Abstract
BACKGROUND
Paragangliomas (PG) are rare neoplasms of neuroendocrine origin that tend to be highly vascularized, slow-growing, and usually sporadic. To date, common treatment options are surgical resection (SR), with or without radiation therapy (RT), and a watch-and-wait approach.
AIM
To evaluate the local control and effectiveness of exclusive fractionated stereotactic RT (FSRT) treatment in unresectable PG (uPG).
METHODS
We retrospectively evaluated patients with uPG (medically inoperable or refused SR) treated with FSRT with a Cyberknife System (Accuray Incorporated, Sunnyvale, California). Toxicity and initial efficacy were evaluated.
RESULTS
From May 2009 to January 2023, 6 patients with a median age of 68 (range 20-84) were treated with FSRT. The median delivered dose was 21 Gy (range 20-30 Gy) at a median isodose line of 75.5% (range 70%-76%) in 4 fractions (range 3-5 fractions). The median volume was 13.6 mL (range 12.4-65.24 mL). The median cumulative biological effective dose and equivalent dose in 2-Gy fractions were 70 Gy and 37.10 Gy respectively. Site of origin involved were the timpa-nojugular glomus (4/6), temporal bone, and cervical spine. In 1 of the 6 patients, the follow-up was insufficient; 5 of 6 patients showed a 5-year overall survival and 5-year progression-free survival of 100%. We observed negligible toxicities during and after RT. The majority of patients showed stable symptoms during follow-up. Only 1 patient developed spine metastases.
CONCLUSION
Our preliminary results on this small cohort of patients suggest that FSRT could be an effective and safe alternative to SR.
Keywords: Unresectable paraganglioma, Fractionated stereotactic radiation therapy, Cyberknife, Neurosurgery, Metastasis
Core Tip: Paragangliomas (PGs) are rare tumors with neuroendocrine origin. These lesions tend to be highly vascular and embryologically arise from the extra-adrenal autonomic nervous system, located in the thoracic, abdominal, and head-neck regions. PGs are usually sporadic, except in a few cases that are genetically determined by gene mutations. The clinical signs and symptoms include pulsatile tinnitus, headache, hearing loss, vocal fold paresis, vertigo, lower cranial nerve palsies, and tachycardias. Radiographic studies are pathognomonic in diagnosis. Treatment with fractionated stereotactic radiation therapy can be an effective option for these lesions, especially in reserving facial nerve function.
INTRODUCTION
Paragangliomas (PGs) are rare non-epithelial neoplasms of neuroendocrine origin, also known as chemodectoma, highly vascular and represent 0.6% of all head-neck tumors[1,2]. They embryologically arise from the autonomic nervous system extra-adrenal, in the thoracic, abdominal, and head-neck regions. 1%-3% of head-neck PGs are associated with catecholamines secretion[3]. The common sites of origin include the vagus nerve (vagal PG), temporal area, including tympanic PG and jugular bulb PG, and carotid artery bifurcation. Furthermore, they can show an intracranial extension that represents the main cause of death[4].
According to updated classification, PGs are not defined as “benign” or “malignant” but tumors of undetermined bio-logic with metastatic potential (especially in lymph nodes, liver, and lung) and a tendency to local infiltration of surrounding tissue such as bones or vessels[5]. PGs are usually sporadic, except in a few cases that are genetically determined by constitutional mutations of the genes that encode for the succinate dehydrogenase enzymes (SDHD and SDH)[6,7]. Patients with a genetic predisposition may present bilateral or even multifocal, recurrent PGs[8-10].
Depending on their location, size, and hormone activity, the clinical presentation may be extremely variable with pulsatile tinnitus, headache, hearing loss, vocal fold paresis, vertigo, lower cranial nerve palsies, tachycardia and la-bile blood pressure in catecholamine-secreting PGs. When the patient presents with classical symptoms related to catecholamine excess, the biochemical screening usually includes the measurement of urinary and plasma catecholamines, urinary fractionated metanephrines, plasma-free metanephrines, and urinary vanillylmandelic acid and other catecholamine metabolites[11].
If the diagnosis of PGs is suspected, fine needle biopsy is not indicated, but radiographic studies are pathognomonic. Computed tomography (CT) with contrast enhances these highly vascular PGs and can be utilized to define bone erosion and any possible skull base involvement[12-14]. A complementary imaging modality is magnetic resonance imaging (MRI) with gadolinium contrast that better demonstrates the relation of the PGs to the adjacent vessels. The PGs are characterized by a “salt and pepper” pattern on T2-weighted MRI, due to the high-flow vascular voids within the vascular tumor[15].
Digital subtraction angiography and 3D time-of-flight magnetic resonance angiography can detect flow dynamics and vascular architecture with high sensitivity and specificity[16,17]. 18Ga-Dotatoc positron emission tomography (PET)-CT, fluorodeoxy-glucose PET/CT, and I-123-metaiodobenzylguanidine single photon emission CT/CT can also be used for detecting secreting PGs[18,19]. The classification of PGs is based on the extension of the tumor to surrounding anatomic structures (Table 1). Fisch classification, Glasscock-Jackson, and Shamblin classification are widely used for temporal PGs and carotid body tumors respectively[3,20-23].
Table 1.
