F. A novel putative tyrosine kinase receptor encoded by the eph gene. Science. 1987;238:1717–1720. doi: 10.1126/science.2825356. [DOI] [PubMed] [Google Scholar]
- 16.Cooke JE, Kemp HA, Moens CB. EphA4 is required for cell adhesion and rhombomere-boundary formation in the zebrafish. Curr Biol. 2005;15:536–542. doi: 10.1016/j.cub.2005.02.019. [DOI] [PubMed] [Google Scholar]
- 17.Qin H, Noberini R, Huan X, Shi J, Pasquale EB, Song J. Structural characterization of the EphA4-Ephrin-B2 complex reveals new features enabling Eph–ephrin binding promiscuity. J Biol Chem. 2009;285:644–654. doi: 10.1074/jbc.M109.064824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Takemoto M, Fukuda T, Sonoda R, Murakami F, Tanaka H, Yamamoto N. Ephrin–B3–EphA4 interactions regulate the growth of specific thalamocortical axon populations in vitro. Eur J Neurosci. 2002;16:1168–1172. doi: 10.1046/j.1460-9568.2002.02166.x. [DOI] [PubMed] [Google Scholar]
- 19.Himanen JP, Chumley MJ, Lackmann M, Li C, Barton WA, Jeffrey PD, Vearing C, Geleick D, Feldheim DA, Boyd AW, Henkemeyer M, Nikolov DB. Repelling class discrimination: ephrin-A5 binds to and activates EphB2 receptor signaling. Nat Neurosci. 2004;7:501–509. doi: 10.1038/nn1237. [DOI] [PubMed] [Google Scholar]
- 20.Himanen JP, Rajashankar KR, Lackmann M, Cowan CA, Henkemeyer M, Nikolov DB. Crystal structure of an Eph receptor–ephrin complex. Nature. 2001;414:933–938. doi: 10.1038/414933a. [DOI] [PubMed] [Google Scholar]
- 21.Labrador JP, Brambilla R, Klein R. The N-terminal globular domain of Eph receptors is sufficient for ligand binding and receptor signaling. EMBO J. 1997;16:3889–3897. doi: 10.1093/emboj/16.13.3889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Himanen JP, Yermekbayeva L, Janes PW, Walker JR, Xu K, Atapattu L, Rajashankar KR, Mensinga A, Lackmann M, Nikolov DB, Dhe-Paganon S. Architecture of Eph receptor clusters. Proc Natl Acad Sci USA. 2010;107:10860–10865. doi: 10.1073/pnas.1004148107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lackmann M, Oates AC, Dottori M, Smith FM, Do C, Power M, Kravets L, Boyd AW. Distinct subdomains of the EphA3 receptor mediate ligand binding and receptor dimerization. J Biol Chem. 1998;273:20228–20237. doi: 10.1074/jbc.273.32.20228. [DOI] [PubMed] [Google Scholar]
- 24.Wimmer-Kleikamp SH, Janes PW, Squire A, Bastiaens PI, Lackmann M. Recruitment of Eph receptors into signaling clusters does not require ephrin contact. J Cell Biol. 2004;164:661–666. doi: 10.1083/jcb.200312001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Seiradake E, Harlos K, Sutton G, Aricescu AR, Jones EY. An extracellular steric seeding mechanism for Eph–ephrin signaling platform assembly. Nat Struct Mol Biol. 2010;17:398–402. doi: 10.1038/nsmb.1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Himanen JP, Nikolov DB. Eph signaling: a structural view. Trends Neurosci. 2003;26:46–51. doi: 10.1016/S0166-2236(02)00005-X. [DOI] [PubMed] [Google Scholar]
- 27.Stapleton D, Balan I, Pawson T, Sicheri F. The crystal structure of an Eph receptor SAM domain reveals a mechanism for modular dimerization. Nat Struct Biol. 1999;6:44–49. doi: 10.1038/4917. [DOI] [PubMed] [Google Scholar]
- 28.Leone M, Cellitti J, Pellecchia M. NMR studies of a heterotypic Sam-Sam domain association: the interaction between the lipid phosphatase Ship2 and the EphA2 receptor. Biochemistry. 2008;47:12721–12728. doi: 10.1021/bi801713f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhuang G, Hunter S, Hwang Y, Chen J. Regulation of EphA2 receptor endocytosis by SHIP2 lipid phosphatase via phosphatidylinositol 3-Kinase-dependent Rac1 activation. J Biol Chem. 2007;282:2683–2694. doi: 10.1074/jbc.M608509200. [DOI] [PubMed] [Google Scholar]
- 30.Davis S, Gale NW, Aldrich TH, Maisonpierre PC, Lhotak V, Pawson T, Goldfarb M, Yancopoulos GD. Ligands for EPH-related receptor tyrosine kinases that require membrane attachment or clustering for activity. Science. 1994;266:816–819. doi: 10.1126/science.7973638. [DOI] [PubMed] [Google Scholar]
- 31.Carter N, Nakamoto T, Hirai H, Hunter T. EphrinA1-induced cytoskeletal re-organization requires FAK and p130(cas) Nat Cell Biol. 2002;4:565–573. doi: 10.1038/ncb823. [DOI] [PubMed] [Google Scholar]
- 32.Dobrzanski P, Hunter K, Jones-Bolin S, Chang H, Robinson C, Pritchard S, Zhao H, Ruggeri B. Antiangiogenic and antitumor efficacy of EphA2 receptor antagonist. Cancer Res. 2004;64:910–919. doi: 10.1158/0008-5472.CAN-3430-2. [DOI] [PubMed] [Google Scholar]
- 33.Lawrenson ID, Wimmer-Kleikamp SH, Lock P, Schoenwaelder SM, Down M, Boyd AW, Alewood PF, Lackmann M. Ephrin-A5 induces rounding, blebbing and de-adhesion of EphA3- expressing 293T and melanoma cells by CrkII and Rho-mediated signalling. J Cell Sci. 2002;115:1059–1072. doi: 10.1242/jcs.115.5.1059. [DOI] [PubMed] [Google Scholar]
- 34.Stein E, Lane AA, Cerretti DP, Schoecklmann HO, Schroff AD, Van Etten RL, Daniel TO. Eph receptors discriminate specific ligand oligomers to determine alternative signaling complexes, attachment, and assembly responses. Genes Dev. 1998;12:667–678. doi: 10.1101/gad.12.5.667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vearing C, Lee FT, Wimmer-Kleikamp S, Spirkoska V, To C, Stylianou C, Spanevello M, Brechbiel M, Boyd AW, Scott AM, Lackmann M. Concurrent binding of anti-EphA3 antibody and ephrin-A5 amplifies EphA3 signaling and downstream responses: potential as EphA3-specific tumor-targeting reagents. Cancer Res. 2005;65:6745–6754. doi: 10.1158/0008-5472.CAN-05-0758. [DOI] [PubMed] [Google Scholar]
- 36.Vearing CJ, Lackmann M. Eph receptor signalling; dimerisation just isn’t enough. Growth Factors. 2005;23:67–76. doi: 10.1080/08977190500055869. [DOI] [PubMed] [Google Scholar]
- 37.Tanaka M, Kamo T, Ota S, Sugimura H. Association of dishevelled with Eph tyrosine kinase receptor and ephrin mediates cell repulsion. EMBO J. 2003;22:847–858. doi: 10.1093/emboj/cdg088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van der Geer P, Hunter T, Lindberg RA. Receptor protein–tyrosine kinases and their signal transduction pathways. Annu Rev Cell Biol. 1994;10:251–337. doi: 10.1146/annurev.cb.10.110194.001343. [DOI] [PubMed] [Google Scholar]
- 39.Zisch AH, Stallcup WB, Chong LD, Dahlin-Huppe K, Voshol J, Schachner M, Pasquale EB. Tyrosine phosphorylation of L1 family adhesion molecules: implication of the Eph kinase Cek5. J Neurosci Res. 1997;47:655–665. doi: 10.1002/(SICI)1097-4547(19970315)47:6<655::AID-JNR12>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 40.Noren NK, Yang NY, Silldorff M, Mutyala R, Pasquale EB. Ephrin-independent regulation of cell substrate adhesion by the EphB4 receptor. Biochem J. 2009;422:433–442. doi: 10.1042/BJ20090014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Day B, To C, Himanen JP, Smith FM, Nikolov DB, Boyd AW, Lackmann M. Three distinct molecular surfaces in ephrin-A5 are essential for a functional interaction with EphA3. J Biol Chem. 2005;280:26526–26532. doi: 10.1074/jbc.M504972200. [DOI] [PubMed] [Google Scholar]
- 42.Smith FM, Vearing C, Lackmann M, Treutlein H, Himanen J, Chen K, Saul A, Nikolov D, Boyd AW. Dissecting the EphA3/Ephrin-A5 interactions using a novel functional mutagenesis screen. J Biol Chem. 2004;279:9522–9531. doi: 10.1074/jbc.M309326200. [DOI] [PubMed] [Google Scholar]
- 43.Chung I, Akita R, Vandlen R, Toomre D, Schlessinger J, Mellman I. Spatial control of EGF receptor activation by reversible dimerization on living cells. Nature. 2010;464:783–787. doi: 10.1038/nature08827. [DOI] [PubMed] [Google Scholar]
- 44.Finger C, Escher C, Schneider D (2009) The single transmembrane domains of human receptor tyrosine kinases encode self-interactions. Sci Signal 2: ra56 [DOI] [PubMed]
- 45.Binns KL, Taylor PP, Sicheri F, Pawson T, Holland SJ. Phosphorylation of tyrosine residues in the kinase domain and juxtamembrane region regulates the biological and catalytic activities of Eph receptors. Mol Cell Biol. 2000;20:4791–4805. doi: 10.1128/MCB.20.13.4791-4805.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Davis TL, Walker JR, Loppnau P, Butler-Cole C, Allali-Hassani A, Dhe-Paganon S. Autoregulation by the juxtamembrane region of the human ephrin receptor tyrosine kinase A3 (EphA3) Structure. 2008;16:873–884. doi: 10.1016/j.str.2008.03.008. [DOI] [PubMed] [Google Scholar]
- 47.Wiesner S, Wybenga-Groot LE, Warner N, Lin H, Pawson T, Forman-Kay JD, Sicheri F. A change in conformational dynamics underlies the activation of Eph receptor tyrosine kinases. EMBO J. 2006;25:4686–4696. doi: 10.1038/sj.emboj.7601315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wybenga-Groot LE, Baskin B, Ong SH, Tong J, Pawson T, Sicheri F. Structural basis for autoinhibition of the Ephb2 receptor tyrosine kinase by the unphosphorylated juxtamembrane region. Cell. 2001;106:745–757. doi: 10.1016/S0092-8674(01)00496-2. [DOI] [PubMed] [Google Scholar]
- 49.Singla N, Erdjument-Bromage H, Himanen Juha P, Muir Tom W, Nikolov Dimitar B. A semisynthetic Eph receptor tyrosine kinase provides insight into ligand- induced kinase activation. Chem Biol. 2011;18:361–371. doi: 10.1016/j.chembiol.2011.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fang WB, Brantley-Sieders DM, Hwang Y, Ham AJ, Chen J. Identification and functional analysis of phosphorylated tyrosine residues within EphA2 receptor tyrosine kinase. J Biol Chem. 2008;283:16017–16026. doi: 10.1074/jbc.M709934200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Warner N, Wybenga-Groot LE, Pawson T. Analysis of EphA4 receptor tyrosine kinase substrate specificity using peptide-based arrays. FEBS J. 2008;275:2561–2573. doi: 10.1111/j.1742-4658.2008.06405.x. [DOI] [PubMed] [Google Scholar]
- 52.Citri A, Yarden Y. EGF–ERBB signalling: towards the systems level. Nat Rev Mol Cell Biol. 2006;7:505–516. doi: 10.1038/nrm1962. [DOI] [PubMed] [Google Scholar]
- 53.Freywald A, Sharfe N, Roifman CM. The kinase-null EphB6 receptor undergoes transphosphorylation in a complex with EphB1. J Biol Chem. 2002;277:3823–3828. doi: 10.1074/jbc.M108011200. [DOI] [PubMed] [Google Scholar]
- 54.Truitt L, Freywald T, DeCoteau J, Sharfe N, Freywald A. The EphB6 receptor cooperates with c-Cbl to regulate the behavior of breast cancer cells. Cancer Res. 2010;70:1141–1153. doi: 10.1158/0008-5472.CAN-09-1710. [DOI] [PubMed] [Google Scholar]
- 55.Bush JO, Soriano P. Ephrin-B1 forward signaling regulates craniofacial morphogenesis by controlling cell proliferation across Eph–ephrin boundaries. Genes Dev. 2010;24:2068–2080. doi: 10.1101/gad.1963210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jorgensen C, Sherman A, Chen GI, Pasculescu A, Poliakov A, Hsiung M, Larsen B, Wilkinson DG, Linding R, Pawson T. Cell-specific information processing in segregating populations of Eph receptor ephrin-expressing cells. Science. 2009;326:1502–1509. doi: 10.1126/science.1176615. [DOI] [PubMed] [Google Scholar]
- 57.Janes PW, Griesshaber B, Atapattu L, Nievergall E, Hii L, Mensinga A, Chheang C, Day B, Boyd AW, Bastiaens PI, Jorgensen C, Pawson T, Lackmann M (2011) Eph receptor function is modulated by hetero-oligomerisation of A and B type Eph receptors. J Cell Biol. doi:10.1083/jcb.201104037 [DOI] [PMC free article] [PubMed]
- 58.Kholodenko BN, Hancock JF, Kolch W. Signalling ballet in space and time. Nat Rev Mol Cell Biol. 2010;11:414–426. doi: 10.1038/nrm2901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fukai J, Yokote H, Yamanaka R, Arao T, Nishio K, Itakura T. EphA4 promotes cell proliferation and migration through a novel EphA4-FGFR1 signaling pathway in the human glioma U251 cell line. Mol Cancer Ther. 2008;7:2768–2778. doi: 10.1158/1535-7163.MCT-07-2263. [DOI] [PubMed] [Google Scholar]
- 60.Yokote H, Fujita K, Jing X, Sawada T, Liang S, Yao L, Yan X, Zhang Y, Schlessinger J, Sakaguchi K. Trans-activation of EphA4 and FGF receptors mediated by direct interactions between their cytoplasmic domains. Proc Natl Acad Sci USA. 2005;102:18866–18871. doi: 10.1073/pnas.0509741102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Park EK, Warner N, Bong YS, Stapleton D, Maeda R, Pawson T, Daar IO. Ectopic EphA4 receptor induces posterior protrusions via FGF signaling in Xenopus embryos. Mol Biol Cell. 2004;15:1647–1655. doi: 10.1091/mbc.E03-09-0674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Poliakov A, Cotrina ML, Pasini A, Wilkinson DG. Regulation of EphB2 activation and cell repulsion by feedback control of the MAPK pathway. J Cell Biol. 2008;183:933–947. doi: 10.1083/jcb.200807151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Larsen AB, Pedersen MW, Stockhausen MT, Grandal MV, van Deurs B, Poulsen HS. Activation of the EGFR gene target EphA2 inhibits epidermal growth factor-induced cancer cell motility. Mol Cancer Res. 2007;5:283–293. doi: 10.1158/1541-7786.MCR-06-0321. [DOI] [PubMed] [Google Scholar]
