This meta-analysis assesses the association of the enhanced recovery after surgery guidelines and hospital length of stay, readmission, complications, and mortality compared with usual care.
Key Points
Question
Does the adoption of enhanced recovery after surgery (ERAS) guidelines decrease hospital length of stay, hospital readmission, complications, and mortality?
Findings
In this meta-analysis of 74 randomized clinical trials with 9076 participants, the ERAS guidelines were associated with decreased hospital length of stay and complications. Type of surgery and number of ERAS elements were associated with estimates of length of stay.
Meaning
These findings suggest that the ERAS guidelines were associated with decreasing hospital length of stay and complications; future research should focus on improving implementation and compliance with ERAS guidelines to improve patient outcomes.
Abstract
Importance
A comprehensive review of the evidence exploring the outcomes of enhanced recovery after surgery (ERAS) guidelines has not been completed.
Objective
To evaluate if ERAS guidelines are associated with improved hospital length of stay, hospital readmission, complications, and mortality compared with usual surgical care, and to understand differences in estimates based on study and patient factors.
Data Sources
MEDLINE, Embase, Cumulative Index to Nursing and Allied Health Literature, and Cochrane Central were searched from inception until June 2021.
Study Selection
Titles, abstracts, and full-text articles were screened by 2 independent reviewers. Eligible studies were randomized clinical trials that examined ERAS-guided surgery compared with a control group and reported on at least 1 of the outcomes.
Data Extraction and Synthesis
Data were abstracted in duplicate using a standardized data abstraction form. The study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Risk of bias was assessed in duplicate using the Cochrane Risk of Bias tool. Random-effects meta-analysis was used to pool estimates for each outcome, and meta-regression identified sources of heterogeneity within each outcome.
Main Outcome and Measures
The primary outcomes were hospital length of stay, hospital readmission within 30 days of index discharge, 30-day postoperative complications, and 30-day postoperative mortality.
Results
Of the 12 047 references identified, 1493 full texts were screened for eligibility, 495 were included in the systematic review, and 74 RCTs with 9076 participants were included in the meta-analysis. Included studies presented data from 21 countries and 9 ERAS-guided surgical procedures with 15 (20.3%) having a low risk of bias. The mean (SD) Reporting on ERAS Compliance, Outcomes, and Elements Research checklist score was 13.5 (2.3). Hospital length of stay decreased by 1.88 days (95% CI, 0.95-2.81 days; I2 = 86.5%; P < .001) and the risk of complications decreased (risk ratio, 0.71; 95% CI, 0.59-0.87; I2 = 78.6%; P < .001) in the ERAS group. Risk of readmission and mortality were not significant.
Conclusions and Relevance
In this meta-analysis, ERAS guidelines were associated with decreased hospital length of stay and complications. Future studies should aim to improve implementation of ERAS and increase the reach of the guidelines.
Introduction
Globally, more than 313 million people undergo lifesaving or life-prolonging surgery yearly and that number is increasing.1,2 While surgery is essential to maintain health and quality of life, it consumes significant health care resources and is not without complications. The mean length of hospital stay for surgery is 5 days equating to more than 7.5 million patient days each year at a cost of at least $1200 per day.1 The length of hospital stay increases for patients undergoing surgery who experience complications postoperatively.3,4,5,6,7,8,9,10 More than a third of patients undergoing surgery experience complications,11,12,13 placing these patients at higher risk of complications than patients not undergoing surgery.3,4,5,14,15 Patients who experience complications are also more likely to require additional interventions, be readmitted to hospital, and die, which are all associated with increased costs.10,16,17,18 An estimated 50% of complications are preventable, which provides an opportunity to use evidence-based strategies to improve outcomes for patients and health care systems.3,4,8,9,10,11,13,14 Moreover, improving surgical outcomes and reducing health care resources can increase the capacity for patients to access surgical care.8,9
As the need for surgery continues to exceed health care capacity, strategies are needed to ensure care is effective, efficient, and safe. One option is implementing the enhanced recovery after surgery (ERAS) guidelines. There are currently ERAS guidelines for 23 types of surgery19 and implementation programs active in more than 20 other types of surgery.20 ERAS guidelines provide recommendations for perioperative care and have been found to reduce complications,21 length of hospital stay,22 and cost.23,24 Several surgery-specific systematic reviews have found positive or mixed effects on outcomes after adopting ERAS guidelines.25,26,27,28,29,30,31 However, a comprehensive review of ERAS efficacy across surgical areas and within heterogenous populations has not been conducted. To our knowledge, there are no systematic reviews examining these outcomes across the breadth of ERAS-guided surgical procedures or comparing outcomes by surgery type, which could provide a better understanding of ERAS efficacy. The objective of this study is to synthesize the evidence on the efficacy of ERAS and determine if the adoption of ERAS guidelines improves hospital length of stay, hospital readmission, mortality, and postoperative complications compared with usual surgical care?
Methods
This systematic review and meta-analysis is reported using the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guideline.32 The protocol was registered with the PROSPERO registry of systematic reviews (CRD42021255973).
Search Strategy
The search strategy combined controlled vocabulary and keywords for the population (patients undergoing surgery), the intervention (ERAS, fast track, enhanced recovery) and outcomes (length of hospital stay, hospital readmission, complications, mortality) and was run in MEDLINE, Embase, the Cumulative Index to Nursing and Allied Health Literature, and Cochrane Central Register of Controlled Trials on June 18, 2021 (eAppendix 1 in Supplement 1). The search strategy was not limited by publication date or language.
Eligibility Criteria
Studies were included if they reported at least 1 of the outcomes among adult patients undergoing surgery before and after ERAS guidelines were implemented. Studies that implemented ERAS guidelines, including ERAS-based guidelines (eg, ERAS, fast-track, enhanced recovery protocol), were included if the protocols contained elements that were ERAS-based. Any study design that compared the intervention (ERAS guidelines) and the comparator group (usual care) were included in the overall systematic review, but only randomized clinical trials were included in the meta-analysis.
Studies with only pediatric patients or those that did not stratify estimates by adults and children were excluded. Studies that did not provide estimates or sufficient data to calculate estimates were excluded. Conference proceedings or abstracts, reviews, guidelines, and commentaries were excluded. Studies with less than 50 patients total were excluded due to the high risk of bias in these studies. Studies published in any language were included; however, it was excluded if we could not translate the article. For studies with multiple manuscripts, the manuscript with the most comprehensive dataset was included and the others were excluded (duplicate data).
Study Selection
Covidence was used to manage study selection.33 At each stage of screening a sample of titles and abstracts (n = 5), and full texts (n = 10) were screened by all reviewers to ensure consistency between reviewers and additional studies were reviewed together until a κ of 0.8 was achieved. Titles, abstracts, and full texts were screened for eligibility by 2 independent reviewers. During the title and abstract screening, references included by at least 1 reviewer were included for full-text screening to decrease the risk of incorrectly excluding studies. Disagreement between reviewers was resolved through discussion or consulting a third reviewer.
Data Collection Process
A standardized database (Microsoft Excel) was used to abstract data from included studies. The data abstraction database was pilot tested on 2 studies. Prior to full data abstraction all reviewers were trained by completing data abstraction for the same 5 studies; training continued until a κ of 0.8 was achieved. Data were abstracted by a single reviewer and 65% of the data were double-checked by an independent reviewer. The data dictionary for the standardized data abstraction form is available in eAppendix 2 in Supplement 1.
Variables
The primary outcome variables were hospital length of stay, readmission within 30 days of index hospital discharge, postoperative complications within 30 days of surgery, and mortality within 30 days of surgery. While the primary outcome variables were limited to 30 days from surgery or index hospitalization, data for other time periods were abstracted as secondary outcomes.
Additional data items abstracted from the studies included bibliographic information, study characteristics, patient data, type of protocol evaluated (ERAS, enhanced recovery, fast-track, other), number of ERAS elements included in the protocol (count) even if compliance was not reported for each element, and data on compliance. A full list of data items abstracted is included in the data dictionary (eAppendix 2 in Supplement 1).
Risk of Bias in Individual Studies
The risk of bias of individual studies was evaluated using the Cochrane Risk of Bias Tool (version 2),34 by 2 independent reviewers with graduate training in epidemiology. Any disagreements between reviewers were resolved through discussion. The Reporting on ERAS Compliance, Outcomes, and Elements Research (RECOvER) checklist assessed completeness of reporting.35
Data Analysis
Study characteristics were described using descriptive statistics as appropriate. A random effects meta-analysis was used to pool estimates for each outcome variable (metan package).36,37 The I2 quantified the magnitude of between-study heterogeneity, and the Cochrane Q statistic determined the significance of heterogeneity. Risk ratios (RR) were calculated for data reported as proportions (ie, mortality, complications, and readmission), and mean differences were calculated for data reported as means (ie, hospital length of stay). Estimates reported as median (IQR) were converted to mean (SD) using the Wan method38 and were pooled.
