Abstract
Atherosclerosis originates in childhood and adolescence. The goal of this review is to highlight how exercise and physical activity during childhood and adolescence, critical periods of growth and development, can prevent adult cardiovascular disease (CVD), particularly through molecular mechanisms of monocytes, a key cell of the innate immune system. Monocytes are heterogeneous and pluripotential cells that can, paradoxically, play a role in both the instigation and prevention of atherosclerosis. Recent discoveries in young adults reveal that brief exercise affects monocyte gene pathways promoting a cell phenotype that patrols the vascular system and repairs injuries. Concurrently, exercise inhibits pro-inflammatory monocytes, cells that contribute to vascular damage and plaque formation. Because CVD is typically asymptomatic in youth, minimally invasive techniques must be honed to study the subtle anatomic and physiologic evidence of vascular dysfunction. Exercise gas exchange and heart rate measures can be combined with ultrasound assessments of vascular anatomy and reactivity, and near-infrared spectroscopy to quantify impaired O2 transport that is often hidden at rest. Combined with functional, transcriptomic, and epigenetic monocyte expression and measures of monocyte–endothelium interaction, molecular mechanisms of early CVD can be formulated, and then translated into effective physical activity-based strategies in youth to prevent adult-onset CVD.
INTRODUCTION AND GOALS OF THE REVIEW
Children are the most naturally physically active human beings, reduced physical activity (PA) is a cardinal sign of childhood disease, and exercise testing provides clues to mechanisms of health and disease that are often hidden when the child is at rest. Despite this, and because mechanistic studies, data analytics, and testing protocols have failed to keep pace with enabling technologies and computing capacity, biomarkers of fitness and PA have yet to be widely incorporated into translational research and clinical practice designed to prevent adult-onset diseases during childhood. Challenges also arise because acceptable standards for research in healthy children are appropriately high. Innovative thinking is required to minimize the invasiveness of any experimental procedure and ensure that protocols are child friendly, engaging not only the volunteers themselves but also their parents or guardians.
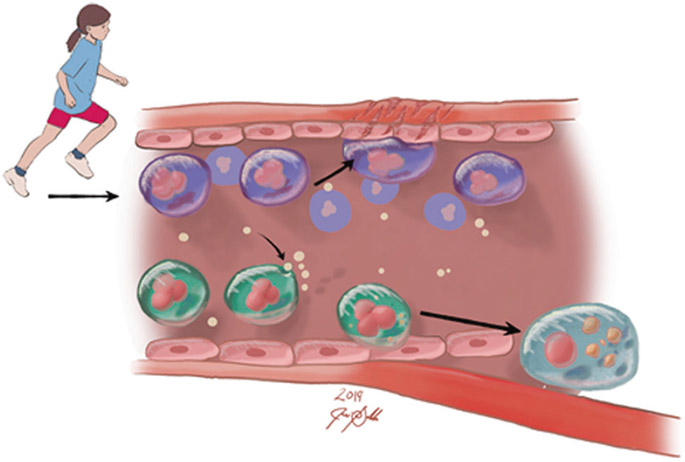
The goal of this review is to focus on a major global health problem, cardiovascular disease (CVD). Adult, clinically symptomatic CVD is a major and tragic cause of morbidity and early mortality throughout the world.1 Surprisingly, CVD starts in childhood even though its symptoms are absent until much later in life.2,3 Large-scale studies of CVD risk suggest that sufficient and sustained levels of PAtivity during childhood may protect against adult CVD.4-8 Because critical gaps still exist in our understanding of the pediatric origins of CVD, specific therapies, precise prescriptions for exercise, nutrition, and other child-appropriate lifestyle interventions are inadequate or lacking. In this review, we focus on novel conceptual and experimental developments involving the potential role of a specific innate immune cell, the monocyte, in the early pathophysiology of adult CVD (Fig. 1).
Fig. 1.
Exercise promotes cardiovascular health through reprogramming of monocytes. Our recently published data show that exercise leads to increased NR4A1, NR4A2, and AREG (the amphiregulin gene) gene expression.13 These are associated with patrolling monocyte subtypes that, in contrast to pro-inflammatory monocyte subtypes, patrol the vascular and repair damaged tissues rather than contribute to atherosclerosis
THE CENTRAL ROLE PLAYED BY MONOCYTES IN TRANSDUCING EXERCISE INTO HEALTH BENEFITS IN CHILD HEALTH AND DISEASE
Once considered to be single-purpose cells that could only react in highly circumscribed ways to invading organisms, monocytes (and other innate immune cell types such as natural killer cells and granulocytes) are now known to be involved in a range of functions that can “learn” through genomic mechanisms and either benefit health or exacerbate disease.9,10 Monocytes are accessible through phlebotomy in pediatric research; thus, like in adults, their function can be studied. Monocytes are the precursors for tissue macrophages. Both monocytes and macrophages play a role in the pathogenesis of atherosclerosis, and while they share many cellular markers, the precise and complex mechanisms through which the monocyte transforms into a macrophage has not yet been fully elucidated.11 New data from a variety of research groups demonstrate the effect of brief exercise on gene and microRNA expression of circulating immune cells in children and adults. These initial studies permit an identification of specific mechanisms in child health that link exercise and innate immune cell function with disease prevention and clinical outcomes.12,13
Many of the genomic and epigenetic pathways identified in leukocytes of children and adults are related to growth and repair, as well as prevention of diseases such as asthma, cancer, and atherosclerosis14,15 (Fig. 2). Advanced use of techniques such as flow cytometry has stimulated research into the effect of acute and chronic exercise on leukocyte function [summarized recently by Gjevestad et al.,16] including the impact of exercise on mitochondrial function17 and oxidative stress,18,19 each of which has been implicated as an essential component of the molecular transduction of PA in other tissues, such as the skeletal muscle.20 Exercise-sensitive monocytes may play a role in vascular health Circulating monocytes are a heterogeneous set of pluripotential innate immune cells that can paradoxically play a role in both the instigation and prevention of atherosclerotic plaques (Fig. 1). Exercise, even very brief exercise lasting only a few minutes, leads to leukocytosis with substantial increases in circulating monocytes in both laboratory and field settings in adults and children.21 The idea that the exercise-associated increase in the number of circulating monocytes would be accompanied by changes in their gene, and microRNA expression was first demonstrated in young adults.13 The response to acute exercise was substantial in the range and magnitude of gene expression (894 genes altered), gene pathways, and microRNA (19 microRNAs). A remarkably consistent pattern of change emerged from our studies, namely, that exercise could shift monocyte gene expression profiles and function to anti-inflammatory and anti-atherogenic activity. Clearly, future research will be necessary to distinguish how monocyte responses to acute exercise are influenced by fitness and training, the latter determined in large measure by chronic exercise.
Fig. 2.
Examples of gene expression changes in circulating leukocytes following brief exercise. These reveal intriguing mechanistic links between physical activity and health. We hypothesize that these effects are influenced by growth and maturational status. Data are from refs. 13,128,129
Several gene pathways known to be related to atherosclerosis were enriched, including mitogen-activated protein kinase signaling pathway [e.g., interleukin-4 (IL-4) stimulation of anti-inflammatory macrophages 22] and apoptosis pathway [upregulated in peripheral blood mononuclear cells in patients with peripheral arterial diseases.23] Two particularly intriguing observations led to the formulation of additional hypotheses to explain potential anti-atherosclerotic effects of exercise on monocytes. First, the transcription factors NR4A1 and NR4A2, belonging to the nuclear hormone receptor superfamily, were upregulated following exercise in monocytes by 3- and 5-fold, respectively. Several investigators have noted that in murine models,24 the NR4A superfamily plays a role in shaping monocytes to become patrolling cells that crawl along the endothelium and survey the capillaries for microparticles, cellular debris, and other signs of endothelial damage and disruption. They found that these patrolling monocytes could, in conjunction with neutrophils, ameliorate endothelial cell necrosis without extravasation or diapedesis. They concluded that, when carefully regulated, the NR4A1-activated nonclassical monocytes could play a protective role in vascular health.
The second compelling observation was that gene expression of key members of the epidermal growth factor family was also upregulated in monocytes by brief exercise [amphiregulin (AREG) by 19.3-fold, heparin-binding growth factor by 7.8-fold, and epiregulin by 6.8-fold]. These pleiotropic growth factors are involved in tissue healing and repair, and vascular smooth muscle growth.25-27] Interestingly, monocyte EGR2 [early-growth response 2, which when upregulated shifts monocytes to macrophages, hastening vascular complications in diabetes28] was reduced in response to exercise by 4.4-fold. In Table 1, we show that acute exercise can alter monocyte gene expression in a way that renders them protective against the development of atherosclerosis. These mechanisms should be studied in healthy children and adolescents, and expand our knowledge of how exercise might play a role in preventing preclinical pediatric origins of atherosclerosis. The role of microRNAs in the regulation of atherosclerosis has also received a great deal of attention over the past several years. As shown in Table 2, a number of the monocyte microRNAs that were affected by brief exercise are involved in the pathogenesis of vascular disease.
