Abstract
Introduction
Infections represent one of the most frequent causes of death of higher-risk MDS patients, as reported previously also by our group. Azacitidine Infection Risk Model (AIR), based on red blood cell (RBC) transfusion dependency, neutropenia <0.8 × 109/L, platelet count <50 × 109/L, albumin <35g/L, and ECOG performance status ≥2 has been proposed based on the retrospective data to estimate the risk of infection in azacitidine treated patients.
Methods
The prospective non-intervention study aimed to identify factors predisposing to infection, validate the AIR score, and assess the impact of antimicrobial prophylaxis on the outcome of azacitidine-treated MDS/AML and CMML patients.
Results
We collected data on 307 patients, 57.6 % males, treated with azacitidine: AML (37.8%), MDS (55.0%), and CMML (7.1%). The median age at azacitidine treatment commencement was 71 (range, 18-95) years. 200 (65%) patients were assigned to higher risk AIR group. Antibacterial, antifungal, and antiviral prophylaxis was used in 66.0%, 29.3%, and 25.7% of patients, respectively. In total, 169 infectious episodes (IE) were recorded in 118 (38.4%) patients within the first three azacitidine cycles. In a multivariate analysis ECOG status, RBC transfusion dependency, IPSS-R score, and CRP concentration were statistically significant for infection development (p < 0.05). The occurrence of infection within the first three azacitidine cycles was significantly higher in the higher risk AIR group – 47.0% than in lower risk 22.4% (odds ratio (OR) 3.06; 95% CI 1.82-5.30, p < 0.05). Administration of antimicrobial prophylaxis did not have a significant impact on all-infection occurrence in multivariate analysis: antibacterial prophylaxis (OR 0.93; 0.41-2.05, p = 0.87), antifungal OR 1.24 (0.54-2.85) (p = 0.59), antiviral OR 1.24 (0.53-2.82) (p = 0.60).
Discussion
The AIR Model effectively discriminates infection-risk patients during azacitidine treatment. Antimicrobial prophylaxis does not decrease the infection rate.
Keywords: azacitidine, infection, myelodysplastic syndromes, acute myeloid leukemia, chronic myelomonocytic leukemia, infection prevention
Introduction
Azacitidine, a hypomethylating agent, is the only drug able to improve overall survival in higher-risk myelodysplastic syndrome (MDS) patients, and hence, it is also the only drug that has been licensed for this indication in Europe since 2010. Following the licensing for MDS, azacitidine has been registered in acute myeloid leukemia (AML) based on its proven efficacy for this indication as well. The incidence of infections in MDS/AML patients treated with azacitidine is very high reaching, especially in real-world practice, up to 62%–75% (1, 2).Infections and transformation to AML represent the main causes of death of higher-risk MDS patients. That is why assessment of the risk of infection is crucial and several studies have aimed to identify factors predisposing to infection, but with divergent results. Recently, the Polish Adult Leukemia Group (PALG), in a retrospective study, has developed a model that stratifies infection risk. The model comprises neutrophil and platelet count, serum albumin level, red blood cell transfusion dependency, and Eastern Cooperative Oncology Group (ECOG) performance status (2). The present study is a prospective validation of the model. It also aims to identify other potential predictive markers.
Antibacterial and antifungal prophylaxis have been shown to reduce the risk of infection- and all-cause-related death incidence in neutropenic patients receiving intensive chemotherapy or allogeneic hematopoietic cell transplantation (allo-HCT) for AML. Therefore, antimicrobial prophylaxis is routinely administered when intensive chemotherapy protocols are used (3). In contrast, no convincing data justifies the administration of antimicrobial prophylaxis in hypomethylating agent-treated patients. Therefore, we aimed to also analyze the impact of antimicrobial agents on infection incidence.
Patients and methods
Patient population
Patients diagnosed with MDS, AML, or CMML according to World Health Organization (WHO) 2016 criteria and treated with 5-azacitidine from 1 January 2018 to 31 December 2022 were included and followed up to February 2023. Nine Polish hematology centers aligned with the PALG participated in this prospective non-interventional trial. The study was approved by each Institution’s Ethics Committee in accordance with national legislation and is conducted in accordance with local legal and regulatory requirements and ethical standards including the 1964 Helsinki Declaration and its later amendments to ensure the protection of the patient’s personal data.
