Abstract
Objectives
To assess the presence and anatomical distribution of activated fibroblasts in the joints and entheses of patients with psoriasis with arthralgia and to test how fibroblast activation visualised by 68gallium-labelled fibroblast activation protein inhibitor-04 (68Ga-FAPI-04)-positron emission tomography (PET)/CT correlates with clinical tenderness, musculoskeletal ultrasound findings and progression to psoriatic arthritis (PsA).
Methods
We conducted a prospective cohort study in patients with psoriasis and arthralgia who underwent clinical and ultrasound evaluation and whole-body PET/CT imaging with 68Ga-FAPI-04. 68Ga-FAPI-04 uptake at synovial and entheseal sites was assessed by maximal standardised uptake values (SUVmax) and PET/CT Joint Index (JI); logistic regression models were used to investigate its correlation with clinical and ultrasound findings. Survival analyses were performed on patients with at least 6 months of follow-up.
Results
36 patients with psoriasis were enrolled. 68Ga-FAPI-04 uptake was found in 318 (7.9%) joints and 369 (7.3%) entheses in 29 (80.6%) participants, with a mean SUVmax (SD) of 3.2 (1.8) for joints and 2.9 (1.6) for entheses. Large joints and the lower limbs were predominantly affected. A significant positive relationship was found between 68Ga-FAPI-04-PET/CT signal intensity and the 68 tender joint count (SUVmax: p<0.001; PET/CT-JI: p<0.001) and tender entheses count (SUVmax: p<0.001; PET/CT-JI: p=0.002). No correlations were found with ultrasound findings (SUVmax: p=0.969; PET/CT-JI: p=0.720). Patients with relevant synovio-entheseal 68Ga-FAPI-04 uptake showed a statistically significant higher risk of developing PsA (p=0.02), independent of ultrasound findings.
Conclusions
Patients with psoriasis presenting with arthralgia show localised signs of resident tissue activation in joints and entheses, which are associated with higher risk of developing PsA.
Keywords: Fibroblasts; Psoriatic Arthritis; Ultrasonography; Outcome Assessment, Health Care; Risk Factors
WHAT IS ALREADY KNOWN ON THIS TOPIC.
Fibroblast activation plays a key role in orchestrating synovitis and enthesitis. Little is known on the role of fibroblast activation in the earliest changes of psoriatic arthritis (PsA) and the progression from psoriasis to PsA. Positron emission tomography (PET)/CT imaging with 68gallium-labelled fibroblast activation protein inhibitor-04 (68Ga-FAPI-04) provides a new molecular imaging modality that enables the visualisation of fibroblast activation in tissues in vivo.
To date, there are no data on the clinical significance of 68Ga-FAPI-04-PET/CT in patients at risk of developing PsA.
WHAT THIS STUDY ADDS
This study shows that fibroblast activation is a frequent finding in the joints and entheses of patients with psoriasis with arthralgia.
The localisation of 68Ga-FAPI-04 uptake correlates with the clinical examination of joint and entheseal tenderness, while no correlation could be found with musculoskeletal ultrasound findings, suggesting that mesenchymal activation may precede inflammatory changes detected by conventional imaging techniques and may underlie the pathophysiology of inflammatory pain in the preclinical phase of PsA.
Synovial and entheseal 68Ga-FAPI-04 uptake in patients with psoriasis has a significant positive predictive value for the development of PsA.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
This study points to fibroblast activation as an initial step on the road to develop PsA. Resident tissue activation in patients with psoriasis may not only explain the site specificity of musculoskeletal inflammation in PsA, but may also help design intervention strategies that prevent development of PsA by targeting fibroblast activation.
Introduction
Psoriatic arthritis (PsA) is a common form of inflammatory arthritis, which afflicts about 30% of patients with psoriasis.1 The manifestations of PsA are considerably heterogeneous but can lead to irreversible joint damage and loss of physical function already in the early stages of disease.2 Identifying patients with psoriasis (PsO) at higher risk of transition to PsA is crucial to define patients, who require a closer follow-up and who could benefit from preventive measures, allowing for earlier detection of musculoskeletal disease and earlier initiation of treatment. The majority of patients with PsO who develop PsA undergo a prodromal state characterised by increasing non-specific musculoskeletal complaints and beginning functional limitations in which inflammation remains subclinical.3 4 In this early stage, patients usually present with unspecific arthralgia, arthralgia fatigue and stiffness, as well as inflammatory changes on imaging, but lack evidence of clinical arthritis.5–7 Several studies performed in patients with PsO before the onset of PsA show evidence of synovitis and enthesitis,8–10 tenosynovitis11 and enthesophytes12 on MRI scans, ultrasound or CT scans, which were associated with a higher risk of transition from PsO to PsA. However, the molecular mechanisms underlying this transition are not fully understood and neither soluble biomarkers nor conventional imaging techniques provide additional insights into the early molecular alterations that predate inflammation and structural damage.13 Previous work on murine models of arthritis showed that cell surface markers of mesenchymal tissue remodelling such as fibroblast activation protein (FAP) are upregulated in enthesitis and synovitis already in the early stages of disease.14–16 Recently, a 68gallium-labelled radionuclide that binds with high affinity to FAP has been developed and approved for human use.17 Accumulation of 68gallium-labelled FAP inhibitor (68Ga-FAPI-04) tracer in target tissues allows visualising pathogenically activated fibroblasts in vivo by positron emission tomography (PET)/CT, enabling a whole-body staging and measurement of mesenchymal tissue activation.17 18 In recent studies, 68Ga-FAPI-04-PET/CT has proven highly valuable for depicting tissue remodelling and inflammation in a number of autoimmune rheumatic diseases.19 20 Luo et al have investigated the use of this imaging technique in patients with rheumatoid arthritis and demonstrated that the extent of joint involvement detected by 68Ga-FAPI-04-PET/CT correlates with conventional clinical and serological markers of disease activity with higher sensitivity compared with 18fluorodeoxyglucose (FDG).21 To date, however, there are no data on the clinical significance of this imaging modality in early arthritis and enthesitis, such as in the preclinical phases of PsA.
In this prospective cohort study, we evaluated whether and where there is evidence for early fibroblast activation in the joints and the entheses of patients with PsO with high risk of developing PsA using 68Ga-FAPI-04-PET/CT. We also tested whether the distribution of tracer uptake correlates with the clinical and ultrasound findings. Furthermore, we aimed to investigate whether the presence of an articular 68Ga-FAPI-04-PET/CT in patients with PsO is associated with the development of PsA.
