Abstract
Background
The rotator cuff (RC) plays a pivotal role in the performance and health of the shoulder and upper extremity. Blood flow restriction training (BFRT) is a modality to improve strength and muscle hypertrophy with even low-load training in healthy and injured individuals. There is minimal evidence examining its effect proximal to the occluded area, and particularly on the RC.
Hypothesis & Purpose
The purpose of this case series is to explore the effects of low-load BFRT on RC strength, hypertrophy, and tendon thickness in asymptomatic individuals.
Study Design
Case series.
Methods
Fourteen participants with asymptomatic, untrained shoulders were recruited to participate. They performed an eight-week low-load shoulder exercise regimen where BFR was applied to the dominant arm only during exercise. The dependent variables were maximal isometric strength of the shoulder external rotators(ER) and elevators (in the scapular plane in full can position) (FC) measured via handheld dynamometry, cross sectional area (CSA) of the supraspinatus and infraspinatus muscles, and supraspinatus tendon thickness measured via ultrasound imaging (US). Mean changes within and between arms were compared after training using paired t-tests. Cohen’s d was used to determine effect sizes.
Results
All participants were able to complete the BFRT regimen without adverse effects. Mean strength and CSA increased for all variables in both arms, however this increase was only significant (p<0.01) for FC strength bilaterally and CSA for the supraspinatus and infraspinatus on the BFRT side. The effect sizes for increased supraspinatus and infraspinatus CSA on the BFRT side were 0.40 (9.8% increase) and 0.46 (11.7% increase) respectively. There were no significant differences when comparing the mean changes of the BFRT side to the non-BFRT side for strength or muscle CSA. There were no significant changes to supraspinatus tendon thickness.
Conclusion
These results suggest variability in response of the RC musculature to low-load BFRT in asymptomatic individuals. The potential for a confounding systemic response in the study design makes determining whether low-load BFRT is more beneficial than low-load non-BFRT difficult. The hypertrophy seen on the BFRT side warrants further study.
Level of Evidence
4
Keywords: blood flow restriction training, BFRT, cross-sectional area, rotator cuff, shoulder
INTRODUCTION
Shoulder pain impacts about 12% of the population, with a variable range of 26% - 81% in high risk populations where rotator cuff (RC) tendinopathy accounts for roughly half of cases.1–3 The rotator cuff plays a pivotal role in the performance of the glenohumeral joint particularly in athletes where it acts to stabilize the humerus during high-load activity.4 Improved rotator cuff performance has been associated with improved outcomes in patients with rotator cuff pathology and following shoulder surgery.5–7
Blood flow restriction training (BFRT) has been performed in the healthy population for over 40 years with recent literature demonstrating relatively strong evidence for its efficacy in developing muscle hypertrophy and force production in healthy individuals with low-load training.8,9 Amplification of the training effects of low-loads may be particularly valuable in a clinical population who are unable to generate higher loads secondary to weakness or inhibition, or in whom high loads would be unsafe in consideration of healing tissues. There is a growing body of evidence indicating the safety and efficacy of generating hypertrophic and strength increases in patient populations, predominantly with lower extremity injury or after lower extremity surgical procedures.10–12 Use of BFRT in sports clinical practice and training communities for the lower and upper extremity has become increasingly popular.
The primary mechanism for muscular adaptation is increased activation of type IIx muscle fibers at lower mechanical loads than normally required via hypoxia locally induced by an inflated specialized pressure cuff at the proximal portion of the limb.13 Secondary mechanisms include mechanotransduction effects from venous pooling and a cascade of local and systemic hormonal responses.14
Some evidence has indicated advantageous muscular adaptations proximal to the placement of the pressure cuff.15–19 Recent authors have indicated shoulder strength gains following BFRT regimens in healthy individuals, but results have been mixed with relatively small effects observed.15,18–21 Additionally, there is early evidence to suggest hypertrophy of shoulder musculature occurs with low-load training in combination with shoulder BFRT,19–21 however the methods used included girth measurement and whole shoulder DEXA scan which are not gold standard measures for muscle hypertrophy and are not specific to the rotator cuff. Existing studies in this area have not examined the effect of BFRT on RC muscle cross-sectional area (CSA).
There is even less evidence exploring the use of BFRT for conditions of the shoulder in patients. To the authors’ knowledge there are only three peer-reviewed articles on the use of BFRT in patients with shoulder problems – one case series in patients following shoulder stabilization surgery,22 one case report on a patient with adhesive capsulitis and rotator cuff involvement,23 and one case report on two patients with subacromial pain.24 Overall, there is limited evidence regarding the effects of upper extremity BFRT on the shoulder, and a further paucity of evidence on its effects specific to the rotator cuff. The lack of clear evidence in this domain combined with reported benefit in patients with lower extremity involvement warrants further investigation. The purpose of this case series is to explore the effects of low-load blood flow restriction training on RC strength, hypertrophy, and tendon thickness.
