Abstract
Introduction
Clozapine is the only antipsychotic approved for treatment-resistant schizophrenia, but without appropriate monitoring, it can be associated with potentially fatal outcomes. An International Adult Clozapine Titration Guideline categorizes patients into normal or slow metabolizers. Categorization provides clozapine titration schedules and recommends regular c-reactive protein (CRP) and clozapine concentration monitoring to reduce the risk of adverse drug reactions (ADRs). The impact of the guideline on clozapine ADRs has not been evaluated.
Methods
A retrospective chart review assessed clozapine titrations, laboratory monitoring, ADRs, and discontinuations for clozapine-naive adult inpatients at a single center from January 1, 2013, to June 1, 2022. Each patient’s cumulative weekly clozapine dosage was compared with their guideline recommended dosage to create a percent accordance. Linear logistic regression evaluated the relationship between titration speed and the presence of an ADR, while descriptive statistics analyzed laboratory monitoring.
Results
Forty-three patients were included, with the majority being White males with schizophrenia. An inverse relationship existed between the last inpatient week clozapine dose percent accordance and the probability of an ADR. Nonobese patients were less likely than obese patients to experience an ADR (odds ratio = 0.17; 95% CI, 0.03-0.99). CRP and clozapine concentration monitoring was suboptimal.
Discussion
Based on our small retrospective review of primarily White males, more aggressive clozapine titrations did not increase ADRs. Future studies with more diverse samples are needed and should focus on specific ADRs, which may have increased occurrence with rapid titrations. Obese patients were at higher risk of ADRs, correlating with the guideline-recommended slower titrations for these patients.
Keywords: antipsychotic agents, clozapine, dose-response relationship, drug, drug-related side effects and adverse reactions, inflammation, schizophrenia spectrum and other psychotic disorders
Introduction
Clozapine is the only antipsychotic approved by the FDA for treatment-resistant schizophrenia and recurrent suicidal behavior in patients with schizophrenia or schizoaffective disorder.1,2 Off-label uses of clozapine include treatment-resistant bipolar disorder, Parkinson disease psychosis, and pediatric psychiatric disorders.3-5 Clozapine’s boxed warnings are severe neutropenia; increased mortality in dementia-related psychosis; orthostatic hypotension, bradycardia, and syncope; seizure; and myocarditis, cardiomyopathy, and mitral valve incompetence.1 Clozapine adverse drug reactions (ADRs) commonly lead to clozapine discontinuation and, in some cases, death,6 with pneumonia being identified as the leading cause of death worldwide in clozapine-treated patients and myocarditis as possibly second, depending on the country.7
Clozapine is primarily metabolized by cytochrome P450 (CYP) 1A2 and to a lesser extent via CYP2C19, CYP2D6, and CYP3A4.1,8 Genetic aspects can lead to altered clozapine pharmacokinetics due to reduced CYP1A2 metabolism, where patients from Asia (Pakistan to Japan) and indigenous people of the Americas may require lower clozapine doses compared with those of European Ancestry, and descendants of sub-Saharan African may require higher clozapine doses.9 Rapid clozapine titrations during initial exposure, particularly when coprescribed with metabolic inhibitors (eg, disulfiram, ciprofloxacin, estrogen, fluvoxamine), have been suggested to result in clozapine toxicity, which might lead to potentially fatal ADRs, such as myocarditis or sudden death.10-16 For drug–drug interactions (DDIs), valproate coprescription with clozapine needs clarification, as valproate can increase the risk of clozapine-induced myocarditis (odd ratio [OR] 2.78-4.11),11 while it is also shown to be both an inhibitor or inducer of clozapine metabolism.17,18 Genetic interpatient variability of CYP1A2 activity, DDIs, and clozapine-induced inflammation, among other factors, can also contribute to elevated clozapine levels and subsequent ADRs.13,18 Slower clozapine titrations may reduce the risk of severe adverse effects, as particularly low rates of clozapine-induced