Methods
Recruitment of participants
Adult patients with a biopsy-proven or dermatologist-confirmed diagnosis of PsO who experienced arthralgia were recruited at the outpatient clinic of the Department of Rheumatology and Immunology at the University Hospital of Erlangen (Germany). All patients had been referred by the Department of Dermatology of the Erlangen Hospital in order to exclude the presence of PsA and were examined clinically and by musculoskeletal ultrasound by an experienced rheumatologist (GC, FF) to rule out present or past clinical or imaging signs of inflammatory joint involvement related to PsA (arthritis, enthesitis, dactylitis, axial involvement). Furthermore, patients with PsO were not allowed to fulfil the Classification Criteria for Psoriatic Arthritis (CASPAR)22 and rheumatoid factor and anti-citrullinated protein antibodies had to be negative in all participants. Use of both conventional synthetic disease-modifying antirheumatic drugs (DMARDs) and biological DMARDs (bDMARDS)/targeted synthetic DMARDs was allowed for the treatment of cutaneous PsO. After 68Ga-FAPI-04-PET/CT imaging, all patients underwent an outpatient rheumatological follow-up at our clinic. Patients who provided at least 6 months of follow-up were considered for the survival analyses. Investigators were not blind to the imaging results at follow-up.
All participants were enrolled in the Psoriasis and Psoriatic Arthritis Cohort of the University Clinic of Erlangen (#86_21 Bc), which has been described elsewhere,12 and provided written informed consent prior to study inclusion.
Measurements
Demographic data such as age, sex, body mass index (BMI) as well as data on PsO subtype, date of onset of PsO and arthralgia were collected. Visual Analogue Scale (VAS) for pain, duration of morning stiffness, PsO therapy, as well as information about family history of PsA, were collected. Joint tenderness (TJC) and swelling were assessed by 66/68 joint count and entheseal tenderness was assessed according to the Maastricht Ankylosing Spondylitis Enthesitis Score (MASES) and Leeds Enthesitis Index (LEI). Psoriatic skin disease activity was assessed by the Psoriasis Area Severity Index. The Health Assessment Questionnaire (HAQ) was used to assess disability. C reactive protein (CRP) levels were recorded.
Ultrasound examination
Musculoskeletal ultrasound assessment of the joints and entheses was performed according to European Alliance of Associations for Rheumatology guidelines.23 All ultrasound examinations were conducted with either a MyLab25 (Esaote Biomedica, Genoa, Italy) or HS40 (Samsung Electronics, Suwon-Shi, Korea) ultrasound system equipped with broadband 4–13 MHz and 3–16 MHz linear transducers, respectively. A corresponding ultrasound preset was used for each joint or enthesis as per the manufacturer’s recommendations. Images were acquired in B mode and in power Doppler (PD) mode. The examined joints were evaluated for the presence of synovitis, which was scored both in B mode and PD mode in four grades (0–3) according to the Outcome Measures in Rheumatology (OMERACT) scoring system.24 Flexor tendons were evaluated for the presence of tenosynovitis both in B mode and PD mode. Entheses were evaluated for the presence of inflammatory (PD sign, thickening, hypoechogenicity) and structural changes (erosions and enthesophytes/calcifications) according to OMERACT recommendations.24 All sonographic examinations were performed by either one of two rheumatologists experienced in musculoskeletal ultrasound (GC, 5 years of experience; FF, 3 years of experience). The following regions of interest (ROIs) were selected for the ultrasound examination in order to reflect the most commonly affected sites of inflammation in PsA that can be detected by ultrasound: (1) joints and tendons: wrists, metacarpophalangeal joints, finger flexor tendons, elbows, knees; and (2) entheses: flexor tendon attachment at the lateral humeral epicondyle, proximal patellar attachment of the quadriceps tendon, distal patellar attachment of the patellar tendon, Achilles tendon attachment on the calcaneus.
68Ga-FAPI-04-PET/CT examination
68Ga-FAPI-04-PET/CT acquisitions were carried out on the same dedicated PET/CT system (Biograph Vision 600, Siemens Healthineers, Erlangen, Germany). In any case, the covered PET field of view (FOV) was from skull to the toes with an additional bed position of the hands (3 min per bed, axial FOV per bed 26.3 cm). PET data were corrected for random and scattered coincidences, as well as for decay during scanning. PET attenuation correction was carried out by the CT portion of the multimodal acquisition. All corrections and reconstructions were obtained using the PET/CT manufacturer’s software. All PET/CT datasets were analysed with commercially available software (Syngo. via, Siemens Molecular Imaging, Hoffman Estates, Illinois, USA), allowing review of PET, CT and fused imaging data. Visual evaluation was performed by two experienced nuclear medicine physicians (AA, CS) and one radiologist. Datasets were analysed by visual interpretation of coronal, sagittal and transverse slices. As described by Luo et al, a circular volume of interest (VOI) was placed around the affected joint in order to achieve the standardised uptake value (SUV).21 In our analysis, we used the maximal SUV (SUVmax), meaning we recorded the measurement of the most intense pixel in the selected VOI. Furthermore, a FAPI-PET/CT Joint Index was used, whereby the intensity of intra-articular tracer uptake is compared with the blood pool as well as with the gluteal muscle as reference organs described previously by Luo et al.21 For background measurement, a circular VOI with a diameter of 1 cm was placed in the ascending aorta and in the gluteal muscle.21 For the conduction of survival analyses, patients were considered ‘68Ga-FAPI-04-PET/CT positive’ when at least one joint or enthesis showed relevant radionuclide uptake, defined as presence of measurable FAPI uptake with a PET/CT Joint Index ≥2. A complete list of the ROIs at which the assessment of FAPI uptake was performed can be found in the online supplemental materials.
rmdopen-2024-004294supp001.pdf (505.5KB, pdf)
Statistical data analysis
Descriptive data were analysed using means and SDs or medians and IQRs for continuous variables, and count data/percentages were used for categorical variables. We employed logistic regression analysis to examine the association between 68Ga-FAPI-04-PET/CT signal and clinical as well as ultrasound findings to gain a comprehensive understanding of the relationships between variables by assessing the interaction between quantitative data and qualitative observations. Under the clinical assumption that the HR for the development of PsA does not undergo major changes over time, we applied the log-rank test to perform survival analyses. Correlations were calculated using Spearman’s rank correlation coefficient. The curves of PsA-free survival were visualised by Kaplan-Meier plots. Group comparisons between patients with and without synovio-entheseal 68Ga-FAPI-04-PET/CT signal were performed using the Mann-Whitney U test after assessing the normality of the distributions. A p value equal to or less than 0.05 was considered statistically significant. All statistical analyses were conducted using the R Statistical language (V.4.3.0; R Core Team, 2023) on Windows V.10×64 (build 19045), using the packages lme4, Matrix, broom.mixed, ggbeeswarm, janitor, polycor, lubridate, directlabels, ggpubr, rstatix, lmerTest, sjPlot, see, modelbased, report, tibble, formattable, broom, ggrepel, ggplot2, stringr, forcats, tidyverse, readxl, dplyr, purrr, readr, tidyr, knitr, cat, sjmisc, effects, survival, survminer and kableExtra.
