Abstract
Photodynamic therapy (PDT) with verteporfin involves intravenous administration of a photosensitizer followed by its laser light activation at the target site to inhibit aberrant choroidal vascularization. This narrative review provides an overview of the role verteporfin PDT plays in the management of chorioretinal conditions. A PubMed literature review of all English-language articles published through October 19, 2023, was conducted to identify relevant references. Verteporfin PDT has been shown to be safe and effective for the treatment of patients with choroidal neovascularization (CNV) due to neovascular age-related macular degeneration and is often used in combination with a vascular endothelial growth factor (VEGF) inhibitor. Additionally, patients with polypoidal choroidal vasculopathy, a subtype of neovascular age-related macular degeneration, also benefit from verteporfin PDT combined with a VEGF inhibitor for improving visual acuity. Verteporfin PDT has also been effective in treating patients with peripapillary CNV, as well as eyes with CNV due to ocular histoplasmosis and pathologic myopia. Reduced dose and/or fluence PDT protocols have been effective in patients with central serous chorioretinopathy while reducing adverse effects. In eyes with choroidal hemangioma, tumor regression and visual outcomes have been improved with verteporfin PDT treatment. Photodynamic therapy with verteporfin continues to play an important role in the management of chorioretinal conditions.
Keywords: choroid, retina, macular degeneration, vascularization, VEGF inhibitor
Introduction
In the 1980s, photodynamic therapy (PDT) was introduced as a topical oncology treatment, given its favorable adverse event (AE) profile and notably decreased skin phototoxicity.1 In the 1990s, PDT was developed as a treatment for subfoveal choroidal neovascularization (CNV) in age-related macular degeneration (AMD).1 In ophthalmologic applications, PDT involves intravenous administration of the nontoxic photosensitizer verteporfin, followed by laser activation.1,2 Verteporfin is transported in the bloodstream by lipoproteins to the ocular pathogenic site of interest and binds to the low-density lipoprotein receptor that is increasingly expressed by the abnormal neovascular endothelial cells at the affected area.1,3
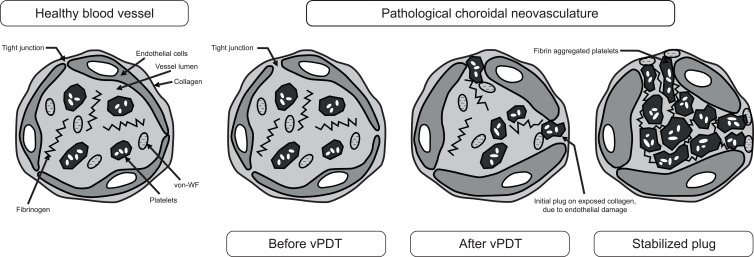
Verteporfin is administered intravenously over 10 minutes, followed by a 5-minute wait to enable localization at the target area before activation using a low-energy laser light (far-red wavelength of 689 nm).1,2 Excitation of verteporfin results in the production of both singlet oxygen and reactive oxygen species at the target site, resulting in vascular endothelial cell damage, apoptosis, and restructuring of the neovascularization at the affected site.1,2,4 Further, by releasing factors such as histamines, thromboxane, and tumor necrosis factor-α, vPDT treatment results in blood flow stasis and occlusion at the CNV site (Figure 1).1,2,5–11 Verteporfin for injection is approved for the treatment of patients with predominantly classic subfoveal CNV due to neovascular AMD, pathologic myopia, or presumed ocular histoplasmosis (POHS).12 Standard PDT with verteporfin (vPDT) involves the intravenous injection of verteporfin 6 mg/m2, followed by targeted irradiation with fluence 50 J/cm2.13 Protocols that involved decreased dosing of verteporfin (eg, to 3 mg/m2) and/or reduced fluence (eg, half-dose/half-fluence) were developed to potentially diminish some of the adverse effects observed with standard vPDT (eg, retinal pigment epithelium [RPE] damage, bacillary retinal detachment).13–15
Figure 1.
Mechanism of action of vPDT at the site of the blood vessel wall.7 Verteporfin PDT induces endothelial damage, including rounding of endothelial cells, which allows for clotting factors to be released at the site of damage and subsequent recruitment of platelets that form an occlusion. Adapted from Debefve E, Pegaz B, van den Bergh H, Wagnieres G, Lange N, Ballini JP. Video monitoring of neovessel occlusion induced by photodynamic therapy with verteporfin (Visudyne), in the CAM model. Angiogenesis. 2008;11(3):235–243. Creative Commons.7
Abbreviations: von-WF, von Willebrand; vPDT, verteporfin photodynamic therapy.
Until the introduction in the mid-2000s of vascular endothelial growth factor (VEGF) inhibitors, vPDT was the treatment of choice for several chorioretinal conditions.16 In the healthy eye, basal VEGF secretion from the RPE maintains the choroidal integrity and supports the survival of the RPE.17,18 However, in eyes with CNV, the polarity of the RPE is compromised, and VEGF secretion is increased.18 VEGF inhibitors target and bind specific VEGF receptors, inhibiting downstream angiogenesis and leakage.19 Although intravitreal VEGF inhibitors are typically used as first-line therapy for neovascular AMD, vPDT remains a viable primary or adjunct treatment option for many chorioretinal conditions.16,20 The aim of this review is to provide an overview of the applications, safety, and efficacy of vPDT, either as monotherapy or combined with VEGF inhibitors, in the treatment of chorioretinal conditions, including choroidal neovascular membranes from neovascular AMD (including polypoidal choroidal vasculopathy [PCV]), POHS, pathologic myopia, and peripapillary CNV, as well as central serous chorioretinopathy (CSC) and choroidal hemangioma.
Methods
A review in PubMed of English-language articles published through October 19, 2023, was conducted. Search terms included “verteporfin”, “photodynamic therapy”, “choroidal neovascularization”, “age-related macular degeneration”, “polypoidal choroidal vasculopathy”, “central serous chorioretinopathy”, “choroidal hemangioma”, “peripapillary choroidal neovascularization”, “presumed ocular histoplasmosis”, and “pathologic myopia”. For CNV associated with AMD, clinical studies identified for inclusion in this review were randomized, controlled studies of ≥150 patients. Inclusion criteria were less stringent for other conditions, given that fewer clinical studies exist for these conditions.
Results
Subfoveal Choroidal Neovascularization Due to Age-Related Macular Degeneration
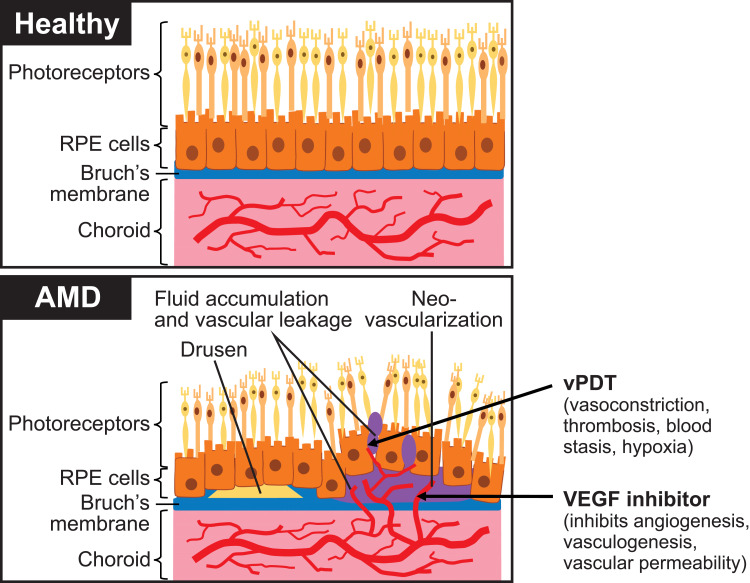
Neovascular AMD is an advanced form of AMD characterized by choroidal neovascular membrane formation under or above the RPE or occasionally within the retina.21 Neovascular AMD occurs in approximately 10% to 15% of patients with AMD, yet accounts for about 90% of all vision loss.22,23 In patients with neovascular AMD, growth of new vasculature from the choroid and through Bruch’s membrane (collagen- and elastin-abundant extracellular matrix layer under the RPE) enables vascular leakage through the now-compromised RPE (Figure 2).14,24,25 Vision loss may occur secondary to RPE detachment, subretinal hemorrhage, and/or photoreceptor cell damage due to vascular leakage.24 Polypoidal choroidal vasculopathy is a subtype of neovascular AMD and is discussed separately in a later section of this article.
Figure 2.
Normal eye compared with choroidal neovascularization in age-related macular degeneration. Arrows show drug targets and mode of action.14,24,25 Adapted from Rastoin O, Pages G, Dufies M. Experimental models in neovascular age related macular degeneration. Int J Mol Sci. 2020;21(13):4627. Creative Commons.24
Abbreviations: AMD, age-related macular degeneration; RPE, retinal pigment epithelium; VEGF, vascular endothelial growth factor; vPDT, verteporfin photodynamic therapy.
Early detection of neovascular AMD is critical, as if not treated promptly, central vision loss might be irreversible.26 The efficacy and safety of vPDT were assessed in several randomized clinical trials (Table 1).27–29 In the Treatment of Age-Related Macular Degeneration With Photodynamic Therapy (TAP) study, vPDT demonstrated efficacy compared with placebo in patients with neovascular AMD (ie, patients lost <15 letters of visual acuity [VA] from baseline) at 12 and 24 months.27,30 Findings of the Verteporfin in Photodynamic Therapy (VIP) study supported findings of the TAP study, with vPDT demonstrating efficacy in VA compared with placebo at 12 and 24 months.29
Table 1.
