Abstract
We performed a meta‐analysis to compare diagnostic performances of computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET or PET/CT), for detection of metastatic lymph nodes in patients with cervical cancer. We searched MEDLINE (PubMed), EMBASE and the Cochrane Review database in December 2007. All articles were independently reviewed and selected by three evaluators. We estimated a summary receiver operating characteristic (sROC) curve. The area under the curve (AUC), Q*, and pooled weighted estimates of sensitivity and specificity for each modality by patient‐based and region‐ or node‐based data analyses and conducted pair‐wise comparisons between modalities using the two‐sample Z‐test. Forty‐one of 768 initially identified studies were included in the meta‐analysis. In a patient‐based data analysis, PET or PET/CT showed the highest pooled sensitivity (82%) and specificity (95%), while CT showed 50% and 92%; and MRI, 56% and 91%, respectively. The AUC (0.9641) and Q* (0.9106) of PET or PET/CT were significantly higher than those of MRI (AUC = 0.8270; Q* = 0.7599), both P < 0.001. In region‐ or node‐based data analysis, sensitivities of CT (52%) and PET or PET/CT (54%) were higher than that of MRI (38%), P < 0.02 and P < 0.001, respectively, while specificities of MRI (97%) and PET or PET/CT (97%) were higher than that of CT (92%), both P < 0.001. The AUC and Q* showed no significant difference among CT, MRI, and PET or PET/CT. PET or PET/CT had an overall higher diagnostic performance than did CT or MRI in detecting metastatic lymph nodes in patients with cervical cancer. (Cancer Sci 2010)
Uterine cervical carcinoma is the second most common malignancy in women worldwide, and is the only major gynecologic malignancy that is staged clinically according to International Federation of Obstetrics and Gynecology (FIGO) recommendations.( 1 ) However, clinical staging of cervical cancer is accurate in only 29% of patients. Undiagnosed lymph node metastases are a major problem( 2 , 3 , 4 , 5 ) because lymph node status is an independent prognostic factor for survival of patients with primary cervical cancer.( 6 , 7 , 8 , 9 ) Although lymph node dissection before radiotherapy results in improved survival of patients with macroscopically enlarged pelvic and para‐aortic lymph nodes, the routine pretreatment surgical staging is not recommended. Thus, an inaccurate evaluation of lymph node metastasis associated with uterine cervical carcinoma often leads to unsatisfactory treatment.( 10 , 11 , 12 , 13 , 14 )
Computed tomography (CT) and magnetic resonance imaging (MRI) have been used to assess the metastatic lymph nodes of cervical cancer patients. A meta‐analysis of studies evaluating these diagnostic methods concluded that these methods have only moderate sensitivity and specificity for detection of metastatic lymph nodes.( 15 ) On the contrary, the recent studies have reported that positron emission tomography (PET or PET/CT), employing [18F]‐fluoro‐2‐deoxy‐d‐glucose (FDG), is more sensitive than CT or MRI for detection of metastatic lymph nodes in patients with cervical cancer.( 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 , 26 , 27 , 28 , 29 , 30 , 31 , 32 , 33 )
Numerous studies have described the use of CT, MRI, and PET or PET/CT for detection of metastatic lymph nodes in patients with cervical cancer, but no studies have compared these three imaging modalities for those patients. Some previous studies have compared PET and MRI and suggested that PET is more accurate than MRI, but the statistical power of the comparisons was too low to assess the significance of this difference.( 20 , 21 , 25 , 26 , 27 , 30 , 34 ) Scheidler et al. ( 15 ) performed a meta‐analysis of studies performed between 1971 and 1997 that compared the accuracy of CT and MRI for detection of metastatic lymph nodes. However, patient heterogeneity, differences in imaging techniques, and differences in diagnostic criteria for metastatic lymph nodes made it difficult to compare these two imaging modalities directly.
Since Scheidler et al.’s meta‐analysis, there have been many technical improvements in CT and MRI, and PET has recently become widely available. The aim of our study was to compare diagnostic performances of CT, MRI, and PET or PET/CT for detection of metastatic lymph nodes in patients with cervical cancer, using a meta‐analysis.
Materials and Methods
Data sources and keywords. We searched MEDLINE (PubMed), EMBASE, and the Cochrane Library in December 2007, using common keywords relevant to cervical cancer, lymph node, and imaging. The keywords for the literature search were “cervical cancer” and “lymph node” for disease factors, and “computed tomography” or “magnetic resonance imaging” or “positron emission tomography” for imaging factors. We scanned bibliographies of relevant articles to identify additional studies. The search period was January 1981 to December 2007 for CT, January 1988 to December 2007 for MRI, and January 2001 to December 2007 for PET or PET/CT, after consideration of the time of introduction of each modality in the clinical setting.
Selection criteria. The selection criteria for the relevant studies were: (i) a minimal sample size of 20 patients; (ii) histological examination of lymph nodes by surgery or biopsy; (iii) use of clear diagnostic criteria; (iv) adequate presentation of data (true‐positive [TP], false‐positive [FP], true‐negative [TN], false‐negative [FN]); and (v) use of the English language.
Selection of relevant studies. All studies retrieved from databases were independently evaluated by the three co‐authors of this study. When there was a disagreement concerning inclusion of a study, it was resolved by discussion. When there were insufficient or missing data in a study, we contacted the authors to request the relevant information. We included only one article, when there were two or more articles with the same data.
Data extraction. From the studies finally selected, we extracted the study name (first author, year of publication), journal name, and comparison methods (patient‐by‐patient, region‐by‐region and node‐by‐node comparison). TP, FP, TN, and FN results were recorded. Because different comparison methods led to different results, even within the same study, results were recorded twice for seven MRI studies( 19 , 27 , 30 , 35 , 36 , 37 , 38 ) and six PET or PET/CT studies.( 19 , 20 , 27 , 28 , 30 , 33 ) In five studies,( 20 , 28 , 33 , 36 , 39 ) pelvic and para‐aortic lymph node metastases were subdivided and results were recorded for each area.
Statistical analyses. We conducted all analyses based on two types of comparison method; one is a patient‐based data analysis, and the other is a region‐ or node‐based data analysis. A patient‐based data analysis uses the pathologically proven positive node in the same patient who had been identified to have metastatic lymph nodes by preoperative imaging, while region‐ or node‐ based analyses use the pathologically proven positive node in the corresponding region or node which had been described as containing positive node by preoperative imaging.
Summary receiver operating characteristic (sROC) curve and the area under the curve (AUC) were calculated. The sROC curve was constructed by using an estimated false positive rate (FPR) and true positive rate (TPR) of each study based on a regression model.( 40 , 41 , 42 ) We also calculated another global measure of test efficacy, Q*; the intersection of the estimated sROC curve with the line where sensitivity equals specificity; values of Q* near 1.0 indicate that sROC curves are snugged up near the desirable north‐west corner where sensitivity and specificity are both 1.0. 42
We obtained summary sensitivity and specificity data with 95% confidence intervals using bivariate analysis for diagnostic meta‐analysis.( 43 ) In order to examine heterogeneity in sensitivity and specificity across studies, Higgins I‐square (I 2) was calculated. An I 2 value greater than 50% was considered to indicate substantial heterogeneity.
We conducted a two‐sample Z‐test in order to evaluate a significant difference in sensitivity, specificity, AUC, and Q* values between any two diagnostic modalities. A P‐value less than 0.05 was considered statistically significant. All of the statistical analyses were conducted using Meta‐DiSc version 1.4.( 44 )
Results
We initially identified 768 studies from the initial search using the keywords. After exclusion of duplicates (n = 97), we reviewed all remaining article titles and abstracts, and 597 additional studies were excluded. We reviewed the remaining 74 articles, and finally included 41 studies in our meta‐analysis (Fig. 1). The main reasons for exclusion of studies were as follows: use of a non‐English language( 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 , 56 , 57 , 58 , 59 , 60 , 61 , 62 , 63 ) (n = 19); insufficient number of patients( 17 , 18 , 65 , 69 , 70 , 71 , 72 ) (n = 7); insufficient data( 64 , 65 , 66 , 67 , 68 ) (n = 5); use of identical populations( 73 ) (n = 1); and non‐use of a histopathological gold standard for lymph node evaluation( 74 ) (n = 1).
Figure 1.
Flow diagram for identification of relevant studies.