Table 1.
Differential exercise regulation of monocyte genes previously associated with inflammation and/or CAD
| Gene | CAD | Exercise | Possible mechanisms |
|---|---|---|---|
| CD36 | ↑ | ↓ | Facilitates scavenging of modified LDL and activates inflammatory pathways131 |
| TLR4 | ↑ | ↓ | Pathogenesis and destabilization of atherosclerotic plaques132 |
| VCAN | ↑ | ↓ | Versican is involved in advanced lesions of atherosclerosis at the borders of lipid-filled necrotic cores as well as at the plaque–thrombus interface133 |
| DNAJB6 | ↑ | ↓ | Controls HSP function, known to be a key immune modulator in atherosclerotic plaques134 |
| FAM198B | ↑ | ↓ | As yet unknown |
| HIST1H2BG | ↑ | ↓ | Histone modification is a critical component of a transcriptional cascade regulating SMC proliferation and might play a role in the development of proliferative vascular diseases135 |
All data from human subjects. Data from coronary artery disease (CAD) patients is derived from previous studies. The exercise data is from our recent study in healthy people
Table 2.
microRNAs in monocytes significantly (FDR ≤0.05) affected (FC) by exercise and their possible connection to atherosclerosis [exercise data from our publication13]
| microRNA | FC | Possible link to atherosclerosis |
|---|---|---|
| miR-130a | ↓1.5 | Involved in angiogenesis in endothelial progenitor cells;136 serum biomarker for atherosclerosis137 |
| miR-221 | ↓1.3 | Involved in vascular remodeling; regulation of monocytes into dendritic cells138 |
| miR-23b | ↓1.3 | Controls immune tolerance in dendritic cells; plays an atheroprotective role in shear stress vascular remodeling139 |
| miR-29b | ↑1.9 | Plays a key role in the mechanisms through which LDLs alter vascular smooth muscle function; significantly upregulated in atherosclerotic aortic aneurysm tissue;140 inhibits migration and proliferation of vascular smooth muscle cells in neointimal formation141 |
| miR-362-3p | ↑1.4 | Downregulated more than twofold in both brain and blood following experimental injury to the cerebral vasculature142 |
| miR-660 | ↑1.4 | Increases the efficiency of ex vivo platelet generation;143 predicts future fatal myocardial infarction in healthy individuals144 |
| miR-140-5p | ↑1.3 | Circulating levels are elevated in severely obese individuals145 |
| miR-532-5p | ↑1.3 | Circulating levels are elevated in severely obese individuals145 |
| miR-30e | ↑1.3 | Substantially downregulated in animal model of atherosclerotic lesions;146 inhibits neointimal hyperplasia by targeting calmodulin-dependent protein kinase147 |
| miR-15a | ↑1.3 | Involved in blood–brain barrier disruption in animal models of vascular injury;148 associated with abdominal aortic aneurysms and peripheral arterial disease149 |
FC fold change
THE PEDIATRIC ORIGINS OF ADULT CVD
Exercise in children and adolescents is not merely play, but is an essential component of growth and development.29-31 Children are among the most spontaneously physically active human beings.32 It is not surprising that participation in PA is a major determinant of health across the lifespan and health-related quality of life in both healthy children and in children with chronic diseases.33,34 Despite this essential biologic role for PA, children have not been spared the relentless reduction in levels of PA that is creating a crisis in health care in our nation and throughout the world.35 Recognition of the enormous morbidity and cost of physical inactivity-related diseases, such as atherosclerosis, type 2 diabetes, and osteoporosis, has spurred new policy initiatives targeting preventive medicine early in life.36
The concept of pediatric origins of adult health and disease is gaining scientific merit,37,38 highlighting the need to transform existing notions of how to evaluate health in a growing child. A physically inactive (even normal-weight) child may have no symptoms of disease, but evidence of deterioration in vascular health may already be present.39,40 Although as yet insufficiently studied, there is increasing evidence that the rapidly changing phenotype associated with normal growth and development is accompanied by global changes in gene expression.41 The pattern of change of gene and epigenetic expression during childhood is likely to influence the response to acute exercise and habitual physical activity. The notion of what it means to be a healthy child must change and include robust metrics of physical fitness and their biologic underpinnings.
Equally worrisome is that the deleterious health effects of physical inactivity and poor fitness are exacerbated in children with chronic disease and/or disabilities42,43 or with environmental–lifestyle conditions like obesity.44 Children with diseases or conditions previously associated with mortality during the first two decades of life (e.g., sickle cell disease, cystic fibrosis) are living longer due to remarkable advances in research and care, but are often unable to achieve levels of PA and fitness associated with health benefits in otherwise healthy children.45,46 Not surprisingly, the healthspan [the period of life free from serious chronic diseases and disability47] of children with chronic diseases is threatened not only by the underlying disease, but also by the compounding effects of insufficient PA and sedentary behavior. Increasing PA and fitness is feasible, but has proven quite challenging to implement in a systematic manner.48 Once a pattern of physical inactivity and a sedentary lifestyle is established, a vicious cycle ensues, in which constraints on PA harm immediate health and contribute to lifelong health impairment ranging from cardiovascular and metabolic disease to osteoporosis.49,50 Exactly what constitutes ideal physical fitness in a child with a chronic condition (or, in fact, a child considered to be otherwise healthy) remains unknown. Finding beneficial levels of PA in children with chronic disease or disability is challenging because the optimal range of exercise is much narrower than in a healthy child (Fig. 3).
Fig. 3.
Health benefits of exercise are determined, in part, by the energy expenditure associated with physical activity. Both too much (excessive) and too little (sedentarism) exercise can impair health. As shown, the range of healthy exercise is narrower in the child with chronic disease or disability
The need to explore mechanisms focused on the earliest origins of CVD is highlighted in children who survive acute lymphocytic leukemia (ALL). The remarkable success in treating children with ALL is among the great achievements of translational and clinical research of the last generation.51 Physical fitness is low in ALL survivors,52,53 and the healthspan of child and adolescent survivors of ALL remains threatened.54,55 In particular, CVD risk is increased and is associated with obesity and metabolic syndrome. Gibson et al.56 outlined the current state of knowledge regarding many of the health threats faced by ALL survivors, “Unfortunately, treatment is not without consequence; 50% of childhood ALL survivors in their 20s will have at least one chronic medical condition. Early death is also a recognized problem; the standardized mortality ratio among those who survive 5 years from diagnosis is 9.5 (8.8–10.2) with non-cancer-related mortality frequently attributed to a cardiovascular cause.” The mechanisms of this increased morbidity and mortality are unknown, but as there is increasing evidence of endothelial dysfunction and increased CVD risk in survivors of childhood ALL,57,58 due, possibly to chronic inflammatory activation and immune dysregulation.54
TRACKING FITNESS AND CVD RISK DURING CHILDHOOD AND ACROSS THE LIFESPAN
Several pioneering, thoughtfully designed, long-term studies now confirm that CVD risk factors begin in youth, track into symptomatic atherosclerosis in adulthood, but are, fortunately, modifiable [e.g., the Muscatine Study,4 the Young Finns Study,5,59 the Bogalusa Heart Study,6 the CARDIA study,7 Pathobiological Determinants of Atherosclerosis in Youth,8 and the Australian Childhood Determinants of Adult Health study44] Efforts to develop childhood- and youth-based preventive interventions focused on nutrition and physical activity, in combination or separately, have not met expectations. For example, Project HEALTHY, the largest NIH school-based study ever undertaken to prevent obesity and type 2 diabetes in children through changes in the school nutrition and physical education did show a modest benefit on obese middle school children, but did not succeed in demonstrating a more robust population effect.48
The current epidemic of childhood obesity has generated much research into the specific relationships between CVD risk, fitness, and nutrition in the overweight child. Childhood obesity is associated with lifelong increased CVD risk, although the associations are only weak to moderate.60 Chronic inflammation characterized by increased levels of circulating inflammatory cytokines and leukocytosis accompanies childhood obesity,61 and chronic inflammation is a clear risk factor for the development of endothelial dysfunction and CVD.62 Particularly intriguing is work by Mattos et al.,63 who compared monocyte inflammatory function (obtained under resting conditions) in 11 obese with 9 normal-weight children and adolescents. All traditional monocyte subsets (classical, intermediate, nonclassical) in the obese participants produced less IL-10, an anti-inflammatory and anti-atherosclerotic cytokine,64 than did the normal-weight children and adolescents.
