Abstract
Context: Peripheral axon injury and degeneration are often mediated by oxidative stress and inflammation. The hydroalcoholic extract of the red propolis (HERP) has attracted great attention because of its antioxidant and anti-inflammatory activities.
Objective: The objective of this work is to study the effect of HERP on nerve repair and functional recovery after sciatic nerve injury (SNI) in rats.
Materials and methods: The chemical markers in HERP were identified using high-resolution mass spectroscopy. After axonotmesis of sciatic nerve, ibuprofen (IBP) and HERP treatments were orally administered for 28 d. Behavioural tests were performed weekly after SNI. The myelinated axon number was counted using morphometric analysis.
Results: The compounds found in HERP were pinocembrin, formononetin, vestitol, and biochanin A. The animals that underwent SNI showed a significant decrease in motor function based on the Basso, Beattie and Bresnahan scale and sciatic functional index compared with sham animals until 7 d after the surgery (p < 0.05). After 14 and 21 d, the SNI groups treated with either HERP or IBP showed significant improvement (p < 0.01), and the SNI group treated with HERP 10 mg/kg showed accelerated motor recovery compared with the other groups (p < 0.01). SNI caused also a reduction in the myelinated axon counts, and treatment with HERP 10 mg/kg induced a significant increase in the number of myelinated fibres compared with all other groups.
Conclusion: HERP promoted regenerative responses and accelerated functional recovery after sciatic nerve crush. Thus, it can be considered to be a new strategy or complementary therapy for treating nerve injuries.
Keywords: Axonotmesis, crush, high-resolution mass spectroscopy, isoflavones, nerve regeneration, sciatic functional index
Introduction
Peripheral nerve injury (PNI) is a very common neurological problem (Bobinski et al. 2011). Because the current array of anti-inflammatory drugs used in PNI exhibits a wide spectrum of adverse effects and/or ineffectiveness, the investigation of new alternatives is ongoing (Ortega-Álvaro et al. 2012).
During the last decade, studies that used natural products showed positive results with regard to the improvement of neuroregeneration after PNI (Jang et al. 2012). For example, it was demonstrated that the epimedium extract facilitated the regeneration of peripheral nerves and coordinated their growth (Kou et al. 2013). Furthermore, the Butea monosperma (L.) Taub. (Fabaceae) extract promoted behavioural, biochemical, and histological changes in animals with sciatic nerve injuries. These results were mainly attributed to the antioxidant and anti-inflammatory properties of the extracts (Thiagarajan et al. 2002). Thus, the use of natural products as pharmacological agents appears to be a promising approach for modulating local inflammation and avoiding secondary damage, which may improve the regeneration process (Tamaddonfard et al. 2013).
Propolis, also termed bee glue, is an important bee product with great potential as a medicine because of its numerous pharmacological activities (Cinegaglia et al. 2012). The red propolis is a new Brazilian variety, which is rich in antioxidant compounds including phenolics, flavonoids, naphthoquinones, and others (Righi et al. 2011; Morsy et al. 2013). Its botanical origin was described to be Dalbergia ecastophyllum (L) Taub. (Fabaceae) (López et al. 2014). Extracts from the red propolis have been shown to demonstrate several biological activities including cytotoxic activity against tumour cells (Awale et al. 2008) and antimicrobial and antioxidant properties (Righi et al. 2011). Recently, our group demonstrated that the hydroethanolic extract of the red propolis (HERP) exhibited healing (Albuquerque-Júnior et al. 2009; Almeida et al. 2013; Souza et al. 2013) and antitumoural (Frozza et al. 2013) activities.
Based on its chemical composition, and considering its activities as a potent anti-inflammatory and antioxidant compound (Tufek et al. 2013), we hypothesized that the administration of HERP could improve the neuroregeneration process after sciatic nerve constriction injury. Thus, the aims of this study were to evaluate the functional recovery, inflammatory profile, and morphological changes in nerve tissue after sciatic nerve injury (SNI) in rats treated with HERP.
Materials and methods
Hydroalcoholic extract of red propolis
Propolis was collected at Brejo Grande/Sergipe/Brazil (10°28′25″S and 36°26′12 ″W) in October 2011. The extraction was performed using propolis samples (1 g) and 70% ethanol (12.5 mL) at room temperature for 1 h in an ultrasound bath. After extraction, the mixture was filtered, and the solvent was evaporated to produce HERP.
High-resolution mass spectrometry (HRMS)
The dry extract was dissolved in a solution of 50% (v/v) chromatographic grade acetonitrile (Tedia, Fairfield, OH), 50% (v/v) deionized water, and 0.1% ammonia hydroxide for negative-ion mode electrospray ionization mass spectrometry [ESI(−)-MS]. The solution was infused directly into the ESI source by means of a syringe pump (Harvard Apparatus, Warner Instruments, Hamden, CT) at a flow rate of 10 μL min−1. ESI(–)-MS and tandem were acquired using a hybrid high-resolution and high accuracy (5 μL/L) microTOF-QII (Q-TOF) mass spectrometer (Bruker Scientific, Billerica, MA). Capillary and cone voltages were set to +3500 V and +40 V, respectively, with a de-solvation temperature of 100 °C. Diagnostic ions in the crude extract were characterized by comparison of their exact mass (m/z) with compounds identified in previous studies. Hystar software (Bruker Scientific, Billerica, MA) was used for data acquisition and processing. The data were collected in the m/z range of 70–900 at a speed of two scans per second, providing a resolution of 50 000 (FWHM) at m/z 200. No relevant ions were observed below m/z 120 or above m/z 500.
Biological assay
Animals
Male Wistar rats weighing 200–300 g were kept in a temperature-controlled room (23 °C ± 2 °C), with a 12 h light/dark cycle. Food and water were provided ad libitum. The experiments were conducted according to the Ethical Principles in Animal Research adopted by the Brazilian College of Animal Experimentation. This study was approved by the local ethics committee for animal research under the protocol number 011213.
SNI
Rats were anesthetized with a mixture of xylazine (14 mg/kg) and ketamine (100 mg/kg) administered intraperitoneally. For the SNI, a 2 cm surgical incision was made, the left sciatic nerve was exposed, and four clampings of 20 s each were made at 5 mm before anatomical bifurcation with a haemostatic clamp. In the sham group, the left nerve was exposed without nerve crush. Subsequently, the incision was sutured.
Experimental groups
A total of 36 rats were used in the study. The animals were randomly divided into six groups (n = 6 per group). The control groups were sham operated and received vehicle (2% Tween 80 in saline solution; Sham/vehicle) or the highest dosage of HERP (10 mg/kg; Sham/HERP10). The groups with lesions were orally treated with vehicle (SNI/vehicle), HERP 1 mg/kg (SNI/HERP1), HERP 10 mg/kg (SNI/HERP10), or ibuprofen 60 mg/kg (SNI/IBP). All animals received their respective treatments once a day for 28 consecutive days.
