Abstract
Background
Constipation that is prolonged and does not resolve with conventional therapeutic measures is called intractable constipation. The treatment of intractable constipation is challenging, involving pharmacological or non‐pharmacological therapies, as well as surgical approaches. Unresolved constipation can negatively impact quality of life, with additional implications for health systems. Consequently, there is an urgent need to identify treatments that are efficacious and safe.
Objectives
To evaluate the efficacy and safety of treatments used for intractable constipation in children.
Search methods
We searched CENTRAL, MEDLINE, Embase, and two trials registers up to 23 June 2023. We also searched reference lists of included studies for relevant studies.
Selection criteria
We included randomised controlled trials (RCTs) comparing any pharmacological, non‐pharmacological, or surgical treatment to placebo or another active comparator, in participants aged between 0 and 18 years with functional constipation who had not responded to conventional medical therapy.
Data collection and analysis
We used standard Cochrane methods. Our primary outcomes were symptom resolution, frequency of defecation, treatment success, and adverse events; secondary outcomes were stool consistency, painful defecation, quality of life, faecal incontinence frequency, abdominal pain, hospital admission for disimpaction, and school absence. We used GRADE to assess the certainty of evidence for each primary outcome.
Main results
This review included 10 RCTs with 1278 children who had intractable constipation. We assessed one study as at low risk of bias across all domains. There were serious concerns about risk of bias in six studies.
One study compared the injection of 160 units botulinum toxin A (n = 44) to unspecified oral stool softeners (n = 44). We are very uncertain whether botulinum toxin A injection improves treatment success (risk ratio (RR) 37.00, 95% confidence interval (CI) 5.31 to 257.94; very low certainty evidence, downgraded due to serious concerns with risk of bias and imprecision). Frequency of defecation was reported only for the botulinum toxin A injection group (mean interval of 2.6 days). The study reported no data for the other primary outcomes.
One study compared erythromycin estolate (n = 6) to placebo (n = 8). The only primary outcome reported was adverse events, which were 0 in both groups. The evidence is of very low certainty due to concerns with risk of bias and serious imprecision.
One study compared 12 or 24 μg oral lubiprostone (n = 404) twice a day to placebo (n = 202) over 12 weeks. There may be little to no difference in treatment success (RR 1.29, 95% CI 0.87 to 1.92; low certainty evidence). We also found that lubiprostone probably results in little to no difference in adverse events (RR 1.05, 95% CI 0.91 to 1.21; moderate certainty evidence). The study reported no data for the other primary outcomes.
One study compared three‐weekly rectal sodium dioctyl sulfosuccinate and sorbitol enemas (n = 51) to 0.5 g/kg/day polyethylene glycol laxatives (n = 51) over a 52‐week period. We are very uncertain whether rectal sodium dioctyl sulfosuccinate and sorbitol enemas improve treatment success (RR 1.33, 95% CI 0.83 to 2.14; very low certainty evidence, downgraded due to serious concerns with risk of bias and imprecision). Results of defecation frequency per week was reported only as modelled means using a linear mixed model. The study reported no data for the other primary outcomes.
One study compared biofeedback therapy (n = 12) to no intervention (n = 12). We are very uncertain whether biofeedback therapy improves symptom resolution (RR 2.50, 95% CI 1.08 to 5.79; very low certainty evidence, downgraded due to serious concerns with risk of bias and imprecision). The study reported no data for the other primary outcomes.
One study compared 20 minutes of intrarectal electromotive botulinum toxin A using 2800 Hz frequency and botulinum toxin A dose 10 international units/kg (n = 30) to 10 international units/kg botulinum toxin A injection (n = 30). We are very uncertain whether intrarectal electromotive botulinum toxin A improves symptom resolution (RR 0.96, 95% CI 0.76 to 1.22; very low certainty evidence) or if it increases the frequency of defecation (mean difference (MD) 0.00, 95% CI −1.87 to 1.87; very low certainty evidence). We are also very uncertain whether intrarectal electromotive botulinum toxin A has an improved safety profile (RR 0.20, 95% CI 0.01 to 4.00; very low certainty evidence). The evidence for these results is of very low certainty due to serious concerns with risk of bias and imprecision. The study did not report data on treatment success.
One study compared the injection of 60 units botulinum toxin A (n = 21) to myectomy of the internal anal sphincter (n = 21). We are very uncertain whether botulinum toxin A injection improves treatment success (RR 1.00, 95% CI 0.75 to 1.34; very low certainty evidence). No adverse events were recorded. The study reported no data for the other primary outcomes.
One study compared 0.04 mg/kg oral prucalopride (n = 107) once daily to placebo (n = 108) over eight weeks. Oral prucalopride probably results in little or no difference in defecation frequency (MD 0.50, 95% CI −0.06 to 1.06; moderate certainty evidence); treatment success (RR 0.96, 95% CI 0.53 to 1.72; moderate certainty evidence); and adverse events (RR 1.15, 95% CI 0.94 to 1.39; moderate certainty evidence). The study did not report data on symptom resolution.
One study compared transcutaneous electrical stimulation to sham stimulation, and another study compared dietitian‐prescribed Mediterranean diet with written instructions versus written instructions. These studies did not report any of our predefined primary outcomes.
Authors' conclusions
We identified low to moderate certainty evidence that oral lubiprostone may result in little to no difference in treatment success and adverse events compared to placebo. Based on moderate certainty evidence, there is probably little or no difference between oral prucalopride and placebo in defecation frequency, treatment success, or adverse events. For all other comparisons, the certainty of the evidence for our predefined primary outcomes is very low due to serious concerns with study limitations and imprecision. Consequently, no robust conclusions could be drawn.
Keywords: Adolescent; Child; Child, Preschool; Humans; Infant; Bias; Botulinum Toxins, Type A; Botulinum Toxins, Type A/therapeutic use; Constipation; Constipation/therapy; Defecation; Defecation/drug effects; Laxatives; Laxatives/therapeutic use; Lubiprostone; Lubiprostone/therapeutic use; Quality of Life; Randomized Controlled Trials as Topic
Plain language summary
Management of intractable constipation in children
Key messages
• There may be little to no difference between lubiprostone and placebo (dummy treatment) in achieving treatment success in children with intractable constipation. We also found that lubiprostone is probably as safe as placebo.
• There is probably little or no difference between prucalopride, also a laxative, and placebo in frequency of defecation per day, treatment success, and safety.
• It is unclear whether any of the other treatments we looked at are helpful. The evidence is uncertain because of the very low number of study participants and problems with how the research was reported.
• Currently, there is not an agreed‐upon definition for intractable constipation. To develop further evidence on therapies for intractable constipation, researchers must use the same definition. We would encourage further work to come up with an agreed‐upon definition.
What is intractable constipation?
Constipation in children is a common problem. If doctors cannot find an underlying cause for the symptoms, it is called functional constipation. The symptoms of constipation include lower frequency of defecation, bowel incontinence or soiling, and stomach pain. Constipation can be mild and respond well to standard treatments, such as behavioural changes or laxatives. However, it can also be more severe or last for a long time. We defined intractable constipation as constipation that persists despite standard treatments.
What did we want to find out?
We wanted to find out which treatments are helpful and safe for treating children (between 0 and 18 years of age) with constipation that has not responded to standard medical treatments (intractable constipation). This extreme level of constipation can impact a child's life in very negative ways, such as keeping them away from school and other daily activities; causing them pain, discomfort, and general distress; and requiring hospital visits for treatment. Consequently, there is a need to investigate if the tested therapies can help resolve the problem.
What did we do?
We considered any medication, surgical procedure, or therapy to help children with intractable constipation. Each study compared a specific treatment to placebo, no treatment, or another treatment. We were interested in whether the treatments increased the frequency of defecation, improved symptoms, or led to treatment success. We also wanted to know if the treatments were safe, so we looked at whether they caused side effects. We searched for randomised controlled trials (studies in which people are randomly assigned to one of two or more treatment groups) comparing any medical, surgical, or complementary therapy versus no treatment, placebo, or another treatment in children with intractable constipation.
What did we find?
We found 10 studies involving a total of 1278 children. The average age of study participants ranged from around 5 to 10 years. The studies were conducted in Iran, Brazil, Colombia, the USA, Canada, Australia, and several European countries. Studies lasted from one to six months.
The studies made the following comparisons:
• botox injection versus stool softeners;
• erythromycin versus placebo;
• lubiprostone versus placebo;
• rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives;
• biofeedback therapy versus no treatment;
• intrarectal electromotive botox injection versus a usual botox injection;
• botox injection versus myectomy of the internal anal sphincter;
• prucalopride versus placebo;
• transcutaneous electrical stimulation versus sham stimulation; and
• dietitian‐prescribed Mediterranean diet with written instructions versus written instructions.
Main results
• There may be little to no difference between lubiprostone and placebo in achieving treatment success, and there is probably little to no difference in adverse events for this comparison.
• There is probably little or no difference between prucalopride and placebo in defecation frequency, treatment success, and adverse events.
• We do not know whether any of the other treatments looked at in this review are safer or more beneficial than others.
What are the limitations of the evidence?
The evidence is limited due to small participant numbers in the included studies, and because each study looked at a different comparison, both of which resulted in the evidence being imprecise. There were also issues with the way the studies were conducted, which led to concerns that study results might be biased.
How up‐to‐date is this evidence?
This review is current to June 2023.
Summary of findings
Summary of findings 1. Botulinum toxin A injection versus stool softeners for intractable constipation in children.
| Botulinum toxin A injection versus stool softeners | |||||
| Patient or population: children with intractable constipation Setting: secondary care (paediatric surgical clinic), single‐centre, Iran Intervention: botulinum toxin A injection Comparison: stool softeners | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with stool softeners | Risk with botulinum toxin A injection | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (6 months' postintervention) | In 1 study (Ahmadi 2013), the mean defecation interval was reported for the botulinum toxin A injection group (2.6 days), but not for the control group. | ‐ | 88 (1 RCT) | ⊕⊝⊝⊝ Very low a |
|
| Treatment success (measured as children with a defecation interval of fewer than 3 days, 6 months after intervention) | Study population | RR 37.00 (5.31 to 257.94) | 88 (1 RCT) | ⊕⊝⊝⊝ Very low a |
|
| 23 per 1000 | 841 per 1000 (121 to 1000) | ||||
| Adverse events (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded twice due to serious concerns with risk of bias, and once for imprecision (small sample size).
Summary of findings 2. Erythromycin estolate versus placebo for intractable constipation in children.
| Erythromycin estolate versus placebo | |||||
| Patient or population: children with intractable constipation Setting: secondary care (paediatric gastroenterology clinic), single‐centre, Brazil Intervention: erythromycin estolate Comparison: placebo | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with placebo | Risk with erythromycin estolate | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Treatment success (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Adverse events (week 4) | 0 adverse events reported | 0 adverse events reported | Not estimable | 18 (1 RCT) | ⊕⊝⊝⊝ Very low a |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded twice for serious imprecision (small sample size and zero events), and once due to concerns with risk of bias.
Summary of findings 3. Lubiprostone versus placebo for intractable constipation in children.
| Lubiprostone versus placebo | |||||
| Patient or population: children with intractable constipation Setting: secondary care, multicentre (96 sites across the USA, Canada, and Europe) Intervention: oral lubiprostone Comparison: placebo | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with placebo | Risk with lubiprostone | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Treatment success (measured as the number of children with an increase in at least 1 spontaneous bowel movement per week compared to baseline and at least 1 spontaneous bowel movement per week for at least 9 weeks, including 3 of the final 4 treatment weeks, at 12 weeks) | Study population | RR 1.29 (0.87 to 1.92) |
606 (1 RCT) | ⊕⊕⊖⊖ Lowa | |
| 143 per 1000 | 185 per 1000 (124 to 275) |
||||
| Adverse events (number of children with at least 1 adverse event, at 12 weeks) | Study population | RR 1.05 (0.91 to 1.21) | 606 (1 RCT) | ⊕⊕⊕⊖ Moderateb | |
| 564 per 1000 | 592 per 1000 514 to 683 |
||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded twice due to serious imprecision (95% CI overlaps no effect; CI ranges from a small benefit that is less than placebo, to significantly large effect that is more than placebo). bDowngraded once due to imprecision (95% CI overlaps no effect; CI ranges from a small harm that is less than placebo, to a small harm that is more than placebo).
Summary of findings 4. Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives for intractable constipation in children.
| Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives | |||||
| Patient or population: children with intractable constipation Setting: tertiary care (outpatient clinic for functional defecation disorders), single‐centre, the Netherlands Intervention: rectal sodium dioctyl sulfosuccinate and sorbitol Comparison: oral polyethylene glycol laxatives | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with oral polyethylene glycol laxatives | Risk with rectal sodium dioctyl sulfosuccinate and sorbitol | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (measured at the end of the 52‐week study period) | In 1 study (Bongers 2009), the modelled mean (using a linear mixed model) frequency per week was 5.3 days for rectal sodium dioctyl sulfosuccinate and sorbitol, and 3.9 days for polyethylene glycol laxatives. No other variances were reported. | ‐ | 102 (1 RCT) | ⊕⊝⊝⊝ Very low a |
|
| Treatment success (measured as the number of children with at least 3 bowel movements per week and no faecal incontinence, at 52 weeks) | Study population | RR 1.33 (0.83 to 2.14) |
102 (1 RCT) | ⊕⊝⊝⊝ Very low a |
|
| 353 per 1000 | 469 per 1000 (293 to 755) |
||||
| Adverse events (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded twice due to serious concerns with risk of bias, and once for imprecision (small sample size).
Summary of findings 5. Biofeedback therapy versus no intervention for intractable constipation in children.
| Biofeedback therapy versus no intervention | |||||
| Patient or population: children with intractable constipation Setting: secondary care (private paediatric gastroenterology centre), single‐centre, Colombia Intervention: biofeedback therapy Comparison: no intervention | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with no intervention | Risk with biofeedback therapy | ||||
| Non‐fulfilment of Rome IV criteria for functional constipation (timing not reported) | Study population | RR 2.50 (1.08 to 5.79) | 24 (1 RCT) | ⊕⊝⊝⊝ Very low a |
|
| 333 per 1000 | 833 per 1000 (360 to 1000) | ||||
| Frequency of defecation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Treatment success (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Adverse events (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded once due to concerns with risk of bias, and twice for serious imprecision (small sample size).
Summary of findings 6. Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection for intractable constipation in children.
| Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection | |||||
| Patient or population: children with intractable constipation Setting: secondary care (paediatric outpatient clinic), single‐centre, Iran Intervention: intrarectal electromotive botulinum toxin A Comparison: botulinum toxin A injection | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with botulinum toxin A injection | Risk with intrarectal electromotive botulinum toxin A | ||||
| Non‐fulfilment of Rome criteria for functional constipation (Rome II criteria, at 1 month) | Study population | RR 0.96 (0.76 to 1.22) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa |
|
| 833 per 1000 | 800 per 1000 (633 to 1000) | ||||
| Frequency of defecation (measured as the interval in days between defecation, at 1 month) | Study population | ‐ | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa |
|
| The mean score in the control group was 7 days. | MD 0.00 (1.87 lower to 1.87 higher) | ||||
| Treatment success (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Adverse events (total number of adverse events, at 6 months) | Study population | RR 0.20 (0.01 to 4.00) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa |
|
| 67 per 1000 | 13 per 1000 (1 to 267) | ||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; MD: mean difference; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded twice due to serious concerns with risk of bias, and once for imprecision (small sample size).
Summary of findings 7. Botulinum toxin A injection versus myectomy of the internal anal sphincter for intractable constipation in children.
| Botulinum toxin A injection versus myectomy of the internal anal sphincter | |||||
| Patient or population: children with intractable constipation Setting: tertiary care (inpatient paediatrics), single‐centre, United Kingdom Intervention: botulinum toxin A injection Comparison: myectomy of the internal anal sphincter | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with myectomy of the internal anal sphincter | Risk with botulinum toxin A injection | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Treatment success (measured as an improvement in composite symptom severity score, at 12 months) | Study population | RR 1.00 (0.75 to 1.34) |
42 (1 RCT) |
⊕⊝⊝⊝ Very low a |
|
| 810 per 1000 | 810 per 1000 (608 to 1000) |
||||
| Adverse events (12 months) | Study population | Not estimable | 42 (1 RCT) | ⊕⊝⊝⊝ Very low a |
|
| 0 to 1000 | 0 per 1000 (0 to 1000) |
||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded once due to concerns with all risk of bias domains, and twice for serious imprecision (low participant numbers).
Summary of findings 8. Prucalopride versus placebo for intractable constipation in children.
| Prucalopride versus placebo | |||||
| Patient or population: children with intractable constipation Setting: secondary care, multicentre (33 centres across Europe) Intervention: prucalopride Comparison: placebo | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with placebo | Risk with prucalopride | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (measured as mean change in defecation frequency per week from baseline, week 8) | Study population | ‐ | 215 (1 RCT) | ⊕⊕⊕⊝ Moderate a | |
| The mean score in the control group was 1 defecation per week. | MD 0.50 higher (0.06 lower to 1.06 higher) | ||||
| Treatment success (measured as at least 3 spontaneous bowel movements per week and no faecal incontinence in a 2‐week period during weeks 5 to 8 of the study, week 8) | Study population | RR 0.96 (0.53 to 1.72) |
215 (1 RCT) | ⊕⊕⊕⊝ Moderate a | |
| 176 per 1000 | 169 per 1000 (93 to 303) |
||||
| Adverse events (measured as number of participants with at least 1 treatment‐emergent event, at week 8) | Study population | RR 1.15 (0.94 to 1.39) |
215 (1 RCT) | ⊕⊕⊕⊝ Moderate a | |
| 611 per 1000 | 703 per 1000 (574 to 849) |
||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; MD: mean difference; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
aDowngraded one level due to imprecision (small sample size).
Summary of findings 9. Transcutaneous electrical stimulation versus sham stimulation for intractable constipation in children.
| Transcutaneous electrical stimulation versus sham stimulation | |||||
| Patient or population: children with intractable constipation Setting: not reported Intervention: transcutaneous electrical stimulation Comparison: sham stimulation | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with sham stimulation | Risk with transcutaneous electrical stimulation | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Treatment success (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Adverse events (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
Summary of findings 10. Dietitian‐prescribed Mediterranean diet with written instructions versus written instructions for intractable constipation in children.
| Dietitian‐prescribed Mediterranean diet with written instructions versus written instructions | |||||
| Patient or population: children with intractable constipation Setting: paediatric gastroenterology clinic, single‐centre, Greece Intervention: dietitian‐prescribed Mediterranean diet with written instructions Comparison: written instructions | |||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | |
| Risk with written instructions | Risk with dietitian‐prescribed Mediterranean diet | ||||
| Non‐fulfilment of Rome criteria for functional constipation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Frequency of defecation (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Treatment success (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| Adverse events (outcome not reported) | ‐ | ‐ | ‐ | ‐ | ‐ |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval | |||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
Background
Description of the condition
Constipation is one of the most common reasons for referrals to paediatricians, accounting for approximately 3% to 5% of general paediatric consultations. Most of these are for chronic constipation without a physical aetiology, known as functional constipation (Philichi 2018). Worldwide, the estimated prevalence of functional constipation is 9.5% (Koppen 2018). Epidemiological data suggests that constipation is becoming increasingly prevalent in South America, Asia, and Europe (Rajindrajith 2016).
As a clinical entity, constipation can range from mild disease, which responds to maintenance laxative drug treatment, to severe disease, requiring aggressive and invasive treatments. The Rome criteria, currently in its fourth iteration, is a clinically useful tool for defining functional constipation in children and is recognised as the reference standard criteria within most major international clinical guidelines (Hyams 2016).
A universal case definition of intractable constipation remains elusive, but is broadly defined as constipation that does not respond to conventional medical therapy (NICE Guideline 2013; Tabbers 2014). The specific transition point between chronic constipation and intractable constipation is not clear. It is also poorly defined in terms of symptom duration and what constitutes intensive or maximal medical therapy. Regardless of the case definition used to define intractable constipation, the impact of constipation on the child and their caregivers is universally recognised (Rajindrajith 2016). Unresolved constipation can negatively impact health‐related quality of life indicators, with additional implications for providing healthcare systems (Vriesman 2019). In children with constipation, the annual cost of providing medical care is approximately twice that of children who do not suffer from constipation, reflecting the increased outpatient and emergency department visits in the constipated child (Shah 2011).
A factor that complicates clinical recommendations for this condition is the range of case definitions used for intractable constipation. For example, the National Institute of Health and Care Excellence (NICE) defines intractable constipation as constipation that does not respond to sustained and fully optimised medical management (NICE Guideline 2013). These guidelines do not comment on the duration of symptoms or the therapeutic pathways. Similarly, the guidelines jointly published by the North American Society for Pediatric Gastroenterology, Hepatology & Nutrition and European Society for Paediatric Gastroenterology Hepatology and Nutrition (NASPGHAN and ESPGHAN) define intractable constipation as constipation that does not respond to optimum medical treatment of at least three months (Tabbers 2014).
Description of the intervention
While several important organic diseases are associated with constipation, including Hirschsprung's disease, coeliac disease, intestinal neuronal dysplasia, and hypothyroidism, most infantile and childhood constipation is thought to be functional in aetiology (Youssef 2001). In the absence of an organic aetiology for constipation, treatment is aimed at contributing factors, such as pain, poor fluid intake, and psychological barriers. This frequently requires a multidisciplinary approach (NICE Guideline 2013). From a therapeutic perspective, laxative therapy represents the mainstay of medical therapy and is used alongside adjuvant therapies, such as dietary and behavioural modification (Gordon 2016). Osmotic laxatives, such as lactulose and polyethylene glycol, are valuable agents since they can be administered easily to young children in an oral solution (Gordon 2016; Rachel 2020). Stimulant laxatives, for which senna and bisacodyl feature commonly within disimpaction regimens, are available in various preparations, including tablet, liquid, and suppository (Portalatin 2012; Southwell 2020; Vriesman 2020).
How the intervention might work
Irrespective of the specific intervention used, treatment aims to achieve complete disimpaction with minimal discomfort. The range of available interventions reflects the complexity of this challenging condition. Whether delivered via the oral or rectal route (i.e. as a suppository, enema, via transanal irrigation, or via an antegrade continent enema), laxative therapy aims to relieve constipation by increasing the water content of stool, making stools softer, or stimulating peristaltic movements. Botulinum toxin is an acetylcholinesterase inhibitor. It can be injected into the internal anal sphincter, causing clinically reversible muscle paralysis (Irani 2008).
The mechanisms of transanal irrigation in alleviating constipation include simple mechanical washout and stimulation of peristalsis (Emmett 2015). In principle, transcutaneous electrical stimulation (TES) generates an electrical impulse that acts as a stimulus for appropriate peripheral nerves (Ng 2016). Other theories postulated are that TES acts centrally and rebalances excitatory and inhibitory signals, resulting in the normalisation of the neural drive (Sluka 2003). Colonic resection, with anastomosis or bowel‐diverting stoma, is thought to be effective by eliminating a dysfunctional mega‐rectum or colon (Siminas 2015).
Why it is important to do this review
The most severe cases of intractable constipation may necessitate invasive treatments, such as antegrade enemas, bowel resection, and the use of neuromodulation. Since many of these interventions are invasive, expensive, and, in some cases, irreversible, it is crucial to identify which interventions are the most effective and in which clearly defined groups of children. Furthermore, there are potential adverse effects associated with all the treatments described. For instance, laxative therapy can cause abdominal discomfort and excessive flatulence (McClung 2004). Transanal irrigation is widely considered a safe treatment, but bowel perforation, water intoxication, and chemical colitis have been reported (Emmanuel 2010). The antegrade continence enema (ACE) procedure is associated with peritonitis, stomal stenosis, stomal leakage, and high relapse rates (Chan 2016; Siddiqui 2014). Surgical interventions may be associated with additional complications, such as infection, bowel adhesion, and chronic pain. Given the risks associated with the spectrum of treatments available for children with intractable constipation, it is crucial to determine their efficacy and evaluate their safety profiles.
There are several Cochrane reviews on paediatric constipation. These include a comparison between osmotic and stimulant laxatives for childhood constipation (Gordon 2016), and an evaluation of the efficacy of probiotics for treating chronic childhood constipation (Gordon 2016a). There is no review on childhood constipation that is considered intractable in severity. Consequently, there is an urgent need to identify the most efficacious management strategies for children with intractable constipation, to reduce the burden of morbidity and long‐term sequelae of refractory disease (Southwell 2020). This review will interest caregivers, frontline clinicians, commissioning groups, and those developing guidelines and policies.
Objectives
To evaluate the efficacy and safety of treatments used for intractable constipation in children.
Methods
Criteria for considering studies for this review
Types of studies
We included all types of randomised controlled trials (RCTs), including cross‐over RCTs. Cross‐over RCTs were only eligible for inclusion if the results were presented separately for each stage of the trial. We planned to include cluster‐RCTs, but did not encounter any.
Quasi‐randomised trials (controlled trials that use inappropriate strategies of allocating interventions) were ineligible (Higgins 2023).
Types of participants
We included participants between 0 and 18 years of age with a diagnosis of intractable functional constipation. The case definition used in this review was constipation that had not responded to conventional medical therapy (or as defined by the individual studies). Studies that included participants with an underlying organic cause for constipation, such as thyroid disease, Hirschsprung's disease, or coeliac disease, were excluded.
For future update(s), if we come across studies involving only specific subsets of eligible participants, they will be included if stratified data are available for extraction and analysis.
Types of interventions
We included studies that compared any pharmacological, non‐pharmacological, or surgical therapy to placebo or another active comparator.
Treatments and interventions that were eligible for inclusion included, but were not limited to, the following.
-
Any pharmacological therapy intended to treat constipation.
These included osmotic laxatives, stimulant laxatives, stool bulking agents, faecal softeners, and cholinergic agents. We considered all dosing regimens and routes of administration, including oral, rectal, or via an antegrade continence enema.
-
Botulinum toxin injection.
We considered all doses, number of treatments, and treatment intervals. Only injection into the internal and/or external anal sphincter was considered.
-
Transanal irrigation.
We considered all irrigant solutions, volume of irrigant, frequency of treatment, and number of treatments.
-
Transcutaneous electric stimulation.
We considered treatment that used any device to deliver electrical stimulation, provided it was applied either transabdominally or sacral. We considered all treatment intensities, number of treatments, and treatment lengths.
-
Any treatment involving needle insertion at acupuncture points, pain points, or trigger points.
We considered any session length, session frequency, and total treatment length.
Pelvic floor physiotherapy of any duration or frequency.
Definitive surgical interventions, including percutaneous endoscopic colostomy, stoma formation, and sphincter reconstruction.
Types of outcome measures
The outcome measures selected in this review incorporated the recommendations of the Rome Foundation Pediatric Subcommittee on Clinical Trials (Koppen 2018a).
Primary outcomes
Symptom resolution, defined as non‐fulfilment of any Rome criteria for functional constipation, measured at the end of the study period.
The frequency of defecation, measured at the end of the study period. This could be either the interval between defecations, the number of defecations per unit of time, or change from baseline.
Treatment success (as defined by the original studies), reported dichotomously.
Adverse events (as defined by the original studies), reported dichotomously.
Secondary outcomes
Stool consistency, reported dichotomously, using a scaled score (i.e. the Bristol stool chart; Lewis 1997), or continuously as a change from baseline.
Painful defecation, reported dichotomously, using a scaled score, or continuously as a change from baseline.
Quality of life, reported dichotomously, using a scaled score, or continuously as a change from baseline.
Faecal incontinence frequency. This could be either the interval between episodes of incontinence, the number of incontinence episodes per unit of time, or a change from baseline.
Abdominal pain, reported dichotomously, using a scaled score, or continuously as a change from baseline.
Admission to hospital for disimpaction, reported dichotomously, continuously, or change from baseline.
School absence, reported dichotomously, continuously, or change from baseline.
Search methods for identification of studies
We used the following methods to identify studies for inclusion.
Electronic searches
We searched the following databases for relevant studies. The search was performed on 23 June 2023.
Cochrane Central Register of Controlled Trials (CENTRAL) via the Cochrane Library (Issue 6 of 12, June 2023; Appendix 1).
MEDLINE via Ovid SP (1946 to 22 June 2023; Appendix 2).
Embase via Ovid SP (1974 to 2023 Week 24; Appendix 3).
ClinicalTrials.gov (www.clinicaltrials.gov; up to 23 June 2023; Appendix 4).
World Health Organization International Clinical Trials Registry Platform (ICTRP; www.who.int/trialsearch/; up to 23 June 2023; Appendix 5).
There were no limitations on document type, publication status, date, or language (Aali 2021). Where possible, studies published in a non‐English language were professionally translated in full.
Searching other resources
We searched the bibliographies of included studies for references to RCTs that were relevant to the review.
Data collection and analysis
We used an intention‐to‐treat analysis for dichotomous outcomes where participants with missing outcomes were assumed to be treatment failures.
