Abstract
Odontogenic myxomas (OMs) represent asymptomatic, slowly expanding gnathic lesions with aggressive biological behaviour. Though the spectrum of OMs remains classical with multilocular radiolucency and presentation of stellate-shaped cells embedded in a mucoid stroma, they may mimic many other lesions radiographically or histopathologically. We hereby discuss a case of OM in a 28-year-old woman with special emphasis on pathogenesis and differential diagnosis.
Keywords: Mesenchymal, myxoma, odontogenic, tennis racquet appearance
INTRODUCTION
Thomas and Goldman (1947) first described odontogenic myxomas (OMs), which account for nearly 3–6% of all odontogenic tumours.[1] The international histological classification of odontogenic tumours describes OM as a benign odontogenic tumour of mesenchymal origin that is locally invasive and consists of rounded and angular cells that lie in the abundant mucoid stroma.[2] OMs predominantly appear between 20 and 40 years of age with a predilection in premolar–molar region and preferentially in females.[1] It is generally depicted as a slow-growing tumour with the potential to attain considerable size without noticeable signs and symptoms.[2]
Odontogenic ectomesenchyme of a developing tooth and undifferentiated mesenchymal cells of the periodontal ligament are the most probable sources of origin of OMs. The odontogenic origin of this neoplasm is supported by its histological similarity to pulp ectomesenchyme along with its exclusive occurrence in close proximity to the tooth-bearing parts of the jaws.[1,2]
Most lesions of OMs are separated by straight septa forming square, rectangular or triangular spaces and show a ‘honeycomb’, ‘soap bubble’ or ‘tennis racquet’ appearance radiographically.[1,2] The pathologists prefer to diagnose the lesion as OM because of its myxoid change under the microscope; however, its histopathology needs to be differentiated from various benign lesions with a myxoid change including the dental papilla of a developing tooth.[1,3] Since histology holds the gold standard for diagnosis, here we present a case of OM with remarkably characteristic clinical and histopathological features giving an insight into the pathogenesis of myxomas.
CASE REPORT
A 28-year-old woman reported to the Department of Oral Medicine and Radiology with a chief complaint of extraoral swelling on the left lower side of the face for 2 years. The patient was relatively asymptomatic 2 years back when she started noticing the swelling, which was small initially and gradually increased to attain the present size. It was not associated with pain or pus discharge. On inspection extra-orally, a diffuse, solitary swelling was noticed in the left lower one-third of the facial region approximately 3x5 cm in dimensions extending antero-posteriorly from the parasymphyseal region to 2 cm ahead of the angle of the mandible and supero-inferiorly the swelling extended from 2 cm below from the corner of the lip to lower border of the mandible. The swelling appeared round to oval in shape [Figure 1a]. Upon palpation, the swelling appeared diffuse, firm to hard, non-tender, non-compressible, non-reducible and non-fluctuant. The temperature of the swelling was not raised, and lymph nodes were non-palpable. Intra-orally, the swelling appeared to extend from the mesial surface of 33 to the mesial surface of 36. It measured approximately 2x3 cm in size with well-defined margins and colour the same as that of the surrounding mucosa [Figure 1b]. There was also grade 1 mobility irt 35, 36 and 37 and the teeth were vital.
Figure 1.
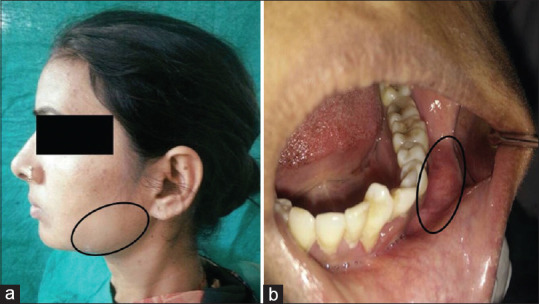
Extraoral (a) and intra-oral (b) clinical presentation
Orthopantomographic evaluation revealed a well-defined multilocular osteolytic radiolucent lesion with evidence of scalloping in a few areas anteriorly and irregular margins in general. The lesion extended antero-posteriorly from the root apex of 34, till 38, involving the body and ramus of the mandible, whereas supero-inferiorly it extended from apical 1/3rd of the root of 36 and 37 till 1.5 cm towards the lower border of the mandible measuring approximately 3x5 cm in size [Figure 2a].