Meta-regression explored potential sources of heterogeneity for each outcome. The potential sources of heterogeneity included patient age and sex, year(s) of data collection, type of surgery, number of ERAS elements included in the protocol, type of protocol (eg, ERAS, enhanced recovery protocol, fast-track, or other), and RECOvER scores. Publication bias was assessed through visual inspection of a funnel plot and statistically using Egger linear regression method. All analyses were performed in Stata 20.0 (StataCorp).36,37 Data were analyzed from September 2023 to February 2024. Statistical tests were 2-sided, and significance was set at P < .05.
Results
The search strategy yielded 12 047 unique references, of which 1493 were reviewed in full-text, 495 were included in the overall systematic review (eFigure in Supplement 1) and 74 RCTs 39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112 were included in the meta-analysis. The characteristics of each individual study are included in eTable 1 in Supplement 1. The included studies represent 9076 patients with 4577 patients in the control (non-ERAS) group and 4375 patients in the ERAS group. In the 54 studies that included age, the mean (SD) age was 50.0 (12.6) years for patients in the control group and 49.2 (12.6) years among patients in the ERAS group.39,41,42,43,44,45,46,47,49,50,51,52,53,54,56,57,59,62,63,64,65,66,67,68,70,73,76,79,81,82,83,84,86,88,89,91,93,94,95,96,97,98,99,100,101,102,103,104,105,106,108,109,110,111
Study Characteristics
The median (range) year of publication was 2019 (2009-2021). Most studies were conducted in high income and high-middle income countries (57 [80.3%]); 22 (29.7%) were published in China,40,46,55,56,67,68,69,70,71,72,75,82,85,86,87,103,107,108,109,112,113 8 (10.8%) in India,44,45,60,76,84,91,97,100 5 (6.8%) in the US.47,49,66,99,104
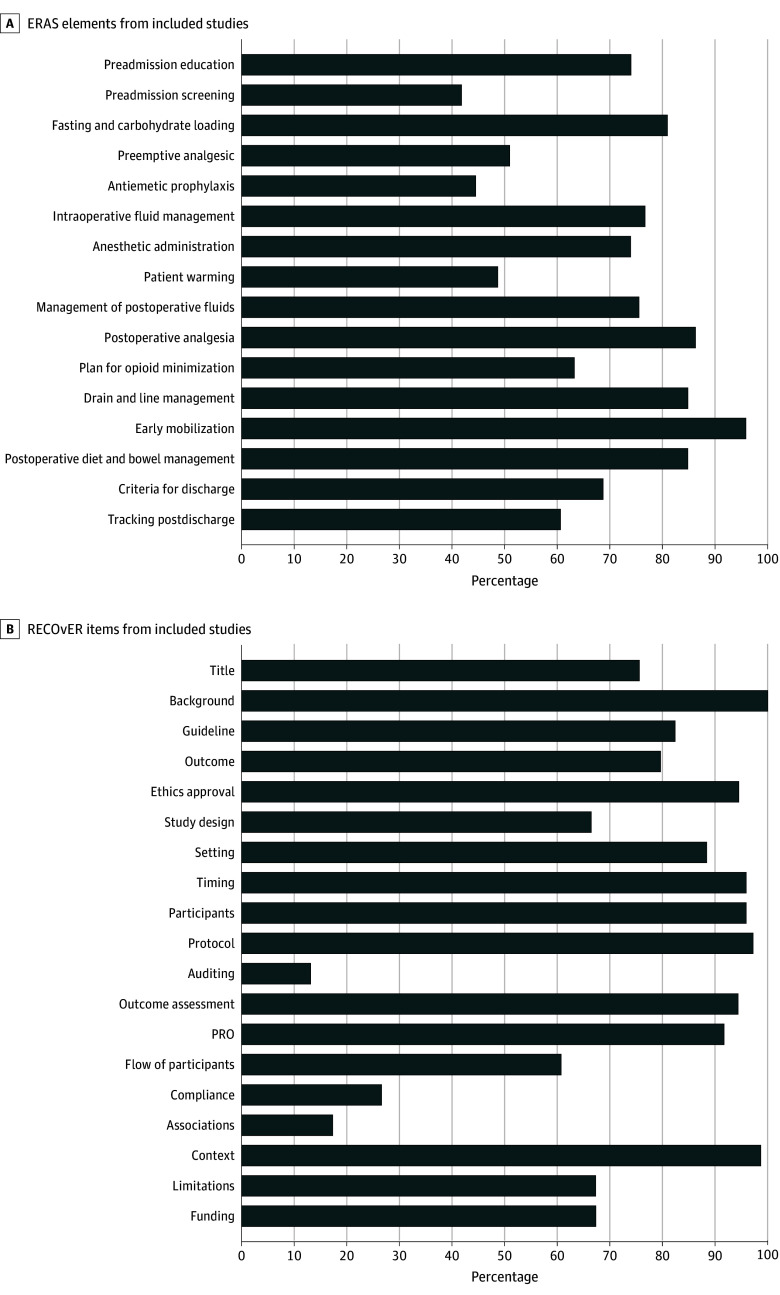
Most of the 74 studies examined the outcomes of ERAS in patients undergoing gastrointestinal surgery (32 [43.2%])39,40,45,47,50,51,53,54,56,58,59,60,73,74,77,79,80,83,87,92,93,94,95,96,97,98,100,102,103,105,112,114 and gynecological surgical procedures (11 [14.9%]),41,43,49,52,65,89,90,110,111,115 with only 1 (1.4%) in cardiac surgery,68 and 2 (2.7%) in urology.44,106 Most of the 74 studies evaluated the outcomes of ERAS guidelines (37 [50.0%]),39,40,41,42,43,44,45,46,48,52,53,54,57,68,69,70,71,73,75,78,82,87,88,89,90,91,98,99,100,101,104,105,106,107,108,110,111 whereas 23 (31.1%) evaluated enhanced recovery protocols,47,49,55,56,58,61,62,63,64,65,66,76,77,79,80,81,83,84,85,95,96,97,102 13 (17.6%) evaluated fast-track protocols,50,51,59,60,72,74,86,92,93,94,103,109,112,116 and 1 (1.4%) evaluated other types of protocols or pathways based on ERAS principles.67 The mean (SD) number of ERAS elements included in the interventions (ERAS, enhanced protocols, fast-track, other) was 11.1 (3.0) elements of a possible 16. Commonly early mobilization (71 [96.0%]), postoperative analgesia management (64 [86.5%]), postoperative diet and bowel management (63 [85.1%]), and drain and tube management (63 [85.1%]) were included (Figure 1). Protocols classified as ERAS included more elements than those classified as fast-track but were no different than enhanced recovery protocols (F1,493 = 9.58; P = .002). The mean (SD) number of elements among studies of ERAS was 11.8 (2.8), compared with 8.9 (2.3) in fast-track protocols, 10.6 (3.7) in enhanced recovery protocols, and 11.4 (3.2) elements with other types of protocols. Compliance with ERAS protocols was reported in 17 (20.3%) studies49,52,53,56,59,67,69,70,77,80,88,89,90,99,101,106,107 but was inconsistently reported and could not be pooled; the mean (SD) compliance reported was 74.7% (10.2%) from the 9 studies from which we could synthesize the data.49,52,67,80,89,90,99,101,106
Figure 1. Study Characteristics.
(A) Among the studies that reported the enhanced recovery after surgery (ERAS) elements, most included early mobilization, postoperative analgesia considerations, and postoperative diet and bowel management. (B) Reporting on ERAS Compliance, Outcomes, and Elements Research (RECOvER) checklist items of included studies. PRO indicates patient reported outcomes.
