Abstract
Objectives
To investigate the influence of preoperative breast MRI on mastectomy and reoperation rates in patients with pure ductal carcinoma in situ (DCIS).
Methods
The MIPA observational study database (7245 patients) was searched for patients aged 18–80 years with pure unilateral DCIS diagnosed at core needle or vacuum-assisted biopsy (CNB/VAB) and planned for primary surgery. Patients who underwent preoperative MRI (MRI group) were matched (1:1) to those who did not receive MRI (noMRI group) according to 8 confounding covariates that drive referral to MRI (age; hormonal status; familial risk; posterior-to-nipple diameter; BI-RADS category; lesion diameter; lesion presentation; surgical planning at conventional imaging). Surgical outcomes were compared between the matched groups with nonparametric statistics after calculating odds ratios (ORs).
Results
Of 1005 women with pure unilateral DCIS at CNB/VAB (507 MRI group, 498 noMRI group), 309 remained in each group after matching. First-line mastectomy rate in the MRI group was 20.1% (62/309 patients, OR 2.03) compared to 11.0% in the noMRI group (34/309 patients, p = 0.003). The reoperation rate was 10.0% in the MRI group (31/309, OR for reoperation 0.40) and 22.0% in the noMRI group (68/309, p < 0.001), with a 2.53 OR of avoiding reoperation in the MRI group. The overall mastectomy rate was 23.3% in the MRI group (72/309, OR 1.40) and 17.8% in the noMRI group (55/309, p = 0.111).
Conclusions
Compared to those going directly to surgery, patients with pure DCIS at CNB/VAB who underwent preoperative MRI had a higher OR for first-line mastectomy but a substantially lower OR for reoperation.
Clinical relevance statement
When confounding factors behind MRI referral are accounted for in the comparison of patients with CNB/VAB-diagnosed pure unilateral DCIS, preoperative MRI yields a reduction of reoperations that is more than twice as high as the increase in overall mastectomies.
Key Points
• Confounding factors cause imbalance when investigating the influence of preoperative MRI on surgical outcomes of pure DCIS.
• When patient matching is applied to women with pure unilateral DCIS, reoperation rates are significantly reduced in women who underwent preoperative MRI.
• The reduction of reoperations brought about by preoperative MRI is more than double the increase in overall mastectomies.
Keywords: Breast neoplasms (biopsy, needle); Carcinoma (intraductal, noninfiltrating); Magnetic resonance imaging; Mastectomy; Reoperation
Introduction
The role of preoperative breast magnetic resonance imaging (MRI) in guiding the treatment of ductal carcinoma in situ (DCIS) diagnosed at core needle biopsy (CNB) or vacuum-assisted biopsy (VAB) is an open issue in clinical practice [1–4] that has been extensively investigated considering different outcomes, e.g., short-term surgical results [5–16], upgrade to invasive cancer at final pathology [17, 18], detection of additional ipsilateral or contralateral disease [19], long-term recurrence [20], and patient preferences [21, 22]. Surgical outcomes are crucial, as the rate of mastectomy and the rate of reoperation after breast-conserving surgery are major indicators of breast care quality and the focus of multidisciplinary efforts towards surgical de-escalation [23–25].
As shown by three systematic reviews published between 2015 and 2021 [26–28], cohort studies that investigated the effects of preoperative MRI on surgical outcomes of DCIS generally exhibit a referral bias towards MRI for young patients with extensive and high-grade DCIS. These characteristics represent strong confounding factors, being intrinsically associated with poor outcomes and ultimately prompting more aggressive surgery [27, 29]. Naturally, these studies outline contrasting scenarios and add to ongoing uncertainty about the impact of preoperative MRI on surgical outcomes [27, 28].
However, different results emerge from the very few studies adopting methods to minimize or remove the referral bias [30]. Save for the very small subgroups of DCIS in the MONET randomized controlled trial [31], as of mid-2023, only two studies present such characteristics: the IRCIS randomized controlled trial [11] and the propensity score matching study by Yoon et al [15]. Pooled together, data from these two studies show that, compared to patients not referred to preoperative MRI, patients with DCIS undergoing preoperative MRI have a 1.52 odds ratio (OR) of undergoing mastectomy as their initial surgery, a 1.89 OR of having negative margins and avoiding reoperation, and a 0.97 OR of overall mastectomy (at first-line surgery or at reoperation). Nonetheless, as noted in the conclusions of a systematic review by the European Commission Initiative on Breast Cancer working groups [27], estimates from these studies remain affected by low statistical power.
In this context, the present study takes advantage of the large cohort of patients with DCIS registered in the database of the Multicenter International Prospective Analysis (MIPA) study [32–34], which allows the application of methods for confounder adjustment while still retaining a large number of patients. Thus, we aimed to compare in matched cohorts the surgical outcomes of patients with DCIS referred or not referred to preoperative MRI, namely the first-line mastectomy rate, the reoperation rate, and the overall mastectomy rate.
Materials and methods
Study design
This is a subgroup analysis of data from the MIPA study (ISRCTN41143178), whose design is detailed in the protocol paper [32]. In summary, MIPA is a prospective observational study conducted in 27 centers worldwide between June 2013 and November 2018, after approval from the Ethics Committee of the coordinating center (Comitato Etico Ospedale San Raffaele, Milano, Italy; protocol number 2784). Each center consecutively enrolled women aged 18–80 years with newly diagnosed breast cancer, without indication for neoadjuvant therapy and amenable to upfront surgery. In accordance with the observational nature of the study, each multidisciplinary team followed local routine practice in the diagnostic and therapeutic pathway, including the decision on whether to refer patients to bilateral contrast-enhanced preoperative MRI after conventional imaging with digital mammography and/or ultrasonography (US).
Study population and endpoints
According to the aforementioned aims, this study focuses on patients with a diagnosis of pure unilateral DCIS at CNB/VAB, as the presence of ipsilateral and/or contralateral invasive cancer and the presence of bilateral DCIS are known to influence surgical management [35, 36], acting as strong potential confounding factors.
Following the study protocol [32], surgical endpoints for all patients in this analysis are (i) first-line mastectomy; (ii) immediate/short-term reoperation for close or positive margins; and (iii) overall mastectomy (i.e., performance of mastectomy as first-line surgery or at reoperation). Due to the focus on unilateral lesions, the secondary endpoint of first-line bilateral mastectomy is excluded from this analysis.