Classification of paragangliomas
|
Glasscock-Jackson classification
|
Fisch classification
|
Shamblin classification
|
||||
| TPG | JPG | JPG | Carotid PG | |||
| Type 1 | Small mass limited to the promontory | Involves jugular bulb, middle ear, mastoid process | Type A | Limited to middle ear cleft | Type 1 | Tumors are localized with minimal vascular attachments. These are easily amenable to complete resection with very little morbidity |
| Type 2 | Tumor completely filling the middle ear | Extends under canal, with or without intracranial extension | Type B | Limited to the tympanomastoid area; cortical bone over jugular bulb intact | Type 2 | Tumors partially surround the carotids. These are careful surgical excisions |
| Type 3 | Tumor filling middle ear and mastoid | Extends into petrous apex with or without intracranial extension | Type C (C1-C2-C3) | Involving the infralabyrinthine compartment and petrous apex of the temporal bone | Type 3 | Tumors encase the carotids. Surgical resection is difficult and may require major vessel reconstruction |
| Type 4 | Further extension through the EAC or anteriorly to the carotid artery | Extends beyond petrous apex into clivus or infratemporal fossa, with or without intracranial extension | Type D (D1-D2-D3) | Glomus jugular tumors with intracranial extensions | ||
TPG: Tympanic paraganglioma; JPG: Jugular bulb paraganglioma; PG: Paraganglioma; EAC: External auditory canal.
For asymptomatic or elderly patients watch and wait approach is reasonable. For symptomatic patients, to date, common treatment options are surgical resection (SR), embolization with or without surgical resection, radiation therapy (RT), as definitive, adjuvant, or salvage therapy. SR represents the first-line treatment but, due to the proximity of the tumor to critical neurovascular structures, it is often complicated especially in larger tumors[24-26].
When PGs are considered unresectable (uPG), alternative treatments are represented by subtotal resection in association with RT (for residual, recurrent tumors or giant tumors) or RT alone. Several studies reported a significant local control with acceptable toxicities in patients who underwent subtotal resection followed by RT or treated with RT only[27-30]. RT treatment can be performed with a Cyberknife System (CK), Gamma Knife System (GKS), and linear accelerator (LINAC)-based stereotactic radiosurgery (SRS) with single or multi-fractions schedules [fractionated stereotactic RT (FSRT)][31-33].
SRS is usually performed when PGs measure less than 3 cm, whereas tumors that are larger or have a component of extracranial spread are suitable for conventionally fractionated radiotherapy [external beam RT (EBRT)][12-14]. The aim of our study is to evaluate the local control and effectiveness of exclusive FSRT treatment in uPG.
MATERIALS AND METHODS
The study was conducted at the Department of Radiation Oncology of the University of Messina, Italy. Patients were retrospectively identified from the institutional register. Written consent was obtained from patients who were able to communicate or from their next of kin if the patient couldn't provide consent. The techniques described are standard and the data collection was retrospective, thus special Institutional Review Board approval was not required. This study was performed according to the ethical standards of our Institutional Review Board and in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
The patients considered included those with PGs radiologically proven, age >18 years, and availability of complete pre- and postoperative clinical and radiological data (contrast- CT scan, MRI, and angiography) for localization of the tumors. The Glasscock-Jackson and Fisch classifications were used. To evaluate the facial nerve damage, the House-Brackmann grading system was used. All patients received exclusive FSRT.
The pretreatment imaging consisted of a thin-section multiplanar reconstruction-gradient echo volumetric study conducted on a Siemens Magnetom 1.5T MRI system (Siemens, Erlangen, Germany), performed with the following parameters: Repetition time 9.7 ms, echo time 4 ms, matrix 200 × 256, flip angle 1, orientation sagittal. A multislice CT was also performed using a multislice scanner, Siemens Sensation 16 (Siemens, Erlangen, Germany).
The Multiplan Treatment Planning System (Accuray Incorporated, Sunnyvale, California) was used for inverse treatment planning. The gross target volume (GTV) was contrast CT and MRI-based and was defined according to the radiological findings. Planning target volume (PTV) was created by defining a 1-mm margin to GTV.
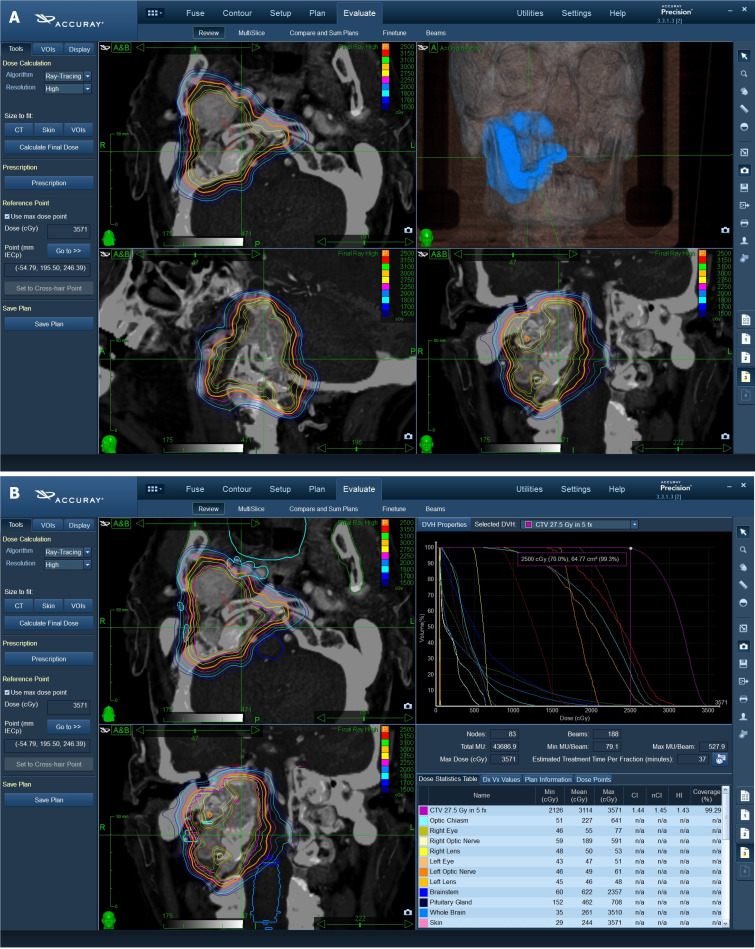
The critical organs at risk (OARs) were outlined in the axial plane with a simultaneous display of contours on re-constructed orthogonal images. OARs included: Normal brain, optic chiasm, brain stem, hypophysis, bilateral eyes and lens, optic nerves, pituitary gland, and cochlea, cranial nerves, oral cavity, mandible, parotid gland, esophagus. The characteristics of treatment are reported in Figure 1.
