Abstract
Paxillin is a ubiquitously expressed adaptor protein integral to focal adhesions, cell motility, and apoptosis. Paxillin has also recently been implicated as a mediator of nongenomic androgen receptor (AR) signaling in prostate cancer and other cells. We sought to investigate the relationship between paxillin and AR in granulosa cells (GCs), where androgen actions, apoptosis, and focal adhesions are of known importance, but where the role of paxillin is understudied. We recently showed that paxillin knockout in mouse GCs increases fertility in older mice. Here, we demonstrate that paxillin knockdown in human granulosa-derived KGN cells, as well as knockout in mouse primary GCs, results in reduced AR protein but not reduced mRNA expression. Further, we find that both AR protein and mRNA half-lives are reduced by approximately one-third in the absence of paxillin, but that cells adapt to chronic loss of paxillin by upregulating AR gene expression. Using co-immunofluorescence and proximity ligation assays, we show that paxillin and AR co-localize at the plasma membrane in GCs in a focal adhesion kinase-dependent way, and that disruption of focal adhesions leads to reduced AR protein level. Our findings suggest that paxillin recruits AR to the GC membrane, where it may be sequestered from proteasomal degradation and poised for nongenomic signaling, as reported in other tissues. To investigate the physiological significance of this in disorders of androgen excess, we tested the effect of GC-specific paxillin knockout in a mouse model of polycystic ovary syndrome (PCOS) induced by chronic postnatal dihydrotestosterone (DHT) exposure. While none of the control mice had estrous cycles, 33% of paxillin knockout mice were cycling, indicating that paxillin deletion may offer partial protection from the negative effects of androgen excess by reducing AR expression. Paxillin-knockout GCs from mice with DHT-induced PCOS also produced more estradiol than GCs from littermate controls. Thus, paxillin may be a novel target in the management of androgen-related disorders in women, such as PCOS.
Keywords: paxillin, androgen receptor, ovary, granulosa, focal adhesion, polycystic ovary syndrome, PCOS
Introduction
Although traditionally regarded as male hormones, androgens play a crucial role in female reproduction. Excessive androgen production is a hallmark of polycystic ovary syndrome (PCOS) (Azziz et al., 2016), which is the leading cause of female infertility (Dennett and Simon, 2015). In addition, PCOS can cause a wide range of symptoms that can range from unpleasant to debilitating, including acne, hair loss, dysmenorrhea, weight gain, insulin resistance, and mental health and mood disorders such as anxiety and depression (Balen et al., 1995; Dennett and Simon, 2015; Hoeger et al., 2021). Furthermore, patients with PCOS are at higher risk for endometrial cancer, Type 2 diabetes mellitus, and major cardiovascular events (Berni et al., 2021). On the other hand, androgen deficiency in mouse androgen receptor (AR) knockout models causes reduced fertility and premature ovarian insufficiency, or diminished ovarian reserve (DOR) (Hu et al., 2004; Sen and Hammes, 2010). The observation that both PCOS and DOR result in impaired fertility highlights the importance of androgen balance in order to maintain female reproductive functionality (Astapova et al., 2019). Thus, studying androgens in the context of female reproductive health is important.
Granulosa cells (GCs) are the main androgen-responsive ovarian cell type. GCs are stimulated by FSH to metabolize androgens, produced by neighboring theca cells, into estrogens. Androgen deficiency in genetic females causes subfertility (Gleicher et al., 2013; Shohat-Tal et al., 2015). Transgenic mice with cell-specific knockout of AR in theca cells (Ma et al., 2017) and in oocytes (Sen and Hammes, 2010) exhibit normal fertility, while both global as well as GC-specific AR knockouts result in impaired fertility as a result of DOR (Hu et al., 2004; Sen and Hammes, 2010; Walters et al., 2012). It is clear that androgen actions in GCs are of major importance for female fertility; however, the mechanisms of androgen actions and the regulation of AR in GCs are not yet known.
Most of our current knowledge of AR comes from prostate cancer studies. In prostate cancer cells, AR action has been found to be regulated by paxillin (Sen et al., 2010). Paxillin (PXN) is an adaptor protein that is involved in the formation of focal adhesion complexes in many cell types. Paxillin binds with cytoskeletal and focal adhesion proteins, such as vinculin, tubulin, and focal adhesion kinase (FAK), and has been found to play an important role in cell motility and cancer cell migration and invasion (Brown et al., 1996; Alpha et al., 2020). In addition to its role in focal adhesions, more recent studies have implicated paxillin in steroid signaling. In prostate cancer cells, paxillin regulates both nuclear and extranuclear androgen actions, serving as a liaison between the two modes of AR signaling (Ma and Hammes, 2018). Specifically, paxillin mediates rapid AR signaling through the epidermal growth factor receptor (EGFR)–mitogen-activated protein kinase (MAPK) pathway, promotes AR nuclear import, and co-regulates androgen-responsive gene networks in prostate cancer, potentiating androgen effects on cell metabolism and proliferation (Kasai et al., 2003; Sen et al., 2010). As in prostate cancer cells, androgen signaling is crucial to GC processes, but it is unknown whether paxillin plays a similar role regulating AR actions in GCs.
Few studies have focused on the role of paxillin in female reproduction. In Xenopus laevis oocytes, paxillin is an important regulator of steroid-mediated development, where paxillin phosphorylation is required for expression of a key protein in oocyte maturation, namely MOS, and MAPK1 (ERK2) activation and oocyte maturation (Miedlich et al., 2017). In mouse GCs, paxillin was found to be required for androgen-stimulated FSH receptor protein translation, likely owing to paxillin’s role in modulating the MAPK pathway (Sen et al., 2014). This suggests that paxillin may module ovarian function. To explore this, we recently created a GC-specific paxillin knockout mouse model and found that paxillin loss in GCs reduces cell death and increases female fertility in advanced reproductive age (Vann et al., 2023). Previously, we showed that genomic AR actions in GCs may be minimal, and that AR localization is a major determinant of its stability, as its half-life increases dramatically when AR is not available for proteasomal degradation in the cytoplasm (Astapova et al., 2021). Here, we elucidate the interaction between paxillin and AR in GCs and show that paxillin recruits AR to the cell membrane, near focal adhesions, thereby protecting it from degradation and promoting its nongenomic activity. Further, we show that this interaction may be relevant in female reproduction by enhancing ovarian sensitivity to excess androgen exposure in a mouse model of PCOS.
Materials and methods
Animals
Mice were maintained in an experimental animal facility according to the protocol for the Care and Use of Laboratory Animals, approved by the University Committee on Animal Resources at the University of Rochester. Mice were group-housed when possible, except during estrous cycling studies during which mice were single-housed, on a 12-h light–dark cycle with lights on at 6:00 AM and ambient temperature of 23 ± 1°C. Standard chow (LabDiet PicoLab® Rodent Diet 20, Marlborough, MA, USA), sterilized by the manufacturer via irradiation, and ultra-filtered (0.1 micron) municipal water were provided ad libitum.
Generation of GC-specific paxillin knockout mice
GC-specific paxillin knockout mice were generated as previously published using the Cre-lox system (Vann et al., 2023). Briefly, paxillin-floxed mice (Pxnfl/fl) were crossed with mice expressing Cre recombinase driven by the anti-Müllerian hormone receptor 2 (Amhr2) promoter (Amhr2Cre/+). Offspring positive for Amhr2-Cre and two paxillin-floxed alleles (Amhr2Cre/+; Pxnfl/fl) were considered GC-specific paxillin knockouts, while offspring negative for Amhr2-Cre and positive for two paxillin-floxed alleles (Amhr2+/+; Pxnfl/fl) were designated as littermate controls. We recently characterized the reproductive effects of this knockout model (Vann et al., 2023). Here, we studied the effects of paxillin on AR expression in GCs using an independent cohort of these transgenic mice.
Cell culture
KGN cells were obtained from RIKEN (Wakō, Saitama, Japan; RRID: CVCL0375) and cultured in Dulbecco’s modified Eagle’s medium (DMEM)/F12 (Invitrogen catalog #11330-057, Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS) at 37○C and 5% CO2. Cells were passaged every 5–6 days, and passage number <20 was used for experiments until clonal selection.
To culture primary GCs, ovaries from 24- to 26-week-old mice were dissected in PBS, then transferred to DMEM/F12. Follicles were punctured under a dissecting microscope with 31-guage needles to extrude GCs into the media, as previously described (Tian et al., 2015). Cells were pelleted at 1000g for 5 min at 4○C, then resuspended in fresh DMEM/F12 supplemented with 10% FBS and cultured at 37○C and 5% CO2 for up to 4 days.