Conversely, data on non-surgical secondary study endpoints based on surgical pathology (such as the upgrade of DCIS to invasive cancer and complete DCIS removal) will not be considered in this analysis and will be separately reported for all patients with pure DCIS at CNB/VAB.
Patient matching
The nonrandomized observational design of the MIPA study implies the existence of different selection biases towards the referral to MRI, yielding a skewed distribution of several characteristics (i.e., covariates) between patients who underwent MRI before surgery (MRI group) and those who did not undergo MRI (noMRI group). Thus, patient matching was implemented to reduce covariate imbalance, to estimate the average effect of preoperative MRI on surgical outcomes in the population at clinical equipoise, i.e., patients with overlapping baseline characteristics [37, 38].
To strictly adhere to the real-world workflow of breast cancer care, the following baseline characteristics—all already available before the decision to refer (or not) patients to MRI—were considered covariates for patient matching (Table 1): age; hormonal status; presence of familial breast cancer risk1; posterior-to-nipple diameter2; highest BI-RADS category at conventional imaging; lesion diameter at conventional imaging; lesion presentation at conventional imaging3; surgical planning after conventional imaging.
Table 1.
Comparison of baseline demographic, clinical, and imaging characteristics in the unmatched and matched cohorts
| Unmatched cohorts | Matched cohorts | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| noMRI | MRI | p | SMD | noMRI | MRI | p | SMD | ||
| Patients | 498 | 507 | – | – | 309 | 309 | – | – | |
| Mean age (SD) | 59 years (10) | 56 years (10) | < 0.001 | 0.267 | 57 years (10) | 57 years (9) | 0.865 | 0.013 | |
| Hormonal status | Premenopausal | 118 (23.8%) | 140 (27.6%) | 0.229 | 0.132 | 85 (27.5%) | 75 (24.3%) | 0.462 | 0.022 |
| Perimenopausal | 49 (9.9%) | 62 (12.2%) | 32 (10.4%) | 43 (13.9%) | |||||
| Receiving HRT | 3 (0.6%) | 2 (0.4%) | 2 (0.6%) | 1 (0.3%) | |||||
| Post-menopausal | 326 (65.7%) | 303 (59.8%) | 190 (61.5%) | 190 (61.5%) | |||||
| Patients with familial breast cancer risk | 5 (1.0%) | 11 (2.2%) | 0.224 | 0.093 | 1 (0.3%) | 1 (0.3%) | 1.000 | < 0.001 | |
| Mean posterior-to-nipple diameter (SD) | 96.2 mm (31.9) | 89.6 mm (30.3) | 0.001 | 0.213 | 92.2 mm (26.5) | 90.9 mm (27.9) | 0.554 | 0.043 | |
| Highest BI-RADS at conventional imaging | BI-RADS 0 | 5 (1.0%) | 10 (2.0%) | < 0.001 | 0.354 | 1 (0.3%) | 1 (0.3%) | 1.000 | < 0.001 |
| BI-RADS 1 | 0 (0.0%) | 4 (0.8%) | 0 (0.0%) | 0 (0.0%) | |||||
| BI-RADS 2 | 0 (0.0%) | 8 (1.6%) | 0 (0.0%) | 0 (0.0%) | |||||
| BI-RADS 3 | 14 (2.8%) | 33 (6.5%) | 7 (2.3%) | 7 (2.3%) | |||||
| BI-RADS 4 | 363 (72.9%) | 319 (63.0%) | 238 (77.0%) | 238 (77.0%) | |||||
| BI-RADS 5 | 116 (23.3%) | 132 (26.1%) | 63 (20.4%) | 63 (20.4%) | |||||
| Lesion presentation at conventional imaging | Unifocal | 433 (86.9%) | 425 (83.8%) | 0.014 | 0.185 | 286 (92.6%) | 286 (92.6%) | 1.000 | < 0.001 |
| Multifocal | 55 (11.0%) | 54 (10.7%) | 21 (6.8%) | 21 (6.8%) | |||||
| Multicentric | 10 (2.0%) | 28 (5.5%) | 2 (0.6%) | 2 (0.6%) | |||||
| Mean lesion diameter at conventional imaging (SD) | 20.9 mm (20.4) | 24.0 mm (20.3) | 0.022 | 0.151 | 18.0 mm (14.0) | 18.3 mm (13.4) | 0.790 | 0.014 | |
| Planned mastectomy after conventional imaging | 67 (13.5%) | 125 (24.7%) | < 0.001 | 0.288 | 31 (10.0%) | 31 (10.0%) | 1.000 | < 0.001 | |
SMD standardized mean difference, SD standard deviation, HRT hormone replacement therapy
Using the “MatchIt” package [39] on R (version 4.2.1, The R Foundation for Statistical Computing), nearest neighbor 1:1 matching with the rank-based robust Mahalanobis distance [40, 41] was performed with specifications chosen to optimize covariate balance by taking advantage of the large number of patients in the MIPA study: (i) exact 1:1 matching was enforced for the following categorical covariates: familial breast cancer risk, highest BI-RADS at conventional imaging, lesion presentation at conventional imaging, and surgical planning after conventional imaging; (ii) calipers were applied in the matching of the following continuous covariates: age (caliper width: ± 0.5 standard deviations), posterior-to-nipple diameter (caliper width: ± 2 standard deviations), maximum lesion diameter at conventional imaging (caliper width: ± 0.5 standard deviations). Matching was performed without replacement, and unmatched patients were discarded.
Covariate balance between the MRI and noMRI group was assessed before and after matching by calculating the standardized mean difference for all variables and by performing two-tailed Pearson’s χ2 or Fisher’s exact tests for categorical variables and the Mann–Whitney U test for continuous variables. In order to consider matching successful, a conservative combined balance threshold was applied, with all covariates having to display standardized mean differences ≤ 0.050 with p values ≥ 0.100 [42].
Comparison of surgical endpoints
Comparisons of the three surgical endpoints in the unmatched and matched cohorts were carried out with two-tailed Pearson’s χ2 or Fisher’s exact tests, as appropriate, after calculation of the respective ORs. To account for multiple testing, the Bonferroni correction was applied considering the 6 comparisons of surgical endpoints in the unmatched and matched cohorts, with an ensuing p < 0.008 threshold for statistical significance. All analyses were performed with R (version 4.2.1, The R Foundation for Statistical Computing) and STATA (version MP 17.1, StataCorp).
