Abstract
Thrombocytopenia is a frequent complication of viral infections providing evidence that interaction of platelets with viruses is an important pathophysiological phenomenon. Multiple mechanisms are involved depending on the nature of the viruses involved. These include immunological platelet destruction, inappropriate platelet activation and consumption, and impaired megakaryopoiesis. Viruses bind platelets through specific receptors and identified ligands, which lead to mutual alterations of both the platelet host and the viral aggressor. We have shown that HIV-1 viruses are internalized specifically in platelets and megakaryocytes, where they can be either sheltered, unaltered (with potential transfer of the viruses into target organs), or come in contact with platelet secretory products leading to virus destruction and facilitated platelet clearance. In this issue, we have reviewed the various pathways that platelets use in order to interact with viruses, HIV and others. This review also shows that more work is still needed to precisely identify platelet roles in viral infections, and to answer the challenge of viral safety in platelet transfusion.
Keywords: Platelet, Megakaryocyte, Virus, Thrombocytopenia, Receptors, Immunity, Transfusion
Introduction
If the critical role of blood platelets in hemostasis and thrombosis is clearly recognized, their function during host defense against infection has received much less attention.
Thrombocytopenia is common during or after viral infections. Several mechanisms have been proposed to contribute to thrombocytopenia: (1) platelet destruction mediated by platelet-associated immunoglobulin G (IgG) or platelet–leukocyte aggregation possibly leading to sequestration by macrophages, (2) sequestration of platelets in the enlarged spleen, (3) impaired production of thrombopoietin, and (4) direct effect of viruses on platelets. Some mechanisms possibly responsible for thrombocytopenia still remain unclear, in particular direct interactions between platelets, megakaryocytes (MKs), and viral particles. In 1959, the first visualization by electron microscopy of influenza viruses incorporating into platelets was published [1]. In 1961, Jerushalmy et al. [2] described the in vitro interaction of myxoviruses with human blood platelets. These studies were the first pieces of evidence for a direct link between viral infection and platelets.
Further, in vitro studies, showed that platelets interact with and internalize foreign particles, for example, inert particles such as latex beads [3]. However, this was interpreted as a passive passage of the particles through the platelet surface connected canalicular system (SCCS). Additional studies demonstrated that platelets interact directly with microorganisms by engulfing them in specific compartments, immunologically distinct from the SCCS [4]. Platelet secretory granules were able to fuse with this compartment, followed by destruction of the microorganism. This observation implies a potential role for platelets in the protection and defense of the host organism against infectious microorganisms. However, it is not yet clear whether platelets, by taking up viruses, contribute to the transport and the dissemination of infection in vivo or if, on the contrary, they help the host organism to defend against infection. Platelet–virus interactions may have an ambivalent role, depending on the virus species and on the platelet and MK environment.
Finally, effective viral inactivation methods have been developed during the last decade to ensure the safety of platelet concentrates from pathogens [5, 6]. It is thus most important to improve our understanding of platelet–virus interactions in order to optimize the safety of platelet transfusion.
Platelets and HIV
The ability of platelets to internalize HIV particles was initially described by Zucker-Franklin et al. [7]. Moreover, several studies have indicated that, during infection with HIV, there is a direct interaction between HIV, MKs, and platelets.
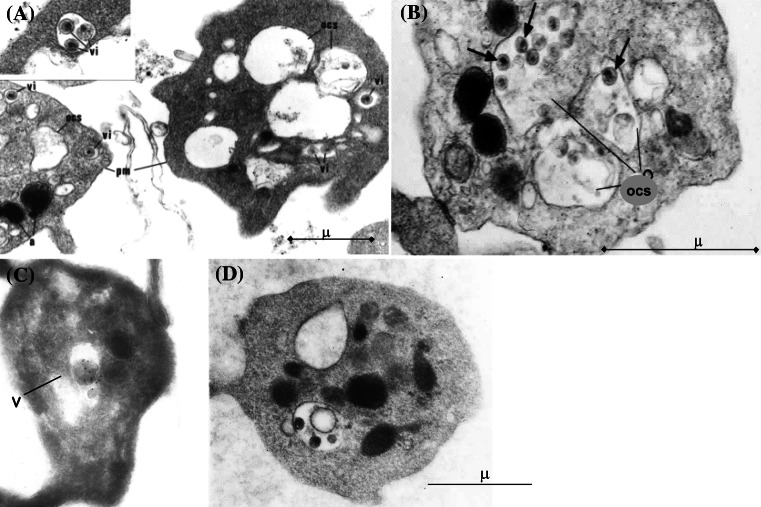
In vitro experiments were done in our laboratory in order to elucidate this potential interaction of platelets with HIV. Ultrastructural examination of platelets incubated with the supernatant of PBMC culture for 30 min demonstrated that HIV are internalized by platelets (Fig. 1). They were internalized entirely, keeping their ultrastructural integrity, including their envelope; no image showing fusion with the platelet plasma membrane could be seen during this short lapse of time. HIV particles were observed in small endocytic vesicles near the plasma membrane. These images were quite characteristic with viral particles either isolated or in groups of up to four, and were tightly enclosed in small vacuoles distinct from the SCCS, in that they were frequently located at the platelet periphery and their content appeared clear, devoid of other content except the viral particles; that is, no cellular debris as observed in the PBMC supernatants indicating a selective uptake of the virions. In addition, several viruses could also be seen in the lumen of the SCCS. At higher magnification, viruses located within endocytic vesicles appeared intact, spherical, and with well-delimited envelopes. Within the SCCS, viral particles appeared swollen and poorly delimited, with irregular shape. Immunolabeling for p24 protein confirmed that the virus-like particles were indeed HIV (Fig. 1c).
Fig. 1.
EM examination of platelets incubated with HIV viruses. Characteristic images of intact HIV viruses are observed: a in small endocytic vesicles (VI) and b,d in the open (surface connected) canalicular system (OCS). The viruses display round shape, dense core and intact envelope. c P24 immunolabeling confirm that HIV viruses are found in endocytic vesicles (V)
Double immunolabeling for p24 protein (protein of the viral core) and fibrinogen or P-selectin (markers of platelet secretion) demonstrated that the intact HIV enclosed in the endocytic vesicles were sheltered from platelet secretory products while, when located in the SCCS, they were in contact with platelet secreted proteins [8] (Fig. 2).