For protein degradation experiments, paxillin knockout KGN cells (see ‘Stable paxillin knockout’) or non-targeting control KGN cells were treated with 50 μM cycloheximide (Fisher 0970100, Waltham MA, USA), diluted in ethanol, for up to 24 h before harvest. For RNA degradation experiments, cells were treated with 5 μM actinomycin D (Sigma-Aldrich catalog #A1410, Burlington, MA, USA), diluted in DMSO, for up to 24 h before harvest. In some experiments, 25 nM dihydrotestosterone (DHT) in ethanol was added to the media.
siRNA knockdown
siRNA targeting human PXN (Dharmacon ON-TARGETplus SMARTpool L-003023-00-0005, Lafayette, CO, USA) was transfected using DharmaFECT3 (3 μL/ml) in Opti-MEM at final amounts of 40 pmol/ml. Non-targeting siRNA (Dharmacon D-001810-01) was used as a non-targeting control in all experiments. Serum-containing media were added after 24 h. The total duration of knockdown was 5 days in all experiments.
Stable paxillin knockout
Paxillin-knockout KGN cells were generated as previously described (Vann et al., 2023). Briefly, KGN cells were transfected using jetPRIME (Polyplus, Illkirch, France) with an all-in-one non-viral vector set containing three single-guide (sg) RNAs targeting the human paxillin gene (ABM catalog #382401110591, Richmond, BC, Canada) or a non-targeting control vector. Two days later, green fluorescent protein (GFP)-positive cells were selected using a BioRad S3e flow cytometer and either seeded into a 96-well at one cell per well to generate single cell-derived clones or cultured for 5 additional days prior to experimentation.
Western blot
After treatments as indicated, cells were lysed directly in the culture plates with lysis buffer containing 50 mM Tris base, 150 mM NaCl, 5 mM EDTA, 1% Triton X-100, and Halt™ Protease and Phosphatase Inhibitor Cocktail (Thermo Fisher Scientific catalog #1861281, Waltham, MA, USA). Cell lysates were boiled for 5 min in sample buffer containing 5% β-mercaptoethanol, then separated in sodium dodecyl sulfate-polyacrylamide gels (4–15% gradient gels, BioRad catalog #4561086, Hercules, CA, USA) and transferred onto polyvinylidene fluoride membranes in 20% methanol. Blots were blocked for 1 h at room temperature with 5% milk in Tris-buffered saline Tween, then probed overnight at 4°C with antibodies for AR (human samples: Cell Signaling Technology catalog #5153, 1:1000, Danvers, MA, USA; mouse samples: Abcam catalog #ab105225, 1:1000, Boston, MA, USA), paxillin (BD Transduction Laboratories catalog #610051, 1:1000, Franklin Lakes, NJ, USA), phospho-paxillin (Cell Signaling Technology catalog #2541, 1:1000), FAK (Invitrogen catalog #701094, 1:1000), phospho-FAK (Invitrogen catalog #700255, 1:1000), or GAPDH (Cell Signaling Technology catalog #2118, 1:4000). Band density was quantified using ImageJ (Schneider et al., 2012) and normalized to GAPDH where indicated.
Quantitative PCR
After treatments as indicated, for quantitative PCR (qPCR), total RNA was isolated from cultured cells using the E.Z.N.A.® Total RNA Kit I (Omega Bio-Tek catalog #R6834, Norcross, GA, USA). RNA samples from KGN cells or primary GCs were reverse-transcribed and amplified in the same reaction using the qScript XLT 1-Step RT-qPCR ToughMix kit (QuantaBio, Beverly, MA, USA) and species-specific ROX-containing TaqMan primers (Applied Biosystems, Waltham, MA; Hs with capital letters for human and Mm with lower case letters for mouse: Mm00448533_m1 [Pxn], Mm00442688_m1 [Ar], Hs00171172_m1 [AR], Mm99999915-g1 [Gapdh], and Hs02786624_g1 [GAPDH]). RNA expression was normalized to GAPDH or to untreated (0 h) samples using the ΔΔCT method.
Immunofluorescence
Cells were seeded on poly-L-lysine-treated glass coverslips to ∼25% confluency. After indicated treatments, cells were fixed with 4% paraformaldehyde, permeabilized with 0.25% Triton X-100, and blocked with 2% bovine serum albumin. The samples were incubated overnight in a humidity chamber at 4°C with mouse anti-PXN antibody (BD Transduction Laboratories catalog #610051, 1:100 or 2.5 μg/ml) and rabbit anti-AR antibody (Santa Cruz AR N-20, catalog #sc-816, 1:50 or 2 μg/ml, Dallas, TX, USA), then for 2 h at room temperature with fluorescein goat anti-rabbit IgG (Invitrogen catalog #A-11008, 1:500) and Texas Red goat anti-mouse IgG (Invitrogen catalog #T-6390, 1:500). IgG controls for the primary antibodies are shown in Supplementary Fig. S1. Samples were mounted in Duolink In Situ Mounting Medium with DAPI (4', 6-diamidino-2-phenylindole; Sigma-Aldrich catalog #DUO82040) or incubated in 1 μM Hoechst 33342 Staining Dye Solution (Abcam catalog #ab228551) for 15 min prior to mounting.
Proximity ligation assay
Primary mouse GCs or KGN cells were seeded as for immunofluorescence (see above) and treated with 5 μM FAK inhibitor PF-573228 (Sigma-Aldrich catalog #PZ0117) in DMSO, and 25 nM DHT in ethanol, or vehicle. Cells were fixed, permeabilized, and probed with primary antibodies as described in the Section ‘Immunofluorescence’. In KGN cells, the Abcam AR antibody SP-107, catalog #ab105225 was used at 1:22 dilution (5 μg/ml). This antibody was validated in our laboratory using AR-expressing and AR-null prostate cancer cells (Supplementary Fig. S2). For DNA methyltransferase (DNMT), the Novus Biologicals catalog #NB100-56519 antibody was used at 1:100 dilution (10 μg/ml, Centennial, CO, USA), and for cytoplasmic poly (A)-binding protein (PABP), the Abcam catalog #21060 antibody was used at 1:100 dilution (10 μg/ml). The Duolink® Proximity Ligation Assay (Sigma-Aldrich, Duolink® In Situ PLA® Probe Anti-Rabbit MINUS: catalog #DUO92005 and Duolink® In Situ PLA® Probe Anti-Mouse PLUS: catalog #DUO92001) was then utilized to image proximity between the following pairs of proteins using specific antibodies: AR and paxillin, AR and DNMT, or paxillin and PABP. In this assay, secondary antibodies are conjugated to complementary oligonucleotides, which generate a fluorescent signal when the two primary antibodies are in close proximity, allowing the complementary oligonucleotides to produce a double-stranded product.
PCOS mouse model
PCOS was induced by chronic postnatal DHT exposure as previously described (van Houten et al., 2012). C57BL/6J pups were weaned on postnatal Day 18. The following day, female pups were placed under general anesthesia (isoflurane/O2), and a 90-day continuous-release pellet containing either 2.5 mg DHT or placebo (Innovative Research of America, Sarasota, FL, USA, catalog #NA-161 and #C-111) was inserted s.c. under the dorsal skin. Mice were randomly assigned to either the DHT or placebo group in a split-litter design, with half of the female pups in each litter receiving DHT pellets and the other half receiving placebo, to account for litter-specific effects. The incision was closed using the Autoclip™ Wound Closing System (BD, catalog #22-275998), and the staple was removed the following week. Immediately following surgery, mice were injected i.p. with 2 μg/g of body weight meloxicam to minimize pain during recovery. After pellet insertion, mice were group-housed within treatment groups as DHT-treated mice tend to become more aggressive.
Estrous cycling
Estrous cycles were monitored using daily vaginal cytology as previously described (Byers et al., 2012) for 2 weeks in 9-week-old (45 days post-pellet) and 15-week-old (90 days post-pellet) mice. Briefly, vaginal lavage samples were collected by repeatedly pipetting a drop of sterile PBS at the vaginal opening, which was then placed on a microscope slide and air-dried. Slides were then processed using Hema 3 Stat Pack (Fisher catalog #22-122911) according to the manufacturer’s instructions and visualized under light microscopy. Similar to a previously developed protocol (McLean et al., 2012), samples containing predominantly neutrophils were classified as diestrus, samples where at least one-third of the cells were nucleated epithelial cells were classified as proestrus, samples with only epithelial cells (both nucleated and keratinized) were classified as estrus, and samples containing both keratinized epithelial cells and neutrophils were classified as metestrus. Mice were single-housed beginning 1 week before and continuing throughout estrous cycle monitoring to minimize group-housing effects.
Statistical analysis
Data are displayed as mean with SEM of the indicated numbers of biological replicates. Data analysis was performed using GraphPad Prism (version 10.0.3, GraphPad Software, Boston, MA, USA, www.graphpad.com). Unless otherwise noted, data were analyzed by the nonparametric Mann–Whitney test. The estrous cycling data analysis was performed using the Mantel–Haenszel Chi-square test. RNA and protein degradation experiments were analyzed using non-linear regression with a one-phase decay model, with Y0 constrained to the t = 0 value and Plateau constrained to 0. A P value <0.05 was considered to be statistically significant.