Summary of Select Clinical Trials of vPDT for the Treatment of Choroidal Neovascularization Associated with Age-Related Macular Degeneration
| Study and Design | Patient Population | Treatments | Efficacy Outcomes | Safety Outcomes |
|---|---|---|---|---|
| vPDT vs placebo | ||||
| TAP 12 mo27 24 mo30 R, MC, DM, PBO-C US (11 sites) Canada (2 sites) Europe (9 sites) N=609 (609 eyes) Age: ≥50 y |
Pts with CNV associated with AMD | vPDT (n=402) vs PBO (n=207) |
vPDT vs PBO % eyes with <15 letters lost (<3 lines) of VA from baseline
|
12 mo (vPDT vs PBO) Tx-related AEs: 46% vs 36% Photosensitivity reactions with vPDT
Tx-related discontinuation (only with vPDT)
24 mo (vPDT vs PBO) Tx-related AEs: 47.8% vs 33.8% Photosensitivity reactions with vPDT
No additional tx-related discontinuations |
| VIP29 R, MC, DM, PBO-C Europe and North America (28 sites) N=339 (339 eyes) |
Pts with CNV associated with AMD | vPDT (n=225) vs PBO (n=114) |
vPDT vs PBO % eyes with ≥15 letters lost of VA from baseline
|
vPDT 10 pts (4%) had severe decrease in VA within 7 d of tx; some pts experienced subsequent improvement in VA <1% pts had photosensitivity reactions |
| VEGF inhibitor vs sham | ||||
| MARINA31 R, MC, DM, Sham-C US (96 sites) N=716 |
Pts with CNV associated with AMD | Ranibizumab 0.5 mg (n=240) Ranibizumab 0.3 mg (n=238) Sham injection (n=238) |
Ranibizumab 0.5 mg and 0.3 mg vs sham injection % pts losing <15 letters of VA from baseline
|
Ranibizumab 0.5 mg and 0.3 mg vs sham injection AEs at 24 mo
|
| vPDT vs VEGF inhibitor | ||||
| ANCHOR 12 mo32 24 mo33 R, MC, DM 83 sites N=423 Age: ≥50 y |
Pts with CNV associated with AMD | Ranibizumab 0.5 mg (n=140) Ranibizumab 0.3 mg (n=140) vPDT (n=143) |
Ranibizumab 0.5 mg and 0.3 mg vs vPDT % pts losing <15 letters of VA from baseline
|
Ranibizumab 0.5 mg and 0.3 mg vs vPDT AEs at 12 mo
AEs at 24 mo
|
| vPDT combination therapy | ||||
| FOCUS 1 yr34 2 yr35 R, SM, MC US (25 sites) N=162 |
Pts with CNV associated with AMD | vPDT + ranibizumab 0.5 mg (n=106) vPDT + sham injection (n=56) |
vPDT + ranibizumab 0.5 mg vs vPDT % pts losing <15 letters of VA from baseline
|
vPDT + ranibizumab 0.5 mg vs vPDT Nonocular AEs more common with combination therapy vs vPDT at 1 y
Combination therapy vs vPDT at 2 y
|
| DENALI36 R, MC, DM, ph 3b N=321 |
Pts with CNV associated with AMD | vPDT + ranibizumab 0.5 mg (n=104) vPDT (reduced fluence) + ranibizumab 0.5 mg (n=105) Ranibizumab 0.5 mg (n=112) |
vPDT + ranibizumab 0.5 mg vs vPDT (reduced-fluence) + ranibizumab 0.5 mg vs ranibizumab 0.5 mg Mean change from baseline in BCVA at 12 mo: 5.3, 4.4, vs 8.1 letters
% pts with ranibizumab tx-free interval of ≥3 mo after third consecutive monthly ranibizumab injection: 92.6% and 83.5% (combination tx only) |
vPDT + ranibizumab 0.5 mg vs vPDT (reduced-fluence) + ranibizumab 0.5 mg vs ranibizumab 0.5 mg Ocular AEs at 12 mo: 60.6%, 52.8%, 54.1%
|
| MONT BLANC37 R, MC, DM Europe (45 sites) N=255 Age: ≥50 y |
Pts with CNV associated with AMD | vPDT + ranibizumab 0.5 mg (n=122) Ranibizumab 0.5 mg (n=133) |
vPDT + ranibizumab 0.5 mg vs ranibizumab 0.5 mg Mean change from baseline in BCVA at 12 mo: 2.5 vs 4.4 letters (P=0.005)
% pts with ranibizumab tx-free interval ≥3 mo after 2 mo: 96% and 92% |
vPDT + ranibizumab 0.5 mg vs ranibizumab 0.5 mg Ocular AEs at 12 mo: 41.8% vs 40.6%
|
| RADICAL38 R, MC, SM, ph 2 N=162 (162 eyes) Age: ≥50 y |
Pts with CNV associated with AMD | vPDT (half-fluence) + ranibizumab 0.5 mg + dexamethasone 0.5 mg (n=39) vPDT (quarter-fluence) + ranibizumab 0.5 mg + dexamethasone 0.5 mg (n=39) vPDT (half-fluence) + ranibizumab 0.5 mg (n=43) Ranibizumab 0.5 mg (n=41) |
vPDT (half-fluence) + ranibizumab + dexamethasone vs vPDT (quarter-fluence) + ranibizumab + dexamethasone vs vPDT (half-fluence) + ranibizumab vs ranibizumab Retreatments
Mean change (95% CI) from baseline in VA letter score
|
vPDT (half-fluence) + ranibizumab + dexamethasone vs vPDT (quarter-fluence) + ranibizumab + dexamethasone vs vPDT (half-fluence) + ranibizumab vs ranibizumab Tx-related AEs at 24 mo: 44%, 49%, 47%, 27%
|
Abbreviations: AE, adverse event; AMD, age-related macular degeneration; ANCHOR, Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in Age-Related Macular Degeneration; BCVA, best-corrected visual acuity; CI, confidence interval; CNV, choroidal neovascularization; DM, double-masked; FOCUS, RhuFab V2 Ocular Treatment Combining the Use of Visudyne to Evaluate Safety; GI, gastrointestinal; IOP, intraocular pressure; MARINA, Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular Age-Related Macular Degeneration; MC, multicenter; PBO, placebo; PBO-C, placebo-controlled; ph, phase; pt, patient; R, randomized; RADICAL, Reduced Fluence Visudyne-Anti-VEGF-Dexamethasone in Combination for AMD Lesions; SAE, serious adverse event; Sham-C, sham-controlled; SM, single-masked; TAP, Treatment of Age-Related Macular Degeneration With Photodynamic Therapy; Tx, treatment; VA, visual acuity; VEGF, vascular endothelial growth factor; VIP, Verteporfin in Photodynamic Therapy; vPDT, verteporfin PDT.
The efficacy of VEGF inhibitors was initially demonstrated in two Phase 3 studies.31–33 The Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular Age-Related Macular Degeneration (MARINA) study demonstrated that a significantly greater number of patients treated with ranibizumab lost <15 letters of VA from baseline compared with sham injection at 12 and 24 months (P<0.001 for both doses of ranibizumab versus sham at 12 and 24 months; Table 1).31 Results of the Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in Age-Related Macular Degeneration (ANCHOR) study led to VEGF inhibitors supplanting vPDT as the treatment of choice for patients with neovascular AMD, as ranibizumab resulted in a significantly greater percentage of patients experiencing improvement in VA compared with vPDT for up to 24 months.32,33 Clinical trials with VEGF inhibitors have demonstrated benefit to patients with neovascular AMD but have shown some limitations, including continuous vision loss despite treatment.39,40 In addition, one study found that only approximately 40% of patients achieved 20/40 vision or better after 12 months.31 Treatment adherence may be negatively affected by the schedule and/or number of injections and the associated financial, social, and caregiver burden.22,39 More recently, combining VEGF inhibitors with vPDT has been shown to improve VA and reduce the number of retreatments compared with VEGF inhibitor monotherapy (Table 1).34–38 Indeed, a systematic review of clinical studies conducted in Japanese patients with neovascular AMD showed that VEGF inhibitors as monotherapy or in combination with vPDT were more effective than vPDT monotherapy.41
Guidelines from the National Institute for Health and Care Excellence (NICE) and the Royal College of Ophthalmologists state that patients with neovascular AMD should be treated with VEGF inhibitors within 2 weeks of referral to a specialist.42,43 VEGF inhibitors currently available for the treatment of AMD include bevacizumab, ranibizumab, aflibercept 2 mg and 8 mg, brolucizumab, and faricimab.39,44–46
Polypoidal Choroidal Vasculopathy
Polypoidal choroidal vasculopathy, a clinically distinct subtype of exudative AMD, is characterized by subretinal polypoidal orange-yellow vascular lesions associated with serous and hemorrhagic RPE and retinal detachments.20,42,43,47–49 Diagnosis of PCV is based on the presence of polypoidal dilatations, which can be observed using optical coherence tomography but more readily with indocyanine green angiography; otherwise, differentiating PCV from AMD can be difficult.50 The etiology and pathophysiology of PCV are not well understood.20 However, the pachychoroid, or thick choroid, concept has emerged to explain, at least partially, the pathogenesis of PCV, with some investigators suggesting that PCV can be further subclassified as pachychoroid or non-pachychoroid PCV.51 However, it should be noted that the presence of a thick choroid on its own is not necessarily pathogenic in nature, as healthy eyes can have pachychoroid.52 In eyes with pachychoroid phenotype, thick choroid is typically observed with abnormally dilated choroidal vessels in Haller’s layer (pachyvessels), along with thinning of Sattler’s layer and the choriocapillaris overlying the pachyvessels.52 In PCV, choroidal hyperpermeability may be detected, hemorrhage from aneurysms can occur, and soft drusen are absent.52
While anti-VEGF inhibition plays an important role in neovascular AMD treatment, it can be less effective in eyes with PCV. The combination of anti-VEGF injections with vPDT could be a preferable therapeutic approach for PCV, potentially resulting in more favorable treatment outcomes.20 PCV is more common in non-white individuals than in whites and patients often present at a younger age than those with AMD.47
According to guidelines from an international panel, patients with PCV should be treated with vPDT plus a VEGF inhibitor.53 The efficacy and safety of vPDT in combination with VEGF inhibitors has been demonstrated in numerous clinical trials, including EVEREST, EVEREST II, Aflibercept in Polypoidal Choroidal Vasculopathy (PLANET), and ATLANTIC (Table 2).54–58 In EVEREST, a significantly greater percentage of adults with PCV receiving vPDT alone or in combination with ranibizumab achieved complete regression of polyps compared with ranibizumab alone after 6 months (71.4% and 77.8%, vs 28.6%, respectively; P<0.005 for both comparisons).57 Similarly, a significantly greater percentage of patients receiving vPDT alone or with ranibizumab achieved complete regression of polyps at least once during the 6-month study compared with ranibizumab alone (85.7% and 83.3%, vs 42.9%; P<0.01 for both comparisons).57 In EVEREST II, achievement of complete polyp regression was significantly greater with vPDT plus ranibizumab compared with ranibizumab alone after 24 months (56.6% vs 26.7%; P<0.0001).55 In the PLANET study, the proportion of patients who achieved complete polyp regression with aflibercept plus vPDT was comparable to that with aflibercept alone at weeks 52 (44.8% vs 38.9%, respectively; P=0.3) and 96 (29.1% vs 33.1%; P=0.6).56,58 Visual acuity remained improved 1 and 2 years after combination therapy in ATLANTIC and PLANET.54,56,58
Table 2.