Treatment
Patients received azacitidine dosed initially 75 mg/m2 subcutaneously for days 1 through 7 or in a “5–2-2” scheme with a 2-day rest over the weekend. Diagnosis of MDS, AML, or CMML was established based on the WHO 2016 classification (4).
Administration of antibacterial, antiviral, and antifungal prophylaxis was at clinician’s discretion based on center policy. Trimethoprim/sulfamethoxazole, levofloxacin, ciprofloxacin, and occasionally other antibiotics were given for antibacterial prophylaxis, while fluconazole, posaconazole, and voriconazole were prescribed as antifungals, and acyclovir was prescribed as antiviral prophylaxis.
Baseline patient demographics, laboratory data, comorbidities, gut colonization with multidrug-resistant bacteria (MRB), ECOG performance status, MDS-specific Comorbidity Index, BMI (body mass index), cigarette smoking, and transfusion dependency were determined for each patient. The International Prognostic Scoring System (IPSS) and IPSS-revised (IPSS-R) were applied to MDS, AML with 20%–30% marrow blasts, and CMML patients (5–7).
An infectious episode was defined as Grade ≥ III infection according to the Common Terminology Criteria for Adverse Events version 3.0—neutrophil count threshold was decreased from 1.0 × 109/L to 0.5 × 109/L for neutropenic fever; consequently, in our analyses, neutropenic fever was defined as neutropenia 38°C persisting for more than 1 h, or body temperature 38.3°C recorded on a single occasion (Freifeld 2010). Invasive fungal infections were defined according to European Organization for Research and Treatment of Cancer (EORTC)/National Institute of Allergy and Infectious Diseases Mycoses Study Group criteria (Pauw 2010). Infectious episodes were classified as microbiologically defined (if positive microbiological samples were available) and/or clinically defined (physician’s judgment based on clinical course, lab tests, imaging, and treatment outcome).
The Azacitidine Infectious Risk Model described in ACLM 2019 was applied to stratify patients. RBC transfusion dependency, neutropenia < 0.8 × 109/L, platelet count < 50 × 109/L, albumin level < 35 g/dL, and ECOG performance status ≥ 2 were assigned one point each, and the infection rate was finally determined for each total score grouping patients into two categories: lower (0–2 score) and higher early infection risk (3–5 score).
Statistical analysis
All analyses were conducted using statistical software R version 4.3 (R Foundation for Statistical Computing, Vienna, Austria). Survival curves were calculated with the Kaplan–Meier estimator, and hazard ratios were calculated with Cox proportional hazards model. Median survival was calculated based on Kaplan–Meier curves. p-values for group comparisons ( Table 1 ) were calculated with Pearson’s χ 2 test for categorical variables and Wilcoxon test for continuous variables. The predictive model was built using a two-step procedure. In the first step (variable filtering), pairwise relations between infections and other variables were calculated. Then, the most significant variables were converted into binary variables. Based on these variables, the predictive model was built using the logistic regression method.
Table 1.
Patients’ and azacitidine treatment characteristics.
| Characteristics | n (%) |
|---|---|
| Sex | |
| Male | 177 (57.6%) |
| Female | 130 (42.4%) |
| Age, median (range); years | 71 (18–95) |
| WHO 2016 diagnosis | |
| AML | 116 (37.8%) |
| MDS | 169 (55.0%) |
| CMML | 22 (7.1%) |
| IPSS | |
| Low | 1 (0.3%) |
| Int 1 | 16 (5.2%) |
| Int 2 | 102 (33.2%) |
| High | 89 (28.9%) |
| NA | 99 (33.2%) |
| IPSS-R | |
| Very Low | 0 (0.0%) |
| Low | 2 (0.6%) |
| Intermediate | 21 (6.8%) |
| High | 75 (24.4%) |
| Very High | 103 (33.5%) |
| NA | 106 (34.5%) |
| ECOG Performance Status | |
| 0–1 | 213 (69.3%) |
| 2–4 | 94 (30.7%) |
| MDS Comorbidity Index | |
| 0-1 2-3 |
231 (75.4%) 76 (24.6%) |
| BMI | |
| <25 kg/m2
≥ 25 kg/m2 No data |
107 (34.8%) 161 (52.4%) 39 (12.8%) |
| Transfusion dependency | |
| RBC TD | 168 (54.7%) |
| PLT TD | 71 (23.1%) |
| Comorbidities | |
| DM | 67 (21.8%) |
| COPD | 22 (7.1%)) |
| Cardiac failure (NYHA III/IV) |
40 (13.2%) |
| Second malignancy | 68 (22.1%) |
| Corticosteroid treatment | 18 (20.4) |
| Autoimmune disease | 41 (13.3%) |
| Tobacco smokers | 86 (28.0%) |
| MRB colonization | 50 (16.2%) |
| Antimicrobial Prophylaxis | |
| Antibacterial | 205 (66.0%) |
| Antifungal Antiviral |
90 (29.3%) 79 (25.7%) |
| 5-AZA administration | |
| Inpatient | 243 (79.2%) |
| Outpatient | 64 (20.8%) |
WHO, World Health Organization; AML, acute myeloid leukemia; MDS, myelodysplastic syndromes; CMML, chronic myelomonocytic leukemia; ECOG, Eastern Cooperative Oncology Group; IPSS, international prognostic scoring system; IPSS-R, revised international prognostic scoring system; MDS, myelodysplastic syndrome; RBC TD, Red Blood Cell Transfusion Dependence; PLT TD, Platelet Transfusion Dependence; NYHA, New York Heart Association; DM, Diabetes mellitus; COPD, Chronic obstructive pulmonary disease; MRB, multidrug-resistant bacteria; 5-AZA, azacitidine.