Results
Study population
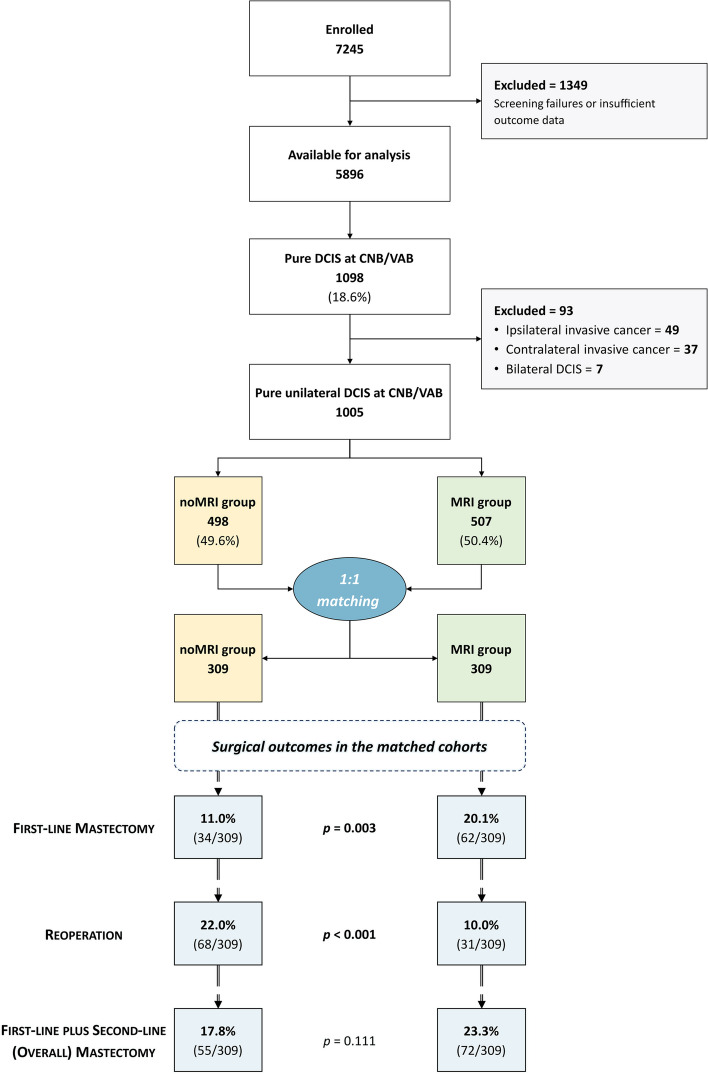
As described in the study flowchart (Fig. 1), 5896 among the 7245 patients enrolled between June 2013 and November 2018 had sufficient information to be considered for this analysis. At least one lesion diagnosed as pure DCIS at CNB/VAB was present in 1098/5896 patients (18.6%): applying the exclusion criteria, 37/1098 (3.4%) patients were excluded because of the presence of a contralateral invasive cancer, 49/1098 (4.5%) because of the presence of another ipsilateral lesion diagnosed as invasive cancer, and 7/1098 (0.6%) because of the presence of bilateral pure DCIS. Thus, 1005 patients with pure unilateral DCIS at CNB/VAB were included in this analysis, 507/1005 (50.4%) in the MRI group and 498/1005 (49.6%) in the noMRI group.
Fig. 1.
Study flowchart and surgical outcomes in the matched cohorts
As detailed in Table 2, tissue sampling was most frequently prompted by mammographic findings (884/1005 patients, 87.9%), either alone (540/1005 patients, 53.7%) or in combination with US or MRI (344/1005 patients, 34.2%). A total of 658/1005 (65.5%) samplings were performed with VAB, while the remaining 347/1005 (34.5%) with CNB: accordingly, stereotaxis was the most frequent biopsy guidance (673/1005 patients, 67.0%), followed by US (310/1005 patients, 30.8%) and MRI (22/1005 patients, 2.2%).
Table 2.
Modality of detection of the findings prompting tissue sampling
| Tissue sampling prompt | Number | % |
|---|---|---|
| Mammography alone | 540/1005 | 53.7% |
| US alone | 93/1005 | 9.3% |
| MRI alone | 20/1005 | 2.0% |
| Mammography + US | 194/1005 | 19.3% |
| Mammography + MRI | 89/1005 | 8.8% |
| US + MRI | 8/1005 | 0.8% |
| Mammography + US + MRI | 61/1005 | 6.1% |
US ultrasonography, MRI magnetic resonance imaging
Unmatched cohorts—baseline characteristics
Before matching, different distributions between the noMRI and the MRI group were observed for six of the eight baseline descriptors (Table 1). Patients in the MRI group were younger than those in the noMRI group (mean age 56 years versus 59 years, p < 0.001) and also differed in DCIS presentation at conventional imaging, displaying larger lesions (mean diameter 24.0 versus 20.9 mm, p = 0.022) that were more frequently multifocal or multicentric (16.2% versus 13.0%, p = 0.014). Finally, patients in the MRI group had a + 11.2% difference in the referral to mastectomy after conventional imaging compared to patients in the noMRI group (125/507 patients in the MRI group, 24.7%, versus 67/498 patients in the noMRI group, 13.5%).
Unmatched cohorts—surgical endpoints
As detailed in Table 3, the + 11.2% difference in the referral to mastectomy in the MRI group after conventional imaging rose to + 19.2% in the evaluation of first-line surgery: specifically, the first-line mastectomy rate was 33.5% in the MRI group (170/507 patients) compared to 14.3% (71/498 patients) in the noMRI group (OR 3.03, p < 0.001). While the 12.4% (63/498 patients) reoperation rate of the MRI group was significantly lower (− 7.7%, OR 0.56, p = 0.001) than the 20.1% reoperation rate of the noMRI group (100/498 patients), the overall rate of mastectomy in the MRI group (190/507 patients, 37.5%) was still 17.2% higher (OR 2.36, p < 0.001) than that of the noMRI group (101/498 patients, 20.3%).
Table 3.