Summary of Select Clinical Trials of vPDT for the Treatment of Polypoidal Choroidal Vasculopathy
| Study and Design | Patient Population | Treatments | Efficacy Outcomes | Safety Outcomes |
|---|---|---|---|---|
| vPDT combination therapy | ||||
| EVEREST57 R, MC, DM, ph 4 Asia (7 sites) N=61 |
Pts with symptomatic macular PCV | Ranibizumab 0.5 mg + vPDT vPDT alone Ranibizumab 0.5 mg alone |
Ranibizumab 0.5 mg + vPDT vs vPDT alone vs ranibizumab 0.5 mg alone % pts with complete polyp regression at 6 mo: 77.8% (P=0.002 vs ranibizumab) vs 71.4% (P=0.004 vs ranibizumab) vs 28.6% |
Ranibizumab 0.5 mg + vPDT vs vPDT alone vs ranibizumab 0.5 mg alone Ocular AEs: 26.3% vs 33.3% vs 19.0% |
| EVEREST II55 R, MC, DM, ph 4 24 mo Asia (42 sites) N=322 |
Pts with symptomatic macular PCV Tx-naïve |
Ranibizumab 0.5 mg with vPDT (combination tx; n=168) vs ranibizumab 0.5 mg with sham PDT (monotherapy; n=154) IV injection on day 1 (baseline) and at 1 mo and 2 mo, followed by PRN regimen with ≥28 d between ranibizumab tx |
Combination vs monotherapy Changes in BCVA during 24 mo
% of pts with complete polypoidal lesion regression (assessed by ICGA) at 24 mo and those with absence of leakage (assessed by FA) at 24 mo
Changes in central subfield thickness from baseline to 24 mo
Number of vPDT treatments received in study eye before 24 mo
|
Combination vs monotherapy Safety and tolerability of both treatments up to 24 mo
Mortality (all unrelated to study treatment): 2/172 (1.2%) vs 1/135 (0.7%) |
| PLANET56 R, MC, DM, ph 3b/4 62 sites: Asia (n=57), Germany (n=1), Hungary (n=4) N=318 pts Age: ≥50 y |
Pts with symptomatic macular PCV | IV aflibercept 2 mg every 4 wk, at wks 0, 4, and 8 (run-in phase) At wk 12, randomization into aflibercept plus rescue PDT (combination therapy) vs aflibercept plus sham PDT (monotherapy) |
Combination vs monotherapy Change from baseline in BCVA (ETDRS letter score) for study eye at 52 wk (primary endpoint)
% of pts without moderate vision loss of ≥15 ETDRS letters from baseline to 52 wk (secondary endpoint)
|
Combination vs monotherapy Safety at 52 wk
|
| ATLANTIC54 R, DM, Sham-C, ph 4 Europe (14 sites) N=50 (50 eyes) |
Caucasian pts with tx-naïve PCV | IV aflibercept 2 mg T&E + vPDT (n=28) IV aflibercept 2 mg T&E + sham PDT (n=28) Monthly IV aflibercept at wk 0, 4, 8, then pts randomized at wk 16 |
IV aflibercept 2 mg T&E + vPDT vs sham PDT Change from baseline in BCVA to 52 wk
Complete polyp occlusion at 52 wk: 68% vs 77% (P=0.5) |
IV aflibercept 2 mg T&E + vPDT vs sham PDT Ocular AEs: 14% vs 32%
Ocular SAEs: 0% vs 0% AE leading to discontinuation: 4% vs 0% |
Abbreviations: AE, adverse event; BCVA, best-corrected visual acuity; CI, confidence interval; DM, double-masked; ETDRS, Early Treatment Diabetic Retinopathy Study; FA, fluorescein angiography; ICGA, indocyanine green angiography; IQR, interquartile range; IV, intravitreal; LS, least-squares; MC, multicenter; PCV, polypoidal choroidal vasculopathy; PDT, photodynamic therapy; Ph, phase; PLANET, Aflibercept in Polypoidal Choroidal Vasculopathy; PRN, pro re nata; pt, patient; R, randomized; SAE, serious adverse event; SD, standard deviation; SE, standard error; Sham-C, sham-controlled; T&E, treat and extend; tx, treatment; vPDT, verteporfin PDT.
Several factors have been associated with improved visual outcomes in PCV following vPDT. In the EVEREST II study, younger age and lower (better) baseline best-corrected VA (BCVA) were significantly associated with improvement in BCVA 12 months after treatment with ranibizumab 0.5 mg plus standard fluence vPDT or ranibizumab 0.5 mg alone (P<0.001 for both).59 Thus, it is apparent that vPDT, typically in combination with VEGF inhibitors, is effective for the treatment of patients with PCV.
Presumed Ocular Histoplasmosis
Presumed ocular histoplasmosis is a clinical diagnosis characterized by the absence of anterior or vitreous segment inflammation and at least 2 of the 3 following characteristics: multifocal chorioretinal scarring of the macula and mid-periphery, chorioretinal peripapillary atrophy, and CNV.60,61 Presumed ocular histoplasmosis is thought to be caused by Histoplasma capsulatum fungal infection of the chorioretinal region.60,61 While Histoplasma capsulatum can be found worldwide, reports of POHS come primarily from the United States and a handful of other countries, including Mexico, India, the United Kingdom, and the Netherlands.61 Analysis of data from >50 million outpatients in the United States included in the 2014–2016 IBM MarketScan Commercial Database and Medicare Supplemental Database found that 6678 (0.01%) had POHS, with 25% having CNV.62 Men and women are equally affected.61 The primary infection is thought to precede symptoms by years. The age at diagnosis is typically between 20 and 50 years, but it also occurs in younger and older individuals.61 Risk factors for developing POHS identified in a US study include smoking, diabetes, and residing in rural areas.63
The VEGF inhibitor aflibercept was shown to be effective and safe for patients with POHS in a masked open-label study; the 5 patients with POHS experienced improvement from baseline in VA and a decrease from baseline in central subfoveal thickness, and reported no systemic or ocular AEs at 12 months.64 Results of a Phase 1 study that included 9 patients with POHS indicated that more than half of patients overall experienced improvement in visual acuity following monthly intravitreal ranibizumab injections (66.7% had ≥15 letter improvement at both 6 and 12 months) or 3 monthly intravitreal ranibizumab injections followed by treatment as needed (64.3% and 57.1% had ≥15 letter improvement at 6 and 12 months, respectively).65 A long-term, retrospective study reported that VEGF inhibitor alone or in combination with vPDT was effective for improving VA for the first few years of a 10-year period, with no significant difference between treatment modalities.66
Verteporfin PDT has demonstrated efficacy and safety in the management of patients with POHS. In an open-label, uncontrolled, multicenter, prospective study of 26 adults with POHS with subfoveal CNV lesions treated with vPDT, more than half (56%) gained ≥7 letters of VA from baseline after 12 months, though this decreased to 10 of 22 patients (45%) at 24 months.67,68 Further, 10 of 23 patients (43%) with fluorescein leakage at baseline had no leakage after 12 months, and 17 of 20 patients (85%) had no leakage after 24 months.67,68 Seven patients (27%) experienced 17 treatment-related adverse events; the most common of these were injection-site adverse events (ie, edema, extravasation, inflammation, injection-site pain, injection-site reaction), which were seen in 4 patients (15%).67 The safety profile was comparable at 24 months.68 Although anti-VEGF monotherapy appears to be effective for POHS-related CNV, vPDT as monotherapy or in combination with anti-VEGF therapy may still be a useful option for patients who do not wish, or are unable to receive, anti-VEGF injections. Results of an open-label, randomized, Phase 2 study comparing vPDT with ranibizumab in 9 patients with POHS-related CNV showed a mean improvement of 21 and 19.6 letters of visual acuity, respectively, at 1-year follow-up.69 A chart review that included 3 eyes with POHS-related CNV receiving concurrent treatment with vPDT every 10–12 weeks and intravitreal bevacizumab every 4–6 weeks showed that this treatment regimen improved not only visual acuity, but eyes had no edema or subretinal fluid and/or leakage at least 5 months after the last treatment.70
Pathologic Myopia
Pathologic myopia, which affects 1% to 3% of individuals, is defined as “excessive axial elongation associated with myopia that leads to structural changes in the posterior segment of the eye”, which can lead to loss of BCVA.71,72 Pathologic myopia can occur in eyes with high myopia (ie, <–5.0 diopters) and is one of the most common causes of blindness, with estimates of its global prevalence suggesting that this trend is likely to continue.71–73 Myopic CNV is a complication of pathologic myopia and is visible as a small, flat lesion close to the fovea that presents with or without bleeding.72,73
In the randomized, controlled, multicenter, Phase 3 Ranibizumab and PDT (verteporfin) Evaluation in Myopic Choroidal Neovascularization (RADIANCE) study in 277 adults with visual impairment due to myopic CNV secondary to myopia, the proportion of patients who gained ≥10 and ≥15 letters or achieved 84 letters (BCVA) was more than two-fold greater in patients receiving ranibizumab 0.5 mg monotherapy compared with patients undergoing vPDT monotherapy at month 3.74 Patients assigned to the vPDT group could receive vPDT, ranibizumab, or both from month 3 onward, at the discretion of the investigator.74 Patients receiving ranibizumab continued to experience improvement in BCVA through month 12, and this was also experienced by those in the vPDT group once ranibizumab was allowed at month 3.74 Ranibizumab and vPDT were well tolerated through month 12.74
Peripapillary Choroidal Neovascularization
