The drug treatment of asthma has remained essentially unchanged over the past three decades in terms of the use of corticosteroid, β2 agonist, and theophylline drugs. Asthma treatment has also been improved by the widespread dissemination and implementation of management guidelines emphasising the pivotal role of first line preventative, anti-inflammatory therapy.1,2 This article provides a brief overview of modern drug treatment for chronic asthma. It does not cover the treatment of acute asthma, which is discussed in detail elsewhere.3
Summary points
The dose of inhaled steroid should be titrated against asthmatic symptoms, peak flow, and usage of β2 agonist drugs
The safest dose of inhaled steroid is the lowest effective maintenance dose producing optimal long term control and quality of life
Adding second line anti-inflammatory controller treatment such as a leukotriene antagonist or theophylline may be an alternative to monotherapy with a high dose of inhaled steroid
If control is inadequate despite optimised anti-inflammatory treatment, it is better to add regular treatment with a long acting β2 agonist drug than a short acting one
Methods
I searched Medline and bids for articles published between 1977 and 1998, using appropriate index terms for each drug or class of drugs. I included key review articles and searched manually for relevant papers and abstracts in recent issues of mainstream journals on general, respiratory, and allergy medicine. This article was also based on personal, long standing, clinical and research interests in the management of allergy and asthma. Some aspects of this review will inevitably be based on personal opinion, particularly where the latest guidelines are already out of date—for example, with the emerging role of leukotriene antagonists.
First principles of treatment
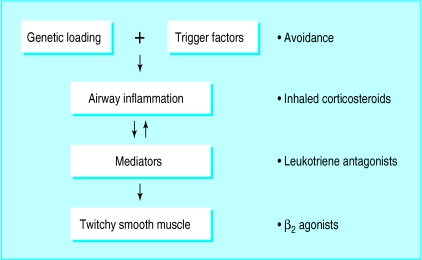
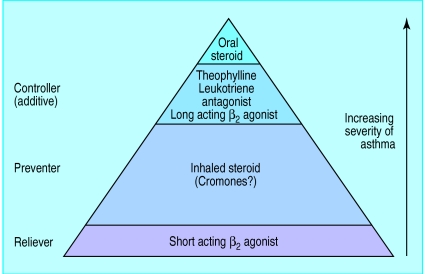
The past decade of research has led to a greater understanding of the pathophysiology of asthma and, in particular, the pivotal role of the underlying inflammatory process (fig 1). Current asthma management guidelines stress the importance of switching off the inflammatory process at the top of the cascade by giving first line preventitive treatment with inhaled corticosteroids, thereby reducing the need to provide symptomatic relief with short acting β2 agonists at the bottom of the cascade. Drugs such as long acting inhaled β2 agonists, theophyllines, and anti-leukotrienes may also be used as second line “controller treatment” when given with inhaled corticosteroids to improve symptom control and reduce diurnal variability (fig 2). These treatment options should be used conjunction with removal of any potential trigger factors (box).
Common reasons for poor response to treatment
Poor compliance—for example, as a result of having to take drugs (such as cromoglycate) four times daily, or polypharmacy
Poor technique in using an inhaler—for example, difficulty with metered dose inhalers
Presence of trigger factors—for example, allergen, smoking, occupation, oesophageal reflux, rhinitis
Wrong diagnosis—for example, having chronic obstructive pulmonary disease, bronchiectasis, or heart failure rather than asthma
Figure 1.
The inflammatory cascade in asthma
Figure 2.
The asthma treatment pyramid
Goals
A number of optimal treatment goals should be set for a given patient within the pharmacoeconomic constraints of the available health service provision. These goals are set out in the box.