Results
Participants
A total of 36 patients with PsO with arthralgia were enrolled. Detailed clinical and demographic characteristics are summarised in table 1. Participants had a mean age (SD) of 49.5 (12.2) years and 52.8% were women. The majority of patients were overweight, with a mean BMI (SD) of 30.0 (8.5). As for occupational and leisure physical activity, the majority of participants had either a sedentary lifestyle (55.5%) or sedentary work (58.3%), with 14 (38.9%) declaring being completely sedentary. The duration of psoriatic skin disease and arthralgia was highly variable throughout the cohort; the mean time (SD) since the first PsO manifestation was 16.7 years (16.8) and the mean (SD) duration of arthralgia was 3.8 (3.4) years. Most of the patients (34 (94.6%)) had inflammatory arthralgia. The average (SD) CRP level was 6.1 (1.9) mg/dL. Patients reported moderate levels of pain, the mean (SD) VAS was 43 (22) mm. Functional assessment with the HAQ revealed only minor impairment, with a median (IQR) value of 0.1 (0.0–0.6). The majority of patients were receiving topical treatment for PsO with vitamin D3 analogues and/or topical glucocorticoids (69.4%). About half were using non-steroidal anti-inflammatory drugs on demand due to joint pain (47.2%), while no participant was receiving oral glucocorticoids. Lastly, less than one in five of the patients received treatment with bDMARDs due to severe skin PsO (19.4%).
Table 1.
Demographic and clinical characteristics
| N=36 | |
| Age, mean (SD) | 49.5 (12.2) |
| Sex, n (%) | |
| Female | 19 (52.8) |
| Male | 17 (47.2) |
| BMI, mean (SD) | 30.0 (8.5) |
| Smoking history, n (%) | |
| Current | 16 (44.4) |
| Ex-smoker | 8 (22.2) |
| Non-smoker | 12 (33.3) |
| Psoriasis type, n (%) | |
| Plaque | 32 (88.9) |
| Nail involvement | 5 (13.9) |
| Scalp involvement | 9 (25.0) |
| Pustular | 4 (11.1) |
| Inversa | 3 (8.3) |
| Duration of skin disease, years, mean (SD) | 16.7 (16.8) |
| Duration of arthralgia, months, mean (SD) | 3.8 (3.4) |
| Inflammatory arthralgia | 34 (94.6%) |
| Non-specific arthralgia | 2 (5.6%) |
| Family history (1st grade), n (%) | |
| Psoriasis | 12 (33.3) |
| Rheumatic disease | 3 (8.3) |
| Physical activity | |
| Sport | |
| Sedentary lifestyle | 20 (55.5%) |
| 1 hour or less weekly | 5 (13.9%) |
| 1–3.5 hours weekly | 5 (13.9%) |
| 3.5–7 hours weekly | 4 (11.1%) |
| More than 7 hours weekly | 2 (5.6%) |
| Occupational | |
| Sedentary work | 21 (58.3%) |
| Some physical activity | 6 (16.7%) |
| Physically demanding work | 9 (25.0%) |
| Disease activity | |
| C reactive protein, mg/dL, mean (SD) | 6.1 (1.9) |
| Pain VAS, mm, mean (SD) | 43 (22) |
| Tender joint count 68, median (IQR) | 1 (0–5) |
| Swollen joint count 66, median (IQR) | 0 (0–0) |
| LEI, median (IQR) | 0.0 (0.0–1.3) |
| MASES, median (IQR) | 0.0 (0.0–1.5) |
| HAQ, median (IQR) | 0.1 (0.0–0.6) |
| Treatment | |
| Topical therapies, n (%) | 25 (69.4) |
| Oral glucocorticoids, n (%) | 0 (0) |
| NSAIDs (occasional use), n (%) | 17 (47.2) |
| csDMARDs, n (%) | 0 (0) |
| bDMARDs, n (%) | 7 (19.5) |
| Ustekinumab | 4 (11.1) |
| Guselkumab | 1 (2.8) |
| Risankizumab | 1 (2.8) |
| Ixekizumab | 1 (2.8) |
bDMARDs, biological disease-modifying antirheumatic drugs; BMI, body mass index; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; HAQ, Health Assessment Questionnaire; LEI, Leeds Enthesitis Index; MASES, Maastricht Ankylosing Spondylitis Enthesitis Score; NSAIDs, non-steroidal anti-inflammatory drugs; VAS, Visual Analogue Scale.