Peripapillary CNV is a chorioretinal condition that involves CNV within 1 disc diameter from the optic nerve.1 Considerable time may elapse between the beginning of anatomical changes and the occurrence of visual impairment.75 The epidemiology of peripapillary CNV is not well documented, but prevalence in the UK has been estimated at 0.29% in individuals ≥65 years of age in the Bridlington eye assessment project.76 The pathogenesis of peripapillary CNV involves damage of the peripapillary RPE–Bruch’s membrane–photoreceptor complex that leads to a wound healing response, which continues until vision is aberrantly affected.75 Some patients are considered to have idiopathic disease, with no known cause for their disease, while in others PCV is a cause of peripapillary CNV.75 While some cases of peripapillary CNV can be monitored without treatment, therapies such as VEGF inhibitors, focal argon laser photocoagulation, or vPDT have been used with varying degrees of success.1 Treatment with VEGF inhibitors may require multiple injections to achieve clinical outcomes, while treatment of peripapillary CNV with vPDT is not currently an indicated use.1,12
In a retrospective case series of 7 patients with peripapillary CNV receiving vPDT, all experienced improvement in VA during the follow-up (range, 3–33 months), with only 2 patients requiring ≥1 vPDT treatment.77 A study of 66 patients (67 eyes) with peripapillary CNV who were treated with the VEGF inhibitor bevacizumab found that while 88% of eyes had inactive disease during follow-up (median, 44 months; range, 7.5–131.8 months), almost half (49%) had disease inactivity after the first treatment cycle of 3 bevacizumab injections.78 Of the eyes that experienced inactive disease, recurrence occurred in 85% within a mean of 9±18 months, with 59% experiencing recurrence in less than 3 months. A shorter time to recurrence was significantly associated with being male (P<0.01) and having a younger age (median age, 79 vs 84 years; P<0.05).78
Central Serous Chorioretinopathy
Central serous chorioretinopathy is a common idiopathic chorioretinal condition, mainly affecting men between 30 and 50 years of age.79,80 Chronic CSC is characterized by persistent retinal detachment, which can result in irreversible vision loss.81,82 The incidence of CSC was found to be 18.3 per 100,000 among active-duty US armed forces service members (2001–2018), with rates increasing with age (the rate among those ≥40 years was ~30 times that of those <20 years).83 CSC is considered to be a disorder on the pachychoroid spectrum, with the pathophysiology of CSC thought to involve choroidal thickening (pachychoroid) and fluid leaking from the choriocapillaris across the RPE into the subretinal space, which, together with increased hydrostatic pressure, can cause a serous retinal detachment.14,52,81,84 Interestingly, only 14% of eyes developing chronic CSC had been previously diagnosed with acute CSC.85
Guideline recommendations regarding the treatment of chronic CSC are lacking; however, both thermal laser as well as PDT have demonstrated efficacy. While focal argon laser photocoagulation is useful for focal leakage away from the fovea, available clinical data indicate that half-dose or half-fluence vPDT can achieve high success for lesions at the central macula as well as those with more diffuse leakage.81 Verteporfin PDT is thought to exert its therapeutic effect through the closure of abnormal, leaking choroidal vessels, allowing remodeling of the choroidal vasculature.1 In a randomized, open-label study of 48 patients with chronic CSC assigned to receive PDT at varying fluence rates, 50% of the standard fluence rate (25 J/cm2) was associated with improved VA and lower rates of recurrence than 30% (15 J/cm2) or 40% (20 J/cm2) of standard fluence PDT.86 A retrospective cohort study of patients with CSC and serous retinal detachment receiving reduced fluence PDT (25 J/cm2; n=74) compared with those who did not receive treatment (n=81) demonstrated that reduced fluence PDT maintained VA in significantly more patients than no treatment after 12 months (93.6% vs 70.9%, respectively; P<0.001) and 24 months (85.7% vs 69.8%; P=0.02).87 Further, patients in the reduced fluence group had a significantly greater rate of complete resolution of the serous retinal detachment compared with the no treatment group (hazard ratio, 5.0; 95% CI, 3.2–7.9; P<0.001).87 A randomized study (n=40) comparing half-dose vPDT with half-fluence PDT showed that both methods were effective for improving VA and reducing the subretinal fluid leakage experienced by patients with chronic CSC.88 A retrospective study of patients with CSC (n=42) who received full-dose vPDT (6 mg/m2), half-dose vPDT (3 mg/m2), or half-dose/half-fluence vPDT (3 mg/m2; 25 J/cm2) showed that full-dose and half-dose vPDT significantly improved VA from baseline (pre-vPDT; P=0.02 and P=0.004, respectively).89 Subretinal fluid thickness was significantly decreased from baseline in the full-dose and half-dose vPDT groups, but not in the half-dose/half-fluence group (P=0.005, P<0.001, and P=0.08).89 In a long-term, retrospective, follow-up study (mean, 58.1 months) of patients with chronic CSC with baseline subretinal fluid (n=94 eyes; n=87 patients) who mostly received half-fluence vPDT (81% of eyes), 11% had subretinal fluid at the final follow-up visit; in this study, 84% of eyes underwent a single vPDT treatment.90 In the randomized, controlled SPECTRA study, a significantly greater percentage of patients with chronic CSC receiving half-dose vPDT achieved complete subretinal fluid resolution at 3 months compared with those receiving oral eplerenone (78% vs 17%, respectively; P<0.001).91 In this short-term trial, VA was comparable between groups at 3 months (P=0.6).91 Finally, in a randomized trial that included patients from SPECTRA, a comparable percentage of patients receiving half-dose vPDT or oral eplerenone maintained complete subretinal fluid resolution after 12 months (89.6% vs 88.1%, respectively; P=0.5), but half-dose vPDT was associated with significantly improved VA compared with oral eplerenone (P=0.03).92
Smaller studies also support the efficacy of vPDT for CSC, including a prospective, comparative study of 10 patients with chronic CSC who had undergone a single successful treatment with half-dose vPDT; vision outcomes were significantly improved from baseline with vPDT (P=0.04), and microstructural and microcirculation changes were observed 1 month after treatment.93 A second study retrospectively analyzed data from 32 patients (34 eyes) with CSC subgrouped by fluorescein angiography (point source leakage in FA [n=17] and diffuse dye oozing [n=16]) and reported that half-dose vPDT was effective for patients with CSC with CNV, with better results in the subgroup of patients with point source leakage, based on fewer half-dose PDT sessions (1.1 vs 1.9, respectively; P=0.006) and better final BCVA findings (logMAR, 0.1 vs 0.3; P=0.02) at year 3.94 A different retrospective study found that vPDT, regardless of the combination of full- or half-dose verteporfin and/or full- or half-dose fluence used, significantly improved BCVA from baseline to posttreatment in patients with CSC (n=30; >1-year follow-up period; 0.5 vs 0.3, respectively; P=0.008).95 Central foveal thickness was also significantly improved in these patients (P=0.0003).95 In this study, 9 of 15 patients with chronic CSC required additional treatment for this condition.95 Finally, a significantly greater percentage of patients with chronic CSC with persistent subretinal fluid following eplerenone treatment who crossed over to half-dose vPDT (n=37) experienced complete resolution of subretinal fluid within 3 months compared with patients who crossed over from vPDT to eplerenone (n=9; 86.5% vs 22.2%, respectively; P=0.03).96
A retrospective analysis of a single-center study of patients with CSC (85 eyes) reported that 20% of eyes with serous retinal detachment had vision loss 6 months after PDT.97 Results of this study indicate that pretreatment retinal layer thickness may be a predictor of vision loss after vPDT.97 Another study found that for patients with chronic CSC who failed to respond to ≥2 PDT sessions (median, 3 previous PDT sessions [range, 2–4]), an additional PDT session resulted in a complete response of subretinal fluid resorption in 8 of 10 patients, with 7 of 8 having an early vessel occlusion in the choriocapillaris 3 days after PDT, suggesting that patients might experience complete response if this anatomical improvement occurs following PDT.82
