Abstract
CD8+ T cells are likely to play an important role in host defense against Salmonella enterica serovar Typhi by several effector mechanisms, including lysis of infected cells (cytotoxicity) and gamma interferon (IFN-γ) secretion. In an effort to better understand these responses, we studied the T-cell receptor (TCR) repertoire of serovar Typhi-specific CD8+ T cells in humans. To this end, we determined the TCR beta chain (Vβ) usage of CD8+ T cells from three volunteers orally immunized with Ty21a typhoid vaccine by flow cytometry using a panel of monoclonal antibodies. Although TCR Vβ usage varied among volunteers, we identified oligoclonal Vβ subset expansions in individual volunteers (Vβ 2, 5.1, 8, 17, and 22 in volunteer 1; Vβ 1, 2, 5.1, 14, 17, and 22 in volunteer 2; and Vβ 3, 8, 14, and 16 in volunteer 3). These subsets were antigen specific, as shown by cytotoxicity and IFN-γ secretion assays on Vβ sorted cells and on T-cell clones derived from these volunteers. Moreover, eight-color flow cytometric analysis showed that these clones exhibited a T effector memory phenotype (i.e., CCR7− CD27− CD45RO+ CD62L−) and coexpressed gut homing molecules (e.g., high levels of integrin α4β7, intermediate levels of CCR9, and low levels of CD103). In conclusion, our results show that long-term T-cell responses to serovar Typhi in Ty21a vaccinees are oligoclonal, involving multiple TCR Vβ families. Moreover, these serovar Typhi-specific CD8+ T cells bearing defined Vβ specificities are phenotypically and functionally consistent with T effector memory cells with preferential gut homing potential.
Typhoid fever remains an important public health priority, particularly in developing countries, with an estimated 16 million new cases annually and 600,000 deaths (17, 21). The appearance of antibiotic-resistant strains of Salmonella enterica serovar Typhi, the causative agent of typhoid fever, has added new urgency for the development of improved typhoid vaccines. New-generation attenuated serovar Typhi vaccine strains have the potential to become the preferred public health tool to immunize against typhoid fever because of their ability to elicit long-lasting protective systemic and mucosal immune responses. In addition, significant efforts have been focused in recent years on the use of attenuated serovar Typhi vaccines as carriers of foreign antigens to deliver foreign genes because of their ability to gain access to dendritic cells (51, 52) and deliver antigens or DNA coding for the antigens (12, 31, 32). These characteristics make the use of Salmonella live vectors particularly attractive for mass immunization programs. Moreover, live attenuated serovar Typhi vaccines are expected to have relatively low manufacturing costs compared to many other types of vaccines.
CD8+ T cells might play an important role in host defense against serovar Typhi by several effector mechanisms, including lysis of infected cells and gamma interferon (IFN-γ) secretion (35, 36, 44, 45). These CD8+ T cells recognize antigenic peptides bound to self-major histocompatibility complex via T-cell antigen receptor (TCR) (26, 48) on the surface of infected cells. The TCR on most peripheral T cells is a heterodimeric molecule, composed of α (Vα) and β (Vβ) chains (7). Functionally, Vβ has been associated with the antigen recognition process, and specific recognition of peptide results in the clonal expansion of a subset of Vβ antigen-specific T cells (7, 26). Thus, direct examination of TCR Vβ specificities has been used to identify and track oligoclonal antigen-specific T-cell responses during infection and after immunization (1, 14, 30). Approximately 60 TCR Vβ gene segments are known in humans (30). TCR Vβ usage has been studied in various systems, and either broad or restricted patterns have been identified in a number of areas, including autoimmunity (29, 53), response to viral (10, 25, 40, 42) or bacterial (8, 11, 16, 27) antigens, and tumor immunity (22).
The induction of strong, long-lived immunologic T- and B-cell memory responses is of paramount importance in the development of effective vaccines. Thus, in the present study, we analyzed the TCR Vβ gene usage of specific CD8+ T cells from healthy adults 5 to 40 months after oral immunization with serovar Typhi strain Ty21a, the only licensed attenuated typhoid vaccine, to gain further insights into the clonality and characteristics of T-cell responses to serovar Typhi. Although the responses were oligoclonal, we observed that CD8+ T cells from several Vβ families participated in the anti-serovar Typhi response. Moreover, we studied the induction of memory T-cell subsets and the homing potential of these cells. The specificity of the expanded cell subsets was demonstrated by functional assays on Vβ sorted cells and on clones. Moreover, we observed that Vβ serovar Typhi-specific CD8+ T cells express surface molecules associated with a T memory effector phenotype with gut homing potential.
MATERIALS AND METHODS
Subjects.
Three healthy volunteers, 32 to 41 years old, recruited from the Baltimore-Washington area and University of Maryland at Baltimore campus, participated in this study. Volunteers were vaccinated following the routine immunization schedule for the Ty21a typhoid vaccine, i.e., four spaced doses of 2 × 109 to 6 × 109 CFU of Ty21a at an interval of 48 h between doses (20, 35). Volunteers were screened for good health by medical history, physical examination, normal laboratory tests, including blood counts, and the absence of antibiotic treatment at the times of leukapheresis. The purpose of this study was explained to volunteers, and they gave informed, signed consent. Cells from volunteers 1, 2, and 3 were collected 5, 25, and 40 months after immunization, respectively, to perform a time course analysis of the immune responses to serovar Typhi. Peripheral blood mononuclear cells (PBMC) were isolated by density gradient centrifugation (35).
Target and stimulator cells.
Blasts were established from PBMC isolated from Ty21a vaccinees following standard procedures (35, 36, 44) and maintained in RPMI 1640 (Gibco, Grand Island, New York) supplemented with 100 U/ml penicillin, 100 μg/ml streptomycin, 50 μg/ml gentamicin, 2 mM l-glutamine, 2.5 mM sodium pyruvate, 10 mM HEPES buffer, 1% nonessential amino acids, and 10% heat-inactivated fetal bovine serum (R10) plus 20 IU/ml recombinant human interleukin-2 (rhIL-2) (Boehringer GmbH, Mannheim, Germany). Blasts were infected by incubating in RPMI (without antibiotics) for 3 h at 37°C with wild-type serovar Typhi strain ISP1820 (35, 36). In some experiments, the following day, blasts were γ-irradiated (4,000 rads) and used as stimulators. For cytotoxic T lymphocyte (CTL) assays, infected and noninfected cells were labeled with 200 μCi of sodium chromate (51Cr) (Amersham Pharmacia Biotech, Piscataway, New Jersey) for 1 h at 37°C, washed, and used as targets.
To confirm that targets were infected with serovar Typhi, cells were stained with anti-CSA-1-fluorescein isothiocyanate (FITC) (KPL, Gaithersburg, MD) and analyzed by flow cytometry (35, 36).
Effector cells.
For cytotoxic assays, both ex vivo and in vitro-expanded PBMC from immunized volunteers were used as effectors. In vitro-expanded effectors were obtained using a modification of a previously described technique (35, 36). Briefly, PBMC were restimulated weekly with irradiated autologous serovar Typhi-infected blasts at an effector-to-stimulator cell ratio of 7:1 for 3 weeks in R10 containing 20 IU/ml of rhIL-2. Aliquots of these effector cells were frozen weekly before each new restimulation. The expression of surface molecules on these cell lines was determined by flow cytometry. To this end, effectors were stained with a panel of anti-TCR Vβ monoclonal antibodies (MAbs) (TCR Vβ Repertoire kit, IO Test Beta Mark; Beckman-Coulter, Miami, FL) and combinations of MAbs to CD3 (clone UCHT1), CD4 (clone IM0704), CD25 (clone B1.49.9), CD28 (clone IM1236), HLA-DR (clone H279) (Beckman-Coulter), CD8 (clone SK1), CD56 (clone B159), CD38 (clone HB7), CD45RO (clone UCHL-1), and CD62L (clone Dreg-56) (BD Pharmingen, San Diego, CA). MAbs or the corresponding isotype controls conjugated to the following fluorochromes were used in these studies: FITC, phycoerythrin (PE), energy-coupled dye (ECD) (PE-Texas Red conjugate), peridinin chlorophyll protein (PerCP), PE-Cy5 conjugate, PE-Cy7 conjugate, allophycocyanin (APC), and APC-Cy7 conjugate or biotin (followed by streptavidin-Pacific blue [Molecular Probes, Eugene, Oregon]). The TCR Vβ panel used in these studies included MAbs that recognize 24 different Vβ specificities (i.e., Vβ 5.3, Vβ 7.1, Vβ 3, Vβ 9, Vβ 17, Vβ 16, Vβ 13.1, Vβ 13.6, Vβ 8, Vβ 18, Vβ 5.1, Vβ 20, Vβ 5.2, Vβ 2, Vβ 12, Vβ 23, Vβ 1, Vβ 21.3, Vβ 11, Vβ 22, Vβ 14, Vβ 13.2, Vβ 4, and Vβ 7.2) (see Table 1 for more details), which cover ∼60% of the known specificities (47) (Table 1). Samples were run in a MoFlo flow cytometer/cell sorter system (Dako-Cytomation, Fort Collins, CO) and analyzed using the WinList flow cytometry software analysis package (Verity Software House, Topsham, ME).