Figure 1.
The characteristics of treatment. A: Target definition and 3D dose distribution; B: Cyberknife treatment planning and dose-volume distribution.
Treatment was delivered using a CyberKnife System (Accuray Incorporated, Sunnyvale, California), an image-guided, frameless, LINAC-based, 6 MV radiosurgery system with Skull Tracking. The ray-tracing algorithm was routinely used for non-isocentric beam delivery.
Clinical and radiologic follow-up with contrast-enhanced MRI T1-T2 weighted, proton density, and fluid-attenuated in-version recovery sequences MRI, were obtained at three months and then every six months for two years followed by yearly evaluations. We included the latest available follow-up in this analysis.
Toxicity and initial efficacy were evaluated. The clinical status of the patients was classified using the Karnofsky Performance Status before treatment and at the last follow-up; new neurologic deficits and any neurological event were recorded separately. The revised NCI Common Terminology Criteria for Adverse Events, version 4.0 was applied to evaluate radiotherapy-related toxicity. RECIST v1.1 criteria were used to evaluate response to treatment.
RESULTS
From May 2009 to January 2023, 6 patients (4 females, 2 males) with a median age of 68 ± 24.4 interquartile range (IQR) (range 20-84) were treated with FSRT. Five of six patients had temporal PGs: In 4 patients, PGs originate from the timpano jugular glomus and one from the temporal bone. Based on Glasscock-Jackson classification, temporal PGs were type 3 (1/6), three jugular PGs were type 3 (3/6), and one type 2. According to Fish classification, 4 patients showed grade D and 1 grade C. One patient had cervical spine PG. At the time of presentation, symptoms included cervical mass with the displacement of critical structure and severe deficit of cranial nerves. Patients referred asymmetry of mouth (VII, facial nerve, House-Brackmann grade 4), hearing loss (VII acoustic nerve), dysphagia (IX, glossopharyngeal nerve), pulsatile tinnitus and swallowing difficulty (X, vagal nerve), and limited forehead movement mouth (XI, hypoglossal nerve). Only 1 patient showed SDHB mutation. The characteristics of patients are summarized in Table 2.
Table 2.
Patients characteristics
|
Patients
|
Age (yr)
|
Site of origin
|
Fisch classification
|
Glasscock-Jackson classification
|
Surgery
|
Volume (mL)
|
Dose (Gy)
|
Isodose (%)
|
Fraction
|
Pathogenesis
|
| 1 | 20 | TPGs | D | 3 | No | 12.4 | 21 | 75 | 3 | Sporadic |
| 2 | 60 | Cervical spine | \ | \ | No | 20.2 | 20 | 77 | 5 | SDHB mut |
| 3 | 68 | JPG | D | 3 | No | 12.7 | 20 | 73 | 3 | Sporadic |
| 4 | 44 | JPG | C | 2 | No | 14.5 | 21 | 76 | 3 | Sporadic |
| 5 | 84 | JPG | D | 3 | No | 64.24 | 25 | 70 | 5 | Sporadic |
| 6 | 74 | JPG | D | 3 | No | 10.99 | 30 | 82.5 | 5 | Sporadic |
TPG: Tympanic paraganglioma; JPG: Jugular bulb paraganglioma.
The median delivered dose was 21 Gy ± 3.75 IQR (range 20-30 Gy) at a median isodose line 75.5% ± 3.25% IQR (range 70%-76%) in 4 fractions (range 3-5 fractions). The median volume was 13.6 mL ± 6.3 IQR (ranging 12.4-65.24 mL). The median cumulative biological effective dose (a/b 4.5) and equivalent dose in 2-Gy fractions were 70 Gy and 37.10 Gy ± 2.76 IQR respectively.
The median follow-up was 72 months ± 66.75 IQR. In 1 of the 6 patients, the follow-up was insufficient. The median overall survival (OS) was 72 months ± 111.75 IQR; the median progression-free survival (PFS) was 72 months ± 66.75 IQR 5 of 6 patients showed a 5-year OS and 5-year PFS of 100%. We observed negligible toxicities during and after RT. Five patients showed mild cranial nerve: 2 to the facial nerve (House-Brackmann grade 2), 1 to the vagal and hypoglossal cranial nerves, and 1 to the acoustic and glossopharyngeal nerves. The majority of patients showed stable symptoms during follow-up. Patient outcomes are summarized in Table 3. Only 1 patient developed spine metastases 12 years after FSRT. A revision of the histopathological sample was required showing a malignant PG. The patient underwent re-irradiation with proton therapy.
Table 3.
