Abstract
There is currently substantial interest in the identification of human tumor antigens for the diagnosis and immunotherapy of cancer. In our previous study, secretion character and up‐regulation of triosephosphate isomerase were observed in lung squamous cell carcinoma, and autoantibodies against triosephosphate isomerase and peroxiredoxin 6 were detected in the sera from over 25% of patients, but in none of the healthy controls. In this study, peroxiredoxin 6 was also found at higher levels in the sera of the patients. Up‐regulated triosephosphate isomerase and peroxiredoxin 6 were further validated by enzyme‐linked immunosorbent assay in an additional 61 lung squamous cell carcinoma patients, 23 lung adenocarcinoma patients, 56 other types of carcinoma patients, 12 benign lung disease patients, and 59 healthy controls. We found that both triosephosphate isomerase and peroxiredoxin 6 were specifically elevated in lung squamous cell carcinoma sera compared with other groups, with the exception of peroxiredoxin 6 in lung adenocarcinoma patients. Positive correlation between triosephosphate isomerase and distant metastasis was found. At the cut‐off point 0.221 (optical density value) on the receiver operating characteristic curve, triosephosphate isomerase could comparatively discriminate lung squamous cell carcinoma from healthy controls with a sensitivity of 65.6%, specificity 84.7%, and total accuracy 75%. For peroxiredoxin 6, at the cut‐off point 0.151, it could discriminate the two groups with a sensitivity of 70.5%, specificity 62.7%, and total accuracy 65.8%. With both triosephosphate isomerase and peroxiredoxin 6, discriminant analysis results showed that 68.9% of the lung squamous cell carcinoma and 83.1% of healthy controls were correctly classified. We concluded that triosephosphate isomerase and peroxiredoxin 6 could be markers for lung squamous cell carcinoma. (Cancer Sci 2009; 100: 2396–2401)
Lung cancer is a worldwide leading cause of cancer death, with gradually increased incidence and mortality. Despite improvements in lung cancer diagnostic imaging and therapy methods in the past decade, little progress has been made in improving survival rates. Early detection of lung cancer remains one of the biggest challenges. Detection of lung cancer at early disease stages is the first step to successful clinical therapy and increased survival. Nevertheless, few of the identified markers for lung cancer, such as neuron‐specific enolase, carcinoembryonic antigen, cytokeratin 19 fragment (CYFRA 21‐1), squamous cell carcinoma antigen, cancer antigen‐125, and tissue polypeptide antigen, have been proven acceptable for routine clinical use, because of conflicting results from different studies or low sensitivity and specificity not reliable for clinical diagnosis.( 1 , 2 ) Therefore, identification of clinical reliable biomarkers is essential for effective early diagnosis of lung cancer.
In our previous study, autoantibodies against triosephosphate isomerase (TIM) and peroxiredoxin 6 (PRDX6) were identified in 25% and 30% of lung squamous cell carcinoma patients but in none of the healthy controls.( 3 ) Moreover, secretion character and increased expression of TIM in lung squamous cell carcinoma were detected.( 3 ) As the secretion character of PRDX6 was reported,( 4 ) we hypothesized that serum TIM and PRDX6 might be useful in the detection of lung squamous cell carcinoma. In this study, we evaluated PRDX6 levels with Western blot analysis and further measured the serum level of TIM and PRDX6 in lung squamous cell carcinoma patients and other control groups using the enzyme‐linked immunosorbent assay (ELISA) method. The potential power of TIM and PRDX6 for discriminating between the lung squamous cell carcinoma group and healthy control group was then evaluated using receiver operating characteristic (ROC) curve analysis and Bayes discriminant analysis.
In addition, the correlations of TIM and PRDX6 with clinical stages and the differentiation of lung squamous cell carcinoma were also evaluated to find their potential diagnostic power.
Materials and Methods
Serum samples. A total of 174 serum samples were applied in this study. There were 61 serum samples from lung squamous cell carcinoma patients with an age range of 24–80 years (median, 56 years); 23 serum samples from patients with lung adenonocarcinoma with an age range of 28–76 years (median, 54 years); and 56 serum samples from patients with other types of carcinoma with an age range of 26–80 years (median, 51 years), including five with nasopharyngeal carcinoma, 20 with esophageal carcinoma, six with breast cancer, seven with gastric carcinoma, and 18 colon cancer. In addition, 59 healthy controls without a prior history of cancer or autoimmune disease, which were gender‐ and age‐matched with 59 of the 61 lung squamous cell carcinoma patients, and 12 patients with lung benign disease, were used. All sera under study were collected between December 2004 and May 2008, as described in detail in Table 1. All the serum specimens were obtained from Xiangya Hospital of Central South University and Hunan Provincial Tumor Hospital, Hunan, China. All samples were collected at the time of diagnosis before any therapy following informed consent, and were prepared as previously described.( 5 )
Table 1.