Results
Paxillin enhances AR expression in human and mouse GCs
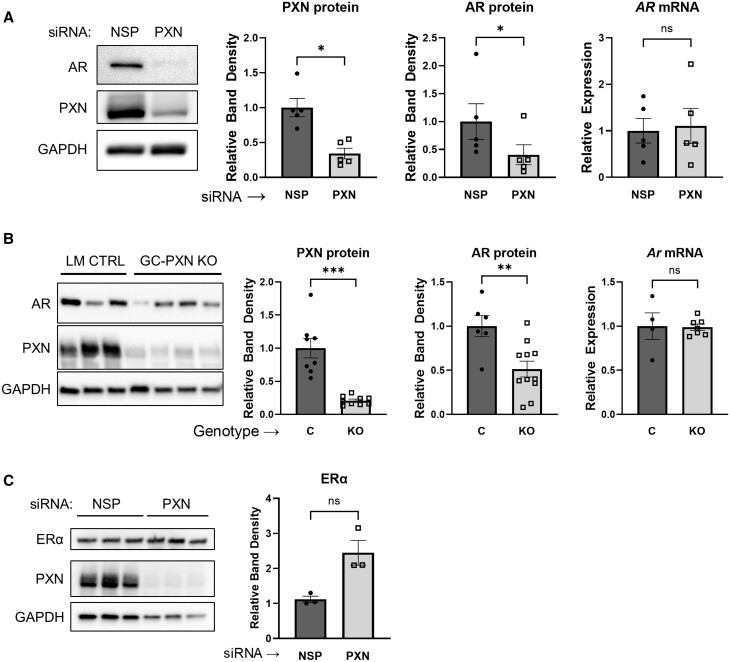
To investigate the role of paxillin in GC androgen signaling, we first knocked down paxillin using siRNA in human granulosa-derived KGN cells. After 5 days, paxillin mRNA and protein content was significantly reduced by the knockdown. This resulted in decreased AR protein, but not mRNA expression (Fig. 1A, Supplementary Fig. S3). We also measured AR expression in primary GCs isolated from GC-specific paxillin knockout mice (PXN-KO GCs), which were created by crossing Amhr2-Cre expressing mice with Pxn-floxed mice as recently reported (Vann et al., 2023). Similar to KGN cells, AR protein content was slightly but statistically significantly decreased, but Ar mRNA was not affected in PXN-KO GCs (Fig. 1B, Supplementary Fig. S3). This suggests that paxillin regulates AR expression in GCs at a post-transcriptional level. To assess whether this effect is specific to AR, we also measured the expression of another nuclear receptor, estrogen receptor α (ERα) in paxillin-knockdown KGN cells. The protein level of ERα, relative to the housekeeping gene GAPDH, was not significantly altered after paxillin knockdown (Fig. 1C, Supplementary Fig. S3).
Figure 1.
Paxillin enhances androgen receptor protein content independently of its mRNA expression in KGN cells and in mouse granulosa cells. (A) Representative western blot, band densitometry, and qPCR of KGN cells after transfection with siRNA targeting non-specific (NSP) control or paxillin (PXN). Data are mean and SEM of five independent experiments. Statistical significance was determined by paired Mann–Whitney test. (B) Representative western blot, band densitometry and qPCR in mouse primary GCs from GC-specific paxillin knockout mice (GC-PXN KO or KO), or littermate controls (LM CTRL or C), n = 3–11 per group. Cells were cultured for 4 days before analysis. Data are mean and SEM of n = 3–11 mice per group. Statistical significance was determined by Mann–Whitney test. (C) ERα protein content in PXN-knockdown KGN cells transfected as in (A) and band densitometry of ERα relative to GAPDH. Statistical significance was determined by Mann–Whitney test. For all comparisons, ns: not significant; *P > 0.05; **P < 0.01; ***P < 0.001. AR: androgen receptor, PXN: paxillin, NSP: nonspecific control, LM CTRL: littermate control, GC-PXN KO: granulosa-specific paxillin knockout, ERα: estrogen receptor alpha. Uncropped blots are shown in Supplementary Fig. S3.
Paxillin and AR co-localize at the mouse and human GC membrane in a focal adhesion-dependent manner
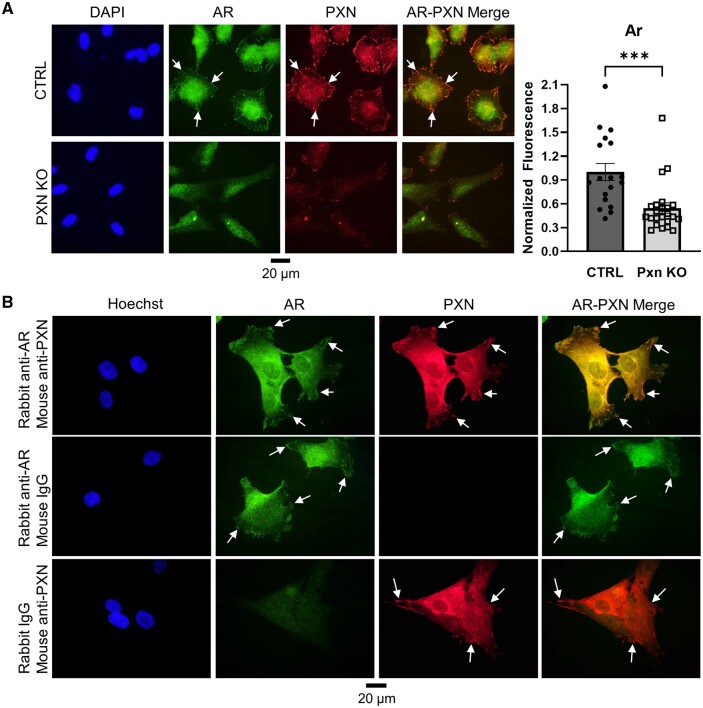
Having found that paxillin enhances AR protein content, we next sought to determine the mechanism by which paxillin regulates AR expression. To determine whether paxillin and AR co-localize in GCs, we performed co-immunofluorescence of both proteins in mouse primary GCs. The purity of our GC isolation was confirmed by enriched FSH receptor expression and low Cyp17 expression compared with theca cells from the same ovaries (Supplementary Fig. S4). Surprisingly, we found that mouse AR and paxillin co-localize in punctate, membrane-associated structures (Fig. 2A), which have the appearance of focal adhesions. We then examined AR in primary GCs from GC-specific paxillin-knockout mice. Owing to the long half-life of paxillin (more than 5 days in our previous studies) and the comparatively short life cycle of a follicle, paxillin protein was depleted but not completely absent in paxillin-knockout GCs, consistent with our prior findings (Vann et al., 2023). Paxillin-knockout GCs did not show appreciable AR near the cell membrane, suggesting that paxillin recruits AR to the cell membrane. Further, AR expression in paxillin-knockout GCs was reduced compared to the littermate control GCs (Fig. 2A), validating our western blot findings from Fig. 1. Similar to mouse primary GCs, we observed co-localization of AR and PXN in human KGN cells (Fig. 2B). To prove that this result is not an artifact of fluorescence cross-over between the red and green spectra, we used non-specific isotype control antibodies for both AR and PXN, which did not produce fluorescent signals (Supplementary Fig. S1 and Fig. 2B).
Figure 2.
Androgen receptor and paxillin co-localize at the granulosa cell membrane. (A) Mouse primary GCs were isolated from GC-specific paxillin knockout mice (PXN KO) or littermate controls (CTRL) and cultured on coverslips before fixation and co-immunofluorescence with antibodies against AR and PXN. AR fluorescence was quantified in 18–23 cells from each sample using Image J and analyzed by Mann–Whitney test. ***P < 0.001. (B) KGN cells were grown on coverslips, fixed, and co-immunoprobed with the primary antibodies listed on the left. All samples were then probed with both anti-mouse and anti-rabbit secondary antibodies. White arrows indicate membrane-localized AR and PXN in all pictures. DAPI: 4',6-diamidino-2-phenylindole, AR: androgen receptor, PXN: paxillin, CTRL: littermate control, PXN KO: granulosa-specific paxillin knockout.