We finally included data from 20 studies for the analysis of CT performance, 31 studies for the analysis of MRI performance, and 20 studies for analysis of PET or PET/CT performance, respectively (1, 2, 3).
Table 1.
Computed tomography studies included in the meta‐analysis (n = 20)*
| Study, year (reference) | Journal | Type of scanner | Comparison | N | Sensitivity, no. (%) | Specificity, no. (%) | Positive likelihood ratio | Negative likelihood ratio |
|---|---|---|---|---|---|---|---|---|
| Grumbine et al., 1981( 83 ) | Gynecol Oncol | Non‐helical | p | 24 | 0/6 (0) | 17/18 (94) | 0.90 | 1.01 |
| Walsh et al., 1981( 84 ) | Am J Roentgenol | Non‐helical | p | 25 | 12/15 (80) | 7/10 (70) | 2.67 | 0.29 |
| Brenner et al., 1982( 85 ) | Cancer | Non‐helical | p | 20 | 4/6 (67) | 13/14 (93) | 9.33 | 0.36 |
| Villasanta et al., 1983( 86 ) | Obstet Gynecol | Non‐helical | p | 42 | 10/13 (77) | 25/29 (86) | 5.58 | 0.27 |
| Van engelshoven et al., 1984( 87 ) | Gynecol Obstet Invest | Non‐helical | p | 20 | 3/7 (43) | 12/13 (92) | 5.57 | 0.62 |
| Bandy et al., 1985( 88 ) | Obstet Gynecol | Non‐helical | p | 44 | 9/12 (75) | 29/32 (91) | 8.00 | 0.28 |
| Vas et al., 1985†( 39 ) | J Comput Tomogr | Non‐helical | p | 30 | 10/16 (63) | 11/14 (79) | 2.92 | 0.48 |
| Vas et al., 1985‡( 39 ) | J Comput Tomogr | Non‐helical | p | 33 | 10/12 (83) | 20/21 (95) | 17.5 | 0.18 |
| Camilien et al., 1988( 89 ) | Gynecol Oncol | Non‐helical | p | 51 | 3/12 (25) | 38/39 (97) | 9.75 | 0.77 |
| Janus et al., 1989( 90 ) | Clin Imaging | Non‐helical | p | 22 | 1/3 (33) | 18/19 (95) | 6.33 | 1.85 |
| Matsukuma et al., 1989( 91 ) | Gynecol Oncol | Non‐helical | p | 70 | 5/7 (71) | 61/63 (97) | 22.50. | 0.30 |
| Kim et al., 1990( 92 ) | Radiology | Single helical | r | 30 | 6/12 (50) | 36/48 (75) | 2.00 | 0.67 |
| Heller et al., 1990( 93 ) | Gynecol Oncol | Single helical | p | 253 | 31/61 (34) | 184/192 (96) | 22.50 | 0.30 |
| Kim et al., 1993( 94 ) | J Comput Tomogr | Single helical | r | 99 | 7/29 (24) | 158/169 (93) | 3.71 | 0.81 |
| Subak et al., 1995( 95 ) | Obstet Gynecol | Single helical | p | 37 | 3/5 (60) | 29/32 (91) | 6.40 | 0.44 |
| Chu et al., 1997‡( 96 ) | Gynecol Oncol | Single helical | p | 28 | 4/10 (40) | 17/18 (94) | 6.67 | 0.64 |
| Yang et al., 2000( 97 ) | Am J Roentgenol | Single helical | r | 43 | 11/17 (62) | 57/59 (97) | 20.67 | 0.39 |
| Hertel et al., 2002†( 36 ) | Gynecol Oncol | Not presented | p | 75 | 3/20 (15) | 47/55 (85) | 1.00 | 1.00 |
| Hertel et al., 2002‡( 36 ) | Gynecol Oncol | Not presented | p | 91 | 3/16 (19) | 66/75 (88) | 1.58 | 0.92 |
| Bellomi et al., 2005( 34 ) | Eur Radiol | Single helical | r | 62 | 31/48 (65) | 418/448 (93) | 9.29 | 0.38 |
*All studies used contrast agents. †Pelvic lymph nodes; ‡para‐aortic lymph nodes. N, number of patients who fulfilled the inclusion criteria; p, patient‐based comparison; r, region‐specific comparison.
Table 2.
Magnetic resonance imaging studies included in the meta‐analysis (n = 31)
| Study, year (reference) | Journal | Strength of magnetic field | Comparison | N | Sensitivity, no. (%) | Specificity, no. (%) | Positive likelihood ratio | Negative likelihood ratio |
|---|---|---|---|---|---|---|---|---|
| Hricak et al., 1988 ( 98 ) | Radiology | 1.5 tesla | p | 57 | 9/11 (82) | 44/46 (96) | 18.82 | 0.19 |
| Waggenspack et al., 1988( 99 ) | J Comput Assist Tomogr | 1.5 tesla | p | 20 | 3/3 (100) | 17/17 (100) | NA | 0 |
| Greco et al., 1989( 100 ) | Clinical Radiology | 1.5 tesla | p | 46 | 3/8 (38) | 32/38 (84) | 2.38 | 0.74 |
| Janus et al., 1989( 90 ) | Clin Imaging | 1.5 tesla | p | 22 | 3/4 (75) | 16/18 (89) | 6.75 | 0.74 |
| Kim et al., 1990( 92 ) | Radiology | 2.0 tesla | p | 30 | 3/15 (20) | 44/45 (98) | 4.00 | 0.79 |
| Ho et al., 1992( 64 ) | J Fromos Med Assoc | 1.5 tesla | p | 20 | 0/5 (0) | 15/15 (100) | 2.67 | 0.95 |
| Kim et al., 1993( 94 ) | J Comput Assist Tomogr | 2.0 tesla | p | 99 | 7/29 (24) | 167/169 (99) | 20.40 | 0.77 |
| Hawnaur et al., 1994( 101 ) | Clin Radiol | 1.5 tesla | p | 49 | 12/16 (75) | 29/33 (88) | 6.19 | 0.28 |
| Kim et al., 1994( 102 ) | Radiology | 1.5 tesla | p | 272 | 23/37 (62) | 223/235 (98) | 29.22 | 0.39 |
| Subak et al., 1995( 95 ) | Obstet Gynecol | 1.5 tesla | p | 71 | 8/13 (62) | 53/58 (91) | 7.14 | 0.42 |
| Hawighorst et al., 1998( 103 ) | J Comput Assist Tomogr | 1.5 tesla | p | 33 | 13/19 (68) | 11/14 (78) | 3.09 | 0.41 |
| Yu et al., 1998( 104 ) | Am J Roentgenol | 1.5 tesla | p | 94 | 9/14 (64) | 73/80 (91) | 7.11 | 0.40 |
| Yang et al., 2000( 97 ) | Am J Roentgenol | 1.5 tesla | r | 43 | 12/17 (71) | 53/59 (90) | 7.11 | 0.32 |
| Narayan et al., 2001( 20 ) | Int J Gynecol Cancer | 1.5 tesla | p | 24 | 6/12 (50) | 12/12 (100) | 6.25 | 0.54 |
| Reinhardt et al., 2001( 19 ) | Radiology | 1.5 tesla | p | 35 | 8/11 (73) | 20/24 (83) | 4.29 | 0.33 |
| Reinhardt et al., 2001( 19 ) | Radiology | 1.5 tesla | r | 35 | 14/22 (67) | 263/270 (97) | 22.33 | 0.34 |
| Sheu et al., 2001( 105 ) | Eur Radiol | 1.5 tesla | p | 41 | 9/11 (82) | 26/30 (87) | 6.31 | 0.21 |
| Hertel et al., 2002†( 36 ) | Gynecol Oncol | Not presented | p | 67 | 3/12 (25) | 48/55 (87) | 1.92 | 0.86 |
| Hertel et al., 2002‡( 36 ) | Gynecol Oncol | Not presented | p | 67 | 0/6 (0) | 60/61 (98) | 0.00 | 1.02 |
| Choi et al., 2004( 106 ) | J Comput Assist Tomogr | 1.5 tesla | r | 113 | 8/22 (36) | 198/204 (97) | 12.00 | 0.29 |
| Bellomi et al., 2005( 34 ) | Eur Radiol | 1.0 tesla | r | 62 | 35/48 (73) | 417/448 (93) | 10.43 | 0.29 |
| Park et al., 2005( 27 ) | Jpn J Clin Oncol | 1.5 tesla | p | 36 | 8/14 (57) | 16/22 (73) | 2.11 | 0.59 |
| Park et al., 2005( 27 ) | Jpn J Clin Oncol | 1.5 tesla | r | 36 | 12/22 (55) | 40/50 (80) | 2.75 | 0.56 |
| Choi et al., 2006( 37 ) | Am J Roentgenol | 1.5 tesla | r | 55 | 12/36 (33) | 346/349 (97) | 11.00 | 0.69 |
| Choi et al., 2006( 37 ) | Am J Roentgenol | 1.5 tesla | n | 55 | 15/62 (24) | 1830/1847 (99) | 24.00 | 0.77 |
| Choi et al., 2006( 30 ) | Cancer | 1.5 tesla | p | 22 | 4/13 (39) | 4/9 (44) | 0.70 | 1.39 |
| Choi et al., 2006( 30 ) | Cancer | 1.5 tesla | r | 22 | 10/33 (30) | 112/121 (93) | 4.29 | 0.75 |
| Chung et al., 2007( 35 ) | Jpn J Clin Oncol | 1.5 tesla | p | 119 | 24/34 (71) | 59/85 (69) | 2.29 | 0.42 |
| Chung et al., 2007( 35 ) | Jpn J Clin Oncol | 1.5 tesla | r | 119 | 30/74 (41) | 693/759 (91) | 4.56 | 0.65 |
| Sahdev et al., 2007( 38 ) | Int J Gynecol Cancer | 1.5 tesla | p | 150 | 7/19 (37) | 120/131 (92) | 4.63 | 0.68 |
| Sahdev et al., 2007( 38 ) | Int J Gynecol Cancer | 1.5 tesla | n | 150 | 12/44 (27) | 1427/1442 (99) | 27.00 | 0.74 |
†Pelvic lymph nodes; ‡para‐aortic lymph nodes. N, number of patients who fulfilled the inclusion criteria; NA, not available: Positive likelihood could not be calculated because specificity was 1.0; p, patient‐based comparison; r, region‐specific comparison; n, node‐based comparison.