Characteristics of serum samples
| Characteristics | No. of samples |
|---|---|
| Lung squamous cell carcinoma | 61 |
| Male | 52 |
| Female | 9 |
| Mean age (years) | 56 (24–80) |
| Disease stage | 61 |
| I | 24 |
| II | 11 |
| III | 17 |
| IV | 9 |
| Differentiation | 61 |
| Well differentiated | 5 |
| Moderately differentiated | 46 |
| Poorly differentiated | 10 |
| Lung adenocarcinoma | 23 |
| Non‐cancer controls | 71 |
| Normal controls | 59 |
| Male | 50 |
| Female | 9 |
| Mean age (years) | 56 (24–71) |
| Benign lung disease | 12 |
| Pneumonia | 6 |
| Tuberculosis | 6 |
| Other types of cancer | 56 |
| Nasopharyngeal carcinoma | 5 |
| Esophageal carcinoma | 20 |
| Breast cancer | 6 |
| Gastric cancer | 7 |
| Colon cancer | 18 |
Coating and detecting antibodies for ELISA. To increase the sensitivity of the detection, we used a third horseradish peroxidase–conjugated antibody against the detecting antibody in the ELISA, and through the process, the reaction was magnified twice. For TIM, mouse antihuman monoclonal antibody (TIM, H00007167‐M01; Abnova, Taipei, Taiwan) was used as the capture antibody. When the antigen was captured, goat anti‐TIM polyclonal antibody (sc‐22031; Santa Cruz Biotechnology, Santa Cruz, CA, USA) was used as the detecting antibody, and then horseradish peroxidase–conjugated rabbit antigoat antibody was used to link the detecting antibody and react with the substrate tetramethylbenzidine (TMB).
For PRDX6, rabbit antihuman polyclonal antibody (PRDX6, ab16824; Abcam, Cambridge, MA, USA) was used as the capture antibody. When the antigen was captured, mouse monoclonal antihuman antibody (PRDX6 ab16947; Abcam) was used as the detecting antibody and then horseradish peroxidase–conjugated rabbit antimouse antibody was used to link the detecting antibody and react with the substrate TMB.
ELISA analysis. Briefly, each well of the microplate was coated with 100 ng (100 μL) of individual antibody or 100 μL coating buffer (as a background) (carbonate buffer, 50 mm sodium carbonate [pH 9.6]) overnight at 4°C, followed by blocking with PBS containing 2% bovine serum albumin for 2 h at room temperature, and then the serum samples (100 μL, 1:80) were added to the coated wells and incubated at 37°C for 2 h. The microplates were washed and incubated with detectable antibody against TIM (dilution, 1:400) and PRDX6 (dilution, 1:2000) for 2 h at 37°C. The microplates were washed and incubated with horseradish peroxidase–conjugated antigoat IgG antibody (for TIM, 1:2000 dilution; Amersham Biosciences, Piscataway, NJ, USA) and horseradish peroxidase–conjugated antimouse IgG antibody (for PRDX6, 1:2000 dilution; Amersham Biosciences) for 1 h at 37°C. After washing, TMB one solution (Promega, Madison, WI, USA) was added to each well. The reaction was stopped by 2 m H2SO4 and the optical density (OD) value was measured at 450 nm. All samples were assayed in duplicate and the results were the mean values of the reading. For each serum sample, the OD value of the background was subtracted from that of the well coated with specific antibody. All analyses were assayed in two parallels.
Statistical analyses. All analyses were run using SPSS 11.0 (SPSS, Chicago, IL, USA). A nonparametric test (Mann–Whitney U‐test) was employed to compare the ELISA results from different groups, including lung squamous cell carcinoma versus normal, lung squamous cell carcinoma versus other types of carcinoma, lung squamous cell carcinoma versus lung adenocarcinoma, and lung squamous cell carcinoma versus lung benign disease. Spearson Rho correlation analysis was applied to find the correlation of TIM and PRDX6 with clinical stages and the differentiation of lung squamous cell carcinoma. The Pearson correlation of TIM and PRDX6 in individual lung squamous cell carcinoma patients was also evaluated. Two‐tailed P‐values of 0.05 or less were considered significant. ROC analysis was used to detect the optimal cut‐off points (i.e. those with the highest total accuracy) for separating lung squamous cell carcinoma from healthy controls. Discriminant analysis was also applied to differentiate lung squamous cell carcinoma patients from healthy controls using TIM and PRDX6. To compare their power of classification, the sensitivity and specificity was given with each analysis.