MATERIALS & METHODS
Study Design
This case series examines the effects of blood flow restriction training of the shoulder on RC musculature hypertrophy and isometric strength in adults with asymptomatic, untrained shoulders. Participants underwent an exam to exclude those for whom upper extremity exercise or BFRT might be unsafe. They performed RC isometric strength testing via fixed dynamometry, and RC muscle CSA measurement via US imaging, before and after an 8-week progressive exercise regimen (Figure 1). Participants performed the exercise regimen with both arms, while BFR was applied proximally, to the dominant arm only. All participants provided informed consent to participate in this study, which was approved by an institutional review board.
FIGURE 1. Study design timeline.
Strength & Hypertrophy Measurement
Isometric Dynamometry
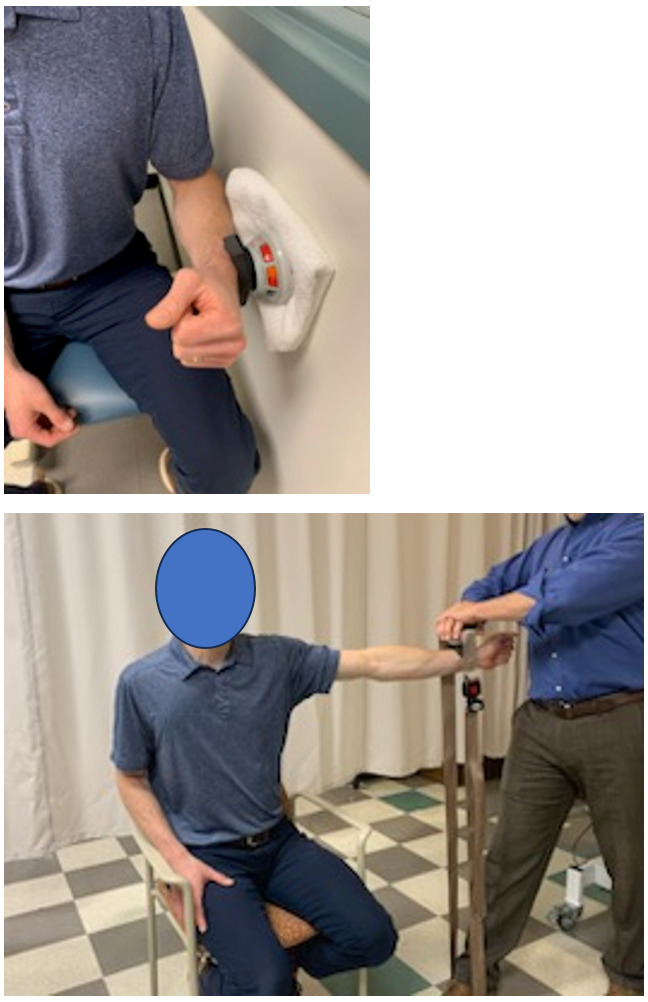
Participants’ maximal volitional isometric contraction (MVIC) strength for external rotators (ER) and elevators (in the full can position) (FC) was assessed with fixed dynamometry using a handheld dynamometer stabilized by a wall or inelastic strap (Figure 2) on both arms. For ER measurement participants were seated upright with their arm at their side, elbow bent to 90 degrees, and the forearm in neutral rotation. The dynamometer was aligned to the ulnar styloid process. For FC measurement, participants were seated with the arm elevated to 90 degrees in the plane of the scapula (40 degrees anterior to the frontal plane) with the dynamometer aligned to the radial styloid process. Participants performed two submaximal trials at 50% effort and then rested for 1 minute. Participants then performed two maximal effort trials with three minutes of rest between each.25 If there was greater than a 10% difference between the two maximal efforts a third maximal effort was performed. Results of maximal efforts were averaged for data and exercise regimen purposes detailed below. Shoulder dynamometry has been shown to have good reliability.26–28
FIGURE 2. Isometric dynamometry for shoulder external rotation (ER) against a wall (top) and elevation in the scapular plane (FC) against an inelastic belt (bottom).