TABLE 1.
Panel of TCR Vβ monoclonal antibodies used in these studies and their expression in CD3+ CD8+ lymphocytes from healthy individuals
| Vβ antibody specificity or total | Fluorochrome | MAb clone | % Vβ expression in CD8+ TCRαβ+ cellsa
|
|
|---|---|---|---|---|
| Median | p10-p90 | |||
| Vβ 1 | PE and FITC | BL37.2 | 4.70 | 3.1-8.5 |
| Vβ 2 | PE and FITC | MPB2D5 | 5.60 | 3.3-8.7 |
| Vβ 3 | FITC | CH92 | 3.60 | 0.5-9.9 |
| Vβ 4 | PE and FITC | WJF24 | NA | |
| Vβ 5.1 | PE and FITC | IMMU157 | 3.50 | 1.6-7.3 |
| Vβ 5.2 | PE | 36213 | NA | |
| Vβ 5.3 | PE | 3D11 | NA | |
| Vβ 7.1 | PE and FITC | ZOE | 3.90 | 1.4-8.0 |
| Vβ 7.2 | FITC | ZIZOU4 | NA | |
| Vβ 8 | FITC | 56C5.2 | 4.60 | 2.6-7.0 |
| Vβ 9 | PE | FIN9 | 2.30 | 1.3-4.4 |
| Vβ 11 | PE | C21 | 0.50 | 0.5-1.1 |
| Vβ 12 | FITC | VER2.32 | 2.50 | 1.0-3.7 |
| Vβ 13.1 | PE | IMMU222 | NA | |
| Vβ 13.2 | PE | H132 | NA | |
| Vβ 13.6 | PE and FITC | JU74.3 | 1.90 | 0.8-3.6 |
| Vβ 14 | FITC | CAS1.1.3 | 4.80 | 1.7-13.4 |
| Vβ 16 | FITC | TAMAYA1.2 | 1.20 | 0.5-2.0 |
| Vβ 17 | PE and FITC | E17.5F3 | 5.00 | 2.5-7.0 |
| Vβ 18 | PE | BA62.6 | 0.50 | 0.5-0.9 |
| Vβ 20 | FITC | ELL1.4 | 1.80 | 0.5-5.1 |
| Vβ 21.3 | FITC | IG125 | 2.90 | 1.4-4.8 |
| Vβ 22 | PE and FITC | IMMU546 | 2.90 | 0.5-5.4 |
| Vβ 23 | PE | AF23 | 2.00 | 0.5-3.4 |
| Total | 54.20 | |||
Reference values (median) in healthy controls are derived from van den Beemd et al. (47). p10, 10th percentile; p90, 90th percentile; NA, not available.
For IFN-γ enzyme-linked immunospot (ELISPOT) assays, both ex vivo and in vitro-expanded PBMC (as described above) were also used as effectors.
T-cell cloning.
T cells from volunteers 1 and 3 were expanded for one cycle in the presence of serovar Typhi-infected autologous blasts and cloned by limiting dilution and polyclonal stimulation with 2.5 × 105 irradiated allogeneic blasts and 2.5 × 105 K562/CD32/4-1 BBL cells (kindly provided by M. Mauer and C. June, University of Pennsylvania, Philadelphia, PA) prepulsed with both anti-CD3 (clone OKT3; ATCC, Rockville, MD) and anti-CD28 (clone 9.3; BD Pharmingen) MAbs (0.5 μg/ml each) (24). These cell lines and clones were cultured in R10 supplemented with 60 IU/ml of rhIL-2 for 10 days and expanded by multiple cycles of in vitro restimulation every 10 days with allogeneic blasts and K562/CD32/4-1 BBL cells at a 2:1 ratio of T cells:stimulator cells. The expression of surface homing and/or effector memory molecules was assessed in these T-cell clones by eight-color flow cytometry (MoFlo). The following MAbs (coupled to the indicated fluorochromes) were used: integrin α4β7 (clone ACT-1) was kindly provided by W. Newman, Leukosite, Cambridge, MA, and conjugated to Alexa 488 using an Alexa 488 labeling kit (Molecular Probes); CD27-PE (clone M-T271); CD62L-PE-Cy5 (clone Dreg-56); CCR7-PE-Cy7 (clone 3D12) and CD8-APC-Cy7 (clone Sk1) (BD Pharmingen); CD45RO-ECD (clone UCHL-1) and CD103-biotin (clone 2G5) (Beckman-Coulter); and CCR9-APC (clone 112509; R&D Systems, Minneapolis, MN). Alexa 405-streptavidin was purchased from Molecular Probes.
Cytotoxicity (chromium release test).
As previously described in detail, cytotoxicity against uninfected or serovar Typhi-infected targets was determined by using a 4-h 51Cr release assay (34-36, 44). Specific cytotoxicity mediated by effector cells was calculated by substracting the lysis of uninfected targets from the lysis of serovar Typhi-infected targets. Lysis of noninfected targets was generally less than 10%. The cutoff for positive responses in CTL assays was defined as >10% specific cytotoxicity (34-36, 44).
IFN-γ ELISPOT assays.
The human IFN-γ ELISPOT assay was carried out as previously described (34-36). Briefly, PBMC from immunized volunteers were incubated with uninfected or serovar Typhi-infected stimulator cells. Effector cells cultured without stimulator cells or with CD3/CD28 beads (0.6 μl/ml; Dynal) were used as negative and positive controls, respectively. After 16 h, cultures were assayed for IFN-γ by ELISPOT. Anti-IFN-γ (5 μg/ml, clone 2G1; Endogen, Woburn, MA) and biotinylated anti-IFN-γ (2 μg/ml, clone B133.5; Endogen) MAbs were used as capture and detection reagents, respectively. Complexes were detected by incubation with avidin-peroxidase conjugate (Sigma), and TrueBlue reagent (KPL) was used as a substrate. Each sample was tested in triplicate. Spots were enumerated using a stereomicroscope. Net frequencies were calculated as previously described (34-36). The cutoff for ELISPOT assays was established as the frequency of IFN-γ spot-forming cells/106 PBMC in cocultures of effectors with noninfected targets plus 3 standard errors (SE).
Statistical analysis.
All tests were performed using SigmaStat software (version 3.0; SPSS Science software products, Chicago, IL). P values that were <0.05 were considered significant.
RESULTS
Generation of serovar Typhi-specific polyclonal cell lines for Vβ analysis.
Our initial approach to evaluating the Vβ repertoire of specific T cells elicited in humans following immunization with the attenuated Ty21a typhoid vaccine involved the enrichment of serovar Typhi-specific T cells by repeated cycles of in vitro restimulation with serovar Typhi-infected polyclonal autologous blasts. We have previously reported the use of serovar Typhi-specific T-cell lines following one cycle of in vitro stimulation with serovar Typhi-infected autologous polyclonal blasts to demonstrate specific cytotoxic activity and IFN-γ production in clinical trials of attenuated serovar Typhi oral live vector vaccines (34-36). In the present studies, serovar Typhi-specific polyclonal cell lines from three Ty21a typhoid vaccinees generated following one cycle of in vitro stimulation were further expanded by weekly restimulation with irradiated autologous serovar Typhi-infected blasts for up to 3 weeks before specificity and TCR Vβ repertoire analysis. Aliquots of these effector cells were frozen weekly before each new restimulation, and the phenotype of these cell lines was determined. The specificity of these cell lines was evaluated by measuring their ability to secrete IFN-γ and to kill serovar Typhi-infected targets.