Results
Baseline patient characteristics
We collected data on 307 patients treated with azacitidine: AML, n = 116 (37.8%); MDS, n = 169 (55.0%); and CMML, n = 22 (7.1%). A total of 231 patients received at least three azacitidine cycles. Median number of administered cycles was 4 (range, 4–5). The median age at azacitidine commencement was 71 (range, 18–95) years and men predominated (n = 177; 57.6%) in the analyzed group. IPSS-R was calculated for 201 (65.5%) patients, and most of them belonged to high-risk (37.3%) or very-high-risk (51.2%) groups. Gut MRB colonization was found in 55 (16.2%) patients before azacitidine treatment and in 55 (17.9%) during the first three azacitidine cycles. Antibacterial, antifungal, and antiviral prophylaxes were used in 66.0%, 29.3%, and in 25.7% patients, respectively. Detailed information on baseline patients’ characteristics is presented in Table 1 .
Infection characteristics
In total, 169 infectious episodes (IEs) were recorded in 118 (38.4%) patients during the first three azacitidine cycles. Clinical characteristic is presented in Table 2 . Most of them [90 IEs (50.0%)] were classified as bacterial, 19 IEs (10.6%) were classified as fungal, and 15 IEs (8.3%) were classified as viral, and 56 IEs (31.1%) were not microbiologically categorized ( Table 3 ). Some episodes were classified by more information simultaneously (e.g., clinical presentation and microbiological characterizations), resulting in 198 clinically characterized and 180 microbiologically classified infections. Two infections were recorded in 27 patients and at least three IEs in 10 patients. Septic shock occurred in 11 IEs (5.5%).
Table 2.
Infection characteristics.
| Infection category | % of infectious episodes |
|---|---|
| Neutropenic fever | 13.1 |
| FUO (without neutropenia) | 3.0 |
| Pneumonia | 40.4 |
| Sepsis (including septic shock) | 8.5 |
| Septic shock | 5.5 |
| Gastrointestinal tract infection | 9.1 |
| Skin and soft tissue infection | 5.6 |
| Urinary tract infection | 8.1 |
| Other | 6.6 |
FUO, fever of unknown origin.
Table 3.
Microbiologically confirmed infection.
| Type of infection | (n) | Pathogen | (n) | |
|---|---|---|---|---|
| Bacterial | 58 | |||
| Gram–positive bacteria | 32 | |||
| Coagulase-negative Staphylococcus |
7 | |||
| Staphylococcus aureus | 6 | |||
| Enterococcus spp. | 8 | |||
| VRE
Clostridioides difficile Streptococcus agalactiae |
5 10 1 |
|||
| Gram-negative bacteria | 26 | |||
|
Escherichia coli
ESBL |
8 4 |
|||
|
Klebsiella pneumoniae
ESBL MBL Citrobacter freundi |
9 6 3 1 |
|||
| Morganella morganii | 1 | |||
| Pseudomonas aeruginosa | 5 | |||
| Stenotrophomonas maltophilia | 1 | |||
| Mycobacterium tuberculosis | 1 | |||
| Fungal | 10 | |||
| Proven | 7 | Aspergillus fumigatus | 2 | |
|
Candida albicans
Candida glabrata Mucor |
3 1 1 |
|||
| Probable | 3 | |||
| Viral | 13 | |||
|
Influenza A
SARS-CoV2 |
1 12 |
VRE, Vancomycin-Resistant Enterococcus; ESBL, extended-spectrum beta-lactamases; MBL, metallo-beta-lactama; SARS-CoV2, severe acute respiratory syndrome coronavirus 2.
