Abstract
Several studies have revealed that midbrain dopamine (DA) neurons, even within a single neuroanatomical area, display heterogeneous properties. In parallel, studies using single cell profiling techniques have begun to cluster DA neurons into subtypes based on their molecular signatures. Recent work has shown that molecularly defined DA subtypes within the substantia nigra (SNc) display distinctive anatomic and functional properties, and differential vulnerability in Parkinson’s disease (PD). Based on these provocative results, a granular understanding of these putative subtypes and their alterations in PD models, is imperative. We developed an optimized pipeline for single-nuclear RNA sequencing (snRNA-seq) and generated a high-resolution hierarchically organized map revealing 20 molecularly distinct DA neuron subtypes belonging to three main families. We integrated this data with spatial MERFISH technology to map, with high definition, the location of these subtypes in the mouse midbrain, revealing heterogeneity even within neuroanatomical sub-structures. Finally, we demonstrate that in the preclinical LRRK2G2019S knock-in mouse model of PD, subtype organization and proportions are preserved. Transcriptional alterations occur in many subtypes including those localized to the ventral tier SNc, where differential expression is observed in synaptic pathways, which might account for previously described DA release deficits in this model. Our work provides an advancement of current taxonomic schemes of the mouse midbrain DA neuron subtypes, a high-resolution view of their spatial locations, and their alterations in a prodromal mouse model of PD.
Introduction
Midbrain DA neurons are traditionally organized in three main anatomical areas – the ventral tegmental area (VTA), substantia nigra pars compacta (SNc) and retrorubral area (RR)1, with further sub-divisions therein2. To explain the disparate roles of DA in normal behavior and disease, additional heterogeneity of midbrain neurons within these areas has been postulated3. Recent studies have indeed revealed diversity within clusters in electrophysiological properties as well as responses during various behavioral paradigms4–15. Complementing these studies, recent evidence using single cell classification has opened the possibility that DA neurons can be clustered based on their molecular signatures16–25. Early studies have begun to suggest that molecularly distinct DA populations may have distinctive anatomical projection patterns, as well as functionally distinct activity patterns4, 25–27. For example, in the SNc, a ventral Anxa1+ population is enriched in acceleration correlated activity, whereas dorsal Calb1+ or lateral Vglut2+ populations are enriched in deceleration correlated activity25. These results suggest that molecularly defined DA neurons need to be considered as a key variable for electrophysiological and behavioral studies.
Based on these exciting results, and the fact that the functional interrogation of DA neurons is happening at rapid speed often agnostic to DA subpopulations, there is a clear need to define DA neuron subtypes with more granularity. Previous studies using single cell sequencing in mice have been limited by the number of cells analyzed, the quality of cDNA library, as well as bias during isolation, thus providing an incomplete picture of DA neuron subtypes17, 19–23. Furthermore, the anatomical distribution of these subtypes in the midbrain remains only partially elucidated. Additionally, given the closely related nature of these subtypes, the stability of these subtypes across pathological conditions remains unclear. In other words, are some of these “subtypes” a representation of cell state rather than cell type?
The unbiased study of the transcriptomic landscape of DA neuron subtypes in the midbrain can inform about the molecular drivers of selective dysfunction of DA neurons in PD. Pathogenic mutations which increase leucine-rich repeat kinase 2 (LRRK2) activity are one of the most common causes of autosomal dominant PD and clinically similar to sporadic cases28, 29,30. Additionally, even in patients with idiopathic PD, LRRK2 kinase activity is increased in DA neurons31, providing justification for studying LRRK2-driven mechanisms in DA neurons. Structural and functional synaptic changes are a recurring theme with LRRK2 mutations32–34. Accordingly, impaired DA transmission is observed with several LRRK2 mouse models including knock-in (KI) mouse lines expressing the most common LRRK2 mutation (G2019S) at physiological levels35–37, suggestive of functional synaptic deficits in dopaminergic circuits in the absence of overt DA neuron loss. Thus, LRRK2 KI mouse models are valuable for investigating early PD mechanisms in vulnerable DA neurons. However, despite the clinical relevance, the cell-autonomous functions of LRRK2 in nigral DA neurons are largely unknown.
Single-nuclear RNA sequencing (snRNA-seq) offers an avenue to begin to understand downstream transcriptomic alterations within DA neuron subtypes. We have used snRNA-seq to surveil DA neurons, which allows greater acquisition of DA numbers, and minimizes isolation bias25. Here, we extend previous studies by optimizing our isolation protocol, enabling sequencing of large numbers of DA nuclei. We provide a high-resolution view of DA subtypes and an online portal to interrogate this dataset. We systematically map DA subtype distribution using MERFISH, providing unprecedented spatial resolution. Finally, we determine the stability of these subtypes in a preclinical mouse model of PD, LRRK2G2019S, and reveal molecular pathways that are altered in locomotion-relevant DA neuron subtypes.
Results
A large snRNA-seq dataset of midbrain dopaminergic neurons reveals novel molecular subtypes
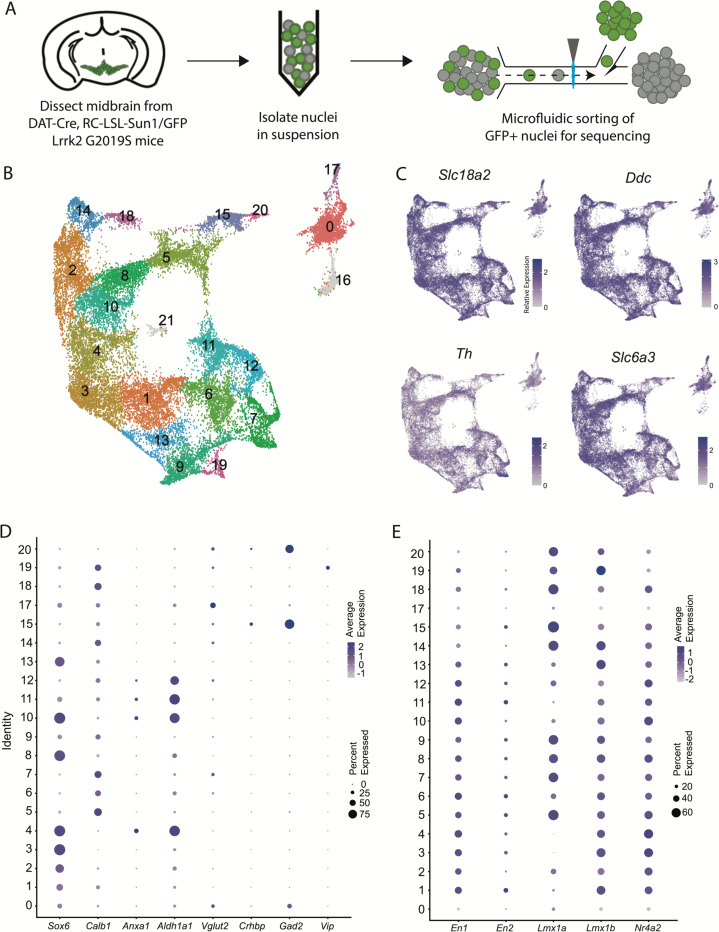
We performed snRNA-seq from dopaminergic nuclei (defined by Dat-Cre expression) in LRRK2G2019S heterozygous mutants and control littermates using a similar technique to our previously published dataset25 but modified to utilize a chip-based microfluidic sorting method which minimizes nuclear stress. This improved protocol enabled increased yield and sample quality (Fig 1A, Fig S1A). Doing so enabled us to generate a dataset with over 28,500 profiled cells, after quality control filtering, with a median UMI count of 7750.5 and median of 3056 genes. Downstream clustering resulted in 22 unique clusters (Fig 1B), of which two clusters (16 and 21) were determined to represent possible dopaminergic/glial doublets based on co-expression of glial markers in these populations and were excluded from downstream analyses (shown in grey in Fig 1B, Fig S1F). The 20 remaining clusters all showed expression of key pan-dopaminergic markers including Slc18a2 (Vmat2), Ddc, Th, and Slc6a3 (Dat) (Fig 1C). These clusters were observed in both males and females, and genotypes, in similar proportions (Fig S1B-E). These subtypes showed distinct expression signatures of many previously described markers of DA neuron subtypes (Fig 1D)17. However, our increased yield and sample quality also allowed explicit detection of known populations that have eluded most prior single-cell studies, such as DA neurons expressing Vip17, 38, thereby demonstrating the strength of this pipeline in detecting rare DA neuron subtypes. Importantly, all the described subtypes showed high expression of midbrain floorplate markers (Fig 1E), with exception of clusters 0 and 17, which possibly represent Slc6a3+ neurons in the supramammillary and/or premamillary regions39, 40. Thus, our increased number of subtypes over prior studies represents increased granularity among classic midbrain DA neuron subtypes, rather than detection of non-floorplate derived populations that express DA markers.
Figure 1.
Generation of a large RNAseq dataset of midbrain dopamine neurons. A) Schematic of single nucleus isolation pipeline using microfluidic chip-based sorting from n=8 mice. B) UMAP representation of clusters. Of note, putative clusters 16 and 21 are shown in grey, and are removed from analyses due to coexpression of glial markers. C) Expression of the defining midbrain dopaminergic neuron markers vesicular monoamine transporter 2 (Vmat2; Slc18a2), DOPA decarboxylase (Ddc), tyrosine hydroxylase (Th), and dopamine transporter (Dat; Slc6a3). Robust expression is observed throughout all clusters. D) Dot plot showing expression of several markers of previously described dopamine neuron subtypes, consistent with finding additional heterogeneity within established subtypes. E) Expression patterns of midbrain floorplate markers. All clusters show robust expression with exception of 0 and 17, which indicate these populations are not midbrain DA neurons but might be located in nearby hypothalamic nuclei.
Analysis of differentially expressed genes reveals families of subtypes and relationships to prior studies
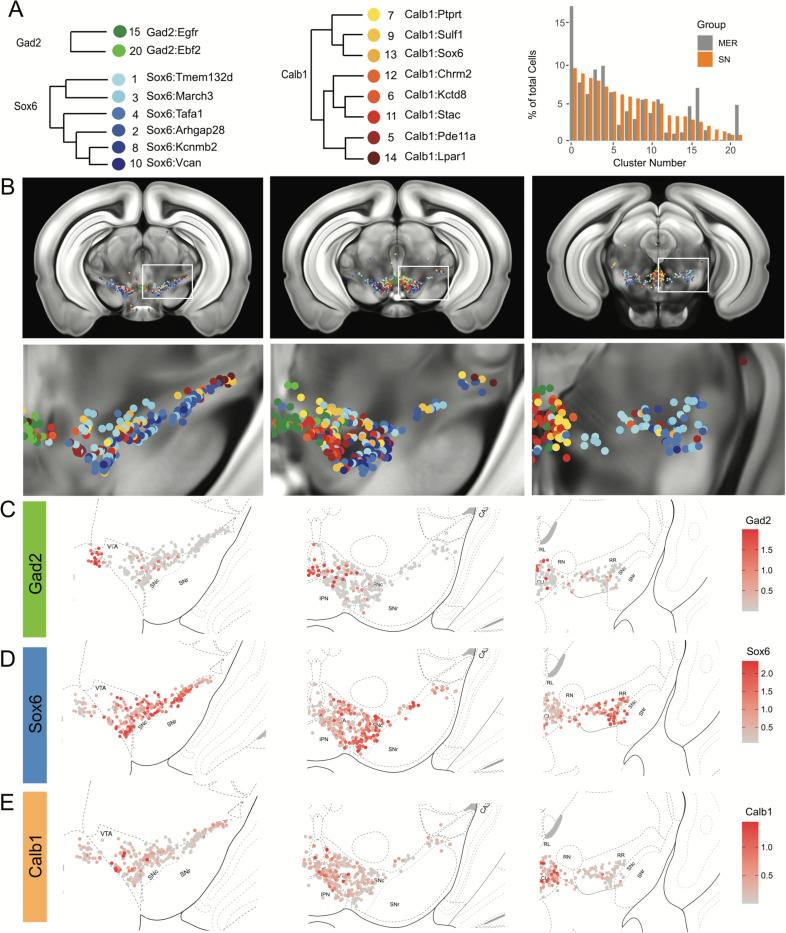
Given the large number of clusters discovered in our dataset, we first sought to organize these subtypes into groups in an unbiased manner. By creating a cluster dendrogram (Fig 2A), we were able to map an approximation of the relative relatedness of different subtypes, which segregated into three main families based on major branch points. By running differential expression on each node of the cluster dendrogram (i.e., exploring differentially expressed genes (DEGs) between the two arms emerging from a given node), we were then able to create a stepwise expression code that can uniquely define any individual cluster (Fig 2A, G). The earliest division point among our clusters was defined by genes Prkcd and Lef1, which are highly expressed only in cluster 17 (Log2 fold changes = 6.279 and 7.246 respectively, BH-corrected p values ≈ 0). Next, a family of clusters defined by Gad2/Kctd16 (deemed Gad2 family, Log2 fold changes = 5.318 and 4.014 respectively, BH-corrected p values ≈ 0) separated from all other clusters, which expressed significantly higher levels of Ddc and Slc6a3 (Log2 fold changes = 1.419 and 1.643 respectively, BH-corrected p values ≈ 0). The first major branch point between Ddc-high/Slc6a3-high clusters was then defined by cells expressing Sox6/Col25a1 (Log2 fold changes = 2.720 and 2.305 respectively, BH-corrected p values ≈ 0) versus those expressing Calb1/Ndst3 (Log2 fold changes = 2.025 and 3.368 respectively, BH-corrected p values ≈ 0), thus creating the Sox6 family and Calb1 families respectively. This division between Sox6 and Calb1 is consistent with our previous work38, as well as a prior large single cell profiling study that found this division to be the central branch point among DA neurons across several species including humans 41. Cluster division can be seen by visualizing coexpression of the DEGs, taken from the branch points labeled in red (Fig 2C-F), on the UMAP plot. Of note, while these branch points markers describe the overall trends of gene expression, these genes are not perfectly distinct or universal among the members of these cluster families. For example, clusters 1, 11 and 13 show expression of both Sox6 and Calb1 (Fig 1D). The co-expression of these markers has previously been described in a fraction of mouse DA neurons38, 42, 43. Furthermore, because the calculations of differential expression at each node take into account only the descendants of that branch point, markers defining a branch point may also be strongly expressed in clusters outside this comparison. Thus, while the final marker(s) listed for each subtype in Fig 2A are not unique to that population, following the sequential branches leading to this marker in a stepwise fashion will specifically define this population (Fig 2G). By doing so, we provide a strategy for potential genetic access to any individual DA subtype using intersectional logic gates.
Figure 2.
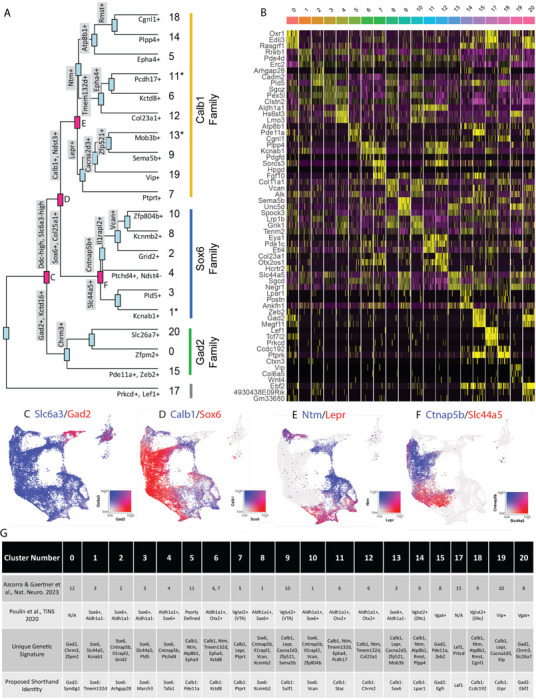
Mapping subtype identities, marker genes, and relationships to previously described populations. A) Dendrogram of DA neuron subtypes in our dataset. Distance between branch points represent approximations of relatedness between subsequent nodes, and genes labeled at each branch point represent the top differentially expressed genes distinguishing the downstream groups. Subtypes fell largely into three “families,” with expression patterns generally defined by Gad2, Sox6, or Calb1. Notably, clusters 1, 11, and 13 are shown with an asterisk as these do not display differential expression in line with the defining genes of their cluster families (i.e. are not significantly enriched for their eponymous family genes). B) Heatmap of top differentially expression genes for each cluster. The top 3 genes for each cluster are shown. C-F) Coexpression of genes at specified branch points denoted in 2A. G) Table describing putative clusters and relation to previous literature. Top row: cluster number. Second row: Equivalent cluster in Azcorra & Gaertner et al25. Third row: stepwise genetic signatures based on following sequential branch points of cluster dendrogram. Bottom row: Proposed shorthand name for subtypes, based on cluster family and top defining genes for that cluster within each family.
