Abstract
Spleen necrosis virus (SNV) and Reticuloendotheliosis virus strain A (REV-A) belong to the family of reticuloendotheliosis viruses and are 90% sequence related. SNV-derived retroviral vectors produced by the REV-A-based D17.2G packaging cell line were shown to infect human cells (H.-M. Koo, A. M. C. Brown, Y. Ron, and J. P. Dougherty, J. Virol. 65:4769–4776, 1991), while similar vectors produced by another SNV-based packaging cell line, DSH134G, are not infectious in human cells (reviewed by R. Dornburg, Gene Ther. 2:301–310, 1995). Here we describe a careful reevaluation of the infectivity of vectors produced from the most commonly used REV-A- or SNV-based packaging cells obtained from various sources with, among them, one batch of D17.2G packaging cells obtained from the American Type Culture Collection. None of these packaging cells produced vectors able to infect human cells. Thus, contrary to previously published data, we conclude that REV-based vectors are not infectious in human cells.
Spleen necrosis virus (SNV) and Reticuloendotheliosis virus strain A (REV-A) belong to the family of reticuloendotheliosis viruses (REVs), a group of closely related amphotropic retroviruses all of which were isolated from birds (3, 29, 35, 37, 40). REVs include chicken syncytial virus, duck infectious anemia virus, SNV, REV-A, and its acutely transforming variant REV-T, which contains the rel oncogene (3, 42).
SNV and REV-A share more than 90% sequence homology, their cis- and trans-acting sequences appear to be exchangeable without impairment of virus replication (10), and they have the same receptor (9, 16, 27). Although originally isolated from avian species, REVs are more closely related to mammalian oncoretroviruses than to other avian retroviruses by sequence homology (21, 22, 38, 39, 43) and serological cross-reactivity (2, 4). Superinfection experiments have shown that SNV and REV-A belong to the same interference subgroup as the simian type D viruses (23, 27).
Using the REV-A-based packaging cell line D17.2G (12), Koo et al. (26) showed that REV-A-based vectors could infect human cells as efficiently as amphotropic murine leukemia virus (ampho-MLV). Contrary to this finding, when the SNV-based packaging cell line DSH134G (30) was used, no infection of human cells was observed (6). Because D17.2G and DSH134G produce REV-A and SNV envelopes, respectively, it was not clear whether the difference in human cell infectivity resulted from sequence differences between the two envelopes or from a special feature imparted to the viral particles produced from D17.2G cells.
To reevaluate the tropism of REV-based vectors, we have compared the infectivity of vectors produced from the most commonly used SNV- or REV-A-based packaging cell lines in human cells, i.e., the C3A2 (40) and D17.2G (12) packaging cell lines, which express the REV-A envelope, and DSN (13) and DSH134G (30) helper cells, which express the SNV envelope. We demonstrate that none of these packaging cell-produced SNV vectors was able to infect human cells. Thus, the ability of D17.2G-produced vectors to infect human cells is probably a special feature of some batches. The discrepancies between our results and those from other labs are precisely discussed.
Infection of human cells with nonreplicative SNV-lacZ vectors produced by various SNV- or REV-A-based packaging cells.
The tropism of REV-based vectors was determined by using an SNV-based gene transfer vector which transduced the bacterial β-galactosidase gene (lacZ) (in the vector pCXL; 32) or very similar SNV-based vectors carrying either a fusion gene between the phleomycin resistance gene and the lacZ gene (SNV-Sh ble::lacZ) (R. Gautier and T. Jaffredo, unpublished data) or a fusion gene consisting of the drosophila alcohol dehydrogenase gene and the phleomycin resistance gene (17). The vectors were transfected into the four most commonly used SNV- or REV-A-based helper cells. These were the SNV-based helper cell lines DSN (obtained from H. Temin's laboratory, University of Wisconsin; reference 13) and DSH134G (30) and the REV-A-based helper cell lines C3A2 (obtained from H. Temin's laboratory; reference 41) and D17.2G (CRL 8468; obtained from the American Type Culture Collection [ATCC]; reference 12). As a control, we also used a helper cell line which expressed the SNV Gag-Pol proteins and the envelope of ampho-MLV (termed DSH-ampho-MLV-env). Vectors were harvested from stably transfected helper cell clones, and infectivity was tested in various human cell lines, e.g., HeLa (18) or HOS (31), or the canine cell line D17 (36). The best producer clones were selected and tested for absence of replication-competent (RC) virus by using QT6 cells (33), an immortalized quail cell line, as naive recipient cells. Virus was harvested from nearly confluent monolayers of packaging cells 16 to 18 h after the medium was changed. The harvested medium was centrifuged at 3,500 × g for 10 min to remove cells and cell debris, and the virus was used for infection of various cells.