Comparison of surgical outcomes in the unmatched and matched cohorts
| Unmatched cohorts 1005 patients |
Matched cohorts 618 patients |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| noMRI group 498 patients |
MRI group 507 patients |
Difference for the MRI group | noMRI group 309 patients |
MRI group 309 patients |
Difference for the MRI group | |||||
| % | OR | p | % | OR | p | |||||
| First-line mastectomy | 71 (14.3%) | 170 (33.5%) | + 19.2% | 3.03 | < 0.001 | 34 (11.0%) | 62 (20.1%) | + 9.1% | 2.03 | 0.003 |
| Reoperation | 100 (20.1%) | 63 (12.4%) | − 7.7% | 0.56 | 0.001 | 68 (22.0%) | 31 (10.0%) | − 12.0% | 0.40 | < 0.001 |
| Overall mastectomy (first-line + second-line) | 101 (20.3%) | 190 (37.5%) | + 17.2% | 2.36 | < 0.001 | 55 (17.8%) | 72 (23.3%) | + 5.5% | 1.40 | 0.111 |
MRI magnetic resonance imaging, OR odds ratio
Matched cohorts—baseline characteristics
A total of 618 patients were matched, 309 in each group. As detailed in Table 1, matched patients had a median age of 57 years, being mostly post-menopausal (380/618, 61.5%). At conventional imaging, the most frequent BI-RADS classifications were BI-RADS 4 in 476/618 patients (77.0%) and BI-RADS 5 in 126/618 patients (20.4%). The vast majority of matched patients (572/618, 92.6%) had unifocal presentation at conventional imaging, 42/618 (6.8%) having multifocal DCIS, and only 4/618 patients (0.6%) presenting with multicentric DCIS.
Matched cohorts—surgical endpoints
Starting from the matched 10.0% rate of referral to mastectomy at conventional imaging (31/309 patients in both groups; Table 3), the MRI group still had a significantly higher first-line mastectomy rate (20.1%, 62/309 patients, OR 2.03) compared to the noMRI group (11.0%, 34/309 patients, p = 0.003). However, the reoperation rate in the MRI group (10.0%, 31/309 patients, OR for reoperation 0.40) was less than half that of the noMRI group (22.0%, 68/309 patients, p < 0.001), corresponding to a 2.53 OR of avoiding reoperation for women in the MRI group. This resulted in a non-significant difference (p = 0.111) in the overall mastectomy rate for the MRI group (23.3%, 72/309 patients, OR 1.40) compared to the noMRI group (17.8%, 55/309 patients).
Discussion
This subgroup analysis of the MIPA study focused on 1005 patients with pure unilateral DCIS at CNB/VAB who underwent (507 patients, 50.4%) or did not undergo (498 patients, 49.6%) preoperative MRI, evaluating differences in surgical outcomes between the MRI and noMRI groups. After 1:1 patient matching according to eight covariates concerning demographic, clinical, and imaging characteristics, 309 patients were matched in each group; the significantly higher first-line mastectomy rate of the MRI group (20.1% versus 11.0% in the noMRI group) was counterbalanced by an even higher decrease of reoperations (10.0% in the MRI group versus 22.0% in the noMRI group), culminating in a 5.5% increase in the overall mastectomy rate for the MRI group (23.3% vs 17.8% in the noMRI group) that will need to be clinically contextualized with follow-up data.
As already mentioned, the interpretation of our results must consider methodological peculiarities and limitations both of this study and of previous ones. In the unmatched cohorts, data from all three surgical outcomes (first-line mastectomy, reoperation, overall mastectomy) are in line with pooled data of previous cohort studies reported by the working groups of the European Commission Initiative on Breast Cancer [27]: in our study—before patient matching—preoperative MRI led patients with pure DCIS at CNB/VAB to an even lower OR for reoperation (0.56 versus a pooled 0.72) but to higher OR for first-line mastectomy (3.03 versus a pooled 2.04) and overall mastectomy (2.36 versus a pooled 1.58).
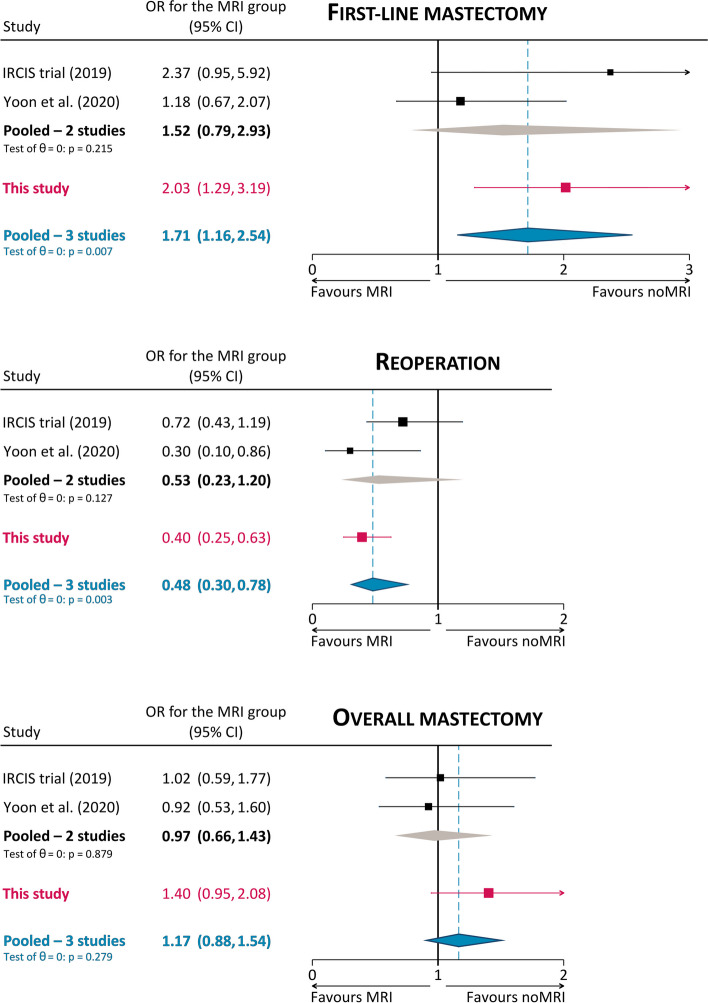
Importantly, in the matched cohort, data from our study confirmed the results obtained by previous studies where randomization [11] or propensity score matching [15] were implemented to deal with confounding factors. As shown in Fig. 2, the 2.03 OR for first-line mastectomy of women in the MRI group was higher than the 1.18 OR obtained by Yoon et al [15] but lower than the 2.37 OR in the IRCIS trial [11]: of note, adding our data to the pooling, the ensuing 1.71 pooled OR reached statistical significance (p = 0.007), substantiating the association between MRI and first-line mastectomy. However, a similar finding could be observed for the protective effect of MRI towards reoperation, underlined by the 0.40 OR in our study, slightly higher than the 0.30 OR obtained by Yoon et al [15] but almost half of the 0.72 OR in the IRCIS trial [11]: adding our data to the pooling confirmed the protective effect of MRI for reoperation, with a significant 0.48 pooled OR (p = 0.003). For the last endpoint (i.e., overall mastectomy), the 1.40 OR of MRI found in our study was higher than the protective 0.92 OR found by Yoon et al [15] and the 1.02 OR found in the IRCIS trial [11]. Nonetheless, as in these two studies, the confidence interval of this OR crosses the no-effect line, not reaching statistical significance (p = 0.111, pooled OR 1.71 with p = 0.279). Notably, our overall mastectomy rates in the matched cohorts (23.3% in the MRI group, 17.8% in the noMRI group) were about half of those found by Yoon et al (38.7% in the MRI group, 40.6% in the noMRI group) [15] and closely comparable to those of the IRCIS trial (17.6% in the MRI group, 17.3% in the noMRI group) [11].