Selection of studies
As described below, two review authors (CGC and SR) independently screened studies for eligibility at the title, abstract, and full‐text review stages. We used the systematic review system Covidence to upload search results, screen abstracts and full‐text study reports, and export data into electronic spreadsheets. We selected studies in accordance with the recommendations in the Cochrane Handbook for Systematic Reviews of Interventions (Lefebvre 2021).
Merged search results from different sources using the reference system, in which duplicate records of the same report were removed (i.e. records reporting the same journal, title, volume, and page range).
Screened the titles and abstracts of all records yielded by the search, discarding those that were clearly irrelevant and progressing all others deemed potentially relevant.
Retrieved the full‐text reports of potentially relevant records.
Linked multiple reports of the same study. We did not discard secondary reports of a study as they may contain valuable information.
We carefully examined the full‐text reports, selecting studies based on our inclusion and exclusion criteria (see Criteria for considering studies for this review). For each study excluded at the full‐text review stage, we assigned a reason for exclusion and entered the record into the Characteristics of excluded studies table.
We corresponded with study authors to request further information when required to clarify study eligibility. We did not exclude studies for the sole reason that measured outcome data were not reported.
Adjudication between review author decisions took place only at the full‐text review stage. Initially, differences in assessment between review authors were managed through discussion. Where disagreement persisted, adjudication by a third review author (MG) took place.
Data extraction and management
We developed a data extraction form a priori, as per the recommendations in the Cochrane Handbook for Systematic Review of Interventions, and piloted the form on two random RCTs to ensure it was fit for purpose (Li 2021). Two review authors (CGC and SR) independently used the data extraction form to extract and record data. Following this, extractions were compared, with any differences resolved through discussion. A third review author adjudicated in instances of persisting disagreement (MG).
We recorded the study title, author list, year of publication, and country of publication for each study. We then extracted the following data for each study.
Methods: study design, setting (i.e. hospital, primary care), and study period (period of time during which participants were enrolled).
Population: age, sex, duration of symptoms, case definition of intractable constipation. We used the explicit definition if stated. If not stated, we recorded the characteristics of participants that led to inclusion as 'intractable' constipation. This included, where applicable, the length of unsuccessful therapy prior to enrolment, the number and detail of therapies trialled without success, or a combination of both, as described by the original study. We also recorded the number of participants originally assigned to each treatment group.
Intervention: number of treatment arms within the trial and a description of the intervention (if treatment was non‐pharmacological). For interventions involving the administration of medications, we recorded the name and therapeutic class of the agent, preparation, route of administration, dose, frequency of administration, and length of treatment.
Control: description of the placebo, active comparator (class of therapy, preparation, route, dose, frequency, and length of treatment).
Outcomes: outcomes reported (as per Primary outcomes and Secondary outcomes) and the timing of study endpoints. We collected outcome data at the study endpoint and other time points, if reported. This included the number of cases that filled the Rome criteria for functional constipation, frequency of defecation, stool consistency, frequency and severity of painful defecation, frequency of faecal incontinence, and frequency and severity of abdominal pain. We also recorded treatment success alongside the definition for this (if different from non‐fulfilment of Rome criteria for functional constipation) and adverse events. We noted whether studies actively monitored for adverse events, or if they simply provided spontaneous reporting of adverse events. For continuous outcomes, we extracted the mean value and standard deviation (SD) at baseline and at study endpoints. If the mean value was not provided, we extracted the median or interquartile range instead.
Other: trial registration details, conflicts of interest, funding details, risk of bias assessments. We also recorded details of any email communication with the study authors.
Assessment of risk of bias in included studies
Two review authors (CGC and SR) independently assessed risk of bias in the included studies using Cochrane's RoB 1 tool, as described in Appendix 6 (Higgins 2011; Higgins 2017). Any differences in judgement between review authors were discussed, with a third review author (MG) adjudicating as needed.
We assessed the following domains as having a low, high, or unclear risk of bias.
Sequence generation (i.e. was the allocation sequence adequately generated?)
Allocation concealment (i.e. was allocation concealment concealed?)
Blinding of participants and personnel and outcome assessors (i.e. was knowledge of the allocated intervention adequately prevented during the study?)
Incomplete outcome data (i.e. were incomplete outcome data adequately addressed?)
Selective outcome reporting (i.e. were study reports free of the suggestion of selective outcome reporting?)
Other potential sources of bias (i.e. did the study appear to be free of other problems that could have put it at a high risk of bias?)
We considered studies that received a judgement of high risk of bias in one or more domain(s) to be at high risk of bias overall; those that received a judgement of low risk of bias in all domains to be at low risk of bias overall; and those that received a judgement of unclear risk of bias in one or more domains to be at unclear risk of bias overall.
Measures of treatment effect
We determined measures of treatment effect as per the recommendations in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2023). We analysed data on an intention‐to‐treat basis using RevMan software (RevMan 2024).
For dichotomous outcomes, we calculated the risk ratio (RR) and corresponding 95% confidence intervals (CI) using a random‐effects model.
For continuous outcomes measured on the same scale, we extracted mean change or endpoint data to calculate a mean difference (MD) with corresponding 95% CIs. Where studies used different scales to evaluate the same outcome, we planned to calculate the standardised mean difference (SMD) and 95% CI.
Unit of analysis issues
The unit of analysis was the participant. For studies comparing more than two intervention groups, we planned to make multiple pairwise comparisons between all possible pairs of intervention groups. To avoid double‐counting, we planned to divide shared intervention groups evenly among the comparisons. For dichotomous outcomes, we planned to divide both the number of events and the total number of participants. For continuous outcomes, we would only divide the total number of participants, and leave the means and SDs unchanged.
We only included cross‐over studies if data were separately reported before and after cross‐over, and only used pre‐cross‐over data. We did not anticipate finding any cluster‐RCTs.
Dealing with missing data
We assessed missing data for each included study. We attempted to contact the primary study authors to request any relevant missing data. If we received no response from study authors after two attempts, we used the information available to us. We estimated missing SDs using relevant statistical methods as described in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2023). We judged studies that failed to report measures of variance as at high risk of reporting bias.
Assessment of heterogeneity
We intended to test for statistical heterogeneity of effect size between studies using the Chi² test, considering a P value of less than 0.10 as indicative of significant heterogeneity. We planned to quantify and represent inconsistency using the I² statistic, interpreting values as follows, as described in the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2021):
0% to 40%: might not be important;
30% to 60%: may represent moderate heterogeneity;
50% to 90%: may represent substantial heterogeneity;
75% to 100%: considerable heterogeneity.
We would not have pooled data in a meta‐analysis if we detected a considerable degree of statistical heterogeneity (I² > 75%). We planned that if there was considerable statistical heterogeneity, we would conduct sensitivity analyses to explore possible explanations.
Assessment of reporting biases
We initially compared the outcomes listed within study protocols to those reported in the published manuscript. If we did not have access to the protocol, we compared the outcomes listed in the methods section of the published manuscript to the results section. We planned that if pooled analyses included 10 or more studies, we would construct funnel plots to investigate publication bias (Egger 1997).
Data synthesis
We provided a narrative synthesis of the key characteristics for the included studies (i.e. the number of included studies; study design of the included studies; characteristics of participants across studies; interventions used in both treatment and control groups; and outcome measures reported).
We planned to combine data for meta‐analysis from individual studies when the interventions, patient groups, and outcomes were similar, as deemed by review author consensus. We planned to use a random‐effects model to pool data. We would not have pooled data in a meta‐analysis if we detected considerable heterogeneity (I² > 75%).
Given the diversity of interventions identified for the treatment of intractable constipation, we grouped interventions for analysis using the following classification.
Laxative therapy (single‐agent therapy)
Laxative therapy (combination therapy)
Non‐laxative pharmacological interventions
Colonic irrigation
Surgical interventions
Alternative therapies
Where we were unable to carry out a meta‐analysis (e.g. data were too heterogeneous, high statistical heterogeneity, or too few studies), we presented a narrative summary of the results, which we have reported according to the Synthesis Without Meta‐analysis (SWiM) guideline (Campbell 2020).
Subgroup analysis and investigation of heterogeneity
We planned to conduct subgroup analyses based on the following factors.
Type of medication preparation
Length of the intervention
Definition used to define intractable constipation
Characteristics of participants' intractable constipation, including length of unsuccessful therapy prior to enrolment and number/type of failed therapies prior to enrolment
Sensitivity analysis
We planned to undertake sensitivity analyses on the primary outcomes to assess whether the findings of the review were robust to the decisions made during the review process. In particular, we planned to exclude studies at high or unclear risk of selection bias (due to the method of allocation concealment) and performance bias. Where data analyses included studies with reported and estimated SDs, we planned to exclude those studies with estimated SDs to assess whether this affected the findings of the review. We also planned to investigate whether the choice of model (fixed‐effect versus random‐effects) may have affected the results.
Summary of findings and assessment of the certainty of the evidence
We created summary of findings tables for our predefined primary outcomes (see Primary outcomes) for all comparisons using GRADEpro GDT software. Two review authors (CGC and SR) independently assessed the overall certainty of the evidence for the primary outcomes using the GRADE approach (Guyatt 2013). We assessed the certainty of the evidence for each outcome as high, moderate, low, or very low based on the five GRADE considerations (risk of bias, inconsistency, imprecision, indirectness, and publication bias), as follows.
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
Results
Description of studies
Results of the search
Our search (see Electronic searches) retrieved 10,023 records. After removal of duplicates, 6834 records remained for screening. We excluded 6785 records based on title and abstract. We obtained the full texts of the remaining 49 records. We excluded 25 studies (see Characteristics of excluded studies). Two records are awaiting classification (Characteristics of studies awaiting classification). We identified one ongoing study (see Characteristics of ongoing studies).
We included 10 studies in the review (see Characteristics of included studies). A study flow diagram is shown in Figure 1.
1.
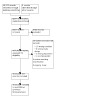
Study flow diagram.
Included studies
A summary of the treatments used in the intervention and control arms and trial registration details is presented in Table 11. Case definitions used for intractable constipation, the age of included participants, time point of outcome measurements, and outcomes reported are presented in Table 12.
1. Characteristics of included studies ‐ interventions, length of intervention, trial registrations.
| Study ID | Intervention group | Description of the intervention | Control group | Description of the control | Concurrent therapy | Length of intervention | Trial registered | Trial registry outcomes published |
| Ahmadi 2013 | Botulinum toxin injection | Botulinum toxin A was injected under general anaesthesia into 3 regions of the anal sphincter (3, 6, and 9 o'clock). Total dose of botulinum toxin A was 160 units (80 units at 6 o'clock, 40 units at 3 o'clock, and 40 units at 9 o'clock). The toxin was injected into both internal and external anal sphincters. | Stool softeners (no further details provided) | Not recorded | Both groups had rectal disimpaction with phosphate and saline enemas before starting the study. | One‐off intervention | Not registered | N/A |
| Bellomo‐Brandão 2003 | Erythromycin estolate | Oral erythromycin estolate 20 mg/kg/day (split into 4 doses), given 6 hours apart. The maximum daily dose was 1000 mg. | Placebo | No further details | Children were admitted to hospital for faecal impaction removal by consecutive enemas. After colonic cleansing, an oral osmotic laxative was prescribed as maintenance therapy to establish a regular frequency of bowel movements. Lactulose (667 mg/mL) or magnesium hydroxide (80 mg/mL) was administered at a daily dose of 2 mL/kg, with a maximum dose of 60 mL. Participants were instructed to use a saline‐glycol enema when there was no spontaneous stool movement after a 72‐hour period. The dose of initially prescribed laxative was maintained, reduced, or increased according to stool frequency, faecal consistency, and stool passage conditions which occurred during the previous 2 weeks. The laxative dose was reduced when participants mentioned liquid faecal consistency and increased evacuation in the presence of hard/scybalous faecal consistency or a stool frequency of less than 3 per week or enema requirement during the previous period. | 4 weeks | Not registered | N/A |
| Benninga 2021 | Lubiprostone | Oral lubiprostone 12 μg twice a day (if weight < 50 kg) or oral lubiprostone 24 μg twice a day (if weight > 50 kg) | Placebo | No further details | Nil | 12 weeks | NCT02042183 (prospectively registered) | Yes |
| Bongers 2009 | Rectal sodium dioctyl sulfosuccinate + sorbitol enema | 3 rectal enemas weekly during the first 3 months. Thereafter, this frequency was reduced by 1 enema per week every 3 months. Rectal enema consisted of 120 mL sodium dioctyl sulfosuccinate and sorbitol. Conventional therapy was delivered alongside the intervention. | Oral polyethylene glycol laxative | Oral laxative therapy consisted of polyethylene glycol, with a starting dose of 0.5 g/kg. If treatment was considered insufficient, the dose was optimised to a maximum of 1.5 g/kg. A rectal enema or bisacodyl suppository of 5 mg was only prescribed in case of reoccurrence of faecal impaction. | All children underwent rectal disimpaction by rectal enema (120 mL sodium dioctyl sulfosuccinate and sorbitol) on 3 consecutive days to achieve an empty rectum before starting the treatment trial. If rectal disimpaction was unsuccessful, rectal enemas were continued for a maximum of 7 days. | 52 weeks | ISRCTN99089299 (retrospectively registered) | Yes |
| Castilla 2021 | Biofeedback therapy | No further details provided. | No intervention | N/A | Nil | Not recorded | Not registered | N/A |
| Kajbafzadeh 2020 | Intrarectal electromotive botulium toxin A | Electromotive therapy involved 20 minutes of current at frequency 2800 Hz, interval 50 μs, and amplitude of 10 to 15 mA (after colonic irrigation). BoNTA was added at a dose of 10 IU/kg to an irrigated rectum that had been filled with saline solution to its maximal capacity. | Botulinum toxin A injection | Internal anal sphincter injection of botulinum toxin A was performed in 4 quadrants at a dose of 10 IU/kg. The procedure was performed under general anaesthetic. | Oral polyethylene glycol (PEG, 1 to 1.5 g/kg for a maximum of 7 days) and bowel washing or rectum irrigation were advised to participant with a large faecal mass in the rectum for faecal disimpaction. The laxative regimen (PEG, 0.2 to 0.6 g/kg/day) was continued for all children during the study. The dose of laxative was reduced in children who responded to the treatment after 1 month follow‐up and was then discontinued in children who sustained their response to treatment after 6 months' follow‐up. Both groups also received conventional treatment including advice to consume a high‐fibre diet, hydration, toilet training, correct defecation posture, to sit on the toilet 3 times a day after mealtimes in a relaxed position for 5 minutes. | One‐off intervention | IRCT20111229008554N4 (retrospectively registered) | Yes |
| Keragiozoglou‐Lampoudi 2012 | Personalised Mediterranean diet | In addition to paediatric gastroenterologist written instruction, each child and their parent had a further same‐day appointment with a registered dietitian who prescribed a personalised diet (7‐day diet plan) based on the Mediterranean‐type eating plan, and calculated to cover the personal energy, nutrient, water, and fibre requirements of paediatric patients. | Parents received written instructions about their children’s diet with examples explained by a paediatric gastroenterologist. | Parents were given written instruction about their children's diet with examples provided by a paediatric gastroenterologist. | Nil | One‐off intervention | Not registered | N/A |
| Keshtgar 2007 | Botulinum toxin A injection | Injection of botulinum toxin into the internal anal sphincter. Performed under general anaesthesia, 15 units of botulinum type A were injected per quadrant (total dose = 60 units) into the internal anal sphincter. | Myectomy of the anal sphincter | Myectomy of the internal anal sphincter. This was achieved by making a small transverse incision on the dentate line at the 3 o'clock position, and a plane developed between the mucosa and muscle. A strip of the internal anal sphincter was excised proximally into the distal rectum with length of approximately 5 cm and a width of approximately 0.5 cm. | Children stayed in the hospital postoperatively and received intensification of laxative treatment, toilet training, and were assessed by a child psychologist. Children were encouraged to establish a regular pattern or bowel movement and laxative intake (including methycellulose as a stool softener and senna as a bowl stimulant). Movicol and sodium picosulfate were given if a child did not pass bowel motion for 3 days. | One‐off intervention | Not registered | N/A |
| Mugie 2014 | Prucalopride | For those weighing ≤ 50 kg, an initial dose of 0.04 mg/kg body weight, once daily was used). After 4 weeks, the dose could be increased to 0.06 mg/kg or decreased to 0.02 mg/kg, based on treatment response and the presence of safety/tolerability. Children who underwent dose adjustment remained on that dose for the remainder of the double‐blind period. Children weighing > 50 kg received 2 mg prucalopride. | Placebo | Oral placebo (identical in appearance and taste to the interventional agent) | Children were not to change their lifestyle or diet. Legal guardians of children aged ≥ 4 years were instructed to continue with toilet training (defined as at least 3, 5‐minute visits to the toilet in a silent, relaxed atmosphere after each meal) during the study. Dosing schedule was 1 to 3 hours before evening meals. Laxatives and agents that influence bowel habits were not permitted during the run‐in period. If the child did not have a bowel movement for ≥ 3 consecutive days, they could take 5 mg bisacodyl or 7.5 mg/mL sodium picosulfate droplets (1 droplet per 5 kg body mass) for rescue purposes. If the standard dose was insufficient, an increase was allowed after discussion with the investigator. If the child had no bowel movements, an enema (e.g. sodium dioctyl sulfosuccinate and sorbitol) or oral agent (e.g. PEG 3350) could be administered to remove the impaction. | 8 weeks | NCT01330381 (prospectively registered) | Yes |
| Southwell 2012 | Transcutaneous electrical stimulation | 12 x 20‐minute sessions of real interferential therapy (IFT) over 4 weeks. 2 electrodes were placed paraspinally and 2 over the abdomen. Active stimulation was applied by physiotherapists at a comfortable intensity (< 40 mA, carrier frequency 4 kHz, varying beat frequency 80 to 150 Hz). | Sham therapy by a physiotherapist | Placebo therapy (as per the intervention group, but with a frequency and sweep of 0) | Nil | 4 weeks | Not registered | N/A |
IU: international units; N/A: not applicable; PEG: polyethylene glycol
2. Characteristics of included studies ‐ method of diagnosis, treatment groups, outcomes reported.
| Study ID | Case definition for intractable constipation | Age | Number of participants randomised | Time points of outcome measurements | Outcomes reported* |
| Ahmadi 2013 | Chronic constipation for more than 3 months, and who had not responded to medical treatment | 2 to 12 years (range), 5 years (mode) | 88: IG 44, CG 44 | 6 months | 1b, 1c, 2a, 2b, 2d |
| Bellomo‐Brandão 2003 | Chronic or lifelong history of constipation, including the presence of megarectum or faecal impaction | IG 9.7 ± 3.0, CG 3.5 ± 2.6 years (mean) | 21 (not split into IG and CG) | Week 2 and 4 | 1d |
| Benninga 2021 | Primary functional constipation according to the Rome III criteria. Email communication with the authors confirmed that children with constipation refractory to medical therapy were considered eligible for inclusion. | IG 11, CG 11 years (mean) | 606: IG 404, CG 202 | Week 2, 4, 6, 8, 10, 12 | 1c, 1d, 2b, 2d, 2e |
| Bongers 2009 | Constipation for at least 2 years and unresponsive to conventional treatment | IG 10.5, CG 11.0 years (mean) | 102: IG 51, CG 51 | Week 12, 26, 39, 52 | 1b, 1c, 2b, 2d, 2e |
| Castilla 2021 | Diagnosis based on Rome IV criteria and pharmacological treatment without response for more than 2 years | 10.6 (median) | 24: IG 12, CG 12 | Not reported | 1a |
| Kajbafzadeh 2020 | Intractable functional constipation based on Rome III criteria. All children had failed to respond to at least 6 months of medical management (dietary modification and use of laxatives), biofeedback, and interferential electrical stimulation. | IG 7.1 ± 2.3, CG 7.3 ± 2.4 (mean) | 60: IG 30, CG 30 | Month 1 and 6 | 1a, 1b, 1d, 2a, 2b, 2c, 2d |
| Keragiozoglou‐Lampoudi 2012 | Functional constipation diagnosed according to the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition criteria. Constipation had to be refractory to treatment by a paediatrician for several months or presented with complications, or both. | 4.4 years (mean) | 86: IG 44, CG 42 | Week 4 | ‐ |
| Keshtgar 2007 | Chronic idiopathic constipation, defined as failure to respond to laxative treatment and manual evacuation of stool with or without anal dilatation under anaesthesia | IG 10, CG 8.6 years (median) | 42: IG 21, CG 21 | Month 1, 6, 12 | 1c, 1d, 2b, 2d |
| Mugie 2014 | Functional constipation for at least 3 months based on Rome III criteria | 8.3 ± 4.5, CG 8.2 ± 4.7 years (mean) | 215: IG 107, CG 108 | Week 2, 4, 8 | 1b, 1c, 1d, 2b, 2c, 2d, 2e |
| Southwell 2012 | Functional constipation for at least 3 months based on Rome II criteria | IG 12.1, CG 11.4 years (mean) | 46 | Week 8 | 2c |
CG: control group; IG: intervention group
*Outcomes reported
1a. Symptom resolution, defined as non‐fulfilment of Rome criteria for functional constipation
1b. Frequency of defecation
1c. Treatment success, as defined by the original study
1d. Adverse events
2a. Stool consistency
2b. Painful defecation
2c. Quality of life
2d. Faecal incontinence
2e. Abdominal pain
2f. Admission to hospital for disimpaction
2g. School absence
Settings
Ten RCTs met our inclusion criteria (Ahmadi 2013; Bellomo‐Brandão 2003; Benninga 2021; Bongers 2009; Castilla 2021; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Keshtgar 2007; Mugie 2014; Southwell 2012). Eight were single‐centre studies (Ahmadi 2013; Bellomo‐Brandão 2003; Bongers 2009; Castilla 2021; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Keshtgar 2007; Southwell 2012), and two were multicentre studies (Benninga 2021; Mugie 2014). One study each was conducted in Australia (Southwell 2012), Colombia (Castilla 2021), Greece (Keragiozoglou‐Lampoudi 2012), Brazil (Bellomo‐Brandão 2003), the Netherlands (Bongers 2009), and the UK (Keshtgar 2007). Two studies were conducted in Iran (Ahmadi 2013; Kajbafzadeh 2020). Mugie 2014 included 33 sites across Europe, and Benninga 2021 included 96 sites across Europe, Canada, and the USA. One study used a cross‐over design (Bellomo‐Brandão 2003); we extracted data relating to the pre‐cross‐over phase of this study, as separate data were presented for each phase.
Participants
The 10 RCTs included 1278 participants, ranging from 21 participants in Bellomo‐Brandão 2003 to 606 participants in Benninga 2021. The mean age of participants was less than 5 years in one study (Keragiozoglou‐Lampoudi 2012), between 5 and 10 years in five studies (Ahmadi 2013, Bellomo‐Brandão 2003; Kajbafzadeh 2020; Keshtgar 2007; Mugie 2014), and greater than 10 years in four studies (Benninga 2021; Bongers 2009; Castilla 2021; Southwell 2012).
Several definitions were used to define intractable constipation. Five studies explicitly referred to Rome criteria, with one referencing Rome II (Southwell 2012); three referencing Rome III (Benninga 2021; Kajbafzadeh 2020; Mugie 2014); and one referencing Rome IV (Castilla 2021). Seven studies defined intractable constipation as refractory to medical treatment and included various minimum durations of chronicity. This was three months in Ahmadi 2013, Keragiozoglou‐Lampoudi 2012, Mugie 2014, and Southwell 2012; six months in Kajbafzadeh 2020; and two years in Bongers 2009 and Castilla 2021. Two studies made no reference to a minimum length of disease or absolute failure to respond to medical therapy, or both, but instead described inclusion based on intractable constipation‐defining clinical features. Bellomo‐Brandão 2003 defined intractable constipation as "chronic or lifelong history of constipation, including the presence of megarectum or faecal impaction". Keshtgar 2007 used the definition "failure to respond to laxative treatment and manual evacuation of stool with or without anal dilation under anaesthesia".
Interventions
One study described the use of single‐agent laxative therapy. In Bongers 2009, participants received three rectal enemas (120 mL sodium dioctyl sulfosuccinate and sorbitol) weekly for three months, reduced by one enema per week every three months, up to 12 months. This was compared to conventional therapy of education, behavioural strategies, and oral laxatives (polyethylene glycol, 0.5 g/kg/day, increased to 1.5 g/kg/day if treatment response was insufficient). No study investigated combination laxative therapy.
Three studies described the use of non‐laxative pharmacological interventions. In Bellomo‐Brandão 2003, participants received oral erythromycin estolate (20 mg/kg/day, split into four doses) for four weeks. This was compared to placebo therapy (no further details provided). In Benninga 2021, participants received oral lubiprostone (12 μg or 24 μg, depending on weight) for 12 weeks. This was compared to placebo (identical soft gelatin capsules). In Mugie 2014, participants received oral prucalopride (0.04 mg/kg once daily for children < 50 kg or 2 mg for children > 50 kg). Participants in the control group received an oral placebo (identical in appearance and taste).
Three studies described surgical interventions. In Ahmadi 2013, participants underwent an injection of botulinum toxin into the anal sphincter, performed under general anaesthetic (160 units in three regions). This was compared to stool softener therapy (no further details provided). In Kajbafzadeh 2020, participants received intrarectal electromotive therapy under general anaesthetic (20 minutes, 2800 Hz, interval 50 μs, amplitude 10 to 15 mA). This was compared to botulinum toxin A injection only. In Keshtgar 2007, participants received an injection of botulinum toxin A into the internal anal sphincter (60 units split into four quadrants). This was compared to myectomy of the internal anal sphincter.
Two studies described alternative therapies. In Castilla 2021, participants received biofeedback therapy (no further details provided). This was compared to no biofeedback therapy. In Southwell 2012, participants received interferential therapy (12 x 20‐minute sessions) or sham therapy, delivered over a four‐week period.
In Keragiozoglou‐Lampoudi 2012, participants and their parents or guardian received an appointment with a registered dietitian who prescribed a personalised Mediterranean diet, in addition to paediatric gastroenterologist written instruction. This was compared to written instruction only.
There was a range of different approaches to pre‐treatment and the use of concurrent therapies across the included studies. In Ahmadi 2013, all participants had rectal disimpaction with phosphate and saline enemas before receiving either botulinum toxin A injection or stool softeners. In Bellomo‐Brandão 2003, all participants were admitted to hospital for faecal impaction removal by enemas, followed by oral osmotic laxatives as maintenance therapy to ensure a regular frequency of bowel movements, before starting either erythromycin or placebo. No pre‐treatment or concurrent therapy was reported in Benninga 2021, Castilla 2021, Kajbafzadeh 2020, and Southwell 2012. In Bongers 2009, all participants underwent rectal disimpaction by rectal enemas on three consecutive days to achieve an empty rectum before continuing with either rectal enemas or oral laxatives. In Keragiozoglou‐Lampoudi 2012, all participants received treatment with pharmacological agents as per NASPGHAN guidelines, alongside either a dietitian‐prescribed Mediterranean diet or written instruction. In Keshtgar 2007, all children received intensification of laxative treatment, toilet training, and child psychologist assessment following either injection of botulinum toxin A into the internal anal sphincter or myectomy of the internal anal sphincter. In Mugie 2014, all participants were instructed to not consume any pharmacological agents that may influence bowel movements during the run‐in period prior to either oral prucalopride or placebo, the only study to do so.
Funding sources and conflicts of interest
Seven studies did not provide details on funding sources (Ahmadi 2013; Bellomo‐Brandão 2003; Benninga 2021; Bongers 2009; Castilla 2021; Keragiozoglou‐Lampoudi 2012; Keragiozoglou‐Lampoudi 2012). Funding was received from a university in one study (Kajbafzadeh 2020), from a pharmaceutical company in one study (Mugie 2014), and a national medical research council in one study (Southwell 2012).
Five studies did not provide details on conflicts of interest (Ahmadi 2013; Bellomo‐Brandão 2003; Benninga 2021; Castilla 2021; Keshtgar 2007). Three studies declared no conflicts of interest (Bongers 2009; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012). Two studies declared conflicts of interest: in Mugie 2014, several authors were employees of the pharmaceutical company providing the interventional agent, and in Southwell 2012, one author co‐owns a patent on the interventional approach (interferential therapy) used within the study.
Excluded studies
We excluded 25 studies at the full‐text stage; they are listed, along with the reasons for their exclusion, in Characteristics of excluded studies.
Fifteen studies did not include participants with intractable constipation, with six including children with disease less than three months in duration, and nine including children who had not previously been treated with optimal medical therapy. One study did not involve an intervention, and a further four studies were excluded due to study design. Two studies did not present data for the paediatric population as a subgroup of the total study population. Two studies focused solely on adults.
Two studies required further discussion among the review authors to determine eligibility. In Keshtgar 2005, there was no description of the minimum duration of symptoms a child must experience to be eligible for inclusion. Within the results section of the report, the length of symptoms is presented as a median and range for both the intervention and control groups. Since at least one participant within the intervention group had a disease duration of fewer than three months, this study was excluded. In Thomson 2008, children receiving doses of stimulant laxatives at the higher end of the dosing spectrum, with no clinical effect, were not considered for inclusion. Given that stimulant laxatives, especially escalating dosages, is a component of optimal medical therapy, we excluded this study as failing to include children with treatment‐resistant intractable constipation.
Studies awaiting classification
Two studies are awaiting classification (see Characteristics of studies awaiting classification). The Australian New Zealand Clinical Trials Registry (ANZCTR) registration ACTRN12620000131954 does not describe the length of disease or previously failed therapy for inclusion. Similarly, the ClinicalTrials.gov record NCT05035784 does not describe the minimum disease length or failed therapy required for inclusion. The full‐text study reports, once available, will allow for judgements on whether participants had disease consistent with intractable constipation. ACTRN12620000131954 was due to complete recruitment in February 2021, and NCT05035784 was due to complete recruitment in March 2022.
Ongoing studies
We identified one ongoing trial (see Characteristics of ongoing studies) (NCT05059756). The study protocol for this trial states an inclusion criterion that aligns with the definition of intractable constipation used within this review. It would be the first study to investigate percutaneous tibial nerve stimulation and pelvic floor rehabilitation in this patient population.
Risk of bias in included studies
We assessed the 10 included studies for risk of bias (see Characteristics of included studies). A graphical summary of the risk of bias for all included studies is presented in Figure 2. The percentages of included studies at low, high, or unclear risk of bias for each risk of bias domain are summarised in Figure 3.
2.