Ultrasound Imaging
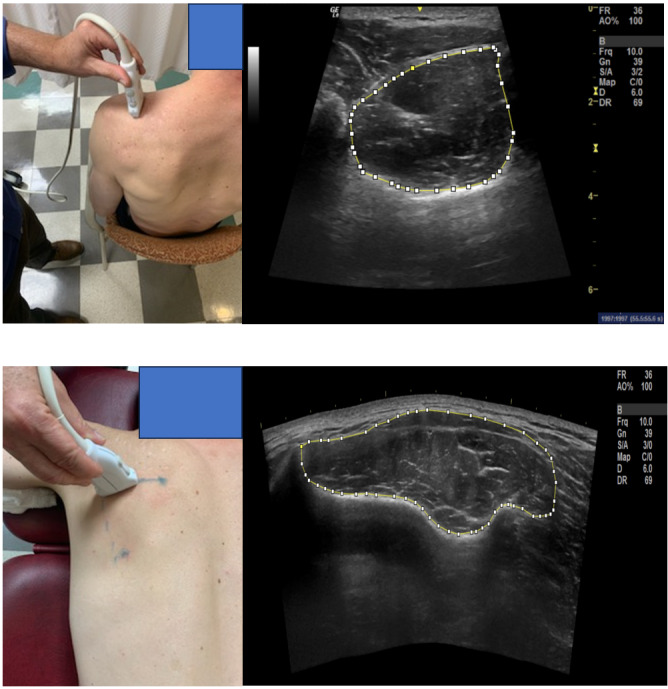
Supraspinatus and infraspinatus CSA and supraspinatus tendon thickness of both shoulders was obtained using GE Logiq-e B-mode ultrasound (GE Healthcare, Wisconsin, USA) with a high resolution, multi-frequency (8-13MHz) linear transducer. The participant was seated comfortably on a stool with the arm held in different positions for the supraspinatus muscle thickness, and tendon thickness measurements.29 The participant was prone for the infraspinatus muscle thickness measurement (Figure 3). At least three scans were performed by an individual trained in the study methods for each structure and an average of three measurements for each structure was used for data analyses. ImageJ [version 1.45s (NIH, Bethesda, MD)] computerized image analysis program was used for size measurements, performed by a different single trained individual. These methods of US data collection have demonstrated good reliability.30
FIGURE 3. Ultrasound Imaging and cross-sectional area (CSA) measurement examples – supraspinatus (top) and infraspinatus (bottom).
Exercise Regimen
Participants performed two common clinical shoulder exercises – sidelying external rotation and standing scaption (elevation in the scapular plane) – on both arms (Figure 4). The sidelying external rotation was performed with a small towel roll under the arm and scaption was performed to 90 degrees of elevation. Participants performed the exercises two times per week with at least one day of rest between sessions for eight weeks. Participants were allowed to miss three sessions from the protocol, and if they missed two sessions consecutively, they completed an additional week of exercise. Participants performed four sets of each exercise – the first set was 30 repetitions, and the remaining sets were 15 repetitions – all with 30 seconds of rest between sets. If a participant failed to complete all repetitions in a set due to fatigue they took the rest allotted and continued to the next set. The exercises were performed with dumbbells at 20% of the average MVIC performed by the participants which was re-assessed every two weeks and loads were adjusted accordingly. The ER MVIC determined the weight for sidelying ER exercise, and the FC MVIC determined the weight for the scaption exercise. The dumbbell selection was rounded to the nearest pound.
FIGURE 4. Sidelying shoulder external rotation (top) and scaption (bottom) exercises. The same exercises were performed without the BFR cuff on the non-dominant arm.

Blood Flow Restriction Protocol
Participants performed the exercise regimen with a Delfi PTS Personalized Tourniquet System II BFR cuff applied to the dominant upper arm at 50% arterial occlusion pressure (AOP). Participants were monitored for excessive pain (>7/10 on a numeric pain rating scale (NPRS)), excessive rate of perceived exertion (RPE) (>8/10), lack of capillary refill, loss of sensation, or any negative systemic response. If these occurred, the pressure was reduced by 10% and exercise was continued. If the symptoms persisted, the session would be ended for the day, however this did not occur during the study. The exercise and blood flow restriction parameters were based on previous studies.8,10,31
Data Analysis
Data was analyzed using Microsoft Excel 2016 MSO (16.0.4266.1001). Descriptive data were represented using means (SD). Paired, two-tailed t-tests were used to assess changes within the same arm pre versus post training, and between arms after training. Cohen’s d (mean difference divided by pooled SD) was used to determine effect size of within group (same arm) changes.
RESULTS
Participants
Fourteen participants were enrolled in the study. All participants completed the study and there were no adverse events. The cuff pressure was reduced by 10% for four participants due to excessive RPE (>8/10) during sessions early in the protocol or when increasing resistance. They were all able to continue with the exercise regimen and returned to ideal cuff pressure in subsequent sessions. The mean age (SD) of the participants was 30.7 (14.8), two were left hand dominant, and three were male.
Strength, Hypertrophy, and Tendon Thickness
The data for strength, hypertrophy, and tendon thickness are shown in Table 1 and Table 2. The mean increase in strength was statistically significant for FC (p<0.01) in both arms. The mean change in strength for ER increased in both arms, but the differences were not statistically significant. There were no statistically significant strength gain differences between the non-BFRT and BFRT sides.