Behavioural evaluation
All tests were performed on the 1st, 7th, 14th, 21st, and 28th days. A baseline performance assessment was taken 1 d before surgeries. All experiments were performed using blind methodology.
Basso, Beattie, and Bresnahan (BBB) test
Functional recovery was assessed using the BBB locomotor rating scale (Basso et al. 1995). Scores of 0 and 21 were given when there were no spontaneous movements and normal movements, respectively. A score of 20 represents full weight support and complete limb coordination, whereas zero represents the absence of movements. The animals were observed and assessed over a 6 min exposure to an open area (transparent box 70 cm × 40 cm, height 20 cm). BBB scores were recorded once before the surgery (pre-test) to establish a baseline control and weekly after the surgery.
Sciatic function index (SFI)
The walking track analysis was performed weekly after surgery to evaluate motor function recovery after treatments. Pre-operatively, the rats were trained to walk down a wooden track (70 cm × 10 cm) into a darkened goal box. The rats’ hind paws were painted with non-toxic finger paint, and any changes in their paw prints that resulted from nerve injury were recorded. The recordings continued until six measurable footprints were collected. A formula used for SFI (Bain et al. 1989) considered the print length as the distance from the heel to the top of the third toe, intermediary toe spread as the distance from the second toe to the fourth toe, and toe spread as the distance between the first toe and the fifth toe, from the operated and non-operated foot. The SFI is an indicator of the degree of nerve dysfunction and varies from 0 to −100 with 0 corresponding to normal function and −100 corresponding to complete dysfunction.
Beam walk performance
The functional test was assessed using a beam walk task where the objective was to record the animals’ walking ability on narrow wooden beams (from 7.7 to 1.7 cm wide). Briefly, the task required the rats to traverse a narrow wooden beam (70 cm in length). The scoring criteria for distance travelled were based on a rating scale from 0 to 12, where 0 indicates an inability to ambulate beyond the 7.7 cm wide wooden beam, 1–5 presents uncoordinated ambulation on the 7.7 cm wide wooden beam and 6–12 indicates traversal of the entire length of progressively narrower wooden beams (from 7.7 to 1.7 cm wide) and entrance into the goal box. Rats were trained prior to injury to perform the tasks without errors.
Withdrawal reflex
Each animal was gently placed on a non-slippery surface, and the interdigital folds of the hind limbs were pressed around the first rack using a Halsted haemostat. The data were recorded when the animal displayed pain, withdrew the paw, made noise, or bit (Von Euler et al. 1996).
Histological evaluation
On day 28 after SNI, the rats were euthanized in a CO2 chamber. The distal segment of the sciatic nerve (2 cm before anatomical bifurcation, pieces of 10 mm) was removed, formalin-fixed, and paraffin-embedded. Histological sections (5 μm) were cut using a microtome, stained with haematoxylin and eosin (HE), and then examined under light microscopy. Nerve sections were analysed qualitatively and quantitatively (percent of preserved area around epineurium, mean number of myelinated fibres in the transversal area [250 μm2], and inflammatory profile [graded as 0 for absent, 1 for mild, 2 for moderate, or 3 for severe]). At least four histological fields in close proximity to the lesion epicenter were selected in each nerve for analysis. Image analysis was performed using a microscope (Nikon, Tokyo, Japan) equipped with a 10 × (for measuring total nerve areas) and 40 × (for axon number counting) objective coupled to a photographic camera (Nikon, Tokyo, Japan) and connected to a computer system. The software Axovision (ZEISS, Monroeville, PA) was used to collect the measurements.
Statistics
All data are expressed as the mean ± standard error of mean. Non-Gaussian data (scores) were analyzed using the Kruskal–Wallis test followed by Dunn’s multiple comparison tests. All statistical analyses were performed using SPSS version 19.0 (SPSS Inc., Chicago, IL). Values of p < 0.05 were considered to represent statistically significant differences.
Results
Chemical composition by HRMS
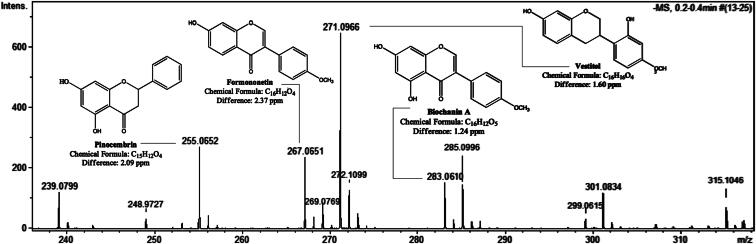
The chemical composition was evaluated by direct infusion-based HRMS. The analysis in the negative-ion mode, using ammonia hydroxide 0.1% in a solution of water:acetonitrile (1:1), was effective for compounds in the crude extract. The region between m/z 230–320, where the major markers were found, is shown in Figure 1. The chemical structures, elemental formulas, and the differences in parts per million (ppm) to exact mass are exhibited in Figure 1. In comparison with results from previously published studies (Righi et al. 2011; Frozza et al. 2013; López et al. 2014), pinocembrin (m/z 255.0652, diff. 2.09 ppm), formononetin (m/z 267.0651, diff. 2.37 ppm), vestitol (m/z 271.0966, diff. 1.60 ppm), and biochanin A (m/z 283.0610, diff. 1.24 ppm) were confirmed in HERP. Because of the widely accepted accuracy threshold for confirmation of elemental compositions, 5 ppm (Lacorte and Fernandez-Alba 2006), this analysis has been associated with highly reliable identification of the target compounds.
Figure 1.
High-resolution mass spectrometry (HRMS) of HERP.
