Abstract
Endocrine gland-derived vascular endothelial growth factor (EG-VEGF) is an angiogenic factor reported to be specific for endocrine tissues, including the placenta. Its biological activity is mediated via two G protein-coupled receptors, prokineticin receptor 1 (PROKR1) and prokineticin receptor 2 (PROKR2). We have recently shown that (i) EG-VEGF expression peaks between the 8th and 11th weeks of gestation, (ii) its mRNA and protein levels are up-regulated by hypoxia, (iii) EG-VEGF is a negative regulator of trophoblast invasion and (iv) its circulating levels are increased in preeclampsia (PE), the most threatening pathology of pregnancy. Here, we investigated the regulation of the expression of EG-VEGF and its receptors by hCG, a key pregnancy hormone that is also deregulated in PE. During the first trimester of pregnancy, hCG and EG-VEGF exhibit the same pattern of expression, suggesting that EG-VEGF is potentially regulated by hCG. Both placental explants (PEX) and primary cultures of trophoblasts from the first trimester of pregnancy were used to investigate this hypothesis. Our results show that (i) LHCGR, the hCG receptor, is expressed both in cyto- and syncytiotrophoblasts, (ii) hCG increases EG-VEGF, PROKR1 and PROKR2 mRNA and protein expression in a dose- and time-dependent manner, (iii) hCG increases the release of EG-VEGF from PEX conditioned media, (iv) hCG effects are transcriptional and post-transcriptional and (v) the hCG effects are mediated by cAMP via cAMP response elements present in the EG-VEGF promoter region. Altogether, these results demonstrate a new role for hCG in the regulation of EG-VEGF and its receptors, an emerging regulatory system in placental development.
Keywords: EG-VEGF, Prokineticin, hCG, Placenta, Pregnancy
Introduction
The process of embryo implantation and trophoblast invasion is the most limiting factor for a successful pregnancy. Molecular interactions at the embryo–maternal interface during the time of adhesion and subsequent invasion are crucial for implantation [1]. Failure of these interactions can lead to preeclampsia (PE), early pregnancy loss or intrauterine growth retardation (IUGR). There is evidence suggesting that cytokines that are produced by the developing placenta play an important role in these processes [1]. We have recently determined the role of a new actor in these processes [2, 3], endocrine gland-derived vascular endothelial growth factor (EG-VEGF), also known as prokineticin 1 (PROK1).
EG-VEGF is a growth factor that was found to be specifically expressed in endocrine tissues, including the testis, adrenal gland, ovary, and placenta [4]. In addition, it was shown to promote tissue-specific angiogenesis in endocrine organs [5–9]. EG-VEGF acts via two G protein-coupled receptors, termed prokineticin receptor 1 (PROKR1) and prokineticin receptor 2 (PROKR2) [10]. Recent data from our group reported the expression of EG-VEGF and its receptors in human and mouse placenta and its role during the first trimester of pregnancy [3, 11, 12]. We have shown that EG-VEGF is localized to the syncytial layer of the human placenta, where the expression levels of both EG-VEGF and its receptors are high, with the strongest expression peaking between the 8th and 11th weeks of gestation (wg). We have also shown that EG-VEGF controls trophoblast invasion and that its circulating levels are significantly elevated in PE [3, 11]. More recently, we have also shown that EG-VEGF is a potent angiogenic factor in the placenta and have established its angiogenic role during pregnancy [2]. Altogether, these findings suggest that EG-VEGF is directly involved in normal placental development and that its expression should be finely regulated. We and others have shown that the expression of EG-VEGF and its receptors is up-regulated by hypoxia [4, 11]. During placental development, the hypoxic environment occurs from the beginning of implantation to the end of the first trimester. However, the strongest expression of EG-VEGF is between the 8th and 11th wg, suggesting that factors other than hypoxia might regulate the EG-VEGF/PROKR1/PROKR2 system. To date, little is known about the regulation of EG-VEGF and its receptors, and there is no known cause for the peak in EG-VEGF expression at the end of the first trimester of pregnancy. During the first trimester of pregnancy, one dominant hormone, hCG (human chorionic gonadotropin), exhibits a similar pattern of expression to that of EG-VEGF and displays similar effects on placental development [13–15]. One of the earliest endocrine roles of hCG is to stimulate the corpus luteum to produce enough progesterone to establish pregnancy at the outset. In the placenta, hCG is well known to facilitate trophoblastic differentiation [14, 15] and has been reported to induce the expression of specific genes, such as vascular endothelial growth factor, leukemia inhibitory factor, and metalloproteinase-9, all of which are central to the establishment of the feto-maternal interface [16, 17]. In human placenta, hCG is primarily produced by the syncytiotrophoblast and, to a certain extent, by the cytotrophoblast [18, 19]. In normal pregnancies, detectable levels of hCG begin to appear in the maternal circulation approximately 2–3 weeks after conception, and the peak in hCG levels is observed at ~8–9 wg before declining significantly in the later stages of pregnancy. There is no discernible rhythmicity in the secretion of hCG during the day, but appreciable fluctuations in the levels of hCG in plasma have been observed over time on the same day in the same pregnant woman. Additionally, it is well known that the half-life of hCG is quite long (24 h) [20], suggesting a maintenance of hCG concentrations even in the intervals between hCG secretion [20]. High serum hCG levels at mid-late pregnancy have been associated with PE, IUGR and Down’s syndrome (DS) [21].
The hormone hCG transduces signals by binding to its specific receptor (LHCGR). Binding of hCG to its receptor generates signal transduction via the activation of the associated heterotrimeric G-proteins and the consequent activation of protein kinase A (PKA) through cAMP mobilization, as well as an increase in intracellular calcium through the inositol triphosphate/phospholipase C pathway [22, 23]. LHCGR expression in human placenta has been reported by many groups [24, 25]; however, the precise sites of its expression and the types of receptors expressed throughout pregnancy are still under investigation [25–28]. This discrepancy is probably due to the multiple isoforms that have been described for this receptor. It has also been reported that, in early pregnancy, the LHCGR is truncated and probably non-functional until 9 wg [26, 27, 29].
Because hCG has long been associated with the initiation and maintenance of pregnancy and because our recent findings propose EG-VEGF as a new factor that is directly involved in human placentation, we hypothesized that hCG might be involved in the regulation of EG-VEGF and its receptors. In the present study, we determined the effect of hCG on EG-VEGF secretion, established its effect on EG-VEGF expression both at the mRNA and protein levels, and determined its effect on the expression of PROKR1 and PROKR2. More importantly, we characterized the molecular mechanism by which hCG regulates this new factor and its receptors.
Materials and methods
Tissue collection
Forty-eight first-trimester human placentas at 6–11 wg were obtained from the elective terminations of pregnancies. Shortly after collection, the tissue was either fixed in paraformaldehyde at room temperature for immunohistochemistry, flash frozen in liquid nitrogen and stored at −80°C to be used for RNA and protein extraction or placed in ice-cold Hanks’ balanced salt solution (Ca2+ and Mg2+ HBSS) and transported to the laboratory for the generation of in vitro and ex vivo primary cultures. Twenty-nine placentas from 6 to 11 wg were used for primary cultures. The collection and processing of human placentas were approved by the University Hospital Ethics Committee, and informed consent was obtained from each patient.
