Abstract
In humans, maternal antibodies inhibit successful immunization against measles, because they interfere with vaccine-induced seroconversion. We have investigated this problem using the cotton rat model (Sigmodon hispidus). As in humans, passively transferred antibodies inhibit the induction of measles virus (MV)-neutralizing antibodies and protection after immunization with MV. In contrast, a recombinant vesicular stomatitis virus (VSV) expressing the MV hemagglutinin (VSV-H) induces high titers of neutralizing antibodies to MV in the presence of MV-specific antibodies. The induction of neutralizing antibodies increased with increasing virus dose, and all doses gave good protection from subsequent challenge with MV. Induction of antibodies by VSV-H was observed in the presence of passively transferred human or cotton rat antibodies, which were used as the models of maternal antibodies. Because MV hemagglutinin is not a functional part of the VSV-H envelope, MV-specific antibodies only slightly inhibit VSV-H replication in vitro. This dissociation of function and antigenicity is probably key to the induction of a neutralizing antibody in the presence of a maternal antibody.
Measles virus (MV) is the single most important cause of infant mortality worldwide. Vaccination with an attenuated live virus vaccine has proven to induce protective immunity in seronegative individuals, and even low titers of neutralizing antibodies seem to be protective (4, 12). In developing countries with a high level of infection, infants below the age of 12 months are at high risk for MV infection. In this age group passively transferred maternal immunoglobulins (Ig) pose a problem because declining maternal antibodies interfere with vaccine-induced seroconversion but do not protect against infection with wild-type MV (13, 15). To induce immunity in the presence of maternal antibodies, high-titer vaccines (>104.7 PFU) were administered to infants at the age of 4 to 6 months (1, 17). These infants showed good serological responses and protection against measles. However, especially in female children, an increased mortality due to infections other than measles was observed after immunization with high-titer vaccines (2, 7), and the use of this vaccine was therefore discontinued.
In order to develop vaccine alternatives which induce MV-neutralizing antibodies in the presence of maternal antibodies, we have used MV infection in the cotton rat model (Sigmodon hispidus, inbred strain Cotton NIco) (9). Cotton rats are the only rodents in which MV replicates in the respiratory tract (18). Here we demonstrate that the passive transfer of human serum containing MV-specific antibodies inhibits vaccine-induced seroconversion and abolishes protection against MV. To induce neutralizing antibodies in the presence of MV-specific antibodies, we tested a recombinant vesicular stomatitis virus (VSV) expressing the MV hemagglutinin (VSV-H) (14). VSV is known for the rapid induction of neutralizing antibodies against its surface protein G, and VSV recombinants expressing influenza virus hemagglutinin induce high neutralizing antibody titers to influenza virus in mice (10, 11). In the recombinant VSV-H, the MV hemagglutinin is incorporated into the bullet-shaped envelope and comprises about one-fourth of the envelope proteins in the envelope but is not needed for replication. Using this vector we show here that intranasal (i.n.) but not intraperitoneal (i.p.) immunization led to the induction of MV-neutralizing antibodies in the presence of maternal antibodies.
MATERIALS AND METHODS
Cotton rats: infection, immunization, serum transfer, and virus titration.
Cotton rats (inbred strain Cotton/NIco) were obtained from Iffa Credo, Lyon, France. Animals were kept under controlled environmental conditions and used at the age of 6 to 8 weeks (60 to 70 g). The i.n. infection, i.p. infection or serum injection, and retro-orbital blood sampling were done under ether narcosis. To mimic maternal MV-specific antibodies, 1 ml of a human serum (antibody concentration of 16 IU/ml by enzyme-linked immunosorbent assay [ELISA]; antibody titer of 320 by neutralization (NT) assay and 256 by hemagglutination inhibition assay) was used. For challenge experiments, 4 days after i.n. infection with 5 × 105 PFU of MV HU2 strain in a volume of 50 to 100 μl, animals were asphyxiated using CO2, lungs were removed, and the 50% tissue culture infectious dose (TCID50) was determined as described previously (9).