Behavioural evaluation
BBB test
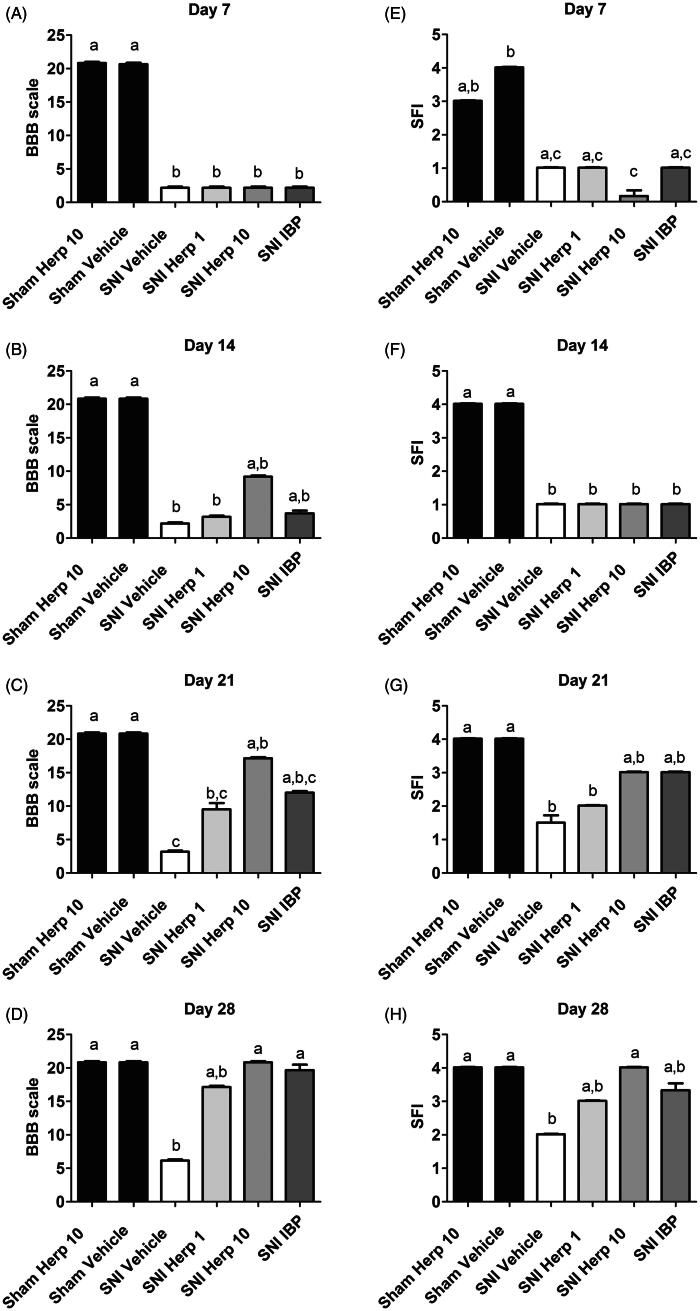
All the animals that underwent SNI showed a significant decrease in motor function when compared with the sham animals 7 d after surgery (p < 0.05; Figure 2A). After 14 d, the SNI/HERP10 and the SNI/IBP groups, despite displaying similar results when compared with the SNI/vehicle group, showed results akin to those found in the sham groups, with gradual improvement in behavioural parameters (Figure 2B). After 21 d, we observed that the SNI/HERP10 group demonstrated a significant increase in the BBB score (p < 0.05) when compared with the SNI/vehicle group (Figure 2C). On day 28 after SNI, both HERP10 and IBP resulted in increased scores statistically on par with those observed in the sham groups (p > 0.05; Figure 2D).
Figure 2.
BBB score: day 7 (A), day 14 (B), day 21 (C), and day 28 (D) after surgery (p <0.05). SFI score: day 7 (E); day 14 (F); day 21 (G), and day 28 (H) after surgery (p <0.05) (SNI, sciatic nerve injury; IBP60, ibuprofen 60 mg/kg treatment; HERP 1, 1 mg/kg treatment; HERP10, 10 mg/kg treatment).
SFI
In the SFI test performed on day 7 after the surgery (Figure 2E), the SNI groups showed a significant reduction in values compared with the sham/vehicle group (p < 0.05). Despite the statistically similar results between the sham/HERP10 group and the sham/vehicle group (p > 0.05), no behavioural differences were detected when compared with the SNI groups with the exception of SNI/HERP10. On day 14 after the surgery (Figure 2F), all SNI groups showed the same results (p > 0.05), which were statistically different from those observed in the sham groups (p < 0.05). On day 21 after the surgery, the SNI/HERP10 and SNI/IBP groups presented similar results when compared with the sham groups (p > 0.05; Figure 2G). After 28 d, all treated groups (SNI/HERP1, SNI/HERP10, and SNI/IBP) reached values statistically equal to those of the sham groups (p > 0.05). The SNI/HERP10 group was the only treated group that presented a significant difference from the SNI/vehicle group (p < 0.001; Figure 2H).
Beam walk performance
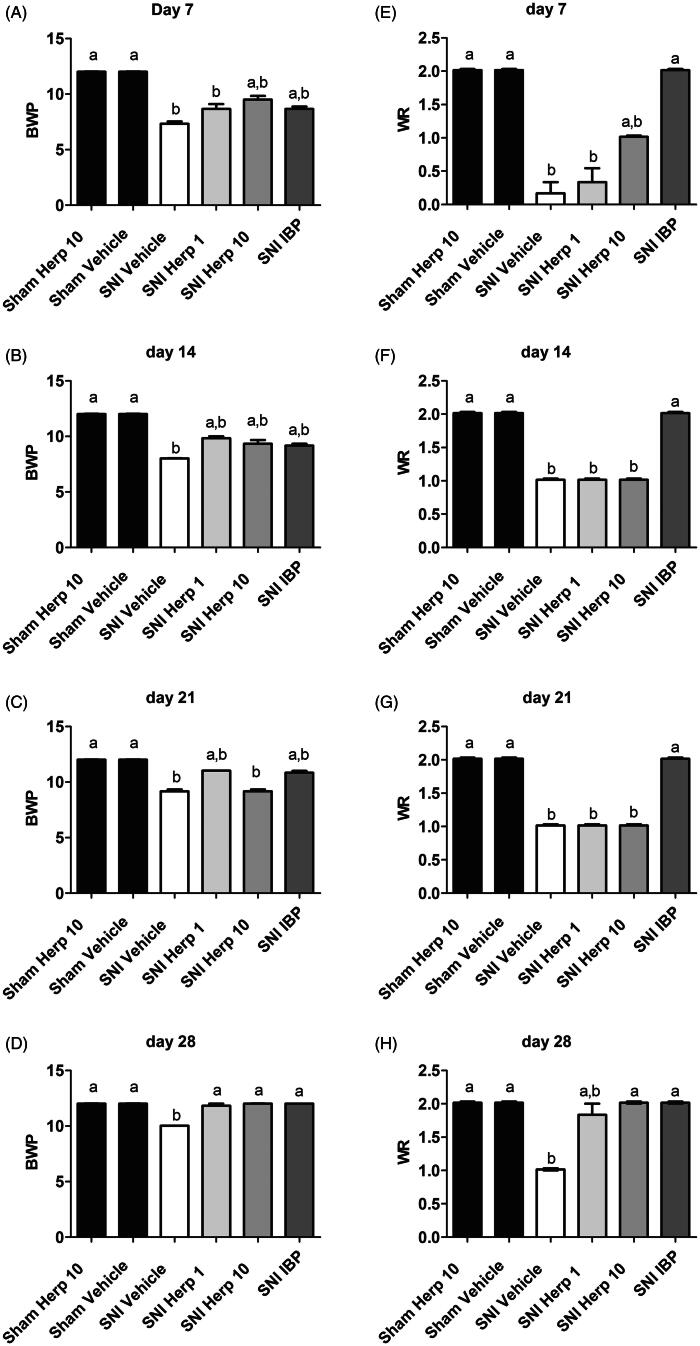
On day 7 after the surgery, we observed that the SNI/HERP10 and SNI/IBP groups presented similar scores in relation to the sham groups (p > 0.05), although they were similar to the SNI/vehicle group scores (p > 0.05; Figure 3A). After 14 d, the SNI/HERP1 group reached the statistical level of performance of the sham groups (p > 0.05; Figure 3B). All treated groups presented their highest scores on day 28 and showed significant differences compared with the SNI vehicle group (p < 0.05; Figure 3D).