Table 1. Pre- and Post-training data for strength, cross-sectional area and tendon thickness for BFRT and non BFRT arms.
| Within Arm | Mean(SD) Before | Mean(SD) After | Mean Difference | % Change | Effect Size | p-value |
|---|---|---|---|---|---|---|
| Strength (lb) | ||||||
| ER BFRT side | 21.1(4.8) | 22.9(6.8) | 1.8 | 8.5% | 0.31 | 0.135 |
| ER non-BFRT side | 20.0(5.4) | 22.0(6.7) | 2.0 | 10.0% | 0.33 | 0.118 |
| FC BFRT side | 19.2(7.6) | 20.5(8.0) | 1.2 | 6.3% | 0.16 | 0.011 |
| FC non-BFRT side | 18.9(7.6) | 20.7(7.1) | 1.8 | 9.6% | 0.25 | 0.001 |
| Cross-Sectional Area (mm2) | ||||||
| Supraspinatus BFRT side | 892(176) | 979(203) | 88 | 9.8% | 0.46 | 0.004 |
| Supraspinatus non-BFRT side | 888(179) | 943(175) | 55 | 6.2% | 0.31 | 0.093 |
| Infraspinatus BFRT side | 1778(509) | 1987(534) | 207 | 11.7% | 0.40 | 0.009 |
| Infraspinatus non-BFRT side | 1808(653) | 1862(482) | 55 | 3.0% | 0.10 | 0.088 |
| Tendon Thickness (mm2) | ||||||
| Supraspinatus BFRT side | 6.7(1.3) | 6.6(0.8) | -0.1 | -1.9% | -0.12 | 0.472 |
| Supraspinatus non-BFRT side | 6.8(0.6) | 6.7(1.0) | -0.1 | -1.6% | -0.13 | 0.572 |
SD = standard deviation, ER = external rotation, FC = full can / elevation in the scapular plane, BFRT = blood flow restricted training, lb = pounds, mm2 = millimeters squared
Table 2. Mean differences between arms due to training.
| Between Arms | Mean Difference | p-value |
|---|---|---|
| Strength (lb) | ||
| External Rotation | -0.2 | 0.890 |
| Full Can | -0.6 | 0.184 |
| Cross-Sectional Area (mm2) | ||
| Supraspinatus | 33 | 0.470 |
| Infraspinatus | 152 | 0.156 |
| Tendon Thickness (mm2) | ||
| Supraspinatus | -0.02 | 0.460 |
Mean difference is non-BFRT side subtracted from BFRT side. lb = pounds, mm2 = millimeters squared
Mean cross-sectional area increased for both the supraspinatus and infraspinatus muscles in both arms, however the increase was only statistically significant (p<0.01) in the BFRT side. The effect sizes (cohen’s d) for increased supraspinatus and infraspinatus CSA on the BFRT side were 0.40 (9.8% increase) and 0.46 (11.7% increase) respectively. The increased mean CSA on the BFRT side was 88mm2 for the supraspinatus and 207mm2 for the infraspinatus. These values are in the range of existing MDC95 for the supraspinatus (70-130mm2) but not the infraspinatus (290-370mm2).30 There were no significant differences in mean change between arms for either muscle. There were no significant changes to supraspinatus tendon thickness on either side. Due to imaging error, data from only 10 participants was included in the tendon analysis.
DISCUSSION
Strength increased relatively symmetrically in the BFRT and non-BFRT arms, and only FC strength gains were statistically significantly different. This could represent a relatively expected response of untrained musculature to training, neural adaptation, motor learning from the biweekly testing, or a systemic response to the BFRT. Other studies examining RC strengthening with BFRT have produced mixed results.18–21 Interestingly, most of these studies showed a similar pattern where strength improved in multi-joint movements such as FC, flexion, or bench press that involve the deltoid and pectoralis musculature where isolated rotation such as ER did not.17,19–21
Increases in muscle hypertrophy were only statistically significant on the BFRT side for both the supraspinatus and infraspinatus. The effect size and percentage increases were 0.40 (9.8% increase) and 0.46 (11.7% increase) respectively, which are moderate and similar to expected increases for traditional high-load or low-load progressive resistance training, and low-load blood flow restriction training in musculature distal to the cuff.8,17,32,33 The difference between sides was not significant.
There was no statistically significant change in tendon thickness on either side. Since the participants had asymptomatic shoulders and likely minimal to no pathology there may not have been much opportunity for change. It seems that use of shoulder BFRT is not deleterious to healthy rotator cuff tendon and further study is warranted in various patient populations.
The results of this study present a confounding comparison. There was significantly increased muscle CSA only in the BFRT side, while there were no between side differences for strength change. There is an apparent decoupling of hypertrophy and strength gain. A primary confounder and limitation of this study is that the BFRT performed on one side may have had a systemic cross over effect boosting the response to the loading stimulus in the non-BFRT side. There is conflicting evidence on the systemic effect of low-load BFRT34 wherein there is an endocrine response similar to high-load training particularly with a large increase in human growth hormone and insulin growth factors.35,36 This is also a mechanism proposed to contribute to advantageous tissue response to BFRT proximal to the cuff.19 The study design cannot tease out the potential impact of this effect. So, while the study design controls for a number of between-individual comparison factors it may be possible that a systemic hormonal response boosted strength and hypertrophy in the non-BFRT side that might not have occurred if it were performed in a different individual.