In addition to PDT, subthreshold laser therapy also is used for the treatment of CSC.98 The first study to compare subthreshold laser therapy with half-dose vPDT demonstrated that patients in both groups experienced improvement from baseline in fluid leakage, and the percentage of patients with a reduction in leakage activity in both groups was greater compared with the control group at 16 weeks (60% [P=0.03, vs baseline] and 66.7% [P=0.008, vs baseline] vs 33.3%, respectively).99 A randomized, double-masked study comparing half-dose vPDT with subthreshold micropulse laser therapy demonstrated that choriocapillaris flow deficit areas were significantly reduced from baseline as early as 1 month posttreatment in the half-dose vPDT group (P=0.049) compared with reductions at 6 months posttreatment in the subthreshold laser group (P=0.005).98 Further, foveal choroidal volume was significantly decreased from baseline at all time points in the half-dose vPDT group (1 month, P=0.003; 3 months, P=0.007; and 6 months, P=0.006); however, foveal choroidal volume did not change over time in the subthreshold laser group.98 In the randomized, open-label PLACE trial of patients with CSC, half-dose vPDT (n=89) was more effective than high-density subthreshold micropulse laser (n=90) for completely resolving subretinal fluid leakage 6 to 8 weeks after treatment (patients with complete absence of subretinal fluid leakage, 51.2% vs 13.8%, respectively; P<0.001) and 7 to 8 months after the initial treatment (67.2% vs 28.8%, respectively; P<0.001).100 The improvement in VA from baseline was significantly greater with half-dose vPDT compared with the micropulse laser after 6 to 8 weeks (P=0.01), but improvement, while evident in both groups 7 to 8 months after the initial treatment, did not differ significantly between groups (P=0.1).100 Finally, results of a randomized, open-label study comparing half-dose vPDT with subthreshold laser therapy showed that VA improved significantly from baseline to 10 months in both groups (P<0.001, for both groups vs baseline).101 Further, subfoveal choroidal thickness improved from baseline to 10 months with both half-dose vPDT and subthreshold laser therapy (P≤0.01, for both groups vs baseline).101
Choroidal Hemangioma
Choroidal hemangioma is a rare, benign, highly vascularized tumor localized to the choroid that is typically asymptomatic in childhood, with symptom onset occurring during adulthood.102–104 Choroidal hemangioma frequently occurs as a circumscribed lesion, but it can also present as the more diffuse form associated with Sturge-Weber syndrome.104 Symptoms can include visual impairment and metamorphopsia.102
Retrospective analysis of data from patients with choroidal hemangioma (n=458 tumors in 457 patients) compared subretinal and intraretinal fluid control and visual outcomes during the pre-PDT era (1967–2001; n=220 tumors in 220 patients) with those of the PDT era (2002–2018; n=238 tumors in 237 patients) and indicated that tumor regression was significantly greater in the PDT era than the pre-PDT era (final diameter, 5.9 vs 6.9 mm [P<0.001]; final thickness, 2.5 vs 3.0 mm [P<0.001]).105 Further, for patients with baseline VA ≥20/40, a significantly greater percentage of patients in the PDT era maintained VA ≥20/40 compared with patients in the pre-PDT era (74.7% vs 59.6%; P<0.001), while a significantly greater percentage of patients in the PDT era with baseline VA 20/50 to 20/200 achieved VA ≥20/40 compared with patients in the pre-PDT era (47.3% vs 25.4%; P<0.001).105
A single-center study of 19 patients with choroidal hemangioma showed that 14 patients (73.7%) experienced improved VA with vPDT treatment, while 4 patients (21.1%) remained stable; only 1 patient (5.2%) had a slight decrease in VA over a mean follow-up of 10.6 months (range, 2–24 months).106 Improvement from baseline of ≥2 lines of VA was observed in 8 patients (42.1%), which was significantly correlated with shorter duration of symptoms (P=0.007).106
The long-term efficacy of vPDT in patients with choroidal hemangioma was shown in a single-center retrospective review of 10 patients (10 eyes) with a mean of 4.5 years of follow-up.104 In this study, vPDT improved BCVA from baseline to last follow-up (mean [SD] logMAR, 0.7 [0.5] vs 0.6 [0.6]), with VA improving in 5 patients (50%) and remaining stable in 4 patients (40%).104 A second retrospective analysis examined long-term outcomes (mean follow-up, 30.8 months) of vPDT in 15 patients with ocular tumors, including 10 with choroidal hemangioma.107 Among patients with choroidal hemangioma, VA improved in 2 patients (20%) and remained stable in 5 patients (50%); tumor size decreased in 5 patients (50%) and remained stable in 3 patients (30%).107
In a retrospective review of patients with choroidal hemangioma (n=79), predictors of VA ≥20/40 compared with ≤20/50 following vPDT included baseline VA ≥20/40, smaller tumor size (ie, mean tumor basal diameter, 5.4 vs 6.2 mm [P=0.03]; mean tumor thickness, 2.9 vs 3.2 mm [P=0.01]), less foveolar cystoid macular edema (ie, abnormal fluid buildup in the retina; 30% vs 70% [P=0.001]), and being treatment-naïve (67% vs 33% [P=0.04]).108
In addition, half-dose vPDT was associated with good anatomical and functional outcomes in a case series that included 4 patients with choroidal hemangioma. Patients experienced tumor shrinkage and fluid resolution after half-dose vPDT, while experiencing no AEs.102
Rare Diseases
Additional uses for vPDT have been reported in rare ophthalmic diseases, including Coats disease, idiopathic macular telangiectasia, and in the management of CNV linked with Stargardt disease and retinitis pigmentosa.109–111 A case report describing a patient with bilateral idiopathic macular telangiectasia showed that vPDT was effective for improving VA over 6 months after treatment.110 Finally, a case report described the efficacy of vPDT for the treatment of a patient with Stargardt disease and two different patients with retinitis pigmentosa.111 The patient with Stargardt disease experienced improvement in VA and foveal hemorrhage 1 month after treatment, and had complete resolution of VA and hemorrhage 3 months after a second round of vPDT when foveal hemorrhage recurred.111 The patients with retinitis pigmentosa also had improved VA 3 months after vPDT.111
Discussion and Conclusions
The prevalence of ophthalmologic conditions that impair vision, including neovascular AMD, is estimated to grow globally over time. Thus, enlisting the use of effective and safe treatments for these conditions is crucial. Anti-VEGF treatments have become first-line therapy for many patients with choroidal neovascular membranes. For the number of patients who require ongoing treatments, and particularly those with PCV, auxiliary treatments might be needed. One such treatment is vPDT, either as monotherapy or in combination with a VEGF inhibitor, which has demonstrated efficacy and safety for patients with CNV due to AMD, PCV, CSC, choroidal hemangioma, peripapillary CNV, POHS, and pathologic myopia. In patients with CNV due to AMD, VEGF inhibitor monotherapy was shown to have greater efficacy than vPDT monotherapy (eg, ANCHOR study). Combination therapy improved visual outcomes in patients with PCV, which is particularly important in these patients as VEGF inhibitors are less effective than in other conditions. Thus, guidelines currently support combination therapy for this patient population. Photodynamic therapy has a clear role in the treatment of CSC, and adjusting protocols (ie, decreasing vPDT dose and/or fluence) in this patient population to diminish adverse effects was effective; however, additional research is warranted to elucidate further optimal dosing and fluence of vPDT in CSC and possibly provide a treatment consensus. Data are limited in patients with choroidal hemangioma, but a comparison of data related to outcomes in the pre-PDT era with those from the PDT era showed that improved vision outcomes were associated with the introduction of vPDT. While anti-VEGF monotherapy is typically effective for other causes of CNV, smaller studies and retrospective analyses indicate that vPDT has favorable efficacy in peripapillary CNV, as well as for eyes with CNV due to POHS and pathologic myopia, and in rare ophthalmic diseases. However, randomized, controlled clinical trials with larger patient populations could further establish and support vPDT treatment of these conditions, either in combination with VEGF inhibitors or independently.
Acknowledgments
Editorial and medical writing assistance was provided under the direction of the authors by Sophie Bolick, PhD, and Pratibha Hebbar, PhD, Synchrony Medical Communications, LLC, West Chester, PA, and funded by Bausch + Lomb.
Disclosure
SJG reports being a consultant for Allergan, Apellis, Bausch + Lomb, American Academy of Ophthalmology, Boehringer Ingelheim, Johnson and Johnson, Merck Manual, Regeneron, Roche/Genentech, and West Pharmaceutical Services; and receiving research grants from Alcon, American Academy of Ophthalmology, Apellis, Boehringer Ingelheim, Kodiak Biosciences, NGM Bio, and Regeneron. In addition, he reports advisory board for and personal fees from Coherus; lecture fees from Canadian Ophthalmological Society, Cole Eye Summit, Physicians’ Education Resource, and Retina Fellows Forum, outside the submitted work. MH reports being a consultant, investigator, or serving on the advisory board for Apellis, ALEXION, Allergan/AbbVie, Bausch + Lomb, Emagix, Topcon, and Protagonist; and is a National Institutes of Health grant recipient. In addition, she has served as a panelist for IvericBio, for which she received compensation.