Goals of treatment
To achieve normal percentage predicted values for forced expiratory volume in one second/peak expiratory flow
Reduce diurnal variability of peak expiratory flow and symptoms
Minimise use of reliever β2 agonist drugs
Optimise quality of life
Reduce risk of severe exacerbation of asthma
Corticosteroids
Inhaled corticosteroids are the most potent anti-inflammatory agents for treating asthma and act in a relatively non-specific manner by inhibiting a variety of inflammatory cells, cytokine expression, and transcription factors which are involved in the inflammatory disease process.4 The anti-inflammatory effects of inhaled corticosteroids have been shown in bronchial biopsy studies.5 The delivery directly to the airway of relatively small doses of topically active corticosteroid, along with an extensive degree of hepatic first pass inactivation of the swallowed moiety, results in a high therapeutic index—the ratio of anti-asthmatic efficacy to systemic adverse effects. The inhaled corticosteroid drugs are also the most cost effective form of treatment for preventing asthma (table 1).
Table 1.
Comparative daily costs for asthma drugs
| Drug | Dosage regimen | Cost per day* |
|---|---|---|
| Inhaled beclomethasone (Qvar) | 200 μg twice daily | 16p |
| Inhaled cromoglycate (Non-proprietary) | 10 mg four times daily | 99p |
| Inhaled salmeterol (Serevent) | 50 μg twice daily | 95p |
| Inhaled salbutamol (Non-proprietary) | 200 μg four times daily | 7p |
| Oral montelukast (Singulair) | 10 mg once daily | 92p |
| Oral theophylline (Uniphyllin) | 200 mg twice daily | 14p |
Calculated from British National Formulary, No 36 (March 1998). The cheapest formulation of each drug was taken.
Dose-response relation
There is considerable variation between patients in the degree of glucocorticoid receptor sensitivity, which in turn determines both the airway and the systemic dose-response relation. For most adults with mild to moderate asthma, the steep part of the dose-response curve for anti-asthmatic efficacy generally occurs at doses below 800 μg/day (400 μg/day in children) for beclomethasone dipropionate, budesonide, and triamcinolone acetonide. However, for systemic adverse effects, the curve tends to become much steeper at doses above 800 μg/day. Even in patients with more severe asthma, the dose-response curve for efficacy may be relatively shallow above 800 μg/day. This dissociation between the airway and systemic dose-response relation results in an inverted U shaped curve for the benefit to risk ratio. This begins to decline above a watershed of approximately 800 μg/day (400 μg/day in children), although the exact point depends on disease severity and individual sensitivity. Because respirable lung dose improves greatly with the hydrofluoroalkane formulation of beclomethasone, half the dose of the chlorofluorocarbon formulation can be given, and it is possible to target delivery to the inflamed small airways.
Regimen
The modern management of persistent asthma with inhaled corticosteroid drugs involves starting treatment with a relatively high dose for four to eight weeks in order to gain rapid optimal control. This is followed by a gradual tapering of the dose to determine the lowest effective maintenance dose for a given person. For people with mild to moderate asthma, effective control can usually be achieved by a once daily regimen when the patient has been stabilised on maintenance treatment at doses of up to 800 μg/day of budesonide or beclomethasone. Evidence also suggests that early intervention with inhaled corticosteroid drugs may prevent any long term decline in lung function resulting from bronchial fibrosis caused by untreated chronic inflammation.4
Systemic effects
The main concern with inhaled corticosteroid treatment is the potential for dose related systemic effects. These include adrenal suppression, osteoporosis, growth suppression, skin bruising, cataracts and ocular hypertension. There is increasing evidence that the newer high potency drugs such as fluticasone propionate show a less favourable systemic bioactivity profile, although this may be partly offset by the ability to use a lower effective maintenance dose. Three separate dose-response studies have shown that even when correcting for differences in topical potency, therapeutically equivalent microgram doses of budesonide and fluticasone (half dose) exhibit at least a 1.5-fold difference in systemic bioactivity as assessed by sensitive measures of adrenal suppression.6–8 This is supported by a meta-analysis of 22 studies in which fluticasone propionate produced significantly steeper dose related adrenal suppression than budesonide (2.5-fold difference), beclomethasone dipropionate (2.1-fold difference), or triamcinolone acetonide (3.6-fold difference)—a finding that was particularly evident at doses above 800 μg/day.9
Available data suggest that the beneficial effects of inhaled corticosteroids on disease control will outweigh any potential systemic bioactivity in terms of long term growth in asthmatic children. No effect of these drugs on the final achieved adult height has been shown.10,11 Indeed, other factors such as socioeconomic status and, perhaps, nutrition may be more important in determining height in asthmatic children taking inhaled corticosteroids.12 Evidence indicates, particularly in at risk groups such as postmenopausal women, that bone density in lumbar vertebrae may be reduced by long term exposure to inhaled corticosteroids, and that this reduction is related to the cumulative dose and duration of treatment.13,14 Corticosteroid induced osteoporosis may be reduced by the use of oestrogen replacement therapy or biphosphonate drugs.