Viruses.
Recombinant VSV and VSV-H (14) were grown and titrated on baby hamster kidney (BHK) cells, and MV strains Edmonston B and HU2 were grown and titrated on Vero cells according to standard procedures (9, 14).
ELISA.
ELISAs were performed according to standard procedures. For ELISA 10 μg of gradient-purified, UV-inactivated MV/ml was coated in 200 mM NaCO3 buffer (pH 9.6) at 4°C overnight, blocked with phosphate-buffered saline–10% fetal calf serum–0.05% Tween 20, and incubated with dilutions of human serum at room temperature for 1 h. After being washed, the plate was incubated for 1 h at room temperature with a horseradish peroxidase-coupled rabbit serum specific for human IgG (Dako, Hamburg, Germany) and was subsequently developed with 0.5 mg of ortho-phenyldiamine/ml in buffer (35 mM citrate, 66 mM Na2HPO4 [pH 5,2])–0.01% H2O2. Human sera were standardized using human anti-MV serum (2nd international standard 1990; 5 IU per ml; National Institute for Biological Standards and Control, Potters Bar, United Kingdom).
To test for MV-specific cotton rat IgG, coated plates were incubated with dilutions of cotton rat serum at 4°C for 1 h. After being washed, the plate was incubated with rabbit serum specific for cotton rat IgG (Virion Systems, Rockville, Md.) for 1 h at room temperature. After being washed, the plate was incubated with horseradish peroxidase-coupled goat serum specific for rabbit IgG (Zymed, San Francisco, Calif.) for 45 min at room temperature and developed as described above.
NT and PRNT assays. (i) NT assay.
Serum dilutions were incubated with 50 PFU of MV Edmonston strain or recombinant VSV or VSV-H for 1 h at 37°C and plated in duplicate onto 104 Vero cells (MV) or BHK cells (VSV) per well of a 96-well plate. Two (VSV) or 5 days (MV) later titers were determined microscopically.
(ii) Plaque reduction neutralization (PRNT) assay.
Serum dilutions were incubated with 100 PFU of MV Edmonston strain for 1 h at 37°C and plated onto Vero cells (80 to 90% density) in a six-well plate. Cells were overlayed with agar, and after 5 days the incubation mixture was stained with neutral red (Merck) solution (0.6% in phosphate-buffered saline) and the plaques were counted.
RESULTS
Both cotton rat and human MV-specific antibodies inhibit vaccine-induced seroconversion in cotton rats.
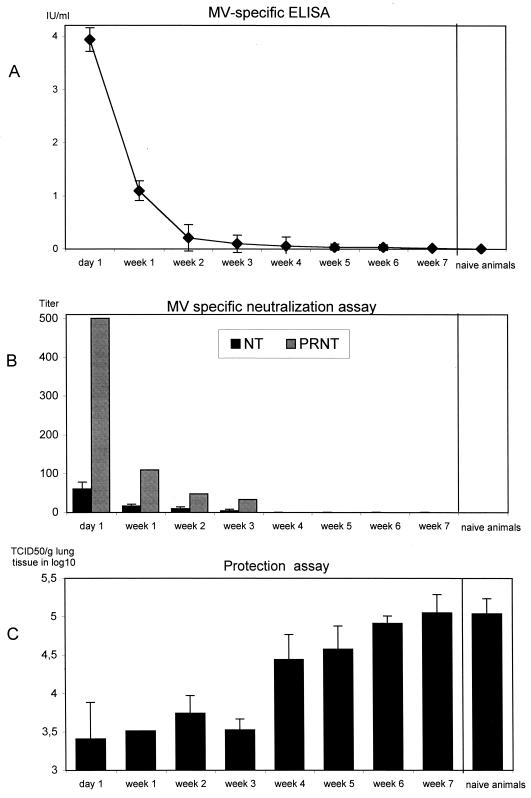
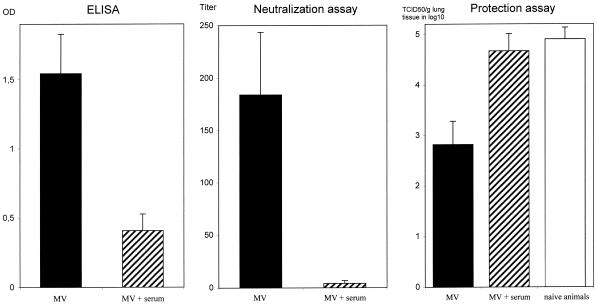
Weanling cotton rats from MV-immune dams have high serum titers of both MV-neutralizing antibodies (NT assay titer, 180 ± 60) and total MV-specific antibodies (optical density, 2.1 ± 0.04) at 3 weeks after birth. As in humans, antibody titers decline over time, and no neutralizing antibodies were detectable after 8 weeks. At 3 weeks after birth, animals with high titers of maternal antibodies were immunized i.p. with the MV Edmonston vaccine strain. Seven weeks later no neutralizing antibodies were detectable (data not shown). In contrast, vaccinated animals lacking maternal