Fig. 2.
a Platelets incubated with VSV lentiviruses, having undergone double immunolabeling for the viral protein P24(10 nm gold, arrows) and the α-granule marker P-selectin (5 nm gold, arrowheads). b Three lentiviruses (1, 2, 3), identified by P24 immunolabeling, can be detected in small endocytic vesicles deprived of α-granule marker. These viruses display round shape and intact envelope. c Double immunolabeling for the viral protein P24 and the α-granule marker P-selectin (b). On the other hand, lentiviruses present in the surface connected canalicular system (SCCS) can only be identified by P24 immunolabeling (arrows). Indeed, they are degraded, having lost their viral envelope and their round morphology. The presence of P-selectin on the surface connected canalicular system (SCCS) membrane (arrowheads) attests for granular secretion. This indicates that contact with platelet secretory products is able to damage internalized viruses (bar 0.125 μm)
These observations indicate that:
Platelets are able to endocytose HIV virions
HIV can be enclosed within platelets, sheltered from host immune system aggression and transported by circulating platelets within the entire human body
Viruses can come in contact with platelet secreted alpha-granule contents leading to their destruction
The above findings appear to be relevant to human pathology since examination of platelets from patients with acquired immunodeficiency syndrome and high viremia showed the same endocytic vacuoles suggesting that HIV endocytosis also occurs in vivo [4] (Fig. 1d).
Moreover, platelet–virus interactions have biological effects on platelets. Indeed, platelets having endocytosed viruses are activated and express P-selectin, the receptor for macrophages, on their membrane. Therefore, they may be cleared from the circulation by macrophages with which they could interact to help HIV killing. Thrombocytopenia as a complication of HIV infection could in part be the result of this defense mechanism.
Modified lentiviruses
Since the experimental model based on the use of intact live HIV is hazardous, in order to continue this study, an experimental model was developed that is safer for the experimentor. This was based on the use of non-replicative, genetically modified lentiviruses bearing either HIV envelope (HIV-e) or vesicular stomatitis virus with protein G envelope (VSV-e) proteins. Indeed, since the objective of the study was to examine precisely the mechanisms of uptake of viral particles, the use of non-replicative lentiviruses is not a hinderance.
Vectors derived from lentiviruses by deletion of the viral coding sequences and of the transcription regulatory sequences of the 3′ LTR [9] were used. These two major modifications rendered them replication defective.
Platelets are able to internalize the recombinant lentiviruses in the same way as HIV, without fusion of their envelope with the cell plasma membrane, since many internalized virus particles displayed a well-preserved envelope.
Ultrastructural examination of platelets incubated with the lentiviral suspension demonstrated that both strains of lentiviruses, HIV and HIV-e, were internalized entirely, keeping their structural integrity, including their envelopes; no images of fusion with the platelet plasma membrane were observed during this short lapse of time and modified lentiviruses observed in the platelet endocytic compartment indeed exhibited well-preserved envelopes. Therefore, the use of lentiviruses with HIV envelopes is a good model for studying platelet interaction with HIV since the capture step is an important one.
Platelet receptors and viruses
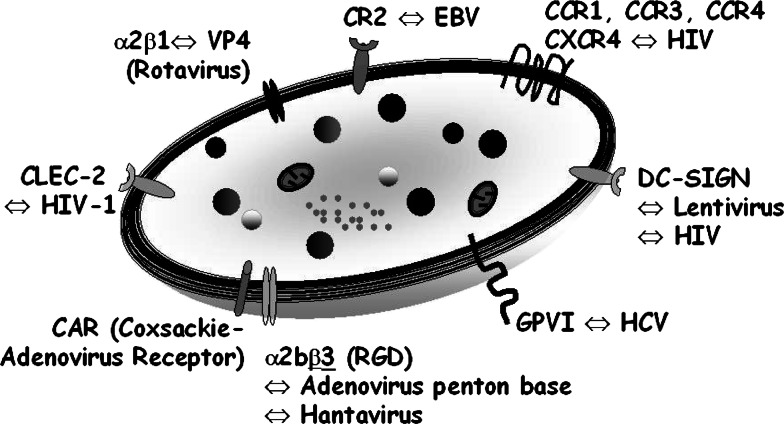
There is increasing evidence that many viruses interact directly with platelets receptors in addition to the receptor FcgR2A (CD32), which is able to bind virus–IgG complexes. Numerous platelet receptors have been implicated in viral particle binding (Fig. 3). Studies have demonstrated that cell–viral particles interactions are mediated by integrins. Rotavirus contains two outer capsid viral proteins, the spike protein VP4 and the major capsid component VP7, both of which are implicated in cell entry. Rotavirus VP4-mediated cell entry involves the alpha2beta1 integrin (α2β1), whereas VP7 appears to interact with alphaXbeta2 and alpha4beta1 integrins [10]. Since α2β1 is expressed on platelets and is a major collagen receptor, implicated in platelet adhesion to fibrillar collagen, one may speculate that rotaviruses interact with platelets. Alpha2b-beta3 (α2bβ3) recognizes the arginine-glycine-aspartic acid (RGD) sequence of ligands such as fibrinogen, laminin, vitronectin, and von Willebrand factor, and α2bβ3 mainly mediates platelet–platelet interaction during aggregation. Because the RGD sequence is found in the adenovirus penton base, adenovirus internalization requires the presence of cell surface integrins that can bind ligands with an RGD sequence [11]. Other integrins such as alpha(v)beta(3) or alpha(v)beta(5) may interact with adenoviruses. Despite this interaction, adenoviral vectors (over a wide concentration range) do not induce, inhibit, or potentiate human platelet aggregation [12]. In contrast, a recent study shows that, after intra-venous administration of recombinant adenovirus serotype 5 (usually used as vectors for gene therapy), the viruses rapidly bind to circulating platelets, which causes their activation/aggregation and subsequent entrapment in liver sinusoids [13]. Virus–platelet aggregates are taken up by Kupffer cells and degraded [13]. Hantaviruses also bind beta3 integrins. The use of these receptors by hantaviruses is likely to be fundamental to hantavirus pathogenesis since beta3 integrins are critical adhesive receptors for platelets and endothelial cells [14]. Finally, since beta1 integrins mediate internalization of mammalian reoviruses to cultured cells, one could also speculate on interactions with platelets [15].
Fig. 3.
Diagram of the various platelet receptors involved in platelet–virus interaction
Along with integrins, other receptors have been described which mediate interaction between platelets and viruses. Human platelets express the complement (C3d) receptor type II (CR2) on their membranes [16], which is also a receptor for Epstein Barr virus (EBV). Monoclonal antibody against CR2 (OKB7) blocks the binding of EBV and also inhibits EBV-mediated release of TGF-beta [17]. One of the principal collagen receptors, glycoprotein VI (GPVI), has been proposed to play a role in viral transport and/or persistence, as hepatitis C virus (HCV) can bind GPVI [18]. Platelets could be potential transporters of infectious virions to the target liver [18]. Recently, a specific virus receptor, coxsackie-adenovirus receptor (CAR), has been identified on platelets [19]. Thrombocytopenia following administration of adenoviral gene transfer vectors is partly due to platelet activation and accelerated platelet clearance by activation of CAR and/or integrin binding.