- 64.Larsen AB, Stockhausen MT, Poulsen HS. Cell adhesion and EGFR activation regulate EphA2 expression in cancer. Cell Signal. 2010;22:636–644. doi: 10.1016/j.cellsig.2009.11.018. [DOI] [PubMed] [Google Scholar]
- 65.Komurov K, Padron D, Cheng T, Roth M, Rosenblatt KP, White MA. Comprehensive mapping of the human kinome to epidermal growth factor receptor signaling. J Biol Chem. 2010;285:21134–21142. doi: 10.1074/jbc.M110.137828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Deribe YL, Wild P, Chandrashaker A, Curak J, Schmidt MH, Kalaidzidis Y, Milutinovic N, Kratchmarova I, Buerkle L, Fetchko MJ, Schmidt P, Kittanakom S, Brown KR, Jurisica I, Blagoev B, Zerial M, Stagljar I, Dikic I (2009) Regulation of epidermal growth factor receptor trafficking by lysine deacetylase HDAC6. Sci Signal 2: ra84 [DOI] [PubMed]
- 67.Thelemann A, Petti F, Griffin G, Iwata K, Hunt T, Settinari T, Fenyo D, Gibson N, Haley JD. Phosphotyrosine signaling networks in epidermal growth factor receptor overexpressing squamous carcinoma cells. Mol Cell Proteomics. 2005;4:356–376. doi: 10.1074/mcp.M400118-MCP200. [DOI] [PubMed] [Google Scholar]
- 68.Brantley-Sieders DM, Zhuang G, Hicks D, Fang WB, Hwang Y, Cates JM, Coffman K, Jackson D, Bruckheimer E, Muraoka-Cook RS, Chen J. The receptor tyrosine kinase EphA2 promotes mammary adenocarcinoma tumorigenesis and metastatic progression in mice by amplifying ErbB2 signaling. J Clin Invest. 2008;118:64–78. doi: 10.1172/JCI33154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kim I, Ryu YS, Kwak HJ, Ahn SY, Oh J-L, Yancopoulos GD, Gale NW, Koh GY. EphB ligand, ephrinB2, suppresses the VEGF- and angiopoietin-1-induced Ras/mitogen-activated protein kinase pathway in venous endothelial cells. FASEB J. 2002;16(9):1126–1128. doi: 10.1096/fj.01-0805fje. [DOI] [PubMed] [Google Scholar]
- 70.Ojima T, Takagi H, Suzuma K, Oh H, Suzuma I, Ohashi H, Watanabe D, Suganami E, Murakami T, Kurimoto M, Honda Y, Yoshimura N. EphrinA1 inhibits vascular endothelial growth factor-induced intracellular signaling and suppresses retinal neovascularization and blood-retinal barrier breakdown. Am J Pathol. 2006;168:331–339. doi: 10.2353/ajpath.2006.050435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Halford MM, Armes J, Buchert M, Meskenaite V, Grail D, Hibbs ML, Wilks AF, Farlie PG, Newgreen DF, Hovens CM, Stacker SA. Ryk-deficient mice exhibit craniofacial defects associated with perturbed Eph receptor crosstalk. Nat Genet. 2000;25:414–418. doi: 10.1038/78099. [DOI] [PubMed] [Google Scholar]
- 72.Halford MM, Stacker SA. Revelations of the RYK receptor. Bioessays. 2001;23:34–45. doi: 10.1002/1521-1878(200101)23:1<34::AID-BIES1005>3.3.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 73.Trivier E, Ganesan TS. RYK, a catalytically inactive receptor tyrosine kinase, associates with EphB2 and EphB3 but does not interact with AF-6. J Biol Chem. 2002;277:23037–23043. doi: 10.1074/jbc.M202486200. [DOI] [PubMed] [Google Scholar]
- 74.Kamitori K, Tanaka M, Okuno-Hirasawa T, Kohsaka S. Receptor related to tyrosine kinase RYK regulates cell migration during cortical development. Biochem Biophys Res Commun. 2005;330:446–453. doi: 10.1016/j.bbrc.2005.02.177. [DOI] [PubMed] [Google Scholar]
- 75.Salvucci O, de la Luz SM, Martina JA, McCormick PJ, Tosato G. EphB2 and EphB4 receptors forward signaling promotes SDF-1-induced endothelial cell chemotaxis and branching remodeling. Blood. 2006;108:2914–2922. doi: 10.1182/blood-2006-05-023341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sharfe N, Freywald A, Toro A, Dadi H, Roifman C. Ephrin stimulation modulates T cell chemotaxis. Eur J Immunol. 2002;32:3745–3755. doi: 10.1002/1521-4141(200212)32:12<3745::AID-IMMU3745>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 77.Duda DG, Kozin SV, Kirkpatrick ND, Xu L, Fukumura D, Jain RK. CXCL12 (SDF1alpha)-CXCR4/CXCR7 pathway inhibition: an emerging sensitizer for anticancer therapies? Clin Cancer Res. 2011;17:2074–2080. doi: 10.1158/1078-0432.CCR-10-2636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Dalva MB, Takasu MA, Lin MZ, Shamah SM, Hu L, Gale NW, Greenberg ME. EphB receptors interact with NMDA receptors and regulate excitatory synapse formation. Cell. 2000;103:945–956. doi: 10.1016/S0092-8674(00)00197-5. [DOI] [PubMed] [Google Scholar]
- 79.Grunwald IC, Korte M, Wolfer D, Wilkinson GA, Unsicker K, Lipp HP, Bonhoeffer T, Klein R. Kinase-independent requirement of EphB2 receptors in hippocampal synaptic plasticity. Neuron. 2001;32:1027–1040. doi: 10.1016/S0896-6273(01)00550-5. [DOI] [PubMed] [Google Scholar]
- 80.Henderson JT, Georgiou J, Jia Z, Robertson J, Elowe S, Roder JC, Pawson T. The receptor tyrosine kinase EphB2 regulates NMDA-dependent synaptic function. Neuron. 2001;32:1041–1056. doi: 10.1016/S0896-6273(01)00553-0. [DOI] [PubMed] [Google Scholar]
- 81.Takasu MA, Dalva MB, Zigmond RE, Greenberg ME. Modulation of NMDA receptor-dependent calcium influx and gene expression through EphB receptors. Science. 2002;295:491–495. doi: 10.1126/science.1065983. [DOI] [PubMed] [Google Scholar]
- 82.Kayser MS, McClelland AC, Hughes EG, Dalva MB. Intracellular and trans-synaptic regulation of glutamatergic synaptogenesis by EphB receptors. J Neurosci. 2006;26:12152–12164. doi: 10.1523/JNEUROSCI.3072-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462–475. doi: 10.1038/nrm1662. [DOI] [PubMed] [Google Scholar]
- 84.Jones TL, Chong LD, Kim J, Xu RH, Kung HF, Daar IO. Loss of cell adhesion in Xenopus laevis embryos mediated by the cytoplasmic domain of XLerk, an erythropoietin-producing hepatocellular ligand. Proc Natl Acad Sci USA. 1998;95:576–581. doi: 10.1073/pnas.95.2.576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Oates AC, Lackmann M, Power MA, Brennan C, Down LM, Do C, Evans B, Holder N, Boyd AW. An early developmental role for eph–ephrin interaction during vertebrate gastrulation. Mech Dev. 1999;83:77–94. doi: 10.1016/S0925-4773(99)00036-2. [DOI] [PubMed] [Google Scholar]
- 86.Winning RS, Scales JB, Sargent TD. Disruption of cell adhesion in Xenopus embryos by Pagliaccio, an Eph- class receptor tyrosine kinase. Dev Biol. 1996;179:309–319. doi: 10.1006/dbio.1996.0262. [DOI] [PubMed] [Google Scholar]
- 87.Brown A, Yates PA, Burrola P, Ortuno D, Vaidya A, Jessell TM, Pfaff SL, O’Leary DD, Lemke G. Topographic mapping from the retina to the midbrain is controlled by relative but not absolute levels of EphA receptor signaling. Cell. 2000;102:77–88. doi: 10.1016/S0092-8674(00)00012-X. [DOI] [PubMed] [Google Scholar]
- 88.Reber M, Burrola P, Lemke G. A relative signalling model for the formation of a topographic neural map. Nature. 2004;431:847–853. doi: 10.1038/nature02957. [DOI] [PubMed] [Google Scholar]
- 89.Flanagan JG. Neural map specification by gradients. Curr Opin Neurobiol. 2006;16:59–66. doi: 10.1016/j.conb.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 90.Lemke G, Reber M. Retinotectal mapping: new insights from molecular genetics. Annu Rev Cell Dev Biol. 2005;21:551–580. doi: 10.1146/annurev.cellbio.20.022403.093702. [DOI] [PubMed] [Google Scholar]
- 91.McLaughlin T, O’Leary DD. Molecular gradients and development of retinotopic maps. Annu Rev Neurosci. 2005;28:327–355. doi: 10.1146/annurev.neuro.28.061604.135714. [DOI] [PubMed] [Google Scholar]
- 92.Mellitzer G, Xu Q, Wilkinson DG. Eph receptors and ephrins restrict cell intermingling and communication. Nature. 1999;400:77–81. doi: 10.1038/21907. [DOI] [PubMed] [Google Scholar]
- 93.Xu Q, Mellitzer G, Robinson V, Wilkinson DG. In vivo cell sorting in complementary segmental domains mediated by Eph receptors and ephrins. Nature. 1999;399:267–271. doi: 10.1038/20452. [DOI] [PubMed] [Google Scholar]
- 94.Nievergall E, Janes PW, Stegmayer C, Vail ME, Haj FG, Teng SW, Neel BG, Bastiaens PI, Lackmann M. PTP1B regulates Eph receptor function and trafficking. J Cell Biol. 2010;191:1189–1203. doi: 10.1083/jcb.201005035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Batlle E, Henderson JT, Beghtel H, van den Born MM, Sancho E, Huls G, Meeldijk J, Robertson J, van de Wetering M, Pawson T, Clevers H. Beta-catenin and TCF mediate cell positioning in the intestinal epithelium by controlling the expression of EphB/ephrinB. Cell. 2002;111:251–263. doi: 10.1016/S0092-8674(02)01015-2. [DOI] [PubMed] [Google Scholar]
- 96.Bundesen LQ, Scheel TA, Bregman BS, Kromer LF. Ephrin-B2 and EphB2 regulation of astrocyte-meningeal fibroblast interactions in response to spinal cord lesions in adult rats. J Neurosci. 2003;23:7789–7800. doi: 10.1523/JNEUROSCI.23-21-07789.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Afshari FT, Kwok JC, Fawcett JW. Astrocyte-produced ephrins inhibit schwann cell migration via VAV2 signaling. J Neurosci. 2010;30:4246–4255. doi: 10.1523/JNEUROSCI.3351-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Parrinello S, Napoli I, Ribeiro S, Digby PW, Fedorova M, Parkinson DB, Doddrell RD, Nakayama M, Adams RH, Lloyd AC. EphB signaling directs peripheral nerve regeneration through Sox2-dependent Schwann cell sorting. Cell. 2010;143:145–155. doi: 10.1016/j.cell.2010.08.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Davy A, Bush JO, Soriano P. Inhibition of gap junction communication at ectopic Eph/ephrin boundaries underlies craniofrontonasal syndrome. PLoS Biol. 2006;4:e315. doi: 10.1371/journal.pbio.0040315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Barrios A, Poole RJ, Durbin L, Brennan C, Holder N, Wilson SW. Eph/Ephrin signaling regulates the mesenchymal-to-epithelial transition of the paraxial mesoderm during somite morphogenesis. Curr Biol. 2003;13:1571–1582. doi: 10.1016/j.cub.2003.08.030. [DOI] [PubMed] [Google Scholar]
- 101.Julich D, Geisler R, Holley SA. Integrinalpha5 and delta/notch signaling have complementary spatiotemporal requirements during zebrafish somitogenesis. Dev Cell. 2005;8:575–586. doi: 10.1016/j.devcel.2005.01.016. [DOI] [PubMed] [Google Scholar]
- 102.Koshida S, Kishimoto Y, Ustumi H, Shimizu T, Furutani-Seiki M, Kondoh H, Takada S. Integrinalpha5-dependent fibronectin accumulation for maintenance of somite boundaries in zebrafish embryos. Dev Cell. 2005;8:587–598. doi: 10.1016/j.devcel.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 103.Stephen LJ, Fawkes AL, Verhoeve A, Lemke G, Brown A. A critical role for the EphA3 receptor tyrosine kinase in heart development. Dev Biol. 2007;302:66–79. doi: 10.1016/j.ydbio.2006.08.058. [DOI] [PubMed] [Google Scholar]
- 104.Frieden LA, Townsend TA, Vaught DB, Delaughter DM, Hwang Y, Barnett JV, Chen J. Regulation of heart valve morphogenesis by Eph receptor ligand, ephrin-A1. Dev Dyn. 2010;239:3226–3234. doi: 10.1002/dvdy.22458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.O’Donnell M, Chance RK, Bashaw GJ. Axon growth and guidance: receptor regulation and signal transduction. Annu Rev Neurosci. 2009;32:383–412. doi: 10.1146/annurev.neuro.051508.135614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Frisen J, Yates PA, McLaughlin T, Friedman GC, O’Leary DD, Barbacid M. Ephrin-A5 (AL-1/RAGS) is essential for proper retinal axon guidance and topographic mapping in the mammalian visual system. Neuron. 1998;20:235–243. doi: 10.1016/S0896-6273(00)80452-3. [DOI] [PubMed] [Google Scholar]
- 107.Dottori M, Hartley L, Galea M, Paxinos G, Polizzotto M, Kilpatrick T, Bartlett PF, Murphy M, Kontgen F, Boyd AW. EphA4 (Sek1) receptor tyrosine kinase is required for the development of the corticospinal tract. Proc Natl Acad Sci USA. 1998;95:13248–13253. doi: 10.1073/pnas.95.22.13248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Cheng HJ, Nakamoto M, Bergemann AD, Flanagan JG. Complementary gradients in expression and binding of ELF-1 and Mek4 in development of the topographic retinotectal projection map. Cell. 1995;82:371–381. doi: 10.1016/0092-8674(95)90426-3. [DOI] [PubMed] [Google Scholar]