Chronic inflammation in the pediatric age group is not observed solely in obese individuals. Inflammatory mediators are higher, for example, in normal-weight, sedentary high school girls compared with age- and weight-matched girls who participate in organized sports.65 Other conditions, such as systemic lupus erythematosus or inflammatory bowel disease, are accompanied by chronic inflammation and a proclivity to CVD.66,67 A possible therapeutic and/or anti-atherosclerotic role for exercise acting, perhaps, as outlined below through circulating monocytes is a prime target for translational research.
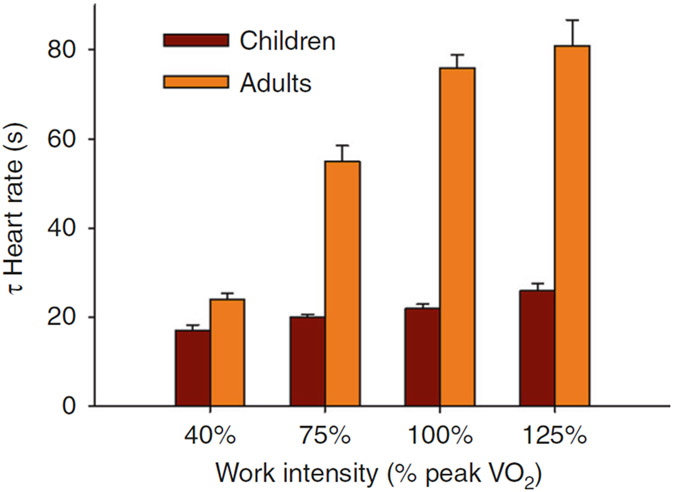
A challenge in research designed to understand the inflammation–fitness connection as a factor in CVD risk early in life is that children are not simply miniature adults [e.g., refs. 68,69] Physical fitness in both adults and children is commonly measured by testing, which gauges systemic physiologic responses to acute exercise. When normalized to body size, strength is lower in children,70 as is the magnitude of the physiologic response to chronic exercise training (both resistance and aerobic).71,72 Children are among the most naturally physically active humans32 and oxygen uptake normalized to work rate is higher than in adults for high-intensity exercise.73 Gas exchange and heart rate (HR) response kinetics are also different in children74-76 (Fig. 4), as are metabolic responses, such as lactate kinetics,77 high-energy intramuscular phosphate dynamics [using 31P-magnetic resonance spectroscopy,78] and CO2 storage capacity.79 Recent work demonstrates that leukocytosis in response to exercise is greater in older compared with younger children.80 Consequently, maturational and developmental factors must be accounted for in any attempt to determine how acute or chronic exercise influences genomic and functional responses of those immune cells, like monocytes, which might play a role in the earliest development of CVD.
Fig. 4.
Differences in physiologic responses to exercise between children and adults. Shown here are the recovery times [as time constants (τ)] to 1-min bouts of exercise of increasing intensity in children and adults. HR recovery times were shorter in children compared with adults for the exercise above the anaerobic (lactate) threshold. Data from ref. 130
METHODS TO DETECT VASCULAR INVOLVEMENT IN THE PEDIATRIC POPULATION
Vascular stiffness
Inflammation is linked to the development of vascular stiffness, a hallmark of CVD.81 In adults, several studies have demonstrated specific correlation between monocyte subtypes and noninvasive metrics of arterial stiffness in patients with CVD.82 Arterial structure and function can be measured noninvasively in children with common carotid artery (CCA) intima–media thickness (IMT), CCA distensibility, and pulse wave velocity (PWV). These measures are functionally linked because both pressure change and cvBRS (cardiovagal baroreflex sensitivity) depend on the arterial elastic properties. cvBRS, CCA distensibility, IMT, and PWV have all demonstrated utility in identifying both positive and negative autonomic and arterial alterations in children in response to physical activity/fitness83 and CVD risk factors, respectively.84-86
The majority of studies in children demonstrate a positive correlation between cardiorespiratory fitness and arterial compliance, and an inverse correlation between cardiorespiratory fitness and stiffness parameters.87,88 Finally, in one recent study Donghui et al.89 demonstrated that a 6-week exercise training and nutrition program improved the reactive hyperemia index (felt to be a measure of microvascular reactivity) in a group of obese adolescents.
Fitness testing and CVD
Gas exchange in response to exercise is increasingly used as a noninvasive, albeit indirect, clinically useful biomarker for CVD. Correlating monocyte function to cardiopulmonary exercise testing (CPET) could serve as a powerful translational and clinical research tool in gauging the mechanism of the pediatric origins of adult CVD. In both adults and children, disambiguating reduced fitness from true CVD using gas exchange and HR data from CPET is challenging. In adult studies, innovative data analytics in which the rich dataset of gas exchange and HR variables obtained during both submaximal and peak CPET are beginning to identify noninvasive biomarkers (such as the dynamic relationship between and ) that correlate with established indexes of CVD (such as stroke volume measured by stress echocardiography).90 In a study in adolescents with high body mass index,91 the pattern of CPET abnormalities suggested a pervasive impairment of O2 delivery—an indirect indicator of vascular or cardiac dysfunction. An easily calculated slope , derived from the submaximal portion of a progressive exercise CPET, is one relatively accessible approach to gauge the effectiveness of O2 delivery during exercise in children and adolescents, and normal values are available.92 As noted above, standardized fitness assessments are associated with CVD risk in children and adults, but these typically weak to modest correlations are found predominantly in large sample studies.
A number of promising technologies are emerging for noninvasively measuring vascular reactivity and the effectiveness of O2 delivery at the level of the working muscle during exercise. Microvascular reactivity can be studied noninvasively in the skin and has been used to test vascular function in both adults and adolescents.93 Near-infrared spectroscopy (NIRS) has been used to gauge the degree of muscle microcirculatory impairment in patients with heart failure.94 NIRS has also been used to assess the effect of exercise training in patients with intermittent claudication.95 Luck et al.96 recently used NIRS in patients with peripheral artery disease to demonstrate a link between working muscle ischemia and systemic responses such as higher blood pressure and HR.
Translational research that combines systemic gas exchange, noninvasive tissue-specific vascular reactivity and oxygen delivery, and functional and genomic expression of circulating leukocytes is certainly feasible in children and adolescents. Such an approach might bring about a better understanding of what constitutes healthy levels of physical activity and fitness in healthy children. Results from healthy children could then be used to improve therapies for a variety of chronic childhood diseases like sickle cell anemia,97 in which exercise is impaired and chronic inflammation [mediated, perhaps, to some degree specifically by monocytes98] contributes to an impaired healthspan. Despite the broad recognition that many children in the United States (and throughout the world) no longer engage in healthy levels of physical activity,99 defining what the level of optimal physical activity should be remains quite vague. For example, in a recent study of 182 9–11 year olds, Füssenich et al.100 noted, “there were no differences between CCVR [composite cardiovascular risk score] of children who undertook 60 min MVPA (moderate to vigorous physical activity) per day in accordance with World Health Organization (WHO) recommendations, and those who did not. This implies that current recommendations may be an underestimation of the PA necessary to reduce clustered CVD risk. A gender difference between the CVD risk in active and inactive children raises the possibility that gender specific guidelines may be needed, although much work is needed to determine if these differences are a result of gender specific responses to PA or sex differences in PA level. Taken together these findings suggest that in order to reduce CVD risk, the current guidelines should be updated.” New approaches to the precise and reproducible assessment of the physiological response to exercise are needed if we are to advance a mechanistic knowledge of the pediatric origins of CVD.
