Abstract
Purpose of review:
To summarize the evidence and clinical implications of weight and body composition changes during midlife in women and provide an overview of weight gain prevention and management in this population.
Recent findings:
Aging-related changes such as decreased energy expenditure and physical activity, are important culprits for weight gain in midlife women. The hormonal changes of menopause also influence body adiposity distribution and increase central adiposity. These body changes can have health consequences including the development of cardiometabolic diseases, osteoarthritis, cancer, worsening in cognition, mental health, and menopause symptoms.
Summary:
Midlife women experience changes related to aging, menopause, and lifestyle which favor weight gain. Clinical practice should focus on early counseling and anticipatory guidance on the importance of dietary changes and physical activity to attenuate this phenomenon. Future research should focus on the longitudinal relationship between weight trends in midlife and health consequences and mortality.
Keywords: Midlife women, Weight gain, Body composition, Metabolic rate, Lifestyle intervention
Introduction
Midlife is the period of life between early adulthood and old age, typically between the ages of 40 and 65. This period is characterized by a variety of physical, psychological, and social changes, particularly in women as it coincides with the menopause transition. Concerns about weight are common among women during midlife, with reports of weight gain and increased abdominal adiposity, occurring in isolation or together. There are several factors that can contribute to weight and body composition changes, including a decrease in physical activity, declining hormone levels (i.e., decreased estrogen and androgen levels, and estrogen to androgen ratio), and dietary changes. Weight and body composition changes in midlife may result in the development of overweight and obesity, contributing to an overall increased risk in cardiovascular disease (CVD) and mortality. In this review, we will summarize the evidence and clinical implications of weight and body composition changes during midlife in women and provide a brief overview of weight gain prevention and weight management in this population.
Weight and Body Composition changes
Weight gain is a common concern for midlife women and has been reported in several studies. In the study of women across the nation (SWAN), midlife women (n=3064; mean age 45.9 years) gained an average of 0.7kg (1.5 pounds) per year, independent of age at baseline or menopause status.[1] Although racial and socioeconomic disparities impacted body weight at baseline (i.e., non-white and lower socioeconomic status are associated with higher baseline weight), in a following study, weight gain occurred in all midlife women, suggesting the uniformity of this trend.[2] Similarly, in the Nurses’ Health Study (NHS), midlife women (n= 41,518; mean age 53.7 years) gained an average of 3kg (6.8 pounds) over 8 years of follow-up, for an average of 0.4 kg (0.85 pounds) per year.[3] Comparably, women without any chronic co-morbidities in the Biobehavioral Health in Diverse Midlife Women Study (n=232; mean age 43.42 years) gained an average of 1.4 kg (3 pounds) over 2 years, for an average of 0.7 kg (1.5 pounds) per year.[4] In another study, almost 70% of women reported gaining weight during the menopause transition,[5] and weight gain appeared to continue even postmenopause.[5, 6] However, weight gain is not limited to midlife; numerous studies have documented an average yearly weight gain of 0.5 kg to 1 kg (1–2 lb) in US adults.[7] In one specific US study, women aged 36–79 experienced an increase of 5.4 kg (approximately 11.9 lb) over the prior 10 years, with the age group of 36–39 years showing the highest weight gain (9 kg or 19.8 lb).[8] Additionally, in another Australian study, women aged 18–23 years gained 8.6 kilograms (approximately 18.9 lb) over a 13-year period, which on average is about 0.66 kg (approximately 1.46 lb) per year.[9]
Nonetheless, persistent weight gain throughout adulthood, particularly when it does not level off during midlife, can significantly contribute to the progression or onset of overweight and obesity. In 2015, the Women’s Health Initiative (WHI) study reported that in women aged 50 to 79 years, one-third had overweight and one-quarter had obesity at baseline.[6] Further, 36% of the participants gained clinically significant weight (>3% of their weight) over 3 years of follow-up.[6] In the US, the prevalence of obesity has increased over the past few decades across all age groups of women; however, the prevalence of obesity among 40–60-year-old women was higher compared to women between the ages of 20 and 39: 43.3 vs. 39.7%, respectively.[10]
In fact, current data may underestimate the incidence of weight gain in midlife women. This may be due to several reasons, the first of which is that some of the studies reporting on this topic included mostly women with preexisting excess adiposity, and many women who experience weight gain during this stage of their lives may still not yet have overweight or obesity.[1, 2, 6, 11] Second, some national registries do not address overweight as part of the spectrum of excess adiposity. For instance, the National Center for Health Statistics (NCHS) only reports the prevalence of obesity without addressing the equally prominent issue of overweight.[10] Third, many studies only report the change in weight in women across the menopause transition, rather than across the midlife stage.[11–13] Therefore, data regarding the average anticipated weight gain in midlife women are incomplete. Nevertheless, weight gain in midlife women is observed in clinical practice, and it predisposes midlife women to obesity and other adverse health outcomes.[10]
Unlike weight gain, a more specific phenomenon to midlife women is the change in body composition.[1] There is a consistent rise in body fat mass over the years in women. However, research indicates that midlife women experience a more precipitous increase of body fat from age 45 to 75 years compared to younger and older women.[14] In addition, when stratifying women by menopause status, peri- and postmenopausal women tend to have significantly higher absolute and relative total body fat than premenopausal women.[12, 13, 15] Similarly, midlife women have an increase in central fat deposition. In the SWAN study (n=3064; mean age 45.9 years), midlife women had an average increase in waist circumference of 2.2 cm (0.9 in) over a span of 3 years.[1] In contrast, the Biobehavioral Health in Diverse Midlife Women study (n=232; mean age 43.42 years) showed no statistically significant difference in waist circumference over a 2-year follow-up. It is important to note that in the SWAN study, after the 3 years of follow-up, 60.9% of the women were perimenopausal, and 13% of women were postmenopausal, while in the Biobehavioral Health in Diverse Midlife Women study, most women (75%) were still premenopausal.[1, 4] This suggests that the change in body composition in midlife may be primarily driven by the hormonal changes that accompany the menopause transition. This theory is supported by the fact that waist circumference progressively increases through the menopause transition, from pre-, to peri-, and then to postmenopause.[15] Finally, midlife women tend to lose lean mass, and postmenopausal women tend to have significantly lower muscle mass than premenopausal women.[16, 17]
Pathophysiology
It can be challenging to discern the effects of aging from the effects of menopause on weight gain in midlife women as these two life events overlap. Women are more likely to report weight gain in midlife, but both men and women gain weight during this stage of life, suggesting the role of aging.[18] A systematic review that included 2472 women revealed that weight gain was attributed to aging rather than menopause status after evaluating different statistical models to compare the two.[12] However, menopause was a strong predictor of increased central fat deposition as waist circumference increased by 4.63 cm (1.82 inches) during the menopause transition.[12] Therefore, aging seems to be the playing a key role in weight gain, although menopause alters body composition in midlife women. However, this may be an oversimplification given the various mechanisms behind weight gain in midlife women (Figure 1).