- 109.Drescher U, Kremoser C, Handwerker C, Loschinger J, Noda M, Bonhoeffer F. In vitro guidance of retinal ganglion cell axons by RAGS, a 25 kDa tectal protein related to ligands for Eph receptor tyrosine kinases. Cell. 1995;82:359–370. doi: 10.1016/0092-8674(95)90425-5. [DOI] [PubMed] [Google Scholar]
- 110.Feldheim DA, Vanderhaeghen P, Hansen MJ, Frisen J, Lu Q, Barbacid M, Flanagan JG. Topographic guidance labels in a sensory projection to the forebrain. Neuron. 1998;21:1303–1313. doi: 10.1016/S0896-6273(00)80650-9. [DOI] [PubMed] [Google Scholar]
- 111.Yates PA, Roskies AL, McLaughlin T, O’Leary DD. Topographic-specific axon branching controlled by ephrin-As is the critical event in retinotectal map development. J Neurosci. 2001;21:8548–8563. doi: 10.1523/JNEUROSCI.21-21-08548.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kullander K, Mather NK, Diella F, Dottori M, Boyd AW, Klein R. Kinase-dependent and kinase-independent functions of EphA4 receptors in major axon tract formation in vivo. Neuron. 2001;29:73–84. doi: 10.1016/S0896-6273(01)00181-7. [DOI] [PubMed] [Google Scholar]
- 113.Yokoyama N, Romero MI, Cowan CA, Galvan P, Helmbacher F, Charnay P, Parada LF, Henkemeyer M. Forward signaling mediated by ephrin-B3 prevents contralateral corticospinal axons from recrossing the spinal cord midline. Neuron. 2001;29:85–97. doi: 10.1016/S0896-6273(01)00182-9. [DOI] [PubMed] [Google Scholar]
- 114.Krull CE, Lansford R, Gale NW, Collazo A, Marcelle C, Yancopoulos GD, Fraser SE, Bronner-Fraser M. Interactions of Eph-related receptors and ligands confer rostrocaudal pattern to trunk neural crest migration. Curr Biol. 1997;7:571–580. doi: 10.1016/S0960-9822(06)00256-9. [DOI] [PubMed] [Google Scholar]
- 115.Smith A, Robinson V, Patel K, Wilkinson DG. The EphA4 and EphB1 receptor tyrosine kinases and ephrin-B2 ligand regulate targeted migration of branchial neural crest cells. Curr Biol. 1997;7:561–570. doi: 10.1016/S0960-9822(06)00255-7. [DOI] [PubMed] [Google Scholar]
- 116.Wang HU, Anderson DJ. Eph family transmembrane ligands can mediate repulsive guidance of trunk neural crest migration and motor axon outgrowth. Neuron. 1997;18:383–396. doi: 10.1016/S0896-6273(00)81240-4. [DOI] [PubMed] [Google Scholar]
- 117.Davy A, Aubin J, Soriano P. Ephrin-B1 forward and reverse signaling are required during mouse development. Genes Dev. 2004;18:572–583. doi: 10.1101/gad.1171704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bush JO, Soriano P. Ephrin-B1 regulates axon guidance by reverse signaling through a PDZ-dependent mechanism. Genes Dev. 2009;23:1586–1599. doi: 10.1101/gad.1807209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Compagni A, Logan M, Klein R, Adams RH. Control of skeletal patterning by ephrinB1-EphB interactions. Dev Cell. 2003;5:217–230. doi: 10.1016/S1534-5807(03)00198-9. [DOI] [PubMed] [Google Scholar]
- 120.Gallarda BW, Bonanomi D, Muller D, Brown A, Alaynick WA, Andrews SE, Lemke G, Pfaff SL, Marquardt T. Segregation of axial motor and sensory pathways via heterotypic trans-axonal signaling. Science. 2008;320:233–236. doi: 10.1126/science.1153758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Klein R. Bidirectional modulation of synaptic functions by Eph/ephrin signaling. Nat Neurosci. 2009;12:15–20. doi: 10.1038/nn.2231. [DOI] [PubMed] [Google Scholar]
- 122.Lai KO, Ip NY. Synapse development and plasticity: roles of ephrin/Eph receptor signaling. Curr Opin Neurobiol. 2009;19:275–283. doi: 10.1016/j.conb.2009.04.009. [DOI] [PubMed] [Google Scholar]
- 123.Kayser MS, Nolt MJ, Dalva MB. EphB receptors couple dendritic filopodia motility to synapse formation. Neuron. 2008;59:56–69. doi: 10.1016/j.neuron.2008.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Torres R, Firestein BL, Dong H, Staudinger J, Olson EN, Huganir RL, Bredt DS, Gale NW, Yancopoulos GD. PDZ proteins bind, cluster, and synaptically colocalize with Eph receptors and their ephrin ligands. Neuron. 1998;21:1453–1463. doi: 10.1016/S0896-6273(00)80663-7. [DOI] [PubMed] [Google Scholar]
- 125.Akaneya Y, Sohya K, Kitamura A, Kimura F, Washburn C, Zhou R, Ninan I, Tsumoto T, Ziff EB. Ephrin-A5 and EphA5 interaction induces synaptogenesis during early hippocampal development. PLoS One. 2010;5:e12486. doi: 10.1371/journal.pone.0012486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ethell IM, Irie F, Kalo MS, Couchman JR, Pasquale EB, Yamaguchi Y. Ephb/syndecan-2 signaling in dendritic spine morphogenesis. Neuron. 2001;31:1001–1013. doi: 10.1016/S0896-6273(01)00440-8. [DOI] [PubMed] [Google Scholar]
- 127.Henkemeyer M, Itkis OS, Ngo M, Hickmott PW, Ethell IM. Multiple EphB receptor tyrosine kinases shape dendritic spines in the hippocampus. J Cell Biol. 2003;163:1313–1326. doi: 10.1083/jcb.200306033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Penzes P, Beeser A, Chernoff J, Schiller MR, Eipper BA, Mains RE, Huganir RL. Rapid induction of dendritic spine morphogenesis by trans-synaptic ephrinB-EphB receptor activation of the Rho-GEF kalirin. Neuron. 2003;37:263–274. doi: 10.1016/S0896-6273(02)01168-6. [DOI] [PubMed] [Google Scholar]
- 129.Segura I, Essmann CL, Weinges S, Acker-Palmer A. Grb4 and GIT1 transduce ephrinB reverse signals modulating spine morphogenesis and synapse formation. Nat Neurosci. 2007;10:301–310. doi: 10.1038/nn1858. [DOI] [PubMed] [Google Scholar]
- 130.Fu WY, Chen Y, Sahin M, Zhao XS, Shi L, Bikoff JB, Lai KO, Yung WH, Fu AK, Greenberg ME, Ip NY. Cdk5 regulates EphA4-mediated dendritic spine retraction through an ephexin1-dependent mechanism. Nat Neurosci. 2007;10:67–76. doi: 10.1038/nn1811. [DOI] [PubMed] [Google Scholar]
- 131.Li W, Zheng Z, Keifer J. Transsynaptic EphB/Ephrin-B signaling regulates growth of presynaptic boutons required for classical conditioning. J Neurosci. 2011;31:8441–8449. doi: 10.1523/JNEUROSCI.6343-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Adams RH, Eichmann A. Axon guidancemolecules in vascular patterning. Cold Spring Harbor Perspect Biol. 2010;2:a001875. doi: 10.1101/cshperspect.a001875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Adams RH, Wilkinson GA, Weiss C, Diella F, Gale NW, Deutsch U, Risau W, Klein R. Roles of ephrinB ligands and EphB receptors in cardiovascular development: demarcation of arterial/venous domains, vascular morphogenesis, and sprouting angiogenesis. Genes Dev. 1999;13:295–306. doi: 10.1101/gad.13.3.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Gerety SS, Anderson DJ. Cardiovascular ephrinB2 function is essential for embryonic angiogenesis. Development. 2002;129:1397–1410. doi: 10.1242/dev.129.6.1397. [DOI] [PubMed] [Google Scholar]
- 135.Gerety SS, Wang HU, Chen ZF, Anderson DJ. Symmetrical mutant phenotypes of the receptor EphB4 and its specific transmembrane ligand ephrin-B2 in cardiovascular development. Mol Cell. 1999;4:403–414. doi: 10.1016/S1097-2765(00)80342-1. [DOI] [PubMed] [Google Scholar]
- 136.Wang HU, Chen ZF, Anderson DJ. Molecular distinction and angiogenic interaction between embryonic arteries and veins revealed by ephrin-B2 and its receptor Eph-B4. Cell. 1998;93:741–753. doi: 10.1016/S0092-8674(00)81436-1. [DOI] [PubMed] [Google Scholar]
- 137.Zhong TP, Childs S, Leu JP, Fishman MC. Gridlock signalling pathway fashions the first embryonic artery. Nature. 2001;414:216–220. doi: 10.1038/35102599. [DOI] [PubMed] [Google Scholar]
- 138.Pitulescu ME, Adams RH. Eph/ephrin molecules—a hub for signaling and endocytosis. Genes Dev. 2010;24:2480–2492. doi: 10.1101/gad.1973910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Adams RH, Diella F, Hennig S, Helmbacher F, Deutsch U, Klein R. The cytoplasmic domain of the ligand ephrinB2 is required for vascular morphogenesis but not cranial neural crest migration. Cell. 2001;104:57–69. doi: 10.1016/S0092-8674(01)00191-X. [DOI] [PubMed] [Google Scholar]
- 140.Foo SS, Turner CJ, Adams S, Compagni A, Aubyn D, Kogata N, Lindblom P, Shani M, Zicha D, Adams RH. Ephrin-B2 controls cell motility and adhesion during blood-vessel-wall assembly. Cell. 2006;124:161–173. doi: 10.1016/j.cell.2005.10.034. [DOI] [PubMed] [Google Scholar]
- 141.Herbert SP, Huisken J, Kim TN, Feldman ME, Houseman BT, Wang RA, Shokat KM, Stainier DY. Arterial-venous segregation by selective cell sprouting: an alternative mode of blood vessel formation. Science. 2009;326:294–298. doi: 10.1126/science.1178577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Cowan CA, Yokoyama N, Saxena A, Chumley MJ, Silvany RE, Baker LA, Srivastava D, Henkemeyer M. Ephrin-B2 reverse signaling is required for axon pathfinding and cardiac valve formation but not early vascular development. Dev Biol. 2004;271:263–271. doi: 10.1016/j.ydbio.2004.03.026. [DOI] [PubMed] [Google Scholar]
- 143.Brantley-Sieders DM, Caughron J, Hicks D, Pozzi A, Ruiz JC, Chen J. EphA2 receptor tyrosine kinase regulates endothelial cell migration and vascular assembly through phosphoinositide 3-kinase-mediated Rac1 GTPase activation. J Cell Sci. 2004;117:2037–2049. doi: 10.1242/jcs.01061. [DOI] [PubMed] [Google Scholar]
- 144.Ogawa K, Pasqualini R, Lindberg RA, Kain R, Freeman AL, Pasquale EB. The ephrin-A1 ligand and its receptor, EphA2, are expressed during tumor neovascularization. Oncogene. 2000;19:6043–6052. doi: 10.1038/sj.onc.1204004. [DOI] [PubMed] [Google Scholar]
- 145.Sawamiphak S, Seidel S, Essmann CL, Wilkinson GA, Pitulescu ME, Acker T, Acker-Palmer A. Ephrin-B2 regulates VEGFR2 function in developmental and tumour angiogenesis. Nature. 2010;465(7297):487–491. doi: 10.1038/nature08995. [DOI] [PubMed] [Google Scholar]
- 146.Wang Y, Nakayama M, Pitulescu ME, Schmidt TS, Bochenek ML, Sakakibara A, Adams S, Davy A, Deutsch U, Luthi U, Barberis A, Benjamin LE, Makinen T, Nobes CD, Adams RH. Ephrin-B2 controls VEGF-induced angiogenesis and lymphangiogenesis. Nature. 2010;465(7297):483–486. doi: 10.1038/nature09002. [DOI] [PubMed] [Google Scholar]
- 147.Gale NW, Baluk P, Pan L, Kwan M, Holash J, DeChiara TM, McDonald DM, Yancopoulos GD. Ephrin-B2 selectively marks arterial vessels and neovascularization sites in the adult, with expression in both endothelial and smooth- muscle cells. Dev Biol. 2001;230:151–160. doi: 10.1006/dbio.2000.0112. [DOI] [PubMed] [Google Scholar]
- 148.Shin D, Garcia-Cardena G, Hayashi S, Gerety S, Asahara T, Stavrakis G, Isner J, Folkman J, Gimbrone MA, Jr, Anderson DJ. Expression of ephrinB2 identifies a stable genetic difference between arterial and venous vascular smooth muscle as well as endothelial cells, and marks subsets of microvessels at sites of adult neovascularization. Dev Biol. 2001;230:139–150. doi: 10.1006/dbio.2000.9957. [DOI] [PubMed] [Google Scholar]
- 149.Salvucci O, Maric D, Economopoulou M, Sakakibara S, Merlin S, Follenzi A, Tosato G. EphrinB reverse signaling contributes to endothelial and mural cell assembly into vascular structures. Blood. 2009;114:1707–1716. doi: 10.1182/blood-2008-12-192294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Kuijper S, Turner CJ, Adams RH. Regulation of angiogenesis by Eph–ephrin interactions. Trends Cardiovasc Med. 2007;17:145–151. doi: 10.1016/j.tcm.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 151.Helbling PM, Saulnier DM, Brandli AW. The receptor tyrosine kinase EphB4 and ephrin-B ligands restrict angiogenic growth of embryonic veins in Xenopus laevis . Development. 2000;127:269–278. doi: 10.1242/dev.127.2.269. [DOI] [PubMed] [Google Scholar]
- 152.Pfaff D, Heroult M, Riedel M, Reiss Y, Kirmse R, Ludwig T, Korff T, Hecker M, Augustin HG. Involvement of endothelial ephrin-B2 in adhesion and transmigration of EphB-receptor-expressing monocytes. J Cell Sci. 2008;121:3842–3850. doi: 10.1242/jcs.030627. [DOI] [PubMed] [Google Scholar]
- 153.Tammela T, Alitalo K. Lymphangiogenesis: molecular mechanisms and future promise. Cell. 2010;140:460–476. doi: 10.1016/j.cell.2010.01.045. [DOI] [PubMed] [Google Scholar]
- 154.Makinen T, Adams RH, Bailey J, Lu Q, Ziemiecki A, Alitalo K, Klein R, Wilkinson GA. PDZ interaction site in ephrinB2 is required for the remodeling of lymphatic vasculature. Genes Dev. 2005;19:397–410. doi: 10.1101/gad.330105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Campbell TN, Robbins SM. The Eph receptor/ephrin system: an emerging player in the invasion game. Curr Issues Mol Biol. 2008;10:61–66. [PubMed] [Google Scholar]