Table 3.
PET or PET/CT studies included in the meta‐analysis (n = 20)
| Study, year (reference) | Journal | Comparison | N | Sensitivity, no. (%) | Specificity, no. (%) | Positive likelihood ratio | Negative likelihood ratio |
|---|---|---|---|---|---|---|---|
| Narayan et al., 2001†( 20 ) | Int J Gynecol Cancer | p | 24 | 10/12 (83) | 11/12 (92) | 10.38 | 0.18 |
| Narayan et al., 2001‡( 20 ) | Int J Gynecol Cancer | r | 24 | 4/7 (57) | 16/17 (94) | 9.5 | 0.46 |
| Reinhardt et al., 2001( 19 ) | Radiology | p | 35 | 10/11 (91) | 24/24 (100) | NA | 0.09 |
| Reinhardt et al., 2001( 19 ) | Radiology | r | 35 | 17/21 (81) | 269/271 (99) | 81 | 0.19 |
| Belhocine et al., 2002( 22 ) | Gynecol Oncol | n | 22 | 19/27 (70) | 184/190 (97) | 23.33 | 0.31 |
| Yeh et al., 2002‡( 21 ) | Oncol Rep | p | 42 | 10/12 (83) | 29/30 (97) | 27.67 | 0.18 |
| Lin et al., 2003‡( 107 ) | Gynecol Oncol | p | 50 | 12/14 (86) | 34/36 (94) | 14.33 | 0.15 |
| Ma et al., 2003( 24 ) | J Nucl Med | p | 104 | 38/38 (100) | 66/66 (100) | NA | 0 |
| Park et al., 2005( 27 ) | Jpn J Clin Oncol | p | 36 | 6/14 (43) | 22/22 (100) | NA | 0.57 |
| Park et al., 2005( 27 ) | Jpn J Clin Oncol | r | 36 | 9/22 (41) | 50/50 (100) | NA | 0.59 |
| Roh et al., 2005( 26 ) | Eur J Cancer | r | 54 | 14/37 (38) | 383/395 (97) | 12.67 | 0.64 |
| Wright et al., 2005†( 28 ) | Cancer | p | 59 | 10/19 (53) | 36/40 (90) | 5.30 | 0.52 |
| Wright et al., 2005‡( 28 ) | Cancer | p | 45 | 1/4 (25) | 40/41 (98) | 12.50 | 0.77 |
| Wright et al., 2005†( 28 ) | Cancer | r | 59 | 12/26 (46) | 84/92 (91) | 5.11 | 5.9 |
| Wright et al., 2005‡( 28 ) | Cancer | r | 45 | 2/5 (40) | 84/85 (99) | 40 | 0.61 |
| Choi et al., 2006( 30 ) | Cancer | p | 22 | 10/13 (77) | 5/9 (56) | 1.75 | 0.41 |
| Choi et al., 2006( 30 ) | Cancer | r | 22 | 19/33 (58) | 112/121 (93) | 8.29 | 0.45 |
| Sironi et al., 2006( 31 ) | Radiology | p | 47 | 13/18 (72) | 18/19 (95) | 14.40 | 0.29 |
| Loft et al., 2007†( 33 ) | Gynecol Oncol | p | 78 | 21/21 (100) | 50/57 (87) | 7.69 | 0 |
| Loft et al., 2007‡( 33 ) | Gynecol Oncol | p | 119 | 15/15 (100) | 103/104 (99) | 100 | 0 |
†Pelvic lymph nodes; ‡para‐aortic lymph nodes. N, number of patients who fulfilled the inclusion criteria; NA, not available: Positive likelihood could not be calculated because specificity was 1.0; p, patient‐based comparison; r, region‐specific comparison; n, node‐based comparison; CT, computed tomography; PET, positron emission tomography.
Patient‐based data analysis. Figure 2 shows the sROC curves of the performance of CT, MRI, and PET or PET/CT for detection of metastatic lymph nodes in cervical cancer patients, based on a patient‐based data analysis. AUCs (SE) of each modality were 0.8998 (0.042), 0.8270 (0.045), and 0.9641 (0.021) for CT, MRI, and PET or PET/CT, respectively; Q* values were 0.8310 (0.0456), 0.7599 (0.0409), and 0.9106 (0.0322), respectively. Table 4 shows summary sensitivity and specificity of three modalities by patient‐based data. A significant heterogeneity was found in most analyses except for the meta‐analysis of CT studies for specificity (I 2 = 31.6). Summary sensitivity and specificity of CT were 50% (95% confidence interval [CI]: 43%, 57%) and 92.0% (95% CI: 90%, 94%). For MRI, summary sensitivity and specificity were 56% (95% CI: 51%, 62%) and 91% (95% CI: 90%, 93%). For PET or PET/CT, summary sensitivity and specificity were 82% (95% CI: 75%, 87%) and 95% (95% CI: 93%, 97%).
Figure 2.
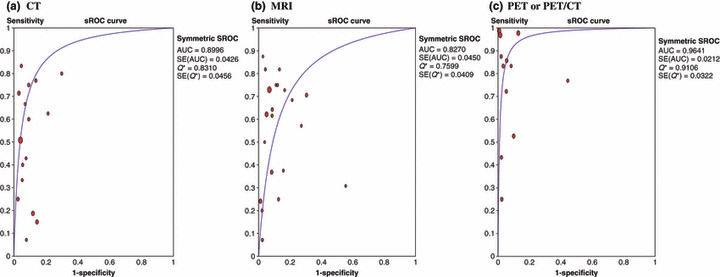
Summary receiver operating characteristic (sROC) curve of the performance of (a) computed tomography (CT), (b) magnetic resonance imaging (MRI), and (c) positron emission tomography (PET or PET/CT) for detection of metastatic lymph nodes in patients with cervical cancer based on a patient‐based data analysis. AUC, area under the curve; Q*, Q* value.
Table 4.