Specificity evaluation of TIM and PRDX6 for lung squamous cell carcinoma and other tumors. As higher expression of TIM is reported in colon cancer, while higher expression of PRDX6 is reported in esophageal cancer and lung adenocarcinoma,( 4 , 5 , 6 ) we selected colon cancer patients as the control serum of patients with other cancers for the specificity evaluation of TIM, and esophageal cancer patients and lung adenocarcinoma patients for the specificity evaluation of PRDX6. An independent samples test and ROC analysis were used. Two‐tailed P‐values of 0.05 or less were considered significant.
Results
Expression of PRDX6 in lung squamous cell carcinoma patient sera. The secretion character of PRDX6 has been previously reported,( 7 ) and our studies also confirmed its secretion in the human lung squamous cell carcinoma cell line HTB‐182. To exclude the possible proteins released from dead cells, cytoskeleton protein β‐actin was used as an internal control. PRDX6 was detected in both the whole‐cell extracts and culture supernatant of HTB 182 cells, whereas β‐actin was only detected in the whole‐cell extracts (Fig. 1). Next, we sought to investigate the expression of PRDX6 in patient sera. Thirty lung squamous cell carcinoma patient sera and 30 normal control sera were immunoblotted with anti‐PRDX6 antibody. Results showed that PRDX6 was detected in sera of all the patients and their paired controls, but was higher in 70% (21/30) of the patients than in their paired controls (Fig. 2).
Figure 1.
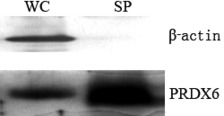
Western blot analysis showing the presence of peroxiredoxin 6 (PRDX6) in the culture supernatant of HTB‐182 cells. Proteins from either whole‐cell extracts (WC) or culture supernatant (SP) were immunodetected for PRDX6 and β‐actin.
Figure 2.
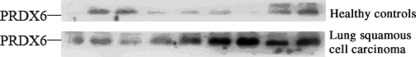
A representative result from Western blot analysis, showing the presence of peroxiredoxin 6 (PRDX6) in the sera of lung squamous cancer patients and their paired healthy controls. Sera (50 μg/well) from patients with lung squamous cell carcinoma or from matched healthy controls were immunodetected with anti‐PRDX6 antibody.
TIM in sera was increased in lung squamous cell carcinoma. To confirm whether the elevation of TIM and PRDX6 was specific in lung squamous cell carcinoma, ELISA assay was used for further validation in 174 serum samples. All values are presented as mean ± SD (Table 2). It was found that TIM in lung squamous cell carcinoma patients (mean, 0.253; range, 0.166–0.410) was significantly higher than that in healthy controls (mean, 0.199; range 0.078–0.378; P < 0.001), other types of carcinoma (mean, 0.219; range, 0.103–0.300; P < 0.05), lung adenocarcinoma patients (mean, 0.205; range, 0.082–0.307; P < 0.01), and lung benign disease patients (mean, 0.177; range, 0.132–0.237; P < 0.001).
Table 2.
Levels of TIM and PRDX6 (mean ± SD) in various groups
| Groups | OD value | |
|---|---|---|
| TIM | PRDX6 | |
| Lung squamous cell carcinoma | 0.253 ± 0.057 | 0.157 ± 0.011 |
| Normal controls | 0.199 ± 0.046 | 0.150 ± 0.006 |
| Other types of cancer | 0.219 ± 0.054 | 0.143 ± 0.102 |
| Nasopharyngeal carcinoma | 0.139 ± 0.026 | 0.147 ± 0.006 |
| Esophageal carcinoma | 0.221 ± 0.029 | 0.141 ± 0.012 |
| Breast cancer | 0.154 ± 0.039 | 0.148 ± 0.006 |
| Gastric cancer | 0.177 ± 0.016 | 0.146 ± 0.006 |
| Colon cancer | 0.275 ± 0.016 | 0.140 ± 0.011 |
| Lung adenocarcinoma | 0.205 ± 0.064 | 0.154 ± 0.008 |
| Benign lung disease | 0.177 ± 0.028 | 0.142 ± 0.011 |
OD, optical density; PRDX6, peroxiredoxin 6; TIM, triosephosphate isomerase.
PRDX6 in sera was increased in lung squamous cell carcinoma. PRDX6 in lung squamous cell carcinoma patients (mean, 0.157; range, 0.142–0.205) was found to be significantly higher than that in healthy controls (mean, 0.150; range, 0.136–0.167; P < 0.001), other types of carcinoma (mean, 0.143; range, 0.111–0.163; P < 0.001), and patients with lung benign disease (mean, 0.142; range, 0.127–0.160; P < 0.001). But there was no significant difference of PRDX6 between lung squamous cell carcinoma patients and lung adenocarcinoma patients (mean, 0.154; range, 0.138–0.172; P > 0.05).
The statistics collected from lung squamous cell carcinoma patients, other types of carcinoma patients, healthy controls, and benign lung disease patients are shown in Table 2 and 3, 4.
Figure 3.