myocarditis have occurred in the Netherlands, who use very slow clozapine titrations.19,20 Recent comments have emerged in literature to modify the clozapine package insert to include ancestry-based titrations, update DDIs, and reassess the inclusion of other important ADRs (eg, pneumonia, inflammation).13,21 Many of these viewpoints are supported in the 2021 International Clozapine Titration Guideline (ICTG), which emphasizes individualized clozapine titrations based on ancestry, obesity status, and ICTG-specific DDIs (ie, valproate, fluvoxamine, estrogen). Additionally, the ICTG recommends weekly c-reactive protein (CRP) and clozapine concentration monitoring when available to improve patient safety.18 A recent retrospective review of Japanese clozapine naive inpatients found serious inflammatory adverse events reduced from 34% to 13% (P < 0.001) when using slower titrations, as recommended by the ICTG, compared with faster titration rates found in Japanese prescribing information.2 This retrospective chart review aimed to determine the probability of major ADRs based on accordance with inpatient clozapine dose titrations with the ICTG.18 This project also evaluated our institution’s clozapine laboratory monitoring compared with those recommended by the ICTG in efforts to highlight areas for pharmacists to combat clozapine underutilization in the United States.22,23
Methods
This institutional review board–approved project collected data from the electronic health record via the retrospective chart review of inpatients initiating their first lifetime exposure to clozapine at a single inpatient psychiatry unit from January 1, 2013, to June 1, 2022. The original search found 81 unique patients prescribed clozapine, with 43 of these patients being clozapine naive. Electronic health record data were collected for the 43 patients from index admission to 30 days postdischarge. Data extracted included age at admission, body mass index (BMI), clozapine concentrations, clozapine initiation date, CRP and troponin monitoring at baseline and weekly intervals, cumulative weekly clozapine dose received, clozapine indication, ICTG-specific DDIs (ie, valproate, fluvoxamine, estrogen), hospital discharge date, race, sex, smoking status before admission, and safety data, which included clozapine discontinuation for any reason, and major ADRs leading to clozapine-dose adjustment, discontinuation, or emergency medical evaluation. Major ADRs were included if they occurred during the inpatient clozapine titration or within 30 days of hospital discharge and included agranulocytosis/severe neutropenia, CRP elevation (≥ 3 mg/L), death, infection (eg, pneumonia), inflammatory conditions (eg, myocarditis, transaminitis, hypertroponinemia [≥ 0.4 ng/mL]), severe gastrointestinal hypomotility (eg, ileus, bowel obstruction or perforation), hypotension requiring medical intervention, severe sedation requiring further medical evaluation, and seizure.
Patients who were obese or concurrently on medications identified as causing ICTG-specific DDIs were categorized as slow clozapine metabolizers, and those without these characteristics were normal metabolizers as per the ICTG.18 A BMI of 30 kg/m2 or higher was used for obesity status stratification. Each patient’s clozapine dose administered during hospitalization was summed weekly and cumulatively for the entire hospitalization. For the primary outcome, clozapine dosages were compared with ICTG-recommended weekly and cumulative doses, which were calculated by averaging the fastest and slowest titrations for each specific ancestry and metabolizer status group. Percent accordance more than 100% indicated the patient’s dose was higher than the average ICTG-recommended dose based on either their last inpatient week of hospitalization or cumulative amount during the hospitalization. The primary outcome variable was the presence of an ADR. The following 4 binary logistic regression models were considered to determine the probability of ADRs:
Linear logistical regression was performed with R 4.2 Software to determine relationships of guideline-emphasized patient characteristics that impacted titration, which was used for similar research.24 ICTG-recommended laboratory monitoring was performed and evaluated with descriptive statistics.