No infection of HeLa or HOS cells was detected with vectors produced from C3A2, D17.2G, DSN, or DSH134G helper cells (Table 1). In contrast, DSH-ampho-MLV-env-produced vectors give titers ranging from 5 × 104 to 1 × 105 LacZ-forming units (LFU)/ml, respectively (Table 1). D17 cells permissive for SNV and ampho-MLV infection served as positive controls. In this cell type, vector virus titers ranged from 105 to 106 LFU/ml (Table 1), except for DSN cells, which are known to give lower vector virus titers than C3A2 or D17.2G cells (13). LacZ staining was never detected in mock-infected D17, HeLa, or HOS cells.
TABLE 1.
Titers of SNV vectorsa packaged by different REV-A- or SNV-based helper cell lines on D17, HeLa, or HOS cells
| Cell lineb | Virus titer (LFU/ml of virus stock)
|
||||
|---|---|---|---|---|---|
| C3A2 | D17.2G (ATCC CRL 8468) | DSN | DSH134G | DSH-ampho- MLV-env | |
| D17 | 106 | 106 | 103 | 106 | 105 |
| HeLa | <1 | <1 | <1 | <1 | 105 |
| HOS | <1 | NDc | <1 | <1 | 5 · 104 |
The vectors were SNV-CXL, SNV-Sh ble::lacZ, and SNV-ADH::Sh ble. No difference in either tropism or expression was detected.
Cell lines are described in the text.
ND, not done.
C3A2-produced vectors express the REV-A envelope from a plasmid identical to that present in D17.2G cells. Neither of these cell lines produced a vector displaying human cell tropism. Thus, the differences in human cell infectivity between our results and those of Koo et al. (26) is likely due to a special feature imparted by some batches of D17.2G packaging cells and certainly did not result from differences between the REV-A and SNV envelope proteins. In addition, when using an RC strain of SNV, those investigators (26) did not find productive infection of human cells and concluded that a posttranscriptional block in SNV replication occurred in these cells. However, they did not investigate whether the virus was integrated. Since the SNV promoter is very strong in human cells, it is very unlikely that the lack of sufficient gene expression accounted for the lack of productive infection.
Comparison of DSH134G- and DSH-ampho-MLV-env-produced SNV-lacZ vector tropism for various human cells.
To further evaluate the tropism of SNV-based vectors for human cells, we compared the infectivity of CXL vectors produced from the solely SNV-based DSH helper cell line with that of vectors produced from the DSH-ampho-MLV-env helper cell line (19), which contains SNV Gag-Pol and the ampho-MLV envelope. Vector particles produced from both packaging lines encapsidated the SNV-based lacZ vector pCXL. Infectivity was determined in human cell lines from various origins (Table 2), as well as human embryo skin primary fibroblasts from a week 21 of amenorrhea female fetus used at the third passage in culture. Human embryo tissue was obtained from voluntary or therapeutic abortions performed in compliance with French legislation after informed consent was obtained from the parents.
TABLE 2.