Meta-Analysis by Outcome
Length of Stay
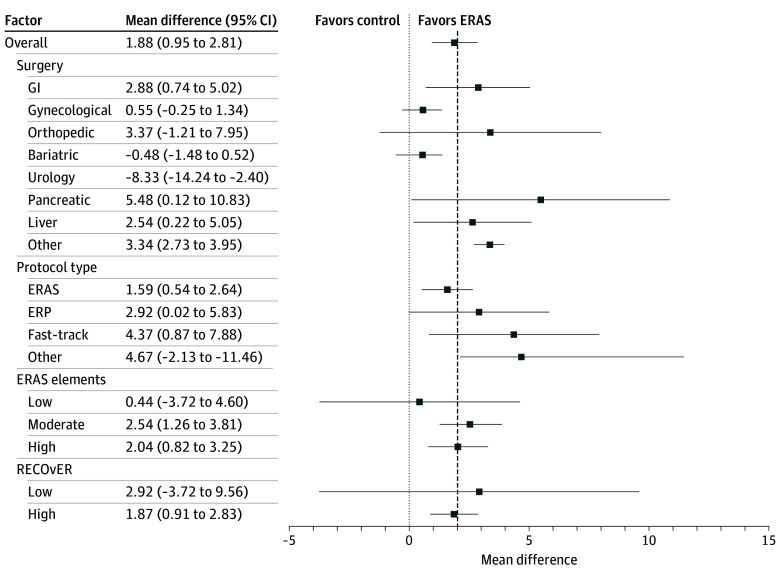
Of the 74 included studies, 44 studies (62.2%) reported an overall median or mean estimate of hospital length of stay.39,40,42,44,45,47,48,49,50,52,53,55,58,60,61,65,66,71,73,74,78,79,82,84,85,88,89,90,91,95,97,98,101,103,105,107,108,109,111,113 The pooled mean length of hospital stay was 1.88 days (95% CI, 0.95-2.81 days; I2 = 86.5%; P < .001) shorter in the ERAS groups compared with control groups. ERAS also decreased the postoperative hospital length of stay (22 studies; mean difference, 2.83 days; 95% CI, 2.10-3.55 days; I2 = 0%; P < .001) compared with control groups. Publication bias was not significant.40,41,53,54,56,63,64,68,71,72,75,79,82,85,87,94,104,105,107,112,113,117
Hospital Readmission
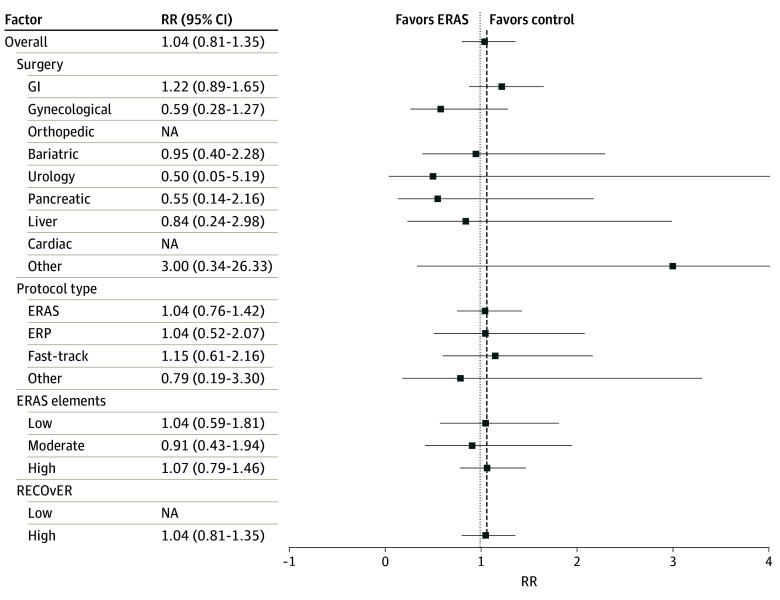
Nineteen of 74 studies (41.3%) reported 30-day hospital readmissions. The pooled RR of 30-day hospital readmission was 1.04 (95% CI, 0.81-1.35; I2 = 0%; P = .74)39,43,44,52,53,54,65,66,67,69,79,80,89,94,97,101,102,105,112; with a protective effect when the time period was not specified (11 studies, RR, 0.61; 95% CI, 0.41-0.93; I2 = 0%; P = .02).41,42,47,56,63,75,82,88,98,106,111 Publication bias was not significant.
Complications
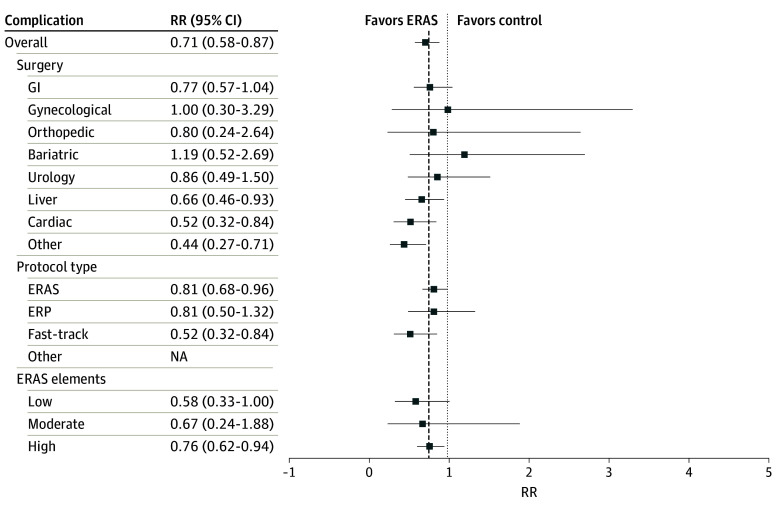
Fourteen of 74 studies (18.9%) reported an overall (unstratified) estimate of complications with a pooled estimate of 0.71 (95% CI, 0.59-0.87; I2 = 78.6%; P < .001),44,46,53,67,68,69,79,80,82,86,87,93,95,96 favoring ERAS compared with control groups. Of the nine studies that reported 30-day complications, the pooled RR was 0.73 (95% CI, 0.56-0.94; I2 = 86.3%; P < .02),53,67,79,80,82,87,93,95,96 favoring ERAS over control groups. There was evidence of publication bias using the Egger test for small study effects (t = 2.24; 95% CI, 0.14-2.15; P = .003); however, estimates were symmetrically distributed upon visual inspection of the funnel plot.
Mortality
There were 19 of 74 studies that reported 30-day postoperative mortality, with a pooled risk of 0.95 (95% CI, 0.48-1.88) with no heterogeneity between estimates (I2 = 0%; P = .89).40,44,50,53,54,57,69,70,77,78,87,89,92,93,95,96,97,98,107 Because many studies reported estimates of 0 mortality, a continuity correction was applied to calculate RRs.118 Publication bias was not significant.
Sources of Heterogeneity Determined by Meta-Regression
There were no differences in the pooled estimates of hospital length of stay based on country income, RECOvER checklist score, year of publication, number of ERAS elements included in the protocol, or risk of bias. The type of surgery accounted for heterogeneity in the estimate of hospital length of stay (coefficient, 0.32; 95% CI, 0.17-0.48; P < .001). The pooled estimate of mean difference in length of hospital stays for patients undergoing pancreatic, orthopedic, and gastrointestinal surgery was larger than other surgical procedures (Figure 2). None of the variables explored accounted for variance in the pooled estimate of 30-day hospital readmission, complications, or mortality. These findings can be found in Figure 3 and Figure 4.
Figure 2. Meta-Analysis of Hospital Length of Stay.
Patients in enhanced recovery after surgery (ERAS) groups stayed 1.9 days less in hospital compared with control groups. Patient undergoing pancreatic, orthopedic, and gastrointestinal (GI) surgical procedures had greater reductions in hospitals stays than patients undergoing gynecological and breast surgery. ERP, enhanced recovery protocol; RECOvER, Reporting on ERAS Compliance, Outcomes, and Elements Research.
Figure 3. Meta-Analysis of Hospital Readmission.
Patients in the enhanced recovery after surgery (ERAS) groups had a lower risk of being readmitted after their index hospitalization than patients in the control group. None of the variables explored were associated with the risk of readmission. ERP indicates enhanced recovery protocol; GI, gastrointestinal; NA, not applicable; RECOvER, Reporting on ERAS Compliance, Outcomes, and Elements Research; RR, risk ratio.
Figure 4. Meta-Analysis of Complications.
Patients in the enhanced recovery after surgery (ERAS) groups had a lower risk of having complications after their surgery than patients in the control group. None of the variables explored were associated with the risk of complications. ERP indicates enhanced recovery protocol; GI, gastrointestinal; NA, not applicable; RR, risk ratio.
Risk of Bias and RECOvER
Among the 74 included studies, 40 (54.1%) had some risk of bias, 19 (25.7%) had high risk of bias, and 15 (20.3%) had low risk of bias. Missingness of outcome data were considered a source of low risk of bias among 64 studies (86.5%) while deviations from the per-protocol intervention was considered a source of high risk of bias in 17 studies (23.0%) (eTable 2 in Supplement 1).
The mean (SD) number of RECOvER checklist items that were included in the studies was 13.5 (2.3). Commonly reported items included providing context and importance within the background (74 [100.0%]), explaining the significance of the study (73 [98.7%]), and reporting the protocol used (72 [97.3%]) (Figure 1). The least commonly reported items were auditing of outcomes (10 [13.5%]), examining associations between outcomes and other variables (13 [17.6%]), and reporting compliance with the ERAS guidelines (20 [27.0%] (Figure 1).
Discussion
This study found that using the ERAS guidelines were associated with a decrease in the length of hospital stay and complications. Hospital length of stay varied by surgery type. There were many studies included, but some evidence gaps remain. For example, there was a paucity of evidence for some surgical procedures (none for head and neck and breast, and few studies for cardiac), fewer studies reported mortality, and compliance was reported inconsistently and poorly.