Long term treatment with high doses of inhaled corticosteroids is associated with an increased risk of a posterior subcapsular cataracts and, to a lesser degree, with ocular hypertension.15,16 Skin bruising, an adverse effect of inhaled corticosteroids, is more prevalent in elderly people and is associated with adrenal suppression.17 Indeed, skin bruising is visible evidence of increased collagen turnover, and should therefore prompt further screening for other tissue specific adverse effects. It may therefore be prudent to perform a regular annual or biennial check up to look for evidence of systemic bioactivity in the skin, eye, adrenal gland, and bone in adults receiving long term, high dose inhaled corticosteroid treatment (>800 μg daily of beclomethasone or budesonide and >400 μg daily of fluticasone), and to monitor growth in children (>400 μg daily beclomethasone or budesonide and >200 μg daily fluticasone).
Local adverse effects
Local adverse effects such as oral candidiasis may be alleviated by using a large volume spacer to reduce the deposition of drug on the oropharynx. The occurrence of oral candidiasis is related to the dose and to the mucosal exposure time to topical corticosteroid. Thus, a once or twice daily dosing regimen will reduce the likelihood, and this can be reduced further by regular mouth rinsing. Using a spacer device has other advantages—there is increased delivery of respirable particles and the coordination problems associated with using metered dose inhalers are reduced.
Cromones
The cromones include sodium cromoglycate and sodium nedocromil, which act predominantly by stabilising mast cells. These drugs are well tolerated and have no systemic adverse effects, but they are less effective in treating asthma than inhaled corticosteroids. The cromones tend to be most effective in patients with mild atopic asthma, particularly in children with an exercise or allergen induced component to their condition. However, there is uncertainty about the degree of anti-inflammatory activity of the cromones, at least on the basis of bronchial biopsy studies.18 Compliance may be a problem with these drugs as they are short acting and need to be taken four times daily. Cromone treatment is also much more expensive than that with low dose inhaled corticosteroid drugs (table 1).
Leukotriene antagonists
The cysteinyl leukotrienes are metabolites of arachidonic acid comprising leukotrienes C4, D4, and E4. The cysteinyl leukotrienes cause smooth muscle constriction and proliferation and are important mediators in the pathophysiology of the inflammatory process. The leukotriene antagonists such as zafirlukast (twice daily) and montelukast (once daily) are well tolerated, seem effective over a wide spectrum of disease severity, and exhibit both bronchodilator and anti-inflammatory activity.19 Responsiveness to leukotriene antagonists varies and may be genetically determined by the degree of leukotriene synthesis resulting from 5-lipoxygenase activity. In the United Kingdom, montelukast is licensed in patients aged 6 years and over as second line asthma controller treatment in combination with inhaled corticosteroids. It is only licensed as monotherapy in the prophylaxis of exercise induced asthma. Zafirlukast, however, is currently licensed in patients aged 12 years and over, including first line use instead of inhaled corticosteroids.
Advantages
One of the main advantages of the leukotriene antagonist drugs is that they are active orally, which avoids the potential compliance problems with the inhaled route. The compliance factor with leukotriene antagonists may also be reinforced by the fact that they work within the first 24 hours, while inhaled corticosteroids take much longer to achieve maximal response. The leukotriene antagonists are comparable in cost with long acting β2 agonists, but are much more expensive than low dose inhaled corticosteroids (table 1).