Figure 3.
Beam walking test day 7 (A), day 14 (B), day 21 (C), and day 28 (D) after surgery (p <0.05); withdrawal reflex test: day 7 (E), day 14 (F), day 21 (G), and day 28 (H) after surgery (p <0.05) (SNI, sciatic nerve injury; IBP60, ibuprofen 60 mg/kg treatment; HERP 1, 1 mg/kg treatment; HERP10, 10 mg/kg treatment).
Withdrawal reflex
On day 7, the SNI/IBP group showed increased reflexes compared with the SNI/vehicle and SNI/HERP1 groups (p < 0.01), and the SNI/HERP10 group presented results statistically similar to those of the sham groups (Figure 3E). After 14 d and 21 d with the injury, the SNI/vehicle, SNI/HERP1, and SNI/HERP10 groups maintained a statistical difference from the sham groups (p < 0.05; Figure 3F and G). By day 28, all SNI-treated groups showed the same scores as sham animals with the exception of the SNI/vehicle group (p < 0.01; Figure 3H).
The SNI/vehicle group presented an increase on day 14, and the score was maintained until day 28 (p < 0.0001). The improvement of this score was observed in all HERP groups (p < 0.01) such that SNI/HERP10 presented improvement by day 7, maintained it up to day 21, and increased it again on day 28 (p < 0.01).
Histological examination
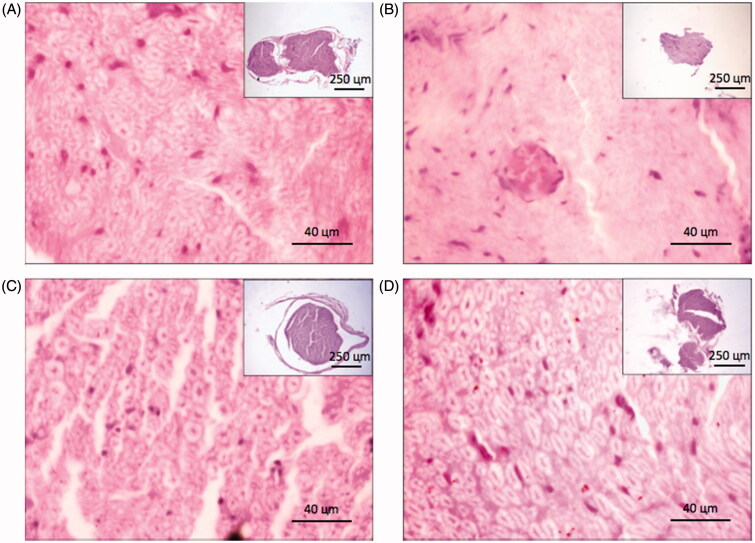
Qualitative analysis revealed that the SNI group treated with vehicle showed nerve derangement. This characteristic was almost absent in groups treated with HERP and IBP (Figure 4).
Figure 4.
Effects of HERP and IBP treatments on morphology of the sciatic nerves in rats at the 4th week post-surgery (hematoxylin–eosin staining). Photomicrographs show the left sciatic nerve (400×), 28 d after surgery. (A) Sham operated; (B) sciatic nerve injured treated with vehicle; (C) sciatic nerve injured treated with ibuprofen (60 mg/kg); (D) sciatic nerve injured treated with hydroethanolic extract of red propolis (10 mg/kg). Upper, the lower magnification (100×) illustrates the total area in each group. The integrity of axon morphology that is evident in sham group (A) (surrounding epineurium, fascicle divisions) is completely loss after SNI (B). It can be also observed the fusion of the extracellular material (B) and hyperemia (dashed with arrow). These characters were improved by IBP (C) and HERP (D) Myelinated axons are indicated by black arrows (in longitudinal and transversal arrangement, mostly in sham and HERP groups) and the inflammatory cells are indicated by white arrows (almost absent in groups sham, IBP, and HERP, but infiltrated in SNI).
Axon number: After post-surgical week 8, the mean axon number in the vehicle-treated SNI group was significantly decreased compared with the other groups. IBP and HERP1 promoted recovery in the axon number, obtaining means near those of the sham group. Moreover, when HERP10 was administered to SNI animals, the mean axon number increased, even compared with sham animals (p < 0.0001; Table 1 and Figure 4).
Table 1.
Histological examination 28 d after surgery.
| Group (n = 6) | % Area | Axon number/ 250 μm2 | Inflammatory profile |
|---|---|---|---|
| Sham/vehicle | 100.00 ± 6.23a | 32.55 ± 1.35a | 0.00 ± 0.00a |
| SNI/vehicle | 62.14 ± 4.04b | 17.15 ± 0.90b | 1.73 ± 0.06b |
| SNI/HERP 1 | 81.52 ± 2.66c | 26.02 ± 1.09a | 1.14 ± 0.05c |
| SNI/HERP 10 | 85.31 ± 2.85c | 49.31 ± 2.49c | 0.08 ± 0.04d |
| SNI/IBP 60 | 88.33 ± 4.51a,c | 31.00 ± 3.39a | 1.19 ± 0.06c |
Different letters in the same column indicates significant difference (p <0.0001, histological examination was submitted to one way ANOVA followed by Tukey post-hoc test. Non-Gaussian data from inflammatory profile were analyzed by the Kruskal–Wallis test followed by Dunn’s multiple comparison tests).
Inflammatory profile: Although the inflammatory infiltrate was not observed (absent) in the sham group, the injured group treated with vehicle presented moderate levels of cellular inflammatory infiltrate. The scores were significantly decreased to mild grade in the SNI/HERP1, SNI/HERP10, and SNI/IBP groups. Moreover, HERP10 reduced the inflammatory profile from moderate to mild–absent (Kruskal–Wallis followed by Dunn’s, p < 0.0001; Table 1).
Discussion
For the rapid analysis of natural metabolites, HRMS has been considered to be an important tool. Considering the complex constituents of HERP, direct infusion-based HRMS ESI(−)-MS was applied. In Brazil, 13 varieties of propolis are found because of the megabiodiversity and wide territorial area of the country. The green variety (botanical source is Baccharis dracunculifolia) has received the most attention in the published literature and presents a different chemical composition from the most recently identified propolis, the red variety. The chemical composition of the green propolis showed phenolic compounds, such as p-coumaric acid and artepillin C (Franchi et al. 2012; Berretta et al. 2013), which are not found in the red variety. In addition, isoflavones including formononetin and biochanin A are already considered to be chemical biomarkers of the red propolis (López et al. 2014).
In this study, we determined the major compounds present in the hydroalcoholic extracts of the Sergipe red propolis to evaluate the activity of these compounds. In comparison with the positive-ion mode, the negative-ion mode was determined to be the most effective, because of the presence of hydroxyls in flavonoids. With m/z 255.0652, 267.0651, 271.0966, and 283.0610, we confirmed the presence of pinocembrin, formononetin, vestitol, and biochanin A, respectively, as components of the red propolis from Sergipe (Figure 1).