Results
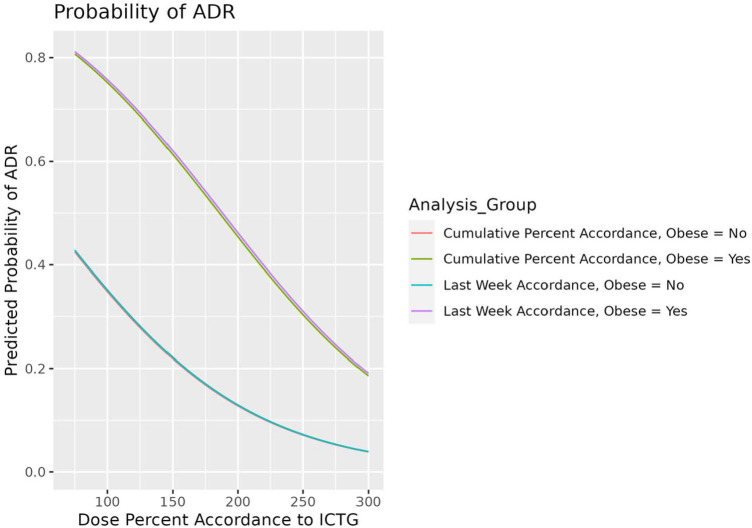
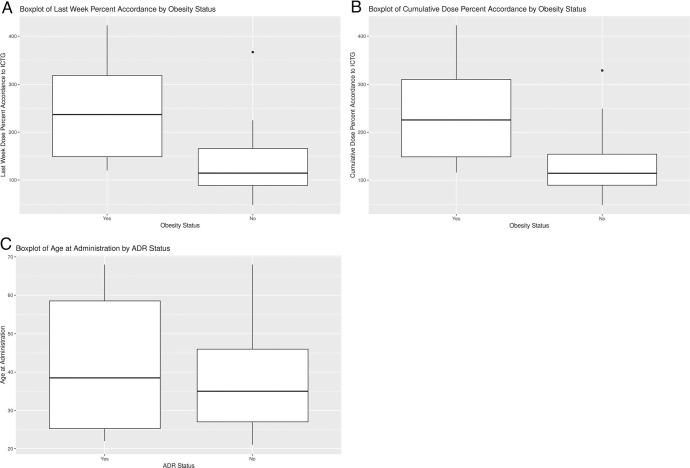
Forty-three patients were included for analysis, with the majority being nonobese males with schizophrenia or schizoaffective disorder and European/Western Asian ancestry (Table 1). Five patients were coprescribed valproate in the non-ADR group, compared with zero in the ADR group (Table 1). The mean BMI was 30.9 kg/m2 (SD 6.7) in the ADR group and 29.7 kg/m2 (SD 7.5) in the non-ADR group. The cumulative and last inpatient week clozapine mean dose percent accordance to ICTG recommendations were 177.3% (SD 92.9) and 180.4% (SD 95.9), respectively. An inverse relationship between both percent dose accordance models and the probability of ADRs was found (Figure 1). Patients with a BMI of 30 kg/m2 or higher had a higher percent clozapine dose accordance compared with those with a BMI of less than 30 kg/m2 (Figure 2A and B) and a higher probability of ADRs (Figure 1). Fourteen patients (32.6%) experienced an ADR, with CRP elevation being the most common (n = 8), where the mean CRP level was 11.2 mg/L (SD 8.8) (Table 2). ADRs, which resulted in clozapine discontinuation, were CRP elevation (n = 4), troponin elevation (n = 3), myocarditis (n = 2), transaminitis (n = 1), and severe sedation (n = 1) (Table 2).
TABLE 1.
Patient demographics, percent of weekly cumulative clozapine dose, and weekly laboratory monitoring in accordance with guideline recommendations
| ADR Patients (n = 14) |
Non-ADR Patients (n = 29) |
All Patients (N = 43) |
|
|---|---|---|---|
| Patient Characteristics | |||
| Mean Age in Years at Admission (SD) | 41.7 (17.2) | 38.9 (14.1) | 42.6 (14.4) |
| Mean BMI kg/m2 (SD) | 30.9 (6.7) | 29.7 (7.5) | 29.8 (7.3) |
| % BMI >30 kg/m2, n (%) | 7 (50) | 11 (37.9) | 18 (41.9) |
| Smoker, n (%) | 6 (42.9) | 16 (55.2) | 22 (51.2) |
| Female, n (%) | 6 (42.9) | 9 (31.1) | 15 (34.9) |
| Diagnosis | |||
| Schizophrenia, n (%) | 9 (64.3) | 11 (37.9) | 20 (46.5) |
| Schizoaffective Disorder, n (%) | 3 (21.4) | 17 (58.6) | 20 (46.5) |
| Bipolar Disorder, n (%) | 2 (14.3) | 1 (3.5) | 3 (7.0) |
| Ancestry | |||
| European/Western Asian, n (%) | 14 (100) | 28 (96.6) | 42 (97.7) |
| Native to the Americas, n (%) | 0 (0) | 1 (3.5) | 1 (2.3) |
| Coprescribed DDIs Mentioned in the ICTG | |||
| Valproate, n (%) | 0 (0) | 5 (17.2) | 5 (11.6) |
| Estrogen, n (%) | 0 (0) | 0 (0) | 0 (0) |
| Fluvoxamine, n (%) | 0 (0) | 0 (0) | 0 (0) |
| Metabolism Status | |||
| Average Metabolism, n (%) | 7 (50) | 15 (51.7) | 22 (51.2) |
| Poor Metabolism, n (%) | 7 (50) | 14 (48.3) | 21 (48.8) |
| Clozapine Dose Percent Accordance/Exposure with ICTG | |||
| Mean Inpatient Week 1. n; % accordance (SD) | 14; 142.1 (70.8) | 29; 181 (91.7) | 43; 168.3 (86.6) |
| Mean Inpatient Week 2. n; % accordance (SD) | 4; 118.4 (66.0) | 13; 232.7 (115.7) | 17; 205.8 (115.6) |
| Mean Inpatient Week 3. n; % accordance (SD) | 2; 125.3 (60.4) | 2; 239.8 (20.9) | 4; 182.5 (75.7) |
| Mean Inpatient Week 4. n; % accordance | 1; 82.5 | 0; N/A | 1; 82.5 |
| Mean Last Week Inpatient. n; % accordance (SD) | 14; 144.6 (65.9) | 29; 197.6 (106) | 43; 180.4 (95.9) |
| Cumulative Exposure for All Weeks of Titration. n; % accordance (SD) | 14; 143.8 (68.1) | 29; 193.5 (99.7) | 43; 177.3 (92.9) |
| Percentage of Laboratory Monitoring Performed During Titration | |||
| Baseline, n (CRP; Trop; ANC; CLZ Level) | 14 (28.6%; 7.1%; 100%; N/A) | 29 (0%; 0%; 100%; N/A) | 43 (9.3%; 2.3%; 100%; N/A) |
| Week 1, n (CRP; Trop; ANC; CLZ Level) | 14 (57.1%; 71.4%; 100%; 0%) | 29 (10.3%; 27.6%; 100%; 0%) | 43 (25.6%; 41.9%; 100%; 0%) |
| Week 2, n (CRP; Trop; ANC; CLZ Level) | 8 (75%; 87.5%; 100%; 0%) | 23 (17.4%; 34.8%; 100%; 4.3%) | 31 (32.3%; 48.4%; 100%; 3.2%) |
| Week 3, n (CRP; Trop; ANC; CLZ Level) | 4 (100%; 100%; 100%; 25%) | 10 (20%; 20%; 100%; 10%) | 14 (42.9%; 42.9%; 100%; 14.3%) |
| Week 4, n (CRP; Trop; ANC; CLZ Level) | 1 (100%; 100%; 100%; 0%) | 0 (0%; 0%; 0%; 0%) | 1 (100%; 100%; 100%; 0%) |
ADR = adverse drug reaction; ANC = absolute neutrophil count; BMI = Body Mass Index; CLZ = clozapine; CRP = c-reactive protein; DDI = drug-drug interaction; ICTG = International Clozapine Titration Guideline; N/A = not applicable; Trop = troponin.