Additive effects
Preliminary data with montelukast and zafirlukast suggest that they show additive effects to low or high dose inhaled corticosteroids.20–23 In multicentre studies, montelukast in children (5 mg once daily) or adults (10 mg once daily) was better than placebo at controlling asthma over eight to 12 weeks, and this effect was seen equally in patients taking or not taking corticosteroids.24,25 In addition, montelukast and placebo showed no differences in adverse effects on biochemical liver function tests. Regular treatment with montelukast produces a sustained, high level of protection against exercise induced bronchoconstriction, in contrast with salmeterol, which induces tolerance.26 Another potential advantage of leukotriene antagonist drugs is that they are effective in treating coexistent allergic rhinitis.27 A Churg-Strauss-like syndrome has been reported with zafirlukast and montelukast; this is probably due to an unmasking of the underlying condition caused by tapering of concomitant oral corticosteroids, and reinforces that leukotriene antagonists should not be used instead of oral corticosteroids in dependent patients with severe asthma.
Need for further data
Thus, there seem to be logical reasons for using leukotriene antagonists as second line controller treatment in that they possess both anti-inflammatory and bronchodilator activity, afford bronchoprotection against allergen and exercise, do not exhibit tachyphylaxis, and are well tolerated (table 2). Bronchial biopsy studies show anti-inflammatory effects of leukotriene antagonists in reducing numbers of T lymphocytes, mast cells, and eosinophils.28 However, further efficacy and safety data from long term studies are needed to establish the appropriate place of leukotriene antagonists in asthma treatment guidelines—in particular, whether they should be used as an alternative to low dose inhaled corticosteroid as first line monotherapy in patients with mild to moderate persistent asthma.
Table 2.
Drugs for second line, additive control of asthma
| Drug | Route and frequency | Therapeutic ratio | Anti-inflammatory activity | Symptom control | Tolerance | Cost |
|---|---|---|---|---|---|---|
| Long acting β2 agonist | Inhaled, twice daily* | High | None | Good | Yes | Expensive |
| Theophylline | Oral, twice daily | Low | Weak | Moderate | No | Cheap |
| Leukotriene antagonist | Oral, once or twice daily | High | Moderate | Good | No | Expensive |
May be used once daily for nocturnal symptoms.
Antihistamines
Cetirizine and loratidine are examples of potent, selective, type I histamine receptor antagonists that are well tolerated and are taken on a once daily basis. They have a limited role in treating asthma in patients with a known allergenic trigger factor such as pollen or animal fur. They may also be of value where there is associated seasonal allergic rhinitis and conjunctivitis or if there is associated allergic urticaria. Preliminary data suggest that antihistamines and leukotriene antagonists may show additive effects on control in asthma and allergic rhinitis.27,29 Antihistamines should never be used as monotherapy for chronic asthma, but only as an adjunct to inhaled corticosteroids.
Theophyllines
The bronchodilator activity of oral theophylline is relatively weak and high doses are needed. Lower doses of theophylline show anti-inflammatory effects, and this finding led to a reappraisal of theophylline’s role as second line controller treatment in addition to inhaled corticosteroids.30,31 The advantages of theophylline are that it may be taken once or twice daily as an oral slow release formulation and is inexpensive (table 1).
The main disadvantages of theophylline are several important potential drug interactions that may result in drug toxicity as well as the expense and inconvenience of therapeutic drug monitoring (table 2). Although the adverse effects of theophylline are related to the plasma concentration, the drug tends to be less well tolerated, even at low doses, than other second line controller drugs such as leukotriene antagonists or long acting β2 agonists.