Summary of patient outcome
|
Patients
|
OS (months)
|
PFS (months)
|
Response treatment
|
| 1 | 180 | 144 | PD |
| 2 | 156 | 96 | PR |
| 3 | 72 | 72 | PR |
| 4 | 72 | 72 | PR |
| 5 | 5 | 5 | NA |
| 6 | 6 | 6 | PR |
OS: Overall survival; PFS: Progression free survival; PD: Progression disease; PR: Partial response; NA: Not available.
DISCUSSION
The management of PGs is controversial. The first option is represented by surgical resection, but it is associated with a high complication rate as nerve damage, stroke, and bleeding, exacerbated by possible previous embolization[34-38]. RT was widely studied as an alternative to surgery and has shown favorable results[39]. Both FSRT and SRS have been investigated and historically considered for recurrent or residual disease[40,41] including PGs, after subtotal resection with high rates of local control[24-26].
SRS shows equal efficacy with lower toxicity rates than FSRT and is reasonable for PGs 3 cm or less in size[42-44]. Fatima et al[45], in their metanalysis, showed a pooled local control of 94.2% with no statistically significant difference in local control between different techniques of SRS (CK, GKS, LINAC). The analysis comprises patients who underwent both SR and embolization and subsequent RT, in a median volume of 8.4 mL and with a median dose of 15 Gy in 1-5 fractions[45].
Several studies showed an optimal local control with negligible toxicities delivering a single fraction in small lesions. Principal methods of SRS include GKS and LINAC radiosurgery[46]. Patel et al[47] evaluated the quality of life in 26 patients treated with RT performed with GKS, alone or in adjuvant modality. The median radiation dose was 16 Gy in PGs with a median tumor volume of 7090 mm3. The study showed better outcomes in patients who underwent primary SRS than adjuvant SRS[47].
Ehret et al[48] evaluated the efficacy of the SRS performed with CK in patients who had prior treatment, SR, or embolization. Ehret et al[48] using a median dose of 16.5 Gy, with a median prescription isodose line of 70% achieved a 5-year actuarial local control (LC) of 100% in PGs of 4.3 mL (median volume). Marchetti et al[31] delivered a median dose of 12 Gy (median isodose line was 78.6%) in PGs with a median volume of 3.6 mL with no radiological progression at the site of the treatment.
For greater lesions, FSRT or EBRT are generally considered. Marchetti et al[31], in patients with greater lesions ( median volume 16 mL), delivered a median dose of 25 Gy in 5 fractions at a median isodose line of 80%.
Tosun et al[49] delivered FSRT in 12 patients. The median dose of 24 Gy with a median isodose line of 75%. The median tumor volume was 35.5 mL (range 5.3-113.8 mL). Of them, 7 underwent SR and according to Fisch classification 6 were D2 and only 1 C1. Only 1 patient with D2 PGs had no SR and was treated with RT alone with a dose of 30 Gy in 10 fractions with the Cyberknife System. In this study, with a median follow-up of 30 months, no acute or late toxicity related to FSRT was observed and LC was 100%[49].
Another study reported a fractionated stereotactic treatment with CK in PGs with volume ranging from 0.84 to 69.3 cm3 (median volume of 4.64 cm3). Out of 36 patients, 12 with no prior treatment, received FSRT (volume ranged from 9.73 to 69.3 cm3). A patient with a volume of 69.3 cm3 was treated with 25 Gy in 5 fractions; another PG of 42 cm3 was divided into two portions (intracranial and cervical) that received 24 Gy in 3 fractions respectively[50].
In a retrospective study, 81 PGs were treated by conventional EBRT in 25 fractions with a median dose of 45 Gy (range 41.4-68 Gy), of whom 60 were treated with exclusive RT and 21 had prior surgery. The median GTV and PTV were 30 cm3. The 5-year and 10-year actuarial LC rates were 100% and 98.7% respectively. In this study, 5 patients showed grade 3 late toxicity and 2 patients developed secondary meningiomas[51].
There is no consensus to define PGs as “giant”. Main et al[30], in their case report, considered giant PG with a volume of 93.553 cm3 that was treated with only SR, whereas López-Arcas et al[52] reported a case of combination treatment in giant PG of a maximum diameter of 4 cm, removed surgically after embolization and treated with FSRT performed with a coverage dose of 14 Gy at an isodose of 83%. Other studies evaluated the efficacy of RT in unresectable or bulky diseases[53,54].
To the best of our knowledge, lack of data on the management of unresectable PGs due to high dimension or bone infiltration. This is the latest analysis of the efficacy and feasibility of exclusive FSRT in unresectable paraganglioma using a CK.
Here we report a case series of PG judged unresectable and treated with exclusive FSRT. The PGs volumes ranged from 12.4 to 65.24 mL. The treatment was delivered with a median dose of 21 Gy in 4 fractions using the CK. No acute and late toxicities were observed. All patients had stable or improved clinical status and LC rates were comparable with literature data. Characteristics of the analyzed studies are summarized in Table 4.
Table 4.