Human villous explant cultures
Villous explant cultures were established from the first-trimester human placentas at 6–11 wg. Small fragments of placental villi (15–20 mg wet weight) were placed into 48-well plates that had been pre-coated with 150 μl per well of diluted Matrigel (Matrigel/DMEM-F12) (Becton–Dickinson, Le Pont-de-Claix, France) and polymerized at 37°C for 30 min. Explants were cultured in DMEM-Ham’s F-12 (DMEM/F12; Invitrogen, Cergy Pontoise, France) medium supplemented with 100 μg/ml streptomycin and 100 U/ml penicillin, pH 7.4. After 24 h of culture, the medium was changed, and the explants were incubated in the absence or presence of hCG (10–100 IU/ml) (Sigma-Aldrich, France) or forskolin (10 μM, Sigma-Aldrich) for 0–48 h. Villous explants were maintained in culture for 72 h. Explants from a single placenta were used in triplicate for each time point. For statistical analysis, the (n) value represents the number of placentas and not explants. In some experiments, explants were treated with the following compounds: 50 μg/ml 5,6-dichloro-1-β-d-ribofuranosylbenzimidazole (DRB), a potent RNA polymerase inhibitor; 10 μg/ml cycloheximide (CHX), a translation inhibitor; 100 μM Rp-cAMP (Rp-diastereomer of adenosine-3′,5′-cyclic monophosphorothioate, Sigma-Aldrich), a diastereomer of cAMP that competitively binds to the regulatory subunit of PKA to prevent cAMP-induced dissociation and activation of the enzyme (30); and 2 μM of the PKA inhibitor (PKI, 14–22 amide, cell-permeable, myristoylated, CALBIOCHEM), a potent competitive, synthetic peptide inhibitor of cAMP-dependent protein kinase A (PKA). Placental explants were treated with Rp-cAMP and PKI for 10 h.
Immunohistochemistry
Placental tissues collected at 6–11 wg were fixed for 24 h at 4°C in 4% (v/v) paraformaldehyde, embedded in paraffin, and cut into 5-μm sections as described previously (11). Adjacent sections were stained using specific antibodies and the avidin–biotin immunoperoxidase detection method. Endogenous peroxidase activity was quenched by pretreatment with 3% (v/v) hydrogen peroxide in methanol for 30 min. Rabbit polyclonal antibodies that were developed in our laboratory were used to detect EG-VEGF, PROKR1 and PROKR2 (Covalab, Lyon, France). The hCG receptor was detected using the human LHCGR antibody peptide 28–77 (Santa Cruz Biotechnology). For immunohistochemical detection, antibodies were incubated with the tissue sections for 18 h at 4°C and used at final concentrations of 0.33 μg/ml for anti-EG-VEGF, 0.84 μg/ml for anti-PROKR1, 0.84 μg/ml for anti-PROKR2, and 1 μg/ml for anti-LHCGR. The tissue sections were subsequently washed three times with PBS and incubated with biotinylated goat anti-rabbit IgG (1:250 dilution in blocking solution; Sigma-Aldrich, Saint-Quentin Fallavier, France) for 1 h at 4°C. After three PBS washes, the slides were incubated with an avidin: biotin complex (Vectastain ABC kit; Vector Laboratories, Burlingame, CA) for 1 h. After a final PBS wash, the immunoreactive proteins were visualized after the addition of 3,3-diaminobenzidine (Dako, Trappes, France) for 2 min and then counterstained with hematoxylin. Control sections were treated with antibodies that had been pre-absorbed overnight at 4°C with the appropriate antigen peptides or without primary antibodies.
Western-blot analysis
Frozen placental samples and cultured trophoblast cells were homogenized in RIPA lysis buffer [50 mM Tris–HCl (pH 7.5), 150 mM NaCl, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate, 1% Triton X-100, 1 mM PMSF, 5 μg/ml leupeptin, 5 μg/ml aprotinin] and processed as previously described (3). Protein extracts were electrophoretically separated on 0.1% sodium dodecyl sulfate-12% polyacrylamide gels and electrically transferred onto 0.45 μm nitrocellulose membranes. The membranes were blotted with antibodies against PROKR1, PROKR2 and LHCGR. The PROKR1 and PROKR2 antibodies were both used at a final concentration of 0.84 μg/ml, and the LHCGR antibody was used at 0.75 μg/ml. The intensities of the immunoreactive bands were measured by scanning the photographic film and analyzing the images on a desktop computer using Scion Image software (version 4.0.2; Scion Corp., Frederick, MD). The mean pixel density for each band was analyzed to obtain relative OD units for the PROKR1 and PROKR2 proteins. To standardize for sample loading, the blots were subsequently stripped using a commercially available kit following the manufacturer’s instructions (Re-blot; Millipore) and re-probed with an anti-β-actin antibody (Sigma-Aldrich, France) as an internal control for protein loading.
EG-VEGF ELISA
EG-VEGF was measured by ELISA (PeproTech, France) in conditioned media from placental explants that were or were not treated with hCG (10–100 IU/ml). Two separate standard curves were constructed to allow accurate readings of samples at the upper and lower ranges of the assay. All samples were in the linear range of the standard curves. The detection limit of the assay was 16 pg/ml.
Isolation and treatment of trophoblasts
Placental cytotrophoblasts were isolated from first-trimester human placentas (9–11 wg, n = 14) and cultured as previously described [30]. Briefly, the tissue was thoroughly washed in 50 ml cold sterile HBSS until the supernatant was nearly free of blood. Areas rich in chorionic villi were selected and were minced into small pieces between scalpels. The tissue was incubated in HBSS containing trypsin (0.25%) and DNase (0.2 mg/ml) for 30-min digestions. The dispersed placental cells were filtered through 100-μm nylon gauze and loaded onto a discontinuous Percoll gradient (5–70% in 5% steps of 3 ml each) and then centrifuged at 1,200 × g for 20 min at room temperature to separate the different cell types. The cytotrophoblast cells that sedimented between the density markers of 1.049 and 1.062 g/ml were collected and washed with DMEM. Isolated cells were then incubated with anti-CD9 antibodies and subjected to negative immunomagnetic separation using MiniMacs columns (Miltenyi Biotech, Paris, France). The dispersed trophoblasts were cultured for 24 h at 37°C in 5% CO2/95% air to allow attachment. The cells were then divided into two groups; one group was treated 1 day after the plating to ensure the cytotrophoblast phenotype, while the other group was allowed to differentiate into a syncytiotrophoblast phenotype and treated 48 h later. Both cultures were treated with hCG (10–100 IU/ml). In some experiments, trophoblast cells were treated with DRB (50 μg/ml) or CHX (10 μg/ml). Cell viability as assessed by Trypan blue exclusion was greater than 95% before and after incubation.
RNA isolation and RT-qPCR analysis
Total RNA was extracted from placental tissue using TRIzol reagent (Invitrogen, USA) according to the manufacturer’s instructions. To remove any genomic DNA contamination, total RNA was treated with RNase-free DNase I treatment (Qiagen). The total RNA concentration was determined using Nanodrop.
Reverse transcription was performed on 0.5 μg of total RNA with Superscript II-RNaseH reverse transcriptase (Invitrogen, Cergy Pontoise, France) under the conditions recommended by the manufacturer. The expression levels of EG-VEGF, PROKR1, and PROKR2 mRNA and 18S rRNA was quantified by real-time RT-PCR using a Light Cycler apparatus (Roche Diagnostics, Meylan, France). The PCR was performed using the primers shown in Table 1 and SYBR green PCR core reagents (LightCycler-FastStart Master SYBR Green I, Roche Diagnostics) according to the manufacturer’s instructions. In addition, several control reactions were routinely run in parallel, including RT-PCR run in the absence of reverse transcriptase to confirm the absence of genomic DNA contamination and reverse transcription reactions without RNA to check for reagent contamination. PCR conditions were as follows: step 1, 94°C for 10 min; step 2, 45 cycles consisting of 95°C for 15 s, the temperature indicated in Table 1 for 5 s, and 72°C for 10 s. The results were normalized to 18S rRNA expression levels. To assess the linearity and efficiency of PCR amplification, standard curves for all transcripts were generated with serial dilutions of trophoblast cDNA. A melt curve analysis was performed on the products of the amplification reaction to ascertain the melting temperatures of the products.
Table 1.