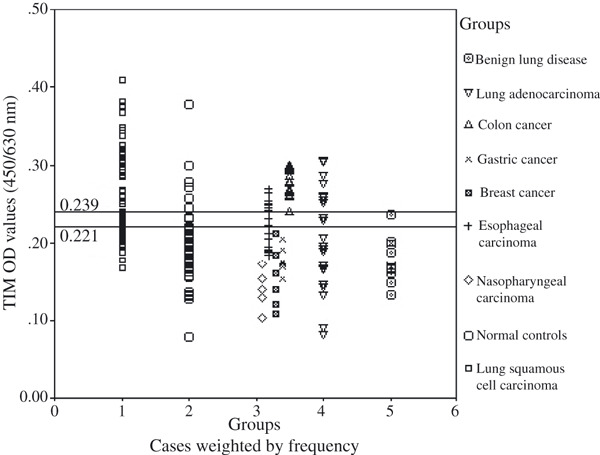
Distribution of the triosephosphate isomerase (TIM) optical density (OD) (y‐axis) in lung squamous cell carcinoma patients, normal controls, other types of carcinoma patients, lung adenocarcinoma patients, and benign lung disease patients.
Figure 4.
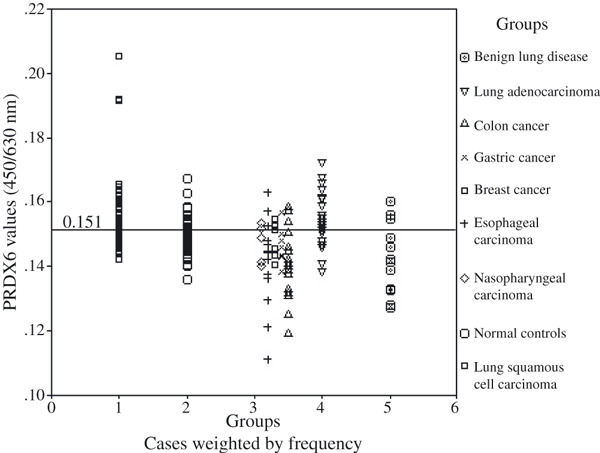
Distribution of the peroxiredoxin 6 (PRDX6) optical density (OD) values (y‐axis) in lung squamous cell carcinoma patients, normal controls, other types of carcinoma patients, lung adenocarcinoma patients, and benign lung disease patients.
In addition, Pearson correlation analysis was applied to find the correlation of TIM and PRDX6 with TNM stages and the differentiation of lung squamous cell carcinoma. Statistic analysis showed that lung squamous cell carcinoma patients with distant metastasis (M1) had a higher level of serum TIM compared with lung squamous cell carcinoma patients with M0, with the Pearson correlation coefficient 0.403 (P = 0.018; P < 0.05). However, no significant correlation was found between TIM or PRDX6 with differentiation of lung squamous cell carcinoma, and there was no significant correlation between TIM and PRDX6 in individual lung squamous cell carcinoma patients (data not shown).
ROC curves of TIM and PRDX6. Although the preliminary results showed that the levels of TIM and PRDX6 were elevated in the majority of lung squamous cell carcinoma patients, there was considerable overlap between lung squamous cell carcinoma and other control groups, especially the healthy controls. Therefore, ROC analyses were used to detect the optimal cut‐off points of TIM and PRDX6 for better discrimination between lung squamous cell carcinoma and healthy individuals. At an optimal cut‐off point of 0.221 (OD value) for TIM levels in serum samples, 40 patients in the lung squamous cell carcinoma group were above this cut‐off and 21 were below it. In the healthy controls group, nine patients were above this cut‐off and 50 were below it. The sensitivity of this cut‐off point was 65.6%, the specificity 84.7%, and total accuracy 75%. The area under the curve was 0.790 (Fig. 5). For PRDX6 levels, at an optimal cut‐off point of 0.151 (OD value), 43 patients in the lung squamous cell carcinoma group were above this cut‐off and 18 were below it. In the healthy controls group, 22 patients were above this cut‐off and 37 were below it. The sensitivity of this cut‐off point was 70.5%, the specificity 62.7%, and total accuracy 66.7%. The area under the curve was 0.721 (Fig. 5). Furthermore, from the figures, we could find few individuals in the other control groups who were above the cut‐off value, with the exception of the lung adenocarcinoma group in the condition of PRDX6. The results suggested that lung squamous cell carcinoma patients could be comparatively discriminated from other control groups at the optimal cut‐off points for TIM or PRDX6 and both TIM and PRDX6 together.
Figure 5.