To enable comparisons of DA populations across existing and future literature, we next sought to better define genetic signatures for each of the individual putative subtypes and to provide a common language for discussing these populations. To do so, we utilized two complementary approaches. First, we explored the top differentially expressed genes with positive relative expression in each cluster (Fig 2B). While many of these markers are expressed in more than one population, some (e.g. Vip) are almost entirely unique to a single cluster. As a complementary approach, we performed differential expression among clusters within each of our three subtype families, thereby creating a shorthand identity based on the cluster family and top DEGs (Fig 2G). Within the Sox6 family, these clusters are Sox6 Tmem132d (listed as cluster 1 in Fig 1 and 2), Sox6Arhgap28 (cluster 2), Sox6March3 (cluster 3), Sox6Tafa1 (cluster 4), Sox6Kcnmb2 (cluster 8), and Sox6Vcan (cluster 10); in the Calb1 family, these are Calb1Pde11a (cluster 5), Calb1Kctd8 (cluster 6), Calb1Ptprt (cluster 7), Calb1Sulf1 (cluster 9), Calb1Stac (cluster 11), Calb1Chrm2 (cluster 12), Calb1Sox6 (cluster 13), Calb1Lpar1 (cluster 14), Calb1Ccdc192 (cluster 18), Calb1Gipr (cluster 19); in the Gad2 family, these are Gad2Syndig1 (cluster 0), Gad2Egfr (cluster 15), and Gad2Ebf2 (cluster 20). Cluster 17 (defined by Lef1) did not have an associated cluster family (Fig 2A). These shorthand identities provide a simple nomenclature for our clusters, which we have utilized throughout the remainder of this paper.
After establishing genetic signatures for each putative subtype, we were next able to correlate our cluster identities to those described in another recent study25, as well as the consensus subtypes proposed in Poulin et al, 2020 (Fig 2G)17. Doing so provides context for discussing our putative DA neuron subtypes with regards to prior literature. For example, cluster Sox6Tafa1 was found to be equivalent to a population we previously defined by high expression of Anxa1, which hold notably distinct functional and anatomical properties25. Several of the subtypes that emerged in our new dataset also appear to be novel divisions within clusters that previously showed evidence of internal heterogeneity based on their cluster stability metrics25. For instance, a Vip+ cluster (Calb1Gipr) emerged from within a highly variable cluster (defined as #10 in 25), as well as additional subdivisions emerged from within Sox6+ clusters (most notably #3 described in 25) (Fig 2G). This emergence of unique populations, such as Calb1Gipr, is likely due to the increased yield and sample quality of our new dataset which has enabled better detection of small populations and gene markers with lower expression level. This is further supported by the high cluster stability of the new Calb1Gipr (Fig S2A,B), resolving the instability of the equivalent cluster in Azcorra & Gaertner et al25. We reclustered our snRNA-seq data into the clusters defined in Azcorra & Gaertner et al. and found our clusters aligned with or further subdivided many of the previously defined clusters (Fig S3A). Lastly, to further corroborate our cluster identities, we used an independent bioinformatic platform, Scanpy, to re-cluster our nuclei (Fig S3B). Comparing the Seurat and Scanpy clusters using a Sankey diagram (Fig S3C) shows a high correlation between the Seurat and Scanpy clusters.
Mapping the spatial location of midbrain DA neurons using MERFISH
Having identified multiple clusters of midbrain DA neurons, we next resolved to determine the spatial distribution of these populations using MERFISH44, a form of spatial transcriptomics that offers subcellular resolution imaging-based RNA quantification. We assembled a 500 gene panel which included 120 of the top DEGs identified within the snRNA-seq dataset, as well as 380 general markers of neuronal and non-neuronal cell populations (Supp Table 1). Seven coronal slices were collected from adult mouse midbrains and processed for MERFISH (Fig 3A). The coronal slices were selected containing ventral midbrain regions between −2.9mm to −3.9mm from bregma. Cells were segmented using the CellPose algorithm based on DAPI and Poly-T staining and passed through quality control filters to remove cells with too large of a volume (potential doublets) or too few transcripts detected (false cells; Fig S4A-C). This resulted in 429,713 identified cells (Fig 3B). Cells from the seven sections displayed significantly correlated gene expression and minimal batch effects (Fig S4D). Cells were integrated in Seurat using reciprocal principal component analysis (RPCA) algorithm and then clustered, resulting in 40 final clusters comprising distinct classes of neurons, oligodendrocytes, oligodendrocyte precursor cells (OPCs), astrocytes, microglia and meningeal cells (Fig 3B). These clusters could be attributed to 25 functional groups based on the expression of cell type markers from Supp. Table 1 and their spatial distribution. Glial cell associated genes were mostly contained within their expected cell type: oligodendrocytes (Mog, Cnp), OPC (Pgfra, Olig2), astrocytes (Aqp4, Gfap), and microglia (Aif1, Tmem119). However, we noted the unexpected distribution of the oligodendrocyte-specific transcript Mbp in non-glial cells which we believe might indicate the presence of this mRNA in glial-processes that overlap non-glial cells45. Within the neuronal class we identified 16 excitatory clusters (presence of Slc17a6 or Slc17a7), 7 inhibitory clusters (presence of Slc32a1, Gad1 or Gad2), one serotonergic cluster and one DAergic cluster (Fig 3B-D, S4G). Though our gene panel was designed to identify neuronal heterogeneity of the midbrain, it offered the resolution to delineate 7 excitatory neuron classes present in distinct layers of the cortex and 6 cortical interneuron classes (Fig S4E,F). Cortical excitatory neurons were absent from subcortical structures such as thalamus, hypothalamus, midbrain and hindbrain. This fundamental dichotomy between cortical and subcortical neurons in the adult mouse brain is supported by recent spatial transcriptomic studies46–48.
Figure 3.
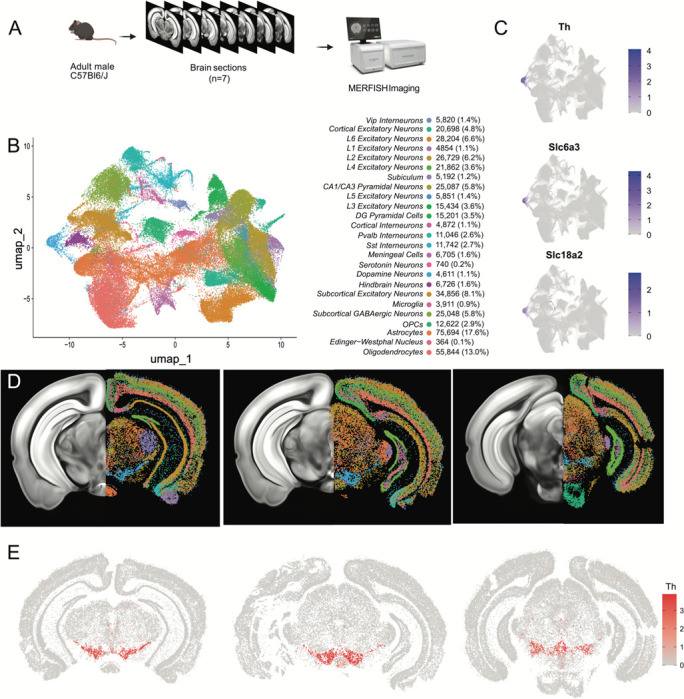
Identification of dopamine neurons in MERFISH. A) Schematic showing the processing and imaging of brain tissue with MERFISH. B) Clustering and annotation of all cells identified by MERFISH. C) Relative expression of Th, Slc6a3 and Slc18a2 shows the presence of a single cluster (blue cluster in B) that expresses all three genes. D) Spatial location of neuronal clusters for whole brain MERFISH along three rostral-caudal delineations associated with −3.0, −3.2 and −3.6mm Bregma. E) Cellular expression of Th in the sections shown in D.
A population of DA neurons was identified among our 40 clusters based on the expression Th, Slc6a3 and Slc18a2 (Fig 3C). The location of the dopaminergic cluster (blue in Fig 3D) considerably overlaps with Th expression (Fig 3E). This cluster comprised 4,532 cells, was entirely located in the midbrain, and was also enriched for Ddc, Dlk1, and Drd2 (not shown). Within this population, we detected expression of genes associated with glutamatergic and GABAergic neurotransmission, in line with previous reports of multi-lingual characteristics of DA neurons49–54, for instance the glutamatergic marker Slc17a6 (Vglut2) and GABAergic markers Gad2 and Slc32a1 (Vgat) (Fig S4G). Markers for cholinergic (Chat) and serotonergic (Tph2) neurotransmission were not detected in putative DA neurons and the glutamatergic transporters Slc17a7 (Vglut1) and Slc17a8 (Vglut3) were also absent. We next wanted to characterize the neuroanatomical location of this DAergic cluster. We manually delineated neuroanatomical boundaries on each of the slices for seven midbrain regions known to contain most dopamine neurons including SNc, VTA, retrorubral area (RR), caudal linear nucleus (CLi), interfascicular nucleus (IF), rostral linear nucleus (RLi), and periaqueductal grey (PAG). These boundaries were delineated based on cell density (dapi), white maker landmarks, and Th transcripts distribution using the Allen Brain Atlas as a reference (Fig S5A). We found that cells in our DA neuron cluster were located in the following locations: 35.2% in the SNc, 19.8% in the VTA, 7.8% in the CLi, 15.3% in the RR, 2.0% in the IF, 0.6% in the RLi, 4% in the PAG and 15.3% located elsewhere (Fig S5B).
Finally, to explore the diversity of DAergic neurons identified by MERFISH we subclustered the 4,532 cells to resolve 12 distinct subgroups (Fig S5C-F). For clarity, we are designating the clusters identified solely with MERFISH transcript levels as MER-0 to 11. Many of the clusters were characterized by increased expression of known subtype markers such as Calb1 (MER-6, MER-8 and MER-10), Aldh1a1 (MER-5), Ndnf (MER-0), Gad2 (MER-7 and MER-9) and Slc17a6 (MER-2).
Cell label transfer allows the mapping of snRNA-seq clusters
To provide the spatial distribution of the clusters identified using snRNA-seq, we sought to project the MERFISH dataset onto the snRNA-seq UMAP space. We first compared the expression level of the 500 genes utilized in our panel to select features for integration with comparable expression levels. We excluded 128 genes based on the deviation of average counts per cell within the datasets, potentially resulting from faulty probes or improper assignment of RNA transcripts to segmented cells. For instance, Sox6 transcripts were not detected across the entire brain (data not shown). We successfully projected the MERFISH dataset onto the snRNA-seq UMAP space and identified 2,297 dopamine neurons with a cell similarity score > 0.5 increasing confidence in the correct assignment to snRNA-seq defined clusters. Almost all clusters were represented by more than 20 cells in our MERFISH dataset (Fig. 4A and S6A). Clusters Calb1Ccdc192 and Calb1Gipr were not resolved in this label transfer either due to their relatively smaller number or location in an under-sampled region. Indeed, the Vip-expressing Calb1Gipr has been shown to be located in the caudal midbrain (PAG/DR)38, a region not well covered in our MERFISH experiment. Comparing snRNA-seq clusters with the ones obtained by clustering the MERFISH dataset alone showed a strong correspondence (Fig S6B). Half of the snRNA-seq clusters were predominantly comprised of single MERFISH clusters (10/20) and 25% (5/20) were comprised of 2 MERFISH clusters, demonstrating that the integration with the snRNA-seq dataset allowed us to further refine our MERFISH-based classification (Fig S6B). We then imputed expression data from the snRNA-seq dataset onto the MERFISH cells to visualize whole genome expression levels. The imputed data across the genes used for data integration correlated strongly with MERFISH transcripts as shown for Calb1, Gad2 and Aldh1a1 genes (Fig S6C-E). Altogether, the two datasets integration allowed us to map the neuroanatomical location of 18 of the clusters identified by snRNA-seq as well as impute expression of the whole transcriptome.
Figure 4.
Mapping of MERFISH data with snRNA-seq clustering: A) Grouping and recoloring of snRNA-seq clusters into distinct families represented by Gad2, Sox6 and Calb1 (Left). Relative proportion of dopamine neurons comprising each cluster from the snRNA-seq (SN, orange) and MERFISH label transfer (MER, grey) data (Right). B) Spatial representation of predicted clustering across the rostral-caudal axis, zoom of one representative hemisphere shown below. C-E) Imputed cellular expression of Gad2, Calb1 and Sox6 across the rostral-caudal axis.
Locating DA neuron clusters within the midbrain
Clusters were sorted into Gad2, Sox6 and Calb1 families based on our hierarchical dendrogram (Fig 4A; Gad2 n=124 cells, Sox6 n=887 cells, Calb1 n=590 cells). Imputed Gad2, Sox6 and Calb1 gene expression within DA neurons showed a distinct medio-lateral and rostro-caudal distribution (Fig 4C-E). The spatial distribution of imputed Sox6 and Calb1 transcripts within DA neurons correlated well with previous reports38,43,55 whereas the distribution of the Gad2 family is predominantly observed in the rostral linear/posterior hypothalamus region and CLi, and to a lesser extent in the VTA (Fig 4B, S7). Each family was overrepresented in a neuroanatomically-defined area; for instance the SNc is composed of 78.4% of neurons from the Sox6 family (Fig S7A). However, each family was also found across multiple neuroanatomical regions (Fig S7A). We also observed spatial heterogeneity within families, for example, one branch of the Calb1 family dendrogram (Fig 4, label in shades of yellow-orange defined by higher Lepr expression) was enriched in the dorsal VTA whereas the other Calb1 branch (Fig 4, label in shades of red defined by higher Ntm expression) populated mostly the ventromedial VTA. Similar observations could be made for the Sox6 family, where subtypes displayed biased distributions. This led us to explore the distribution of each individual cluster and representative markers toward the goal of generating a granular map of the DAergic system.