These chemical differences between the red and green propolis are responsible for the biological efficiency of the bee glue. In 2013, Almeida et al. showed that red propolis treatment was associated with faster resolution of the healing process compared with green propolis treatment. This finding was attributed to the chemical composition of the red propolis. While studying the antitumor activity of propolis extracts in 2012, Franchi et al. demonstrated that the red propolis was more cytotoxic than the green variety in several human leukaemia cell lines and established that their positive results were associated with one or more of the chemical ingredients of the red propolis.
In this study, HERP attenuated the behavioural and histological changes induced by sciatic nerve crush injury. The axonal growth associated with functional recovery was observed after SNI using natural products and flavonoids (Thiagarajan et al. 2002; Quintans et al. 2014). These actions are frequently attributed to their antioxidant and anti-inflammatory activities because the influence of the microenvironment (oxidative potential, presence of inflammatory and glial cells, and altered calcium levels) in upholding or reducing the overall regeneration of peripheral nerves has been found to be essential (Muthuraman and Singh 2012; Kou et al. 2013; Tufek et al. 2013).
Here, the BBB, SFI, and beam walking performance tests were conducted to assess the peripheral nerve recovery that influences sensorimotor functions (Dinh et al. 2009). The higher dose of HERP (10 mg/kg) accelerated functional recovery after SNI compared with all other treatments and presented results similar to those of the sham group in 4 weeks. The lower dose (1 mg/kg) showed a profile similar to IBP with the best outcome compared with the vehicle treated group. These results indicated an important neuroprotective effect of HERP after SNI.
In agreement with our results, it has been demonstrated that the constituents of the red propolis have beneficial actions against neuronal cell death and degeneration. Formononetin, an isoflavone present in our sample and described as a marker in the red propolis (Righi et al. 2011), demonstrated neuroprotective action against neuronal hypoxia (Sun et al. 2012), glutamate-induced toxicity (Chen et al. 2008), and superoxide-induced damage (Yu et al. 2009) in vitro. Furthermore, formononetin protected the rat brain from oxidative stress in an animal model of Alzheimer's disease (Occhiuto et al. 2009). After traumatic brain injury in rats, formononetin reduced inflammatory responses by decreasing malondialdehyde, tumour necrosis factor-α, and interleukin-6 concentrations and the mRNA levels of cyclooxygenase-2 in the brain, while increasing the activities of glutathione peroxidase, superoxide dismutase, and the protein expression of nuclear factor E2-related factor 2 (Li et al. 2014) for neuroprotection.
Furthermore, the compounds pinocembrin and biochanin A were identified in our sample and have demonstrated neuroprotective actions against important neuronal damage factors including apoptosis and mitochondrial dysfunction (Liu et al. 2012). Pinocembrin was found to protected brain tissue from ischemia/reperfusion injury (Liu et al. 2008) by attenuating endoplasmic reticulum stress-induced apoptosis (Wu et al. 2013), attenuating blood–brain barrier injury induced by global cerebral ischemia–reperfusion in rats (Meng et al. 2011), and prevention of glutamate-induced apoptosis in neuronal cells (Gao et al. 2008). Biochanin A ameliorated behavioural and neurochemical deficits in a model for Alzheimer's disease (Biradar et al. 2014) and protected PC12 cells against Aβ-induced toxicity (Choi et al. 2010). Additionally, this compound has demonstrated neuroprotective activity against oxidative stress (Occhiuto et al. 2009) and lipopolysaccharide-induced damage through inhibition of microglia activation and proinflammatory factor generation (Chen et al. 2007).
Two types of events occur after nerve injury. The distal stump (also termed Wallerian degeneration), which is important for the preparation of the regeneration microenvironment, contributes to the removal of degenerated myelin and the recruitment of new Schwann cells. Furthermore, the degeneration of the distal stump is involved in the functional recovery of the target organs and the sprouting of new axons in the proximal stumps (Sta et al. 2014). Oxidative stress is markedly generated in peripheral nerve injury, interfering with the accumulation of macrophages that clear myelin debris (Zhang et al. 2013) during the first stages of Wallerian degeneration. Therefore, antioxidant agents such as HERP (Frozza et al. 2013) may be helpful in the clearance of degenerated myelin by macrophages and thus accelerate the regenerative process.
Furthermore, the Schwann cells in the distal nerve initially raise an effective response that promotes axon regeneration; however, their ability to survive and support axon growth declines within 8 weeks of denervation (Sulaiman and Gordon 2000) because of apoptotic cell death (Sulaiman and Gordon 2002). This time-dependent loss of support in the distal nerve is one factor that limits successful long-distance peripheral axon regeneration. Thus, the anti-apoptotic properties of HERP compounds (4 weeks of treatment) may have an important role in the management of axonal growth.
The HERP compounds may be related to a regenerative response, as formononetin has been shown to accelerate the differentiation of neural stem cells into neurons by increasing the transcription of pro-neural genes through neurogenin-2 promoter activity (Arai et al. 2013). Moreover, the protective mechanism of action of a formononetin sulfonate derivate in the central nervous system was attributed to the suppression of cell apoptosis and improved angiogenesis (Zhu et al. 2014). These events are closely related to the latter phase of nerve regeneration at the distal end (Heumann et al. 1987), suggesting that HERP may have neuroregenerative potential and plastic effects.
Compatible with this consideration, results found in the histological analysis revealed conserved nerve area, preserved number of myelinated axons, and decreased inflammatory infiltrate 28 d after SNI in groups treated with HERP and IBP. Because HERP 1 mg/kg presented very similar results to IBP, we performed histological analyses only for HERP 10 mg/kg. Notably, HERP10 induced an increase in the number of myelinated axons compared with the sham treatment.
Therefore, it is possible that HERP accelerated axonal growth and promoted sprouting. Sprouting is considered a hallmark of regeneration and usually appears 2 weeks from chronic nerve compression injury in animals (Mozaffar et al. 2009). During the initial stages of peripheral nerve regeneration, each injured axon produces collaterals at the injury site; however, many of the new axons do not reach the distal nerve ends. Thus, the distal nerve segments are characterized by a prominent mass of interlaced connective tissue with a small number of nerve fibres organized in typical mini fascicles (Keilhoff et al. 2007).
IBP was able to induce sprouting and regeneration in the central nervous system (Wang et al. 2009). After peripheral nerve injury, IBP improved functional recovery and axon regeneration, effects attributed to the inhibition of the small GTP-binding protein Rho (Madura et al. 2011). Based on our results, HERP was more effective in improving axonal growth and guidance which helps to explain the accelerated functional recovery from 1 to 4 weeks after surgery.
An analysis of paw withdrawal reflexes was used to assess the integrity of the afferent pathways and spinal reflexes related to axonal recovery (Metz et al. 2000). IBP showed the best effects, although all treatments reached values equal to sham within 28 d. However, it is possible that IBP and HERP act in different aspects of afferent transmission after SNI. Although the effects of IBP are mediated by COX enzymes (Carty et al. 2011), the effects of propolis and formononetin seem to indicate an interference with cytokine (IL-1β and TNF-α) production (Wang et al. 2012; Li et al. 2014). Furthermore, HERP may have an analgesic action against inflammatory pain by interfering with sensorial responses and reducing free radicals (Franchin et al. 2013).