Infectious risk
Univariate analysis showed that ECOG status, cytogenetics IPSS R score, marrow blast percentage, ferritin, IgG and CRP concentration, red blood cell, platelet transfusion dependency, and serum albumin level were associated with the risk of infection (for details, please see Table 4 ).
Table 4.
Univariate analysis of risk factors for infection within the first 3 cycles of azacitidine treatment.
| Characteristics | With infection in the first 3 cycles N (%) or median (range) |
Without infection in the first 3 cycles N (%) or median (range) |
p-value |
|---|---|---|---|
| Sex | |||
| Male | 66 (55.9) | 111 (58.7) | 0.716 |
| Female | 52 (44.1) | 78 (41.2) | |
| Age, median, range | 72 (33–88) | 71 (18–95) | 0.251 |
| WHO 2016 diagnosis | 0.024* | ||
| AML | 52 (44.0) | 64 (33.8) | |
| MDS | 54 (45.7) | 115 (60.8) | |
| CMML | 12 (10.1) | 10 (5.2) | |
| IPSS | 0.22 | ||
| Low | 0 (0.0) | 1 (0.5) | |
| Int 1 | 5 (4.2) | 11 (5.8) | |
| Int 2 | 36 (30.5) | 66 (34.9) | |
| High | 39 (33.0) | 50 (26.4) | |
| NA | 38 (32.2) | 61 (32.2) | |
| IPSS-R | 0.21 | ||
| Very Low | 0 (0.0) | 0 (0.0) | |
| Low | 2 (0.8) | 1 (0.5) | |
| Intermediate | 7 (5.9) | 14 (7.4) | |
| High | 23 (19.5) | 52 (27.5) | |
| Very High | 47 (39.8) | 56 (29.6) | |
| NA | 40 (33.9) | 66 (34.9) | |
| ECOG Performance Status | 0+ | ||
| 0–1 | 61 (51.6) | 152 (80.4) | |
| 2–4 | 57 (48.3) | 37 (19.5) | |
| IPSS cytogenetics | 0.055 | ||
| Low | 38 (32.2) | 70 (37.0) | |
| Intermediate | 18 (15.2) | 40 (21.1) | |
| High | 20 (16.9) | 28 (14.8) | |
| NA | 42 (35.6) | 51 (26.9) | |
| IPSS-R cytogenetics | 0.031* | ||
| Very Good | 38 (32.2) | 70 (37.0) | |
| Good | 1 (0.8) | 3 (1.5) | |
| Intermediate | 19 (16.1) | 45 (23.8) | |
| Poor/Very Poor | 25 (21.1) | 35 (18.5) | |
| NA | 35 (29.6) | 36 (19.0) | |
| Vit D3 | 18.0 (7.2-40) | 20.0 (8.0-80.1) | 0.179 |
| RBC TD | 82 (70.0) | 86 (46.0) | 0+ |
| Diabetes mellitus | 25 (21.5) | 42 (22.5) | 0.946 |
| COPD | 13 (11.2) | 9 (4.8) | 0.067 |
| Cardiac failure (NYHA III/IV) |
21 (18.1) | 19 (10.1) | 0.070 |
| Second malignancy | 24 (20.6) | 44 (23.4) | 0.681 |
| Corticosteroid treatment | 10 (8.5) | 10 (5.3) | 0.384 |
| Autoimmune disease | 16 (13.9) | 25 (13.2) | 1 |
| CRP (mg/L) | 20.5 (0-242) | 6 (0-200) | 0+ |
| Hemoglobin (g/dl) | 8.9 (4.7–97.0) | 9.0 (4.5–91.5) | 0.172 |
| Platelets (× 109/l) | 45 (4–828) | 61 (5–628) | 0.162 |
| Neutrophils (× 109/l) | 0.63 (0.0–56.8) | 0.87 (0.02–39.0) | 0.143 |
| Lymphocytes (× 109/l) | 1.16 (0.0–9.5) | 1.30 (0.1–11.0) | 0.516 |
| Monocytes (× 109/l) | 0.35 (0.0–41.0) | 0.30 (0.0–34.5) | 0.837 |
| Bone marrow blasts(%) | 16.7 (1.0–96.2) | 14.0 (0.0–93.0) | 0.016 |
| IgG (g/l) | 1121 (100-3937) | 900(100–1180) | 0.026* |
| Ferritin (ng/ml) | 1144 (61–11099) | 574 (11–7932) | 0.001* |
| Iron (g/dl) | 121 (27–267) | 124 (6.8–291) | 0.428 |
| Albumin (g/dl) | 3.6 (1.9–5.4) | 3.9 (2.6–9.97) | 0.0004* |
AML, acute myeloid leukemia; CMML, chronic myelomonocytic leukemia; CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; IgG, immunoglobulin G; Int, intermediate; IPSS, international prognostic scoring system; IPSS-R, revised international prognostic; scoring system; MDS, myelodysplastic syndrome; NA, not available; NYHA, New York Heart Association; RBC TD, Red Blood Cell Transfusion Dependence; COPD, Chronic obstructive pulmonary disease.