- 156.Chiari R, Hames G, Stroobant V, Texier C, Maillere B, Boon T, Coulie PG. Identification of a tumor-specific shared antigen derived from an Eph receptor and presented to CD4 T cells on HLA class II molecules. Cancer Res. 2000;60:4855–4863. [PubMed] [Google Scholar]
- 157.Easty DJ, Bennett DC. Protein tyrosine kinases in malignant melanoma. Melanoma Res. 2000;10:401–411. doi: 10.1097/00008390-200010000-00001. [DOI] [PubMed] [Google Scholar]
- 158.Hess AR, Margaryan NV, Seftor EA, Hendrix MJ. Deciphering the signaling events that promote melanoma tumor cell vasculogenic mimicry and their link to embryonic vasculogenesis: role of the Eph receptors. Dev Dyn. 2007;236:3283–3296. doi: 10.1002/dvdy.21190. [DOI] [PubMed] [Google Scholar]
- 159.Easty DJ, Herlyn M, Bennett DC. Abnormal protein tyrosine kinase gene expression during melanoma progression and metastasis. Int J Cancer. 1995;60:129–136. doi: 10.1002/ijc.2910600119. [DOI] [PubMed] [Google Scholar]
- 160.Vecchi M, Confalonieri S, Nuciforo P, Vigano MA, Capra M, Bianchi M, Nicosia D, Bianchi F, Galimberti V, Viale G, Palermo G, Riccardi A, Campanini R, Daidone MG, Pierotti MA, Pece S, Di Fiore PP. Breast cancer metastases are molecularly distinct from their primary tumors. Oncogene. 2008;27:2148–2158. doi: 10.1038/sj.onc.1210858. [DOI] [PubMed] [Google Scholar]
- 161.Zelinski DP, Zantek ND, Stewart JC, Irizarry AR, Kinch MS. EphA2 overexpression causes tumorigenesis of mammary epithelial cells. Cancer Res. 2001;61:2301–2306. [PubMed] [Google Scholar]
- 162.Fournier MV, Martin KJ, Kenny PA, Xhaja K, Bosch I, Yaswen P, Bissell MJ. Gene expression signature in organized and growth-arrested mammary acini predicts good outcome in breast cancer. Cancer Res. 2006;66:7095–7102. doi: 10.1158/0008-5472.CAN-06-0515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Wu Q, Suo Z, Risberg B, Karlsson MG, Villman K, Nesland JM. Expression of Ephb2 and Ephb4 in breast carcinoma. Pathol Oncol Res. 2004;10:26–33. doi: 10.1007/BF02893405. [DOI] [PubMed] [Google Scholar]
- 164.Kumar SR, Singh J, Xia G, Krasnoperov V, Hassanieh L, Ley EJ, Scehnet J, Kumar NG, Hawes D, Press MF, Weaver FA, Gill PS. Receptor tyrosine kinase EphB4 is a survival factor in breast cancer. Am J Pathol. 2006;169:279–293. doi: 10.2353/ajpath.2006.050889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Munarini N, Jager R, Abderhalden S, Zuercher G, Rohrbach V, Loercher S, Pfanner-Meyer B, Andres AC, Ziemiecki A. Altered mammary epithelial development, pattern formation and involution in transgenic mice expressing the EphB4 receptor tyrosine kinase. J Cell Sci. 2002;115:25–37. doi: 10.1242/jcs.115.1.25. [DOI] [PubMed] [Google Scholar]
- 166.Brantley DM, Cheng N, Thompson EJ, Lin Q, Brekken RA, Thorpe PE, Muraoka RS, Cerretti DP, Pozzi A, Jackson D, Lin C, Chen J. Soluble Eph A receptors inhibit tumor angiogenesis and progression in vivo. Oncogene. 2002;21:7011–7026. doi: 10.1038/sj.onc.1205679. [DOI] [PubMed] [Google Scholar]
- 167.Ruiz JC, Robertson EJ. The expression of the receptor-protein tyrosine kinase gene, eck, is highly restricted during early mouse development. Mech Dev. 1994;46:87–100. doi: 10.1016/0925-4773(94)90078-7. [DOI] [PubMed] [Google Scholar]
- 168.Chen J, Nachabah A, Scherer C, Ganju P, Reith A, Bronson R, Ruley HE. Germ-line inactivation of the murine Eck receptor tyrosine kinase by gene trap retroviral insertion. Oncogene. 1996;12:979–988. [PubMed] [Google Scholar]
- 169.Cheng N, Brantley D, Fang WB, Liu H, Fanslow W, Cerretti DP, Bussell KN, Reith A, Jackson D, Chen J. Inhibition of VEGF-dependent multistage carcinogenesis by soluble EphA receptors. Neoplasia. 2003;5:445–456. doi: 10.1016/s1476-5586(03)80047-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Brantley-Sieders DM, Fang WB, Hicks DJ, Zhuang G, Shyr Y, Chen J. Impaired tumor microenvironment in EphA2-deficient mice inhibits tumor angiogenesis and metastatic progression. FASEB J. 2005;19:1884–1886. doi: 10.1096/fj.05-4038fje. [DOI] [PubMed] [Google Scholar]
- 171.Brantley-Sieders DM, Fang WB, Hwang Y, Hicks D, Chen J. Ephrin-A1 facilitates mammary tumor metastasis through an angiogenesis-dependent mechanism mediated by EphA receptor and vascular endothelial growth factor in mice. Cancer Res. 2006;66:10315–10324. doi: 10.1158/0008-5472.CAN-06-1560. [DOI] [PubMed] [Google Scholar]
- 172.Deroanne C, Vouret-Craviari V, Wang B, Pouyssegur J. EphrinA1 inactivates integrin-mediated vascular smooth muscle cell spreading via the Rac/PAK pathway. J Cell Sci. 2003;116:1367–1376. doi: 10.1242/jcs.00308. [DOI] [PubMed] [Google Scholar]
- 173.Hayashi S, Asahara T, Masuda H, Isner JM, Losordo DW. Functional ephrin-B2 expression for promotive interaction between arterial and venous vessels in postnatal neovascularization. Circulation. 2005;111:2210–2218. doi: 10.1161/01.CIR.0000163566.07427.73. [DOI] [PubMed] [Google Scholar]
- 174.Foubert P, Silvestre JS, Souttou B, Barateau V, Martin C, Ebrahimian TG, Lere-Dean C, Contreres JO, Sulpice E, Levy BI, Plouet J, Tobelem G, Le Ricousse-Roussanne S. PSGL-1-mediated activation of EphB4 increases the proangiogenic potential of endothelial progenitor cells. J Clin Invest. 2007;117:1527–1537. doi: 10.1172/JCI28338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Mansson-Broberg A, Siddiqui AJ, Genander M, Grinnemo KH, Hao X, Andersson AB, Wardell E, Sylven C, Corbascio M. Modulation of ephrinB2 leads to increased angiogenesis in ischemic myocardium and endothelial cell proliferation. Biochem Biophys Res Commun. 2008;373:355–359. doi: 10.1016/j.bbrc.2008.06.036. [DOI] [PubMed] [Google Scholar]
- 176.Noren NK, Foos G, Hauser CA, Pasquale EB. The EphB4 receptor suppresses breast cancer cell tumorigenicity through an Abl–Crk pathway. Nat Cell Biol. 2006;8:815–825. doi: 10.1038/ncb1438. [DOI] [PubMed] [Google Scholar]
- 177.Erber R, Eichelsbacher U, Powajbo V, Korn T, Djonov V, Lin J, Hammes HP, Grobholz R, Ullrich A, Vajkoczy P. EphB4 controls blood vascular morphogenesis during postnatal angiogenesis. EMBO J. 2006;25:628–641. doi: 10.1038/sj.emboj.7600949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Masood R, Xia G, Smith DL, Scalia P, Still JG, Tulpule A, Gill PS. Ephrin B2 expression in Kaposi sarcoma is induced by human herpesvirus type 8: phenotype switch from venous to arterial endothelium. Blood. 2005;105:1310–1318. doi: 10.1182/blood-2004-03-0933. [DOI] [PubMed] [Google Scholar]
- 179.Hainaud P, Contreres JO, Villemain A, Liu LX, Plouet J, Tobelem G, Dupuy E. The role of the vascular endothelial growth factor-Delta-like 4 ligand/Notch4-ephrin B2 cascade in tumor vessel remodeling and endothelial cell functions. Cancer Res. 2006;66:8501–8510. doi: 10.1158/0008-5472.CAN-05-4226. [DOI] [PubMed] [Google Scholar]
- 180.Scehnet JS, Ley EJ, Krasnoperov V, Liu R, Manchanda PK, Sjoberg E, Kostecke AP, Gupta S, Kumar SR, Gill PS. The role of Ephs, Ephrins, and growth factors in Kaposi sarcoma and implications of EphrinB2 blockade. Blood. 2009;113:254–263. doi: 10.1182/blood-2008-02-140020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Guo H, Miao H, Gerber L, Singh J, Denning MF, Gilliam AC, Wang B. Disruption of EphA2 receptor tyrosine kinase leads to increased susceptibility to carcinogenesis in mouse skin. Cancer Res. 2006;66:7050–7058. doi: 10.1158/0008-5472.CAN-06-0004. [DOI] [PubMed] [Google Scholar]
- 182.Batlle E, Bacani J, Begthel H, Jonkheer S, Gregorieff A, van de Born M, Malats N, Sancho E, Boon E, Pawson T, Gallinger S, Pals S, Clevers H. EphB receptor activity suppresses colorectal cancer progression. Nature. 2005;435:1126–1130. doi: 10.1038/nature03626. [DOI] [PubMed] [Google Scholar]
- 183.Dopeso H, Mateo-Lozano S, Mazzolini R, Rodrigues P, Lagares-Tena L, Ceron J, Romero J, Esteves M, Landolfi S, Hernandez-Losa J, Castano J, Wilson AJ, Ramon y Cajal S, Mariadason JM, Schwartz S, Jr, Arango D. The receptor tyrosine kinase EPHB4 has tumor suppressor activities in intestinal tumorigenesis. Cancer Res. 2009;69:7430–7438. doi: 10.1158/0008-5472.CAN-09-0706. [DOI] [PubMed] [Google Scholar]
- 184.Xia G, Kumar SR, Masood R, Zhu S, Reddy R, Krasnoperov V, Quinn DI, Henshall SM, Sutherland RL, Pinski JK, Daneshmand S, Buscarini M, Stein JP, Zhong C, Broek D, Roy-Burman P, Gill PS. EphB4 expression and biological significance in prostate cancer. Cancer Res. 2005;65:4623–4632. doi: 10.1158/0008-5472.CAN-04-2667. [DOI] [PubMed] [Google Scholar]
- 185.Stephenson SA, Slomka S, Douglas EL, Hewett PJ, Hardingham JE. Receptor protein tyrosine kinase EphB4 is up-regulated in colon cancer. BMC Mol Biol. 2001;2:15. doi: 10.1186/1471-2199-2-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Takai N, Miyazaki T, Fujisawa K, Nasu K, Miyakawa I. Expression of receptor tyrosine kinase EphB4 and its ligand ephrin-B2 is associated with malignant potential in endometrial cancer. Oncol Rep. 2001;8:567–573. doi: 10.3892/or.8.3.567. [DOI] [PubMed] [Google Scholar]
- 187.Berclaz G, Flutsch B, Altermatt HJ, Rohrbach V, Djonov V, Ziemiecki A, Dreher E, Andres AC. Loss of EphB4 receptor tyrosine kinase protein expression during carcinogenesis of the human breast. Oncol Rep. 2002;9:985–989. [PubMed] [Google Scholar]
- 188.Guan M, Xu C, Zhang F, Ye C. Aberrant methylation of EphA7 in human prostate cancer and its relation to clinicopathologic features. Int J Cancer. 2009;124:88–94. doi: 10.1002/ijc.23890. [DOI] [PubMed] [Google Scholar]
- 189.Wang J, Kataoka H, Suzuki M, Sato N, Nakamura R, Tao H, Maruyama K, Isogaki J, Kanaoka S, Ihara M, Tanaka M, Kanamori M, Nakamura T, Shinmura K, Sugimura H. Downregulation of EphA7 by hypermethylation in colorectal cancer. Oncogene. 2005;24:5637–5647. doi: 10.1038/sj.onc.1208720. [DOI] [PubMed] [Google Scholar]
- 190.Fox BP, Kandpal RP. Invasiveness of breast carcinoma cells and transcript profile: Eph receptors and ephrin ligands as molecular markers of potential diagnostic and prognostic application. Biochem Biophys Res Commun. 2004;318:882–892. doi: 10.1016/j.bbrc.2004.04.102. [DOI] [PubMed] [Google Scholar]
- 191.Nosho K, Yamamoto H, Takahashi T, Mikami M, Taniguchi H, Miyamoto N, Adachi Y, Arimura Y, Itoh F, Imai K, Shinomura Y. Genetic and epigenetic profiling in early colorectal tumors and prediction of invasive potential in pT1 (early invasive) colorectal cancers. Carcinogenesis. 2007;28:1364–1370. doi: 10.1093/carcin/bgl246. [DOI] [PubMed] [Google Scholar]
- 192.Dottori M, Down M, Huttmann A, Fitzpatrick DR, Boyd AW. Cloning and characterization of EphA3 (Hek) gene promoter: DNA methylation regulates expression in hematopoietic tumor cells. Blood. 1999;94:2477–2486. [PubMed] [Google Scholar]
- 193.Kuang SQ, Bai H, Fang ZH, Lopez G, Yang H, Tong W, Wang ZZ, Garcia-Manero G. Aberrant DNA methylation and epigenetic inactivation of Eph receptor tyrosine kinases and ephrin ligands in acute lymphoblastic leukemia. Blood. 2010;115:2412–2419. doi: 10.1182/blood-2009-05-222208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Dong Y, Wang J, Sheng Z, Li G, Ma H, Wang X, Zhang R, Lu G, Hu Q, Sugimura H, Zhou X. Downregulation of EphA1 in colorectal carcinomas correlates with invasion and metastasis. Mod Pathol. 2009;22:151–160. doi: 10.1038/modpathol.2008.188. [DOI] [PubMed] [Google Scholar]
- 195.Herath NI, Doecke J, Spanevello MD, Leggett BA, Boyd AW. Epigenetic silencing of EphA1 expression in colorectal cancer is correlated with poor survival. Br J Cancer. 2009;100:1095–1102. doi: 10.1038/sj.bjc.6604970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Clevers H, Batlle E. EphB/EphrinB receptors and Wnt signaling in colorectal cancer. Cancer Res. 2006;66:2–5. doi: 10.1158/0008-5472.CAN-05-3849. [DOI] [PubMed] [Google Scholar]
- 197.Cortina C, Palomo-Ponce S, Iglesias M, Fernandez-Masip JL, Vivancos A, Whissell G, Huma M, Peiro N, Gallego L, Jonkheer S, Davy A, Lloreta J, Sancho E, Batlle E. EphB–ephrin-B interactions suppress colorectal cancer progression by compartmentalizing tumor cells. Nat Genet. 2007;39:1376–1383. doi: 10.1038/ng.2007.11. [DOI] [PubMed] [Google Scholar]