Figure 1:
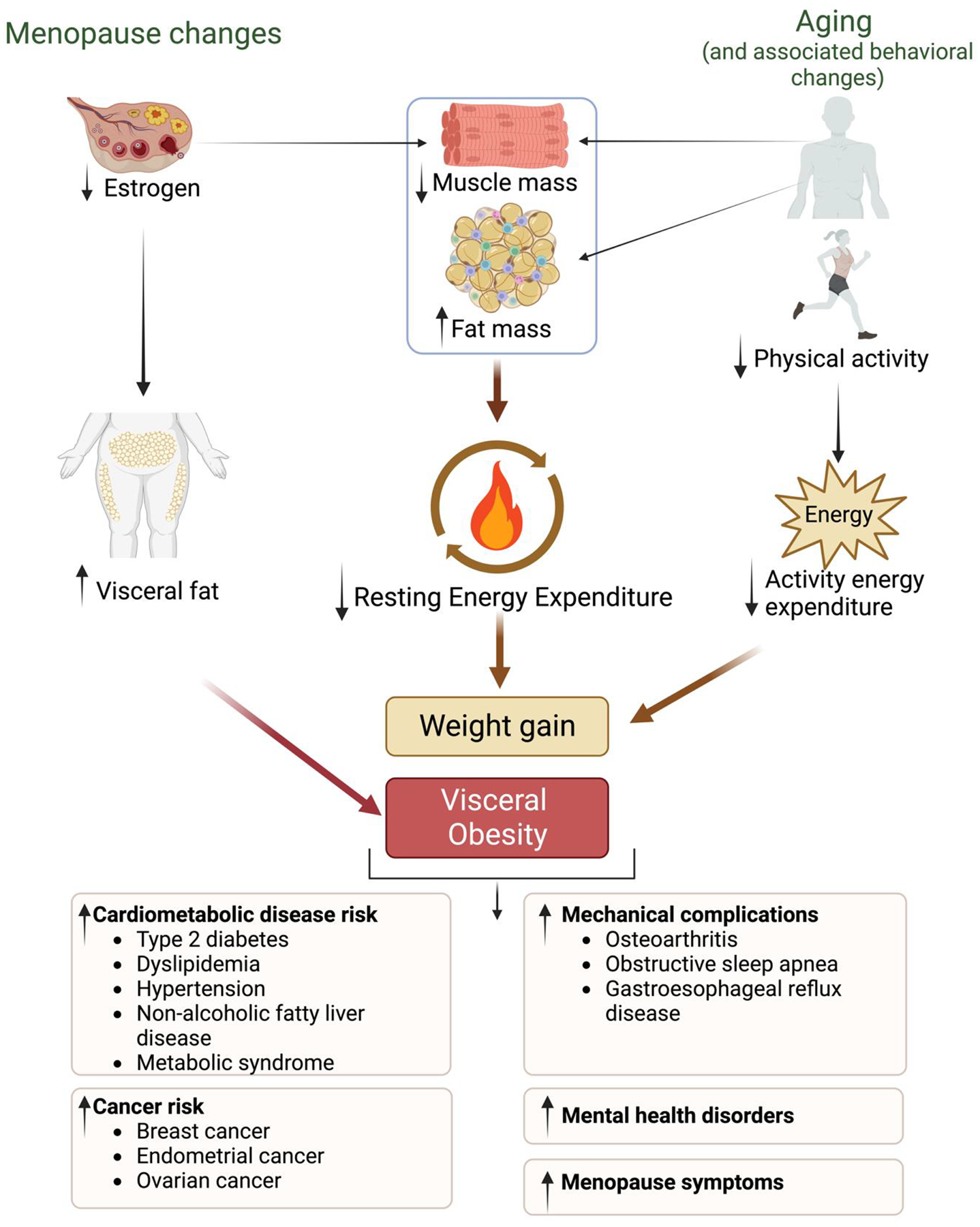
Changes in Weight and Body Composition in Midlife Women and Clinical Consequences
Energy balance
Increases in stored energy—often presenting as weight gain—results from the discordance between calorie intake and total energy expenditure (TEE). TEE is determined by the basal metabolic rate (BMR), activity energy expenditure, and dietary induced thermogenesis (DIT).[19] BMR accounts for 60–80% of TEE, and it is used interchangeably with resting metabolic rate (RMR) in the literature.[20, 21] Both measure oxygen consumption and/or carbon dioxide production via indirect calorimetry, with the only difference being that BMR is measured under stricter conditions, i.e., in the morning after an overnight fast and after a 24-hr exercise- and stress-free period.[22] In general, women have lower absolute BMR and RMR than men which may be explained by the differences in body composition (lower lean mass and higher fat mass).[22][23] On the other hand, DIT contributes to 10% of TEE, while activity-related energy expenditure encompasses a range of 15–30% of TEE.
BMR and RMR decline linearly with age, consequently resulting in a decline in TEE. [20–22] Consequently, as a woman ages, weight gain can naturally occur if dietary habits and physical activity remain unchanged. Body lean mass, i.e., muscle mass, is one of the strongest determinants of BMR and the major contributor to the decline in BMR with age. As such, age-related muscle mass loss may explain most of the decline in BMR in midlife women[19] After the age of 30, muscle mass loss occurs at a rate of 3–8% per decade, making it more challenging to maintain muscle mass.[17] This is further exacerbated by the hormonal changes during menopause.[16] Estrogen deficit after menopause is associated with increased inflammatory markers that are associated with muscle degradation.[24] Moreover, estrogen acts directly on muscle stem cells to promote muscle maintenance, regeneration, and repair, effects that are attenuated after menopause.[25] In addition, women experience androgen loss as they age, mainly during their early reproductive years[26], with a slower decline during midlife, and no apparent additional effect of the menopause transition.[27] In women from the Melbourne Women’s Midlife Health Study, testosterone levels were stable during midlife, but dehydroepiandrosterone sulfate (DHEAS) declined by 1.5% annually.[27] While the literature is mixed, there is some evidence that lower testosterone levels are associated with lower body lean mass,[28] and increased risk of sarcopenia in women[29]. Therefore, androgen loss may contribute to the muscle loss in midlife women, and hence to the decrease in BMR.
Further, physical activity and resistance training play a key role in slowing muscle loss and increasing BMR and active energy expenditure. Almost 30% of adults ≥50 years of age in the US report no physical activity outside of work.[30] The Physical Activity Guidelines Advisory Committee recommends at least 150 minutes (about 2 and a half hours) a week of moderate-intensity exercise.[31] In the SWAN cohort, only 7.2% of the study population reported physical activity levels that consistently met the recommendation over their midlife span.[32] Similarly, two studies based on the SWAN population reported that 25% of midlife women who participated in the registry had decreasing physical activity over a 15-year follow-up, and that the decrease in physical activity was associated with the most weight gain, when compared to unchanged or increased level of physical activity.[1, 33]
Privilege
In terms of dietary changes, lower intake of dietary fiber and higher intake of dietary fat in midlife is associated with increased risk of weight gain, but longitudinal studies report a decrease rather than an increase in calorie intake in midlife women.[6, 34, 35] Moreover, weight gain in midlife women has been better explained by decreased levels of physical activity as opposed to increased caloric intake in regression models.[35]
Vasomotor Symptoms and Sleep
The SWAN cohort has provided evidence that frequent VMS (hot flashes and night sweats) are associated with subsequent weight gain, higher BMI, and higher waist circumference in midlife women.[36] Both the onset of high-frequency VMS and the increase in VMS frequency over time were associated with greater weight gain during subsequent visits.[37] This is relevant as VMS are reported by up to 75% of women during the menopause transition.[38] The mechanisms behind this association have not been established. VMS have been associated with decreased exercise and overall physical activity, important risk factors for weight gain.[37, 39–42] Additionally, data support that poor sleep quality may have an impact on weight gain rather than sleep duration, as another report from the SWAN cohort revealed that sleep duration was not associated with prospective weight gain.[36, 43] In the SWAN cohort, self-reported sleep problems, defined as trouble falling asleep, waking up early, and/or waking up several times per night occurring three times or more, accounted for almost 30% of the effect of VMS on weight gain.[36] This suggests that sleep disruptions may significantly contribute to weight gain but are not the sole factor in women experiencing high-frequency VMS.