Characteristics of the analyzed studies
|
Ref.
|
PGs
Resectable |
Embolization
|
Median dose (Gy)
|
Isodose (%)
|
Median fractions
|
Technique
|
Median volume (mL)/
size (cm) |
Outcome
|
| Fatima et al[45] | Yes (NA) | Yes (NA) | 15 | NA | 1-5 | CK-GKS-LINAC | 8.4 mL | 94.2% LC |
| Patel et al[47] | Yes (10/26) | Yes (1/26) | 16 | NA | 1 | GKS | 7090 mL | NA |
| Ehret et al[48] | Yes (20/53) | Yes (8/53) | 16.5 | 70 | 1 | CK | 4.3 mL | 100% 5-year LC |
| Marchetti et al[31] | Yes (NA) | No | 12.6 | 78.6 | 1 | CK | 3.6 mL |
33% LC (35 months) |
| 25 | 80 | 5 | 16.1 mL | |||||
| Tosun et al[49] | Yes (NA) | No | 24 | 75 | 3 | CK | 35.5 mL | 100% LC |
| Lieberson et al[50] | 24/12 | 20 | 80 | 1-5 | CK | 4.64 mL | 100% LC | |
| Dupin et al[51] | Yes (21/81) No (60/21) |
Yes (1/81) | 25 | \ | 25 |
60Co machine/ LINAC |
30 mL | 100% 5-year LC; 98.7% 10-year LC |
| Main et al[30] | Yes (1/1) | Yes (1/1) | \ | \ | \ | \ | 93.553 mL | NA |
| López-Arcas et al[52] | Yes (1/1) | Yes (1/1) | 14 | 2 | CK | 4 cm | NA | |
| This study | No | No | 21 | 75.5 | 4 | CK | 13 mL | 100% |
PG: Paragangliomas; NA: Not available; CK: Cyberknife System; GKS: Gamma Knife System; LINAC: Linear accelerator; LC: Local control.
The present study has several limitations; the small series of patients does not allow to perform powered statistical analyses and the minimum follow-up period is too short to evaluate long-term recurrent. Therefore, our results must be confirmed by studies with larger patient sizes and longer follow-ups.
CONCLUSION
In conclusion, despite the limitation of the study, our results suggest that exclusive FSRT could be an effective and safe alternative when SR is excluded.
Footnotes
Institutional review board statement: The surgical techniques are standard and the data collection was retrospective, thus special IRB approval was not required. This study was performed according to the ethical standards of our Institutional Review Board and in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent statement: Patients were not required to give informed consent to the study because the analysis used anonymous clinical data that were obtained after each patient agreed to treatment by written consent.
Conflict-of-interest statement: The authors declare no conflict of interest.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Medicine, research and experimental
Country of origin: Italy
Peer-review report’s classification
Scientific Quality: Grade A
Novelty: Grade C
Creativity or Innovation: Grade C
Scientific Significance: Grade B
P-Reviewer: Ren G, China S-Editor: Che XX L-Editor: A P-Editor: Zhao S
Contributor Information
Antonio Pontoriero, Radiation Oncology Unit, Department of Biomedical, Dental Science and Morphological and Functional Images, University of Messina, Messina 98125, Italy.
Paola Critelli, Radiation Oncology Unit, Department of Biomedical, Dental Science and Morphological and Functional Images, University of Messina, Messina 98125, Italy.
Marco Zeppieri, Department of Ophthalmology, University Hospital of Udine, Udine 33100, Italy. markzeppieri@hotmail.com.
Filippo Flavio Angileri, Neurosurgery Unit, Department of Biomedical, Dental Science and Morphological and Functional Images, University of Messina, Messina 98125, Italy.
Tamara Ius, Neurosurgery Unit, Head-Neck and NeuroScience Department, University Hospital of Udine, Udine 33100, Italy.
Data sharing statement
Data is available upon written request.
References
- 1.Taïeb D, Kaliski A, Boedeker CC, Martucci V, Fojo T, Adler JR Jr, Pacak K. Current approaches and recent developments in the management of head and neck paragangliomas. Endocr Rev. 2014;35:795–819. doi: 10.1210/er.2014-1026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pellitteri PK, Rinaldo A, Myssiorek D, Gary Jackson C, Bradley PJ, Devaney KO, Shaha AR, Netterville JL, Manni JJ, Ferlito A. Paragangliomas of the head and neck. Oral Oncol. 2004;40:563–575. doi: 10.1016/j.oraloncology.2003.09.004. [DOI] [PubMed] [Google Scholar]
- 3.Offergeld C, Brase C, Yaremchuk S, Mader I, Rischke HC, Gläsker S, Schmid KW, Wiech T, Preuss SF, Suárez C, Kopeć T, Patocs A, Wohllk N, Malekpour M, Boedeker CC, Neumann HP. Head and neck paragangliomas: clinical and molecular genetic classification. Clinics (Sao Paulo) 2012;67 Suppl 1:19–28. doi: 10.6061/clinics/2012(Sup01)05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Boedeker CC, Ridder GJ, Neumann HP, Maier W, Schipper J. [Diagnosis and management of cervical paragangliomas: the Freiburg experience] Laryngorhinootologie. 2004;83:585–592. doi: 10.1055/s-2004-814466. [DOI] [PubMed] [Google Scholar]