Risk of bias summary.
3.

Risk of bias graph.
For studies with an unclear risk of bias assessment, we sought clarification from the study authors. Where we received responses, we have reported the changes made to our initial assessments (see Characteristics of included studies). We contacted the authors of eight included studies (Ahmadi 2013; Bellomo‐Brandão 2003; Benninga 2021; Bongers 2009; Castilla 2021; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Southwell 2012). We received responses from Benninga 2021, Bongers 2009, and Mugie 2014.
Allocation
Five studies described a random component sufficient for adequate sequence generation and were judged to be at low risk of bias. All five studies used computer‐generated random assignments (Benninga 2021; Bongers 2009; Kajbafzadeh 2020; Keshtgar 2007; Mugie 2014). We assessed five studies as at unclear risk of bias for methods of sequence generation (Ahmadi 2013; Bellomo‐Brandão 2003; Castilla 2021; Keragiozoglou‐Lampoudi 2012; Southwell 2012). In three of these studies, no details were provided on the methods of randomisation (Bellomo‐Brandão 2003 ; Keragiozoglou‐Lampoudi 2012; Southwell 2012). In Castilla 2021, while simple random allocation was reported, the adequacy of sequence generation was deemed to be unclear. Similarly, in Ahmadi 2013, the random division of participants into case and control groups was described, but the reported detail was insufficient to permit a definitive judgement.
Four studies described an allocation sequence concealment technique that securely implemented the sequence. In Benninga 2021, the stratification scheme was generated by a trial supply management system. Two studies used opaque and sequentially numbered, sealed envelopes (Bongers 2009; Keshtgar 2007). In Mugie 2014, the study authors confirmed via written communication this was achieved by "a computer system". In six studies, no details were provided on the methods used to conceal the allocation sequence, and no responses from study authors were received to requests made for further detail (Ahmadi 2013; Bellomo‐Brandão 2003; Castilla 2021; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Southwell 2012).
Blinding
Three studies clearly described the tools used to address blinding of participants and personnel. In Benninga 2021 and Mugie 2014, blinding was maintained using a placebo medication that was identical in appearance and taste to the interventional agent. In Keragiozoglou‐Lampoudi 2012, blinding of participants and personnel was not possible, given the fact the intervention involved a dietitian‐prescribed diet, in addition to paediatric gastroenterologist written instruction; however, we perceived this lack of blinding to have a minimal influence on the reported outcomes.
We deemed four studies as at high risk for performance bias due to additional invasive measures received by those in the intervention groups, which prevented blinding of study personnel to treatment allocations. In Ahmadi 2013, children in the intervention group received botulinum toxin injection under general anaesthetic, whereas children in the control group received stool softeners. In Bongers 2009, children in the intervention group received rectal enemas, whereas children in the control group received oral laxatives only. In Kajbafzadeh 2020, children in the intervention group received intrarectal electromotive treatment, whereas children in the control group received intrasphincteric botulinum toxin A injection. In Keshtgar 2007, children in the intervention group received botulinum toxin injection under general anaesthetic, whereas children in the control group underwent myectomy of the internal anal sphincter, for which all procedures were performed by the study authors.
In three studies no details were provided on the methods used to blind participants and personnel (Bellomo‐Brandão 2003; Castilla 2021; Southwell 2012), and no responses were received to our requests for additional information.
Three studies clearly described the tools used to address blinding of outcome assessors. In Benninga 2021, the study endpoints were assessed based on the content recorded in a diary completed by caregivers, who were blinded to treatment allocations. In Keshtgar 2007, outcome assessments were undertaken by a paediatrician blind to treatment allocations. In Mugie 2014, the study authors confirmed by written communication that the outcome assessors were blind to treatment allocations.
We assessed two studies as at high risk for detection bias. In Bongers 2009, the study authors confirmed by written communication that the outcome assessors were not blind to the treatment allocations. In Keragiozoglou‐Lampoudi 2012, outcome assessment was performed by a registered dietitian, which given that the intervention involved a dietitian‐prescribed diet, is likely to have introduced bias.
In five studies no details were provided on the methods used to blind outcome assessors (Ahmadi 2013; Bellomo‐Brandão 2003; Castilla 2021; Kajbafzadeh 2020; Southwell 2012), and no responses were received to our requests for additional information.
Incomplete outcome data
We assessed seven studies as at low risk of attrition bias. In Benninga 2021, the study authors provided written communication clarifying attrition rates. In the other six studies, all participants randomised to the intervention and control groups were transparently reported and equally balanced between treatment arms (Castilla 2021; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Keshtgar 2007; Mugie 2014; Southwell 2012).
We assessed two studies as at unclear risk of attrition bias. In Ahmadi 2013, no data were provided on the number of participants included in the analyses, and the authors were unresponsive to communication. In Bongers 2009, the reasons for 13 children not completing the study were not reported. We contacted the study authors for clarification, but they were not able to locate the data. We decided to assign an unclear risk of bias to this study as the attrition rates between the intervention and control groups were similar.
We assessed one study as at a high risk of attrition bias. In Bellomo‐Brandão 2003, one‐third of the randomised children were not included in the final analysis, with no description of the numbers excluded in the intervention and control groups.
Selective reporting
We judged six studies to be sufficiently transparent and complete in the reporting of outcomes, permitting a judgement of low risk of reporting bias. No study protocol was published for Bellomo‐Brandão 2003; however, the outcomes reported in the results section of the study report matched those described in the methods section. Furthermore, the authors utilised a 12‐point scoring system, which incorporated many of the outcomes routinely measured in this research area. Similarly, in Keshtgar 2007, a composite score involving eight clinical outcomes was produced, again encompassing the range of clinical measures expected to be reported. The outcomes reported by Southwell 2012 included a spectrum of expected outcomes, including painful defecation and faecal incontinence. In Castilla 2021, while there was no published protocol, it was the only study to include non‐fulfilment of Rome criteria for functional constipation as a primary outcome. In two studies, the outcomes reported within the study report matched those specified within a prospectively registered trial protocol (Benninga 2021; Mugie 2014).
We judged one study as at unclear risk of selective reporting bias. In Keragiozoglou‐Lampoudi 2012, no clinically meaningful outcomes were reported, aside from compliance with a prescribed diet. It would be expected that in studies including children with disease of high acuity, commonly reported outcomes such as symptom improvement would be described. Since we were unable to obtain a response from the study authors, we judged this study as at unclear risk of bias.
We judged three studies as at high risk of reporting bias. In Bongers 2009, the study protocol was retrospectively registered. Furthermore, the definition used to define treatment success was not typical within the field. In Kajbafzadeh 2020, the study protocol was retrospectively registered, in which defecation frequency was the only specified outcome. In the methods section of the study report, the primary outcome was defined as the resolution of symptoms. Improvement in symptoms was the only outcome reported within the results section of the study report. In Ahmadi 2013, the study authors provided outcome data for defecation interval, but only for the intervention group. This is likely to represent selective reporting, as the authors repeatedly cite improvement in defecation within the treatment group as a significant outcome of the study.
Other potential sources of bias
We assessed eight studies as at low risk of other potential sources of bias (Ahmadi 2013; Benninga 2021; Bongers 2009; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Keshtgar 2007; Mugie 2014; Southwell 2012). In Bellomo‐Brandão 2003 and Castilla 2021, there was minimal description of the baseline characteristics of participants within the intervention and control groups, therefore it was not possible to determine the extent to which the treatment groups were equivalent to each other. As such, an unclear risk of bias was assigned to these two studies.
Effects of interventions
See: Table 1; Table 2; Table 3; Table 4; Table 5; Table 6; Table 7; Table 8; Table 9; Table 10
The primary and secondary outcomes reported by the included studies are tabulated in Table 13 and Table 14.
3. Primary outcome data.
| Study ID | Symptom resolution (non‐fulfilment of Rome criteria for functional constipation) | Frequency of defecation | Treatment success | Adverse events |
| Ahmadi 2013 | NR |
Mean defecation interval (6 months) IG: 2.6 days CG: NR |
Defecation interval of fewer than 3 days (6 months) IG: 37/44 CG: 1/44 |
NR |
| Bellomo‐Brandão 2003 | NR | NR | NR |
(4 weeks) IG 0 (number randomised not reported) CG 0 (number randomised not reported) |
| Benninga 2021 | NR | NR |
Increase in ≥ 1 spontaneous bowel movement/week compared with baseline and ≥ 1 spontaneous bowel movement/week for at least 9 weeks, including 3 of the final 4 treatment weeks (12 weeks) IG: 75/404 CG: 29/202 |
Children with ≥ 1 adverse event (12 weeks) IG1 (lubiprostone 12 μg): 142 IG2 (lubiprostone 24 μg): 97 CG: 114 Children with ≥ 1 severe adverse event (12 weeks) IG1: 10 IG2: 4 CG: 12 Children with ≥ 1 treatment‐related adverse event (12 weeks) IG1: 76 IG2: 58 CG: 49 Children with ≥ 1 serious adverse event (12 weeks) IG1: 9 IG2: 2 CG: 7 Children with ≥ 1 treatment‐related serious adverse event (12 weeks) IG1: 4 IG2: 0 CG: 2 Children who discontinued therapy because of an adverse event (12 weeks) IG1: 9 IG2: 8 CG: 6 Children who discontinued because of a treatment‐related adverse event (12 weeks) IG1: 6 IG2: 6 CG: 3 |
| Bongers 2009 | NR |
Mean defecation interval (52 weeks) IG: 5.3 days CG: 3.9 days |
≥ 3 bowel movements per week and < 1 faecal incontinence episode per week, irrespective of laxative use (52 weeks) IG: 24/51 CG: 18/51 |
NR |
| Castilla 2021 |
(Timing not reported) IG: 10/12 CG: 4/12 |
NR | NR | NR |
| Kajbafzadeh 2020 |
Rome II criteria (1 month) IG: 24/30 CG: 25/30 |
Median (IQR) (1 month) IG: 7 (5) days CG: 7 (5) days (6 months) IG: 7 (6) days CG: 7 (6) days |
NR |
Major treatment‐related adverse events IG: 0 CG: 0 Total adverse events IG 0: CG: 2 (rectal prolapse) |
| Keragiozoglou‐Lampoudi 2012 | NR | NR | NR | NR |
| Keshtgar 2007 | NR | NR |
Improvement in symptom severity score (12 months) IG: 17/21 CG: 17/21 |
(12 months) IG: 0/21 CG: 0/21 |
| Mugie 2014 | NR |
Mean change (SD) in defecation frequency per week from baseline (week 8) IG: 1.5 ± 2.35 CG 1.0 ± 1.78 |
Mean of ≥ 3 spontaneous bowel movements per week and ≤ 1 faecal incontinence episode/2 weeks during weeks 5 to 8 (week 8) IG: 18/107 (17.0%) CG: 19/108 (17.8%) |
Children with ≥ 1 treatment‐emergent adverse events IG: 74/106 CG: 65/107 Children experiencing ≥ 1 serious adverse events IG: 5/106 CG: 2/107 |
| Southwell 2012 | NR | NR | NR | NR |
CG: control group; IG: intervention group; IQR: interquartile range; NR: not reported; SD: standard deviation
4. Secondary outcome data.
| Study ID | Stool consistency | Painful defecation | Quality of life | Faecal incontinence | Abdominal pain | Admission to hospital for disimpaction | School absence |
| Ahmadi 2013 |
Stool condensation (hard stools, dichotomous yes/no) (6 months) IG: 12/44 (28%) CG: 34/44 (78%) |
Painful defecation (dichotomous yes/no) (6 months) IG: 7/44 (15%) CG: 39/44 (86%) |
NR |
Soiling (dichotomous yes/no) (6 months) IG: 3/44 (7.5%) CG: 19/44 (42.5%) |
NR | NR | NR |
| Bellomo‐Brandão 2003 | NR | NR | NR | NR | NR | NR | NR |
| Benninga 2021 | NR |
Mean change (SD) from baseline using a 5‐point scale (0 = not at all; 4 = extreme) (12 weeks) IG: −0.81 ± 1.02 CG: −0.65 ± 1.10 |
NR |
Mean (SD) change in the number of episodes of faecal incontinence per day from baseline (12 weeks) IG: 0.04 ± 0.37 CG: 0.07 ± 0.48 |
Mean change (SD) from baseline using a 5‐point scale (0 = none; 4 = very severe) (12 weeks) IG: −0.42 ± 0.84 CG: −0.35 ± 0.76 |
NR | NR |
| Bongers 2009 | NR |
Panful defecation (binary yes/no) (week 52) IG: 4/51 (8.8%) CG: 11/51 (20.9%) |
NR |
Faecal incontinence (binary yes/no) (week 52) IG: 28/51 (54.9%) CG: 26/51 (50.9%) |
Abdominal pain (dichotomous yes/no) (week 52) IG: 10/51 (20.3%) CG: 16/51 (30.6%) |
NR | NR |
| Castilla 2021 | NR | NR | NR | NR | NR | NR | NR |
| Kajbafzadeh 2020 |
Normal stools (Bristol stool chart form 4, dichotomous yes/no) (1 month) IG: 22/30 CG: 24/30 (6 months) IG: 17/30 CG: 16/30 |
Painful defecation (dichotomous yes/no) (1 month) IG: 7/44 CG: 4/44 (6 months) IG: 7/44 CG: 5/44 |
Median constipation‐related quality of life score (IQR) (6 months) IG: 55 (13) CG: 55 (14) |
Median (IQR) episodes of faecal incontinence per day (1 month) IG: 0 (4) CG: 0 (5) (6 months) IG: 0 (4) CG: 0 (5) |
NR | NR | NR |
| Keragiozoglou‐Lampoudi 2012 | NR | NR | NR | NR | NR | NR | NR |
| Keshtgar 2007 | NR |
Improved symptom severity score in the painful defecation domain (12 months) IG: 13/21 CG: 9/21 |
NR |
Improved symptom severity score in the soiling domain (12 months) IG: 14/21 CG: 11/21 |
NR | NR | NR |
| Mugie 2014 | NR |
Mean change (SD) in level of pain during defecation using a scaled score (0 = no pain; 5 = severe pain)(week 8) IG: 0.6 ± 1.36 CG: 0.4 ± 1.19 |
Mean (SD) improvement in child‐reported total score (week 8) IG: 3.9 ± 13.8 CG: 2.7 ± 12.4 Mean (SD) improvement in parent‐reported total score (week 8) IG: 6.5 ± 13.9 CG: 4.1 ± 14.2 |
Mean change (SD) in the number of episodes of faecal incontinence during the preceding 2 weeks from baseline (week 8) IG: 8.7 ± 36.85 CG: 13.9 ± 64.91 |
Mean change (SD) in abdominal pain score (0 to 5) from baseline (8 weeks) IG: −0.20 ± 0.76 CG: −0.30 ± 0.94 |
NR | NR |
| Southwell 2012 | NR | NR |
Mean parent‐reported quality of life score using generic quality of life questionnaire (8 weeks) IG: 70.1 CG: 70.2 |
NR | NR | NR | NR |
CG: control group; IG: intervention group; IQR: interquartile range; NR: not reported; SD: standard deviation
Comparison 1. Botulinum toxin A injection versus stool softeners
One study compared botulinum toxin injection (n = 44) to unspecified stool softeners (n = 44), delivered as a one‐off intervention, and included 88 children (Ahmadi 2013). Outcome measures were recorded six months following the intervention. Children were asked questions about the signs of constipation, but no detail is provided on the format of these questions. See Table 1.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Ahmadi 2013.
Frequency of defecation
In Ahmadi 2013, the mean defecation interval was reported for the botulinum toxin A injection group (2.6 days) only and not for the control group. The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Table 1).
Treatment success
We are very uncertain whether botulinum toxin A injection improves treatment success, measured as children with a defecation interval of fewer than three days, compared to unspecified stool softeners (37/44 children with botulinum toxin A injection versus 1/44 children with stool softeners; risk ratio (RR) 37.00, 95% confidence interval (CI) 5.31 to 257.94). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 1.1; Table 1).
1.1. Analysis.

Comparison 1: Botulinum toxin A injection versus stool softeners, Outcome 1: Treatment success
Adverse events
This outcome was not reported by Ahmadi 2013.
Secondary outcomes
Stool consistency
We are very uncertain whether botulinum toxin A injection improves stool consistency, measured as children with stool condensation, compared to unspecified stool softeners (12/44 children with botulinum toxin A injection versus 34/44 children with stool softeners; RR 0.35, 95% CI 0.21 to 0.59). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 1.2).
1.2. Analysis.

Comparison 1: Botulinum toxin A injection versus stool softeners, Outcome 2: Stool consistency
Painful defecation
We are very uncertain whether botulinum toxin A injection reduces painful defecation, measured as children with painful defecation, compared to unspecified stool softeners (7/44 children with botulinum toxin A injection versus 39/44 children with stool softeners; RR 0.18, 95% CI 0.09 to 0.36). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 1.3).
1.3. Analysis.

Comparison 1: Botulinum toxin A injection versus stool softeners, Outcome 3: Painful defecation
Quality of life
This outcome was not reported by Ahmadi 2013.
Faecal incontinence
We are very uncertain whether botulinum toxin A injection reduces faecal incontinence, measured as children with faecal soiling, compared to unspecified stool softeners (3/44 children with botulinum toxin A injection versus 19/44 children with stool softeners; RR 0.16, 95% CI 0.05 to 0.50). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 1.4).
1.4. Analysis.