Clinical implications
The accumulation of excess adiposity can contribute to the development of cardiometabolic diseases, mechanical complications, cancer, changes in cognition and mental health, and worse menopause symptoms. While there is robust evidence on the association between obesity (primarily based on BMI) and these disorders, there is a paucity of data on the longitudinal relationship between weight gain and the incidence of these conditions throughout women’s lifespans, particularly as it relates to the midlife stage. Adiposity-associated diseases can increase with age, but as summarized below, there is a rapid increase in their prevalence during midlife in women, with research indicating a potential role of hormonal changes as women go through menopause. [44]
Cardiometabolic Risk Factors
Hypertension
It is not surprising that the incidence of hypertension increases in midlife as the relationship between aging and increased blood pressure has been well established.[45] However, the incidence of hypertension varies across women’s lifespans, with a precipitous rise in its prevalence in early to mid-adulthood, a rise that gradually becomes less pronounced in midlife and late adulthood.[46] In the United States, between 2017 and 2020, the prevalence of hypertension was 15% in women aged 20–34 years old, 32% in women aged 35–44 years old, 49% in women aged 45–54 years, 63% in women aged 55–64 years old, 75% in women aged 65–74 years old, and 81% in women aged ≥75 years old.[46] In addition to aging, weight gain is a strong predictor of hypertension. Particularly, younger age at onset of weight gain, i.e., early adulthood and extending into early midlife, is associated with a higher risk of incident hypertension, whereas late-onset weight gain is not.[47, 48] Sundstrom et al., reported that every 10 kg (22 pounds) of weight gain in women, starting at age 20 and throughout midlife, was associated with a 3.2 and 2.4 mmHg increase in systolic and diastolic blood pressures, respectively.[48] Similarly, data from the NHS revealed that every 4.5 kg (10 pounds) of weight gained during early midlife was associated with a 30% increased risk of hypertension.[49] The changes in blood pressure across the midlife period in women has been shown to be independent of menopause status.[50]
Metabolic Dysfunction-Associated Steatotic Liver Disease
In women younger than 50 years old the prevalence of metabolic dysfunction-associated steatotic liver disease (MASLD-previously referred to as non-alcoholic fatty liver disease) tends to be stable around 10–15% and increases steadily afterwards, reaching a peak of 20–35% at the age of 60 years.[51–54] Although it could be theorized that aging could explain this upward trend, at least in part, this phenomenon is not seen in men aged 50 or older, in whom MASLD prevalence remains stable.[52] Research indicates that the menopause transition may be a major risk factor, with the prevalence of MASLD doubling in postmenopausal women compared to premenopausal women.[55] MASLD is the leading cause of chronic hepatic disease, cirrhosis, and liver cancer globally.[56] In women, MASLD is the leading indication for liver transplantation.[57] Like in the general population, weight gain and increased adiposity, particularly in the central distribution, are associated with increased risk of MASLD in midlife women. However, their effects have not been quantified in this population.[58, 59]
Dysglycemia and Dyslipidemia
As with other cardiometabolic risk factors, diabetes and dyslipidemia incidence vary across women’s lifespans, slowly rising in early adulthood, then rapidly increasing between the ages 40 and 55 years and slowly decreasing thereafter.[60] Consequently, compared to women aged 20–40 years, the prevalence of diabetes and dyslipidemia in midlife women increases by 3- to 4-fold, and compared to women aged 65 years and older, the prevalence of both diseases is slightly lower during midlife.[61–64] All published data on dysglycemia and dyslipidemia assess the effect of baseline weight or BMI rather than the longitudinal relationship between weight gain and the incidence of dysglycemia and dyslipidemia. For instance, data from the SWAN cohort has revealed that over the span of a follow up of 16 years, the incidence of type 2 diabetes was 37% in midlife women, with women with incident diabetes having a 14% higher BMI at baseline.[62] Similarly, data from multiple cohort studies, including the SWAN, the Dutch Lifelines Cohort, and the UK Medical Research Council National Survey of Health Development, indicate that an elevated BMI during women’s midlife, particularly as they approach the menopause transition, is associated with a greater increase in total cholesterol, low density lipoprotein cholesterol, and apolipoprotein A and B, independent of age.[65–67]
Cardiovascular disease
CVD is the leading cause of death among women.[44] In women, the prevalence of CVD increases with age, particularly after midlife, and it is estimated as follows: 17% in women aged 20–39 years, >50% in women aged 40–59 years, 77% in women aged 60–79 years, and >90 after age 80 years.[46] Evidence suggests that menopause is a major and independent risk factor for CVD, with an inverse linear association between age at menopause onset and CVD risk.[68–70] Weight gain and changes in body composition among midlife women are associated with increased risk of CVD and mortality.[44] Multiple studies have reported a strong association between obesity and central obesity (independent of BMI) and increased CVD mortality in midlife women.[71, 72] Indeed, data suggest that women with normal BMI but with central obesity may have a greater CVD risk compared to women with BMI in the obesity range, and without central obesity.[72] This risk appears to be the result of a combination of midlife metabolic and cardiovascular changes which are exacerbated by the menopause transition which is now considered a female-specific CVD risk factor.[44] Throughout the menopause transition, independent of age, women experience an increase in carotid intima media thickness, carotid atherosclerosis and remodeling, arterial stiffness, and fat deposition within and outside the pericardial sac, changes that are exacerbated by excess adiposity.[73, 74]
Cancer
Excess adiposity poses a risk for esophageal, gastric, colorectal, hepatic, gallbladder, pancreatic, breast, endometrial, ovarian, and kidney cancers, among others.[75] A study by Chadid et al., reported that in midlife women, weight gain of more than 0.45 kg (1 pound) per year is associated with a 30% increased risk of obesity-related cancers.[76] Furthermore, weight gain combined with metabolic dysfunction (e.g., prediabetes, type 2 diabetes, dyslipidemia, hypertension), increased this risk by 77%.[76] In this study, women transitioning from a BMI in the normal range to the overweight range during midlife had a 60% greater risk of obesity-related cancer.[76] These findings are concordant with the evidence on weight gain and breast cancer risk, the most common cancer in women. Data from the National Institutes of Health-AARP Diet and Health Study showed that in postmenopausal women not on hormone therapy (HT), weight gain of 10–19.9 kg (22–44 pounds) from age 35 to 50 increased the risk of postmenopausal breast cancer by 40%, whereas weight gain greater than 50 kg (110 pounds) more than doubled this risk.[77] Along the same lines, another study reported that every 5 kg (11 pounds) increase in weight was associated with a 39% increased risk of postmenopausal endometrial cancer, a 13% increased risk of postmenopausal ovarian cancer, and a 4% increased risk of colorectal cancer.[78, 79] In midlife women, the changes in body composition are of importance as studies have reported a strong association between central obesity, independent of BMI, and increased cancer mortality, particularly among postmenopausal women.[71] Notably, while weight gain and excess adiposity late in midlife, particularly after menopause, is associated with an increased risk of breast cancer, excess adiposity during puberty and early adulthood may confer protection against breast cancer, and specifically against hormone receptor-positive breast cancer.[80, 81]
Obstructive sleep apnea
Excess adiposity has been identified as major risk factor for obstructive sleep apnea (OSA) across adulthood, irrespective of sex.[82] The global prevalence of OSA among women varies between 3% and 25%, and data reveal that its prevalence increases significantly in midlife women.[83] For instance, in a large population-based study, the prevalence of mild to severe OSA among women aged less than 45 was calculated at 24%, compared to 56% in women aged 45–54, and 75% in women aged between 55–70.[84] The effect of weight and body composition changes that women experience in midlife on the risk of sleep apnea incidence and severity has not been studied.