- 5.Williams MD, Tischler AS. Update from the 4th Edition of the World Health Organization Classification of Head and Neck Tumours: Paragangliomas. Head Neck Pathol. 2017;11:88–95. doi: 10.1007/s12105-017-0786-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Burnichon N, Abermil N, Buffet A, Favier J, Gimenez-Roqueplo AP. The genetics of paragangliomas. Eur Ann Otorhinolaryngol Head Neck Dis. 2012;129:315–318. doi: 10.1016/j.anorl.2012.04.007. [DOI] [PubMed] [Google Scholar]
- 7.Timmers HJ, Gimenez-Roqueplo AP, Mannelli M, Pacak K. Clinical aspects of SDHx-related pheochromocytoma and paraganglioma. Endocr Relat Cancer. 2009;16:391–400. doi: 10.1677/ERC-08-0284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Grufferman S, Gillman MW, Pasternak LR, Peterson CL, Young WG Jr. Familial carotid body tumors: case report and epidemiologic review. Cancer. 1980;46:2116–2122. doi: 10.1002/1097-0142(19801101)46:9<2116::aid-cncr2820460934>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 9.Netterville JL, Jackson CG, Miller FR, Wanamaker JR, Glasscock ME. Vagal paraganglioma: a review of 46 patients treated during a 20-year period. Arch Otolaryngol Head Neck Surg. 1998;124:1133–1140. doi: 10.1001/archotol.124.10.1133. [DOI] [PubMed] [Google Scholar]
- 10.Netterville JL, Reilly KM, Robertson D, Reiber ME, Armstrong WB, Childs P. Carotid body tumors: a review of 30 patients with 46 tumors. Laryngoscope. 1995;105:115–126. doi: 10.1288/00005537-199502000-00002. [DOI] [PubMed] [Google Scholar]
- 11.Kiernan CM, Solórzano CC. Pheochromocytoma and Paraganglioma: Diagnosis, Genetics, and Treatment. Surg Oncol Clin N Am. 2016;25:119–138. doi: 10.1016/j.soc.2015.08.006. [DOI] [PubMed] [Google Scholar]
- 12.Hu K, Persky MS. Multidisciplinary management of paragangliomas of the head and neck, Part 1. Oncology (Williston Park) 2003;17:983–993. [PubMed] [Google Scholar]
- 13.Hu K, Persky MS. The multidisciplinary management of paragangliomas of the head and neck, Part 2. Oncology (Williston Park) 2003;17:1143–1153; discussion 1154, 1158, 1161. [PubMed] [Google Scholar]
- 14.Persky MS, Hu KS. Paragangliomas of the head and neck. In: Harrison LB, Hong WK, Sessions RB, editors. Head and Neck Cancer: A Multidisciplinary Approach. 3rd ed. Philadephia: Lippincott Williams & Wilkins; 2009: 655-687. [Google Scholar]
- 15.Olsen WL, Dillon WP, Kelly WM, Norman D, Brant-Zawadzki M, Newton TH. MR imaging of paragangliomas. AJR Am J Roentgenol. 1987;148:201–204. doi: 10.2214/ajr.148.1.201. [DOI] [PubMed] [Google Scholar]
- 16.Neves F, Huwart L, Jourdan G, Reizine D, Herman P, Vicaut E, Guichard JP. Head and neck paragangliomas: value of contrast-enhanced 3D MR angiography. AJNR Am J Neuroradiol. 2008;29:883–889. doi: 10.3174/ajnr.A0948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van den Berg R, Schepers A, de Bruïne FT, Liauw L, Mertens BJ, van der Mey AG, van Buchem MA. The value of MR angiography techniques in the detection of head and neck paragangliomas. Eur J Radiol. 2004;52:240–245. doi: 10.1016/j.ejrad.2003.12.002. [DOI] [PubMed] [Google Scholar]
- 18.Kong G, Schenberg T, Yates CJ, Trainer A, Sachithanandan N, Iravani A, Ravi Kumar A, Hofman MS, Akhurst T, Michael M, Hicks RJ. The Role of 68Ga-DOTA-Octreotate PET/CT in Follow-Up of SDH-Associated Pheochromocytoma and Paraganglioma. J Clin Endocrinol Metab. 2019;104:5091–5099. doi: 10.1210/jc.2019-00018. [DOI] [PubMed] [Google Scholar]
- 19.Chang CA, Pattison DA, Tothill RW, Kong G, Akhurst TJ, Hicks RJ, Hofman MS. (68)Ga-DOTATATE and (18)F-FDG PET/CT in Paraganglioma and Pheochromocytoma: utility, patterns and heterogeneity. Cancer Imaging. 2016;16:22. doi: 10.1186/s40644-016-0084-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jackson CG, Glasscock ME 3rd, Harris PF. Glomus Tumors. Diagnosis, classification, and management of large lesions. Arch Otolaryngol. 1982;108:401–410. doi: 10.1001/archotol.1982.00790550005002. [DOI] [PubMed] [Google Scholar]
- 21.Oldring D, Fisch U. Glomus tumors of the temporal region: surgical therapy. Am J Otol. 1979;1:7–18. [PubMed] [Google Scholar]
- 22.Fisch U. Infratemporal fossa approach for glomus tumors of the temporal bone. Ann Otol Rhinol Laryngol. 1982;91:474–479. doi: 10.1177/000348948209100502. [DOI] [PubMed] [Google Scholar]
- 23.Shamblin WR, ReMine WH, Sheps SG, Harrison EG Jr. Carotid body tumor (chemodectoma). Clinicopathologic analysis of ninety cases. Am J Surg. 1971;122:732–739. doi: 10.1016/0002-9610(71)90436-3. [DOI] [PubMed] [Google Scholar]