Nerve injury initiates an inflammatory response and induces expression of pro-inflammatory cytokines including TNF-α, IL-1β, and IL-6, and induces a significant accumulation of inflammatory cells (Ruohonen et al. 2005). These inflammatory cells and mediators not only cause tissue damage and secondary injury but also play an important role in the regenerative process. For example, inflammatory cells and mediators are responsible for the recruitment of immature Schwann cells and the accelerated rapid migration of Schwann cells (Shamash et al. 2002).
However, the IL-1 and TNF pathways are involved in the recruitment of cells such as neutrophils and proinflammatory M1 macrophages (Nadeau et al. 2011). Moreover, these cytokines play a key role in the development and maintenance of pain after peripheral nerve injury (Scholz and Woolf 2007).
The recruitment of inflammatory cells, such as neutrophils, into the injured site contributes to the generation of altered behavioral and sensorial parameters and has been associated with the inflammatory response after SNI (Liou et al. 2013). Interestingly, the main phenolic compounds vestitol and neovestitol, discovered in the ethanolic extract of the red propolis (Inui et al. 2014), have demonstrated anti-inflammatory actions by inhibiting neutrophil migration (Franchin et al. 2013). This effect would be beneficial in reducing the exacerbation of secondary inflammatory response and damage. In agreement with this assertion, the inflammatory infiltrate, although discrete, was reduced after 28 d.
Recently, da Silva et al. (2015) evaluated the acute and sub-acute toxicities of HERP and observed that this extract showed no lethal effects in rats orally administered with 300 mg/kg, indicating that HERP has an LD50 above this value. Toxic signals were observed only at doses higher than 100 mg/kg in a sub-acute study, whereas 10 mg/kg was shown to be safe. Because of the multiple pathways involved in the pathophysiology of nerve injury (Sulaiman and Gordon 2002), a natural product such as the red propolis, with a wide variety of activities and low toxicity, may represent a novel therapeutic approach in the management of various aspects relevant for nerve regeneration.
Conclusion
Treatment with HERP promoted protective effects as evidenced by improved sensorimotor recovery, decreased inflammatory grade, preserved nerve area, and increased myelinated axons after SNI. These results suggested that HERP may be a novel therapeutic approach that should be considered for the treatment of SNI.
Declaration of interest
The authors report that they have no conflicts of interest. This work was supported by FAPITEC/SE (Government Foundation to Research Development of Sergipe State). F. F. Padilha, R. L. C. Albuquerque-Júnior, J. C. Cardoso, and L. S. S. Quintans-Júnior are recipients of CNPq productivity grants. Roberta A. Barbosa received scholarship from FAPITEC/SE and Tássia G. M. Nunes, Tâmara G. M. Nunes, and R. Belo-Neto received scholarship from CNPq.
References
- Albuquerque-Júnior RLC, Barreto ALS, Pires JA, Reis FP, Lima SO, Ribeiro MAG, Cardoso JC. 2009. Effect of bovine type-I collagen-based films containing red propolis on dermal healing in rodent model. Int J Morphol. 27:1105–1110. [Google Scholar]
- Almeida EB, Cardoso JC, Lima AK, Oliveira NL, Pontes-Filho NT, Lima SO, Souza ICL, Albuquerque-Junior RL. 2013. The incorporation of Brazilian propolis into collagen-based dressing films improves dermal burn healing. J Ethnopharmacol. 147:419–425. [DOI] [PubMed] [Google Scholar]
- Arai MA, Koryudzu K, Koyano T, Kowithavakorn T, Ishibashi M. 2013. Naturally occurring Ngn2 promoter activators from Butea superba. Mol Biosyst. 9:2489–2497. [DOI] [PubMed] [Google Scholar]
- Awale S, Li F, Onozuka H, Esumi H, Tezuka Y, Kadota S. 2008. Constituents of Brazilian red propolis and their preferential cytotoxic activity against human pancreatic PANC-1 cancer cell line in nutrient-deprived condition. Bioorg Med Chem Lett. 16:181–189. [DOI] [PubMed] [Google Scholar]
- Bain JR, Mackinnon SE, Hunter DA.. 1989. Functional evaluation of complete sciatic peroneal and posterior tibial nerve lesions in the rat. Plast Reconstr Surg. 83:129–138. [DOI] [PubMed] [Google Scholar]
- Basso DM, Beattie MS, Bresnahan JC.. 1995. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 12:1–21. [DOI] [PubMed] [Google Scholar]
- Biradar S, Joshi H, Chheda T.. 2014. Biochanin-A ameliorates behavioural and neurochemical derangements in cognitive-deficit mice for the betterment of Alzheimer's disease. Hum Exp Toxicol. 33:369–382. [DOI] [PubMed] [Google Scholar]
- Berretta AA, Castro PA, Cavalheiro AH, Fontes VS, Bom VP, Nascimento AP, Marquelle-Oliveira F, Pedrazzi V, Ramalho LN, Goldman GH. 2013. Evaluation of mucoadhesive gels with propolis (EPP-AF) in preclinical treatment of candidiasis vulvovaginal infection. Evid Based Complement Alternat Med. 18. Article ID641480, pages 10.1155/2013/641480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bobinski F, Martins DF, Bratty T, Mazzardo-Martins L, Winkelmann-Duarte EC, Guglielmo LG, Santos AR. 2011. Neuroprotective and neuroregenerative effects of low intensity aerobic exercise on sciatic nerve crush injury in mice. Neuroscience. 194:337–348. [DOI] [PubMed] [Google Scholar]
- Carty ML, Wixey JA, Reinebrant HE, Gobe G, Colditz PB, Buller KM. 2011. Ibuprofen inhibits neuroinflammation and attenuates white matter damage following hypoxia-ischemia in the immature rodent brain. Brain Res. 1402:9–19. [DOI] [PubMed] [Google Scholar]
- Chen HQ, Jin ZY, Li GH.. 2007. Biochanin A protects dopaminergic neurons against lipopolysaccharide-induced damage through inhibition of microglia activation and proinflammatory factors generation. Neurosci Lett. 417:112–117. [DOI] [PubMed] [Google Scholar]
- Chen J, Long Y, Han M, Wang T, Chen Q, Wang R. 2008. Water-soluble derivative of propolis mitigates scopolamine-induced learning and memory impairment in mice. Pharmacol Biochem Behav. 90:441–446. [DOI] [PubMed] [Google Scholar]