Comparison 1: Botulinum toxin A injection versus stool softeners, Outcome 4: Faecal incontinence
Abdominal pain
This outcome was not reported by Ahmadi 2013.
Admission to hospital for disimpaction
This outcome was not reported by Ahmadi 2013.
School absence
This outcome was not reported by Ahmadi 2013.
Comparison 2. Erythromycin estolate versus placebo
One study compared oral erythromycin estolate to placebo, administered over four weeks, and included 21 randomised children (Bellomo‐Brandão 2003). Fourteen children completed the study, six in the erythromycin group and eight in the placebo group. The reasons for postrandomisation exclusions are described in Characteristics of included studies. Outcome measures were recorded at the end of the four‐week study period. A 12‐point composite score system was used which was based on stool frequency, soiling, pain or difficulty at stool passage, faecal consistency, and daily laxative dose. The breakdown score for each item of the composite was not reported. See Table 2.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Bellomo‐Brandão 2003.
Frequency of defecation
This outcome was not reported by Bellomo‐Brandão 2003.
Treatment success
This outcome was not reported by Bellomo‐Brandão 2003.
Adverse events
No adverse events were recorded in either the erythromycin estolate or placebo arm of Bellomo‐Brandão 2003. The evidence is of very low certainty due to concerns with risk of bias and serious imprecision. See Table 2.
Secondary outcomes
Stool consistency
This outcome was not reported by Bellomo‐Brandão 2003.
Painful defecation
This outcome was not reported by Bellomo‐Brandão 2003.
Quality of life
This outcome was not reported by Bellomo‐Brandão 2003.
Faecal incontinence
This outcome was not reported by Bellomo‐Brandão 2003.
Abdominal pain
This outcome was not reported by Bellomo‐Brandão 2003.
Admission to hospital for disimpaction
This outcome was not reported by Bellomo‐Brandão 2003.
School absence
This outcome was not reported by Bellomo‐Brandão 2003.
Comparison 3. Lubiprostone versus placebo
One study compared oral lubiprostone (n = 404) with a placebo (n = 202), administered over 12 weeks, and included 606 children (Benninga 2021). Outcome measures were recorded at the end of the 12‐week study period. A daily e‐diary was completed by the parent or child. See Table 3.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Benninga 2021.
Frequency of defecation
This outcome was not reported by Benninga 2021.
Treatment success
There may be little to no difference in treatment success between lubiprostone and placebo, measured as the number of children with an increase in at least one spontaneous bowel movement per week compared to baseline and at least one spontaneous bowel movement per week for at least nine weeks, including three of the final four treatment weeks (75/404 children with lubiprostone versus 29/202 children with placebo; RR 1.29, 95% CI 0.87 to 1.92). The evidence is of low certainty, downgraded twice due to serious imprecision (Analysis 2.1; Table 3).
2.1. Analysis.

Comparison 2: Lubiprostone versus placebo, Outcome 1: Treatment success
Adverse events
Lubiprostone probably results in little to no difference in adverse events, measured as the number of children with at least one adverse event, as compared to placebo (239/404 versus 114/202; RR 1.05, 95% CI 0.91 to 1.21). The evidence is of moderate certainty due to imprecision (Analysis 2.2; Table 3).
2.2. Analysis.

Comparison 2: Lubiprostone versus placebo, Outcome 2: Adverse events
The most frequently reported adverse events in the lubiprostone group were nausea (n = 59, 14.5%), vomiting (n = 48, 11.9%), headache (n = 40, 9.9%), diarrhoea (n = 30, 7.4%), gastroenteritis (n = 20, 5.0%), pyrexia (n = 19, 4.7%), sinusitis (n = 13, 3.2%), chest pain (n = 10, 2.5%), rash (n = 8, 2.0%). In the placebo group, the equivalent adverse events were nausea (n = 14, 6.9%), vomiting (n = 12, 5.9%), headache (n = 15, 7.4%), diarrhoea (n = 6, 3.0%), gastroenteritis (n = 4, 2.0%), pyrexia (n = 7, 3.5%), sinusitis (n = 3, 1.5%), chest pain (n = 1, < 1%), rash (n = 3, 1.5%).
Secondary outcomes
Stool consistency
This outcome was not reported by Benninga 2021.
Painful defecation
There may be little to no difference in painful defecation between the lubiprostone and placebo groups, measured as mean change in defecation pain from baseline using a five‐point severity scale, where 0 = no pain; 4 = extreme pain (−0.81 ± 1.02 versus −0.65 ± 1.10; mean difference (MD) −0.16, 95% CI −0.35 to 0.03). The evidence is of low certainty, downgraded twice due to serious imprecision (Analysis 2.3).
2.3. Analysis.

Comparison 2: Lubiprostone versus placebo, Outcome 3: Painful defecation
Quality of life
This outcome was not reported by Benninga 2021.
Faecal incontinence
There may be little to no difference in faecal incontinence between the lubiprostone and placebo groups, measured as mean change from baseline in the number of episodes of faecal incontinence per day (0.04 ± 0.37 versus 0.07 ± 0.48; MD −0.03, 95% CI −0.11 to 0.05). The evidence is of low certainty, downgraded twice due to serious imprecision (Analysis 2.4).
2.4. Analysis.

Comparison 2: Lubiprostone versus placebo, Outcome 4: Faecal incontinence
Abdominal pain
There may be little to no difference in abdominal pain between the lubiprostone and placebo groups, measured as mean change from baseline in the severity of abdominal pain using a four‐point severity scale (−0.42 ± 0.84 versus −0.35 ± 0.76; MD −0.07, 95% CI −0.20 to 0.06). The evidence is of low certainty, downgraded twice due to serious imprecision (Analysis 2.5).
2.5. Analysis.

Comparison 2: Lubiprostone versus placebo, Outcome 5: Abdominal pain
Admission to hospital for disimpaction
This outcome was not reported by Benninga 2021.
School absence
This outcome was not reported by Benninga 2021.
Comparison 4. Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives
One study compared rectal sodium dioctyl sulfosuccinate and sorbitol enemas to oral polyethylene glycol laxatives, administered over 52 weeks, and included 102 children (Bongers 2009). Outcome measures were recorded at the end of the 52‐week study period. Participants recorded metrics relating to defecation using a daily diary. See Table 4.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Bongers 2009.
Frequency of defecation
In Bongers 2009, the study investigators reported the modelled mean (using a linear mixed model) frequency of defecation per week: 5.3 days for rectal sodium dioctyl sulfosuccinate and sorbitol, and 3.9 days for polyethylene glycol laxatives. Since no variance was provided, we were unable to conduct an analysis. The evidence is of very low certainty due to serious concerns with risk of bias and imprecision.
Treatment success
We are very uncertain whether rectal sodium dioctyl sulfosuccinate and sorbitol improves treatment success, measured as the number of children with at least three bowel movements per week and no faecal incontinence, compared to oral polyethylene glycol laxatives (24/51 children with rectal sodium dioctyl sulfosuccinate and sorbitol enemas versus 18/51 children with oral polyethylene glycol laxatives; RR 1.33, 95% CI 0.83 to 2.14). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 3.1; Table 4).
3.1. Analysis.

Comparison 3: Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives, Outcome 1: Treatment success
Adverse events
This outcome was not reported by Bongers 2009.
Secondary outcomes
Stool consistency
This outcome was not reported by Bongers 2009.
Painful defecation
We are very uncertain whether rectal sodium dioctyl sulfosuccinate and sorbitol improves painful defecation, measured as children with painful defecation, compared to oral polyethylene glycol laxatives (4/51 with rectal sodium dioctyl sulfosuccinate and sorbitol versus 11/51 with oral polyethylene glycol laxatives; RR 2.50, 95% CI 0.84 to 7.46). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 3.2).
3.2. Analysis.

Comparison 3: Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives, Outcome 2: Painful defecation
Quality of life
This outcome was not reported by Bongers 2009.
Faecal incontinence
We are very uncertain whether rectal sodium dioctyl sulfosuccinate and sorbitol reduces faecal incontinence, measured as the number of children with faecal incontinence, compared to oral polyethylene glycol laxatives (28/51 children with rectal sodium dioctyl sulfosuccinate and sorbitol enemas versus 26/51 children with oral polyethylene glycol laxatives; RR 1.08, 95% CI 0.75 to 1.55). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 3.3).
3.3. Analysis.

Comparison 3: Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives, Outcome 3: Faecal incontinence
Abdominal pain
We are very uncertain whether sodium dioctyl sulfosuccinate and sorbitol reduces abdominal pain, measured as children with abdominal pain, compared to oral polyethylene glycol laxatives (16/51 children with rectal sodium dioctyl sulfosuccinate and sorbitol enemas versus 10/51 children with oral polyethylene glycol laxatives; RR 1.60, 95% CI 0.80 to 3.18). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 3.4).
3.4. Analysis.

Comparison 3: Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives, Outcome 4: Abdominal pain
Admission to hospital for disimpaction
This outcome was not reported by Bongers 2009.
School absence
This outcome was not reported by Bongers 2009.
Comparison 5. Biofeedback therapy versus no intervention
One study compared biofeedback therapy (n = 12) to no intervention (n = 12), delivered as a one‐off intervention, and included 24 children (Castilla 2021). The timing of the outcome measure assessment was not reported. The method of outcome assessment was not reported. See Table 5.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
We are very uncertain whether biofeedback therapy improves symptom resolution, measured as non‐fulfilment of Rome IV criteria, compared to no intervention (10/12 children with biofeedback therapy versus 4/12 children with no intervention; RR 2.50, 95% CI 1.08 to 5.79). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 4.1; Table 5).
4.1. Analysis.

Comparison 4: Biofeedback therapy versus no intervention, Outcome 1: Non‐fulfilment of Rome criteria
Frequency of defecation
This outcome was not reported by Castilla 2021.
Treatment success
This outcome was not reported by Castilla 2021.
Adverse events
This outcome was not reported by Castilla 2021.
Secondary outcomes
Stool consistency
This outcome was not reported by Castilla 2021.
Painful defecation
This outcome was not reported by Castilla 2021.
Quality of life
This outcome was not reported by Castilla 2021.
Faecal incontinence
This outcome was not reported by Castilla 2021.
Abdominal pain
This outcome was not reported by Castilla 2021.
Admission to hospital for disimpaction
This outcome was not reported by Castilla 2021.
School absence
This outcome was not reported by Castilla 2021.
Comparison 6. Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection
One study compared intrarectal electromotive botulinum toxin A (n = 30) to botulinum toxin A injection (n = 30), delivered as a one‐off intervention, and included 60 children (Kajbafzadeh 2020). Outcome measures were recorded one month and six months following the intervention. All outcomes except for quality of life were assessed at one month. Quality of life assessments were recorded six months after the intervention. Defecation metrics were recorded by parents using a diary. A quality of life questionnaire was completed for children before and six months after treatment. See Table 6.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
We are very uncertain whether intrarectal electromotive botulinum toxin A improves symptoms, measured as non‐fulfilment of Rome II criteria, compared to botulinum toxin A injection (24/30 children with intrarectal electromotive botulinum toxin A versus 25/30 children with botulinum toxin A injection; RR 0.96, 95% CI 0.76 to 1.22). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.1; Table 6).
5.1. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 1: Non‐fulfilment of Rome criteria
Frequency of defecation
We are very uncertain whether intrarectal electromotive botulinum toxin A improves the frequency of defecation, measured as the interval in days between defecation, compared to botulinum toxin A injection (7.0 ± 3.7 in children receiving intrarectal electromotive botulinum toxin versus 7.0 ± 3.7 in children receiving botulinum toxin A injection; MD 0.00, 95% CI −1.87 to 1.87). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.2; Table 6).
5.2. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 2: Frequency of defecation
Treatment success
This outcome was not reported by Kajbafzadeh 2020.
Adverse events
We are very uncertain whether intrarectal electromotive botulinum toxin A has an improved safety profile, measured as the total number of adverse events, compared to botulinum toxin A injection (0/30 children with intrarectal electromotive botulinum toxin A versus 2/30 children with botulinum toxin A injection; RR 0.20, 95% CI 0.01 to 4.00). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.3; Table 6).
5.3. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 3: Adverse events
Secondary outcomes
Stool consistency
We are very uncertain whether intrarectal electromotive botulinum toxin A improves stool consistency, measured as children with Bristol stool chart form 4, compared to botulinum toxin A injection (22/30 children with electromotive botulinum toxin A versus 24/30 children with botulinum toxin A injection; RR 0.92, 95% CI 0.69 to 1.21). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.4).
5.4. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 4: Stool consistency
Painful defecation
We are very uncertain whether intrarectal electromotive botulinum toxin A improves painful defecation, measured as children with painful defecation, compared to botulinum toxin A injection (7/30 children with intrarectal electromotive botulinum toxin A versus 4/30 children with botulinum toxin A injection; RR 1.75, 95% CI 0.57 to 5.36). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.5).
5.5. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 5: Painful defecation
Quality of life
We are very uncertain whether intrarectal electromotive botulinum toxin A improves quality of life, measured on a constipation‐related quality of life score, compared to botulinum toxin A injection (55 ± 9.6 with intrarectal electromotive botulinum toxin A versus 55 ± 10.3 with botulinum toxin A injection; MD 0.00, 95% CI −5.04 to 5.04). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.6).
5.6. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 6: Quality of life
Faecal incontinence
We are very uncertain whether intrarectal electromotive botulinum toxin A reduces faecal incontinence, measured as median daily episodes of faecal incontinence, compared to botulinum toxin A injection (0.00 ± 2.96 with intrarectal electromotive botulinum toxin A versus 0.00 ± 3.70 with botulinum toxin A injection; MD 0.00, 95% CI −1.70 to 1.70). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 5.7).
5.7. Analysis.

Comparison 5: Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection, Outcome 7: Faecal incontinence
Abdominal pain
This outcome was not reported by Kajbafzadeh 2020.
Admission to hospital for disimpaction
This outcome was not reported by Kajbafzadeh 2020.
School absence
This outcome was not reported by Kajbafzadeh 2020.
Comparison 7. Botulinum toxin A injection versus myectomy of the internal anal sphincter
One study compared botulinum toxin injection (n = 21) to myectomy of the internal anal sphincter (n = 21), delivered as a one‐off intervention, and included 42 children (Keshtgar 2007). Outcome measures were assessed at 12 months following the intervention. A questionnaire that included key defecation metrics was completed by the child or their parent. See Table 7.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Keshtgar 2007.
Frequency of defecation
This outcome was not reported by Keshtgar 2007.
Treatment success
We are very uncertain whether botulinum toxin A injection improves treatment success, measured as an improvement in composite symptom severity score, compared to myectomy of the internal anal sphincter (17/21 children with botulinum toxin A injection versus 17/21 children with myectomy of the internal anal sphincter; RR 1.00, 95% CI 0.75 to 1.34). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 6.1; Table 7).
6.1. Analysis.

Comparison 6: Botulinum toxin A injection versus myectomy of the internal anal sphincter, Outcome 1: Treatment success
Adverse events
No adverse events were recorded in either the botulinum toxin A injection arm (0/21 children) or the myectomy of the internal anal sphincter arm (0/21 children). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 6.2; Table 7).
6.2. Analysis.

Comparison 6: Botulinum toxin A injection versus myectomy of the internal anal sphincter, Outcome 2: Adverse events
Secondary outcomes
Stool consistency
This outcome was not reported by Keshtgar 2007.
Painful defecation
We are very uncertain whether botulinum toxin A injection improves painful defecation, measured as improvement in painful defecation domain of symptom severity score, compared to myectomy of the internal anal sphincter (13/21 children with botulinum toxin A injection versus 9/21 children with myectomy of the internal anal sphincter; RR 1.44, 95% CI 0.8 to 2.62). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 6.3).
6.3. Analysis.

Comparison 6: Botulinum toxin A injection versus myectomy of the internal anal sphincter, Outcome 3: Painful defecation
Quality of life
This outcome was not reported by Keshtgar 2007.
Faecal incontinence
We are very uncertain whether botulinum toxin A injection reduces faecal incontinence, measured as improvement in faecal soiling domain of symptom severity score, compared to myectomy of the internal anal sphincter (14/21 children with botulinum toxin A injection versus 11/21 children with myectomy of the internal anal sphincter; RR 1.27, 95% CI 0.77 to 2.11). The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Analysis 6.4).
6.4. Analysis.

Comparison 6: Botulinum toxin A injection versus myectomy of the internal anal sphincter, Outcome 4: Faecal incontinence
Abdominal pain
This outcome was not reported by Keshtgar 2007.
Admission to hospital for disimpaction
This outcome was not reported by Keshtgar 2007.
School absence
This outcome was not reported by Keshtgar 2007.
Comparison 8. Prucalopride versus placebo
One study compared oral prucalopride (n = 107) to placebo (n = 108), administered over eight weeks, and included 215 children (Mugie 2014). Outcome measures were assessed at the end of the eight‐week study period. The outcome assessments involved participant interviews on defecation metrics and quality of life questionnaires. See Table 8.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Mugie 2014.
Frequency of defecation
There is probably little or no difference between oral prucalopride and placebo in the frequency of defecation, measured as mean change in defecation frequency per week from baseline (1.50 ± 2.35 versus 1.00 ± 1.78; MD 0.50, 95% CI −0.06 to 1.06). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.1; Table 8).
7.1. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 1: Frequency of defecation
Treatment success
There is probably little or no difference between oral prucalopride and placebo in treatment success, measured as at least three spontaneous bowel movements per week and no faecal incontinence over a two‐week period during weeks 5 to 8 of the study (18/107 with prucalopride versus 19/108 with placebo; RR 0.96, 95% CI 0.53 to 1.72). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.2; Table 8).
7.2. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 2: Treatment success
Adverse events
There is probably little or no difference between oral prucalopride and placebo in adverse events, measured as children with at least one treatment‐emergent adverse event (75/107 with prucalopride versus 66/108 with placebo; RR 1.15, 95% CI 0.94 to 1.39). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.3; Table 8).
7.3. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 3: Adverse events
Secondary outcomes
Stool consistency
This outcome was not reported by Mugie 2014.
Painful defecation
There is probably little or no difference between oral prucalopride and placebo in painful defecation, measured as mean change from baseline in the severity of pain during defecation using a 6‐point scale, where 0 = no pain; 5 = severe pain (0.60 ± 1.36 with prucalopride versus 0.40 ± 1.19 with placebo; MD −0.20, 95% CI −0.54 to 0.14). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.4).
7.4. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 4: Painful defecation
Quality of life
There is probably little or no difference between oral prucalopride and placebo in quality of life, measured as improvement in child‐reported score (3.9 ± 13.8 with prucalopride versus 2.7 ± 12.4 with placebo; MD 1.20, 95% CI −2.31 to 4.71) and improvement in parent‐reported score (6.5 ± 13.9 with prucalopride versus 4.1 ± 14.2 with placebo; MD 2.40, 95% CI −1.36 to 6.16). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.5; Analysis 7.6).
7.5. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 5: Quality of life (child‐reported)
7.6. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 6: Quality of life (parent‐reported)
Faecal incontinence
There is probably little or no difference between oral prucalopride and placebo in faecal incontinence, measured as mean change from baseline in the number of episodes of faecal incontinence during the preceding two weeks (−8.70 ± 36.85 with prucalopride versus −13.90 ± 64.91 with placebo; MD 5.20, 95% CI −8.89 to 19.29). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.7).
7.7. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 7: Faecal incontinence
Abdominal pain
There is probably little or no difference between oral prucalopride and placebo in abdominal pain, measured as mean change from baseline in the severity of abdominal pain using a 5‐point scale (−0.20 ± 0.76 with prucalopride versus −0.30 ± 0.94 with placebo; MD 0.10, 95% CI −0.13 to 0.33). The evidence is of moderate certainty due to concerns with imprecision (Analysis 7.8).
7.8. Analysis.