Primers used for RT-qPCR
| Gene | Forward primer (5′–3′) | Reverse primer (5′–3′) | (°C) |
|---|---|---|---|
| EG-VEGF | AGGTCCCCTTCTTCAGGAAACG | TCCAGGCTGTGCTCAGGAAAAG | 56 |
| PROKR1 | GTCCTCGTCATTGTCAAGAGCC | AAACACGGTGGGGAAGAAGTCG | 58 |
| PROKR2 | CATCCCATCGCCTTACTTTGC | CTTTTCCTTCACGAACACAGTGG | 58 |
| 18S | TTGTTGGTTTTCGGAACTGAGGC | GGCAAATGCTTTCGCTCTGGTC | 60 |
DNA transfection and dual-luciferase activity assay
Cos7 cells (100,000 cells/12-well plates) were transfected in Opti-MEM (Invitrogen) using Lipofectamine-2,000 (Invitrogen) with 0.285 μg pGL3b-luc and 0.500 μg pGL3-pEG-luc. The pGL3b plasmid was purchased from Promega (Madison, WI, USA). Twenty-four hours after transfection, the cells were treated with or without 8-bromo-cAMP (Sigma-Aldrich, France) for 4 or 6 h. Firefly luciferase activities were measured sequentially with the Dual-Luciferase reporter assay (Promega). The results are expressed as the ratios of firefly luciferase activity per μg of proteins.
Mutagenesis and functional analysis
The cAMP binding sites were individually mutated using site-directed mutagenesis. Complementary primers were designed for each mutation. Mutant constructs were generated by PCR with PFU turbo (Promega, Madison, WI) using 60 ng of each primer, 20 ng of pGL3-pEG-luc as the template, 250 μM dNTP, 2 U PFU turbo polymerase and 1 × PFU buffer in a total volume of 50 μl. The recommended cycling conditions were adapted as follows to permit amplification of the full plasmid containing the desired mutation: denaturation at 95°C for 60 s; 18 cycles of 95°C for 60 s, 55°C for 60 s, 68°C for 10 min; final extension at 72°C for 10 min. A DpnI digestion was performed prior to bacterial transformation of the plasmid DNA to eliminate the parent promoter plasmid. All oligonucleotides used for these procedures are given in Fig. 5A (mutations highlighted).
Fig. 5.
Mechanism by which hCG up-regulates EG-VEGF expression. A Effect of 8-bromo-cAMP on EG-VEGF promoter activation in Cos7 cells transfected with pGL3b pEG-VEGF or pGL3b (used as control). Luciferase activity was measured after 4 h and 6 h of cAMP stimulation. Data represent the mean ± SEM from three independent experiments. (*p < 0.05). B Effect of 8-bromo-cAMP on EG-VEGF promoter activation in Cos7 cells transfected with the following plasmids: pGL3b, pGL3b pEG-VEGF, pGL3b pEG-VEGF mutated at the CRE1 site, pGL3b pEG-VEGF mutated at the CRE2 site, and pGL3b pEG-VEGF mutated at the CRE1 and CRE2 sites. Luciferase activity was measured after 6 h of cAMP stimulation. Data represent the mean ± SEM from three independent experiments
For functional analysis, Cos7 cells were plated in a 24-well plate the day before transfection to achieve a density of 70–80% confluence at the day of the transfection. The cells were transiently transfected with 500 ng of the pGL3-pEG-luc expression plasmid or empty pGL3 using Lipofectamine-2,000 (Invitrogen) according to the manufacturer’s instructions. After 48 h, the transfection medium was removed and replaced with fresh medium with or without 500 μM 8-bromo-cAMP and 200 μM IBMX. After 6 h of incubation, the cells were lysed using passive lysis buffer (Promega, Madison, WI) as per the manufacturer’s instructions. The Dual-Luciferase assay system (Promega, Madison, WI) was used to assess the luciferase activity in the cell lysates using a Tecan Infinite M200 microplate reader (Tecan, France). The fold stimulation of the promoter activity was calculated after normalizing the reporter firefly luciferase values. The results are expressed as the ratios of firefly luciferase activity per μg of protein.
Statistical analysis
All data are expressed as the mean ± SE. Statistical comparisons were made using non-parametric and Mann–Whitney tests. Calculations were performed using SigmaStat (Jandel Scientific Software, San Rafael, CA).
Results
Expression of LHCGR in human placenta
The first series of experiments was conducted to determine the sites and levels of expression of hCG receptors in human placenta during the first and third trimesters of pregnancy. Figure 1A shows representative photograph of LHCGR immunolocalization within the placental villi of human placenta at 8 wg (a) and 10 wg (b). Immunostainings show that LHCGR is highly expressed by cyto- and syncytiotrophoblast cells. Strong expression could also be observed in Hofbauer cells. Negative controls are shown in (c) and (d). Figure 1B shows a representative Western blot that illustrates the levels of LHCGR in human first trimester and term placentas. Two major bands with molecular masses of 55 and 70 kDa are observed. A slight but non-significant decrease in the intensity of the bands is observed toward the end of the first trimester. The strongest expression, however, is observed in term placentas.
Fig. 1.
LHCGR protein expression in placental tissue at 8 and 10 weeks of gestation. A LHCGR expression in human placental sections. The photographs in a and b show chorionic villi sections immunostained with anti-LHCGR antibody at 8 wg and 10 wg, respectively. The photographs in c and d show tissue sections incubated with pre-immune sera at 8 and 10 wg, respectively. Cytotrophoblast (Ct), Hofbauer cells (Ho), syncytiotrophoblast (St). Scale bar 50 μm. B Representative Western-blot analysis of LHCGR expression in human placental extracts collected at 6–8 wg, 9–11 wg and at term. Two major bands with molecular masses of 55 and 70 kDa were observed at all gestational periods
Effect of hCG on EG-VEGF protein expression and secretion
In previous reports from our group [11], we have shown that EG-VEGF is highly expressed by the syncytiotrophoblast layer in the human placenta during the first trimester of pregnancy. We have also shown that this cytokine is secreted and can be measured in human serum. We therefore investigated the effect of hCG on EG-VEGF expression and secretion in the conditioned media of explant cultures (6–11 wg) that were or were not treated with hCG. It was particularly relevant to study the effects of hCG on EG-VEGF expression in an organotypic system in which the villous tissue architecture is maintained. Placental villous explants in culture preserve the topology of the intact villi and mimic physiological responses.
Effect of hCG on EG-VEGF protein expression
The analysis of the effects of hCG on EG-VEGF protein expression in placentas from 6 to 11 wg showed differential responses with respect to the gestational age of the placentas examined. The placentas that were collected from 6 to 8 wg were less sensitive to hCG treatment compared to those collected from 9 to 11 wg. Figure 2 shows representative photographs of control and treated placental explants (10 and 100 IU/ml of hCG) at 8 and 10 wg. Whereas a concentration of 10 IU/ml was sufficient to show a significant increase in EG-VEGF protein expression at 10 wg (h), a ten-fold higher concentration was needed to see a significant effect at 8 wg (c). Photographs in (d, e, f) and (j, k, l) show negative stainings in placental sections of 8 and 10 wg, respectively.
Fig. 2.
Effect of hCG on EG-VEGF expression. The photographs in this figure show EG-VEGF staining in sections of placental explants that have been cultured in the absence or presence of hCG (10 and 100 IU/ml) at 8 and 10 wg. a and g show non-treated explant sections at 8 and 10 wg, respectively. b and h show sections of explants that have been treated with 10 IU/ml hCG for 48 h at 8 and 10 wg, respectively. c and i show sections of explants that have been treated with 100 IU/ml hCG for 48 h at 8 and 10 wg, respectively. Undersized photographs in d–f and in j–l show tissue sections incubated with pre-immune serum to EG-VEGF at 8 wg and 10 wg, respectively. Cytotrophoblast (Ct), Hofbauer cells (Ho), syncytiotrophoblast (St). Scale bar 50 μm, (d–f) and (j–l)
Effect of hCG on EG-VEGF protein secretion
We observed similar effects of hCG on EG-VEGF secretion as we observed on EG-VEGF expression. Figure 3A and B show the temporal and dose–response regulations of EG-VEGF secretion by hCG in explants from placentas of 6–8 wg and 9–11 wg, respectively. In the 6–8 wg placentas, hCG increased EG-VEGF secretion in a dose-dependent manner with significant effects observed at 50 and 100 IU/ml. In the 9–11 wg placentas, hCG exhibited an opposite dose–response effect, with the lowest concentration of hCG being the most effective.