Figure 2.

Orthopantomogram (a) and CT scan (b)
Upon cone beam computed tomography (CBCT) analysis (using NewTom GiANO with selected field of view (FOV) of 11*8 cm at 90 kv, 12.9 mAs and exposure time of 3.6 seconds), a radiolucent lesion measuring approx. 71.1 mm antero-posteriorly, 23 mm medio-laterally and 50.7 mm supero-inferiorly in the left posterior body of the mandible and left ramus region at the level of crestal–middle–apical thirds of the alveolus and basal mandible-ramus body; extending from distal aspect of 34 till the posterior border of the ramus and supero-inferiorly extending up to the sigmoid notch and abutting the infero-posterior thirds of the left coronoid process was noticed. The internal structure of the lesion appeared radiolucent and multilocular with multiple, fine, thin, straight septae arranged in a reticular pattern with few septae intersecting with each other. There was evidence of scalloping, thinning, effacement and expansion of the bucco-lingual cortices at the level of cervical–middle–apical thirds of the alveolus and basal mandible and ramus. Spiking root resorption irt 35, 36 and 37 and knife edge root resorption irt 38 were also evident. The inferior alveolar canal could not be appreciated within the lesion [Figure 2b].
Upon the clinicoradiological investigation, a differential diagnosis of OM, central giant cell granuloma, ameloblastoma and osteogenic sarcomas was considered. Ruling out the other possibilities, the provisional diagnosis of OM was considered [Table 1]. An incisional biopsy was performed, and multiple soft tissue bits were received. The tissues were processed for histopathological evaluation, and the submitted hematoxylin and eosin (H&E)-stained sections revealed round, spindle-shaped or angular cells, which were evenly spaced within the fibrillar matrix composed of loose, abundantly myxoid connective tissue stroma, closely resembling the mesenchymal portion of a developing tooth. Some areas showed moderately dense collagen fibres. A few strands of inactive odontogenic epithelium were also seen within the connective tissue stroma accompanied by uniformly dispersed mast cells. Overall histological appearance of the lesion revealed more amount of myxoid stroma in a sparsely fibrous and acellular background. The diagnosis of myxoma was given. Based on the above findings, an excisional biopsy was performed and one hemimandibulectomy specimen extending from the midline to the left condylar area with associated teeth and soft tissues was received [Figure 3a]. Tissues were harvested from multiple sites [Figure 3b] for histopathologic evaluation, and the submitted H&E-stained sections revealed abundant loose mucoid stroma containing rounded, spindle-shaped or angular cells, which were evenly spaced within the fibrillar matrix. Focal areas showed delicate fibrous component and nests of odontogenic rests. A few mild chronic inflammatory cells and delicate capillaries lined by endothelial cells were also evident [Figure 3c and d].
Table 1.
Clinicoradiological differential diagnosis of odontogenic myxoma
| Odontogenic myxoma | Ameloblastoma | Central giant cell granuloma | Osteogenic sarcoma | |
|---|---|---|---|---|
| Clinical features | Grow slowly✓ Overlying mucosa is pale pink✓ Female. > male✓ 10–30 years of age✓ | Aggressive in nature✗ Male > female✗ 4th to 6th decades of life✗ | Painless swelling✓ Overlying mucosa has reddish-purple colour✗ Female > male✓ Younger than 20 years✗ | Swelling is usually rapid ✗ Erythema of overlying mucosa✗ Male > female✗ 4th decade of life✗ |
| Radiographic finding/location | Mand: max—3:1✓ Between premolar and molar region✓ Rarely in the ramus and condyle✗ | Mand: max—5 : 1✓ Molar, ramus region most common✗ | More predilection for mandible✓ Anterior to molar in mandibular lesion✗ | More predilection for mandible✓ Posterior body including the tooth-bearing region, angle, ramus✗ |
| Effect on surrounding structure | Spiking root resorption✓ Knife edge root resorption✓ | Knife edge root resorption✓ | Resorption of tooth roots is not a constant feature✗ | Widening of the periodontal membrane✓ |
| Periphery | Usually is well defined May be ill-defined✓ May not have corticated margin✓ | Well-defined frequently delineated by cortical margins✗ | Well-defined radiographic margin✓ Periphery shows no evidence of cortication✓ | Ill-defined border✓ ‘Hair-on-end’ trabeculae✗ |
| Internal structure | Sharper and straighter thin etched septa✓ Finding one or two of these straight septa will help in the identification✓ | Coarse, curved, well-defined trabeculae✗ubble✗ect✗ Honeycomb appearance✗ | Wispy, ill-defined trabeculae✗ Some are at right angles to the periphery✓ | Granular sclerotic-appearing bone✗ Cotton balls, wisps or honeycombed✗ |
Figure 3.