Comparison 7: Prucalopride versus placebo, Outcome 8: Abdominal pain
Admission to hospital for disimpaction
This outcome was not reported by Mugie 2014.
School absence
This outcome was not reported by Mugie 2014.
Comparison 9. Transcutaneous electrical stimulation versus sham stimulation
One study compared transcutaneous electric stimulation to sham stimulation, administered over four weeks, and included 46 children (Southwell 2012). See Table 9.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Southwell 2012.
Frequency of defecation
This outcome was not reported by Southwell 2012.
Treatment success
This outcome was not reported by Southwell 2012.
Adverse events
This outcome was not reported by Southwell 2012.
Secondary outcomes
Stool consistency
This outcome was not reported by Southwell 2012.
Painful defecation
This outcome was not reported by Southwell 2012.
Quality of life
This outcome was not reported by Southwell 2012.
Faecal incontinence
This outcome was not reported by Southwell 2012.
Abdominal pain
This outcome was not reported by Southwell 2012.
Admission to hospital for disimpaction
This outcome was not reported by Southwell 2012.
School absence
This outcome was not reported by Southwell 2012.
Comparison 10. Dietitian‐prescribed Mediterranean and physician‐explained written instructions diet versus physician‐explained written instructions
One study compared a dietitian‐prescribed Mediterranean diet with physician‐explained written instructions (n = 44) to physician‐explained written instructions (n = 42), delivered as a one‐off intervention, and included 86 children (Keragiozoglou‐Lampoudi 2012). See Table 10.
Primary outcomes
Symptom resolution ‐ non‐fulfilment of Rome criteria
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Frequency of defecation
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Treatment success
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Adverse events
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Secondary outcomes
Stool consistency
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Painful defecation
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Quality of life
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Faecal incontinence
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Abdominal pain
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Admission to hospital for disimpaction
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
School absence
This outcome was not reported by Keragiozoglou‐Lampoudi 2012.
Discussion
Summary of main results
A variety of therapies are used for the treatment of intractable constipation. The heterogeneity in outcome measures reported and interventions used severely limited our scope for meta‐analysis. Given the broad scope of interventions used, we were unable to combine studies for the purpose of meta‐analysis. This review included 10 RCTs with 1278 children who had intractable constipation.
One study compared the injection of botulinum toxin A to unspecified oral stool softeners (Ahmadi 2013). We are very uncertain whether botulinum toxin A injection improves frequency of defecation (data reported for the experimental group only), treatment success, stool consistency, painful defecation, and faecal incontinence. The evidence is of very low certainty due to concerns with study limitations (risk of bias) and imprecision (Table 1).
One study compared oral erythromycin estolate to placebo (Bellomo‐Brandão 2003). No adverse events were recorded in either the erythromycin estolate or placebo group; the evidence is very uncertain due to concerns with risk of bias and imprecision (Table 2).
One study compared oral lubiprostone to placebo (Benninga 2021). There may be little to no difference between groups in treatment success, painful defecation, faecal incontinence, and abdominal pain. The evidence is of low certainty due to imprecision. Based on moderate‐certainty evidence, we also found that lubiprostone probably results in little to no difference in adverse events when compared to placebo. See Table 3.
One study compared rectal sodium dioctyl sulfosuccinate and sorbitol enemas to polyethylene glycol laxatives (Bongers 2009). We are very uncertain whether rectal sodium dioctyl sulfosuccinate and sorbitol improves frequency of defecation, treatment success, painful defecation, faecal incontinence, and abdominal pain. The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Table 4).
One study compared biofeedback therapy to no intervention (Castilla 2021). We are very uncertain whether biofeedback therapy improves symptom resolution. The evidence is of very low certainty due to serious concerns with risk of bias and imprecision (Table 5).
One study compared intrarectal electromotive botulinum toxin A to botulinum toxin A injection (Kajbafzadeh 2020). We are very uncertain whether intrarectal electromotive botulinum toxin A improves symptom resolution, frequency of defecation, stool consistency, painful defecation, quality of life, and faecal incontinence. We are very uncertain whether intrarectal electromotive botulinum toxin A has an improved safety profile. The evidence is of very low certainty due to serious concerns with risk of bias and imprecision.
One study compared the injection of botulinum toxin A to myectomy of the internal anal sphincter (Keshtgar 2007). We are very uncertain whether botulinum toxin A injection improves treatment success, painful defecation, and faecal incontinence. No adverse events were recorded in either the botulinum toxin A injection or myectomy of the internal anal sphincter group.
One study compared prucalopride to placebo (Mugie 2014). There is probably little or no difference between oral prucalopride and placebo in improving the frequency of defecation, treatment success, painful defecation, quality of life, faecal incontinence, and abdominal pain. There is also probably little or no difference between oral prucalopride and placebo in adverse events. The evidence is of moderate certainty due to concerns with imprecision.
The primary outcomes for this review were not reported by the study comparing interferential therapy to sham therapy (Southwell 2012), or the study comparing a dietitian‐prescribed personalised Mediterranean diet and physician‐explained written instruction to physician‐explained written instruction (Keragiozoglou‐Lampoudi 2012).
None of the included studies reported on the outcomes of admission to hospital for disimpaction and school absence.
Overall completeness and applicability of evidence
The completeness and applicability of the evidence were affected by several issues.
First, there was significant clinical heterogeneity across studies. This is primarily related to the nature of the interventions delivered. One study investigated single‐agent laxative therapy. Three studies described non‐laxative pharmacological interventions, including oral erythromycin estolate, oral lubiprostone, and oral prucalopride. Three studies described surgical intervention, including botulinum toxin A injection into the anal sphincter, intrarectal electromotive therapy, and myectomy of the internal anal sphincter. Three studies described alternative therapy, including biofeedback therapy, interferential therapy, and dietitian‐prescribed diets. Due to this heterogeneity, meta‐analysis was not feasible.
Second, the reporting of relevant primary outcomes was a significant issue for the completeness of the evidence. For instance, only two studies reported the number of participants whose symptoms had resolved, using relevant Rome criteria as the case definition (Castilla 2021; Kajbafzadeh 2020). Only four studies reported the frequency of defecation at the study endpoint (Ahmadi 2013; Kajbafzadeh 2020; Mugie 2014). In Bongers 2009, no measure of spread was provided, meaning no further analysis could be performed. In Kajbafzadeh 2020, this was reported as the median interval between defecations, while in Mugie 2014, this was reported as the mean change in defecation frequency from baseline. In Ahmadi 2013, defecation interval was also reported, but only for the intervention group, which was ultimately felt to confer a high risk of selective reporting bias. Half of the included studies reported treatment success, each using different definitions. The simplest definition was used by Ahmadi 2013, that is a defecation interval of fewer than three days. In Keshtgar 2007, treatment success was defined as any improvement in symptom severity score. In Bongers 2009 and Mugie 2014, treatment success was defined as three or more bowel movements per week with no episodes of faecal incontinence, with the latter study requiring these conditions to be met in each of the last four weeks of the study period. Benninga 2021 defined success relative to the baseline metric. An increase in spontaneous bowel movements per week by one constituted success, which had to be present for at least nine weeks of the study period and at least three of the final four weeks. Five studies reported on adverse events (Bellomo‐Brandão 2003; Benninga 2021; Kajbafzadeh 2020; Keshtgar 2007; Mugie 2014). In three of these studies, no adverse events were recorded in the treatment groups (Bellomo‐Brandão 2003; Kajbafzadeh 2020; Keshtgar 2007). Benninga 2021 and Mugie 2014 provided detailed reporting of treatment‐related adverse events, serious adverse events, and discontinuations due to treatment‐related adverse events.
The third major issue, mainly relating to the applicability of the evidence, was the case definitions used to define intractable constipation. Five explicitly referred to Rome criteria, split across Rome II (Southwell 2012), Rome III (Benninga 2021; Kajbafzadeh 2020; Mugie 2014), and Rome IV (Castilla 2021). Eight referred to a minimum duration of chronicity, ranging from three months to at least two years (Ahmadi 2013; Bongers 2009; Castilla 2021; Kajbafzadeh 2020; Keragiozoglou‐Lampoudi 2012; Keshtgar 2007; Mugie 2014; Southwell 2012). The two studies that made no reference to chronicity, instead described intractable constipation using indicators of severity, such as the presence of megarectum or the failure to respond to invasive surgical procedures (Bellomo‐Brandão 2003; Keshtgar 2007). The lack of a uniform definition is a major factor limiting research in this area, as the differentiation between chronic constipation and intractable constipation has not been clearly delineated.
Fourth, there was a variation in pre‐treatment or concurrent therapy, or both, across the included studies. Several studies administered faecal disimpaction treatments prior to children receiving trial therapies, several of which involved hospital admissions for repeated enemas and colonic irrigation. Only in Mugie 2014 were participants instructed to not consume any agents that influence bowel movements prior to receiving any intervention.
Quality of the evidence
There were significant issues related to risk of bias in most of the included studies. Despite requests made to study authors, only three provided data and responses that allowed us to modify our risk of bias judgements (Benninga 2021; Bongers 2009; Mugie 2014). We assessed two studies as having a low risk of bias across all domains assessed (Benninga 2021; Mugie 2014). Six of the remaining eight studies received at least one judgement of high risk of bias. Of these, two studies received two high risk of bias judgements (Ahmadi 2013; Kajbafzadeh 2020), and one study received three high risk of bias judgements (Bongers 2009).
Half of the included studies did not provide details on the measures used to ensure a random component sufficient for adequate sequence generation (selection bias). Seven studies did not describe the allocation sequence concealment techniques used to securely implement the sequence (selection bias). Due to the paucity of information provided, no studies were assessed as at high risk of bias for either of these domains.
The domain with the highest number of high risk of bias judgements was blinding of participants and personnel, totalling four (Ahmadi 2013; Bongers 2009; Kajbafzadeh 2020; Keshtgar 2007). In all cases, this was due to the invasive nature of the interventions received by participants. One of these studies was also judged to be at high risk for blinding of outcome assessment (Bongers 2009), as was a further study (Keragiozoglou‐Lampoudi 2012).
We assessed seven studies as at low risk of attrition bias since they transparently described attrition rates. One study received a high risk of bias judgement for this domain (Bellomo‐Brandão 2003).
Selective reporting was also an area of concern in three studies, with each assessed as at high risk of reporting bias (Ahmadi 2013; Bongers 2009; Kajbafzadeh 2020). In Ahmadi 2013, this was due to reporting the primary outcome for just the intervention group and not the control group. In the other two studies, this was because of retrospective trial registrations.
Potential biases in the review process
Clinical heterogeneity is a concern in this review. The inclusion of a wide range of pharmacological, non‐pharmacological, and surgical interventions, together with the non‐uniformity of case definitions used to define intractable constipation in children, is an issue. Even with the publication of identified ongoing studies, the biases are unlikely to be mitigated. The lack of clarity on what constitutes intractable constipation is a concern, and clarification of nomenclature is essential to ensure future research has applicability.
Agreements and disagreements with other studies or reviews
This is the first Cochrane review focusing on constipation that is intractable in severity.
Authors' conclusions
Implications for practice.
We found that oral lubiprostone may be little to no different to placebo in achieving treatment success or in improving painful defecation, faecal incontinence, and abdominal pain. Oral lubiprostone probably results in little to no difference in adverse events as compared to placebo. We also found that there is probably little or no difference between oral prucalopride and placebo in defecation frequency, treatment success, painful defecation, quality of life, faecal incontinence, abdominal pain, and adverse events. These results may be used to inform clinicians and those involved in guideline development.
It is uncertain whether any of the other interventions included in this review are safe or efficacious in treating intractable constipation in children, either because they did not report data on our predefined outcomes of interest, or when they did, the certainty of evidence was downgraded due to very serious imprecision and risk of bias.
Although many of the therapeutic interventions included are at present being used in managing children with intractable constipation in real‐world clinical practice, paediatric gastroenterologists, patients, and their families/carers need to be aware of the lack of clear evidence for any of these management strategies.
Implications for research.
Further well‐designed, appropriately powered randomised controlled trials are essential to generate more robust evidence‐based clinical interventions for the management of intractable constipation.
Before such studies, work is needed to clarify a consensus definition of intractable constipation to ensure that populations being considered in future reviews are homogenous. Without this, such studies could include highly diverse populations of children with very different journeys.
The evidence base would be strengthened if researchers considered the possibility of bias in their reporting and also reported data on the primary and secondary outcomes defined by the Rome foundation paediatric subcommittee on clinical trials. This approach would undoubtedly provide uniformity of data in different studies.
The importance of sample size cannot be overstated. All but two of the studies included in this review had a very small sample size. It is imperative to perform a power calculation to have a sample that provides an adequate number of subjects and controls to give adequate power to the study to detect the effectiveness of an intervention. Such precise calculations are critical for putting an end to the proliferation of underpowered studies and increasing the precision of findings.
Finally, other therapeutic modalities, both surgical and medical, are being used to treat children with intractable constipation. Researchers are encouraged to conduct trials using these interventions, thereby expanding the therapeutic armoury for children with intractable constipation.
History
Protocol first published: Issue 5, 2021
Acknowledgements
We thank Farhad Shokraneh for his assistance with the search strategies in May 2021 and for running the updated searches in June 2023.
Editorial and peer‐reviewer contributions
Cochrane Gut supported the authors in the development of this review. Morris Gordon and Anthony Akobeng are members of Cochrane Gut but were not involved in the editorial process or decision‐making for this review.
The following people conducted the editorial process for this review.
Sign‐off Editor (final editorial decision): Grigorios Leontiadis, Department of Medicine, Division of Gastroenterology, McMaster University
Managing Editor (selected peer reviewers, provided comments, collated peer‐reviewer comments, provided editorial guidance to authors, edited the article): Joey Kwong, Cochrane Central Editorial Service
Editorial Assistant (conducted editorial policy checks and supported the editorial team): Lisa Wydrzynski, Cochrane Central Editorial Service
Copy Editor (copy‐editing and production): Lisa Winer, Cochrane Central Production Service
Peer reviewers (provided comments and recommended an editorial decision): David Avelar Rodiguez, The Hospital for Sick Children, Toronto, Ontario (clinical/content review), Maria Angela Bellomo‐Brandao, School of Medical Science, University of Campinas ‐ UNICAMP (clinical/content review), Karla Duque J. (consumer review), Nuala Livingstone, Cochrane Evidence Production and Methods Directorate (methods review), Yuan Chi, Cochrane Campbell Global Ageing Partnership (search review)
Appendices
Appendix 1. CENTRAL Ovid search strategy
Date Run: 23/06/2023 21:20:01
#1 [mh Constipation] or [mh "Fecal Impaction"] or (Constipat* or Fecalith or Dyschezia or Coprostasis or Colonic Inertia or ((F?ecal or F?eces or Stool?) NEAR/3 (Impact* or Retention or Retain* or Evacuat*)) or ((Bowel or Intestinal) NEAR/3 (Delayed or Retention or Retain* or Evacuat* or Function* or Habit* or Movement* or Symptom* or Motility)) or Obstipat* or Colon Transit or Def?ecat*):ti,ab,kw 24078
#2 [mh Recurrence] or [mh "Salvage Therapy"] or [mh Retreatment] or (Intractable or Unmanageable or Uncontrollable or Difficult to Control or Chronic or ((Non or "Not") NEAR/3 Respon*) or Unrespon* or Nonrespon* or Recurr* or Relaps* or Recrudescence* or Refractor* or Re?occur* or Redelop* or Exacerbate* or Re?appear* or Return* or Progress* or Periodic or Persist* or Deteriorat* or Failure or Failed or ((After or Following) NEAR/2 (Fail* or First Line or Withdr* or Cessation or Stop* or Discontin* or De‐Escalation)) or Re?treat* or Salvage or Rescue or Re?introduc*):ti,ab,kw 585799
#3 [mh Adolescent] or [mh Child] or [mh Infant] or [mh Minors] or [mh Pediatrics] or [mh Puberty] or [mh ^Schools] or [mh "Schools, Nursery"] or (Baby or Babies or Child* or P?ediatric* or P?adiatric* or Infan* or Neo?nat* or Post?Nat* or New?born* or Kid or Kids or Adolescen* or Pre?school* or Toddler* or Post?matur* or Pre?matur* or Pre?term* or Preemie or Perinat* or Boy* or Girl* or Teen* or Minors or Prepubescen* or Postpubescen* or Prepuberty or Pubescen* or Puber* or Elementary School* or High?School* or Kinder* or Jugend* or Nurser* or Primary School* or Secondary School* or Middle School* or Youth* or Young* or Student* or Juvenil* or School?Age* or Under?age* or Schoolchild* or Under 16 or Under 18):ti,ab,kw 515316
#4 #1 AND #2 AND #3 in Trials 2553
Appendix 2. MEDLINE Ovid search strategy
Database: Ovid MEDLINE(R) ALL <1946 to June 22, 2023>
1 exp Constipation/ or Fecal Impaction/ or (Constipat* or Fecalith or Dyschezia or Coprostasis or Colonic Inertia or ((F?ecal or F?eces or Stool?) adj3 (Impact* or Retention or Retain* or Evacuat*)) or ((Bowel or Intestinal) adj3 (Delayed or Retention or Retain* or Evacuat* or Function* or Habit* or Movement* or Symptom* or Motility)) or Obstipat* or Colon Transit or Def?ecat*).tw,kw. (76698)
2 exp Recurrence/ or exp Salvage Therapy/ or exp Retreatment/ or (Intractable or Unmanageable or Uncontrollable or Difficult to Control or Chronic or ((Non or "Not") adj3 Respon*) or Unrespon* or Nonrespon* or Recurr* or Relaps* or Recrudescence* or Refractor* or Re?occur* or Redelop* or Exacerbate* or Re?appear* or Return* or Progress* or Periodic or Persist* or Deteriorat* or Failure or Failed or ((After or Following) adj2 (Fail* or First Line or Withdr* or Cessation or Stop* or Discontin* or De‐Escalation)) or Re?treat* or Salvage or Rescue or Re?introduc*).tw,kw. (5468582)
3 exp Adolescent/ or exp Child/ or exp Infant/ or exp Minors/ or exp Pediatrics/ or exp Puberty/ or Schools/ or Schools, Nursery/ or (Baby or Babies or Child* or P?ediatric* or P?adiatric* or Infan* or Neo?nat* or Post?Nat* or New?born* or Kid or Kids or Adolescen* or Pre?school* or Toddler* or Post?matur* or Pre?matur* or Pre?term* or Preemie or Perinat* or Boy* or Girl* or Teen* or Minors or Prepubescen* or Postpubescen* or Prepuberty or Pubescen* or Puber* or Elementary School* or High?School* or Kinder* or Jugend* or Nurser* or Primary School* or Secondary School* or Middle School* or Youth* or Young* or Student* or Juvenil* or School?Age* or Under?age* or Schoolchild* or Under 16 or Under 18).tw,kw. (5712466)
4 1 and 2 and 3 (7107)
Note: 4 is Cochrane Highly Sensitive Search Strategy for identifying randomised trials in MEDLINE: sensitivity‐maximizing version (2008 revision); Ovid format
Appendix 3. Embase Ovid search strategy
Database: Embase <1974 to 2023 Week 24>
1 exp Constipation/ or Fecal Impaction/ or (Constipat* or Fecalith or Dyschezia or Coprostasis or Colonic Inertia or ((F?ecal or F?eces or Stool?) adj3 (Impact* or Retention or Retain* or Evacuat*)) or ((Bowel or Intestinal) adj3 (Delayed or Retention or Retain* or Evacuat* or Function* or Habit* or Movement* or Symptom* or Motility)) or Obstipat* or Colon Transit or Def?ecat*).tw,kw. (183058)
2 exp Recurrent Disease/ or exp Salvage Therapy/ or exp Retreatment/ or (Intractable or Unmanageable or Uncontrollable or Difficult to Control or Chronic or ((Non or "Not") adj3 Respon*) or Unrespon* or Nonrespon* or Recurr* or Relaps* or Recrudescence* or Refractor* or Re?occur* or Redelop* or Exacerbate* or Re?appear* or Return* or Progress* or Periodic or Persist* or Deteriorat* or Failure or Failed or ((After or Following) adj2 (Fail* or First Line or Withdr* or Cessation or Stop* or Discontin* or De‐Escalation)) or Re?treat* or Salvage or Rescue or Re?introduc*).tw,kw. (7674281)
3 exp Adolescence/ or exp Adolescent/ or exp Child/ or exp Kindergarten/ or exp Pediatrics/ or exp Puberty/ or School/ or High School/ or Middle School/ or Nursery School/ or Primary School/ or (Baby or Babies or Child* or P?ediatric* or P?adiatric* or Infan* or Neo?nat* or Post?Nat* or New?born* or Kid or Kids or Adolescen* or Pre?school* or Toddler* or Post?matur* or Pre?matur* or Pre?term* or Preemie or Perinat* or Boy* or Girl* or Teen* or Minors or Prepubescen* or Postpubescen* or Prepuberty or Pubescen* or Puber* or Elementary School* or High?School* or Kinder* or Jugend* or Nurser* or Primary School* or Secondary School* or Middle School* or Youth* or Young* or Student* or Juvenil* or School?Age* or Under?age* or Schoolchild* or Under 16 or Under 18).tw,kw. (6283592)
4 Randomized controlled trial/ or Controlled clinical study/ or randomization/ or intermethod comparison/ or double blind procedure/ or human experiment/ or (random$ or placebo or (open adj label) or ((double or single or doubly or singly) adj (blind or blinded or blindly)) or parallel group$1 or crossover or cross over or ((assign$ or match or matched or allocation) adj5 (alternate or group$1 or intervention$1 or patient$1 or subject$1 or participant$1)) or assigned or allocated or (controlled adj7 (study or design or trial)) or volunteer or volunteers).ti,ab. or (compare or compared or comparison or trial).ti. or ((evaluated or evaluate or evaluating or assessed or assess) and (compare or compared or comparing or comparison)).ab. (6335080)
5 (random$ adj sampl$ adj7 ("cross section$" or questionnaire$1 or survey$ or database$1)).ti,ab. not (comparative study/ or controlled study/ or randomi?ed controlled.ti,ab. or randomly assigned.ti,ab.) (9485)
6 Cross‐sectional study/ not (randomized controlled trial/ or controlled clinical study/ or controlled study/ or (randomi?ed controlled or control group$1).ti,ab.) (350346)
7 (((case adj control$) and random$) not randomi?ed controlled).ti,ab. (21660)
8 (Systematic review not (trial or study)).ti. (263055)
9 (nonrandom$ not random$).ti,ab. (18985)
10 ("Random field$" or (random cluster adj3 sampl$)).ti,ab. (4511)
11 (review.ab. and review.pt.) not trial.ti. (1123686)
12 "we searched".ab. and (review.ti. or review.pt.) (49996)
13 ("update review" or (databases adj4 searched)).ab. (63082)
14 (rat or rats or mouse or mice or swine or porcine or murine or sheep or lambs or pigs or piglets or rabbit or rabbits or cat or cats or dog or dogs or cattle or bovine or monkey or monkeys or trout or marmoset$1).ti. and animal experiment/ (1229787)
15 Animal experiment/ not (human experiment/ or human/) (2583504)
16 or/5‐15 (4348940)
17 4 not 16 (5590377)
18 and/1‐3,17 (3279)
19 limit 18 to (embase or conference abstracts) (3076)
Note: the latest RCT filter for Embase is from Cochrane Handbook for Systematic Reviews of Interventions.
Appendix 4. ClinicalTrials.gov search strategy
Advanced Search (Classic)
Condition or disease: Constipation
Study type: Interventional Studies (Clinical Trials)
Eligibility Criteria: Age: Child (birth–17)
156 Studies
Appendix 5. WHO ICTRP search strategy
Advanced Search
Constipation in the Condition
Search for clinical trials in children
Recruitment status is ALL
268 records for 242 trials
Appendix 6. Criteria for assigning risk of bias judgements using Cochrane’s RoB 1 tool
Sequence generation (i.e. was the allocation sequence adequately generated?). We will only consider randomised controlled trials for inclusion in the review. We will assess randomisation as at low risk of bias where the procedure for random sequence generation was explicitly described (e.g. computer‐generated random numbers). Where no description is given, we will contact the authors for further information. If no response is received, we will allocate a judgement of unclear risk of bias. We will judge any non‐random process (e.g. odd or even date of birth; hospital or clinic record number) as at high risk of bias.
Allocation concealment (i.e. was allocation concealment concealed?). We will assess concealment of treatment allocation as at low risk of bias if the procedure was explicitly described, and adequate efforts were made to ensure that intervention allocations could not have been foreseen in advance of, or during, enrolment (e.g. centralised randomisation, numbered or coded containers). Procedures considered to have a high risk of bias include alternation or reference to case record number or date of birth. Where no description of the method of allocation concealment is provided, we will contact the study authors. If we do not receive a response, we will assign a judgement of unclear risk of bias.
Blinding of participants, caregivers, and health professionals. Since the participants in this review are children, the caregivers will need to be blinded to the treatment allocations, alongside the healthcare professionals and study personnel, to permit a judgement of low risk of bias. We will judge open‐label trials as at high risk of bias for this domain. Where no description is given of the blinding of participants, caregivers, and health professionals, we will contact the study authors. If we do not receive a response, we will assign a judgement of unclear risk of bias.
Blinding of outcome assessment (i.e. were outcome assessors aware of the allocations?). We will describe the methods used, if any, to blind the outcome assessors from knowledge of which intervention a participant received. We will judge studies to be at low risk of bias if outcome assessors were blinded, or where we consider that lack of blinding would not have affected the results. If there was no blinding, or blinding was not possible because of the nature of the intervention, or was broken during the study, we will judge the study to be at high risk of bias, due to the possibility that the lack of blinding influenced the results. If no description is given, we will contact the study authors for more information. If we do not receive a response, we will assign a judgement of unclear risk of bias.
Incomplete outcome data (i.e. were incomplete outcome data adequately addressed?). We will assign a judgement of low risk of bias in the following instances.
If participants included in the analysis and those randomised into the trial are exactly the same.
If missing outcome data are balanced in terms of numbers across the intervention and control groups, with similar reasons.
If, for dichotomous outcome data, the proportion of missing outcomes compared with the observed event risk is not sufficient to have a clinically relevant impact on the intervention effect estimate.
If, for continuous outcome data, the plausible effect size (mean difference) among missing outcomes is not sufficient to have a clinically relevant impact on the observed effect size.
If missing data were imputed using appropriate methods.
We will assign a judgement of high risk of bias in the following instances.
When reasons for missing outcome data are likely to be related to the true outcome, with either an imbalance in numbers or reasons for missing data across intervention groups.
