Abstract
Chondrosarcoma (ChS), a malignant cartilage-producing tumor, is the second most frequently diagnosed osseous sarcoma after osteosarcoma. It represents a very heterogeneous group of malignant chemo- and radiation-resistant neoplasms, accounting for approximately 20% of all bone sarcomas. The majority of ChS patients have a good prognosis after a complete surgical resection, as these tumors grow slowly and rarely metastasize. Conversely, patients with inoperable disease, due to the tumor location, size, or metastases, represent a great clinical challenge. Despite several genetic and epigenetic alterations that have been described in distinct ChS subtypes, very few therapeutic options are currently available for ChS patients. Therefore, new prognostic factors for tumor progression as well as new treatment options have to be explored, especially for patients with unresectable or metastatic disease. Recent studies have shown that a correlation between immune infiltrate composition, tumor aggressiveness, and survival does exist in ChS patients. In addition, the intra-tumor microvessel density has been proven to be associated with aggressive clinical behavior and a high metastatic potential in ChS. This review will provide an insight into the ChS microenvironment, since immunotherapy and antiangiogenic agents are emerging as interesting therapeutic options for ChS patients.
Keywords: chondrosarcoma, tumor microenvironment, angiogenesis, immune checkpoint inhibitors
1. Introduction
1.1. Incidence and Histopathology
Chondrosarcoma (ChS) is the second most frequent malignant bone tumor, accounting for approximately 20% of all bone sarcomas [1]. These tumors constitute a heterogeneous group of mesenchymal neoplasms producing a non-osteoid cartilage matrix [2]. Epidemiologically, ChS seems to exhibit a weak predilection for males, with a mean age of incidence rate of 51 years [3]. Furthermore, rare ChS subtypes may also occur at a younger age, with a peak incidence in the second and third decades of life [4]. Recent data from the literature report that ChS rates are increasing, but this is probably due to the better characterization of the atypical cartilaginous tumor (ACT), which is a low-grade malignant ChS often localized in the appendicular skeleton [5,6].
The most common ChS localizations include bones of the axial skeleton, pelvis, scapula, sternum, and ribs, followed by the proximal humerus and femur. Of note, hands and feet are only rarely involved [7]. Macroscopically, ChSs are large tumors, usually greater than 4 cm in size, having a translucent lobular, white, or blue–grey cut surface due to the presence of hyaline cartilage [8].
Primary ChSs that arise de novo account for 20–27% of all primary malignant bone tumors, whereas secondary ChSs, accounting for approximately 1% of bone tumors, arise from pre-existing enchondroma or osteochondroma [9].
Grade evaluation of ChS is based on cellularity, nuclear features, and mitotic rate, as well as tumor matrix characteristics [10,11,12]. According to the 2020 WHO classification updated by Choi J. et al. [13,14], eight ChS subtypes have been identified. The conventional ChS is the most frequent subtype, accounting for about 25% of all primary bone tumors in adults. The majority of conventional ChSs are low- or intermediate-grade tumors characterized by an indolent clinical course and low metastatic potential. However, 5–10% of them exhibit high metastatic potential and a poor prognosis. The main site of metastatic disease is the lung, with regional lymph nodes less commonly involved [15]. According to their bone location, conventional ChS subtypes may be further classified as central (the most frequent) when they arise in the intramedullary cavity of bones, peripheral when they arise within the cartilage cap of a pre-existing osteochondroma, or periosteal (the rarest) when they localize on the surface of the bones. While the peripheral tumors are intermediate-grade or high-grade malignancies, the periosteal ChS seen on the external surface of the bone is Grade I or II. Cortical invasion, soft tissue extension, and size (>5 cm), as well as the presence of periostin, a stromal-related protein absent in enchondromas, can be helpful in distinguishing them from periosteal chondromas [16,17,18].
Dedifferentiated (dd) ChSs that occur in the fifth–seventh decades of life are rare, aggressive tumors accounting for approximately 10% of all ChSs [19]. Firstly described by Dahlin D.C. and Beabout J.W. in 1971 [20], ddChSs generally have a poor prognosis due to their strong tendency to metastasize. This histotype represents a great challenge for clinical management due to the coexistence of a high-grade non-cartilaginous sarcoma in the context of a low-grade chondrogenic component, with one blending of the first into the other [21]. The non-cartilaginous portion is frequently constituted by conventional osteosarcoma (70% of cases) and less frequently by malignant fibrous histiocytoma or fibrosarcoma [21].
Mesenchymal ChSs that frequently involve the axial skeleton are rare, high-grade tumors with a poor prognosis due to their strong tendency to metastasize. They can originate from either bone or soft tissues and are characterized by the presence of differentiated cartilage mixed with solid areas in which undifferentiated small round cells simulate Ewing’s sarcoma [9,22].
Finally, the very rare Clear Cell ChS is a low-grade malignant tumor occurring between the third and fifth decades of life. This subtype, which preferentially involves the epiphyses of long bones [23], is characterized by the presence of round or polygonal cells with large nuclei, centrally placed nucleoli, and abundant vacuolated, clear cytoplasm, arranged in lobules and sheets [16]. Woven bone formation and osteoclast-like giant cells are common features, but prominent osteoid formation should be considered for a differential diagnosis of osteosarcoma. Areas of conventional low-grade ChSs can be observed in approximately half of cases; mitotic figures are rare, but dedifferentiation can also occur [16].
1.2. Clinical Features and Therapeutic Management
The clinical presentation of ChSs often consists of persistent pain due to cartilaginous bone lesions with bone destruction, lytic, or blastic lesions with reactive thickening of the cortex or periosteum [24].
The prognosis of ChSs mainly depends on tumor grade and subtype. With respect to overall survival (OS), the ACT and secondary ChSs have a good survival rate, with a 5-year OS in more than 90% of cases [4,25]. Periosteal and conventional ChS subtypes have a good five-year OS (about 68%), followed by clear cell ChS (62.3%). One- and five-year OS in myxoid (64.1%, 49.8%) and mesenchymal (49.2%, 37.6%) subtypes are relatively lower but still better than in dedifferentiated ChSs, with reported five-year survival rates between 11% and 24% [26]. Survival decreases with increasing tumor grade, and the 5-year OS for grade 2 and grade 3 is 75% and 30%, respectively [4]. At initial presentation, only about 6% of ChS patients have a diagnosis of distant metastasis [7]. However, patients with metastatic disease at presentation have a 5-year OS of 28%, with a median OS of only 14 months [27]. Other negative prognostic factors include old age, tumor size, and tumor location in the axial skeleton [4].
At present, conventional radiation therapy is limited to patients with incomplete resection, inoperable tumors, or metastases [28], whereas systemic chemotherapy is only for the treatment of mesenchymal and ddChSs [29]. Nowadays, as discussed in a recent review study by Gilbert A. and colleagues [11], the introduction of hadron therapy seems to be a promising strategy for improving the local control and OS of ChS patients that are surgically inoperable. The chemo- and radio-resistance of ChS cells is mainly due to the presence of either a primary tumor mass or a metastatic niche of cancer stem cells (CSCs) that contributes to genetic and molecular heterogeneity [30]. This CSC subpopulation has been shown to play an important role in tumor progression, chemoresistance, recurrence, and metastasis [31,32]. Moreover, overexpression of the P-glycoprotein involved in the human multidrug resistance gene [33], a dense extracellular matrix, as well as poor vascularity that may establish a hypoxic microenvironment, contribute to the chemo- and radio-resistance of ChSs [34,35].
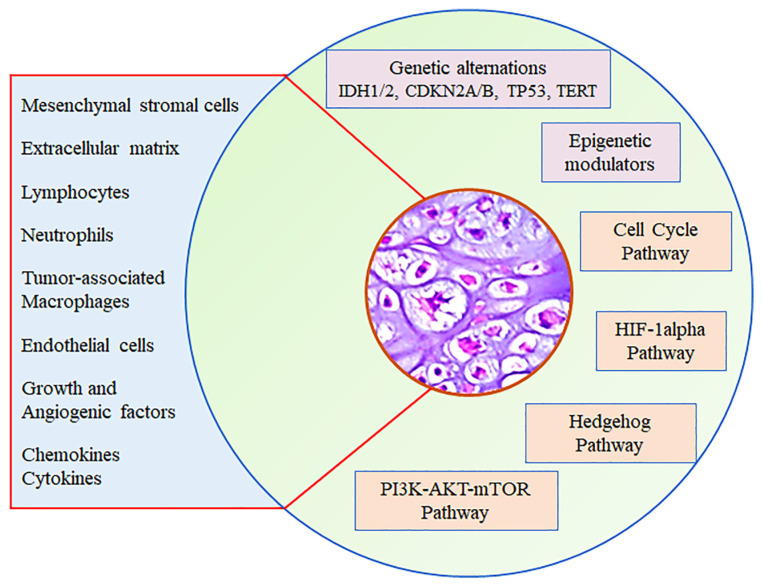
Nowadays, in addition to stem-like characteristics, it has been ascertained that genomic heterogeneity and epigenetic modifications in ChS cells, as well as deregulation of pivotal signal transduction pathways, are implicated in the drug- and radio-resistance of ChS cells [8,30,36]. This provided the rationale for developing new drugs, such as inhibitors of tyrosine kinase, activating mutated genes, mammalian target of rapamycin (mTOR), or histone deacetylase (HDAC) [37] (Figure 1). Unfortunately, most of them, including Hedgehog inhibitors that elicited good pre-clinical responses, failed to demonstrate a survival benefit when translated into clinical trials [38]. Although recent studies have demonstrated good short-term results after intralesional surgical procedures for low-grade ChSs treated with curettage and adjuvant treatments consisting of phenol application, cauterization, or cryotherapy, the primary and preferred treatment for ChS patients with localized disease remains radical surgical resection with wide margins [27,39,40,41]. Of note, for the management of extremity ChSs, amputation is only reserved for patients with extensive and invasive disease [42]. In this regard, the limited efficacy of available treatments combined with the clinical management of extensive and invasive ChSs remain challenging, and new therapeutic approaches are urgently needed [26].
Figure 1.
Schematic representation of multiple mechanisms by which genetic and epigenetic alterations, dysregulated signaling pathways, as well as soluble and cellular components within the TME, sustain the initiation and progression of chondrosarcomas.
In the last two decades, the role of the tumor microenvironment (TME) in governing the fate of solid tumors has been extensively investigated, and it is now clear that tumor cells establish an intricate crosstalk with cellular and soluble components within the TME, favoring tumor growth and progression [43]. In addition to tumor cells, the immune cells of the TME, depending on their nature and functional state, can exert cytotoxic activity, ensuring efficient immune surveillance or promoting tumor progression, making the TME immune-tolerant. The composition of the TME varies between tumor types, and hallmark features include the extracellular matrix (ECM), normal and transformed stromal cells, endothelial cells, and immune cells, as well as soluble components, including cytokines, chemokines, growth, and angiogenic factors (Figure 1) [44].
This review aims to provide insights into the components of the TME that govern ChS growth and progression, highlighting the mechanisms of action and clinical usage of antiangiogenic and immunotherapeutic agents.
2. Tumor Microenvironment
Although relatively few studies have allowed broadening the knowledge of the TME in ChSs, to date, it is clear that a plethora of signaling molecules within the TME cooperate to support ChS progression and metastasis through a variety of mechanisms [30].