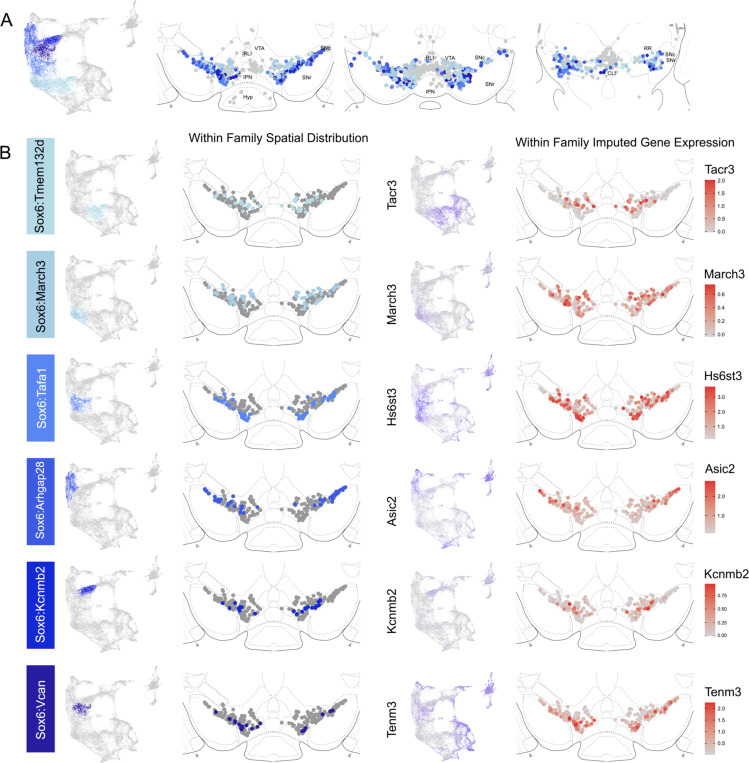
The hierarchical clustering of the Sox6 family reveals two major branches (Fig 2A and 4A): 1) Branch 1 defined by higher levels of Slc44a5 and composed of subtypes Sox6Tmem132d and Sox6March3 and 2) Branch 2 defined by Cntnap5 and composed of subtypes Sox6Tafa1, Sox6Arhgap28, Sox6Kcnmb2 and Sox6Vcan. We observed an important discrepancy between these two branches, with branch 1 being located more dorsal and medial than branch 2 (Fig 5A). Interestingly, this dorsoventral organization breaks down in the caudal SNc and RR where neurons from both branches are mostly intermixed. Only minor differences in location were observed between clusters of Branch 1, maybe reflecting their molecular similarities. Of note, whereas Sox6Tmem132d is predominant in the rostral SNc and VTA for this branch, neurons of Sox6March3 are more numerous in the RR (Fig 5B, Fig S7B). Within Branch 2, the molecular identity correlated with the medio-lateral distribution. For instance, neurons of cluster Sox6Arhgap28 were found to be more lateral in the SNc compared to neurons of Sox6Tafa1, Sox6Kcnmb2 or Sox6Vcan. This medio-lateral distribution of neurons of the SNc ventral tier might reflect the topographical nigrostriatal projections previously reported26.
Figure 5.
Spatial localization of Sox6 family of DA neurons. A) Distribution of Sox6 family DA neuron subtypes relative to all other DA neurons along the rostral-caudal axis. DA cells belonging to the Calb1 and Gad2 family are colored in light grey. B) Location of individual subtype within the Sox6 family (left two panels) with other cells in the Sox6 family shown in dark grey. Relative cellular expression of genetic markers associated with each subfamily shown in a UMAP and a midbrain section.
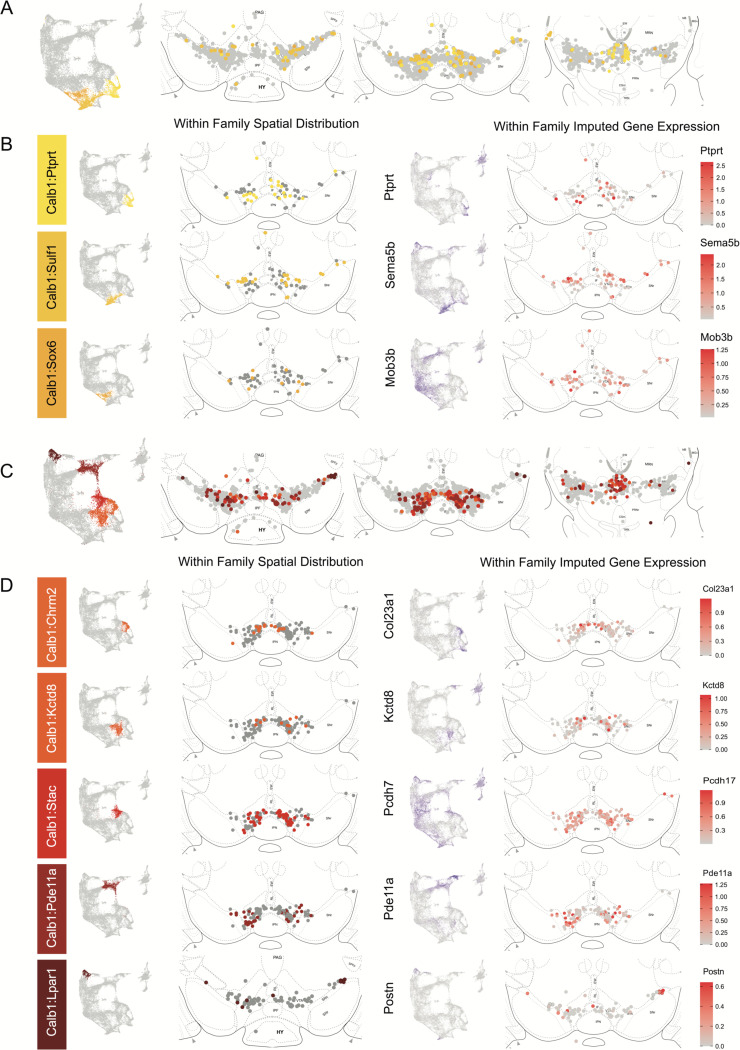
The Calb1 family was the most molecularly diverse which was also reflected in its localization (Fig 6). Two major branches of Calb1 DA neurons were identified: 1) Branch #1 (Fig 6A-B) containing subtypes Calb1Ptprt, Calb1Sulf1, and Calb1Sox6 and 2) Branch #2 (Fig 6C-D) containing subtypes Calb1Chrm2, Calb1Kctd8, Calb1Stac, Calb1Pde11a and Calb1Lpar1. Broadly speaking, Calb1 subtypes were predominantly localized to the VTA and CLi with some neurons showing localization to the dorsal SNc, SNpl, or RR (Fig S7). Within the VTA, Branch #1 Calb1 neurons tended to localize to the dorsal VTA (Fig 6A-B) while Branch #2 Calb1 neurons showed distinct ventromedial localization (Fig 6C-D). In more caudal sections the difference was even more pronounced, although intermingling was always observed. Within branch #1, subtype Calb1Ptprt and Calb1Sox6 were largely absent from the SNc dorsal tier and RR, whereas subtype Calb1Sulf1 was present. Some notable distinctions were observed in both groups, specifically subtype Calb1Sulf1 and Calb1Lpar1 which were each observed in significant numbers in the dorsolateral SNc/SNpl region. Of these, only Calb1Lpar1 expressed significant Slc17a6 (data not shown), making it a likely candidate for the SNpl Vglut2+ DA neuron population found to innervate the tail of the striatum27. Indeed, these laterally located neurons have been genetically targeted with a Slc17a6-Cre driver, and display deceleration correlated responses, and robust responses to physically aversive stimuli25, 27.
Figure 6.
Spatial localization of Calb1 family DA neurons. A) Calb1 family of neurons were divided into two distinct branches based on hierarchical clustering. Distribution of Branch 1 of Calb1 family DA neurons along the rostral-caudal axis with all other DA cells colored in grey. B) Location of individual Calb1-Branch1 clusters within the Calb1-Branch1 family. Only cells in the Calb1-Branch1 family are shown. Relative cellular expression of genetic markers associated with each subtype shown in a UMAP and brain section. C) and D) Same as A and B, but for Branch 2 of Calb1 family DA neurons. Relative cellular expression of genetic markers associated with each subfamily shown in a UMAP and a midbrain section.
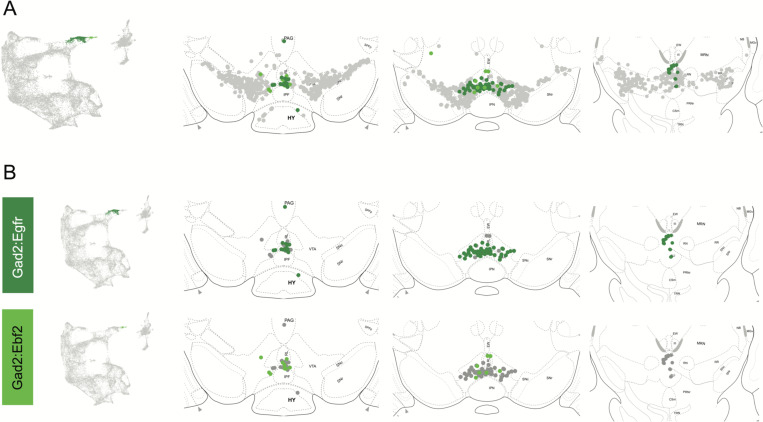
Within the Gad2 family we were able to resolve two subtypes. Subtypes Gad2Egfr and Gad2Ebf2 had very similar spatial distributions and were prominent in the posterior IF, RLi, and CLi (Fig 7, Fig S7). These regions have been associated with small DA neurons of lower Th and Slc6a3 levels. Subtype Gad2Egfr neurons were more plentiful than cluster Gad2Ebf2, with the laker being absent in more caudal sections. All Gad2 subtypes expressed Slc32a1, opening the possibility that these neurons co-release GABA. Further, they also expressed Slc17a6, revealing a potentially multilingual DA neuron subtype.
Figure 7.
Spatial location of Gad2 family DA neurons. A) Distribution of Gad2 family DA neurons along the rostral-caudal axis with other DA cells colored in grey. B) Location of individual subtypes within the Gad2 family with only cells of the Gad2 family shown in grey.
LRRK2G2019S KI results in pan-dopaminergic gene expression changes consistent with deficits observed in this model, without altering DA subtype proportions
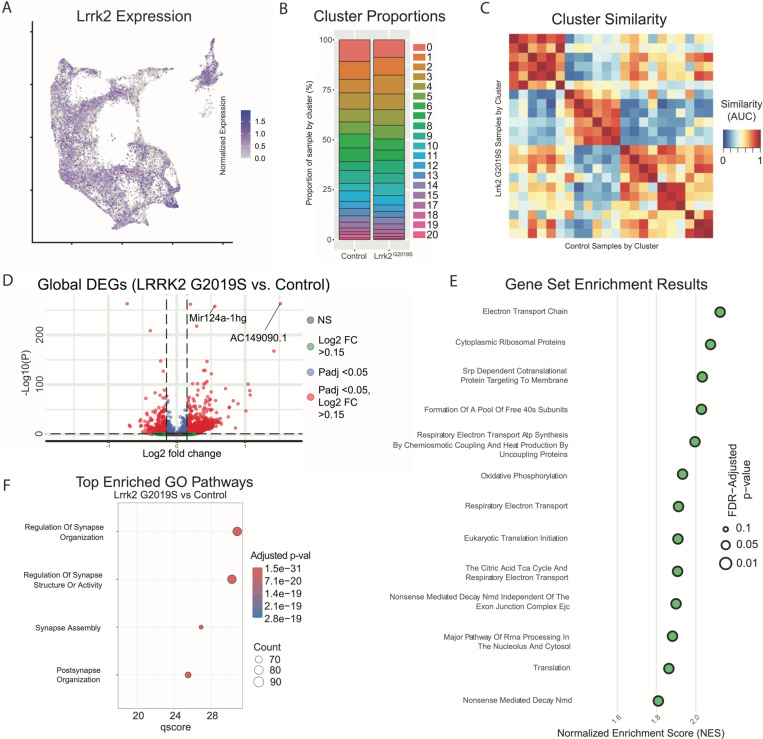
With cluster identities and locations defined, we next sought to explore the downstream gene expression characteristics of DA neurons in Lrrk2 mutants. The role of Lrrk2 in PD pathophysiology remains unclear56, and Lrrk2 mutations are often proposed as being an indirect source of dysfunction for the DA system, such as through its high expression in glial or striatal cells34, 57–60. However, previous studies have shown alterations in DA signaling or gene expression even when the mutant LRRK2 was expressed specifically in DA neurons61, 62. Thus, as a first step in demonstrating the possibility that mutant Lrrk2 in DA neurons themselves can contribute to dysfunction, we mapped the expression of Lrrk2 across all DA neuron subtypes (Fig 8A). We found detectable expression across most subtypes, with the notable exception of Gad2Egfr neurons.
Figure 8.
Global effects of Lrrk2G2019S mutation are observed across all clusters. A) Expression pattern of Lrrk2 RNA, which is present in all clusters, though notably low in Gad2Egfr (cluster 15). B) Cluster proportions in control and mutant samples. Relative proportions of each subtype within the dataset are relatively unchanged. C) Cluster similarity heatmap shows that each cluster from control samples is more similar to its corresponding cluster in the mutant samples, suggesting there is no large-scale change in cell type across samples. D) Volcano plot of differentially expressed genes across conditions. Each point is colored according to its significance and magnitude of fold change. E) Gene set enrichment analysis (GSEA) results comparing all clusters across conditions. Several top enriched pathways in Lrrk2 mutants are related to mitochondrial energy production/oxidative phosphorylation. F) Gene ontology (GO) results comparing all clusters across conditions. Top enriched pathways are related to synapse organization and function, consistent with previously described synaptic dysfunction in Lrrk2G2019S mutant mice.
Given the distinctive properties of different DA neuron subtypes, we next sought to test if phenotypic changes in Lrrk2 mutant mice are driven by changes in the relative proportions of said subtypes. While previous studies have shown no overt DA neuron loss, it is possible that there was a selective reduction of a subtype(s) that would be undetectable by previous methods. We first graphed the proportions of each subtype within our control and mutant datasets (Fig 8B), and found remarkable similarity across these samples, consistent with previous reports showing no DA neuron loss in these mice36. To further assess for changes in cell state among clusters across conditions, we next compared the similarity of each cluster to all other clusters across conditions (Fig 8C). Meta-neighbor analysis revealed that the most similar population to each wildtype cluster was its corresponding subtype in the Lrrk2 mutant samples. This suggests that the general subtype organization is impervious to LRRK2G2019S perturbation, and that our clustering scheme is more likely representative of cell type rather than cell state.
To assess pan-dopaminergic gene expression changes as a function of Lrrk2 genotype, we next looked at globally differentially expressed genes across our mutant and control samples. Calculating differentially expressed genes across all clusters revealed a large number of significantly enriched or diminished genes as a function of Lrrk2 mutation (1646 genes in total with BH corrected p-value <0.05, Wilcoxon rank sum test). The results of this differential expression analysis are plotted in Fig 8D, with full results and statistics available in Extended Data. Notably, most of these changes appear to be small in magnitude; among FDR-significant genes, only 51 showed increased expression and 7 showed decreased expression with log2 fold changes greater than 0.5 (Fig 8D). Two genes with high enrichment in Lrrk2 mutants, Mir124a-1hg and AC149090.1 were both of particular interest due to their potential relevance to PD pathogenesis. AC149090.1 is orthologous to the human PISD gene, which is involved in autophagy and implicated in mitochondrial dysfunction63–65. This gene has been proposed as a sensitive marker of biological aging in the brain63, 66, an interesting finding given that age is the single largest risk factor for PD. Also of note, Mir124a-1hg is a host gene (though not the exclusive gene for) miR-124, a microRNA with purported neuroprotective effects that has been found to be downregulated in PD patients or some PD mouse models67–70.