- 198.Genander M, Halford MM, Xu NJ, Eriksson M, Yu Z, Qiu Z, Martling A, Greicius G, Thakar S, Catchpole T, Chumley MJ, Zdunek S, Wang C, Holm T, Goff SP, Pettersson S, Pestell RG, Henkemeyer M, Frisen J. Dissociation of EphB2 signaling pathways mediating progenitor cell proliferation and tumor suppression. Cell. 2009;139:679–692. doi: 10.1016/j.cell.2009.08.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Merlos-Suarez A, Barriga FM, Jung P, Iglesias M, Cespedes MV, Rossell D, Sevillano M, Hernando-Momblona X, Silva-Diz V, Munoz P, Clevers H, Sancho E, Mangues R, Batlle E. The Intestinal Stem Cell Signature Identifies Colorectal Cancer Stem Cells and Predicts Disease Relapse. Cell Stem Cell. 2011;8(5):511–524. doi: 10.1016/j.stem.2011.02.020. [DOI] [PubMed] [Google Scholar]
- 200.Oricchio E, Nanjangud G, Wolfe AL, Schatz JH, Mavrakis KJ, Jiang M, Liu X, Bruno J, Heguy A, Olshen AB, Socci ND, Teruya-Feldstein J, Weis-Garcia F, Tam W, Shaknovich R, Melnick A, Himanen JP, Chaganti RS, Wendel HG. The Eph-receptor A7 is a soluble tumor suppressor for follicular lymphoma. Cell. 2011;147:554–564. doi: 10.1016/j.cell.2011.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Zantek ND, Azimi M, Fedor-Chaiken M, Wang B, Brackenbury R, Kinch MS. E-cadherin regulates the function of the EphA2 receptor tyrosine kinase. Cell Growth Differ. 1999;10:629–638. [PubMed] [Google Scholar]
- 202.Noren NK, Pasquale EB. Paradoxes of the EphB4 receptor in cancer. Cancer Res. 2007;67:3994–3997. doi: 10.1158/0008-5472.CAN-07-0525. [DOI] [PubMed] [Google Scholar]
- 203.Li X, Wang L, Gu JW, Li B, Liu WP, Wang YG, Zhang X, Zhen HN, Fei Z. Up-regulation of EphA2 and down-regulation of EphrinA1 are associated with the aggressive phenotype and poor prognosis of malignant glioma. Tumour Biol. 2010;31:477–488. doi: 10.1007/s13277-010-0060-6. [DOI] [PubMed] [Google Scholar]
- 204.Noren NK, Lu M, Freeman AL, Koolpe M, Pasquale EB. Interplay between EphB4 on tumor cells and vascular ephrin-B2 regulates tumor growth. Proc Natl Acad Sci USA. 2004;101:5583–5588. doi: 10.1073/pnas.0401381101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Alazzouzi H, Davalos V, Kokko A, Domingo E, Woerner SM, Wilson AJ, Konrad L, Laiho P, Espin E, Armengol M, Imai K, Yamamoto H, Mariadason JM, Gebert JF, Aaltonen LA, Schwartz S, Jr, Arango D. Mechanisms of inactivation of the receptor tyrosine kinase EPHB2 in colorectal tumors. Cancer Res. 2005;65:10170–10173. doi: 10.1158/0008-5472.CAN-05-2580. [DOI] [PubMed] [Google Scholar]
- 206.Davalos V, Dopeso H, Velho S, Ferreira AM, Cirnes L, Diaz-Chico N, Bilbao C, Ramirez R, Rodriguez G, Falcon O, Leon L, Niessen RC, Keller G, Dallenbach-Hellweg G, Espin E, Armengol M, Plaja A, Perucho M, Imai K, Yamamoto H, Gebert JF, Diaz-Chico JC, Hofstra RM, Woerner SM, Seruca R, Schwartz S, Jr, Arango D. High EPHB2 mutation rate in gastric but not endometrial tumors with microsatellite instability. Oncogene. 2006;26:308–311. doi: 10.1038/sj.onc.1209780. [DOI] [PubMed] [Google Scholar]
- 207.Kan Z, Jaiswal BS, Stinson J, Janakiraman V, Bhatt D, Stern HM, Yue P, Haverty PM, Bourgon R, Zheng J, Moorhead M, Chaudhuri S, Tomsho LP, Peters BA, Pujara K, Cordes S, Davis DP, Carlton VE, Yuan W, Li L, Wang W, Eigenbrot C, Kaminker JS, Eberhard DA, Waring P, Schuster SC, Modrusan Z, Zhang Z, Stokoe D, de Sauvage FJ, Faham M, Seshagiri S. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature. 2010;466:869–873. doi: 10.1038/nature09208. [DOI] [PubMed] [Google Scholar]
- 208.Sjoblom T, Jones S, Wood LD, Parsons DW, Lin J, Barber TD, Mandelker D, Leary RJ, Ptak J, Silliman N, Szabo S, Buckhaults P, Farrell C, Meeh P, Markowitz SD, Willis J, Dawson D, Willson JK, Gazdar AF, Hartigan J, Wu L, Liu C, Parmigiani G, Park BH, Bachman KE, Papadopoulos N, Vogelstein B, Kinzler KW, Velculescu VE. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314:268–274. doi: 10.1126/science.1133427. [DOI] [PubMed] [Google Scholar]
- 209.Ding L, Getz G, Wheeler DA, Mardis ER, McLellan MD, Cibulskis K, Sougnez C, Greulich H, Muzny DM, Morgan MB, Fulton L, Fulton RS, Zhang Q, Wendl MC, Lawrence MS, Larson DE, Chen K, Dooling DJ, Sabo A, Hawes AC, Shen H, Jhangiani SN, Lewis LR, Hall O, Zhu Y, Mathew T, Ren Y, Yao J, Scherer SE, Clerc K, Metcalf GA, Ng B, Milosavljevic A, Gonzalez-Garay ML, Osborne JR, Meyer R, Shi X, Tang Y, Koboldt DC, Lin L, Abbott R, Miner TL, Pohl C, Fewell G, Haipek C, Schmidt H, Dunford-Shore BH, Kraja A, Crosby SD, Sawyer CS, Vickery T, Sander S, Robinson J, Winckler W, Baldwin J, Chirieac LR, Dutt A, Fennell T, Hanna M, Johnson BE, Onofrio RC, Thomas RK, Tonon G, Weir BA, Zhao X, Ziaugra L, Zody MC, Giordano T, Orringer MB, Roth JA, Spitz MR, Wistuba II, Ozenberger B, Good PJ, Chang AC, Beer DG, Watson MA, Ladanyi M, Broderick S, Yoshizawa A, Travis WD, Pao W, Province MA, Weinstock GM, Varmus HE, Gabriel SB, Lander ES, Gibbs RA, Meyerson M, Wilson RK. Somatic mutations affect key pathways in lung adenocarcinoma. Nature. 2008;455:1069–1075. doi: 10.1038/nature07423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Balakrishnan A, Bleeker FE, Lamba S, Rodolfo M, Daniotti M, Scarpa A, van Tilborg AA, Leenstra S, Zanon C, Bardelli A. Novel somatic and germline mutations in cancer candidate genes in glioblastoma, melanoma, and pancreatic carcinoma. Cancer Res. 2007;67:3545–3550. doi: 10.1158/0008-5472.CAN-07-0065. [DOI] [PubMed] [Google Scholar]
- 211.Corbo V, Ritelli R, Barbi S, Funel N, Campani D, Bardelli A, Scarpa A. Mutational profiling of kinases in human tumours of pancreatic origin identifies candidate cancer genes in ductal and ampulla of vater carcinomas. PLoS One. 2010;5:e12653. doi: 10.1371/journal.pone.0012653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Bonifaci N, Gorski B, Masojc B, Wokolorczyk D, Jakubowska A, Debniak T, Berenguer A, Serra Musach J, Brunet J, Dopazo J, Narod SA, Lubinski J, Lazaro C, Cybulski C, Pujana MA. Exploring the link between germline and somatic genetic alterations in breast carcinogenesis. PLoS One. 2010;5:e14078. doi: 10.1371/journal.pone.0014078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Davies H, Hunter C, Smith R, Stephens P, Greenman C, Bignell G, Teague J, Butler A, Edkins S, Stevens C, Parker A, O’Meara S, Avis T, Barthorpe S, Brackenbury L, Buck G, Clements J, Cole J, Dicks E, Edwards K, Forbes S, Gorton M, Gray K, Halliday K, Harrison R, Hills K, Hinton J, Jones D, Kosmidou V, Laman R, Lugg R, Menzies A, Perry J, Petty R, Raine K, Shepherd R, Small A, Solomon H, Stephens Y, Tofts C, Varian J, Webb A, West S, Widaa S, Yates A, Brasseur F, Cooper CS, Flanagan AM, Green A, Knowles M, Leung SY, Looijenga LH, Malkowicz B, Pierotti MA, Teh BT, Yuen ST, Lakhani SR, Easton DF, Weber BL, Goldstraw P, Nicholson AG, Wooster R, Stratton MR, Futreal PA. Somatic mutations of the protein kinase gene family in human lung cancer. Cancer Res. 2005;65:7591–7595. doi: 10.1158/0008-5472.CAN-05-1855. [DOI] [PubMed] [Google Scholar]
- 214.Bardelli A, Parsons DW, Silliman N, Ptak J, Szabo S, Saha S, Markowitz S, Willson JK, Parmigiani G, Kinzler KW, Vogelstein B, Velculescu VE. Mutational analysis of the tyrosine kinome in colorectal cancers. Science. 2003;300:949. doi: 10.1126/science.1082596. [DOI] [PubMed] [Google Scholar]
- 215.Lin J, Gan CM, Zhang X, Jones S, Sjoblom T, Wood LD, Parsons DW, Papadopoulos N, Kinzler KW, Vogelstein B, Parmigiani G, Velculescu VE. A multidimensional analysis of genes mutated in breast and colorectal cancers. Genome Res. 2007;17:1304–1318. doi: 10.1101/gr.6431107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Gylfe AE, Sirkia J, Ahlsten M, Jarvinen H, Mecklin JP, Karhu A, Aaltonen LA. Somatic mutations and germline sequence variants in patients with familial colorectal cancer. Int J Cancer. 2010;127:2974–2980. doi: 10.1002/ijc.25529. [DOI] [PubMed] [Google Scholar]
- 217.Zogopoulos G, Jorgensen C, Bacani J, Montpetit A, Lepage P, Ferretti V, Chad L, Selvarajah S, Zanke B, Hudson TJ, Pawson T, Gallinger S. Germline EPHB2 receptor variants in familial colorectal cancer. PLoS One. 2008;3:e2885. doi: 10.1371/journal.pone.0002885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Huusko P, Ponciano-Jackson D, Wolf M, Kiefer JA, Azorsa DO, Tuzmen S, Weaver D, Robbins C, Moses T, Allinen M, Hautaniemi S, Chen Y, Elkahloun A, Basik M, Bova GS, Bubendorf L, Lugli A, Sauter G, Schleutker J, Ozcelik H, Elowe S, Pawson T, Trent JM, Carpten JD, Kallioniemi OP, Mousses S. Nonsense-mediated decay microarray analysis identifies mutations of EPHB2 in human prostate cancer. Nat Genet. 2004;36:979–983. doi: 10.1038/ng1408. [DOI] [PubMed] [Google Scholar]
- 219.Ruhe JE, Streit S, Hart S, Wong CH, Specht K, Knyazev P, Knyazeva T, Tay LS, Loo HL, Foo P, Wong W, Pok S, Lim SJ, Ong H, Luo M, Ho HK, Peng K, Lee TC, Bezler M, Mann C, Gaertner S, Hoefler H, Iacobelli S, Peter S, Tay A, Brenner S, Venkatesh B, Ullrich A. Genetic alterations in the tyrosine kinase transcriptome of human cancer cell lines. Cancer Res. 2007;67:11368–11376. doi: 10.1158/0008-5472.CAN-07-2703. [DOI] [PubMed] [Google Scholar]
- 220.Wimmer-Kleikamp SH, Lackmann M. Eph-modulated cell morphology, adhesion and motility in carcinogenesis. IUBMB Life. 2005;57:421–431. doi: 10.1080/15216540500138337. [DOI] [PubMed] [Google Scholar]
- 221.Holmberg J, Clarke DL, Frisen J. Regulation of repulsion versus adhesion by different splice forms of an Eph receptor. Nature. 2000;408:203–206. doi: 10.1038/35041577. [DOI] [PubMed] [Google Scholar]
- 222.Dravis C, Yokoyama N, Chumley MJ, Cowan CA, Silvany RE, Shay J, Baker LA, Henkemeyer M. Bidirectional signaling mediated by ephrin-B2 and EphB2 controls urorectal development. Dev Biol. 2004;271:272–290. doi: 10.1016/j.ydbio.2004.03.027. [DOI] [PubMed] [Google Scholar]
- 223.Birgbauer E, Cowan CA, Sretavan DW, Henkemeyer M. Kinase independent function of EphB receptors in retinal axon pathfinding to the optic disc from dorsal but not ventral retina. Development. 2000;127:1231–1241. doi: 10.1242/dev.127.6.1231. [DOI] [PubMed] [Google Scholar]
- 224.Gu C, Park S. The EphA8 receptor regulates integrin activity through p110gamma phosphatidylinositol-3 kinase in a tyrosine kinase activity-independent manner. Mol Cell Biol. 2001;21:4579–4597. doi: 10.1128/MCB.21.14.4579-4597.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Huynh-Do U, Stein E, Lane AA, Liu H, Cerretti DP, Daniel TO. Surface densities of ephrin-B1 determine EphB1-coupled activation of cell attachment through alphavbeta3 and alpha5beta1 integrins. EMBO J. 1999;18:2165–2173. doi: 10.1093/emboj/18.8.2165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Hansen MJ, Dallal GE, Flanagan JG. Retinal axon response to ephrin-as shows a graded, concentration-dependent transition from growth promotion to inhibition. Neuron. 2004;42:717–730. doi: 10.1016/j.neuron.2004.05.009. [DOI] [PubMed] [Google Scholar]
- 227.Matsuoka H, Obama H, Kelly ML, Matsui T, Nakamoto M. Biphasic functions of the kinase-defective EphB6 receptor in cell adhesion and migration. J Biol Chem. 2005;280:29355–29363. doi: 10.1074/jbc.M500010200. [DOI] [PubMed] [Google Scholar]
- 228.von Philipsborn AC, Lang S, Loeschinger J, Bernard A, David C, Lehnert D, Bonhoeffer F, Bastmeyer M. Growth cone navigation in substrate-bound ephrin gradients. Development. 2006;133:2487–2495. doi: 10.1242/dev.02412. [DOI] [PubMed] [Google Scholar]
- 229.Holmberg J, Frisen J. Ephrins are not only unattractive. Trends Neurosci. 2002;25:239–243. doi: 10.1016/S0166-2236(02)02149-5. [DOI] [PubMed] [Google Scholar]
- 230.Wimmer-Kleikamp SH, Nievergall E, Gegenbauer K, Adikari S, Mansour M, Yeadon T, Boyd AW, Patani NR, Lackmann M. Elevated protein tyrosine phosphatase activity provokes Eph/ephrin-facilitated adhesion of pre-B leukemia cells. Blood. 2008;112:721–732. doi: 10.1182/blood-2007-11-121681. [DOI] [PubMed] [Google Scholar]
- 231.Shintani T, Ihara M, Sakuta H, Takahashi H, Watakabe I, Noda M. Eph receptors are negatively controlled by protein tyrosine phosphatase receptor type O. Nat Neurosci. 2006;9:761–769. doi: 10.1038/nn1697. [DOI] [PubMed] [Google Scholar]
- 232.Astin JW, Batson J, Kadir S, Charlet J, Persad RA, Gillatt D, Oxley JD, Nobes CD. Competition amongst Eph receptors regulates contact inhibition of locomotion and invasiveness in prostate cancer cells. Nat Cell Biol. 2010;12:1194–1204. doi: 10.1038/ncb2122. [DOI] [PubMed] [Google Scholar]