Fig. 2.
Explorative comparison of surgical outcomes with previous controlled analyses. Odds ratios (OR) refer to women with DCIS undergoing preoperative MRI, with the noMRI group as the reference
At an overall appraisal, results from our study emphasize that when CNB/VAB yields a DCIS diagnosis, women undergoing preoperative MRI have a 2.03 OR of receiving mastectomy as their first-line surgery that is counterbalanced by the 2.53 OR of avoiding reoperation. The focus on biopsy data represents a strong point of our study and allows for the translation of these results into clinical practice, as multidisciplinary teams (and surgeons in particular) do not know in advance the final pathology report—where the case under discussion would be confirmed as a pure DCIS or a DCIS associated with an invasive cancer—but base their decision-making on CNB/VAB results and on what is suggested by imaging findings, in particular extent of calcifications on mammograms, hypoechoic findings at US, or enhancement at MRI. Finally, the non-significant 1.40 OR of the MRI group for overall mastectomy—also due to the loss of cases from the matching process—leaves open the question on whether this OR and the absolute percentage differences found for this surgical outcome (5.5% in our study, 1.9% and 0.3% in the other two studies [11, 15]) are clinically relevant (also in a long-term perspective that will be explored with follow-up data) and justify the conduction of studies adequately powered to detect significant differences in this endpoint, as highlighted by a cost-effectiveness analysis conducted on data from the IRCIS trial [43].
The limitations of this work can be ascribed to two macro-areas, i.e., general limitations of the MIPA study itself and limitations specifically pertaining this subgroup analysis. As for the general limitations of the MIPA study, its nonrandomized observational design remains the chief obstacle to a controlled evaluation of surgical outcomes. However, we addressed this issue with the aid of patient matching according to a large number of covariates, also employing conservative matching methods with strict criteria. While this represents a potential solution to avoid some issues of randomized controlled trials such as high costs, statistical power issues, and poor external validity and representativeness of results obtained in highly controlled settings [44, 45], we acknowledge that our analysis could not account for several other patient-specific or institutional potentially confounding factors. Of these, the most difficult to model remains the effect of the surgical habits at each institution and even of each surgeon in a given institution [46–50]. Another general limitation of the MIPA study is its wide enrolment timeframe, during which the quick expansion of the role of digital breast tomosynthesis and of MRI itself in the diagnostic setting could have created hidden imbalances between and inside subgroups.
The main specific limitation of this targeted analysis on needle biopsy–diagnosed DCIS is the fact that the MIPA study database did not collect information on DCIS grade and receptor status at CNB/VAB, acquiring these data only from surgical pathology. Had it been available, DCIS grade at CNB/VAB would have represented a covariate for matching, considering not only its prognostic implications [51] but also its specific influence on the accuracy of MRI [52] and on the interplay between preoperative MRI and surgical outcomes [10, 12, 16]. Again referring to the 2013–2018 enrolment timeframe of the MIPA study, this subgroup analysis could not account for the impact of the progressive clinical introduction of 3-T MRI systems, which are known to improve DCIS differential diagnosis and the accuracy of lesion sizing [53–55], nor for the potential competition in the preoperative setting between MRI and other contrast-enhanced imaging modalities, such as contrast-enhanced mammography [56–58].
In conclusion, this subgroup analysis of the MIPA study showed that, when surgical outcomes of women diagnosed with pure DCIS at CNB/VAB are compared in matched cohorts, the increase in the overall mastectomy rate engendered by preoperative MRI is less than half the corresponding reduction in reoperation rates.
Acknowledgements
The MIPA study was promoted by the European Network for the Assessment of Imaging in Medicine (EuroAIM)—a joint initiative of the European Institute for Biomedical Imaging Research (EIBIR)—and was endorsed by the European Society of Breast Imaging.
The authors thank Bayer AG that provided an unconditional research grant, in particular Dr. Stephanie Schermuck-Joschko (who passed away due to a car accident after the study started) and Dr. Jan Endrikat.
The authors also thank Monika Hierath, Eva Haas, Katharina Krischak, and Peter Gordebeke from the EIBIR staff which managed all the administrative work of this study.
The following persons collaborated at individual centers: Lucia Camera, MD, Department of Radiology, Azienda Ospedaliera Universitaria Integrata, Verona, Italy; Sara Mirandola, MD, Department of Surgery, Azienda Ospedaliera Universitaria Integrata, Verona, Italy; Marta M. Panzeri, MD, Department of Breast Radiology, IRCCS Ospedale San Raffaele, Milan, Italy; Danúbia A. de Andrade, MD, PhD, and Alfredo Carlos S. D. Barros, MD, PhD, Department of Breast Surgery, Hospital Sírio Libanês, São Paulo, Brazil; Katja Siegmann-Luz, MD, and Benjamin Wiesinger, MD, Department of Diagnostic and Interventional Radiology, University Hospital of Tübingen, Germany; James M. Anderson, Max Hobbs, and Wanda Gunawan, Royal Perth Hospital, Perth, Australia.