When, for dichotomous outcome data, the proportion of missing outcomes compared with the observed event risk is sufficient to induce clinically relevant bias in the intervention effect estimate.
When, for continuous outcome data, the plausible effect size (mean difference) among missing outcomes is sufficient to induce clinically relevant bias in the observed effect size.
When an 'as‐treated' analysis is carried out in cases where there is a substantial departure of the intervention received from that assigned at randomisation.
When there is a potentially inappropriate application of simple imputation.
We will assign a judgement of unclear risk of bias in the following instances.
When there is insufficient reporting of attrition or exclusions, or both, to permit a judgement of low or high risk of bias.
When the study reported incomplete outcome data.
When the trial did not clearly report the numbers randomised to the intervention and control groups.
Selective outcome reporting (i.e. are reports of the study free of the suggestion of selective outcome reporting?). We will assess the reporting of outcomes as at low risk of bias if all outcomes prespecified in the study protocol are reported in the study manuscript or secondary publications. If no protocol exists, we will assign a rating of low risk of bias if the authors report on the outcomes described in the methods section of the study manuscript. We will evaluate all study publications (primary and secondary) to ensure that there is no evidence of selective outcome reporting. If no description is given, we will contact the authors for more information. If we do not receive a response, we will assign a judgement of unclear risk of bias. If there is evidence of selective reporting (deviation from protocol, key planned outcomes not reported), we will assign a judgement of high risk of bias.
Other potential sources of bias (i.e. did the study appear to be free of other problems that could put it at a high risk of bias?). We will consider other potential sources of bias, including early trial termination (e.g. if a study was stopped early due to a data‐dependent process) and baseline imbalance between treatment groups. If we perceive that these other sources of bias are likely to threaten validity, we will assign a high risk of bias. We will assign a judgement of low risk of bias if the study appears to be free from such threats to validity. When the risk of bias is unclear from the published information, we will attempt to contact the study authors for clarification. If this is not forthcoming, we will assess these studies as being at unclear risk of bias.
See Higgins 2011; Higgins 2017.
Data and analyses
Comparison 1. Botulinum toxin A injection versus stool softeners.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Treatment success | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 37.00 [5.31, 257.94] |
| 1.2 Stool consistency | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.21, 0.59] |
| 1.3 Painful defecation | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 0.18 [0.09, 0.36] |
| 1.4 Faecal incontinence | 1 | 88 | Risk Ratio (M‐H, Random, 95% CI) | 0.16 [0.05, 0.50] |
Comparison 2. Lubiprostone versus placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Treatment success | 1 | 606 | Risk Ratio (M‐H, Random, 95% CI) | 1.29 [0.87, 1.92] |
| 2.2 Adverse events | 1 | 606 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.91, 1.21] |
| 2.3 Painful defecation | 1 | 606 | Mean Difference (IV, Random, 95% CI) | ‐0.16 [‐0.35, 0.03] |
| 2.4 Faecal incontinence | 1 | 606 | Mean Difference (IV, Random, 95% CI) | ‐0.03 [‐0.11, 0.05] |
| 2.5 Abdominal pain | 1 | 606 | Mean Difference (IV, Random, 95% CI) | ‐0.07 [‐0.20, 0.06] |
Comparison 3. Rectal sodium dioctyl sulfosuccinate and sorbitol versus oral polyethylene glycol laxatives.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 3.1 Treatment success | 1 | 102 | Risk Ratio (M‐H, Random, 95% CI) | 1.33 [0.83, 2.14] |
| 3.2 Painful defecation | 1 | 102 | Risk Ratio (M‐H, Random, 95% CI) | 2.50 [0.84, 7.46] |
| 3.3 Faecal incontinence | 1 | 102 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.75, 1.55] |
| 3.4 Abdominal pain | 1 | 102 | Risk Ratio (M‐H, Random, 95% CI) | 1.60 [0.80, 3.18] |
Comparison 4. Biofeedback therapy versus no intervention.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 4.1 Non‐fulfilment of Rome criteria | 1 | 24 | Risk Ratio (M‐H, Random, 95% CI) | 2.50 [1.08, 5.79] |
Comparison 5. Intrarectal electromotive botulinum toxin A versus botulinum toxin A injection.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 5.1 Non‐fulfilment of Rome criteria | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.76, 1.22] |
| 5.2 Frequency of defecation | 1 | 60 | Mean Difference (IV, Random, 95% CI) | 0.00 [‐1.87, 1.87] |
| 5.3 Adverse events | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.20 [0.01, 4.00] |
| 5.4 Stool consistency | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.69, 1.21] |
| 5.5 Painful defecation | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.75 [0.57, 5.36] |
| 5.6 Quality of life | 1 | 60 | Mean Difference (IV, Random, 95% CI) | 0.00 [‐5.04, 5.04] |
| 5.7 Faecal incontinence | 1 | 60 | Mean Difference (IV, Random, 95% CI) | 0.00 [‐1.70, 1.70] |
Comparison 6. Botulinum toxin A injection versus myectomy of the internal anal sphincter.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 6.1 Treatment success | 1 | 42 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.75, 1.34] |
| 6.2 Adverse events | 1 | 42 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 6.3 Painful defecation | 1 | 42 | Risk Ratio (M‐H, Random, 95% CI) | 1.44 [0.80, 2.62] |
| 6.4 Faecal incontinence | 1 | 42 | Risk Ratio (M‐H, Random, 95% CI) | 1.27 [0.77, 2.11] |
Comparison 7. Prucalopride versus placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 7.1 Frequency of defecation | 1 | 215 | Mean Difference (IV, Random, 95% CI) | 0.50 [‐0.06, 1.06] |
| 7.2 Treatment success | 1 | 215 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.53, 1.72] |
| 7.3 Adverse events | 1 | 215 | Risk Ratio (M‐H, Random, 95% CI) | 1.15 [0.94, 1.39] |
| 7.4 Painful defecation | 1 | 215 | Mean Difference (IV, Random, 95% CI) | ‐0.20 [‐0.54, 0.14] |
| 7.5 Quality of life (child‐reported) | 1 | 215 | Mean Difference (IV, Random, 95% CI) | 1.20 [‐2.31, 4.71] |
| 7.6 Quality of life (parent‐reported) | 1 | 215 | Mean Difference (IV, Random, 95% CI) | 2.40 [‐1.36, 6.16] |
| 7.7 Faecal incontinence | 1 | 215 | Mean Difference (IV, Random, 95% CI) | 5.20 [‐8.89, 19.29] |
| 7.8 Abdominal pain | 1 | 215 | Mean Difference (IV, Random, 95% CI) | 0.10 [‐0.13, 0.33] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Ahmadi 2013.
| Study characteristics | ||
| Methods |
Study design: randomised, open‐label trial Setting: secondary care (paediatric surgical clinic), single‐centre (Tehran University of Medical Sciences, Iran) Study period: not explicitly stated |
|
| Participants |
Case definition: chronic constipation for more than 3 months, and who had not responded to medical treatment Baseline investigations: perineal examination, anal manometry, and rectal biopsy. All had to be normal before entry into the study. Inclusion criteria: chronic constipation for more than 3 months that had not responded to medical treatment; ganglion cells in their rectal biopsy; high threshold recto‐anal inhibitory reflex (RAIR) or absent RAIR in manometry Exclusion criteria: abnormal anus or perineum, transition zone in barium enema Number randomised: IG 44, CG 44 Number analysed: IG not reported, CG not reported Postrandomisation dropouts (DO)/exclusions (E): IG not reported, CG not reported Number completing the trial: IG not reported, CG not reported Sex (M/F): IG 18/22, CG not reported Age: 2 to 12 years (range), 5 years (mode) Age of onset of constipation: not reported Duration of constipation: not reported |
|
| Interventions | Both groups had rectal disimpaction with phosphate and saline enemas before starting the study. Intervention group: in addition to stool softeners, botulinum toxin A was injected under general anaesthesia into 3 regions of the anal sphincter (3, 6, and 9 o'clock). The total dose of botulinum toxin was 160 units (80 units at 6 o'clock, 40 units at 3 o'clock, and 40 units at 9 o'clock). The toxin was injected into both internal and external anal sphincters. Control group: stool softeners (no further detail provided) Length of the intervention: one‐off procedure Timing of follow‐up: 6 months |
|
| Outcomes |
Method of outcome assessment: participants were asked questions about the presence of the signs of constipation including painful defecation, vomiting, stool consistency, soiling and defecation intervals. Note, no detail is provided on how these questions were asked (i.e. questionnaire, diary, interview). 1b. Frequency of defecation 1c. Treatment success 2a. Stool consistency 2b. Painful defecation 2d. Faecal incontinence |
|
| Notes |
Funding source: not stated Conflict of interest: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk |
Quote: "patients were randomly divided into cases and control group" Comment: no detail was provided on the specific method of randomisation. We attempted to contact the authors, but no response was received |
| Allocation concealment (selection bias) | Unclear risk | Comment: no detail was provided on the processes to conceal allocations. We attempted to contact the authors, but no response was received |
| Blinding of participants and personnel (performance bias) All outcomes | High risk |
Quote: "case group received botulinum toxin injection in addition to this therapy (stool softeners)" Comment: open‐label trial and therefore, high‐risk of bias |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: no reference was made to the tools that addressed the blinding of outcome assessors. We attempted to contact the authors, but no response was received |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: no data was presented on attrition, so unable to determine the risk of bias. We attempted to contact the authors, but no response was received |
| Selective reporting (reporting bias) | High risk | Comment: whilst no protocol is available in the public domain, the outcomes reported in the Results section of the study report match those described within the Methods section. The study authors report on the improvement in defecation frequency for the intervention group, but do not report the value for the control group. This is likely to represent a high risk of reporting bias |
| Other bias | Low risk | No concerns for other biases |
Bellomo‐Brandão 2003.
| Study characteristics | ||
| Methods |
Study design: randomised double‐blind, placebo‐controlled, cross‐over trial Setting: secondary care (paediatric gastroenterology outpatient clinic), single‐centre (State University of Campinas Medical School Hospital, Portugal) Study period: October 1998 to September 2000 |
|
| Participants |
Case definition: children with a chronic or lifelong history of constipation Baseline investigations: anorectal manometry, balloon expulsion test, liver function tests Inclusion criteria: stool passage frequency less than 3 times per week; the presence of diurnal and nocturnal soiling; the presence of faecal impaction as a filled rectum and identified by a palpable abdominal mass or by digital rectal examination; no previous or current disease known to affect gastrointestinal motility Exclusion criteria: previous or current disease known to affect gastrointestinal motility; history of gastrointestinal sub‐occlusive episodes; mechanical obstruction as determined by barium enema; outlet obstruction evaluated by defecography; absence of rectal anal inhibitory reflex detected by anorectal manometry; abnormal balloon expulsion test Number randomised: 21 (not split into IG and CG) Number analysed: IG 6, CG 8 Postrandomisation dropouts (DO)/exclusions (E): 7 (3, refusal to take any medication; 3, failure to return for a scheduled second medical visit; 1, failure to take the 4 daily doses of the test drug) Number completing the trial: IG 6, CG 8 Sex (M/F): IG 5/1, CG 6/2 Age (mean ± SD): IG 9.7 ± 3.0, CG 3.5 ± 2.6 years Age of onset of constipation (mean ± SD): IG 5.1 ± 3.8, CG 3.5 ± 2.6 years Duration of constipation (mean ± SD): IG 4.6 ± 3.6, CG 5.8 ± 1.9 years |
|
| Interventions | Children were admitted to hospital for faecal impaction removal by consecutive enemas. After colonic cleansing, an oral osmotic laxative was prescribed as maintenance therapy to establish a regular frequency of bowel movements. Lactulose (667 mg/mL) or magnesium hydroxide (80 mg/mL) was administered at a daily dose of 2 mL/kg, with a maximum dose of 60 mL. Children were instructed to use a saline‐glycol enema when there was no spontaneous stool movement after a 72‐hour period. The dose of the initially prescribed laxative was maintained, reduced, or increased according to stool frequency, faecal consistency, and stool passage conditions which occurred during the previous 2 weeks. The laxative dose was reduced when children mentioned liquid faecal consistency and increased evacuation in the presence of hard/scybalous faecal consistency or a stool frequency of less than 3 per week or enema requirement during the previous period. Intervention group: oral erythromycin estolate 20 mg/kg/day (split into 4 doses), given 6 hours apart. The maximum daily dose was 1000 mg. CG: placebo (no further details provided) Length of intervention: 4 weeks Measurement of outcome: week 2 and 4 |
|
| Outcomes |
Method of outcome assessment: children returned every 15 days, when they were asked about symptoms. A 12‐point score system was used which was based on stool frequency, faecal impaction, soiling, pain or difficulty at stool passage, faecal consistency, and daily laxative dose. 1d. Adverse events |
|
| Notes |
Funding source: not stated Conflict of interest: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: no details were provided on how the randomisation process was operationalised. We attempted to contact the authors, but no response was received |
| Allocation concealment (selection bias) | Unclear risk | Comment: no details were provided on the processes implemented to conceal the allocation. We attempted to contact the authors, but no response was received |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: no details were provided on the blinding processes. We attempted to contact the authors, but no response was received |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: no details were provided on the processes implemented to blind the assessors to the outcome. We attempted to contact the authors, but no response was received |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: 21 patients were randomised. Seven of these were excluded and not included in the analysis. Whilst the reasons for exclusion are described, the reasons per group are not. We attempted to contact the authors, but no response was received |
| Selective reporting (reporting bias) | Low risk | Comment: no protocol available. The outcomes described within the Methods section match the outcomes reported within the Results section |
| Other bias | Unclear risk | Comment: the baseline characteristics between the two groups are limited in information. It is therefore not possible to determine whether they were equivalent to each other. We attempted to contact the authors, but no response was received |
Benninga 2021.
| Study characteristics | ||
| Methods |
Study design: multicentre, double‐blind, placebo‐controlled trial Setting: secondary care, multicentre (96 sites across the USA, Canada, Europe) Duration: December 2013 to July 2016 |
|
| Participants |
Case definition: confirmed diagnosis of primary functional constipation according to the Rome III criteria. Email communication with the authors confirmed that children with constipation refractory to medical therapy were considered eligible for inclusion. Baseline investigations: not reported Inclusion criteria: children aged 6 to 17 years of age; patient diary (e‐diary) had to reflect > 70% compliant in recording evening or end‐of‐day assessments during the screening period, and indicate an average of less than 3 weekly spontaneous bowel movements, with < 25% of spontaneous bowel movements involving at least some straining and/or a 5‐point modified Bristol Stool Form Scale type 1 or 2. Exclusion criteria: child’s constipation was attributed to a physical, mental, or cognitive condition; inflammatory bowel disease; medication; or anatomic, neurological, or endocrine or metabolic factors; child was a candidate for or had undergone abdominal surgery, or had any condition other than constipation that could affect gastrointestinal motility or defecation; child had Hirschsprung’s disease; patient‐reported episodes of faecal incontinence not associated with stool retention; child experienced an unexplained significant weight loss; there was evidence of untreated faecal impaction at the time of screening. Number randomised: IG1 233, IG2 171, IG(combined) 404, CG 202 Number analysed (mITT population, at least 1 dose and 1 efficacy assessment): IG1 107, IG2 292, IG(combined) 399, CG 195 Number analysed (safety population, week 12, at least 1 dose): IG(combined) 400, CG 195 Number completing (at least 86 days of therapy): IG1 not reported, IG2 not reported, IG(combined) 297, CG 147 Reasons for difference between mITT population (1 dose and 1 efficacy assessment) and children completing treatment (at least 86 days of therapy): IG(combined): dose escalation 124, discontinued 65; CG: dose escalation 0, discontinued 55 Sex (M/F): IG1 54/53, IG2 129/163, IG(combined) 183/216, CG 89/106 Age (mean, years): IG1 9, IG2 12, IG(combined) 11, CG 11 Age of onset of constipation: not reported Duration of constipation: not reported |
|
| Interventions | Instructed to administer the doses of intervention/control at least 5 hours apart with at least 240 mL of fluid Intervention group 1 (IG1): oral lubiprostone 12 μg, twice a day (if weight < 50 kg) Intervention group 2 (IG2): oral lubiprostone 24 μg, twice a day (if weight > 50 kg) Control group: placebo (identical soft gelatin capsules) Length of the intervention: 12 weeks Time of follow‐up: week 2, 4, 6, 8, 10, 12 |
|
| Outcomes |
Method of outcome assessment: daily e‐diary by the parent or legal guardian or the child, as appropriate, from the initial screening visit through the end of the treatment period. 1c. Treatment success 1d. Adverse events 2b. Painful defecation 2d. Faecal incontinence 2e. Abdominal pain |
|
| Notes |
Funding source: not stated Conflict of interest: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "patients were assigned to treatment groups using a randomization code and stratification scheme generated by the Randomization and Trial Supply Management system" |
| Allocation concealment (selection bias) | Low risk | Quote: "randomization code and stratification scheme generated by the Randomization and Trial Supply Management system" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "treatment blinding among patients and their parents or legal guardians was maintained by providing study treatments in identical containers and as identical soft gelatin capsules" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: the primary endpoint was assessed based on the content recorded in the daily e‐diary by the parent or legal guardian or the patient from the initial screening visit through the end of the treatment period, who were blinded |
| Incomplete outcome data (attrition bias) All outcomes | Low risk |
Comment: within Supplementary Figure 1, the flow of patients through the study is presented. Based on the information within the flow diagram, 55 of the 202 patients randomised to the placebo arm did not complete treatment (defined as completing 84 days of treatment). Similarly, 107 of the 404 patients randomised to the Lubiprostone arm did not complete treatment. In the corresponding breakdown, the 'discontinued' numbers are given as 55/202 (placebo) and 65/404 (Lubiprostone). The figure legend states that the breakdown values refer to the reasons from the sponsor (based on 505 completes determined by the site investigators rather than the 444 participants who completed the study, defined as completing 84 days of treatment The sponsor for the trial responded to our request for clarification and explained that patients who were treated for <84 days were not counted within the 'discontinued' breakdown (equal to 19 in the placebo group and 42 in the treatment group). We received a breakdown with all the reasons for discontinuation in the placebo (n = 36) and intervention (n = 65) arms |
| Selective reporting (reporting bias) | Low risk | Comment: outcomes reported as per the trial registration (NCT02042183) |
| Other bias | Low risk | No concerns for other biases |
Bongers 2009.
| Study characteristics | ||
| Methods |
Study design: randomised, open‐label trial Setting: tertiary care (outpatient clinic for functional defecation disorders), single‐centre (Emma Children's Hospital Academic Medical Centre, the Netherlands) Study period: September 2001 to November 2005 |
|
| Participants |
Case definition: children with chronic constipation for at least 2 years and unresponsive to conventional treatment Baseline investigations: not reported Inclusion criteria: children between 8 and 18 years with functional constipation for at least 2 years and unresponsive to conventional treatment (functional constipation was defined as the presence of at least 2 of the following 4 symptoms: (1) spontaneous defecation frequency < 3 per week, (2) faecal incontinence episodes ≥ 2 per week, (3) passage of large‐diameter stools that might obstruct the toilet, and (4) palpable abdominal or rectal mass on physical examination) Exclusion criteria: children who had been treated with enemas on a regular basis (more than 3 rectal enemas in previous treatment); children with organic causes of constipation, including Hirschsprung’s disease, muscle disorders, prior rectoanal surgery, spina bifida, mental retardation, or hypothyroidism Number randomised: IG 51, CG 51 Number analysed: IG 50, CG 51 Postrandomisation dropouts (DO)/exclusions (E): IG 1 (DO), CG 1 (DO) Number completing the trial: IG 44, CG 43 Sex (M/F): IG 32/18, CG 33/17 Age (years): IG 10.5 (mean), 9.5 to 12.5 (range); CG 11.0 (mean), 9.2 to 11.8 (range) Age of onset of constipation (median (IQR)): IG 4.0 (3.0 to 5.0), CG 4.0 (3.0 to 5.0) years Duration of constipation (median (IQR)): IG 7.0 (5.1 to 9.3), CG 6.5 (4.7 to 9.3) years |
|
| Interventions | All children underwent rectal disimpaction by rectal enema (120 mL sodium dioctyl sulfosuccinate and sorbitol) on 3 consecutive days to achieve an empty rectum before starting the treatment trial. If rectal disimpaction was unsuccessful, rectal enemas were continued for a maximum of 7 days. Intervention group: 3 rectal enemas weekly during the first 3 months. Thereafter, this frequency was reduced by 1 enema per week every 3 months. Rectal enema consisted of 120 mL sodium dioctyl sulfosuccinate and sorbitol. Conventional therapy was delivered alongside the intervention. Control group: conventional therapy consisted of education, behavioural strategies, and oral laxatives. Oral laxative therapy consisted of polyethylene glycol, with a starting dose of 0.5 g/kg. If treatment was considered insufficient, the dose was optimised to a maximum of 1.5 g/kg. A rectal enema or bisacodyl suppository of 5 mg was only prescribed in case of reoccurrence of faecal impaction (control group only). Length of the intervention: 52 weeks Measurement of outcomes: week 12, 26, 39, 52 |
|
| Outcomes |
Method of outcome measurement: participants kept a bowel diary to record daily defecation frequency and faecal incontinence episodes, painful defecation, and abdominal pain. Visits to the outpatient clinic for evaluation of defecation pattern and laxative use were scheduled for all children at 2, 4, 6, 12, 26, 39, and 52 weeks. 1b. Frequency of defecation 1c. Treatment success 2b. Painful defecation 2d. Faecal incontinence 2e. Abdominal pain |
|
| Notes |
Funding source: not reported Conflicts of interest: declared ‐ no conflicts of interest |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "computerized randomisation was used to generate a sequence of random group assignment for consecutive patients" |
| Allocation concealment (selection bias) | Low risk | Comment: the study authors were contacted and confirmed this was done via sealed envelopes |
| Blinding of participants and personnel (performance bias) All outcomes | High risk |
Quote: "in addition to conventional treatment, three rectal enemas weekly during the first three months" Comment: open‐label trial and therefore, high‐risk of bias |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: the study authors were contacted and confirmed the study assessor was not blinded either |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Comment: six patients in the intervention group and seven in the control group discontinued the study (as per the published report). It is not clear by reading the treatment adherence paragraph in the Results section what happened to these 13 patients. We contacted the authors for clarification ‐ they were unable to find the data |
| Selective reporting (reporting bias) | High risk | Comment: the trial was registered retrospectively (ISRCTN99089299) |
| Other bias | Low risk | No concerns for other biases |
Castilla 2021.
| Study characteristics | ||
| Methods |
Study design: randomised trial Setting: secondary care (private paediatric gastroenterology centre), single‐centre (El Bosque University, Pediatric Gastroenterology, Hepatology and Nutrition, Colombia) Study period: April 2019 to December 2019 |
|
| Participants |
Case definition: Rome IV criteria and had been treated pharmacologically for more than 2 years Baseline investigations: high‐resolution anorectal manometry Inclusion criteria: Rome IV constipation, no response to pharmacological therapy (for more than 2 years) Exclusion criteria: not reported Number randomised: IG 12, CG 12 Number analysed: IG 12, CG 12 Postrandomisation dropouts (DO)/exclusions (E): 0 Number completing the trial: not reported Sex (M/F): 10/15 Age (median (IQR)): 10.6 (6) Age of onset of constipation: not reported Duration of constipation: not reported |
|
| Interventions |
Intervention group: biofeedback ‐ no further detail provided, apart from that 10 sessions (mean) were delivered per child Control group: not described Length of the intervention: not reported Timing of follow‐up: not reported |
|
| Outcomes |
Method of outcome measurement: not described 1a. Non‐fulfilment of the Rome IV criteria for functional constipation |
|
| Notes |
Funding source: not reported Conflicts of interest: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: no detail provided, apart from "simple random allocation". We attempted to contact the authors, but no response was received |
| Allocation concealment (selection bias) | Unclear risk | Comment: no detail provided. We attempted to contact the authors, but no response was received |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: no detail provided. We attempted to contact the authors, but no response was received |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: no detail provided. We attempted to contact the authors, but no response was received |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: all those randomised to the treatment arms were included in the final analyses |
| Selective reporting (reporting bias) | Low risk | Comment: the outcomes are not entirely clear in the Methods section; however, recovery from chronic constipation is defined and presented in the Results section |
| Other bias | Unclear risk | Comment: no baseline characteristic data was presented to be able to judge homogeneity between groups, apart from manometry sensitivity which was balanced between groups. We attempted to contact the authors, but no response was received |
Kajbafzadeh 2020.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial Setting: secondary care (paediatric outpatient clinic), single‐centre (Children's Medical Centre in Tehran, Iran) Study period: 2017 to 2019 |
|
| Participants |
Case definition: intractable functional constipation based on Rome III criteria. All children had failed to respond to at least 6 months of medical management (dietary modification and use of laxatives), biofeedback, and interferential electrical stimulation. Baseline investigations: measurement of rectum diameter by ultrasound and anorectal manometry Inclusion criteria: at least 2 of the following symptoms for at least 2 months: a maximum of 2 defecation times per week, at least 1 episode of incontinence after toilet training, painful defecation and passing hard stool with large diameter, positive history of faecal impaction or bowel movements that clogged the toilet; defecation frequency of fewer than 3 times per week; positive history for the passing of hard stool; episodes of faecal soiling; abnormal stool form (Bristol Stool Form 1 to 3); painful defecation; and a high anal sphincter pressure Exclusion criteria: inflammatory or metabolic diseases, neurologic and psychiatric disorders, Hirschsprung disease, and positive history of abdominal or anal sphincter surgeries Number randomised: IG 30, CG 30 Number analysed: IG 30, CG 30 Postrandomisation dropouts (DO)/exclusions (E): IG 0, CG 0 Number completing the trial: IG 30, CG 30 Sex (M/F): IG 16/14, CG 17/12 Age (mean ± SD, years): IG 7.1 ± 2.3 (range 4 to 12), CG 7.3 ± 2.4 (range 4 to 13) Age of onset of constipation: not reported Duration of constipation (median (IQR)): not reported |
|