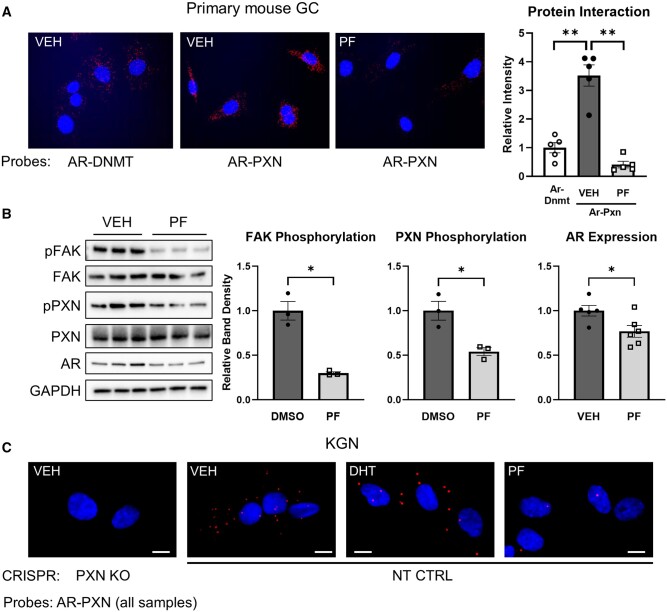
To test whether the AR–paxillin co-localization sites may indeed be focal adhesions, we next disrupted focal adhesions in wild-type mouse primary GCs using a FAK inhibitor, PF-573228 (PF). We performed a proximity ligation assay (PLA) to evaluate protein–protein interactions between AR and paxillin (Fig. 3A). In this assay, the secondary antibodies used for immunofluorescence are conjugated to complementary oligonucleotides, which bind each other and produce a fluorescent signal only when the two primary antibodies are in close proximity. The interaction of AR with DNMT1 was included as the negative control. We found that PXN and AR significantly interact in GCs, and that PF strongly disrupts this interaction (Fig. 3A). As expected, PF inhibited FAK phosphorylation and PXN phosphorylation (Fig. 3B, Supplementary Fig. S5). Notably, AR protein content also decreased in PF-treated cells, suggesting that paxillin’s regulation of AR may be dependent on focal adhesions. Likewise, although to a smaller degree, we observed AR–PXN interaction in KGN cells (Fig. 3C), which was present in both untreated (vehicle; VEH) and DHT-treated cells, and inhibited by PF. No interaction was detected in paxillin-negative control cells, which had undergone clustered regularly interspaced short palindromic repeats (CRISPR)-Cas9 gene editing to delete PXN, confirming antibody specificity.
Figure 3.
Androgen receptor and paxillin interaction requires intact granulosa cell focal adhesions. (A) Proximity ligation assay (PLA) in primary mouse GCs treated for 24 h with 5 μM PF-573228 (PF) or DMSO (VEH) and probed with antibodies against AR and PXN or AR and DNMT as negative control. Protein proximity signal produces red fluorescence, merged with DAPI indicating cell nuclei. Right: red fluorescence was quantified in five randomly selected cells in each sample using Image J and analyzed with Mann–Whitney tests. Magnification: ×40. **P < 0.01. (B) Representative western blot and band densitometry of primary mouse GCs (n = 3–6 mice per group) treated for 24 h with 5 μM PF-573228 (PF) or DMSO (VEH) and analyzed by Mann–Whitney tests. *P < 0.05. (C) PLA assay in KGN cells edited with CRISPR to delete paxillin (PXN KO) or non-targeting control (NT CTRL) and treated with vehicle (VEH), 25 nM dihydrotestosterone (DHT) or 5 μM PF-573228 (PF). All samples were probed with PXN and AR antibodies. Protein proximity signal produces red fluorescence, merged with DAPI indicating cell nuclei. Scale bar: 10 μm. AR: androgen receptor, PXN: paxillin, DNMT: DNA methyltransferase 1, GC: granulosa cells, VEH: vehicle, PF: PF-573228, FAK: focal adhesion kinase, CRISPR: clustered regularly interspaced short palindromic repeats, PXN KO: paxillin knockout, NT CTRL: non-targeting control. Uncropped blots are shown in Supplementary Fig. S5.
Human GCs compensate for PXN KO-mediated AR deficiency
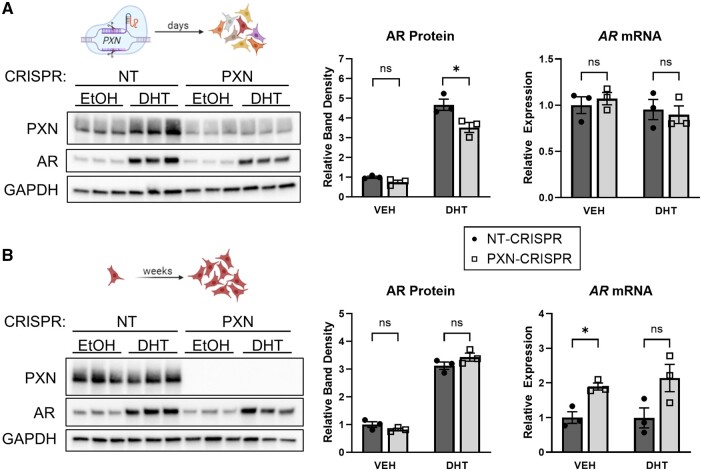
To evaluate the long-term effects of paxillin loss in GCs, we developed stable paxillin knockout clonal cell lines using the CRISPR-Cas9 system with PXN-specific (PXN-KO) or non-targeting control (NT CTRL) sgRNA in KGN cells. As previously published, PXN-KO KGN cells proliferate normally in culture (Vann et al., 2023). CRISPR-transfected cells were selected using GFP flow cytometry, then AR protein content was measured 7 days after transfection. In these polyclonal PXN-KO cells (Fig. 4A, Supplementary Fig. S6), there was some persistent PXN expression, and AR protein was less affected than in transient PXN knockdown KGN cells (Fig. 1). Only cells treated with DHT, which stabilizes AR protein levels (Astapova et al., 2021), showed a significantly reduced AR expression after PXN knockout (Fig. 4A, Supplementary Fig. S6). Similar to our findings in the transient knockdown and Cre-mediated GC-specific knockout (Fig. 1), AR mRNA expression was unchanged in KGN cells shortly after CRISPR-Cas9 deletion of paxillin (Fig. 4A). We then generated PXN-null cell lines by clonal expansion of single cells. After several weeks of clonal selection and multiple passages, AR protein content normalized to control levels in paxillin-null KGN clonal lines, while AR mRNA increased significantly in VEH-treated cells and tended to increase but did not reach statistical significance in DHT-treated cells (Fig. 4B, Supplementary Fig. S6). This suggests that GCs may compensate for chronic loss of AR protein levels by increasing mRNA transcription.
Figure 4.
Androgen receptor expression is rescued after chronic paxillin loss in CRISPR-Cas9-generated paxillin-null KGN clones. (A): KGN cells were transfected with paxillin-targeting or non-targeting control sgRNA and CRISPR-Cas9 lentivirus (PXN-CRISPR or NT-CRISPR, respectively). Successfully transfected cells were selected using GFP-positive flow cytometry 2 days later, cultured for 5 additional days, then treated with 0.1% ethanol (VEH) or 25 nM dihydrotestosterone (DHT) for 24 h before western blotting and qPCR. Band densitometry was analyzed by Mann–Whitney tests. (B) Clonal populations were obtained from single CRISPR-edited KGN cells by expansion in culture over a period of several weeks, then treated and analyzed the same way as in (A). *P < 0.05, ns: not significant. Pictures drawn in Biorender. CRISPR: clustered regularly interspaced short palindromic repeats, PXN: paxillin, NT: non-targeting control, EtOH: ethanol, DHT: dihydrotestosterone, AR: androgen receptor, VEH: vehicle, GFP: green fluorescent protein. Uncropped blots are shown in Supplementary Fig. S6.
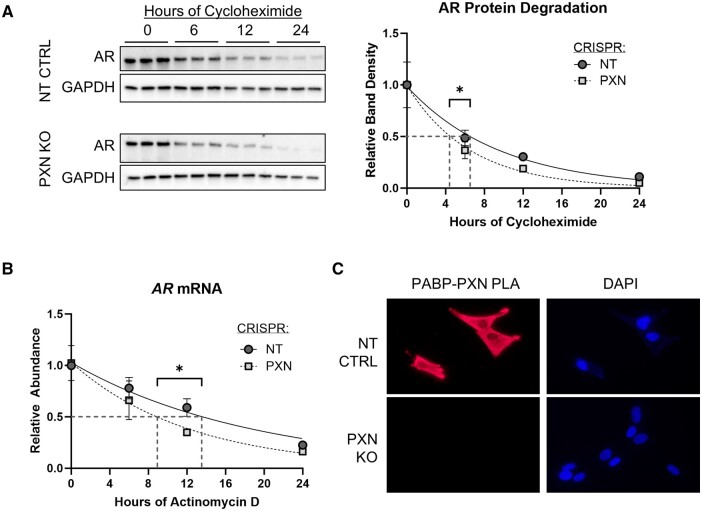
Paxillin prolongs AR protein and mRNA half-lives in human GCs
To better understand the mechanisms by which PXN regulates AR expression in GCs, we next measured the effect of paxillin on the half-lives of AR protein and mRNA. We used cycloheximide and actinomycin D to halt protein and mRNA synthesis, respectively, and measured AR degradation over 24 h. We found that both AR protein and mRNA degrade more quickly in the absence of paxillin: AR protein half-life is reduced from 6.7 to 4.5 h (Fig. 5A and Supplementary Fig. S7), and AR mRNA half-life is reduced from 13.1 to 8.5 h (Fig. 5B) in PXN-KO KGN cells compared with NT-CTRL KGN cells. We repeated this experiment using a different set of clonal cell lines and found similar results (Supplementary Fig. S8). Paxillin has been shown to interact with polyadenylation binding protein (PABP) in Xenopus levis oocytes to increase the mRNA translation of key reproductive genes (Miedlich et al., 2017). To further investigate the relationship between PXN and AR mRNA in GCs, we assessed the interaction between PXN and PABP in KGN cells. We used the PLA assay to show that PXN and PABP interact significantly in NT-CTRL KGN cells, while this interaction is absent in PXN-KO KGN cells, confirming assay specificity (Fig. 5C). These results suggest that paxillin prolongs both AR protein and mRNA half-lives, likely through different mechanisms, as discussed below.