antibodies developed antibody titers that were detectable for at least 10 months. Although the protection by maternal antibodies is similar to the situation in humans, it is difficult in our model system to distinguish between actively induced antibodies and passively transferred maternal antibodies. Because human and cotton rat antibodies can be distinguished by ELISA, we chose in all initial experiments to use an MV-specific human serum (containing 16 IU/ml as defined by comparison to a World Health Organization standard serum) to mimic maternal antibodies. After transfer of 1 ml (16 IU of antibodies) of human serum into cotton rats, MV-specific antibodies declined over 6 weeks (Fig. 1a). By using the sensitive PRNT assay, neutralizing antibodies were detected for 3 weeks after serum transfer (Fig. 1b). Some of the animals given the neutralizing antibody were also challenged with MV at the time points indicated (Fig. 1c). The presence of antibodies correlated with greater-than-10-fold reductions in viral titers in lungs of infected animals for the first 3 weeks. With declining antibody titers, protection from MV infection declined also and was gone by week 7. As in humans, the NT assay was relatively insensitive compared to the PRNT assay and ELISA and significant reduction in virus replication was still seen when antibody titers were below the threshold of detection. We also measured the inhibition of measles vaccine-induced seroconversion by MV-specific antibodies. One day after serum transfer animals were immunized with 105 PFU of MV i.p., which induced high titers of MV-specific and neutralizing antibodies (Fig. 2).. In the presence of transferred human MV-specific antibodies, however, the generation of total MV-specific antibody titers was reduced more than fourfold and neutralizing antibody titers were abrogated (Fig. 2). In consequence, subsequent protection in these animals was severely reduced compared to that in animals immunized in the absence of MV-specific antibodies (Fig. 2).
FIG. 1.
MV-specific human antibodies mimic maternal antibodies and decline after transfer into cotton rats. One milliliter of human serum containing MV-specific antibodies (16 IU/ml) was transferred into naive cotton rats (60 to 70 g). One day later and at weekly intervals from weeks 1 to 7, human MV-specific antibody titers (six animals per time point) were measured by ELISA (a) and virus NT and PRNT assays (b). At the same time points animals were challenged i.n. with MV, and virus titers (TCID50/g of lung tissue ± standard deviation) were determined by titration 4 days later (four animals per time point, except week 1 [one animal]) (c). By NT assay neutralizing antibodies can be demonstrated until week 2, by the more-sensitive PRNT assay they can be demonstrated until week 3, and by ELISA they can be demonstrated until week 6 (26 mIU/ml). In the presence of neutralizing antibodies the virus reduction is more than 10-fold and declines from week 4.
FIG. 2.