We next performed pathway enrichment analyses using gene set enrichment analysis (GSEA) and gene ontology (GO) to compare our Lrrk2 mutant samples to our controls. Using GSEA, we found 18 gene sets with significant differential expression via GSEA (FDR adjusted p-values < 0.05). The top 13 of these enriched pathways (those with NES scores > 1.8) are shown in Fig 8E. While a broad range of biological domains are found among these results, many of the top enriched pathways were related to energy production via mitochondrial oxidative respiration, remarkably similar to prior comparisons between vulnerable and resistant DA neuron populations43. Mitochondrial energy production pathways, and specifically the extraordinary bioenergetic burdens of DA neurons, have been heavily implicated in PD vulnerability71, 72. Thus, the enrichment of these pathways in Lrrk2 mutants is particularly intriguing as it may reflect the structural and functional alterations reported with LRRK2 mutations in patients73 and preclinical models74, 75 and further predispose these cells to dysfunction and ultimately degeneration. Utilizing GO, we again found many pathways displaying significant differential expression between Lrrk2 mutants and controls. Among these, the top pathways are all associated with synaptic organization and function (Fig 8F). This was particularly intriguing given that pre-synaptic dysfunction in Lrrk2 mutants such as endocytosis76–78 or trafficking of axonal cargo impairments79 could underlie the mechanisms for the deficits in DA signaling in these mice80, 81. Of note, decreased endocytosis was observed in DA and not in cortical or hippocampal LRRK2G2019S primary neurons82. These results, although in vitro, suggest cell type specific synaptic effects of dopamine neurons, consistent with their vulnerability in LRRK2-related PD.
Comparing individual subtypes across conditions allows insights into subtype-specific dysfunction
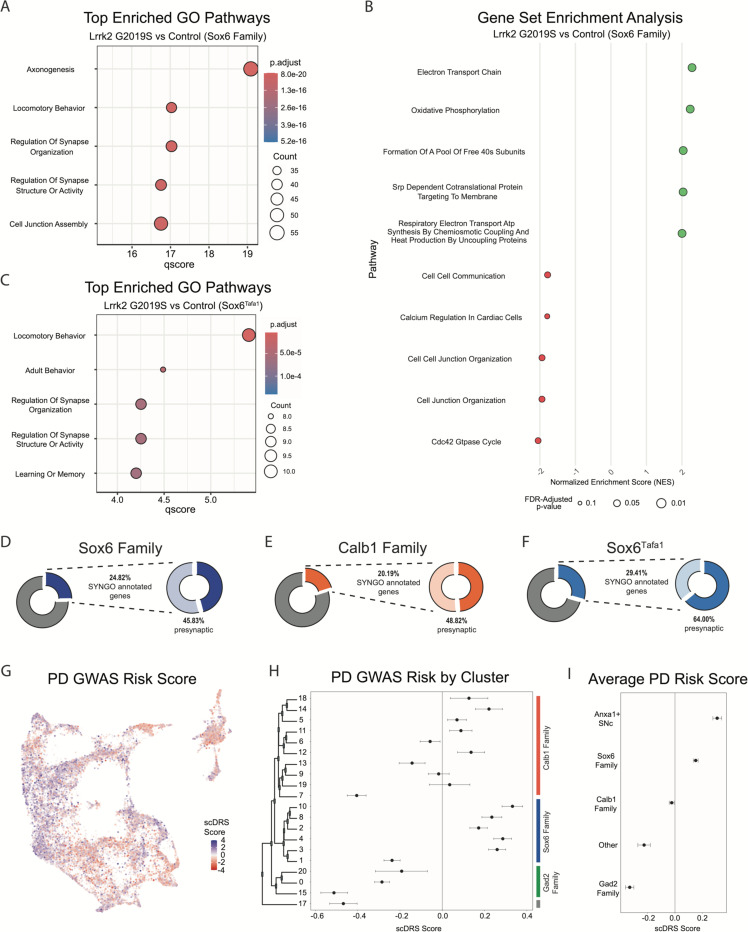
Given that global gene expression differences across our conditions appear to hold relevance for putative mechanisms of PD pathogenesis, we next sought to address the question of whether gene expression within any given subtype is specifically altered in our PD model. Although Lrrk2 mRNA seemed uniform across subtypes, downstream effects could be subtype-specific secondary to differential Lrrk2 kinase activity, distinct kinase targets, or superimposed differences in other properties of each subtype that culminate in distinct downstream transcriptional alterations. Given the proposed vulnerability of Sox6+ DA neurons and relative resilience of Calb1+ neurons in PD3, 41, 43, we first compared these two cluster families across Lrrk2 mutants versus control mice to see if any changes in DA neurons are specific to vulnerable or resistant cell types. We found 729 DEGs among the Sox6 family, and 679 among the Calb1 family (BH corrected P value <0.05). Among those genes, we found 327 DEGs were shared between the Sox6 and Calb1 families (Fig S8A). The full list of DEGs and statistics is available in Extended Data. Notably, AC149090.1 was highly significant in both cohorts, implying that the pan-DA differential expression of this gene was not driven by a particular subset of DA neurons. To further compare changes within the Sox6 family, we once again performed GSEA and GO but on these groups of clusters in isolation (Fig 9A-B). In line with our previous results, GO revealed pathways that largely corresponded to synaptic and axonal function. However, the pathway with second largest enrichment now corresponded to locomotory behavior (Fig 9A). Finally, to extend the granularity our analyses, we applied the same pipelines to one individual cluster, Sox6Tafa1, which has the highest expression of Anxa1. This SNc subtype was of particular interest to us given recent results showing that Anxa1+ ventral tier SNc neuronal activity is selectively correlated with acceleration in mice running on a treadmill, leading to the hypothesis that degeneration of these neurons may, in part, underpin PD motor dysfunction25. The top enriched GO pathway in our analysis was locomotory behavior (FDR-corrected p value = 3.91E-6), with several significant pathways once again corresponding to synaptic function.
Figure 9.
Changes in gene expression are observed in PD-implicated DA neuron subtypes in Lrrk2G2019S mice. A) GO results comparing Sox6 cluster family across conditions. Several pathways are highly relevant to locomotor behavior and previously described dysfunction (such as synaptic organization pathways) in Lrrk2G2019S mutant mice. B) GSEA results comparing Sox6 cluster family across conditions. While twenty pathways were positively enriched in mutant Sox6 family clusters (BH-corrected p-values < 0.05), several additional pathways were enriched in either direction using a less conservative cutoff of p < 0.1. The top five positively and negatively enriched pathways with this less conservative cutoff are shown. Pathway enrichment was similar to global changes, but more pronounced in the Sox6 family of clusters. C) GO results comparing the Sox6Tafa1 cluster, which showed the highest expression of Anxa1, across conditions. This population has previously been shown to selectively signal during motor acceleration and shows enrichment for motor pathways as a function of Lrrk2 genotype, which could suggest DA signaling deficits in acceleration-correlated circuits and a potential substrate for subsequent motor deficits in Lrrk2G2019S DA neurons. D) Results of SynGO analyses for Sox6 family, Calb1 family, or Sox6Tafa1 clusters. SynGO-annotated (i.e. synaptic) genes were enriched among DEGs for ventral SNc populations, particularly Sox6Tafa1. Of note, among these synaptic genes, Sox6Tafa1 cluster showed much heavier enrichment for those localized to the presynapse, which may suggest subtype-specific presynaptic function in acceleration-corrected subtypes. G) Feature plot of each cell’s association with PD risk loci proposed by GWAS as calculated by scDRS scores. Clear differences are observed across clusters. H) Mean and 95% confidence intervals for PD GWAS risk (scDRS scores) for each cluster. Sox6 family of clusters are particularly elevated. I) Average risk scores with 95% confidence intervals among cluster families, as well as clusters Sox6Tafa1 and Sox6Vcan, the two Anxa1-expressing SNc clusters, which showed the highest average risk scores.
Next, we compared the synaptic compartment and biological functions of DEGs (BH adjusted p values < 0.05) in Lrrk2G2019S mutants versus controls using SynGO, a database for systematic annotation of synaptic genes83. We focused primarily on the Sox6 and Calb1 families, but also on cluster Sox6Tafa1 given that this population (SNc neurons with highest expression of Anxa1) is specifically linked to movement25. Of the DEGs in the Sox6 family, Calb1 family, and Sox6Tafa1 cluster, 24.82%, 20.19% and 29.41% of these genes had synaptic localizations, respectively, with more significant synaptic associations among populations localized to ventral tier SNc (Fig 9D-F, Fig S8B). Further compartment categorization revealed that among these synaptic DEGs annotated by SynGO, 45.83% of Sox6 family, 64.0% of Sox6Tafa1 and 48.82% of Calb1 family DEGs have presynaptic localizations, raising the possibility of enhanced disruption of presynaptic function in acceleration-correlated DA neurons. From these presynaptic DEGs, 20/77 in the Sox6 family, 5/16in Sox6Tafa1, and 16/62 in the Calb1 family have specific annotations for active zones, which are the site of dopamine release. Annotations based on the biological functions showed that 20.83%,28.0% and 15.74% of Sox6, Sox6Tafa1 and Calb1 DEGs, respectively, are associated with processes in the presynapse (Fig S8C). Overall, greater proportions of DEGs are associated with presynaptic locations in cells from vulnerable DA neurons (Sox6 family, and in particular, Sox6Tafa1), compared to less vulnerable ones (Calb1 family).
Given the differential effects of Lrrk2 mutation across subtypes, we also sought to map the association of gene expression in our dataset with co-expression of PD-associated risk loci identified by GWAS84 using a polygenic enrichment score method, scDRS85. We first calculated a PD GWAS risk score for each individual cell, which revealed clear bias for higher risk scores among clusters that localize to the ventral SNc (Fig 9G). Calculating the mean PD GWAS risk score for each cluster and corresponding 95% confidence intervals confirmed significant risk associations in several clusters, particularly those in the Sox6 family (Fig 9H). Interestingly, the Gad2 family of clusters all showed significant negative associations with PD risk loci (Fig 9H). Of note, the highest scDRS scores were in clusters Sox6Tafa1 and Sox6Vcan, the only two SNc clusters that express Anxa1. By Calculating the average scores for each cluster family as a whole, as well as these two Anxa1-expressing clusters, we found that only the Sox6 family (and Anxa1-expressing SNc clusters within it) show significant associations with PD risk loci (Fig 9I).
Finally, due to the large number of potential comparisons to be made across clusters, cluster families, and genotypes, we have also developed an online tool that allows for exploration of our datasets, which we have termed Dopabase (URL: https://awatramanilab.shinyapps.io/shinyapp-2/). Utilizing this tool, users have the ability to access many analysis and plotting functions to continue to explore populations of interest.
Discussion
Our work provides five key advances. 1. We provide a large snRNAseq dataset that provides high granularity to our taxonomic schemes. 2. We provide a user-friendly portal to query this dataset. 3. We plot the spatial location of most identified DA subtypes, showing heterogeneity even within sub-domains in traditional anatomical areas. 4. We demonstrate that cluster proportions do not change in LRRK2G2019S mice, although gene expression changes are observed, most notably in mitochondrial bioenergetics and synapse organization pathways in vulnerable DA subtypes. 5. PD GWAS risk is most prominent in Anxa1 SNc neurons.
We demonstrate the presence of 20 molecularly distinct clusters of midbrain DA neurons. These are hierarchically organized, the dendrogram being split into three levels, which we refer to as family, branch and subtype. Family (Level 1) distinctions are driven by the expression of Gad2/Kctd16 vs Slc6a3/Ddc high expressing cells. Slc6a3/Ddc high cells are further divided by Sox6/Col25a1 and Calb1/Ndst3 (Level 2) among other genes. It is likely that these fundamental divisions are, at least in part, set early in development in the mesodiencephalic floor plate, where Sox6 demarcates progenitors with a substantially higher probability of ventral SNc and lateral VTA fate, and conversely, Sox6- progenitors have a higher probability of a Calb1+ fate43, 55. The Sox6 family is split into two branches with six subtypes, the Calb1 family is split into two branches with ten subtypes, whereas the Gad2 family can be further split into two subtypes. This scheme represents a more granular taxonomy than previous studies19–21, 23, 25, 38, 41, and bears some resemblance to a recent study86. Key congruencies between these studies include: 1) parsing midbrain DAergic system into broad groups of subtypes based on expression of Sox6 and Calb1, and to a lesser extent Gad2, populations, 2) greater diversity observed in Calb1+ clusters, 3) the identification of some consensus and homogenous subtypes. Key discrepancies include the extent to which each cluster further subdivided and the final number and molecular signature of DA subtypes. In the laker study86, they refer to levels as territories and neighborhoods. For simplicity of communication, we refer to the lowest level of our dendrogram of molecularly defined DA neuron clusters, as subtypes. However, we acknowledge that because of the common developmental origin from the floor plate87–90, their high relatedness, the blurriness of boundaries between clusters, and the polythetic nature of clustering, referring to these groups as neighborhoods is an alternative and reasonable possibility. Notwithstanding these considerations, defining markers of some of these clusters and generating Cre/Flp lines, may be useful to at very least enrich for DA neuron populations with distinctive properties as has recently been shown for the Anxa1+ SNc neurons, which at a population level are selectively correlated with acceleration, while reward responses are almost entirely absent.
Our spatial MERFISH analysis provides the most granular representation of DA subtype locations to date. This resolution was obtained by the integration of snRNA-Seq and MERFISH datasets. Our data complement current spatial transcriptomic datasets46–48. For instance, using MERFISH, Zishen et al. identified 10 cluster in the VTA (A10), 9 clusters in the SNc (A9), and 2 clusters in the RR although little detail on the molecular identity or location of these subtypes is provided. In addition, Langlieb et al 48, provide a molecular atlas of the adult mouse brain in which they identified 13 clusters of midbrain DA neurons and located these using Slide-Seq. By contrast, Salmani et al.86 used established markers to locate seven DA neuron territories and 16 neighborhoods. They report a distribution of Gad2+ cells in the midline including CLi, RLi and IF, which bears similarity with our Gad2 family. They also Pdia5+/Calb1+ DA neurons in the pars lateralis, likely representing an overlapping population with our Calb1Postn and Calb1Sulf1. Within the SNc, our data fits also well with the location of DA neurons that could be labeled with Anxa1-Cre, although here we show that in our Seurat based clustering, Anxa1 expression is detected in Sox6Tafa1 and to a lesser extent in Sox6Vcan cells. Taken together, with our increasing appreciation of DA neuron heterogeneity, a consensus is bound to arise on both the identity and location of DA neuron subtypes. IntegraRon of all these emerging datasets will be necessary to achieve a comprehensive DAergic neuron taxonomy that will ultimately encompass all morphological, connectional, physiological, molecular, and neuroanatomical parameters.
A key finding of this study is the heterogeneity observed even within sub-domains of the traditional anatomical clusters. An example of this is the lateral SNc/SNpl. Current models depict the SN heterogeneity as a mediolateral gradient91. Our data substantially refines this by showing that even within the lateral SNc/SNpl region, there are several distinct subtypes. One interesting finding is that two of the lateral subtypes from distinct families (Sox6 and Calb1), show some common gene expression signatures. For example, Asic2 or Ankfn1 are highly expressed in Calb1Lpar1 and Sox6Arhgap28 subtype, both of which are laterally located. One interpretation of this is that secondary gene expression programs may have been superimposed upon the early developmental sub-divisions, for instance during circuit assembly. A second example of heterogeneity within sub-domains is in the dorsolateral VTA. Again, we find enormous heterogeneity in this region, which is unaccounted for in previous models. It is likely that neurons in this region will have vastly distinct anatomical and functional properties, as exemplified by the complex intermixing of neurons with diverging axonal projections7. Careful intersectionally based interrogation methods will need to be developed to distinguish these populations.
We address whether our clustering schemes are more reflective of cell types or cell states. We show that the proportions and definitions of subtypes are consistent between controls and mutants, and that mutant subtypes are most closely related to their control counterpart. This suggests that our taxonomic scheme is impervious to the context of a mild perturbation such as LRRK2G2019S, suggesting that our clusters are reflective of cell types, rather than cell states. It is likely, that with more severe perturbations, such as a toxin lesion, more substantial alterations of taxonomic schemes are observed86, 92. However, we expect that for mild insults, day to day behavioral changes, or pharmacological paradigms, our clusters will be resistant to changes, although individual genes may vary.