Osteoarthritis
Osteoarthritis is the leading cause of chronic pain and disability in the US, affecting at least 19% of adults over the age of 45 years, with a higher prevalence among women.[85–88] Increased body adiposity is one of the early established risk factors for osteoarthritis in the scientific literature.[89] In the Melbourne Women’s Midlife Health Project, BMI was one of factors contributing to the increased prevalence of osteoarthritis in otherwise healthy middle-aged women over an 11-year follow-up period.[87] Importantly, the increased mechanical joint load is not the only mechanism explaining this association given that increased adiposity has been shown to enhance the risk of arthritis in non-weight bearing joints such as hand osteoarthritis.[90] One explanation is that the increase in inflammatory cytokines from excess adipose tissue, especially visceral adipose tissue, mediates synovial inflammation.[87, 91]
Cognition
Overweight and obesity during midlife have been associated with a decline in cognitive function later in life in both men and women. In one study, male and female participants who had overweight/obesity during midlife (mean age 41.6 years) had worse cognitive function with a steeper decline later in life (over a 20-year period).[92] Similarly, in another study, overweight/obesity during late midlife (mean age 61.1 years) was associated with worse cognitive function later in life, but a steeper decline only in verbal abilities and memory.[93] The influence of weight change during midlife on cognitive function is less understood. In the latter study, weight loss rather than weight gain during midlife was associated with cognitive function decline later in life.[93] However, in this study, those who lost weight across the midlife stage had the highest BMI in early midlife. Therefore, BMI may be a generic parameter that does not accurately reflect the cardiometabolic changes taking place. In a recent cross-sectional analysis from the Kronos Early Estrogen Prevention Study (KEEPS), in 621 recently menopausal women, mean age 52.7years, central obesity rather than overall obesity was associated with lower cognitive function measured by Modified Mini Mental Status score. These findings underscore the need for considering adiposity measures beyond BMI to assess and manage the negative consequences of excess adiposity.[94]
Psychosocial Considerations
There is no data reporting on the effect of weight gain and mental health diseases in midlife women. A complex and bidirectional interaction exists between body weight and mental health, and it is usually reciprocal. In midlife women, higher BMI may be associated with an increased prevalence of depressive symptoms and major depression diagnosis.[95] Further, midlife women with higher BMI are more likely to have lower life satisfaction and higher physical and psychological health stress such as body aches, difficulty relaxing, headaches, or feeling stressed and nervous.[96]
Excess adiposity has been associated with sexual dysfunction in midlife women, which based on the NHS cohort, is reported by 50% of sexually active women.[97] In general, adiposity per se can pose physical limitations for sexual activity in women, without necessarily affecting sexual desire and satisfaction.[98] However, women with excess adiposity are more likely to experience negative body image that is in turn associated with lower arousal, sexual desire, lubrication, orgasm, and sexual satisfaction.[99] In midlife women, baseline BMI is inversely associated with frequency of intercourse but there is no association between BMI and sexual desire, arousal, and orgasm.[100] Further, longitudinal analysis over a 14-year study period showed that sexual desire, arousal, orgasm, and frequency of intercourse declined over time, and while BMI increased over time, there was no association between the changes in BMI and sexual function variables.[100] Studies assessing the effect of weight loss on sexual function suggest that lifestyle interventions, pharmacotherapy with naltrexone/bupropion, and bariatric surgery improve sexual function in women with overweight and obesity.[101–103]
Menopause Symptoms
Vasomotor Symptoms
Traditionally, in women with overweight and obesity, increased estrogen production via androgen aromatization in the adipose tissue was thought to protect against VMS. Interestingly, the association between excess adiposity and VMS may vary by menopause stage. On one hand, data from several large cohort studies suggest that higher adiposity is associated with lower odds of VMS during the late menopause, and on the other hand, women with excess adiposity, particularly in the central and subcutaneous distribution, report more frequent and more severe VMS early in the menopause transition. There is no robust evidence suggesting an association between weight gain or increased abdominal adiposity and incidence, frequency, or intensity of VMS.[104] However, lifestyle changes resulting in weight loss have been shown to decrease the number and intensity of VMS.[105] The connection between excess adiposity and increased VMS is not well understood. Thermodysregulation may explain, at least in part, the connection between excess adiposity and increased VMS during early menopause. Thermodysregulation could be the result of decreased heat dissipation from excess fat accumulation and altered adipokine secretion, particularly increased leptin levels.[106]
Urogenital Symptoms
Excess adiposity has been associated with increased urogenital symptoms. The genitourinary syndrome of menopause (GSM) is an umbrella term that encompasses vaginal, genital, and urinary symptoms affecting up to 85% of women in the menopause transition.[107] Compared to normal-weight women, postmenopausal women with obesity are four times more likely to have vulvovaginal itching and irritation.[108] Similarly, women with obesity have a higher prevalence of urinary incontinence and urinary urgency.[40]
Weight Management in Midlife Women
Weight Gain Prevention
The US Preventive Services Task Force (USPSTF) recommends interventions for adults with overweight or obesity but provides no guidance for prevention of weight gain in adults with a BMI below 25 kg/m2.[109] A recent systemic review by the Women’s Perspective Services Initiative (WPSI) showed that counseling women on weight loss had favorable weight outcomes with minimal adverse events.[110] Given these data and the weight and body composition trends in midlife women, in 2022, the WPSI provided guidelines to counsel midlife women with normal or overweight BMI (18.5–29.9 kg/m2) to maintain weight or limit weight gain to prevent obesity.[111] Counseling should be proactive and started in women in their late 30’s, while in their early 40’s, an individualized weight management intervention plan should be instituted.[112] Despite these general guidelines, there is limited data supporting specific recommendations on interventions in midlife women.[113] Based on data from the Pittsburgh Women’s Healthy Life Project, a hypocaloric diet of 1300 Kcal/day with reduced saturated fat and cholesterol throughout the menopause transition can prevent weight gain over a 4.5-year follow-up period.[114] Similarly, based on the WHI, a low-fat diet paired with an increased intake of vegetables, fruits, and grains, and without purposeful caloric restriction, prevents weight gain in postmenopausal women.[115] Importantly, interventions focusing on education, coaching, motivational interviewing, and dietary changes, alone or combined with physical activity, are consistently more effective than physical activity interventions alone.[114–119]
Management of Menopause Symptoms
As changes associated with the menopause transition, particularly in relation to VMS, can significantly impact multiple aspects of a woman’s lifestyle and contribute to weight gain and body composition changes, special considerations should be given to this aspect. There is robust evidence supporting that HT decreases VMS and can lead to improved sleep, increased activity, and overall increased quality of life, all factors that can mitigate weight gain in midlife women.[120] As such, HT should be considered for all women with bothersome VMS.[120] If HT is contraindicated, women should be offered alternative treatment modalities. Cognitive behavioral therapy and hypnosis are effective interventions that improve the severity and severity and frequency of VMS, respectively.[121, 122] Similarly, nonhormonal pharmacologic interventions have been shown to be effective for the management of VMS.[123] Particular attention should be paid to avoiding weight promoting medications. If weight gain is a major concern, weight neutral medications including venlafaxine, desvenlafaxine, oxybutynin, and fezolinetant are preferred, whereas gabapentin, clonidine, paroxetine, citalopram, and escitalopram should be avoided.[120, 123, 124]
HT has been associated with decreased visceral adiposity, decreased total adiposity, decreased muscle mass loss, and a favorable effect on energy expenditure; however, these associations were of low magnitude.[125–128] HT use has also been associated with improved metabolic parameters, such as a reduced risk of insulin resistance and new onset diabetes.[129] While HT provides some body composition and metabolic benefits, it should not be prescribed for these indications, especially as these effects have not been proven to have any positive clinical impact on major health outcomes in women. For clinical awareness, HT is weight neutral, and while some women report weight gain after HT initiation, there is no evidence that HT increases adiposity. The reported weight gain associated with HT use may be due to the effect of estrogen and/or progestogen-mediated fluid retention.[130]
Overweight and Obesity Treatment
Screening for overweight and obesity should be performed throughout a woman’s lifespan. While BMI is the universal screening tool, given the changes in body composition and body fat redistribution that occur in midlife women, other measures such as waist-to-hip ratio or measurement of whole-body adiposity by dual-x-ray absorptiometry (DXA) or bioimpedance could be considered for screening and monitoring of progress.[112, 113] In women living with overweight or obesity (BMI of 25 kg/m2 or greater, waist-to-hip ratio of 0.85 or greater, and/or body fat for greater than 30%), each visit with a health care practitioner should be considered an opportunity to assess awareness and knowledge of the disease and readiness to discuss and to start a weight management program.[112] Excess adiposity, from overweight to obesity, is a complex and multifactorial disease that can affect virtually all body systems and as such, requires an interdisciplinary approach.[112]
In general, weight management in midlife women should not be different than in the general population with a few special considerations. Comprehensive lifestyle interventions remain the backbone of any treatment plan for weight gain prevention or weight loss, and should include medical nutrition therapy, exercise, and behavioral interventions. A protein intake of at least 30% of total daily caloric intake or at least 1.2 g/kg of body weight has demonstrated favorable effects on body composition during a weight loss intervention in middle aged women.[131] Exercise has a modest effect on weight loss but improves body composition by reducing visceral adiposity and protecting against excessive lean mass loss, which is of particular importance as midlife women go through the menopause transition.[132, 133] In midlife women, cognitive behavioral therapy for weight loss has not only shown to result in weight reduction but is also associated with improved restrained eating and quality of life.[134]
Due to metabolic and behavioral adaptations in response to caloric restriction, some women may not achieve any weight loss, and among the ones who do, most will experience weight regain with lifestyle interventions. Hence, in many cases, second level therapies including antiobesity medications, bariatric endoscopic procedures, and metabolic/bariatric surgery need to be considered in conjunction with lifestyle changes. There are established indications for the use of these tools (Table 1). As we aim to improve the treatment of overweight/obesity and its metabolic complications, it is customary practice to use multimodal approaches for treatment of this disease. There are no studies assessing the short- and long-term outcomes of these interventions in midlife women specifically. For now, when determining the appropriate intervention, factors such as the goal weight loss, co-existing health conditions, and potential complications should be considered.