- 24.Lim JY, Kim J, Kim SH, Lee S, Lim YC, Kim JW, Choi EC. Surgical treatment of carotid body paragangliomas: outcomes and complications according to the shamblin classification. Clin Exp Otorhinolaryngol. 2010;3:91–95. doi: 10.3342/ceo.2010.3.2.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sanna M, Jain Y, De Donato G, Rohit, Lauda L, Taibah A. Management of jugular paragangliomas: the Gruppo Otologico experience. Otol Neurotol. 2004;25:797–804. doi: 10.1097/00129492-200409000-00025. [DOI] [PubMed] [Google Scholar]
- 26.Ivan ME, Sughrue ME, Clark AJ, Kane AJ, Aranda D, Barani IJ, Parsa AT. A meta-analysis of tumor control rates and treatment-related morbidity for patients with glomus jugulare tumors. J Neurosurg. 2011;114:1299–1305. doi: 10.3171/2010.9.JNS10699. [DOI] [PubMed] [Google Scholar]
- 27.Anderson JL, Khattab MH, Anderson C, Sherry AD, Luo G, Manzoor N, Attia A, Netterville J, Cmelak AJ. Long-term Outcomes for the Treatment of Paragangliomas in the Upfront, Adjuvant, and Salvage Settings With Stereotactic Radiosurgery and Intensity-modulated Radiotherapy. Otol Neurotol. 2020;41:133–140. doi: 10.1097/MAO.0000000000002461. [DOI] [PubMed] [Google Scholar]
- 28.Papaspyrou K, Mewes T, Rossmann H, Fottner C, Schneider-Raetzke B, Bartsch O, Schreckenberger M, Lackner KJ, Amedee RG, Mann WJ. Head and neck paragangliomas: Report of 175 patients (1989-2010) Head Neck. 2012;34:632–637. doi: 10.1002/hed.21790. [DOI] [PubMed] [Google Scholar]
- 29.Künzel J, Iro H, Hornung J, Koch M, Brase C, Klautke G, Zenk J. Function-preserving therapy for jugulotympanic paragangliomas: a retrospective analysis from 2000 to 2010. Laryngoscope. 2012;122:1545–1551. doi: 10.1002/lary.23268. [DOI] [PubMed] [Google Scholar]
- 30.Main AM, Benndorf G, Feldt-Rasmussen U, Fugleholm K, Kistorp T, Loya AC, Poulsgaard L, Rasmussen ÅK, Rossing M, Sølling C, Klose MC. Case Report: Giant Paraganglioma of the Skull Base With Two Somatic Mutations in SDHB and PTEN Genes. Front Endocrinol (Lausanne) 2022;13:857504. doi: 10.3389/fendo.2022.857504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Marchetti M, Pinzi V, Tramacere I, Bianchi LC, Ghielmetti F, Fariselli L. Radiosurgery for Paragangliomas of the Head and Neck: Another Step for the Validation of a Treatment Paradigm. World Neurosurg. 2017;98:281–287. doi: 10.1016/j.wneu.2016.10.132. [DOI] [PubMed] [Google Scholar]
- 32.Huo M, Sahgal A, Pryor D, Redmond K, Lo S, Foote M. Stereotactic spine radiosurgery: Review of safety and efficacy with respect to dose and fractionation. Surg Neurol Int. 2017;8:30. doi: 10.4103/2152-7806.200581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Patel AK, Rodríguez-López JL, Hirsch BE, Burton SA, Flickinger JC, Clump DA. Long term outcomes with linear accelerator stereotactic radiosurgery for treatment of jugulotympanic paragangliomas. Head Neck. 2021;43:449–455. doi: 10.1002/hed.26497. [DOI] [PubMed] [Google Scholar]
- 34.Papaspyrou K, Mann WJ, Amedee RG. Management of head and neck paragangliomas: review of 120 patients. Head Neck. 2009;31:381–387. doi: 10.1002/hed.20967. [DOI] [PubMed] [Google Scholar]
- 35.Langerman A, Athavale SM, Rangarajan SV, Sinard RJ, Netterville JL. Natural history of cervical paragangliomas: outcomes of observation of 43 patients. Arch Otolaryngol Head Neck Surg. 2012;138:341–345. doi: 10.1001/archoto.2012.37. [DOI] [PubMed] [Google Scholar]
- 36.Persky MS, Setton A, Niimi Y, Hartman J, Frank D, Berenstein A. Combined endovascular and surgical treatment of head and neck paragangliomas--a team approach. Head Neck. 2002;24:423–431. doi: 10.1002/hed.10068. [DOI] [PubMed] [Google Scholar]
- 37.Suárez C, Rodrigo JP, Bödeker CC, Llorente JL, Silver CE, Jansen JC, Takes RP, Strojan P, Pellitteri PK, Rinaldo A, Mendenhall WM, Ferlito A. Jugular and vagal paragangliomas: Systematic study of management with surgery and radiotherapy. Head Neck. 2013;35:1195–1204. doi: 10.1002/hed.22976. [DOI] [PubMed] [Google Scholar]
- 38.Green JD Jr, Brackmann DE, Nguyen CD, Arriaga MA, Telischi FF, De la Cruz A. Surgical management of previously untreated glomus jugulare tumors. Laryngoscope. 1994;104:917–921. doi: 10.1288/00005537-199408000-00001. [DOI] [PubMed] [Google Scholar]
- 39.Conti A, Pontoriero A, Iatì G, Cardali SM, Brogna A, Friso F, Rosetti V, Zoli M, Parisi S, Cacciola A, Lillo S, Pergolizzi S, Mazzatenta D. Image-Guided Multisession Radiosurgery of Skull Base Meningiomas. Cancers (Basel) 2020;12:3569. doi: 10.3390/cancers12123569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pontoriero A, Critelli P, Conti A, Cardali S, Angileri FF, Germanò A, Lillo S, Carretta A, Brogna A, Santacaterina A, Parisi S, Pergolizzi S. The "Combo" radiotherapy treatment for high-risk grade 2 meningiomas: dose escalation and initial safety and efficacy analysis. J Neurooncol. 2023;161:203–214. doi: 10.1007/s11060-022-04107-3. [DOI] [PubMed] [Google Scholar]