Fig. 3.
Effect of hCG on EG-VEGF secretion. A, B Temporal (0–48 h) and dose-dependence analysis of the effect of hCG (10–100 IU/ml) on EG-VEGF secretion in placental conditioned media collected from placental explants at 6–8 wg and 9–11 wg, respectively. C Time course (0–48 h) of the effect of forskolin (10 μM) on EG-VEGF secretion. D Effects of the PKA inhibitors Rp-cAMP (100 μM) and PKI (2 μM) on the effects of hCG (25 μM) and forskolin (10 μM) on EG-VEGF secretion. The treatments lasted for 10 h. Data represent the mean ± SEM from three independent experiments (*p < 0.05, **p < 0.001)
The effects of hCG on EG-VEGF secretion were confirmed using forskolin, an activator of protein kinase A (PKA). Because both the hCG and EG-VEGF expression profiles show the highest levels of expression between 9 and 11 wg, we used placentas at this period of gestation for all mechanistic experiments. Figure 3C shows a time course of the effect of forskolin on EG-VEGF secretion. There was a significant increase in EG-VEGF secretion in response to forskolin treatment, suggesting an activation of the cAMP pathway. To further demonstrate that the effect of hCG on EG-VEGF secretion involves the cAMP pathway, we performed a set of experiments in which the effects of hCG (25 IU/ml) and forskolin (10 μM) were tested in the absence or presence of protein kinase A inhibitors. Both Rp-cAMP (100 μM) and PKA inhibitor (PKI, 2 μM) were used. The treatments lasted for 10 h. As expected, both hCG and forskolin significantly increased EG-VEGF secretion; these effects were abolished in the presence of the protein kinase A inhibitors PKI and Rp-cAMP (Fig. 3D).
hCG effect on EG-VEGF mRNA expression
To investigate the effect of hCG on EG-VEGF mRNA expression, both primary human placental explants and primary trophoblast cells isolated from first-trimester placentas were used. Figure 4A, B and C show the temporal and dose–response regulation of EG-VEGF by hCG in explants and syncytiotrophoblasts, respectively. In placental explants, 10 IU/ml of hCG significantly increased EG-VEGF expression with a peak at 12 h (Fig. 4A). In the trophoblasts, the highest effect was induced by the addition of 10 IU/ml of hCG with a decrease in the response to hCG at higher concentrations (Fig. 4B, C). The temporal and dose–response experiments both demonstrated the positive effect of hCG on EG-VEGF mRNA expression but also divulged the existence of a self-regulatory phenomenon in the effect of hCG on EG-VEGF expression when the treatment lasted over 12 h (Fig. 4A) or was present at high concentrations (Fig. 4B, C). A similar type of regulation has been attributed to a temporary internalization of LHCGR from the plasma membrane [31–33]. As for the effect of hCG on EG-VEGF secretion, we also investigated how PKA inhibitors modulated the effect of hCG on EG-VEGF mRNA expression. Figure 4D shows that both PKI and Rp-cAMP abolished the effects of hCG and forskolin on EG-VEGF mRNA expression, suggesting the direct involvement of the cAMP pathway in this regulation. To further characterize the mechanism by which hCG regulates EG-VEGF mRNA expression, we examined the effects of transcription and translation inhibitors on the hCG regulation of EG-VEGF expression in syncytiotrophoblast cells. Both the RNA polymerase inhibitor DRB (50 μg/ml) and the translation inhibitor CHX (10 μg/ml) abolished hCG induction of EG-VEGF (Fig. 4E). These data suggest that hCG regulates EG-VEGF gene expression at the transcriptional level and that this effect requires protein neosynthesis.
Fig. 4.
Effect of hCG on EG-VEGF mRNA expression in placental explants and in syncytiotrophoblasts. A Temporal effect of hCG (10 IU/ml) on EG-VEGF mRNA expression measured by qRT-PCR in placental explants. Data represent the means ± SEM from 3 independent experiments (*p < 0.05). B Representative analysis of the effect of hCG (10, 50 and 100 IU/ml) on EG-VEGF mRNA expression in syncytiotrophoblast cultures (12 h). C Dose–response effect of hCG (10, 50 and 100 IU/ml) on EG-VEGF mRNA expression in syncytiotrophoblast cultures (12 h) monitored by q-PCR. D Effects of the PKA inhibitors Rp-cAMP (100 μM) and PKI (2 μM) on the effects of hCG (25 μM) and forskolin (10 μM) on EG-VEGF mRNA expression. The treatments lasted for 10 h. E Effects of DRB (50 μg/ml) and CHX (10 μg/ml) on basal and hCG-induced EG-VEGF mRNA expression in primary trophoblast cells. Data represent the mean ± SEM from three independent experiments (*p < 0.05, **p < 0.001)
Mechanism by which hCG up-regulates EG-VEGF expression
It is well established that physiological concentrations of hCG elevate cAMP levels and induce the activation of the protein kinase A signaling pathway (34, 35). Therefore, we evaluated whether the up-regulation of EG-VEGF gene expression by hCG in placental cells was mediated by this pathway. To investigate the involvement of cAMP in the effect of hCG on EG-VEGF expression, we cloned the promoter region of the EG-VEGF gene (−3,000 bp), which contains two canonical cAMP response elements: CRE1 (cyclic AMP responsive element 1), located at −1,728 bp, and CRE2 (cyclic AMP responsive element-like 2), located at −383 bp (Fig. 5A). We then performed gene reporter assays with Cos7 cells that transiently expressed the luciferase gene under the control of the EG-VEGF promoter. Twenty-four hours after transfection, the cells were treated with or without 8-bromo-cAMP for 4 or 6 h. Figure 5B shows that cAMP treatment significantly increased EG-VEGF promoter activity both at 4 and 6 h compared to the control condition without the EG-VEGF promoter. These results indicate that cAMP enhances EG-VEGF promoter activity and regulates EG-VEGF promoter expression at the transcriptional level. To determine whether CRE1 or CRE2 or both are involved in EG-VEGF activation in response to cAMP, we mutated CRE1, CRE2 or both. Mutation of CRE1 slightly modulated the effect of cAMP on the EG-VEGF promoter; however, the mutation of CRE2 completely abolished this effect. The loss of an effect of cAMP on the EG-VEGF promoter was also observed when both CRE1 and CRE2 were mutated (Fig. 5C). Altogether, these results demonstrate that the increase in EG-VEGF mRNA production in response to hCG is largely mediated via the activation of CRE2.
hCG effects on the expression of PROKR1 receptors
Because of the significant effect of hCG on EG-VEGF expression in placental cells, we wondered whether this key hormone also regulates the expression of the EG-VEGF receptors PROKR1 and PROKR2. Figure 6 shows the effect of hCG on PROKR1 expression in placental explants and in cytotrophoblast cells collected at 9–11 wg. Figure 6A shows the effect of hCG on PROKR1 mRNA expression. hCG treatment (12 h) significantly increased PROKR1 mRNA levels. This effect was, however, only observed at 100 IU/ml and not at lower concentrations. This effect was substantiated at the protein level. Protein expression in placental explants was assessed by immunohistochemistry on explants that had been incubated in the absence or presence of hCG (10 and 100 IU/ml) for 48 h (Fig. 6B), while protein expression in cytotrophoblasts was examined by Western blot on protein extracted from cells that had been incubated in the absence or presence of hCG (100 IU/ml) for 48 h (Fig. 6C).
Fig. 6.
