Abstract
Background
Neurocardiogenic syncope is a common condition with significant associated psychological and physical morbidity. The effectiveness of therapeutic options for neurocardiogenic syncope beyond placebo remains uncertain.
Methods
The primary endpoint was the risk ratio (RR) of spontaneously recurring syncope following any therapeutic intervention. We also examined the effect of blinding on treatment efficacy. We identified all randomised trials which evaluated the effect of any pharmacological, device-based or supportive intervention on patients with a history of syncope. A systematic search was conducted on Medline, Embase, PubMed databases and Cochrane Central Register for Controlled Trials from 1950 to 25 April 2023. Event rates, their RRs and 95% CIs were calculated, and a random-effects meta-analysis was conducted for each intervention. Data analysis was performed in R using RStudio.
Results
We identified 47 eligible trials randomising 3518 patients. Blinded trials assessing syncope recurrence were neutral for beta blockers, fludrocortisone and conventional dual-chamber pacing but were favourable for selective serotonin reuptake inhibitors (SSRIs) (RR 0.40, 95% CI 0.26 to 0.63, p<0.001), midodrine (RR 0.70, 95% CI 0.53 to 0.94, p=0.016) and closed-loop stimulation (CLS) pacing (RR 0.15, 95% CI 0.07 to 0.35, p<0.001). Unblinded trials reported significant benefits for all therapy categories other than beta blockers and consistently showed larger benefits than blinded trials.
Conclusions
Under blinded conditions, SSRIs, midodrine and CLS pacing significantly reduced syncope recurrence. Future trials for syncope should be blinded to avoid overestimating treatment effects.
PROSPERO registration number
CRD42022330148.
Keywords: SYNCOPE; Meta-Analysis; Quality of Health Care; Pacemaker, Artificial
WHAT IS ALREADY KNOWN ON THIS TOPIC
Syncope is a common problem with significant physical and psychological comorbidity.
WHAT THIS STUDY ADDS
This study shows that unblinded syncope trials consistently overestimate treatment effect.
The only therapies that effectively reduced syncope recurrence beyond placebo were selective serotonin reuptake inhibitors, midodrine and closed-loop stimulation pacing.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
This work highlights the gap in evidence for blinded randomised controlled trials in neurocardiogenic syncope.
Introduction
Neurocardiogenic syncope is common with a lifetime incidence of >35% with one-third of these individuals suffering significant psychological and physical injury.1
Syncope typically results from a vasodepressor response (hypotension), a cardioinhibitory response (bradycardia) or a combination of both mechanisms. Despite the great variety of proposed therapies, including medications, cardiac pacemaker devices and physical training programmes, it is not clear which therapies are effective beyond placebo.2
Syncope could be considered an objective endpoint because loss of consciousness can be thought of as binary, and it might be argued that blinding therefore is unnecessary. Furthermore, blinding and placebo control introduce additional cost and complexity in trial. However, it has been repeatedly found within cardiology that even with endpoints thought to be objective, for example, blood pressure, blinding is essential for reliable results.3 4 The merits and need for blinded, placebo-controlled trials have not been tested when evaluating syncope management approaches.
We therefore conducted a meta-analysis of randomised controlled trials of therapies to prevent recurrent syncope. We specifically test whether blinding makes an important difference.
Methods
Search strategy
We performed a systematic search of Medline, Embase, PubMed databases and Cochrane Central Register for Controlled Trials from 1950 to 25 April 2023 for all randomised studies conducted in humans.
The search strategy was devised by DK, MJS-S and NK. The search, carried out by MS and NK, included the search strings ‘syncope’ AND ‘vasovagal’ OR ‘neurocardiogenic’ OR ‘reflex mediated’ OR ‘reflex-mediated’ OR ‘carotid sinus’ combined with the Cochrane Central Register’s highly specific search string for randomised controlled trials. MS and NK also manually searched the bibliographies of selected studies as well as previous systematic reviews and meta-analyses to identify any other remaining studies eligible for inclusion. Disputes were resolved through discussion with two other authors, DK and MJS-S.
Inclusion criteria
We included all randomised studies which prospectively evaluated the effect of any pharmacological,5–23 device-based24–44 or supportive interventions45–53 for neurocardiogenic, reflex, vasovagal, carotid sinus or orthostatic syncope (online supplemental eTable 1a–i). Abstracts were reviewed for suitability and full-text articles evaluated accordingly. For full-text studies to be eligible for inclusion, studies had to have a minimum follow-up time of 4 weeks and to report outcomes of spontaneously recurring syncope. To be eligible for inclusion in the meta-analysis (and not only the systematic review), we chose interventions that had at least more than one comparable study on the topic.
openhrt-2024-002669supp001.pdf (433.9KB, pdf)
Exclusion criteria
In order to optimise the real-world relevance of our systematic review and meta-analysis, studies which reported outcomes measured exclusively in an acute tilt-table test setting were excluded from analysis. Studies which evaluated the therapeutic effects of interventions in healthy volunteers or in blood donors were also excluded.
Interventions and endpoints
The primary endpoint was the risk ratio (RR) of spontaneously recurring syncope following any pharmacological, device-based or supportive therapeutic intervention. Supportive interventions included the use of physical manoeuvres, yoga, and tilt or orthostatic training exercises (supervised or self-conducted). We did not abstract, report or analyse the number of recurrent episodes of syncope due to a limited number of studies reporting this endpoint.
Data abstraction and analysis
MS and NK independently extracted information from all selected articles, verified by a third author (MJS-S). Included trials were assessed using the Cochrane risk of bias tool by two independent reviewers (online supplemental eTable 2a–f). NK, MJS-S, DPF and DK formulated the analysis plan.
Study population information was retrieved including participant age, sex and number of episodes of syncope prior to enrolment in the trial (online supplemental eTable 3).
We abstracted event rates, their RRs and 95% CIs. Where event rates were reported both at intervals as well as at the end of the study, only end-of-study event rates were recorded.