- Choi YH, Hong SS, Shin YS, Hwang BY, Park SY, Lee D. 2010. Phenolic compounds from Pueraria lobata protect PC12 cells against Aβ-induced toxicity. Arch Pharm Res. 33:1651–1654. [DOI] [PubMed] [Google Scholar]
- Cinegaglia NC, Bersano PRO, Búfalo MC, Sforcin JM. 2012. Cytotoxic action of Brazilian propolis in vitro on canine osteosarcoma cells. Phytother Res. 27:1277–1281. [DOI] [PubMed] [Google Scholar]
- da Silva RO, Andrade VM, Bullé Rêgo ES, Azevedo Dória GA, Santos Lima BD, da Silva FA, de Souza Araújo AA, de Albuquerque Júnior RL, Cordeiro Cardoso J, Zanardo Gomes M. 2015. Acute and sub-acute oral toxicity of Brazilian red propolis in rats. J Ethnopharmacol. 170:66–71. [DOI] [PubMed] [Google Scholar]
- Dinh P, Hazel A, Palispis W, Survadevara S, Gupta R. 2009. Functional assessment after sciatic nerve injury in a rat model. Microsurgery. 29:644–649. [DOI] [PubMed] [Google Scholar]
- Franchi GC, Jr Moraes CS, Toreti VC, Daugsch A, Nowill AE, Park YK. 2012. Comparison of effects of the ethanolic extracts of Brazilian propolis on human leukemic cells as assessed with the MTT assay. Evid Based Complement Alternat Med. 6. Article ID 918956, doi: 10.1155/2012/918956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franchin M, Cunha MG, Denny C, Napimoga NH, Cunha TM, Bueno-Silva B, Alencar SM, Ikegaki M, Rosalen PL. 2013. Bioactive fraction of geopropolis from Melipona scutellaris decreases neutrophils migration in the inflammatory process: involvement of nitric oxide pathway. Evid Based Complement Alternat Med. [Online]. Available from: http://www.hindawi.com/journals/ecam/2013/907041/. http://www.hindawi.com/journals/ecam/2013/907041/. Accessed on 12 October 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frozza CO, Garcia CS, Gambato G, de Souza MD, Salvador M, Moura S, Padilha FF, Seixas FK, Collares T, Borsuk S. 2013. Chemical characterization, antioxidant and cytotoxic activities of Brazilian red propolis. Food Chem Toxicol. 52:137–142. [DOI] [PubMed] [Google Scholar]
- Gao M, Zhang WC, Liu QS, Hu JJ, Liu GT, Du GH. 2008. Pinocembrin prevents glutamate-induced apoptosis in SH-SY5Y neuronal cells via decrease of bax/bcl-2 ratio. Eur J Pharmacol. 591:73–79. [DOI] [PubMed] [Google Scholar]
- Heumann R, Lindholm D, Bandtlow C, Meyer M, Radeke MJ, Misko TP, Shooter E, Thoenen H. 1987. Differential regulation of mRNA encoding nerve growth factor and its receptor in rat sciatic nerve during development, degeneration, and regeneration: role of macrophages. Proc Natl Acad Sci USA. 84:8735–8739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inui S, Hatano A, Yoshino M, Hosoya T, Shimamura Y, Masuda S, Ahn MR, Tazawa S, Araki Y, Kumazawa S. 2014. Identification of the phenolic compounds contributing to antibacterial activity in ethanol extracts of Brazilian red propolis. Nat Prod Res. 28:1293–1296. [DOI] [PubMed] [Google Scholar]
- Jang CH, Cho YB, Choi CH.. 2012. Effect of Ginkgo biloba extract on recovery after facial nerve crush injury in the rat. Int J Pediatr Otorhinolaryngol. 76:1823–1826. [DOI] [PubMed] [Google Scholar]
- Keilhoff G, Langnaese K, Wolf G, Fansa H. 2007. Inhibiting effect of minocycline on the regeneration of peripheral nerves. Dev Neurobiol. 67:1382–1395. [DOI] [PubMed] [Google Scholar]
- Kou V, Wang Z, Wu Z, Zhang, Zhang P, Zhang Y, Yin X, Wong X, Qiu G, Jiang B. 2013. Epimedium extract promotes peripheral nerve regeneration in rats. Evid Based Complement Alternat Med. [Online]. Available from: http://www.hindawi.com/journals/ecam/2013/954798/.Accessedon20August2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacorte S, Fernandez-Alba AR.. 2006. Time of flight mass spectrometry applied to the liquid chromatographic analysis of pesticides in water and food. Mass Spectrom Rev. 25:866–880. [DOI] [PubMed] [Google Scholar]
- Li Z, Dong X, Zhang J, Zeng G, Zhao H, Liu Y, Qiu R, Mo L, Ye Y. 2014. Formononetin protects TBI rats against neurological lesions and the underlying mechanism. J Neurol Sci. 15:112–117. [DOI] [PubMed] [Google Scholar]
- Liou JT, Lee CM, Lin YC, Chen CY, Liao CC, Lee HC, Day YJ. 2013. P-Selectin is required for neutrophils and macrophage infiltration into injured site and contributes to generation of behavioral hypersensitivity following peripheral nerve injury in mice. Pain. 154:2150–2159. [DOI] [PubMed] [Google Scholar]
- Liu R, Gao M, Yang ZH, Du GH. 2008. Pinocembrin protects rat brain against oxidation and apoptosis induced by ischemia-reperfusion both in vivo and in vitro. Brain Res. 1216:104–115. [DOI] [PubMed] [Google Scholar]
- Liu R, Wu CX, Zhou D, Yang F, Tian S, Zhang L, Zhang TT, Du GH. 2012. Pinocembrin protects against β-amyloid-induced toxicity in neurons through inhibiting receptor for advanced glycation end products (RAGE)-independent signaling pathways and regulating mitochondrion-mediated apoptosis. BMC Med. 10:105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López BGC, Schmidt EM, Eberlin MN, Sawaya ACHF. 2014. Phytochemical markers of different types of red propolis. Food Chem. 146:174–180. [DOI] [PubMed] [Google Scholar]
- Madura T, Tomita K, Terenghi G.. 2011. Ibuprofen improves functional outcome after axotomy and immediate repair in the peripheral nervous system. J Plast Reconstr Aesthet Surg. 64:1641–1646. [DOI] [PubMed] [Google Scholar]
- Meng F, Liu R, Gao M, Wang Y, Yu X, Xuan Z, Sun J, Yang F, Wu C, Du G. 2011. Pinocembrin attenuates blood–brain barrier injury induced by global cerebral ischemia–reperfusion in rats. Brain Res. 1391:93–101. [DOI] [PubMed] [Google Scholar]
- Metz GA, Merkler D, Dietz V, Schwab ME, Fouad K. 2000. Efficient testing of motor function in spinal cord injured rats. Brain Res. 883:165–177. [DOI] [PubMed] [Google Scholar]
- Morsy AS, Abdalla AL, Soltan YA, Sallam SM, El-Azrak KD, Louvandini H, Alencar SM. 2013. Effect of Brazilian red propolis administration on hematological, biochemical variables and parasitic response of Santa Inês ewes during and after flushing period. Trop Anim Health Prod. 45:1609–1618. [DOI] [PubMed] [Google Scholar]