Summary sensitivity and specificity of CT, MRI, and PET or PET/CT
| Category | No. of studies | Summary sensitivity, % (95% CI) | I 2* (%) | Summary specificity, % (95% CI) | I 2* (%) |
|---|---|---|---|---|---|
| Patient‐based comparison | |||||
| CT | 16 | 50 (43–57) | 71.1 | 92 (90–94) | 31.6 |
| MRI | 21 | 56 (51–62) | 70.7 | 91 (90–93) | 80.1 |
| PET or PET/CT | 12 | 82 (75–87) | 80.7 | 95 (93–97) | 69.7 |
| Region/node‐based comparison | |||||
| CT | 4 | 52 (42–62) | 78.0 | 92 (90–94) | 81.5 |
| MRI | 9 | 38 (32–43) | 67.7 | 97 (97–98) | 95.0 |
| PET or PET/CT | 8 | 54 (46–61) | 57.3 | 97 (96–98) | 70.9 |
*Test for heterogeneity: An I 2 value greater than 50% was considered to indicate substantial heterogeneity across the studies included in the analysis. CT, computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography.
Table 5 shows pair‐wise comparisons between modalities for sensitivity, specificity, AUC, and Q* values by patient‐based data. Statistically significant differences were observed in the following comparisons: CT versus PET or PET/CT (50%vs 82%, P < 0.001) and MRI versus PET or PET/CT (56%vs 82%, P < 0.001) for sensitivity; CT versus PET or PET/CT (92%vs 95%, P = 0.04) and MRI versus PET or PET/CT (91%vs 95%, P < 0.001) for specificity; MRI versus PET or PET/CT (0.8270 vs 0.9641, <0.001) for AUC; and MRI versus PET or PET/CT (0.7599 vs 0.9106, P < 0.001) for Q* values.
Table 5.
Pair‐wise comparisons between modalities for sensitivity, specificity, AUC, and Q*
| Category | Sensitivity | Specificity | AUC | Q* |
|---|---|---|---|---|
| Patient‐based comparison | ||||
| CT vs MRI | 50%vs 56% (0.19) | 92%vs 91% (0.43) | 0.8998 vs 0.8270 (0.28) | 0.8310 vs 0.7599 (0.25) |
| CT vs PET or PET/CT | 50%vs 82% (<0.001†) | 92%vs 95% (0.04†) | 0.8998 vs 0.9641 (0.18) | 0.8310 vs 0.9160 (0.15) |
| MRI vs PET or PET/CT | 56%vs 82% (<0.001†) | 91%vs 95% (<0.001†) | 0.8270 vs 0.9641 (<0.001†) | 0.7599 vs 0.9160 (<0.001†) |
| Region/node‐based comparison | ||||
| CT vs MRI | 52%vs 38% (0.02†) | 92%vs 97% (<0.001†) | 0.8085 vs 0.7450 (0.79) | 0.7433 vs 0.6893 (0.80) |
| CT vs PET or PET/CT | 52%vs 54% (0.75) | 92%vs 97% (<0.001†) | 0.8085 vs 0.8181 (0.97) | 0.7433 vs 0.7519 (0.97) |
| MRI vs PET or PET/CT | 38%vs 54% (<0.001†) | 97%vs 97% (1.00) | 0.7450 vs 0.8181 (0.72) | 0.6893 vs 0.7519 (0.72) |
*By two‐sample Z‐test, values of parenthesis indicate P‐value; †significant, P < 0.05. Q* corresponds to the point on the sROC curve where sensitivity and specificity are equal. AUC, area under the curve; CT, computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography; sROC, summary receiver operating characteristic curve.
Region‐ or node‐based data analysis. Figure 3 shows the sROC curves of the performance of CT, MRI, and PET or PET/CT for detection of metastatic lymph nodes in cervical cancer patients, based on region‐ or node‐based data analysis. AUCs (SE) of each modality were 0.8085 (0.2273), 0.7450 (0.0877), and 0.8181 (0.1815) for CT, MRI, and PET or PET/CT, respectively; Q* values were 0.7433 (0.2008), 0.6893 (0.0722), and 0.7519 (0.1626), respectively. Table 4 shows summary sensitivity and specificity of three modalities by region‐ or node‐based data. Summary sensitivity and specificity of CT were 52% (95% CI: 42%, 62%) and 92% (95% CI: 90%, 94%). For MRI, summary sensitivity and specificity were 38% (95% CI: 32%, 43%) and 97% (95% CI: 97%, 98%). For PET or PET/CT, summary sensitivity and specificity were 54% (95% CI: 46%, 61%) and 97% (95% CI: 96%, 98%).
Figure 3.
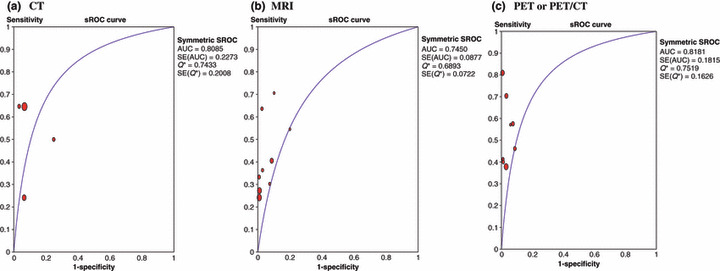
Summary receiver operating characteristic curve of the performance of (a) computed tomography (CT), (b) magnetic resonance imaging (MRI), and (c) positron emission tomography (PET or PET/CT) for detection of metastatic lymph nodes in patients with cervical cancer based on a region‐ or node‐based analysis. AUC, area under the curve; Q*, Q* value.
Table 5 shows pair‐wise comparisons between modalities for sensitivity, specificity, AUC, and Q* values by region‐ or node‐based data. Statistically significant differences were observed in the following comparisons: CT versus MRI (52%vs 38%, P = 0.02) and MRI versus PET or PET/CT (38%vs 54%, P < 0.001) for sensitivity; and CT versus MRI (92%vs 97%, P < 0.001) and CT versus PET or PET/CT (92%vs 97%, P < 0.001) for specificity. However, there was no significant difference regarding AUC and Q* values.
Discussion
Positron emission tomography using FDG is well known for a technique being used for the assessment of primary tumors and metastases, and prognostic stratifications and planning and monitoring of tumor therapy, as well as for the early detection of recurrent tumor growth.( 75 ) Also, simultaneous acquisition of co‐registered PET and CT images (PET/CT) enables more precise discrimination between physiologic and malignant FDG uptake and more accurate localization of lesions.( 76 ) Our meta‐analysis showed that PET or PET/CT had overall higher sensitivity, specificity, AUC, and Q* than did CT or MRI in a patient‐based data analysis; and a higher sensitivity than did MRI and a higher specificity than did CT in region‐ or node‐based data analyses, for detection of metastatic lymph nodes in patients with cervical cancer. Our findings are similar to those of the previous meta‐analyses. Regarding the performance of PET or PET/CT, a recent meta‐analysis by Gu et al. reported that AUC of PET, whether interpreted with or without the use of CT, was higher than that of CT or MR (P < 0.05) in diagnosing recurrent ovarian carcinoma.( 77 ) Also, Zhang et al.’s meta‐analysis reported that PET had a quite high sensitivity (0.91) and specificity (0.83) for detecting distant metastasis in recurrent colorectal carcinoma.( 78 )
Clinically, the presence of metastatic lymph nodes significantly influences the prognosis of cervical cancer patients.( 12 , 13 , 14 , 79 , 80 , 81 ) In patients with surgically staged and locally advanced cervical carcinoma, 5‐year survival rates of patients without a metastatic lymph node, with a pelvic metastatic lymph node, or with a para‐aortic metastatic lymph node, were reported to be 57%, 34%, and 12%, respectively.( 82 ) Nevertheless, lymph node metastasis is not used in the FIGO staging of cervical cancer.( 1 )
Before the use of PET, three diagnostic modalities such as lymphangiography (LAG), CT, and MRI had been used to assess the metastatic lymph nodes of cervical cancer patients. Scheidler et al. compared the utility of LAG, CT, and MRI for the diagnosis of metastatic lymph nodes in patients with cervical cancer via a meta‐analysis in 1997.( 15 ) They reported a trend toward better performance for MRI than for LAG or CT, even though sROC analyses showed no significant differences in the overall performance of these modalities. However, those findings are less informative for the following reasons: first, LAG is no longer used in clinical settings because of significant improvements in CT and MRI techniques; second, many of the studies included in the cited overview adopted per‐patient‐based comparisons and this makes it difficult to directly compare the diagnostic performance of CT or MRI in the detection of metastatic lymph nodes when researchers adopt per‐region or per‐node‐based comparisons; third, since 2001, a lot of studies have reported that PET or PET/CT, as a state‐of‐the art noninvasive diagnostic tool, showed a better diagnostic performance for the detection of metastatic lymph nodes in patients with cervical cancer, compared with CT or MRI.