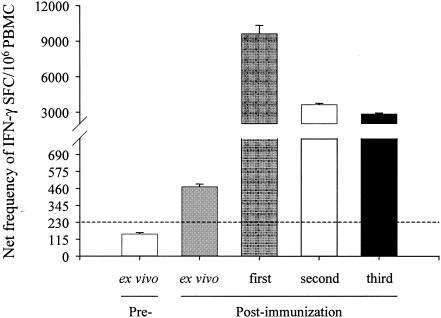
Secretion of IFN-γ by effector cells in response to stimulation with serovar Typhi-infected targets was measured by ELISPOT in ex vivo PBMC and in polyclonal cell lines after one, two, and three cycles of in vitro restimulation. Marked increases in the frequency of IFN-γ-secreting cells in response to stimulation with serovar Typhi-infected targets were observed. The largest increase of IFN-γ production occurred after one cycle of in vitro restimulation. Similarities in the levels of IFN-γ production following two and three cycles of in vitro restimulation suggest a stabilization of these responses with continued in vitro stimulation. Compared with preimmunization levels without in vitro restimulation, postimmunization PBMC without in vitro restimulation showed a 3-fold increase, while polyclonal cell lines showed an average 35-fold increase in serovar Typhi-responsive cells (63-, 24-, and 19-fold after one, two, and three cycles of in vitro restimulation, respectively) (Fig. 1). This corresponds to 0.3 to 1% of the total cell population (2,853 to 9,640 spots/106 cells in these polyclonal cell lines).
FIG. 1.
Frequency of IFN-γ-producing cells before and after immunization with Ty21a ex vivo or following in vitro expansion. Net frequencies of IFN-γ spot-forming cells (SFC) were assessed by an IFN-γ ELISPOT assay using PBMC from volunteer 2 as effectors and autologous blasts infected with wild-type serovar Typhi as stimulators. PBMC were evaluated ex vivo or following coculture with stimulators weekly for up to 3 weeks. The dashed line represents the cutoff for positive ELISPOT assays determined as described in the text. These data are representative of two individual volunteers with similar results.
51Cr release assays and correlation with IFN-γ-secreting effector T-cell populations.
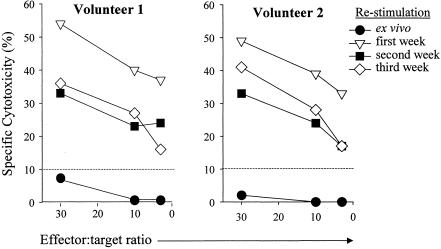
Cytolytic function of the polyclonally expanded cell lines was measured by standard 51Cr release assays. After 3 weeks of in vitro restimulation with autologous serovar Typhi-infected targets, we observed that these cell lines maintain high levels of CTL activity against serovar Typhi-infected targets. As shown in Fig. 2, similar results were obtained in volunteers 1 and 2. Consistent with our previous results, ex vivo PBMC from both volunteers were unable to lyse autologous blasts infected with serovar Typhi (35, 36). However, both volunteers showed increased levels of cytotoxic activity after in vitro restimulation. Although CTL activity is at best semiquantitative, it is important to note that, as observed with IFN-γ production, the highest increase of the cytotoxicity levels occurred after one in vitro restimulation followed by stabilization after two and three cycles of in vitro restimulation (e.g., volunteer 2 exhibited 33, 17, and 17% of specific lysis at an effector-to-target cell [E:T] ratio of 3:1 after one, two, and three in vitro restimulation cycles, respectively). Moreover, as we have previously described (35), when we compared the results obtained by ELISPOT with those obtained by 51Cr release assays, a positive correlation (R2 = 0.910, P < 0.046) was observed between the frequencies of serovar Typhi antigen-responsive cells, as evidenced by IFN-γ production and the cytotoxic activity of these cell lines at E:T ratios of 3:1 (data not shown).
FIG. 2.
Ability of effectors to lyse serovar Typhi-infected target cells during the generation of polyclonal cell lines. Pre- or postimmunization PBMC from volunteers 1 and 2 were evaluated ex vivo or after coculture with serovar Typhi-infected autologous cells for up to three cycles of in vitro weekly restimulation for the ability to lyse targets by 51Cr release assays. The dashed line represents the cutoff for positive 51Cr release assays as described in the text.
Taken together, the results from IFN-γ ELISPOT and CTL assays clearly show that the polyclonal cell lines remain functional after repeated cycles of specific antigenic stimulation.
Characterization of the effector population.
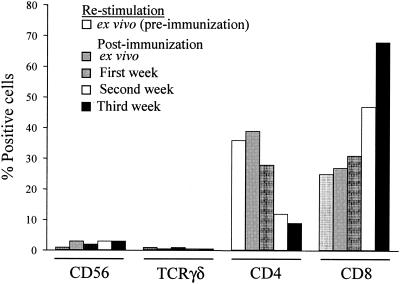
We next investigated which cell populations in PBMC from immunized volunteers were preferentially expanded after several cycles of in vitro restimulation with serovar Typhi-infected autologous blasts. In agreement with previous results using a similar system (34-36), a major expansion in CD8+ T-cell populations, as determined by flow cytometry, occurred during in vitro restimulations. The percentage of CD8+ T-cell populations increased progressively after each additional in vitro restimulation (e.g., 26, 44, and 63% after one, two, and three cycles of in vitro restimulation, respectively). These increases were accompanied by concomitant decreases in the percentage of CD4+ cells at the same time points (28, 13, and 9% after one, two, and three cycles of in vitro restimulation, respectively) (Fig. 3). No changes were observed in the percentage of NK (CD56+) or T cells bearing γ/δ T-cell receptors (Fig. 3).
FIG. 3.
Distribution of lymphocyte subpopulations in ex vivo PBMC or after different cycles of in vitro restimulation. PBMC from volunteer 1 were collected before and after immunization with attenuated serovar Typhi strain Ty21a typhoid vaccine, and cells were evaluated ex vivo or after weekly restimulation with autologous serovar Typhi-infected targets for up to 3 weeks. Percentages of lymphocyte subpopulations were determined by flow cytometry. These data are representative of two individual experiments using cells from the same volunteer with similar results.
To further characterize the composition of polyclonal cell lines after in vitro restimulation, the proportion of cells expressing molecules involved in T-cell activation and those associated with memory effector T cells in populations following three cycles of in vitro expansion were compared to those present in PBMC without in vitro restimulation. To this end, PBMC from Ty21a vaccinees ex vivo or cocultured with stimulators for up to 3 weeks were stained with MAbs to CD3, CD4, CD8, CD56, CD25, CD28, CD38, CD45RO, CD62L, and HLA-DR and analyzed by multicolor flow cytometry. The results are shown in Table 2. Of the various CD3+ CD8+ cell subpopulations expressing CD45RO+, a T-cell memory marker, only the percentages of CD38+ and HLA-DR+ differed significantly between ex vivo and in vitro-restimulated PBMC. Five-color flow cytometric analysis showed that in vitro-expanded CD3+ CD8+ CD45RO+ T cells contained a greater proportion of cells bearing activation molecules (HLA-DR, 97%; CD38+, 39%) than did the initial ex vivo population (HLA-DR, 24%; CD38+, 9%). Smaller increases were also observed in the percentages of CD3+ CD4+ CD45RO+ populations coexpressing HLA-DR and CD38 after each cycle of restimulation (Table 2). Of note is that no major differences were observed in the expression of CD62L and CD28 in either cell population (Table 2).
TABLE 2.