Table 1.
Guidelines for Antiobesity Treatments
| BMI╲ Intervention | Lifestyle | Anti-obesity medications | Sleeve Gastroplasty | Bariatric Surgery |
|---|---|---|---|---|
| 25–26.9 | Yes | |||
| 27–29.9 | Yes | Yes, with adiposity-related comorbiditiesa | Yes, with adiposity-related comorbiditiesa | |
| 30–34.9 | Yes | Yes | Yes | Yes, with metabolic syndrome |
| 35–39.9 | Yes | Yes | Yes | Yes, with adiposity-related comorbiditiesa |
| >40 | Yes | Yes | Yes | Yes |
| Estimated weight loss at 1 year (%)[135] | 5–7% | 5–15% | 15–17% | 25–30% |
Indicated if adiposity-related comorbidities are present. These include type 2 diabetes, hypertension, dyslipidemia, cardiovascular disease, obstructive sleep apnea, gastroesophageal acid reflux, osteoarthritis, among others.
Conclusions
Weight gain experienced by women during midlife is a result of declining BMR, decrease in physical activity, and changes in body composition that include muscle mass loss and increased fat mass. The weight gain and body composition change that women experience during midlife increase the risk of developing overweight or obesity and its clinical ramifications, leading to increased morbidity and mortality. As the prevalence of overweight and obesity in women rise during and after midlife, it is of utmost importance to implement strategic interventions to prevent weight gain and manage overweight and obesity in this population. Efforts should focus on behavioral and lifestyle changes, particularly decreasing caloric intake, and increasing physical activity, along with second level antiobesity treatments when needed. While there are robust data on the effect of obesity on adiposity-related diseases in midlife women, there remains a paucity of data on the longitudinal relationship between weight trends in midlife and the incidence of cardiometabolic risk factors, CVD, and cardiovascular and overall mortality.
Funding:
This publication was supported by Grant Number K12 HD065987
Footnotes
Competing Interests:
MDH: None
MS: None
EK: Ekta Kapoor has no conflicts of interest related to the subject of this manuscript. However, over the past 36 months she has had the following conflicts of interest: She has been a consultant for Astellas and Mithra Pharmaceuticals, Scynexis and Womaness. She receives grant support from Mithra Pharmaceuticals. She has received payment for development of educational content from Med Learning Group and Academy of Continued Healthcare Learning. She has received honoraria for CME activity from PriMed and OBG Management.
CLS: None
SSF: Stephanie Faubion has no conflicts of interest related to the subject of this manuscript. However, over the past 36 months, she has received honoraria for lectures from PriMed. She is also the medical director of The Menopause Society.
References
Papers of particular interest, have been highlighted as:
● of importance
- 1.Sternfeld B, et al. , Physical activity and changes in weight and waist circumference in midlife women: findings from the Study of Women’s Health Across the Nation. Am J Epidemiol, 2004. 160(9): p. 912–22. [DOI] [PubMed] [Google Scholar]
- 2.Lewis TT, et al. , Race, Education, and Weight Change in a Biracial Sample of Women at Midlife. Archives of Internal Medicine, 2005. 165(5): p. 545–551. [DOI] [PubMed] [Google Scholar]
- 3.Field AE, et al. , Dietary fat and weight gain among women in the Nurses’ Health Study. Obesity (Silver Spring), 2007. 15(4): p. 967–76. [DOI] [PubMed] [Google Scholar]
- 4.Choi J, et al. , Physical activity, weight, and waist circumference in midlife women. Health Care Women Int, 2012. 33(12): p. 1086–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pimenta F, et al. , Predictors of weight variation and weight gain in peri- and post-menopausal women. J Health Psychol, 2014. 19(8): p. 993–1002. [DOI] [PubMed] [Google Scholar]
- 6.Jung SY, et al. , Risk profiles for weight gain among postmenopausal women: a classification and regression tree analysis approach. PLoS One, 2015. 10(3): p. e0121430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hutfless S, et al. , AHRQ Comparative Effectiveness Reviews, in Strategies to Prevent Weight Gain Among Adults. 2013, Agency for Healthcare Research and Quality (US): Rockville (MD). [PubMed] [Google Scholar]
- 8.Tucker LA and Parker K, 10-Year Weight Gain in 13,802 US Adults: The Role of Age, Sex, and Race. Journal of Obesity, 2022. 2022: p. 7652408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Holowko N, et al. , Educational mobility and weight gain over 13 years in a longitudinal study of young women. BMC Public Health, 2014. 14: p. 1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hales CM, et al. , Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017–2018. NCHS Data Brief, 2020(360): p. 1–8. [PubMed] [Google Scholar]
- 11.Greendale GA, et al. , Changes in body composition and weight during the menopause transition. JCI Insight, 2019. 4(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ambikairajah A, et al. , Fat mass changes during menopause: a metaanalysis. Am J Obstet Gynecol, 2019. 221(5): p. 393–409.e50. [DOI] [PubMed] [Google Scholar]
- 13.Genazzani AR and Gambacciani M, Effect of climacteric transition and hormone replacement therapy on body weight and body fat distribution. Gynecol Endocrinol, 2006. 22(3): p. 145–50. [DOI] [PubMed] [Google Scholar]
- 14.Kyle UG, et al. , Fat-free and fat mass percentiles in 5225 healthy subjects aged 15 to 98 years. Nutrition, 2001. 17(7–8): p. 534–41. [DOI] [PubMed] [Google Scholar]
- 15.Dmitruk A, et al. , Body composition and fatty tissue distribution in women with various menstrual status. Rocz Panstw Zakl Hig, 2018. 69(1): p. 95–101. [PubMed] [Google Scholar]
- 16.Sipilä S, et al. , Muscle and bone mass in middle-aged women: role of menopausal status and physical activity. J Cachexia Sarcopenia Muscle, 2020. 11(3): p. 698–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Volpi E, Nazemi R, and Fujita S, Muscle tissue changes with aging. Curr Opin Clin Nutr Metab Care, 2004. 7(4): p. 405–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zheng Y, et al. , Associations of Weight Gain From Early to Middle Adulthood With Major Health Outcomes Later in Life. Jama, 2017. 318(3): p. 255–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Heydenreich J, et al. , Total Energy Expenditure, Energy Intake, and Body Composition in Endurance Athletes Across the Training Season: A Systematic Review. Sports Med Open, 2017. 3(1): p. 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Roberts SB and Dallal GE, Energy requirements and aging. Public Health Nutr, 2005. 8(7a): p. 1028–36. [DOI] [PubMed] [Google Scholar]
- 21.BASAL METABOLISM IN HEALTH AND DISEASE. Archives of Internal Medicine, 1924. 34(6): p. 890–890. [Google Scholar]
- 22.McMurray RG, et al. , Examining variations of resting metabolic rate of adults: a public health perspective. Med Sci Sports Exerc, 2014. 46(7): p. 1352–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ferraro R, et al. , Lower sedentary metabolic rate in women compared with men. J Clin Invest, 1992. 90(3): p. 780–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Geraci A, et al. , Sarcopenia and Menopause: The Role of Estradiol. Front Endocrinol (Lausanne), 2021. 12: p. 682012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kitajima Y and Ono Y, Estrogens maintain skeletal muscle and satellite cell functions. J Endocrinol, 2016. 229(3): p. 267–75. [DOI] [PubMed] [Google Scholar]
- 26.Davison SL, et al. , Androgen Levels in Adult Females: Changes with Age, Menopause, and Oophorectomy. The Journal of Clinical Endocrinology & Metabolism, 2005. 90(7): p. 3847–3853. [DOI] [PubMed] [Google Scholar]