While the ERAS guidelines were established with the publication of the ERAS Society guideline for perioperative care of patients undergoing colonic surgery,119 enhanced recovery as a concept was based on previously described fast-track protocols.120,121,122 Accordingly, it is not surprising that many included studies were labeled as fast-track and that a large proportion were focused on gastrointestinal surgery. However, this study did not identify any differences in estimates of hospital length of stay, readmission, complications, or mortality related to the protocol name. While the name given to the protocol examined did not account for heterogeneity between estimates, the number of ERAS elements included in the protocols did for length of hospital stay. This suggests that the number of elements included in the protocol is likely more important than the branding of the protocol, which is supported by previous studies.123,124 Encouragingly, most studies reported their protocol elements (90%), which is important for evaluating ERAS outcomes,35 but very few studies reported compliance with each element. Unfortunately, most studies included less than 70% (11) of the traditional ERAS elements. While the number of ERAS elements included in the protocol is important, it was not possible to assess what elements are most important for the outcomes of ERAS. Future studies should focus on evaluating the relative outcomes of each ERAS element to understand the core elements required to achieve the benefits of ERAS. This will simplify ERAS and promote adoption in lower-resource settings.125,126
Like with other guidelines, ERAS outcomes depends on adoption of ERAS guidelines.127,128,129 Indeed, greater compliance with ERAS has been shown to improve outcomes.129,130,131 Since only about 20% of included studies reported compliance with ERAS, and reporting of these estimates were too heterogeneous to include in the analysis, it is likely that mean compliance was lower than 75% and that our findings underestimate the outcomes of ERAS. If compliance is important for ERAS outcomes, how can compliance be improved? Strategies for implementing ERAS have been outlined, with a focus on team building and audit of compliance-and-feedback.126,132 These strategies are aligned with principles of implementation science, and evidence-informed strategies for effective guideline implementation.127,128 Additional strategies that should be considered when implementing ERAS include identifying barriers and facilitators to implementation, including end-users in developing strategies, education, and prompts-and-reminders.127,133,134 Taken together, future work should focus on strategies to improve implementation of and compliance with ERAS guidelines; the importance of which has been noted.126 Additionally, in agreement with the RECOvER checklist, we advocate for improved reporting of overall compliance and compliance with each ERAS element in studies exploring the outcomes of ERAS guidelines.
Limitations
This study has limitations. While the search strategy was developed by a librarian with expertise in systematic reviews, some studies may not have been identified. Using the Egger statistic and a visual assessment of funnel plots, we did not find evidence of bias toward studies with large effect sizes. While the comprehensiveness of the systematic review is a strength, it can also be limiting because included studies were heterogeneous based on the type of ERAS protocol, surgery type, and outcomes. Additionally, analysis is limited by the data provided within the included studies. As such, variables that may be associated with our outcome variables, including socioeconomic status, insurance status, and surgical complexity, were not accounted for in the analysis. Similarly, patients undergoing ERAS-guided surgical procedures may be a highly selected group, which we could not control for. Finally, these findings may not be generalized to all types of surgery because most studies explored the efficacy of ERAS in colorectal surgery and obstetric or gynecologic surgery. Indeed our findings suggested that the type of surgery was associated with length of hospital stay, but the reason for the variation was unclear.
Conclusions
This meta-analysis found that ERAS was associated with a decreased length of hospital stay (without a significant increase in hospital readmissions) and complications. Given the improvements across specialties, future research should aim to apply ERAS to new surgical specialties and in more clinical settings globally.
eAppendix 1. Search Strategy
eAppendix 2. Data Dictionary
eTable 1. Characteristics of Included Studies
eTable 2. Risk of Bias
eFigure. PRISMA Flow Diagram
Data Sharing Statement
References
- 1.Inpatient hospitalization, surgery, and newborn statistics, 2019-2020. Canadian Institute for Health Information. Accessed May 30, 2024. https://www.cihi.ca/en/hospital-stays-in-canada-series
- 2.Weiser TG, Haynes AB, Molina G, et al. Estimate of the global volume of surgery in 2012: an assessment supporting improved health outcomes. Lancet. 2015;385(suppl 2):S11. doi: 10.1016/S0140-6736(15)60806-6 [DOI] [PubMed] [Google Scholar]
- 3.Baker GRN, Norton PG, Flintoft V, et al. The Canadian Adverse Events Study: the incidence of adverse events among hospital patients in Canada. CMAJ. 2004;170(11):1678-1686. doi: 10.1503/cmaj.1040498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brennan TAL, Leape LL, Laird NM, et al. Incidence of adverse event and negligence in hospitalized patients. Results of the Harvard Medical Practice Study I. N Engl J Med. 1991;324(6):370-376. doi: 10.1056/NEJM199102073240604 [DOI] [PubMed] [Google Scholar]
- 5.Forster AJA, Asmis TR, Clark HD, et al. ; Ottawa Hospital Patient Safety Study . Ottawa Hospital Patient Safety Study: incidence and timing of adverse events in patients admitted to a Canadian teaching hospital. CMAJ. 2004;170(8):1235-1240. doi: 10.1503/cmaj.1030683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sauro KM, Machan M, Whalen-Browne L, Owen V, Wu G, Stelfox HT. Evolving trends in hospital safety: a systematic review and meta-analysis. J Patient Saf. 2021. 17(8):e1285-1295. doi: 10.1097/PTS.0000000000000889 [DOI] [PubMed] [Google Scholar]
- 7.Sauro KM, Quan H, Sikdar KC, Faris P, Jette N. Hospital safety among neurologic patients: a population-based cohort study of adverse events. Neurology. 2017;89(3):284-290. doi: 10.1212/WNL.0000000000004111 [DOI] [PubMed] [Google Scholar]
- 8.Sauro KM, Soo A, de Grood C, et al. Adverse events after transition from ICU to hospital ward: a multicenter cohort study. Crit Care Med. 2020;48(7):946-953. doi: 10.1097/CCM.0000000000004327 [DOI] [PubMed] [Google Scholar]
- 9.Sauro KM, Soo A, Quan H, Stelfox HT. Adverse events among hospitalized critically ill patients: a retrospective cohort study. Med Care. 2020;58(1):38-44. doi: 10.1097/MLR.0000000000001238 [DOI] [PubMed] [Google Scholar]
- 10.Zegers M, de Bruijne MC, de Keizer B, et al. The incidence, root-causes, and outcomes of adverse events in surgical units: implication for potential prevention strategies. Patient Saf Surg. 2011;5(1):13. doi: 10.1186/1754-9493-5-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Healey MA, Shackford SR, Osler TM, Rogers FB, Burns E. Complications in surgical patients. Arch Surg. 2002;137(5):611-617. doi: 10.1001/archsurg.137.5.611 [DOI] [PubMed] [Google Scholar]
- 12.Tevis SE, Kennedy GD. Postoperative complications and implications on patient-centered outcomes. J Surg Res. 2013;181(1):106-113. doi: 10.1016/j.jss.2013.01.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Woodfield JC, Jamil W, Sagar PM. Incidence and significance of postoperative complications occurring between discharge and 30 days: a prospective cohort study. J Surg Res. 2016;206(1):77-82. doi: 10.1016/j.jss.2016.06.073 [DOI] [PubMed] [Google Scholar]
- 14.Baines RJ, Langelaan M, de Bruijne MC, et al. Changes in adverse event rates in hospitals over time: a longitudinal retrospective patient record review study. BMJ Qual Saf. 2013;22(4):290-298. doi: 10.1136/bmjqs-2012-001126 [DOI] [PubMed] [Google Scholar]