MV-specific human antibodies inhibit vaccine-induced seroconversion and protection in cotton rats. One milliliter of human serum containing MV-specific antibodies (16 IU/ml) was transferred into naive cotton rats. One day later animals were immunized i.p. with 105 PFU of MV and challenged after 6 weeks with MV i.n. The total MV-specific antibodies were measured by ELISA (1:100 dilution), and the neutralizing antibodies were measured by NT assay. Virus titers in lung tissue (± standard deviations) were determined on day 5 after i.n. infection. Data represent the averages for five animals. The difference in protection was significant between groups with and without serum transfer (P < 0.0003; two-sided paired t test). OD, optical density.
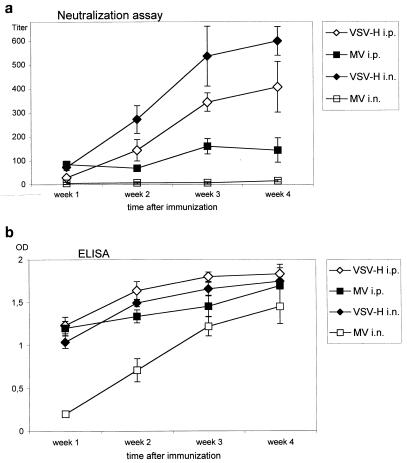
The i.n. immunization with VSV-H results in more-rapid generation of neutralizing antibodies and higher titers than does immunization with MV.
Recombinant VSV-H (14) was chosen to be tested for the induction of MV-neutralizing antibodies for the following reasons: the MV hemagglutinin is the most important antigen for the induction of neutralizing antibodies (6), VSV is known for rapid and efficient induction of neutralizing antibody responses directed against its own surface G protein (3), and MV hemagglutinin comprises 25% of the total envelope protein in the recombinant VSV-H (14). First, the ability of VSV-H to induce neutralizing antibodies in comparison to MV was tested after i.p. and i.n. immunization. In comparison with the MV Edmonston strain, VSV-H induced higher titers of neutralizing and total MV-specific antibodies (Fig. 3). The difference was especially striking after i.n. immunization, indicating that VSV-H is a vector well suited for i.n. immunization.
FIG. 3.
VSV-H induces higher titers of neutralizing antibodies than MV. After inoculation of 5 × 106 PFU of VSV-H or MV i.p. or i.n., MV-specific antibodies were measured by NT assay (a) and by ELISA (1:100 dilution; b) in weekly intervals from week 1 to 4 after immunization. All data represent the means for five animals ± standard deviations. Four weeks after i.n. immunization with MV, the neutralizing antibody titer, 15, was above the threshold level of detection (>10). The percentage of MV-specific IgA antibodies in whole serum remained constant irrespective of the virus or route of immunization used. OD, optical density.
MV-specific antibodies do not inhibit VSV-H replication in vitro.
To induce MV-neutralizing antibodies in vivo, in the presence of MV-specific antibodies, VSV-H would have to escape neutralization by MV-specific antibodies. Although MV hemagglutinin is expressed in considerable quantities as part of the envelope of VSV-H, it is a passenger protein with no function in viral replication. We assumed that replication of VSV-H might therefore not be inhibited by MV-specific antibodies to the same degree as MV replication. In vitro, serum containing antibodies against glycoprotein G of VSV effectively neutralized VSV-H (data not shown). Human and cotton rat sera containing high titers of MV-neutralizing antibodies (1:640) neutralized MV effectively, but only very high serum concentrations neutralized VSV-H (1:20). Thus VSV-H effectively escapes neutralization by antibody concentrations that completely neutralize MV in vitro.
VSV-H induces MV-neutralizing antibodies in the presence of MV-specific antibodies.