From the abstracted event rates, or RR and 95% CIs, we performed a random-effects meta-analysis for each intervention. We stratified by the presence of blinding and then tested if this had a significant impact and then went on to quantify the placebo effect seen.
We used the I2 statistic to assess for heterogeneity. Data analysis was performed in R using RStudio, with the ‘metafor’ package, and results were reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. This meta-analysis was registered on PROSPERO on 4 May 2022 (CRD42022330148).
Results
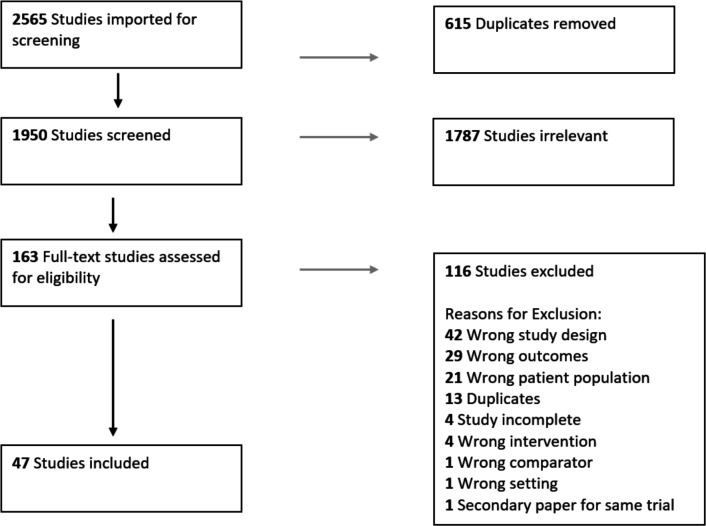
The search strategy identified 2565 studies for screening. After the removal of duplicates and screening of titles and abstracts, 163 full-text studies were assessed for eligibility. Of these, 47 met the inclusion criteria (figure 1). Of these 47 studies eligible for inclusion in our systematic review, 39 were included in the meta-analysis. This included 17 trials randomising 1793 patients testing pharmacological therapies,5–19 21–23 14 trials randomising 1015 patients testing pacing therapies24–30 32–39 and 8 trials randomising 690 patients which tested other interventions which can be broadly described as physical interventions.45–53
Figure 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram summarising the search outcomes. A total of 2565 papers were identified through the systematic search. Duplicates were removed leaving a total of 1950 papers to be screened of which 163 were relevant to the purpose of this paper and 47 met the inclusion criteria.
Across all studies, 55.8% of participants were female, the mean age was 46.8 and the mean follow-up time was 15.5 months (online supplemental eTable 3).
Ignoring blinding as the impact of this is formally assessed in this paper; overall, the risk of bias in this study was classed as medium-high risk of bias (online supplemental eTable 2a–f).
Blinded placebo-controlled trials
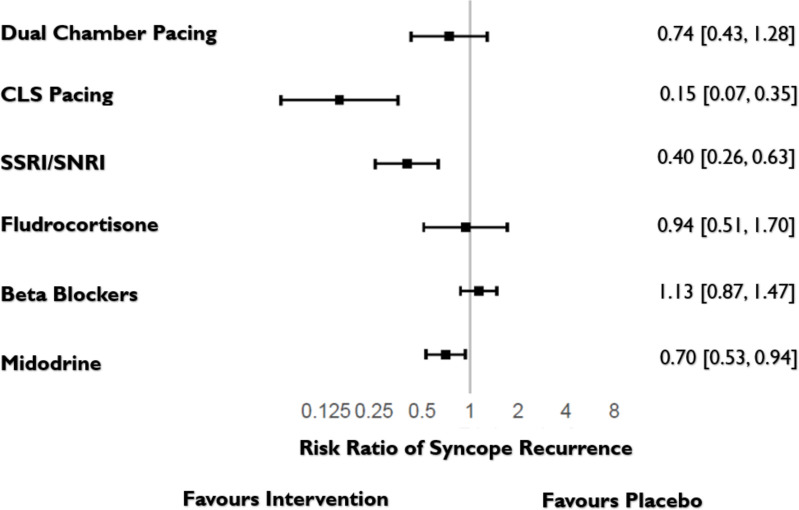
There were 17 blinded placebo-controlled trials totalling 1399 patients. 11 trials (1025 patients) were of medications8–10 13–18 and six trials (374 patients) were of implantable cardiac devices25 26 32 33 36 37 (figure 2 and online supplemental eFigure 1).
Figure 2.
Summary effect of therapies on syncope recurrence in blinded trials with placebo control. Blinded trials were neutral for conventional pacing, beta blockers and fludrocortisone but favourable for closed-loop stimulation (CLS) pacing, selective serotonin reuptake inhibitors/serotonin norepinephrine reuptake inhibitors (SSRIs/SNRIs) and midodrine.
Placebo-controlled drug trials
There were three placebo-controlled trials with beta blockers,5 9 13 totalling 304 patients, which showed no significant reduction in syncope recurrence (RR 1.11, 95% CI 0.85 to 1.45, p=0.358, I2=0%). Two placebo-controlled trials including 242 patients assessing fludrocortisone therapy6 10 also failed to show significant therapeutic effect (RR 0.83, 95% CI 0.63 to 1.11, p=0.827, I2=44%).
Four trials of selective serotonin reuptake inhibitors and serotonin norepinephrine reuptake inhibitors (SSRI/SNRI),13–15 17 totalling 417 patients, showed significant reduction in the risk of syncope recurrence (RR 0.40, 95% CI 0.25 to 0.63, p<0.001, I2=0%). Similarly, midodrine had favourable results in its two placebo-controlled trials,8 18 totalling 179 patients (RR 0.70, 95% CI 0.53 to 0.94, p=0.016, I2=0%).
Placebo-controlled pacemaker trials
Placebo-controlled trials of pacemaker therapies addressed two main types of pacing therapy. The first was conventional dual-chamber (DDD) pacing, of which there were three trials randomising 206 patients.25 26 32 Trials are detailed in figure 4. The meta-analysis found no significant change in the risk of syncope recurrence with conventional pacing in placebo-controlled trials (RR 0.70, 95% CI 0.43 to 1.28, p=0.459, I2=51%).