| Interventions | Oral polyethylene glycol (PEG, 1 to 1.5 g/kg for a maximum of 7 days) and bowel washing or rectum irrigation were advised for children with a large faecal mass in the rectum for faecal disimpaction. The laxative regimen (PEG, 0.2 to 0.6 g/kg/day) was continued for all children during the study. The dose of laxative was reduced in children who responded to the treatment after 1‐month follow‐up and was then discontinued in children who sustained their response to treatment after 6 months of follow‐up. Both groups also received conventional treatment including advice to consume a high‐fibre diet, hydration, toilet training, correct defecation posture, to sit on the toilet 3 times a day after mealtimes in a relaxed position for 5 minutes. Intervention group: intrarectal electromotive botulinum toxin A. Electromotive therapy involved 20 minutes of current at a frequency of 2800 Hz, interval 50 μs, and amplitude of 10 to 15 mA (after colonic irrigation). BoNTA (Dysport, Ipsen Limited, Slough, United Kingdom) was added at a dose of 10 IU/kg to an irrigated rectum that had been filled with saline solution to its maximal capacity. Control group: routine intrasphincteric botulinum toxin A administration (as above) Length of the intervention: one‐off intervention Timing of follow‐up: month 1 and 6 Method of outcome measurement: parent diary of bowel habits, frequency of defecation per week, stool form, and the number of painful defecation episodes. Investigators recorded a paediatric incontinence/constipation score questionnaire (scale 0 to 29) and a visual pain scale (0 to 10, 10 being the worst) according to the child's/parent's report. A constipation‐related quality of life questionnaire was completed for participants in both groups before and 6 months after treatment. The quality of life questionnaire comprised 37 questions, containing 4 fields: constipation‐related (3 items), emotional functioning (13 items), social functioning (11 items), and treatment/intervention (10 items). |
|
| Outcomes |
1a. Non‐fulfilment of Rome IV criteria 1b. Defecation frequency 1c. Adverse events 2a. Stool consistency 2b. Painful defecation 2c. Quality of life 2d. Faecal incontinence |
|
| Notes |
Funding source: Vice Chancellor for Research of Tehran University of Medical Sciences supported the study by providing a financial grant. Conflicts of interest: declared ‐ nil reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "a computer‐generated list of random numbers was used" |
| Allocation concealment (selection bias) | Unclear risk | Comment: no detail provided. We attempted to contact the authors, but no response was received |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: this study was not blinded, as the intervention group received an additional procedure to the control group |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: the measures implemented to blind the assessors to the allocations was not described. We attempted to contact the authors, but no response was received |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: all those randomised to both the intervention and control groups were accounted for in the analyses |
| Selective reporting (reporting bias) | High risk | The trial registration had been registered retrospectively, and the only outcome reported in this protocol was defecation frequency per week. In the Methods section of the study report, the primary outcome is defined as the resolution of symptoms. In the Results section, data is presented related to improvement in symptoms, but not resolution. We attempted to contact the authors, but no response was received |
| Other bias | Low risk | No concerns for other biases |
Keragiozoglou‐Lampoudi 2012.
| Study characteristics | ||
| Methods |
Study design: prospective randomised trial Setting: secondary care (outpatient paediatric gastroenterology clinic), single‐centre (Department of Nutrition and Dietetics, Alexander Technological Educational Institute of Thessaloniki, Greece) Study period: not reported |
|
| Participants |
Case definition: functional constipation diagnosed according to the NASPGHAN criteria. Constipation had to be refractory to treatment by a paediatrician for several months or presented with complications, or both. Baseline investigations: not reported Inclusion criteria: not stated Exclusion criteria: not stated Number randomised: IG 44, CG 42 Number analysed: IG 44, CG 42 Postrandomisation dropouts (DO)/exclusions (E): not reported Number completing the trial: IG 44, CG 42 Sex (M/F): 44/42 (not reported for IG and CG) Age (mean (range)): 4.4 (1 to 11) years (not reported for IG and CG) Age of onset of constipation: not reported Duration of constipation: not reported |
|
| Interventions | Both groups were treated according to NASPGHAN guidelines with pharmaceutical treatment, including lactulose together with instruction for diet modification. Intervention group: in addition to paediatric gastroenterologist written instruction, each child and their parent had a further same‐day appointment with a registered dietitian who prescribed a personalised diet (7‐day diet plan) based on the Mediterranean‐type eating plan and calculated to cover the personal energy, nutrient, water, and fibre requirements of paediatric patients. Control group: parents were given written instruction about their children's diet with examples provided by a paediatric gastroenterologist. Length of intervention: one‐off intervention Measurement of outcome: 4 weeks |
|
| Outcomes | Method of outcome measurement: 24‐hour diet recall by a registered dietitian | |
| Notes |
Funding source: not reported Conflicts of interest: declared ‐ no conflicts of interest |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: no details were provided on the specific method of randomisation. We attempted to contact the authors, but no response was received |
| Allocation concealment (selection bias) | Unclear risk | Comment: no details were provided on the allocation concealment processes. We attempted to contact the authors, but no response was received |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: whilst blinding was not performed, it was not felt to influence the outcomes of this study |
| Blinding of outcome assessment (detection bias) All outcomes | High risk |
Quote: "dietary intake was assessed using a 24‐hour diet recall by a registered dietician" Comment: given this study was comparing physician instruction to dietician‐provided diet plans, outcome assessment by a dietician risks detection bias in favour of the intervention group |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: all those randomised to both the intervention and control groups were accounted for in the analyses |
| Selective reporting (reporting bias) | Unclear risk | Comment: whilst no protocol for this study is available, all outcomes reported within the Results section of the written report match those described within the Methods section. Despite this, for a study involving those with intractable constipation, it would be expected that outcomes with a clinical focus would be reported |
| Other bias | Low risk | No concerns for other biases |
Keshtgar 2007.
| Study characteristics | ||
| Methods |
Study design: double‐blind, randomised trial Setting: tertiary care (inpatient paediatrics), single‐centre (Guy's and St Thomas' Hospital, United Kingdom) Study period: April 2001 to April 2003 |
|
| Participants |
Case definition: chronic idiopathic constipation, defined as failure to respond to laxative treatment and manual evacuation of stool with or without anal dilatation under anaesthesia. Duration of laxative therapy (median (IQR)): IG 80 (5 to 144), CG 66 (13 to 117) months Baseline investigations: intestinal transit study, anorectal manometry, anal endosonography, plain film radiography Inclusion criteria: 4 to 16 years of age with chronic idiopathic constipation. At least 2 of the following 4 criteria should have been fulfilled for the definition of constipation and faecal soiling: stool frequency of fewer than 3 times per week; soiling frequency of 2 or more times per week; periodic passage of large amounts of stool at least once every 7 to 30 days; and presence of a palpable abdominal or rectal mass. Exclusion criteria: underlying anorectal anomaly or endocrine abnormality; neuropathic bowel; learning difficulties; and evidence of anal sphincter damage on endosonography or a low anal sphincter pressure of less than 30 mmHg on manometry Number randomised: IG 21, CG 21 Number analysed: IG 21, CG 21 (3 months); IG 20 (1 = lost to follow‐up), CG 20 (1 = lost to follow‐up) (12 months) Postrandomisation dropouts (DO)/exclusions (E): IG 0, CG 0 Number completing the trial: IG 21, CG 21 Sex (M/F): IG 11/10, CG 14/7 Age (median (IQR)): IG 10 (6.0 to 14.7), CG 8.6 (4.0 to 14.9) years Age of onset of constipation: not reported Duration of constipation: not reported Age of onset of constipation: not reported Duration of constipation (median (IQR)): IG 94 (5 to 156), CG 74 (13 to 149) months |
|
| Interventions | Children stayed in the hospital postoperatively and received intensification of laxative treatment and toilet training, and were assessed by a child psychologist. Children were encouraged to establish a regular pattern of bowel movement and laxative intake (including methylcellulose as a stool softener and senna as a bowel stimulant). Movicol and sodium picosulfate were given if the child did not pass bowel motion for 3 days. Intervention group: injection of botulinum toxin into the internal anal sphincter. Performed under general anaesthesia, 15 units of botulinum type A were injected per quadrant (total dose = 60 units) into the internal anal sphincter. Control group: myectomy of the internal anal sphincter. This was achieved by making a small transverse incision on the dentate line at the 3 o'clock position, and a plane developed between the mucosa and muscle. A strip of the internal anal sphincter was excised proximally into the distal rectum with a length of approximately 5 cm and a width of approximately 0.5 cm. Length of the intervention: one‐off procedure Timing of follow‐up: 3, 6, and 12 months |
|
| Outcomes |
Method of outcome measurement: symptom severity score questionnaire (0 = best, 65 = worst) 1c. Treatment success 1d. Adverse events 2b. Painful defecation 2d. Faecal incontinence |
|
| Notes |
Funding source: not reported Conflict of interest: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "computer‐generated sealed‐envelope allocation system" |
| Allocation concealment (selection bias) | Low risk | Quote: "computer‐generated sealed‐envelope allocation system" |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: given the invasive nature of the procedures, it would not have been possible to blind the surgeons to the allocations. Of note, all procedures were carried out by authors of this review |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "post‐operative assessments were done by a paediatrician who was blinded to the treatment randomisation" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: full reporting of patient flow, data, and outcomes through the study |
| Selective reporting (reporting bias) | Low risk | Comment: whilst no protocol for this study is available, all outcomes reported within the Results section of the written report match those described within the Methods section |
| Other bias | Low risk | No concerns for other biases |
Mugie 2014.
| Study characteristics | ||
| Methods |
Study design: double‐blind, placebo‐controlled and an open‐label, active‐controlled period Setting: secondary care, multicentre (33 centres across Europe) Study period: April 2011 to March 2013 |
|
| Participants |
Case definition: functional constipation based on the Rome III criteria. Duration of symptoms (mean ± SD): IG 4.4 ± 4.2, CG 4.2 ± 3.9 years. The majority of children had 'severe' or 'very severe' constipation (81.7%), painful or hard bowel movements (85.9%), and a large faecal mass in the rectum (79.7%). Baseline investigations: not reported Inclusion criteria: aged between 6 months and 18 years with functional constipation, defined as < 3 spontaneous bowel movements/week with at least 1 of the following during the previous month (for children aged < 4 years) or 2 months (for children aged ≥ 4 years): ≥ 1 episode of faecal incontinence/week (after the acquisition of toileting skills); retentive posturing or excessive volitional stool retention; painful or hard bowel movements; large‐diameter stools; or a large faecal mass in the rectum Exclusion criteria: underlying cause of defecation disorder (e.g. Hirschsprung’s disease, spina bifida occulta, cystic fibrosis, or gastrointestinal malformations); significant developmental delays associated with musculoskeletal or neurologic conditions affecting the gastrointestinal tract; constipation secondary to endocrine, metabolic, neurologic, organic, autoimmune disorders, surgery, or drugs; clinically significant cardiac, vascular, liver, pulmonary, or psychiatric disorders; severe renal insufficiency; HIV; AIDS; hepatitis B; hepatitis C; or clinically significant abnormalities of haematology, urinalysis, or blood biochemistry at screening. Children with known lactose intolerance for whom it was expected that low doses of lactose could lead to diarrhoea or those who were known to have an allergy to one of the investigational drugs or its excipients were also excluded. Children who were breastfed during the study or who used any investigational drug within the 30 days preceding screening were also excluded. Number randomised: IG 107, CG 108 Number analysed (efficacy): IG 107, CG 108 Number analysed (safety, at least 1): IG 106, CG 107 Postrandomisation dropouts (DO)/exclusions (E): IG 21 (10, early withdrawal; 6, withdrew consent; 2, non‐compliance; 1, ineffective treatment; 1, adverse event; 1, lost to follow‐up; 1, did not fulfil the inclusion criteria); CG 13 (6, early withdrawal; 6, withdrew consent; 1, non‐compliance; 1, ineffective treatment; 1, adverse event) Number completing the trial: IG 96, CG 101 Sex (M/F): IG 46/60, CG 49/58 Age (mean ± SD): 8.3 ± 4.5, CG 8.2 ± 4.7 years Age of onset of constipation (mean ± SD): IG 4.4 ± 5.5, CG 4.2 ± 3.9 years Duration of constipation (median (IQR)): not reported |
|
| Interventions | Children were not to change their lifestyle or diet. Legal guardians of children aged ≥ 4 years were instructed to continue with toilet training (defined as at least 3, 5‐minute visits to the toilet in a silent, relaxed atmosphere after each meal) during the study. The dosing schedule was 1 to 3 hours before evening meals. Laxatives and agents that influence bowel habits were not permitted during the run‐in period. If the child did not have a bowel movement for ≥ 3 consecutive days, they could take 5 mg bisacodyl or 7.5 mg/mL sodium picosulfate droplets (1 droplet per 5 kg body mass) for rescue purposes. If the standard dose was insufficient, an increase was allowed after discussion with the investigator. If the child had no bowel movements, an enema (e.g. sodium dioctyl sulfosuccinate and sorbitol) or oral agent (e.g. polyethylene glycol 3350) could be administered to remove the impaction. Intervention group: oral prucalopride (prucalopride succinate oral solution). For children weighing ≤ 50 kg, an initial dose of 0.04 mg/kg body weight, once daily was used. After 4 weeks, the dose could be increased to 0.06 mg/kg or decreased to 0.02 mg/kg, based on treatment response and the presence of safety/tolerability. Children who underwent dose adjustment remained on that dose for the remainder of the double‐blind period. Children weighing > 50 kg received 2 mg prucalopride. Control group: oral placebo (identical in appearance and taste to the interventional agent) Length of the intervention: 8 weeks Timing of follow‐up: week 2, 4, 8 |
|
| Outcomes |
Method of outcome measurement: participants visited the study centre for efficacy and safety assessments. Quality of life was evaluated using the Pediatric Quality of Life Inventory Generic Core Scales Version 4.0 (PedsQOL) and the PedsQL gastrointestinal symptoms module. 1b. Frequency of defecation 1c. Treatment success 1d. Adverse events 2b. Painful defecation 2c. Quality of life 2d. Faecal incontinence frequency 2e. Abdominal pain |
|
| Notes |
Funding source: declared ‐ study was sponsored by Shire Development LCC Conflict of interest: these authors disclose the following: Alexandra Green and Amy Levine are employees of Shire. Rene Kerstens, Jannie Ausma, and Magnus Ruth are former employees of Shire‐Movetis. Marc A Benninga is a consultant for AstraZeneca, Danone, Shire, Sucampo, and Zeria. The remaining authors disclose no conflicts. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "randomization was organized using a central interactive web‐based, voice‐response system, which applied a minimization algorithm and generated a medication number to ensure blinding" |
| Allocation concealment (selection bias) | Low risk |
Quote: "patients and investigators were blinded to treatment allocation" Comment: whilst stated in the study report, no detail was provided on how this was achieved. We contacted the study authors who confirmed this was achieved via a computer system |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Quote: "patients and investigators were blinded to treatment allocation; placebo was identical in taste and appearance to prucalopride" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: blinding of the outcome assessors is not explicitly referred to within the study report. We contacted the study authors who confirmed the outcome assessors were blinding to participant allocations |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: full reporting of patient flow through the study. Equal and transparently reported attrition between the intervention and control groups |
| Selective reporting (reporting bias) | Low risk | Comment: outcomes reported as described in the prospectively registered trial protocol (NCT01330381) |
| Other bias | Low risk | No concern for other biases |
Southwell 2012.
| Study characteristics | ||
| Methods |
Study design: randomised, placebo‐controlled trial Setting: tertiary care (surgical outpatient clinic), single‐centre (Royal Children's Hospital, Melbourne, Australia) Study period: not explicitly stated |
|
| Participants |
Case definition: defined by the Rome II criteria. Average duration of symptoms (mean): IG 10.6, CG 8.5 years Baseline investigations: slow‐transit constipation confirmed by radioisotope nuclear transit study Inclusion criteria: aged between 7 and 18 years of age and had been treated for constipation for a minimum of 2 years Exclusion criteria: metabolic or hormonal causes for constipation; cognitive impairment or anorectal retention/normal transit on their nuclear transit study were included Number randomised: 46 (not split into IG and CG) Number analysed: not reported Postrandomisation dropouts (DO)/exclusions (E): not reported Number completing the trial: IG 21, CG 21 Sex (M/F): not reported Age (mean): IG 12.1 (range 7.4 to 17.7), CG 11.4 (range 7.8 to 16.5) Age of onset of constipation (mean): IG 8.5, CG 10.6 years Duration of constipation (range): IG 2.7 to 14.4, CG 4.4 to 15.1 years |
|
| Interventions |
Intervention group: 12 x 20‐minute sessions of real interferential therapy (IFT) over 4 weeks. 2 electrodes were placed paraspinally and 2 over the abdomen. Active stimulation was applied by physiotherapists at a comfortable intensity (< 40 mA, carrier frequency 4 kHz, varying beat frequency 80 to 150 Hz). Control group: placebo therapy (as above, but with a frequency and sweep of 0) Length of the intervention: 4 weeks Timing of follow‐up: 8 weeks |
|
| Outcomes | ||
| Notes |
Funding source: NHMRC and Murdoch Children's Research Institute Australia Conflict of interest: BR has received research funding from NHMRC Australia and co‐owns a patent on this method. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: not described within the study report. We attempted to contact the study authors for further information, but no response was received |
| Allocation concealment (selection bias) | Unclear risk | Comment: not described within the study report. We attempted to contact the study authors for further information, but no response was received |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Comment: no information provided on the measures to blind participants and personnel. Aside from stating 'double‐blind', no further information was provided. We attempted to contact the authors for further information, but no response was received |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Comment: no information provided on whether the outcome assessors were blinded to the treatment allocations. We attempted to contact the authors for further information, but no response was received |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: full flow of participants through the study is described. Clear and transparent reporting |
| Selective reporting (reporting bias) | Low risk | Comment: Retrospective trial registration available. The outcomes described within the Methods section match the outcomes reported within the Results section |
| Other bias | Low risk | No concerns for other biases |
CG: control group IG: intervention group IQR: interquartile range IU: international units mTT: modified intention‐to‐treat NASPGHAN: North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition NHMRC: National Health and Medical Research Council SD: standard deviation
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bekkali 2012 | The study reports including people with functional constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Broide 2001 | The study reports including people with functional constipation, but does not provide detail on previous therapy delivered. We are not able to describe this population as intractable constipation. |
| Burnett 2004 | Variety of treatments offered within a nurse‐led clinic, compared to attendance to a general paediatric clinic (consultant‐led). This is not an interventional study. |
| Clarke 2012 | Not a randomised controlled trial. Case series of children with slow‐transit constipation treated with interferential current. |
| Dehghani 2012 | This study focused on children with chronic constipation for more than 3 months' duration, but does not describe the previous therapy tried. It is therefore not possible to classify this cohort as intractable constipation. |
| Di Lorenzo 2020 | Trial included people with functional constipation of at least 2 months' duration prior to enrolment. This does not meet the criteria for intractable constipation used in the review. |
| Díaz 2020 | The study reports including people with functional constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Gondo 2020 | Trial included people with functional constipation of at least 2 months' duration prior to enrolment. This does not meet the criteria for intractable constipation used in the review. |
| Gremse 2000 | The study reports including people with chronic constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Heemskerk 2021 | This study protocol proposes including participants aged 14 to 80 years of age, with no indication of subgroup analyses for participants less than 18 years. It therefore lies outside the scope of this review. |
| Ismail 2009 | This study included people who relapsed (constipation) following cessation of transcutaneous electric stimulation, received as part of another RCT. Within this study, people who previously completed another RCT were offered the opportunity to continue therapy within their home environment. We have excluded this study on the grounds of incorrect study design. |
| ISRCTN24521269 | Trial protocol was registered in 2005. No publication since, and the authors listed were not contactable. We have excluded this as incorrect study design (protocol only, with no study report). |
| Keshtgar 2005 | The study reports including people with chronic constipation, but does not provide detail on the chronicity. From the baseline demographic results, it is noted that the range of symptoms in the control group includes 2 months, which falls outside the case definition used for intractable constipation in this review. |
| Khan 2020 | This study is not a randomised trial. Participants were split into 4 groups; however, a randomised methodology was not incorporated. We have excluded this study on grounds of incorrect study design. |
| Loening‐Baucke 2006 | Trial included people with functional constipation of at least 2 months' duration prior to enrolment. This does not meet the criteria for intractable constipation used in the review. |
| Modin 2018 | The study reports including people with functional constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Nakajima 2019 | This study included participants ≥ 15 years of age, therefore does not focus exclusively on a paediatric population. |
| NCT03054805 | Trial protocol; no study report published. Trial included people with functional constipation of at least 1 month duration prior to enrolment. This does not meet the criteria for intractable constipation used in the review. |
| Nurko 2000 | The study reports including people with functional constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Peng 2006 | This study included people above the age of 18 and thus did not include any children. It therefore lies outside the scope of this review. |
| Radwan 2021 | The study reports including people with functional constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Strisciuglio 2021 | The study reports including people with functional constipation, but does not provide detail on the chronicity or how refractory the disease was. |
| Thomson 2008 | The study reports including people with functional constipation, but does not provide detail on previous therapy delivered. We note from the baseline demographic tables that ~92% participants had tried laxative therapy, meaning not all participants have tried conventional laxative therapy. The study therefore does not meet the inclusion criteria of the review due to failure to meet the case definition for intractable constipation. |
| Velasco‐Benitez 2023 | Not an RCT |
| Weifeng 2021 | This study included people above the age of 18 years and thus did not include any children. It therefore lies outside the scope of this review. |
RCT: randomised controlled trial
Characteristics of studies awaiting classification [ordered by study ID]
ACTRN12620000131954.
| Methods |
Study design: double‐blind, randomised, placebo‐controlled Setting: secondary care, single‐centre (Waikato Hospital, Australia) Study period (proposed): August 2020 to February 2021 |
| Participants |
Case definition: Rome IV criteria. No detail provided on the chronicity or length of disease. This would need clarifying before inclusion in subsequent reviews. Inclusion criteria: children aged between 5 and 15 years; primary diagnosis of chronic idiopathic constipation. Exclusion criteria: people with an identified organic cause for constipation Sample size target: 108 |
| Interventions |
Intervention group: parents of children will be trained to use the transcutaneous electrical stimulator at home. Devices will deliver 4‐kilohertz carrier frequency, a beat frequency of 80 to 160 Hz with an intensity of less than 33 mA. Control group: the control arm will be given identical devices modified by the manufacturer to deliver a similar 4‐kilohertz carrier frequency with a beat frequency of 80 to 160 Hz with an intensity of less than 33 mA. This is identical to the treatment arm. After the first minute of application, devices provided to the control arm will be modified to gradually ramp down and stop all current delivered over a period of 5 minutes. Length of the intervention: 1 hour of treatment per day over a 6‐week period Timing of follow‐up: 6 weeks |
| Outcomes |
Primary outcomes: mean number of spontaneous bowel motions per week between treatment and control groups (measured using bowel diaries) Secondary outcomes: mean number of soiling accidents per week (measured using bowel diaries) |
| Notes |
NCT05035784.
| Methods |
Study design: randomised, double‐blind controlled trial Setting: secondary care, single‐centre Study period (proposed): August 2019 to March 2022 Status: recruiting |
| Participants |
Case definition: Rome IV criteria. No detail provided on the chronicity or length of the disease. This would need clarifying before inclusion in subsequent reviews. Inclusion criteria: 4 to 14 years of age. Rome IV criteria for childhood constipation; after a course of polyethylene glycol and a course of Chinese medicine treatment were ineffective; barium enema showing faecal impaction. Exclusion criteria: congenital and/or acquired intestinal diseases, such as congenital megacolon, intestinal stenosis, polyps, Crohn's disease, tuberculosis, inflammation, and tumours; anorectal diseases, such as anal atresia, fistula, abscess, and tumour; neurological diseases, such as brain and spinal cord diseases; genetic metabolic diseases; psychosocial and behavioural diseases; other systemic diseases. Sample size target: 110 |
| Interventions |
Intervention group: faecal supernatant Control group: a placebo designed to match the faecal bacterial transplantation + retrograde colonic enema group based on appearance Length of the intervention: one‐off procedure Timing of follow‐up: week 4 and 12 |
| Outcomes |
Primary outcome: frequency of defecation Secondary outcomes: frequency of bowel movements per week, painful or hard bowel movements, large‐diameter or scybalous stools, excessive volitional stool retention, encopresis |
| Notes |
Characteristics of ongoing studies [ordered by study ID]
NCT05059756.
| Study name | PTNS and PFR in the treatment of childhood constipation |
| Methods |
Study design: randomised, sham‐controlled trial Setting: secondary care, single‐centre Study period (proposed): September 2020 to May 2020 Status: recruiting |
| Participants |