Effect of hCG on PROKR1 mRNA and protein expression. A Dose–response effect of hCG (12 h) on PROKR1 mRNA expression measured by qRT-PCR in cytotrophoblast cells. B PROKR1 staining in sections of placental explants that have been cultured in the absence or presence of hCG (10 and 100 IU/ml) at 10 wg. Non-treated explant sections are shown in a, and treated explant sections with hCG at 10 IU/ml and 100 IU/ml are shown in b and c, respectively. Undersized photographs in d, e, and f show tissue sections incubated with pre-immune serum to PROKR1. Cytotrophoblast (Ct), Hofbauer cells (Ho), syncytiotrophoblast (St). Scale bar 50 μm. C Representative Western-blot analysis of PROKR1 expression in cytotrophoblasts incubated in the absence or presence of hCG (100 IU/ml). D Histograms of the mean relative OD of PROKR1 protein signals normalized to β-actin. Data are mean ± SEM (**p < 0.001)
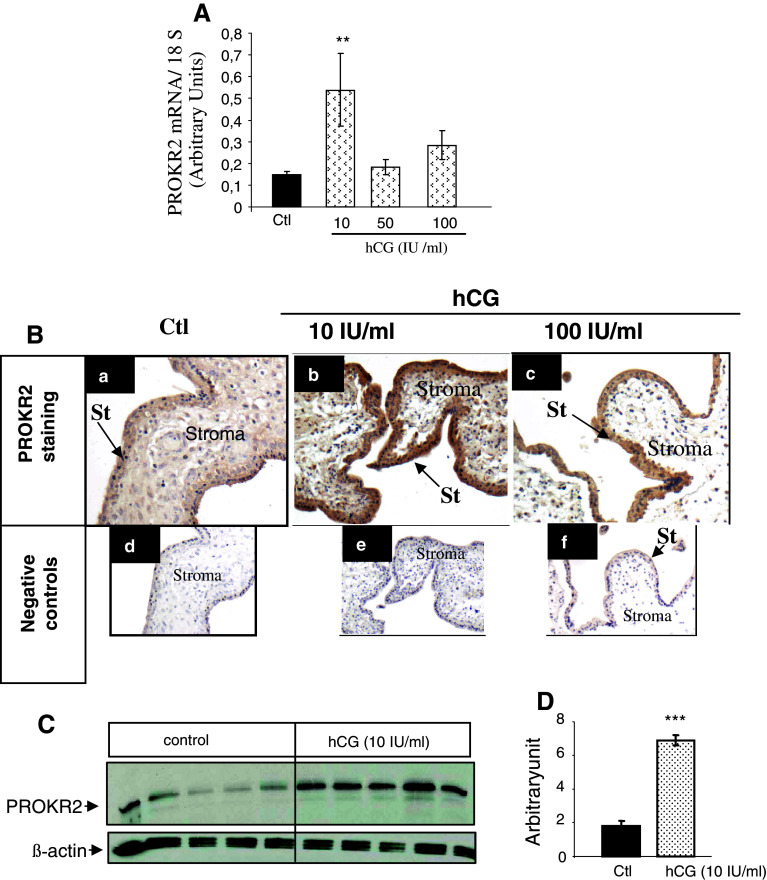
Effects of hCG on the expression of PROKR2 receptors
The effect of hCG on PROKR2 expression was also investigated in placental explants and in syncytiotrophoblast cells. Figure 7 shows the effect of hCG on PROKR2 expression in syncytiotrophoblasts and in placental explants collected at 9–11 wg. Figure 7A shows the effect of hCG treatment (12 h) on PROKR2 mRNA expression. In contrast to the effects of hCG on PROKR1, its effects on PROKR2 mRNA levels were significantly increased in response to ten-fold lower concentrations of hCG. This effect was substantiated at the protein level. Protein expression in placental explants was assessed by immunohistochemistry on explants that had been incubated in the absence or presence of hCG (10 and 100 IU/ml) for 48 h (Fig. 7B). In the syncytiotrophoblasts protein expression was assessed by Western blot on cells that had been incubated in the absence or presence of 10 IU/ml hCG (Fig. 7C). Altogether, these findings suggest that PROKR1 and PROKR2 are finely regulated by hCG.
Fig. 7.
Effect of hCG on PROKR2 mRNA and protein expression. A Dose–response effect of hCG (12 h) on PROKR2 mRNA expression, measured by qRT-PCR, in syncytiotrophoblast cells. B PROKR2 staining in sections of placental explants that have been cultured in the absence or presence of hCG (10 and 100 IU/ml) at 10 wg. Non-treated explants sections are shown in a and treated explant sections with hCG at 10 IU/ml and (100 IU/ml) are shown in b and c, respectively. Undersized photographs in d, e, and f show tissue sections incubated with pre-immune serum to PROKR2. Cytotrophoblast (Ct), Hofbauer cells (Ho), syncytiotrophoblast (St). Scale bar 50 μm. C Representative Western-blot analysis of PROKR2 expression in syncytiotrophoblast incubated in the absence or presence of hCG (10 IU/ml). D Histograms of the mean relative OD of PROKR2 protein signals normalized to β-actin protein. Data are mean ± SEM (**p < 0.001)
Discussion
During the last decade, several reports have shed light on the role of the new angiogenic factor EG-VEGF in reproductive organs. It is now well established that EG-VEGF plays a role in physiological and pathological ovarian activities [36–38] and in testicular angiogenesis [9] and is involved in the success of endometrial implantation [5, 39, 40]. In the placenta, we [2, 3, 11, 12] and others [41, 42] have demonstrated the role of EG-VEGF during early and late pregnancy, respectively. In four recent publications from our group, we have shown that EG-VEGF and its receptors are abundant in the human placenta during the first trimester of pregnancy, with the highest levels of expression found in the syncytiotrophoblast layer [3, 11]. We have also reported that EG-VEGF controls trophoblast invasion and has angiogenic effects in microvascular endothelial cells [2, 3]. Furthermore, we reported the dysregulation of circulating levels of EG-VEGF in preeclampsia [3]. The regulation of the expression of both EG-VEGF and its receptors was, however, still unknown. We and others have shown that the EG-VEGF gene is actively transcribed under hypoxic conditions via mechanisms that are common to other hypoxia-inducible genes [4, 11]. Nevertheless, additional regulation of EG-VEGF and its receptors by an important pregnancy signal was predictable but had not been investigated.
Here, we demonstrate that the key hormone of placentation, hCG, significantly increases the expression of EG-VEGF and its receptors during the first trimester of pregnancy. The effect of hCG on EG-VEGF was assessed in two physiological systems, placental explants and primary trophoblast cultures. The present findings demonstrate a new physiological regulation between an endocrine hormone released by the syncytiotrophoblast layer that acts in an autocrine manner on the same cell type to increase the expression of EG-VEGF and its receptor PROKR2, which is preferentially expressed by this differentiated cell type (3, 11). hCG also acts in a paracrine manner on the undifferentiated cell type of the placenta, the cytotrophoblast, where it stimulates the expression of PROKR1. Altogether, these results reveal a new role for hCG in human placentation through its activation of the EG-VEGF/PROKR1/PROKR2 system and might explain the peak of expression of this ligand and its receptors during the first trimester of pregnancy (8–11 wg). More importantly, we demonstrated that the effect of hCG on EG-VEGF is transcriptional and post-transcriptional and determined the mechanism by which this hormone up-regulates EG-VEGF production. In silico analysis of the EG-VEGF promoter revealed consensus response elements for different transcription factors, including CREB. Mutation of these sites showed that the effect of hCG on EG-VEGF expression is mediated via the activation of CRE2, which is located at −383 bp in the EG-VEGF promoter region, and not CRE1, which is located at −1,728 bp. Therefore, these data provide evidence of the direct regulation of EG-VEGF transcription upon stimulation by hCG in trophoblast cells (Fig. 8).
Fig. 8.