68Ga-FAPI-04-PET/CT findings of joints and entheses
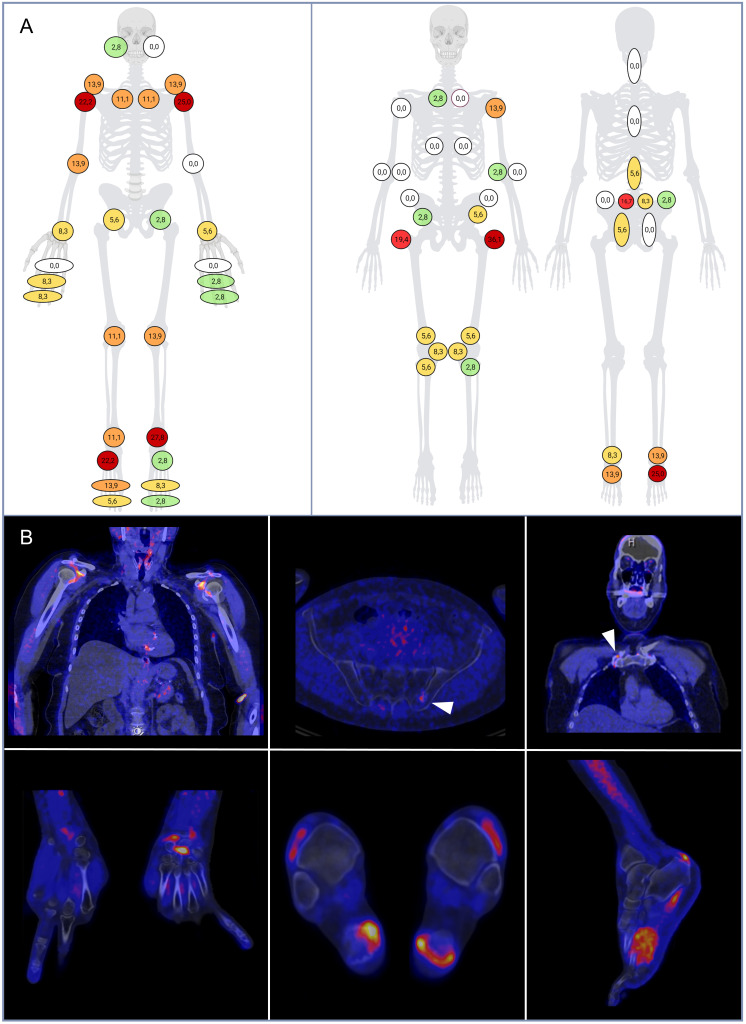
A total of 4019 synovial ROIs and 4949 entheseal ROIs from 36 68Ga-FAPI-04-PET/CT scans from all participants were evaluated. The PET/CT scans were positive for presence of any relevant synovial and/or entheseal 68Ga-FAPI-04 uptake in 80.6% (n=29) of participants and a significant burden of both synovial and entheseal radionuclide uptake could be observed throughout the cohort. A total of 318 (7.9%) joints and 369 (7.3%) entheses demonstrated 68Ga-FAPI-04 uptake in 68Ga-FAPI-04-PET/CT, with mean (SD) SUVmax of 3.2 (1.8) for joints and 2.9 (1.6) for entheses. On a patient basis, the median (IQR) PET joint count was 2.0 (0.0–4.0) and the median (IQR) PET entheses count was 1.0 (0.0–3.0). Joints and entheses of the lower limbs seemed to be predominantly affected in 68Ga-FAPI-04-PET/CT compared with the upper limbs. The distribution of radionuclide uptake in joints, entheses and in the axial skeleton is shown in figure 1, alongside examples of tracer accumulation.
Figure 1.
(A) Distribution of 68Ga-FAPI-04-PET/CT findings in the joints (A), entheses and axial skeleton (B). The frequency of PET/CT findings for each site is expressed as percentage on the total of available examination per site (0%: white; ≤5%: green; 5–10%: yellow; 10–15%: orange; 15–20%: light red; >20%: dark red). (B) PET/CT images of patients with psoriasis showing increased 68Ga-FAPI-04 uptake in the left sacroiliac joint (left upper quadrant), at the Achilles tendon enthesis and plantar fascia (right upper quadrant), in the synovial recesses of the shoulder joints (left lower quadrant) and in the left wrist joint (right lower quadrant). 68Ga-FAPI-04, 68gallium-labelled fibroblast activation protein inhibitor-04; PET, positron emission tomography.
68Ga-FAPI-04-PET/CT results in relation to clinical characteristics and therapy
We found no significant relations between 68Ga-FAPI-04 uptake and physical activity by stratifying patients by levels of occupational and leisure physical activity. However, patients who exhibited a relevant synovio-entheseal 68Ga-FAPI-04 uptake had a significantly higher mean (SD) BMI (30.6 (7.7)) compared with 68Ga-FAPI-04-PET/CT-negative patients (25.5 (3.2)) (p=0.029). Accordingly, a moderate correlation between BMI and the burden of synovio-entheseal 68Ga-FAPI-04 uptake expressed by PET/CT Joint Index was found with a Spearman’s coefficient r=0.38 (p=0.026) (online supplemental figure 1).
rmdopen-2024-004294supp002.pdf (349.2KB, pdf)
Regarding therapy, we observed a numerically lower cumulative PET/CT Joint Index in patients who were under treatment with bDMARD with a mean (SD) of 7.0 (±6.9), while patients who were not receiving DMARDs had a mean (SD) PET/CT Joint Index of 11.8 (±11.6). However, the variability was high due to the small sample sizes and differences were not found to be significant (p=0.175).
68Ga-FAPI-04-PET/CT results in relation to physical findings
Overall, 1320 joint sites and 1080 entheseal sites were clinically assessed. Clinical examination showed a total of 95 (7.8%) tender joints, no swollen joints and 56 (5.2%) tender entheses. The median TJC (IQR) was 1 (0–5), the median LEI (IQR) was 0 (0–1.3) and the median MASES (IQR) was 0 (0–1.5). Joint tenderness was predominantly found at the upper extremities, and mostly at the metacarpophalangeal joints (n=15), shoulder (n=15), wrist (n=14) and proximal interphalangeal joints (PIPs) (n=10). The most frequently affected entheses were the lateral humeral epicondyle (n=18), the spinous process of the fifth lumber vertebra (n=14) and the Achilles tendons (n=12). A detailed overview of clinical findings is reported in online supplemental table 1. An increased 68Ga-FAPI-04 uptake was found in corresponding sites of joint and entheseal tenderness in 1.5% of all sites (20.3% of tender sites). 68Ga-FAPI-04 uptake was found in non-tender joints and entheseal sites in 6.2% of all sites (6.7% of non-tender sites). Another 5.9% of articular sites were tender but were not associated with 68Ga-FAPI-04-PET/CT signal. The remaining 86.4% of sites were neither tender nor showed an increased 68Ga-FAPI-04 uptake (table 2). Of note, the two patients with non-inflammatory non-specific arthralgia also exhibited no relevant 68Ga-FAPI-04 uptake.
Table 2.
Agreement of clinical and ultrasound findings with 68Ga-FAPI-04 uptake
| 68Ga-FAPI-04 uptake | ||||
| Absent | Present | Total | ||
| Physical examination at baseline | Non-tender | 2074 86.4% |
149 6.2% |
2223 92.6% |
| Tender | 141 5.9% |
36 1.5% |
177 7.4% |
|
| Total | 2215 92.3% |
185 7.7% |
2400 100% |
|
| 68Ga-FAPI-04 uptake | ||||
| Absent | Present | Total | ||
| Ultrasound | Changes absent | 235 75.6% |
13 4.2% |
248 79.8% |
| Changes present | 58 18.6% |
5 1.6% |
63 20.2% |
|
| Total | 293 94.2% |
18 5.8% |
311 100% |
|
68Ga-FAPI-04, 68gallium-labelled fibroblast activation protein inhibitor-04.