There are several other theories that have been presented to explain hypertrophy and strength gains proximal to the BFR cuff. Common ones include increased EMG activation of proximal musculature, remote ischemic preconditioning, and a backflow effect where vascular pressure builds proximal to the cuff.13–15,37,38 Both shoulders were untrained and there were no significant differences between sides at baseline despite the mean strength being 5% and 1.5% lower on the nondominant side for ER and FC respectively. Even so, the slightly higher mean strength gain on the non-BFRT side may be somewhat due to an inherently less trained state since it was the non-dominant side with greater potential for neural adaptation with training which is also a limitation of this research. These mechanisms and factors may contribute to the apparent decoupling of hypertrophy and strength gain between sides.
There are additional limitations to this research. While the parameters used for the BFRT regimen are commonly recommended, the 20%1RM is the lower end of what is reported as effective in the literature. That said, the potential for gains with low-load training is part of what makes BFRT potentially advantageous for clinical practice. Similarly, to achieve clinical feasibility, training was performed by rounding dynamometry measurements to the nearest pound for dumbbell use. This may have blunted training intensity increases that should have been associated with small strength gains found at the biweekly strength re-assessments. Furthermore, increased exercise volume might have produced more pronounced effects.
Another limitation is that the use of an isometric strength measure to approximate the resistance for isotonic exercise may carry inherent error. However, the effect of this is likely small and the repeated assessment and matched progression of resistance in the exercise regimen would likely minimize this.
While reliability of this study’s measurement tools is strong,26,27,29,30,39–42 a higher number of participants may have demonstrated a clearer between side difference for CSA. Furthermore, most participants were female and there is conflicting evidence to suggest that the menstrual cycle has an impact on response to strength training.43–45 This was not controlled and so may be a confounding factor. However, it is unlikely that this would be controlled in a current clinical environment.
This study was performed in individuals with asymptomatic untrained shoulders. Those with trained shoulders or patients with symptomatic shoulders and/or local pathology may respond differently. There may be other neurosensory and psychological mechanisms and benefits related to application of BFRT that were not directly examined in this study.
The exercise regimen of this study was designed to be simple and clinically feasible. The frequency, exercise selection, and equipment used aside from the BFR device are all very common in clinical practice for the shoulder. Various BFR devices are also becoming more common and accessible in clinical environments. When applied appropriately, BFRT has been reportedly relatively safe,22,23,31,46,47 and in this study all participants were able to complete the study without adverse events. Given these factors, it seems likely that a similar regimen could be applied in clinical practice.
CONCLUSION
This study and others examining the response of RC tissue to BFRT have produced mixed results.17–21,37 This study is the first to examine the hypertrophic response specifically of the supraspinatus and infraspinatus to low-load BFRT as measured by US. The potential for a confounding systemic response makes determining if low-load BFRT is more beneficial than low-load non-BFRT in asymptomatic untrained shoulders difficult. However, the hypertrophy gains on the BFRT side along with strength gains in a similar pattern to other studies are encouraging and warrant further study.
Conflict of Interest
All authors, their immediate family, and any research foundation with which they are affiliated did not receive any financial payments or other benefits from any commercial entity related to the subject of this article.
References
- Incidence of shoulder pain in repetitive work. Leclerc A., Chastang J. F., Niedhammer I., Landre M. F., Roquelaure Y. 2004Occup Environ Med. 61(1):39–44. [PMC free article] [PubMed] [Google Scholar]