To the best of our knowledge, the current study is the first meta‐analysis of the diagnostic performance of CT, MRI, and PET in the detection of metastatic lymph nodes in cervical cancer patients. In our study, we subdivided the included studies into a per‐patient comparison group and per‐region or per‐node comparison groups. CT and MRI had similar diagnostic performance but both had lower sensitivity and higher specificity than had PET. This may be because the criteria of the lymph node size were used differently in each study, and also CT and MRI cannot readily differentiate metastatic nodes from hyperplastic nodes of the similar size. In some studies, the mean size of a metastatic lymph node was smaller than 1 cm (largest short axis diameter).( 37 ) This may also explain the low performance of CT and MRI when the largest short axis diameter is the diagnostic size criterion. Even though our study showed that the diagnostic performance of PET was significantly better than that of CT and MRI in both a per‐patient based data analysis and region‐ or node‐based data analyses, PET has a lower diagnostic performance than surgical staging for the detection of metastatic lymph nodes in cervical cancer patients.
Also, we performed subgroup analyses based on the type of CT scanner (not shown due to space limitation). Sensitivity was 39% (95% CI: 30%, 49%; n = 5) for helical CT (per‐patient based comparison) and 61% (95% CI: 52%, 71%; n = 11) for non‐helical CT (per‐patient based comparison), while specificity was similar between them: 92% (95% CI: 89%, 95%) and 92% (88%, 95%), respectively. We could guess that a possible reason for the discrepancy between helical and non‐helical CT might be associated with publication bias in the early period. That is, studies showing low sensitivity might not be published.
More importantly, especially in regard to the detection of metastatic lymph nodes, we think that diagnostic performance in CT imaging is not that much related with technical development because the important criteria for metastatic lymph nodes is only the size of lymph node; CT imaging in the early period might also be able to detect metastatic lymph nodes more than 1 cm in size as well as those modalities in the recent period. Again, although recent studies involved more advanced imaging with higher image quality than the previous ones, and lymph nodes detected additionally tended to be smaller (<1 cm in a short axis diameter), these additionally detected lymph nodes might not change the diagnostic decision (size criteria for lymph node metastasis is usually more than 1 cm) in imaging analyses (CT or MRI).
Our study had several limitations. First, we did not evaluate a region‐based or node‐based data analysis separately because only three studies( 22 , 37 , 38 ) had reported the findings from a node‐based data analysis. This could be evaluated after more individual studies are conducted. Second, we could not present the exact reasons for heterogeneity which was observed in the meta‐analysis for most summary sensitivity and specificity of CT, MRI, and PET, except for summary specificity of CT (I 2 = 31.6). However, this may be associated with the variation in patient characteristics or quality of studies. Third, we did not evaluate diagnostic performances by type of MRI scanner. It is not likely that subgroup analyses by type of MRI scanner would provide additional useful information because most studies (26 of 32 MRI studies) used 1.5‐tesla MR scanners. Last, there could be reporting or publication bias on this topic. Further research is required to determine this.
In conclusion, this meta‐analysis is the first study to evaluate the diagnostic performance of CT, MRI, and PET in the detection of metastatic lymph nodes in cervical cancer patients. Our study showed that PET or PET/CT had an overall higher diagnostic performance than those of CT or MRI in detecting metastatic lymph nodes in patients with cervical cancer. These findings have a clinical implication in that they may give useful information to not only radiologists in interpretation of images but also gynecologic oncologists in choosing imaging modality in the management of cervical cancer patients.
Contributorship statement and guarantor
S.K. Myung and H.J. Choi were responsible for the initial plan, study design, statistical analysis, and the conduct of the study. H.J. Choi, S.K. Myung, and W. Ju were responsible for data collection, data extraction, data interpretation, and manuscript drafting. H.J. Choi, W. Ju, Y. Kim, and S.K. Myung were responsible for data interpretation and manuscript drafting. S.K. Myung is the guarantor for this paper and has full responsibility for this study.
Disclosure Statement
The authors have no conflict of interest.
H. J. Choi and W. Ju equally contributed to this article.
References
- 1. Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer 2001; 94: 153–6. [DOI] [PubMed] [Google Scholar]
- 2. Ballon SC, Berman ML, Lagasse LD, Petrilli ES, Castaldo TW. Survival after extraperitoneal pelvic and paraaortic lymphadenectomy and radiation therapy in cervical carcinoma. Obstet Gynecol 1981; 57: 90–5. [PubMed] [Google Scholar]
- 3. Averette HE, Dudan RC, Ford JH Jr. Exploratory celiotomy for surgical staging of cervical cancer. Am J Obstet Gynecol 1972; 113: 1090–6. [DOI] [PubMed] [Google Scholar]
- 4. Takeshima N, Yanoh K, Tabata T, Nagai K, Hirai Y, Hasumi K. Assessment of the revised International Federation of Gynecology and obstetrics staging for early invasive squamous cervical cancer. Gynecol Oncol 1999; 74: 165–9. [DOI] [PubMed] [Google Scholar]
- 5. Hricak H, Gatsonis C, Chi DS et al. Role of imaging in pretreatment evaluation of early invasive cervical cancer: results of the intergroup study American College of Radiology Imaging Network 6651‐Gynecologic Oncology Group 183. J Clin Oncol 2005; 23: 9329–37. [DOI] [PubMed] [Google Scholar]
- 6. Hopkins MP, Morley GW. Prognostic factors in advanced stage squamous cell cancer of the cervix. Cancer 1993; 72: 2389–93. [DOI] [PubMed] [Google Scholar]
- 7. Stehman FB, Bundy BN, DiSaia PJ, Keys HM, Larson JE, Fowler WC. Carcinoma of the cervix treated with radiation therapy. I. A multi‐variate analysis of prognostic variables in the Gynecologic Oncology Group. Cancer 1991; 67: 2776–85. [DOI] [PubMed] [Google Scholar]
- 8. Sakuragi N, Satoh C, Takeda N et al. Incidence and distribution pattern of pelvic and paraaortic lymph node metastasis in patients with Stages IB, IIA, and IIB cervical carcinoma treated with radical hysterectomy. Cancer 1999; 85: 1547–54. [DOI] [PubMed] [Google Scholar]
- 9. Lai CH, Hong JH, Hsueh S et al. Preoperative prognostic variables and the impact of postoperative adjuvant therapy on the outcomes of Stage IB or II cervical carcinoma patients with or without pelvic lymph node metastases: an analysis of 891 cases. Cancer 1999; 85: 1537–46. [DOI] [PubMed] [Google Scholar]
- 10. Downey GO, Potish RA, Adcock LL, Prem KA, Twiggs LB. Pretreatment surgical staging in cervical carcinoma: therapeutic efficacy of pelvic lymph node resection. Am J Obstet Gynecol 1989; 160: 1055–61. [DOI] [PubMed] [Google Scholar]