Our work describes a rich heterogeneity of DA neurons in the murine SNc that might serve as a reference for understanding patterns of degeneration in PD. The majority of SNc neurons belong to the Sox6 family, but some are from the Calb1 family. Several studies on post-mortem human PD brain have demonstrated a relative resilience of Calb1+ DA neurons43, 93, 94. Calb1+ neurons in mice SNc and SNpl project most densely to the DMS and tail of striatum (TS) respectively3, 27. This could match the patterns of axon preservation observed in post-mortem PD brain, which show relative sparing of medial, ventral and caudal regions of the caudate-putamen95, 96. Indeed it is plausible that patterns of degeneration could be associated with distinct clinical presentations of PD – a recent study suggests that patients with relatively spared caudate projections have a higher probability of tremor97. Additionally, we postulate that spared DA neurons projections may be a cellular substrate mediating the side-effects of levodopa particularly at high doses. In line with this, high DA release in the TS is associated with hallucination like behaviors in mice98.
Given the relevance of LRRK2 to genetic and idiopathic PD, we examined the effects of LRRK2 on DA neuron gene expression to start appreciating the molecular pathophysiology of relevant subpopulations in PD. We relied on a KI LRRK2 model, expressing the G2019S pathogenic mutation due to the highest prevalence of this mutation over other mutations. Although these mice do not exhibit DA neuron loss, they do have nigrostriatal synaptic alterations within a physiological range35. Our transcriptomic interrogation has three main takeaways. First, the Lrrk2 gene is expressed in most DA neurons in mice; in humans Lrrk2 transcript is enriched in the Sox6 population41. While previous work has shown the LRRK2 protein expression in midbrain DA neurons60, 99, its relative expression across DA neuron subtypes in mice requires further investigation. Thus, transcriptomic alterations observed within DA neurons are potentially cell-autonomous effects, albeit indirect, since LRRK2 functions as a protein kinase with phosphoregulation primarily affected. Second, while individual genes showed only modest differences, pathway analysis showed a strong dysregulation of synaptic pathways. This is conceptually aligned with prior work describing that LRRK2 phosphorylates various key presynaptic targets (not necessarily in DA axons) associated with SV cycle100. Impairments of these processes at the nigrostriatal terminals may contribute to the DA release deficits reported in LRRK2 mutants35–37. Third, alterations in pathways associated with oxidative phosphorylation and energy production are in line with the described LRRK2 impact on mitochondrial structure and mitophagy in the LRRK2G2019S KI mice. As DA transmission changes and mitochondrial abnormalities are the two main and consistent phenotypes with the KI mouse models, our transcriptomic findings indicate that functional changes are at least partly linked with gene expression changes.
Taken together, our work complements perfectly the recent taxonomy of mouse46–48 and human brain cell types101. Focusing our efforts on the DAergic system allowed us to reveal a comprehensive transcriptomic signature of midbrain DA neurons as well as ascribe with precision their spatial location. Moreover, we provide a web resource to explore this dataset through multiple angles. We also uncovered cell type specific transcriptomic changes in a prodromal model of PD revealing the molecular consequences of higher LRRK2 activity. Disentangling the molecular diversity of DA neurons, now defining 20 transcriptomic subtypes, raises important questions. For instance, what are the functional implications of this diversity and are these transcriptomic differences reflected in other cellular features? Decades of experiments might be needed to answer such questions, however, by defining genetic anchor points unique to each subtype, we have provided an avenue to target these populations in the mouse.
Methods
snRNASeq
Animals
All animals used in this study were maintained and cared following protocols approved by the Northwestern Animal Care and Use Committee. Cre mouse lines were maintained heterozygous by breeding to wild-type C57BL6 mice (RRID:IMSR_CRL:027), with the exception of DAT-IRES-Cre, CAG-Sun1/sfGFP, which was maintained homozygous and crossed to Lrrk2G2019S mice (RRID:IMSR_JAX:030961)only for generation of experimental mice for nuclei isolation. Both males and females were used in equal number for all RNAseq experiments.
Sample Preparation
To isolate nuclei for snRNA-seq library generation, n=8 6-month old DAT-IRES-CRE (RRID:IMSR_JAX:027178), CAG-Sun1/sfGFP (RRID:IMSR_JAX:021039), Lrrk2G2019S+/wt or littermate Lrrk2wt/wt mice (2 female control, 2 male control, 2 female mutant, 2 male mutant) were sacrificed and rapidly decapitated for extraction of brain tissue as previously described for isolation of GFP+ (i.e. dopaminergic) nuclei. A 2mm thick block of ventral midbrain tissue was dissected out from each mouse and collected for isolation. Tissue was dounce homogenized in a nuclear extraction buffer (10mM Tris, 146mM NaCl, 1mM CaCl2, 21mM MgCl2, 0.1% NP-40, 40u/mL Protector RNAse inhibitor (Roche 3335399001). Dounce homogenizer was washed with 4mL of a washing buffer (10mM Tris, 146mM NaCl, 1mM CaCl2, 21mM MgCl2, 0.01% BSA, 40U/mL Protector RNAse inhibitor) and filtered through a 30uM cell strainer. After three rounds of washing by centrifugation at 500g for 5 minutes, nuclei pellets were resuspended resuspension buffer (10mM Tris, 146mM NaCl, 1mM CaCl2, 21mM MgCl2, 2% BSA, 0.02% Tween-20) and filtered through a 20uM strainer. This nuclei suspension was loaded onto a MACSQuant Tyto HS chip and diluted with 1x PBS. Nuclei were sorted using gates set for isolation of GFP+ singlet nuclei. Sorted nuclei were subsequently used for preparation of four 10X Genomics Chromium libraries. Protocol can be found at dx.doi.org/10.17504/protocols.io.14egn6wryl5d/v1.
Library preparations were performed by the Northwestern University NUSeq Core Facility. Nuclei number and viability were first analyzed using Nexcelom Cellometer Auto2000 with AOPI fluorescent staining method. Sixteen thousand nuclei were loaded into the Chromium Controller (10X Genomics, PN-120223) on a Chromium Next GEM Chip G (10X Genomics, PN-1000120), and processed to generate single nucleus gel beads in the emulsion (GEM) according to the manufacturer’s protocol. The cDNA and library were generated using the Chromium Next GEM Single Cell 3’ Reagent Kits v3.1 (10X Genomics, PN-1000286) and Dual Index Kit TT Set A (10X Genomics, PN-1000215) according to the manufacturer’s manual with following modification: PCR cycle used for cDNA generation was 16 and the resulting PCR products was size-selected using 0.8X SPRI beads instead of 0.6X SPRI beads as stated in protocol. Quality control for constructed library was performed by Agilent Bioanalyzer High Sensitivity DNA kit (Agilent Technologies, 5067–4626) and Qubit DNA HS assay kit for qualitative and quantitative analysis, respectively.
The multiplexed libraries were pooled and sequenced on an Illumina Novaseq6000 sequencer with paired-end 50 kits using the following read length: 28 bp Read1 for cell barcode and UMI and 91 bp Read2 for transcript. Raw sequence reads were then demultiplexed and transcript reads were aligned to mm10 genome using CellRanger (v7.0.1 – RRID:SCR_017344 – https://www.10xgenomics.com/support/software/cell-ranger/latest) with --include-introns function. snRNA datasets can be found at GEO accession GSE271781.
Data curation and analysis
Outputs from CellRanger were read into Seurat version 5.0.1 using the Read10X command for each sample. Numbers of UMIs, features and ribosomal reads and mitochondrial reads were plotted for each dataset and used to determine cutoffs for quality control pre-filtering of each sample. The following QC filtering commands were used in R: control1 <- subset(control1, subset = nFeature_RNA > 1200 & nFeature_RNA < 7800 & percent.mt < 0.5 & nCount_RNA < 29000 & percent.ribo < 0.5); lrrk1 <- subset(lrrk1, subset = nFeature_RNA > 1200 & nFeature_RNA < 6500 & percent.mt < 0.5 & nCount_RNA < 26000 & percent.ribo < 0.5); control2 <- subset(control2, subset = nFeature_RNA > 800 & nFeature_RNA < 5000 & percent.mt < 0.5 & nCount_RNA < 15000 & percent.ribo < 0.5); lrrk2 <- subset(lrrk2, subset = nFeature_RNA > 1000 & nFeature_RNA < 7500 & percent.mt < 0.5 & nCount_RNA < 20000 & percent.ribo < 0.5) (post-filtering violin plots shown in Fig S1A). The male and female datasets were then normalized and integrated using the SCTransform V2 method102 in Seurat V5103. Cells were initially clustered using 35 principal components at a resolution of 0.8. Plotting clusters on UMAP showed one small cluster exceedingly distant from all other clusters, which was suspected to represent EW nucleus cells and was subsequently removed. Afterwards, the integration and clustering pipeline was repeated without these cells to remove their impact on PCA. Final clustering was achieved using the FindNeighbors() command with the following parameters: dims = 1:32, reduction = “cca”, n.trees = 500, k.param = 40, and with the FindClusters() command with the following parameters: resolution = 0.8, algorithm = 1, group.singletons = TRUE, graph.name = “SCT_snn”. UMAP reduction was calculated using the RunUMAP command with the following parameters: reduction = “cca”, dims = 1:32, reduction.name = “umap.cca”, n.epochs = 500, min.dist = .2, n.neighbors = 1000. In total, the integration resulted in a final dataset of 28532 nuclei, with a median UMI count of 7750.5 and median of 3056 features. All clusters were represented in both male and female samples (Fig S1B). Sex of original mice was inferred for each cell by Calculating the expression ratio of genes Uty and Eif relative to Tsix and Xist for each cell, revealing roughly equal proportions of male and female cells in the final dataset (Fig S1C). All clusters were represented in each individual sample (Fig S1D), and with no overt differences in proportions across conditions (Fig S1E, Fig 8C). Clusters 21 and 16 were removed from downstream analyses due to high expression of glial markers Atp1a2 and Mbp, respectively (Fig S1F). All differential expression calculations were also performed in Seurat using the FindMarkers() command; individual parameters for differential expression at each step of our analysis are provided in the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
ShinyCell web browser was created using the SCT assay of the Seurat Object described above104. The code can be found at the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
Mapping data query sets were done using TranserData() seurat function. The dataset and clusters from Azcorra & Gaertner et al. (Dataset can be found in the Gene Expression Omnibus GSE222558) 25 was used as the reference, and the query dataset was the one described in Figure 1. Any cells clustered with a prediction.max.score <.5 were removed from the data set (2.4% of cells removed). The exact workflow can be found at the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
Sankey plots were made using the Network3D package in R and the sankeyNetwork function. The ‘source’ and ‘target’ nodes are the corresponding cluster labels. ‘Value’ is set as the number of cells clustered in both the source and target nodes. The exact workflow can be found at the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
Cluster stability and homogeneity analyses
To quantify how homogenous each cluster is and uncover potential further subdivisions that may exist within our clusters, we applied two approaches highly similar to those used for our previously described dataset25. First, we generated a measure of stability for each cluster through random downsampling and reclustering of the dataset using the same custom R scripts as previously described25. Doing so provided cluster stability metrics (Fig S2A) representing the propensity for cells within a cluster to continue to cocluster when data is removed.
To understand potential sources of lower cluster stability values (e.g. two clusters being grouped together as one, or additional small clusters being divided into two adjacent clusters), we utilized the ClusTree (v0.5.1 – RRID:SCR_016293 – https://github.com/lazappi/clustree) R package to plot clustering at ten incremental resolutions of 0.1 through 1.1 (Fig S2B).
Scanpy
Cells were clustered using the scanpy toolkit (Version #1.10.1 RRID: SCR_018139 https://scanpy.readthedocs.io/en/stable/ on Python 3.10.13105. Outputs from CellRanger were read into Scanpy using scanpy.read_10x_mtx(). Cells were filtered with the same parameters as described above in the Seurat workflow. Datasets were individually clustered using the Leiden clustering algorithm. The four datasets were then integrated using scanpy.tl.ingest.(). Each dataset was mapped onto Control 1. The exact workflow can be found at the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
Pathway Enrichment Analyses
To evaluate pathways and gene sets enriched in subtypes or across conditions, we utilized the fgsea (v3.19; https://bioconductor.org/packages/release/bioc/html/fgsea.html; SCR_020938) and clusterProfiler (v4.8.3 – RRID:SCR_016884 – https://doi.org/doi:10.18129/B9.bioc.clusterProfiler) R packages. Pathways for GSEA analysis (mouse canonical pathways gene sets) were obtained from MSigDB (v2023.2.Hs, 2023.2.Mm; https://www.gsea-msigdb.org/gsea/msigdb; SCR_016863). Custom R scripts were created for visualization of GSEA results and are available in the deposited code for this paper. fgsea was run using the following parameters: nperm=1000, stats=ranks, minSize = 25, maxSize = 500. For gene ontology, the biological processes gene sets were obtained using the org.Mm.eg.db R package (v3.19; https://bioconductor.org/packages/release/data/annotation/html/org.Mm.eg.db.html; SCR_002 774), and GO (part of clusterProfiler package) was run using default settings with BH adjustment of p-values and utilizing FDR-adjusted significant (p < 0.05) DEGs for the respective comparisons. Results were visualized using the enrichplot R package (v3.19; https://www.bioconductor.org/packages/release/bioc/html/enrichplot.html; SCR_006442).
scDRS Cell-level Scoring of GWAS Risk Associations
Gene expression data (‘corrected’ UMI counts) was extracted from the SCT. Expression matrices, cell metadata, and feature metadata were loaded using Pandas (v2.2.2; https://pandas.pydata.org/; SCR_018214) in Jupyter (Jupyter: v7.2.1; https://jupyter.org/install; SCR_018315) notebook. The data were converted into an AnnData object using the anndata package (AnnData v0.10.8; https://anndata.readthedocs.io/en/latest/; SCR_018209) and stored in HDF5 format. Normalization and scaling were performed using the Scanpy package (v1.8.1) (PMID: 29409532). Total counts for each cell were normalized to 10,000 reads using the sc.pp.normalize_total function and log-transformed using sc.pp.log1p. The sc.pp.highly_variable_genes function (parameters: min_mean=0.0125, max_mean=3, and min_disp=0.5) was used to identify highly variable genes. Finally, the anndata object was scaled using the sc.pp.scale function, with a max_value parameter of 10.
GWAS summary statistics for Parkinson’s Disease risk were obtained from EBI GWAS Catalog Study ID: GCST009325 (PMID: 31701892) and processed using the MAGMA tool on FUMA’s online platform (v1.5.2; https://fuma.ctglab.nl/snp2gene; SCR_017521) to generate a scored list (z-scores) of significant genes. Default settings were used with a 10×10 gene window. Scored gene list was processed using the munge-gs function of the scDRS package (v1.0.2; https://martinjzhang.github.io/scDRS/) to create .gs gene sets (PMID: 34431100) and loaded into the Jupyter Notebook using the scdrs.uRl.load_gs function. To align with the anndata object data, the gene set’s human genes were mapped to mouse orthologs and intersected with genes present in the dataset. Preprocessing was completed using the scdrs.preprocess function, binning genes by mean and variance with parameters n_mean_bin=20 and n_var_bin=20. Scoring for Parkinson’s disease was performed using the scdrs.score_cell function (parameters: ctrl_match_key=mean_var, n_ctrl=1000, weight_opt=vs, return_ctrl_raw_score=False, and return_ctrl_norm_score=True).
Bootstrapped 95% confidence intervals for mean score within clusters or cluster families were calculated in R using 10,000 permutations for each group and plotted as mean scDRS score per group with CI represented as error bars.
Parkinson’s Disease MAGMA Analysis Ranked Genes dataset can be found at 10.5281/zenodo.13076447
SynGO Analyses
SynGO analysis protocol is described in detail at dx.doi.org/10.17504/protocols.io.x54v92r94l3e/v1.