Figure 5.
Paxillin prolongs androgen receptor mRNA and protein half-lives. (A) CRISPR-Cas9-generated clonal paxillin-knockout (PXN KO) and non-targeting control (NT CTRL) KGN cells were treated with 50 μM cycloheximide for indicated times before western blotting. Band densitometry was fitted to a one-phase decay with least squares fit, analyzed by ANOVA and protein half-life was calculated by GraphPad Prism. (B) AR mRNA was measured by qPCR from cells generated as in (A) and treated with 5 μM actinomycin D for up to 24 h, then analyzed the same way as in (A) to determine the mRNA half-life. (C) PLA assay of PABP and PXN and DAPI staining in PXN KO and NT CTRL samples. Red fluorescent signal represents PABP and PXN proximity. Magnification: ×40. NT CTRL: non-targeting control, PXN KO: paxillin knockout, AR: androgen receptor, CRISPR: clustered regularly interspaced short palindromic repeats, PABP: poly-A binding protein, PLA: proximity ligation assay, DAPI: DAPI: 4',6-diamidino-2-phenylindole. Uncropped blots are shown in Supplementary Fig. S7.
PXN loss in GCs partially rescues estrous cyclicity in a PCOS mouse model
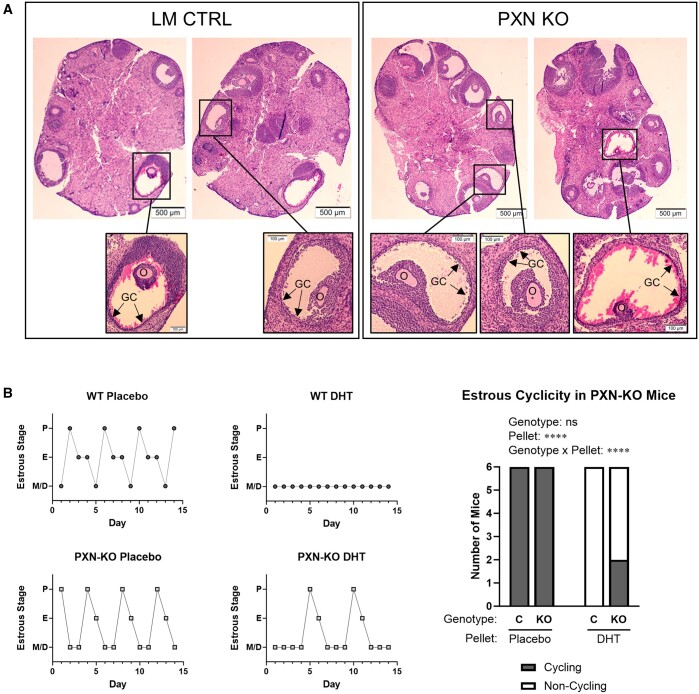
Since paxillin enhances AR expression in GCs, we sought to determine whether the GC-PXN KO mouse model’s AR-deficient GCs would be protective in androgen excess-related diseases such as PCOS. We implanted female C57BL/6J 19-day-old GC-PXN KO mice and female Cre–/– littermate controls (LM CTRLs) with s.c. pellets containing either 2.5 mg DHT or placebo. This treatment was developed by van Houten et al. and extensively characterized by that group and others to induce a PCOS-like phenotype in mice, including anovulation, cystic follicles, and metabolic derangements (van Houten et al., 2012; Caldwell et al., 2014; Xue et al., 2018; Rodriguez Paris et al., 2021). To confirm the PCOS-like ovarian morphology in this mouse model, we examined several ovarian sections from one PXN KO and one LM CTRL mouse treated with DHT and observed no corpora lutea and abundant cystic follicles, characterized by thin GC layers and degenerating oocytes, in both genotypes (Fig. 6A). We then evaluated estrous cyclicity at 76–90 days after pellet insertion. All placebo-treated mice demonstrated normal estrous cycles, defined as proestrus followed by estrus (Fig. 6B). As we previously published (Vann et al., 2023), PXN KO had no effect on cyclicity in placebo-treated mice. As expected, all LM CTRL mice treated with DHT were in persistent diestrus, demonstrating complete acyclicity. Remarkably, two out of six DHT-treated knockout mice exhibited at least one estrous cycle (Fig. 6B). This suggests that PXN loss in GCs may partially rescue anovulation induced by chronic DHT exposure by reducing GC AR expression and consequently reducing GC sensitivity to excess androgens.
Figure 6.
Reproductive effects of granulosa cell-specific paxillin knockout in a mouse PCOS model. GC-specific paxillin knockout mice (PXN-KO) and littermate controls (WT) were injected with s.c. pellets containing placebo or 2.5 mg DHT on postnatal Day 19. (A) Representative ovary sections stained with hematoxylin and eosin from adult PXN-KO and WT mice treated with DHT as above. High-magnification insets show cystic follicles characterized by thin GC layers (labeled GC) and degenerating oocytes (labeled O). No corpora lutea were observed in any of the ovary sections. (B) Estrous stage was determined daily at 76–90 days after pellet insertion via visual cytology assessment of vaginal lavage. Left, representative estrous cycles. P: proestrus; E: estrus; M/D: metestrus or diestrus. Right, number of mice that experienced at least one estrous cycle (‘Cycling’, defined as proestrus followed immediately by estrus) during the 14-day cycling study. Data were analyzed by Fisher’s exact test (C versus KO and placebo versus DHT) and by Mantel–Haenszel Chi-square test (interaction of pellet treatment and genotype). ns: not significant; ****P < 0.0001. LM CTRL: littermate control, PXN KO: paxillin knockout, GC: granulosa cells, O: oocyte, WT: wild-type, P: proestrus, E: estrus, M/D: metestrus/diestrus, C: control, KO: knockout, DHT: dihydrotestosterone.
Estradiol production is increased in GCs from PXN KO mice with DHT-induced PCOS
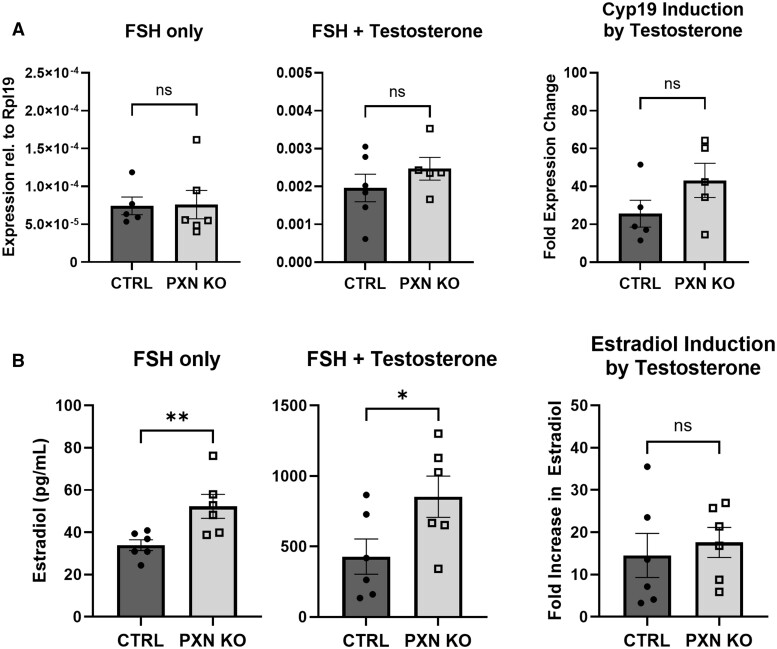
To determine whether PXN KO affects steroidogenesis in PCOS GCs, we measured the expression of aromatase (Cyp19) and estradiol production in primary GCs isolated from mice with DHT-induced PCOS. GCs from PXN KO and LM CTRL mice exposed to chronic DHT treatment as described above were cultured for 2 days, then stimulated with FSH with or without testosterone (T), which is the substrate for aromatase-mediated estradiol production. After 2 days of treatment, Cyp19 gene expression was dramatically induced by T, but was similar in PXN KO and LM CTRL GCs (Fig. 7A). However, estradiol production was increased in the PXN KO GCs compared with LM CTRL GCs cultured with and without T (Fig. 7B). The relative increases in both Cyp19 and estradiol production after treatment with T were similar in PXN KO and LM CTRL GCs. These findings suggest that PXN KO in mouse GCs may be protective from PCOS-related defects in steroidogenesis, and this may result in improved estrous cyclicity, as above. However, since the response to T was not affected by PXN KO, these effects may occur through indirect cellular mechanisms, rather than directly through reduced AR expression.