* - statistically significant meaning p value < 0,05.
In a multivariate analysis, only ECOG status, RBC transfusion dependency, IPSS-R score, and CRP concentration retained significance ( Table 5 ).
Table 5.
Multivariate generalized mixed model (logistic), odds of getting infection in any cycle.
| Parameter | Cutoff | Odds Ratio | 95% confidence interval | Significance P-value |
|---|---|---|---|---|
| CRP | > 20 mg/L | 2.68 | 1.39–5.19 | 0.003* |
| ECOG | > 2 | 2.73 | 1.42–5.31 | 0.002* |
| WBC | > 6.5 x109/L | 2.27 | 1.07-4.73 | 0.03* |
| RBC TD | Yes | 3.07 | 1.37–7.85 | 0.01* |
| R-IPSS | > 2 | 2.37 | 1.23–4.68 | 0.01* |
CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; IPSS-R, revised international prognostic; WBC, White Blood Cells; RBC TD, Red blood cells transfusion dependency.
* - statistically significant meaning p value < 0,05.
Most patients [200 (65%)] were assigned to the higher-risk AIR Model group. The occurrence of infection within the first three azacitidine cycles was significantly higher in the higher-risk group (47.0%) than in the lower-risk group (22.4%) [odds ratio 3.06 (1.82–5.30) (p < 0.05)]. The sensitivity of the model was 80, and specificity was 44.
Antimicrobial prophylaxis
In most patients (224, 73%), antimicrobial prophylaxis was administered during the first three azacitidine cycles. Infections occurred more frequently in patients with prophylaxis (44%) than in patients without prophylaxis (22%) (p = 0.001) during any of the first three AZA cycles; the difference was significant in the first cycle but it disappeared in the second and the third cycle, if analyzed separately ( Table 6 ).
Table 6.
Relative frequency of infection in patients with and without prophylaxis.
| Infection | No prophylaxis (n-72) |
Prophylaxis (n-197) |
p-value |
|---|---|---|---|
| Infection during any of the first 3 cycles (total) | 22% | 44% | 0.001* |
|
Infection
1-st cycle |
13.8% | 29.1% | 0.001* |
|
Infection
2-nd cycle |
16.2% | 24.3% | 0.14 |
|
Infection
3-rd cycle |
10.1% | 13.3% | 0.6 |
* - statistically significant meaning p value < 0,05.
The cohorts with prophylaxis vs. no prophylaxis differed significantly in terms of (1) underlying diagnosis—more frequent administration of prophylaxis in AML (62%) than in MDS (43%) or CMML (36%), (2) previous treatment—more prevalent in patients exposed previously to chemotherapy (62.9% vs. 46.3%), (3) prevalence of neutropenia (neutropenia below 0.8 × 109/L present in 57.6% vs. 40.6%), and (4) platelet transfusion dependency (60.8% vs. 46.0% in non-platelet transfused patients).
Administration of antimicrobial prophylaxis did not have a significant impact on all-infection occurrence in multivariate analysis: antibacterial prophylaxis OR 0.93 (0.41–2.05), p = 0.87; antifungal OR 1.24 (0.54–2.85), p = 0.59; and antiviral OR 1.24 (0.53–2.82), p = 0.60.
Clinical outcome
Median follow-up was 16 months (95% CI, 13–18) and 214 patients (70.0%) died during that time. Median overall survival was 11 months (95% CI, 9–14).