- 11. Potish RA, Twiggs LB, Okagaki T, Prem KA, Adcock LL. Therapeutic implications of the natural history of advanced cervical cancer as defined by pretreatment surgical staging. Cancer 1985; 56: 956–60. [DOI] [PubMed] [Google Scholar]
- 12. Lagasse LD, Creasman WT, Shingleton HM, Ford JH, Blessing JA. Results and complications of operative staging in cervical cancer: experience of the Gynecologic Oncology Group. Gynecol Oncol 1980; 9: 90–8. [DOI] [PubMed] [Google Scholar]
- 13. Kupets R, Covens A. Is the International Federation of Gynecology and Obstetrics staging system for cervical carcinoma able to predict survival in patients with cervical carcinoma?: an assessment of clinimetric properties. Cancer 2001; 92: 796–804. [DOI] [PubMed] [Google Scholar]
- 14. Chung CK, Nahhas WA, Zaino R, Stryker JA, Mortel R. Histologic grade and lymph node metastasis in squamous cell carcinoma of the cervix. Gynecol Oncol 1981; 12: 348–54. [DOI] [PubMed] [Google Scholar]
- 15. Scheidler J, Hricak H, Yu KK, Subak L, Segal MR. Radiological evaluation of lymph node metastases in patients with cervical cancer. A meta‐analysis. JAMA 1997; 278: 1096–101. [PubMed] [Google Scholar]
- 16. Sugawara Y, Eisbruch A, Kosuda S, Recker BE, Kison PV, Wahl RL. Evaluation of FDG PET in patients with cervical cancer. J Nucl Med 1999; 40: 1125–31. [PubMed] [Google Scholar]
- 17. Rose PG, Adler LP, Rodriguez M, Faulhaber PF, Abdul‐Karim FW, Miraldi F. Positron emission tomography for evaluating para‐aortic nodal metastasis in locally advanced cervical cancer before surgical staging: a surgicopathologic study. J Clin Oncol 1999; 17: 41–5. [DOI] [PubMed] [Google Scholar]
- 18. Umesaki N, Tanaka T, Miyama M et al. The role of 18F‐fluoro‐2‐deoxy‐D‐glucose positron emission tomography (18F‐FDG‐PET) in the diagnosis of recurrence and lymph node metastasis of cervical cancer. Oncol Rep 2000; 7: 1261–4. [DOI] [PubMed] [Google Scholar]
- 19. Reinhardt MJ, Ehritt‐Braun C, Vogelgesang D et al. Metastatic lymph nodes in patients with cervical cancer: detection with MR imaging and FDG PET. Radiology 2001; 218: 776–82. [DOI] [PubMed] [Google Scholar]
- 20. Narayan K, Hicks RJ, Jobling T, Bernshaw D, McKenzie AF. A comparison of MRI and PET scanning in surgically staged loco‐regionally advanced cervical cancer: potential impact on treatment. Int J Gynecol Cancer 2001; 11: 263–71. [DOI] [PubMed] [Google Scholar]
- 21. Yeh LS, Hung YC, Shen YY, Kao CH, Lin CC, Lee CC. Detecting para‐aortic lymph nodal metastasis by positron emission tomography of 18F‐fluorodeoxyglucose in advanced cervical cancer with negative magnetic resonance imaging findings. Oncol Rep 2002; 9: 1289–92. [PubMed] [Google Scholar]
- 22. Belhocine T, Thille A, Fridman V et al. Contribution of whole‐body 18FDG PET imaging in the management of cervical cancer. Gynecol Oncol 2002; 87: 90–7. [DOI] [PubMed] [Google Scholar]
- 23. Singh AK, Grigsby PW, Dehdashti F, Herzog TJ, Siegel BA. FDG‐PET lymph node staging and survival of patients with FIGO stage IIIb cervical carcinoma. Int J Radiat Oncol Biol Phys 2003; 56: 489–93. [DOI] [PubMed] [Google Scholar]
- 24. Ma SY, See LC, Lai CH et al. Delayed (18)F‐FDG PET for detection of paraaortic lymph node metastases in cervical cancer patients. J Nucl Med 2003; 44: 1775–83. [PubMed] [Google Scholar]
- 25. Tsai C‐S, Chang T‐C, Lai C‐H et al. Preliminary report of using FDG‐PET to detect extrapelvic lesions in cervical cancer patients with enlarged pelvic lymph nodes on MRI/CT. Int J Radiat Oncol Biol Phys 2004; 58: 1506–12. [DOI] [PubMed] [Google Scholar]
- 26. Roh JW, Seo SS, Lee S et al. Role of positron emission tomography in pretreatment lymph node staging of uterine cervical cancer: a prospective surgicopathologic correlation study. Eur J Cancer 2005; 41: 2086–92. [DOI] [PubMed] [Google Scholar]
- 27. Park W, Park YJ, Huh SJ et al. The usefulness of MRI and PET imaging for the detection of parametrial involvement and lymph node metastasis in patients with cervical cancer. Jpn J Clin Oncol 2005; 35: 260–4. [DOI] [PubMed] [Google Scholar]
- 28. Wright JD, Dehdashti F, Herzog TJ et al. Preoperative lymph node staging of early‐stage cervical carcinoma by [18F]‐fluoro‐2‐deoxy‐D‐glucose‐positron emission tomography. Cancer 2005; 104: 2484–91. [DOI] [PubMed] [Google Scholar]
- 29. Unger JB, Ivy JJ, Ramaswamy MR, Charrier A, Connor P. Whole‐body [18F]fluoro‐2‐deoxyglucose positron emission tomography scan staging prior to planned radical hysterectomy and pelvic lymphadenectomy. Int J Gynecol Cancer 2005; 15: 1060–4. [DOI] [PubMed] [Google Scholar]
- 30. Choi HJ, Roh JW, Seo SS et al. Comparison of the accuracy of magnetic resonance imaging and positron emission tomography/computed tomography in the presurgical detection of lymph node metastases in patients with uterine cervical carcinoma: a prospective study. Cancer 2006; 106: 914–22. [DOI] [PubMed] [Google Scholar]
- 31. Sironi S, Buda A, Picchio M et al. Lymph node metastasis in patients with clinical early‐stage cervical cancer: detection with integrated FDG PET/CT. Radiology 2006; 238: 272–9. [DOI] [PubMed] [Google Scholar]
- 32. Zaspel U, Hamm B. Current relative values of MRI, CT and PET in the diagnosis of cervical carcinoma. Onkologe 2006; 12: 854–68. [Google Scholar]
- 33. Loft A, Berthelsen AK, Roed H et al. The diagnostic value of PET/CT scanning in patients with cervical cancer: a prospective study. Gynecol Oncol 2007; 106: 29–34. [DOI] [PubMed] [Google Scholar]
- 34. Bellomi M, Bonomo G, Landoni F et al. Accuracy of computed tomography and magnetic resonance imaging in the detection of lymph node involvement in cervix carcinoma. Eur Radiol 2005; 15: 2469–74. [DOI] [PubMed] [Google Scholar]
- 35. Chung HH, Kang SB, Cho JY et al. Can preoperative MRI accurately evaluate nodal and parametrial invasion in early stage cervical cancer? Jpn J Clin Oncol 2007; 37: 370–5. [DOI] [PubMed] [Google Scholar]
- 36. Hertel H, Kohler C, Elhawary T, Michels W, Possover M, Schneider A. Laparoscopic staging compared with imaging techniques in the staging of advanced cervical cancer. Gynecol Oncol 2002; 87: 46–51. [DOI] [PubMed] [Google Scholar]
- 37. Choi HJ, Kim SH, Seo SS et al. MRI for pretreatment lymph node staging in uterine cervical cancer. AJR Am J Roentgenol 2006; 187: W538–43. [DOI] [PubMed] [Google Scholar]
- 38. Sahdev A, Sohaib SA, Wenaden AE, Shepherd JH, Reznek RH. The performance of magnetic resonance imaging in early cervical carcinoma: a long‐term experience. Int J Gynecol Cancer 2007; 17: 629–36. [DOI] [PubMed] [Google Scholar]
- 39. Vas W, Wolverson M, Freel J, Salimi Z, Sundaram M. Computed tomography in the pretreatment assessment of carcinoma of the cervix. J Comput Tomogr 1985; 9: 359–68. [DOI] [PubMed] [Google Scholar]
- 40. Irwig L, Macaskill P, Glasziou P, Fahey M. Meta‐analytic methods for diagnostic test accuracy. J Clin Epidemiol 1995; 48: 119–30; discussion 31–2. [DOI] [PubMed] [Google Scholar]
- 41. Littenberg B, Moses LE. Estimating diagnostic accuracy from multiple conflicting reports: a new meta‐analytic method. Med Decis Making 1993; 13: 313–21. [DOI] [PubMed] [Google Scholar]