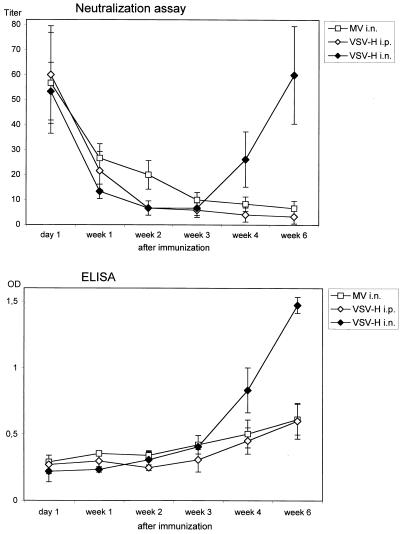
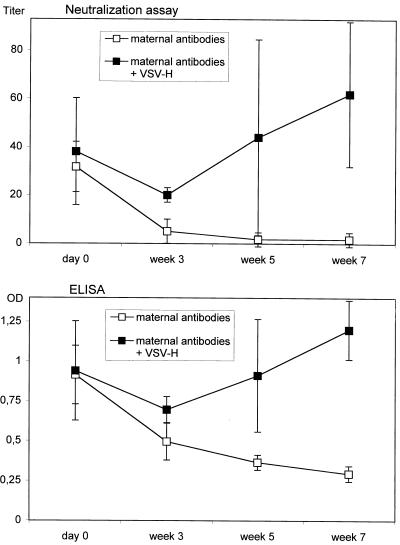
To test whether VSV-H is able to replicate in the presence of MV-specific antibodies in vivo, cotton rats were immunized with VSV-H either i.p. or i.n. or with MV i.n. 1 day after serum transfer. After i.p. immunization with VSV-H or i.n. immunization with MV, antibody production was severely suppressed as measured by NT assay and ELISA (Fig. 4) compared to immunization in the absence of MV-specific antibodies (Fig. 3). In contrast, after i.n. immunization with VSV-H, total MV-specific as well as neutralizing antibody production started after 4 weeks. This effect was dose dependent. An inoculum of 5 × 105 PFU of VSV-H overcame MV-specific antibodies (NT assay titer, 17.5), but a 40-fold increase in the virus inoculum gave approximately 3-fold-increased titers of neutralizing antibodies (Table 1). Viral replication was shown to be critical for antibody induction, as UV-inactivated VSV-H was not able to induce neutralizing antibodies (data not shown). Although the titers of neutralizing antibodies in animals without serum transfer were higher, no difference in protection between animals with and without serum transfer was seen (Table 1).
FIG. 4.
Immunization via the i.n. route with VSV-H overcomes maternal antibodies. After transfer of 16 IU of MV-specific human antibodies into cotton rats, animals were immunized 1 day later with 2 × 107 PFU of MV i.n. (three animals) or 2 × 107 PFU of VSV-H i.n. (four animals per group) or 5 × 107 PFU of VSV-H i.p. (four animals per group). On the day of immunization (day 1) and after 1, 2, 3, 4, and 6 weeks serum samples were obtained and tested for the presence of neutralizing antibodies by NT assay (top) and of MV-specific cotton rat antibodies by ELISA (bottom). After 3 weeks neutralizing antibodies fell below the level of detection indicating that neutralizing antibodies from week 4 onwards were produced by cotton rats. This is confirmed by the ELISA data. OD, optical density.
TABLE 1.