- 233.Reiss K, Saftig P. The “a disintegrin and metalloprotease” (ADAM) family of sheddases: Physiological and cellular functions. Semin Cell Dev Biol. 2009;20:126–137. doi: 10.1016/j.semcdb.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 234.Hattori M, Osterfield M, Flanagan JG. Regulated cleavage of a contact-mediated axon repellent. Science. 2000;289:1360–1365. doi: 10.1126/science.289.5483.1360. [DOI] [PubMed] [Google Scholar]
- 235.Janes PW, Saha N, Barton WA, Kolev MV, Wimmer-Kleikamp SH, Nievergall E, Blobel CP, Himanen JP, Lackmann M, Nikolov DB. Adam meets Eph: an ADAM substrate recognition module acts as a molecular switch for ephrin cleavage in trans. Cell. 2005;123:291–304. doi: 10.1016/j.cell.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 236.Salaita K, Nair PM, Petit RS, Neve RM, Das D, Gray JW, Groves JT. Restriction of receptor movement alters cellular response: physical force sensing by EphA2. Science. 2010;327:1380–1385. doi: 10.1126/science.1181729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Janes PW, Wimmer-Kleikamp SH, Frangakis AS, Treble K, Griesshaber B, Sabet O, Grabenbauer M, Ting AY, Saftig P, Bastiaens PI, Lackmann M. Cytoplasmic relaxation of active Eph controls ephrin shedding by ADAM10. PLoS Biol. 2009;7:e1000215. doi: 10.1371/journal.pbio.1000215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Reddy P, Slack JL, Davis R, Cerretti DP, Kozlosky CJ, Blanton RA, Shows D, Peschon JJ, Black RA. Functional analysis of the domain structure of tumor necrosis factor-alpha converting enzyme. J Biol Chem. 2000;275:14608–14614. doi: 10.1074/jbc.275.19.14608. [DOI] [PubMed] [Google Scholar]
- 239.Blobel CP. ADAMs: key components in EGFR signalling and development. Nat Rev Mol Cell Biol. 2005;6:32–43. doi: 10.1038/nrm1548. [DOI] [PubMed] [Google Scholar]
- 240.Litterst C, Georgakopoulos A, Shioi J, Ghersi E, Wisniewski T, Wang R, Ludwig A, Robakis NK. Ligand binding and calcium influx induce distinct ectodomain/gamma-secretase-processing pathways of EphB2 receptor. J Biol Chem. 2007;282:16155–16163. doi: 10.1074/jbc.M611449200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Kopan R, Ilagan MXG. [gamma]-Secretase: proteasome of the membrane? Nat Rev Mol Cell Biol. 2004;5:499–504. doi: 10.1038/nrm1406. [DOI] [PubMed] [Google Scholar]
- 242.Wei S, Xu G, Bridges LC, Williams P, White JM, DeSimone DW. ADAM13 induces cranial neural crest by cleaving class B ephrins and regulating Wnt signaling. Dev Cell. 2010;19:345–352. doi: 10.1016/j.devcel.2010.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Georgakopoulos A, Litterst C, Ghersi E, Baki L, Xu C, Serban G, Robakis NK. Metalloproteinase/presenilin1 processing of ephrinB regulates EphB-induced Src phosphorylation and signaling. EMBO J. 2006;25:1242–1252. doi: 10.1038/sj.emboj.7601031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Tomita T, Tanaka S, Morohashi Y, Iwatsubo T. Presenilin-dependent intramembrane cleavage of ephrin-B1. Mol Neurodegener. 2006;1:2. doi: 10.1186/1750-1326-1-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Inoue E, Deguchi-Tawarada M, Togawa A, Matsui C, Arita K, Katahira-Tayama S, Sato T, Yamauchi E, Oda Y, Takai Y. Synaptic activity prompts gamma-secretase-mediated cleavage of EphA4 and dendritic spine formation. J Cell Biol. 2009;185:551–564. doi: 10.1083/jcb.200809151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Lin KT, Sloniowski S, Ethell DW, Ethell IM. Ephrin-B2-induced cleavage of EphB2 receptor is mediated by matrix metalloproteinases to trigger cell repulsion. J Biol Chem. 2008;283:28969–28979. doi: 10.1074/jbc.M804401200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Pascall JC, Brown KD. Intramembrane cleavage of ephrinB3 by the human rhomboid family protease, RHBDL2. Biochem Biophys Res Commun. 2004;317:244–252. doi: 10.1016/j.bbrc.2004.03.039. [DOI] [PubMed] [Google Scholar]
- 248.Marston DJ, Dickinson S, Nobes CD. Rac-dependent trans-endocytosis of ephrinBs regulates Eph–ephrin contact repulsion. Nat Cell Biol. 2003;5:879–888. doi: 10.1038/ncb1044. [DOI] [PubMed] [Google Scholar]
- 249.Zimmer M, Palmer A, Kohler J, Klein R. EphB–ephrinB bi-directional endocytosis terminates adhesion allowing contact mediated repulsion. Nat Cell Biol. 2003;5:869–878. doi: 10.1038/ncb1045. [DOI] [PubMed] [Google Scholar]
- 250.Kasemeier-Kulesa JC, Bradley R, Pasquale EB, Lefcort F, Kulesa PM. Eph/ephrins and N-cadherin coordinate to control the pattern of sympathetic ganglia. Development. 2006;133:4839–4847. doi: 10.1242/dev.02662. [DOI] [PubMed] [Google Scholar]
- 251.Orsulic S, Kemler R. Expression of Eph receptors and ephrins is differentially regulated by E-cadherin. J Cell Sci. 2000;113:1793–1802. doi: 10.1242/jcs.113.10.1793. [DOI] [PubMed] [Google Scholar]
- 252.Hess AR, Seftor EA, Gruman LM, Kinch MS, Seftor RE, Hendrix MJ. VE-cadherin regulates EphA2 in aggressive melanoma cells through a novel signaling pathway: implications for vasculogenic mimicry. Cancer Biol Ther. 2006;5:228–233. doi: 10.4161/cbt.5.2.2510. [DOI] [PubMed] [Google Scholar]
- 253.van de Wetering M, Sancho E, Verweij C, de Lau W, Oving I, Hurlstone A, van der Horn K, Batlle E, Coudreuse D, Haramis AP, Tjon-Pon-Fong M, Moerer P, van den Born M, Soete G, Pals S, Eilers M, Medema R, Clevers H. The beta-catenin/TCF-4 complex imposes a crypt progenitor phenotype on colorectal cancer cells. Cell. 2002;111:241–250. doi: 10.1016/S0092-8674(02)01014-0. [DOI] [PubMed] [Google Scholar]
- 254.Solanas G, Cortina C, Sevillano M, Batlle E. Cleavage of E-cadherin by ADAM10 mediates epithelial cell sorting downstream of EphB signalling. Nat Cell Biol. 2011;13:1100–1107. doi: 10.1038/ncb2298. [DOI] [PubMed] [Google Scholar]
- 255.Walker-Daniels J, Hess AR, Hendrix MJ, Kinch MS. Differential regulation of EphA2 in normal and malignant cells. Am J Pathol. 2003;162:1037–1042. doi: 10.1016/S0002-9440(10)63899-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Yuan W, Chen Z, Wu S, Ge J, Chang S, Wang X, Chen J. Expression of EphA2 and E-cadherin in gastric cancer: correlated with tumor progression and lymphogenous metastasis. Pathol Oncol Res. 2009;15:473–478. doi: 10.1007/s12253-008-9132-y. [DOI] [PubMed] [Google Scholar]
- 257.Miao H, Strebhardt K, Pasquale EB, Shen TL, Guan JL, Wang B. Inhibition of integrin-mediated cell adhesion but not directional cell migration requires catalytic activity of EphB3 receptor tyrosine kinase: role of Rho family small GTPases. J Biol Chem. 2005;280:923–932. doi: 10.1074/jbc.M411383200. [DOI] [PubMed] [Google Scholar]
- 258.Bourgin C, Murai KK, Richter M, Pasquale EB. The EphA4 receptor regulates dendritic spine remodeling by affecting beta1-integrin signaling pathways. J Cell Biol. 2007;178:1295–1307. doi: 10.1083/jcb.200610139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Miao H, Burnett E, Kinch M, Simon E, Wang B. Activation of EphA2 kinase suppresses integrin function and causes focal-adhesion-kinase dephosphorylation. Nat Cell Biol. 2000;2:62–69. doi: 10.1038/35000008. [DOI] [PubMed] [Google Scholar]
- 260.Miao H, Li DQ, Mukherjee A, Guo H, Petty A, Cutter J, Basilion JP, Sedor J, Wu J, Danielpour D, Sloan AE, Cohen ML, Wang B. EphA2 mediates ligand-dependent inhibition and ligand-independent promotion of cell migration and invasion via a reciprocal regulatory loop with Akt. Cancer Cell. 2009;16:9–20. doi: 10.1016/j.ccr.2009.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Masuda J, Usui R, Maru Y. Fibronectin type I repeat is a nonactivating ligand for EphA1 and inhibits ATF3-dependent angiogenesis. J Biol Chem. 2008;283:13148–13155. doi: 10.1074/jbc.M702164200. [DOI] [PubMed] [Google Scholar]
- 262.Nagashima K, Endo A, Ogita H, Kawana A, Yamagishi A, Kitabatake A, Matsuda M, Mochizuki N. Adaptor protein Crk is required for ephrin-B1-induced membrane ruffling and focal complex assembly of human aortic endothelial cells. Mol Biol Cell. 2002;13:4231–4242. doi: 10.1091/mbc.E02-04-0181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Huang X, Wu D, Jin H, Stupack D, Wang JY. Induction of cell retraction by the combined actions of Abl-CrkII and Rho-ROCK1 signaling. J Cell Biol. 2008;183:711–723. doi: 10.1083/jcb.200801192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Li W, Fan J, Woodley DT. Nck/Dock: an adapter between cell surface receptors and the actin cytoskeleton. Oncogene. 2001;20:6403–6417. doi: 10.1038/sj.onc.1204782. [DOI] [PubMed] [Google Scholar]
- 265.Holland SJ, Gale NW, Gish GD, Roth RA, Songyang Z, Cantley LC, Henkemeyer M, Yancopoulos GD, Pawson T. Juxtamembrane tyrosine residues couple the Eph family receptor EphB2/Nuk to specific SH2 domain proteins in neuronal cells. EMBO J. 1997;16:3877–3888. doi: 10.1093/emboj/16.13.3877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Stein E, Huynh-Do U, Lane AA, Cerretti DP, Daniel TO. Nck recruitment to Eph receptor, EphB1/ELK, couples ligand activation to c-Jun kinase. J Biol Chem. 1998;273:1303–1308. doi: 10.1074/jbc.273.3.1303. [DOI] [PubMed] [Google Scholar]
- 267.Vindis C, Teli T, Cerretti DP, Turner CE, Huynh-Do U. EphB1-mediated cell migration requires the phosphorylation of paxillin at Tyr-31/Tyr-118. J Biol Chem. 2004;279:27965–27970. doi: 10.1074/jbc.M401295200. [DOI] [PubMed] [Google Scholar]
- 268.Fawcett JP, Georgiou J, Ruston J, Bladt F, Sherman A, Warner N, Saab BJ, Scott R, Roder JC, Pawson T. Nck adaptor proteins control the organization of neuronal circuits important for walking. Proc Natl Acad Sci USA. 2007;104:20973–20978. doi: 10.1073/pnas.0710316105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Hu T, Shi G, Larose L, Rivera GM, Mayer BJ, Zhou R. Regulation of process retraction and cell migration by EphA3 is mediated by the adaptor protein Nck1. Biochemistry. 2009;48:6369–6378. doi: 10.1021/bi900831k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Noren NK, Pasquale EB. Eph receptor-ephrin bidirectional signals that target Ras and Rho proteins. Cell Signal. 2004;16:655–666. doi: 10.1016/j.cellsig.2003.10.006. [DOI] [PubMed] [Google Scholar]
- 271.Jaffe AB, Hall A. Rho GTPases: biochemistry and biology. Annu Rev Cell Dev Biol. 2005;21:247–269. doi: 10.1146/annurev.cellbio.21.020604.150721. [DOI] [PubMed] [Google Scholar]
- 272.Moeller ML, Shi Y, Reichardt LF, Ethell IM. EphB receptors regulate dendritic spine morphogenesis through the recruitment/phosphorylation of focal adhesion kinase and RhoA activation. J Biol Chem. 2006;281:1587–1598. doi: 10.1074/jbc.M511756200. [DOI] [PubMed] [Google Scholar]
- 273.Shi Y, Pontrello CG, DeFea KA, Reichardt LF, Ethell IM. Focal adhesion kinase acts downstream of EphB receptors to maintain mature dendritic spines by regulating cofilin activity. J Neurosci. 2009;29:8129–8142. doi: 10.1523/JNEUROSCI.4681-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Wahl S, Barth H, Ciossek T, Aktories K, Mueller BK. Ephrin-A5 induces collapse of growth cones by activating Rho and Rho kinase. J Cell Biol. 2000;149:263–270. doi: 10.1083/jcb.149.2.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Yamazaki T, Masuda J, Omori T, Usui R, Akiyama H, Maru Y. EphA1 interacts with integrin-linked kinase and regulates cell morphology and motility. J Cell Sci. 2009;122:243–255. doi: 10.1242/jcs.036467. [DOI] [PubMed] [Google Scholar]
- 276.Wickstrom SA, Lange A, Montanez E, Fassler R. The ILK/PINCH/parvin complex: the kinase is dead, long live the pseudokinase! EMBO J. 2010;29:281–291. doi: 10.1038/emboj.2009.376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Groeger G, Nobes CD. Co-operative Cdc42 and Rho signalling mediates ephrinB-triggered endothelial cell retraction. Biochem J. 2007;404:23–29. doi: 10.1042/BJ20070146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Irie F, Yamaguchi Y. EphB receptors regulate dendritic spine development via intersectin, Cdc42 and N-WASP. Nat Neurosci. 2002;5:1117–1118. doi: 10.1038/nn964. [DOI] [PubMed] [Google Scholar]
- 279.Bisson N, Poitras L, Mikryukov A, Tremblay ML, Moss T. EphA4 signaling regulates blastomere adhesion in the Xenopus embryo by recruiting Pak1 to suppress Cdc42 function. Mol Biol Cell. 2007;18:1030–1043. doi: 10.1091/mbc.E06-04-0294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Jurney WM, Gallo G, Letourneau PC, McLoon SC. Rac1-mediated endocytosis during ephrin-A2- and semaphorin 3A-induced growth cone collapse. J Neurosci. 2002;22:6019–6028. doi: 10.1523/JNEUROSCI.22-14-06019.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Ostman A, Frijhoff J, Sandin A, Bohmer FD. Regulation of protein tyrosine phosphatases by reversible oxidation. J Biochem. 2011;150:345–356. doi: 10.1093/jb/mvr104. [DOI] [PubMed] [Google Scholar]