MONOCYTE FUNCTION: ROLE IN CVD ACROSS THE LIFESPAN AND METHODS FOR ASSESSMENT
Monocyte function by flow cytometry
Although it is known that monocyte subtype changes in response to exercise, the literature is scant and this remains a topic ripe for additional research. The overall concentration of monocytes doubles in the circulation as a result of the brief exercise protocol in both children and adults, reflecting the well-described acute effect of physical activity on leukocytes in general and monocytes in particular.13,21 Using flow cytometry, we classified subtypes of circulating monocytes. The emerging paradigm identifies classical (CD14++CD16−), intermediate (CD14++CD16+), and nonclassical (CD14+CD16++) subsets.101 The incidence of ischemic cardiovascular events in a retrospective study was associated with an increased number of classical monocytes.102 Some investigators postulate that the classical monocytes represent a more proinflammatory population of cells, and consequently are more likely to promote rather than attenuate atherosclerosis, but more work is needed to determine whether monocyte subtypes are useful biomarkers of clinically apparent cardiovascular disease or disease risk.103 Devêvre et al.104 have shown that obesity is associated with an increased proportion of intermediate and nonclassical monocytes in adults. There is evidence to support an association between intermediate monocyte subtypes and lipid levels in people with stable atherosclerosis.105
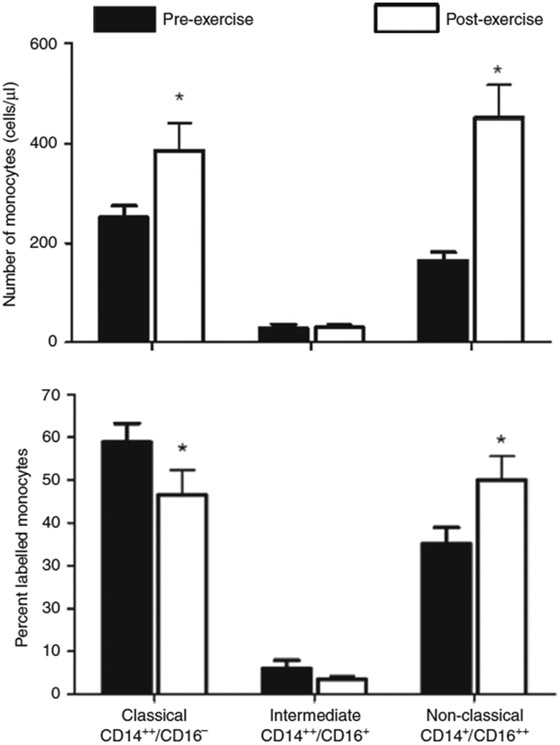
Consistent with studies done previously in other laboratories,106 we noted a significant increase in nonclassical monocytes after exercise and a parallel reduction in the proportion of classical monocytes in young adults (Fig. 5). Whether these observed differences in CD14 and CD16 monocyte surface markers are accompanied by changes in gene expression has not been fully elucidated, although Wong et al.107 found that the monocyte subtypes appeared to have distinct gene expression patterns. More recent work using single-cell RNA-sequencing supports an even wider array of distinct gene expression patterns among monocytes.108 Whether these patterns are influenced by exercise in children and adolescents is unknown. Monocyte function by tissue-engineered microfluidic endothelial cell systems One promising and emerging approach for studying monocyte behavior in a biologically relevant context is the development of vascularized micro-organ platform that utilizes microfluidics to drive vessel network formation within a fibrin gel matrix.109 Specifically, such devices incorporate arteriole (high pressure) and venule (low pressure) microfluidic channels that flank a central cell chamber where vascular network formation occurs. In preliminary studies (Fig. 6), we have demonstrated the feasibility of perfusing purified, fluorescently labeled human monocytes through these microvascular networks. Over time, a portion of these monocytes adheres to the endothelial lining of the vascular network. Closer examination of monocytes after 18 h of perfusion indicates extravasation of some cells. At both early and late time points, adherent and extravasated monocytes are quantified within each chamber, providing our research with an in vitro quantification of monocyte function.
Fig. 5.
Effect of brief exercise (pre vs. post) on circulating monocyte subtypes (classical, CD14++/CD16−; intermediate, CD14++/CD16+; nonclassical, CD14+/CD16++) in young adults. Top panel shows the absolute numbers. Bottom panel shows the percent changes. The brief exercise bout increased both the classical and nonclassical absolute number of monocyte subtype. *P < 0.01, before vs. after exercise. Data from ref. 13
Fig. 6.
A vascularized micro-organ platform showing promise for studying monocyte–endothelial cell (EC) interactions. Unpublished data: a a microfluidics device incorporating arteriole (high pressure) and venule (low pressure) fluid channels drives fluid flow across and b angiogenesis within a flanking cell chamber (red, ECs). c Anastomosis with these channels and physiological tightness of the vessels is shown by perfusion with 70 kDa rhodamine-dextran (green). d Perfusion of 100,000 monocytes per mL demonstrates monocyte adhesion after 18 h of perfusion (green, arrowheads). Closer examination within the dashed area reveals e both adherent (arrowheads) and extravasated (arrows) monocytes. f Both monocyte populations are readily quantified over time (***both adherent and extravasated monocytes increased over time in this preliminary study)
MONOCYTE GENE EXPRESSION RESPONSE TO EXERCISE: POSSIBLE MECHANISMS
Changes in monocyte gene expression can result from the physiological impact of exercise itself [e.g., heat,110 pH,111 hypoxia,112 turbulence/shear stress,113 and the effect of hormones and other mediators.114] Changes can also result from the shifting of populations of immune cells whose gene expression patterns in their marginated pools (e.g., lung, lymph, bone marrow, vasculature) differ from cells that were in the circulation prior to exercise.115 Our recent data13 permit us to draw some inferences concerning possible mechanisms. As noted, we discovered that AREG gene expression in monocytes increased by 19.3-fold increase following brief, intense exercise. The circulating monocyte count doubled in response to exercise. If the effect of exercise on AREG gene expression was solely mediated by the addition of marginal monocytes to the circulating pool and not by some direct effect of exercise on monocyte gene expression, then the 19.3-fold increase that we observed for this gene could occur only if the monocytes that entered the circulation had been expressing AREG at levels ~40-fold greater than the circulating monocytes, a highly unlikely scenario. Similarly, the 4.4-fold decrease in another gene, EGR2, would be difficult to explain only on the basis of shifting monocytes into the circulating pool. In the extreme case that the extra-circulatory monocytes had no detectable expression of this gene, the lowest possible reduction in gene expression would be a 2-fold decrease. Thus, it is reasonable to speculate that exercise has some direct effect on gene expression in the circulating monocytes. Whatever the mechanism(s) may be, it is clear that relatively brief exercise alters the gene expression profile of the circulating pool of monocytes, and it is this pool that will most likely interact with the endothelium in the prevention or pathogenesis of atherosclerosis.
A number of intriguing observations have been made in recent years suggesting that systemic neuroadrenergic and hormonal responses to exercise may attenuate possible harmful inflammatory effects that typically accompany an outpouring of monocytes and other inflammatory cells into the circulation. Dimitrov et al.116 demonstrated in adults that the proportion of tumor necrosis factor producing monocytes was suppressed by exercise. Further, using an elegant in vitro model, they showed that exercise-associated increases in epinephrine might be responsible for the modulating effect on the monocytes. In a group of adults with and without type 2 diabetes (mean age about 56 years old), Durrer et al.117 found that one bout of low-volume, high-intensity interval training (HIIT) reduced TLR2 expression in monocytes [toll-like receptor-2, known to play a role in atherosclerosis118] with no effect on neutrophils. The authors suggested that HIIT might be a useful adjunctive therapy to recued chronic inflammation in patients with type 2 diabetes. It is not yet understood at what point during childhood and/or adolescence these exercise-monocyte interactions become manifest.
THE BRIEF LIFE OF THE MONOCYTE; SEX, AGING, AND THE IMPACT ON THE LONG-TERM DURATION OF EXERCISE EFFECTS
We are only beginning to study and appreciate the profound impact that sex has on CVD-related biomarkers. In a recent study, Lew et al.119 discovered from an existing cohort of thousands of adults that cardiometabolic biomarker profiles differ significantly between women and men in the general population. Sex differences were most apparent for biomarkers of adiposity, endothelial dysfunction, inflammatory cell recruitment, and cardiac stress and injury. Included in the differences were higher levels in females of monocyte chemoattractant protein-1. Campesi et al.130 recently demonstrated that LPS affects ERα (ER–estrogen receptor) but not ERβ activation status in monocyte-derived macrophages (MDM) from young men and women. The significant role of ERα in LPS-mediated inflammatory responses in MDMs may represent an initial step in elucidating the effect of sex in the relationship between LPS and ERα, and, ultimately, in the sex-related differences in the clinical manifestation of atherosclerosis. When sex differences in monocyte/macrophage function become apparent during childhood is not known.
It is well established that immune function is influenced by sex and maturational status early in life.121 These changes continue as humans age.122 Less is known about the specific functional characteristics of monocytes. The proportion of the various circulating leukocyte subtypes changes during puberty and is influenced by sex.123 Interestingly, in adults, Metcalf et al.124 found that unstimulated monocyte subsets did not reveal significant age-related alternations. However, agonist-stimulated monocytes isolated from adults and old subjects did show alternations at transcriptional and functional levels implicating dynamic age-related changes in regulation of the adaptive immune response and monocyte function associated with defense mechanisms against bacteria and viruses. To the extent that acute exercise stimulates monocytes, exercise may prove to be useful at uncovering elements of monocyte function related to inflammation and atherosclerosis that would be hidden at rest.