FIGURE 1.
Probability of adverse drug reactions (ADRs) based on last week International Clozapine Titration Guideline (ICTG) percent dose accordance, cumulative ICTG percent dose accordance, and obesity status. Four binary logistic regression models evaluated the probability of ADR for obese and nonobese patients. Last week ICTG percent dose accordance was taken from the clozapine exposure for the last week inpatient, while cumulative ICTG percent dose accordance encompassed total clozapine exposure through the last inpatient week
FIGURE 2.
Last week International Clozapine Titration Guideline (ICTG) percent dose accordance and cumulative ICTG percent dose accordance percentage by obesity status; age at administration by adverse drug reaction (ADR) status. (A) Last week percent accordance was taken from the clozapine exposure for the last week inpatient. On average, obese patients did have a higher last week percent accordance. One mild outlier existed, within median ± 1.5 × (Q3-Q1), in the non-ADR group. (B) Cumulative ICTG percent dose accordance encompassed total clozapine exposure through the last inpatient week. On average, obese patients did have higher cumulative ICTG percent dose accordance. One mild outlier existed, within median ± 1.5 × (Q3-Q1), in the non-ADR group. (C) Age in years at the time of admission was compared with ADR occurrence (yes or no). The patients in the ADR group were slightly older; the, average age in ADR group was 41.7 versus 38.9 years in the non-ADR group
TABLE 2.
Major adverse drug reactions (ADRs) reported during clozapine (CLZ) initiation
| ADR | Mean Age in Years at Admission (SD) | No. of Patients with ADR | Mean BMI kg/m2 (SD) | Mean Days to ADR (SD) | Average Last Cumulative CLZ Week Dose Accordance to Guideline (SD) | Management |
|---|---|---|---|---|---|---|
| All ADRs | 41.7 (17.2) | 14 | 31 (6.5) | 15 (9.4) | 144.6% (65.9) | |
| CLZ Concentration >1000 µg/L | 62 | 1 | 33.6 | 25 | 130.6% | CLZ dose reduced by 40% (n = 1) |
| CRP Elevation ≥3 mg/L | 34.8 (12.9) | 8 | 33.1 (6.9) | 19 (9.5) | 165.2% (80.5) | CLZ dose reduced by 40% (n = 1); CLZ discontinued (n = 4); negative myocarditis workup, continued CLZ dose (n = 1); CLZ continued (n = 2) |
| Myocarditis | 38.5 (0.7) | 2 | 34.2 (2.3) | 20 (2.8) | 241.1% (103.4) | CLZ discontinued (n = 2) |
| Pneumonia | 26 | 1 | 26.8 | 15 | 94.3% | CLZ continued at same dose (n = 1) |
| Severe Sedation | 42.3 (23.2) | 3 | 28.9 (9.2) | 9 (5.2) | 170.5% (72.5) | ER visit to rule out CVA, CLZ continued at same dose (n = 1); CLZ discontinued (n = 1); CLZ dose reduced 50% (n = 1) |
| Transaminitis | 48 | 1 | 25.9 | 24 | 82.5% | CLZ discontinued (n = 1) |
| Troponin Elevation | 41 (16.2) | 4 | 32.1 (3.4) | 20 (13.2) | 178% (97.8) | CLZ discontinued (n = 3); negative myocarditis workup, continued CLZ dose (n = 1) |
BMI = Body Mass Index; CRP = c-reactive protein; CVA = cerebrovascular accident; ER = emergency room.