The second tested pacemaker therapy was pacing using a closed-loop stimulation (CLS) algorithm. The placebo arm for this was either conventional DDD pacing or a pacemaker implant but with pacing therapies switched off.
Four placebo-controlled trials of 345 patients evaluated CLS pacing. Three of these trials compared CLS with conventional DDD pacing,33 36 37 totalling 218 patients. They showed a significant reduction in syncope recurrence (RR 0.15, 95% CI 0.07 to 0.35, p<0.001, I2=0%). One trial of 127 patients compared CLS pacing to pacemaker implantation but with pacing therapies switched off.31 This also showed a significant reduction in syncope recurrence (RR 0.30, 95% CI 0.16 to 0.55, p<0.0001, I2=0%).
Non-placebo-controlled trials
There were six therapeutic options with more than one trial testing an intervention on syncope recurrence rates, totalling 22 trials of 1630 patients. The categories of therapy included: (1) DDD (dual chamber pacing) / VVI (ventricular pacing) (611 patients)24 27–31 35 38; (2) beta blocker medication (84 patients)11 12; (3) midodrine medication (80 patients)7 19; (4) oral rehydration salt medication (271 patients)21 23; (5) tilt training (251 patients)46–49; and (6) physical interventions (439 patients)45 50 51 53 (online supplemental eFigure 2). All assessed treatment categories, with the exception of beta blocker medication, showed statistically significant reduction in syncope recurrence when tested in non-placebo-controlled trials (figure 3).
Figure 3.
Summary effect of therapies on syncope recurrence in unblinded trials without placebo control. Unblinded trials without placebo control were significant for every category of therapy with the exception of beta blockers.
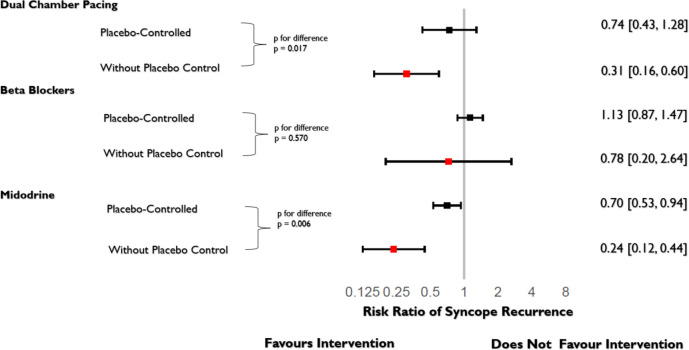
Differences between placebo-controlled trials and non-placebo-controlled trials
Three categories had undergone trialling with both placebo-controlled and non-placebo-controlled designs (figure 4). In two of these three categories, the non-placebo-controlled trials showed a much larger effect size with a significantly different result from the placebo-controlled trials: DDD pacing (p=0.017) and midodrine (p=0.006). Of the three categories, only midodrine showed both a significant reduction in syncope recurrence in both placebo-controlled trials as well as non-placebo-controlled trials.
Figure 4.
Comparison of trials with and without placebo control across therapies. Midodrine, beta blockers and dual-chamber pacing have been assessed in studies using placebo control and in others without placebo control. All three interventions have been reportedly more effective in studies without placebo control. Dual-chamber pacing showed significant reduction in syncope recurrence in studies without placebo control but failed to demonstrate this under placebo control. Midodrine has been found to reduce syncope recurrence even in placebo-controlled studies; however, the magnitude of risk ratio reduction was less than in studies without placebo.
Quantification of the placebo effect on syncope recurrence
There were two therapies which showed numerical reductions in syncope in both placebo-controlled and non-placebo-controlled trials: cardiac pacing and midodrine. It was, therefore, possible to visualise the impact of the placebo/nocebo effect of the patient knowing whether they were receiving active treatment.
For pacing, syncope freedom rates in the control arms averaged 62%. With blinded pacing this improved by 14 percentage points, and if the pacing was unblinded this was a further 15 percentage point improvement (figure 5).
Figure 5.
Placebo effect on syncope recurrence. The overall ‘placebo effect’ in the treatment of cardiogenic syncope is estimated in this figure. For instance, about 15% of pacing effect in reducing syncope recurrence can be attributed to a placebo effect, and this is even higher in some pharmacological interventions, as shown with midodrine where 23.1% of treatment effect can be attributed to a placebo effect.
For midodrine, there was an even more interesting pattern. In the blinded trials the syncope freedom rate rose from 38% to 57% with midodrine and increment of 19 percentage points. However, in the unblinded trials the situation was more extreme. The control arm patients, who knew they were not receiving the drug, had even worse syncope freedom (16%), the active arm patients who knew they were receiving the drug had even better syncope freedom (80%), so that the difference between arms was vastly magnified at 54 percentage points.
Discussion
This meta-analysis found that under rigorous placebo-controlled trial conditions, CLS pacing (but not DDD pacing), midodrine and SSRIs/SNRIs significantly reduced syncope recurrence. CLS pacing reduced syncope risk by fourfold. Furthermore, non-placebo-controlled trials consistently showed a large bias to more positive results than placebo-controlled trials of the same therapy. Beta blockers and fludrocortisone do not show a beneficial effect when tested in placebo-controlled trials.
Insights into syncope from pacing trials
Both conventional DDD pacing and CLS pacing deliver the same intervention, namely an increase in heart rate.54 The difference between the two therapies is that a conventional DDD pacemaker (even when using specially designed rate-drop-response algorithms) makes the decision to pace solely by observing heart rate and therefore targets cardioinhibitory syncope. The CLS algorithm, in contrast, specifically monitors for changes in right ventricular volume as an additional trigger for pacing.33 Although in neurocardiogenic syncope, there is often bradycardia which can be intense, there are many patients in whom there is either no substantial bradycardia or the bradycardia occurs late in the pathogenesis, after the patient is already unconscious or almost unconscious.55 In them, the early pathogenesis of syncope may be due to vasodilatation and vascular volume redistribution, which can cause a profound fall in blood pressure without a bradycardia.2 56 Even if a conventional DDD pacemaker would eventually pace during syncope, the CLS algorithm has the theoretical advantage of commencing the pacing without waiting for the heart rate to collapse.37 57–59 This meta-analysis indicates that this theoretical advantage successfully manifests as reduced syncope recurrence rates.