The lifespan of the monocyte is short, ranging from hours to several days. This raises the question of whether an active lifestyle during childhood is effective only insofar as the child or adolescent exercises regularly and frequently enough to alter the genomic profile and function of the extant population of circulating monocytes at the moment that they are tested. Investigators are just beginning to examine whether the state of physical fitness is associated with changes in gene expression in leukocytes, and some intriguing data are emerging. For example, Queiroga et al.125 found that gene expression (from whole blood) of PPAR-γ was associated with fitness (as ) in discordant monozygotic twins. Flynn et al.126 showed that resistance exercise training appeared to lower TLR4 and CD14 mRNA (from whole blood) in older women. Longer-term effects of a physically active lifestyle on short-lived cells like monocytes could occur if exercise somehow influenced bone marrow stem cells, and while much work needs to be done, Emmons et al.127 showed in murine models that exercise can alter trafficking of bone marrow derived hematopoietic stem cells.
TOWARD THE FUTURE
The data reviewed herein suggest the need to examine the pediatric origins of CVD in novel ways. The review highlights the value of exercise in eliciting monocyte function that might be hidden at rest. Although the value of studies directly in children in terms of relevance to the development of new therapeutic approaches is clear, any research in pediatric populations must take advantage of new technologies that expand the reach of minimally invasive approaches. Advances in understanding inflammatory mechanisms that contribute to the earliest manifestations of CVD will likely come from studies that combine a variety of technologies. Much work needs to be done. Dataintense CPET involving gas exchange, HR, and blood pressure variables must incorporate protocols that reflect real world patterns of exercise in children. Machine learning data analytics should now be applied to CPET to gain insights into gas exchange signals heretofore impossible to achieve with standardized approaches. Noninvasive measures of vascular anatomy and responsiveness using advanced ultrasound technology along with NIRS approaches to quantify blood flow and dynamic patterns of hemoglobin can be done in healthy children and in children with a variety of conditions (obesity, ALL survivors, or children with sickle cell disease) to yield a better understanding of abnormal exercise responses in specific diseases and conditions. Functional and genomic monocyte responses should be gauged in the context of these advanced dynamic phenotypic metrics. It is this synergy of tools that will ultimately lead to a better understanding of the earliest pathogenesis of CVD in children, and shed light on disease and risk progression across the lifespan.
ACKNOWLEDGEMENTS
This work was supported by NIH grants: P01HD048721, U01TR002004, and UL1TR001414.
Footnotes
Competing interests: The authors declare no competing interests.
REFERENCES
- 1.Ralston J, Reddy KS, Fuster V & Narula J Cardiovascular diseases on the global agenda. Glob. Heart 11, 375–379 (2016). [DOI] [PubMed] [Google Scholar]
- 2.Heindel JJ & Vandenberg LN Developmental origins of health and disease: a paradigm for understanding disease cause and prevention. Curr. Opin. Pediatr 27, 248–253 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Visentin S. et al. Early origins of adult disease: low birth weight and vascular remodeling. Atherosclerosis 237, 391–399 (2014). [DOI] [PubMed] [Google Scholar]
- 4.Mahoney LT et al. Coronary risk factors measured in childhood and young adult life are associated with coronary artery calcification in young adults: the Muscatine Study. J. Am. Coll. Cardiol 27, 277–284 (1996). [DOI] [PubMed] [Google Scholar]
- 5.Raitakari OT et al. Cardiovascular risk factors in childhood and carotid artery intima–media thickness in adulthood: the Cardiovascular Risk in Young Finns Study. JAMA 290, 2277–2283 (2003). [DOI] [PubMed] [Google Scholar]
- 6.Li S. et al. Childhood cardiovascular risk factors and carotid vascular changes in adulthood: the Bogalusa Heart Study. JAMA 290, 2271–2276 (2003). [DOI] [PubMed] [Google Scholar]
- 7.Loria CM et al. Early adult risk factor levels and subsequent coronary artery calcification: the CARDIA Study. J. Am. Coll. Cardiol 49, 2013–2020 (2007). [DOI] [PubMed] [Google Scholar]
- 8.Gidding SS et al. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) risk score in young adults predicts coronary artery and abdominal aorta calcium in middle age: the CARDIA study. Circulation 133, 139–146 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.van der Meer JWM, Joosten LAB, Riksen N & Netea MG Trained immunity: a smart way to enhance innate immune defence. Mol. Immunol 68, 40–44 (2015). [DOI] [PubMed] [Google Scholar]
- 10.Rigamonti E, Zordan P, Sciorati C, Rovere-Querini P & Brunelli S Macrophage plasticity in skeletal muscle repair. Biomed. Res. Int 2014, 560629 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Epelman S, Lavine KJ & Randolph GJ Origin and functions of tissue macrophages. Immunity 41, 21–35 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Radom-Aizik S, Zaldivar F Jr., Leu S-YY & Cooper DMM Brief bout of exercise alters gene expression in peripheral blood mononuclear cells of early- and late-pubertal males. Pediatr. Res 65, 447–452 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Radom-Aizik S, Zaldivar FP Jr., Haddad F & Cooper DM Impact of brief exercise on circulating monocyte gene and microRNA expression: implications for atherosclerotic vascular disease. Brain Behav. Immun 39, 121–129 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hallstrand TS, Altemeier WA, Aitken ML & Henderson WR Role of cells and mediators in exercise-induced bronchoconstriction. Immunol. Allergy Clin. N. Am 33, 313–328 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Idzkowska E. et al. The role of different monocyte subsets in the pathogenesis of therosclerosis and acute coronary syndromes. Scand. J. Immunol 82, 163–173 (2015). [DOI] [PubMed] [Google Scholar]
- 16.Gjevestad GO, Holven KB & Ulven SM Effects of exercise on gene expression of inflammatory markers in human peripheralblood cells: a systematic review. Curr. Cardiovasc. Risk Rep 9, 34 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Krüger K & Mooren FC Exercise-induced leukocyte apoptosis. Exerc. Immunol. Rev 20, 117–134 (2014). [PubMed] [Google Scholar]
- 18.Gano LB et al. Increased proinflammatory and oxidant gene expression in circulating mononuclear cells in older adults: amelioration by habitual exercise. Physiol. Genomics 43, 895–902 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fisher G. et al. Lymphocyte enzymatic antioxidant responses to oxidative stress following high-intensity interval exercise. J. Appl. Physiol 110, 730–737 (2011). [DOI] [PubMed] [Google Scholar]
- 20.Perez-Schindler J & Philp A Regulation of skeletal muscle mitochondrial function by nuclear receptors: implications for health and disease. Clin. Sci. (Lond.) 129, 589–599 (2015). [DOI] [PubMed] [Google Scholar]
- 21.Perez CJJ et al. Effects of laboratory versus field exercise on leukocyte subsets and cell adhesion molecule expression in children. Eur. J. Appl. Physiol 86, 34–39 (2001). [DOI] [PubMed] [Google Scholar]
- 22.Zhao XNN, Li YNN & Wang YTT Interleukin-4 regulates macrophage polarization via the MAPK signaling pathway to protect against atherosclerosis. Genet. Mol. Res http://www.ncbi.nlm.nih.gov/pubmed/26910000, 15 (2016). [DOI] [PubMed] [Google Scholar]
- 23.Masud R, Shameer K, Dhar A, Ding K & Kullo IJ Gene expression profiling of peripheral blood mononuclear cells in the setting of peripheral arterial disease. J. Clin. Bioinforma 2, 6 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hamers AAJ, Hanna RN, Nowyhed H, Hedrick CC & de Vries CJM NR4A nuclear receptors in immunity and atherosclerosis. Curr. Opin. Lipidol 24, 381–385 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kyotani Y. et al. Intermittent hypoxia induces the proliferation of rat vascular smooth muscle cell with the increases in epidermal growth factor family and erbB2 receptor. Exp. Cell Res 319, 3042–3050 (2013). [DOI] [PubMed] [Google Scholar]