Proposed model of hCG stimulation of the EG-VEGF/PROKR1 and PROKR2 system in human placenta during the first trimester of pregnancy. A Illustration of placental villi with the new physiological regulation between hCG and EG-VEGF. hCG released from the syncytiotrophoblast layer will activate EG-VEGF and PROKR2 expression in the ST layer in an autocrine manner and PROKR1 expression in the cytotrophoblast layer in a paracrine manner. B Summary of the proposed model according to which PROKR1, expressed by cytotrophoblasts, is only increased in response to high hCG levels. In contrast, EG-VEGF and PROKR2, which are mainly expressed by differentiated syncytiotrophoblasts, are increased in response to low hCG levels
In earlier work from our group, we have shown that EG-VEGF could be measured in circulating serum with the highest levels found during the first trimester of pregnancy (3). These data demonstrate that, during this period of gestation, the placenta is an important source of EG-VEGF and that its local production should be finely controlled. Here, we show for the first time in an ex vivo model, the placental explant, that EG-VEGF is secreted and can be measured in the explant conditioned medium. Moreover, we show that hCG significantly increases this secretion.
During normal pregnancy, hCG secretion peaks at ~8–9 wg with a half-life of 24 h (20), which correlates with the period of EG-VEGF production. Consistent with these observations is the demonstration that placentas collected at 6–8 wg and treated with physiological concentrations of hCG (80–100 IU/ml) had five-fold higher levels of EG-VEGF secretion, which explains the exponential increase in EG-VEGF secretion that was reported earlier at this gestational period [11]. At 9–11 wg, the same concentration of hCG only increased EG-VEGF secretion 1.5-fold, which might explain the gradual decline also reported at this period of gestation [11]. Therefore, the congruent secretion of hCG and EG-VEGF suggests that some of the recently described effects of hCG on placental villi development, such as its effects on placental angiogenesis [17, 43], might be mediated by EG-VEGF. Consistent with these data, Jabbour and colleagues have recently shown that EG-VEGF stimulation of LIF (leukemia inhibitory factor) expression during endometrial implantation in the baboon is controlled by embryonic hCG [39, 40].
When the effect of hCG on EG-VEGF expression and secretion in placental explants was analyzed, a significant up-regulation was observed, and this effect was dose- and time-dependent. Nevertheless, we observed that, at higher doses (100 IU/ml), hCG stimulated EG-VEGF to a lower degree. This phenomenon is probably due to a transient loss of second-messenger production (desensitization), internalization, and/or loss of cell surface receptors (down-regulation) in response to physiological doses of the ligand [32–35, 44]. In our experiments, the decrease in LHCGR response was only observed when explant tissues or isolated syncytiotrophoblasts were used, not when cytotrophoblast cells were used. This observation supports the hypothesis that cyto- and syncytiotrophoblast cells might not express the same forms of LHCGR. These speculations are consistent with previous reports showing that LHCGR might exist as multiple splicing forms in a given tissue [45, 46] and with data showing differential expression levels of LHCGR in cyto- and syncytiotrophoblast cells [18, 19, 47]. Furthermore, we observed a discrepancy in the response to hCG between placentas of 6–8 wg and those of 9–11 wg. This discrepancy could be due to differences in the types of cells and their contents between 6–8 wg and 9–11 wg. In fact, it is well established that the 6–8 wg placentas contain more cytotrophoblast cells than syncytiotrophoblasts, which is due to the higher proliferative index of cytotrophoblast at this gestational age [48–50], whereas the 9–11 wg placentas contain more of the syncytial type of cells. Interestingly, Peter Husslein’s group [51] has clearly demonstrated that syncytiotrophoblast nuclei contain significantly higher levels of transcriptional complexes compared to cytotrophoblasts. In particular, they demonstrated that phosphorylated CREB-1 and ATF-1 (dependent on protein kinase A), which bind cAMP response elements, and the coactivator CBP are primarily located in syncytial nuclei. Furthermore, it is has also been shown that endogenous cAMP levels spontaneously increase sixfold during in vitro syncytium formation [47–49]. Therefore, one can speculate that the discrepancy in the responses between 6–8 wg and 9–11 wg placentas might be due to a difference in cell types and in the transcriptional machinery that they contain. Consistent with this hypothesis, we found that hCG stimulated EG-VEGF and PROKR2 expression at low concentrations (10 IU/ml), whereas PROKR1 stimulation was only observed at higher concentrations of hCG (100 IU/ml). Given the preferential expression of EG-VEGF and PROKR2 in the syncytiotrophoblast and that of PROKR1 in the cytotrophoblast, one can speculate that differential hCG effects are due to differences in the transcriptional machinery present in cyto- and syncytiotrophoblasts. The discrepancy in the responses of PROKR1 and PROKR2 to the same concentrations of hCG might also be explained by the differential composition of their promoter regions. In silico analysis showed that the PROKR2 promoter region exhibits additional potential response elements to cAMP (CRE and Sp1) compared to the PROKR1 promoter.
To date, it is well established that dysregulation in hCG production is tightly associated with the development of major pregnancy pathologies, such as preeclampsia [52–54]. Interestingly, we have shown that EG-VEGF levels are also dysregulated in preeclampsia (3), suggesting that the recently described loop of regulation between hCG and EG-VEGF might contribute to mechanisms underlying the development of placental pathologies during early human pregnancy. In the present work, we provide evidence that hCG up-regulates the expression of EG-VEGF and its receptors PROKR1 and PROKR2 in human placenta and that the hCG regulatory effect on EG-VEGF expression involves the cAMP signal transduction pathway. In summary, our results further support the importance of EG-VEGF in pregnancy and reveal a new loop of physiological regulation between two key factors within the placental villi.
Acknowledgments
We thank the staff of the Department of Gynecology/Obstetrics (Pr. J.P. Schaal) at the University Hospital of Grenoble for giving us access to human placentas. We acknowledge the following sources of funding: INSERM (U1036), University Joseph Fourier, Commissariat à l’Energie Atomique (DSV/iRTSV/BCI), the Région Rhône-Alpes (CIBLE-2008). S.B. was supported by doctoral scholarships from the French Ministry of Education and Research and from the Fondation pour la Recherche Médicale.