Induction of MV-neutralizing antibodies and protection against MV-infection by VSV-H in the presence of MV-specific antibodiesa
| Immunization (PFU/virus) | Amt of human MV-specific antibodies (IU) | Result of:
|
Virus titer (log10 TCID50/g of lung tissue ± SD) | Virus reduction (%) | No. of animals | |
|---|---|---|---|---|---|---|
| ELISA (OD ± SD)b | NT assay (titer ± SD) | |||||
| None | 0.2 ± 0.05 | <10 | 4.3 ± 0.22 | 4 | ||
| None | 16 | 0.3 ± 0.02 | <10 | 4.3 ± 0.25 | 4 | |
| 5 × 107/WTc VSV | 0.3 ± 0.04 | <10 | 4.3 ± 0.10 | 3 | ||
| 5 × 107/VSV-H | 1.6 ± 0.1 | 320 ± 69 | 3.1 ± 0.14 | 93 | 3 | |
| 5 × 105/VSV-H | 16 | 1.1 ± 0.2 | 17.5 ± 3.5 | 3.6 ± 0.04 | 76 | 2 |
| 5 × 106/VSV-H | 16 | 1.2 ± 0.1 | 20 ± 0.0 | 3.6 ± 0.26 | 79 | 2 |
| 2 × 107/VSV-H | 16 | 1.3 ± 0.1 | 60 ± 16.3 | 3.2 ± 0.27 | 92 | 4 |
| 5 × 107/VSV-H | 16 | 1.5 ± 0.1 | 60 ± 20 | 3.0 ± 0.14 | 94 | 4 |
After transfer of 16 IU into cotton rats, animals were immunized with increasing doses of VSV-H. After 7 weeks serum samples were taken and analysed by ELISA and NT assay. In addition, animals were challenged i.n. with MV, and virus titers in lung tissue were determined 4 days later. Although immunization with 2 × 107 and 5 × 107 PFU of VSV-H in the presence of MV-specific antibodies led to a reduction in neutralizing antibody titers compared to immunization in the absence of MV-specific antibodies, levels of protection were comparable.
OD, optical density.
WT, wild type.
Interestingly, serum transfer of MV-specific antibodies inhibited the generation of hemagglutinin-specific antibodies, as the titers of VSV-neutralizing antibodies were the same in both groups.
Immunization with VSV-H also overcomes maternal (MV-specific) cotton rat antibodies.
We performed an experiment to determine if VSV-H was able to overcome homologous cotton rat anti-MV antibodies in addition to human MV-specific antibodies. Cotton rats from MV-immune dams with high titers of maternally derived MV-specific antibodies were given VSV-H i.n. As shown in Fig. 5 an increase in anti-MV antibodies was detectable by NT assay and by ELISA in the vaccinated animals after 3 to 7 weeks, while the maternal antibodies declined in the control animals.
FIG. 5.
Induction of neutralizing antibodies by VSV-H in the presence of maternal (cotton rat) antibodies. Five-week-old cotton rats from MV-immune dams were immunized with 5 × 106 PFU of VSV-H (four animals) or left untreated (three animals) (day 0). At various time points MV-specific antibody titers were tested by NT assay (top) and ELISA (bottom). OD, optical density.
DISCUSSION
Maternal antibodies are an early line of specific immune defense to protect the newborn against bacterial and viral pathogens which have been encountered by the mother. To immunize against measles in the presence of maternal antibodies has proven to be difficult because declining MV-specific maternal antibodies inhibit vaccine-induced seroconversion but do not confer protection against wild-type MV infection. In addition, maternal antibody titers differ between infants from immunized versus naturally infected mothers. In the latter, titers are higher and persist longer (8). Therefore it is desirable to be able to use a measles vaccine irrespective of the titer of maternal antibodies.
The cotton rat proved to be a suitable animal model to test vaccine candidates. Similar to the situation in human populations, the declining maternal MV-specific antibodies inhibit vaccine-induced seroconversion in cotton rats and protection against MV is severely reduced. In an experimental study with monkeys it was shown that a prime boost strategy (two immunizations with a 4-week interval) overcomes maternal antibodies (16). After immunization with MV or immune system-stimulating complexes containing the MV glycoproteins hemagglutinin and fusion protein, neutralizing antibodies were induced and protection against MV challenge was observed. However, repeated vaccinations are difficult in countries with migrating populations or when mass vaccination campaigns are undertaken. In humans, attempts have been made to immunize in the presence of maternal antibodies by a single aerosol dose of vaccine (for a review see reference 5) or by subcutaneous (s.c.) injection of a high-titer vaccine (2, 7). Whereas vaccination by aerosol has not given results superior to s.c. application, high-titer vaccines can achieve seroconversion in the presence of maternal antibodies and protect against measles. However, probably because of the marked immunosuppressive effect of this high-titer vaccine, a subsequent higher mortality due to secondary infections was noted.