Comparison of infection of human cells from various origins by DSH134G- and DSH-ampho-Env-produced SNV-lacZa
| Packaging cell line producing SNV-lacZ | Virus titer (LFU/ml of virus stock)
|
|||||||
|---|---|---|---|---|---|---|---|---|
| HeLa | SK BRK-3 | COLO 320DM | MDA-MB453 | KG1a | Daudi | HPFc | D17 | |
| DSH134G | 1b | <10 | <10 | <10 | <1 | <1 | 1 | 106 |
| DSH-ampho-Env | NDd | 6 · 104 | 4 · 104 | 4 · 103 | 1 · 102 | 4 · 102 | ND | 1 · 105 |
The vector was either SNV-CXL or SNV-Sh ble::lacZ. No difference in expression or viral titer was detected. Cell lines: HeLa, epithelium-like cervical carcinoma (18); SK BRK-3, human breast cancer (1); COLO 320DM, colon carcinoma (36); MDA-MB453, human breast cancer (ATCC HTB131); KG1a, hematopoietic B lymphoma (24); Daudi, hematopoietic B lymphoma (25); D17, dog osteosarcoma.
Virus titers were obtained from at least two independent experiments with no significant variation.
Human embryo skin primary fibroblasts.
ND, not done.
Coinciding with earlier observations (5–7, 19), no or only background (up to 10 LFU) infectivity in the human cells was observed with vectors produced by DSH134G packaging cells. Dog D17 cells, which served as a positive control, were infected with titers of 106 LFU (Table 2). However, CXL vectors pseudotyped with ampho-MLV Env infected all of the human nonhematopoietic cell lines investigated with titers ranging from 4 · 103 to 105 LFU/ml (Table 2). The hematopoietic cell lines KG1a (24) and Daudi (25) were poorly infected. These cells are known to express low levels of ampho-MLV receptors (34). Thus, low levels of infectivity of these hematopoietic cells correlate with low-level receptor expression, coinciding with other reports (34). This conclusion is further supported by recent findings that human hematopoietic cells can be efficiently infected with SNV when a targeting envelope capable of binding to a cell surface protein is expressed on the viral surface (19). For example, KG1a cells which express the CD34 receptor were infected with SNV vectors displaying anti-CD34 single-chain antibodies with titers above 105 (19). It is worthy of note that D17.2G-produced vectors (obtained from reference 26) infected HeLa and HPF cells with titers similar to those obtained with ampho-MLV Env-pseudotyped SNV vectors, e.g., 104 and 5 · 105 LFU/ml, while the same vector produced by D17.2G cells originating from the ATCC did not (data not shown).
Amphotropic envelope detection on SNV-based helper cells: FACS and immunocytochemical analyses.
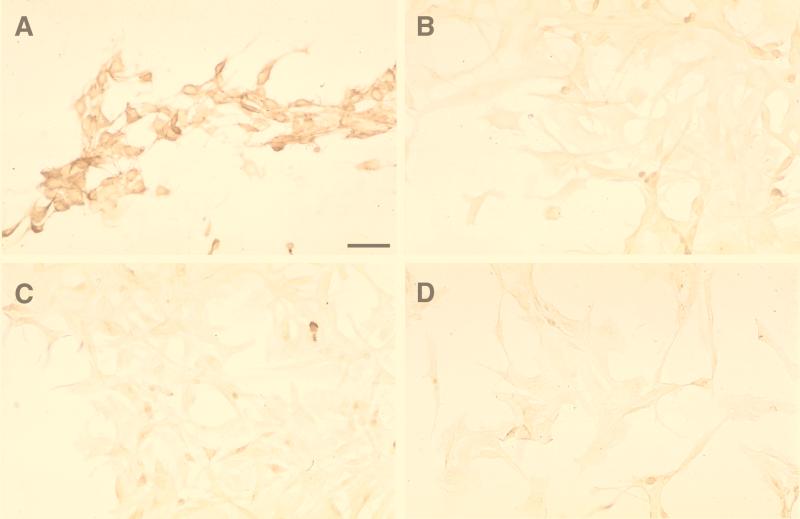
Since we wanted to be sure that the packaging cells we used were free of amphotropic contamination, we tested the cells for the presence of ampho-MLV Env at the cell surface by immunocytochemistry analysis. psi CRIP, an ampho-MLV-based packaging cell line (8); D17.2G; DSH134G; and D17 cells were plated on eight-well Lab Tek chamber slides (Nunc) with appropriate culture media. The next day, they were fixed with 4% paraformaldehyde in phosphate-buffered saline and incubated overnight with a 1/500 dilution of a goat polyclonal antibody directed against ampho-MLV (Quality Biotech) revealed with a sheep anti-goat peroxidase-coupled secondary antibody (Biosys). Tyramide Signal Amplification (NEN Life Science) was used to increase the signal-to-noise ratio in accordance with the manufacturer's recommendations. No staining was found on D17.2G, DSH134G, or D17 cells. As expected, the psi CRIP cells were strongly stained (Fig. 1).