References
- 1.Fitzgerald B, Levytska K, Kingdom J, Walker M, Baczyk D, Keating S. Villous trophoblast abnormalities in extremely preterm deliveries with elevated second trimester maternal serum hCG or inhibin-A. Placenta. 2011;32:339–345. doi: 10.1016/j.placenta.2011.01.018. [DOI] [PubMed] [Google Scholar]
- 2.Brouillet S, Hoffmann P, Benharouga M, Salomon A, Schaal JP, Feige JJ, Alfaidy N. Molecular characterization of EG-VEGF-mediated angiogenesis: differential effects on microvascular and macrovascular endothelial cells. Mol Biol Cell. 2010;21:2832–2843. doi: 10.1091/mbc.E10-01-0059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hoffmann P, Saoudi Y, Benharouga M, Graham CH, Schaal JP, Mazouni C, Feige JJ, Alfaidy N. Role of EG-VEGF in human placentation: Physiological and pathological implications. J Cell Mol Med. 2009;13:2224–2235. doi: 10.1111/j.1582-4934.2008.00554.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.LeCouter J, Kowalski J, Foster J, Hass P, Zhang Z, Dillard-Telm L, Frantz G, Rangell L, DeGuzman L, Keller GA, Peale F, Gurney A, Hillan KJ, Ferrara N. Identification of an angiogenic mitogen selective for endocrine gland endothelium. Nature. 2001;412:877–884. doi: 10.1038/35091000. [DOI] [PubMed] [Google Scholar]
- 5.Battersby S, Critchley HO, Morgan K, Millar RP, Jabbour HN. Expression and regulation of the prokineticins (endocrine gland-derived vascular endothelial growth factor and Bv8) and their receptors in the human endometrium across the menstrual cycle. J Clinical Endocrinol Metabolism. 2004;89:2463–2469. doi: 10.1210/jc.2003-032012. [DOI] [PubMed] [Google Scholar]
- 6.Ferrara N, Frantz G, LeCouter J, Dillard-Telm L, Pham T, Draksharapu A, Giordano T, Peale F. Differential expression of the angiogenic factor genes vascular endothelial growth factor (VEGF) and endocrine gland-derived VEGF in normal and polycystic human ovaries. Am J Pathol. 2003;162:1881–1893. doi: 10.1016/S0002-9440(10)64322-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.LeCouter J, Zlot C, Tejada M, Peale F, Ferrara N. Bv8 and endocrine gland-derived vascular endothelial growth factor stimulate hematopoiesis and hematopoietic cell mobilization. Proc Natl Acad Sci USA. 2004;101:16813–16818. doi: 10.1073/pnas.0407697101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Podlovni H, Ovadia O, Kisliouk T, Klipper E, Zhou QY, Friedman A, Alfaidy N, Meidan R. Differential expression of prokineticin receptors by endothelial cells derived from different vascular beds: a physiological basis for distinct endothelial function. Cell Physiol Biochem. 2006;18:315–326. doi: 10.1159/000097609. [DOI] [PubMed] [Google Scholar]
- 9.Samson M, Peale FV, Jr, Frantz G, Rioux-Leclercq N, Rajpert-De Meyts E, Ferrara N. Human endocrine gland-derived vascular endothelial growth factor: expression early in development and in Leydig cell tumors suggests roles in normal and pathological testis angiogenesis. J Clinical Endocrinol Metabolism. 2004;89:4078–4088. doi: 10.1210/jc.2003-032024. [DOI] [PubMed] [Google Scholar]
- 10.Lin DC, Bullock CM, Ehlert FJ, Chen JL, Tian H, Zhou QY. Identification and molecular characterization of two closely related G protein-coupled receptors activated by prokineticins/endocrine gland vascular endothelial growth factor. J Biological Chem. 2002;277:19276–19280. doi: 10.1074/jbc.M202139200. [DOI] [PubMed] [Google Scholar]
- 11.Hoffmann P, Feige JJ, Alfaidy N. Expression and oxygen regulation of endocrine gland-derived vascular endothelial growth factor/prokineticin-1 and its receptors in human placenta during early pregnancy. Endocrinology. 2006;147:1675–1684. doi: 10.1210/en.2005-0912. [DOI] [PubMed] [Google Scholar]
- 12.Hoffmann P, Feige JJ, Alfaidy N. Placental expression of EG-VEGF and its receptors PKR1 (prokineticin receptor-1) and PKR2 throughout mouse gestation. Placenta. 2007;28:1049–1058. doi: 10.1016/j.placenta.2007.03.008. [DOI] [PubMed] [Google Scholar]
- 13.Herr F, Baal N, Reisinger K, Lorenz A, McKinnon T, Preissner KT, Zygmunt M. HCG in the regulation of placental angiogenesis. Results of an in vitro study. Placenta. 2007;28(Suppl A):S85–S93. doi: 10.1016/j.placenta.2007.02.002. [DOI] [PubMed] [Google Scholar]
- 14.Licht P, Russu V, Wildt L. On the role of human chorionic gonadotropin (hCG) in the embryo-endometrial microenvironment: implications for differentiation and implantation. Semin Reprod Med. 2001;19:37–47. doi: 10.1055/s-2001-13909. [DOI] [PubMed] [Google Scholar]
- 15.Malassine A, Cronier L. Hormones and human trophoblast differentiation: a review. Endocrine. 2002;19:3–11. doi: 10.1385/ENDO:19:1:3. [DOI] [PubMed] [Google Scholar]
- 16.Licht P, Fluhr H, Neuwinger J, Wallwiener D, Wildt L. Is human chorionic gonadotropin directly involved in the regulation of human implantation? Mol Cell Endocrinol. 2007;269:85–92. doi: 10.1016/j.mce.2006.09.016. [DOI] [PubMed] [Google Scholar]
- 17.Zygmunt M, Herr F, Keller-Schoenwetter S, Kunzi-Rapp K, Munstedt K, Rao CV, Lang U, Preissner KT. Characterization of human chorionic gonadotropin as a novel angiogenic factor. J Clinical Endocrinol Metabolism. 2002;87:5290–5296. doi: 10.1210/jc.2002-020642. [DOI] [PubMed] [Google Scholar]
- 18.Rao CV, Lei ZM. The past, present and future of nongonadal LH/hCG actions in reproductive biology and medicine. Mol Cell Endocrinol. 2007;269:2–8. doi: 10.1016/j.mce.2006.07.007. [DOI] [PubMed] [Google Scholar]
- 19.Shi QJ, Lei ZM, Rao CV, Lin J. Novel role of human chorionic gonadotropin in differentiation of human cytotrophoblasts. Endocrinology. 1993;132:1387–1395. doi: 10.1210/en.132.3.1387. [DOI] [PubMed] [Google Scholar]
- 20.Yoshida Y. Secretion of human chorionic gonadotropin in early pregnancy. Med Mol Morphol. 2005;38:104–111. doi: 10.1007/s00795-004-0258-z. [DOI] [PubMed] [Google Scholar]
- 21.Frendo JL, Olivier D, Cheynet V, Blond JL, Bouton O, Vidaud M, Rabreau M, Evain-Brion D, Mallet F. Direct involvement of HERV-W Env glycoprotein in human trophoblast cell fusion and differentiation. Mol Cell Biol. 2003;23:3566–3574. doi: 10.1128/MCB.23.10.3566-3574.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ryu KS, Gilchrist RL, Koo YB, Ji I, Ji TH. Gene, interaction, signal generation, signal divergence and signal transduction of the LH/CG receptor. Int J Gynaecology and obstetrics: the off organ of the Int Federation of Gynaecology and Obstetrics. 1998;60(Suppl 1):S9–S20. doi: 10.1016/S0020-7292(98)80001-5. [DOI] [PubMed] [Google Scholar]
- 23.Ryu KS, Ji I, Chang L, Ji TH. Molecular mechanism of LH/CG receptor activation. Mol Cell Endocrinol. 1996;125:93–100. doi: 10.1016/S0303-7207(96)03951-2. [DOI] [PubMed] [Google Scholar]
- 24.Alsat E, Cedard L. Demonstration of a specific fixation of radio-iodinated human chorionic gonadotropin (HCG I-125) in fragments of human placentas. C R Acad Sci Hebd Seances Acad Sci D. 1974;278:2665–2668. [PubMed] [Google Scholar]
- 25.Reshef E, Lei ZM, Rao CV, Pridham DD, Chegini N, Luborsky JL. The presence of gonadotropin receptors in nonpregnant human uterus, human placenta, fetal membranes, and decidua. J Clinical Endocrinol Metabolism. 1990;70:421–430. doi: 10.1210/jcem-70-2-421. [DOI] [PubMed] [Google Scholar]
- 26.Jauniaux E, Bao S, Eblen A, Li X, Lei ZM, Meuris S, Rao CV. HCG concentration and receptor gene expression in placental tissue from trisomy 18 and 21. Mol Hum Reprod. 2000;6:5–10. doi: 10.1093/molehr/6.1.5. [DOI] [PubMed] [Google Scholar]