Figure 7.
Granulosa cell-specific paxillin knockout increases estradiol production in mice with dihydrotestosterone-induced PCOS. (A): Cyp19 gene expression was measured by qPCR in primary GCs from littermate control (CTRL) and GC paxillin knockout (PXN KO) mice cultured for 4 days and treated as indicated (FSH: 100 ng/ml, Testosterone: 100nM) during the last 2 days of culture. (B) Estradiol concentration was measured in the culture media collected from cells described in (A). All comparisons were analyzed by Mann–Whitney test; ns: not significant; *P < 0.05; **P < 0.01. Rpl19: ribosomal protein L19, CTRL: control, PXN KO: paxillin knockout.
Discussion
Although paxillin is primarily known as a focal adhesion protein, recent research has begun to reveal its role in modulation of steroid signaling. Paxillin has been implicated in androgen signaling in several cell types (Sen et al., 2014; Ma et al., 2019), but the extent and mechanism of this interaction is poorly understood. Our findings show that paxillin modulates AR expression in GCs. We showed for the first time that AR co-localizes with paxillin at the plasma membrane, associated with focal adhesions in GCs. We also found that paxillin enhances AR protein in GCs and extends both AR protein and mRNA half-lives. Additionally, we found that a GC-specific PXN knockout in mice may be protective against the reproductive consequences of androgen excess, such as in PCOS. Therefore, paxillin regulation of AR may have physiological relevance, and paxillin may be a potential target in developing novel PCOS therapies.
We found that acute paxillin loss, shown using siRNA transfection and early CRISPR-modified KGN cells, decreases AR protein content. This effect may be specific to AR: the expression of ERα was not reduced after paxillin knockdown in KGN cells. AR protein was also decreased in GCs from GC-PXN KO mice, where we have previously shown that paxillin is depleted late in the life cycle of a follicle (Vann et al., 2023). Our previous study also showed that PXN knockout does not affect GC viability or proliferation and does not increase apoptosis (Vann et al., 2023). In wild-type mouse GCs, AR protein was reduced after inhibition of FAK, which also disrupted AR–paxillin co-localization in focal adhesions. Notably, AR mRNA levels were unaffected by paxillin loss in all of the above experimental models. Taken together, these data show that paxillin enhances AR expression at the protein level. We hypothesize that part of this effect may be caused by protection of AR from proteasomal degradation by sequestering it near focal adhesions.
Our studies suggest that paxillin may regulate AR through multiple mechanisms. In addition to AR protein, paxillin also extends AR mRNA half-life in our experiments. The mechanism by which this occurs may involve enhanced stabilization of the mRNA molecule through the interaction of paxillin and PABP. This mechanism was reported in In X. laevis oocytes, where paxillin was found to elongate the poly-A tail and enhance translation of Mos mRNA, a key gene in oocyte maturation, through interaction with PABP (Miedlich et al., 2017). Accordingly, we confirmed co-localization of these two proteins through the PLA assay in primary mouse GCs, pointing toward polyadenylation as a potential mechanism by which paxillin enhances AR mRNA stability.
Despite the role of paxillin in AR mRNA stability, cells with paxillin loss through knockdown or knockout maintain normal expression of AR mRNA, suggesting that mRNA synthesis is increased to compensate for its accelerated degradation. In fact, in our clonal selection and propagation of paxillin-null KGN cell lines, we observed that chronic and complete loss of paxillin resulted in increased AR mRNA levels relative to non-targeted controls, whereas AR protein was normalized to control levels. These results suggest that a compensation mechanism exists to ensure a certain level of AR expression in these cancer-derived cells. AR-dependent proliferation has been demonstrated in several cancer cell models, including castration-sensitive prostate cancer (Culig et al., 1994; Migliaccio et al., 2006; Sen et al., 2010, 2011) and, notably, ER-negative breast cancer, where AR is upregulated and may be a treatment target (Barton et al., 2015; He et al., 2017; Christenson et al., 2021). KGN cells may therefore demonstrate a compensatory activation of AR gene transcription, which drives their survival in the face of chronic paxillin loss.
While steroid hormone receptors are classically known to be nuclear receptors that exert their actions by regulating gene transcription, growing evidence over the past two decades points to the presence of these receptors at or near the cell membrane, where they exert rapid signaling which can act in concert with their nuclear actions (Mauvais-Jarvis et al., 2021). There has been increasing focus on non-classical AR actions caused by AR activation at the cell membrane (Evaul et al., 2007; Lange et al., 2007; Hammes and Levin, 2011), where it can promote growth factor signaling. AR is reported to associate with membrane-bound proteins, such as caveolin-1 in prostate cancer and Sertoli cells (Lu et al., 2001; Deng et al., 2017), two cell types in which membrane-associated androgen signaling is crucial to cellular function (Cheng et al., 2007). There have also been increasing reports of membrane-bound AR variants. These variants are typically encoded by distinct genes and are very different in structure from the nuclear (classical) AR, with many lacking N-terminal or DNA-binding domains and some categorized as ion channels or G-protein coupled receptors (Yang et al., 2011; Garza-Contreras et al., 2017; Mauvais-Jarvis et al., 2021). These variants are not expected to be recognized by the AR antibodies used in our studies. AR and its variants have been reported to be present and have biological activity in lipid rafts in the cell membrane (Garza-Contreras et al., 2017; Grolez et al., 2019; Aflatounian et al., 2020). Membrane-associated AR and possibly some AR variants are responsible for rapid, non-transcriptional steroid hormone signaling, and this signaling has been shown to have differential yet critical roles in androgen action and cellular function, particularly in the context of prostate cancer (Sen et al., 2012; Li et al., 2018), where paxillin mediates these important non-classical actions of AR (Sen et al., 2010). We previously showed that AR has minimal genomic activity in GCs (Astapova et al., 2021), suggesting that a major part of its effect in GCs, similar to prostate cancer cells, may be nongenomic. This is the first report of AR interacting with membrane structures that appear and function as focal adhesions based on our previous studies of paxillin co-localization with the focal adhesion marker vinculin (Vann et al., 2023), and the first study to identify membrane-associated AR in GCs. Of note, while focal adhesions are traditionally known as cell–cell contacts, we only observed these structures in cell–coverslip contacts. In our samples, we are unable to visualize cell–cell focal adhesions either with paxillin or AR immunofluorescence. We presume these cell–coverslip contact structures to be focal adhesions based on previous literature showing similar appearing paxillin-containing structures in cultured cells that are not in direct contact with each other (Cao et al., 2016; Yang et al., 2016; Legerstee and Houtsmuller, 2021). Furthermore, AR localization near the cell membrane is ablated with either the loss of paxillin or the inhibition of FAK, suggesting that paxillin plays a key role in this interaction, similar to the role of caveolin-1 in Sertoli and prostate cancer cells. Future studies are needed to determine whether this paradigm contributes to nongenomic androgen signaling in GCs.
We found PXN knockout in GCs to reduce AR expression; therefore, we sought to determine whether this downregulation of AR would be protective in androgen excess disorders. We recently reported that GC-specific PXN knockout increases fecundity in female mice (Vann et al., 2023). Here, we found that after chronic prepubertal DHT exposure, 33% of GC-specific PXN KO mice exhibited at least one estrous cycle, in comparison to wild-type DHT-treated mice, which in our experience and in other reports (van Houten et al., 2012; Aflatounian et al., 2020; Cox et al., 2020; Rodriguez Paris et al., 2021) are completely acyclic. This is especially striking because chronic postnatal exposure to DHT, a potent and non-aromatizable androgen, is a more extreme model of PCOS relative to other models such as prenatal androgenization or exposure to weaker androgens, such as dehydroepiandrosterone (Caldwell et al., 2014). Therefore, we speculate that this protective effect may be even more pronounced in other models of PCOS. We postulate that a decreased expression of ARs in androgen-sensitive reproductive tissues, such as the GC, provides resistance to the reproductive consequences of androgen excess; in this case, acyclicity. On the other hand, although estradiol production was higher both under basal and T-stimulated conditions, T treatment did not result in a higher relative induction of Cyp19 or higher relative increase in estradiol production in PXN-knockout GCs. This suggests that the effects of PXN knockout on estradiol production may not be through direct effects on AR stimulation by T, but rather through other, indirect pathways affecting estradiol production.