- 15.Davis P, Lay-Yee R, Briant R, Ali W, Scott A, Schug S. Adverse events in New Zealand public hospitals II: preventability and clinical context. N Z Med J. 2003;116(1183):U624. [PubMed] [Google Scholar]
- 16.Vonlanthen R, Slankamenac K, Breitenstein S, et al. The impact of complications on costs of major surgical procedures: a cost analysis of 1200 patients. Ann Surg. 2011;254(6):907-913. doi: 10.1097/SLA.0b013e31821d4a43 [DOI] [PubMed] [Google Scholar]
- 17.Seese L, Sultan I, Gleason TG, et al. The impact of major postoperative complications on long-term survival after cardiac surgery. Ann Thorac Surg. 2020;110(1):128-135. doi: 10.1016/j.athoracsur.2019.09.100 [DOI] [PubMed] [Google Scholar]
- 18.Patel AS, Bergman A, Moore BW, Haglund U. The economic burden of complications occurring in major surgical procedures: a systematic review. Appl Health Econ Health Policy. 2013;11(6):577-592. doi: 10.1007/s40258-013-0060-y [DOI] [PubMed] [Google Scholar]
- 19.Guidelines. ERAS Society. Accessed May 13, 2024. https://erassociety.org/guidelines/list-of-guidelines/
- 20.Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152(3):292-298. doi: 10.1001/jamasurg.2016.4952 [DOI] [PubMed] [Google Scholar]
- 21.Varadhan KK, Neal KR, Dejong CH, Fearon KC, Ljungqvist O, Lobo DN. The enhanced recovery after surgery (ERAS) pathway for patients undergoing major elective open colorectal surgery: a meta-analysis of randomized controlled trials. Clin Nutr. 2010;29(4):434-440. doi: 10.1016/j.clnu.2010.01.004 [DOI] [PubMed] [Google Scholar]
- 22.Senagore AJ, Whalley D, Delaney CP, Mekhail N, Duepree HJ, Fazio VW. Epidural anesthesia-analgesia shortens length of stay after laparoscopic segmental colectomy for benign pathology. Surgery. 2001;129(6):672-676. doi: 10.1067/msy.2001.114648 [DOI] [PubMed] [Google Scholar]
- 23.Nelson G, Kiyang LN, Crumley ET, et al. Implementation of enhanced recovery after surgery (ERAS) across a provincial healthcare system: the ERAS Alberta colorectal surgery experience. World J Surg. 2016;40(5):1092-1103. doi: 10.1007/s00268-016-3472-7 [DOI] [PubMed] [Google Scholar]
- 24.Thanh N, Nelson A, Wang X, et al. Return on investment of the enhanced recovery after surgery (ERAS) multiguideline, multisite implementation in Alberta, Canada. Can J Surg. 2020;63(6):E542-E550. doi: 10.1503/cjs.006720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Azhar RA, Bochner B, Catto J, et al. Enhanced recovery after urological surgery: a contemporary systematic review of outcomes, key elements, and research needs. Eur Urol. 2016;70(1):176-187. doi: 10.1016/j.eururo.2016.02.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bisch SP, Jago CA, Kalogera E, et al. Outcomes of enhanced recovery after surgery (ERAS) in gynecologic oncology - A systematic review and meta-analysis. Gynecol Oncol. 2021;161(1):46-55. doi: 10.1016/j.ygyno.2020.12.035 [DOI] [PubMed] [Google Scholar]
- 27.Greco M, Capretti G, Beretta L, Gemma M, Pecorelli N, Braga M. Enhanced recovery program in colorectal surgery: a meta-analysis of randomized controlled trials. World J Surg. 2014;38(6):1531-1541. doi: 10.1007/s00268-013-2416-8 [DOI] [PubMed] [Google Scholar]
- 28.Huang ZD, Gu HY, Zhu J, et al. The application of enhanced recovery after surgery for upper gastrointestinal surgery: meta-analysis. BMC Surg. 2020;20(1):3. doi: 10.1186/s12893-019-0669-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Puccetti F, Wijnhoven BPL, Kuppusamy M, Hubka M, Low DE. Impact of standardized clinical pathways on esophagectomy: a systematic review and meta-analysis. Dis Esophagus. 2022;35(2):doab027. doi: 10.1093/dote/doab027 [DOI] [PubMed] [Google Scholar]
- 30.Lee Y, Yu J, Doumouras AG, Li J, Hong D. Enhanced recovery after surgery (ERAS) versus standard recovery for elective gastric cancer surgery: a meta-analysis of randomized controlled trials. Surg Oncol. 2020;32:75-87. doi: 10.1016/j.suronc.2019.11.004 [DOI] [PubMed] [Google Scholar]
- 31.Ahmed OS, Rogers AC, Bolger JC, Mastrosimone A, Robb WB. Meta-analysis of enhanced recovery protocols in bariatric surgery. J Gastrointest Surg. 2018;22(6):964-972. doi: 10.1007/s11605-018-3709-x [DOI] [PubMed] [Google Scholar]
- 32.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372(71):n71. doi: 10.1136/bmj.n71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Covidence systematic review software. Covidence. Accessed May 13, 2024. https://www.covidence.org/
- 34.Higgins JPT, Altman DG, Gøtzsche PC, et al. ; Cochrane Bias Methods Group; Cochrane Statistical Methods Group . The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Elias KM, Stone AB, McGinigle K, et al. ; ERAS Society and ERAS USA . The reporting on ERAS Compliance, Outcomes, and Elements Research (RECOvER) checklist: a joint statement by the ERAS and ERAS USA Societies. World J Surg. 2019;43(1):1-8. doi: 10.1007/s00268-018-4753-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kontopantelis E, Reeves D. metaan:Random-effects meta-analysis. Stata J. 2010;10(3):395-407. doi: 10.1177/1536867X1001000307 [DOI] [Google Scholar]
- 37.Nyaga VN, Arbyn M, Aerts M. Metaprop: a Stata command to perform meta-analysis of binomial data. Arch Public Health. 2014;72(1):39. doi: 10.1186/2049-3258-72-39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14(1):135. doi: 10.1186/1471-2288-14-135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Abd ElRahman EM, Kharoub MS, Shora A, Emara NA, Balbaa MA. Early outcome of enhanced recovery programs versus conventional perioperative care in elective open left side colonic carcinoma surgery: analysis of 80 cases. Indian J Surg Oncol. 2020;11(3):372-377. doi: 10.1007/s13193-020-01074-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Abdikarim I, Cao XY, Li SZ, Zhao YQ, Taupyk Y, Wang Q. Enhanced recovery after surgery with laparoscopic radical gastrectomy for stomach carcinomas. World J Gastroenterol. 2015;21(47):13339-13344. doi: 10.3748/wjg.v21.i47.13339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Akca A,, yilmAz G. Enhanced recovery after surgery (ERAS) improves SF-12 scores in patients undergoing gynecologic surgery: positive butterfly effect. Acta Med Mediter. 2019;35(4):2277-2282. [Google Scholar]
- 42.Akhtar MS, Khan N, Qayyum A, Khan SZ; Journal of Ayub Medical College . Cost difference of enhanced recovery after surgery pathway vs. conventional care in elective laparoscopic cholecystectomy. J Ayub Med Coll Abbottabad. 2020;32(4):470-475. [PubMed] [Google Scholar]
- 43.Baluku M, Bajunirwe F, Ngonzi J, Kiwanuka J, Ttendo S. A randomized controlled trial of enhanced recovery after surgery versus standard of care recovery for emergency cesarean deliveries at Mbarara Hospital, Uganda. Anesth Analg. 2020;130(3):769-776. doi: 10.1213/ANE.0000000000004495 [DOI] [PubMed] [Google Scholar]
- 44.Bansal D, Nayak B, Singh P, Nayyar R, Ramachandran R, Kumar R, et al. Randomized controlled trial to compare outcomes with and without the enhanced recovery after surgery protocol in patients undergoing radical cystectomy. Indian J Urol. 2020;36(2):95-100. doi: 10.4103/iju.IJU_11_20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chndan MN, Khakholia M, Bhuyan K. Enhanced recovery after surgery (ERAS) versus standard care in patients undergoing emergency surgery for perforated peptic ulcer. Indian J Surg. 2020;82(6):1044-1049. doi: 10.1007/s12262-020-02195-x [DOI] [Google Scholar]
- 46.Cui H, Sun Z, Ruan J, Yu Y, Fan C.. Effect of enhanced recovery after surgery (ERAS) pathway on the postoperative outcomes of elbow arthrolysis: a randomized controlled trial. Int J Surg. 2019;68(101228232):78-84. [DOI] [PubMed] [Google Scholar]
- 47.Damadi AA, Lax EA, Smithson L, Pearlman RD. Comparison of therapeutic benefit of bupivacaine HCL transversus abdominis plane (TAP) block as part of an enhanced recovery pathway versus traditional oral and intravenous pain control after minimally invasive colorectal surgery: a prospective, randomized, double-blind trial. Am Surg. 2019;85(12):1363-1368. doi: 10.1177/000313481908501230 [DOI] [PubMed] [Google Scholar]