β2 agonists
The β2 agonist drugs act primarily on airway smooth muscle and are the most effective form of bronchodilator treatment. Their effects on smooth muscle and mast cells result in protection against several stimuli of bronchoconstriction. For example, inhaled β2 agonists are highly effective at preventing bronchoconstriction when used shortly before exercise or exposure to known allergens. Evidence suggests that regular use of short acting β2 agonists may worsen control of asthma, although this claim remains controversial.32 The requirement for regular use of reliever treatment with short acting β2 agonists marks an activated inflammatory cascade and hence the need to step up the dose of inhaled corticosteroid (fig 1).
Long acting β2 agonists
The long acting β2 agonists salmeterol and eformoterol became available against this background of recommending short acting β2 agonists for occasional use as a reliever. These drugs are now included in the guidelines for regular, twice daily use as second line controller treatment in conjunction with a low dose of inhaled corticosteroid.1,2 This recommendation is based on studies which showed that adding a long acting β2 agonist to a low dose of inhaled corticosteroid drug produced comparable control to monotherapy with a higher dose of inhaled corticosteroid.33
Concerns
There is concern that the regular use of long acting β2 agonists may simply palliate the sequelae of an activated inflammatory cascade without suppressing the underlying inflammatory process, particularly as β2 agonists have no anti-inflammatory activity (table 2).34 None the less, long acting β2 agonists may be valuable when given regularly for persistent symptoms in patients who would otherwise need to use short acting β2 agonists frequently, provided adequate anti-inflammatory treatment is also being taken.
We also know that tolerance to the airway effects of β2 agonists develops when these drugs are given regularly and that this is more pronounced for loss of bronchoprotective activity than bronchodilator activity.35,36 Moreover, the development of tolerance with long acting β2 agonists occurs in genetically predisposed people. They have a particular variant of the β2 adrenoceptor, which occurs in up to 50% of white people in the United Kingdom.37 This genetic polymorphism of the β2 adrenoceptor may also explain individual variation in the clinical response to long acting β2 agonist treatment. Eformoterol is a more potent drug and has a faster onset of action than salmeterol. It may therefore be used as required for reliever treatment up to the maximum recommended twice daily dose. This type of “as required” dosing regimen with eformoterol is not recommended in current asthma guidelines. Nor, however, does it seem appropriate to advocate its regular use in every case, particularly if it is not needed all the time. New fixed dose combinations of fluticasone and salmeterol will soon become available in the United Kingdom. Although they might improve compliance, such formulations are less flexible and may inadvertently result in patients taking unnecessary long term treatment with long acting β2 agonists.
Controlled release oral salbutamol or oral bambuterol (a prodrug of terbutaline) may, like long acting β2 agonists, be used to treat nocturnal symptoms. Although the oral formulations tend to be associated with systemic effects such as tremor and tachycardia, these usually wear off because of tolerance. None the less, drugs such as bambuterol are less expensive than long acting β2 agonists and, like theophylline, have the advantage of being taken as a once daily tablet.
Conclusions
Inhaled corticosteroids should be used as early as possible as first line anti-inflammatory treatment for patients presenting with symptoms of persistent asthma. For patients who have mild to moderate asthma, there is no proved therapeutic benefit in using more potent and expensive drugs such as fluticasone propionate than older and cheaper drugs such as beclomethasone dipropionate. The therapeutic index for inhaled corticosteroids can be optimised by tapering the dose until the lowest effective maintenance dose is achieved and by using a metered dose inhaler with a spacer device. Second line anti-inflammatory controller treatment may be added as an alternative to monotherapy with a high dose of inhaled corticosteroid in order to avoid any local and systemic adverse effects with the latter treatment. In this respect, there seems to be a rationale for adding treatment with leukotriene antagonists such as montelukast or zafirlukast. These have anti-inflammatory and bronchodilator properties, do not induce tolerance, and can be taken as a once or twice daily tablet. Further long term studies are required to evaluate the position of leukotriene antagonists as first line preventer treatment instead of low dose inhaled corticosteroid drugs in patients with mild to moderate asthma. Theophylline is a cheaper alternative for second line combined anti-inflammatory treatment, but has a lower therapeutic index that requires drug monitoring and shows several important potential drug interactions. If asthma control is inadequate and reliever drugs need to be taken frequently, despite optimised anti-inflammatory treatment, regular treatment with long acting β2 agonists is preferred to that with short acting β2 agonists.