In summary, paxillin loss in our studies is beneficial against the negative effects of both aging (Vann et al., 2023) and androgen excess on female fertility. This protection may be linked to reduced GC oxidative stress (Gozin et al., 1998; Mahdi et al., 2000), which involves paxillin activation downstream of nuclear factor-κB signaling (Zhang et al., 2015) and is associated with PCOS in humans (González, 2012). The interaction between paxillin and AR near GC focal adhesions may represent an important target for modulating folliculogenesis in fertility treatments.
Supplementary Material
Author Biography
 Dr. Olga Astapova: Dr. Astapova is an assistant professor at the University of Rochester Medical Center (Rochester, NY, USA). She received her MD and PhD from Wayne State University in Detroit, MI, USA, before training in Internal Medicine and Endocrinology at the University of Rochester Medical Center. Dr. Astapova is a physician–scientist who specializes in disorders of sex steroids, including polycystic ovary syndrome (PCOS) and hypogonadism, as well as gender-affirming hormone therapy. She runs a basic research laboratory studying androgen actions in the ovary using a mouse model of PCOS and actively participates in education and mentoring of trainees at multiple levels.
Dr. Olga Astapova: Dr. Astapova is an assistant professor at the University of Rochester Medical Center (Rochester, NY, USA). She received her MD and PhD from Wayne State University in Detroit, MI, USA, before training in Internal Medicine and Endocrinology at the University of Rochester Medical Center. Dr. Astapova is a physician–scientist who specializes in disorders of sex steroids, including polycystic ovary syndrome (PCOS) and hypogonadism, as well as gender-affirming hormone therapy. She runs a basic research laboratory studying androgen actions in the ovary using a mouse model of PCOS and actively participates in education and mentoring of trainees at multiple levels.
Contributor Information
Adelaide E Weidner, Division of Endocrinology, Department of Medicine, University of Rochester Medical Center, Rochester, NY, USA.
Anna Roy, Division of Endocrinology, Department of Medicine, University of Rochester Medical Center, Rochester, NY, USA.
Kenji Vann, Division of Endocrinology, Department of Medicine, University of Rochester Medical Center, Rochester, NY, USA.
Ariana C Walczyk, Division of Endocrinology, Department of Medicine, University of Rochester Medical Center, Rochester, NY, USA.
Olga Astapova, Division of Endocrinology, Department of Medicine, University of Rochester Medical Center, Rochester, NY, USA.
Supplementary data
Supplementary data are available at Molecular Human Reproduction online.
Data availability
All data are available upon request from the corresponding author.
Authors’ roles
A.E.W.: major part of experimentation and manuscript writing. A.R.: protein and RNA degradation experiments, estrous cycling study. K.V.: immunofluorescence and proximity ligation assays, mouse colony management, and estrous cycling study. A.C.W.: proximity ligation assays, western blotting, and qPCR. O.A.: experimental design, immunofluorescence, western blotting, qPCR, and manuscript editing.
Funding
National Institutes of Health NICHD Award Number 1K08HD107134-01A1 and University of Rochester Department of Medicine Pilot Award Program.
Conflict of interest
The authors disclose no conflicts of interest.
References
- Aflatounian A, Edwards MC, Rodriguez Paris V, Bertoldo MJ, Desai R, Gilchrist RB, Ledger WL, Handelsman DJ, Walters KA.. Androgen signaling pathways driving reproductive and metabolic phenotypes in a PCOS mouse model. J Endocrinol 2020;245:381–395. [DOI] [PubMed] [Google Scholar]
- Alpha KM, Xu W, Turner CE.. Paxillin family of focal adhesion adaptor proteins and regulation of cancer cell invasion. Int Rev Cell Mol Biol 2020;355:1–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astapova O, Minor BMN, Hammes SR.. Physiological and pathological androgen actions in the ovary. Endocrinology 2019;160:1166–1174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astapova O, Seger C, Hammes SR.. Ligand binding prolongs androgen receptor protein half-life by reducing its degradation. J Endocr Soc 2021;5:bvab035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azziz R, Carmina E, Chen Z, Dunaif A, Laven JS, Legro RS, Lizneva D, Natterson-Horowtiz B, Teede HJ, Yildiz BO.. Polycystic ovary syndrome. Nat Rev Dis Primers 2016;2:16057. [DOI] [PubMed] [Google Scholar]
- Balen AH, Conway GS, Kaltsas G, Techatrasak K, Manning PJ, West C, Jacobs HS.. Andrology: polycystic ovary syndrome: the spectrum of the disorder in 1741 patients. Hum Reprod 1995;10:2107–2111. [DOI] [PubMed] [Google Scholar]
- Barton VN, D’Amato NC, Gordon MA, Lind HT, Spoelstra NS, Babbs BL, Heinz RE, Elias A, Jedlicka P, Jacobsen BM. et al. Multiple molecular subtypes of triple-negative breast cancer critically rely on androgen receptor and respond to enzalutamide in vivo. Mol Cancer Ther 2015;14:769–778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berni TR, Morgan CL, Aled Rees D.. Women with polycystic ovary syndrome have an increased risk of major cardiovascular events: a population study. J Clin Endocrinol Metab 2021;106:E3369–E3380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown MC, Perrotta JA, Turner CE.. Identification of LIM3 as the principal determinant of paxillin focal adhesion localization and characterization of a novel motif on paxillin directing vinculin and focal adhesion kinase binding. J Cell Biol 1996;135:1109–1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byers SL, Wiles MV, Dunn SL, Taft RA.. Mouse estrous cycle identification tool and images. PLoS One 2012;7:e35538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldwell ASL, Middleton LJ, Jimenez M, Desai R, McMahon AC, Allan CM, Handelsman DJ, Walters KA.. Characterization of reproductive, metabolic, and endocrine features of polycystic ovary syndrome in female hyperandrogenic mouse models. Endocrinology 2014;155:3146–3159. [DOI] [PubMed] [Google Scholar]
- Cao L, Zhang L, Zhao X, Zhang Y.. A hybrid chalcone combining the trimethoxyphenyl and isatinyl groups targets multiple oncogenic proteins and pathways in hepatocellular carcinoma cells. PLoS One 2016;11:e0161025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng J, Watkins SC, Walker WH.. Testosterone activates mitogen-activated protein kinase via Src kinase and the epidermal growth factor receptor in sertoli cells. Endocrinology 2007;148:2066–2074. [DOI] [PubMed] [Google Scholar]
- Christenson JL, O’Neill KI, Williams MM, Spoelstra NS, Jones KL, Trahan GD, Reese J, Van Patten ET, Elias A, Eisner JR. et al. Activity of combined androgen receptor antagonism and cell cycle inhibition in androgen receptor positive triple negative breast cancer. Mol Cancer Ther 2021;20:1062–1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox MJ, Edwards MC, Rodriguez Paris V, Aflatounian A, Ledger WL, Gilchrist RB, Padmanabhan V, Handelsman DJ, Walters KA.. Androgen action in adipose tissue and the brain are key mediators in the development of PCOS traits in a mouse model. Endocrinology 2020;161:bqaa061. [DOI] [PubMed] [Google Scholar]
- Culig Z, Hobisch A, Cronauer M, Radmayr C, Trapman J, Hittmair A, Bartsch G, Klocker H.. Androgen receptor activation in prostatic tumor cell lines by insulin-like growth factor-I, keratinocyte growth factor, and epidermal growth factor. Cancer Res 1994;54:5474–5478. [PubMed] [Google Scholar]
- Deng Q, Wu Y, Zhang Z, Wang Y, Li M, Liang H, Gui Y.. Androgen receptor localizes to plasma membrane by binding to caveolin-1 in mouse sertoli cells. Int J Endocrinol 2017;2017:3985916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dennett CC, Simon J.. The role of polycystic ovary syndrome in reproductive and metabolic health: overview and approaches for treatment. Diabetes Spectr 2015;28:116–120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evaul K, Jamnongjit M, Bhagavath B, Hammes SR.. Testosterone and progesterone rapidly attenuate plasma membrane Gβγ-mediated signaling in Xenopus laevis oocytes by signaling through classical steroid receptors. Mol Endocrinol 2007;21:186–196. [DOI] [PubMed] [Google Scholar]
- Garza-Contreras J, Duong P, Snyder BD, Schreihofer DA, Cunningham RL.. Presence of androgen receptor variant in neuronal lipid rafts. eNeuro 2017;4:ENEURO.0109-0117.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleicher N, Kim A, Weghofer A, Kushnir VA, Shohat-Tal A, Lazzaroni E, Lee H-J, Barad DH.. Hypoandrogenism in association with diminished functional ovarian reserve. Hum Reprod 2013;28:1084–1091. [DOI] [PubMed] [Google Scholar]