References
- 1.Newman DK. Photodynamic therapy: current role in the treatment of chorioretinal conditions. Eye (Lond). 2016;30(2):202–210. doi: 10.1038/eye.2015.251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.van Dijk EHC, van Rijssen TJ, Subhi Y, Boon CJF. Photodynamic therapy for chorioretinal diseases: a practical approach. Ophthalmol Ther. 2020;9(2):329–342. doi: 10.1007/s40123-020-00250-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chowdhary RK, Sharif I, Chansarkar N, et al. Correlation of photosensitizer delivery to lipoproteins and efficacy in tumor and arthritis mouse models; comparison of lipid-based and Pluronic P123 formulations. J Pharm Pharm Sci. 2003;6(2):198–204. [PubMed] [Google Scholar]
- 4.Weishaupt KR, Gomer CJ, Dougherty TJ. Identification of singlet oxygen as the cytotoxic agent in photoinactivation of a murine tumor. Cancer Res. 1976;36(7 pt 1):2326–2329. [PubMed] [Google Scholar]
- 5.Michels S, Schmidt-Erfurth U. Sequence of early vascular events after photodynamic therapy. Invest Ophthalmol Vis Sci. 2003;44(5):2147–2154. doi: 10.1167/iovs.02-0604 [DOI] [PubMed] [Google Scholar]
- 6.Schmidt-Erfurth U, Michels S, Barbazetto I, Laqua H. Photodynamic effects on choroidal neovascularization and physiological choroid. Invest Ophthalmol Vis Sci. 2002;43(3):830–841. [PubMed] [Google Scholar]
- 7.Debefve E, Pegaz B, van den Bergh H, Wagnieres G, Lange N, Ballini JP. Video monitoring of neovessel occlusion induced by photodynamic therapy with verteporfin (Visudyne), in the CAM model. Angiogenesis. 2008;11(3):235–243. doi: 10.1007/s10456-008-9106-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kramer M, Miller JW, Michaud N, et al. Liposomal benzoporphyrin derivative verteporfin photodynamic therapy. Selective treatment of choroidal neovascularization in monkeys. Ophthalmology. 1996;103(3):427–438. doi: 10.1016/S0161-6420(96)30675-1 [DOI] [PubMed] [Google Scholar]
- 9.Miller JW, Walsh AW, Kramer M, et al. Photodynamic therapy of experimental choroidal neovascularization using lipoprotein-delivered benzoporphyrin. Arch Ophthalmol. 1995;113(6):810–818. doi: 10.1001/archopht.1995.01100060136048 [DOI] [PubMed] [Google Scholar]
- 10.Schmidt-Erfurth U, Hasan T. Mechanisms of action of photodynamic therapy with verteporfin for the treatment of age-related macular degeneration. Surv Ophthalmol. 2000;45(3):195–214. doi: 10.1016/S0039-6257(00)00158-2 [DOI] [PubMed] [Google Scholar]
- 11.Husain D, Miller JW, Michaud N, Connolly E, Flotte TJ, Gragoudas ES. Intravenous infusion of liposomal benzoporphyrin derivative for photodynamic therapy of experimental choroidal neovascularization. Arch Ophthalmol. 1996;114(8):978–985. doi: 10.1001/archopht.1996.01100140186012 [DOI] [PubMed] [Google Scholar]
- 12.Visudyne (verteporfin for injection), for intravenous use [package insert]. Bridgewater, NJ: Bausch & Lomb Americas Inc.; February 2023. [Google Scholar]
- 13.Desmettre T, Mainster MA, Ledesma-Gil G. Half-fluence, half-dose photodynamic therapy: less direct damage but more inflammation? Pharmaceuticals (Basel). 2023;16(4):494. doi: 10.3390/ph16040494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Iacono P, Da Pozzo S, Varano M, Parravano M. Photodynamic therapy with verteporfin for chronic central serous chorioretinopathy: a review of data and efficacy. Pharmaceuticals (Basel). 2020;13(11):349. doi: 10.3390/ph13110349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ledesma-Gil G, Desmettre T, Mainster MA. Bacillary layer detachment after photodynamic therapy for central serous chorioretinopathy. Retin Cases Brief Rep. 2023;17(3):239–241. doi: 10.1097/ICB.0000000000001190 [DOI] [PubMed] [Google Scholar]
- 16.Gao Y, Yu T, Zhang Y, Dang G. Anti-VEGF monotherapy versus photodynamic therapy and anti-VEGF combination treatment for neovascular age-related macular degeneration: a meta-analysis. Invest Ophthalmol Vis Sci. 2018;59(10):4307–4317. doi: 10.1167/iovs.17-23747 [DOI] [PubMed] [Google Scholar]
- 17.Saint-Geniez M, Maldonado AE, D’Amore PA. VEGF expression and receptor activation in the choroid during development and in the adult. Invest Ophthalmol Vis Sci. 2006;47(7):3135–3142. doi: 10.1167/iovs.05-1229 [DOI] [PubMed] [Google Scholar]
- 18.Moon BH, Kim Y, Kim SY. Twenty years of anti-vascular endothelial growth factor therapeutics in neovascular age-related macular degeneration treatment. Int J Mol Sci. 2023;24(16):13004. doi: 10.3390/ijms241613004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Helotera H, Kaarniranta K. A linkage between angiogenesis and inflammation in neovascular age-related macular degeneration. Cells. 2022;11(21):3453. doi: 10.3390/cells11213453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Avaylon J, Gallemore RP. Verteporfin photodynamic therapy for the treatment of choroidal conditions—an overview. US Ophthalmic Rev. 2019;12(2):69–78. doi: 10.17925/USOR.2019.12.2.69. [DOI] [Google Scholar]
- 21.Guymer RH, Campbell TG. Age-related macular degeneration. Lancet. 2023;401(10386):1459–1472. doi: 10.1016/S0140-6736(22)02609-5 [DOI] [PubMed] [Google Scholar]
- 22.Apte RS. Age-related macular degeneration. N Engl J Med. 2021;385(6):539–547. doi: 10.1056/NEJMcp2102061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ferris FL 3rd, Fine SL, Hyman L. Age-related macular degeneration and blindness due to neovascular maculopathy. Arch Ophthalmol. 1984;102(11):1640–1642. doi: 10.1001/archopht.1984.01040031330019 [DOI] [PubMed] [Google Scholar]
- 24.Rastoin O, Pages G, Dufies M. Experimental models in neovascular age related macular degeneration. Int J Mol Sci. 2020;21(13):4627. doi: 10.3390/ijms21134627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Song D, Liu P, Shang K, Ma YB. Application and mechanism of anti-VEGF drugs in age-related macular degeneration. Front Bioeng Biotechnol. 2022;10:943915. doi: 10.3389/fbioe.2022.943915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schmidt-Erfurth U, Chong V, Loewenstein A, et al. Guidelines for the management of neovascular age-related macular degeneration by the European Society of Retina Specialists (EURETINA). Br J Ophthalmol. 2014;98(9):1144–1167. doi: 10.1136/bjophthalmol-2014-305702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Treatment of Age-Related Macular Degeneration With Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin. Arch Ophthalmol. 1999;117(10):1329–1345. doi: 10.1001/archopht.117.10.1329 [DOI] [PubMed] [Google Scholar]
- 28.Soubrane G, Bressler NM. Treatment of subfoveal choroidal neovascularisation in age related macular degeneration: focus on clinical application of verteporfin photodynamic therapy. Br J Ophthalmol. 2001;85(4):483–495. doi: 10.1136/bjo.85.4.483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Verteporfin in Photodynamic Therapy Study Group. Verteporfin therapy of subfoveal choroidal neovascularization in age-related macular degeneration: two-year results of a randomized clinical trial including lesions with occult with no classic choroidal neovascularization--verteporfin in photodynamic therapy report 2. Am J Ophthalmol. 2001;131(5):541–560. doi: 10.1016/s0002-9394(01)00967-9 [DOI] [PubMed] [Google Scholar]
- 30.Bressler NM; Treatment of Age-Related Macular Degeneration With Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin. Arch Ophthalmol. 2001;119(2):198–207. [PubMed] [Google Scholar]
- 31.Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1419–1431. doi: 10.1056/NEJMoa054481 [DOI] [PubMed] [Google Scholar]
- 32.Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1432–1444. doi: 10.1056/NEJMoa062655 [DOI] [PubMed] [Google Scholar]
- 33.Brown DM, Michels M, Kaiser PK, Heier JS, Sy JP, Ianchulev T. Ranibizumab versus verteporfin photodynamic therapy for neovascular age-related macular degeneration: two-year results of the ANCHOR study. Ophthalmology. 2009;116(1):57–65.e55. doi: 10.1016/j.ophtha.2008.10.018 [DOI] [PubMed] [Google Scholar]
- 34.Heier JS, Boyer DS, Ciulla TA, et al. Ranibizumab combined with verteporfin photodynamic therapy in neovascular age-related macular degeneration: year 1 results of the FOCUS Study. Arch Ophthalmol. 2006;124(11):1532–1542. doi: 10.1001/archopht.124.11.1532 [DOI] [PubMed] [Google Scholar]
- 35.Antoszyk AN, Tuomi L, Chung CY, Singh A, Group FS. Ranibizumab combined with verteporfin photodynamic therapy in neovascular age-related macular degeneration (FOCUS): year 2 results. Am J Ophthalmol. 2008;145(5):862–874. doi: 10.1016/j.ajo.2007.12.029 [DOI] [PubMed] [Google Scholar]
- 36.Kaiser PK, Boyer DS, Cruess AF, et al. Verteporfin plus ranibizumab for choroidal neovascularization in age-related macular degeneration: twelve-month results of the DENALI study. Ophthalmology. 2012;119(5):1001–1010. doi: 10.1016/j.ophtha.2012.02.003 [DOI] [PubMed] [Google Scholar]
- 37.Larsen M, Schmidt-Erfurth U, Lanzetta P, et al. Verteporfin plus ranibizumab for choroidal neovascularization in age-related macular degeneration: twelve-month MONT BLANC study results. Ophthalmology. 2012;119(5):992–1000. doi: 10.1016/j.ophtha.2012.02.002 [DOI] [PubMed] [Google Scholar]
- 38.Gallemore RP, Wallsh J, Hudson HL, Ho AC, Chace R, Pearlman J. Combination verteporfin photodynamic therapy ranibizumab-dexamethasone in choroidal neovascularization due to age-related macular degeneration: results of a phase II randomized trial. Clin Ophthalmol. 2017;11:223–231. doi: 10.2147/OPTH.S119510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Arepalli S, Kaiser PK. Pipeline therapies for neovascular age related macular degeneration. Int J Retina Vitreous. 2021;7(1):55. doi: 10.1186/s40942-021-00325-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Heier JS, Brown DM, Chong V, et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012;119(12):2537–2548. doi: 10.1016/j.ophtha.2012.09.006 [DOI] [PubMed] [Google Scholar]
- 41.Takahashi K, Iida T, Ishida S, et al. Effectiveness of current treatments for wet age-related macular degeneration in Japan: a systematic review and pooled data analysis. Clin Ophthalmol. 2022;16:531–540. doi: 10.2147/OPTH.S345403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chandra S, McKibbin M, Mahmood S, et al. The Royal College of Ophthalmologists Commissioning guidelines on age macular degeneration: executive summary. Eye (Lond). 2022;36(11):2078–2083. doi: 10.1038/s41433-022-02095-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.National Institute for Clinical Excellence. Age-related macular degeneration: NICE guideline NG82. 2018; Available from: https://www.nice.org.uk/guidance/ng82. Accessed June 21, 2023.