- 26.Zhang H. et al. Heparin-binding epidermal growth factor-like growth factor signaling in flow-induced arterial remodeling. Circ. Res 102, 1275–1285 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nguyen BK et al. Improved function and myocardial repair of infarcted heart by intracoronary injection of mesenchymal stem cell-derived growth factors. J. Cardiovasc. Transl. Res 3, 547–558 (2010). [DOI] [PubMed] [Google Scholar]
- 28.Jaedicke KM et al. Leptin up-regulates TLR2 in human monocytes. J. Leukoc. Biol 93, 561–571 (2013). [DOI] [PubMed] [Google Scholar]
- 29.Klentrou P. Influence of exercise and training during critical stages of bone growth and development. Pediatr. Exerc. Sci http://www.ncbi.nlm.nih.gov/pubmed/26884506 (2016). [DOI] [PubMed] [Google Scholar]
- 30.Shay CM et al. Status of cardiovascular health in US adolescents: prevalence estimates from the National Health and Nutrition Examination Surveys (NHANES) 2005–2010. Circulation 127, 1369–1376 (2013). [DOI] [PubMed] [Google Scholar]
- 31.Gates PE, Strain WD & Shore AC Human endothelial function and microvascular ageing. Exp. Physiol 94, 311–316 (2009). [DOI] [PubMed] [Google Scholar]
- 32.Mielgo-Ayuso J. et al. Physical activity patterns of the Spanish population are mostly determined by sex and age: findings in the ANIBES study. PLoS ONE 11, e0149969 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Maher CA, Toohey M & Ferguson M Physical activity predicts quality of life and happiness in children and adolescents with cerebral palsy. Disabil. Rehabil 38, 865–869 (2016). [DOI] [PubMed] [Google Scholar]
- 34.Pacheco DRR, Silva MJB, Alexandrino AMS & Torres RMT Exercise-related quality of life in subjects with asthma: a systematic review. J. Asthma 49, 487–495 (2012). [DOI] [PubMed] [Google Scholar]
- 35.Kohl HW et al. The pandemic of physical inactivity: global action for public health. Lancet 380, 294–305 (2012). [DOI] [PubMed] [Google Scholar]
- 36.Briggs AM et al. Musculoskeletal health conditions represent a global threat to healthy aging: a report for the 2015 World Health Organization World Report on ageing and health. Gerontologist 56 (Suppl. 2), S243–S255 (2016). [DOI] [PubMed] [Google Scholar]
- 37.Sata F. Developmental Origins of Health and Disease (DOHaD) and Epidemiology. Nihon Eiseigaku Zasshi 71, 41–46 (2016). [DOI] [PubMed] [Google Scholar]
- 38.Chehade H, Simeoni U, Guignard J-P & Boubred F Preterm birth: long term cardiovascular and renal consequences. Curr. Pediatr. Rev 14, 219–226 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Stary HC Lipid and macrophage accumulations in arteries of children and the development of atherosclerosis. Am. J. Clin. Nutr 72, 1297S–1306S (2000). [DOI] [PubMed] [Google Scholar]
- 40.Noma K, Kihara Y & Higashi Y Outstanding effect of physical exercise on endothelial function even in children and adolescents. Circ. J 81, 637–639 (2017). [DOI] [PubMed] [Google Scholar]
- 41.Stevens A. et al. Human growth is associated with distinct patterns of gene expression in evolutionarily conserved networks. BMC Genomics 14, 547 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hoffman MC et al. Deficits in physical function among young childhood cancer survivors. J. Clin. Oncol 31, 2799–2805 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Schneiderman JE et al. Longitudinal relationship between physical activity and lung health in patients with cystic fibrosis. Eur. Respir. J 43, 817–823 (2014). [DOI] [PubMed] [Google Scholar]
- 44.Schmidt MD, Magnussen CG, Rees E, Dwyer T & Venn AJ Childhood fitness reduces the long-term cardiometabolic risks associated with childhood obesity. Int. J. Obes 40, 1134–1140 (2016). [DOI] [PubMed] [Google Scholar]
- 45.Liem RI et al. Reduced fitness and abnormal cardiopulmonary responses to maximal exercise testing in children and young adults with sickle cell anemia. Physiol. Rep http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4425953&tool=pmcentrez&rendertype=abstract, 3 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Elmesmari R, Reilly JJ, Martin A & Paton JY Accelerometer measured levels of moderate-to-vigorous intensity physical activity and sedentary time in children and adolescents with chronic disease: a systematic review and metaanalysis. PLoS ONE 12, e0179429 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Seals DR & Melov S Translational geroscience: emphasizing function to achieve optimal longevity. Aging (Albany, NY) 6, 718–730 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Foster GD et al. A school-based intervention for diabetes risk reduction. N. Engl. J. Med 363, 443–453 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Magnussen CG, Smith KJ & Juonala M When to prevent cardiovascular disease? As early as possible: lessons from prospective cohorts beginning in childhood. Curr. Opin. Cardiol 28, 561–568 (2013). [DOI] [PubMed] [Google Scholar]
- 50.Cheung PC, Cunningham SA, Naryan KMV & Kramer MR Childhood obesity incidence in the United States: a systematic review. Child Obes. 12, 1–11 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ness KK, Armenian SH, Kadan-Lottick N & Gurney JG Adverse effects of treatment in childhood acute lymphoblastic leukemia: general overview and implications for long-term cardiac health. Expert Rev. Hematol 4., 185–197 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lemay V. et al. Prevention of long-term adverse health outcomes with cardiorespiratory fitness and physical activity in childhood acute lymphoblastic leukemia survivors. J. Pediatr. Hematol. Oncol http://www.ncbi.nlm.nih.gov/pubmed/30688830, 1 (2019). [DOI] [PubMed] [Google Scholar]
- 53.Yildiz Kabak V, Calders P, Duger T, Mohammed J & van Breda E Short and long-term impairments of cardiopulmonary fitness level in previous childhood cancer cases: a systematic review. Support Care Cancer 27, 69–86 (2019). [DOI] [PubMed] [Google Scholar]
- 54.Sulicka J. et al. Elevated markers of inflammation and endothelial activation and increased counts of intermediate monocytes in adult survivors of childhood acute lymphoblastic leukemia. Immunobiology 218, 810–816 (2013). [DOI] [PubMed] [Google Scholar]
- 55.Perkins SM, Dewees T, Shinohara ET, Reddy MM & Frangoul H Risk of subsequent malignancies in survivors of childhood leukemia. J. Cancer Surviv 7, 544–550 (2013). [DOI] [PubMed] [Google Scholar]
- 56.Gibson TM, Ehrhardt MJ & Ness KK Obesity and metabolic syndrome among adult survivors of childhood leukemia. Curr. Treat. Options Oncol 17, 17 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sadurska E, Zaucha-Prażmo A, Brodzisz A, Kowalczyk J & Beń-Skowronek I Premature atherosclerosis after treatment for acute lymphoblastic leukemia in childhood. Ann. Agric. Environ. Med 25, 71–76 (2017). [DOI] [PubMed] [Google Scholar]
- 58.Giordano P. et al. Endothelial dysfunction and cardiovascular risk factors in childhood acute lymphoblastic leukemia survivors. Int. J. Cardiol 228, 621–627 (2017). [DOI] [PubMed] [Google Scholar]
- 59.Laitinen TT et al. Lifetime measures of ideal cardiovascular health and their association with subclinical atherosclerosis: the Cardiovascular Risk in Young Finns Study. Int. J. Cardiol 185, 186–191 (2015). [DOI] [PubMed] [Google Scholar]
- 60.Ajala O, Mold F, Boughton C, Cooke D & Whyte M Childhood predictors of cardiovascular disease in adulthood. A systematic review and meta-analysis. Obes. Rev http://www.ncbi.nlm.nih.gov/pubmed/28545166 (2017). [DOI] [PubMed] [Google Scholar]
- 61.Zaldivar F. et al. Body fat and circulating leukocytes in children. Int. J. Obes 30, 906–911 (2006). [DOI] [PubMed] [Google Scholar]
- 62.Engin A. in: Advances in Experimental Medicine and Biology 345–379 (2017).http://www.ncbi.nlm.nih.gov/pubmed/28585207. [DOI] [PubMed] [Google Scholar]
- 63.Mattos RT et al. Chronic low-grade inflammation in childhood obesity is associated with decreased IL-10 expression by monocyte subsets. PLoS ONE 11, e0168610 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Han X & Boisvert WA Interleukin-10 protects against atherosclerosis by modulating multiple atherogenic macrophage function. Thromb. Haemost 113, 505–512 (2014). [DOI] [PubMed] [Google Scholar]
- 65.Ischander M. et al. Physical activity, growth, and inflammatory mediators in BMI-matchedfemale adolescents. Med. Sci. Sports Exerc 39, 1131–1138 (2007). [DOI] [PubMed] [Google Scholar]
- 66.Motoki Y. et al. Anti-phospholipid antibodies contribute to arteriosclerosis in patients with systemic lupus erythematosus through induction of tissue factor expression and cytokine production from peripheral blood mononuclear cells. Thromb. Res 130, 667–673 (2012). [DOI] [PubMed] [Google Scholar]