2.1. Extracellular Matrix
The microenvironment of ChSs is characterized by a dense, heterogeneous ECM that provides a physical scaffold to support cellular functions and tissue integrity. In low-grade ChSs, the ECM is commonly organized in irregularly shaped and sized lobules separated by fibrous bands, and the occurrence of calcification areas arising from bone destruction suggests their enchondroma origin [5,12,21]. The ECM is a complex network in which several macromolecules, including collagen members, glycoproteins, and proteoglycans, as well as soluble factors, govern its continuous remodeling, playing important roles in angiogenesis, cancer invasion, and metastasis [45]. The ECM is continually remodeled by matrix metalloproteinases (MMPs) [46]. MMP-1, MMP-2, and MMP-13 regulate ECM degradation, and their high expression well correlates with ChS cell invasiveness and progression disease (PD), whereas MMP-9 expression has been documented to be associated with better survival in ChS patients [47,48,49].
Unlike normal chondrocytes that synthesize collagen types II, IX, X, XI, and proteoglycans [50], ChS cells produce a much more compact cartilaginous matrix, consisting of type II collagen, hydrophobic proteoglycans, and hyaluronan, a large negatively charged polysaccharide. In virtue of its tendency to aggregate, hyaluronan forms supramolecular complexes that contribute to the pericellular matrix organization [30]. By comparing hyaluronan content in ChS tissues with normal chondroid tissue as a control, Fede C. and colleagues found that a correlation exists between hyaluronan content, ChS aggressiveness, and chemoresistance [51]. Accordingly, the 4-methylumbellifer that is an inhibitor of hyaluronan synthesis prevents ECM formation, reducing in vitro and in vivo proliferation and invasion of rat ChS cells [52]. Recently, Nota S.P.F.T. and coworkers analyzed the expression levels of the Chondroitin Sulfate ProteoGlycan 4 (CSPG4) in a tissue microarray built with 29 primary conventional and 47 ddChS tissues by immunohistochemistry (IHC). The authors found high expression of CSPG4 in 71% of conventional ChS tissues and in 15% of ddChS tumors. In addition, CSPG4 expression was found to positively correlate with the time to metastasis and the survival of either conventional or ddChS patients [53]. The lysyl oxidase (LOX) family maintains the rigidity and structural stability of the ECM by catalyzing the cross-linking of collagen and elastin. Several studies have shown that aberrant expression of LOX in solid tumors, including ChSs, is associated with tumor progression and metastasis in virtue of the LOX’s ability to alter the ECM, increasing tumor cell migration and invasion [54,55].
Among the cytokines released in the ECM, Interleukin (IL)-1beta contributes to ChS vascularization and progression by inducing ChS cells to secrete the vascular endothelial growth factor (VEGF) [56]. Similarly, the CCL-5 chemokine (otherwise named RANTES) correlates with the tumor stage of ChS patients by inducing VEGF release [57]. All this evidence suggests that perturbation of the ECM is a possible strategy for inhibiting tumor growth and that deregulation of secretory trafficking of ECM molecules may provide viable therapeutic targets for the treatment of ChSs.
2.2. Hypoxia
The hypoxic environment that characterizes ChSs is mainly due to an imbalance occurring between oxygen delivery and consumption [58]. Low oxygen levels activate hypoxia-inducible factors (HIFs), which affect key stages of the ChSs’ metastatic process, including angiogenesis, invasion, tumor cell dormancy, release of extracellular vesicles, and progression of ChS metastases [59]. Low oxygen levels may increase HIF-1alpha expression in both normal and malignant chondrocytes. Analyzing 20 human ChS tissues, Kubo and colleagues found that both HIF-1alpha and HIF-2alpha are expressed in ChS cells and that HIF-1alpha content positively correlates with a shorter disease-free survival (DFS) [60]. Furthermore, a hypoxic microenvironment may induce chemo-resistance by allowing and selecting the survival of CSCs [61]. HIF-1 alpha and HIF-2 alpha factors trigger CSC proliferation via the activation of the PI3K/AKT and NF-κB pathways [62,63], allowing activation of Notch and Hedgehog pathways that maintain the stem potential of tumor cells [11]. Hypoxia also promotes cell invasion through HIF-1alpha, which increases MMP1 and activates the CXCR4/SDF-1 chemokine receptor axis in human ChS cell lines [64]. ChS vascularity increases with an increased histologic grade. This is due to the fact that, under hypoxic conditions, HIF-1alpha increases the release of VEGF, which, in turn, promotes tumor vascularization [65,66]. Kim H. and coworkers have documented that, like HIF-1alpha, HIF-2alpha upregulation is associated with a poor prognosis in high-grade ChS patients by conferring on ChS cells the capacity to grow, invade, and metastasize [67]. Furthermore, Hou S.-M. and coworkers recently found that, when cultured under hypoxic conditions, ChS cells secrete more exosomes that induce the polarization of macrophages into an M2-like phenotype, ultimately promoting a metastatic behavior in ChS tissues [68]. In the future, considering the pivotal role of hypoxia in sustaining the metastatic process, we foresee that the development of HIF-1alpha and/or HIF-2 alpha inhibitors could provide new therapeutic options for patients with metastatic ChS.
2.3. Adipose Tissue
The adipose tissue remodels the ECM through the secretion of MMPs, contributing to the adhesion and migration of ChS cells. Indeed, adiponectin levels released by differentiated adipocytes have been shown to correlate with the tumor stage of human ChSs. Lee HP and coworkers demonstrated that adiponectin induces ChS cells to secrete VEGF-A that, in turn, triggers migration and tube formation of human endothelial progenitor cells through PI3K, Akt, mTOR, and HIF-1alpha pathways [69]. Moreover, Chiu YC and colleagues found that adiponectin-dependent ChS cell migration is mediated by alpha2beta1 integrin upregulation [70]. The other adipocyte-derived cytokine, leptin, is increased in ChS tissues, and its content correlates with the ChS grade [71]. There is also evidence that leptin enhances angiogenesis and lymphangiogenesis in ChSs by promoting the secretion of VEGF-A and VEGF-C factors [72,73].
2.4. Angiogenesis
Although originating from avascular cartilaginous tissues, emerging data from the literature show that an intense microvessel density in ChS tissues is associated with aggressive clinical behavior, higher metastatic potential, and poor progression-free survival (PFS) [74]. Ayala G. and coworkers demonstrated that ChS vascularity increases with increased histologic grade and that blood vessels in ChSs may be located either in peri- or intra-cartilage [65]. Minopoli M. and coworkers investigated the relationship between clinical outcome and tumor vascularization in 18 ChS tissues, including 12 conventional and 6 ddChSs. They found that both ChS histotypes exhibit an appreciable intratumoral microvessel density, with vessels uniformly distributed in ddChSs or mainly localized at the margin of cartilaginous nodules in conventional ChS tissues. Data analysis supports the notion that an inverse correlation between high vascularization and shorter PFS occurs [75]. In addition to the direct and indirect contribution of the ECM and hypoxia in inducing angiogenesis, as already discussed above, other possible mechanisms are involved in ChS vascularization. For instance, Ingangi V. and coworkers demonstrated that peptide inhibitors of cell migration suppress neovascularization and intravasation of human ChS cells injected in nude mice [76,77], with a mechanism involving the urokinase receptor, formyl-peptide receptor type 1, and the integrin alphavbeta3 [78,79]. Another mechanism of angiogenesis in ChSs has been recently described by Cheng C. and colleagues. They found that exosomes derived from ChS cells are able to transport the long non-coding RNA (lncRNA) RAMP2-AS1 into human umbilical vein endothelial cells (HUVECs), promoting VEGFR2-dependent tube formation. Interestingly, the same authors also showed that elevated RAMP2-AS1 levels found in sera from ChS patients correlate with metastatic behavior and poor prognosis, suggesting that exosomal RAMP2-AS1 may be considered a novel biomarker and therapeutic target for ChS patients [80]. In addition to VEGFR, crucial mediators of ChS angiogenesis and metastasis are the platelet-derived growth factor (PDGF) receptors. A positive correlation has been observed between PDGFR-alpha and PDGFR-beta levels and ChS aggressiveness [81]. More recently, Wang CQ and coworkers documented that sphingosine-1-phosphate enhances expression of PDGF-alpha in human ChS cells, inducing angiogenesis through Ras, Raf, MEK, ERK, and AP-1 signaling pathways [82]. Once established the pivotal role of angiogenesis in affecting the PFS of ChS patients, efforts to develop antiangiogenic therapies have provided many agents, including small molecules, tyrosine kinase inhibitors, or fully human monoclonal antibodies such as bevacizumab that inhibit angiogenesis [83], and several of them have been introduced into clinical trials (Table 1). Firstly, imatinib, a multikinase inhibitor targeting PDGFR, was administered to 16 patients with unresectable or metastatic conventional ChSs without exerting meaningful clinical activity in terms of objective response (OR) and PFS lengthening [84]. Later, Phase 2 studies with other multikinase inhibitors, including pazopanib [85], regorafenib [86], and anlotinib [87], all targeting VEGF receptors, were found to improve the DFS of unresectable or metastatic conventional ChSs. Of note, the clinical trial with pazopanib in patients with unresectable or metastatic CS has completed accrual and is currently undergoing analysis for safety and efficacy. Finally, bevacizumab, a recombinant, humanized monoclonal antibody recognizing all four VEGF isoforms, has been included in several clinical trials since it provided beneficial effects when administered in combination with other drugs or as a maintenance regimen (Table 1). These recent results are very promising and will allow the inclusion of VEGF/VEGFR inhibitors in daily practice. Approaches based on the association of treatments are also in development, including clinical trials with chemotherapy and/or immune checkpoint inibitors (ICIs) associated with antiangiogenic agents (Table 1).
2.5. Immune Environment
Despite impressive advances in the understanding of how immune cells govern the progression of solid tumors [88], there are still few, sometimes contradictory, studies describing the immune landscape of ChSs. Nevertheless, increasing evidence seems to prove that a correlation between tumor tissue immunogenicity and the clinical course of the disease occurs in ChSs. In this regard, some authors reported that the immune infiltrate of ChS may include exhausted CD8+ T-cells [89] and that therefore some of these hard-to-treat patients could be treated with immunotherapeutic drugs. Moreover, given the importance of the localization of tumor-infiltrating lymphocytes (TILs) for response to immunotherapy in other solid tumors, it would be crucial to investigate the spatial distribution of these cells in ChS. Some observations exist for conventional (low-grade) tumors, where immune cells frequently localize at the periphery of the cartilaginous islands, probably due to the presence of a dense hyaline ECM that makes the tumor impenetrable [90,91]. Conversely, in some aggressive ddChSs with a more disorganized architecture, immune cells were also found in close proximity and intermingled with tumor cells [91], suggesting a potential responsiveness of these tumors to immunotherapy.
The TME is rich in immune infiltrates, including lymphocytes, neutrophils, natural killer cells, and macrophages. In ChSs, tumor-associated macrophages (TAMs) represent the most abundant immune cell population. TAMs mainly exhibit M2-like phenotypes that promote angiogenesis, extracellular matrix remodeling, cancer cell proliferation, metastasis, and immunosuppression, as well as resistance to chemotherapeutic agents [90,92,93]. Immunosuppression exerted by TAMs is mediated by the secretion of cytokines, chemokines, and MMPs that activate regulatory T-cells (Tregs) and block cytotoxic CD8+ T-cells through the secretion of NO and ROS metabolites [94]. Moreover, TAMs can recruit Tregs through CCL22, which further suppresses the antitumor immune response of T-cells [95]. It has been shown that a high number of CD68+/CD163+ M2-like TAMs positively correlate with metastatic diseases at diagnosis and shorter survival in sarcoma patients [90,92].