- 27.Burger HG, et al. , A prospective longitudinal study of serum testosterone, dehydroepiandrosterone sulfate, and sex hormone-binding globulin levels through the menopause transition. J Clin Endocrinol Metab, 2000. 85(8): p. 2832–8. [DOI] [PubMed] [Google Scholar]
- 28.van Geel TA, et al. , Measures of bioavailable serum testosterone and estradiol and their relationships with muscle mass, muscle strength and bone mineral density in postmenopausal women: a cross-sectional study. Eur J Endocrinol, 2009. 160(4): p. 681–7. [DOI] [PubMed] [Google Scholar]
- 29.Yuki A, et al. , Low free testosterone is associated with loss of appendicular muscle mass in Japanese community-dwelling women. Geriatr Gerontol Int, 2015. 15(3): p. 326–33. [DOI] [PubMed] [Google Scholar]
- 30.Watson KB, et al. Physical Inactivity Among Adults Aged 50 Years and Older - United States, 2014. MMWR. Morbidity and mortality weekly report, 2016. 65, 954–958 DOI: 10.15585/mmwr.mm6536a3. [DOI] [PubMed] [Google Scholar]
- 31.Piercy KL, et al. , The Physical Activity Guidelines for Americans. JAMA, 2018. 320(19): p. 2020–2028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang D, et al. , Healthy Lifestyle During the Midlife Is Prospectively Associated With Less Subclinical Carotid Atherosclerosis: The Study of Women’s Health Across the Nation. J Am Heart Assoc, 2018. 7(23): p. e010405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pettee Gabriel K, et al. , Physical activity trajectories during midlife and subsequent risk of physical functioning decline in late mid-life: The Study of Women’s Health Across the Nation (SWAN). Prev Med, 2017. 105: p. 287–294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Duval K, et al. , Effects of the menopausal transition on dietary intake and appetite: a MONET Group Study. Eur J Clin Nutr, 2014. 68(2): p. 271–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Macdonald HM, et al. , Longitudinal changes in weight in perimenopausal and early postmenopausal women: effects of dietary energy intake, energy expenditure, dietary calcium intake and hormone replacement therapy. Int J Obes Relat Metab Disord, 2003. 27(6): p. 669–76. [DOI] [PubMed] [Google Scholar]
- 36.Gibson CJ, et al. , Association between vasomotor symptom frequency and weight gain in the Study of Women’s Health Across the Nation. Menopause, 2023. 30(7): p. 709–716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gibson C, Matthews K, and Thurston R, Daily physical activity and hot flashes in the Study of Women’s Health Across the Nation (SWAN) Flashes Study. Fertil Steril, 2014. 101(4): p. 1110–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gold EB, et al. , Longitudinal analysis of the association between vasomotor symptoms and race/ethnicity across the menopausal transition: study of women’s health across the nation. Am J Public Health, 2006. 96(7): p. 1226–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thurston RC, et al. , Abdominal adiposity and hot flashes among midlife women. Menopause, 2008. 15(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Saccomani S, et al. , Does obesity increase the risk of hot flashes among midlife women?: a population-based study. Menopause, 2017. 24(9): p. 1065–1070. [DOI] [PubMed] [Google Scholar]
- 41.Koo S, et al. , Obesity associates with vasomotor symptoms in postmenopause but with physical symptoms in perimenopause: a cross-sectional study. BMC Womens Health, 2017. 17(1): p. 126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Romani WA, Gallicchio L, and Flaws JA, The association between physical activity and hot flash severity, frequency, and duration in mid-life women. Am J Hum Biol, 2009. 21(1): p. 127–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ogilvie RP and Patel SR, The epidemiology of sleep and obesity. Sleep Health, 2017. 3(5): p. 383–388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Khoudary SRE, et al. , Menopause Transition and Cardiovascular Disease Risk: Implications for Timing of Early Prevention: A Scientific Statement From the American Heart Association. Circulation, 2020. 142(25): p. e506–e532. [DOI] [PubMed] [Google Scholar]
- 45.Sun Z, Aging, Arterial Stiffness, and Hypertension. Hypertension, 2015. 65(2): p. 252–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tsao CW, et al. , Heart Disease and Stroke Statistics—2023 Update: A Report From the American Heart Association. Circulation, 2023. 147(8): p. e93–e621. [DOI] [PubMed] [Google Scholar]
- 47.Li W, et al. , Association between age at onset of overweight and risk of hypertension across adulthood. Heart, 2022. 108(9): p. 683–688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sundström J, et al. , Weight gain and blood pressure. Journal of Hypertension, 2020. 38(3): p. 387–394. [DOI] [PubMed] [Google Scholar]
- 49.Field AE, et al. , Weight Cycling, Weight Gain, and Risk of Hypertension in Women. American Journal of Epidemiology, 1999. 150(6): p. 573–579. [DOI] [PubMed] [Google Scholar]
- 50.Matthews KA, et al. , Are Changes in Cardiovascular Disease Risk Factors in Midlife Women Due to Chronological Aging or to the Menopausal Transition? Journal of the American College of Cardiology, 2009. 54(25): p. 2366–2373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wong VW, et al. , Prevalence of non-alcoholic fatty liver disease and advanced fibrosis in Hong Kong Chinese: a population study using proton-magnetic resonance spectroscopy and transient elastography. Gut, 2012. 61(3): p. 409–15. [DOI] [PubMed] [Google Scholar]
- 52.Wang Z, et al. , Sex-specific prevalence of fatty liver disease and associated metabolic factors in Wuhan, south central China. Eur J Gastroenterol Hepatol, 2014. 26(9): p. 1015–21. [DOI] [PubMed] [Google Scholar]
- 53.Kojima S, et al. , Increase in the prevalence of fatty liver in Japan over the past 12 years: analysis of clinical background. J Gastroenterol, 2003. 38(10): p. 954–61. [DOI] [PubMed] [Google Scholar]
- 54.Wang Z, et al. , Prevalence of nonalcoholic fatty liver disease and its metabolic risk factors in women of different ages and body mass index. Menopause, 2015. 22(6): p. 667–73. [DOI] [PubMed] [Google Scholar]
- 55.DiStefano JK, NAFLD and NASH in Postmenopausal Women: Implications for Diagnosis and Treatment. Endocrinology, 2020. 161(10). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cotter TG and Rinella M, Nonalcoholic Fatty Liver Disease 2020: The State of the Disease. Gastroenterology, 2020. 158(7): p. 1851–1864. [DOI] [PubMed] [Google Scholar]
- 57.Noureddin M, et al. , NASH Leading Cause of Liver Transplant in Women: Updated Analysis of Indications For Liver Transplant and Ethnic and Gender Variances. Am J Gastroenterol, 2018. 113(11): p. 1649–1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Janssen I, et al. , Testosterone and visceral fat in midlife women: the Study of Women’s Health Across the Nation (SWAN) fat patterning study. Obesity (Silver Spring), 2010. 18(3): p. 604–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhao D, et al. , Abdominal obesity, chronic inflammation and the risk of non-alcoholic fatty liver disease. Ann Hepatol, 2022: p. 100726. [DOI] [PubMed] [Google Scholar]
- 60.Gregg EW, et al. , Trends in lifetime risk and years of life lost due to diabetes in the USA, 1985–2011: a modelling study. Lancet Diabetes Endocrinol, 2014. 2(11): p. 867–74. [DOI] [PubMed] [Google Scholar]