Abbreviations
- CNB
Core needle biopsy
- DCIS
Ductal carcinoma in situ
- MIPA
Multicenter International Prospective Analysis
- MRI
Magnetic resonance imaging
- OR
Odds ratio
- US
Ultrasonography
- VAB
Vacuum-assisted biopsy
Funding
Open access funding provided by Università degli Studi di Milano within the CRUI-CARE Agreement. This study received an unconditional research grant from Bayer AG. This company did not have any influence on the study protocol planning, did not have any access to the study database, and was not involved in any way in the manuscript writing or submission phases.
Declarations
Guarantor
The scientific guarantor of this publication is Prof. Francesco Sardanelli, MD.
Conflict of interest
Outside the present work, the authors declare the following relation with companies and institutions:
Francesco Sardanelli declares relationships with Bayer Healthcare (consultation/speaker fees), Bracco Imaging (grant/research support), and General Electric Healthcare (speaker fee).
Nehmat Houssami receives research funding via a National Breast Cancer Foundation (NBCF Australia) Breast Cancer Research Leadership Fellowship.
Fiona J. Gilbert received research grants from General Electric Healthcare, GSK, and Hologic, and had research collaborations with Volpara and Bayer AG. She is an NIHR senior investigator and receives funding from the Cambridge BRC.
Marc B. I. Lobbes received research grants from and is a member of the speakers’ bureau of General Electric Healthcare.
Katja Pinker declares being part of speakers bureaus for the European Society of Breast Imaging (active), Bayer AG (ended), Siemens Healthineers (ended), DKD 2019 (ended), and Olea Medical (ended); consulting for Genentech, Merantix Healthcare, AURA Health Technologies, and Guerbet.
Paola Clauser and Katja Pinker are part of the Scientific Editorial Board of European Radiology, and Rossano Girometti is Deputy Editor of European Radiology. As such, none of them had any role in handling this manuscript and none of them took part in the decision processes.
All other authors declare that they have no conflict of interest related to the present work, and that they have nothing to disclose.
Statistics and biometry
The first three authors (A.C., G.D.L, N.H.) have significant statistical expertise.
Informed consent
Written informed consent was obtained from all patients in this study, unless waived by local Ethics Committees.
Ethical approval
This study was approved by the Ethics Committee of the coordinating center on January 29, 2013 (Comitato Etico Ospedale San Raffaele, Milano, Italy; protocol number 2784), and thereafter by local Ethics Committees of participating centers.
Study subjects or cohorts overlap
This paper is a targeted subanalysis of 1005 patients that were previously included in the main paper of the MIPA study (Sardanelli et al, European Radiology 2022; 10.1007/s00330-021-08240-x) and in the paper comparing patients with different MRI referrals (Cozzi et al, European Radiology 2023; 10.1007/s00330-023-09600-5).
Methodology
• prospective
• observational
• multicenter study
Footnotes
Defined as the presence of 3 or more first-degree relatives with breast or ovarian cancer.
Measured on the craniocaudal mammographic view from the posterior image limit to the basis of the nipple.
Unifocal; multifocal; multicentric.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Grimm LJ, Rahbar H, Abdelmalak M, Hall AH, Ryser MD. Ductal carcinoma in situ: state-of-the-art review. Radiology. 2022;302:246–255. doi: 10.1148/radiol.211839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.van Seijen M, Lips EH, Thompson AM, et al. Ductal carcinoma in situ: to treat or not to treat, that is the question. Br J Cancer. 2019;121:285–292. doi: 10.1038/s41416-019-0478-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Solin LJ. Management of ductal carcinoma in situ (DCIS) of the breast: present approaches and future directions. Curr Oncol Rep. 2019;21:33. doi: 10.1007/s11912-019-0777-3. [DOI] [PubMed] [Google Scholar]
- 4.Wright JL, Rahbar H, Obeng-Gyasi S, Carlos R, Tjoe J, Wolff AC. Overcoming barriers in ductal carcinoma in situ management: from overtreatment to optimal treatment. J Clin Oncol. 2022;40:225–230. doi: 10.1200/JCO.21.01674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Allen LR, Lago-Toro CE, Hughes JH, et al. Is there a role for MRI in the preoperative assessment of patients with DCIS? Ann Surg Oncol. 2010;17:2395–2400. doi: 10.1245/s10434-010-1000-9. [DOI] [PubMed] [Google Scholar]
- 6.Itakura K, Lessing J, Sakata T, et al. The impact of preoperative magnetic resonance imaging on surgical treatment and outcomes for ductal carcinoma in situ. Clin Breast Cancer. 2011;11:33–38. doi: 10.3816/CBC.2011.n.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Davis KL, Barth RJ, Gui J, Dann E, Eisenberg B, Rosenkranz K. Use of MRI in preoperative planning for women with newly diagnosed DCIS: risk or benefit? Ann Surg Oncol. 2012;19:3270–3274. doi: 10.1245/s10434-012-2548-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kropcho LC, Steen ST, Chung AP, Sim M-S, Kirsch DL, Giuliano AE. Preoperative breast MRI in the surgical treatment of ductal carcinoma in situ. Breast J. 2012;18:151–156. doi: 10.1111/j.1524-4741.2011.01204.x. [DOI] [PubMed] [Google Scholar]
- 9.Pilewskie M, Kennedy C, Shappell C, et al. Effect of MRI on the management of ductal carcinoma in situ of the breast. Ann Surg Oncol. 2013;20:1522–1529. doi: 10.1245/s10434-012-2771-y. [DOI] [PubMed] [Google Scholar]
- 10.Hajaj M, Karim A, Pascaline S, Noor L, Patel S, Dakka M. Impact of MRI on high grade ductal carcinoma in situ (HG DCIS) management, are we using the full scope of MRI? Eur J Radiol. 2017;95:271–277. doi: 10.1016/j.ejrad.2017.08.027. [DOI] [PubMed] [Google Scholar]