We have chosen the highly attenuated recombinant VSV-H to immunize cotton rats against MV challenge (14). In cotton rats the attenuated recombinant VSV-H induces, after i.p. and especially after i.n. immunization, much higher titers of MV-neutralizing antibodies than MV itself. This response could be due to rapid replication and high-level MV hemagglutinin production in vivo by the VSV-H vector, but other factors may be involved also. From studies with mice it is known that VSV induces a T-cell-independent IgM response and high titers of neutralizing antibodies specific for the VSV G protein (3). This response may be due to the highly organized assembly of VSV G protein in the rigid bullet-shaped envelope. In contrast, MV has a very flexible pleomorphic envelope. The incorporation of MV hemagglutinin into the VSV envelope might therefore render MV hemagglutinin more antigenic. Immunization (i.n.) with VSV-H but not with MV induces neutralizing antibodies in the presence of MV-specific antibodies. VSV-H replicates for 5 days in lung tissue at lower titers than MV and does not spread into peripheral organs (data not shown). Immunization with UV-inactivated virus demonstrates that virus replication is essential to induce neutralizing antibodies in the presence of MV-specific antibodies. This is confirmed by the fact that after i.p. inoculation VSV-H replicates abortively (data not shown). MV hemagglutinin is part of the VSV-H envelope but has no function in viral replication (14). The ability of VSV-H to escape MV-specific antibodies in vitro is mirrored in the in vivo situation. These data indicate that a replication-competent vector is required to successfully immunize in the presence of maternal MV-specific antibodies. The strategy of having the appropriate antigen encoded as a passenger protein on the surface of a virus particle may be effective in other cases where a strong antibody response must be generated in the presence of maternal antibodies.
ACKNOWLEDGMENTS
This work was in part supported by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung, Wissenschaft, Forschung und Technologie, Pfleger-Stiftung, and by World Health Organization and National Institutes of Health grants to J.K.R.
REFERENCES
- 1.Aaby P, Jensen T, Hansen H. Trial of high dose Edmonston-Zagreb measles vaccine in Guinea-Bissau: protective efficacy. Lancet. 1988;ii:809–811. doi: 10.1016/s0140-6736(88)92780-8. [DOI] [PubMed] [Google Scholar]
- 2.Aaby P, Knudsen K, Whittle H, et al. Long-term survival after Edmonston-Zagreb measles vaccination in Guinea-Bissau: increased female mortality rate. J Pediatr. 1993;122:904–908. doi: 10.1016/s0022-3476(09)90015-4. [DOI] [PubMed] [Google Scholar]
- 3.Bachmann M F, Zinkernagel R M. Neutralizing antiviral B cell responses. Annu Rev Immunol. 1997;15:235–270. doi: 10.1146/annurev.immunol.15.1.235. [DOI] [PubMed] [Google Scholar]
- 4.Chen R T, Markowitz L E, Albrecht P, Stewart J A, Mofenson L M, Preblud S R, Orenstein W A. Measles antibody: reevaluation of protective titers. J Infect Dis. 1990;162:1036–1042. doi: 10.1093/infdis/162.5.1036. [DOI] [PubMed] [Google Scholar]
- 5.Clements C J, Cutts F T. The epidemiology of measles: thirty years of vaccination. Curr Top Microbiol Immunol. 1995;191:13–33. doi: 10.1007/978-3-642-78621-1_2. [DOI] [PubMed] [Google Scholar]
- 6.Griffin D E. Immune responses during measles virus infection. Curr Top Microbiol Immunol. 1995;191:117–134. doi: 10.1007/978-3-642-78621-1_8. [DOI] [PubMed] [Google Scholar]