Hemimandibulectomy specimen (a), specimens collected from different sites (b), loose fibrillar mucoid stroma with stellate cells and few capillaries (H&E stain; 4x, 40x) (c and d)
The tissue was then subjected to Alcian blue [Figure 4a and b] and periodic acid schiff (PAS) staining [Figure 4c and d] to confirm the nature of the ground substance, that is, hyaluronic acid and chondroitin sulphate. The positivity for the same in OMs and lesions, which also show myxomatous changes such as myxofibroma, chondromyxoid fibroma, myxoid neurofibroma and myxoid sarcomas, however, is also significant. The lesions were then ruled out based on their histopathology [Table 2] and through confirmation of immunohistochemical staining for alpha-smooth muscle actin (SMA) [Figure 5a and b] and S100 [Figure 5c and d] for myxofibroma and myxoid neurofibroma, respectively, as the staining was negative, thereby confirming a true myxomatous origin of the lesion. A final diagnosis of OM was then given, and the patient was kept under regular follow-up to check recurrences, if any, for 6 months. The recovery was uneventful.
Figure 4.

Alcian blue stain in control and case (a and b); PAS stain in control and case (c and d)
Table 2.
Histopathological differential diagnosis of odontogenic myxoma
| Lesions | Features | Possibility in diagnosis |
|---|---|---|
| Myxofibroma | Shows cellular pleomorphism along with the presence of binucleate, bizarre and multinucleated cells | Absent in case of odontogenic myxomas |
| Chondromyxoid fibroma | Represents a well-circumscribed tumour consisting of nodular myxoid tissue with scattered giant cells and areas of cartilaginous differentiation | Absent in case of odontogenic myxomas |
| Myxoid neurofibroma | Shows the presence of long wavy nuclei and positivity for S100 protein with the abundance of mast cells. | Absent in case of odontogenic myxomas |
| Myxoid sarcomas | Shows an abundance of atypical cells | Absent in case of odontogenic myxomas |
Figure 5.

Alpha-SMA in control and case (a and b); S100 in control and case (c and d)
DISCUSSION
OM represents less than 10% of all odontogenic tumours and is considered locally invasive, slow-growing, with well-defined pathologic characteristics and generally associated with tooth germ.[4] OM can occur at any age from childhood to the elderly, and mandibular lesions appear to cross the midline.[2] Patients afflicted with an OM generally notice a painless, slowly enlarging expansion of the jaw with possible tooth loosening or displacement,[1] which was also noted in our case. Displacement of teeth is relatively common, though root resorption is less frequently seen. It is reported that involved teeth could be mobile but are usually vital.[2] Similar findings were noted in our case.