Expression of activation molecules on CD3+ CD4+ CD45RO+ and CD3+ CD8+ CD45RO+ T-cell memory subsets before and after in vitro restimulation
| Molecule | In vitro restimulationa
|
|||
|---|---|---|---|---|
| None | 1 | 2 | 3 | |
| CD25 | ||||
| CD4+ | 7 | 21 | 13 | 17 |
| CD8+ | 4 | 33 | 15 | 5 |
| CD28 | ||||
| CD4+ | 71 | 76 | 74 | 83 |
| CD8+ | 74 | 81 | 79 | 89 |
| CD38 | ||||
| CD4+ | 6 | 19 | 40 | 29 |
| CD8+ | 9 | 37 | 69 | 39 |
| HLA-DR | ||||
| CD4+ | 8 | 28 | 26 | 55 |
| CD8+ | 24 | 54 | 75 | 97 |
PBMC from volunteer 2 were collected 25 months following immunization with attenuated serovar Typhi strain Ty21a typhoid vaccine and restimulated weekly with autologous serovar Typhi-infected targets for up to 3 weeks. Percentages of positive cells were determined by flow cytometry. Data in columns show the percentage of positive cells.
Taken together, the results from the functional assays and the analysis of surface expression of activation/memory molecules suggest that activated CD8+ T cells coexpressing memory markers remain functional after several rounds of in vitro stimulation and are therefore appropriate for studying the TCR Vβ repertoire induced by immunization of volunteers with the Ty21a typhoid vaccine.
Flow cytometric Vβ analysis in polyclonal cell lines.
Following functional validation and characterization of the polyclonal cell lines, we next evaluated the TCR Vβ repertoire of these populations. First, we examined whether CD8+ T cells with a broad TCR Vβ repertoire or a limited selected repertoire are involved in the host's serovar Typhi response after administration of the Ty21a typhoid vaccine. To this end, we studied the TCR Vβ usage of these cell lines by flow cytometry using a panel of 24 MAbs which cover ∼60% of the known specificities (47).
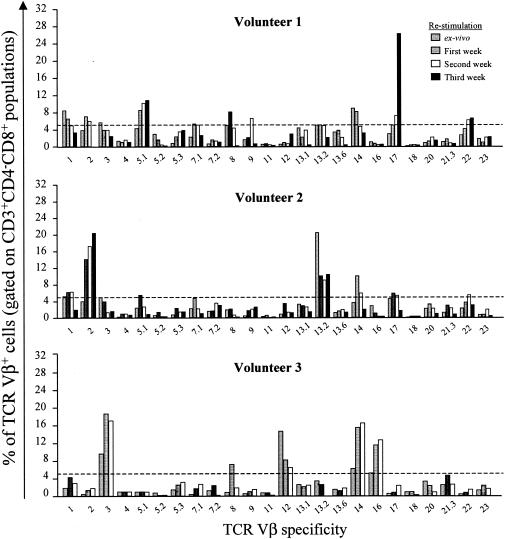
Five-color cytofluorometric analysis was performed to determine whether CD4+ or CD8+ T cells of defined Vβ were preferentially expanded by in vitro restimulation. Changes above the mean plus 3 SE of the ex vivo effector cells (controls) were defined as being increased. Although TCR Vβ diversity varied greatly from volunteer to volunteer, we observed the oligoclonal expansion of particular Vβ subsets in each individual (Vβ 2, 5.1, 8, 17, and 22 in volunteer 1; Vβ 1, 2, 5.1, 14, 17, and 22 in volunteer 2; and Vβ 3, 8, 14, and 16 in volunteer 3). As shown in Fig. 4, volunteer 1 exhibited a marked increase in the percentage of CD8+ T cells expressing TCR Vβ 17 after three in vitro restimulations (26.4%) compared to postimmunization levels without in vitro restimulation (3.0%), whereas CD4+ Vβ 17+ cells did not change significantly over time (5.8, 5.4, 5.4, and 5.8% ex vivo and after one, two, and three restimulations, respectively) (data not shown). In volunteer 2, a major increase in the percentage of CD8+ T cells expressing TCR Vβ 2 was found even after a single cycle of in vitro restimulation (14.3%), and it continued to rise after three successive cycles of in vitro restimulation (20.5%), compared to postimmunization levels without in vitro restimulation (4.1%) (Fig. 4). CD4+ Vβ 2+ cells did not change significantly over time (4.2, 6.3, 5.3, and 6.5% ex vivo and after one, two, and three restimulations, respectively) (data not shown). Taken together, the most prominent changes were observed in the Vβ 2, 3, 5.1, 14, 16, and 17 families. Increases in Vβ 2, Vβ 5.1, and Vβ 17 were observed in both volunteer 1 and volunteer 2. Of note is that although in volunteer 3 the Vβ 2 and Vβ 17 changes did not reach the mean percentage plus 3 SE of ex vivo effector cells used as a cutoff level of positivity, increases of 5 to 18 times over the values observed ex vivo without restimulation were observed after the first and second restimulations (i.e., for Vβ 2, 0.1, 1.43, and 1.85% and for Vβ 17, 0.19, 1.02, and 2.43% of positive cells were observed ex vivo, after one cycle, and after two cycles, respectively).
FIG. 4.
TCR Vβ repertoire analysis of serovar Typhi-specific CD8+ T cells in volunteers immunized with the attenuated serovar Typhi strain Ty21a typhoid vaccine. PBMC from volunteers 1, 2, and 3 were analyzed ex vivo or after weekly restimulation with autologous serovar Typhi-infected targets for up to 3 weeks, and the percentage of 24 TCR Vβ specificities was determined by flow cytometry. Data are presented as the percentage of TCR Vβ+ cells in CD3+ CD4− CD8+ gated populations. Only the percentages of TCR Vβ+ cells above the mean plus 3 SE of all Vβ specificities ex vivo postimmunization (dashed lines) were considered to be significant. The percentage of TCR Vβ 2 in volunteer 1 after three cycles of restimulation is not available because of technical difficulties.
It is important to note that except for the overrepresentation of Vβ 13.2 in volunteer 2 and Vβ 12 in volunteer 3, the reported distributions of Vβ usage observed in ex vivo effectors (controls) are comparable to the Vβ usage previously reported for healthy controls (Table 1) (47). Moreover, the sum average of the percentages of all Vβ specificities in ex vivo CD8+ PBMC after immunization was 69% (volunteer 1, 72%; volunteer 2, 72%; and volunteer 3, 61%), further supporting the contention that the Vβ usage in these volunteers is similar to that previously reported for healthy individuals (∼60% of CD8+ PBMC) (Table 1) (47).
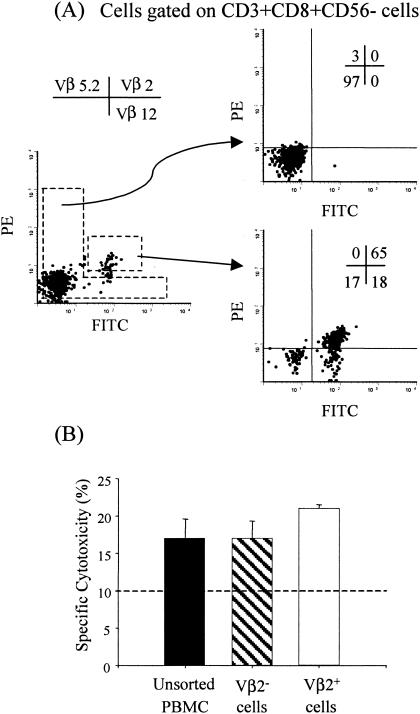
We next evaluated whether, following the remarkable expansion of CD8+ TCR Vβ 2 T cells, they remain functional in mediating CTL activity against serovar Typhi-infected cells. To this end, all three in vitro-restimulated postimmunization PBMC aliquots from volunteer 2 were pooled and surface-stained for CD8, CD56, CD3, Vβ 2, Vβ 5.2, and Vβ 12 and the TCR Vβ 2+ CD8+ CD56− CD3+ and TCR Vβ 2− CD8+ CD56− CD3+ populations sorted (Fig. 5A). The CTL activity of sorted as well as unsorted populations was measured by 51Cr release assays against serovar Typhi-infected autologous blasts. More than 10% specific lysis was observed in both sorted and unsorted PBMC at an E:T ratio of 3:1 (Fig. 5B). These results demonstrate that both TCR Vβ 2+ CD3+ CD8+ and TCR Vβ 2− CD3+ CD8+ populations are functional and specific for serovar Typhi-infected targets. Moreover, these results also indicate that TCR Vβ specificities other than Vβ 2 are also able to mediate CTL activity. Taken together, these observations support the notion that more than one TCR Vβ family is involved in the host's response to serovar Typhi after vaccination with the Ty21a typhoid vaccine.