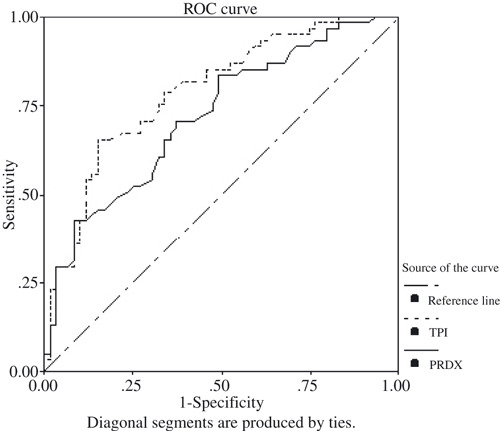
Receiver operating characteristic (ROC) analysis curves for the optimal cut‐off points of triosephosphate isomerase (TIM) (optical density [OD] value, 0.221) and peroxiredoxin 6 (PRDX6) (OD value, 0.151) for discriminating between lung squamous cell carcinoma group and healthy controls. Area under the curves (AUCs) are 0.790 (0.790 ± 0.0041) and 0.721 (0.721 ± 0.046), respectively.
Discriminant analysis with the two variables. Given the higher levels of TIM and PRDX6 in lung squamous cell carcinoma than those in the healthy controls, discriminant analysis using the two variables together was applied to discriminate between lung squamous cell carcinoma and healthy controls.
From the ROC analysis above, we also evaluated sensitivity and specificity when either TIM > 0.221 or PRDX6 > 0.151 were classified as lung squamous cell carcinoma, and when both of the variables above the cut‐off values were classified as lung squamous cell carcinoma. Results showed that with either of the variables, the sensitivity was 90.8 (56/61), the specificity 59.3 (29/59), and the accuracy 70.8%, and with both of the variables above the cut‐off point, the sensitivity was 44.3% (27/61), the specificity 98.5% (58/59), and the accuracy 70.8%. The results showed that with increasing sensitivity, there decreasing specificity, and vice versa.
Through discriminant analysis, the canonical discriminant function coefficient was found to be D = 15.562* TIM + 66.250*PRDX6 − 13.692. With the function, each case could be classified as lung squamous cell carcinoma with D > 0 and as be healthy control with D < 0. As a result, 42/61 (sensitivity, 68.9%) of the lung squamous cell carcinoma patients and 49/59 (specificity, 83.1%) of the healthy controls were correctly classified, and the total accuracy was 75.8%. In addition, the results of cross‐validation showed classification results as being the same.
This indicated that with the two variables together, discriminant analysis may increase the classification power with an increase of the Youden index, compared with other conditions (Table 3).
Table 3.
Sensitivity and specificity of TIM and PRDX6 discriminating between lung squamous cell carcinoma and healthy controls under different conditions
| Standard | Sensitivity (%) | Specificity (%) | Youden index (Sensitivity + specificity − 1) (%) |
|---|---|---|---|
| TIM† | 65.6 | 84.7 | 50.3 |
| PRDX‡ | 70.5 | 62.7 | 33.2 |
| Either§ | 91.8 | 49.1 | 48.4 |
| Both¶ | 44.3 | 98.5 | 42.8 |
| Discrimiant analysis†† | 68.9 | 83.1 | 52.0 |
(†–††) indicate different conditions for classification. (†) indicates use of the cut‐off point of TIM only. (‡) the cut‐off point of PRDX only. (§) either TIM or PRDX above their cut‐off points and (¶) both TIM and PRDX above their cut‐off points classified as lung squamous cell carcinoma. (††) indicates use of TIM and PRDX6 together in discriminant analysis.
Specificity evaluation of TIM and PRDX6 for lung squamous cell carcinoma and other tumors. As shown above, there was no significant difference in PRDX6 between lung squamous cell carcinoma patients and lung adenocarcinoma patients. We also compared PRDX6 levels between lung squamous cell carcinoma patients and esophageal carcinoma patients, as well as TIM levels between lung squamous cell carcinoma patients and colon cancer patients. PRDX6 was found to be significantly higher in lung squamous cell carcinoma patients (mean, 0.157; range, 0.142–0.205) than in esophageal carcinoma patients (mean, 0.141; range, 0.111–0.163; P < 0.01). For the specificity evaluation of TIM, it was found that TIM was significantly higher in colon cancer patients (mean, 0.275; range, 0.240–0.300; P = 0.01 < 0.05) than in lung squamous cell carcinoma patients (mean, 0.253, range, 0.166–0.410). From Figure 3 we could identify TIM OD values of all of the 18 colon patients above the cut‐off point of 0.221. To discriminate between colon cancer patients and lung squamous cell carcinoma patients, a cut‐off point of 0.239 (above the cut‐off point, colon cancer classified; under the cut‐off point, lung squamous cell carcinoma classified) was found by ROC analysis, with the area under the curve 0.698 (Fig. 6).
Figure 6.