- 48.Dambaev GT, Shefer NA, Ena II, Kondaurov AG, Strezh VA. [ERAS protocol for perioperative management of patients with non-small cell lung cancer]. Khirurgiia (Mosk). 2020;(12):52-58. doi: 10.17116/hirurgia202012152 [DOI] [PubMed] [Google Scholar]
- 49.Dickson EL, Stockwell E, Geller MA, et al. Enhanced recovery program and length of stay after laparotomy on a gynecologic oncology service: a randomized controlled trial. Obstet Gynecol. 2017;129(2):355-362. doi: 10.1097/AOG.0000000000001838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fei Y, Zong G-q, Chen J, Liu R-m. Fast-track protocols in devascularization for cirrhotic portal hypertension. Rev Assoc Med Bras 1992. 2015;61(3):250-257. doi: 10.1590/1806-9282.61.03.250 [DOI] [PubMed] [Google Scholar]
- 51.Feng F, Li XH, Shi H, et al. Fast-track surgery combined with laparoscopy could improve postoperative recovery of low-risk rectal cancer patients: a randomized controlled clinical trial. J Dig Dis. 2014;15(6):306-313. doi: 10.1111/1751-2980.12142 [DOI] [PubMed] [Google Scholar]
- 52.Ferrari F, Forte S, Sbalzer N, Zizioli V, Mauri M, Maggi C, et al. Validation of an enhanced recovery after surgery protocol in gynecologic surgery: an Italian randomized study. Am J Obstet Gyn. 2020;223(4):543.e1-543.e14. doi: 10.1016/j.ajog.2020.07.003 [DOI] [PubMed] [Google Scholar]
- 53.Forsmo HM, Pfeffer F, Rasdal A, Ostgaard G, Mohn AC, Korner H, et al. Compliance with enhanced recovery after surgery criteria and preoperative and postoperative counselling reduces length of hospital stay in colorectal surgery: results of a randomized controlled trial. Colorectal Dis. 2016;18(6):603-611. doi: 10.1111/codi.13253 [DOI] [PubMed] [Google Scholar]
- 54.Forsmo HM, Pfeffer F, Rasdal A, Sintonen H, Korner H, Erichsen C. Pre- and postoperative stoma education and guidance within an enhanced recovery after surgery (ERAS) programme reduces length of hospital stay in colorectal surgery. Int J Surg. 2016;36(Pt A):121-126. doi: 10.1016/j.ijsu.2016.10.031 [DOI] [PubMed] [Google Scholar]
- 55.Han H, Guo S, Jiang H, Wu X. Feasibility and efficacy of enhanced recovery after surgery protocol in Chinese elderly patients with intracranial aneurysm. Clin Interv Aging. 2019;14(101273480):203-207. doi: 10.2147/CIA.S187967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.He F, Lin X, Xie F, Huang Y, Yuan R. The effect of enhanced recovery program for patients undergoing partial laparoscopic hepatectomy of liver cancer. Clin Transl Oncol. 2015;17(9):694-701. doi: 10.1007/s12094-015-1296-9 [DOI] [PubMed] [Google Scholar]
- 57.Hwang DW, Kim HJ, Lee JH, et al. Effect of enhanced recovery after surgery program on pancreaticoduodenectomy: a randomized controlled trial. J Hepatobiliary Pancreat Sci. 2019;26(8):360-369. doi: 10.1002/jhbp.641 [DOI] [PubMed] [Google Scholar]
- 58.Ibrahim H, Kotb S, Abd Allah A, et al. Enhanced recovery protocol versus standard protocol for patients undergoing radical cystectomy: results of a prospective randomized study. Afr J Urol. 2020;26(1):2. doi: 10.1186/s12301-019-0012-3 [DOI] [Google Scholar]
- 59.Ionescu D, Iancu C, Ion D, et al. Implementing fast-track protocol for colorectal surgery: a prospective randomized clinical trial. World J Surg. 2009;33(11):2433-2438. doi: 10.1007/s00268-009-0197-x [DOI] [PubMed] [Google Scholar]
- 60.Iyer SP, Kareem Z. Fast-track protocol versus conventional protocol on patient outcome: a randomized clinical trial. Niger J Surg. 2019;25(1):36-41. doi: 10.4103/njs.NJS_34_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jones CN, Morrison BL, Kelliher LJ, et al. Hospital costs and long-term survival of patients enrolled in an enhanced recovery program for open liver resection: prospective randomized controlled trial. JMIR Perioper Med. 2021;4(1):e16829. doi: 10.2196/16829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kang SH, Lee Y, Min SH, et al. Multimodal enhanced recovery after surgery (ERAS) program is the optimal perioperative care in patients undergoing totally laparoscopic distal gastrectomy for gastric cancer: a prospective, randomized, clinical trial. Ann Surg Oncol. 2018;25(11):3231-3238. doi: 10.1245/s10434-018-6625-0 [DOI] [PubMed] [Google Scholar]
- 63.Kapritsou M, Korkolis DP, Giannakopoulou M, Kaklamanos I, Konstantinou M, Katsoulas T, et al. Fast-track recovery program after major liver resection: a randomized prospective study. Gastroenterol Nurs. 2018;41(2):104-110. doi: 10.1097/SGA.0000000000000049 [DOI] [PubMed] [Google Scholar]
- 64.Kapritsou M, Papathanassoglou ED, Konstantinou EA, Korkolis DP, Mpouzika M, Kaklamanos I, et al. Effects of the enhanced recovery program on the recovery and stress response in patients with cancer undergoing pancreatoduodenectomy. Gastroenterol Nurs. 2020;43(2):146-155. doi: 10.1097/SGA.0000000000000417 [DOI] [PubMed] [Google Scholar]
- 65.Kilpiö O, Härkki PSM, Mentula MJ, Väänänen A, Pakarinen PI. Recovery after enhanced versus conventional care laparoscopic hysterectomy performed in the afternoon: a randomized controlled trial. Int J Gynaecol Obstet. 2020;151(3):392-398. doi: 10.1002/ijgo.13382 [DOI] [PubMed] [Google Scholar]
- 66.Lavu H, McCall NS, Winter JM, et al. Enhancing patient outcomes while containing costs after complex abdominal operation: a randomized controlled trial of the whipple accelerated recovery pathway. J Am Coll Surg. 2019;228(4):415-424. doi: 10.1016/j.jamcollsurg.2018.12.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lemanu DP, Singh PP, Berridge K, et al. Randomized clinical trial of enhanced recovery versus standard care after laparoscopic sleeve gastrectomy. Br J Surg. 2013;100(4):482-489. doi: 10.1002/bjs.9026 [DOI] [PubMed] [Google Scholar]
- 68.Li M, Zhang J, Gan TJ, Qin G, Wang L, Zhu M, et al. Enhanced recovery after surgery pathway for patients undergoing cardiac surgery: a randomized clinical trial. Eur J Cardiothorac Surg. 2018;54(3):491-497. doi: 10.1093/ejcts/ezy100 [DOI] [PubMed] [Google Scholar]
- 69.Liang X, Ying H, Wang H, et al. Enhanced recovery care versus traditional care after laparoscopic liver resections: a randomized controlled trial. Surg Endosc. 2018;32(6):2746-2757. doi: 10.1007/s00464-017-5973-3 [DOI] [PubMed] [Google Scholar]
- 70.Liu B, Liu S, Wang Y, et al. Neurosurgical enhanced recovery after surgery (ERAS) programme for elective craniotomies: are patients satisfied with their experiences: a quantitative and qualitative analysis. BMJ Open. 2019;9(11):e028706. doi: 10.1136/bmjopen-2018-028706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Liu B, Liu S, Wang Y, et al. Enhanced recovery after intraspinal tumor surgery: a single-institutional randomized controlled study. World Neurosurg. 2020;136(101528275):e542-e552. doi: 10.1016/j.wneu.2020.01.067 [DOI] [PubMed] [Google Scholar]
- 72.Liu G, Jian F, Wang X, Chen L. Fast-track surgery protocol in elderly patients undergoing laparoscopic radical gastrectomy for gastric cancer: a randomized controlled trial. Onco Targets Ther. 2016;9(101514322):3345-3351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mari G, Costanzi A, Crippa J, Falbo R, Miranda A, Rossi M, et al. Surgical stress reduction in elderly patients undergoing elective colorectal laparoscopic surgery within an ERAS protocol. Chirurgia (Bucur). 2016;111(6):476-480. doi: 10.21614/chirurgia.111.6.476 [DOI] [PubMed] [Google Scholar]
- 74.Mari GM, Costanzi A, Maggioni D, et al. Fast-track versus standard care in laparoscopic high anterior resection: a prospective randomized-controlled trial. Surg Laparosc Endosc Percutan Tech. 2014;24(2):118-121. doi: 10.1097/SLE.0b013e3182a50e3a [DOI] [PubMed] [Google Scholar]
- 75.Mingjie X, Luyao Z, Ze T, YinQuan Z, Quan W. Laparoscopic radical gastrectomy for resectable advanced gastric cancer within enhanced recovery programs: a prospective randomized controlled trial. J Laparoendosc Adv Surg Tech A. 2017;27(9):959-964. doi: 10.1089/lap.2016.0057 [DOI] [PubMed] [Google Scholar]
- 76.Mohsina S, Shanmugam D, Sureshkumar S, Kundra P, Mahalakshmy T, Kate V. Adapted ERAS pathway vs. standard care in patients with perforated duodenal ulcer-a randomized controlled trial. J Gastrointest Surg. 2018;22(1):107-116. doi: 10.1007/s11605-017-3474-2 [DOI] [PubMed] [Google Scholar]