FIG. 1.
Immunohistochemical detection of an MLV-related amphotropic envelope on different cell types. Tyramide amplification and diaminobenzidine staining were used. Panels: A, psi CRIP cells; B, D17.2G cells (ATCC); C, DSH134G cells; D, D17 cells. Only psi CRIP cells were strongly immunostained. Bar, 60 μm.
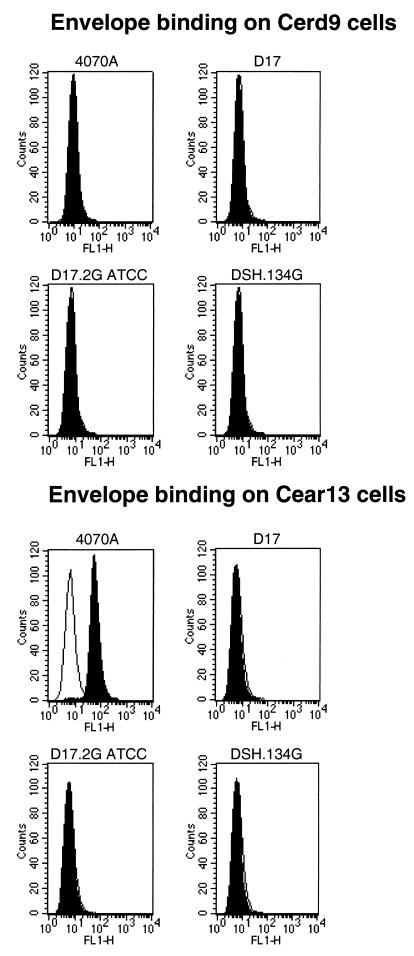
To further corroborate this finding, fluorescence-activated cell sorter (FACS) analysis by the method of Kadan et al. (20) was performed. This assay is based on the use of two different cell lines: a naive one which expresses neither SNV nor ampho-MLV receptors (28) and another expressing the ampho-MLV receptor PIT-2. When added to these cells, ampho-MLV envelope proteins or virions bind to the receptor. Material bound to the cells is recognized by incubation with monoclonal antibody 83A25 (15) against the MLV envelope for 45 min at 4°C. Cells are then incubated with anti-rat immunoglobulin fluorescein isothiocyanate-conjugated antibodies (Dako) and subjected to FACS analysis. Incubation of CERD9 (negative control) and CEAR13 (expressing PIT-2) cells with the supernatant from D17, D17.2G, or DSH134G cells or that of the viral MLV envelope 4070A was followed by detection of ampho-MLV Env. This test enables the detection of ampho-MLV Env present in the culture medium. No FACS signal could be detected at any time with CERD9. CEAR13 gave a very strong signal with the 4070A envelope (positive control), but no signal was detected with D17, D17.2G, or DSH134G-conditioned medium (Fig. 2). Surprisingly, D17.2G cells, from reference 26, displayed a strong signal in both tests which should be correlated with the ability of these cells to produce virus able to infect human cells (data not shown).
FIG. 2.