By IHC analysis of a large cohort of ChS tissues, including all ChS histotypes, Kostine M. and coworkers found that 85% of ddChSs are highly infiltrated by CD163/CD68-positive TAMs [93]. Accordingly, Simard FA and coauthors, using a swarm rat ChS model, showed that tumor size decreases and tumor progression delays upon either TAM depletion with clodronate-containing liposomes or their conversion into the pro-inflammatory M1 phenotype with mifamurtide [90]. Minopoli M. and coworkers demonstrated in multicellular 3D-organotypic models that human primary ChS cells induce monocyte-derived primary macrophages to acquire a M2-like phenotype, which, in turn, increases the invasive capability of ChS cells [75]. The authors also documented that, when human primary ChS cells were engrafted in nude mice, peptide inhibitors of cell migration reduced tumor size and vascularization by preventing recruitment and infiltration of murine monocytes into the tumor tissue [75]. A retrospective evaluation of a tissue microarray from 52 conventional and 24 ddChS tissues allowed Nota S.P.F.T. and coworkers to ascertain that an initially immunogenic environment may be reverted when ChS cells begin to utilize escape mechanisms that prevent their immune recognition and destruction [96]. TAMs can induce immunosuppression through the expression of the colony stimulating factor 1 receptor (CSF1R), also known as CD115. CSF1R may be engaged by two ligands, both of which are secreted by ChS cells: colony stimulating factor 1 (CSF-1) and interleukin-IL-34. Ligand-engaged CSF1R induces macrophages to release tumorigenic cytokines, including TGF-β and IL-10, which convert the immune microenvironment into a protumoral phenotype [97]. Richert J. and coauthors found that 84.6% of ddChSs exhibit a strong positivity for CSF1R macrophages, whereas in conventional ChSs it was only 64 % [91]. Importantly, CSF1R has been proposed to be an effective target for blocking TAMs that infiltrate the tumor stroma [98].
Several CSF1R inhibitors, including small molecules and neutralizing antibodies, have been developed, and many drugs targeting CSF1R are currently in clinical development or approved for treating solid tumors [99]. Therefore, agents blocking CSF1R could represent a novel therapeutic approach for ChS patients in the future.
Within the TME, Tregs have been shown to promote tumor progression by quenching antitumor immune responses. Furthermore, Treg may indirectly support the survival of cancer cells through their interaction with mesenchymal stromal cells [100].
In order to study the impact of TAMs and TILs on the clinical outcome of ChS patients, Richert J. and colleagues analyzed the phenotype and density of immune cells in 27 conventional and 49 ddChS tissues by IHC and RT-qPCR. The authors confirmed that TAMs are the major immune cell population in ChSs. Furthermore, they documented that the immune infiltrate composition in ddChSs correlates with patient outcome, as high levels of CD68+ macrophages are associated with the presence of metastases at diagnosis, whereas a high density of CD3+ TILs and CD8+ cytotoxic T-cells is associated with a better OS [91]. TAMs may lead to immunosuppression in sarcomas through the expression of several markers, including PD-L1, CD80, CD206, CD163, and CSF1R; the release of cytokines including IL-1, IL-6, IL-8, IL-10, TNF-alpha, TGF-beta, and VEGF; and chemokines such as CCL2, CCL5, CCL18, and MMP-9 [101]. Given the abundance of TAMs and the high level of expression of CSF1R reported, Richert J. and collaborators proposed that immunomodulation with CSF1R inhibitors may be a promising therapeutic approach in both conventional and ddChS ChS subtypes [91].
Discordant results have been reported from a multi-omic analysis of a large cohort of fresh ChS tumors. Based on the integrated data of single-cell CyTOF and flow cytometry, Li B. and coworkers provided the immune characterization of 98 conventional CHS cases. They classified the ChS immune microenvironment into three subtypes: the cluster “granulocytic–myeloid-derived suppressor cell (G-MDSC) dominant”, with a high number of myeloid cells; the immune exhausted cluster, with high exhausted T-cell and dendritic-cell infiltration; and the immune desert cluster, with few immune cells, the last subtype being frequently found in high-grade ChS. The results allowed authors to discourage immunotherapies targeting TAMs since few macrophages and monocytes were found in tumor tissues, while encouraging immunotherapeutic approaches with ICIs for ChS patients bearing the “immune exhausted” subtype [89].
In the TME, following TGF-β stimulation, neutrophils can be polarized toward N1 antitumor or N2 protumor phenotypes [102,103,104]. Like TAMs, tumor-associated neutrophils (TANs) impair antitumor immunity, stimulate angiogenesis, and accelerate tumor growth, invasion, and ultimately metastasis by remodeling the ECM and producing growth factors, including VEGF, a variety of cytokines, and chemokines such as IL-17 and MMP9, as well as the high mobility group box 1 [102,103,104]. Although many studies have evaluated the prognostic value of TANs as well as the neutrophil/lymphocyte ratio in a variety of solid tumors [105], to the best of our knowledge, only a few studies have been conducted in sarcoma, and none of them included ChS cases.
There is increasing evidence that the complement system is activated in various cancer tissues and has a role in the modulation of the tumor microenvironment. For instance, using U2-OS osteosarcoma cells in an in vitro model, Jeon H. and coworkers found that complement activation promotes angiogenesis by increasing the production of the proangiogenic factors FGF1 and VEGF-A [106], suggesting that a linkage between the complement system and angiogenesis exists and that the related mechanisms could be potential targets for ChS treatment.
C3a and its cognate receptor C3aR are key components of the complement system, having a role in tumor promotion and immunosuppression. C3a has been suggested to exert immunosuppressive activity by promoting neutrophil recruitment and CD4+ T-cell activation, as well as changing TAM polarization status and recruitment in a variety of solid tumors [107,108,109]. In this regard, Magrini E. et al. have recently ascertained that the C3a–C3aR axis plays a role in driving immunosuppression and tumor promotion in transplanted sarcoma models [109]. Therefore, the C3a–C3aR complex should be considered as a therapeutic target for counteracting the immunosuppressive activity of the tumor microenvironment.
3. Immunotherapy
The programmed cell death 1 (PD-1) receptor expressed on the surface of activated T-cells recognizes two ligands: PD-L1, otherwise named B7-H1, and PD-L2, otherwise named B7-DC. The interaction of these ligands with the immune checkpoint PD-1 on T-cells induces anergy in lymphocytes, counteracting T-cell-mediated immune responses [110]. When PD-L1 binds to PD-1, an inhibitory signal in T-cells blocks their proliferation and reduces cytokine production. Consequently, either the cytotoxic or the memory potential of CD8+ T-cells are impaired, resulting in the blocking of immune surveillance [111]. In recent years, blocking the PD-1/PD-L1 pathways with ICIs has changed the clinical management and fate of a variety of solid tumors [112]. The best-known ICIs are antibodies targeting PD-1, its ligand PD-L1, as well as the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4). In this regard, the FDA, EMA, and other regulatory agencies approved several ICIs, including monoclonal antibodies targeting PD-1 (nivolumab pembrolizumab), PD-L1 (atezolizumab), and CTLA-4 (ipilimumab). Their impressive ability to elicit antitumor immune responses in solid tumors has encouraged investigations into their potential beneficial effects for ChS patients. Furthermore, aldesleukin, a recombinant form of human interleukin-2 that stimulates proliferation and maturation of the CD8-positive T- and natural killer-cells, has been shown to elicit promising outcomes in advanced solid tumors [113].
PD-1-positive T-cells have been found in both conventional and ddChS tissues [114]. Firstly, some years ago, Kostine M and coworkers, using a large tissue microarray from conventional, mesenchymal, and ddChS tissues, reported that PD-L1 is expressed in about 40% of ddChS tissues while it is absent in conventional and mesenchymal ChS tissues. The authors also found that PD-L1 levels in ddChSs are associated with the number of tumor-infiltrating leukocytes expressing the HLA-1 antigen [93]. Later, Yang X. and colleagues analyzed by IHC the expression levels of PD-L1 and PD-L2 in 40 human ChS tissues. Although the authors did not provide information on the analyzed histotypes, they found that PD-L1 and PD-L2 are expressed in 68% and 42% of ChS tissues, respectively, and that PD-L1 expression levels correlate with shorter survival [115]. Importantly, while PD-L2 expression levels only associate with earlier recurrence and lower age in ChS patients [115], PD-L1 expression has been found to be associated with larger tumor size, advanced tumor grade, earlier recurrence, and shorter OS in ChS patients [116]. Accordingly, a meta-analysis of 39 independent studies on 3680 patients with bone and soft tissue sarcomas, including 81 ChS cases, allowed Wang F. and coworkers to conclude that PD-L1 overexpression correlates with a higher rate of tumor metastasis, a more advanced tumor grade, and higher T lymphocyte infiltration [117].
These observations provided the rationale for testing the clinical efficacy of ICIs in counteracting ChS aggressiveness. Paoluzzi I. and coworkers reported in a retrospective study the first evidence of effects elicited by immunotherapy in ChS patients. The authors analyzed a cohort of 28 patients with relapsed, metastatic, or unresectable soft or bone sarcomas, including one mesenchymal ChS (primary site, skull) and two ddChSs (primary site, lower extremity of buttock). Patients received intravenous administration of 3 mg/kg nivolumab every 2 weeks. Two ChS patients had stable disease, whereas a partial response (PR) to nivolumab was achieved by a 74-year-old male with metastatic ddChS. Interestingly, higher PD-L1 levels in ChS cells from this patient were found, as compared with PD-L1 levels assessed in the tumor cells from the other ChS patients [118]. Subsequently, Tawbi H.A. and coauthors published the first results from the SARC028 clinical trial. This is the first multicenter, prospective Phase 2 study designed in order to examine the efficacy of pembrolizumab in bone and soft tissue sarcomas. Eighty-four patients with histological evidence of metastatic, surgically unresectable, or locally advanced sarcoma received an intravenous infusion of 200 mg pembrolizumab every 3 weeks until PD or unacceptable toxicity was ascertained. PD was assessed using computed tomography or magnetic resonance imaging at baseline, after 8 weeks, and then every 12 weeks until PD. Among 40 patients with bone sarcoma, 5 had ddChSs. For one of them, the response observed was satisfying, as a 50% tumor reduction and stable disease for more than 6 months were achieved. A second patient with ChS had stable disease, whereas the remaining three patients manifested progression disease, with a median follow-up of 18.7 months [119]. Furthermore, the correlative analyses of the SARC028 study documented that a higher density of cytotoxic tumor-infiltrating T-cells correlates with a better PFS [120]. Encouraging results were also derived from a Phase 1/2 nonrandomized clinical trial of doxorubicin plus pembrolizumab combined treatment in patients with advanced sarcoma, including one clear cell ChS, three conventional, and 4 ddChSs. As a result, three ChS patients had tumor regression, including one conventional ChS with a 26% decrease in size, and two ChS patients had durable disease regression, suggesting that doxorubicin plus pembrolizumab may provide benefit to these patients [121]. Further interesting evidence comes from a recent case report of a patient with femoral ddChS, multiple lung nodules, and a pelvic soft tissue mass. At the diagnosis, the authors reported PD-L1 expression in 95% of tumor cells and 20% in tumor-associated immune cells. The patient received 200 mg of pembrolizumab every 3 weeks. After five weeks, computed tomography showed a stable necrotic right pelvic lymph node and resolution of multiple lung nodules. After ten months, the patient reported the presence of an unchanged 4 mm pulmonary nodule and no metastatic disease in the abdomen and pelvis. Surprisingly, although after 29 cycles he discontinued the pembrolizumab treatment due to immunotherapy-induced psoriatic arthritis, the patient continued to have a complete response on surveillance imaging [122].