The most clinically relevant variables found to affect clozapine adverse events or drug discontinuation were the last week ICTG percent dose accordance and cumulative ICTG percent dose accordance (Figure 1). The 2 dosage variables were highly correlated and were fit in separate models to avoid a high degree of multicollinearity (Figure 1). Nonobese patients’ clozapine titrations were more likely to be in accordance with ICTG-recommended titrations (Figure 2A and B) and less likely than obese patients to experience an ADR (OR = 0.1737; 95% CI, 0.03-0.99) (Figure 1). Mean age was similar between patients with or without an ADR or drug discontinuation, but greater dispersion to the upper quartile was seen in patients with an ADR or drug discontinuation (Figure 2c). Laboratory monitoring of absolute neutrophil count was obtained weekly for 100% of patients, but CRP and clozapine concentrations were not routinely obtained (Table 1).
Discussion
Of the 43 patients initiated on clozapine, 14 experienced serious ADRs, with 11 of these discontinuing clozapine. Elevated CRP or troponin (n = 7) were the most common reasons for clozapine discontinuation, followed by myocarditis, transaminitis, and severe sedation. The probability of an ADR revealed an inverse relationship between both cumulative and last week ICTG percent dose accordance (Figure 1). This may have been related to reverse causation, where patients deemed likely to have an ADR received smaller doses or slowed titrations and should be interpreted with caution because of our small sample size. Patients with a BMI of 30 kg/m2 or higher had a nearly 2-fold increase in percent clozapine dose accordance compared with those with a BMI of less than 30 kg/m2 (Figure 2A and B) and a higher probability of ADRs (Figure 1). Higher dose accordance observed in obese patients was likely due to clozapine titrations started before the publication of the ICTG, which categorized obese patients as poor metabolizers and recommended slower titrations.18 While the ICTG categorizes patients with a BMI of 29 kg/m2 or higher as a poor metabolizer,18 this project used a BMI of 30 kg/m2 or higher per standard obesity status definitions.25 In the ICTG supplemental data, a BMI of 29 kg/m2 or higher is stated to account for a less than 1% reason for poor metabolism,18 but human studies suggest decreased CYP3A4, 2D6, and 1A2 activity in obese individuals, which is relevant for clozapine metabolism.8,26 Thus, obese patients were at higher risk of an ADR, and slower titrations, as recommended in the ICTG, appeared to be justified.
Several clozapine ADRs have been shown or are theorized to be related to titration speed or occur during titration, including myocarditis, inflammatory conditions (eg, hepatitis, nephritis, pancreatitis),27,28 seizures,29,30 hypotension,31-33 and pneumonia.11,18,34-36 While several small studies suggest faster clozapine titrations are safe and provide the benefit of shorter hospital stays, more research is needed in this area.36-38 Our incidence of myocarditis (4.6%) was higher than the worldwide estimate of 3%,10 that in other Western countries (< 1%),39 and the Netherlands (0.03%),39 who use slow outpatient titrations.20 No patients developed agranulocytosis/severe neutropenia during their initial titration, which could be expected as the incidence is 0.3 to 0.4 cases per 100-person years.40 An opportunity exists in future studies to evaluate the probability of specific ADRs in relationship with titration speed, which was not performed because of the small sample size.
ICTG-recommended laboratory monitoring was suboptimal, with only weekly absolute neutrophil count monitoring occurring 100% of the time (Table 1). CRP monitoring was performed in 9.3% of patients before initiating clozapine and 25.6% at titration week (Table 1). Given the impact of clozapine-related mortality from myocarditis and pneumonia, increased frequency of CRP monitoring is an area where pharmacists can not only improve patient safety but also improve provider comfort.7,22,41
Limitations of this study include its retrospective design and the inability to draw causal conclusions. Linear logistical regression used ADRs as the primary outcome, with elevated biomarkers (ie, CRP and troponin) being the most common reasons for clozapine discontinuation. While elevated biomarkers do not constitute an ADR by most definitions, we included them in the primary outcome as they represent poor clozapine tolerability. Future studies are needed to provide evidence of thresholds to discontinue clozapine or coordinate continued care with specialists (eg, cardiology consults for myocarditis) to avoid unnecessary discontinuation of clozapine. Our small sample size and primarily European/Western Asian ancestry population limited generalizability and power to evaluate specific ADR probability, such as myocarditis. Additionally, the short duration of hospitalization limited our ability to follow titration progression and clozapine tolerance, which is demonstrated by weekly attrition (Table 1). We did not include patient age in our logistic regression model (Figure 2C), as there are no age-related recommendations in the ICTG18; however, age has been shown to impact clozapine metabolism through an inverse relationship and may warrant further investigation.42,43 Finally, we only included ICTG-specific DDIs, but other clinically relevant medications impact clozapine metabolism, which we did not evaluate (eg, ciprofloxacin, disulfiram, viloxazine, ritonavir).15,16,21
Based on our small retrospective chart review of primarily White males, more aggressive clozapine dose relationships did not increase the incidence of ADRs. Patients with a BMI of 30 kg/m2 or higher had a nearly 2-fold increase in percent clozapine dose accordance compared with those with a BMI of less than 30 kg/m2 (Figure 2A and B) and higher probability of ADRs (Figure 1), supporting ICTG-recommended slower titrations for these patients. ICTG-recommended laboratory monitoring was suboptimal, providing an opportunity for pharmacists to improve laboratory monitoring. Future studies with more diverse samples are needed and should focus on specific ADRs, such as inflammatory conditions, which may have increased occurrence with rapid titrations.