Footnotes
Funding: None.
Competing interests: BL has been reimbursed by the following companies for speaking at educational symposiums, consultancy work, research funding, or attending scientific meetings: Rhone Poulenc Rorer, Astra Draco, Boehringer Ingelheim, 3M Health Care, ML Laboratories, Novartis, Merck, Glaxo-Wellcome, Zeneca, Shering Plough. His spouse currently has shares in Glaxo-Wellcome and Astra.
References
- 1.British guidelines on asthma management: 1995 review and position statement. Thorax. 1997;52(suppl 1):S1–21. [Google Scholar]
- 2.National Asthma Education and Prevention Programme Expert Panel. National Asthma Education and Prevention Programme Expert Panel report II. Guidelines for the diagnosis and management of asthma. Bethesda, MD: National Institutes of Health; 1997. (Publication 97 4051.) [Google Scholar]
- 3.Lipworth BJ. Treatment of acute asthma. Lancet. 1997;350(suppl II):18–23. doi: 10.1016/s0140-6736(97)90032-5. [DOI] [PubMed] [Google Scholar]
- 4.Barnes PJ, Pedersen S, Busse WW. Efficacy and safety of inhaled corticosteroids: new developments. Am J Respir Crit Care Med. 1998;157(suppl):S1–53. doi: 10.1164/ajrccm.157.3.157315. [DOI] [PubMed] [Google Scholar]
- 5.Laitinen LA, Laitinen A, Haahtela T. A comparative study of the effects of inhaled corticosteroid budesonide and β2-agonist terbutaline on airway inflammation in newly diagnosed asthma. J Allergy Clin Immunol. 1992;90:32–42. doi: 10.1016/s0091-6749(06)80008-4. [DOI] [PubMed] [Google Scholar]
- 6.Clark DJ, Lipworth BJ. Adrenal suppression with chronic dosing of fluticasone propionate compared with budesonide in adult asthmatic patients. Thorax. 1997;52:55–58. doi: 10.1136/thx.52.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Boorsma M, Andersson N, Larsson P, Ullman A. Assessment of the relative systemic potency of inhaled fluticasone and budesonide. Eur Respir J. 1996;9:1427–1432. doi: 10.1183/09031936.96.09071427. [DOI] [PubMed] [Google Scholar]
- 8.Donnelly R, Williams KM, Baker B, Badcock CA, Day RO, Seale PJ. Effects of budesonide and fluticasone on 24 hour plasma cortisol: a dose response study. Am J Respir Crit Care Med. 1997;156:1746–1751. doi: 10.1164/ajrccm.156.6.9703003. [DOI] [PubMed] [Google Scholar]
- 9.Lipworth BJ, Wilson AM. Dose response to inhaled corticosteroids: benefits and risks. In: Chapman K, editor. Semin Respir Crit Care Med. 1998. p. 19. :633-54. [Google Scholar]
- 10.Allen DB, Mullen M, Mullen B. A meta-analysis of the effect of oral and inhaled corticosteroids on growth. J Allergy Clin Immunol. 1994;93:967–976. doi: 10.1016/s0091-6749(94)70043-5. [DOI] [PubMed] [Google Scholar]
- 11.Silverstein MD, Yunginger JW, Reed CE, Patterson T, Zimmerman D, Li JTC, et al. Attained adult height after childhood asthma: effect of glucocorticoid therapy. J Allergy Clin Immunol. 1997;99:466–474. doi: 10.1016/s0091-6749(97)70072-1. [DOI] [PubMed] [Google Scholar]