These aforementioned results evidence that some subtypes of ChSs may effectively benefit from treatment with immunotherapy, especially in the case of high-grade ChSs and ddChSs, as PD-1 inhibitors have been proven to stabilize the disease or elicit partial responses. Although responding patients seem to be associated with a higher number of tumor-infiltrating activated T-cells [89,116], it remains to definitively ascertain the potential predictive values of PD-L1 or PD-1 in ChSs. Based on these results, several clinical trials aimed at investigating the possibility that ChSs may effectively benefit from immunotherapeutic treatments, especially in the case of high-grade ones, have been planned, and most of them are ongoing (Table 1). However, it must be taken into account that most clinical studies include all sarcoma histotypes that should otherwise be considered distinct identities since sarcomas comprise more than a hundred histological subtypes and that, among these, ChSs comprise eight distinct histotypes. Therefore, our ability to select patients who will benefit from immune checkpoint blockade will remain poor until collaborative/multicenter studies allow us to enroll larger cohorts of ChS patients.
Table 1.
Recent, relevant, complete, and ongoing clinical trials with antiangiogenic and/or immune checkpoint inhibitors for ChS patients.
| NCT Number | Clinical Trial | Phase | Drug Class | Drug | Patients (n. ChSs) | Outcomes | Status | Ref. |
|---|---|---|---|---|---|---|---|---|
| NCT00928525 | Open-label trial of imatinib in patients with desmoid tumors and ChSs | 2 | Antiangiogenic MTKI | Imatinib– Mesylate |
34 (16 ChSs) |
Efficacy ORR | Completed | [84] |
| NCT02982486 | Phase II of nivolumab plus ipilimumab in non-resectable sarcoma and endometrial carcinoma | 2 | Anti-PD-L1 Anti-CTLA-4 |
Nivolumab Ipilimumab | Soft and bone sarcomas |
Efficacy Safety PFS OS |
Unknown status | [123] |
| NCT00464620 | Trial of dasatinib in advanced Sarcomas |
2 | Antiangiogenic MTKI | Dasatinib | 109 (33 ChSs) |
ORR CBR RR PFSR OS |
Completed | [124] |
| NCT02301039 | SARC028: A Phase II study of the anti-PD1 antibody pembrolizumab (MK-3475) in patients with advanced sarcomas | 2 | Anti-PD-1 | Pembrolizumab | Soft tissue and bone sarcoma (n. 42 bone sarcomas) |
OR CR PR PFS RR OS |
Completed | [119] |
| NCT03449108 | LN-145 or LN-145-S1 in treating patients with relapsed or refractory ovarian cancer, triple-negative breast cancer (TNBC), anaplastic thyroid cancer, osteosarcoma, or other bone and soft tissue sarcomas | 2 | Recombinant IL-2 BCL2 Inhibitor Anti-PD-1 Anti-CTLA-4 chemotherapy |
Aldesleukin ALN-145 LN-145-S1 Cyclophosphamide Fludarabine Ipilimumab Nivolumab |
30 (bone sarcoma, ddChS, giant cell tumor of bone, and 13 more) | Efficacy Safety ORR DCR PFS PR CR, SD DoR CR PR PD, PFS OS |
Active, not recruiting | [125] |
| NCT03474640 | Safety, tolerability, and pharmacokinetics of an anti-PD-1 monoclonal antibody in subjects with advanced malignancies | 1/2 | Humanized anti-PD-1 | Toripalimab | Bone sarcoma |
Safety Toxicity PFS PFSR OS |
Closed for enrollment | [126] |
| NCT01330966 | Study of pazopanib in the treatment of surgically unresectable or metastatic ChSs | 1/2 | Antiangiogenic MTKI | Pazopanib | 47 ChSs | Safety Efficacy DCR PFS CR SD PD PR OS |
Completed | [85] |
| NCT03277924 | Trial of sunitinib and/or nivolumab plus chemotherapy in advanced soft tissue and bone sarcomas (ImmunoSarc) | 1/2 | Antiangiogenic MTKI Anti-PD-L1 chemotherapy |
Sunitinib Nivolumab Epirubicin |
200 (1 clear cell ChS, 6 dd-ChSs) |
Safety Toxicity PFS OR CR PR OS |
Recruiting | [127] |
| NCT02888665 | Pembrolizumab and doxorubicin hydrochloride in treating patients with sarcomas that are metastatic or cannot be removed by surgery | 1/2 | Anti-PD-1 chemotherapy |
Pembrolizumab Doxorubicin |
Bone sarcomas (1 clear cell ChS, 3 Conventional, and 4 ddChSs) |
ORR CR PR PFS MTD OS |
Completed | [121] |
| NCT02389244 | A Phase II study evaluating the efficacy and safety of regorafenib in patients with metastatic bone sarcomas (REGOBONE) | 2 | Antiangiogenic MTKI | Regorafenib | Metastatic bone sarcomas (46 ChSs) |
PFS | Recruiting | [86] |
| NCT05193188 | A multicenter clinically controlled study of anlotinib combined with PD-1 antibody on unresectable high-grade ChS with different IDH denotypes | 2 | Antiangiogenic MTKI PD-1 Ab |
Anlotinib PD-1 Ab |
high-grade, conventional, and ddChSs |
PFSR ORR DCR |
Recruiting | [87] |
| NCT04055220 | Efficacy and safety of regorafenib as maintenance therapy after first-line treatment in patients with bone sarcomas (REGOSTA) | 2 | Antiangiogenic MTKI | Regorafenib | Bone sarcomas |
Efficacy Safety RFS OS |
Recruiting | [128] |
| NCT04553692 | Phase I study of IGM-8444 as a single agent and in combination with subjects with relapsed and/or refractory solid cancers | 1 | Anti-DR5 Antiangiogenic chemotherapies BCL2 inhibitor |
IGM-8444 Folfiri Bevacizumab Birinapant Venetoclax |
Relapsed, refractory, or newly diagnosed solid cancers, including ChSs |
ORR DoR PFS |
Recruiting | [129,130] |
| NCT03282344 | A study of NKTR-214 in combination with nivolumab in patients with metastatic and/or locally advanced sarcomas | 2 | IL2 pathway agonist, ICI |
NKTR-214 Nivolumab | Metastatic and/or locally advanced soft and bone sarcoma |
PR CR PD SD |
Active, not recruiting | [131] |
| NCT04458922 | Testing atezolizumab in people 2-17 years old with clear cell sarcoma or advanced ChS | 2 | Anti-PD-L1 | Atezolizumab | 27 ChSs | ORR CR PR, PD PFS RR OS |
Active, not recruiting | [132] |
| NCT03414229 | A study of epacadostat, an IDO1 inhibitor, in combination with pembrolizumab in patients with metastatic and/or locally advanced sarcomas | 2 | IDO1 Inhibitor Anti-PD-1 | Epacadostat Pembrolizumab | Metastatic and/or locally advanced sarcoma |
Efficacy Safety ORR PFS RFS OS |
Active, not recruiting | [133] |
Abbreviations: overall response rate (ORR), disease-free survival (DFS), clinical benefit rate (CBR), progression-free survival (PFS), progression-free survival rate (PFSR), disease control rate (DCR), duration of response (DoR), partial response (PR), complete response (CR), stable disease (SD), progressive disease (PD), response rate (RR), relapse-free survival (RFS), maximum tolerated dose (MTD), overall survival (OS).
4. Conclusions and Future Perspectives
Chondrosarcomas (ChSs) are a heterogeneous group of rare, cartilage-forming neoplasms, representing the second most common primary bone malignancy. Today, surgical management remains the main choice of treatments, as ChSs are resistant to both radiation and chemotherapy. Recently, the understanding of the immune landscape of ChSs has opened new immunotherapeutic options that will probably change the standard of care for high-grade ChSs and ddChSs in the future.