MERFISH
Animals
Adult C57Bl/6 male mice (RRID:IMSR_CRL:027) aged 2–3 months were used for the MERFISH experiments. Animals were housed at the Montreal Neurological Institute (MNI) on a 12h-12h light-dark cycle (light cycle 7:00 to 19:00) with ad libitum access to food and water. Experiments were approved by the MNI Animal Care Committee and conducted according to guidelines and regulations from the Canadian Council on Animal Care.
Gene selection and panel assembly for MERFISH
Hybridization probes were generated by Vizgen using their custom gene portal. DA neuron specific genes were selected based on differential gene expression analysis from the 20 clusters identified in the single-nuclei sequencing data. Also included were: 1) general markers of glutamatergic, GABAergic, cholinergic, serotoninergic neuron populations, 2) markers of non-neuronal cell types, 3) neuropeptide precursor genes and neuropeptide receptors and 4) general ion channels. The list of all probes on the panel can be found in Supplemental Table 1.
Tissue preparation and tissue imaging for MERFISH
Mice were perfused with 1x PBS followed by 4% paraformaldehyde (PFA). Their brains were extracted and post-fixed in PFA for 16 hours, embedded in O.C.T. (Tissue-Tek O.C.T.; 25608–930, VWR), and stored at −80 °C until sectioning. Frozen brains were sectioned at −18 °C on a cryostat (Leica CM3050S). Within 3 animals we collected 10um-thick sequential coronal brain sections every 200μm on specialized glass slides (approximately 5–6 sections per mouse). Seven sections were chosen with the best morphology across the midbrain for processing. Sample preparation was completed using Vizgen gene imaging kits provided according to manufacturer. All steps were performed with RNAse free conditions. Sections were first washed with 1x PBS three times before incubation in 70% Ethanol overnight at 4°C to permeabilize cell membranes. The following day each section was washed with sample wash buffer followed by an incubation in formamide wash buffer at 37 °C for 30 minutes. The gene panel mix containing hybridization probes was then placed on top of the tissue and placed in a humidified 37 °C chamber for 36–48h. Afterwards the tissue was incubated with formamide buffer for 30m at 47 °C twice before washing with sample wash buffer. The hybridized Tissue was then embedded in a 0.05% ammonium persulfate gel and cleared overnight in a solution of Proteinase K (NEB Cat# P8107S) at 37 °C. The following morning the sample was incubated in a solution containing DAPI and PolyT stain to aid in cell segmentation. Once processed, brain sections were prepped for imaging using the commercial Merscope system developed by Vizgen. 500-gene panel cartridges were thawed in a 37°C water bath for 1h before the start of imaging. Mouse RNAse inhibitor (NEB Cat# M0314) was added to imaging activation buffer and added to the cartridge fluidic system before loading into the Merscope to prime the fluidics chamber. Slides were removed from wash buffer and placed in the imaging capsule, connected to the fluidics chamber. Sections were imaged according to the manufacturer (Vizgen). The exact protocol for tissue preparation and tissue imaging for MERFISH can be found at dx.doi.org/10.17504/protocols.io.14egn6opyl5d/v1.
Preprocessing and quality control of MERFISH data
Cell by gene matrices were processed in R using the Seurat V5 package103. To exclude cells that underwent poor cell segmentation we removed cells that had >500μm3 and <4000μm3 volumes. To remove cells that show low transcript identity or falsely identified cells we removed any cell that had <40 total RNA transcripts detected. The remaining cells then underwent SCT normalization102 before running RPCA integration to integrate datasets from individual sections. Shared nearest neighbour clustering was completed using the first 37 principle components using a resolution of k=0.7. Genetic markers of each cluster were determined using a roc test and was used to annotate our dataset. High-level annotations for glia and neuron types were confirmed using the SCType package in R (v3.0; https://sctype.app/). The putative DA cluster was subsetted, re-normalized and subclustered using the first 19 principle components and a resolution of k=0.65. Gene markers of each subcluster was determined using a ROC test and the top three genes from each cluster were identified based on the high positive log-fold change with a low p-values (adjusted p-value<10−50). Code can be found on the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
Integration of MERFISH data with scRNA-seq data
To integrate MERFISH data with snRNA-seq data we utilized the weighted-nearest neighbour algorithm defined in Stuart, Butler, et al 106. Briefly, anchor points were established between the two datasets using the FindTransferAnchors and MapQuery functions in Seurat. This allowed for a Projection of the MERFISH data into the scRNA-seq space and a transfer of cluster identity between the two. Importantly, a label transfer prediction score was assigned to each MERFISH cell and cells with scores greater than 0.5 were classified as high confidence cells. Whole genome expression was imputed onto MERFISH cells using snRNA-seq data using the TransferData function in Seurat using the snRNA-seq normalized data as a reference. Cluster identifications were compared between transfer data and MERFISH subclustering using a Sankeyplot in R. Briefly, the number of cells that transferred between two pairs of clusters were calculated and expressed as a proportion within each MERFISH cluster. Only flows >10% were included in the visualization. Spatial visualizations of DA subtypes were generated using the ImageDimPlot function in Seurat and overlaid on the Allen Brain Atlas mouse anatomical template or the Paxinos mouse brain coronal axis using affinity designer. Gene expression values were obtained using the ImageFeaturePlot function in Seurat. Code can be found on the Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal).
MERFISH anatomical localization
We utilized the Vizualizer software (v2.2, Vizgen Inc. https://portal.vizgen.com/resources/software) to delineate mouse midbrain regions into 11 distinct regions: the caudal linear nucleus (CLi), interfascicular nucleus (IF), interpeduncular nucleus (IPN), periaqueductal grey region (PAG), posterior hypothalamus (PH), rostral linear nucleus (RLi), retrorubral field (RR), substantia nigra pars compacta (SNc), Substantia nigra pars reticulata (SNr), Superior Colliculus (SC) and the ventral tegmental area (VTA). Brain regions were manually sectioned based on the intensity of DAPI imaging as well as the presence of Th transcripts compared to the Allen Brain Atlas. Cells within each ROI were counted to generate the localization of each family and cluster by counts and proportions.
MERFISH data can be found at DOI #: 10.5281/zenodo.12636328.
Supplementary Material
Acknowledgements
ZG and CO performed snRNAseq and MERFISH experiments and data analysis. CB, ES, AS and CC performed data analysis and shinycell app construction. LP and DD provided scientific input on DA classification and LRRK2 alterations. ZG, CO, LP, JFP, RA wrote the manuscript. We would like to acknowledge Dr. Kevin Petrecca, Dr. Phuong Le and Michael Luo for generously providing access to Merscope and technical advice. RA, DD, AS and LP were funded by Aligning Science Across Parkinson’s (ASAP-020600). For the purpose of open access, the authors have applied a CC-BY 4.0 public copyright license to this manuscript. RA was also funded by 1R01NS119690-01, P50 DA044121-01A1. LP was funded by R01 NS097901. ZG was funded by NINDS 1F31NS115524-01A1. JFP was funded by CIHR, HBHL, Parkinson Canada. CB was funded by Parkinson Canada and FRQS.
Footnotes
Teaser: Using snRNASeq and MERFISH we identified midbrain DA subtypes, mapped their spatial location, and identified alterations in a LRRK2 model
Code Availability Statement
Code is available at Awatramani Lab Github page (github.com/AwatramaniLab/Gaertner_Oram_etal) and DOI # 10.5281/zenodo.13820931.
Extended Data:
Extended data files contain raw differential expression analysis results in Excel format. Tabs within the file are provided for various comparisons referenced/used throughout the text.
References
- 1.Björklund A, Dunnek SB. Dopamine neuron systems in the brain: an update. Trends Neurosci. 2007;30(5):194–202. Epub 20070403. doi: 10.1016/j.tins.2007.03.006. [DOI] [PubMed] [Google Scholar]
- 2.Fu Y, Yuan Y, Halliday G, Rusznák Z, Watson C, Paxinos G. A cytoarchitectonic and chemoarchitectonic analysis of the dopamine cell groups in the substantia nigra, ventral tegmental area, and retrorubral field in the mouse. Brain Struct Funct. 2012;217(2):591–612. Epub 20110921. doi: 10.1007/s00429-011-0349-2. [DOI] [PubMed] [Google Scholar]
- 3.Gaertner Z, Azcorra M, Dombeck DA, Awatramani R. Molecular heterogeneity in the substantia nigra: A roadmap for understanding PD motor pathophysiology. Neurobiol Dis. 2022;175:105925. Epub 20221111. doi: 10.1016/j.nbd.2022.105925. [DOI] [PubMed] [Google Scholar]
- 4.Evans RC, Zhu M, Khaliq ZM. Dopamine Inhibition Differentially Controls Excitability of Substantia Nigra Dopamine Neuron Subpopulations through T-Type Calcium Channels. J Neurosci. 2017;37(13):3704–20. Epub 20170306. doi: 10.1523/jneurosci.0117-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Menegas W, Akiti K, Amo R, Uchida N, Watabe-Uchida M. Dopamine neurons projecting to the posterior striatum reinforce avoidance of threatening stimuli. Nat Neurosci. 2018;21(10):1421–30. Epub 20180903. doi: 10.1038/s41593-018-0222-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Engelhard B, Finkelstein J, Cox J, Fleming W, Jang HJ, Ornelas S, Koay SA, Thiberge SY, Daw ND, Tank DW, Wiken IB. Specialized coding of sensory, motor and cognitive variables in VTA dopamine neurons. Nature. 2019;570(7762):509–13. Epub 20190529. doi: 10.1038/s41586-019-1261-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lammel S, Hetzel A, Häckel O, Jones I, Liss B, Roeper J. Unique properties of mesoprefrontal neurons within a dual mesocorticolimbic dopamine system. Neuron. 2008;57(5):760–73. doi: 10.1016/j.neuron.2008.01.022. [DOI] [PubMed] [Google Scholar]
- 8.da Silva JA, Tecuapetla F, Paixão V, Costa RM. Dopamine neuron activity before action initiation gates and invigorates future movements. Nature. 2018;554(7691):244–8. Epub 20180131. doi: 10.1038/nature25457. [DOI] [PubMed] [Google Scholar]
- 9.Matsumoto M, Hikosaka O. Two types of dopamine neuron distinctly convey positive and negative motivational signals. Nature. 2009;459(7248):837–41. Epub 20090517. doi: 10.1038/nature08028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Beier KT, Steinberg EE, DeLoach KE, Xie S, Miyamichi K, Schwarz L, Gao XJ, Kremer EJ, Malenka RC, Luo L. Circuit Architecture of VTA Dopamine Neurons Revealed by Systematic Input-Output Mapping. Cell. 2015;162(3):622–34. doi: 10.1016/j.cell.2015.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Avvisati R, Kaufmann AK, Young CJ, Portlock GE, Cancemi S, Costa RP, Magill PJ, Dodson PD. Distributional coding of associative learning in discrete populations of midbrain dopamine neurons. Cell Rep. 2024;43(4):114080. Epub 20240404. doi: 10.1016/j.celrep.2024.114080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brischoux F, Chakraborty S, Brierley DI, Ungless MA. Phasic excitation of dopamine neurons in ventral VTA by noxious stimuli. Proc Natl Acad Sci U S A. 2009;106(12):4894–9. Epub 20090304. doi: 10.1073/pnas.0811507106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Heymann G, Jo YS, Reichard KL, McFarland N, Chavkin C, Palmiter RD, Soden ME, Zweifel LS. Synergy of Distinct Dopamine Projection Populations in Behavioral Reinforcement. Neuron. 2020;105(5):909–20.e5. Epub 20191223. doi: 10.1016/j.neuron.2019.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Howe MW, Dombeck DA. Rapid signalling in distinct dopaminergic axons during locomotion and reward. Nature. 2016;535(7613):505–10. Epub 20160711. doi: 10.1038/nature18942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lerner TN, Shilyansky C, Davidson TJ, Evans KE, Beier KT, Zalocusky KA, Crow AK, Malenka RC, Luo L, Tomer R, Deisseroth K. Intact-Brain Analyses Reveal Distinct Information Carried by SNc Dopamine Subcircuits. Cell. 2015;162(3):635–47. doi: 10.1016/j.cell.2015.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Phillips RA 3rd, Tuscher JJ, Black SL, Andraka E, Fitzgerald ND, Ianov L, Day JJ. An atlas of transcriptionally defined cell populations in the rat ventral tegmental area. Cell Rep. 2022;39(1):110616. doi: 10.1016/j.celrep.2022.110616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Poulin JF, Gaertner Z, Moreno-Ramos OA, Awatramani R. Classification of Midbrain Dopamine Neurons Using Single-Cell Gene Expression Profiling Approaches. Trends Neurosci. 2020;43(3):155–69. Epub 20200211. doi: 10.1016/j.Rns.2020.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Garritsen O, van Bakum EY, Grossouw LM, Pasterkamp RJ. Development, wiring and function of dopamine neuron subtypes. Nat Rev Neurosci. 2023;24(3):134–52. Epub 20230118. doi: 10.1038/s41583-022-00669-3. [DOI] [PubMed] [Google Scholar]