Profiles of the ampho-MLV envelope in the supernatant of cells. Indirect immunofluorescence flow cytometry patterns on CEAR13 cells expressing the amphotropic receptor PIT-2 and CERD9 cells which do not express it are shown. Supernatants are those of 4070A (ampho-MLV strain 4070A envelope; positive control) and D17.2G (ATCC), DSH134G, and D17 cells. The white profiles indicate cells stained by propidium iodide alone. The black profiles shows cells incubated with monoclonal antibody 83A25 against the ampho-MLV envelope (20). Only 4070A gave a strong positive signal. Two independent analysis were performed which yielded similar results.
Taken together, these results indicate that some batches of D17.2G cells produce an ampho-MLV Env glycoprotein which confers the characteristics of an amphotropic packaging cell line on D17.2G, thus enabling infection of human cells. This finding is further supported by the fact that D17.2G and psi CRIP cells display strong inhibition of infection by, respectively, psi CRIP- and D17.2G-produced virus (superinfection interference; data not shown). Since (i) SNV and REV-A and (ii) ampho-MLV belong to different interference subgroups (37), this indicates the presence of an envelope different from that of REVs in some D17.2G helper cell stocks. Therefore, we conclude that the ability of some batches of D17.2G-produced vectors to efficiently infect human cells is due to contamination with ampho-MLV Env.
The nature of the envelope present on some batches D17.2G cells has remained elusive. It might be a fragment of ampho-MLV Env inserted into the SNV wild-type envelope, a complete envelope together with that of SNV, or a fully RC ampho-MLV. Furthermore, we do not know whether the D17.2G samples distributed in various laboratories are equally contaminated. In this respect, it is important to note that infection experiments performed by different groups with the same cell types have yielded different results. For instance, Mikawa et al. (32) found that infection of NIH 3T3 cells by D17.2G-CXL (bearing a lacZ gene) was negligible, as previously reported (14). Koo et al. (26) reported a titer of 6 · 103 transducing vectors per ml of viral supernatant. Both groups, however, found similar titers on D17 dog cells and on HeLa cells (23, 26).
The SNV envelope displays 42 to 49% amino acid sequence identity with a prototype simian D-type virus envelope (23, 27). Interference experiments using molecular clones of RC strains of SNV and REV-A have shown that SNV and simian D-type viruses belong to a single receptor interference subgroup. Thus, they appear to use the same receptor (23, 27). However, it has been shown that the other members of this interference subgroup can infect human cells (37). Considering these findings, how can the lack of human cell infection by SNV be explained? REVs may use the same cell surface protein as other D-type viruses for envelope docking but may use other protein domains for binding. This would explain why simian D-type virus-infected cells are resistant to SNV infection. Moreover, Dornburg and coworkers have found that the infectivity of SNV particles displaying single-chain antibodies was greatly enhanced when the SNV wild-type envelope was also present (5, 7). It was hypothesized that a cellular receptor for the wild-type SNV envelope that is homologous to that expressed on dog D17 cells is present on human cells but that the human receptor is mutated, preventing high-affinity binding and virus entry. The single-chain antibodies displayed on the viral surface may anchor the vector to the cell surface, hence mediating interaction with the envelope and the wild-type receptor, enabling membrane fusion (11). Since all members of the simian D-type interference subgroup except SNV infect human cells, this hypothesis appears the most likely. Cloning of the receptor should help to elucidate this problem.
In summary, the present study clearly shows that the REVs REV-A and SNV do not infect human cells due to the inability to bind to a cell surface receptor. However, vectors derived from these viruses can infect human cells when they are pseudotyped with envelopes that mediate receptor binding and membrane fusion, e.g., that of ampho-MLV. Thus, SNV or REV-A is suitable for the development of a powerful, hazard-free vector for gene transduction.
Acknowledgments
We are indebted to F. Dieterlen for her constant support and critical reading of the manuscript. We are grateful to F.-L. Cosset for his invaluable help in FACS analysis. We thank M. C. Labastie for providing us with human embryo skin (obtained according to the rules defined by the French Comité d'Ethique) and A. Burns for reading the manuscript. We also thank C. Batejat for Tyramide Signal Amplification staining, M.-F. Meunier for help in manuscript preparation, and F. Viala for excellent photographic assistance.