DC-SIGN
Dendritic cell-specific ICAM-grabbing non-integrin (DC-SIGN), a C-type lectin receptor present on both macrophages and dendritic cells, has been identified on platelets [8]. This molecule recognizes and binds to mannose type carbohydrates, a class of pathogen-associated molecular patterns commonly found on viruses, bacteria, and fungi. Retrovirus-like lentivirus as HIV-1 bound to platelet by DC-SIGN [8, 20]. MKs express the CD4 receptor for HIV [21] as well as the co-receptors CXCR1, 2, 4, and CCR3 [22]. Platelets do not express CD4 but only bear the co-receptors CXCR1, 2, and 4 [22]. These are devoted to virus fusion with the plasma membrane and subsequent delivery of the virus genetic material into the host cell.
An alternative receptor-mediated pathway of HIV penetration within host cells was recently described in dendritic cells. DC-SIGN is a C-type (Ca2+-dependent) lectin receptor, which selectively recognizes high-mannose oligosaccharides [23] and mediates virus capture and internalization within cells (Fig. 4). DC-SIGN is expressed on dendritic cells, including those derived from blood monocytes or found in lymphoid tissues and beneath mucosal surfaces. DC-SIGN-expressing cells internalize HIV-1 virions into a trypsin-resistant compartment, and viral infectivity can be retained for several days before transmission to T cells [24]. In downstream DC-SIGN-mediated internalization within dendritic cells, most of the virions are degraded in an acidic compartment [25]. This is a pathogen receptor, initially described in dendritic cells. Viral particles are internalized in the endocytic vesicles via DC-SIGN in dendritic cells. After internalization, most of the virions are degraded in an acidic compartment but some virions escape lysosomal degradation. They are transmitted to permissive CD4+ lymphocytes, whereas another part will gain access to the dendritic cell cytoplasm. Cytosolic viral material may be the source of low levels of productive infection of dendritic cells. The proteasome also degrades cytosolic virions, leading to the MHC-I-restricted presentation of viral epitopes [25]. DC-SIGN binding to ICAM-3 plays an important role in establishing the first contact between dendritic cells and resting T cells [26].
Fig. 4.
The two pathways of HIV internalization within host cells
The question could then be raised whether lentiviral particle internalization is a specific function of platelets and MKs. The trafficking of viruses within endocytic vesicles resembles the phenomenon of macropinocytosis described in dendritic cells and mediated by the receptor DC-SIGN. As in dendritic cells, an intracellular trafficking pathway leading either to lentivirus homing or to lentivirus inactivation was identified in platelets and MKs. The presence and the functionality of the pathogen receptor DC-SIGN on platelets and MKs was demonstrated by various technical approaches.
For this purpose, highly sensitive and specific techniques such as flow cytometry, immunoEM, western blotting and RT-PCR were used to show the presence of DC-SIGN in platelets. By flow cytometry, 15% of platelets are DC-SIGN positive, and this proportion is quite important since it is equivalent to the proportion in dendritic cells. DC-SIGN immunolabeling showed that the protein is mainly localised on the platelet surface. Western blotting revealed and confirmed the presence of DC-SIGN protein in platelets (Fig. 5).
Fig. 5.

Demonstration of DC-SIGN expression on platelets: a Detection of DC-SIGN expression by flow cytometry on platelets and dendritic cells. Platelets: DC-SIGN labeling shows that 15% of platelets are DC-SIGN positive. Dendritic cells: as a positive control, an equivalent proportion of dendritic cells express DC-SIGN by flow cytometry. b Immunogold labeling for DC-SIGN: DC-SIGN antigen (arrows) is detected on platelet membrane, on SCCS membrane, and on some limiting membrane α-granules (bar 0.3 lm). c DC-SIGN detection in platelets by western blot. DC-SIGN, a 44-kDa protein, is detected in platelets, and in Hela-DC-SIGN and THP1-DC-SIGN cells (positive control). Hela and THP-1 cells are negative controls
Platelets can endocytose and store plasma proteins such as fibrinogen [27]. Thus, to rule out the hypothesis that DC-SIGN might be adsorbed on platelet surfaces, we studied their MK precursors. MKs were obtained from cultured cord blood precursors and cultured without plasma or serum. Under these technical conditions, cultured MKs DC-SIGN expression was detected by western blotting. In addition, MKs were shown to have the capacity to synthesize DC-SIGN since its RNA was amplified by RT-PCR. This finding confirmed that DC-SIGN expression observed on platelets and MKs was specific, since it is the consequence of endogenous synthesis.
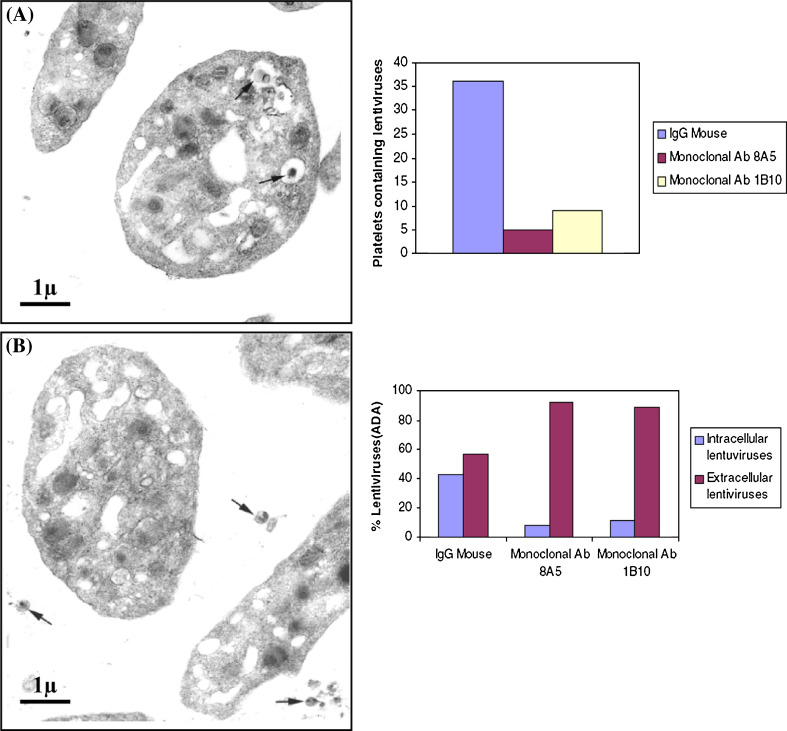
To show whether the receptor DC-SGN is functional in platelets, blocking experiments were performed with specific antibodies. Monoclonal anti-DC-SIGN antibody 1B10 was added to platelets, then they were incubated with a lentivirus suspension. The effect of anti-DC-SIGN antibody on virus internalization was assessed semi-quantitatively. EM examination showed that control platelets consistently internalized HIV-e virions, whereas in the presence of anti-DC SIGN antibody virtually no viral internalization was detected but numerous viral particles were found in the extracellular medium (Fig. 6).