FIG. 5.
Functional analyses of sorted Vβ 2+ CD8+ and Vβ 2− CD8+ cells from volunteer 2. (A) Sorting strategy to isolate Vβ 2+ CD8+ and Vβ 2− CD8+ cells. Expanded cells from volunteer 2 were stained with anti-CD3-ECD, anti-CD8-PerCP, anti-Vβ 5.2-PE, anti-Vβ 12-FITC, and anti Vβ 2-PE/FITC MAbs and sorted by flow cytometry into CD3+ CD8+ CD56− cells expressing or not expressing Vβ 2. The percentages of each cell population following sorting are shown in the upper right corners of individual histograms. (B) Unfractionated PBMC, Vβ 2+ CD3+ CD8+ CD56−, or Vβ 2− CD3+ CD8+ CD56− sorted cell populations were used as effector cells against serovar Typhi-infected autologous blasts, and the cytotoxicity was measured by 51Cr release assays. Bars represent the mean percent specific cytotoxicity observed for each target at an E:T ratio of 3:1. Error bars show the standard deviations of triplicate cultures. The dashed line represents the cutoff for positive 51Cr release assays.
TCR Vβ repertoire and expression of molecules involved in homing and effector and memory function in T-cell clones specific for serovar Typhi antigens.
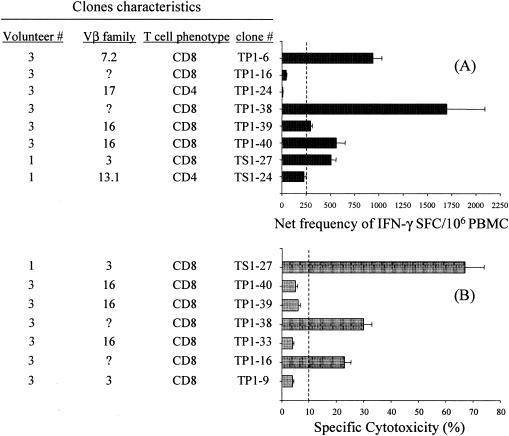
To confirm the Vβ usage of T cells elicited by immunization, as well as to identify additional TCR Vβ specificities that could be involved in specific responses to serovar Typhi, T-cell clones were derived from PBMC from volunteers 2 and 3. T-cell clones were derived by limiting dilution, as described in Materials and Methods, and established cell lines were examined for TCR Vβ usage, a procedure that also helped to confirm the clonality of the populations. The functional activity of these T-cell clones was examined by specific cytotoxicity and frequency of IFN-γ-secreting cells against serovar Typhi-infected targets by 51Cr release and IFN-γ ELISPOT assays, respectively. Figure 6 summarizes the IFN-γ production, cytotoxicity, and TCR Vβ usage of eight CD8+ T-cell clones. In ELISPOT assays, two CD4+ T-cell clones (TP1-24 and TS1-24) were used as negative controls. The clones that either killed infected targets or produced IFN-γ were considered responders. Among the six responder CD8+ T-cell clones (TP1-6, TP1-16, TP1-38, TP1-39, TP1-40, and TS1-27), one clone expressed Vβ 7.2, one expressed Vβ 3, and two expressed Vβ 16. For two clones (TP1-16 and TP1-38), no reactivity was observed with any of the anti-TCR Vβ MAbs in our panel. This is likely the result of the lack of a MAb for the appropriate Vβ specificity in the panel, which in the case of volunteer 3 identified 61% of the TCR Vβ repertoire. Of note is that while some T-cell clones (e.g., TP1-38 and TS1-27) exhibited both CTL and IFN-γ production capabilities, other clones (e.g., TP1-16) were only either cytotoxic or able to produce IFN-γ, suggesting heterogeneity in the effector function of different T-cell subpopulations following immunization with serovar Typhi vaccines.
FIG. 6.
Functional analyses of T-cell clones. (A) The frequency of IFN-γ-producing clones in the presence of autologous blasts infected with serovar Typhi was evaluated by IFN-γ ELISPOT assays. The dashed line represents the cutoff for positive ELISPOT assays determined as described in the text. (B) Specific cytotoxicity against serovar Typhi-infected targets was assessed by 51Cr release assays. Bars represent the mean percent specific cytotoxicity observed for each target at an E:T ratio of 10:1. Error bars show the standard deviations of triplicate cultures. The dashed line represents the cutoff for positive 51Cr release assays.
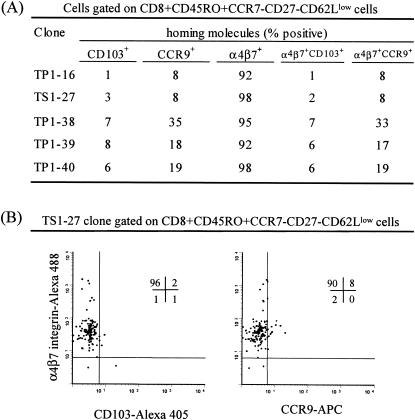
It is widely recognized that oral immunization induces effector memory cells carrying distinct homing receptors (e.g., integrin α4β7, CCR9, and CD103) that might endow the effector cells with the capability to migrate preferentially to the gut (13, 18, 33, 39). Thus, it was important to assess whether these molecules are present on the T-cell clones from volunteers immunized orally with the Ty21a typhoid vaccine described above. Moreover, it was also important to determine whether these T-cell clones exhibit an effector memory phenotype. To this end, clones were stained with MAbs to CD8, CD27, CCR7, CD45RO, CD62L, α4β7, CCR9, and CD103 and analyzed by eight-color flow cytometry. Despite the observed functional and TCR Vβ usage differences among these clones, there were remarkable similarities in their phenotype and expression of homing molecules. All serovar Typhi-specific T-cell clones were found to be CCR7− CD27− CD45RO+ CD62Llow, i.e., exhibit an effector memory phenotype (9, 37). Moreover, clones expressed high levels of integrin α4β7 (>92%), moderate levels of CCR9 (8 to 35%), and low levels of CD103 (1 to 8%) (Fig. 7). Taken together, these results confirm an oligoclonal expansion of Vβ serovar Typhi-specific CD8+ T cells, which have a phenotype consistent with T effector memory cells with preferential gut homing potential.
FIG. 7.
Expression of surface homing, effector, and memory markers as assessed by eight-color flow cytometry. (A) T-cell clones were stained with MAbs to integrin α4β7, CD27, CD62L, CCR7, CD8, CD45RO, CD103, and CCR9 and the data analyzed by flow cytometry. Results are presented as percentages of positive cells in CD8+ CD45RO+ CCR7− CD27− CD62Llow gated populations. (B) Expression of homing markers on the TS1-27 clone. The percentages of positive cells in each quadrant are shown in the upper right corners of individual histograms.
DISCUSSION
In the present study, we evaluated the TCR Vβ specificities of CD8+ T cells from three healthy adults orally immunized with the attenuated serovar Typhi Ty21a strain typhoid vaccine. PBMC from Ty21a typhoid vaccinees were expanded by weekly restimulation with irradiated autologous serovar Typhi-infected blasts for up to 3 weeks and used as effector cells. We found that CD8+ T cells from several Vβ families participated in the anti-serovar Typhi response. Although TCR Vβ usage varied from one volunteer to another, our results are consistent with an oligoclonal Vβ subset expansion in each volunteer (Vβ 2, 5.1, 8, 17, and 22 in volunteer 1; Vβ 1, 2, 5.1, 14, 17, and 22 in volunteer 2; and Vβ 3, 8, 14, and 16 in volunteer 3). Moreover, we observed that these subsets were antigen specific, as shown by functional assays on cells sorted based on their Vβ expression and on T-cell clones. Furthermore, we show that Vβ serovar Typhi-specific CD8+ T cells exhibit a phenotype consistent with effector memory cells with gut homing potential.