This work was supported by the Centre National de la Recherche Scientifique, by AFM and ARC grants 587132 and 9787 to T.J. and R.G., and by a grant from the National Institutes of Health (1RO1AI41899-01) to R.D. and A.J.
REFERENCES
- 1.Alper O, Yamaguchi K, Hitomi J, Honda S, Matsushima T, Abe K. The presence of c-erbB-2 gene product-related protein in culture medium conditioned by breast cancer cell line SK-BR-3. Cell Growth Differ. 1990;1:591–599. [PubMed] [Google Scholar]
- 2.Barbacid M, Hunter E, Aaronson S A. Avian reticuloendotheliosis viruses: evolutionary linkage with mammalian type C retroviruses. J Virol. 1979;30:508–514. doi: 10.1128/jvi.30.2.508-514.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bose H R. The rel family: models for transcriptional regulation and oncogenic transformation. Biochim Biophys Acta. 1992;1114:1–7. doi: 10.1016/0304-419x(92)90002-g. [DOI] [PubMed] [Google Scholar]
- 4.Charman H P, Gilden R V, Oroszlan S. Reticuloendotheliosis virus: detection of immunological relationship to mammalian type C retroviruses. J Virol. 1979;29:1221–1225. doi: 10.1128/jvi.29.3.1221-1225.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chu T-H T, Dornburg R. Retroviral vector particles displaying the antigen-binding site of an antibody enable cell-type-specific gene transfer. J Virol. 1995;69:2659–2663. doi: 10.1128/jvi.69.4.2659-2663.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chu T-H T, Dornburg R. Toward highly efficient cell-type-specific gene transfer with retroviral vectors displaying single-chain antibodies. J Virol. 1997;71:720–725. doi: 10.1128/jvi.71.1.720-725.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chu T-H T, Martinez I, Sheay W C, Dornburg R. Cell targeting with retroviral vector particles containing antibody-envelope fusion proteins. Gene Ther. 1994;1:292–299. [PubMed] [Google Scholar]
- 8.Danos O, Mulligan R C. Safe and efficient generation of recombinant retroviruses with amphotropic and ecotropic host ranges. Proc Natl Acad Sci USA. 1988;85:6460–6464. doi: 10.1073/pnas.85.17.6460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Delwart E L, Panganiban A T. Role of reticuloendotheliosis virus envelope glycoprotein in superinfection interference. J Virol. 1989;63:272–280. doi: 10.1128/jvi.63.1.273-280.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dornburg R. Reticuloendotheliosis viruses and derived vectors. Gene Ther. 1995;2:301–310. [PubMed] [Google Scholar]
- 11.Dornburg R. From the natural evolution to the genetic manipulation of the host-range of retroviruses. Biol Chem. 1997;378:457–468. [PubMed] [Google Scholar]
- 12.Dougherty J P, Temin H M. Determination of the rate of base-pair substitution and insertion mutations in retrovirus replication. J Virol. 1988;62:2817–2822. doi: 10.1128/jvi.62.8.2817-2822.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dougherty J P, Wisniewski R, Yang S, Rhode B W, Temin H M. New retrovirus helper cells with almost no nucleotide sequence homology to retrovirus vectors. J Virol. 1989;63:3209–3212. doi: 10.1128/jvi.63.7.3209-3212.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Embretson J E, Temin H M. Transcription from a spleen necrosis virus 5′ long terminal repeat is suppressed in mouse cells. J Virol. 1987;61:3454–3462. doi: 10.1128/jvi.61.11.3454-3462.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Evans L H, Morrison R P, Malik F G, Portis J, Britt W. A neutralizable epitope common to the envelope glycoproteins of ecotropic, polytropic, xenotropic, and amphotropic murine leukemia viruses. J Virol. 1990;64:6176–6183. doi: 10.1128/jvi.64.12.6176-6183.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Federspiel M J, Crittenden L B, Hughes S H. Expression of avian reticuloendotheliosis virus envelope confers host resistance. Virology. 1989;173:167–177. doi: 10.1016/0042-6822(89)90232-8. [DOI] [PubMed] [Google Scholar]