- 282.Chiarugi P, Pani G, Giannoni E, Taddei L, Colavitti R, Raugei G, Symons M, Borrello S, Galeotti T, Ramponi G. Reactive oxygen species as essential mediators of cell adhesion: the oxidative inhibition of a FAK tyrosine phosphatase is required for cell adhesion. J Cell Biol. 2003;161:933–944. doi: 10.1083/jcb.200211118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Buricchi F, Giannoni E, Grimaldi G, Parri M, Raugei G, Ramponi G, Chiarugi P. Redox regulation of ephrin/integrin cross-talk. Cell Adh Migr. 2007;1:33–42. [PMC free article] [PubMed] [Google Scholar]
- 284.Hiramoto-Yamaki N, Takeuchi S, Ueda S, Harada K, Fujimoto S, Negishi M, Katoh H. Ephexin4 and EphA2 mediate cell migration through a RhoG-dependent mechanism. J Cell Biol. 2010;190:461–477. doi: 10.1083/jcb.201005141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Sahin M, Greer PL, Lin MZ, Poucher H, Eberhart J, Schmidt S, Wright TM, Shamah SM, O’Connell S, Cowan CW, Hu L, Goldberg JL, Debant A, Corfas G, Krull CE, Greenberg ME. Eph-dependent tyrosine phosphorylation of ephexin1 modulates growth cone collapse. Neuron. 2005;46:191–204. doi: 10.1016/j.neuron.2005.01.030. [DOI] [PubMed] [Google Scholar]
- 286.Shamah SM, Lin MZ, Goldberg JL, Estrach S, Sahin M, Hu L, Bazalakova M, Neve RL, Corfas G, Debant A, Greenberg ME. EphA receptors regulate growth cone dynamics through the novel guanine nucleotide exchange factor ephexin. Cell. 2001;105:233–244. doi: 10.1016/S0092-8674(01)00314-2. [DOI] [PubMed] [Google Scholar]
- 287.Knoll B, Drescher U. Src family kinases are involved in EphA receptor-mediated retinal axon guidance. J Neurosci. 2004;24:6248–6257. doi: 10.1523/JNEUROSCI.0985-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Ogita H, Kunimoto S, Kamioka Y, Sawa H, Masuda M, Mochizuki N. EphA4-mediated Rho activation via Vsm-RhoGEF expressed specifically in vascular smooth muscle cells. Circ Res. 2003;93(1):23–31. doi: 10.1161/01.RES.0000079310.81429.C8. [DOI] [PubMed] [Google Scholar]
- 289.Cowan CW, Shao YR, Sahin M, Shamah SM, Lin MZ, Greer PL, Gao S, Griffith EC, Brugge JS, Greenberg ME. Vav family GEFs link activated Ephs to endocytosis and axon guidance. Neuron. 2005;46:205–217. doi: 10.1016/j.neuron.2005.03.019. [DOI] [PubMed] [Google Scholar]
- 290.Hunter SG, Zhuang G, Brantley-Sieders D, Swat W, Cowan CW, Chen J. Essential role of Vav family guanine nucleotide exchange factors in EphA receptor-mediated angiogenesis. Mol Cell Biol. 2006;26:4830–4842. doi: 10.1128/MCB.02215-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Tanaka M, Ohashi R, Nakamura R, Shinmura K, Kamo T, Sakai R, Sugimura H. Tiam1 mediates neurite outgrowth induced by ephrin-B1 and EphA2. EMBO J. 2004;23:1075–1088. doi: 10.1038/sj.emboj.7600128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Beg AA, Sommer JE, Martin JH, Scheiffele P. alpha2-Chimaerin is an essential EphA4 effector in the assembly of neuronal locomotor circuits. Neuron. 2007;55:768–778. doi: 10.1016/j.neuron.2007.07.036. [DOI] [PubMed] [Google Scholar]
- 293.Iwasato T, Katoh H, Nishimaru H, Ishikawa Y, Inoue H, Saito YM, Ando R, Iwama M, Takahashi R, Negishi M, Itohara S. Rac-GAP alpha-chimerin regulates motor-circuit formation as a key mediator of EphrinB3/EphA4 forward signaling. Cell. 2007;130:742–753. doi: 10.1016/j.cell.2007.07.022. [DOI] [PubMed] [Google Scholar]
- 294.Shi L, Fu WY, Hung KW, Porchetta C, Hall C, Fu AK, Ip NY. Alpha2-chimaerin interacts with EphA4 and regulates EphA4-dependent growth cone collapse. Proc Natl Acad Sci USA. 2007;104:16347–16352. doi: 10.1073/pnas.0706626104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Wegmeyer H, Egea J, Rabe N, Gezelius H, Filosa A, Enjin A, Varoqueaux F, Deininger K, Schnutgen F, Brose N, Klein R, Kullander K, Betz A. EphA4-dependent axon guidance is mediated by the RacGAP alpha2-chimaerin. Neuron. 2007;55:756–767. doi: 10.1016/j.neuron.2007.07.038. [DOI] [PubMed] [Google Scholar]
- 296.Ho SK, Kovacevic N, Henkelman RM, Boyd A, Pawson T, Henderson JT. EphB2 and EphA4 receptors regulate formation of the principal inter-hemispheric tracts of the mammalian forebrain. Neuroscience. 2009;160:784–795. doi: 10.1016/j.neuroscience.2009.03.013. [DOI] [PubMed] [Google Scholar]
- 297.Takeuchi S, Yamaki N, Iwasato T, Negishi M, Katoh H. [beta]2-Chimaerin binds to EphA receptors and regulates cell migration. FEBS Lett. 2009;583:1237–1242. doi: 10.1016/j.febslet.2009.03.032. [DOI] [PubMed] [Google Scholar]
- 298.Fang WB, Ireton RC, Zhuang G, Takahashi T, Reynolds A, Chen J. Overexpression of EPHA2 receptor destabilizes adherens junctions via a RhoA-dependent mechanism. J Cell Sci. 2008;121:358–368. doi: 10.1242/jcs.017145. [DOI] [PubMed] [Google Scholar]
- 299.Kinbara K, Goldfinger LE, Hansen M, Chou F-L, Ginsberg MH. Ras GTPases: integrins’ friends or foes? Nat Rev Mol Cell Biol. 2003;4:767–778. doi: 10.1038/nrm1229. [DOI] [PubMed] [Google Scholar]
- 300.Elowe S, Holland SJ, Kulkarni S, Pawson T. Downregulation of the Ras-mitogen-activated protein kinase pathway by the EphB2 receptor tyrosine kinase is required for ephrin-induced neurite retraction. Mol Cell Biol. 2001;21:7429–7441. doi: 10.1128/MCB.21.21.7429-7441.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Tong J, Elowe S, Nash P, Pawson T. Manipulation of EphB2 regulatory motifs and SH2 binding sites switches MAPK signaling and biological activity. J Biol Chem. 2003;278:6111–6119. doi: 10.1074/jbc.M208972200. [DOI] [PubMed] [Google Scholar]
- 302.Zou JX, Wang B, Kalo MS, Zisch AH, Pasquale EB, Ruoslahti E. An Eph receptor regulates integrin activity through R-Ras. Proc Natl Acad Sci USA. 1999;96:13813–13818. doi: 10.1073/pnas.96.24.13813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Dail M, Richter M, Godement P, Pasquale EB. Eph receptors inactivate R-Ras through different mechanisms to achieve cell repulsion. J Cell Sci. 2006;119:1244–1254. doi: 10.1242/jcs.02842. [DOI] [PubMed] [Google Scholar]
- 304.Nakada M, Niska JA, Tran NL, McDonough WS, Berens ME. EphB2/R-Ras signaling regulates glioma cell adhesion, growth, and invasion. Am J Pathol. 2005;167:565–576. doi: 10.1016/S0002-9440(10)62998-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Roselli S, Wallez Y, Wang L, Vervoort V, Pasquale EB. The SH2 domain protein Shep1 regulates the in vivo signaling function of the scaffolding protein Cas. Cell Signal. 2010;22:1745–1752. doi: 10.1016/j.cellsig.2010.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Dodelet VC, Pazzagli C, Zisch AH, Hauser CA, Pasquale EB. A novel signaling intermediate, SHEP1, directly couples Eph receptors to R-Ras and Rap1A. J Biol Chem. 1999;274:31941–31946. doi: 10.1074/jbc.274.45.31941. [DOI] [PubMed] [Google Scholar]
- 307.Riedl JA, Brandt DT, Batlle E, Price LS, Clevers H, Bos JL. Down-regulation of Rap1 activity is involved in ephrinB1-induced cell contraction. Biochem J. 2005;389:465–469. doi: 10.1042/BJ20050048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Richter M, Murai KK, Bourgin C, Pak DT, Pasquale EB. The EphA4 receptor regulates neuronal morphology through SPAR-mediated inactivation of Rap GTPases. J Neurosci. 2007;27:14205–14215. doi: 10.1523/JNEUROSCI.2746-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Aoki M, Yamashita T, Tohyama M. EphA receptors direct the differentiation of mammalian neural precursor cells through a mitogen-activated protein kinase-dependent pathway. J Biol Chem. 2004;279:32643–32650. doi: 10.1074/jbc.M313247200. [DOI] [PubMed] [Google Scholar]
- 310.Davy A, Soriano P. Ephrin signaling in vivo: look both ways. Dev Dyn. 2005;232:1–10. doi: 10.1002/dvdy.20200. [DOI] [PubMed] [Google Scholar]
- 311.Egea J, Klein R. Bidirectional Eph–ephrin signaling during axon guidance. Trends Cell Biol. 2007;17:230–238. doi: 10.1016/j.tcb.2007.03.004. [DOI] [PubMed] [Google Scholar]
- 312.Marquardt T, Shirasaki R, Ghosh S, Andrews SE, Carter N, Hunter T, Pfaff SL. Coexpressed EphA receptors and ephrin-A ligands mediate opposing actions on growth cone navigation from distinct membrane domains. Cell. 2005;121:127–139. doi: 10.1016/j.cell.2005.01.020. [DOI] [PubMed] [Google Scholar]
- 313.Campbell TN, Davy A, Liu Y, Arcellana-Panlilio M, Robbins SM. Distinct membrane compartmentalization and signaling of ephrin-A5 and ephrin-B1. Biochem Biophys Res Commun. 2008;375:362–366. doi: 10.1016/j.bbrc.2008.08.002. [DOI] [PubMed] [Google Scholar]
- 314.Gauthier LR, Robbins SM. Ephrin signaling: one raft to rule them all? One raft to sort them? One raft to spread their call and in signaling bind them? Life Sci. 2003;74:207–216. doi: 10.1016/j.lfs.2003.09.029. [DOI] [PubMed] [Google Scholar]
- 315.Davy A, Gale NW, Murray EW, Klinghoffer RA, Soriano P, Feuerstein C, Robbins SM. Compartmentalized signaling by GPI-anchored ephrin-A5 requires the Fyn tyrosine kinase to regulate cellular adhesion. Genes Dev. 1999;13:3125–3135. doi: 10.1101/gad.13.23.3125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Kao TJ, Kania A. Ephrin-mediated cis-attenuation of Eph receptor signaling is essential for spinal motor axon guidance. Neuron. 2011;71:76–91. doi: 10.1016/j.neuron.2011.05.031. [DOI] [PubMed] [Google Scholar]
- 317.Bruckner K, Pasquale EB, Klein R. Tyrosine phosphorylation of transmembrane ligands for Eph receptors. Science. 1997;275:1640–1643. doi: 10.1126/science.275.5306.1640. [DOI] [PubMed] [Google Scholar]
- 318.Holland SJ, Gale NW, Mbamalu G, Yancopoulos GD, Henkemeyer M, Pawson T. Bidirectional signalling through the EPH-family receptor Nuk and its transmembrane ligands. Nature. 1996;383:722–725. doi: 10.1038/383722a0. [DOI] [PubMed] [Google Scholar]
- 319.Palmer A, Zimmer M, Erdmann KS, Eulenburg V, Porthin A, Heumann R, Deutsch U, Klein R. EphrinB phosphorylation and reverse signaling: regulation by Src kinases and PTP-BL phosphatase. Mol Cell. 2002;9:725–737. doi: 10.1016/S1097-2765(02)00488-4. [DOI] [PubMed] [Google Scholar]
- 320.Chong LD, Park EK, Latimer E, Friesel R, Daar IO. Fibroblast growth factor receptor-mediated rescue of x-ephrin B1-induced cell dissociation in Xenopus embryos. Mol Cell Biol. 2000;20:724–734. doi: 10.1128/MCB.20.2.724-734.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Cowan CA, Henkemeyer M. The SH2/SH3 adaptor Grb4 transduces B-ephrin reverse signals. Nature. 2001;413:174–179. doi: 10.1038/35093123. [DOI] [PubMed] [Google Scholar]
- 322.Bruckner K, Pablo Labrador J, Scheiffele P, Herb A, Seeburg PH, Klein R. EphrinB ligands recruit GRIP family PDZ adaptor proteins into raft membrane microdomains. Neuron. 1999;22:511–524. doi: 10.1016/S0896-6273(00)80706-0. [DOI] [PubMed] [Google Scholar]
- 323.Lin D, Gish GD, Songyang Z, Pawson T. The carboxyl terminus of B class ephrins constitutes a PDZ domain binding motif. J Biol Chem. 1999;274:3726–3733. doi: 10.1074/jbc.274.6.3726. [DOI] [PubMed] [Google Scholar]
- 324.Lu Q, Sun EE, Klein RS, Flanagan JG. Ephrin-B reverse signaling is mediated by a novel PDZ-RGS protein and selectively inhibits G protein-coupled chemoattraction. Cell. 2001;105:69–79. doi: 10.1016/S0092-8674(01)00297-5. [DOI] [PubMed] [Google Scholar]
- 325.Bochenek ML, Dickinson S, Astin JW, Adams RH, Nobes CD. Ephrin-B2 regulates endothelial cell morphology and motility independently of Eph-receptor binding. J Cell Sci. 2010;123:1235–1246. doi: 10.1242/jcs.061903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Dravis C, Henkemeyer M. Ephrin-B reverse signaling controls septation events at the embryonic midline through separate tyrosine phosphorylation-independent signaling avenues. Dev Biol. 2011;355:138–151. doi: 10.1016/j.ydbio.2011.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Xu NJ, Sun S, Gibson JR, Henkemeyer M. A dual shaping mechanism for postsynaptic ephrin-B3 as a receptor that sculpts dendrites and synapses. Nat Neurosci. 2011;14:1421–1429. doi: 10.1038/nn.2931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Vihanto MM, Plock J, Erni D, Frey BM, Frey FJ, Huynh-Do U. Hypoxia up-regulates expression of Eph receptors and ephrins in mouse skin. Faseb J. 2005;19:1689–1691. doi: 10.1096/fj.04-3647fje. [DOI] [PubMed] [Google Scholar]