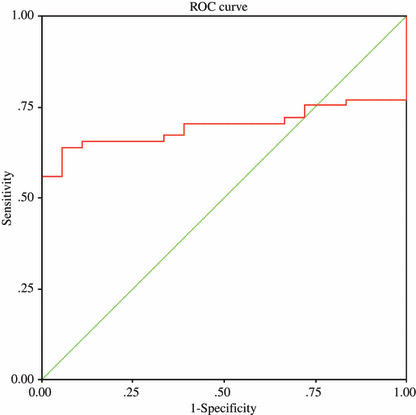
Receiver operating characteristic (ROC) analysis curve for the optimal cut‐off point of triosephosphate isomerase (TIM) (optical density [OD] value, 0.239) for discriminating between lung squamous cell carcinoma group and colon cancer. Area under the curve (AUC) is 0.698 (0.698 ± 0.056).
Discussion
In the present report we show elevated expression of TIM and PRDX6 in lung squamous cell carcinoma when compared with healthy controls. ROC curve analysis was applied to find the optical cut‐off value of serum TIM and PRDX6 to discriminate between lung squamous cell carcinoma and healthy controls. At the same time, the sensitivity and specificity were evaluated. As ROC curve analysis showed, when TIM was used at an optical cut‐off point of 0.221, 65.6.2% (sensitivity, 40/61) of lung squamous cell carcinoma patients and 84.7% (specificity, 50/59) of healthy controls were properly classified, with an accuracy of 75%. With regard to PRDX6, 70.5% (sensitivity, 43/61) of lung squamous cell carcinoma patients and 62.7% (specificity, 37/59) of healthy controls were correctly classified, with an accuracy of 66.7%. Although both could help in discriminating between lung squamous cell carcinoma and healthy controls, the data showed that TIM could give a higher specificity and total accuracy. With the variables together, we evaluated the discriminating power under different conditions. The results showed us we could use different standards for different purposes. We could use either TIM or PRDX6 to increase sensitivity and we should use both TIM and PRDX6 to increase specificity, and to increase the sum of sensitivity and specificity, the last condition (discriminant analysis) would be the best choice.
TIM is a ubiquitously expressed enzyme that catalyses interconversion between dihydroxyacetone phosphate and glycerol dehyde‐3‐phosphate in the energy metabolism pathway. It is ubiquitously distributed in the cytoplasm of all tissues, and generally, with high levels in tissues with high glycolytic activity. Previous reports have shown its overexpression in lung adenocarcinoma and squamous cell carcinoma of the bladder,( 8 , 9 ) our previous results also found up‐regulation of TIM expression in lung squamous cell carcinoma and its secretion in the culture medium of a cultured lung squamous cell carcinoma cell line.( 3 ) It was reported that higher expression of TIM in at the gene level was found in a high‐metastatic mouse melanoma cell line; and at the protein level, higher expression of TIM was reported in a colon cancer cell line with higher metastatic potentials, which indicated some association of TIM with tumor metastasis.( 4 , 10 ) In our previous study, aside from finding anti‐TIM autoantibody and the antigen itself in lung squamous cell carcinoma patients, we also found up‐regulation of TIM expression in lung squamous cell carcinoma and its secretion in the culture medium of a cultured lung squamous cell carcinoma cell line.( 3 ) In the present study, it was confirmed that there was higher serum TIM in lung squamous cell carcinoma patients than in the healthy control group. Moreover, it was interesting for us to note the positive correlation of TIM and lung squamous cell carcinoma distant metastasis, indicating the utility of TIM for lung squamous carcinoma detection.
PRDX6 is a member of the Prxs, which have received considerable attention in recent years as a new and expanding family of thiol‐specific antioxidant proteins.( 11 ) Prxs are ubiquitous enzymes, comprising 0.1% to 0.8% of the soluble proteins in many mammalian cells, and are classified as either 1‐Cys or 2‐Cys Prxs based on whether the protein contains one or two conserved cysteine residues.( 11 ) In mammalian cells, six members of the Prx family have been described.