- Periarthritis of the shoulder. Trial of treatments investigated by multivariate analysis. Lee P. N., Lee M., Haq A. M., Longton E. B., Wright V. 1974Ann Rheum Dis. 33(2):116–9. doi: 10.1136/ard.33.2.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The relationship between training load and injury in men's professional basketball. Weiss K. J., Allen S. V., McGuigan M. R., Whatman C. S. 2017Int J Sports Phys Perf. 12(9):1238–42. doi: 10.1123/ijspp.2016-0726. doi: 10.1123/ijspp.2016-0726. [DOI] [PubMed] [Google Scholar]
- Mechanisms of rotator cuff tendinopathy: intrinsic, extrinsic, or both? Seitz A. L., McClure P. W., Finucane S., Boardman N. D., 3rd, Michener L. A. 2011Clinical Biomech. 26(1):1–12. doi: 10.1016/j.clinbiomech.2010.08.001. doi: 10.1016/j.clinbiomech.2010.08.001. [DOI] [PubMed] [Google Scholar]
- Does strength deficit correlate with shoulder function in patients with rotator cuff tears? Characteristics of massive tears. Oh J. H., Yoon J. P., Kim D. H.., et al. 2019J Shoulder Elbow Surg. 28(10):1861–68. doi: 10.1016/j.jse.2019.03.015. doi: 10.1016/j.jse.2019.03.015. [DOI] [PubMed] [Google Scholar]
- Prognostic factors affecting clinical outcomes after arthroscopic rotator cuff repair: importance of functional recovery by 3 months after surgery. Nakamura Y., Gotoh M., Mitsui Y., et al. 2018J Orthop Surg Res. 13(1):310. doi: 10.1186/s13018-018-1014-8. doi: 10.1186/s13018-018-1014-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehabilitation of shoulder impingement syndrome and rotator cuff injuries: an evidence-based review. Ellenbecker T. S., Cools A. 2010Br J Sports Med. 44(5):319–27. doi: 10.1136/bjsm.2009.058875. doi: 10.1136/bjsm.2009.058875. [DOI] [PubMed] [Google Scholar]
- The efficacy of blood flow restricted exercise: A systematic review & meta-analysis. Slysz J., Stultz J., Burr J.F. 2016J Sci Med Sport. 19(8):669–75. doi: 10.1016/j.jsams.2015.09.005. doi: 10.1016/j.jsams.2015.09.005. [DOI] [PubMed] [Google Scholar]
- Low intensity blood flow restriction training: a meta-analysis. Loenneke J. P., Wilson J. M., Marin P. J., Zourdos M. C., Bemben M. G. 2012Eur J Appl Physiol. 112(5):1849–59. doi: 10.1007/s00421-011-2167-x. doi: 10.1007/s00421-011-2167-x. [DOI] [PubMed] [Google Scholar]
- Blood flow restriction training in clinical musculoskeletal rehabilitation: a systematic review and meta-analysis. Hughes L., Paton B., Rosenblatt B., Gissane C., Patterson S. D. 2017Brit J Sports Med. 51(13):1003–11. doi: 10.1136/bjsports-2016-097071. doi: 10.1136/bjsports-2016-097071. [DOI] [PubMed] [Google Scholar]
- Blood flow restriction training after knee arthroscopy: A randomized controlled pilot study. Tennent D. J., Hylden C. M., Johnson A. E., Burns T. C., Wilken J. M., Owens J. G. 2017Clinical J Sport Med. 27(3):245–52. doi: 10.1097/jsm.0000000000000377. doi: 10.1097/jsm.0000000000000377. [DOI] [PubMed] [Google Scholar]
- Use and safety of KAATSU training: Results of a national survey. Nakajima T., KM, Iida H., Takano H.., et al. 2006Inter J Kaatsu Train Res. 2:5–13. doi: 10.3806/ijktr.2.5. [DOI] [Google Scholar]
- Blood flow restriction: the metabolite/volume threshold theory. Loenneke J. P., Fahs C. A., Wilson J. M., Bemben M. G. 2011Med Hypoth. 77(5):748–52. doi: 10.1016/j.mehy.2011.07.029. doi: 10.1016/j.mehy.2011.07.029. [DOI] [PubMed] [Google Scholar]
- A review on the mechanisms of blood-flow restriction resistance training-induced muscle hypertrophy. Pearson S. J., Hussain S. R. 2015Sports Med. 45(2):187–200. doi: 10.1007/s40279-014-0264-9. doi: 10.1007/s40279-014-0264-9. [DOI] [PubMed] [Google Scholar]
- The effects of blood flow restriction on upper-body musculature located distal and proximal to applied pressure. Dankel S. J., Jessee M. B., Abe T., Loenneke J. P. 2016Sports Med. 46(1):23–33. doi: 10.1007/s40279-015-0407-7. [DOI] [PubMed] [Google Scholar]
- Skeletal muscle size and circulating IGF-1 are increased after two weeks of twice daily 'KAATSU' resistance training. Abe T., YT, Midorikawa T., Sato Y.., et al. 2005Inter J Kaatsu Train Res. 200(1):6–12. doi: 10.3806/ijktr.1.6. [DOI] [Google Scholar]
- The effects of upper body blood flow restriction training on muscles located proximal to the applied occlusive pressure: A systematic review with meta-analysis. Pavlou K., Korakakis V., Whiteley R., Karagiannis C., Ploutarchou G., Savva C. 2023PLoS One. 18(3):e0283309. doi: 10.1371/journal.pone.0283309. doi: 10.1371/journal.pone.0283309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blood flow restriction training for the rotator cuff: a randomized controlled trial. Brumitt J., Hutchison M. K., Kang D., et al. 2020Inter J Sports Phys Perf. 15(8):1175–80. doi: 10.1123/ijspp.2019-0815. doi: 10.1123/ijspp.2019-0815. [DOI] [PubMed] [Google Scholar]