- 12.McCowan C, Neville RG, Thomas GE, Crombie IK, Clark RA, Ricketts IW, et al. Effect of asthma and its treatment in growth: four year follow-up of cohort of children from general practices in Tayside, Scotland. BMJ. 1998;316:668–672. doi: 10.1136/bmj.316.7132.668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Toogood JH, Baskerville JC, Markov AE, Hodesman AB, Frarher LG, Jennings B, et al. Bone mineral density and the risk of fracture in patients receiving long-term inhaled steroid therapy for asthma. J Allergy Clin Immunol. 1995;96:157–166. doi: 10.1016/s0091-6749(95)70003-x. [DOI] [PubMed] [Google Scholar]
- 14.Wisniewski AF, Lewis SA, Green DJ, Maslanka W, Burrel H, Tattersfield AE. Crossectional investigation of the effects of inhaled corticosteroids on bone density and bone metabolism in patients with asthma. Thorax. 1997;52:853–860. doi: 10.1136/thx.52.10.853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cumming RG, Leeder SR, Mitchell P. Use of inhaled corticosteroids and the risks of cataracts. N Engl J Med. 1997;3:3378–4014. doi: 10.1056/NEJM199707033370102. [DOI] [PubMed] [Google Scholar]
- 16.Garbe E, Le Lorier J, Boivin JF, Suissa S. Inhaled and nasal glucocorticoids and the risks of ocular hypertension or open angle glaucoma. JAMA. 1997;277:722–727. [PubMed] [Google Scholar]
- 17.Roy A, LeBlanc C, Paquete L, Ghazzo M, Cole J, Cartier A, et al. Skin bruising in asthmatic subjects treated with high doses of inhaled steroids: frequency in association with adrenal function. Eur Respir J. 1996;9:226–231. doi: 10.1183/09031936.96.09020226. [DOI] [PubMed] [Google Scholar]
- 18.Manolitsas ND, Wang G, Devalia JL, Trigg CJ, McAulay AE, Davies RJ, et al. Regular albuterol, nedocromil sodium and bronchial inflammation in asthma. Am J Respir Crit Care Med. 1995;152:1925–1930. doi: 10.1164/ajrccm.151.6.7767541. [DOI] [PubMed] [Google Scholar]
- 19.Sampson A, Holgate S. Leukotriene modifiers in the treatment of asthma. BMJ. 1998;16:1257–1258. doi: 10.1136/bmj.316.7140.1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Virchow JC, Hassal SM, Summerton L, Harris A. Improved asthma control over 6 weeks with Accolate (zafirlukast) in patients on high dose inhaled corticosteroids [abstract] J Invest Med. 1997;45:286A. [Google Scholar]
- 21.Leff JA, Israel E, Noonan MJ, Finn AF, Godard P, Lufdahl CG, et al. Montelukast (MK-0476) allows tapering of inhaled corticosteroids (ICS) in asthmatic patients while maintaining clinical stability [abstract] Am J Respir Crit Care Med. 1997;155(suppl):976. [Google Scholar]
- 22.Nayak AS, Anderson P, Charous BL, Williams F, Simonson K. Equivalence of adding zafirlukast versus double-dose inhaled corticosteroids in asthmatic patients symptomatic on low dose inhaled corticosteroids. J Allergy Clin Immunol. 1998;101(suppl):S233. [Google Scholar]
- 23.Merck Sharp & Dohme. Singulair product data sheet. Hoddesdon: MSD; 1998. Data on file. [Google Scholar]
- 24.Knorr B, Matz J, Bernstein JA, Nguyen H, Seidenberg BC, Reiss T, et al. Montelukast for chronic asthma in 6-14 year old children. JAMA. 1998;279:1181–1186. doi: 10.1001/jama.279.15.1181. [DOI] [PubMed] [Google Scholar]
- 25.Reiss TF, Chervinsky P, Dockhorn KJ, Shingo S, Seidenberg B, Edwards TB. Montelukast, a once daily leukotriene receptor antagonist, in the treatment of chronic asthma: a multicenter, randomised, double-blind trial. Arch Intern Med. 1998;158:1213–1220. doi: 10.1001/archinte.158.11.1213. [DOI] [PubMed] [Google Scholar]