- 7.Halsey N. Increased mortality following high titer measles vaccines: too much of a good thing. Pediatr Infect Dis J. 1993;12:462–465. doi: 10.1097/00006454-199306000-00002. [DOI] [PubMed] [Google Scholar]
- 8.Katz S L, Gellin B G. Measles vaccine: do we need new vaccines or new programs. Science. 1994;265:1391–1392. doi: 10.1126/science.8073281. [DOI] [PubMed] [Google Scholar]
- 9.Niewiesk S, Eisenhuth I, Fooks A, Clegg J C S, Schnorr J-J, Schneider-Schaulies S, ter Meulen V. Measles virus-induced immune suppression in the cotton rat (Sigmodon hispidus) model depends on viral glycoproteins. J Virol. 1997;71:7214–7219. doi: 10.1128/jvi.71.10.7214-7219.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roberts A J, Buoconore L, Price R, Forman J, Rose J K. Attenuated vesicular stomatitis viruses as vaccine vectors. J Virol. 1999;73:3723–3732. doi: 10.1128/jvi.73.5.3723-3732.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Roberts A J, Kretzschmar E, Perkins A S, Forman J, Price R, Buoconore L, Kawaoka Y, Rose J K. Vaccination with a recombinant vesicular stomatitis virus expressing an influenza virus hemagglutinin provides complete protection from influenza virus challenge. J Virol. 1998;72:4704–4711. doi: 10.1128/jvi.72.6.4704-4711.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Samb B, Aaby P, Whittle H C, Seck A M, Rahman S, Bennett J, Markowitz L, Simondon F. Serologic status and measles attack rates among vaccinated and unvaccinated children in rural Senegal. Pediatr Infect Dis J. 1995;14:203–209. doi: 10.1097/00006454-199503000-00007. [DOI] [PubMed] [Google Scholar]
- 13.Sato H, Albrecht P, Reynolds D W, Stagno S, Ennis F A. Transfer of measles, mumps and rubella antibodies from mother to infant. Its effect on measles, mumps and rubella immunization. Am J Dis Child. 1979;133:1240–1243. doi: 10.1001/archpedi.1979.02130120032005. [DOI] [PubMed] [Google Scholar]
- 14.Schnell M J, Buonocore L, Kretzschmar E, Johnson E, Rose J K. Foreign glycoproteins expressed from recombinant vesicular stomatitis viruses are incorporated efficiently into virus particles. Proc Natl Acad Sci USA. 1996;93:11359–11365. doi: 10.1073/pnas.93.21.11359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schoub B D, Johnson S, McAnerney J M, Wagstaff L A, Matsie W, Reinach S G, et al. Measles, mumps and rubella immunisation at nine months in a developing country. Ped Infect Dis J. 1990;9:263–267. doi: 10.1097/00006454-199004000-00008. [DOI] [PubMed] [Google Scholar]
- 16.van Binnendijk R S, Poelen M C M, van Amerongen G, de Vries P, Osterhaus A D M E. Protective immunity in macaaques vaccinated with live attenuated, recombinant, and subunit measles vaccines in the presence of passively acquired antibodies. J Infect Dis. 1997;175:524–532. doi: 10.1093/infdis/175.3.524. [DOI] [PubMed] [Google Scholar]
- 17.Whittle H, Hanlon P, O'Neil K, Hanlon L, Marsh V, Jupp E. Trial of high-dose Edmonston-Zagreb measles vaccine in Gambia: antibody response and side-effects. Lancet. 1988;ii:811–814. doi: 10.1016/s0140-6736(88)92781-x. [DOI] [PubMed] [Google Scholar]
- 18.Wyde P R, Ambrosi M W, Voss T G, Meyer H L, Gilbert B F. Measles virus replication in lungs of hispid cotton rats after intranasal inoculation. Proc Soc Exp Biol Med. 1992;201:80–87. doi: 10.3181/00379727-201-43483. [DOI] [PubMed] [Google Scholar]