Abbreviations
| ACT | Atypical cartilaginous tumor |
| CBR | clinical benefit rate |
| ChS | Chondrosarcoma |
| CR | Complete response |
| CSC | Cancer stem cell |
| CSF1R | Colony stimulating factor 1 receptor |
| CSPG4 | ProteoGlycan 4 |
| DCR | Disease control rate |
| ddChS Dedifferentiated ChS | |
| DFS | Disease-free survival |
| DoR | Duration of response |
| ECM | Extracellular matrix |
| HDAC | Histone deacetylase |
| HIF | Hypoxia-inducible factor |
| ICI | Immune checkpoint inhibitor |
| IHC | Immunohistochemistry |
| IL | Interleukin |
| LOX | Lysyl oxidase |
| Matrix metalloproteinases | |
| MTD | Maximum tolerated dose |
| mTOR | Mammalian target of rapamycin |
| OR | Objective response |
| ORR | Overall response rate |
| OS | Overall survival |
| PD | Progressive disease |
| PDGF | Platelet-derived growth factor |
| PFS | Progression-free survival |
| PFSR | Progression-free survival rate |
| PR | Partial response |
| RFS | Relapse-free survival |
| RR | Response rate |
| SD | Stable disease |
| TAMs | Tumor-associated macrophages |
| TANs | Tumor-associated neutrophils |
| TME | Tumor microenvironment |
| Human umbilical vein endothelial cells | |
| VEGF | Vascular endothelial growth factor |
Author Contributions
Conceptualization, V.I., M.M., M.V.C. and I.M.; extensive literature searches, V.I., M.M., M.G., G.D.C., A.C. and E.P.; manuscript writing, V.I., M.M., M.V.C. and A.D.C.; critical revision of the work and final version approval, V.I., A.D.C., G.F., M.G., A.C., F.F., G.D.C., E.P., I.M., M.V.C. and M.M. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Funding Statement
This work was funded by the Italian Ministry of Health “Ricerca Corrente” project #2611752, “RITMES”, and by Alleanza Contro il Cancro Italian Network-Working Group Sarcomas: Project RCR-2021- WP13, funded by the Italian Ministry of Health.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Rozeman L.B., Cleton-Jansen A.M., Hogendoorn P.C.W. Pathology of Primary Malignant Bone and Cartilage Tumours. Int. Orthop. 2006;30:437–444. doi: 10.1007/s00264-006-0212-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hogendoorn P.C.W., Bovée J., Nielsen G.P., Fletcher C.D.M., Bridge J.A., Hogendoorn P.C.W., Mertens F. WHO Classification of Tumours of Soft Tissue and Bone. 4th ed. IARC; Lyon, France: 2013. Chondrosarcoma (grades I–III), including primary and secondary variants and periosteal chondrosarcoma; pp. 264–268. [Google Scholar]
- 3.Giuffrida A.Y., Burgueno J.E., Koniaris L.G., Gutierrez J.C., Duncan R., Scully S.P. Chondrosarcoma in the United States (1973 to 2003): An Analysis of 2890 Cases from the SEER Database. J. Bone Jt. Surg. Am. 2009;91:1063–1072. doi: 10.2106/JBJS.H.00416. [DOI] [PubMed] [Google Scholar]
- 4.Van Praag Veroniek V.M., Rueten-Budde A.J., Ho V., Dijkstra P.D.S., Study Group Bone and Soft Tissue Tumours (WeBot) Fiocco M., van de Sande M.A.J. Incidence, Outcomes and Prognostic Factors during 25 Years of Treatment of Chondrosarcomas. Surg. Oncol. 2018;27:402–408. doi: 10.1016/j.suronc.2018.05.009. [DOI] [PubMed] [Google Scholar]
- 5.Wells M.E., Childs B.R., Eckhoff M.D., Rajani R., Potter B.K., Polfer E.M. Atypical Cartilaginous Tumors: Trends in Management. J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 2021;5:e21.00277. doi: 10.5435/JAAOSGlobal-D-21-00277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Thorkildsen J., Taksdal I., Bjerkehagen B., Haugland H.K., Børge Johannesen T., Viset T., Norum O.-J., Bruland Ø., Zaikova O. Chondrosarcoma in Norway 1990–2013; an Epidemiological and Prognostic Observational Study of a Complete National Cohort. Acta Oncol. 2019;58:273–282. doi: 10.1080/0284186X.2018.1554260. [DOI] [PubMed] [Google Scholar]
- 7.Damron T.A., Ward W.G., Stewart A. Osteosarcoma, Chondrosarcoma, and Ewing’s Sarcoma: National Cancer Data Base Report. Clin. Orthop. Relat. Res. 2007;459:40–47. doi: 10.1097/BLO.0b013e318059b8c9. [DOI] [PubMed] [Google Scholar]
- 8.Zając A.E., Kopeć S., Szostakowski B., Spałek M.J., Fiedorowicz M., Bylina E., Filipowicz P., Szumera-Ciećkiewicz A., Tysarowski A., Czarnecka A.M., et al. Chondrosarcoma-from Molecular Pathology to Novel Therapies. Cancers. 2021;13:2390. doi: 10.3390/cancers13102390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim J.-H., Lee S.K. Classification of Chondrosarcoma: From Characteristic to Challenging Imaging Findings. Cancers. 2023;15:1703. doi: 10.3390/cancers15061703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tosun I., Naderi S. Approach to Primary Vertebral Tumors in the Light of the 2020 Updated World Health Organization Classification of Bone Tumors. Turk. Neurosurg. 2023;33:1–9. doi: 10.5137/1019-5149.JTN.36208-21.2. [DOI] [PubMed] [Google Scholar]
- 11.Gilbert A., Tudor M., Montanari J., Commenchail K., Savu D.I., Lesueur P., Chevalier F. Chondrosarcoma Resistance to Radiation Therapy: Origins and Potential Therapeutic Solutions. Cancers. 2023;15:1962. doi: 10.3390/cancers15071962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Limaiem F., Davis D.D., Sticco K.L. StatPearls. StatPearls Publishing; Treasure Island, FL, USA: 2024. Chondrosarcoma. [PubMed] [Google Scholar]
- 13.WHO Classification of Tumours Editorial Board . Soft Tissue and Bone Tumours. IARC; Lyon, France: 2013. [Google Scholar]
- 14.Choi J.H., Ro J.Y. The 2020 WHO Classification of Tumors of Bone: An Updated Review. Adv. Anat. Pathol. 2021;28:119–138. doi: 10.1097/PAP.0000000000000293. [DOI] [PubMed] [Google Scholar]
- 15.Zając A., Król S.K., Rutkowski P., Czarnecka A.M. Biological Heterogeneity of Chondrosarcoma: From (Epi) Genetics through Stemness and Deregulated Signaling to Immunophenotype. Cancers. 2021;13:1317. doi: 10.3390/cancers13061317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Douis H., Saifuddin A. The Imaging of Cartilaginous Bone Tumours. II. Chondrosarcoma. Skelet. Radiol. 2013;42:611–626. doi: 10.1007/s00256-012-1521-3. [DOI] [PubMed] [Google Scholar]
- 17.Righi A., Pacheco M., Cocchi S., Asioli S., Gambarotti M., Donati D.M., Evangelista A., Gnoli M., Locatelli M., Mordenti M., et al. Secondary Peripheral Chondrosarcoma Arising in Solitary Osteochondroma: Variables Influencing Prognosis and Survival. Orphanet J. Rare Dis. 2022;17:74. doi: 10.1186/s13023-022-02210-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nakagawa M., Endo M., Susuki Y., Yokoyama N., Maekawa A., Nabeshima A., Iida K., Fujiwara T., Setsu N., Matsunobu T., et al. Clinical, Radiological, and Histopathological Characteristics of Periosteal Chondrosarcoma with a Focus on the Frequency of Medullary Invasion. J. Clin. Med. 2022;11:2062. doi: 10.3390/jcm11072062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Miao R., Choy E., Raskin K.A., Schwab J.H., Nielsen G.P., Deshpande V., Chebib I., DeLaney T.F., Hornicek F.J., Cote G.M., et al. Prognostic Factors in Dedifferentiated Chondrosarcoma: A Retrospective Analysis of a Large Series Treated at a Single Institution. Sarcoma. 2019;2019:9069272. doi: 10.1155/2019/9069272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dahlin D.C., Beabout J.W. Dedifferentiation of Low-Grade Chondrosarcomas. Cancer. 1971;28:461–466. doi: 10.1002/1097-0142(197108)28:2<461::aid-cncr2820280227>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 21.Murphey M.D., Walker E.A., Wilson A.J., Kransdorf M.J., Temple H.T., Gannon F.H. From the Archives of the AFIP: Imaging of Primary Chondrosarcoma: Radiologic-Pathologic Correlation. Radiographics. 2003;23:1245–1278. doi: 10.1148/rg.235035134. [DOI] [PubMed] [Google Scholar]
- 22.Swanson P.E., Lillemoe T.J., Manivel J.C., Wick M.R. Mesenchymal Chondrosarcoma. An Immunohistochemical Study. Arch. Pathol. Lab. Med. 1990;114:943–948. [PubMed] [Google Scholar]
- 23.Kumar R., David R., Cierney G. Clear Cell Chondrosarcoma. Radiology. 1985;154:45–48. doi: 10.1148/radiology.154.1.3964950. [DOI] [PubMed] [Google Scholar]
- 24.Amer K.M., Munn M., Congiusta D., Abraham J.A., Basu Mallick A. Survival and Prognosis of Chondrosarcoma Subtypes: SEER Database Analysis. J. Orthop. Res. 2020;38:311–319. doi: 10.1002/jor.24463. [DOI] [PubMed] [Google Scholar]
- 25.Lin P.P., Moussallem C.D., Deavers M.T. Secondary Chondrosarcoma. J. Am. Acad. Orthop. Surg. 2010;18:608–615. doi: 10.5435/00124635-201010000-00004. [DOI] [PubMed] [Google Scholar]
- 26.Gazendam A., Popovic S., Parasu N., Ghert M. Chondrosarcoma: A Clinical Review. J. Clin. Med. 2023;12:2506. doi: 10.3390/jcm12072506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang Z., Chen G., Chen X., Huang X., Liu M., Pan W., Yan X., Lin N., Ye Z. Predictors of the Survival of Patients with Chondrosarcoma of Bone and Metastatic Disease at Diagnosis. J. Cancer. 2019;10:2457–2463. doi: 10.7150/jca.30388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Catanzano A.A., Kerr D.L., Lazarides A.L., Dial B.L., Lane W.O., Blazer D.G., Larrier N.A., Kirsch D.G., Brigman B.E., Eward W.C. Revisiting the Role of Radiation Therapy in Chondrosarcoma: A National Cancer Database Study. Sarcoma. 2019;2019:4878512. doi: 10.1155/2019/4878512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Italiano A., Mir O., Cioffi A., Palmerini E., Piperno-Neumann S., Perrin C., Chaigneau L., Penel N., Duffaud F., Kurtz J.E., et al. Advanced Chondrosarcomas: Role of Chemotherapy and Survival. Ann. Oncol. 2013;24:2916–2922. doi: 10.1093/annonc/mdt374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Walter S.G., Knöll P., Eysel P., Quaas A., Gaisendrees C., Nißler R., Hieggelke L. Molecular In-Depth Characterization of Chondrosarcoma for Current and Future Targeted Therapies. Cancers. 2023;15:2556. doi: 10.3390/cancers15092556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Boeuf S., Kunz P., Hennig T., Lehner B., Hogendoorn P., Bovée J., Richter W. A Chondrogenic Gene Expression Signature in Mesenchymal Stem Cells Is a Classifier of Conventional Central Chondrosarcoma. J. Pathol. 2008;216:158–166. doi: 10.1002/path.2389. [DOI] [PubMed] [Google Scholar]