Guideline recommendations for pacing
Currently, clinical guidelines do recommend DDD pacing for recurrent syncope, on the strength of there having been three placebo-controlled trials, with the largest and most recent being positive and prompting a guideline recommendation.25 Although CLS pacing appears to have a much larger effect size, its large trials have been more recent, and this may explain why it has therefore not yet received a guideline recommendation.
Pharmacological therapies
Alpha agonists showed good efficacy. The alpha agonist, midodrine, is recommended in guidelines and widely used clinically.2 A second alpha agonist, etilefrine, has also shown positive results in a single conducted placebo-controlled trial.16 This suggests that alpha agonists may help sustain vascular tone and thus prevent syncope recurrence.60 Further research on the underlying mechanism(s) of syncope may aid in the development of effective and targeted therapies.
Remarkably, SSRIs and SNRIs were the class of drugs showing the largest statistical benefit, reducing syncope recurrence by more than half.13 15 Guidelines issued after those trials, however, do not yet recommend their use, perhaps because the mechanism is not as intuitive.2 Serotonin whose reuptake is inhibited by these agents plays many roles in the nervous system, which may explain their efficacy in neurocardiogenic syncope.
Beta blockers were not effective, and guidelines, therefore up-to-date guidelines, have now stopped recommending them.2
Fludrocortisone did not show effectiveness in its trials.61 There has been some post hoc commentary that in the 'Clinical Trial for the Prevention of Vasovagal Syncope' (POST II) trial, there was significant efficacy after an initial period of dose escalation. However, we have used the prespecified primary endpoint for our analysis. For drugs where there is a significant period of dose escalation, it may be advisable for future triallists to prespecify the primary endpoint to exclude that early period, perhaps using the time course observed in POST II as a guide.
Necessity for placebo control
The placebo effect of device implantation on syncope is more powerful than previously assumed. Non-placebo-controlled trials of conventional DDD pacing appeared to show remarkable efficacy.26 29 30 35 39 43 However, with placebo control, this efficacy is almost halved, this likely reflects the ‘placebo effect’.26 32 62
Many factors may underlie the ‘placebo effect’ ranging from patient expectations, and treatment administration and clinician biases. There is a possibility that between the placebo-controlled and non-placebo-controlled studies, there was a difference in treatment regimen. For example, for the midodrine trials, some were 5 mg two times per day,8 and two others with doses up to 10–15 mg three times a day.7 18
Patient expectations also play an important part, and most trials included in this meta-analysis relied on symptom diaries or patient-reported episodes of syncope for their primary endpoint. Patients may report syncope recurrence differently if they know they are allocated to active treatment; for example, those who believe they are receiving an effective treatment may perceive improvements in their symptoms, including a reduction in the frequency or severity of syncope episodes. This can lead to biased self-reporting of syncope recurrence, as patients may attribute any improvement to the intervention, regardless of its actual efficacy. Similarly, patients who believe they are receiving an effective treatment may have an improved sense of well-being or confidence, which may lead them to overlook or downplay episodes of syncope recurrence. They may interpret transient symptoms differently or attribute them to factors unrelated to syncope, leading to under-reporting of recurrence events.
Furthermore, the methods with which syncope recurrence was detected may impact the results, and clinician biases may play a role here, if those undertaking the follow-up were aware of which patients were allocated to active intervention. This may have been mitigated in other studies where a blinded clinician undertook the follow-ups or where a blinded adjudication committee was used.26 32
For some therapies, it is difficult to construct a placebo control. For example, if the intervention is a tilt-training programme, it requires an alternative programme that is equally impressive to the patient and gives equal reassurance but does not have the key physiological effect being tested. Tan et al were testing a protocol of carefully selected orthostatic and postural training exercises, and so they used for the placebo arm a session of simple activities such as merely standing, which was not expected to be beneficial.48
Clinicians vary in their acceptance of a necessity of placebo control in trial design. Some have argued that it is unethical.63 It should be remembered though that patients coming to us for clinical advice are expecting to receive recommendations for therapies that are more effective than placebo. It could be argued to be deceptive or even unethical to offer them therapies whose only benefit is placebo. In contrast, it is certainly not deceptive or unethical to invite patients to participate in a placebo-controlled trial which can calculate whether the therapy is beneficial beyond placebo, for the benefit of future patients.
At first sight, syncope might be considered immune to the placebo effect. However, our meta-analysis suggests otherwise. There are two possible mechanisms through which event counts might be lowered by the knowledge of the presence of intervention. First, the autonomic nervous system can readily be affected by psychological factors leading to an actual difference in haemodynamic events. Second, if the patient experiences dizziness or a fall, whether that becomes categorised as a presyncope/syncope event or not will be affected by the patient and physician’s knowledge of whether the patient was receiving active treatment or not. Our meta-analysis cannot identify the mechanism but can only document the implied size of the effect.
Limitations
Syncope is a heterogenous condition affecting different patient cohorts in distinct ways with multiple clinical manifestations and definitions of ‘recurrent syncope’ (presyncopal vs true syncopal episodes, number of episodes, etc). The trials were heterogenous in nature and this is a limitation of interpretation of our results and real-world applicability of our analysis. For example, in the pacing trials, the number of episodes of syncope required for trial inclusion ranged from a minimum of 1 up to 6 episodes, and some trials required participants to have a positive head-up tilt-test result.7 9 12 30 Similarly, one unblinded trial of midodrine focused on a paediatric population while other trials of midodrine tested the intervention in adults only. The trials did not show any clear and consistent differences between those with and without placebo control; rather, the differences in inclusion criteria and cohorts were noted across both categories. However, as the trials are heterogenous, we should therefore be cautious in making comparisons between the reported efficacies of different therapies, since the cause of the difference could lie in the differences in trial design and populations.64 In principle, this means we should also be cautious about interpreting the differences between the placebo and non-placebo-controlled trials. Nevertheless, the differences observed are very large and it is implausible that multiple treatment categories that have had multiple trials of each type could have, by coincidence, other explanations for the large bias towards positivity in the non-placebo-controlled trials.