Fig. 6.
DC-SIGN in platelets is functional. a Platelet samples incubated with HIV-e lentiviruses in the absence of anti-DC-SIGN antibody: virtually no extra-cellular viruses are found, whereas numerous images of intracellular viruses can be seen (arrows) (A = α-granules). b Platelet sample incubated with HIV-e lentiviruses in the presence of anti-DC-SIGN antibody: most viruses were observed outside platelets (arrows), and virtually none inside them. Graph Comparison of the respective proportions of extracellular and intracellular lentiviruses as observed on the control samples and in the presence of anti-DC SIGN antibody
These findings support the functionality of the DC-SIGN receptor in platelets where it modulates HIV endocytosis.
Finally, platelets also express the C-type lectin-like receptor 2 (CLEC-2) and a combination of DC-SIGN and CLEC-2 inhibitors strongly reduced the association of HIV-1 with platelets, indicating that these lectins are required for efficient HIV-1 binding to platelets [20].
Megakaryocytes and viruses
Platelets are derived from progenitors, the MKs, located in the bone marrow. Virus interactions with platelets and MKs have been described for a long time and various viruses have been implicated such as paramyxovirus (Newcastle disease virus, etc.) [28], retroviruses including HIV-1 [7, 8], and Herpes viruses (CMV, HHV-6, HHV-7) [29–31].
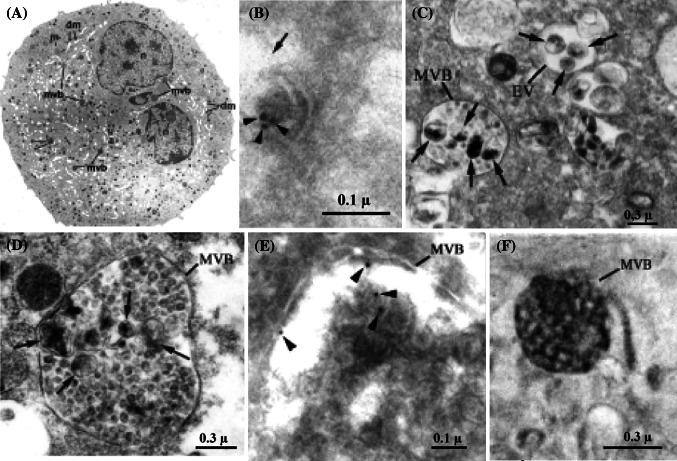
The receptors found to mediate mature platelet–virus interactions may also be involved in virus binding to platelet progenitors. There are differences between MKs and mature platelet receptors and antigens. Immature human MKs express the CD4 antigen on their surface, capable of binding HIV-1, but this antigen is not expressed on fully mature MKs and platelets. CD4 expression occurs during hemopoietic differentiation and is an early step in MK maturation [21]. Using this receptor, HIV-1 can infect cells in the MKs lineage [32]. In addition, we could demonstrate the presence of DC-SIGN in human MKs. DC-SIGN could be detected in cultured MKs by western blotting. In addition, MKs were shown to be able to synthesize DC-SIGN, since its RNA was revealed by RT-PCR. When exposed, in vitro, to a suspension of HIV-e, MKs captured and endocytosed the entire viral particles, including envelopes. Virions were retrieved both in the lumen of the demarcation membrane system were they kept their integrity and within multivesicular bodies whose acidic content led to their destruction (Fig. 7). It seems that the endocytic pathway is relevant in human pathology since ultrastructural images of intact viruses were found in bone marrow MKs from HIV infected patients [7].
Fig. 7.
a EM view of a characteristic mature cultured MK showing demarcation membranes (dm), alpha granules (a), multivesicular bodies (mvb), and incubated with VSV-e lentivirus suspension: b Lentiviruses located in endocytic vacuoles are identified thanks to their size and specific P24 immunolabeling (arrowheads). c They are also found (arrows) in small endocytic vesicles and multivesicular bodies (MVB). In the EV (arrow), viruses seem intact, displaying spherical shape and well-limited envelope. d When located in the MVB, lentiviruses (arrows) appear swollen and poorly limited, with irregular shape. e P24 immunolabeling is detected in MVB (arrowheads), confirming the presence of lentiviruses in these structures. fMVBs are positive for acid phosphatase (revealed by the cerium cytochemical technique) which contributes to viral destruction
Viral replication in MKs has been demonstrated for several viruses like HIV-1 [33], HCV [34], and human CMV (HCMV) [29]. For example, in purified marrow MKs from HIV-seropositive patients with immune thrombocytopenic purpura, viral transcripts, and small amounts of HIV glycoproteins were detected although heterogeneity among MK was observed [35]. These results demonstrate that MKs are susceptible to viral infection and that direct infection of these cells in vivo may contribute to the thrombocytopenia observed in infected patients.
Moreover, in allogeneic bone marrow transplantation, HCMV is frequently associated with graft failure. This could be due to a direct suppressive effect on thrombopoiesis [29]. Down-regulation of haematopoiesis is probably a protective mechanism of the microenvironment that limits injury to the marrow stem/progenitor cell compartment during the subsequent process of elimination of infected cells as has been suggested in dengue virus infection [36]. A study found that HHV-6 viral load was significantly correlated with delayed platelet engraftment and to the number of platelet transfusions required in allogeneic stem cell transplantion [37]. The replication of numerous viruses has been proposed in delayed engraftment after stem cell transplantation. Some in vitro studies have shown that HHV-6 but not HHV-7 is one of the major causes of thrombopoiesis suppression by a direct effect on hematopoietic progenitors [30], whereas another in vitro study suggested that HHV-7 impairs the survival/differentiation of megakaryocytic cells [31].