- 32.Martínez-Delgado P., Lacerenza S., Obrador-Hevia A., Lopez-Alvarez M., Mondaza-Hernandez J.L., Blanco-Alcaina E., Sanchez-Bustos P., Hindi N., Moura D.S., Martin-Broto J. Cancer Stem Cells in Soft-Tissue Sarcomas. Cells. 2020;9:1449. doi: 10.3390/cells9061449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Terek R.M., Schwartz G.K., Devaney K., Glantz L., Mak S., Healey J.H., Albino A.P. Chemotherapy and P-Glycoprotein Expression in Chondrosarcoma. J. Orthop. Res. 1998;16:585–590. doi: 10.1002/jor.1100160510. [DOI] [PubMed] [Google Scholar]
- 34.Jeong W., Kim H.-J. Biomarkers of Chondrosarcoma. J. Clin. Pathol. 2018;71:579–583. doi: 10.1136/jclinpath-2018-205071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang H., Jiang H., Van De Gucht M., De Ridder M. Hypoxic Radioresistance: Can ROS Be the Key to Overcome It? Cancers. 2019;11:112. doi: 10.3390/cancers11010112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bereza M., Dembiński M., Zając A.E., Piątkowski J., Dudzisz-Śledź M., Rutkowski P., Czarnecka A.M. Epigenetic Abnormalities in Chondrosarcoma. Int. J. Mol. Sci. 2023;24:4539. doi: 10.3390/ijms24054539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Miwa S., Yamamoto N., Hayashi K., Takeuchi A., Igarashi K., Tsuchiya H. Therapeutic Targets and Emerging Treatments in Advanced Chondrosarcoma. Int. J. Mol. Sci. 2022;23:1096. doi: 10.3390/ijms23031096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wood G.E., Graves L.A., Rubin E.M., Reed D.R., Riedel R.F., Strauss S.J. Bad to the Bone: Emerging Approaches to Aggressive Bone Sarcomas. Am. Soc. Clin. Oncol. Educ. Book. 2023;43:e390306. doi: 10.1200/EDBK_390306. [DOI] [PubMed] [Google Scholar]
- 39.Schwab J.H., Wenger D., Unni K., Sim F.H. Does Local Recurrence Impact Survival in Low-Grade Chondrosarcoma of the Long Bones? Clin. Orthop. Relat. Res. 2007;462:175–180. doi: 10.1097/BLO.0b013e3180caac2c. [DOI] [PubMed] [Google Scholar]
- 40.Shemesh S.S., Pretell-Mazzini J., Quartin P.A.J., Rutenberg T.F., Conway S.A. Surgical Treatment of Low-Grade Chondrosarcoma Involving the Appendicular Skeleton: Long-Term Functional and Oncological Outcomes. Arch. Orthop. Trauma. Surg. 2019;139:1659–1666. doi: 10.1007/s00402-019-03184-w. [DOI] [PubMed] [Google Scholar]
- 41.Görgün B., Özşahin M.K., Tok O., Davulcu C.D., Karaismailoğlu B., Hız M. Intralesional Curettage and Cementation of Low-Grade Chondrosarcomas of the Appendicular Skeleton: Long-Term Results from a Single Center. Acta Orthop. Traumatol. Turc. 2022;56:402–407. doi: 10.5152/j.aott.2022.22091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Erstad D.J., Ready J., Abraham J., Ferrone M.L., Bertagnolli M.M., Baldini E.H., Raut C.P. Amputation for Extremity Sarcoma: Contemporary Indications and Outcomes. Ann. Surg. Oncol. 2018;25:394–403. doi: 10.1245/s10434-017-6240-5. [DOI] [PubMed] [Google Scholar]
- 43.Mayer S., Milo T., Isaacson A., Halperin C., Miyara S., Stein Y., Lior C., Pevsner-Fischer M., Tzahor E., Mayo A., et al. The Tumor Microenvironment Shows a Hierarchy of Cell-Cell Interactions Dominated by Fibroblasts. Nat. Commun. 2023;14:5810. doi: 10.1038/s41467-023-41518-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Anderson N.M., Simon M.C. The Tumor Microenvironment. Curr. Biol. 2020;30:R921–R925. doi: 10.1016/j.cub.2020.06.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bonnans C., Chou J., Werb Z. Remodelling the Extracellular Matrix in Development and Disease. Nat. Rev. Mol. Cell Biol. 2014;15:786–801. doi: 10.1038/nrm3904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hua H., Li M., Luo T., Yin Y., Jiang Y. Matrix Metalloproteinases in Tumorigenesis: An Evolving Paradigm. Cell Mol. Life Sci. 2011;68:3853–3868. doi: 10.1007/s00018-011-0763-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Malcherczyk D., Heyse T.J., El-Zayat B.F., Kunzke V., Moll R., Fuchs-Winkelmann S., Paletta J.R.J. Expression of MMP-9 Decreases Metastatic Potential of Chondrosarcoma: An Immunohistochemical Study. BMC Musculoskelet. Disord. 2018;19:9. doi: 10.1186/s12891-017-1920-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yao M., Wang X., Zhao Y., Wang X., Gao F. Expression of MMPs Is Dependent on the Activity of Mitogen-Activated Protein Kinase in Chondrosarcoma. Mol. Med. Rep. 2017;15:915–921. doi: 10.3892/mmr.2016.6077. [DOI] [PubMed] [Google Scholar]
- 49.Higuchi T., Takeuchi A., Munesue S., Yamamoto N., Hayashi K., Kimura H., Miwa S., Inatani H., Shimozaki S., Kato T., et al. Anti-tumor Effects of a Nonsteroidal Anti-inflammatory Drug Zaltoprofen on Chondrosarcoma via Activating Peroxisome Proliferator-activated Receptor Gamma and Suppressing Matrix Metalloproteinase-2 Expression. Cancer Med. 2018;7:1944–1954. doi: 10.1002/cam4.1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Söderström M., Böhling T., Ekfors T., Nelimarkka L., Aro H.T., Vuorio E. Molecular Profiling of Human Chondrosarcomas for Matrix Production and Cancer Markers. Int. J. Cancer. 2002;100:144–151. doi: 10.1002/ijc.10457. [DOI] [PubMed] [Google Scholar]
- 51.Fede C., Stecco C., Angelini A., Fan C., Belluzzi E., Pozzuoli A., Ruggieri P., De Caro R. Variations in Contents of Hyaluronan in the Peritumoral Micro-Environment of Human Chondrosarcoma. J. Orthop. Res. 2019;37:503–509. doi: 10.1002/jor.24176. [DOI] [PubMed] [Google Scholar]
- 52.Hamada S., Nishida Y., Zhuo L., Shinomura T., Ikuta K., Arai E., Koike H., Kimata K., Ushida T., Ishiguro N. Suppression of Hyaluronan Synthesis Attenuates the Tumorigenicity of Low-Grade Chondrosarcoma. J. Orthop. Res. 2018;36:1573–1580. doi: 10.1002/jor.23794. [DOI] [PubMed] [Google Scholar]
- 53.Nota S.P.F.T., Osei-Hwedieh D.O., Drum D.L., Wang X., Sabbatino F., Ferrone S., Schwab J.H. Chondroitin Sulfate Proteoglycan 4 Expression in Chondrosarcoma: A Potential Target for Antibody-Based Immunotherapy. Front. Oncol. 2022;12:939166. doi: 10.3389/fonc.2022.939166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Johnston K.A., Lopez K.M. Lysyl Oxidase in Cancer Inhibition and Metastasis. Cancer Lett. 2018;417:174–181. doi: 10.1016/j.canlet.2018.01.006. [DOI] [PubMed] [Google Scholar]
- 55.Tzeng H.-E., Lin S.-L., Thadevoos L.A., Lien M.-Y., Yang W.-H., Ko C.-Y., Lin C.-Y., Huang Y.-W., Liu J.-F., Fong Y.-C., et al. Nerve Growth Factor Promotes Lysyl Oxidase-Dependent Chondrosarcoma Cell Metastasis by Suppressing miR-149-5p Synthesis. Cell Death Dis. 2021;12:1101. doi: 10.1038/s41419-021-04392-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kalinski T. Inflammation as molecular target in chondrosarcoma. Pathologe. 2014;35((Suppl. S2)):249–253. doi: 10.1007/s00292-014-1968-1. [DOI] [PubMed] [Google Scholar]
- 57.Liu G.-T., Huang Y.-L., Tzeng H.-E., Tsai C.-H., Wang S.-W., Tang C.-H. CCL5 Promotes Vascular Endothelial Growth Factor Expression and Induces Angiogenesis by Down-Regulating miR-199a in Human Chondrosarcoma Cells. Cancer Lett. 2015;357:476–487. doi: 10.1016/j.canlet.2014.11.015. [DOI] [PubMed] [Google Scholar]
- 58.Höckel M., Vaupel P. Tumor Hypoxia: Definitions and Current Clinical, Biologic, and Molecular Aspects. J. Natl. Cancer Inst. 2001;93:266–276. doi: 10.1093/jnci/93.4.266. [DOI] [PubMed] [Google Scholar]
- 59.Di Pompo G., Cortini M., Baldini N., Avnet S. Acid Microenvironment in Bone Sarcomas. Cancers. 2021;13:3848. doi: 10.3390/cancers13153848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kubo T., Sugita T., Shimose S., Matsuo T., Arihiro K., Ochi M. Expression of Hypoxia-Inducible Factor-1alpha and Its Relationship to Tumour Angiogenesis and Cell Proliferation in Cartilage Tumours. J. Bone Jt. Surg. Br. 2008;90:364–370. doi: 10.1302/0301-620X.90B3.19806. [DOI] [PubMed] [Google Scholar]
- 61.Sun X., Lv X., Yan Y., Zhao Y., Ma R., He M., Wei M. Hypoxia-Mediated Cancer Stem Cell Resistance and Targeted Therapy. Biomed. Pharmacother. 2020;130:110623. doi: 10.1016/j.biopha.2020.110623. [DOI] [PubMed] [Google Scholar]
- 62.Marhold M., Tomasich E., El-Gazzar A., Heller G., Spittler A., Horvat R., Krainer M., Horak P. HIF1α Regulates mTOR Signaling and Viability of Prostate Cancer Stem Cells. Mol. Cancer Res. 2015;13:556–564. doi: 10.1158/1541-7786.MCR-14-0153-T. [DOI] [PubMed] [Google Scholar]
- 63.Wu S.-L., Li Y.-J., Liao K., Shi L., Zhang N., Liu S., Hu Y.-Y., Li S.-L., Wang Y. 2-Methoxyestradiol Inhibits the Proliferation and Migration and Reduces the Radioresistance of Nasopharyngeal Carcinoma CNE-2 Stem Cells via NF-κB/HIF-1 Signaling Pathway Inactivation and EMT Reversal. Oncol. Rep. 2017;37:793–802. doi: 10.3892/or.2016.5319. [DOI] [PubMed] [Google Scholar]
- 64.Sun X., Wei L., Chen Q., Terek R.M. CXCR4/SDF1 Mediate Hypoxia Induced Chondrosarcoma Cell Invasion through ERK Signaling and Increased MMP1 Expression. Mol. Cancer. 2010;9:17. doi: 10.1186/1476-4598-9-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ayala G., Liu C., Nicosia R., Horowitz S., Lackman R. Microvasculature and VEGF Expression in Cartilaginous Tumors. Hum. Pathol. 2000;31:341–346. doi: 10.1016/s0046-8177(00)80248-8. [DOI] [PubMed] [Google Scholar]
- 66.Lin C., McGough R., Aswad B., Block J.A., Terek R. Hypoxia Induces HIF-1alpha and VEGF Expression in Chondrosarcoma Cells and Chondrocytes. J. Orthop. Res. 2004;22:1175–1181. doi: 10.1016/j.orthres.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 67.Kim H., Cho Y., Kim H.-S., Kang D., Cheon D., Kim Y.-J., Chang M.J., Lee K.M., Chang C.B., Kang S.-B., et al. A System-Level Approach Identifies HIF-2α as a Critical Regulator of Chondrosarcoma Progression. Nat. Commun. 2020;11:5023. doi: 10.1038/s41467-020-18817-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hou S.-M., Lin C.-Y., Fong Y.-C., Tang C.-H. Hypoxia-Regulated Exosomes Mediate M2 Macrophage Polarization and Promote Metastasis in Chondrosarcoma. Aging. 2023;15:13163–13175. doi: 10.18632/aging.205230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lee H.-P., Lin C.-Y., Shih J.-S., Fong Y.-C., Wang S.-W., Li T.-M., Tang C.-H. Adiponectin Promotes VEGF-A-Dependent Angiogenesis in Human Chondrosarcoma through PI3K, Akt, mTOR, and HIF-α Pathway. Oncotarget. 2015;6:36746–36761. doi: 10.18632/oncotarget.5479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chiu Y.-C., Shieh D.-C., Tong K.-M., Chen C.-P., Huang K.-C., Chen P.-C., Fong Y.-C., Hsu H.-C., Tang C.-H. Involvement of AdipoR Receptor in Adiponectin-Induced Motility and Alpha2beta1 Integrin Upregulation in Human Chondrosarcoma Cells. Carcinogenesis. 2009;30:1651–1659. doi: 10.1093/carcin/bgp156. [DOI] [PubMed] [Google Scholar]