Restricted T-cell Vβ repertoires have been shown in infections with bacteria, including Streptococcus (27, 46), Staphylococcus (8, 38), Mycobacterium tuberculosis (11, 28), Listeria (15), and Shigella (16). Although the TCR Vβ repertoire observed with these pathogens was somewhat diverse, TCR Vβ 2 and Vβ 3 usage appears to be favored. The observation in our studies with volunteers immunized with serovar Typhi that Vβ 2 and Vβ 3 specificities were preferentially used appears to support this notion. Interestingly, a study with Shigella (16), a pathogen that shares 80% homology with serovar Typhi, showed a pattern of TCR Vβ usage surprisingly similar to that observed in our expanded T-cell lines. Despite the fact that in this study a limited panel composed of just nine MAbs to different TCR Vβ specificities was used (compared to the 24 Vβ specificities evaluated in our study), the increases in TCR Vβ 2, Vβ 3, Vβ 5.1, Vβ 13.6, and Vβ 17 reported were remarkably similar to the ones observed in our study (TCR Vβ 1, Vβ 2, Vβ 3, Vβ 5.1, Vβ 8, Vβ 14, Vβ 16, Vβ 17, and Vβ 22). Taken together, these observations support the notion that it is likely that T-cell responses against some, if not most, bacteria might be oligoclonal in nature and use a relatively restricted set of TCR Vβ specificities.
The aim of the studies presented in this paper was to analyze the TCR Vβ usage in healthy adults 5 to 40 months after oral immunization with serovar Typhi strain Ty21a to evaluate the persistence of serovar Typhi-specific responses over time. Because the postimmunization specimens were collected long after the T-cell memory contraction phase took place, it is not surprising that we did not observe differences when comparing the ex vivo TCR Vβ usage before and after immunization in two of the volunteers (volunteers 1 and 2) (data not shown). To overcome this difficulty, we evaluated the Vβ repertoire of specific T cells after repeated cycles of in vitro restimulation with serovar Typhi-infected polyclonal autologous blasts. Although this procedure does not allow us to estimate their frequency in vivo, available evidence with other gram-negative bacteria suggests that it should not have negatively affected the identification of at least some of the different specificities elicited by immunization with serovar Typhi. In this context, although some reports studying virus-specific CTL populations showed that in vitro expansion of lymphocytes can introduce a significant bias in TCR usage (41), a previous study in Shigella (16) showed that the in vitro TCR Vβ repertoire was similar to that observed in vivo. By comparing the in vivo and in vitro changes in the TCR Vβ repertoire over time, the authors concluded that the in vitro response induced by Shigella antigens showed a TCR Vβ distribution similar to that observed in vivo. Interestingly, the changes in TCR Vβ usage observed in that study peaked 8 to 11 days after the onset of disease, returning to baseline levels after 30 days. Islam et al. hypothesized that this kinetics was either due to homing of circulating cells to the gut-associated lymphoid tissues or, alternatively, due to apoptotic cell death (16). Interestingly, in the present study, we also observed that the expansion of some Vβ specificities after one cycle of in vitro restimulation (about day 8) was followed by contraction and/or stabilization of these same Vβ specificities during the next rounds of in vitro restimulation. Of note is that after one cycle of in vitro restimulation, we observed stabilization in the cytotoxicity of and IFN-γ production by serovar Typhi-specific CD8+ T cells, despite an increase in the percentage of CD8+ T cells up to the third week of restimulation. These results may reflect the loss of some clones (e.g., by apoptosis death or loss of specificity) and their replacement by others following successive in vitro restimulations, a phenomenon frequently observed with T-cell clones. Finally, it should be emphasized that it is presently not possible to conclusively assess to what extent the specificities that we observed following in vitro expansion are representative of those present in vivo following immunization. This assertion is based on the lack of a complete understanding of the full complement of immune responses to serovar Typhi following immunization that depend, at least in part, on the set of antigens presented during the various stages of in vivo serovar Typhi infection (50), the presence of variables during in vitro expansion that are difficult to control, and technological limitations. These limitations include the following: (i) in most cases, TCR Vβ antigen-specific T-cell precursors exist at frequencies below the level of detection by standard ex vivo methodologies, e.g., tetrameric complexes; (ii) even in the case of high in vivo frequency of certain TCR Vβ antigen-specific T-cell precursors, T-cell stimulation may require a particular TCR conformation or conformational change that is found only on the surface of certain presenting cells (6); and (iii) while Bouneaud et al. (2) showed that nearly half of their tetramer-binding T cells were unable to expand in response to physiological levels of antigen, Bullock et al. (4) showed that the reverse may also occur, i.e., T cells that show strong functional reactivity towards target cells may not necessarily be detected ex vivo by major histocompatibility complex-tetramer staining.
It is well established that lymphocytes continuously recirculate from peripheral blood to secondary lymphoid tissues (3). This recirculation is essential for homing of effector memory cells, which do not express CCR7 or CD27 and are mostly CD45RO+ CD62Llow, to effector sites (9, 13, 37). Serovar Typhi cells are highly invasive bacteria that rapidly and efficiently colonize and invade the mucosa of the small intestine following ingestion (19). Thus, in Salmonella infections, it is likely that specific memory effector T cells in circulation will be homing to effector sites in the gut where they might encounter Salmonella antigens. Selective homing of effector memory cells to the lamina propria of the small intestine is driven, to a large extent, by the expression of the integrin α4β7, which binds MAdCAM-1, a molecule selectively expressed in the venules of normal and inflamed intestinal endothelium (5). In this regard, a study by Lundin et al. has shown that immunization with the Ty21a typhoid vaccine induces antigen-specific T cells that preferentially express integrin α4β7 and are found in circulation up to 21 days following immunization (23). However, other molecules have also recently been shown to participate in this homing process. For example, expression of CCR9 in subsets of circulating integrin α4β7hi or αEβ7 (CD103) lymphocytes plays an important role in homing of these cells to the small intestine (13, 18, 43). Thus, it was important to evaluate the patterns of expression of these homing molecules in Vβ serovar Typhi-specific CD8+ T cells. We observed that T-cell clones, regardless of their TCR Vβ usage, exhibited an effector memory phenotype (i.e., CCR7− CD27− CD45RO+ CD62Llow) and coexpressed high levels of integrin α4β7, intermediate levels of CCR9, and low levels of CD103 gut homing molecules, suggesting that these TCR Vβ-specific clones have the potential to home to the gut-associated lymphoid tissues.
It has been reported that only a few Vβ families predominate in the human gut mucosa (1), with the most frequent being Vβ 1, Vβ 2, Vβ 3, and Vβ 6 (49). Because we studied peripheral blood, we have to consider the possibility that our observations might not reflect the full spectrum of TCR Vβ usage by serovar Typhi-specific CD8+ T cells in the gut microenvironment in vivo. However, our observations that (i) serovar Typhi-specific CD8+ T cells from Ty21a vaccinees exhibited gut homing potential and (ii) except for Vβ 6, which was not present in our MAb panel, we observed all other Vβ specificities (Vβ 1, Vβ 2, and Vβ 3, with the most prominent changes in Vβ 2 and Vβ 3) support the contention that the TCR Vβ usage that we observed in circulation might reflect to a large extent that found in the gut mucosa microenvironment.
Taken together, our observations suggest that the CD8+ effector memory T-cell subpopulations of the restricted TCR Vβ specificities described in this paper are likely to be the effector cells that mediate anti-serovar Typhi long-term cell-mediated immune responses in the local microenvironment of the gut mucosa following oral immunization with attenuated strains of serovar Typhi.
Acknowledgments
We are indebted to the volunteers who allowed us to perform this study. We also thank Bernadette McConnell and the staff from the Blood Bank of the University of Maryland hospital for their help in collecting leukapheresis specimens and Regina Harley for excellent technical assistance in flow cytometric determinations and sorting.