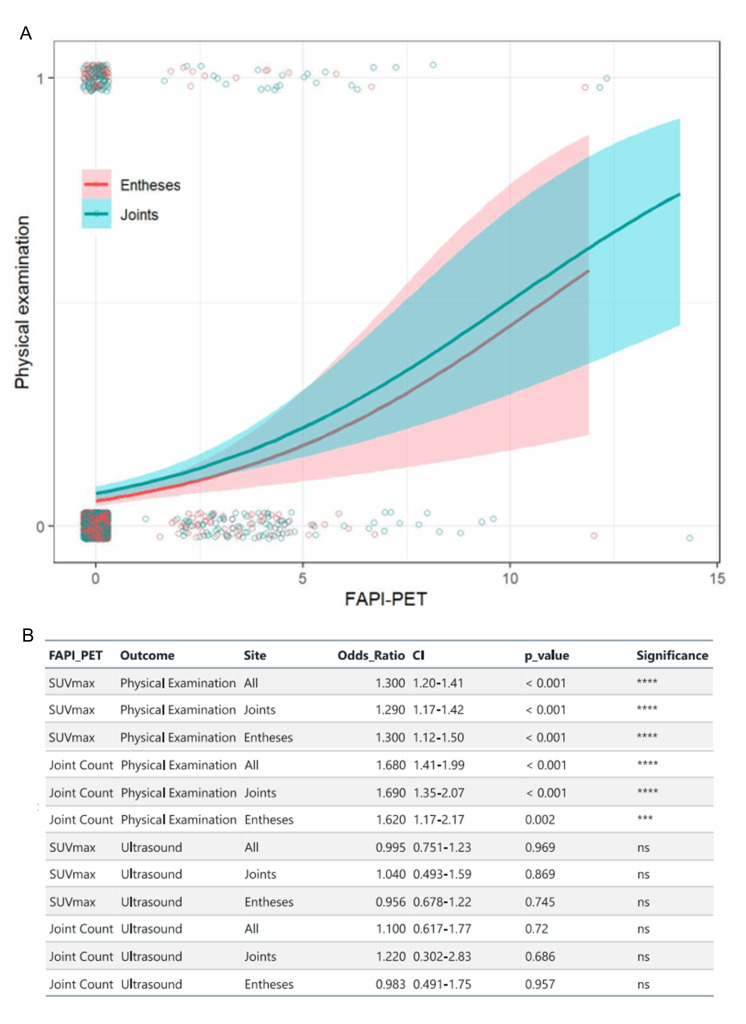
A significant positive relationship between 68Ga-FAPI-04-PET/CT and the 68/66 joint count was found both for SUVmax and for the PET/CT Joint Index in the regression models. The odds of having a tender joint in the joint count increased by 29% (OR=1.29, 95% CI=1.17 to 1.42, p<0.001) for each unit increase in the SUVmax and by 69% (OR=1.69, 95% CI=1.35 to 2.07, p<0.001) for each unit increase in the 68Ga-FAPI-04-PET/CT Joint Index. For entheses, the odds of entheseal tenderness increased by 30% (OR=1.30, 95% CI=1.12 to 1.50, p<0.001) for each unit increase in the SUVmax and by 62% (OR=1.62, 95% CI=1.17 to 2.17, p=0.002) for each unit increase in the 68Ga-FAPI-04-PET/CT Joint Index. Figure 2 shows regression curves for SUVmax and clinical findings and an overview of significant relations between clinical and PET/CT parameters.
Figure 2.
(A) Logistic regression model of clinical examination findings in joints and entheses with 68Ga-FAPI-04 uptake. Regression curves are represented in blue for joints and red for entheses. Shaded areas represent the 95% CI. Points represent individual clinical and imaging findings. (B) Relationships between clinical and ultrasound findings and maximal standardised uptake values (SUVmax) and PET/CT Joint Index. 68Ga-FAPI-04, 68gallium-labelled FAPI-04; FAPI, fibroblast activation protein inhibitor; ns, not significant; PET, positron emission tomography. ****: p<0.001; ***: p<0.01.
68Ga-FAPI-04-PET/CT in relation to ultrasound findings
A total of 311 ultrasound scans were obtained and assessed at the above-mentioned ROIs from the study participants. Abnormal ultrasound findings were fairly frequent but mild, and were found in 25.4% (n=15) of the joints and in 19.0% (n=48) of the entheses. The most commonly affected joints were wrists (4.2%) and knees (1.8%) and synovial hypertrophy was the only finding. All cases were scored as a grade 1 synovial hypertrophy except for one knee joint scored as grade 2. Entheseal anomalies were most frequent at the lateral humeral epicondyle (7.5%) and at the Achilles tendon (5.9%), and mainly consisted in hypoechogenicity (66.7%), calcifications (50.0%) and thickening (29.2%). A detailed overview of the distribution of ultrasound changes is reported in online supplemental table 2. Ultrasound anomalies corresponded to a 68Ga-FAPI-04 uptake in 1.6% of all sites (7.9% of ultrasound-positive sites), while in 4.2% of all sites, 68Ga-FAPI-04 uptake was found in sites with normal ultrasound (5.2% of ultrasound-negative sites). In 18.6% of all sites, no 68Ga-FAPI-04-PET/CT signal was found despite ultrasound changes. The remaining 75.6% of sites were neither tender nor showed an increased 68Ga-FAPI-04 uptake (table 2).