- Mozaffar T, Strandberg E, Abe K, Hilgenberg LG, Smith MA, Gupta R. 2009. Neuromuscular junction integrity after chronic nerve compression injury. J Orthop Res. 7:114–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muthuraman A, Singh N.. 2012. Neuroprotective effect of saponin rich extract of Acorus calamus L. in rat model of chronic constriction injury (CCI) of sciatic nerve-induced neuropathic pain. J Ethnopharmacol. 142:723–731. [DOI] [PubMed] [Google Scholar]
- Nadeau S, Filali M, Zhang J, Kerr BJ, Rivest S, Soulet D, Iwakura Y, de RiveroVaccari JP, Keane RW, Lacroix S. 2011. Functional recovery after peripheral nerve injury is dependent on the pro-inflammatory cytokines IL-1β and TNF: implications for neuropathic pain. J Neurosci. 31:12533–12542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Occhiuto F, Palumbo DR, Samperi S, Zangla G, Pino A, De Pasquale R, Circosta C. 2009. The isoflavones mixture from Trifolium pratense L. protects HCN 1-A neurons from oxidative stress. Phytother Res. 23:192–196. [DOI] [PubMed] [Google Scholar]
- Ortega-Álvaro A, Berrocoso E, Rey-Brea R, Leza JC, Mico JÁ. 2012. Comparison of the antinociceptive effects of ibuprofen arginate and ibuprofen in rat models of inflammatory and neuropathic pain. J Life Sci. 90:13–20. [DOI] [PubMed] [Google Scholar]
- Quintans JSS, Antoniolli AR, Almeida JR, Santana-Filho VJ, Quintans-Júnior LJ. 2014. Natural products evaluated in neuropathic pain models – a systematic review. Basic Clin Pharmacol Toxicol. 114:442–450. [DOI] [PubMed] [Google Scholar]
- Righi AA, Alves TR, Negri G, Marques LM, Breyer H, Salatino A. 2011. Brazilian red propolis: unreported substances, antioxidant and antimicrobial activities. J Sci Food Agric. 91:363–370. [DOI] [PubMed] [Google Scholar]
- Ruohonen S, Khademi M, Jagodic M, Tashiken HS, Olsson T, Roytta M. 2005. Cytokine responses during chronic denervation. J Neuroinflammation. 2:26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scholz J, Woolf CJ.. 2007. The neuropathic pain triad: neurons, immune cells and glia. Nat Neurosci. 10:1361–1368. [DOI] [PubMed] [Google Scholar]
- Shamash S, Reichert F, Rotshenker S.. 2002. The cytokine network of Wallerian degeneration: tumor necrosis factor-alpha, interleukin-1alpha, and interleukin-1beta. J Neurosci. 22:3052–3060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Souza ICL, Nascimento M, Souza-Neta RG, Santos JC, Costa LP, Cardoso JC, Albuquerque-Junior RL. 2013. Effect of the maltodextrin-induced chemical reticulation on the physical properties and healing potential of collagen-based membranes containing Brazilian red propolis extract. Int J Med Med Sci. 5:514–524. [Google Scholar]
- Sta M, Cappaert NL, Ramekers D, Baas F, Wadman WJ. 2014. The functional and morphological characteristics of sciatic nerve degeneration and regeneration after crush injury in rats. J Neurosci Methods. 222:189–198. [DOI] [PubMed] [Google Scholar]
- Sulaiman OA, Gordon T.. 2000. Effects of short-and long-term Schwann cell denervation on peripheral nerve regeneration, myelination, and size. GLIA. 32:234–246. [DOI] [PubMed] [Google Scholar]
- Sulaiman OA, Gordon T.. 2002. Transforming growth factor-beta and forskolin attenuate the adverse effects of long-term Schwann cell denervation on peripheral nerve regeneration in vivo. GLIA. 37:206–218. [DOI] [PubMed] [Google Scholar]
- Sun M, Zhou T, Zhou L, Chen Q, Yu Y, Yang H, Zhong K, Zhang X, Xu F, Cai S. 2012. Formononetin protects neurons against hypoxia-induced cytotoxicity through upregulation of ADAM10 and sAβPPα. J Alzheimers Dis. 28:795–808. [DOI] [PubMed] [Google Scholar]
- Tamaddonfard E, Farshid AA, Ahmadian E, Hamidhoseyni A. 2013. Crocin enhanced functional recovery after sciatic nerve crush injury in rats. Iran J Basic Med Sci. 16:83–90. [PMC free article] [PubMed] [Google Scholar]
- Thiagarajan VRK, Shanmugam P, Krishnan UM, Muthuraman A, Singh N. 2002. Ameliorative potential of Butea monosperma on chronic constriction injury of sciatic nerve induced neuropathic pain in rats. An Acad Bras Cienc. 84:1091–1104. [DOI] [PubMed] [Google Scholar]
- Tufek A, Kaya S, Tokgöz O, Firat U, Evliyaoglu O,Celik F, Karaman H. 2013. The protective effect of dexmedetomidine on bupivacaine-induced sciatic nerve inflammation is mediated by mast cells. J Clin Inv Med. 36:95–102. [DOI] [PubMed] [Google Scholar]
- Wang X, Budel S, Baughman K, Gould G, Song KH, Strittmatter SM. 2009. Ibuprofen enhances recovery from spinal cord injury by limiting tissue loss and stimulating axonal growth. J Neurotrauma. 26:81–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Zhu Y, Gao L, Yin H, Xie Z, Wang D, Zhu Z, Han X. 2012. Formononetin attenuates IL-1β-induced apoptosis and NF-κB activation in INS-1 cells. Molecules. 17:10052–10064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CX, Liu R, Gao M, Zhao G, Wu S, Wu CF, Du GH. 2013. Pinocembrin protects brain against ischemia/reperfusion injury by attenuating endoplasmic reticulum stress induced apoptosis. Neurosci Lett. 546:57–62. [DOI] [PubMed] [Google Scholar]
- Von Euler M, Akesson E, Samuelsson EB, Seiger A, Sundstrom E. 1996. Motor performance score: a new algorithm for accurate behavioral testing of spinal cord injury in rats. Exp Neurol. 137:242–254. [DOI] [PubMed] [Google Scholar]
- Yu DH, Bao YM, An LJ, Yang M. 2009. Protection of PC12 cells against superoxide-induced damage by isoflavonoids from Astragalus mongholicus. Biomed Environ Sci. 22:50–54. [DOI] [PubMed] [Google Scholar]
- Zhang L, Johnson D, Johnson JA.. 2013. Deletion of Nrf2 impairs functional recovery, reduces clearance of myelin debris and decreases axonal remyelination after peripheral nerve injury. Neurobiol Dis. 54:329–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu X, Yao L, Yang X, Sun H, Guo A, Li A, Yang H. 2014. Spatiotemporal expression of KHSRP modulates Schwann cells and neuronal differentiation after sciatic nerve injury. Int J Biochem Cell Biol. 48:1–10. [DOI] [PubMed] [Google Scholar]