- Blood flow restriction training for the shoulder: A case for proximal benefit. Lambert B., Hedt C., Daum J., et al. 2021Am J Sports Med. 49(10):2716–28. doi: 10.1177/03635465211017524. doi: 10.1177/03635465211017524. [DOI] [PubMed] [Google Scholar]
- Upper-extremity blood flow restriction: the proximal, distal, and contralateral effects-a randomized controlled trial. Bowman E. N., Elshaar R., Milligan H., et al. 2020J Shoulder Elbow Surg. 29(6):1267–74. doi: 10.1016/j.jse.2020.02.003. doi: 10.1016/j.jse.2020.02.003. [DOI] [PubMed] [Google Scholar]
- Rotator cuff training with upper extremity blood flow restriction produces favorable adaptations in division IA collegiate pitchers: a randomized trial. Lambert B. S., Hedt C., Ankersen J. P., et al. 2023J Shoulder Elbow Surg. 32(6):e279–e92. doi: 10.1016/j.jse.2023.02.116. doi: 10.1016/j.jse.2023.02.116. [DOI] [PubMed] [Google Scholar]
- The effect of blood flow restriction therapy on shoulder function following shoulder stabilization surgery: A case series. McGinniss J. H., Mason J. S., Morris J. B., Pitt W., Miller E. M., Crowell M. S. 2022Int J Sports Phys Ther. 17(6):1144–55. doi: 10.26603/001c.37865. doi: 10.26603/001c.37865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blood flow restriction training as an adjunct to conventional care in a patient with adhesive capsulitis and rotator cuff tendinopathy: A case report. Safford D. W., Shah K. M., McClure P. 2021J Orthop Sports Phys Ther Cases. 1(4):4. [Google Scholar]
- Resident case series: Blood flow restriction as an adjunct to strengthening exercises in two patients with subacromial impingement and high irritability. Ceballos A., Zeppieri G., Jr., Bialosky J. 2022Int J Sports Phys Ther. 17(5):931–40. doi: 10.26603/001c.36526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rest interval between sets in strength training. de Salles B. F., Simão R., Miranda F., da Silva Novaes J., Lemos A., Willardson J. 2009Sports Med. 39(9):765–77. doi: 10.2165/11315230-000000000-00000. doi: 10.2165/11315230-000000000-00000. [DOI] [PubMed] [Google Scholar]
- Concurrent validity and reliability of a handheld dynamometer in measuring isometric shoulder rotational strength. Chen B., Liu L., Bin Chen L.., et al. 2021J Sport Rehabil. 30(6):965–68. doi: 10.1123/jsr.2020-0021. doi: 10.1123/jsr.2020-0021. [DOI] [PubMed] [Google Scholar]
- Absolute reliability and concurrent validity of hand-held dynamometry in shoulder rotator strength assessment: Systematic review and meta-analysis. Chamorro C., Arancibia M., Trigo B., Arias-Poblete L., Jerez-Mayorga D. 2021Int J Environ Res Pub Health. 18(17) doi: 10.3390/ijerph18179293. doi: 10.3390/ijerph18179293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assessing the reliability of shoulder strength seasurement: hand-held versus fixed dynamometry. Beshay N., Lam P. H., Murrell G. A. C. 2011Shoulder Elbow. 3(4):244–51. doi: 10.1111/j.1758-5740.2011.00137.x. doi: 10.1111/j.1758-5740.2011.00137.x. [DOI] [Google Scholar]
- Relationship between skin intrinsic fluorescence--an indicator of advanced glycation end products-and upper extremity impairments in individuals with diabetes mellitus. Shah K. M., Clark B. R., McGill J. B., Lang C. E., Maynard J., Mueller M. J. 2015Phys Ther. 95(8):1111–9. doi: 10.2522/ptj.20140340. doi: 10.2522/ptj.20140340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [Poster Abstract] Reliability of ultrasound measurements of rotator cuff muscle cross sectional area in healthy adults. Shah K. M., Brennan M., Bucantis J., Kust E., Washatka J., McClure P. W. 2017J Orthop Sports Phys Ther. 47(1):A58–A161. doi: 10.2519/jospt.2017.47.1.A58. [DOI] [Google Scholar]
- Blood flow restriction therapy: An evidence-based approach to postoperative rehabilitation. Watson R., Sullivan B., Stone A.., et al. 2022JBJS Rev. 10(10) doi: 10.2106/jbjs.rvw.22.00062. doi: 10.2106/jbjs.rvw.22.00062. [DOI] [PubMed] [Google Scholar]