- 77.Muller S, Zalunardo MP, Hubner M, Clavien PA, Demartines N; Zurich Fast Track Study Group . A fast-track program reduces complications and length of hospital stay after open colonic surgery. Gastroenterology. 2009;136(3):842-847. doi: 10.1053/j.gastro.2008.10.030 [DOI] [PubMed] [Google Scholar]
- 78.Nakanishi W, Miyagi S, Tokodai K, et al. Effect of enhanced recovery after surgery protocol on recovery after open hepatectomy: a randomized clinical trial. Ann Surg Treat Res. 2020;99(6):320-328. doi: 10.4174/astr.2020.99.6.320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Nunez-Venzor A, Trejo-Avila M, Cardenas-Lailson E, Valenzuela-Salazar C, Herrera-Esquivel J, Moreno-Portillo M. Effect of enhanced recovery after surgery on laparoscopic appendectomy outcomes in patients with complicated appendicitis: a randomized controlled trial. SN Compr Clin Med. 2021;3(2):646-653. doi: 10.1007/s42399-021-00788-5 [DOI] [Google Scholar]
- 80.Ostermann S, Morel P, Chalé JJ, et al. Randomized controlled trial of enhanced recovery program dedicated to elderly patients after colorectal surgery. Dis Colon Rectum. 2019;62(9):1105-1116. doi: 10.1097/DCR.0000000000001442 [DOI] [PubMed] [Google Scholar]
- 81.Pan X, Ma Y, Fang M, Jiang J, Shen J, Zhan R. Improvement in the quality of early postoperative course after endoscopic transsphenoidal pituitary surgery: description of surgical technique and outcome. Front Neurol. 2020;11(101546899):527323. doi: 10.3389/fneur.2020.527323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Peng J, Dong R, Jiao J, et al. Enhanced recovery after surgery impact on the systemic inflammatory response of patients following gynecological oncology surgery: a prospective randomized study. Cancer Manag Res. 2021;13(101512700):4383-4392. doi: 10.2147/CMAR.S294718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pirzada MT, Naseer F, Haider R, et al. Enhanced recovery after surgery (ERAS) protocol in stoma reversals. J Pak Med Assoc. 2017;67(11):1674-1678. [PubMed] [Google Scholar]
- 84.Prabhakaran S, Misra S, Magila M, et al. Randomized controlled trial comparing the outcomes of enhanced recovery after surgery and standard recovery pathways in laparoscopic sleeve gastrectomy. Obes Surg. 2020;30(9):3273-3279. doi: 10.1007/s11695-020-04585-2 [DOI] [PubMed] [Google Scholar]
- 85.Qu L, Liu B, Zhang H, et al. Management of postoperative pain after elective craniotomy: a prospective randomized controlled trial of a neurosurgical enhanced recovery after surgery (ERAS) program. Int J Med Sci. 2020;17(11):1541-1549. doi: 10.7150/ijms.46403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rao JH, Zhang F, Lu H, et al. Effects of multimodal fast-track surgery on liver transplantation outcomes. Hepatobiliary Pancreat Dis Int. 2017;16(4):364-369. doi: 10.1016/S1499-3872(17)60020-1 [DOI] [PubMed] [Google Scholar]
- 87.Ren L, Zhu D, Wei Y, et al. Enhanced recovery after surgery (ERAS) program attenuates stress and accelerates recovery in patients after radical resection for colorectal cancer: a prospective randomized controlled trial. World J Surg. 2012;36(2):407-414. doi: 10.1007/s00268-011-1348-4 [DOI] [PubMed] [Google Scholar]
- 88.Ruiz-Tovar J, Garcia A, Ferrigni C, Gonzalez J, Castellon C, Duran M. Impact of implementation of an enhanced recovery after surgery (ERAS) program in laparoscopic Roux-en-Y gastric bypass: a prospective randomized clinical trial. Surg Obes Relat Dis. 2019;15(2):228-235. doi: 10.1016/j.soard.2018.11.002 [DOI] [PubMed] [Google Scholar]
- 89.Sanchez-Iglesias JL, Carbonell-Socias M, Perez-Benavente MA, Monreal Clua S, Manrique-Munoz S, Garcia Gorriz M, et al. PROFAST: a randomised trial implementing enhanced recovery after surgery for highcomplexity advanced ovarian cancer surgery. Eur J Cancer. 2020;136:149-158. doi: 10.1016/j.ejca.2020.06.011 [DOI] [PubMed] [Google Scholar]
- 90.Sanchez-Iglesias JL, Gomez-Hidalgo NR, Perez-Benavente A, Carbonell-Socias M, Manrique-Munoz S, Serrano MP, et al. Importance of enhanced recovery after surgery (ERAS) protocol compliance for length of stay in ovarian cancer surgery. Ann Surg Onc. 2021;28(13):8979-8986. doi: 10.1245/s10434-021-10228-2 [DOI] [PubMed] [Google Scholar]
- 91.Saurabh K, Sureshkumar S, Mohsina S, Mahalakshmy T, Kundra P, Kate V. Adapted ERAS pathway versus standard care in patients undergoing emergency small bowel surgery: a randomized controlled trial. J Gastrointest Surg. 2020;24(9):2077-2087. doi: 10.1007/s11605-020-04684-6 [DOI] [PubMed] [Google Scholar]
- 92.Sazhin AV, Nechay TV, Titkova SM, et al. Fast-track rehabilitation for acute cholecystitis: intermediate outcomes of prospective randomized trial. Khirurgiia (Mosk). 2018;(12):13-20. doi: 10.17116/hirurgia201812113 [DOI] [PubMed] [Google Scholar]
- 93.Sazhin AV, Nechay TV, Titkova SM, et al. Comparison of standard and fast track rehabilitation in patients with acute appendicitis: intermediate results of multicenter prospective randomized trial. Khirurgiia (Mosk). 2018;(9):15-23. doi: 10.17116/hirurgia2018090115 [DOI] [PubMed] [Google Scholar]
- 94.Scioscia M, Ceccaroni M, Gentile I, et al. Randomized trial on fast track care in colorectal surgery for deep infiltrating endometriosis. J Minim Invasive Gynecol. 2017;24(5):815-821. doi: 10.1016/j.jmig.2017.04.004 [DOI] [PubMed] [Google Scholar]
- 95.Serclova Z, Dytrych P, Marvan J, Nova K, Hankeova Z, Ryska O, et al. Fast-track in open intestinal surgery: prospective randomized study. Clin Nutr. 2009;28(6):618-624. [DOI] [PubMed] [Google Scholar]
- 96.Serclova Z, Dytrych P, Marvan J, Nova K, Hankeova Z, Ryska O, et al. Tolerance of accelerated postoperative rehabilitation following intestinal resections. Rozhl Chir. 2009;88(4):178-184. [PubMed] [Google Scholar]
- 97.Sharma J, Kumar N, Huda F, Payal YS. Enhanced recovery after surgery protocol in emergency laparotomy: a randomized control study. Surg J (N Y). 2021;7(2):e92-e99. doi: 10.1055/s-0041-1725156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Shetiwy M, Fady T, Shahatto F, Setit A. Standardizing the protocols for enhanced recovery from colorectal cancer surgery: are we a step closer to ideal recovery? Ann Coloproctol. 2017;33(3):86-92. doi: 10.3393/ac.2017.33.3.86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Soffin EM, Beckman JD, Tseng A, et al. Enhanced recovery after lumbar spine fusion: a randomized controlled trial to assess the quality of patient recovery. Anesthesiology. 2020;133(2):350-363. doi: 10.1097/ALN.0000000000003346 [DOI] [PubMed] [Google Scholar]
- 100.Swaminathan N, Kundra P, Ravi R, Kate V. ERAS protocol with respiratory prehabilitation versus conventional perioperative protocol in elective gastrectomy- a randomized controlled trial. Int J Surg. 2020;81:149-157. doi: 10.1016/j.ijsu.2020.07.027 [DOI] [PubMed] [Google Scholar]
- 101.Takagi K, Yoshida R, Yagi T, Umeda Y, Nobuoka D, Kuise T, et al. Effect of an enhanced recovery after surgery protocol in patients undergoing pancreaticoduodenectomy: a randomized controlled trial. Clin Nutr. 2019;38(1):174-181. doi: 10.1016/j.clnu.2018.01.002 [DOI] [PubMed] [Google Scholar]
- 102.Tanaka R, Lee S-W, Kawai M, Tashiro K, Kawashima S, Kagota S, et al. Protocol for enhanced recovery after surgery improves short-term outcomes for patients with gastric cancer: a randomized clinical trial. Gastric Cancer. 2017;20(5):861-871. doi: 10.1007/s10120-016-0686-1 [DOI] [PubMed] [Google Scholar]
- 103.Taupyk Y, Cao X, Zhao Y, Wang C, Wang Q. Fast-track laparoscopic surgery: a better option for treating colorectal cancer than conventional laparoscopic surgery. Oncol Lett. 2015;10(1):443-448. doi: 10.3892/ol.2015.3166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Teigen NC, Sahasrabudhe N, Doulaveris G, Xie X, Negassa A, Bernstein J, et al. Enhanced recovery after surgery at cesarean delivery to reduce postoperative length of stay: a randomized controlled trial. Am J Obstet Gynecol. 2020;222(4):372.e1-372.e10. doi: 10.1016/j.ajog.2019.10.009 [DOI] [PubMed] [Google Scholar]