Regression models showed no significant relation between the presence of ultrasound findings at synovial and entheseal sites with neither SUVmax (p=0.969) nor with the PET/CT Joint Index (p=0.720) (online supplemental figure 2).
rmdopen-2024-004294supp003.pdf (25.2KB, pdf)
Predictive value of synovio-entheseal 68Ga-FAPI-04-PET/CT signal for PsA development
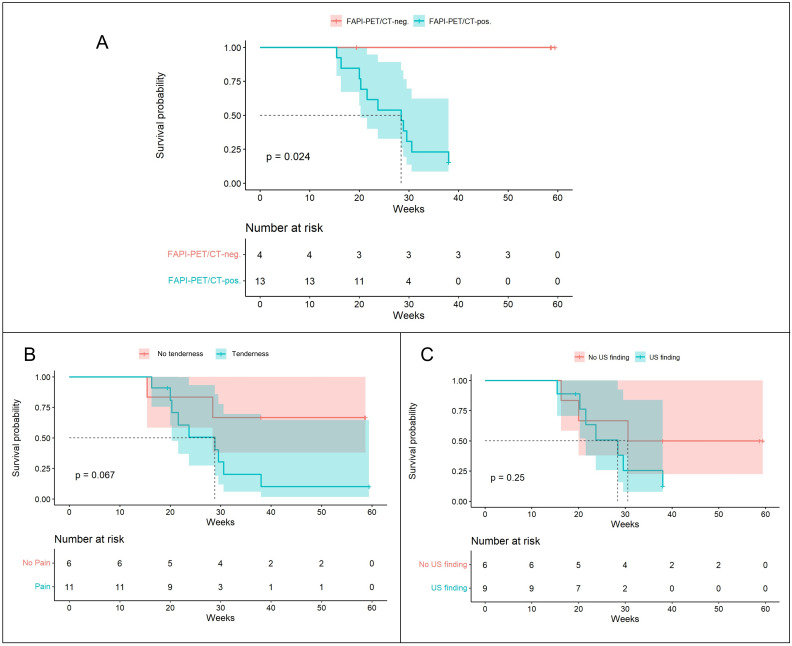
Survival analyses were conducted on a subset of 17 patients for whom a follow-up of at least 6 months after the 68Ga-FAPI-04-PET/CT scan was available. Of the 13 patients with positive 68Ga-FAPI-04 tracer accumulation, only 2 were not diagnosed with PsA during the 38-week observation period. 11 patients had received a diagnosis of PsA and fulfilled CASPAR criteria with presence of both clinical and imaging (ie, ultrasound, MRI) findings compatible with PsA during this period. Further details on the clinical manifestations and performed imaging leading to PsA diagnosis can be found in online supplemental tables 3 and 4. The median time to PsA diagnosis was 23.7 weeks. In all 11 incident PsA cases, the baseline PET/CT scan had shown relevant radionuclide uptake in synovial and entheseal sites, as opposed to no patient in the 68Ga-FAPI-04-PET/CT-negative group. Comparing the PsA-free survival of participants based on their 68Ga-FAPI-04-PET/CT results, patients with relevant synovial and entheseal 68Ga-FAPI-04 uptake had a statistically significant higher risk of developing PsA (p=0.02). When the survival analysis for the subgroups with and without presence of joint/entheseal tenderness was done, progression to PsA was higher, though not significant (p=0.067) in patients exhibiting joint/entheseal tenderness, reflecting the aforementioned findings of an association between 68Ga-FAPI-04-PET/CT findings and tenderness. Subgroup analysis with respect to presence versus absence of ultrasound changes did not reveal significant differences (p=0.25), indicating that a positive 68Ga-FAPI-04-PET/CT signal predicts progression to PsA independent of ultrasound changes (figure 3B).
Figure 3.
(A) Kaplan-Meier curves of the PsA-free survival in patients with psoriasis with positive 68Ga-FAPI-04-PET/CT scans (blue line) versus negative 68Ga-FAPI-04-PET/CT scans (red line) at synovial and entheseal sites. (B) PsA-free survival in patients with presence (blue line) versus absence of synovial or entheseal tenderness (red line). (C) PsA-free survival in patients with presence (blue line) versus absence of ultrasound (US) anomalies (red line). 68Ga-FAPI-04, 68gallium-labelled FAPI-04; FAPI, fibroblast activation protein inhibitor; PET, positron emission tomography; PsA, psoriatic arthritis.
Regarding body weight, 9 of 11 (81.8%) progressors had a BMI >25, whereas all 6 non-progressors had a normal BMI. Furthermore, patients who progressed to PsA seemed to have overall lower exposure to occupational and leisure physical activity compared with non-progressors. Regular physical activity was practised by two progressors (18.2%) compared with 4 non-progressors (66.7%), whereas only 2 of 11 progressors (18.2%) had a physically active occupation as opposed to 2 of 6 (33.3%) non-progressors.
Discussion
Tissue-resident fibroblasts have been strongly implicated in the pathogenesis of inflammatory arthritis and accumulating evidence is unveiling their role as key actors in the attraction and activation of immune cells to the joints. In synovitis, fibroblasts sustain inflammation and osteoclast-mediated bone resorption by secreting proinflammatory cytokines, such as interleukin 6, and a variety of chemokines that allow recruiting immune cells.25 26 Likewise, enthesitis is characterised by a distinct microenvironment, in which fibroblast activation induces new bone formation and promotes the homing of innate immune cells.27 Hence, visualising fibroblast activation in humans in vivo could allow depicting the initial changes in the joints and the entheses that ultimately lead to the development of PsA.
Here, we show substantial signs of fibroblast activation at synovial and entheseal sites demonstrated by an increased 68Ga-FAPI-04 uptake in PET/CT imaging in patients with PsO who experience arthralgia and are therefore at higher risk of developing PsA. Of note, none of these patients had signs of PsA at time of imaging. Fibroblast activation was most frequently found in the joints and entheses of the lower limbs. This observation supports the findings from preclinical studies that showed increased FAP expression in the entheses of murine models of spondyloarthritis after exposure to mechanical strain,14 which is expected to be high in the lower extremities.
The intensity of the 68Ga-FAPI-04 uptake, both as absolute value as well as adjusted by background muscle uptake, positively correlated with the number of clinically tender joints and entheses. In the majority of cases, however, 68Ga-FAPI-04 uptake was present in the absence of tenderness of the respective joint, supporting the notion that mesenchymal activation predates the onset of symptoms. On the other hand, no relation was found between ultrasound-detected inflammatory changes and 68Ga-FAPI-04 uptake in joints or entheses. A possible explanation is that tissue changes related to fibroblast activation in the joints are not reflected by a sufficient degree of morphological and vascular changes that can be detected by ultrasound, but still may lead to substantial inflammatory pain. Also, despite standardisation efforts, operator-dependent biases and technical limitations intrinsic to the ultrasound device may have contributed to a loss of accuracy.
Interestingly, we observed a numerically lower burden of 68Ga-FAPI-04 uptake in patients with PsO treated with bDMARDs compared with the untreated. Statistical significance was not reached mainly due to the small sample size of bDMARD-treated group; however, this might indicate an influence of cytokine-blocking therapies on activated fibroblasts in PsA and should be confirmed in larger studies.