- 27.Licht P, Cao H, Lei ZM, Rao CV, Merz WE. Novel self-regulation of human chorionic gonadotropin biosynthesis in term pregnancy human placenta. Endocrinology. 1993;133:3014–3025. doi: 10.1210/en.133.6.3014. [DOI] [PubMed] [Google Scholar]
- 28.Pidoux G, Gerbaud P, Tsatsaris V, Marpeau O, Ferreira F, Meduri G, Guibourdenche J, Badet J, Evain-Brion D, Frendo JL. Biochemical characterization and modulation of LH/CG-receptor during human trophoblast differentiation. J Cell Physiol. 2007;212:26–35. doi: 10.1002/jcp.20995. [DOI] [PubMed] [Google Scholar]
- 29.Rao CV. The beginning of a new era in reproductive biology and medicine : Expression of low functional luteinizing hormone/human chorionic gonadotropin receptors in nongonadal tissues. J Physiol Pharmacol. 1996;47:41–53. [Google Scholar]
- 30.Botelho LH, Webster LC, Rothermel JD, Baraniak J, Stec WJ. Inhibition of cAMP-dependent protein kinase by adenosine cyclic 3′-, 5′-phosphorodithioate, a second cAMP antagonist. J Biological Chem. 1988;263:5301–5305. [PubMed] [Google Scholar]
- 31.Alfaidy N, Gupta S, DeMarco C, Caniggia I, Challis JR. Oxygen regulation of placental 11 beta-hydroxysteroid dehydrogenase 2: physiological and pathological implications. J Clinical Endocrinol Metabolism. 2002;87:4797–4805. doi: 10.1210/jc.2002-020310. [DOI] [PubMed] [Google Scholar]
- 32.Bradbury FA, Menon KM. Evidence that constitutively active luteinizing hormone/human chorionic gonadotropin receptors are rapidly internalized. Biochemistry. 1999;38:8703–8712. doi: 10.1021/bi990169t. [DOI] [PubMed] [Google Scholar]
- 33.Menon KM, Menon B, Wang L, Gulappa T, Harada M. Molecular regulation of gonadotropin receptor expression: relationship to sterol metabolism. Mol Cell Endocrinol. 2010;329:26–32. doi: 10.1016/j.mce.2010.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Menon KM, Munshi UM, Clouser CL, Nair AK. Regulation of luteinizing hormone/human chorionic gonadotropin receptor expression: a perspective. Biol Reprod. 2004;70:861–866. doi: 10.1095/biolreprod.103.024471. [DOI] [PubMed] [Google Scholar]
- 35.Conti M, Harwood JP, Hsueh AJ, Dufau ML, Catt KJ. Gonadotropin-induced loss of hormone receptors and desensitization of adenylate cyclase in the ovary. J Biological Chem. 1976;251:7729–7731. [PubMed] [Google Scholar]
- 36.Kisliouk T, Levy N, Hurwitz A, Meidan R. Presence and regulation of endocrine gland vascular endothelial growth factor/prokineticin-1 and its receptors in ovarian cells. J Clinical Endocrinol Metabolism. 2003;88:3700–3707. doi: 10.1210/jc.2003-030492. [DOI] [PubMed] [Google Scholar]
- 37.Kisliouk T, Podlovni H, Meidan R. Unique expression and regulatory mechanisms of EG-VEGF/prokineticin-1 and its receptors in the corpus luteum. Ann Anat. 2005;187:529–537. doi: 10.1016/j.aanat.2005.07.005. [DOI] [PubMed] [Google Scholar]
- 38.Kisliouk T, Podlovni H, Spanel-Borowski K, Ovadia O, Zhou QY, Meidan R. Prokineticins (endocrine gland-derived vascular endothelial growth factor and BV8) in the bovine ovary: expression and role as mitogens and survival factors for corpus luteum-derived endothelial cells. Endocrinology. 2005;146:3950–3958. doi: 10.1210/en.2005-0297. [DOI] [PubMed] [Google Scholar]
- 39.Evans J, Catalano RD, Brown P, Sherwin R, Critchley HO, Fazleabas AT, Jabbour HN. Prokineticin 1 mediates fetal-maternal dialogue regulating endometrial leukemia inhibitory factor. FASEB J. 2009;23:2165–2175. doi: 10.1096/fj.08-124495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Evans J, Catalano RD, Morgan K, Critchley HO, Millar RP, Jabbour HN. Prokineticin 1 signaling and gene regulation in early human pregnancy. Endocrinology. 2008;149:2877–2887. doi: 10.1210/en.2007-1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Catalano RD, Lannagan TR, Gorowiec M, Denison FC, Norman JE, Jabbour HN. Prokineticins: novel mediators of inflammatory and contractile pathways at parturition? Mol Hum Reprod. 2010;16:311–319. doi: 10.1093/molehr/gaq014. [DOI] [PubMed] [Google Scholar]
- 42.Denison FC, Battersby S, King AE, Szuber M, Jabbour HN. Prokineticin-1: a novel mediator of the inflammatory response in third-trimester human placenta. Endocrinology. 2008;149:3470–3477. doi: 10.1210/en.2007-1695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Islami D, Bischof P, Chardonnens D. Modulation of placental vascular endothelial growth factor by leptin and hCG. Mol Hum Reprod. 2003;9:395–398. doi: 10.1093/molehr/gag053. [DOI] [PubMed] [Google Scholar]
- 44.Menon KM, Nair AK, Wang L. A novel post-transcriptional mechanism of regulation of luteinizing hormone receptor expression by an RNA binding protein from the ovary. Mol Cell Endocrinol. 2006;246:135–141. doi: 10.1016/j.mce.2005.11.026. [DOI] [PubMed] [Google Scholar]
- 45.Bukovsky A, Indrapichate K, Fujiwara H, Cekanova M, Ayala ME, Dominguez R, Caudle MR, Wimalsena J, Elder RF, Copas P, Foster JS, Fernando RI, Henley DC, Upadhyaya NB. Multiple luteinizing hormone receptor (LHR) protein variants, interspecies reactivity of anti-LHR mAb clone 3B5, subcellular localization of LHR in human placenta, pelvic floor and brain, and possible role for LHR in the development of abnormal pregnancy, pelvic floor disorders and Alzheimer’s disease. Reprod Biol Endocrinol. 2003;1:46. doi: 10.1186/1477-7827-1-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Misrahi M, Beau I, Ghinea N, Vannier B, Loosfelt H, Meduri G, Vu Hai MT, Milgrom E. The LH/CG and FSH receptors: different molecular forms and intracellular traffic. Mol Cell Endocrinol. 1996;125:161–167. doi: 10.1016/S0303-7207(96)03953-6. [DOI] [PubMed] [Google Scholar]
- 47.Pidoux G, Gerbaud P, Marpeau O, Guibourdenche J, Ferreira F, Badet J, Evain-Brion D, Frendo JL. Human placental development is impaired by abnormal human chorionic gonadotropin signaling in trisomy 21 pregnancies. Endocrinology. 2007;148:5403–5413. doi: 10.1210/en.2007-0589. [DOI] [PubMed] [Google Scholar]
- 48.Aplin JD. Developmental cell biology of human villous trophoblast: current research problems. Int J Developmental Biol. 2010;54:323–329. doi: 10.1387/ijdb.082759ja. [DOI] [PubMed] [Google Scholar]
- 49.Chan CC, Lao TT, Cheung AN. Apoptotic and proliferative activities in first trimester placentae. Placenta. 1999;20:223–227. doi: 10.1053/plac.1998.0375. [DOI] [PubMed] [Google Scholar]
- 50.Kar M, Ghosh D, Sengupta J. Histochemical and morphological examination of proliferation and apoptosis in human first trimester villous trophoblast. Hum Reproduction (Oxf, Engl) 2007;22:2814–2823. doi: 10.1093/humrep/dem284. [DOI] [PubMed] [Google Scholar]
- 51.Knofler M, Saleh L, Bauer S, Vasicek R, Griesinger G, Strohmer H, Helmer H, Husslein P. Promoter elements and transcription factors involved in differentiation-dependent human chorionic gonadotrophin-alpha messenger ribonucleic acid expression of term villous trophoblasts. Endocrinology. 2000;141:3737–3748. doi: 10.1210/en.141.10.3737. [DOI] [PubMed] [Google Scholar]
- 52.Gaspard U, Foidart JM, Lambotte R, Reuter AM, Franchimont P. Human chorionic gonadotropin and its subunits in normal and pathologic pregnancies. Ann Endocrinol. 1984;45:269–280. [PubMed] [Google Scholar]
- 53.Goldstein DP, Berkowitz RS. Current management of complete and partial molar pregnancy. J Reproduct Med. 1994;39:139–146. [PubMed] [Google Scholar]
- 54.Myatt L, Miodovnik M. Prediction of preeclampsia. Semin Perinatol. 1999;23:45–57. doi: 10.1016/S0146-0005(99)80059-7. [DOI] [PubMed] [Google Scholar]