This study does not attempt to distinguish between ‘placebo control’ and ‘sham control’. Our reason for this choice is that there is no inherent difference. Introduction of the word ‘sham’ for ‘placebo’ does little more than attempt to discredit the placebo-controlling process by bringing connotations of deceptions or dishonesty.
This meta-analysis used recurrence of syncope rather than counts of syncope as its primary endpoint. This is because this endpoint was reported in all studies, and therefore this choice maximises the inclusivity of the analysis. The reason to not favour episode counts as the outcome is that they have a very skewed distribution, with the majority of syncope episodes being concentrated in a small minority of patients. Nevertheless, numbers of syncope episodes do matter: we rely on the principle that therapies that reduce event counts to zero in some patients are likely to be reducing event counts in the other patients, too.
Possible future directions for treatment of syncope
Cardioneuroablation (CNA) is a novel emerging therapy for the treatment of cardioinhibitory vasovagal syncope. This therapeutic concept aims for vagal denervation through the targeting of ganglionated plexi located on the epicardial surface of the heart’s atrium. Observational data of 465 cases of radiofrequency CNA have been documented to date with meta-analysis suggesting subsequent high rates of freedom from syncope.65 The first controlled trial of this therapeutic procedure has since been published, randomising 48 patients to either ablation or optimal non-pharmacological therapy, again suggesting significant improvement in freedom from syncope.66 While larger randomised trials are needed to support these findings, it is equally important to consider the potential placebo effect of the procedure, and this should be considered in future trial designs.
Other future therapeutic approaches, particularly where vasodepressor components predominate, may focus on reducing peripheral blood pooling, for example, within the splanchnic circulation, and aim to ensure appropriate blood distribution should this mechanism cause a reduction in systemic blood pressure.
Conclusions
Cardiac pacing can reduce risk of syncope fourfold, but only if the trigger for pacing includes right ventricular volume sensing, and not when it is merely heart rate. The most effective drug class in preventing recurrent syncope is SSRIs/SNRIs, with alpha agonists (primarily midodrine) coming second. Three classes of therapy (conventional DDD pacing, beta blockers, midodrine) have undergone both non-placebo-controlled and placebo-controlled trials. The non-placebo-controlled trials reported larger effects. All future trials of syncope treatment should be placebo controlled.
Footnotes
Contributors: All authors of this paper have contributed significantly to the conception, design, execution or interpretation of the reported work. Each author has reviewed the final version of the manuscript and approves it for submission. DK has overseen the production of this manuscript and is the guarantor for its contents.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. The datasets analysed during the current study are available from the corresponding author upon reasonable request. Additionally, all included studies are cited in the references section, and readers are encouraged to refer to the original sources for further information on the data used in this meta-analysis.
Ethics statements
Patient consent for publication
Not applicable.
References
- 1. Saklani P, Krahn A, Klein G. Syncope. Circulation 2013;127:1330–9. 10.1161/CIRCULATIONAHA.112.138396 [DOI] [PubMed] [Google Scholar]
- 2. Brignole M, Moya A, de Lange FJ, et al. 2018 ESC guidelines for the diagnosis and management of syncope. Eur Heart J 2018;39:1883–948. 10.1093/eurheartj/ehy037 [DOI] [PubMed] [Google Scholar]
- 3. Miller FG, Wendler D, Swartzman LC. Deception in research on the placebo effect. PLoS Med 2005;2:e262. 10.1371/journal.pmed.0020262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ahmad Y, Francis DP, Bhatt DL, et al. Renal denervation for hypertension: a systematic review and meta-analysis of randomized, blinded, placebo-controlled trials. JACC Cardiovasc Interv 2021;14:2614–24. 10.1016/j.jcin.2021.09.020 [DOI] [PubMed] [Google Scholar]
- 5. Sheldon R, Connolly S, Rose S, et al. Prevention of syncope trial (POST): a randomized, placebo-controlled study of metoprolol in the prevention of vasovagal syncope. Circulation 2006;113:1164–70. 10.1161/CIRCULATIONAHA.105.535161 [DOI] [PubMed] [Google Scholar]
- 6. Sheldon R, Raj SR, Rose MS, et al. Fludrocortisone for the prevention of vasovagal syncope: a randomized. Placebo-Controlled Trial 2016;1:1–9. [DOI] [PubMed] [Google Scholar]
- 7. Perez-Lugones A, Schweikert R, Pavia S, et al. Usefulness of Midodrine in patients with severely symptomatic neurocardiogenic syncope: a randomized control study. J Cardiovasc Electrophysiol 2001;12:935–8. 10.1046/j.1540-8167.2001.00935.x [DOI] [PubMed] [Google Scholar]
- 8. Romme JJCM, van Dijk N, Go-Schön IK, et al. Effectiveness of Midodrine treatment in patients with recurrent vasovagal syncope not responding to non-pharmacological treatment (STAND-trial). Europace 2011;13:1639–47. 10.1093/europace/eur200 [DOI] [PubMed] [Google Scholar]