Lastly, the presence of synovio-entheseal 68Ga-FAPI-04-PET/CT signal was associated with a high risk of developing PsA over time. Of note, this progression was observed in the presence or absence of ultrasound changes. These findings suggest that a key checkpoint in the development of PsA is resident tissue activation, which not only precedes the onset of PsA but also may emerge before the onset of subclinical inflammation in the joints and entheses. Furthermore, these observations support the concept of synovio-entheseal stress as a key pathophysiological mechanism in PsA, as coined by McGonagle et al, stating that increased stress stimuli on synovio-entheseal complexes lead to local changes in the tissue that render it susceptible to inflammation28 (online supplemental figure 3). Interestingly, however, the pattern of 68Ga-FAPI-04 uptake did not always predict the exact clinical pattern of joint involvement once a PsA diagnosis was established, suggesting that increased tissue remodelling at these sites does not inevitably translate to the development of a clinically evident arthritis at a later point.
rmdopen-2024-004294supp004.pdf (3.5MB, pdf)
Our study has some limitations. First, imaging results were not kept blind to the investigator at follow-up, which might represent a source of confirmation bias. Second, a certain degree of mesenchymal activation may also be found in the context of osteoarthritis. However, our study population was relatively young and bone structure as assessed by CT imaging did not show any signs of osteoarthritis. Hence, a relevant influence of osteoarthritis on our results appears unlikely. Also further aspects such as body weight, metabolic factors, treatments and exposure to physical strain might be able to influence fibroblast activation and consequently the generation of a 68Ga-FAPI-04 signal of the joints. In this cohort, longitudinal data with a long enough follow-up time were only available in a limited number of patients with PsO, which reduces the power of our analysis. Thus, a more refined prediction model for the development of PsA, which includes 68Ga-FAPI-04-PET/CT, would need the analysis of a larger patient cohort. On the other hand, these data provide a conceptual model of the development of PsA, which can be validated in further studies. Lastly, 68Ga-FAPI-04-PET/CT will need to be validated by comparing it with other conventional imaging techniques such as MRI and 18FDG-PET/CT. An exemplary comparison of 68Ga-FAPI-04-PET/CT, ultrasound and MRI findings in a patient with highly active PsA is shown in online supplemental figure 4.
rmdopen-2024-004294supp005.pdf (1.5MB, pdf)
In conclusion, our data suggest that the earliest changes in the joints and entheses of patients with PsO transitioning to PsA are hallmarked by the activation of tissue-resident mesenchymal cells, which can be detected by FAP-specific tracer accumulation. Mesenchymal activation may then drive the local influx of immune cells and site-specific development of PsA. This concept integrates local aspects of PsA such as local tissue stress, which leads to priming of resident tissue and site-specific lowering of the inflammation threshold. The observation that patients with PsO both with and without ultrasound-detected inflammation progress to PsA if a positive 68Ga-FAPI-04-PET/CT signal is present suggests that mesenchymal activation may precede inflammation in the course to PsA. Hence, this evidence paves the way for potential uses of mesenchymal cell targeting in the prevention and therapy of early psoriatic disease.
Acknowledgments
We sincerely thank our patients for their participation in this study. Figure 1 was created with BioRender.com.
Footnotes
Contributors: Study design—GC, AR, DS, GS and FF. Data collection—GC, AA, RNdS, VS, MGR, AK, DS, MS and FF. Ultrasound examinations and scoring—GC and FF. PET/CT evaluation—AA, CS and TK. Tables and figures—GC, AA, SAT, CS, GS and FF. Statistical analysis—SAT. Conceptualising and writing of the manuscript—GC, AA, DS, MS, CS, GS and FF. Guarantor—GS
Funding: The study was supported by the Deutsche Forschungsgemeinschaft (DFG-FOR2886 PANDORA-405969122 and the CRC1181 Checkpoints for Resolution of Inflammation–261193037; Leibniz Award to GS). Additional funding was received by the Bundesministerium für Bildung und Forschung (project MASCARA), the ERC Synergy grant 4D Nanoscope and the Emerging Fields Initiative MIRACLE of Friedrich-Alexander University Erlangen-Nürnberg. This project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking (JU) under grant agreement no 101007757 (HIPPOCRATES). The JU receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA. DS was supported by the 2022 GRAPPA Pilot Research Grant. GC, AA and FF are funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)–493624887 (Clinician Scientist Program NOTICE)).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. Data are available from the corresponding author upon reasonable request.
Ethics statements
Patient consent for publication
Consent obtained directly from patient(s).
Ethics approval
This study involves human participants and was approved by the Ethics Committee of the University of Erlangen-Nuremberg (#23-359-Bp). Participants gave informed consent to participate in the study before taking part.