Footnotes
Disclosures: This research was funded in part by a grant from the AAPP Foundation. Ian McGrane receives royalties from Hogrefe Publishing Corp. All other authors have no conflicts of interest to disclose.
References
- 1.Accord Healthcare Inc. Bethesda (MD): National Library of Medicine (US) ; [updated 2023 Jan; cited 2023 Feb 23]. Clozapine tablet; [about 2 screens]. Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=25c0c6d5-f7b0-48e4-e054-00144ff8d46c
- 2.Kikuchi Y, Komatsu H, Otsuka Y, Ito F, Kanahara N, Tanifuji H, et al. Slower clozapine titration than the official Japanese protocol led to fewer inflammatory adverse effects: A retrospective chart review of seven hospitals. Schizophr Res. Online ahead of print June 16, 2023. 10.1016/j.schres.2023.06.003 [DOI] [PubMed] [Google Scholar]
- 3.Yatham LN, Kennedy SH, Parikh SV, Schaffer A, Bond DJ, Frey BN, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2018;20(2):97-170. 10.1111/bdi.12609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yunusa I, Rashid N, Seyedin R, Paratane D, Rajagopalan K. Comparative efficacy, safety, and acceptability of pimavanserin and other atypical antipsychotics for Parkinson’s disease psychosis: Systematic review and network meta-analysis. J Geriatr Psychiatry Neurol. 2023:8919887231154933. 10.1177/08919887231154933 [DOI] [PubMed] [Google Scholar]
- 5.Steinauer LM, Leung JG, Burkey BW, McGrane IR, Letts V, Goren JL, et al. A retrospective multicenter evaluation of clozapine use in pediatric patients admitted for acute psychiatric hospitalization. J Child Adolesc Psychopharmacol. 2018;28(9):615-9. 10.1089/cap.2018.0036 [DOI] [PubMed] [Google Scholar]
- 6.Pai NB, Vella SC. Reason for clozapine cessation. Acta Psychiatr Scand. 2012;125(1):39-44. 10.1111/j.1600-0447.2011.01776.x [DOI] [PubMed] [Google Scholar]
- 7.de Leon J, Arrojo-Romero M, Verdoux H, Ruan C-J, Schoretsanitis G, Rohde C, et al. Escaping the long shadow cast by agranulocytosis. Reflections on clozapine pharmacovigilance focused on the United Kingdom. J Clin Psychopharmacol. 2023;43(3):239-45. 10.1097/JCP.0000000000001678 [DOI] [PubMed] [Google Scholar]
- 8.Bertilsson L, Carrillo JA, Dahl ML, Llerena A, Alm C, Bondesson U, et al. Clozapine disposition covaries with CYP1A2 activity determined by a caffeine test. Br J Clin Pharmacol. 1994;38(5):471-3. 10.1111/j.1365-2125.1994.tb04385.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Leon J. Reflections on the lack of consideration of ethnic ancestry to stratify clozapine dosing. Psychiatry Investig. 2023;20(3):183-95. 10.30773/pi.2022.0293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ronaldson KJ, Fitzgerald PB, McNeil JJ. Clozapine-induced myocarditis, a widely overlooked adverse reaction. Acta Psychiatr Scand. 2015;132(4):231-40. 10.1111/acps.12416 [DOI] [PubMed] [Google Scholar]
- 11.Ronaldson KJ, Fitzgerald PB, Taylor AJ, Topliss DJ, Wolfe R, McNeil JJ. Rapid clozapine dose titration and concomitant sodium valproate increase the risk of myocarditis with clozapine: a case-control study. Schizophr Res. 2012;141(2-3):173-8. 10.1016/j.schres.2012.08.018 [DOI] [PubMed] [Google Scholar]
- 12.Ertuğrul A, Anıl Yağcıoğlu AE, Ağaoğlu E, Karakaşlı AA, Ak S, Yazıcı MK, Leon J. Valproate, obesity and other causes of clozapine poor metabolism in the context of rapid titration may explain clozapine-induced myocarditis: A re-analysis of a Turkish case series. Rev Psiquiatr Salud Ment (Engl Ed). 2022;15(4):281-6. 10.1016/j.rpsmen.2021.10.001 [DOI] [PubMed] [Google Scholar]
- 13.de Leon J. Reflections on the complex history of the concept of clozapine-induced inflammation during titration. Psychiatr Danub. 2022;34(3):411-21. 10.24869/psyd.2022.411 [DOI] [PubMed] [Google Scholar]