OMs can be extensive including the ramus and the condyle. The cortex of bone may appear thinned due to expansion and may perforate at a later stage of tumour progression. The internal structure of OM is described as either a unilocular or an expansile multilocular radiolucency. Multilocular lesions with fine and/or rough trabeculations are described as ‘tennis racket’, ‘soap bubble’ or ‘honeycomb’ to depict the internal structure.[2]
Microscopic examination in OMs shows coarse well-defined peripheral septae, which are reoriented residual lamellar cortical bone. However, most of the delicate internal septa could be dense fibrous partitions that divided the OM into myxomatous lobules.[2] Histologically, myxoma is largely an unencapsulated tumour composed of spindled- or stellate-shaped cells in a mucoid-rich intercellular matrix composed of glycosaminoglycan (GAG) and rich in hyaluronic acid with mitotic figures. The stroma contains a small amount of collagen fibrils; however, some cases contain a larger amount of collagen bundles and are thus referred to as myxofibroma or fibromyxoma.[5]
The tumour also has connective tissue fibres, bony trabeculae, irregular calcifications, scant blood vessels and sparse capillaries. Nests of odontogenic epithelium are occasionally seen but are not essential for diagnosis. The presence of mast cells may also be reported.[6] All of these classical features were also seen in our case. Immunohistochemistry studies of OMs are thought to be of little value in differentiating these lesions from other nonodontogenic cell tumours; however, they may aid to differentiate true myxomas from lesions with myxomatous changes as was helpful in our case.
OM is considered locally aggressive and shows infiltration in adjacent tissues, which could possibly be due to its gelatinous nature and absence of a capsule.[4] It has a hypocellular matrix and mucoid ground substance of GAGs and chondroitin sulphate, which accounts for its rapid growth.[7] Also, it is now well known that the extracellular matrix (ECM) molecules produced by tumour cells may play important roles in modulating the growth and differentiation of the tumour cells and then influence the biological behaviour of tumours. Furthermore, neoplasms may contain varying mucosubstances depending on the tissue of origin and differentiation.[8] It is therefore speculated that knowledge of characteristics of GAGs and proteoglycans (PGs) in the mucosubstances of these myxoid tumours might shed light on their histogenesis, morphological formation, biological behaviour and differential diagnosis.[9]
Previous histochemical studies have revealed that OM contained hyaluronic acid (HA) and chondroitin sulfate (CS) [Chondroitin Sulfate-4 (CS-4) and/or CS-6], with HA as the main component.[9] In tooth germs, the intense expression of proteoglycan-M (PG-M) or versican, which is expressed as CS-4 and/or CS-6, was restricted to the dental papilla, strongly supporting the origin of this tumour from dental papilla rather than dental sac.[9] However, a biochemical study by Slootweg and colleagues concluded that the GAG content of OM is much higher than that reported for other soft dental tissues, including dental pulp, gingiva and periodontal ligament, thus arguing against the odontogenic origin of this tumour.[10] High recurrences may also be attributed to tumour location such as invasion through cancellous bone[7] and inadequate removal.[4]
Positive staining of the mucoid-rich ECM for Alcian blue and periodic acid–Schiff has been well reported. Tumour cells show uniform positivity on immunostaining with vimentin and patchy staining with smooth muscle actin, although they were negative for desmin, neuron-specific enolase, glial fibrillary acid protein, neurofilaments and S100.[5] Similar staining patterns were observed in our case.