- 44.Vabysmo (faricimab-svoa) injection, for intravitreal use [package insert]. South San Francisco, CA: Genentech, Inc.; October 2023. [Google Scholar]
- 45.Eylea (aflibercept) injection, for intravitreal use [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc.; August 2023. [Google Scholar]
- 46.Eylea HD (aflibercept) injection, for intravitreal use [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc.; August 2023. [Google Scholar]
- 47.Anantharaman G, Sheth J, Bhende M, et al. Polypoidal choroidal vasculopathy: pearls in diagnosis and management. Indian J Ophthalmol. 2018;66(7):896–908. doi: 10.4103/ijo.IJO_1136_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ruamviboonsuk P, Lai TYY, Chen SJ, et al. Polypoidal choroidal vasculopathy: updates on risk factors, diagnosis, and treatments. Asia Pac J Ophthalmol (Phila). 2023;12(2):184–195. doi: 10.1097/APO.0000000000000573 [DOI] [PubMed] [Google Scholar]
- 49.Cheung CMG, Lai TYY, Teo K, et al. Polypoidal choroidal vasculopathy: consensus nomenclature and non-indocyanine green angiograph diagnostic criteria from the Asia-Pacific Ocular Imaging Society PCV Workgroup. Ophthalmology. 2021;128(3):443–452. doi: 10.1016/j.ophtha.2020.08.006 [DOI] [PubMed] [Google Scholar]
- 50.Cheung CMG, Lai TYY, Ruamviboonsuk P, et al. Polypoidal choroidal vasculopathy: definition, pathogenesis, diagnosis, and management. Ophthalmology. 2018;125(5):708–724. doi: 10.1016/j.ophtha.2017.11.019 [DOI] [PubMed] [Google Scholar]
- 51.Yamashiro K, Yanagi Y, Koizumi H, et al. Relationship between pachychoroid and polypoidal choroidal vasculopathy. J Clin Med. 2022;11(15):4614. doi: 10.3390/jcm11154614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cheung CMG, Lee WK, Koizumi H, Dansingani K, Lai TYY, Freund KB. Pachychoroid disease. Eye (Lond). 2019;33(1):14–33. doi: 10.1038/s41433-018-0158-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Koh AH, Chen LJ, Chen SJ, et al. Polypoidal choroidal vasculopathy: evidence-based guidelines for clinical diagnosis and treatment. Retina. 2013;33(4):686–716. doi: 10.1097/IAE.0b013e3182852446 [DOI] [PubMed] [Google Scholar]
- 54.Silva R, Arias L, Nunes S, et al. Efficacy and safety of intravitreal aflibercept treat and extend for polypoidal choroidal vasculopathy in the ATLANTIC Study: a randomized clinical trial. Ophthalmologica. 2022;245(1):80–90. doi: 10.1159/000518235 [DOI] [PubMed] [Google Scholar]
- 55.Lim TH, Lai TYY, Takahashi K, et al. Comparison of ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: the EVEREST II randomized clinical trial. JAMA Ophthalmol. 2020;138(9):935–942. doi: 10.1001/jamaophthalmol.2020.2443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee WK, Iida T, Ogura Y, et al. Efficacy and safety of intravitreal aflibercept for polypoidal choroidal vasculopathy in the PLANET study: a randomized clinical trial. JAMA Ophthalmol. 2018;136(7):786–793. doi: 10.1001/jamaophthalmol.2018.1804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Koh A, Lee WK, Chen LJ, et al. EVEREST study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina. 2012;32(8):1453–1464. doi: 10.1097/IAE.0b013e31824f91e8 [DOI] [PubMed] [Google Scholar]
- 58.Wong TY, Ogura Y, Lee WK, et al. Efficacy and safety of intravitreal aflibercept for polypoidal choroidal vasculopathy: two-year results of the Aflibercept in Polypoidal Choroidal Vasculopathy Study. Am J Ophthalmol. 2019;204:80–89. doi: 10.1016/j.ajo.2019.02.027 [DOI] [PubMed] [Google Scholar]
- 59.Cheung CMG, Tan CS, Patalauskaite R, Margaron P, Lai TYY. Ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: predictors of visual and anatomical response in the EVEREST II Study. Retina. 2021;41(2):387–392. doi: 10.1097/IAE.0000000000002902 [DOI] [PubMed] [Google Scholar]
- 60.Xu TT, Reynolds MM, Hodge DO, Smith WM. Epidemiology and clinical characteristics of presumed ocular histoplasmosis in Olmsted County, Minnesota. Ocul Immunol Inflamm. 2022;30(5):1039–1043. doi: 10.1080/09273948.2020.1859549 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Diaz RI, Sigler EJ, Rafieetary MR, Calzada JI. Ocular histoplasmosis syndrome. Surv Ophthalmol. 2015;60(4):279–295. doi: 10.1016/j.survophthal.2015.02.005 [DOI] [PubMed] [Google Scholar]
- 62.Benedict K, Shantha JG, Yeh S, Beer KD, Jackson BR. Presumed ocular histoplasmosis syndrome in a commercially insured population, United States. PLoS One. 2020;15(3):e0230305. doi: 10.1371/journal.pone.0230305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Richey BF, Obrock RS, Gee ZM, Lu DY, Jacobsen GJ, Richards SC. Smoking, rural residence and diabetes as risk factors for presumed ocular histoplasmosis syndrome. Retina. 2022;42(2):369–374. doi: 10.1097/IAE.0000000000003322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Walia HS, Shah GK, Blinder KJ. Treatment of CNV secondary to presumed ocular histoplasmosis with intravitreal aflibercept 2.0 mg injection. Can J Ophthalmol. 2016;51(2):91–96. doi: 10.1016/j.jcjo.2015.11.007 [DOI] [PubMed] [Google Scholar]
- 65.Heier JS, Brown D, Ciulla T, et al. Ranibizumab for choroidal neovascularization secondary to causes other than age-related macular degeneration: a phase I clinical trial. Ophthalmology. 2011;118(1):111–118. doi: 10.1016/j.ophtha.2010.04.016 [DOI] [PubMed] [Google Scholar]
- 66.Dudenhoefer NE, Noble CW, Petersen MR, et al. Intravitreal antivascular endothelial growth factor for the treatment of choroidal neovascularization secondary to ocular histoplasmosis: ten-year follow-up. Retina. 2022;42(8):1568–1573. doi: 10.1097/IAE.0000000000003488 [DOI] [PubMed] [Google Scholar]
- 67.Saperstein DA, Rosenfeld PJ, Bressler NM, et al. Photodynamic therapy of subfoveal choroidal neovascularization with verteporfin in the ocular histoplasmosis syndrome: one-year results of an uncontrolled, prospective case series. Ophthalmology. 2002;109(8):1499–1505. doi: 10.1016/S0161-6420(02)01103-X [DOI] [PubMed] [Google Scholar]
- 68.Rosenfeld PJ, Saperstein DA, Bressler NM, et al. Photodynamic therapy with verteporfin in ocular histoplasmosis: uncontrolled, open-label 2-year study. Ophthalmology. 2004;111(9):1725–1733. doi: 10.1016/j.ophtha.2004.02.014 [DOI] [PubMed] [Google Scholar]
- 69.Ramaiya K, Blinder KJ, Ciulla T, Cooper B, Shah GK. Ranibizumab versus photodynamic therapy for presumed ocular histoplasmosis syndrome. Ophthalmic Surg Lasers Imaging Retina. 2013;44(1):17–21. doi: 10.3928/23258160-20121221-07 [DOI] [PubMed] [Google Scholar]
- 70.Han DP, McAllister JT, Weinberg DV, Kim JE, Wirostko WJ. Combined intravitreal anti-VEGF and verteporfin photodynamic therapy for juxtafoveal and extrafoveal choroidal neovascularization as an alternative to laser photocoagulation. Eye (Lond). 2010;24(4):713–716. doi: 10.1038/eye.2009.122 [DOI] [PubMed] [Google Scholar]
- 71.Nemeth J, Tapaszto B, Aclimandos WA, et al. Update and guidance on management of myopia. European Society of Ophthalmology in cooperation with International Myopia Institute. Eur J Ophthalmol. 2021;31(3):853–883. doi: 10.1177/1120672121998960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ueta T, Makino S, Yamamoto Y, Fukushima H, Yashiro S, Nagahara M. Pathologic myopia: an overview of the current understanding and interventions. Glob Health Med. 2020;2(3):151–155. doi: 10.35772/ghm.2020.01007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ohno-Matsui K, Lai TY, Lai CC, Cheung CM. Updates of pathologic myopia. Prog Retin Eye Res. 2016;52:156–187. doi: 10.1016/j.preteyeres.2015.12.001 [DOI] [PubMed] [Google Scholar]
- 74.Wolf S, Balciuniene VJ, Laganovska G, et al. RADIANCE: a randomized controlled study of ranibizumab in patients with choroidal neovascularization secondary to pathologic myopia. Ophthalmology. 2014;121(3):682–692.e682. doi: 10.1016/j.ophtha.2013.10.023 [DOI] [PubMed] [Google Scholar]
- 75.Jutley G, Jutley G, Tah V, Lindfield D, Menon G. Treating peripapillary choroidal neovascular membranes: a review of the evidence. Eye (Lond). 2011;25(6):675–681. doi: 10.1038/eye.2011.24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wilde C, Poostchi A, Mehta RL, et al. Prevalence of peripapillary choroidal neovascular membranes (PPCNV) in an elderly UK population-the Bridlington eye assessment project (BEAP): a cross-sectional study (2002-2006). Eye (Lond). 2019;33(3):451–458. doi: 10.1038/s41433-018-0232-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bernstein PS, Horn RS. Verteporfin photodynamic therapy involving the optic nerve for peripapillary choroidal neovascularization. Retina. 2008;28(1):81–84. doi: 10.1097/IAE.0b013e31815e9351 [DOI] [PubMed] [Google Scholar]
- 78.Stanescu N, Friehmann A, Nemet A, et al. Long-term outcomes of anti-vascular endothelial growth factor treatment in peripapillary choroidal neovascularisation due to age-related macular degeneration. Eye (Lond). 2023;37(6):1202–1206. doi: 10.1038/s41433-022-02089-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Liegl R, Ulbig MW. Central serous chorioretinopathy. Ophthalmologica. 2014;232(2):65–76. doi: 10.1159/000360014 [DOI] [PubMed] [Google Scholar]