Thrombocytopenia in HCV infection is complex and multiple mechanisms are involved [38]. Indirect evidence supports the hypothesis of bone marrow suppression by HCV, partly mediated by a reduction of thrombopoetin (TPO) production due to liver cirrhosis and/or fibrosis. The ability of eltrombopag (a selective TPO-receptor agonist) to facilitate initiation and maintenance of interferon-based anti-HCV therapy in patients with thrombocytopenia associated with HCV-related cirrhosis has been evaluated. Promising results were recently published and require confirmation in phase 3 trials [39].
Platelet–virus interactions trigger inflammation
Inflammation and thrombosis are intimately connected. Inflammation-induced changes of the vessel wall may promote platelet adhesion, activation, coagulation and leukocyte recruitment enhancing various pathological states. Activated platelets are the procoagulant surface on which thrombin, the central coagulation protease, is generated. However, thrombin is also a potent proinflammatory mediator, mainly by binding to proteinase-activated receptors (PAR 1–4) which are widely expressed as well as on platelets.
In vitro infection of human umbilical vein endothelial cells (HUVEC) with dengue-2 viruses, stimulated the cells as monitored by expression of E-selectin. Platelets that adhered to dengue-2-stimulated HUVEC were activated as shown by P-selectin (CD62) expression on their membranes. CD62 is not expressed on resting platelets, thus it is a marker of platelet activation. Activated platelet interactions with endothelium could contribute to the thrombocytopenia observed during dengue infection [40] and promote the formation of platelet–leukocyte association, presumably by binding to the leukocyte P-selectin glycoprotein ligand-1 (PSGL1). This result was confirmed by studies with other viruses. Adenovirus-induced endothelial cell activation was shown by VCAM-1 expression and by the release of ultra-large multimers of von Willebrand factor within 1–2 h of virus administration to cultured endothelial cells and in an animal model (mouse) [19]. In this study, the viruses activate platelets and induce microparticle release and platelet–leukocyte complex formation.
These complexes probably participate in the accelerated clearance of platelets by the mononuclear macrophage system. Various microorganisms have been suspected to play a role in pathogenesis and progression of atherosclerosis. Among infectious agents, Chlamydiae pneumoniae are the most plausible pathogens with a causative role [41]. HCMV has also been proposed to be implicated in the pathogenesis of vascular diseases. In vitro studies show that HCMV infection of endothelial cells triggers platelet adhesion and aggregation [42]. Viral infection of endothelial cells activates coagulation cascades and contributes to thrombus formation, which can precipitate atherothrombosis in patients with atherosclerotic disease.
Platelets can also facilitate the accumulation of cytotoxic T lymphocytes (CTL) at sites of infection as was demonstrated for hepatitis B [43]. Changes in the vessel wall due to inflammation favor the exit of virus-specific CTLs from the bloodstream and their accumulation within the liver parenchyma where HBV is replicating.
The role of platelets in antimicrobial host defense
Platelets share structural and functional similarities with granulocytes known to participate in antimicrobial host defense. Tang et al. [44] suggest a direct antimicrobial role for platelets as they are activated and release peptides in response to trauma or mediators of inflammation. Platelet alpha-granules contain a number of antimicrobial peptides with a broad spectrum against Gram-negative and Gram-positive bacteria and fungi [44]. There is little information available about the potential virucidal activity of platelet proteins. Some authors evoke a possible virucidal effect of platelets concentrates, which has not yet been confirmed [45]. Nevertheless, Boukour et al. [8] indicate that platelets and MKs can internalize lentiviruses, which either provide a shelter to lentiviral particles (easily identifiable in the SCCS) or, alternatively, disrupts viral integrity (rupture of the envelope, loss of round shape, and degradation of the viral particle) (Fig. 2). This could be due to direct action of α-granule microbicidal peptides.
Adenovirus injected into mice can induce rapid thrombocytopenia. Soon after virus injection, platelets were cleared from the circulation, sticking around spleen macrophages, ready to be phagocytosed. Some platelets showing signs of activation contained adenovirus-like particles (Fig. 8). This observation confirms that, during adenovirus infection, platelets with a viral load are cleared by macrophages. The fate of intra-platelet viruses after internalization by phagocytes remains to be determined.
Fig. 8.

EM view of a macrophage from a mouse infected by adenoviruses: numerous platelets (P) adhere to the macrophage surface, one of them being partly degranulated displays an intracellular virus-like particle (red arrow). Inset EM aspect of the adenovirus preparation
Moreover, platelets are a significant source of RANTES (regulated upon activation normal T cell expressed and secreted or CCL5) [44, 46, 47] and MIP-1 [47]. Cocchi et al. [48] demonstrated that the chemokines RANTES and MIP-1 are the major HIV-suppressive factors produced by CD8-positive T cells and speculated that chemokine-mediated control of HIV may occur either directly, through their inherent anti-retroviral activity, or indirectly, through their ability to chemoattract T cells and monocytes to the proximity of the infection foci. However, RANTES released by platelets acting as a chemoattractant for different cells could also have the opposite effect of providing new uninfected targets for viral infection.
In addition, the recent study of Iannacone et al. [49] in a mouse model indicate that platelets glycoproteins such as integrin 3 and CD40L are required for protecting the host by clearing lymphocytic choriomeningitis virus infection through cytotoxic T lymphocytes.
Viruses and platelet transfusion
Transfusion of platelets has been implicated in the transmission of viruses, bacteria, protozoa, and potentially prions. The safety of platelet transfusion has been substantially improved over the last few years [5]. A combination of serologic determination and nucleic acid testing (NAT) in small pools of samples has decreased the residual risk of contamination for selected viruses to less than 1:2,000,000 for HIV-1 and HIV-2 and human T-lymphotropic virus (HTLV), less than 1:1,000,000 for HCV, and less than 1:200,000 for HBV [50].
Next to careful selection of donors and leukoreduction, considerable effort has been devoted to develop methods for pathogen inactivation in platelet concentrates [5, 6]. The methods are based on photochemical treatment (PCT) and can be divided into two, psoralens and riboflavin (B2 vitamin).
Psoralen and its derivatives are heterocyclic planar compounds found in many plants. Psoralens intercalate between the bases of nucleic acids and upon exposure to ultraviolet light (UV) can form mono-adducts and cross-links with pyrimidines. The ability of psoralens to target nucleic acids is an obvious advantage for decontamination of platelets, which lack a nucleus. A PCT process with amino-psoralen (Helinx technology) and long-wavelength UV-A light has been shown to inactivate a wide variety of blood-born pathogens in platelets including enveloped and non-enveloped viruses, and Gram-positive and negative bacteria [6]. An integrated system, the INTERCEPT® Platelet System has been developed and uses amotosalen chlorhydrate, S-59 (Helinx technology) with UV-A light. A European trial (EUROSPRITE) and a US study (SPRINT) demonstrated that platelets treated with S-59 and stored for up to 5 days were comparable to untreated platelets [51, 52]. The PCT would reduce the residual risk of pathogens contamination of platelets concentrates because it would also work with virus internalized in platelets.