- Resistance exercise load does not determine training-mediated hypertrophic gains in young men. Mitchell C. J., Churchward-Venne T. A., West D. W.., et al. 2012J Appl Physiol (1985) 113(1):71–7. doi: 10.1152/japplphysiol.00307.2012. doi: 10.1152/japplphysiol.00307.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Resistance training load effects on muscle hypertrophy and strength gain: Systematic review and network meta-analysis. Lopez P., Radaelli R., Taaffe D.R., et al. 2021Med Sci Sports Exerc. 53(6):1206–16. doi: 10.1249/mss.0000000000002585. doi: 10.1249/mss.0000000000002585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blood flow-restricted exercise does not induce a cross-transfer of effect: A randomized controlled trial. Ampomah K., Amano S., Wages N. P., et al. 2019Med Sci Sports Exerc. 51(9):1817–27. doi: 10.1249/mss.0000000000001984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cross-transfer effects of resistance training with blood flow restriction. Madarame H., Neya M., Ochi E., Nakazato K., Sato Y., Ishii N. 2008Med Sci Sports Exerc. 40(2):258–63. doi: 10.1249/mss.0b013e31815c6d7e. doi: 10.1249/mss.0b013e31815c6d7e. [DOI] [PubMed] [Google Scholar]
- Comparison of hormone responses following light resistance exercise with partial vascular occlusion and moderately difficult resistance exercise without occlusion. Reeves G. V., Kraemer R. R., Hollander D. B., et al. 2006J Appl Physiol (1985) 101(6):1616–22. doi: 10.1152/japplphysiol.00440.2006. doi: 10.1152/japplphysiol.00440.2006. [DOI] [PubMed] [Google Scholar]
- Optimal blood flow restriction occlusion pressure for shoulder muscle recruitment with upper extremity exercise. Roehl T., Lambert B. S., Ankersen J., Hernandez K., McCulloch P. C., Hedt C. 2023Am J Sports Med. 51(7):1859–71. doi: 10.1177/03635465231166959. doi: 10.1177/03635465231166959. [DOI] [PubMed] [Google Scholar]
- Blood flow restriction enhances rehabilitation and return to sport: The paradox of proximal performance. Hedt C., McCulloch P. C., Harris J. D., Lambert B. S. 2022Arthrosc Sports Med Rehabil. 4(1):e51–e63. doi: 10.1016/j.asmr.2021.09.024. doi: 10.1016/j.asmr.2021.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Absolute reliability and concurrent validity of hand held dynamometry and isokinetic dynamometry in the hip, knee and ankle joint: Systematic review and meta-analysis. Chamorro C., Armijo-Olivo S., De la Fuente C., Fuentes J., Chirosa L. Javier. 2017Open Med. 12:359–75. doi: 10.1515/med-2017-0052. doi: 10.1515/med-2017-0052. published Online First: 2017/10/27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OP0180 Reliability and comparison of fixed versus handheld shoulder dynamometry in student physical therapists. Safford D. W., SK, Garant L. G.., et al. 2023J Orthop Sports Phys Ther. 53(2):CSM55–CSM146. doi: 10.2519/jospt.2023.53.2.CSM55. [DOI] [Google Scholar]
- High-frequency quantitative ultrasound imaging of human rotator cuff muscles: Assessment of repeatability and reproducibility. Wu Y., Barrere V., Ashir A., et al. 2023 [2023-11-20];Ultrason Imaging. :1617346231207404. doi: 10.1177/01617346231207404. doi: 10.1177/01617346231207404. [DOI] [PMC free article] [PubMed]
- Rehabilitative ultrasound imaging of the supraspinatus muscle: Intra- and interrater reliability of thickness and cross-sectional area. Schneebeli A., Egloff M., Giampietro A., Clijsen R., Barbero M. 2014J Bodyw Mov Ther. 18(2):266–72. doi: 10.1016/j.jbmt.2013.09.009. doi: 10.1016/j.jbmt.2013.09.009. [DOI] [PubMed] [Google Scholar]
- Current evidence shows no influence of women's menstrual cycle phase on acute strength performance or adaptations to resistance exercise training. Colenso-Semple L. M., D'Souza A. C., Elliott-Sale K. J., Phillips S. M. 2023Front Sports Act Living. 5:1054542. doi: 10.3389/fspor.2023.1054542. doi: 10.3389/fspor.2023.1054542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The resistance training effects of different weight level during menstrual cycle in female. Sung E. S., Kim J. H. 2019J Exerc Rehabil. 15(2):249–53. doi: 10.12965/jer.193808.024. doi: 10.12965/jer.193808.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The effects of menstrual cycle phase on exercise performance in eumenorrheic women: A systematic review and meta-analysis. McNulty K. L., Elliott-Sale K. J., Dolan E., et al 2020Sports Med. 50(10):1813–27. doi: 10.1007/s40279-020-01319-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The safety of blood flow restriction training as a therapeutic intervention for patients with musculoskeletal disorders: A systematic review. Minniti M. C., Statkevich A. P., Kelly R. L., et al. 2020Am J Sports Med. 48(7):1773–85. doi: 10.1177/0363546519882652. doi: 10.1177/0363546519882652. [DOI] [PubMed] [Google Scholar]
- Blood flow restriction exercise: considerations of methodology, application, and safety. Patterson S.D., Hughes L., Warmington S.., et al. 2019Front Physiol. 10:533. doi: 10.3389/fphys.2019.00533. doi: 10.3389/fphys.2019.00533. [DOI] [PMC free article] [PubMed] [Google Scholar]