This work was supported by a grant from the National Institute of Allergy and Infectious Diseases, NIH (R01 AI36525 to M.B.S.).
Editor: J. D. Clements
REFERENCES
- 1.Blumberg, R. S., C. E. Yockey, G. G. Gross, E. C. Ebert, and S. P. Balk. 1993. Human intestinal intraepithelial lymphocytes are derived from a limited number of T cell clones that utilize multiple V beta T cell receptor genes. J. Immunol. 150:5144-5153. [PubMed] [Google Scholar]
- 2.Bouneaud, C., P. Kourilsky, and P. Bousso. 2000. Impact of negative selection on the T cell repertoire reactive to a self-peptide: a large fraction of T cell clones escapes clonal deletion. Immunity 13:829-840. [DOI] [PubMed] [Google Scholar]
- 3.Brandtzaeg, P., I. N. Farstad, and G. Haraldsen. 1999. Regional specialization in the mucosal immune system: primed cells do not always home along the same track. Immunol. Today 20:267-277. [DOI] [PubMed] [Google Scholar]
- 4.Bullock, T. N., D. W. Mullins, T. A. Colella, and V. H. Engelhard. 2001. Manipulation of avidity to improve effectiveness of adoptively transferred CD8(+) T cells for melanoma immunotherapy in human MHC class I-transgenic mice. J. Immunol. 167:5824-5831. [DOI] [PubMed] [Google Scholar]
- 5.Butcher, E. C., M. Williams, K. Youngman, L. Rott, and M. Briskin. 1999. Lymphocyte trafficking and regional immunity. Adv. Immunol. 72:209-253. [DOI] [PubMed] [Google Scholar]
- 6.Coles, R. M., C. M. Jones, A. G. Brooks, P. U. Cameron, W. R. Heath, and F. R. Carbone. 2003. Virus infection expands a biased subset of T cells that bind tetrameric class I peptide complexes. Eur. J. Immunol. 33:1557-1567. [DOI] [PubMed] [Google Scholar]
- 7.Davis, M. M., and P. J. Bjorkman. 1988. T-cell antigen receptor genes and T-cell recognition. Nature 334:395-402. [DOI] [PubMed] [Google Scholar]
- 8.Ericson, M. L., N. A. Mooney, and D. J. Charron. 1996. Prokaryotic production of the human T cell receptor Vbeta2 chain and binding to toxic-shock syndrome toxin-1. Gene 168:257-260. [DOI] [PubMed] [Google Scholar]
- 9.Esser, M. T., R. D. Marchese, L. S. Kierstead, L. G. Tussey, F. Wang, N. Chirmule, and M. W. Washabaugh. 2003. Memory T cells and vaccines. Vaccine 21:419-430. [DOI] [PubMed] [Google Scholar]
- 10.Gagnon, S. J., A. Leporati, S. Green, S. Kalayanarooj, D. W. Vaughn, H. A. Stephens, S. Suntayakorn, I. Kurane, F. A. Ennis, and A. L. Rothman. 2001. T cell receptor Vbeta gene usage in Thai children with dengue virus infection. Am. J. Trop. Med. Hyg. 64:41-48. [DOI] [PubMed] [Google Scholar]
- 11.Gambón-Deza, F., M. Pacheco Carracedo, T. Cerd á Mota, and J. Montes Santiago. 1995. Lymphocyte populations during tuberculosis infection: Vβ repertoires. Infect. Immun. 63:1235-1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gomez-Duarte, O. G., M. F. Pasetti, A. Santiago, M. B. Sztein, S. L. Hoffman, and M. M. Levine. 2001. Expression, extracellular secretion, and immunogenicity of the Plasmodium falciparum sporozoite surface protein 2 in Salmonella vaccine strains. Infect. Immun. 69:1192-1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hamann, A., D. P. Andrew, D. Jablonski-Westrich, B. Holzmann, and E. C. Butcher. 1994. Role of alpha 4-integrins in lymphocyte homing to mucosal tissues in vivo. J. Immunol. 152:3282-3293. [PubMed] [Google Scholar]
- 14.Hingorani, R., I. H. Choi, P. Akolkar, B. Gulwani-Akolkar, R. Pergolizzi, J. Silver, and P. K. Gregersen. 1993. Clonal predominance of T cell receptors within the CD8+ CD45RO+ subset in normal human subjects. J. Immunol. 151:5762-5769. [PubMed] [Google Scholar]
- 15.Huleatt, J. W., I. Pilip, K. Kerksiek, and E. G. Pamer. 2001. Intestinal and splenic T cell responses to enteric Listeria monocytogenes infection: distinct repertoires of responding CD8 T lymphocytes. J. Immunol. 166:4065-4073. [DOI] [PubMed] [Google Scholar]
- 16.Islam, D., B. Wretlind, A. A. Lindberg, and B. Christensson. 1996. Changes in the peripheral blood T-cell receptor Vβ repertoire in vivo and in vitro during shigellosis. Infect. Immun. 64:1391-1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ivanoff, B., M. M. Levine, and P. H. Lambert. 1994. Vaccination against typhoid fever: present status. Bull. W. H. O. 72:957-971. [PMC free article] [PubMed] [Google Scholar]
- 18.Kantele, A., J. Zivny, M. Hakkinen, C. O. Elson, and J. Mestecky. 1999. Differential homing commitments of antigen-specific T cells after oral or parenteral immunization in humans. J. Immunol. 162:5173-5177. [PubMed] [Google Scholar]
- 19.Levine, M., and M. Sztein. 2000. Shigella, Salmonella typhi, and Escherichia coli, p. 171-194. In M. Cunningham and R. Fujinami (ed.), Effects of microbes on the immune system. Lippincott Williams & Wilkins, Philadelphia, Pa.
- 20.Levine, M. M., C. Ferreccio, P. Abrego, O. S. Martin, E. Ortiz, and S. Cryz. 1999. Duration of efficacy of Ty21a, attenuated Salmonella typhi live oral vaccine. Vaccine 17:S22-S27. [DOI] [PubMed] [Google Scholar]
- 21.Levine, M. M., and O. S. Levine. 1997. Influence of disease burden, public perception, and other factors on new vaccine development, implementation, and continued use. Lancet 350:1386-1392. [DOI] [PubMed] [Google Scholar]
- 22.Lim, A., L. Trautmann, M. A. Peyrat, C. Couedel, F. Davodeau, F. Romagne, P. Kourilsky, and M. Bonneville. 2000. Frequent contribution of T cell clonotypes with public TCR features to the chronic response against a dominant EBV-derived epitope: application to direct detection of their molecular imprint on the human peripheral T cell repertoire. J. Immunol. 165:2001-2011. [DOI] [PubMed] [Google Scholar]
- 23.Lundin, B. S., C. Johansson, and A.-M. Svennerholm. 2002. Oral immunization with a Salmonella enterica serovar Typhi vaccine induces specific circulating mucosa-homing CD4+ and CD8+ T cells in humans. Infect. Immun. 70:5622-5627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Maus, M. V., A. K. Thomas, D. G. Leonard, D. Allman, K. Addya, K. Schlienger, J. L. Riley, and C. H. June. 2002. Ex vivo expansion of polyclonal and antigen-specific cytotoxic T lymphocytes by artificial APCs expressing ligands for the T-cell receptor, CD28 and 4-1BB. Nat. Biotechnol. 20:143-148. [DOI] [PubMed] [Google Scholar]
- 25.McCloskey, T. W., V. Haridas, R. Pahwa, and S. Pahwa. 2002. T cell receptor V beta repertoire of the antigen specific CD8 T lymphocyte subset of HIV infected children. AIDS 16:1459-1465. [DOI] [PubMed] [Google Scholar]
- 26.Meuer, S. C., K. A. Fitzgerald, R. E. Hussey, J. C. Hodgdon, S. F. Schlossman, and E. L. Reinherz. 1983. Clonotypic structures involved in antigen-specific human T cell function. Relationship to the T3 molecular complex. J. Exp. Med. 157:705-719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ohara-Nemoto, Y., and M. Kaneko. 1996. Expression of T-cell receptor V beta 2 and type 1 helper T-cell-related cytokine mRNA in streptococcal pyrogenic exotoxin-C-activated human peripheral blood mononuclear cells. Can. J. Microbiol. 42:1104-1111. [DOI] [PubMed] [Google Scholar]
- 28.Ohmen, J. D., P. F. Barnes, C. L. Grisso, B. R. Bloom, and R. L. Modlin. 1994. Evidence for a superantigen in human tuberculosis. Immunity 1:35-43. [DOI] [PubMed] [Google Scholar]
- 29.Paliard, X., S. G. West, J. A. Lafferty, J. R. Clements, J. W. Kappler, P. Marrack, and B. L. Kotzin. 1991. Evidence for the effects of a superantigen in rheumatoid arthritis. Science 253:325-329. [DOI] [PubMed] [Google Scholar]
- 30.Pannetier, C., J. Even, and P. Kourilsky. 1995. T-cell repertoire diversity and clonal expansions in normal and clinical samples. Immunol. Today 16:176-181. [DOI] [PubMed] [Google Scholar]
- 31.Pasetti, M. F., R. J. Anderson, F. R. Noriega, M. M. Levine, and M. B. Sztein. 1999. Attenuated ΔguaBA Salmonella typhi vaccine strain CVD 915 as a live vector utilizing prokaryotic or eukaryotic expression systems to deliver foreign antigens and elicit immune responses. Clin. Immunol. 92:76-89. [DOI] [PubMed] [Google Scholar]
- 32.Pasetti, M. F., M. M. Levine, and M. B. Sztein. 2003. Animal models paving the way for clinical trials of attenuated Salmonella enterica serovar Typhi live oral vaccines and live vectors. Vaccine 21:401-418. [DOI] [PubMed] [Google Scholar]
- 33.Pasetti, M. F., R. Salerno-Gonçalves, and M. B. Sztein. 2005. Mechanisms of adaptive immunity that prevent colonization at mucosal surfaces, p. 35-47. In J. P. Nataro, P. S. Cohen, H. L. T. Mobley, and J. N. Weiser (ed.), Colonization of mucosal surfaces. American Society for Microbiology, Washington, D.C.