- 67.Aloi M. et al. Aortic intima–media thickness as an early marker of atherosclerosis in children with inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr 61, 1 (2015). [DOI] [PubMed] [Google Scholar]
- 68.Curcio S. et al. Growing-related changes in arterial properties of healthy children, adolescents, and young adults nonexposed to cardiovascular risk factors: analysis of gender-related differences. Int. J. Hypertens 2016, 4982676 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Armstrong N, Barker AR & McManus AM Muscle metabolism changes with age and maturation: How do they relate to youth sport performance? Br. J. Sports Med 49, 860–864 (2015). [DOI] [PubMed] [Google Scholar]
- 70.Blimkie CJ Resistance training during preadolescence. Issues controversies. Sports Med. 15, 389–407 (1993). [DOI] [PubMed] [Google Scholar]
- 71.Behringer M, Vom Heede A, Yue Z & Mester J Effects of resistance training in children and adolescents: a meta-analysis. Pediatrics 126, e1199–e1210 (2010). [DOI] [PubMed] [Google Scholar]
- 72.Armstrong N & Barker AR Endurance training and elite young athletes. Med. Sport Sci 56, 59–83 (2011). [DOI] [PubMed] [Google Scholar]
- 73.Zanconato S, Cooper DM & Armon Y Oxygen cost and oxygen uptake dynamics and recovery with 1 min of exercise in children and adults. J. Appl. Physiol 71, 993–998 (1991). [DOI] [PubMed] [Google Scholar]
- 74.Armstrong N & Barker AR Oxygen uptake kinetics in children and adolescents: a review. Pediatr. Exerc. Sci 21, 130–147 (2009). [DOI] [PubMed] [Google Scholar]
- 75.Armon Y, Cooper DMM & Zanconato S Maturation of ventilatory responses to 1-minute exercise. Pediatr. Res 29, 362–368 (1991). [DOI] [PubMed] [Google Scholar]
- 76.Cooper DM et al. Coupling of ventilation and CO2 production during exercise in children. Pediatr. Res 21, 568–572 (1987). [DOI] [PubMed] [Google Scholar]
- 77.Dotan R & Falk B Discussion: “The kinetics of blood lactate in boys during and following a single and repeated all-out sprints of cycling are different than in men”—Do children indeed release and remove lactate faster than adults? Appl . Physiol. Nutr. Metab 40, 632–633 (2015). [DOI] [PubMed] [Google Scholar]
- 78.Zanconato S, Buchthal S, Barstow TJJ & Cooper DMM 31P-magnetic resonance spectroscopy of leg muscle metabolism during exercise in children and adults. J. Appl. Physiol 74, 2214–2218 (1993). [DOI] [PubMed] [Google Scholar]
- 79.Armon Y. et al. Oral [13C]bicarbonate measurement of CO2 stores and dynamics in children and adults. J. Appl. Physiol 69, 1754–1760 (1990). [DOI] [PubMed] [Google Scholar]
- 80.Chen JJ et al. Age, sex and weight effects on lactate and leukocyte response to exercise in children and adolescents. Eur. J. Sport Exerc. Sci 6, 1–10 (2018). [PMC free article] [PubMed] [Google Scholar]
- 81.Laina A, Stellos K & Stamatelopoulos K Vascular ageing: underlying mechanisms and clinical implications. ExpGerontol. http://www.ncbi.nlm.nih.gov/pubmed/28624356 (2017). [DOI] [PubMed] [Google Scholar]
- 82.Gomez-Sanchez L. et al. Leukocyte subtype counts and its association with vascular structure and function in adults with intermediate cardiovascular risk. MARK Study. PLoS ONE 10, e0119963 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Brown RA, Shantsila E, Varma C & Lip GYH Epidemiology and pathogenesis of diffuse obstructive coronary artery disease: the role of arterial stiffness, shear stress, monocyte subsets and circulating microparticles. Ann. Med 48, 444–455 (2016). [DOI] [PubMed] [Google Scholar]
- 84.Núñez F. et al. Carotid artery stiffness as an early marker of vascular lesions in children and adolescents with cardiovascular risk factors. Rev. Esp. Cardiol 63, 1253–1260 (2010). [DOI] [PubMed] [Google Scholar]
- 85.Riggio S. et al. Pulse wave velocity and augmentation index, but not intima–media thickness, are early indicators of vascular damage in hypercholesterolemic children. Eur. J. Clin. Invest 40, 250–257 (2010). [DOI] [PubMed] [Google Scholar]
- 86.Genovesi S. et al. Analysis of heart period and arterial pressure variability in childhood hypertension: key role of baroreflex impairment. Hypertension 51, 1289–1294 (2008). [DOI] [PubMed] [Google Scholar]
- 87.Weberruß H. et al. Reduced arterial stiffness in very fit boys and girls. Cardiol. Young 27, 117–124 (2017). [DOI] [PubMed] [Google Scholar]
- 88.Ried-Larsen M, Grøntved A, Froberg K, Ekelund U & Andersen LB Physical activity intensity and subclinical atherosclerosis in Danish adolescents: the European Youth Heart Study. Scand. J. Med. Sci. Sports 23, e168–e177 (2013). [DOI] [PubMed] [Google Scholar]
- 89.Donghui T. et al. Improvement of microvascular endothelial dysfunction induced by exercise and diet is associated with microRNA-126 in obese adolescents. Microvasc. Res 123, 86–91 (2019). [DOI] [PubMed] [Google Scholar]
- 90.Rozenbaum Z. et al. Discriminating circulatory problems from deconditioning. Chest 151, 431–440 (2017). [DOI] [PubMed] [Google Scholar]
- 91.Cooper DMDM et al. Cardiopulmonary exercise testing in children and adolescents with high body mass index. Pediatr. Exerc. Sci 28, 98–108 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Cooper DM, Leu SY, Galassetti P & Radom-Aizik S Dynamic interactions of gas exchange, body mass, and progressive exercise in children. Med. Sci. Sports Exerc 46, 877–886 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Adamska A. et al. Dermal microvessel density and maturity is closely associated with atherogenic dyslipidemia and accumulation of advanced glycation end products in adult patients with type 1 diabetes. Microvasc. Res 121, 46–51 (2019). [DOI] [PubMed] [Google Scholar]
- 94.Manetos C. et al. Skeletal muscle microcirculatory abnormalities are associated with exercise intolerance, ventilatory inefficiency, and impaired autonomic control in heart failure. J. Heart Lung Transpl 30, 1403–1408 (2011). [DOI] [PubMed] [Google Scholar]
- 95.Manfredini F. et al. Near-infrared spectroscopy assessment following exercise training in patients with intermittent claudication and in untrained healthy participants. Vasc. Endovasc. Surg 46, 315–324 (2012). [DOI] [PubMed] [Google Scholar]
- 96.Luck JC et al. Blood pressure and calf muscle oxygen extraction during plantar flexion exercise in peripheral artery disease. J. Appl. Physiol 123, 2–10 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Liem RI et al. The acute phase inflammatory response to maximal exercise testing in children and young adults with sickle cell anaemia. Br. J. Haematol 171, 854–861 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ragab SM & Soliman MA Tissue factor-positive monocytes expression in children with sickle cell disease. Blood Coagul. Fibrinolysis 27, 862–869 (2016). [DOI] [PubMed] [Google Scholar]
- 99.Kohl III et al. The pandemic of physical inactivity: global action for public health. Lancet 380, 294–305 (2012). [DOI] [PubMed] [Google Scholar]
- 100.Füssenich LM et al. Physical activity guidelines and cardiovascular risk in children: a cross sectional analysis to determine whether 60 minutes is enough. BMC Public Health 16, 67 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wong KL et al. The three human monocyte subsets: implications for health and disease. Immunol.Res 53, 41–57 (2012). [DOI] [PubMed] [Google Scholar]
- 102.Berg KE et al. Elevated CD14++. Circ. Cardiovasc. Genet 5, 122–131 (2012). [DOI] [PubMed] [Google Scholar]
- 103.Mehta NN & Reilly MP Monocyte mayhem: do subtypes modulate distinct atherosclerosis phenotypes? Circ. Cardiovasc. Genet 5, 7–9 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Devêvre EF et al. Profiling of the three circulating monocyte subpopulations in human obesity. J. Immunol 194, 3917–3923 (2015). [DOI] [PubMed] [Google Scholar]
- 105.Rogacev KS et al. CD14++CD16+ monocytes independently predict cardiovascular events: a cohort study of 951 patients referred for elective coronary angiography. J. Am. Coll. Cardiol 60, 1512–1520 (2012). [DOI] [PubMed] [Google Scholar]
- 106.Heimbeck I. et al. Standardized single-platform assay for human monocyte subpopulations: lower CD14+CD16++ monocytes in females. Cytom. A 77, 823–830 (2010). [DOI] [PubMed] [Google Scholar]
- 107.Wong KL et al. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood 118, e16–e31 (2011). [DOI] [PubMed] [Google Scholar]