PRDX6 is a bifunctional 25‐kDa protein with both glutathinone peroxidase and phospholipase A2 activities. The peroxidase activity is about 50‐fold greater than the phospholipase activity. A major difference is the pH optimum for the two activities; PLA2 activity is maximal at pH 4, compatible with a lysosomal localization, whereas peroxidase activity is maximal above pH 7, compatible with function in the cytosol.( 12 ) PRDX6 is the only mammalian 1‐Cys member of the peroxiredoxin superfamily and is expressed in all major organs, with a particularly high level in the lung. Numerous studies have provided evidence that PRDX6 can function in vivo as an antioxidant enzyme. Its overexpression could protect H441 cells against peroxide or hydroxyl‐mediated oxidative stress, and overexpressing cells showed less lipid peroxidation and decreased damage to cellular plasma membranes.( 13 ) Overexpression of PRDX6 through adenoviral‐mediated transfer of the PRDX6 gene to the lungs of intact mice could protect them against hyperoxic injury.( 14 ) There have been numerous reports documenting a link between PRDX6 and certain diseases, although for most the mechanism for the linkage is not known to be related to either the peroxidase or the phospholipase activities of the protein. Its elevated expression was found in lungs with malignant mesothelioma( 15 ) or high‐grade squamous cell carcinoma.( 16 ) Previous studies reported that autoantibodies against PRDX6 were present in patients with systemic autoimmune diseases.( 17 ) As its autoantibody has also been detected in esophageal squamous cell carcinoma and in lung adenocarcinoma,( 5 , 6 ) we detected autoantibody against PRDX6 in the sera of lung squamous cell carcinoma (30%) patients but not in the normal controls in our previous study. In this study, our results showed higher levels of PRDX6 in the sera of lung squamous cell carcinoma patients compared with the normal controls, and ROC analysis results showed that PRDX6 may be a biomarker of lung squamous cell carcinoma. However, considering that there was no significant difference in PRDX6 between lung squamous cell carcinoma and lung adenocarcinoma, it needs further investigation.
ROC analysis is very useful in comparing the discriminant power of different markers for certain diseases and the variable with higher area under the curve (AUC) would be more powerful in differentiating. Through the analysis, an optical cut‐off value can be found to discriminate different conditions, which has also been used in many studies on lung tumors and other lung diseases.( 18 , 19 , 20 , 21 ) Discriminant analysis is a mathematical method, which may be applied to a set of markers and which may improve the classification power of any single variable, increasing the final discrimination rate.( 22 , 23 , 24 , 25 ) In our study, we found higher serum levels of TIM and PRDX6 in lung squamous cell carcinoma than in other control groups with the exception of TIM in patients with lung adenocarcinoma, indicating the potential of TIM and PRDX6 as biomarkers. Further, through ROC analysis and discriminant analysis, we found that both TIM and PRDX6 as the only variable or working together could differentiate between most of the lung squamous cell carcinoma patients and the healthy controls.
As our results showed that a much higher TIM level was found in colon cancer patients than in lung squamous cell carcinoma patients, and that no significant difference in PRDX6 was found between lung squamous cell carcinoma patients and lung adenocarcinoma patients, TIM and PRDX6 together may increase their efficiency as biomarkers. Furthermore, the results of the discriminate analysis indicated the same.
The results showed that TIM and PRDX6 could be explored more as biomarkers for lung squamous cell carcinoma and may be utilized in its screening and diagnosis. To our knowledge, this is the first time that a close relationship has been revealed between higher TIM and PRDX6 serum levels and lung squamous cell carcinoma. Still, further studies on a larger scale are needed to confirm the results.
Acknowledgment
This work was supported by grants from the National Natural Sciences Foundation of China (nos. 30500558, 30670990) and the Program for New Century Excellent Talents in University (NCET) (2007‐70).
References
- 1. Stieber P, Aronsson AC, Bialk P et al. Tumor markers in lung cancer: EGTM recommendations. Anticancer Res 1999; 19: 2817–9. [PubMed] [Google Scholar]
- 2. Alaiya AA, Franzen B, Auer G, Linder S. Cancer proteomics: from identification of novel markers to creation of artificial learning models for tumor classification. Electrophoresis 2000; 21: 1210–7. [DOI] [PubMed] [Google Scholar]
- 3. Yang F, Xiao ZQ, Zhang XZ et al. Identification of tumor antigens in human lung squamous cell carcinoma by serological proteome analysis. J Proteome Res 2007; 6 (2): 751–8. [DOI] [PubMed] [Google Scholar]