- 71.Yang S.-N., Chen H.-T., Tsou H.-K., Huang C.-Y., Yang W.-H., Su C.-M., Fong Y.-C., Tseng W.-P., Tang C.-H. Leptin Enhances Cell Migration in Human Chondrosarcoma Cells through OBRl Leptin Receptor. Carcinogenesis. 2009;30:566–574. doi: 10.1093/carcin/bgp023. [DOI] [PubMed] [Google Scholar]
- 72.Yang W.-H., Chen J.-C., Hsu K.-H., Lin C.-Y., Wang S.-W., Wang S.-J., Chang Y.-S., Tang C.-H. Leptin Increases VEGF Expression and Enhances Angiogenesis in Human Chondrosarcoma Cells. Biochim. Biophys. Acta (BBA)–Gen. Subj. 2014;1840:3483–3493. doi: 10.1016/j.bbagen.2014.09.012. [DOI] [PubMed] [Google Scholar]
- 73.Yang W.-H., Chang A.-C., Wang S.-W., Wang S.-J., Chang Y.-S., Chang T.-M., Hsu S.-K., Fong Y.-C., Tang C.-H. Leptin Promotes VEGF-C Production and Induces Lymphangiogenesis by Suppressing miR-27b in Human Chondrosarcoma Cells. Sci. Rep. 2016;6:28647. doi: 10.1038/srep28647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.McGough R.L., Aswad B.I., Terek R.M. Pathologic Neovascularization in Cartilage Tumors. Clin. Orthop. Relat. Res. 2002;397:76–82. doi: 10.1097/00003086-200204000-00011. [DOI] [PubMed] [Google Scholar]
- 75.Minopoli M., Sarno S., Di Carluccio G., Azzaro R., Costantini S., Fazioli F., Gallo M., Apice G., Cannella L., Rea D., et al. Inhibiting Monocyte Recruitment to Prevent the Pro-Tumoral Activity of Tumor-Associated Macrophages in Chondrosarcoma. Cells. 2020;9:1062. doi: 10.3390/cells9041062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ingangi V., Bifulco K., Yousif A.M., Ragone C., Motti M.L., Rea D., Minopoli M., Botti G., Scognamiglio G., Fazioli F., et al. The Urokinase Receptor-Derived Cyclic Peptide [SRSRY] Suppresses Neovascularization and Intravasation of Osteosarcoma and Chondrosarcoma Cells. Oncotarget. 2016;7:54474–54487. doi: 10.18632/oncotarget.9976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Carriero M.V., Bifulco K., Ingangi V., Costantini S., Botti G., Ragone C., Minopoli M., Motti M.L., Rea D., Scognamiglio G., et al. Retro-Inverso Urokinase Receptor Antagonists for the Treatment of Metastatic Sarcomas. Sci. Rep. 2017;7:1312. doi: 10.1038/s41598-017-01425-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bifulco K., Longanesi-Cattani I., Gala M., DI Carluccio G., Masucci M.T., Pavone V., Lista L., Arra C., Stoppelli M.P., Carriero M.V. The Soluble Form of Urokinase Receptor Promotes Angiogenesis through Its Ser88-Arg-Ser-Arg-Tyr92 Chemotactic Sequence. J. Thromb. Haemost. 2010;8:2789–2799. doi: 10.1111/j.1538-7836.2010.04075.x. [DOI] [PubMed] [Google Scholar]
- 79.Minopoli M., Polo A., Ragone C., Ingangi V., Ciliberto G., Pessi A., Sarno S., Budillon A., Costantini S., Carriero M.V. Structure-Function Relationship of an Urokinase Receptor-Derived Peptide Which Inhibits the Formyl Peptide Receptor Type 1 Activity. Sci. Rep. 2019;9:12169. doi: 10.1038/s41598-019-47900-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cheng C., Zhang Z., Cheng F., Shao Z. Exosomal lncRNA RAMP2-AS1 Derived from Chondrosarcoma Cells Promotes Angiogenesis Through miR-2355-5p/VEGFR2 Axis. OncoTargets Ther. 2020;13:3291–3301. doi: 10.2147/OTT.S244652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sulzbacher I., Birner P., Trieb K., Mühlbauer M., Lang S., Chott A. Platelet-Derived Growth Factor-Alpha Receptor Expression Supports the Growth of Conventional Chondrosarcoma and Is Associated with Adverse Outcome. Am. J. Surg. Pathol. 2001;25:1520–1527. doi: 10.1097/00000478-200112000-00008. [DOI] [PubMed] [Google Scholar]
- 82.Wang C.-Q., Lin C.-Y., Huang Y.-L., Wang S.-W., Wang Y., Huang B.-F., Lai Y.-W., Weng S.-L., Fong Y.-C., Tang C.-H., et al. Sphingosine-1-Phosphate Promotes PDGF-Dependent Endothelial Progenitor Cell Angiogenesis in Human Chondrosarcoma Cells. Aging. 2019;11:11040–11053. doi: 10.18632/aging.102508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zafar S.Y., Malin J.L., Grambow S.C., Abbott D.H., Schrag D., Kolimaga J.T., Zullig L.L., Weeks J.C., Fouad M.N., Ayanian J.Z., et al. Early Dissemination of Bevacizumab for Advanced Colorectal Cancer: A Prospective Cohort Study. BMC Cancer. 2011;11:354. doi: 10.1186/1471-2407-11-354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Grignani G., Palmerini E., Stacchiotti S., Boglione A., Ferraresi V., Frustaci S., Comandone A., Casali P.G., Ferrari S., Aglietta M. A Phase 2 Trial of Imatinib Mesylate in Patients with Recurrent Nonresectable Chondrosarcomas Expressing Platelet-Derived Growth Factor Receptor-α or -β: An Italian Sarcoma Group Study. Cancer. 2011;117:826–831. doi: 10.1002/cncr.25632. [DOI] [PubMed] [Google Scholar]
- 85.Chow W., Frankel P., Ruel C., Araujo D.M., Milhem M., Okuno S., Hartner L., Undevia S., Staddon A. Results of a Prospective Phase 2 Study of Pazopanib in Patients with Surgically Unresectable or Metastatic Chondrosarcoma. Cancer. 2020;126:105–111. doi: 10.1002/cncr.32515. [DOI] [PubMed] [Google Scholar]
- 86.Duffaud F., Italiano A., Bompas E., Rios M., Penel N., Mir O., Piperno-Neumann S., Chevreau C., Delcambre C., Bertucci F., et al. Efficacy and Safety of Regorafenib in Patients with Metastatic or Locally Advanced Chondrosarcoma: Results of a Non-Comparative, Randomised, Double-Blind, Placebo Controlled, Multicentre Phase II Study. Eur. J. Cancer. 2021;150:108–118. doi: 10.1016/j.ejca.2021.03.039. [DOI] [PubMed] [Google Scholar]
- 87.Liu Z., Gao S., Zhu L., Wang J., Zhang P., Li P., Zhang F., Yao W. Efficacy and Safety of Anlotinib in Patients with Unresectable or Metastatic Bone Sarcoma: A Retrospective Multiple Institution Study. Cancer Med. 2021;10:7593–7600. doi: 10.1002/cam4.4286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhang Y., Zhang Z. The History and Advances in Cancer Immunotherapy: Understanding the Characteristics of Tumor-Infiltrating Immune Cells and Their Therapeutic Implications. Cell Mol. Immunol. 2020;17:807–821. doi: 10.1038/s41423-020-0488-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Li B., Li G., Yan X., Zhu D., Lin P.P., Wang Z., Qu H., He X., Fu Y., Zhu X., et al. Fresh Tissue Multi-Omics Profiling Reveals Immune Classification and Suggests Immunotherapy Candidates for Conventional Chondrosarcoma. Clin. Cancer Res. 2021;27:6543–6558. doi: 10.1158/1078-0432.CCR-21-1893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Simard F.A., Richert I., Vandermoeten A., Decouvelaere A.-V., Michot J.-P., Caux C., Blay J.-Y., Dutour A. Description of the Immune Microenvironment of Chondrosarcoma and Contribution to Progression. Oncoimmunology. 2017;6:e1265716. doi: 10.1080/2162402X.2016.1265716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Iseulys R., Anne G.-B., Corinne B., Gonzague D.B.D.P., Marie K., Jean-Yves B., Aurélie D. The Immune Landscape of Chondrosarcoma Reveals an Immunosuppressive Environment in the Dedifferentiated Subtypes and Exposes CSFR1+ Macrophages as a Promising Therapeutic Target. J. Bone Oncol. 2020;20:100271. doi: 10.1016/j.jbo.2019.100271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Mantovani A., Allavena P., Marchesi F., Garlanda C. Macrophages as Tools and Targets in Cancer Therapy. Nat. Rev. Drug Discov. 2022;21:799–820. doi: 10.1038/s41573-022-00520-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kostine M., Cleven A.H., de Miranda N.F.C.C., Italiano A., Cleton-Jansen A.-M., Bovée J.V.M.G. Analysis of PD-L1, T-Cell Infiltrate and HLA Expression in Chondrosarcoma Indicates Potential for Response to Immunotherapy Specifically in the Dedifferentiated Subtype. Mod. Pathol. 2016;29:1028–1037. doi: 10.1038/modpathol.2016.108. [DOI] [PubMed] [Google Scholar]
- 94.Pan Y., Yu Y., Wang X., Zhang T. Tumor-Associated Macrophages in Tumor Immunity. Front. Immunol. 2020;11:583084. doi: 10.3389/fimmu.2020.583084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Curiel T.J., Coukos G., Zou L., Alvarez X., Cheng P., Mottram P., Evdemon-Hogan M., Conejo-Garcia J.R., Zhang L., Burow M., et al. Specific Recruitment of Regulatory T Cells in Ovarian Carcinoma Fosters Immune Privilege and Predicts Reduced Survival. Nat. Med. 2004;10:942–949. doi: 10.1038/nm1093. [DOI] [PubMed] [Google Scholar]
- 96.Nota S.P.F.T., Al-Sukaini A., Patel S.S., Sabbatino F., Nielsen G.P., Deshpande V., Yearley J.H., Ferrone S., Wang X., Schwab J.H. High TIL, HLA, and Immune Checkpoint Expression in Conventional High-Grade and Dedifferentiated Chondrosarcoma and Poor Clinical Course of the Disease. Front. Oncol. 2021;11:598001. doi: 10.3389/fonc.2021.598001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Tie Y., Tang F., Wei Y.-Q., Wei X.-W. Immunosuppressive Cells in Cancer: Mechanisms and Potential Therapeutic Targets. J. Hematol. Oncol. 2022;15:61. doi: 10.1186/s13045-022-01282-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Cannarile M.A., Weisser M., Jacob W., Jegg A.-M., Ries C.H., Rüttinger D. Colony-Stimulating Factor 1 Receptor (CSF1R) Inhibitors in Cancer Therapy. J. Immunother. Cancer. 2017;5:53. doi: 10.1186/s40425-017-0257-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wen J., Wang S., Guo R., Liu D. CSF1R Inhibitors Are Emerging Immunotherapeutic Drugs for Cancer Treatment. Eur. J. Med. Chem. 2023;245:114884. doi: 10.1016/j.ejmech.2022.114884. [DOI] [PubMed] [Google Scholar]