- 9. Madrid AH, Ortega J, Rebollo JG, et al. Lack of efficacy of atenolol for the prevention of neurally mediated syncope in a highly symptomatic population: a prospective, double-blind, randomized and placebo-controlled study. J Am Coll Cardiol 2001;37:554–9. 10.1016/s0735-1097(00)01155-4 [DOI] [PubMed] [Google Scholar]
- 10. Salim MA, Di Sessa TG. Effectiveness of fludrocortisone and salt in preventing syncope recurrence in children: a double-blind, placebo-controlled, randomized trial. J Am Coll Cardiol 2005;45:484–8. 10.1016/j.jacc.2004.11.033 [DOI] [PubMed] [Google Scholar]
- 11. Zhang Q, Jin H, Wang L, et al. Randomized comparison of metoprolol versus conventional treatment in preventing recurrence of vasovagal syncope in children and adolescents. Med Sci Monit 2008;14:CR199–203. [PubMed] [Google Scholar]
- 12. Ventura R, Maas R, Zeidler D, et al. A randomized and controlled pilot trial of beta-blockers for the treatment of recurrent syncope in patients with a positive or negative response to head-up tilt test. Pacing Clin Electrophysiol 2002;25:816–21. 10.1046/j.1460-9592.2002.t01-1-00816.x [DOI] [PubMed] [Google Scholar]
- 13. Theodorakis GN, Leftheriotis D, Livanis EG, et al. Fluoxetine vs. propranolol in the treatment of vasovagal syncope: a prospective, randomized, placebo-controlled study. Europace 2006;8:193–8. 10.1093/europace/euj041 [DOI] [PubMed] [Google Scholar]
- 14. Di Girolamo E, Di Iorio C, Sabatini P, et al. Effects of paroxetine hydrochloride, a selective serotonin reuptake inhibitor, on refractory vasovagal syncope: a randomized, double-blind, placebo-controlled study. J Am Coll Cardiol 1999;33:1227–30. 10.1016/s0735-1097(98)00694-9 [DOI] [PubMed] [Google Scholar]
- 15. Flevari P, Leftheriotis D, Repasos E, et al. Fluoxetine vs. placebo for the treatment of recurrent vasovagal syncope with anxiety sensitivity. Europace 2017;19:127–31. 10.1093/europace/euw153 [DOI] [PubMed] [Google Scholar]
- 16. Raviele A, Brignole M, Sutton R, et al. Effect of Etilefrine in preventing syncopal recurrence in patients with vasovagal syncope: a double-blind, randomized, placebo-controlled trial. The Vasovagal Syncope International Study 1999;1:1452–7. 10.1161/01.CIR.99.11.1452 [DOI] [PubMed] [Google Scholar]
- 17. Tajdini M, Aminorroaya A, Rahimi B, et al. Comparison of Trinitroglycerin and adenosine as provocative agents for head-up tilt test in patients with unexplained syncope: a semi-crossover randomized clinical trial with prospective follow-up. J Interv Card Electrophysiol 2021;60:31–9. 10.1007/s10840-019-00652-6 [DOI] [PubMed] [Google Scholar]
- 18. Sheldon R, Faris P, Tang A, et al. Midodrine for the prevention of vasovagal syncope: a randomized clinical trial. Ann Intern Med 2021;174:1349–56. 10.7326/M20-5415 [DOI] [PubMed] [Google Scholar]
- 19. Qingyou Z, Junbao D, Chaoshu T. The efficacy of Midodrine hydrochloride in the treatment of children with vasovagal syncope. J Pediatr 2006;149:777–80. 10.1016/j.jpeds.2006.07.031 [DOI] [PubMed] [Google Scholar]
- 20. Morillo CA, Leitch JW, Yee R, et al. A placebo-controlled trial of intravenous and oral disopyramide for prevention of neurally mediated syncope induced by head-up tilt. J Am Coll Cardiol 1993;22:1843–8. 10.1016/0735-1097(93)90767-u [DOI] [PubMed] [Google Scholar]
- 21. Li W, Wang S, Liu X, et al. Assessment of efficacy of oral rehydration salts in children with neurally mediated syncope of different hemodynamic patterns. J Child Neurol 2019;34:5–10. 10.1177/0883073818803035 [DOI] [PubMed] [Google Scholar]
- 22. Sheldon RS, Amuah JE, Connolly SJ, et al. Effect of metoprolol on quality of life in the prevention of syncope trial. J Cardiovasc Electrophysiol 2009;20:1083–8. 10.1111/j.1540-8167.2009.01518.x [DOI] [PubMed] [Google Scholar]
- 23. Chu W, Wang C, Wu L, et al. Oral rehydration salts: an effective choice for the treatment of children with vasovagal syncope. Pediatr Cardiol 2015;36:867–72. 10.1007/s00246-015-1097-5 [DOI] [PubMed] [Google Scholar]
- 24. Ammirati F, Colivicchi F, Santini M, et al. Permanent cardiac pacing versus medical treatment for the prevention of recurrent vasovagal syncope: a multicenter, randomized, controlled trial. Circulation 2001;104:52–7. 10.1161/hc2601.091708 [DOI] [PubMed] [Google Scholar]
- 25. Brignole M, Menozzi C, Moya A, et al. Pacemaker therapy in patients with Neurally mediated syncope and documented asystole. Circulation 2012;125:2566–71. 10.1161/CIRCULATIONAHA.111.082313 [DOI] [PubMed] [Google Scholar]
- 26. Connolly SJ, Sheldon R, Thorpe KE, et al. Pacemaker therapy for prevention of syncope in patients with recurrent severe vasovagal syncope: second vasovagal pacemaker study (VPS II): a randomized trial. JAMA 2003;289:2224–9. 10.1001/jama.289.17.2224 [DOI] [PubMed] [Google Scholar]
- 27. Sutton R, Brignole M, Menozzi C, et al. Dual-chamber pacing in the treatment of neurally mediated tilt-positive cardioinhibitory syncope: pacemaker versus no therapy: a multicenter randomized study. The Vasovagal Syncope International Study (VASIS) Investigators 2000;1:294–9. [DOI] [PubMed] [Google Scholar]