- 4. Katayama M, Nakano H, Ishiuchi A et al. Protein pattern difference in the colon cancer cell lines examined by two‐dimensional differential in‐gel electrophoresis and mass spectrometry. Surg Today 2006; 36: 1085–93. [DOI] [PubMed] [Google Scholar]
- 5. Fujita Y, Nakanishi T, Hiramatsu M et al. Proteomics‐based approach identifying autoantibody against peroxiredoxin VI as a novel serum marker in esophageal squamous cell carcinoma. Clin Cancer Res 2006; 12 (21): 6415–20. [DOI] [PubMed] [Google Scholar]
- 6. Nakanishi T, Takeuchi T, Ueda K, Murao H, Shimizu A. Detection of eight antibodies in cancer patients’ sera against proteins derived from the adenocarcinoma A549 cell line using proteomics‐based analysis. J Chromatogr B Analyt Technol Biomed Life Sci 2006; 838: 15–20. [DOI] [PubMed] [Google Scholar]
- 7. Okado‐Matsumoto A, Matsumoto A, Fujii J, Taniguchi N. Peroxiredoxin IV is a secretable protein with heparin‐binding properties under reduced conditions. J Biochem 2000; 127: 493–501. [DOI] [PubMed] [Google Scholar]
- 8. Chen G, Gharib TG, Huang CC et al. Proteomic analysis of lung adenocarcinoma: identification of a highly expressed set of proteins in tumors. Clin Cancer Res 2002; 8: 2298–305. [PubMed] [Google Scholar]
- 9. Montgomerie JZ, Gracy RW, Holshuh HJ, Keyser AJ, Bennett CJ, Schick DG. The 28K protein in urinary bladder, squamous metaplasia and urine is triosephosphate isomerase. Clin Biochem 1997; 30: 613–8. [DOI] [PubMed] [Google Scholar]
- 10. Ishiguro T, Nakajima M, Naito M, Muto T, Tsuruo T. Identification of genes differentially expressed in B16 murine melanoma sublines with different metastatic potentials. Cancer Res 1996; 56 (4): 875–9. [PubMed] [Google Scholar]
- 11. Wood ZA, Schroder E, Robin Harris J, Poole LB. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem Sci 2003; 28: 32–40. [DOI] [PubMed] [Google Scholar]
- 12. Chen JW, Dodia C, Feinstein ST, Jain MK, Fisher AB. 1‐Cys Peroxiredoxin, a bifunctional enzyme with glutathione peroxidase and phospholipase A2 activities. J Biol Chem 2000; 275: 28421–7. [DOI] [PubMed] [Google Scholar]
- 13. Manevich Y, Sweitzer T, Pak JH, Feinstein SI, Muzykantov V, Fisher AB. 1‐Cys peroxiredoxin overexpression protects cells against phospholipid peroxidation‐mediated membrane damage. Proc Natl Acad Sci USA 2002; 99: 11599–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wang Y, Manevich Y, Feinstein SI, Fisher AB. Adenovirus‐mediated transfer of the 1‐cys peroxiredoxin gene to mouse lung protects against hyperoxic injury. Am J Physiol Lung Cell Mol Physiol 2004; 286: L1188–93. [DOI] [PubMed] [Google Scholar]
- 15. Kinnula VL, Lehtonen S, Sormunen R et al. Overexpression of peroxiredoxins I, II, III, V, and VI in malignant mesothelioma. J Pathol 2002; 196: 316–23. [DOI] [PubMed] [Google Scholar]
- 16. Lehtonen ST, Svensk AM, Soini Y et al. Peroxiredoxins,a novel protein family in lung cancer. Int J Cancer 2004; 111: 514–21. [DOI] [PubMed] [Google Scholar]
- 17. Karasawa R, Ozaki S, Nishioka K, Kato T. Autoantibodies to peroxiredoxin I and IV in patients with systemic autoimmune diseases. Microbiol Immunol 2005; 49: 57–65. [DOI] [PubMed] [Google Scholar]
- 18. Zhong L, Coe SP, Stromberg AJ, Khattar NH, Jett JR, Hirschowitz EA. Profiling tumor‐associated antibodies for early detection of non‐small cell lung cancer. J Thorac Oncol 2006; 1: 513–9. [PubMed] [Google Scholar]
- 19. Chen Y, Zhang H, Xu AJ et al. Elevation of serum l‐lactate dehydrogenase B correlated with the clinical stage of lung cancer. Lung Cancer 2006; 54: 95–102. [DOI] [PubMed] [Google Scholar]
- 20. Sheu CC, Yu YP, Tsai JR et al. Development of a membrane array‐based multimarker assay for detection of circulating cancer cells in patients with non small cell lung cancer. Int J Cancer 2006; 119 (6): 1419–26. [DOI] [PubMed] [Google Scholar]
- 21. Satoh H, Kurishima K, Ishikawa H, Ohtsuka M. Increased levels of KL‐6 and subsequent mortality in patients with interstitial lung diseases. J Intern Med 2006; 260: 429–34. [DOI] [PubMed] [Google Scholar]
- 22. Ameglio F, Abbolito MR, Giannarelli D et al. Detection of Helicobacter pylori carriers by discriminate analysis of urea and pH levels in gastric juices. J Clin Pathol 1991; 44: 689–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Merucci P, Taggi F, Marolla A et al. Discriminant analysis of Hodgkin’s lymphomas by age and serum proteins. Eur J Cancer Clin Oncol 1984; 20: 1243–7. [DOI] [PubMed] [Google Scholar]
- 24. Shinozaki T, Chigira MK. Mutivariate analysis of serum tumor markers for diagnosis of skeletal metastases. Cancer 1992; 69: 108–12. [DOI] [PubMed] [Google Scholar]
- 25. Ameglio F, Giannarelli D, Cordiali‐Fei P et al. Use of discriminant analysis to assess disease activity in pulmonary tuberculosis with a panel of specific and non‐specific serum markers. Am J Clin Pathol 1994; 101: 719–25. [DOI] [PubMed] [Google Scholar]