- 108.Villani A-C et al. Single-cell RNA-seq reveals new types of human blood dendritic cells, monocytes, and progenitors. Science 356, eaah4573 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Wang X. et al. An on-chip microfluidic pressure regulator that facilitates reproducible loading of cells and hydrogels into microphysiological system platforms. Lab Chip 16, 868–876 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sonna LA et al. Exertional heat injury and gene expression changes: a DNA microarray analysis study. J. Appl. Physiol 96, 1943–1953 (2004). [DOI] [PubMed] [Google Scholar]
- 111.Jancic CC et al. Low extracellular pH stimulates the production of IL-1beta by human monocytes. Cytokine 57, 258–268 (2012). [DOI] [PubMed] [Google Scholar]
- 112.Park KH, Lee TH, Kim CW & Kim J Enhancement of CCL15 expression and monocyte adhesion to endothelial cells (ECs) after hypoxia/reoxygenation and induction of ICAM-1 expression by CCL15 via the JAK2/STAT3 pathway in ECs. J. Immunol 190, 6550–6558 (2013). [DOI] [PubMed] [Google Scholar]
- 113.Mitchell MJ, Lin KS & King MR Fluid shear stress increases neutrophil activation via platelet-activating factor. Biophys. J 106, 2243–2253 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Horstmann J-P, Marzi I & Relja B Adrenergic stimulation alters the expression of inflammasome components and interleukins in primary human monocytes. Exp. Ther. Med 11, 297–302 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Takeda K. et al. IFN-gamma production by lung NK cells is critical for the natural resistance to pulmonary metastasis of B16 melanoma in mice. J. Leukoc. Biol 90, 777–785 (2011). [DOI] [PubMed] [Google Scholar]
- 116.Dimitrov S, Hulteng E & Hong S Inflammation and exercise: Inhibition of monocytic intracellular TNF production by acute exercise via β2-adrenergic activation. Brain Behav. Immun 61, 60–68 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Durrer C, Francois M, Neudorf H & Little JP Acute high-intensity interval exercise reduces human monocyte Toll-like receptor 2 expression in type 2 diabetes. Am. J. Physiol. Regul. Integr. Comp. Physiol 312, R529–R538 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Patel VK, Williams H, Li SCH, Fletcher JP & Medbury HJ Monocyte inflammatory profile is specific for individuals and associated with altered blood lipid levels. Atherosclerosis 263, 15–23 (2017). [DOI] [PubMed] [Google Scholar]
- 119.Lew J. et al. Sex-based differences in cardiometabolic biomarkers-clinical perspective.Circulation 135, 544–555 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Campesi I, Marino M, Montella A, Pais S & Franconi F Sex differences in estrogen receptor α and β levels and activation status in LPS-stimulated human macrophages. J. Cell. Physiol 232, 340–345 (2017). [DOI] [PubMed] [Google Scholar]
- 121.Klein SL & Flanagan KL Sex differences in immune responses. Nat. Rev. Immunol 16, 626–638 (2016). [DOI] [PubMed] [Google Scholar]
- 122.Ostan R. et al. Gender, aging and longevity in humans: an update of an intriguing/neglected scenario paving the way to a gender-specific medicine. Clin. Sci 130, 1711–1725 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Pérez-de-Heredia F. et al. Influence of sex, age, pubertal maturation and body mass index on circulating white blood cell counts in healthy European adolescents—the HELENA study. Eur. J. Pediatr 174, 999–1014 (2015). [DOI] [PubMed] [Google Scholar]
- 124.Metcalf TU et al. Human monocyte subsets are transcriptionally and functionally altered in aging in response to pattern recognition receptor agonists. J. Immunol http://www.ncbi.nlm.nih.gov/pubmed/28696254 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Queiroga MR et al. Influence of cardiorespiratory fitness on PPARG mRNA expression using monozygotic twin case control. J. Diabetes Res 2015, 538732 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Flynn MG, McFarlin BK, Phillips MD, Stewart LK & Timmerman KL Toll-like receptor 4 and CD14 mRNA expression are lower in resistive exercise-trained elderly women. J. Appl. Physiol 95, 1833–1842 (2003). [DOI] [PubMed] [Google Scholar]
- 127.Emmons R, Niemiro GM, Owolabi O & De Lisio M Acute exercise mobilizes hematopoietic stem and progenitor cells and alters the mesenchymal stromal cell secretome. J. Appl. Physiol 120, 624–632 (2016). [DOI] [PubMed] [Google Scholar]
- 128.Radom-Aizik S, Zaldivar FP, Haddad F & Cooper DMM Impact of brief exercise on peripheral blood NK cell gene and microRNA expression in young adults. J. Appl. Physiol 114, 628–636 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Radom-Aizik S, Zaldivar F Jr., Leu S-YY, Galassetti P & Cooper DMM Effects of 30 min of aerobic exercise on gene expression in human neutrophils. J. Appl. Physiol 104, 236–243 (2008). [DOI] [PubMed] [Google Scholar]
- 130.Baraldi E, Cooper DMM, Zanconato S & Armon Y Heart rate recovery from 1 minute of exercise in children and adults. Pediatr. Res 29, 575–579 (1991). [DOI] [PubMed] [Google Scholar]
- 131.Febbraio M & Silverstein RL CD36: implications in cardiovascular disease. Int. J. Biochem. Cell Biol 39, 2012–2030 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.den Dekker WK, Cheng C, Pasterkamp G & Duckers HJ Toll like receptor 4 in atherosclerosis and plaque destabilization. Atherosclerosis 209, 314–320 (2010). [DOI] [PubMed] [Google Scholar]
- 133.Wight TN & Merrilees MJ Proteoglycans in atherosclerosis and restenosis: key roles for versican. Circ. Res 94, 1158–1167 (2004). [DOI] [PubMed] [Google Scholar]
- 134.Lu X & Kakkar V The role of heat shock protein (HSP) in atherosclerosis: pathophysiology and clinical opportunities. Curr. Med. Chem 17, 957–973 (2010). [DOI] [PubMed] [Google Scholar]
- 135.Findeisen HM et al. Epigenetic regulation of vascular smooth muscle cell proliferation and neointima formation by histone deacetylase inhibition. Arterioscler. Thromb. Vasc. Biol 31, 851–860 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Zhang Q, Kandic I & Kutryk MJ Dysregulation of angiogenesis-related microRNAs in endothelial progenitor cells from patients with coronary artery disease. Biochem. Biophys. Res. Commun 405, 42–46 (2011). [DOI] [PubMed] [Google Scholar]
- 137.Li T. et al. Identification of miR-130a, miR-27b and miR-210 as serum biomarkers for atherosclerosis obliterans. Clin. Chim. Acta 412, 66–70 (2011). [DOI] [PubMed] [Google Scholar]
- 138.Chistiakov DA, Sobenin IA, Orekhov AN & Bobryshev YV Human miR-221/222 in physiological and atherosclerotic vascular remodeling. Biomed. Res Int 2015, 354517 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zheng J. et al. MicroRNA-23b promotes tolerogenic properties of dendritic cells in vitro through inhibiting Notch1/NF-kappaB signalling pathways. Allergy 67, 362–370 (2012). [DOI] [PubMed] [Google Scholar]
- 140.Chen KC et al. OxLDL up-regulates microRNA-29b, leading to epigenetic modifications of MMP-2/MMP-9 genes: a novel mechanism for cardiovascular diseases. FASEB J. 25, 1718–1728 (2011). [DOI] [PubMed] [Google Scholar]
- 141.Lee J. et al. MicroRNA-29b inhibits migration and proliferation of vascular smooth muscle cells in neointimal formation. J. Cell. Biochem 116, 598–608 (2015). [DOI] [PubMed] [Google Scholar]
- 142.Liu DZ et al. Brain and blood microRNA expression profiling of ischemic stroke, intracerebral hemorrhage, and kainate seizures. J. Cereb. Blood Flow. Metab 30, 92–101 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Emmrich S. et al. miRNAs can increase the efficiency of ex vivo platelet generation. Ann. Hematol 91, 1673–1684 (2012). [DOI] [PubMed] [Google Scholar]
- 144.Bye A. et al. Circulating microRNAs predict future fatal myocardial infarction in healthy individuals—The HUNT study. J. Mol. Cell. Cardiol http://www.ncbi.nlm.nih.gov/pubmed/27192016 (2016). [DOI] [PubMed] [Google Scholar]
- 145.Ortega FJ et al. Targeting the circulating microRNA signature of obesity. Clin. Chem 59, 781–792 (2013). [DOI] [PubMed] [Google Scholar]
- 146.Han H. et al. Differentiated miRNA expression and validation of signaling pathways in apoE gene knockout mice by cross-verification microarray platform. Exp. Mol. Med 45, e13 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Liu YF et al. MicroRNA-30 inhibits neointimal hyperplasia by targeting Ca(2+)/ calmodulin-dependent protein kinase IIδ (CaMKIIδ). Sci. Rep 6, 26166 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Rink C & Khanna S MicroRNA in ischemic stroke etiology and pathology. Physiol. Genomics 43, 521–528 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Stather PW et al. Identification of microRNAs associated with abdominal aortic aneurysms and peripheral arterial disease. Br. J. Surg 102, 755–766 (2015). [DOI] [PubMed] [Google Scholar]