- 19.La Manno G, Gyllborg D, Codeluppi S, Nishimura K, Salto C, Zeisel A, Borm LE, Stok SRW, Toledo EM, Villaescusa JC, Lönnerberg P, Ryge J, Barker RA, Arenas E, Linnarsson S. Molecular Diversity of Midbrain Development in Mouse, Human, and Stem Cells. Cell. 2016;167(2):566–80.e19. doi: 10.1016/j.cell.2016.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kramer DJ, Risso D, Kosillo P, Ngai J, Bateup HS. Combinatorial Expression of Grp and Neurod6 Defines Dopamine Neuron Populations with Distinct Projection Patterns and Disease Vulnerability. eNeuro. 2018;5(3). Epub 20180613. doi: 10.1523/eneuro.0152-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hook PW, McClymont SA, Cannon GH, Law WD, Morton AJ, Goff LA, McCallion AS. Single-Cell RNA-Seq of Mouse Dopaminergic Neurons Informs Candidate Gene Selection for Sporadic Parkinson Disease. Am J Hum Genet. 2018;102(3):427–46. doi: 10.1016/j.ajhg.2018.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tiklová K, Björklund Å K, LahR L, Fiorenzano A, Nolbrant S, Gillberg L, Volakakis N, Yokota C, Hilscher MM, Hauling T, Holmström F, Joodmardi E, Nilsson M, Parmar M, Perlmann T. Single-cell RNA sequencing reveals midbrain dopamine neuron diversity emerging during mouse brain development. Nat Commun. 2019;10(1):581. Epub 20190204. doi: 10.1038/s41467-019-08453-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Saunders A, Macosko EZ, Wysoker A, Goldman M, Krienen FM, de Rivera H, Bien E, Baum M, Bortolin L, Wang S, Goeva A, Nemesh J, Kamitaki N, Brumbaugh S, Kulp D, McCarroll SA. Molecular Diversity and Specializations among the Cells of the Adult Mouse Brain. Cell. 2018;174(4):1015–30.e16. doi: 10.1016/j.cell.2018.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yaghmaeian Salmani B, Lahti L, Gillberg L, Jacobsen JK, Mantas I, Svenningsson P, Perlmann T. Transcriptomic atlas of midbrain dopamine neurons uncovers differential vulnerability in a Parkinsonism lesion model. Cold Spring Harbor Laboratory; 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Azcorra M, Gaertner Z, Davidson C, He Q, Kim H, Nagappan S, Hayes CK, Ramakrishnan C, Fenno L, Kim YS, Deisseroth K, Longnecker R, Awatramani R, Dombeck DA. Unique functional responses differentially map onto genetic subtypes of dopamine neurons. Nat Neurosci. 2023;26(10):1762–74. Epub 20230803. doi: 10.1038/s41593-023-01401-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu J, Kung J, Dong J, Chang L, Xie C, Habib A, Hawes S, Yang N, Chen V, Liu Z, Evans R, Liang B, Sun L, Ding J, Yu J, Saez-ARenzar S, Tang B, Khaliq Z, Lin DT, Le W, Cai H. Distinct Connectivity and Functionality of Aldehyde Dehydrogenase 1a1-Positive Nigrostriatal Dopaminergic Neurons in Motor Learning. Cell Rep. 2019;28(5):1167–81.e7. doi: 10.1016/j.celrep.2019.06.095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Poulin JF, Caronia G, Hofer C, Cui Q, Helm B, Ramakrishnan C, Chan CS, Dombeck DA, Deisseroth K, Awatramani R. Mapping projections of molecularly defined dopamine neuron subtypes using intersectional genetic approaches. Nat Neurosci. 2018;21(9):1260–71. Epub 20180813. doi: 10.1038/s41593-018-0203-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Greggio E, Jain S, Kingsbury A, Bandopadhyay R, Lewis P, Kaganovich A, van der Brug MP, Beilina A, Blackinton J, Thomas KJ, Ahmad R, Miller DW, Kesavapany S, Singleton A, Lees A, Harvey RJ, Harvey K, Cookson MR. Kinase activity is required for the toxic effects of mutant LRRK2/dardarin. Neurobiol Dis. 2006;23(2):329–41. Epub 20060605. doi: 10.1016/j.nbd.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 29.Tokars V, Chen C, Parisiadou L. Closing the structure-to-function gap for LRRK2. Trends Biochem Sci. 2022;47(3):187–8. Epub 20211028. doi: 10.1016/j.Rbs.2021.10.003. [DOI] [PubMed] [Google Scholar]
- 30.Taymans JM, Fell M, Greenamyre T, Hirst WD, Mamais A, Padmanabhan S, Peter I, Rideout H, Thaler A. Perspective on the current state of the LRRK2 field. NPJ Parkinsons Dis. 2023;9(1):104. Epub 20230701. doi: 10.1038/s41531-023-00544-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Di Maio R, Hoffman EK, Rocha EM, Keeney MT, Sanders LH, De Miranda BR, Zharikov A, Van Laar A, Stepan AF, Lanz TA, Kofler JK, Burton EA, Alessi DR, Hastings TG, Greenamyre JT. LRRK2 activation in idiopathic Parkinson’s disease. Sci Transl Med. 2018;10(451). doi: 10.1126/scitranslmed.aar5429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Khan SS, Jaimon E, Lin Y-E, Nikoloff J, Tonelli F, Alessi DR, Pfeffer SR. Loss of primary cilia and dopaminergic neuroprotection in pathogenic LRRK2-driven and idiopathic Parkinson’s disease. bioRxiv. 2024:2024.01.15.575737. doi: 10.1101/2024.01.15.575737. [DOI] [PMC free article] [PubMed]
- 33.Matikainen-Ankney BA, Kezunovic N, Mesias RE, Tian Y, Williams FM, Huntley GW, Benson DL. Altered Development of Synapse Structure and Function in Striatum Caused by Parkinson’s Disease-Linked LRRK2-G2019S Mutation. J Neurosci. 2016;36(27):7128–41. doi: 10.1523/jneurosci.3314-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen C, Soto G, Dumrongprechachan V, Bannon N, Kang S, Kozorovitskiy Y, Parisiadou L. Pathway-specific dysregulation of striatal excitatory synapses by LRRK2 mutations. Elife. 2020;9. Epub 20201002. doi: 10.7554/eLife.58997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xenias HS, Chen C, Kang S, Cherian S, Situ X, Shanmugasundaram B, Liu G, Scesa G, Savio Chan C, Parisiadou L. R1441C and G2019S LRRK2 knockin mice have distinct striatal molecular, physiological, and behavioral alterations. Commun Biol. 2022;5(1):1211. Epub 20221110. doi: 10.1038/s42003-022-04136-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yue M, Hinkle KM, Davies P, Trushina E, Fiesel FC, Christenson TA, Schroeder AS, Zhang L, Bowles E, Behrouz B, Lincoln SJ, Beevers JE, Milnerwood AJ, Kurti A, McLean PJ, Fryer JD, Springer W, Dickson DW, Farrer MJ, Melrose HL. Progressive dopaminergic alterations and mitochondrial abnormalities in LRRK2 G2019S knock-in mice. Neurobiol Dis. 2015;78:172–95. Epub 20150331. doi: 10.1016/j.nbd.2015.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tozzi A, Tantucci M, Marchi S, Mazzocche P, Morari M, Pinton P, Mancini A, Calabresi P. Dopamine D2 receptor-mediated neuroprotection in a G2019S Lrrk2 genetic model of Parkinson’s disease. Cell Death Dis. 2018;9(2):204. Epub 20180212. doi: 10.1038/s41419-017-0221-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Poulin JF, Zou J, Drouin-Ouellet J, Kim KY, Cicchetti F, Awatramani RB. Defining midbrain dopaminergic neuron diversity by single-cell gene expression profiling. Cell Rep. 2014;9(3):930–43. Epub 20141030. doi: 10.1016/j.celrep.2014.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Soden ME, Miller SM, Burgeno LM, Phillips PEM, Hnasko TS, Zweifel LS. Genetic Isolation of Hypothalamic Neurons that Regulate Context-Specific Male Social Behavior. Cell Rep. 2016;16(2):304–13. Epub 20160623. doi: 10.1016/j.celrep.2016.05.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nouri N, Awatramani R. A novel floor plate boundary defined by adjacent En1 and Dbx1 microdomains distinguishes midbrain dopamine and hypothalamic neurons. Development. 2017;144(5):916–27. Epub 20170207. doi: 10.1242/dev.144949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kamath T, Abdulraouf A, Burris SJ, Langlieb J, Gazestani V, Nadaf NM, Balderrama K, Vanderburg C, Macosko EZ. Single-cell genomic profiling of human dopamine neurons identifies a population that selectively degenerates in Parkinson’s disease. Nat Neurosci. 2022;25(5):588–95. Epub 20220505. doi: 10.1038/s41593-022-01061-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Anderegg A, Poulin JF, Awatramani R. Molecular heterogeneity of midbrain dopaminergic neurons--Moving toward single cell resolution. FEBS Lek. 2015;589(24 Pt A):3714–26. Epub 20151023. doi: 10.1016/j.febslet.2015.10.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pereira Luppi M, Azcorra M, Caronia-Brown G, Poulin JF, Gaertner Z, Gatica S, Moreno-Ramos OA, Nouri N, Dubois M, Ma YC, Ramakrishnan C, Fenno L, Kim YS, Deisseroth K, Cicchetti F, Dombeck DA, Awatramani R. Sox6 expression distinguishes dorsally and ventrally biased dopamine neurons in the substanRa nigra with distinctive properties and embryonic origins. Cell Rep. 2021;37(6):109975. doi: 10.1016/j.celrep.2021.109975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen KH, Boettiger AN, Moffitt JR, Wang S, Zhuang X. RNA imaging. Spatially resolved, highly multiplexed RNA profiling in single cells. Science. 2015;348(6233):aaa6090. Epub 20150409. doi: 10.1126/science.aaa6090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bradl M, Lassmann H. Oligodendrocytes: biology and pathology. Acta Neuropathol. 2010;119(1):37–53. Epub 20091022. doi: 10.1007/s00401-009-0601-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhang M, Pan X, Jung W, Halpern AR, Eichhorn SW, Lei Z, Cohen L, Smith KA, Tasic B, Yao Z, Zeng H, Zhuang X. Molecularly defined and spatially resolved cell atlas of the whole mouse brain. Nature. 2023;624(7991):343–54. Epub 20231213. doi: 10.1038/s41586-023-06808-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yao Z, van Velthoven CTJ, Kunst M, Zhang M, McMillen D, Lee C, Jung W, Goldy J, Abdelhak A, Aitken M, Baker K, Baker P, Barkan E, Bertagnolli D, Bhandiwad A, Bielstein C, Bishwakarma P, Campos J, Carey D, Casper T, Chakka AB, Chakrabarty R, Chavan S, Chen M, Clark M, Close J, Crichton K, Daniel S, DiValenRn P, Dolbeare T, Ellingwood L, Fiabane E, Fliss T, Gee J, Gerstenberger J, Glandon A, Gloe J, Gould J, Gray J, Guilford N, Guzman J, Hirschstein D, Ho W, Hooper M, Huang M, Hupp M, Jin K, Kroll M, Lathia K, Leon A, Li S, Long B, Madigan Z, Malloy J, Malone J, Maltzer Z, Martin N, McCue R, McGinty R, Mei N, Melchor J, Meyerdierks E, Mollenkopf T, Moonsman S, Nguyen TN, Oko S, Pham T, Rimorin C, Ruiz A, Sanchez R, Sawyer L, Shapovalova N, Shepard N, Slaughterbeck C, Sulc J, Tieu M, Torkelson A, Tung H, Valera Cuevas N, Vance S, Wadhwani K, Ward K, Levi B, Farrell C, Young R, Staats B, Wang MM, Thompson CL, Mumi S, Pagan CM, Kruse L, Dee N, Sunkin SM, Esposito L, Hawrylycz MJ, Waters J, Ng L, Smith K, Tasic B, Zhuang X, Zeng H. A high-resolution transcriptomic and spatial atlas of cell types in the whole mouse brain. Nature. 2023;624(7991):317–32. Epub 20231213. doi: 10.1038/s41586-023-06812-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Langlieb J, Sachdev NS, Balderrama KS, Nadaf NM, Raj M, Murray E, Webber JT, Vanderburg C, Gazestani V, Tward D, Mezias C, Li X, Flowers K, Cable DM, Norton T, Mitra P, Chen F, Macosko EZ. The molecular cytoarchitecture of the adult mouse brain. Nature. 2023;624(7991):333–42. Epub 20231213. doi: 10.1038/s41586-023-06818-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Trudeau LE, Hnasko TS, Wallén-Mackenzie A, Morales M, Rayport S, Sulzer D. The multilingual nature of dopamine neurons. Prog Brain Res. 2014;211:141–64. doi: 10.1016/b978-0-444-63425-2.00006-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Descarries L, Bérubé-Carrière N, Riad M, Bo GD, Mendez JA, Trudeau LE. Glutamate in dopamine neurons: synaptic versus diffuse transmission. Brain Res Rev. 2008;58(2):290–302. Epub 20071026. doi: 10.1016/j.brainresrev.2007.10.005. [DOI] [PubMed] [Google Scholar]
- 51.Morales M, Root DH. Glutamate neurons within the midbrain dopamine regions. Neuroscience. 2014;282:60–8. Epub 20140527. doi: 10.1016/j.neuroscience.2014.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Morales M, Margolis EB. Ventral tegmental area: cellular heterogeneity, Connectivity and behaviour. Nat Rev Neurosci. 2017;18(2):73–85. Epub 20170105. doi: 10.1038/nrn.2016.165. [DOI] [PubMed] [Google Scholar]
- 53.Tritsch NX, Granger AJ, SabaRni BL. Mechanisms and functions of GABA co-release. Nat Rev Neurosci. 2016;17(3):139–45. Epub 20160211. doi: 10.1038/nrn.2015.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Conrad WS, Oriol L, Faget L, Hnasko TS. Proportion and Distribution of neurotransmiker-defined cell types in the ventral tegmental area and substantia nigra pars compacta. bioRxiv. 2024:2024.02.28.582356. doi: 10.1101/2024.02.28.582356. [DOI]
- 55.Panman L, Papathanou M, Laguna A, Oosterveen T, Volakakis N, Acampora D, Kurtsdoker I, Yoshitake T, Kehr J, Joodmardi E, Muhr J, Simeone A, Ericson J, Perlmann T. Sox6 and Otx2 control the specification of substantia nigra and ventral tegmental area dopamine neurons. Cell Rep. 2014;8(4):1018–25. Epub 20140807. doi: 10.1016/j.celrep.2014.07.016. [DOI] [PubMed] [Google Scholar]
- 56.Singh A, Zhi L, Zhang H. LRRK2 and mitochondria: Recent advances and current views. Brain Research. 2019;1702:96–104. doi: 10.1016/j.brainres.2018.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Choi I, Kim B, Byun JW, Baik SH, Huh YH, Kim JH, Mook-Jung I, Song WK, Shin JH, Seo H, Suh YH, Jou I, Park SM, Kang HC, Joe EH. LRRK2 G2019S mutation akenuates microglial motility by inhibiting focal adhesion kinase. Nat Commun. 2015;6:8255. Epub 20150914. doi: 10.1038/ncomms9255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Parisiadou L, Yu J, Sgobio C, Xie C, Liu G, Sun L, Gu XL, Lin X, Crowley NA, Lovinger DM, Cai H. LRRK2 regulates synaptogenesis and dopamine receptor activation through modulation of PKA activity. Nat Neurosci. 2014;17(3):367–76. Epub 20140126. doi: 10.1038/nn.3636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cook DA, Kannarkat GT, Cintron AF, Butkovich LM, Fraser KB, Chang J, Grigoryan N, Factor SA, West AB, Boss JM, Tansey MG. LRRK2 levels in immune cells are increased in Parkinson’s disease. NPJ Parkinsons Dis. 2017;3:11. Epub 20170328. doi: 10.1038/s41531-017-0010-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.West AB, Cowell RM, Daher JP, Moehle MS, Hinkle KM, Melrose HL, Standaert DG, Volpicelli-Daley LA. Differential LRRK2 expression in the cortex, striatum, and substantia nigra in transgenic and nontransgenic rodents. J Comp Neurol. 2014;522(11):2465–80. Epub 20140412. doi: 10.1002/cne.23583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pallos J, Jeng S, McWeeney S, Martin I. Dopamine neuron-specific LRRK2 G2019S effects on gene expression revealed by translatome profiling. Neurobiol Dis. 2021;155:105390. Epub 20210511. doi: 10.1016/j.nbd.2021.105390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liu G, Sgobio C, Gu X, Sun L, Lin X, Yu J, Parisiadou L, Xie C, Sastry N, Ding J, Lohr KM, Miller GW, Mateo Y, Lovinger DM, Cai H. Selective expression of Parkinson’s disease-related Leucine-rich repeat kinase 2 G2019S missense mutation in midbrain dopaminergic neurons impairs dopamine release and dopaminergic gene expression. Hum Mol Genet. 2015;24(18):5299–312. Epub 20150629. doi: 10.1093/hmg/ddv249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Buckley MT, Sun ED, George BM, Liu L, Schaum N, Xu L, Reyes JM, Goodell MA, Weissman IL, Wyss-Coray T, Rando TA, Brunet A. Cell-type-specific aging clocks to quantify aging and rejuvenation in neurogenic regions of the brain. Nat Aging. 2023;3(1):121–37. Epub 20221219. doi: 10.1038/s43587-022-00335-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhao T, Goedhart CM, Sam PN, Sabouny R, Lingrell S, Cornish AJ, Lamont RE, Bernier FP, Sinasac D, Parboosingh JS, Vance JE, Claypool SM, Innes AM, Shuk TE. PISD is a mitochondrial disease gene causing skeletal dysplasia, cataracts, and white maker changes. Life Sci Alliance. 2019;2(2). Epub 20190311. doi: 10.26508/lsa.201900353. [DOI] [PMC free article] [PubMed]