Inclusion criteria: 4 to 14 years old; meeting the Roman IV criteria for childhood constipation; 1 course of polyethylene glycol and 1 course of Chinese medicine treatment (ineffective); pelvic floor surface electromyography (EMG) and 3‐D manometry of the anus revealed pelvic floor dysfunction. Exclusion criteria: intestinal stenosis due to organic diseases (e.g. anal fissure, inflammation, intestinal polyps, intestinal adhesion, Crohn's disease, intestinal tuberculosis, tumour, etc.); constipation due to congenital diseases (e.g. congenital megacolon, sigmoid colon, etc.); metabolic endocrine diseases, neurological diseases, and mental diseases; children diagnosed as outlet obstructive constipation and mixed functional constipation; children with severe systemic diseases; children with positive occult blood in stool routine examination. Sample size target: 84 |
| Interventions |
Intervention group: PTNS and PFR (twice daily) Control group: sham PTNS and PFR (twice daily) Length of the intervention: not described Timing of follow‐up: week 4 and 12 |
| Outcomes |
Primary outcome: frequency of defecation Secondary outcomes: frequency of bowel movements per week, painful or hard bowel movements, large‐diameter or scybalous stools, excessive volitional stool retention, encopresis |
| Starting date | 8 September 2020 |
| Contact information | Shucheng Zhang (zhangshucheng76@126.com) |
| Notes |
PFR: pelvic floor rehabilitation PTNS: percutaneous tibial nerve stimulation
Differences between protocol and review
The number of included studies and heterogeneity in trial interventions and reported outcomes precluded meta‐analyses and subsequent subgroup or sensitivity analyses.
For completeness, we decided to present summary of findings tables for every comparison, even if none of the primary outcomes were reported.
We made a minor change from the study protocol for the primary outcome symptom resolution, defined as non‐fulfilment of the Rome criteria for functional constipation. We have now removed the iteration of the Rome criteria, which was specified as Rome IV criteria in the protocol. Since many of the studies were published before the Rome IV criteria, we have instead extracted data for non‐fulfilment based on any of the Rome criteria.
Contributions of authors
MG: secured funding, designed and developed, screened, extracted, resolved conflicts, assessed the certainty of the evidence, contributed to writing and editing, advised on, approved the final version prior to submission, and is a guarantor of the review.
CGC: screened, extracted, resolved conflicts, assessed the certainty of the evidence, contributed to writing and editing, advised on, approved the final version prior to submission.
SR: screened, extracted, resolved conflicts, assessed the certainty of the evidence, contributed to writing and editing, advised on, approved the final version prior to submission.
MB: contributed to writing and editing, advised on, approved the final version prior to submission.
VS: screened, extracted, resolved conflicts, assessed the certainty of the evidence, contributed to writing and editing, advised on, approved the final version prior to submission.
AA: advised on, approved the final version prior to submission.
Sources of support
Internal sources
-
LIFE Research Starter Award, UK
A University of Central Lancashire award for early career researchers
External sources
-
None, Other
No external sources of support
Declarations of interest
MG: no relevant interests; published multiple Cochrane reviews relevant to the interventions in this review; NHS consultant; Co‐ordinating Editor for Cochrane Gut but was not involved in the editorial process or decision‐making for this review.
CGC: none known.
SR: no relevant interests; Professor of Paediatrics, Faculty of Medicine, University of Colombo, Sri Lanka.
MB: no relevant interests; Paediatric Gastroenterologist in an academic medical hospital.
VS: none known.
AA: no relevant interests; Physician, Sidra Medicine; Co‐ordinating Editor for Cochrane Gut but was not involved in the editorial process or decision‐making for this review.
New
References
References to studies included in this review
Ahmadi 2013 {published data only}
- Ahmadi J, Azary S, Ashjaei B, Paragomi P, Khalifeh-Soltan A. Intrasphincteric botulinum toxin injection in treatment of chronic idiopathic constipation in children. Iranian Journal of Pediatrics 2013;23(5):574-8. [PMID: ] [PMC free article] [PubMed] [Google Scholar]
Bellomo‐Brandão 2003 {published data only}
- Bellomo-Brandao MA, Collares EF, da-Costa-Pinto EAL. Use of erythromycin for the treatment of severe chronic constipation in children. Brazilian Journal of Medical and Biological Research 2003;36(10):1391-6. [DOI: 10.1590/s0100-879x2003001000016] [PMID: ] [DOI] [PubMed] [Google Scholar]
Benninga 2021 {published data only}
- Benninga MA, Hussain SZ, Sood MR, Nurko S, Hyman P, Clifford RA, et al. Lubiprostone for pediatric functional constipation: randomized, controlled, double-blind study with long-term extension. Clinical Gastroenterology and Hepatology 2021;20(3):602-10. [DOI: 10.1016/j.cgh.2021.04.005] [PMID: ] [DOI] [PubMed] [Google Scholar]
- NCT02042183. Lubiprostone for children with constipation [A multicentre, long-term safety, efficacy, and pharmacokinetics study of lubiprostone in paediatric subjects aged ≥ 6 years to < 18 years with functional constipation]. clinicaltrials.gov/ct2/show/NCT02138136 (first received 14 May 2014).
Bongers 2009 {published data only}
- Bongers MEJ, den Berg MM, Reitsma JB, Voskuijl WP, Benninga MA. A randomized controlled trial of enemas in combination with oral laxative therapy for children with chronic constipation. Clinical Gastroenterology and Hepatology 2009;7(10):1069-74. [DOI: 10.1016/j.cgh.2009.06.018] [PMID: ] [DOI] [PubMed] [Google Scholar]
- ISRCTN99089299. The effect of additional use of enemas versus the standard treatment of chronic constipation in children. www.isrctn.com/ISRCTN99089299 (first received 04 August 2005).
Castilla 2021 {published data only}
- Castilla LKS, Serrano CAM, Figueroa AMC. Biofeedback therapy in children with chronic functional constipation and sensitivity disorders according to London criteria. Journal of Pediatric Gastroenterology and Nutrition 2021;72(Supplement 1):603. [Google Scholar]
Kajbafzadeh 2020 {published data only}
- IRCT20111229008554N4. Intrarectal electromotive botulinum toxin type A administration in children with refractory constipation. https://www.irct.ir/trial/28186 (first received 5 May 2018).
- Kajbafzadeh AM, Sharifi-Rad L, Nabavizadeh B, Ladi-Seyedian SS, Alijani M, Farahmand F, et al. Intrarectal electromotive botulinum toxin type A administration in children with intractable constipation: a randomized clinical trial. American Journal of Gastroenterology 2020;115(12):2060-7. [DOI: 10.14309/ajg.0000000000000940] [PMID: ] [DOI] [PubMed] [Google Scholar]
Keragiozoglou‐Lampoudi 2012 {published data only}
- Karagiozoglou-Lampoudi T, Daskalou E, Agakidis C, Savvidou A, Apostolou A, Vlahavas G. Personalized diet management can optimise compliance to a high-fiber, high-water diet in children with refractory functional constipation. Journal of the Academy of Nutrition and Dietetics 2012;112(5):725-9. [DOI: 10.1016/j.jand.2012.01.021] [PMID: ] [DOI] [PubMed] [Google Scholar]
Keshtgar 2007 {published data only}
- Keshtgar As, Ward HC, Sanei A, Clayden GS. Botulinum toxin, a new treatment modality for chronic idiopathic constipation in children: long-term follow-up of a double-blind randomized trial. Journal of Pediatric Surgery 2007;42(4):672-80. [DOI: 10.1016/j.jpedsurg.2006.12.045] [PMID: ] [DOI] [PubMed] [Google Scholar]
Mugie 2014 {published data only}
- Mugie SM, Korczowski B, Bodi P, Green A, Kerstens R, Ausma J, et al. Prucalopride is no more effective than placebo for children with functional constipation. Gastroenterology 2014;147(6):1285-95. [DOI: 10.1053/j.gastro.2014.09.005] [PMID: ] [DOI] [PubMed] [Google Scholar]
- NCT01330381. Prucalopride in pediatric subjects with functional constipation [Trial consisting of an 8-week double-blind placebo-controlled part to evaluate efficacy, safety, tolerability and pharmacokinetics of prucalopride in paediatric subjects with functional constipation, aged ≥6 months to <18 years, followed by a 16-week open-label comparator (PEG) controlled part, to document safety and tolerability up to 24 weeks]. clinicaltrials.gov/ct2/show/NCT01330381 (first received 6 April 2011).
Southwell 2012 {published and unpublished data}
- ACTRN12610000418077. Children with slow transit constipation treated with transcutaneous electrical stimulation using interferrential current across the abdomen at T9-L2 compared to sham electrical stimulation - change in defecation, soiling, colonic transit and colonic activity. [TICTOC 1 - Transcutaneous interferrential current to overcome constipation- 1 phyiostherapist clinic based]. https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=335524&isReview=true (first received 25 May 2010).
- Clarke MCC, Chase JW, Gibb S, Hutson JM, Southwell BR. Improvement of quality of life in children with slow transit constipation after treatment with transcutaneous electrical stimulation. Journal of Pediatric Surgery 2009;44(6):1268-72. [DOI: 10.1016/j.jpedsurg.2009.02.031] [PMID: ] [DOI] [PubMed] [Google Scholar]
- Clarke MCC, Chase JW, Gibb S, Robertson VJ, Cato-Smith A, Hutson JM, et al. Decreased colonic transit time after transcutaneous interferential electrical stimulation in children with slow transit constipation. Journal of Pediatric Surgery 2009;44(2):408-12. [DOI: 10.1016/j.jpedsurg.2008.10.100] [PMID: ] [DOI] [PubMed] [Google Scholar]
- Leong LCY, Yik YI, Catto-Smith AG, Robertson VJ, Hutson JM, Southwell BR. Long-term effects of transabdominal electrical stimulation in treating children with slow-transit constipation. Journal of Pediatric Surgery 2011;46(12):2309-12. [DOI: 10.1016/j.jpedsurg.2011.09.022] [DOI] [PubMed] [Google Scholar]
- Southwell B. Transabdominal electrical stimulation to treat slow transit constipation in children. Neuromodulation 2012;15:63. [Google Scholar]
References to studies excluded from this review
Bekkali 2012 {published data only}
- Bekkali N, Liem O, Bongers MEJ, Van Wijk MP, Zegers B, Pelleboer R, et al. One year treatment of childhood constipation comparing PEG 3350 with electrolytes versus PEG 4000: a double-blind randomized controlled trial. Gastroenterology 2012;142(5):S821-22. [DOI: 10.1016/S0016-5085(12)63192-8] [PMID: 28906317] [DOI] [Google Scholar]
Broide 2001 {published data only}
- Broide E, Pintov S, Portnoy S, Barg J, Klinowski E, Scapa E. Effectiveness of acupuncture for treatment of childhood constipation. Digestive Diseases and Sciences 2001;46(6):1270-5. [DOI: 10.1023/a:1010619530548] [PMID: ] [DOI] [PubMed] [Google Scholar]
Burnett 2004 {published data only}
- Burnett CA, Juszczak E, Sullivan PB. Nurse management of intractable functional constipation: A randomised controlled trial. Archives of Disease in Childhood 2004;89(8):717-22. [DOI: 10.1136/adc.2002.025825] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Clarke 2012 {published data only}
- Clarke MCC, Catto-Smith AG, King SK, Dinning PG, Cook IJ, Chase JW, et al. Transabdominal electrical stimulation increases colonic propagating pressure waves in paediatric slow transit constipation. Journal of Pediatric Surgery 2012;47(12):2279-84. [DOI: 10.1016/j.jpedsurg.2012.09.021] [PMID: ] [DOI] [PubMed] [Google Scholar]
Dehghani 2012 {published data only}
- Dehghani SM, Ahmadpour B, Haghighat M, Kashef S, Imanieh MH, Soleimani M. The role of cow's milk allergy in pediatric chronic constipation: A randomized clinical trial. Iran Journal of Pediatrics 2012;22(4):468-74. [PMID: ] [PMC free article] [PubMed] [Google Scholar]
Díaz 2020 {published data only}
- Díaz MB, García CB, Ramírez DKS, Rodriguez AMR. Manual physical therapy in the treatment of functional constipation in children: a pilot randomized controlled trial. Journal of Alternative Complementary Medicine 2020;26(7):620-7. [DOI: 10.1089/acm.2020.0047] [PMID: ] [DOI] [PubMed] [Google Scholar]
Di Lorenzo 2020 {published data only}
- Di Lorenzo C, Nurko S, Hyams JS, Weissman T, Borgstein N, Bartolini W, et al. Linaclotide safety and efficacy in children aged 7 to 17 years with irritable bowel syndrome with constipation. Gastroenterology 2020;158(6):S182-3. [DOI: 10.1016/S0016-5085(20)31142-2] [DOI] [PubMed] [Google Scholar]
- NCT02559817. A safety and efficacy study of a range of linaclotide doses administered orally to children ages 7-17 years, with irritable bowel syndrome with constipation (LIN-MD-63) [A multicenter, randomized, double-blind, placebo-controlled, parallel-group, safety and efficacy study of a range of linaclotide doses administered orally to children, ages 6 to 17 years, a multicenter, randomized, double-blind, placebo-controlled safety and efficacy study of a range of linaclotide doses administered orally to children ages 7-17 years, with irritable bowel syndrome with constipation (IBS-C) (ie, fulfil Rome III criteria for child/adolescent IBS and fulfil modified Rome III Criteria for child/adolescent functional constipation]. clinicaltrials.gov/ct2/show/NCT02559817 (first received 24 September 2015).
Gondo 2020 {published data only}
- Gondo M, Nagata S, Shinbo K, Oota A, Tomomasa T. Polyethylene glycol 3350 plus electrolytes for pediatric chronic constipation: an open-label clinical study in Japan. Pediatrics International 2020;62(5):600-8. [DOI: 10.1111/ped.14102] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gremse 2000 {published data only}
- Gremse D, Hixon J. Comparison of polyethylene glycol 3350, NF powder and lactulose for treatment of chronic constipation in children. Journal of Pediatric Gastroenterology and Nutrition 2000;31:S131. [DOI] [PubMed] [Google Scholar]
- Gremse DA, Hixon J, Crutchfield A. Comparison of polyethylene glycol 3350 and lactulose for treatment of chronic constipation in children. Clinical Pediatrics 2002;41(4):225-9. [DOI: 10.1177/000992280204100405] [PMID: ] [DOI] [PubMed] [Google Scholar]
Heemskerk 2021 {published data only}
- Heemskerk S, Dirksen C, Van Kuijk S, Benninga M, Baeten C, Masclee A, et al. No.2-Trial: A multicenter, open-label, pragmatic randomized controlled trial on sacral neuromodulation versus personalized conservative treatment in slow-transit constipation. Colorectal Disease 2021;23(Supplement 2):4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heemskerk SCM, Rotteveel AH, Benninga MA, Baeten CIM, Masclee AAM, Melenhorst J, et al. Sacral neuromodulation versus personalized conservative treatment in patients with idiopathic slow-transit constipation: study protocol of the No.2-trial, a multicenter open-label randomized controlled trial and cost-effectiveness analysis. International Journal of Colorectal Disease 2018;33(4):493-501. [DOI: 10.1007/s00384-018-2978-x] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ismail 2009 {published data only}
- Ismail KA, Chase J, Gibb S, Clarke M, Catto-Smith AG, Robertson VJ, et al. Daily transabdominal electrical stimulation at home increased defecation in children with slow-transit constipation: a pilot study. Journal of Pediatric Surgery 2009;44(12):2388-92. [DOI: 10.1016/j.jpedsurg.2009.07.063] [PMID: ] [DOI] [PubMed] [Google Scholar]
ISRCTN24521269 {published data only}
- ISRCTN24521269. Randomised controlled trial of injection of botulinum toxin into the internal anal sphincter versus control in treatment of chronic idiopathic constipation in children. www.isrctn.com/ISRCTN24521269 (first received 30 September 2005). [DOI: 10.1186/ISRCTN24521269] [DOI]
Keshtgar 2005 {published data only}
- Keshtgar AS, Ward HC, Clayden GS, Sanei A. Role of anal dilatation in treatment of idiopathic constipation in children: long-term follow-up of a double-blind randomized controlled study. Pediatric Surgery International 2005;21(2):100-5. [DOI: 10.1007/s00383-004-1336-y] [PMID: ] [DOI] [PubMed] [Google Scholar]
Khan 2020 {published data only}
- Khan MA, Chubarova AI, Rassulova MA, Talkovsky EM, Dedurina AV, Novikova EV. Modern possibilities of cryotherapy for chronic functional constipation in children. Vopr Kurortol Fizioter Lech Fiz Kult 2020;97(3):68-75. [DOI: 10.17116/kurort20209703168] [PMID: ] [DOI] [PubMed] [Google Scholar]
Loening‐Baucke 2006 {published data only}
- Loening-Baucke V, Pashankar DS. A randomized, prospective, comparison study of polyethylene glycol 3350 without electrolytes and milk of magnesia for children with constipation and fecal incontinence. Pediatrics 2006;118(2):528-35. [DOI: 10.1542/peds.2006-0220] [PMID: ] [DOI] [PubMed] [Google Scholar]
Modin 2018 {published data only}
- Modin L, Walsted AM, Dalby K, Jakobsen MS. Polyethylene glycol maintenance treatment for childhood functional constipation: a randomized, placebo-controlled trial. Journal of Pediatric Gastroenterology and Nutrition 2018;67(6):732-37. [DOI: 10.1097/MPG.0000000000002070] [PMID: ] [DOI] [PubMed] [Google Scholar]
Nakajima 2019 {published data only}
- Nakajima A, Shinbo K, Ota A, Kinoshita Y. Polyethylene glycol 3350 plus electrolytes for chronic constipation: a randomized, double-blind, placebo-controlled medium-term study with an extensional 52 weeks open-label, long-term study. Journal of Gastroenterology 2019;54(9):792-803. [DOI: 10.1007/s00535-019-01581-x] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
NCT03054805 {published data only}
- NCT03054805. The effect of probiotics on constipation, and intestinal microflora in children with functional constipation. www.clinicaltrials.gov/ct2/show/NCT03054805 (first received 14 August 2019).
Nurko 2000 {published data only}
- Nurko S, Garcia-Aranda JA, Worona LB, Zlochisty O. Cisapride for the treatment of constipation in children: a double-bind study. Journal of Pediatrics 2000;136(1):35-40. [DOI: 10.1016/s0022-3476(00)90046-5] [PMID: ] [DOI] [PubMed] [Google Scholar]
Peng 2006 {published data only}
- Peng HY, Xu AZ. Colonic exclusion and combined therapy for refractory constipation. World Journal of Gastroenterology 2006;12(48):7864-8. [DOI: 10.3748/wjg.v12.i48.7864] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Radwan 2021 {published data only}
- Radwan AB, Gadallah MA, Shahawy MR, Albagdady AA, Talaat AA. Can botulinum toxin help in managing children with functional constipation and obstructed defecation? Journal of Pediatric Surgery 2021;56(4):750-3. [DOI: 10.1016/j.jpedsurg.2020.06.044] [PMID: ] [DOI] [PubMed] [Google Scholar]
Strisciuglio 2021 {published data only}
- Strisciuglio C, Coppola V, Russo M, Tolone C, Marseglia GL, Verrotti A, et al. Promelaxin microenemas are non-inferior to oral polyethylene glycol for the treatment of functional constipation in young children: a randomized clinical trial. Frontiers in Pediatrics 2021;9:753938. [DOI: 10.3389/fped.2021.753938] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Thomson 2008 {published data only}
- Thomson MA, Jenkins HR, Bisset WM, Heuschkel R, Kalra DS, Green MR, et al. Polyethylene glycol 3350 plus electrolytes for chronic constipation in children: a double blind, placebo controlled, crossover study. Archives of Disease in Childhood 2007;92(11):996-1000. [DOI: 10.1136/adc.2006.115493] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Velasco‐Benitez 2023 {published data only}
- Velasco-Benitez C, Villamarin E, Mendez M, Linero A, Hungria G, Saps M. Efficacy of transcutaneous posterior tibial nerve stimulation in functional constipation. European Journal of Pediatrics 2023;182(3):1309-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
Weifeng 2021 {published data only}
- Weifeng H, Xioming Y, Jialei S, Binghua F, Rubao G. Self-administered acupressure for chronic severe functional constipation: a study protocol for a randomized controlled trial. Medicine 2021;100(25):e26349. [DOI: 10.1097/MD.0000000000026349] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies awaiting assessment
ACTRN12620000131954 {published data only}
- ACTRN12620000131954. Tranabdominal electric stimulation in the treatment of chronic constipation in children - TESCCO trial [Double blind randomised, placebo-controlled clinical trial of transcutaneous electric stimulation in the treatment of chronic constipation in children]. https://anzctr.org.au/Trial/Registration/TrialReview.aspx?ACTRN=12620000131954 (first received 17 December 2019).
NCT05035784 {published data only}
- NCT05035784. RCE With FMT in the treatment of childhood constipation [Retrograde colonic enema with fecal microbiota transplantation vs retrograde colonic enema only in the treatment of childhood constipation]. clinicaltrials.gov/ct2/show/NCT05035784 (first received 5 September 2021).
References to ongoing studies
NCT05059756 {published data only}
- NCT05059756. PTNS and PFR in the treatment of childhood constipation [Percutaneous tibial nerve stimulation and pelvic floor rehabilitation in the treatment of childhood constipation]. clinicaltrials.gov/ct2/show/NCT05059756 (first received 28 September 2021).
Additional references
Aali 2021
- Aali G, Shokraneh F. No limitations to language, date, publication type, and publication status in search step of systematic reviews. Journal of Clinical Epidemiology 2021;133:165-7. [DOI: 10.1016/j.jclinepi.2021.02.002] [PMID: ] [DOI] [PubMed] [Google Scholar]
Campbell 2020
- Campbell M, McKenzie JE, Sowden A, Katikireddi SV, Brennan SE, Ellis S, et al. Synthesis without meta-analysis (SWiM) in systematic reviews: reporting guideline. BMJ 2020;368:l6890. [DOI: 10.1136/bmj.l6890] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Chan 2016
- Chan DS, Delicata RJ. Meta-analysis of antegrade continence enema in adults with faecal incontinence and constipation. British Journal of Surgery March 2016;103(4):322-7. [DOI: 10.1002/bjs.10051] [PMID: ] [DOI] [PubMed] [Google Scholar]
Covidence [Computer program]
- Covidence. Version accessed 09 April 2022. Melbourne, Australia: Veritas Health Innovation. Available at covidence.org.
Deeks 2021
- Deeks JJ, Higgins JP, Altman DG, editor(s). Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Egger 1997
- Egger M, Smith GD, Schinder M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315(7109):629-34. [DOI: 10.1136/bmj.315.7109.629] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Emmanuel 2010
- Emmanuel A. Review of the efficacy and safety of transanal irrigation for neurogenic bowel dysfunction. Spinal Cord 2010;48(9):664-73. [DOI: 10.1038/sc.2010.5] [PMID: ] [DOI] [PubMed] [Google Scholar]
Emmett 2015
- Emmett CD, Close HJ, Yiannakou Y, Mason JM. Trans-anal irrigation therapy to treat adult chronic functional constipation: systematic review and meta-analysis. BMC Gastroenterology 2015;15(1):1-8. [DOI: 10.1186/s12876-015-0354-7] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gordon 2016
- Gordon M, MacDonald JK, Parker CE, Akobeng AK, Thomas AG. Osmotic and stimulant laxatives for the management of childhood constipation. Cochrane Database of Systematic Reviews 2016, Issue 8. Art. No: CD009118. [DOI: 10.1002/14651858.CD009118.pub3] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gordon 2016a
- Gordon M, Wallace C, Stone J, Thomas A, Akobeng A. Probiotics for treatment of chronic constipation in children: a Cochrane systematic review. British Paediatric Allergy Immunology and Infection and British Society of Paediatric Gastroenterology, Hepatology and Nutrition 2016;101:A25. [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT. Version accessed 31 May 2022. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Guyatt 2013
- Guyatt GH, Oxman AD, Santesso N, Helfand M, Vist G, Kunz R, et al. GRADE guidelines: 12. Preparing summary of findings tables - binary outcomes. Journal of Clinical Epidemiology 2013;66(2):158-72. [DOI: 10.1016/j.jclinepi.2012.01.012] [PMID: ] [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from training.cochrane.org/handbook/archive/v5.2.
Higgins 2017
- Higgins JP, Altman DG, Sterne JA, editor(s). Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017). Cochrane, 2017. Available from training.cochrane.org/handbook/archive/v5.2.
Higgins 2023
- Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Cochrane Handbook for Systematic Reviews of Interventions version 6.4 (updated August 2023). Cochrane, 2023. Available from www.training.cochrane.org/handbook.
Hyams 2016
- Hyams JS, Di Lorenzo C, Saps M, Shulman RJ, Staiano A, Tilburg M. Functional disorders: children and adolescents. Gastroenterology May 2016;150:1456-68. [DOI: 10.1053/j.gastro.2016.02.015] [PMID: ] [DOI] [PubMed] [Google Scholar]
Irani 2008
- Irani K, Rodriguez L, Doody DP, Goldstein AM. Botulinum toxin for the treatment of chronic constipation in children with internal anal sphincter dysfunction. Pediatric Surgery International July;24(7):779-83. [DOI: 10.1007/s00383-008-2171-3] [PMID: ] [DOI] [PubMed] [Google Scholar]
Koppen 2018
- Koppen IJ, Vriesman MH, Saps M, Rajindrajith S, Shi X, Etten Jamaludin FD, et al. Prevalence of functional defecation disorders in children: a systematic review and meta-analysis. Journal of Pediatrics 2018;198:121-30. [DOI: 10.1016/j.jpeds.2018.02.029] [PMID: ] [DOI] [PubMed] [Google Scholar]
Koppen 2018a
- Koppen IJN, Saps M, Lavigne JV, Nurko S, Taminiau JA, Di Lorenzo C, et al. Recommendations for pharmacological clinical trials in children with functional constipation: the Rome foundation pediatric subcommittee on clinical trials. Neurogastroenterology and Motility 2018;30(4):e12394. [DOI: 10.1111/nmo.13294] [PMID: ] [DOI] [PubMed] [Google Scholar]
Lefebvre 2021
- Lefebvre C, Glanville J, Briscoe S, Littlewood A, Marshall C, Metzendorf M-I, et al. Chapter 4: Searching for and selecting studies. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Lewis 1997
- Lewis SK, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scandinavian Journal of Gastroenterology 1997;32(9):920-4. [DOI: 10.3109/00365529709011203] [PMID: ] [DOI] [PubMed] [Google Scholar]
Li 2021
- Li T, Higgins JP, Deeks JJ, editor(s). Chapter 5: Collecting data. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
McClung 2004
- McClung HJ, Potter C. Rational use of laxatives in children. Advanced Pediatrics 2004;51:231-62. [PMID: ] [PubMed] [Google Scholar]
Ng 2016
- Ng RT, Lee WS, Ang HL, Teo KM, Yik YI, Lai NM. Transcutaneous electrical stimulation (TES) for treatment of constipation in children. Cochrane Database of Systematic Reviews 2016, Issue 7. Art. No: CD010873. [DOI: 10.1002/14651858.CD010873.pub2] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
NICE Guideline 2013
- National Institute for Health and Care Excellence (NICE). Constipation in children and young people. https://www.nice.org.uk/guidance/qs62/documents/constipation-in-children-and-young-people-briefing-paper2 2013. [PubMed]
Philichi 2018
- Philichi L. Management of childhood functional constipation. Journal of Pediatric Health Care 2018;32(1):103-11. [DOI: 10.1016/j.pedhc.2017.08.008] [PMID: ] [DOI] [PubMed] [Google Scholar]
Portalatin 2012
- Portalatin M, Winstead N. Medical management of constipation. Clinics in Colon and Rectal Surgery 2012;25(1):12-9. [DOI: 10.1055/s-0032-1301754] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rachel 2020
- Rachel H, Griffith AF, Teague WJ, Hutson JM, Gibb S, Goldfeld S, et al. Polyethylene glycol dosing for constipation in children younger than 24 months: a systematic review. Journal of Pediatric Gastroenterology and Nutrition 2020;71(2):171-5. [DOI: 10.1097/MPG.0000000000002786] [PMID: ] [DOI] [PubMed] [Google Scholar]
Rajindrajith 2016
- Rajindrajith S, Devanarayana NM, Perera BJC, Benninga MA. Childhood constipation as an emerging public health problem. World Journal of Gastroenterology 2016;22(30):6864-75. [DOI: 10.3748/wjg.v22.i30.6864] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
RevMan 2024 [Computer program]
- Review Manager (RevMan). Version 7.2.0. The Cochrane Collaboration, 2024. Available at revman.cochrane.org.
Shah 2011
- Shah ND, Chitkara D, Branda ME, Van Tilburg MA, Whitehead WE, Katusic SK, et al. Direct medical costs of constipation from childhood to early adulthood: a population-based birth cohort study. Journal of Pediatric Gastroenterology and Nutrition 2011;52(1):47-54. [DOI: 10.1097/MPG.0b013e3181e67058] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Siddiqui 2014
- Siddiqui A, Fishman SJ, Bauer SB, Nurko S. Long-term follow-up of patients after antegrade continence enema procedure. Journal of Pediatric Gastroenterology and Nutrition 2014;52(5):574-80. [DOI: 10.1097/MPG.0b013e3181ff6042] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Siminas 2015
- Siminas S, Losty PD. Current surgical management of pediatric idiopathic constipation: a systematic review of published studies. Annals of Surgery 2015;262(6):925-33. [DOI: 10.1097/SLA.0000000000001191] [PMID: ] [DOI] [PubMed] [Google Scholar]
Sluka 2003
- Sluka KA, Walsh D. Transcutaneous electrical nerve stimulation: basic science mechanisms and clinical effectiveness. Journal of Pain 2003;4(3):109-21. [DOI: 10.1054/jpai.2003.434] [PMID: ] [DOI] [PubMed] [Google Scholar]
Southwell 2020
- Southwell BR. Treatment of childhood constipation: a synthesis of systematic reviews and meta-analyses. Expert Review of Gastroenterology and Hepatology 2020;14(3):163-74. [DOI: 10.1080/17474124.2020.1733974] [PMID: ] [DOI] [PubMed] [Google Scholar]
Tabbers 2014
- Tabbers MM, DiLorenzo C, Berger MY, Faure C, Langendam MW, Nurko S, et al. Evaluation and treatment of functional constipation in infants and children: evidence-based recommendations from ESPGHAN and NASPGHAN. Journal of Pediatric Gastroenterology and Nutrition 2014;58(2):258-74. [DOI: 10.1097/MPG.0000000000000266] [PMID: ] [DOI] [PubMed] [Google Scholar]
Vriesman 2019
- Vriesman MH, Rajindrajith S, Koppen IJN, Etten Jamaludin FS, Dijk M, et al. Quality of life in children with functional constipation: a systematic review and meta-analysis. Journal of Pediatrics 2019;214:141-50. [DOI: 10.1016/j.jpeds.2019.06.059] [PMID: ] [DOI] [PubMed] [Google Scholar]
Vriesman 2020
- Vriesman MH, Wang L, Park C, Diefenbach KA, Levitt MA, Wood RJ, et al. Comparison of antegrade continence enema treatment and sacral nerve stimulation for children with severe functional constipation and fecal incontinence. Neurogastroenterology and Motility 2020;32(8):e13809. [DOI: 10.1111/nmo.13809] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Youssef 2001
- Youssef NN, Di Lorenzo C. Childhood constipation: evaluation and treatment. Journal of Clinical Gastroenterology 2001;33(3):199-205. [DOI: 10.1097/00004836-200109000-00006] [PMID: ] [DOI] [PubMed] [Google Scholar]