- 329.Bonaparte MI, Dimitrov AS, Bossart KN, Crameri G, Mungall BA, Bishop KA, Choudhry V, Dimitrov DS, Wang LF, Eaton BT, Broder CC. Ephrin-B2 ligand is a functional receptor for Hendra virus and Nipah virus. Proc Natl Acad Sci USA. 2005;102:10652–10657. doi: 10.1073/pnas.0504887102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 330.Negrete OA, Levroney EL, Aguilar HC, Bertolotti-Ciarlet A, Nazarian R, Tajyar S, Lee B. EphrinB2 is the entry receptor for Nipah virus, an emergent deadly paramyxovirus. Nature. 2005;436:401–405. doi: 10.1038/nature03838. [DOI] [PubMed] [Google Scholar]
- 331.Lupberger J, Zeisel MB, Xiao F, Thumann C, Fofana I, Zona L, Davis C, Mee CJ, Turek M, Gorke S, Royer C, Fischer B, Zahid MN, Lavillette D, Fresquet J, Cosset FL, Rothenberg SM, Pietschmann T, Patel AH, Pessaux P, Doffoel M, Raffelsberger W, Poch O, McKeating JA, Brino L, Baumert TF. EGFR and EphA2 are host factors for hepatitis C virus entry and possible targets for antiviral therapy. Nat Med. 2011;17:589–595. doi: 10.1038/nm.2341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Holen HL, Zernichow L, Fjelland KE, Evenroed IM, Prydz K, Tveit H, Aasheim HC. Ephrin-B3 binds to a sulfated cell-surface receptor. Biochem J. 2011;433:215–223. doi: 10.1042/BJ20100865. [DOI] [PubMed] [Google Scholar]
- 333.Irie F, Okuno M, Matsumoto K, Pasquale EB, Yamaguchi Y. Heparan sulfate regulates ephrin-A3/EphA receptor signaling. Proc Natl Acad Sci USA. 2008;105:12307–12312. doi: 10.1073/pnas.0801302105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Davy A, Robbins SM. Ephrin-A5 modulates cell adhesion and morphology in an integrin-dependent manner. EMBO J. 2000;19:5396–5405. doi: 10.1093/emboj/19.20.5396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Holen HL, Shadidi M, Narvhus K, Kjosnes O, Tierens A, Aasheim HC. Signaling through ephrin-A ligand leads to activation of Src-family kinases, Akt phosphorylation, and inhibition of antigen receptor-induced apoptosis. J Leukoc Biol. 2008;84(4):1183–1191. doi: 10.1189/jlb.1207829. [DOI] [PubMed] [Google Scholar]
- 336.Lim YS, McLaughlin T, Sung TC, Santiago A, Lee KF, O’Leary DD. p75(NTR) mediates ephrin-A reverse signaling required for axon repulsion and mapping. Neuron. 2008;59:746–758. doi: 10.1016/j.neuron.2008.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Marler KJ, Becker-Barroso E, Martinez A, Llovera M, Wentzel C, Poopalasundaram S, Hindges R, Soriano E, Comella J, Drescher U. A TrkB/EphrinA interaction controls retinal axon branching and synaptogenesis. J Neurosci. 2008;28:12700–12712. doi: 10.1523/JNEUROSCI.1915-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Meyer S, Hafner C, Guba M, Flegel S, Geissler EK, Becker B, Koehl GE, Orso E, Landthaler M, Vogt T. Ephrin-B2 overexpression enhances integrin-mediated ECM-attachment and migration of B16 melanoma cells. Int J Oncol. 2005;27:1197–1206. [PubMed] [Google Scholar]
- 339.Huynh-Do U, Vindis C, Liu H, Cerretti DP, McGrew JT, Enriquez M, Chen J, Daniel TO. Ephrin-B1 transduces signals to activate integrin-mediated migration, attachment and angiogenesis. J Cell Sci. 2002;115:3073–3081. doi: 10.1242/jcs.115.15.3073. [DOI] [PubMed] [Google Scholar]
- 340.Xu Z, Lai KO, Zhou HM, Lin SC, Ip NY. Ephrin-B1 reverse signaling activates JNK through a novel mechanism that is independent of tyrosine phosphorylation. J Biol Chem. 2003;278:24767–24775. doi: 10.1074/jbc.M302454200. [DOI] [PubMed] [Google Scholar]
- 341.Julich D, Mould AP, Koper E, Holley SA. Control of extracellular matrix assembly along tissue boundaries via integrin and Eph/Ephrin signaling. Development. 2009;136:2913–2921. doi: 10.1242/dev.038935. [DOI] [PubMed] [Google Scholar]
- 342.Prevost N, Woulfe DS, Tognolini M, Tanaka T, Jian W, Fortna RR, Jiang H, Brass LF. Signaling by ephrinB1 and Eph kinases in platelets promotes Rap1 activation, platelet adhesion, and aggregation via effector pathways that do not require phosphorylation of ephrinB1. Blood. 2004;103:1348–1355. doi: 10.1182/blood-2003-06-1781. [DOI] [PubMed] [Google Scholar]
- 343.Lee HS, Bong YS, Moore KB, Soria K, Moody SA, Daar IO. Dishevelled mediates ephrinB1 signalling in the eye field through the planar cell polarity pathway. Nat Cell Biol. 2006;8:55–63. doi: 10.1038/ncb1344. [DOI] [PubMed] [Google Scholar]
- 344.Xu NJ, Henkemeyer M. Ephrin-B3 reverse signaling through Grb4 and cytoskeletal regulators mediates axon pruning. Nat Neurosci. 2009;12:268–276. doi: 10.1038/nn.2254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Nakada M, Drake KL, Nakada S, Niska JA, Berens ME. Ephrin-B3 ligand promotes glioma invasion through activation of Rac1. Cancer Res. 2006;66:8492–8500. doi: 10.1158/0008-5472.CAN-05-4211. [DOI] [PubMed] [Google Scholar]
- 346.Steinle JJ, Meininger CJ, Chowdhury U, Wu G, Granger HJ. Role of ephrin B2 in human retinal endothelial cell proliferation and migration. Cell Signal. 2003;15:1011–1017. doi: 10.1016/S0898-6568(03)00072-X. [DOI] [PubMed] [Google Scholar]
- 347.Steinle JJ, Meininger CJ, Forough R, Wu G, Wu MH, Granger HJ. Eph B4 receptor signaling mediates endothelial cell migration and proliferation via the phosphatidylinositol 3-kinase pathway. J Biol Chem. 2002;277:43830–43835. doi: 10.1074/jbc.M207221200. [DOI] [PubMed] [Google Scholar]
- 348.Watanabe T, Sato Y, Saito D, Tadokoro R, Takahashi Y. EphrinB2 coordinates the formation of a morphological boundary and cell epithelialization during somite segmentation. Proc Natl Acad Sci USA. 2009;106:7467–7472. doi: 10.1073/pnas.0902859106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Lee HS, Mood K, Battu G, Ji YJ, Singh A, Daar IO. Fibroblast growth factor receptor-induced phosphorylation of ephrinB1 modulates its interaction with dishevelled. Mol Biol Cell. 2009;20:124–133. doi: 10.1091/mbc.E08-06-0662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Moore KB, Mood K, Daar IO, Moody SA. Morphogenetic movements underlying eye field formation require interactions between the FGF and ephrinB1 signaling pathways. Dev Cell. 2004;6:55–67. doi: 10.1016/S1534-5807(03)00395-2. [DOI] [PubMed] [Google Scholar]
- 351.Tanaka M, Kamata R, Sakai R. Phosphorylation of ephrin-B1 via the interaction with claudin following cell-cell contact formation. EMBO J. 2005;24:3700–3711. doi: 10.1038/sj.emboj.7600831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Lee HS, Nishanian TG, Mood K, Bong YS, Daar IO. EphrinB1 controls cell-cell junctions through the Par polarity complex. Nat Cell Biol. 2008;10:979–986. doi: 10.1038/ncb1758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Lee HS, Daar IO. EphrinB reverse signaling in cell-cell adhesion: is it just par for the course? Cell Adh Migr. 2009;3:250–255. doi: 10.4161/cam.3.3.8211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Campbell TN, Attwell S, Arcellana-Panlilio M, Robbins SM. Ephrin A5 expression promotes invasion and transformation of murine fibroblasts. Biochem Biophys Res Commun. 2006;350:623–628. doi: 10.1016/j.bbrc.2006.09.085. [DOI] [PubMed] [Google Scholar]
- 355.Jiang G, Freywald T, Webster J, Kozan D, Geyer R, DeCoteau J, Narendran A, Freywald A. In human leukemia cells ephrin-B-induced invasive activity is supported by Lck and is associated with reassembling of lipid raft signaling complexes. Mol Cancer Res. 2008;6:291–305. doi: 10.1158/1541-7786.MCR-07-0047. [DOI] [PubMed] [Google Scholar]
- 356.Nakada M, Anderson EM, Demuth T, Nakada S, Reavie LB, Drake KL, Hoelzinger DB, Berens ME. The phosphorylation of ephrin-B2 ligand promotes glioma cell migration and invasion. Int J Cancer. 2009;126:1155–1165. doi: 10.1002/ijc.24849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.Tanaka M, Sasaki K, Kamata R, Sakai R. The C-terminus of ephrin-B1 regulates metalloproteinase secretion and invasion of cancer cells. J Cell Sci. 2007;120:2179–2189. doi: 10.1242/jcs.008607. [DOI] [PubMed] [Google Scholar]
- 358.de Saint-Vis B, Bouchet C, Gautier G, Valladeau J, Caux C, Garrone P. Human dendritic cells express neuronal Eph receptor tyrosine kinases: role of EphA2 in regulating adhesion to fibronectin. Blood. 2003;102:4431–4440. doi: 10.1182/blood-2003-02-0500. [DOI] [PubMed] [Google Scholar]
- 359.Becker E, Huynh-Do U, Holland S, Pawson T, Daniel TO, Skolnik EY. Nck-interacting Ste20 kinase couples Eph receptors to c-Jun N-terminal kinase and integrin activation. Mol Cell Biol. 2000;20:1537–1545. doi: 10.1128/MCB.20.5.1537-1545.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Parri M, Taddei ML, Bianchini F, Calorini L, Chiarugi P. EphA2 reexpression prompts invasion of melanoma cells shifting from mesenchymal to amoeboid-like motility style. Cancer Res. 2009;69:2072–2081. doi: 10.1158/0008-5472.CAN-08-1845. [DOI] [PubMed] [Google Scholar]
- 361.Parri M, Buricchi F, Giannoni E, Grimaldi G, Mello T, Raugei G, Ramponi G, Chiarugi P. EphrinA1 activates a Src/focal adhesion kinase-mediated motility response leading to rho-dependent actino/myosin contractility. J Biol Chem. 2007;282:19619–19628. doi: 10.1074/jbc.M701319200. [DOI] [PubMed] [Google Scholar]
- 362.Zimmer G, Kastner B, Weth F, Bolz J. Multiple effects of ephrin-A5 on cortical neurons are mediated by SRC family kinases. J Neurosci. 2007;27:5643–5653. doi: 10.1523/JNEUROSCI.0954-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Vindis C, Cerretti DP, Daniel TO, Huynh-Do U. EphB1 recruits c-Src and p52Shc to activate MAPK/ERK and promote chemotaxis. J Cell Biol. 2003;162:661–671. doi: 10.1083/jcb.200302073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Clifford N, Smith LM, Powell J, Gattenlohner S, Marx A, O’Connor R. The EphA3 receptor is expressed in a subset of rhabdomyosarcoma cell lines and suppresses cell adhesion and migration. J Cell Biochem. 2008;105:1250–1259. doi: 10.1002/jcb.21926. [DOI] [PubMed] [Google Scholar]
- 365.Hjorthaug HS, Aasheim HC. Ephrin-A1 stimulates migration of CD8+CCR7+ T lymphocytes. Eur J Immunol. 2007;37:2326–2336. doi: 10.1002/eji.200737111. [DOI] [PubMed] [Google Scholar]
- 366.Miao H, Nickel CH, Cantley LG, Bruggeman LA, Bennardo LN, Wang B. EphA kinase activation regulates HGF-induced epithelial branching morphogenesis. J Cell Biol. 2003;162:1281–1292. doi: 10.1083/jcb.200304018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Gallo G, Yee HF, Jr, Letourneau PC. Actin turnover is required to prevent axon retraction driven by endogenous actomyosin contractility. J Cell Biol. 2002;158:1219–1228. doi: 10.1083/jcb.200204140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Puschmann TB, Turnley AM. Eph receptor tyrosine kinases regulate astrocyte cytoskeletal rearrangement and focal adhesion formation. J Neurochem. 2010;113:881–894. doi: 10.1111/j.1471-4159.2010.06655.x. [DOI] [PubMed] [Google Scholar]
- 369.Ogawa K, Wada H, Okada N, Harada I, Nakajima T, Pasquale EB, Tsuyama S. EphB2 and ephrin-B1 expressed in the adult kidney regulate the cytoarchitecture of medullary tubule cells through Rho family GTPases. J Cell Sci. 2006;119:559–570. doi: 10.1242/jcs.02777. [DOI] [PubMed] [Google Scholar]
- 370.Yang NY, Pasquale EB, Owen LB, Ethell IM. The EphB4 receptor-tyrosine kinase promotes the migration of melanoma cells through Rho-mediated actin cytoskeleton reorganization. J Biol Chem. 2006;281:32574–32586. doi: 10.1074/jbc.M604338200. [DOI] [PubMed] [Google Scholar]
- 371.Tolias KF, Bikoff JB, Kane CG, Tolias CS, Hu L, Greenberg ME. The Rac1 guanine nucleotide exchange factor Tiam1 mediates EphB receptor-dependent dendritic spine development. Proc Natl Acad Sci USA. 2007;104:7265–7270. doi: 10.1073/pnas.0702044104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 372.Huai J, Drescher U. An ephrin-A-dependent signaling pathway controls integrin function and is linked to the tyrosine phosphorylation of a 120 kDa protein. J Biol Chem. 2001;276:6689–6694. doi: 10.1074/jbc.M008127200. [DOI] [PubMed] [Google Scholar]
- 373.Hamada K, Oike Y, Ito Y, Maekawa H, Miyata K, Shimomura T, Suda T. Distinct roles of ephrin-B2 forward and EphB4 reverse signaling in endothelial cells. Arterioscler Thromb Vasc Biol. 2003;23:190–197. doi: 10.1161/01.ATV.0000055440.89758.C2. [DOI] [PubMed] [Google Scholar]