- González F. Inflammation in polycystic ovary syndrome: underpinning of insulin resistance and ovarian dysfunction. Steroids 2012;77:300–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gozin A, Franzini E, Andrieu V, Da Costa L, Rollet-Labelle E, Pasquier C.. Reactive oxygen species activate focal adhesion kinase, paxillin and P130CAS tyrosine phosphorylation in endothelial cells. Free Radic Biol Med 1998;25:1021–1032. [DOI] [PubMed] [Google Scholar]
- Grolez GP, Gordiendko DV, Clarisse M, Hammadi M, Desruelles E, Fromont G, Prevarskaya N, Slomianny C, Gkika D.. TRPM8-androgen receptor association within lipid rafts promotes prostate cancer cell migration. Cell Death Dis 2019;10:652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammes SR, Levin ER.. Minireview: recent advances in extranuclear steroid receptor actions. Endocrinology 2011;152:4489–4495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He L, Du Z, Xiong X, Ma H, Zhu Z, Gao H, Cao J, Li T, Li H, Yang K. et al. Targeting androgen receptor in treating HER2 positive breast cancer. Sci Rep 2017;7:14584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoeger KM, Dokras A, Piltonen T.. Update on PCOS: consequences, challenges, and guiding treatment. J Clin Endocrinol Metab 2021;106:e1071–e1083. [DOI] [PubMed] [Google Scholar]
- Hu Y-C, Wang P-H, Yeh S, Wang R-S, Xie C, Xu Q, Zhou X, Chao H-T, Tsai M-Y, Chang C.. Subfertility and defective folliculogenesis in female mice lacking androgen receptor. Proc Natl Acad Sci U S A 2004;101:11209–11214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasai M, Guerrero-Santoro J, Friedman R, Leman E, Getzenberg R, DeFranco D.. The Group 3 LIM domain protein paxillin potentiates androgen receptor transactivation in prostate cancer cell lines. Cancer Res 2003;63:4927–4935. [PubMed] [Google Scholar]
- Lange CA, Gioeli D, Hammes SR, Marker PC.. Integration of rapid signaling events with steroid hormone receptor action in breast and prostate cancer. Annu Rev Physiol 2007;69:171–199. [DOI] [PubMed] [Google Scholar]
- Legerstee K, Houtsmuller AB.. A layered view on focal adhesions. Biology (Basel) 2021;10:1189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Fu X, Cao S, Li J, Xing S, Li D, Dong Y, Cardin D, Park H-W, Mauvais-Jarvis F. et al. Membrane-associated androgen receptor (AR) potentiates its transcriptional activities by activating heat shock protein 27 (HSP27). J Biol Chem 2018;293:12719–12729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu ML, Schneider MC, Zheng Y, Zhang X, Richie JP.. Caveolin-1 interacts with androgen receptor: a positive modulator of androgen receptor mediated transactivation. J Biol Chem 2001;276:13442–13451. [DOI] [PubMed] [Google Scholar]
- Ma X, Biswas A, Hammes SR.. Paxillin regulated genomic networks in prostate cancer. Steroids 2019;151:108463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma X, Hammes SR.. Paxillin actions in the nucleus. Steroids 2018;133:87–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Y, Andrisse S, Chen Y, Childress S, Xue P, Wang Z, Jones D, Ko C, Divall S, Wu S.. Androgen receptor in the ovary theca cells plays a critical role in androgen-induced reproductive dysfunction. Endocrinology 2017;158:98–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahdi MHB, Andrieu V, Pasquier C.. Focal adhesion kinase regulation by oxidative stress in different cell types. IUBMB Life 2000;50:291–299. [DOI] [PubMed] [Google Scholar]
- Mauvais-Jarvis F, Lange CA, Levin ER.. Membrane-initiated estrogen, androgen, and progesterone receptor signaling in health and disease. Endocr Rev 2021;43:720–742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLean AC, Valenzuela N, Fai S, Bennett SAL.. Performing vaginal lavage, crystal violet staining, and vaginal cytological evaluation for mouse estrous cycle staging identification. J Vis Exp 2012;e4389. doi: 10.3791/4389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miedlich SU, Taya M, Young MR, Hammes SR.. Paxillin and embryonic PolyAdenylation Binding Protein (ePABP) engage to regulate androgen-dependent Xenopus laevis oocyte maturation—a model of kinase-dependent regulation of protein expression. Mol Cell Endocrinol 2017;448:87–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Migliaccio A, Castoria G, Domenico MD, Ciociola A, Lombardi M, De Falco A, Nanayakkara M, Bottero D, De Stasio R, Varricchio L. et al. Crosstalk between EGFR and extranuclear steroid receptors. Ann N Y Acad Sci 2006;1089:194–200. [DOI] [PubMed] [Google Scholar]
- Rodriguez Paris V, Edwards MC, Aflatounian A, Bertoldo MJ, Ledger WL, Handelsman DJ, Gilchrist RB, Walters KA.. Pathogenesis of reproductive and metabolic PCOS traits in a mouse model. J Endocr Soc 2021;5:bvab060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider CA, Rasband WS, Eliceiri KW.. NIH image to ImageJ: 25 years of image analysis. Nat Methods 2012;9:671–675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen A, De Castro I, Defranco DB, Deng FM, Melamed J, Kapur P, Raj GV, Rossi R, Hammes SR.. Paxillin mediates extranuclear and intranuclear signaling in prostate cancer proliferation. J Clin Invest 2012;122:2469–2481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen A, Hammes SR.. Granulosa cell-specific androgen receptors are critical regulators of ovarian development and function. Mol Endocrinol 2010;24:1393–1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen A, O’Malley K, Wang Z, Raj GV, Defranco DB, Hammes SR.. Paxillin regulates androgen- and epidermal growth factor-induced MAPK signaling and cell proliferation in prostate cancer cells. J Biol Chem 2010;285:28787–28795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen A, Prizant H, Hammes SR.. Understanding extranuclear (nongenomic) androgen signaling: What a frog oocyte can tell us about human biology. Steroids 2011;76:822–828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen A, Prizant H, Light A, Biswas A, Hayes E, Lee H-J, Barad D, Gleicher N, Hammes SR.. Androgens regulate ovarian follicular development by increasing follicle stimulating hormone receptor and microRNA-125b expression. Proc Natl Acad Sci U S A 2014;111:3008–3013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shohat-Tal A, Sen A, Barad DH, Kushnir V, Gleicher N.. Genetics of androgen metabolism in women with infertility and hypoandrogenism. Nat Rev Endocrinol 2015;11:429–441. [DOI] [PubMed] [Google Scholar]
- Tian Y, Shen W, Lai Z, Shi L, Yang S, Ding T, Wang S, Luo A.. Isolation and identification of ovarian theca-interstitial cells and granulose cells of immature female mice. Cell Biol Int 2015;39:584–590. [DOI] [PubMed] [Google Scholar]
- van Houten EL, Kramer P, McLuskey A, Karels B, Themmen AP, Visser JA.. Reproductive and metabolic phenotype of a mouse model of PCOS. Endocrinology 2012;153:2861–2869. [DOI] [PubMed] [Google Scholar]
- Vann K, Weidner AE, Walczyk AC, Astapova O.. Paxillin knockout in mouse granulosa cells increases fecundity. Biol Reprod 2023;109:669–683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walters KA, Middleton LJ, Joseph SR, Hazra R, Jimenez M, Simanainen U, Allan CM, Handelsman DJ.. Targeted loss of androgen receptor signaling in murine granulosa cells of preantral and antral follicles causes female subfertility1. Biol Reprod 2012;87:151. [DOI] [PubMed] [Google Scholar]
- Xue P, Wang Z, Fu X, Wang J, Punchhi G, Wolfe A, Wu S.. A hyperandrogenic mouse model to study polycystic ovary syndrome. J Vis Exp 2018;e58379. doi: 10.3791/58379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang C, DelRio FW, Ma H, Killaars AR, Basta LP, Kyburz KA, Anseth KS.. Spatially patterned matrix elasticity directs stem cell fate. Proc Natl Acad Sci U S A 2016;113:E4439–4445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X, Guo Z, Sun F, Li W, Alfano A, Shimelis H, Chen M, Brodie AMH, Chen H, Xiao Z. et al. Novel membrane-associated androgen receptor splice variant potentiates proliferative and survival responses in prostate cancer cells. J Biol Chem 2011;286:36152–36160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L-L, Mu G-G, Ding Q-S, Li Y-X, Shi Y-B, Dai J-F, Yu H-G.. Phosphatase and tensin homolog (PTEN) represses colon cancer progression through inhibiting paxillin transcription via PI3K/AKT/NF-κB pathway. J Biol Chem 2015;290:15018–15029. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are available upon request from the corresponding author.