- 17.Gautier R, Drocourt D, Jaffredo T. Generation of small fusion genes carrying phleomycin resistance and Drosophila alcohol dehydrogenase reporter properties, their application in retroviral vectors. Exp Cell Res. 1996;224:291–301. doi: 10.1006/excr.1996.0139. [DOI] [PubMed] [Google Scholar]
- 18.Gey G O, Coffman W D, Kubicek M T. Tissue culture studies of the proliferative capacity of cervical carcinoma and normal epithelium. Cancer Res. 1952;12:264–269. [Google Scholar]
- 19.Jiang A, Chu T-H T, Nocken F, Cichutek K, Dornburg R. Cell-type-specific gene transfer into human cells with retroviral vectors that display single-chain antibodies. J Virol. 1998;72:10148–10156. doi: 10.1128/jvi.72.12.10148-10156.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kadan M J, Sturm S, Anderson W F, Eglitis M A. Detection of receptor-specific murine leukemia virus binding to cells by immunofluorescence analysis. J Virol. 1992;66:2281–2287. doi: 10.1128/jvi.66.4.2281-2287.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kang C-Y, Temin H M. Lack of sequence homology among RNAs of avian leukosis-sarcoma viruses, reticuloendotheliosis viruses, and chicken endogenous RNA-directed DNA polymerase activity. J Virol. 1973;12:1314–1324. doi: 10.1128/jvi.12.6.1314-1324.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kato S, Matsuo K, Nishimura N, Takahashi N, Takano T. The entire nucleotide sequence of baboon endogenous virus DNA: a chimeric genome structure of murine type C and simian type D retroviruses. Jpn J Genet. 1987;62:127–137. [Google Scholar]
- 23.Kewalramani V N, Panganiban A T, Emerman M. Spleen necrosis virus, an avian immunosuppressive retrovirus, shares a receptor with the type D simian retroviruses. J Virol. 1992;66:3026–3031. doi: 10.1128/jvi.66.5.3026-3031.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Klein E, Klein G, Nadkarni J J, Wigzell H, Clifford P. Surface IgM-kappa specificity on a Burkitt lymphoma cell in vivo and in derived culture lines. Cancer Res. 1968;28:1300–1310. [PubMed] [Google Scholar]
- 25.Koeffler H P. Induction of differentiation of human acute myelogenous leukemia cells: therapeutic implications. Blood. 1983;62:709–721. [PubMed] [Google Scholar]
- 26.Koo H-M, Brown A M C, Ron Y, Dougherty J P. Spleen necrosis virus, an avian retrovirus, can infect primate cells. J Virol. 1991;65:4769–4776. doi: 10.1128/jvi.65.9.4769-4776.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Koo H-M, Gu J, Varela-Echavarria A, Ron Y, Dougherty J P. Reticuloendotheliosis type C and primate type D oncoretroviruses are members of the same receptor interference group. J Virol. 1992;66:3448–3454. doi: 10.1128/jvi.66.6.3448-3454.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kozak S L, Siess D C, Kavanaugh M P, Miller A D, Kabat D. The envelope glycoprotein of an amphotropic murine retrovirus binds specifically to the cellular receptor/phosphate transporter of susceptible species. J Virol. 1995;69:3433–3440. doi: 10.1128/jvi.69.6.3433-3440.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Maldonado R L, Bose H R., Jr Group-specific antigen shared by the members of the reticuloendotheliosis virus complex. J Virol. 1976;17:983–990. doi: 10.1128/jvi.17.3.983-990.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Martinez I, Dornburg R. Improved retroviral packaging lines derived from spleen necrosis virus. Virology. 1995;208:234–241. doi: 10.1006/viro.1995.1147. [DOI] [PubMed] [Google Scholar]