- 80.Zhang X, Lim CZF, Chhablani J, Wong YM. Central serous chorioretinopathy: updates in the pathogenesis, diagnosis and therapeutic strategies. Eye Vis (Lond). 2023;10(1):33. doi: 10.1186/s40662-023-00349-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.van Rijssen TJ, van Dijk EHC, Yzer S, et al. Central serous chorioretinopathy: towards an evidence-based treatment guideline. Prog Retin Eye Res. 2019;73:100770. doi: 10.1016/j.preteyeres.2019.07.003 [DOI] [PubMed] [Google Scholar]
- 82.Moreno-Morillo FJ, Fernández-Vigo JI, Burgos-Blasco B, Moriche-Carretero M, López-Guajardo L, Donate-López J. Utility of an additional photodynamic therapy session after multiple failed PDT treatments in chronic central serous chorioretinopathy. Photodiagnosis Photodyn Ther. 2022;39:102953. doi: 10.1016/j.pdpdt.2022.102953 [DOI] [PubMed] [Google Scholar]
- 83.Reynolds ME, Karesh JW, Oh GT, Stahlman S. Incident and recurrent cases of central serous chorioretinopathy, active component, U.S. Armed Forces, 2001-2018. MSMR. 2019;26(9):31–34. [PubMed] [Google Scholar]
- 84.Goté JT, Singh SR, Chhablani J. Comparing treatment outcomes in randomized controlled trials of central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2023;261(8):2135–2168. doi: 10.1007/s00417-023-05996-4 [DOI] [PubMed] [Google Scholar]
- 85.Mohabati D, van Rijssen TJ, van Dijk EH, et al. Clinical characteristics and long-term visual outcome of severe phenotypes of chronic central serous chorioretinopathy. Clin Ophthalmol. 2018;12:1061–1070. doi: 10.2147/OPTH.S160956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Park DG, Jeong S, Noh D, Sagong M. Optimal fluence rate of photodynamic therapy for chronic central serous chorioretinopathy. Br J Ophthalmol. 2021;105(6):844–849. doi: 10.1136/bjophthalmol-2020-316837 [DOI] [PubMed] [Google Scholar]
- 87.Aisu N, Miyake M, Hosoda Y, et al. Effectiveness of reduced-fluence photodynamic therapy for chronic central serous chorioretinopathy: a propensity score analysis. Ophthalmol Sci. 2022;2(2):100152. doi: 10.1016/j.xops.2022.100152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Cheng CK, Chang CK, Peng CH. Comparison of photodynamic therapy using half-dose of verteporfin or half-fluence of laser light for the treatment of chronic central serous chorioretinopathy. Retina. 2017;37(2):325–333. doi: 10.1097/IAE.0000000000001138 [DOI] [PubMed] [Google Scholar]
- 89.Park W, Kim M, Kim RY, Park YH. Comparing effects of photodynamic therapy in central serous chorioretinopathy: full-dose versus half-dose versus half-dose-half-fluence. Graefes Arch Clin Exp Ophthalmol. 2019;257(10):2155–2161. doi: 10.1007/s00417-019-04426-8 [DOI] [PubMed] [Google Scholar]
- 90.Park YJ, Kim YK, Park KH, Woo SJ. Long-term efficacy and safety of photodynamic therapy in patients with chronic central serous chorioretinopathy. Ophthalmic Surg Lasers Imaging Retina. 2019;50(12):760–770. doi: 10.3928/23258160-20191119-03 [DOI] [PubMed] [Google Scholar]
- 91.van Rijssen TJ, van Dijk EHC, Tsonaka R, et al. Half-dose photodynamic therapy versus eplerenone in chronic central serous chorioretinopathy (SPECTRA): a randomized controlled trial. Am J Ophthalmol. 2022;233:101–110. doi: 10.1016/j.ajo.2021.06.020 [DOI] [PubMed] [Google Scholar]
- 92.Feenstra HMA, van Dijk EHC, van Rijssen TJ, et al. Long-term follow-up of chronic central serous chorioretinopathy patients after primary treatment of oral eplerenone or half-dose photodynamic therapy and crossover treatment: SPECTRA trial report No. 3. Graefes Arch Clin Exp Ophthalmol. 2023;261(3):659–668. doi: 10.1007/s00417-022-05836-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Christou EE, Katsanos A, Georgalas I, Kozobolis V, Kalogeropoulos C, Stefaniotou M. The choroid after half-dose photodynamic therapy in chronic central serous chorioretinopathy. Acta Medica (Hradec Kralove). 2022;65(3):89–98. doi: 10.14712/18059694.2022.24 [DOI] [PubMed] [Google Scholar]
- 94.Hu YC, Chen YL, Chen YC, Chen SN. 3-year follow-up of half-dose verteporfin photodynamic therapy for central serous chorioretinopathy with OCT-angiography detected choroidal neovascularization. Sci Rep. 2021;11(1):13286. doi: 10.1038/s41598-021-92693-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hurley DJ, Gallagher D, Petronzi V, O’Rourke M, Kinsella F, Townley D. Examining the efficacy of verteporfin photo-dynamic therapy (PDT) at different dose & fluence levels. Photodiagnosis Photodyn Ther. 2023;44:103848. doi: 10.1016/j.pdpdt.2023.103848 [DOI] [PubMed] [Google Scholar]
- 96.Feenstra HMA, van Dijk EHC, van Rijssen TJ, et al. Crossover to half-dose photodynamic therapy or eplerenone in chronic central serous chorioretinopathy patients. Ophthalmol Retina. 2022;6(10):930–938. doi: 10.1016/j.oret.2022.04.014 [DOI] [PubMed] [Google Scholar]
- 97.Funatsu R, Terasaki H, Sonoda S, Shiihara H, Mihara N, Sakamoto T. Characteristics related to visual acuity loss after successful photodynamic therapy for eyes with central serous chorioretinopathy. Am J Ophthalmol. 2023;256:P164–P174. [DOI] [PubMed] [Google Scholar]
- 98.Ho M, Lai FHP, Ng DSC, et al. Analysis of choriocapillaris perfusion and choroidal layer changes in patients with chronic central serous chorioretinopathy randomised to micropulse laser or photodynamic therapy. Br J Ophthalmol. 2021;105(4):555–560. doi: 10.1136/bjophthalmol-2020-316076 [DOI] [PubMed] [Google Scholar]
- 99.Kretz FTA, Beger I, Koch F, Nowomiejska K, Auffarth GU, Koss MJ. Randomized clinical trial to compare micropulse photocoagulation versus half-dose verteporfin photodynamic therapy in the treatment of central serous chorioretinopathy. Ophthalmic Surg Lasers Imaging Retina. 2015;46(8):837–843. doi: 10.3928/23258160-20150909-08 [DOI] [PubMed] [Google Scholar]
- 100.van Dijk EHC, Fauser S, Breukink MB, et al. Half-dose photodynamic therapy versus high-density subthreshold micropulse laser treatment in patients with chronic central serous chorioretinopathy: the PLACE Trial. Ophthalmology. 2018;125(10):1547–1555. doi: 10.1016/j.ophtha.2018.04.021 [DOI] [PubMed] [Google Scholar]
- 101.Russo A, Turano R, Morescalchi F, et al. Comparison of half-dose photodynamic therapy and 689 nm laser treatment in eyes with chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2017;255(6):1141–1148. doi: 10.1007/s00417-017-3626-9 [DOI] [PubMed] [Google Scholar]
- 102.Pérez-González D, Goldstein M, Iglicki M, Zur D. Half-dose photodynamic therapy as a novel treatment protocol for circumscribed choroidal hemangioma. Life (Basel). 2022;12(11):1748. doi: 10.3390/life12111748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kumar A, Shankar S, Singh A, Mishra S, Kumar P, Arora A. Photodynamic therapy in the treatment of circumscribed choroidal hemangioma: current perspectives. Photodiagnosis Photodyn Ther. 2022;39:103000. doi: 10.1016/j.pdpdt.2022.103000 [DOI] [PubMed] [Google Scholar]
- 104.Murro V, Mucciolo DP, Giorgio D, et al. Long-term follow-up and “double layer sign” in patients affected by circumscribed choroidal hemangioma. Photodiagnosis Photodyn Ther. 2020;31:101960. doi: 10.1016/j.pdpdt.2020.101960 [DOI] [PubMed] [Google Scholar]
- 105.Shields CL, Dalvin LA, Lim LS, et al. Circumscribed choroidal hemangioma: visual outcome in the pre-photodynamic therapy era versus photodynamic therapy era in 458 cases. Ophthalmol Retina. 2020;4(1):100–110. doi: 10.1016/j.oret.2019.08.004 [DOI] [PubMed] [Google Scholar]
- 106.Jurklies B, Anastassiou G, Ortmans S, et al. Photodynamic therapy using verteporfin in circumscribed choroidal haemangioma. Br J Ophthalmol. 2003;87(1):84–89. doi: 10.1136/bjo.87.1.84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Karaca BO, Türkoğlu EB, Doğan ME. Long term results of photodynamic therapy in intraocular tumors. Photodiagnosis Photodyn Ther. 2023;42:103564. doi: 10.1016/j.pdpdt.2023.103564 [DOI] [PubMed] [Google Scholar]
- 108.Di Nicola M, Williams BK, Srinivasan A, et al. Photodynamic therapy for circumscribed choroidal hemangioma in 79 consecutive patients: comparative analysis of factors predictive of visual outcome. Ophthalmol Retina. 2020;4(10):1024–1033. doi: 10.1016/j.oret.2020.04.018 [DOI] [PubMed] [Google Scholar]
- 109.Namba M, Shiode Y, Morizane Y, et al. Successful resolution of Coats disease by photodynamic therapy: a case report. BMC Ophthalmol. 2018;18(1):264. doi: 10.1186/s12886-018-0930-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Dave V, Chhablani J, Narayanan R. Different treatment modalities for choroidal neovascularization in two eyes of one patient with bilateral type 2A parafoveal telangiectasia. Indian J Ophthalmol. 2013;61(7):353–355. doi: 10.4103/0301-4738.99851 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Cheng JY, Adrian KH. Photodynamic therapy for choroidal neovascularization in stargardt disease and retinitis pigmentosa. Retin Cases Brief Rep. 2009;3(4):388–390. doi: 10.1097/ICB.0b013e31817f2e3f [DOI] [PubMed] [Google Scholar]