- 42. Moses LE, Shapiro D, Littenberg B. Combining independent studies of a diagnostic test into a summary ROC curve: data‐analytic approaches and some additional considerations. Stat Med 1993; 12: 1293–316. [DOI] [PubMed] [Google Scholar]
- 43. Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH. Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol 2005; 58: 982–90. [DOI] [PubMed] [Google Scholar]
- 44. Zamora J, Abraira V, Muriel A, Khan K, Coomarasamy A. Meta‐DiSc: a software for meta‐analysis of test accuracy data. BMC Med Res Methodol 2006; 6: 31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Ishii C, Tada S, Tsukioka M, Tanaka H. [CT diagnosis of uterine cancer]. Gan To Kagaku Ryoho 1982; 9: 204–8. [PubMed] [Google Scholar]
- 46. Kanehira C, Arai T, Suda Y, Suzuki M. [CT diagnosis and treatment of lymph node metastases from carcinoma of the cervix]. Rinsho Hoshasen 1983; 28: 285–92. [PubMed] [Google Scholar]
- 47. Kolesnikova EK. Computed tomography in the diagnosis of cervical cancer. Akush Ginekol 1986; 11: 18–23. [PubMed] [Google Scholar]
- 48. Ono M. Diagnostic values and limits on X‐ray computed tomography in untreated uterine cervical cancer. Nippon Igaku Hoshasen Gakkai zasshi. Nippon Acta Radiologica 1986; 46: 478–91. [PubMed] [Google Scholar]
- 49. Raber G, Luning M, Potzschke B, Natho W. [The place of lymphography and computed tomography in the determination of lymph node status in primary cancer of the cervix]. Radiol Diagn (Berl) 1986; 27: 449–59. [PubMed] [Google Scholar]
- 50. Zapf S, Halbsguth A, Schweden F et al. [Problem of pretherapeutic staging of cervical carcinoma. Studies of the diagnostic value of computerized tomography and magnetic resonance tomography in comparison with gynecologic palpation findings and pathologic‐anatomic diagnosis]. Geburtshilfe Frauenheilkd 1987; 47: 838–42. [DOI] [PubMed] [Google Scholar]
- 51. Kojima Y. [Role of X‐ray computed tomography in the staging of cervical carcinoma]. Nippon Sanka Fujinka Gakkai Zasshi 1989; 41: 341–8. [PubMed] [Google Scholar]
- 52. Raber G, Potzschke B. [The status of computerized tomography in the diagnosis of primary cervix cancer]. Geburtshilfe Frauenheilkd 1989; 49: 17–23. [DOI] [PubMed] [Google Scholar]
- 53. Kon Y, Saida Y, Kurosaki Y et al. [Psoas lymph node metastasis in patients with carcinoma of the uterine cervix]. Nippon Igaku Hoshasen Gakkai Zasshi 1990; 50: 1237–42. [PubMed] [Google Scholar]
- 54. La Fianza A, Dore R, Di Giulio G et al. [Lymph node metastasis of carcinoma of the cervix uteri. Role of lymphography and computerized tomography]. Radiol Med (Torino) 1990; 80: 486–91. [PubMed] [Google Scholar]
- 55. Yamada E. [Role of X‐ray computed tomography in the evaluation of prognosis of cervical carcinoma]. Nippon Sanka Fujinka Gakkai Zasshi 1991; 43: 205–12. [PubMed] [Google Scholar]
- 56. Matsubara M. [Clinical significance of magnetic resonance imaging (MRI) in evaluation of the extension of uterine cervical cancer]. Nippon Sanka Fujinka Gakkai Zasshi 1993; 45: 1115–22. [PubMed] [Google Scholar]
- 57. Bautz W, Merl T, Kersting‐Sommerhoff B. [Computerized tomography staging of carcinomas of the uterus]. Radiologe 1994; 34: 370–6. [PubMed] [Google Scholar]
- 58. Scheidler J, Heuck A, Reiser M. [MR tomography in staging of carcinomas of the uterus]. Radiologe 1994; 34: 377–83. [PubMed] [Google Scholar]
- 59. Keskin N, Buyru F, Baysal B, Berkman S. Computed tomographic imaging in the management of cervical cancer. Istanbul Tip Fakultesi Mecmuasi 1996; 59: 41–3. [Google Scholar]
- 60. Hawighorst H, Knapstein PG, Weikel W et al. [Invasive cervix carcinoma (pT2b‐pT4a). Value of conventional and pharmacokinetic magnetic resonance tomography (MRI) in comparison with extensive cross sections and histopathologic findings]. Radiologe 1997; 37: 130–8. [DOI] [PubMed] [Google Scholar]
- 61. Heuck A, Scheidler J, Kimmig R et al. [Lymph node staging in cervix carcinomas: the results of high‐resolution magnetic resonance tomography (MRT) with a phased‐array body coil]. Rofo 1997; 166: 210–4. [DOI] [PubMed] [Google Scholar]
- 62. Kuhnel G, Horn LC, Fischer U et al. [18F‐FDG positron‐emission‐tomography in cervical carcinoma: preliminary findings]. Zentralbl Gynakol 2001; 123: 229–35. [DOI] [PubMed] [Google Scholar]
- 63. Michniewicz K, Oellinger J. [Diagnostic imaging in invasive cervical carcinoma: MRI, CT, and ultrasonography]. Zentralbl Gynakol 2001; 123: 222–8. [DOI] [PubMed] [Google Scholar]
- 64. Ho CM, Chien TY, Jeng CM, Tsang YM, Shih BY, Chang SC. Staging of cervical cancer: comparison between magnetic resonance imaging, computed tomography and pelvic examination under anesthesia. J Formos Med Assoc 1992; 91: 982–90. [PubMed] [Google Scholar]
- 65. Parker LA, McPhail AH, Yankaskas BC, Mauro MA. Computed tomography in the evaluation of clinical stage IB carcinoma of the cervix. Gynecol Oncol 1990; 37: 332–4. [DOI] [PubMed] [Google Scholar]
- 66. Ng HT, Chen SL, Wang JC, Sheu MH. Preoperative examination with CT, MRI and comparison of both to histopathologic findings in cervical carcinoma. CME J Gynecol Oncol 1998; 3: 256–7. [Google Scholar]
- 67. Ozsarlak O, Tjalma W, Schepens E et al. The correlation of preoperative CT, MR imaging, and clinical staging (FIGO) with histopathology findings in primary cervical carcinoma. Eur Radiol 2003; 13: 2338–45. [DOI] [PubMed] [Google Scholar]
- 68. DeSouza NM, Dina R, McIndoe GA, Soutter WP. Cervical cancer: value of an endovaginal coil magnetic resonance imaging technique in detecting small volume disease and assessing parametrial extension. Gynecol Oncol 2006; 102: 80–5. [DOI] [PubMed] [Google Scholar]
- 69. Whitley NO, Brenner DE, Francis A. Computed tomographic evaluation of carcinoma of the cervix. Radiology 1982; 142: 439–46. [DOI] [PubMed] [Google Scholar]
- 70. Soeters RP, Beningfield SJ, Dehaeck K, Levin W, Bloch B. The value of magnetic resonance imaging in patients with carcinoma of the cervix (a pilot study). Eur J Surg Oncol 1991; 17: 119–24. [PubMed] [Google Scholar]
- 71. Murmu D, Bhatla N, Bhargava VL et al. Correlation of radiological and pathological assessment of tumour volume with lymph node involvement in cancer cervix. Indian J Cancer 1998; 35: 27–32. [PubMed] [Google Scholar]
- 72. Oellinger JJ, Blohmer JU, Michniewicz K et al. Pre‐operative staging of cervical cancer: comparison of magnetic resonance imaging (MRI) and computed tomography (CT) with histologic results. Zentralbl Gynakol 2000; 122: 82–91. [PubMed] [Google Scholar]
- 73. Sheu MH, Chang CY, Wang JH, Yen MS. Cervical carcinoma: assessment of parametrial invasion and lymph node metastasis with magnetic resonance imaging. Zhonghua Yi Xue Za Zhi (Taipei) 2000; 63: 634–40. [PubMed] [Google Scholar]
- 74. Grigsby PW, Siegel BA, Dehdashti F. Lymph node staging by positron emission tomography in patients with carcinoma of the cervix. J Clin Oncol 2001; 19: 3745–9. [DOI] [PubMed] [Google Scholar]