- 11.Balleyguier C, Dunant A, Ceugnart L, et al. Preoperative breast magnetic resonance imaging in women with local ductal carcinoma in situ to optimize surgical outcomes: results from the randomized phase III trial IRCIS. J Clin Oncol. 2019;37:885–892. doi: 10.1200/JCO.18.00595. [DOI] [PubMed] [Google Scholar]
- 12.Lehman CD, Gatsonis C, Romanoff J, et al. Association of magnetic resonance imaging and a 12-gene expression assay with breast ductal carcinoma in situ treatment. JAMA Oncol. 2019;5:1036. doi: 10.1001/jamaoncol.2018.6269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Keymeulen KBIM, Geurts SME, Lobbes MBI, et al. Population-based study of the effect of preoperative breast MRI on the surgical management of ductal carcinoma in situ. Br J Surg. 2019;106:1488–1494. doi: 10.1002/bjs.11299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lam DL, Smith J, Partridge SC, et al. The impact of preoperative breast MRI on surgical management of women with newly diagnosed ductal carcinoma in situ. Acad Radiol. 2019;27:478–486. doi: 10.1016/j.acra.2019.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yoon GY, Choi WJ, Kim HH, Cha JH, Shin HJ, Chae EY. Surgical outcomes for ductal carcinoma in situ: impact of preoperative MRI. Radiology. 2020;295:296–303. doi: 10.1148/radiol.2020191535. [DOI] [PubMed] [Google Scholar]
- 16.Healy NA, Parag Y, Soppelsa G, et al. Does pre-operative breast MRI have an impact on surgical outcomes in high-grade DCIS? Br J Radiol. 2022;95:20220306. doi: 10.1259/bjr.20220306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hong M, Fan S, Yu Z, et al. Evaluating upstaging in ductal carcinoma in situ using preoperative MRI-based radiomics. J Magn Reson Imaging. 2022 doi: 10.1002/jmri.28539. [DOI] [PubMed] [Google Scholar]
- 18.Chou SHS, Romanoff J, Lehman CD, et al. Preoperative breast MRI for newly diagnosed ductal carcinoma in situ: imaging features and performance in a multicenter setting (ECOG-ACRIN E4112 Trial) Radiology. 2021;301:66–77. doi: 10.1148/radiol.2021204743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Keymeulen KBIM, Geurts SME, Kooreman LFS, et al. Clinical value of contralateral breast cancers detected by pre-operative MRI in patients diagnosed with DCIS: a population-based cohort study. Eur Radiol. 2022;33:2209–2217. doi: 10.1007/s00330-022-09115-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Luo J, Johnston BS, Kitsch AE, et al. Ductal carcinoma in situ: quantitative preoperative breast MR imaging features associated with recurrence after treatment. Radiology. 2017;285:788–797. doi: 10.1148/radiol.2017170587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fazeli S, Snyder BS, Gareen IF, et al. Patient-reported testing burden of breast magnetic resonance imaging among women with ductal carcinoma in situ. JAMA Netw Open. 2021;4:e2129697. doi: 10.1001/jamanetworkopen.2021.29697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fazeli S, Snyder BS, Gareen IF, et al. Association between surgery preference and receipt in ductal carcinoma in situ after breast magnetic resonance imaging. JAMA Netw Open. 2022;5:e2210331. doi: 10.1001/jamanetworkopen.2022.10331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cody HS, Van Zee KJ. Reexcision — the other breast cancer epidemic. N Engl J Med. 2015;373:568–569. doi: 10.1056/NEJMe1507190. [DOI] [PubMed] [Google Scholar]
- 24.Kaczmarski K, Wang P, Gilmore R, et al. Surgeon re-excision rates after breast-conserving surgery: a measure of low-value care. J Am Coll Surg. 2019;228:504–512e2. doi: 10.1016/j.jamcollsurg.2018.12.043. [DOI] [PubMed] [Google Scholar]
- 25.Shubeck SP, Morrow M, Dossett LA. De-escalation in breast cancer surgery. NPJ Breast Cancer. 2022;8:25. doi: 10.1038/s41523-022-00383-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fancellu A, Turner RM, Dixon JM, Pinna A, Cottu P, Houssami N. Meta-analysis of the effect of preoperative breast MRI on the surgical management of ductal carcinoma in situ. Br J Surg. 2015;102:883–893. doi: 10.1002/bjs.9797. [DOI] [PubMed] [Google Scholar]
- 27.Canelo-Aybar C, Taype-Rondan A, Zafra-Tanaka JH, et al. Preoperative breast magnetic resonance imaging in patients with ductal carcinoma in situ: a systematic review for the European Commission Initiative on Breast Cancer (ECIBC) Eur Radiol. 2021;31:5880–5893. doi: 10.1007/s00330-021-07873-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bartram A, Gilbert F, Thompson A, Mann GB, Agrawal A. Breast MRI in DCIS size estimation, breast-conserving surgery and oncoplastic breast surgery. Cancer Treat Rev. 2021;94:102158. doi: 10.1016/j.ctrv.2021.102158. [DOI] [PubMed] [Google Scholar]
- 29.Pinder SE, Thompson AM, Wesserling J. Low-risk DCIS. What is it? Observe or excise? Virchows Arch. 2022;480:21–32. doi: 10.1007/s00428-021-03173-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pinker K. Preoperative MRI improves surgical planning and outcomes for ductal carcinoma in situ. Radiology. 2020;295:304–306. doi: 10.1148/radiol.2020200076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Peters NHGM, van Esser S, van den Bosch MAAJ, et al. Preoperative MRI and surgical management in patients with nonpalpable breast cancer: the MONET – randomised controlled trial. Eur J Cancer. 2011;47:879–886. doi: 10.1016/j.ejca.2010.11.035. [DOI] [PubMed] [Google Scholar]
- 32.Sardanelli F, Trimboli RM, Houssami N, et al. Solving the preoperative breast MRI conundrum: design and protocol of the MIPA study. Eur Radiol. 2020;30:5427–5436. doi: 10.1007/s00330-020-06824-7. [DOI] [PubMed] [Google Scholar]