- 14.Leung JG, de Leon J, Frye MA, Singh B, Cotes RO, McElroy SL. The modernization of clozapine: A recapitulation of the past in the United States and the view forward. J Clin Psychopharmacol. 2022;42(6):565-80. 10.1097/JCP.0000000000001606 [DOI] [PubMed] [Google Scholar]
- 15.Hahl-Häkkinen L, Rask SM, Solismaa A, Ruuhonen S, Leinonen E. Co-medication with disulfiram markedly increased serum clozapine levels: Two case reports . J Psychopharmacol. 2023;37(2):229-33. 10.1177/02698811221148611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Meyer JM, Proctor G, Cummings MA, Dardashti LJ, Stahl SM. Ciprofloxacin and clozapine: A potentially fatal but underappreciated interaction. Case Rep Psychiatry. 2016;2016:5606098. 10.1155/2016/5606098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.de Leon J. Future studies on the interaction between clozapine and valproic acid should aspire to include longitudinal designs and free valproate concentrations, and should consider that inducer and/or inhibitory effects may vary with time, the individual, and the auto-induction of valproic acid. Ther Drug Monit. 2020;42(1):159-61. 10.1097/FTD.0000000000000705 [DOI] [PubMed] [Google Scholar]
- 18.de Leon J, Schoretsanitis G, Smith RL, et al. An international adult guideline for making clozapine titration safer by using six ancestry-based personalized dosing titrations, CRP, and clozapine levels. Pharmacopsychiatry. 2022;55(2):73-86. Erratum in: Pharmacopsychiatry. 2022;55(2):e1. [DOI] [PubMed] [Google Scholar]
- 19.De Las Cuevas C, Sanz EJ, Ruan CJ, de Leon J. Clozapine-associated myocarditis in the World Health Organization’s pharmacovigilance database: Focus on reports from various countries. Rev Psiquiatr Salud Ment (Engl Ed). 2022;15:238-50. 10.1016/j.rpsmen.2021.07.005 [DOI] [PubMed] [Google Scholar]
- 20.Netherlands clozapine collaboration group , editors. Guideline for the use of clozapine [Internet]. The Netherlands: Clozapine Plus Werkgroep; 2013. [cited 2022 Dec 28]. Available from: http://www.clozapinepluswerkgroep.nl/wp-content/uploads/2013/07/Guideline-for-the-use-of-Clozapine-2013.pdf [Google Scholar]
- 21.Leung JG, McGrane IR. Improving the clozapine prescribing information to enhance clozapine safety and address barriers: clozapine-induced inflammation, pneumonia, and REMS. Psychiatr Danub. 2022;34(3):422-7. PMID: 36256976 [PubMed] [Google Scholar]
- 22.Goldstone LW, DiPaula BA, Werremeyer A, Botts S, Hepburn B, Liu HY, et al. The role of board-certified psychiatric pharmacists in expanding access to care and improving patient outcomes. Psychiatr Serv. 2021;72(7):794-801. 10.1176/appi.ps.202000066 [DOI] [PubMed] [Google Scholar]
- 23.Kelly DL, Freudenreich O, Sayer MA, Love RC. Addressing barriers to clozapine underutilization: a national effort. Psychiatr Serv. 2018;69(2):224-7. 10.1176/appi.ps.201700162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fujii K, Okayasu H, Shinozaki T, Shimoda K, Ozeki Y. Slower titration of lamotrigine reduces the risk of rash. Psychiatry Clin Neurosci. 2020;74(4):282-3. Erratum in: Psychiatry Clin Neurosci. 2021 Oct;75(10):324. 10.1111/pcn.12987 [DOI] [PubMed] [Google Scholar]
- 25.Centers for Disease Control and Prevention [Internet]. Atlanta: National Institutes of Health ; [updated 2022 Jun 3; cited 2023 Feb 22]. About adult BMI; [about 9 screens]. Available from: https://www.cdc.gov/healthyweight/assessing/bmi/adult_bmi/index.html
- 26.Zarezadeh M, Saedisomeolia A, Shekarabi M, Khorshidi M, Emami MR, Müller DJ. The effect of obesity, macronutrients, fasting and nutritional status on drug-metabolizing cytochrome P450s: a systematic review of current evidence on human studies. Eur J Nutr. 2021;60(6):2905-21. 10.1007/s00394-020-02421-y [DOI] [PubMed] [Google Scholar]