The molecular pathogenesis of OMs is still poorly understood, and its clonal nature is still controversial. Protein kinase cyclic adenosine monophosphate (cAMP)-dependent type I regulatory subunit alpha (PRKAR1A) mutations have been described in sporadic and Carney syndrome cardiac myxomas, and such genetic alterations have been reported in a low proportion of OMs. Guanine nucleotide binding protein (GNAS1) mutations were previously reported in intramuscular myxomas but could not be detected in OMs. In the epigenetics field, hypomethylation of the genes cyclin dependent kinase inhibitor (P27), tumor prote (TP53) and Retinoblastoma1 (RB1) has been reported.[11]
Small myxomas are generally treated by curettage, but careful follow-up is necessary for at least 5 years. Larger lesions need extensive resection as myxomas tend to infiltrate the surrounding bone, and hence, complete removal of a large tumour by curettage is often difficult to achieve. The advocated treatment of myxoma is surgical resection with a minimum of 1 cm of bone margin and one tumour-free anatomic barrier because of its high recurrence rate. Liquid nitrogen cryosurgery may limit recurrence because of its ability to devitalise the organic content while leaving the inorganic framework intact.[7] The overall recurrence rate is approximately 25% and usually occurs during the first 2 years after removal. In this case, resection of the affected mandible and application of Carnoy’s solution on the edge of the remaining bone were carried out.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
CONCLUSION
OMs abundantly rich in ECM molecules such as hyaluronic acid and chondroitin sulphate show infiltration within marrow spaces, leading to displacement and resorption of teeth, bone expansion and perforation, facial deformation and even patient morbidity. These probably arise due to developmental replication errors and harbouring a quiet genome. Multiple cases have shown recurrences despite an aggressive treatment approach. Every case of OM therefore requires a prompt and early diagnosis and a correct surgical intervention to avoid recurrences and patient morbidity.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Wang K, Guo W, You M, Liu L, Tang B, Zheng G. Characteristic features ofthe odontogenic myxoma on cone beam computed tomography. Dentomaxillofac Radiol. 2017;46:20160232. doi: 10.1259/dmfr.20160232. doi:10.1259/dmfr.20160232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Noffke CE, Raubenheimer EJ, Chabikuli NJ, Bouckaert MM. Odontogenic myxoma:Review ofthe literature and report of 30 cases from South Africa. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:101–9. doi: 10.1016/j.tripleo.2007.01.026. [DOI] [PubMed] [Google Scholar]
- 3.Titinchi F, Hassan BA, Morkel JA, Nortje C. Odontogenic myxoma:A clinicopathological study in a South African population. J Oral Pathol Med. 2016;45:599–604. doi: 10.1111/jop.12421. [DOI] [PubMed] [Google Scholar]
- 4.Francisco AL, Chulam TC, Silva FO, Ribeiro DG, Pinto CA, Gondak RO, et al. Clinicopathologic analysis of 14 cases of odontogenic myxoma and review of the literature. J Clin Exp Dent. 2017;9:e560–3. doi: 10.4317/jced.52953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rowland A, Benjamin F, Athanasius-Chukwudi O, Uchenna-Kevin O, Modupeola-Omotara S. Central Myxoma/Myxofibroma of the jaws:A clinico-epidemiologic review. Iran J Otorhinolaryngol. 2017;29:35–42. [PMC free article] [PubMed] [Google Scholar]
- 6.Shivashankara C, Nidoni M, Patil S, Shashikala KT. Odontogenic myxoma:A review with report of an uncommon case with recurrence in the mandible of a teenage male. Saudi Dent J. 2017;29:93–101. doi: 10.1016/j.sdentj.2017.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kumar N, Kohli M, Pandey S, Agarwal P. Odontogenic myxoma. J Maxillofac Oral Surg. 2014;13:222–6. doi: 10.1007/s12663-010-0107-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kindblom LG, Angervall L. Histochemical characterization of mucosubstances in bone and soft tissue tumors. Cancer. 1975;36:985–94. doi: 10.1002/1097-0142(197509)36:3<985::aid-cncr2820360322>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 9.Zhao M, Lu Y, Takata T, Ogawa I, Miyauchi M, Mock D, et al. Immunohistochemical and histochemical characterization of the mucosubstances of odontogenicmyxoma:Histogenesis and differential diagnosis. Pathol Res Pract. 1999;195:391–7. doi: 10.1016/S0344-0338(99)80012-0. [DOI] [PubMed] [Google Scholar]
- 10.Slootweg PJ, van den Bos T, Straks W. Glycosaminoglycans in myxoma of the jaw:A biochemical study. J Oral Pathol. 1985;14:299–306. doi: 10.1111/j.1600-0714.1985.tb00497.x. [DOI] [PubMed] [Google Scholar]
- 11.Santos JN, Sousa Neto ES, França JA, Diniz MG, Moreira RG, Castro WH, et al. Next-generation sequencing of oncogenes and tumor suppressor genes in odontogenic myxomas. J Oral Pathol Med. 2017;46:1036–9. doi: 10.1111/jop.12598. [DOI] [PubMed] [Google Scholar]