- 28. Brignole M, Menozzi C, Gianfranchi L, et al. A controlled trial of acute and long-term medical therapy in tilt-induced neurally mediated syncope. Am J Cardiol 1992;70:339–42. 10.1016/0002-9149(92)90615-6 [DOI] [PubMed] [Google Scholar]
- 29. Flammang D, Antiel M, Church T, et al. Is a pacemaker indicated for vasovagal patients with severe cardioinhibitory reflex as identified by the ATP test? A preliminary randomized trial. Europace 1999;1:140–5. 10.1053/eupc.1998.0021 [DOI] [PubMed] [Google Scholar]
- 30. Ryan DJ, Nick S, Colette SM, et al. Carotid sinus syndrome, should we pace? A multicentre, randomised control trial. Heart 2010;96:347–51. 10.1136/hrt.2009.176206 [DOI] [PubMed] [Google Scholar]
- 31. Brignole M, Russo V, Arabia F, et al. Cardiac pacing in severe recurrent reflex syncope and tilt-induced asystole. Eur Heart J 2021;42:508–16. 10.1093/eurheartj/ehaa936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Raviele A, Giada F, Menozzi C, et al. A randomized, double-blind, placebo-controlled study of permanent cardiac pacing for the treatment of recurrent tilt-induced vasovagal syncope. Eur Heart J 2004;25:1741–8. 10.1016/j.ehj.2004.06.031 [DOI] [PubMed] [Google Scholar]
- 33. Baron-Esquivias G, Morillo CA, Moya-Mitjans A, et al. Dual-chamber pacing with closed loop stimulation in recurrent reflex vasovagal syncope: the SPAIN study. J Am Coll Cardiol 2017;70:1720–8. 10.1016/j.jacc.2017.08.026 [DOI] [PubMed] [Google Scholar]
- 34. Parry SW, Steen N, Bexton RS, et al. Pacing in elderly recurrent Fallers with carotid sinus hypersensitivity: a randomised, double-blind, placebo controlled crossover trial. Heart 2009;95:405–9. 10.1136/hrt.2008.153189 [DOI] [PubMed] [Google Scholar]
- 35. Claesson J-E, Kristensson B-E, Edvardsson N, et al. Less syncope and milder symptoms in patients treated with pacing for induced cardioinhibitory carotid sinus syndrome: a randomized study. Europace 2007;9:932–6. 10.1093/europace/eum180 [DOI] [PubMed] [Google Scholar]
- 36. Occhetta E, Bortnik M, Audoglio R, et al. Closed loop stimulation in prevention of vasovagal syncope. Inotropy controlled pacing in vasovagal syncope (INVASY): a multicentre randomized, single blind, controlled study. Europace 2004;6:538–47. 10.1016/j.eupc.2004.08.009 [DOI] [PubMed] [Google Scholar]
- 37. Russo V, Rago A, Papa AA, et al. The effect of dual-chamber closed-loop stimulation on syncope recurrence in healthy patients with tilt-induced vasovagal cardioinhibitory syncope: a prospective, randomised, single-blind, crossover study. Heart 2013;99:1609–13. 10.1136/heartjnl-2013-303878 [DOI] [PubMed] [Google Scholar]
- 38. Connolly SJ, Sheldon R, Roberts RS, et al. The North American Vasovagal pacemaker study (VPS). A randomized trial of permanent cardiac pacing for the prevention of vasovagal syncope. J Am Coll Cardiol 1999;33:16–20. 10.1016/s0735-1097(98)00549-x [DOI] [PubMed] [Google Scholar]
- 39. Kenny RA, Richardson DA, Steen N, et al. Carotid sinus syndrome: a modifiable risk factor for nonaccidental falls in older adults (SAFE PACE). J Am Coll Cardiol 2001;38:1491–6. 10.1016/s0735-1097(01)01537-6 [DOI] [PubMed] [Google Scholar]
- 40. Flammang D, Church TR, De Roy L, et al. Treatment of unexplained syncope: a multicenter, randomized trial of cardiac pacing guided by adenosine 5'-Triphosphate testing. Circulation 2012;125:31–6. 10.1161/CIRCULATIONAHA.111.022855 [DOI] [PubMed] [Google Scholar]
- 41. McLeod CJ, Trusty JM, Jenkins SM, et al. Method of pacing does not affect the recurrence of syncope in carotid sinus syndrome. Pacing Clin Electrophysiol 2012;35:827–33. 10.1111/j.1540-8159.2012.03375.x [DOI] [PubMed] [Google Scholar]
- 42. Deharo J-C, Brunetto AB, Bellocci F, et al. DDDR pacing driven by contractility versus DDI pacing in vasovagal syncope: a multicenter, randomized study. Pacing Clin Electrophysiol 2003;26:447–50. 10.1046/j.1460-9592.2003.00068.x [DOI] [PubMed] [Google Scholar]
- 43. Ammirati F, Colivicchi F, Toscano S, et al. DDD pacing with rate drop response function versus DDI with rate hysteresis pacing for cardioinhibitory vasovagal syncope. Pacing Clin Electrophysiol 1998;21:2178–81. 10.1111/j.1540-8159.1998.tb01148.x [DOI] [PubMed] [Google Scholar]
- 44. Brignole M, Menozzi C, Gaggioli G, et al. Effects of long-term vasodilator therapy in patients with carotid sinus hypersensitivity. Am Heart J 1998;136:264–8. 10.1053/hj.1998.v136.89911 [DOI] [PubMed] [Google Scholar]
- 45. Alizadeh A, Peighambari M, Keikhavani A, et al. The role of acute physical maneuver in preventing vasovagal syncope: a randomized clinical trial. Clinic Cardia Electrophysiology 2016;1. [Google Scholar]
- 46. Duygu H, Zoghi M, Turk U, et al. The role of tilt training in preventing recurrent syncope in patients with vasovagal syncope: a prospective and randomized study. Pacing Clin Electrophysiol 2008;31:592–6. 10.1111/j.1540-8159.2008.01046.x [DOI] [PubMed] [Google Scholar]
- 47. On YK, Park J, Huh J, et al. Is home orthostatic self-training effective in preventing neurally mediated syncope? Pacing Clin Electrophysiol 2007;30:638–43. 10.1111/j.1540-8159.2007.00725.x [DOI] [PubMed] [Google Scholar]
- 48. Tan MP, Newton JL, Chadwick TJ, et al. Home orthostatic training in vasovagal syncope modifies autonomic tone: results of a randomized, placebo-controlled pilot study. Europace 2010;12:240–6. 10.1093/europace/eup368 [DOI] [PubMed] [Google Scholar]