- 41.Parisi S, Ferini G, Lillo S, Brogna A, Chillari F, Ferrantelli G, Settineri N, Santacaterina A, Platania A, Leotta S, Casablanca G, Russo A, Pontoriero A, Adamo V, Minutoli F, Bottari A, Cacciola A, Pergolizzi S. Stereotactic boost on residual disease after external-beam irradiation in clinical stage III non-small cell lung cancer: mature results of stereotactic body radiation therapy post radiation therapy (SBRTpostRT) study. Radiol Med. 2023;128:877–885. doi: 10.1007/s11547-023-01659-w. [DOI] [PubMed] [Google Scholar]
- 42.Hu K, Persky MS. Treatment of Head and Neck Paragangliomas. Cancer Control. 2016;23:228–241. doi: 10.1177/107327481602300306. [DOI] [PubMed] [Google Scholar]
- 43.Pollock BE. Stereotactic radiosurgery in patients with glomus jugulare tumors. Neurosurg Focus. 2004;17:E10. doi: 10.3171/foc.2004.17.2.10. [DOI] [PubMed] [Google Scholar]
- 44.Sahyouni R, Mahboubi H, Moshtaghi O, Goshtasbi K, Sahyouni S, Lin HW, Djalilian HR. Radiosurgery of Glomus Tumors of Temporal Bone: a Meta-analysis. Otol Neurotol. 2018;39:488–493. doi: 10.1097/MAO.0000000000001737. [DOI] [PubMed] [Google Scholar]
- 45.Fatima N, Pollom E, Soltys S, Chang SD, Meola A. Stereotactic radiosurgery for head and neck paragangliomas: a systematic review and meta-analysis. Neurosurg Rev. 2021;44:741–752. doi: 10.1007/s10143-020-01292-5. [DOI] [PubMed] [Google Scholar]
- 46.Dharnipragada R, Butterfield JT, Dhawan S, Adams ME, Venteicher AS. Modern Management of Complex Tympanojugular Paragangliomas: Systematic Review and Meta-Analysis. World Neurosurg. 2023;170:149–156.e3. doi: 10.1016/j.wneu.2022.11.037. [DOI] [PubMed] [Google Scholar]
- 47.Patel NS, Link MJ, Tombers NM, Pollock BE, Carlson ML. Quality of Life in Jugular Paraganglioma Following Radiosurgery. Otol Neurotol. 2019;40:820–825. doi: 10.1097/MAO.0000000000002250. [DOI] [PubMed] [Google Scholar]
- 48.Ehret F, Kufeld M, Fürweger C, Haidenberger A, Schichor C, Tonn JC, Muacevic A, Hempel JM. Single-session image-guided robotic radiosurgery and quality of life for glomus jugulare tumors. Head Neck. 2020;42:2421–2430. doi: 10.1002/hed.26231. [DOI] [PubMed] [Google Scholar]
- 49.Tosun İ, Atalar B, Şahin B, Güngör G, Aydin G, Yapici B, Özyar E. Robotic radiosurgery of head and neck paragangliomas: a single institution experience. Asia Pac J Clin Oncol. 2018;14:e3–e7. doi: 10.1111/ajco.12695. [DOI] [PubMed] [Google Scholar]
- 50.Lieberson RE, Adler JR, Soltys SG, Choi C, Gibbs IC, Chang SD. Stereotactic radiosurgery as the primary treatment for new and recurrent paragangliomas: is open surgical resection still the treatment of choice? World Neurosurg. 2012;77:745–761. doi: 10.1016/j.wneu.2011.03.026. [DOI] [PubMed] [Google Scholar]
- 51.Dupin C, Lang P, Dessard-Diana B, Simon JM, Cuenca X, Mazeron JJ, Feuvret L. Treatment of head and neck paragangliomas with external beam radiation therapy. Int J Radiat Oncol Biol Phys. 2014;89:353–359. doi: 10.1016/j.ijrobp.2014.02.010. [DOI] [PubMed] [Google Scholar]
- 52.López-Arcas JM, Colmenero CM, Martínez R, Martín-Hernán F, Ruiz-Sánchez B, Aragoneses JM. Giant carotid chemodectoma treated with a combination of surgery and CyberKnife radiotherapy: a case report and review of the literature. J Med Case Rep. 2022;16:92. doi: 10.1186/s13256-021-03237-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Parisi S, Ferini G, Cacciola A, Lillo S, Tamburella C, Santacaterina A, Bottari A, Brogna A, Ferrantelli G, Pontoriero A, Minutoli F, Pergolizzi S. A non-surgical COMBO-therapy approach for locally advanced unresectable pancreatic adenocarcinoma: preliminary results of a prospective study. Radiol Med. 2022;127:214–219. doi: 10.1007/s11547-021-01441-w. [DOI] [PubMed] [Google Scholar]
- 54.Ferini G, Parisi S, Lillo S, Viola A, Minutoli F, Critelli P, Valenti V, Illari SI, Brogna A, Umana GE, Ferrantelli G, Lo Giudice G, Carrubba C, Zagardo V, Santacaterina A, Leotta S, Cacciola A, Pontoriero A, Pergolizzi S. Impressive Results after "Metabolism-Guided" Lattice Irradiation in Patients Submitted to Palliative Radiation Therapy: Preliminary Results of LATTICE_01 Multicenter Study. Cancers (Basel) 2022;14:3909. doi: 10.3390/cancers14163909. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data is available upon written request.