- 65.Thomas HE, Zhang Y, Stefely JA, Veiga SR, Thomas G, Kozma SC, Mercer CA. Mitochondrial Complex I Activity Is Required for Maximal Autophagy. Cell Rep. 2018;24(9):2404–17.e8. doi: 10.1016/j.celrep.2018.07.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jin K, Yao Z, van Velthoven CTJ, Kaplan ES, Glattfelder K, Barlow ST, Boyer G, Carey D, Casper T, Chakka AB, Chakrabarty R, Clark M, Departee M, Desierto M, Gary A, Gloe J, Goldy J, Guilford N, Guzman J, Hirschstein D, Lee C, Liang E, Pham T, Reding M, Ronellenfitch K, Ruiz A, Sevigny J, Shapovalova N, Shulga L, Sulc J, Torkelson A, Tung H, Levi B, Sunkin SM, Dee N, Esposito L, Smith K, Tasic B, Zeng H. Cell-type specific molecular signatures of aging revealed in a brain-wide transcriptomic cell-type atlas. bioRxiv. 2023. Epub 20230727. doi: 10.1101/2023.07.26.550355. [DOI]
- 67.Zhang F, Yao Y, Miao N, Wang N, Xu X, Yang C. Neuroprotective effects of microRNA 124 in Parkinson’s disease mice. Archives of Gerontology and Geriatrics. 2022;99:104588. doi: 10.1016/j.archger.2021.104588. [DOI] [PubMed] [Google Scholar]
- 68.Yao L, Ye Y, Mao H, Lu F, He X, Lu G, Zhang S. MicroRNA-124 regulates the expression of MEKK3 in the inflammatory pathogenesis of Parkinson’s disease. J Neuroinflammation. 2018;15(1):13. Epub 20180112. doi: 10.1186/s12974-018-1053-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Han D, Dong X, Zheng D, Nao J. MiR-124 and the Underlying Therapeutic Promise of Neurodegenerative Disorders. Front Pharmacol. 2019;10:1555. Epub 20200117. doi: 10.3389/fphar.2019.01555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Angelopoulou E, Paudel YN, Piperi C. miR-124 and Parkinson’s disease: A biomarker with therapeutic potential. Pharmacol Res. 2019;150:104515. Epub 20191107. doi: 10.1016/j.phrs.2019.104515. [DOI] [PubMed] [Google Scholar]
- 71.González-Rodríguez P, Zampese E, Stout KA, Guzman JN, Ilijic E, Yang B, Tkatch T, Stavarache MA, Wokosin DL, Gao L, Kaplik MG, López-Barneo J, Schumacker PT, Surmeier DJ. Disruption of mitochondrial complex I induces progressive parkinsonism. Nature. 2021;599(7886):650–6. Epub 20211103. doi: 10.1038/s41586-021-04059-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Surmeier DJ, Obeso JA, Halliday GM. Selective neuronal vulnerability in Parkinson disease. Nat Rev Neurosci. 2017;18(2):101–13. doi: 10.1038/nrn.2016.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mortiboys H, Johansen KK, Aasly JO, Bandmann O. Mitochondrial impairment in patients with Parkinson disease with the G2019S mutation in LRRK2. Neurology. 2010;75(22):2017–20. doi: 10.1212/WNL.0b013e3181ff9685. [DOI] [PubMed] [Google Scholar]
- 74.Singh F, Prescok AR, Rosewell P, Ball G, Reith AD, Ganley IG. Pharmacological rescue of impaired mitophagy in Parkinson’s disease-related LRRK2 G2019S knock-in mice. Elife. 2021;10. Epub 20210803. doi: 10.7554/eLife.67604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Liu H, Ho PW, Leung CT, Pang SY, Chang EES, Choi ZY, Kung MH, Ramsden DB, Ho SL. Aberrant mitochondrial morphology and function associated with impaired mitophagy and DNM1L-MAPK/ERK signaling are found in aged mutant Parkinsonian LRRK2(R1441G) mice. Autophagy. 2021;17(10):3196–220. Epub 20201210. doi: 10.1080/15548627.2020.1850008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nguyen M, Krainc D. LRRK2 phosphorylation of auxilin mediates synaptic defects in dopaminergic neurons from patients with Parkinson’s disease. Proc Natl Acad Sci U S A. 2018;115(21):5576–81. Epub 20180507. doi: 10.1073/pnas.1717590115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Soukup SF, Kuenen S, Vanhauwaert R, Manetsberger J, Hernández-Díaz S, Swerts J, Schoovaerts N, Vilain S, Gounko NV, Vints K, Geens A, De Strooper B, Verstreken P. A LRRK2-Dependent EndophilinA Phosphoswitch Is Critical for Macroautophagy at Presynaptic Terminals. Neuron. 2016;92(4):829–44. Epub 20161006. doi: 10.1016/j.neuron.2016.09.037. [DOI] [PubMed] [Google Scholar]
- 78.Islam MS, Nolte H, Jacob W, Ziegler AB, Pütz S, Grosjean Y, Szczepanowska K, Trifunovic A, Braun T, Heumann H, Heumann R, Hovemann B, Moore DJ, Krüger M. Human R1441C LRRK2 regulates the synaptic vesicle proteome and phosphoproteome in a Drosophila model of Parkinson’s disease. Hum Mol Genet. 2016;25(24):5365–82. doi: 10.1093/hmg/ddw352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Boecker CA, Goldsmith J, Dou D, Cajka GG, Holzbaur ELF. Increased LRRK2 kinase activity alters neuronal autophagy by disrupting the axonal transport of autophagosomes. Curr Biol. 2021;31(10):2140–54.e6. Epub 20210324. doi: 10.1016/j.cub.2021.02.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Skelton PD, Tokars V, Parisiadou L. LRRK2 at Striatal Synapses: Cell-Type Specificity and Mechanistic Insights. Cells. 2022;11(1). Epub 20220105. doi: 10.3390/cells11010169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pischedda F, Piccoli G. LRRK2 at the pre-synaptic site: A 16-years perspective. Journal of Neurochemistry. 2021;157(2):297–311. doi: 10.1111/jnc.15240. [DOI] [PubMed] [Google Scholar]
- 82.Pan PY, Li X, Wang J, Powell J, Wang Q, Zhang Y, Chen Z, Wicinski B, Hof P, Ryan TA, Yue Z. Parkinson’s Disease-Associated LRRK2 Hyperactive Kinase Mutant Disrupts Synaptic Vesicle Trafficking in Ventral Midbrain Neurons. J Neurosci. 2017;37(47):11366–76. Epub 20171020. doi: 10.1523/jneurosci.0964-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Koopmans F, van Nierop P, Andres-Alonso M, Byrnes A, Cijsouw T, Coba MP, Cornelisse LN, Farrell RJ, Goldschmidt HL, Howrigan DP, Hussain NK, Imig C, de Jong APH, Jung H, Kohansalnodehi M, Kramarz B, Lipstein N, Lovering RC, MacGillavry H, Mariano V, Mi H, Ninov M, Osumi-Sutherland D, Pielot R, Smalla KH, Tang H, Tashman K, Toonen RFG, Verpelli C, Reig-Viader R, Watanabe K, van Weering J, Achsel T, Ashrafi G, Asi N, Brown TC, De Camilli P, Feuermann M, Foulger RE, Gaudet P, Joglekar A, Kanellopoulos A, Malenka R, Nicoll RA, Pulido C, de Juan-Sanz J, Sheng M, Südhof TC, Tilgner HU, Bagni C, Bayés À, Biederer T, Brose N, Chua JJE, Dieterich DC, Gundelfinger ED, Hoogenraad C, Huganir RL, Jahn R, Kaeser PS, Kim E, Kreutz MR, McPherson PS, Neale BM, O’Connor V, Posthuma D, Ryan TA, Sala C, Feng G, Hyman SE, Thomas PD, Smit AB, Verhage M. SynGO: An Evidence-Based, Expert-Curated Knowledge Base for the Synapse. Neuron. 2019;103(2):217–34.e4. Epub 20190603. doi: 10.1016/j.neuron.2019.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Nalls MA, Blauwendraat C, Vallerga CL, Heilbron K, Bandres-Ciga S, Chang D, Tan M, Kia DA, Noyce AJ, Xue A, Bras J, Young E, von Coelln R, Simon-Sanchez J, Schulte C, Sharma M, Krohn L, Pihlstrom L, Siitonen A, Iwaki H, Leonard H, Faghri F, Gibbs JR, Hernandez DG, Scholz SW, BoRa JA, Martinez M, Corvol JC, Lesage S, Jankovic J, Shulman LM, Sutherland M, Tienari P, Majamaa K, Tom M, Andreassen OA, Bangale T, Brice A, Yang J, Gan-Or Z, Gasser T, HeuRnk P, Shulman JM, Wood NW, Hinds DA, Hardy JA, Morris HR, Graken J, Visscher PM, Graham RR, Singleton AB, andMe Research T, System Genomics of Parkinson’s Disease C, International Parkinson’s Disease Genomics C. Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: a meta-analysis of genome-wide association studies. Lancet Neurol. 2019;18(12):1091–102. doi: 10.1016/S1474-4422(19)30320-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zhang MJ, Hou K, Dey KK, Sakaue S, Jagadeesh KA, Weinand K, Taychameekiatchai A, Rao P, Pisco AO, Zou J, Wang B, Gandal M, Raychaudhuri S, Pasaniuc B, Price AL. Polygenic enrichment distinguishes disease associations of individual cells in single-cell RNA-seq data. Nature GeneRcs. 2022;54(10):1572–80. doi: 10.1038/s41588-022-01167-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Yaghmaeian Salmani B, LahR L, Gillberg L, Jacobsen JK, Mantas I, Svenningsson P, Perlmann T. Transcriptomic atlas of midbrain dopamine neurons uncovers differential vulnerability in a Parkinsonism lesion model. Elife. 2024;12. Epub 20240408. doi: 10.7554/eLife.89482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Blaess S, Bodea GO, Kabanova A, Chanet S, Mugniery E, Derouiche A, Stephen D, Joyner AL. Temporal-spatial changes in Sonic Hedgehog expression and signaling reveal different potentials of ventral mesencephalic progenitors to populate distinct ventral midbrain nuclei. Neural Dev. 2011;6:29. Epub 20110620. doi: 10.1186/1749-8104-6-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Joksimovic M, Anderegg A, Roy A, Campochiaro L, Yun B, Kikappa R, McKay R, Awatramani R. Spatiotemporally separable Shh domains in the midbrain define distinct dopaminergic progenitor pools. Proc Natl Acad Sci U S A. 2009;106(45):19185–90. Epub 20091022. doi: 10.1073/pnas.0904285106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yan CH, Levesque M, Claxton S, Johnson RL, Ang SL. Lmx1a and lmx1b function cooperatively to regulate proliferation, specification, and differentiation of midbrain dopaminergic progenitors. J Neurosci. 2011;31(35):12413–25. doi: 10.1523/jneurosci.1077-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Andersson E, Tryggvason U, Deng Q, Friling S, Alekseenko Z, Robert B, Perlmann T, Ericson J. Identification of intrinsic determinants of midbrain dopamine neurons. Cell. 2006;124(2):393–405. doi: 10.1016/j.cell.2005.10.037. [DOI] [PubMed] [Google Scholar]
- 91.Cox J, Wiken IB. Striatal circuits for reward learning and decision-making. Nat Rev Neurosci. 2019;20(8):482–94. doi: 10.1038/s41583-019-0189-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Tang L, Xu N, Huang M, Yi W, Sang X, Shao M, Li Y, Hao ZZ, Liu R, Shen Y, Yue F, Liu X, Xu C, Liu S. A primate nigrostriatal atlas of neuronal vulnerability and resilience in a model of Parkinson’s disease. Nat Commun. 2023;14(1):7497. Epub 20231118. doi: 10.1038/s41467-023-43213-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Yamada T, McGeer PL, Baimbridge KG, McGeer EG. Relative sparing in Parkinson’s disease of substantia nigra dopamine neurons containing calbindin-D28K. Brain Res. 1990;526(2):303–7. doi: 10.1016/0006-8993(90)91236-a. [DOI] [PubMed] [Google Scholar]
- 94.Damier P, Hirsch EC, Agid Y, Graybiel AM. The substantia nigra of the human brain. I. Nigrosomes and the nigral matrix, a compartmental organization based on calbindin D(28K) immunohistochemistry. Brain. 1999;122 (Pt 8):1421–36. doi: 10.1093/brain/122.8.1421. [DOI] [PubMed] [Google Scholar]
- 95.Kish SJ, Shannak K, Hornykiewicz O. Uneven pakern of dopamine loss in the striatum of patients with idiopathic Parkinson’s disease. Pathophysiologic and clinical implications. N Engl J Med. 1988;318(14):876–80. doi: 10.1056/nejm198804073181402. [DOI] [PubMed] [Google Scholar]
- 96.Kordower JH, Olanow CW, Dodiya HB, Chu Y, Beach TG, Adler CH, Halliday GM, Bartus RT. Disease duration and the integrity of the nigrostriatal system in Parkinson’s disease. Brain. 2013;136(Pt 8):2419–31. doi: 10.1093/brain/awt192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mendonça MD, Ferreira P, Oliveira F, Barbosa R, Meira B, Costa D, Oliveira-Maia A, Silva JAd. Integrity of dopaminergic terminals in the caudate nucleus is relevant for rest tremor in Parkinson’s disease. medRxiv. 2024:2024.04.04.24305353. doi: 10.1101/2024.04.04.24305353. [DOI] [PMC free article] [PubMed]
- 98.Schmack K, Bosc M, Ok T, Sturgill JF, Kepecs A. Striatal dopamine mediates hallucination-like perception in mice. Science. 2021;372(6537). doi: 10.1126/science.abf4740. [DOI] [PubMed] [Google Scholar]
- 99.Mandemakers W, Snellinx A, O’Neill MJ, de Strooper B. LRRK2 expression is enriched in the striosomal compartment of mouse striatum. Neurobiol Dis. 2012;48(3):582–93. Epub 20120729. doi: 10.1016/j.nbd.2012.07.017. [DOI] [PubMed] [Google Scholar]
- 100.Pischedda F, Piccoli G. LRRK2 at the pre-synaptic site: A 16-years perspective. J Neurochem. 2021;157(2):297–311. Epub 20210205. doi: 10.1111/jnc.15240. [DOI] [PubMed] [Google Scholar]
- 101.Sile K, Hodge R, Mossi Albiach A, Lee KW, Ding SL, Hu L, Lönnerberg P, Bakken T, Casper T, Clark M, Dee N, Gloe J, Hirschstein D, Shapovalova NV, Keene CD, Nyhus J, Tung H, Yanny AM, Arenas E, Lein ES, Linnarsson S. Transcriptomic diversity of cell types across the adult human brain. Science. 2023;382(6667):eadd7046. Epub 20231013. doi: 10.1126/science.add7046. [DOI] [PubMed] [Google Scholar]
- 102.Choudhary S, Satija R. Comparison and evaluation of statistical error models for scRNA-seq. Genome Biol. 2022;23(1):27. Epub 20220118. doi: 10.1186/s13059-021-02584-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hao Y, Stuart T, Kowalski MH, Choudhary S, Hoffman P, Hartman A, Srivastava A, Molla G, Madad S, Fernandez-Granda C, Satija R. Dictionary learning for integrative, multimodal and scalable single-cell analysis. Nature Biotechnology. 2024;42(2):293–304. doi: 10.1038/s41587-023-01767-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ouyang JF, Kamaraj US, Cao EY, Rackham OJL. ShinyCell: simple and sharable visualization of single-cell gene expression data. Bioinformatics. 2021;37(19):3374–6. doi: 10.1093/bioinformatics/btab209. [DOI] [PubMed] [Google Scholar]
- 105.Wolf FA, Angerer P, Theis FJ. SCANPY: large-scale single-cell gene expression data analysis. Genome Biol. 2018;19(1):15. Epub 20180206. doi: 10.1186/s13059-017-1382-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Stuart T, Butler A, Hoffman P, Hafemeister C, Papalexi E, Mauck WM 3rd, Hao Y, Stoeckius M, Smibert P, Satija R. Comprehensive Integration of Single-Cell Data. Cell. 2019;177(7):1888–902.e21. Epub 20190606. doi: 10.1016/j.cell.2019.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.