- 49. Zeng H, Ge K, Zhang W, et al. The effect of orthostatic training in the prevention of vasovagal syncope and its influencing factors. Int Heart J 2008;49:707–12. 10.1536/ihj.49.707 [DOI] [PubMed] [Google Scholar]
- 50. van Dijk N, Quartieri F, Blanc J-J, et al. Effectiveness of physical counterpressure maneuvers in preventing vasovagal syncope: the physical counterpressure manoeuvres trial (PC-trial). J Am Coll Cardiol 2006;48:1652–7. 10.1016/j.jacc.2006.06.059 [DOI] [PubMed] [Google Scholar]
- 51. He L, Wang L, Li L, et al. A single-center randomized controlled trial observing the safety and efficacy of modified step-up graded Valsalva Manoeuver in patients with vasovagal syncope. PLoS One 2018;13:e0191880. 10.1371/journal.pone.0191880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Aghajani F, Tavolinejad H, Sadeghian S, et al. Implementation of supervised physical training to reduce vasovagal syncope recurrence: a randomized controlled trial. J Cardiovasc Electrophysiol 2022;33:1863–70. 10.1111/jce.15578 [DOI] [PubMed] [Google Scholar]
- 53. Sharma G, Ramakumar V, Sharique M, et al. Effect of yoga on clinical outcomes and quality of life in patients with vasovagal syncope (LIVE-yoga). JACC Clin Electrophysiol 2022;8:141–9. 10.1016/j.jacep.2021.09.007 [DOI] [PubMed] [Google Scholar]
- 54. Palmisano P, Dell’Era G, Russo V, et al. Effects of closed-loop stimulation vs. DDD pacing on haemodynamic variations and occurrence of syncope induced by head-up tilt test in older patients with refractory cardioinhibitory vasovagal syncope: the tilt test-induced response in closed-loop stimulation multicentre, prospective, single blind, randomized study. Europace 2018;20:859–66. 10.1093/europace/eux015 [DOI] [PubMed] [Google Scholar]
- 55. Gopinathannair R, Salgado BC, Olshansky B. Pacing for vasovagal syncope. Arrhythm Electrophysiol Rev 2018;7:95–102. 10.15420/aer.2018.22.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Wieling W, Jardine DL, de Lange FJ, et al. Cardiac output and vasodilation in the vasovagal response: an analysis of the classic papers. Heart Rhythm 2016;13:798–805. 10.1016/j.hrthm.2015.11.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Brignole M, Tomaino M, Aerts A, et al. Benefit of dual-chamber pacing with closed loop stimulation in tilt-induced cardio-inhibitory reflex syncope (Biosync trial): study protocol for a randomized controlled trial. Trials 2017;18:208. 10.1186/s13063-017-1941-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Palmisano P, Zaccaria M, Luzzi G, et al. Closed-loop cardiac pacing vs. conventional dual-chamber pacing with specialized sensing and pacing algorithms for syncope prevention in patients with refractory vasovagal syncope: results of a long-term follow-up. Europace 2012;14:1038–43. 10.1093/europace/eur419 [DOI] [PubMed] [Google Scholar]
- 59. Rattanawong P, Riangwiwat T, Chongsathidkiet P, et al. Closed-looped stimulation cardiac pacing for recurrent vasovagal syncope: a systematic review and meta-analysis. J Arrhythm 2018;34:556–64. 10.1002/joa3.12102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Liao Y, Li X, Zhang Y, et al. Alpha-adrenoceptor agonists for the treatment of vasovagal syncope: a meta-analysis of worldwide published data. Acta Paediatr 2009;98:1194–200. 10.1111/j.1651-2227.2009.01289.x [DOI] [PubMed] [Google Scholar]
- 61. Sheldon R, Raj SR, Rose MS, et al. Fludrocortisone for the prevention of vasovagal syncope: a randomized, placebo-controlled trial. J Am Coll Cardiol 2016;68:1–9. 10.1016/j.jacc.2016.04.030 [DOI] [PubMed] [Google Scholar]
- 62. Brignole M, Menozzi C, Moya A, et al. Pacemaker therapy in patients with neurally mediated syncope and documented asystole: third international study on syncope of uncertain etiology (ISSUE-3): a randomized trial. Circulation 2012;125:2566–71. 10.1161/CIRCULATIONAHA.111.082313 [DOI] [PubMed] [Google Scholar]
- 63. Stang A, Hense H-W, Jöckel K-H, et al. Is it always unethical to use a placebo in a clinical trial? PLoS Med 2005;2:e72. 10.1371/journal.pmed.0020072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Imrey PB. Limitations of meta-analyses of studies with high heterogeneity. JAMA Netw Open 2020;3:e1919325. 10.1001/jamanetworkopen.2019.19325 [DOI] [PubMed] [Google Scholar]
- 65. Vandenberk B, Lei LY, Ballantyne B, et al. Cardioneuroablation for vasovagal syncope: a systematic review and meta-analysis. Heart Rhythm 2022;19:1804–12. 10.1016/j.hrthm.2022.06.017 [DOI] [PubMed] [Google Scholar]
- 66. Piotrowski R, Baran J, Sikorska A, et al. Cardioneuroablation for reflex syncope: efficacy and effects on autonomic cardiac regulation-A prospective randomized trial. JACC Clin Electrophysiol 2023;9:85–95. 10.1016/j.jacep.2022.08.011 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
openhrt-2024-002669supp001.pdf (433.9KB, pdf)
Data Availability Statement
Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. The datasets analysed during the current study are available from the corresponding author upon reasonable request. Additionally, all included studies are cited in the references section, and readers are encouraged to refer to the original sources for further information on the data used in this meta-analysis.