Riboflavin binds nucleic acids and intercalates between DNA and RNA bases. On activation of cross-linked riboflavin with UV or visible light, guanosine bases are oxidized resulting in single-strand breaks in the nucleic acids. The mechanisms of damage to the nucleic acids is due to direct electron transfer reaction by the oxidation of guanosine, then to generation of oxygen radical, and finally production of peroxide and hydroxyl radicals. The damaged and disrupted nucleic acids are incapable of repair and replication. This process has been commercialized with the MIRASOL Pathogen Reduction Technology (PRT) System. Data of an in vitro study indicated that the device successfully reduced the number of selected pathogens in platelet concentrates [53] whereas it seems less effective on non-enveloped viruses. Platelet functions are slightly affected by MIRASOL™ PRT system [54]. Clinical trials are underway to validate this process in patients who require platelet transfusion.
Finally, another PCT is under development, the association of thionine with yellow light followed by UV-B irradiation [55].
Pathogen inactivation in platelet concentrates as in other blood products is an innovative approach to blood safety. This approach is clearly important since understanding the mechanisms underlying platelet–virus interaction, although improving, is still incomplete (surface binding, internalization, etc.). Pathogen inactivation may also eliminate viruses that are not detected by serologic methods or NAT and emerging viruses where a link with platelets is obviously unknown.
Perspectives
Platelets circulate within the entire human body in their main role of protection against bleeding. In addition, they have many receptors and intracellular glycoproteins, which can interfere with inflammatory responses. This gives platelets a major role in unspecific immunity. In this review, we have surveyed the various pathways that platelets use to interact with viruses. Interactions are specific, mediated by well-defined functional receptors, and lead to mutual alterations of the platelet host and the viral aggressor. However, much work still needs to be performed to identify precisely the platelet role in viral infections, either beneficial and protective or malevolent and pro-infectious. The response might be complex and dependent on the type of virus involved.
Acknowledgments
The authors acknowledge Dr. Karim Benhioud (Institut Gustave Roussy, Villejuif) for providing the mouse model of adenovirus infection and Miss Laurence Momeux for photographic help.
References
- 1.Danon D, Jerushalmy Z, De Vries A. Incorporation of influenza virus in human blood platelets in vitro electron microscopical observation. Virology. 1959;9:719–722. doi: 10.1016/0042-6822(59)90168-0. [DOI] [PubMed] [Google Scholar]
- 2.Jerushalmy Z, Kohn A, De Vries A. Interaction of myxoviruses with human blood platelets in vitro. Proc Soc Exp Biol Med. 1961;106:462–466. doi: 10.3181/00379727-106-26370. [DOI] [PubMed] [Google Scholar]
- 3.White JG, Clawson CC. Effects of small latex particle uptake on the surface connected canalicular system of blood platelets: a freeze-fracture and cytochemical study. Diagn Histopathol. 1982;5:3–10. [PubMed] [Google Scholar]
- 4.Youssefian T, Drouin A, Masse JM, Guichard J, Cramer EM. Host defense role of platelets: engulfment of HIV and Staphylococcus aureus occurs in a specific subcellular compartment and is enhanced by platelet activation. Blood. 2002;99:4021–4029. doi: 10.1182/blood-2001-12-0191. [DOI] [PubMed] [Google Scholar]
- 5.Pelletier JP, Transue S, Snyder EL. Pathogen inactivation techniques. Best Pract Res Clin Haematol. 2006;19:205–242. doi: 10.1016/j.beha.2005.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lin L, Hanson CV, Alter HJ, Jauvin V, Bernard KA, Murthy KK, Metzel P, Corash L. Inactivation of viruses in platelet concentrates by photochemical treatment with amotosalen and long-wavelength ultraviolet light. Transfusion. 2005;45:580–590. doi: 10.1111/j.0041-1132.2005.04316.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zucker-Franklin D, Seremetis S, Zheng ZY. Internalization of human immunodeficiency virus type I and other retroviruses by megakaryocytes and platelets. Blood. 1990;75:1920–1923. [PubMed] [Google Scholar]
- 8.Boukour S, Masse JM, Benit L, Dubart-Kupperschmitt A, Cramer EM. Lentivirus degradation and DC-SIGN expression by human platelets and megakaryocytes. J Thromb Haemost. 2006;4:426–435. doi: 10.1111/j.1538-7836.2006.01749.x. [DOI] [PubMed] [Google Scholar]
- 9.Kung SK, An DS, Chen IS. A murine leukemia virus (MuLV) long terminal repeat derived from rhesus macaques in the context of a lentivirus vector and MuLV gag sequence results in high-level gene expression in human T lymphocytes. J Virol. 2000;74:3668–3681. doi: 10.1128/JVI.74.8.3668-3681.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Coulson BS, Londrigan SL, Lee DJ. Rotavirus contains integrin ligand sequences and a disintegrin-like domain that are implicated in virus entry into cells. Proc Natl Acad Sci USA. 1997;94:5389–5394. doi: 10.1073/pnas.94.10.5389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang Y, Bergelson JM. Adenovirus receptors. J Virol. 2005;79:12125–12131. doi: 10.1128/JVI.79.19.12125-12131.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eggerman TL, Mondoro TH, Lozier JN, Vostal JG. Adenoviral vectors do not induce, inhibit, or potentiate human platelet aggregation. Hum Gene Ther. 2002;13:125–128. doi: 10.1089/10430340152712674. [DOI] [PubMed] [Google Scholar]