- 61.Fang L, et al. , Prevalence of diabetes in the USA from the perspective of demographic characteristics, physical indicators and living habits based on NHANES 2009–2018. Front Endocrinol (Lausanne), 2023. 14: p. 1088882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Karvonen-Gutierrez CA, et al. , Low grip strength predicts incident diabetes among mid-life women: the Michigan Study of Women’s Health Across the Nation. Age Ageing, 2018. 47(5): p. 685–691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang M, et al. , Gender heterogeneity in dyslipidemia prevalence, trends with age and associated factors in middle age rural Chinese. Lipids in Health and Disease, 2020. 19(1): p. 135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.NHANES, QuickStats: Prevalence of High Total Cholesterol Among Adults Aged ≥20 Years, by Age Group and Sex — National Health and Nutrition Examination Survey, 2015–2018., 69:690., Editor. 2020: MMWR Morb Mortal Wkly Rep 69:690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.de Kat AC, et al. , Unraveling the associations of age and menopause with cardiovascular risk factors in a large population-based study. BMC Med, 2017. 15(1): p. 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Badon SE, et al. , Dual trajectories of physical activity and blood lipids in midlife women: The Study of Women’s Health Across the Nation. Maturitas, 2021. 146: p. 49–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Thurston RC, et al. , Menopause versus chronologic aging: their roles in women’s health. Menopause, 2018. 25(8): p. 849–854. [DOI] [PubMed] [Google Scholar]
- 68.Kannel WB, et al. , Menopause and Risk of Cardiovascular Disease. Annals of Internal Medicine, 1976. 85(4): p. 447–452. [DOI] [PubMed] [Google Scholar]
- 69.Dam V, et al. , Association of menopausal characteristics and risk of coronary heart disease: a pan-European case–cohort analysis. International Journal of Epidemiology, 2019. 48(4): p. 1275–1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhu D, et al. , Age at natural menopause and risk of incident cardiovascular disease: a pooled analysis of individual patient data. The Lancet Public Health, 2019. 4(11): p. e553–e564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sun Y, et al. , Association of Normal-Weight Central Obesity With All-Cause and Cause-Specific Mortality Among Postmenopausal Women. JAMA Network Open, 2019. 2(7): p. e197337–e197337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hamer M, et al. , Normal-Weight Central Obesity and Risk for Mortality. Ann Intern Med, 2017. 166(12): p. 917–918. [DOI] [PubMed] [Google Scholar]
- 73.Khan ZA, et al. , Serial Studies in Subclinical Atherosclerosis During Menopausal Transition (from the Study of Women’s Health Across the Nation). Am J Cardiol, 2018. 122(7): p. 1161–1168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.El Khoudary SR, et al. , Postmenopausal Women With Greater Paracardial Fat Have More Coronary Artery Calcification Than Premenopausal Women: The Study of Women’s Health Across the Nation (SWAN) Cardiovascular Fat Ancillary Study. J Am Heart Assoc, 2017. 6(2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Arnold M, et al. , Global burden of cancer attributable to high body-mass index in 2012: a population-based study. Lancet Oncol, 2015. 16(1): p. 36–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chadid S, et al. , Midlife weight gain is a risk factor for obesity-related cancer. British Journal of Cancer, 2018. 118(12): p. 1665–1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ahn J, et al. , Adiposity, Adult Weight Change, and Postmenopausal Breast Cancer Risk. Archives of Internal Medicine, 2007. 167(19): p. 2091–2102. [DOI] [PubMed] [Google Scholar]
- 78.Keum N, et al. , Adult Weight Gain and Adiposity-Related Cancers: A Dose-Response Meta-Analysis of Prospective Observational Studies. JNCI: Journal of the National Cancer Institute, 2015. 107(2). [DOI] [PubMed] [Google Scholar]
- 79.Schlesinger S, et al. , Body weight gain and risk of colorectal cancer: a systematic review and meta-analysis of observational studies. Obes Rev, 2015. 16(7): p. 607–19. [DOI] [PubMed] [Google Scholar]
- 80.Kothari C, Diorio C, and Durocher F, The Importance of Breast Adipose Tissue in Breast Cancer. Int J Mol Sci, 2020. 21(16). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Munsell MF, et al. , Body mass index and breast cancer risk according to postmenopausal estrogen-progestin use and hormone receptor status. Epidemiol Rev, 2014. 36(1): p. 114–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.de Araujo Dantas AB, et al. , Worldwide prevalence and associated risk factors of obstructive sleep apnea: a meta-analysis and meta-regression. Sleep Breath, 2023. [DOI] [PubMed] [Google Scholar]
- 83.Senaratna CV, et al. , Prevalence of obstructive sleep apnea in the general population: A systematic review. Sleep Medicine Reviews, 2017. 34: p. 70–81. [DOI] [PubMed] [Google Scholar]
- 84.Franklin KA, et al. , Sleep apnoea is a common occurrence in females. Eur Respir J, 2013. 41(3): p. 610–5. [DOI] [PubMed] [Google Scholar]
- 85.Lawrence RC, et al. , Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum, 2008. 58(1): p. 26–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Lange-Maia BS, et al. , Factors Influencing Longitudinal Stair Climb Performance from Midlife to Early Late Life: The Study of Women’s Health Across the Nation Chicago and Michigan Sites. J Nutr Health Aging, 2019. 23(9): p. 821–828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Szoeke CEI, et al. , Factors affecting the prevalence of osteoarthritis in healthy middle-aged women: data from the longitudinal Melbourne Women’s Midlife Health Project. Bone, 2006. 39(5): p. 1149–1155. [DOI] [PubMed] [Google Scholar]
- 88.Lange-Maia BS, et al. , Contribution of common chronic conditions to midlife physical function decline: The Study of Women’s Health Across the Nation. Womens Midlife Health, 2020. 6: p. 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Felson DT, The epidemiology of knee osteoarthritis: results from the Framingham Osteoarthritis Study. Semin Arthritis Rheum, 1990. 20(3 Suppl 1): p. 42–50. [DOI] [PubMed] [Google Scholar]
- 90.Reyes C, et al. , Association Between Overweight and Obesity and Risk of Clinically Diagnosed Knee, Hip, and Hand Osteoarthritis: A Population-Based Cohort Study. Arthritis Rheumatol, 2016. 68(8): p. 1869–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Issa RI and Griffin TM, Pathobiology of obesity and osteoarthritis: integrating biomechanics and inflammation. Pathobiol Aging Age Relat Dis, 2012. 2(2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Dahl A, et al. , Being overweight in midlife is associated with lower cognitive ability and steeper cognitive decline in late life. J Gerontol A Biol Sci Med Sci, 2010. 65(1): p. 57–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Dahl AK, et al. , Body mass index across midlife and cognitive change in late life. Int J Obes (Lond), 2013. 37(2): p. 296–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Pal L, et al. , Cardiometabolic measures and cognition in early menopause - Analysis of baseline data from a randomized controlled trial. Maturitas, 2022. 162: p. 58–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Simon GE, et al. , Association between obesity and depression in middle-aged women. Gen Hosp Psychiatry, 2008. 30(1): p. 32–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Darling CA, Coccia C, and Senatore N, Women in midlife: stress, health and life satisfaction. Stress Health, 2012. 28(1): p. 31–40. [DOI] [PubMed] [Google Scholar]
- 97.von Hippel C, et al. , Sexual Function among Women in Midlife: Findings from the Nurses’ Health Study II. Womens Health Issues, 2019. 29(4): p. 291–298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Steffen KJ, et al. , Sexual functioning of men and women with severe obesity before bariatric surgery. Surg Obes Relat Dis, 2017. 13(2): p. 334–343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Erbil N, The Relationships Between Sexual Function, Body Image, and Body Mass Index Among Women. Sexuality and Disability, 2013. 31(1): p. 63–70. [Google Scholar]