- 33.Sardanelli F, Trimboli RM, Houssami N, et al. Magnetic resonance imaging before breast cancer surgery: results of an observational multicenter international prospective analysis (MIPA) Eur Radiol. 2022;32:1611–1623. doi: 10.1007/s00330-021-08240-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cozzi A, Di Leo G, Houssami N et al (2023) Screening and diagnostic breast MRI: how do they impact surgical treatment? Insights from the MIPA study. Eur Radiol. 10.1007/s00330-023-09600-5 [DOI] [PMC free article] [PubMed]
- 35.Wadasadawala T, Lewis S, Parmar V, et al. Bilateral breast cancer after multimodality treatment: a report of clinical outcomes in an Asian population. Clin Breast Cancer. 2018;18:e727–e737. doi: 10.1016/j.clbc.2017.11.003. [DOI] [PubMed] [Google Scholar]
- 36.Jiang H, Zhang R, Liu X, et al. Bilateral breast cancer in China: a 10-year single-center retrospective study (2006–2016) Cancer Med. 2021;10:6089–6098. doi: 10.1002/cam4.4141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.King G, Nielsen R. Why propensity scores should not be used for matching. Polit Anal. 2019;27:435–454. doi: 10.1017/pan.2019.11. [DOI] [Google Scholar]
- 38.Greifer N, Stuart EA (2021) Choosing the estimand when matching or weighting in observational studies. 10.48550/arXiv.2106.10577
- 39.Ho DE, Imai K, King G, Stuart EA. MatchIt: nonparametric preprocessing for parametric causal inference. J Stat Softw. 2011;42:1–28. doi: 10.18637/jss.v042.i08. [DOI] [Google Scholar]
- 40.Rubin DB. Bias reduction using Mahalanobis-metric matching. Biometrics. 1980;36:293. doi: 10.2307/2529981. [DOI] [Google Scholar]
- 41.Greifer N, Stuart EA. Matching methods for confounder adjustment: an addition to the epidemiologist’s toolbox. Epidemiol Rev. 2021;43:118–129. doi: 10.1093/epirev/mxab003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Austin PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med. 2009;28:3083–3107. doi: 10.1002/sim.3697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kandel M, Dunant A, Balleyguier C, Bonastre J. Cost-effectiveness of preoperative magnetic resonance imaging to optimize surgery in ductal carcinoma in situ of the breast. Eur J Radiol. 2020;129:109058. doi: 10.1016/j.ejrad.2020.109058. [DOI] [PubMed] [Google Scholar]
- 44.Rothwell PM. External validity of randomised controlled trials: “to whom do the results of this trial apply?”. Lancet. 2005;365:82–93. doi: 10.1016/S0140-6736(04)17670-8. [DOI] [PubMed] [Google Scholar]
- 45.Djurisic S, Rath A, Gaber S, et al. Barriers to the conduct of randomised clinical trials within all disease areas. Trials. 2017;18:360. doi: 10.1186/s13063-017-2099-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hershman DL, Buono D, Jacobson JS, et al. Surgeon characteristics and use of breast conservation surgery in women with early stage breast cancer. Ann Surg. 2009;249:828–833. doi: 10.1097/SLA.0b013e3181a38f6f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee J, Tanaka E, Eby PR, et al. Preoperative breast MRI: surgeons’ patient selection patterns and potential bias in outcomes analyses. AJR Am J Roentgenol. 2017;208:923–932. doi: 10.2214/AJR.16.17038. [DOI] [PubMed] [Google Scholar]
- 48.Valero MG, Mallory MA, Losk K, et al. Surgeon variability and factors predicting for reoperation following breast-conserving surgery. Ann Surg Oncol. 2018;25:2573–2578. doi: 10.1245/s10434-018-6526-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Myers PL, Park RH, Mitchell DC, Nghiem BT, Amalfi AN. Would plastic surgeons choose breast conservation therapy? Ann Plast Surg. 2019;82:S202–S207. doi: 10.1097/SAP.0000000000001922. [DOI] [PubMed] [Google Scholar]
- 50.Ryan JF, Lesniak DM, Cordeiro E, Campbell SM, Rajaee AN. Surgeon factors influencing breast surgery outcomes: a scoping review to define the modern breast surgical oncologist. Ann Surg Oncol. 2023 doi: 10.1245/s10434-023-13472-w. [DOI] [PubMed] [Google Scholar]
- 51.Shaaban AM, Hilton B, Clements K, et al. Pathological features of 11,337 patients with primary ductal carcinoma in situ (DCIS) and subsequent events: results from the UK Sloane Project. Br J Cancer. 2021;124:1009–1017. doi: 10.1038/s41416-020-01152-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kuhl CK, Schrading S, Bieling HB, et al. MRI for diagnosis of pure ductal carcinoma in situ: a prospective observational study. Lancet. 2007;370:485–492. doi: 10.1016/S0140-6736(07)61232-X. [DOI] [PubMed] [Google Scholar]
- 53.Rahbar H, DeMartini WB, Lee AY, Partridge SC, Peacock S, Lehman CD. Accuracy of 3T versus 1.5T breast MRI for pre-operative assessment of extent of disease in newly diagnosed DCIS. Eur J Radiol. 2015;84:611–616. doi: 10.1016/j.ejrad.2014.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Greenwood HI, Wilmes LJ, Kelil T, Joe BN. Role of breast MRI in the evaluation and detection of DCIS: opportunities and challenges. J Magn Reson Imaging. 2020;52:697–709. doi: 10.1002/jmri.26985. [DOI] [PubMed] [Google Scholar]
- 55.Roque R, Cordeiro MR, Armas M, Caramelo F, Caseiro-Alves F, Figueiredo-Dias M. The accuracy of magnetic resonance imaging in predicting the size of pure ductal carcinoma in situ: a systematic review and meta-analysis. NPJ Breast Cancer. 2022;8:77. doi: 10.1038/s41523-022-00441-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cozzi A, Schiaffino S, Sardanelli F. The emerging role of contrast-enhanced mammography. Quant Imaging Med Surg. 2019;9:2012–2018. doi: 10.21037/qims.2019.11.09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cozzi A, Magni V, Zanardo M, Schiaffino S, Sardanelli F. Contrast-enhanced mammography: a systematic review and meta-analysis of diagnostic performance. Radiology. 2022;302:568–581. doi: 10.1148/radiol.211412. [DOI] [PubMed] [Google Scholar]
- 58.European Commission Initiative on Breast Cancer (2022) Planning surgical treatment: contrast-enhanced spectral mammography. https://healthcare-quality.jrc.ec.europa.eu/european-breast-cancer-guidelines/surgical-planning/CESM. Accessed 30 Jun 2023