- 27.Lally J, Al Kalbani H, Krivoy A, Murphy KC, Gaughran F, MacCabe JH. Hepatitis, interstitial nephritis, and pancreatitis in association with clozapine treatment: A systematic review of case series and reports. J Clin Psychopharmacol. 2018;38(5):520-7. 10.1097/JCP.0000000000000922 [DOI] [PubMed] [Google Scholar]
- 28.Verdoux H, Quiles C, de Leon J. Clinical determinants of fever in clozapine users and implications for treatment management: A narrative review. Schizophr Res. 2019;211:1-9. 10.1016/j.schres.2019.07.040 [DOI] [PubMed] [Google Scholar]
- 29.Devinsky O, Honigfeld G, Patin J. Clozapine-related seizures. Neurology. 1991;41(3):369-71. 10.1212/wln.41.3.369 [DOI] [PubMed] [Google Scholar]
- 30.Lee KC, Finley PR, Alldredge BK. Risk of seizures associated with psychotropic medications: emphasis on new drugs and new findings. Expert Opin Drug Saf. 2003;2(3):233-47. 10.1517/14740338.2.3.233 [DOI] [PubMed] [Google Scholar]
- 31.Lieberman JA, Safferman AZ. Clinical profile of clozapine: adverse reactions and agranulocytosis. Psych Quart. 1992;63:51-70. 10.1007/BF01064682 [DOI] [PubMed] [Google Scholar]
- 32.Tanzer TD, Brouard T, Pra SD, Warren N, Barras M, Kisely S, Brooks E, Siskind D. Treatment strategies for clozapine-induced hypotension: a systematic review. Ther Adv Psychopharmacol. 2022;12:20451253221092931. 10.1177/20451253221092931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Oyewumi LK, Cernovsky ZZ, Freeman DJ. Autonomic signs and dosing during the initial stages of clozapine therapy. Med Sci Monit. 2004;10(2):PI19-23. PMID: 14737054 [PubMed] [Google Scholar]
- 34.Kuo CJ, Yang SY, Liao YT, Chen WJ, Lee, W-C, Shau W-Y, et al. Second-generation antipsychotic medications and risk of pneumonia in schizophrenia. Schizophr Bull. 2013;39(3):648-57. 10.1093/schbul/sbr202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rohde C, Polcwiartek C, Kragholm K, Ebdrup BH, Siskind D, Nielsen J. Adverse cardiac events in out-patients initiating clozapine treatment: a nationwide register-based study. Acta Psychiatr Scand. 2018;137(1):47-53. 10.1111/acps.12827 [DOI] [PubMed] [Google Scholar]
- 36.Poyraz CA, Özdemir A, Sağlam NG, et al. Rapid clozapine titration in patients with treatment refractory schizophrenia. Psychiatr Q. 2016;87(2):315-22. 10.1007/s11126-015-9394-y [DOI] [PubMed] [Google Scholar]
- 37.Ifteni P, Correll CU, Nielsen J, Burtea V, Kane JM, Manu P. Rapid clozapine titration in treatment-refractory bipolar disorder. J Affect Disord. 2014;166:168-72. 10.1016/j.jad.2014.04.020 [DOI] [PubMed] [Google Scholar]
- 38.Ifteni P, Nielsen J, Burtea V, Correll CU, Kane JM, Manu P. Effectiveness and safety of rapid clozapine titration in schizophrenia. Acta Psychiatr Scand. 2014;130(1):25-9. 10.1111/acps.12241 [DOI] [PubMed] [Google Scholar]
- 39.Cohen D, Bogers JP, van Dijk D, Bakker B, Schulte PF. Beyond white blood cell monitoring: screening in the initial phase of clozapine therapy. J Clin Psychiatry. 2012;73(10):1307-12. 10.4088/JCP.11r06977 [DOI] [PubMed] [Google Scholar]
- 40.Schulte P. Risk of clozapine-associated agranulocytosis and mandatory white blood cell monitoring. Ann Pharmacother. 2006;40(4):683-8. 10.1345/aph.1G396 [DOI] [PubMed] [Google Scholar]
- 41.Pandarakalam JP. The art of clozapine therapy and “clozaphobia”. BMJ. 2019;364:l484. 10.1136/bmj.l484 [DOI] [PubMed] [Google Scholar]
- 42.Ismail Z, Wessels AM, Uchida H, Ng W, Mamo DC, Rajji TK, et al. Age and sex impact clozapine plasma concentrations in inpatients and outpatients with schizophrenia. Am J Geriatr Psychiatry. 2012;20(1):53-60. 10.1097/JGP.0b013e3182118318 [DOI] [PubMed] [Google Scholar]
- 43.Reeves S, Bertrand J, Obee SJ, Hunter S, Howard R, Flanagan RJ. A population pharmacokinetic model to guide clozapine dose selection, based on age, sex, ethnicity, body weight and smoking status. Br J Clin Pharmacol. 2023. Feb 15. 10.1111/bcp.15691. Epub ahead of print. [DOI] [PubMed] [Google Scholar]