- 13.Stone D, Liu Y, Shayakhmetov D, Li ZY, Ni S, Lieber A. Adenovirus-platelet interaction in blood causes virus sequestration to the reticuloendothelial system of the liver. J Virol. 2007;81:4866–4871. doi: 10.1128/JVI.02819-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mackow ER, Gavrilovskaya IN. Cellular receptors and hantavirus pathogenesis. Curr Top Microbiol Immunol. 2001;256:91–115. doi: 10.1007/978-3-642-56753-7_6. [DOI] [PubMed] [Google Scholar]
- 15.Maginnis MS, Forrest JC, Kopecky-Bromberg SA, Dickeson SK, Santoro SA, Zutter MM, Nemerow GR, Bergelson JM, Dermody TS. Beta1 integrin mediates internalization of mammalian reovirus. J Virol. 2006;80:2760–2770. doi: 10.1128/JVI.80.6.2760-2770.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nunez D, Charriaut-Marlangue C, Barel M, Benveniste J, Frade R. Activation of human platelets through gp140, the C3d/EBV receptor (CR2) Eur J Immunol. 1987;17:515–520. doi: 10.1002/eji.1830170413. [DOI] [PubMed] [Google Scholar]
- 17.Ahmad A, Menezes J. Binding of the Epstein-Barr virus to human platelets causes the release of transforming growth factor-beta. J Immunol. 1997;159:3984–3988. [PubMed] [Google Scholar]
- 18.Zahn A, Jennings N, Ouwehand WH, Allain JP. Hepatitis C virus interacts with human platelet glycoprotein VI. J Gen Virol. 2006;87:2243–2251. doi: 10.1099/vir.0.81826-0. [DOI] [PubMed] [Google Scholar]
- 19.Othman M, Labelle A, Mazzetti I, Elbatarny HS, Lillicrap D. Adenovirus-induced thrombocytopenia: the role of von Willebrand factor and P-selectin in mediating accelerated platelet clearance. Blood. 2007;109:2832–2839. doi: 10.1182/blood-2006-06-032524. [DOI] [PubMed] [Google Scholar]
- 20.Chaipan C, Soilleux EJ, Simpson P, Hofmann H, Gramberg T, Marzi A, Geier M, Stewart EA, Eisemann J, Steinkasserer A, et al. DC-SIGN and CLEC-2 mediate human immunodeficiency virus type 1 capture by platelets. J Virol. 2006;80:8951–8960. doi: 10.1128/JVI.00136-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Basch RS, Kouri YH, Karpatkin S. Expression of CD4 by human megakaryocytes. Proc Natl Acad Sci USA. 1990;87:8085–8089. doi: 10.1073/pnas.87.20.8085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Riviere C, Subra F, Cohen-Solal K, Cordette-Lagarde V, Letestu R, Auclair C, Vainchenker W, Louache F. Phenotypic and functional evidence for the expression of CXCR4 receptor during megakaryocytopoiesis. Blood. 1999;93:1511–1523. [PubMed] [Google Scholar]
- 23.Feinberg H, Mitchell DA, Drickamer K, Weis WI. Structural basis for selective recognition of oligosaccharides by DC-SIGN and DC-SIGNR. Science. 2001;294:2163–2166. doi: 10.1126/science.1066371. [DOI] [PubMed] [Google Scholar]
- 24.Geijtenbeek TB, Kwon DS, Torensma R, van Vliet SJ, van Duijnhoven GC, Middel J, Cornelissen IL, Nottet HS, KewalRamani VN, Littman DR, et al. DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell. 2000;100:587–597. doi: 10.1016/S0092-8674(00)80694-7. [DOI] [PubMed] [Google Scholar]
- 25.Moris A, Nobile C, Buseyne F, Porrot F, Abastado JP, Schwartz O. DC-SIGN promotes exogenous MHC-I-restricted HIV-1 antigen presentation. Blood. 2004;103:2648–2654. doi: 10.1182/blood-2003-07-2532. [DOI] [PubMed] [Google Scholar]
- 26.Geijtenbeek TB, Torensma R, van Vliet SJ, van Duijnhoven GC, Adema GJ, van Kooyk Y, Figdor CG. Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell. 2000;100:575–585. doi: 10.1016/S0092-8674(00)80693-5. [DOI] [PubMed] [Google Scholar]
- 27.Cramer EM, Vainchenker W, Vinci G, Guichard J, Breton-Gorius J. Gray platelet syndrome: immunoelectron microscopic localization of fibrinogen and von Willebrand factor in platelets and megakaryocytes. Blood. 1985;66:1309–1316. [PubMed] [Google Scholar]
- 28.Jerushalmy Z, Kaminski E, Kohn A, Devries A. Interaction of Newcastle disease virus with megakaryocytes in cell cultures of Guinea pig bone marrow. Proc Soc Exp Biol Med. 1963;114:687–690. doi: 10.3181/00379727-114-28771. [DOI] [PubMed] [Google Scholar]
- 29.Crapnell K, Zanjani ED, Chaudhuri A, Ascensao JL, St Jeor S, Maciejewski JP. In vitro infection of megakaryocytes and their precursors by human cytomegalovirus. Blood. 2000;95:487–493. [PubMed] [Google Scholar]
- 30.Isomura H, Yoshida M, Namba H, Fujiwara N, Ohuchi R, Uno F, Oda M, Seino Y, Yamada M. Suppressive effects of human herpesvirus-6 on thrombopoietin-inducible megakaryocytic colony formation in vitro. J Gen Virol. 2000;81:663–673. doi: 10.1099/0022-1317-81-3-663. [DOI] [PubMed] [Google Scholar]
- 31.Gonelli A, Mirandola P, Grill V, Secchiero P, Zauli G. Human herpesvirus 7 infection impairs the survival/differentiation of megakaryocytic cells. Haematologica. 2002;87:1223–1225. [PubMed] [Google Scholar]
- 32.Kouri YH, Borkowsky W, Nardi M, Karpatkin S, Basch RS. Human megakaryocytes have a CD4 molecule capable of binding human immunodeficiency virus-1. Blood. 1993;81:2664–2670. [PubMed] [Google Scholar]
- 33.Chelucci C, Federico M, Guerriero R, Mattia G, Casella I, Pelosi E, Testa U, Mariani G, Hassan HJ, Peschle C. Productive human immunodeficiency virus-1 infection of purified megakaryocytic progenitors/precursors and maturing megakaryocytes. Blood. 1998;91:1225–1234. [PubMed] [Google Scholar]
- 34.Li X, Jeffers LJ, Garon C, Fischer ER, Scheffel J, Moore B, Reddy KR, Demedina M, Schiff ER. Persistence of hepatitis C virus in a human megakaryoblastic leukaemia cell line. J Viral Hepat. 1999;6:107–114. doi: 10.1046/j.1365-2893.1999.00140.x. [DOI] [PubMed] [Google Scholar]
- 35.Louache F, Bettaieb A, Henri A, Oksenhendler E, Farcet JP, Bierling P, Seligmann M, Vainchenker W. Infection of megakaryocytes by human immunodeficiency virus in seropositive patients with immune thrombocytopenic purpura. Blood. 1991;78:1697–1705. [PubMed] [Google Scholar]