- 34.Salerno-Goncalves, R., M. Fernandez-Vina, D. M. Lewinsohn, and M. B. Sztein. 2004. Identification of a human HLA-E-restricted CD8+ T cell subset in volunteers immunized with Salmonella enterica serovar Typhi strain Ty21a typhoid vaccine. J. Immunol. 173:5852-5862. [DOI] [PubMed] [Google Scholar]
- 35.Salerno-Goncalves, R., M. F. Pasetti, and M. B. Sztein. 2002. Characterization of CD8(+) effector T cell responses in volunteers immunized with Salmonella enterica serovar Typhi strain Ty21a typhoid vaccine. J. Immunol. 169:2196-2203. [DOI] [PubMed] [Google Scholar]
- 36.Salerno-Goncalves, R., T. L. Wyant, M. F. Pasetti, M. Fernandez-Vina, C. O. Tacket, M. M. Levine, and M. B. Sztein. 2003. Concomitant induction of CD4(+) and CD8(+) T cell responses in volunteers immunized with Salmonella enterica serovar Typhi strain CVD 908-htrA. J. Immunol. 170:2734-2741. [DOI] [PubMed] [Google Scholar]
- 37.Sallusto, F., D. Lenig, R. Forster, M. Lipp, and A. Lanzavecchia. 1999. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401:708-712. [DOI] [PubMed] [Google Scholar]
- 38.Seth, A., L. J. Stern, T. H. Ottenhoff, I. Engel, M. J. Owen, J. R. Lamb, R. D. Klausner, and D. C. Wiley. 1994. Binary and ternary complexes between T-cell receptor, class II MHC and superantigen in vitro. Nature 369:324-327. [DOI] [PubMed] [Google Scholar]
- 39.Shaw, S. K., and M. B. Brenner. 1995. The beta 7 integrins in mucosal homing and retention. Semin. Immunol. 7:335-342. [DOI] [PubMed] [Google Scholar]
- 40.Soroosh, P., F. Shokri, M. Azizi, and M. Jeddi-Tehrani. 2003. Analysis of T-cell receptor beta chain variable gene segment usage in healthy adult responders and nonresponders to recombinant hepatitis B vaccine. Scand. J. Immunol. 57:423-431. [DOI] [PubMed] [Google Scholar]
- 41.Sourdive, D. J., K. Murali-Krishna, J. D. Altman, A. J. Zajac, J. K. Whitmire, C. Pannetier, P. Kourilsky, B. Evavold, A. Sette, and R. Ahmed. 1998. Conserved T cell receptor repertoire in primary and memory CD8 T cell responses to an acute viral infection. J. Exp. Med. 188:71-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Stewart-Jones, G. B., A. J. McMichael, J. I. Bell, D. I. Stuart, and E. Y. Jones. 2003. A structural basis for immunodominant human T cell receptor recognition. Nat. Immunol. 4:657-663. [DOI] [PubMed] [Google Scholar]
- 43.Svensson, M., J. Marsal, A. Ericsson, L. Carramolino, T. Broden, G. Marquez, and W. W. Agace. 2002. CCL25 mediates the localization of recently activated CD8alphabeta(+) lymphocytes to the small-intestinal mucosa. J. Clin. Investig. 110:1113-1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sztein, M. B., M. K. Tanner, Y. Polotsky, J. M. Orenstein, and M. M. Levine. 1995. Cytotoxic T lymphocytes after oral immunization with attenuated vaccine strains of Salmonella typhi in humans. J. Immunol. 155:3987-3993. [PubMed] [Google Scholar]
- 45.Sztein, M. B., S. S. Wasserman, C. O. Tacket, R. Edelman, D. Hone, A. A. Lindberg, and M. M. Levine. 1994. Cytokine production patterns and lymphoproliferative responses in volunteers orally immunized with attenuated vaccine strains of Salmonella typhi. J. Infect. Dis. 170:1508-1517. [DOI] [PubMed] [Google Scholar]
- 46.Tomai, M. A., J. A. Aelion, M. E. Dockter, G. Majumdar, D. G. Spinella, and M. Kotb. 1991. T cell receptor V gene usage by human T cells stimulated with the superantigen streptococcal M protein. J. Exp. Med. 174:285-288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.van den Beemd, R., P. P. Boor, E. G. van Lochem, W. C. Hop, A. W. Langerak, I. L. Wolvers-Tettero, H. Hooijkaas, and J. J. van Dongen. 2000. Flow cytometric analysis of the Vbeta repertoire in healthy controls. Cytometry 40:336-345. [DOI] [PubMed] [Google Scholar]
- 48.van den Elsen, P. J., and A. Rudensky. 2004. Antigen processing and recognition. Recent developments. Curr. Opin. Immunol. 16:63-66. [Google Scholar]
- 49.Van Kerckhove, C., G. J. Russell, K. Deusch, K. Reich, A. K. Bhan, H. DerSimonian, and M. B. Brenner. 1992. Oligoclonality of human intestinal intraepithelial T cells. J. Exp. Med. 175:57-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Waterman, S. R., and D. W. Holden. 2003. Functions and effectors of the Salmonella pathogenicity island 2 type III secretion system. Cell. Microbiol. 5:501-511. [DOI] [PubMed] [Google Scholar]
- 51.Wick, M. J. 2002. The role of dendritic cells during Salmonella infection. Curr. Opin. Immunol. 14:437-443. [DOI] [PubMed] [Google Scholar]
- 52.Wick, M. J. 2003. The role of dendritic cells in the immune response to Salmonella. Immunol. Lett. 85:99-102. [DOI] [PubMed] [Google Scholar]
- 53.Zissel, G., I. Baumer, B. Fleischer, M. Schlaak, and J. Muller-Quernheim. 1997. TCR V beta families in T cell clones from sarcoid lung parenchyma, BAL, and blood. Am. J. Respir. Crit. Care Med. 156:1593-1600. [DOI] [PubMed] [Google Scholar]