References
- 1. Ritchlin CT, Colbert RA, Gladman DD. Psoriatic arthritis. N Engl J Med 2017;376:957–70. 10.1056/NEJMra1505557 [DOI] [PubMed] [Google Scholar]
- 2. Gladman DD, Antoni C, Mease P, et al. Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis 2005;64 Suppl 2:ii14–7. 10.1136/ard.2004.032482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. De Marco G, Zabotti A, Baraliakos X, et al. Characterisation of Prodromal and very early Psoriatic arthritis: a systematic literature review informing a EULAR Taskforce. RMD Open 2023;9:e003143. 10.1136/rmdopen-2023-003143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Eder L, Polachek A, Rosen CF, et al. The development of Psoriatic arthritis in patients with psoriasis is preceded by a period of nonspecific musculoskeletal symptoms: A prospective cohort study. Arthritis & Rheumatology 2017;69:622–9. 10.1002/art.39973 [DOI] [PubMed] [Google Scholar]
- 5. Wilson FC, Icen M, Crowson CS, et al. Incidence and clinical predictors of Psoriatic arthritis in patients with psoriasis: a population-based study. Arthritis Rheum 2009;61:233–9. 10.1002/art.24172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Zabotti A, De Marco G, Gossec L, et al. n.d. EULAR points to consider for the definition of clinical and imaging features suspicious for progression from psoriasis to Psoriatic arthritis. Ann Rheum Dis:ard–2023. 10.1136/ard-2023-224148 [DOI] [PubMed] [Google Scholar]
- 7. Scher JU, Ogdie A, Merola JF, et al. Preventing Psoriatic arthritis: focusing on patients with psoriasis at increased risk of transition. Nat Rev Rheumatol 2019;15:153–66. 10.1038/s41584-019-0175-0 [DOI] [PubMed] [Google Scholar]
- 8. Faustini F, Simon D, Oliveira I, et al. Subclinical joint inflammation in patients with psoriasis without concomitant Psoriatic arthritis: a cross-sectional and longitudinal analysis. Ann Rheum Dis 2016;75:2068–74. 10.1136/annrheumdis-2015-208821 [DOI] [PubMed] [Google Scholar]
- 9. Zuliani F, Zabotti A, Errichetti E, et al. Ultrasonographic detection of Subclinical Enthesitis and Synovitis: a possible stratification of Psoriatic patients without clinical musculoskeletal involvement. Clin Exp Rheumatol 2019;593–9. [PubMed] [Google Scholar]
- 10. Naredo E, Moller I, de Miguel E, et al. High prevalence of ultrasonographic Synovitis and Enthesopathy in patients with psoriasis without Psoriatic arthritis: a prospective case-control study. Rheumatology 2011;50:1838–48. 10.1093/rheumatology/ker078 [DOI] [PubMed] [Google Scholar]
- 11. Zabotti A, McGonagle DG, Giovannini I, et al. Transition phase towards Psoriatic arthritis: clinical and ultrasonographic Characterisation of Psoriatic arthralgia. RMD Open 2019;5:e001067. 10.1136/rmdopen-2019-001067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Simon D, Tascilar K, Kleyer A, et al. Association of structural Entheseal lesions with an increased risk of progression from psoriasis to Psoriatic arthritis. Arthritis Rheumatol 2022;74:253–62. 10.1002/art.41239 [DOI] [PubMed] [Google Scholar]
- 13. Generali E, Scirè CA, Favalli EG, et al. Biomarkers in Psoriatic arthritis: a systematic literature review. Expert Review of Clinical Immunology 2016;12:651–60. 10.1586/1744666X.2016.1147954 [DOI] [PubMed] [Google Scholar]
- 14. Cambré I, Gaublomme D, Schryvers N, et al. Running promotes Chronicity of arthritis by local modulation of complement Activators and impairing T regulatory feedback loops. Ann Rheum Dis 2019;78:787–95. 10.1136/annrheumdis-2018-214627 [DOI] [PubMed] [Google Scholar]
- 15. Ritchlin C. Navigating the diverse immune landscapes of Psoriatic arthritis. Semin Immunopathol 2021;43:279–90. 10.1007/s00281-021-00848-x [DOI] [PubMed] [Google Scholar]
- 16. Jacob M, Chang L, Puré E. Fibroblast activation protein in remodeling tissues. Curr Mol Med 2012;12:1220–43. 10.2174/156652412803833607 [DOI] [PubMed] [Google Scholar]
- 17. Lindner T, Loktev A, Altmann A, et al. Development of quinoline-based Theranostic ligands for the targeting of fibroblast activation protein. J Nucl Med 2018;59:1415–22. 10.2967/jnumed.118.210443 [DOI] [PubMed] [Google Scholar]
- 18. Kuwert T, Schmidkonz C, Prante O, et al. FAPI PET opens a new window to understanding immune-mediated inflammatory diseases. J Nucl Med 2022;63:1136–7. 10.2967/jnumed.122.263922 [DOI] [PubMed] [Google Scholar]
- 19. Schmidkonz C, Atzinger A, Ramming A, et al. FAPI PET/CT immune-fibrosis imaging for new insights into rheumatologic disorders. J Nucl Med 2023;64:1674–5. 10.2967/jnumed.123.266182 [DOI] [PubMed] [Google Scholar]
- 20. Bergmann C, Distler JHW, Treutlein C, et al. 68Ga-FAPI-04 PET-CT for molecular assessment of fibroblast activation and risk evaluation in systemic sclerosis-associated interstitial lung disease: a single-centre, pilot study. The Lancet Rheumatology 2021;3:e185–94. 10.1016/S2665-9913(20)30421-5 [DOI] [PubMed] [Google Scholar]
- 21. Luo Y, Pan Q, Zhou Z, et al. 68Ga-FAPI PET/CT for rheumatoid arthritis: a prospective study. Radiology 2023;307:e222052. 10.1148/radiol.222052 [DOI] [PubMed] [Google Scholar]
- 22. Taylor W, Gladman D, Helliwell P, et al. Classification criteria for Psoriatic arthritis: development of new criteria from a large international study. Arthritis & Rheumatism 2006;54:2665–73. 10.1002/art.21972 [DOI] [PubMed] [Google Scholar]
- 23. Möller I, Janta I, Backhaus M, et al. The 2017 EULAR standardised procedures for ultrasound imaging in rheumatology. Ann Rheum Dis 2017;76:1974–9. 10.1136/annrheumdis-2017-211585 [DOI] [PubMed] [Google Scholar]
- 24. Bruyn GA, Iagnocco A, Naredo E, et al. OMERACT definitions for ultrasonographic Pathologies and elementary lesions of rheumatic disorders 15 years on. J Rheumatol 2019;46:1388–93. 10.3899/jrheum.181095 [DOI] [PubMed] [Google Scholar]
- 25. Mizoguchi F, Slowikowski K, Wei K, et al. Functionally distinct disease-associated fibroblast Subsets in rheumatoid arthritis. Nat Commun 2018;9:789:789. 10.1038/s41467-018-02892-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Croft AP, Campos J, Jansen K, et al. Distinct fibroblast Subsets drive inflammation and damage in arthritis. Nature 2019;570:246–51. 10.1038/s41586-019-1263-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Schett G, Lories RJ, D’Agostino M-A, et al. Enthesitis: from pathophysiology to treatment. Nat Rev Rheumatol 2017;13:731–41. 10.1038/nrrheum.2017.188 [DOI] [PubMed] [Google Scholar]
- 28. McGonagle D, Lories RJU, Tan AL, et al. “The concept of a “Synovio‐Entheseal complex” and its implications for understanding joint inflammation and damage in Psoriatic arthritis and beyond”. Arthritis & Rheumatism 2007;56:2482–91. 10.1002/art.22758 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
rmdopen-2024-004294supp001.pdf (505.5KB, pdf)
rmdopen-2024-004294supp002.pdf (349.2KB, pdf)
rmdopen-2024-004294supp003.pdf (25.2KB, pdf)
rmdopen-2024-004294supp004.pdf (3.5MB, pdf)
rmdopen-2024-004294supp005.pdf (1.5MB, pdf)
Data Availability Statement
Data are available upon reasonable request. Data are available from the corresponding author upon reasonable request.