- 105.Trejo-Ávila ME, Romero-Loera S, Cárdenas-Lailson E, et al. Enhanced recovery after surgery protocol allows ambulatory laparoscopic appendectomy in uncomplicated acute appendicitis: a prospective, randomized trial. Surg Endosc. 2019;33(2):429-436. doi: 10.1007/s00464-018-6315-9 [DOI] [PubMed] [Google Scholar]
- 106.Vlad O, Catalin B, Mihai H, et al. Enhanced recovery after surgery (ERAS) protocols in patients undergoing radical cystectomy with ileal urinary diversions: A randomized controlled trial. Medicine (Baltimore). 2020;99(27):e20902. doi: 10.1097/MD.0000000000020902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Wang Y, Liu B, Zhao T, et al. Safety and efficacy of a novel neurosurgical enhanced recovery after surgery protocol for elective craniotomy: a prospective randomized controlled trial. J Neurosurg. 2018;130(5):1-12. [DOI] [PubMed] [Google Scholar]
- 108.Wu XF, Kong WF, Wang WH, et al. Enhanced recovery after surgery protocols in functional endoscopic sinus surgery for patients with chronic rhinosinusitis with nasal polyps: a randomized clinical trial. Chin Med J (Engl). 2019;132(3):253-258. doi: 10.1097/CM9.0000000000000060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yang G, Chen W, Chen W, Tang X, Huang Y, Zhang L. Feasibility and safety of 2-day discharge after fast-track total hip arthroplasty: a Chinese experience. J Arthroplasty. 2016;31(8):1686-1692.e1. doi: 10.1016/j.arth.2016.02.011 [DOI] [PubMed] [Google Scholar]
- 110.Yilmaz G, Akça A, Aydin N. Enhanced recovery after surgery (ERAS) versus conventional postoperative care in patients undergoing abdominal hysterectomies. Ginekol Pol. 2018;89(7):351-356. doi: 10.5603/GP.a2018.0060 [DOI] [PubMed] [Google Scholar]
- 111.Yilmaz G, Akca A, Kiyak H, Salihoglu Z. Comparison of enhanced recovery protocol with conventional care in patients undergoing minor gynecologic surgery. Wideochir Inne Tech Maloinwazyjne. 2020;15(1):220-226. doi: 10.5114/wiitm.2019.85464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zhao G, Cao S, Cui J. Fast-track surgery improves postoperative clinical recovery and reduces postoperative insulin resistance after esophagectomy for esophageal cancer. Support Care Cancer. 2014;22(2):351-358. doi: 10.1007/s00520-013-1979-0 [DOI] [PubMed] [Google Scholar]
- 113.Pan J, Hei Z, Li L, et al. The advantage of implementation of enhanced recovery after surgery (ERAS) in acute pain management during elective cesarean delivery: a prospective randomized controlled trial. Ther Clin Risk Manag. 2020;16(101253281):369-378. doi: 10.2147/TCRM.S244039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Liu XX, Pan HF, Jiang ZW, et al. Fast-track and minimally invasive surgery for gastric cancer. Chin Med J (Engl). 2016;129(19):2294-2300. doi: 10.4103/0366-6999.190659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Pang Q, Duan L, Jiang Y, Liu H. Oncologic and long-term outcomes of enhanced recovery after surgery in cancer surgeries: a systematic review. World J Surg Oncol. 2021;19(1):191. doi: 10.1186/s12957-021-02306-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50. doi: 10.1186/1748-5908-4-50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Kang Y, Liu J, Chen H, et al. Enhanced recovery after surgery (ERAS) in elective intertrochanteric fracture patients result in reduced length of hospital stay (LOS) without compromising functional outcome. J Orthop Surg Res. 2019;14(1):209. doi: 10.1186/s13018-019-1238-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wei JJ, Lin EX, Shi JD, et al. Meta-analysis with zero-event studies: a comparative study with application to COVID-19 data. Mil Med Res. 2021;8(1):41. doi: 10.1186/s40779-021-00331-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Fearon KC, Ljungqvist O, Von Meyenfeldt M, et al. Enhanced recovery after surgery: a consensus review of clinical care for patients undergoing colonic resection. Clin Nutr. 2005;24(3):466-477. doi: 10.1016/j.clnu.2005.02.002 [DOI] [PubMed] [Google Scholar]
- 120.Engelman RM, Rousou JA, Flack JE III, et al. Fast-track recovery of the coronary bypass patient. Ann Thorac Surg. 1994;58(6):1742-1746. doi: 10.1016/0003-4975(94)91674-8 [DOI] [PubMed] [Google Scholar]
- 121.Bardram L, Funch-Jensen P, Jensen P, Crawford ME, Kehlet H. Recovery after laparoscopic colonic surgery with epidural analgesia, and early oral nutrition and mobilisation. Lancet. 1995;345(8952):763-764. doi: 10.1016/S0140-6736(95)90643-6 [DOI] [PubMed] [Google Scholar]
- 122.Møiniche S, Bülow S, Hesselfeldt P, Hestbaek A, Kehlet H. Convalescence and hospital stay after colonic surgery with balanced analgesia, early oral feeding, and enforced mobilisation. Eur J Surg. 1995;161(4):283-288. [PubMed] [Google Scholar]
- 123.ERAS Compliance Group . The impact of enhanced recovery protocol compliance on elective colorectal cancer resection: results from an international registry. Ann Surg. 2015;261(6):1153-1159. doi: 10.1097/SLA.0000000000001029 [DOI] [PubMed] [Google Scholar]
- 124.Wijk L, Udumyan R, Pache B, Altman AD, Williams LL, Elias KM, et al. International validation of enhanced recovery after surgery society guidelines on enhanced recovery for gynecologic surgery. Am J Obstet Gynecol. 2019;221(3):237.e1-237.e11. doi: 10.1097/SGA.0000000000000417 [DOI] [PubMed] [Google Scholar]
- 125.Oodit R, Biccard B, Brindle M, Ljungqvist O, Nelson G. ERAS Society recommendations for improving perioperative care in low- and middle-income countries through implementation of existing tools and programs: an urgent need for the surgical safety checklist and enhanced recovery after surgery. World J Surg. 2022;46(5):1247. doi: 10.1007/s00268-022-06450-y [DOI] [PubMed] [Google Scholar]
- 126.Ljungqvist O, de Boer HD, Balfour A, et al. Opportunities and challenges for the next phase of enhanced recovery after surgery: a review. JAMA Surg. 2021;156(8):775-784. doi: 10.1001/jamasurg.2021.0586 [DOI] [PubMed] [Google Scholar]
- 127.Grimshaw JM, Thomas RE, MacLennan G, et al. Effectiveness and efficiency of guideline dissemination and implementation strategies. Health Technol Assess. 2004;8(6):iii-iv, 1-72. doi: 10.3310/hta8060 [DOI] [PubMed] [Google Scholar]
- 128.Peters S, Sukumar K, Blanchard S, et al. Trends in guideline implementation: an updated scoping review. Implement Sci. 2022;17(1):50. doi: 10.1186/s13012-022-01223-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Gustafsson UO, Oppelstrup H, Thorell A, Nygren J, Ljungqvist O. Adherence to the ERAS protocol is associated with 5-year survival after colorectal cancer surgery: a retrospective cohort study. World J Surg. 2016;40(7):1741-1747. doi: 10.1007/s00268-016-3460-y [DOI] [PubMed] [Google Scholar]
- 130.Twomey R, Matthews TW, Nakoneshny S, et al. Impact of early mobilization on recovery after major head and neck surgery with free flap reconstruction. Cancers (Basel). 2021;13(12):2852. doi: 10.3390/cancers13122852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Twomey R, Matthews TW, Nakoneshny SC, et al. From pathways to practice: impact of implementing mobilization recommendations in head and neck cancer surgery with free flap reconstruction. Cancers (Basel). 2021;13(12):2890. doi: 10.3390/cancers13122890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Francis NK, Walker T, Carter F, et al. Consensus on training and implementation of enhanced recovery after surgery: a Delphi study. World J Surg. 2018;42(7):1919-1928. doi: 10.1007/s00268-017-4436-2 [DOI] [PubMed] [Google Scholar]
- 133.Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines: a framework for improvement. JAMA. 1999;282(15):1458-1465. doi: 10.1001/jama.282.15.1458 [DOI] [PubMed] [Google Scholar]
- 134.Farquhar CM, Kofa EW, Slutsky JR. Clinicians’ attitudes to clinical practice guidelines: a systematic review. Med J Aust. 2002;177(9):502-506. doi: 10.5694/j.1326-5377.2002.tb04920.x [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eAppendix 1. Search Strategy
eAppendix 2. Data Dictionary
eTable 1. Characteristics of Included Studies
eTable 2. Risk of Bias
eFigure. PRISMA Flow Diagram
Data Sharing Statement