- 31.McAllister R M, Gardner M B, Greene A E, Bradt C, Nichols W W, Landing B H. Cultivating in vitro of cells derived from a human osteosarcoma. Cancer. 1971;27:397–402. doi: 10.1002/1097-0142(197102)27:2<397::aid-cncr2820270224>3.0.co;2-x. [DOI] [PubMed] [Google Scholar]
- 32.Mikawa T, Fischman D A, Dougherty J P, Brown A M C. In vivo analysis of a new lacZ retrovirus vector suitable for cell lineage marking in avian and other species. Exp Cell Res. 1991;195:516–523. doi: 10.1016/0014-4827(91)90404-i. [DOI] [PubMed] [Google Scholar]
- 33.Moscovici C, Moscovici M G, Jimenez H, Lai M C C, Hayman M J, Vogt P K. Continuous tissue culture cell lines derived from chemically induced tumors of Japanese quail. Cell. 1977;11:95–103. doi: 10.1016/0092-8674(77)90320-8. [DOI] [PubMed] [Google Scholar]
- 34.Orlic D, Girard L J, Jordan C T, Anderson S M, Cline A P, Bodine D M. The level of mRNA encoding the amphotropic retrovirus receptor in mouse and human hematopoietic stem cells is low and correlates with the efficiency of retrovirus transduction. Proc Natl Acad Sci USA. 1996;93:11097–11102. doi: 10.1073/pnas.93.20.11097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Purchase H G, Witter R L. The reticuloendotheliosis viruses. Curr Top Microbiol Immunol. 1975;71:103–124. doi: 10.1007/978-3-642-66193-8_3. [DOI] [PubMed] [Google Scholar]
- 36.Quinn L A, Moore G E, Morgan R T, Woods L K. Cell lines from human colon carcinoma with unusual cell products, double minutes, and homogeneously staining regions. Cancer Res. 1979;39:4914–4924. [PubMed] [Google Scholar]
- 37.Sommerfelt M A, Weiss R A. Receptor interference groups of 20 retroviruses plating on human cells. Virology. 1990;176:58–69. doi: 10.1016/0042-6822(90)90230-o. [DOI] [PubMed] [Google Scholar]
- 38.Sonigo P, Barker C, Hunter E, Wain-Hobson S. Nucleotide sequence of Mason-Pfizer monkey virus: an immunosuppressive D-type retrovirus. Cell. 1986;45:375–385. doi: 10.1016/0092-8674(86)90323-5. [DOI] [PubMed] [Google Scholar]
- 39.Thayer R M, Power M D, Bryant M L, Gardner M B, Barr P J, Luciw P A. Sequence relationships of type D retroviruses which cause simian acquired immunodeficiency syndrome. Virology. 1987;157:317–329. doi: 10.1016/0042-6822(87)90274-1. [DOI] [PubMed] [Google Scholar]
- 40.Varmus H E, Brown P. Retroviruses. In: Berg D E, Howe M M, editors. Mobile DNA. Washington, D.C.: American Society for Microbiology; 1988. pp. 53–108. [Google Scholar]
- 41.Watanabe S, Temin H M. Construction of a helper cell line for avian reticuloendotheliosis virus cloning vectors. Mol Cell Biol. 1983;3:2241–2249. doi: 10.1128/mcb.3.12.2241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Weiss R. Experimental biology and assay of RNA tumor viruses. In: Weiss R, Teich N, Varmus H, Coffin J, editors. RNA tumor viruses. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1984. pp. 65–67. [Google Scholar]
- 43.Wilhelmsen K C, Eggleton K, Temin H M. Nucleic acid sequences of the oncogene v-rel in reticuloendotheliosis virus strain T and its cellular homolog, the proto-oncogene c-rel. J Virol. 1984;52:172–182. doi: 10.1128/jvi.52.1.172-182.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]