- 100.Nackers LM, et al. , Associations between body mass index and sexual functioning in midlife women: the Study of Women’s Health Across the Nation. Menopause, 2015. 22(11): p. 1175–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wing RR, et al. , Effect of intensive lifestyle intervention on sexual dysfunction in women with type 2 diabetes: results from an ancillary Look AHEAD study. Diabetes Care, 2013. 36(10): p. 2937–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Halseth A, et al. , Quality of life, binge eating and sexual function in participants treated for obesity with sustained release naltrexone/bupropion. Obes Sci Pract, 2018. 4(2): p. 141–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sarwer DB, et al. , 4-Year Changes in Sex Hormones, Sexual Functioning, and Psychosocial Status in Women Who Underwent Bariatric Surgery. Obes Surg, 2018. 28(4): p. 892–899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Gold EB, et al. , Longitudinal analysis of changes in weight and waist circumference in relation to incident vasomotor symptoms: the Study of Women’s Health Across the Nation (SWAN). Menopause, 2017. 24(1): p. 9–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Thurston RC, et al. , Behavioral weight loss for the management of menopausal hot flashes: a pilot study. Menopause, 2015. 22(1): p. 59–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Alexander C, et al. , Serum leptin levels, hormone levels, and hot flashes in midlife women. Fertility and Sterility, 2010. 94(3): p. 1037–1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Krychman M, et al. , The Women’s EMPOWER Survey: Women’s Knowledge and Awareness of Treatment Options for Vulvar and Vaginal Atrophy Remains Inadequate. J Sex Med, 2017. 14(3): p. 425–433. [DOI] [PubMed] [Google Scholar]
- 108.Pastore LM, et al. , Self-reported urogenital symptoms in postmenopausal women: Women’s Health Initiative. Maturitas, 2004. 49(4): p. 292–303. [DOI] [PubMed] [Google Scholar]
- 109.Curry SJ, et al. , Behavioral Weight Loss Interventions to Prevent Obesity-Related Morbidity and Mortality in Adults: US Preventive Services Task Force Recommendation Statement. Jama, 2018. 320(11): p. 1163–1171. [DOI] [PubMed] [Google Scholar]
- 110•.Cantor AG, et al. , Preventing Obesity in Midlife Women: A Systematic Review for the Women’s Preventive Services Initiative. Ann Intern Med, 2022. 175(9): p. 1275–1284. [DOI] [PubMed] [Google Scholar]; Counseling to prevent on results in less weight gain with relatively no harms.
- 111.Chelmow D, et al. , Preventing Obesity in Midlife Women: A Recommendation From the Women’s Preventive Services Initiative. Ann Intern Med, 2022. 175(9): p. 1305–1309. [DOI] [PubMed] [Google Scholar]
- 112.Ranjan P, et al. , Evidence and consensus-based clinical practice guidelines for management of overweight and obesity in midlife women: An AIIMS-DST initiative. J Family Med Prim Care, 2022. 11(12): p. 7549–7601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Jull J, et al. , Lifestyle interventions targeting body weight changes during the menopause transition: a systematic review. J Obes, 2014. 2014: p. 824310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Simkin-Silverman LR, et al. , Lifestyle intervention can prevent weight gain during menopause: results from a 5-year randomized clinical trial. Ann Behav Med, 2003. 26(3): p. 212–20. [DOI] [PubMed] [Google Scholar]
- 115.Howard BV, et al. , Low-fat dietary pattern and weight change over 7 years: the Women’s Health Initiative Dietary Modification Trial. Jama, 2006. 295(1): p. 39–49. [DOI] [PubMed] [Google Scholar]
- 116.Lombard C, et al. , Preventing Weight Gain in Women in Rural Communities: A Cluster Randomised Controlled Trial. PLoS Med, 2016. 13(1): p. e1001941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Williams LT, et al. , Can a relatively low-intensity intervention by health professionals prevent weight gain in mid-age women? 12-Month outcomes of the 40-Something randomised controlled trial. Nutr Diabetes, 2014. 4(5): p. e116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lawton BA, et al. , Exercise on prescription for women aged 40–74 recruited through primary care: two year randomised controlled trial. Bmj, 2008. 337: p. a2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bea JW, et al. , Resistance training predicts 6-yr body composition change in postmenopausal women. Med Sci Sports Exerc, 2010. 42(7): p. 1286–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Khan SJ, et al. , Vasomotor Symptoms During Menopause: A Practical Guide on Current Treatments and Future Perspectives. Int J Womens Health, 2023. 15: p. 273–287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Ayers B, et al. , Effectiveness of group and self-help cognitive behavior therapy in reducing problematic menopausal hot flushes and night sweats (MENOS 2): a randomized controlled trial. Menopause, 2012. 19(7): p. 749–59. [DOI] [PubMed] [Google Scholar]
- 122.Elkins GR, et al. , Clinical hypnosis in the treatment of postmenopausal hot flashes: a randomized controlled trial. Menopause, 2013. 20(3): p. 291–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.The 2023 nonhormone therapy position statement of The North American Menopause Society. Menopause, 2023. 30(6): p. 573–590. [DOI] [PubMed] [Google Scholar]
- 124.Wharton S, et al. , Medications that cause weight gain and alternatives in Canada: a narrative review. Diabetes Metab Syndr Obes, 2018. 11: p. 427–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Chen Z, et al. , Postmenopausal hormone therapy and body composition--a substudy of the estrogen plus progestin trial of the Women’s Health Initiative. Am J Clin Nutr, 2005. 82(3): p. 651–6. [DOI] [PubMed] [Google Scholar]
- 126.Papadakis GE, et al. , Menopausal Hormone Therapy Is Associated With Reduced Total and Visceral Adiposity: The OsteoLaus Cohort. The Journal of Clinical Endocrinology & Metabolism, 2018. 103(5): p. 1948–1957. [DOI] [PubMed] [Google Scholar]
- 127.Javed AA, et al. , Association Between Hormone Therapy and Muscle Mass in Postmenopausal Women: A Systematic Review and Meta-analysis. JAMA Network Open, 2019. 2(8): p. e1910154–e1910154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Weidlinger S, et al. , Impact of estrogens on resting energy expenditure: A systematic review. Obes Rev, 2023. 24(10): p. e13605. [DOI] [PubMed] [Google Scholar]
- 129.Salpeter SR, et al. , Meta-analysis: effect of hormone-replacement therapy on components of the metabolic syndrome in postmenopausal women. Diabetes Obes Metab, 2006. 8(5): p. 538–54. [DOI] [PubMed] [Google Scholar]
- 130.Stachenfeld NS, Hormonal changes during menopause and the impact on fluid regulation. Reprod Sci, 2014. 21(5): p. 555–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Arciero PJ, et al. , Increased protein intake and meal frequency reduces abdominal fat during energy balance and energy deficit. Obesity, 2013. 21(7): p. 1357–1366. [DOI] [PubMed] [Google Scholar]
- 132.Bellicha A, et al. , Effect of exercise training on weight loss, body composition changes, and weight maintenance in adults with overweight or obesity: An overview of 12 systematic reviews and 149 studies. Obes Rev, 2021. 22 Suppl 4(Suppl 4): p. e13256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Yeh ML, et al. , Exercises improve body composition, cardiovascular risk factors and bone mineral density for menopausal women: A systematic review and meta-analysis of randomized controlled trials. Appl Nurs Res, 2018. 40: p. 90–98. [DOI] [PubMed] [Google Scholar]
- 134.Pimenta F, et al. , Brief cognitive-behavioral therapy for weight loss in midlife women: a controlled study with follow-up. Int J Womens Health, 2012. 4: p. 559–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Bray GA and Ryan DH, Evidence-based weight loss interventions: Individualized treatment options to maximize patient outcomes. Diabetes Obes Metab, 2021. 23 Suppl 1: p. 50–62. [DOI] [PubMed] [Google Scholar]