- 100.Harris F., Berdugo Y.A., Tree T. IL-2-Based Approaches to Treg Enhancement. Clin. Exp. Immunol. 2023;211:149–163. doi: 10.1093/cei/uxac105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zając A.E., Czarnecka A.M., Rutkowski P. The Role of Macrophages in Sarcoma Tumor Microenvironment and Treatment. Cancers. 2023;15:5294. doi: 10.3390/cancers15215294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Fridlender Z.G., Sun J., Kim S., Kapoor V., Cheng G., Ling L., Worthen G.S., Albelda S.M. Polarization of Tumor-Associated Neutrophil Phenotype by TGF-Beta: “N1” versus “N2” TAN. Cancer Cell. 2009;16:183–194. doi: 10.1016/j.ccr.2009.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Shaul M.E., Fridlender Z.G. Tumour-Associated Neutrophils in Patients with Cancer. Nat. Rev. Clin. Oncol. 2019;16:601–620. doi: 10.1038/s41571-019-0222-4. [DOI] [PubMed] [Google Scholar]
- 104.Fridlender Z.G., Albelda S.M. Tumor-Associated Neutrophils: Friend or Foe? Carcinogenesis. 2012;33:949–955. doi: 10.1093/carcin/bgs123. [DOI] [PubMed] [Google Scholar]
- 105.Masucci M.T., Minopoli M., Carriero M.V. Tumor Associated Neutrophils. Their Role in Tumorigenesis, Metastasis, Prognosis and Therapy. Front. Oncol. 2019;9:1146. doi: 10.3389/fonc.2019.01146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Jeon H., Han S.R., Lee S., Park S.J., Kim J.H., Yoo S.-M., Lee M.-S. Activation of the Complement System in an Osteosarcoma Cell Line Promotes Angiogenesis through Enhanced Production of Growth Factors. Sci. Rep. 2018;8:5415. doi: 10.1038/s41598-018-23851-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Nabizadeh J.A., Manthey H.D., Steyn F.J., Chen W., Widiapradja A., Md Akhir F.N., Boyle G.M., Taylor S.M., Woodruff T.M., Rolfe B.E. The Complement C3a Receptor Contributes to Melanoma Tumorigenesis by Inhibiting Neutrophil and CD4+ T Cell Responses. J. Immunol. 2016;196:4783–4792. doi: 10.4049/jimmunol.1600210. [DOI] [PubMed] [Google Scholar]
- 108.Guglietta S., Chiavelli A., Zagato E., Krieg C., Gandini S., Ravenda P.S., Bazolli B., Lu B., Penna G., Rescigno M. Coagulation Induced by C3aR-Dependent NETosis Drives Protumorigenic Neutrophils during Small Intestinal Tumorigenesis. Nat. Commun. 2016;7:11037. doi: 10.1038/ncomms11037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Magrini E., Di Marco S., Mapelli S.N., Perucchini C., Pasqualini F., Donato A., Guevara Lopez M.d.l.L., Carriero R., Ponzetta A., Colombo P., et al. Complement Activation Promoted by the Lectin Pathway Mediates C3aR-Dependent Sarcoma Progression and Immunosuppression. Nat. Cancer. 2021;2:218–232. doi: 10.1038/s43018-021-00173-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Latchman Y., Wood C.R., Chernova T., Chaudhary D., Borde M., Chernova I., Iwai Y., Long A.J., Brown J.A., Nunes R., et al. PD-L2 Is a Second Ligand for PD-1 and Inhibits T Cell Activation. Nat. Immunol. 2001;2:261–268. doi: 10.1038/85330. [DOI] [PubMed] [Google Scholar]
- 111.Han Y., Liu D., Li L. PD-1/PD-L1 Pathway: Current Researches in Cancer. Am. J. Cancer Res. 2020;10:727–742. [PMC free article] [PubMed] [Google Scholar]
- 112.Taefehshokr N., Baradaran B., Baghbanzadeh A., Taefehshokr S. Promising Approaches in Cancer Immunotherapy. Immunobiology. 2020;225:151875. doi: 10.1016/j.imbio.2019.11.010. [DOI] [PubMed] [Google Scholar]
- 113.Ko B., Takebe N., Andrews O., Makena M.R., Chen A.P. Rethinking Oncologic Treatment Strategies with Interleukin-2. Cells. 2023;12:1316. doi: 10.3390/cells12091316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Thanindratarn P., Dean D.C., Nelson S.D., Hornicek F.J., Duan Z. Advances in Immune Checkpoint Inhibitors for Bone Sarcoma Therapy. J. Bone Oncol. 2019;15:100221. doi: 10.1016/j.jbo.2019.100221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yang X., Zhu G., Yang Z., Zeng K., Liu F., Sun J. Expression of PD-L1/PD-L2 Is Associated with High Proliferation Index of Ki-67 but not with TP53 Overexpression in Chondrosarcoma. Int. J. Biol. Markers. 2018;33:507–513. doi: 10.1177/1724600818774464. [DOI] [PubMed] [Google Scholar]
- 116.Zhu Z., Jin Z., Zhang M., Tang Y., Yang G., Yuan X., Yao J., Sun D. Prognostic Value of Programmed Death-Ligand 1 in Sarcoma: A Meta-Analysis. Oncotarget. 2017;8:59570–59580. doi: 10.18632/oncotarget.19168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Wang F., Yu T., Ma C., Yuan H., Zhang H., Zhang Z. Prognostic Value of Programmed Cell Death 1 Ligand-1 in Patients with Bone and Soft Tissue Sarcomas: A Systemic and Comprehensive Meta-Analysis Based on 3,680 Patients. Front. Oncol. 2020;10:749. doi: 10.3389/fonc.2020.00749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Paoluzzi L., Cacavio A., Ghesani M., Karambelkar A., Rapkiewicz A., Weber J., Rosen G. Response to Anti-PD1 Therapy with Nivolumab in Metastatic Sarcomas. Clin. Sarcoma Res. 2016;6:24. doi: 10.1186/s13569-016-0064-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Tawbi H.A., Burgess M., Bolejack V., Van Tine B.A., Schuetze S.M., Hu J., D’Angelo S., Attia S., Riedel R.F., Priebat D.A., et al. Pembrolizumab in Advanced Soft-Tissue Sarcoma and Bone Sarcoma (SARC028): A Multicentre, Two-Cohort, Single-Arm, Open-Label, Phase 2 Trial. Lancet Oncol. 2017;18:1493–1501. doi: 10.1016/S1470-2045(17)30624-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Keung E.Z., Burgess M., Salazar R., Parra E.R., Rodrigues-Canales J., Bolejack V., Van Tine B.A., Schuetze S.M., Attia S., Riedel R.F., et al. Correlative Analyses of the SARC028 Trial Reveal an Association Between Sarcoma-Associated Immune Infiltrate and Response to Pembrolizumab. Clin. Cancer Res. 2020;26:1258–1266. doi: 10.1158/1078-0432.CCR-19-1824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Pollack S.M., Redman M.W., Baker K.K., Wagner M.J., Schroeder B.A., Loggers E.T., Trieselmann K., Copeland V.C., Zhang S., Black G., et al. Assessment of Doxorubicin and Pembrolizumab in Patients with Advanced Anthracycline-Naive Sarcoma: A Phase 1/2 Nonrandomized Clinical Trial. JAMA Oncol. 2020;6:1778–1782. doi: 10.1001/jamaoncol.2020.3689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Singh A., Thorpe S.W., Darrow M., Carr-Ascher J.R. Case Report: Treatment of Metastatic Dedifferentiated Chondrosarcoma with Pembrolizumab Yields Sustained Complete Response. Front. Oncol. 2022;12:991724. doi: 10.3389/fonc.2022.991724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Le D.T., Uram J.N., Wang H., Bartlett B.R., Kemberling H., Eyring A.D., Skora A.D., Luber B.S., Azad N.S., Laheru D., et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015;372:2509–2520. doi: 10.1056/NEJMoa1500596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Schuetze S.M., Wathen J.K., Lucas D.R., Choy E., Samuels B.L., Staddon A.P., Ganjoo K.N., von Mehren M., Chow W.A., Loeb D.M., et al. SARC009: Phase 2 Study of Dasatinib in Patients with Previously Treated, High-Grade, Advanced Sarcoma. Cancer. 2016;122:868–874. doi: 10.1002/cncr.29858. [DOI] [PubMed] [Google Scholar]
- 125.Amaria R.N., Bernatchez C., Forget M.-A., Haymaker C.L., Conley A.P., Livingston J.A., Varadhachary G.R., Javle M.M., Maitra A., Tzeng C.-W.D., et al. Adoptive Transfer of Tumor-Infiltrating Lymphocytes in Patients with Sarcomas, Ovarian, and Pancreatic Cancers. JCO. 2019;37:TPS2650. doi: 10.1200/JCO.2019.37.15_suppl.TPS2650. [DOI] [Google Scholar]
- 126.Yang J., Dong L., Yang S., Han X., Han Y., Jiang S., Yao J., Zhang Z., Zhang S., Liu P., et al. Safety and Clinical Efficacy of Toripalimab, a PD-1 mAb, in Patients with Advanced or Recurrent Malignancies in a Phase I Study. Eur. J. Cancer. 2020;130:182–192. doi: 10.1016/j.ejca.2020.01.028. [DOI] [PubMed] [Google Scholar]
- 127.Martin-Broto J., Hindi N., Grignani G., Martinez-Trufero J., Redondo A., Valverde C., Stacchiotti S., Lopez-Pousa A., D’Ambrosio L., Gutierrez A., et al. Nivolumab and Sunitinib Combination in Advanced Soft Tissue Sarcomas: A Multicenter, Single-Arm, Phase Ib/II Trial. J. Immunother. Cancer. 2020;8:e001561. doi: 10.1136/jitc-2020-001561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Duffaud F., Chabaud S., Gautier J., Ferlay C., Vizoso S., Brahmi M., Benezech S., Dufresne A., Marec-Berard P., Ray-Coquard I.L., et al. REGOSTA: A Randomized, Placebo-Controlled, Double-Blinded, Multicenter Study Evaluating the Efficacy and Safety of Regorafenib (REGO) as Maintenance Therapy after First-Line Treatment in Patients (Pts) with Osteosarcoma (OS) and Non-Osteosarcomas (Non-OS) of Bone (Non-Ewing, Non-Chondrosarcomas and Non-Chordomas) JCO. 2021;39:TPS11576. doi: 10.1200/JCO.2021.39.15_suppl.TPS11576. [DOI] [Google Scholar]
- 129.Wang B.T., Kothambawala T., Wang L., Matthew T.J., Calhoun S.E., Saini A.K., Kotturi M.F., Hernandez G., Humke E.W., Peterson M.S., et al. Multimeric Anti-DR5 IgM Agonist Antibody IGM-8444 Is a Potent Inducer of Cancer Cell Apoptosis and Synergizes with Chemotherapy and BCL-2 Inhibitor ABT-199. Mol. Cancer Ther. 2021;20:2483–2494. doi: 10.1158/1535-7163.MCT-20-1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Desbois M., Calhoun S.E., Hart K.C., Denson C., Yakkundi P., Matthew T.J., Leabman M.K., Billic S., Hernandez G., Humke E.W., et al. Novel Combinations of Aplitabart, a DR5 Agonist IgM Antibody, with ADCs or Chemotherapeutic Agents Lead to Robust Anti-Tumor Responses in Solid Tumor Models; Proceedings of the 115th Annual Meeting of the American Association for Cancer Research 2024; San Diego, CA, USA. 5–10 April 2024. [Google Scholar]
- 131.D’Angelo S.P., Richards A.L., Conley A.P., Woo H.J., Dickson M.A., Gounder M., Kelly C., Keohan M.L., Movva S., Thornton K., et al. Pilot Study of Bempegaldesleukin in Combination with Nivolumab in Patients with Metastatic Sarcoma. Nat. Commun. 2022;13:3477. doi: 10.1038/s41467-022-30874-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Salkeni M.A., Ko B., Van Tine B.A., Conley A.P., Davis E.J., Burgess M.A., Close J.L., Foster J.C., O’Sullivan Coyne G.H., Moore N., et al. A Phase 2 Study of an Anti–PD-L1 Antibody (Atezolizumab) in Dedifferentiated Chondrosarcoma. JCO. 2023;41:11533. doi: 10.1200/JCO.2023.41.16_suppl.11533. [DOI] [Google Scholar]
- 133.Kelly C.M., Qin L.X., Whiting K.A., Richards A.L., Avutu V., Chan J.E., Chi P., Dickson M.A., Gounder M.M., Keohan M.L., et al. A Phase II Study of Epacadostat and Pembrolizumab in Patients with Advanced Sarcoma. Clin. Cancer Res. 2023;29:2043–2051. doi: 10.1158/1078-0432.CCR-22-3911. [DOI] [PMC free article] [PubMed] [Google Scholar]