- 26.Turpin JA, Edelman JM, DeLucca PT, Pearlman DS. Chronic administration of montelukast (MK-0476) is superior to inhaled salmeterol in the prevention of exercise induced bronchoconstriction (EIB) [abstract] Am J Respir Crit Care Med. 1998;157(suppl):A456. [Google Scholar]
- 27.Malmstrom K, Meltzer E, Prenner B, Lu S, Weinstein S, Wolfe J, et al. Effects of montelukast (a leukotriene receptor antagonist), loratidine, montelukast and loratidine and placebo in seasonal allergic rhinitis and conjunctivitis. J Allergy Clin Immunol. 1998;101(suppl):S97. doi: 10.1067/mai.2000.106040. [DOI] [PubMed] [Google Scholar]
- 28.Nakamura Y, Hoshino M, Sim JJ, Ishii K, Hosaka K, Sakamoto T. Effect of the leukotriene receptor antagonist pranlukase on cellular infiltration in the bronchial mucosa of patients with asthma. Thorax. 1998;53:835–841. doi: 10.1136/thx.53.10.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reicin AS, Wenstein SF, White R, Fineman SM, Nguyen H, Geissler L, et al. Montelukast and loratidine compared to montelukast alone produces additional benefit in the treatment of chronic asthma [abstract] Am J Respir Crit Care Med. 1998;157(suppl):A416. [Google Scholar]
- 30.Kidney J, Diminguez M, Taylor PM, Rose M, Chung KF, Barnes PJ, et al. Immunomodulation by theophylline in asthma, demonstration by withdrawal of therapy. Am J Respir Crit Care Med. 1995;151:1907–1914. doi: 10.1164/ajrccm.151.6.7767539. [DOI] [PubMed] [Google Scholar]
- 31.Evans DJ, Taylor DA, Zetterstrom U, Fan Chung K, O’Connor BJ, Barnes PJ. A comparison of low dose inhaled budesonide plus theophylline and high dose inhaled budesonide for moderate asthma. N Engl J Med. 1997;337:1412–1418. doi: 10.1056/NEJM199711133372002. [DOI] [PubMed] [Google Scholar]
- 32.Sears MR, Taylor DR, Print CG, Lake DC, Li QQ, Flannery EM, et al. Regular inhaled beta-agonist treatment in bronchial asthma. Lancet. 1990;336:1391–1396. doi: 10.1016/0140-6736(90)93098-a. [DOI] [PubMed] [Google Scholar]
- 33.Pauwels R, Lofdahl CG, Postma D, Tattersfield AE, O’Byrne P, Barnes PJ, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med. 1997;337:1405–1411. doi: 10.1056/NEJM199711133372001. [DOI] [PubMed] [Google Scholar]
- 34.McIvor RA, Pizzichini E, Turner MO, Hussack P, Hargreave FE, Sears MR. Potential masking effect of salmeterol in airway inflammation in asthma. Am J Respir Crit Care Med. 1998;158:924–930. doi: 10.1164/ajrccm.158.3.9802069. [DOI] [PubMed] [Google Scholar]
- 35.Ramage L, Lipworth BJ, Ingham CG, Cree IA, Dhillon DP. Reduced protection against exercise induced bronchoconstriction after chronic dosing with salmeterol. Respir Med. 1994;88:363–368. doi: 10.1016/0954-6111(94)90042-6. [DOI] [PubMed] [Google Scholar]
- 36.Lipworth B, Tan S, Devlin M, Aiken T, Baker R, Hendrick D. Effects of treatment with formoterol on bronchoprotection against methacholine. Am J Med. 1998;104:431–438. doi: 10.1016/s0002-9343(98)00086-2. [DOI] [PubMed] [Google Scholar]
- 37.Tan KS, Hall IP, Dewar J, Dow E, Lipworth BJ. Association between β2-adrenoceptor polymorphism and susceptibility to bronchodilator desensitisation on moderately severe stable asthmatics. Lancet. 1997;350:995–999. doi: 10.1016/S0140-6736(97)03211-X. [DOI] [PubMed] [Google Scholar]