- 36.La Russa VF, Innis BL. Mechanisms of dengue virus-induced bone marrow suppression. Baillieres Clin Haematol. 1995;8:249–270. doi: 10.1016/S0950-3536(05)80240-9. [DOI] [PubMed] [Google Scholar]
- 37.Ljungman P, Wang FZ, Clark DA, Emery VC, Remberger M, Ringden O, Linde A. High levels of human herpesvirus 6 DNA in peripheral blood leucocytes are correlated to platelet engraftment and disease in allogeneic stem cell transplant patients. Br J Haematol. 2000;111:774–781. doi: 10.1046/j.1365-2141.2000.02422.x. [DOI] [PubMed] [Google Scholar]
- 38.Weksler BB. Review article: the pathophysiology of thrombocytopenia in hepatitis C virus infection and chronic liver disease. Aliment Pharmacol Ther. 2007;26(Suppl 1):13–19. doi: 10.1111/j.1365-2036.2007.03512.x. [DOI] [PubMed] [Google Scholar]
- 39.McHutchison JG, Dusheiko G, Shiffman ML, Rodriguez-Torres M, Sigal S, Bourliere M, Berg T, Gordon SC, Campbell FM, Theodore D, et al. Eltrombopag for thrombocytopenia in patients with cirrhosis associated with hepatitis C. N Engl J Med. 2007;357:2227–2236. doi: 10.1056/NEJMoa073255. [DOI] [PubMed] [Google Scholar]
- 40.Krishnamurti C, Peat RA, Cutting MA, Rothwell SW. Platelet adhesion to dengue-2 virus-infected endothelial cells. Am J Trop Med Hyg. 2002;66:435–441. doi: 10.4269/ajtmh.2002.66.435. [DOI] [PubMed] [Google Scholar]
- 41.Kalvegren H, Majeed M, Bengtsson T. Chlamydia pneumoniae binds to platelets and triggers P-selectin expression and aggregation: a causal role in cardiovascular disease? Arterioscler Thromb Vasc Biol. 2003;23:1677–1683. doi: 10.1161/01.ATV.0000084810.52464.D5. [DOI] [PubMed] [Google Scholar]
- 42.Rahbar A, Soderberg-Naucler C. Human cytomegalovirus infection of endothelial cells triggers platelet adhesion and aggregation. J Virol. 2005;79:2211–2220. doi: 10.1128/JVI.79.4.2211-2220.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Iannacone M, Sitia G, Isogawa M, Marchese P, Castro MG, Lowenstein PR, Chisari FV, Ruggeri ZM, Guidotti LG. Platelets mediate cytotoxic T lymphocyte-induced liver damage. Nat Med. 2005;11:1167–1169. doi: 10.1038/nm1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tang YQ, Yeaman MR, Selsted ME. Antimicrobial peptides from human platelets. Infect Immun. 2002;70:6524–6533. doi: 10.1128/IAI.70.12.6524-6533.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Maurice A, Marchand-Arvier M, Edert D, Le Faou A, Gondrexon G, Vigneron C. The virucidal effect of platelet concentrates: preliminary study and first conclusions. Platelets. 2002;13:219–222. doi: 10.1080/0953371027000. [DOI] [PubMed] [Google Scholar]
- 46.Kameyoshi Y, Dorschner A, Mallet AI, Christophers E, Schroder JM. Cytokine RANTES released by thrombin-stimulated platelets is a potent attractant for human eosinophils. J Exp Med. 1992;176:587–592. doi: 10.1084/jem.176.2.587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Klinger MH, Wilhelm D, Bubel S, Sticherling M, Schroder JM, Kuhnel W. Immunocytochemical localization of the chemokines RANTES and MIP-1 alpha within human platelets and their release during storage. Int Arch Allergy Immunol. 1995;107:541–546. doi: 10.1159/000237097. [DOI] [PubMed] [Google Scholar]
- 48.Cocchi F, De Vico AL, Garzino-Demo A, Arya SK, Gallo RC, Lusso P. Identification of RANTES, MIP-1 alpha, and MIP-1 beta as the major HIV-suppressive factors produced by CD8+ T cells. Science. 1995;270:1811–1815. doi: 10.1126/science.270.5243.1811. [DOI] [PubMed] [Google Scholar]
- 49.Iannacone M, Sitia G, Isogawa M, Whitmire JK, Marchese P, Chisari FV, Ruggeri ZM, Guidotti LG. Platelets prevent IFN-alpha/beta-induced lethal hemorrhage promoting CTL-dependent clearance of lymphocytic choriomeningitis virus. Proc Natl Acad Sci USA. 2008;105:629–634. doi: 10.1073/pnas.0711200105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dodd RY, Notari EPt, Stramer SL. Current prevalence and incidence of infectious disease markers and estimated window-period risk in the American Red Cross blood donor population. Transfusion. 2002;42:975–979. doi: 10.1046/j.1537-2995.2002.00174.x. [DOI] [PubMed] [Google Scholar]
- 51.van Rhenen D, Gulliksson H, Cazenave JP, Pamphilon D, Ljungman P, Kluter H, Vermeij H, Kappers-Klunne M, de Greef G, Laforet M, et al. Transfusion of pooled buffy coat platelet components prepared with photochemical pathogen inactivation treatment: the euroSPRITE trial. Blood. 2003;101:2426–2433. doi: 10.1182/blood-2002-03-0932. [DOI] [PubMed] [Google Scholar]
- 52.McCullough J, Vesole DH, Benjamin RJ, Slichter SJ, Pineda A, Snyder E, Stadtmauer EA, Lopez-Plaza I, Coutre S, Strauss RG, et al. Therapeutic efficacy and safety of platelets treated with a photochemical process for pathogen inactivation: the SPRINT trial. Blood. 2004;104:1534–1541. doi: 10.1182/blood-2003-12-4443. [DOI] [PubMed] [Google Scholar]
- 53.Ruane PH, Edrich R, Gampp D, Keil SD, Leonard RL, Goodrich RP. Photochemical inactivation of selected viruses and bacteria in platelet concentrates using riboflavin and light. Transfusion. 2004;44:877–885. doi: 10.1111/j.1537-2995.2004.03355.x. [DOI] [PubMed] [Google Scholar]
- 54.AuBuchon JP, Herschel L, Roger J, Taylor H, Whitley P, Li J, Edrich R, Goodrich RP. Efficacy of apheresis platelets treated with riboflavin and ultraviolet light for pathogen reduction. Transfusion. 2005;45:1335–1341. doi: 10.1111/j.1537-2995.2005.00202.x. [DOI] [PubMed] [Google Scholar]
- 55.Mohr H. Methylene blue and thionine in pathogen inactivation of plasma and platelet concentrates. Transfus Apher Sci. 2001;25:183–184. doi: 10.1016/S1473-0502(01)00117-3. [DOI] [PubMed] [Google Scholar]