- 75. Ak I, Stokkel MP, Pauwels EK. Positron emission tomography with 2‐[18F]fluoro‐2‐deoxy‐D‐glucose in oncology. Part II. The clinical value in detecting and staging primary tumours. J Cancer Res Clin Oncol 2000; 126: 560–74. [DOI] [PubMed] [Google Scholar]
- 76. Kluetz PG, Meltzer CC, Villemagne VL et al. Combined PET/CT imaging in oncology. Impact on patient management. Clin Positron Imaging 2000; 3: 223–30. [DOI] [PubMed] [Google Scholar]
- 77. Gu P, Pan LL, Wu SQ, Sun L, Huang G. CA 125, PET alone, PET‐CT, CT and MRI in diagnosing recurrent ovarian carcinoma: a systematic review and meta‐analysis. Eur J Radiol 2009; 71: 164–74. [DOI] [PubMed] [Google Scholar]
- 78. Zhang C, Chen Y, Xue H et al. Diagnostic value of FDG‐PET in recurrent colorectal carcinoma: a meta‐analysis. Int J Cancer 2009; 124: 167–73. [DOI] [PubMed] [Google Scholar]
- 79. Liu MT, Hsu JC, Liu WS et al. Prognostic factors affecting the outcome of early cervical cancer treated with radical hysterectomy and post‐operative adjuvant therapy. Eur J Cancer Care (Engl) 2008; 17: 174–81. [DOI] [PubMed] [Google Scholar]
- 80. Van Nagell JR Jr, Roddick JW Jr, Lowin DM. The staging of cervical cancer: inevitable discrepancies between clinical staging and pathologic findinges. Am J Obstet Gynecol 1971; 110: 973–8. [DOI] [PubMed] [Google Scholar]
- 81. Novakovic P, Mandic A, Vujkov T et al. Radical hysterectomy for stage IB1 cervical carcinoma: lymph node metastasis as a prognostic factor. J BUON 2002; 7: 247–50. [PubMed] [Google Scholar]
- 82. Lanciano RM, Corn BW. The role of surgical staging for cervical cancer. Semin Radiat Oncol 1994; 4: 46–51. [DOI] [PubMed] [Google Scholar]
- 83. Grumbine FC, Rosenshein NB, Zerhouni EA, Siegelman SS. Abdominopelvic computed tomography in the preoperative evaluation of early cervical cancer. Gynecol Oncol 1981; 12: 286–90. [DOI] [PubMed] [Google Scholar]
- 84. Walsh JW, Goplerud DR. Prospective comparison between clinical and CT staging in primary cervical carcinoma. AJR Am J Roentgenol 1981; 137: 997–1003. [DOI] [PubMed] [Google Scholar]
- 85. Brenner DE, Whitley NO, Prempree T, Villasanta U. An evaluation of the computed tomographic scanner for the staging of carcinoma of the cervix. Cancer 1982; 50: 2323–8. [DOI] [PubMed] [Google Scholar]
- 86. Villasanta U, Whitley NO, Haney PJ, Brenner D. Computed tomography in invasive carcinoma of the cervix: an appraisal. Obstet Gynecol 1983; 62: 218–24. [PubMed] [Google Scholar]
- 87. Van Engelshoven JM, Versteege CW, Ruys JH, De Haan J, Sanches H. Computed tomography in staging untreated patients with cervical cancer. Gynecol Obstet Invest 1984; 18: 289–95. [DOI] [PubMed] [Google Scholar]
- 88. Bandy LC, Clarke‐Pearson DL, Silverman PM, Creasman WT. Computed tomography in evaluation of extrapelvic lymphadenopathy in carcinoma of the cervix. Obstet Gynecol 1985; 65: 73–6. [PubMed] [Google Scholar]
- 89. Camilien L, Gordon D, Fruchter RG, Maiman M, Boyce JG. Predictive value of computerized tomography in the presurgical evaluation of primary carcinoma of the cervix. Gynecol Oncol 1988; 30: 209–15. [DOI] [PubMed] [Google Scholar]
- 90. Janus CL, Mendelson DS, Moore S, Gendal ES, Dottino P, Brodman M. Staging of cervical carcinoma: accuracy of magnetic resonance imaging and computed tomography. Clin Imaging 1989; 13: 114–6. [DOI] [PubMed] [Google Scholar]
- 91. Matsukuma K, Tsukamoto N, Matsuyama T, Ono M, Nakano H. Preoperative CT study of lymph nodes in cervical cancer‐‐its correlation with histological findings. Gynecol Oncol 1989; 33: 168–71. [DOI] [PubMed] [Google Scholar]
- 92. Kim SH, Choi BI, Lee HP et al. Uterine cervical carcinoma: comparison of CT and MR findings. Radiology 1990; 175: 45–51. [DOI] [PubMed] [Google Scholar]
- 93. Heller PB, Maletano JH, Bundy BN, Barnhill DR, Okagaki T. Clinical‐pathologic study of stage IIB, III, and IVA carcinoma of the cervix: extended diagnostic evaluation for paraaortic node metastasis‐‐a Gynecologic Oncology Group study. Gynecol Oncol 1990; 38: 425–30. [DOI] [PubMed] [Google Scholar]
- 94. Kim SH, Choi BI, Han JK et al. Preoperative staging of uterine cervical carcinoma: comparison of CT and MRI in 99 patients. J Comput Assist Tomogr 1993; 17: 633–40. [DOI] [PubMed] [Google Scholar]
- 95. Subak LL, Hricak H, Powell CB, Azizi L, Stern JL. Cervical carcinoma: computed tomography and magnetic resonance imaging for preoperative staging. Obstet Gynecol 1995; 86: 43–50. [DOI] [PubMed] [Google Scholar]
- 96. Chu KK, Chang SD, Chen FP, Soong YK. Laparoscopic surgical staging in cervical cancer – preliminary experience among Chinese. Gynecol Oncol 1997; 64: 49–53. [DOI] [PubMed] [Google Scholar]
- 97. Yang WT, Lam WW, Yu MY, Cheung TH, Metreweli C. Comparison of dynamic helical CT and dynamic MR imaging in the evaluation of pelvic lymph nodes in cervical carcinoma. AJR Am J Roentgenol 2000; 175: 759–66. [DOI] [PubMed] [Google Scholar]
- 98. Hricak H, Lacey CG, Sandles LG, Chang YC, Winkler ML, Stern JL. Invasive cervical carcinoma: comparison of MR imaging and surgical findings. Radiology 1988; 166: 623–31. [DOI] [PubMed] [Google Scholar]
- 99. Waggenspack GA, Amparo EG, Hannigan EV. MR imaging of uterine cervical carcinoma. J Comput Assist Tomogr 1988; 12: 409–14. [DOI] [PubMed] [Google Scholar]
- 100. Greco A, Mason P, Leung AWL, Dische S, McIndoe GAJ, Anderson MC. Staging of carcinoma of the uterine cervix: MRI‐surgical correlation. Clin Radiol 1989; 40: 401–5. [DOI] [PubMed] [Google Scholar]
- 101. Hawnaur JM, Johnson RJ, Buckley CH, Tindall V, Isherwood I. Staging, volume estimation and assessment of nodal status in carcinoma of the cervix: comparison of magnetic resonance imaging with surgical findings. Clin Radiol 1994; 49: 443–52. [DOI] [PubMed] [Google Scholar]
- 102. Kim SH, Kim SC, Choi BI, Han MC. Uterine cervical carcinoma: evaluation of pelvic lymph node metastasis with MR imaging. Radiology 1994; 190: 807–11. [DOI] [PubMed] [Google Scholar]
- 103. Hawighorst H, Schoenberg SO, Knapstein PG et al. Staging of invasive cervical carcinoma and of pelvic lymph nodes by high resolution MRI with a phased‐array coil in comparison with pathological findings. J Comput Assist Tomogr 1998; 22: 75–81. [DOI] [PubMed] [Google Scholar]
- 104. Yu KK, Hricak H, Subak LL, Zaloudek CJ, Powell CB. Preoperative staging of cervical carcinoma: phased array coil fast spin‐echo versus body coil spin‐echo T2‐weighted MR imaging. AJR Am J Roentgenol 1998; 171: 707–11. [DOI] [PubMed] [Google Scholar]
- 105. Sheu MH, Chang CY, Wang JH, Yen MS. Preoperative staging of cervical carcinoma with MR imaging: a reappraisal of diagnostic accuracy and pitfalls. Eur Radiol 2001; 11: 1828–33. [DOI] [PubMed] [Google Scholar]
- 106. Choi SH, Kim SH, Choi HJ, Park BK, Lee HJ. Preoperative magnetic resonance imaging staging of uterine cervical carcinoma: results of prospective study. J Comput Assist Tomogr 2004; 28: 620–7. [DOI] [PubMed] [Google Scholar]
- 107. Lin WC, Hung YC, Yeh LS, Kao CH, Yen RF, Shen YY. Usefulness of (18)F‐fluorodeoxyglucose positron emission tomography to detect para‐aortic lymph nodal metastasis in advanced cervical cancer with negative computed tomography findings. Gynecol Oncol 2003; 89: 73–6. [DOI] [PubMed] [Google Scholar]