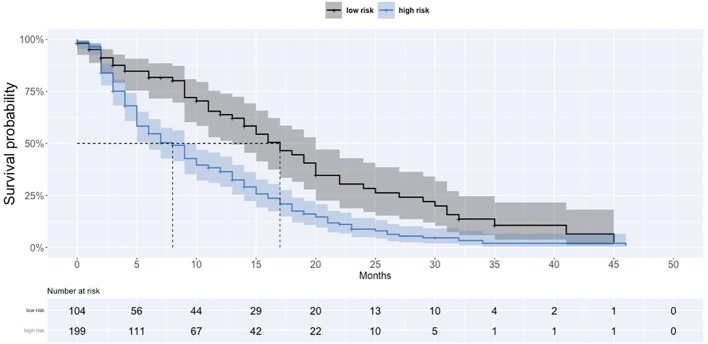
The median overall survival in the higher-risk group was 9 months (6–10), while that in the lower-risk group is 19 months (15–24) ( Figure 1 ). Fatal outcome was recorded in 25 out of 169 IEs (14.7%). Infection-related attributable mortality was 8.1% (25/307) in the whole group and 21.5% (25/118) in patients who developed infections. The median survival in patients with infection was 6 months (5–9), while that in patients without infection was 15 months (12–17).
Figure 1.
Survival curves in high and low infection risk according to AIR group.
Discussion
Azacitidine is associated with an overall survival advantage in comparison to other regimens in AML/MDS/CMML patients unfit for intensive chemotherapy and allogeneic hematopoietic cell transplantation (8, 9). Its favorable effect is probably counterbalanced by the risk of development of serious infections. The incidence of infectious episodes in MDS patients is significantly increased when compared to the age-matched general population (10).It remains the subject of debate if administration of hypomethylating agents (HMAs)/azacitidine per se is associated with grade III/IV infection. In Shargian-Alon et al.’s meta-analysis covering nine randomized controlled trials comparing HMA-containing regimens with other regimens administered to MDS/AML patients, it was found that HMA treatment increases the risk of serious infection. However, the comparator group was very divergent in the studies, including best supportive care. It is worth noting that all-cause mortality was reduced in the HMA group (11).
In our study almost 40% of patients developed grade III/IV infection during the first three azacitidine cycles. It is in line with previous reports where also most infections occurred within the first three azacitidine cycles (12, 13). Prediction of infection is crucial in patients treated with azacitidine. In our cohort ECOG status, RBC transfusion dependency, IPSS R-score, and CRP concentration were significant predictive factors in multivariate analysis. Previously, our group developed the Azacitidine Infection Risk (AIR) Model including ECOG performance status, platelet and neutrophil count, albumin serum concentration, and RBC transfusion dependency with a sensitivity of 62% and a specificity of 86% (Madry et al.) (2). In the present study, albumin concentration retained its significance only in univariate analysis, while patients with lower neutrophil and platelet count had a tendency for higher infection occurrence, but the difference was insignificant. Nonetheless, using the combination of factors applied in the AIR Model, we confirmed the validity of this model in the current analysis with a sensitivity of 80% and a specificity of 44%. Two times more patients (47%) suffered from infection in the higher-risk group in comparison with the lower-risk group (22%). Patients assigned to the higher-risk group lived significantly shorter, with a median OS of 9 months vs. 19 months.
Serum concentration of C-reactive protein is commonly used as an infection marker but with an undefined practical role according to guidelines addressed to patients with hematologic malignancies and febrile neutropenia (14). In our analysis, elevated CRP > 20 mg/L before the commencement of AZA was found to be an independent predictor of infection in patients with no other clinical manifestation of infection. In the Sugiura et al. study, asymptomatic CRP elevation anteceded febrile neutropenia occurrence in 55.6% of cases, and prophylactic application of broad-spectrum antibiotics in patients with elevated CRP reduced infection rates from 31% to 6.7% (15). It is interesting to note that the ratio of CRP to albumin was found to be associated with worse response rate and shorter overall survival in newly diagnosed AML patients (16).
Our patients were relatively well microbiologically characterized with available microbiological results in 45% of cases in comparison to other studies, where such a confirmation was documented only in 15%–54% of infectious episodes (12, 17).Pneumonia was the most frequent clinical presentation (40%), with the rate being much higher than in other studies (16%–30%). It may probably be attributable to the SARS-CoV-2 pandemic era (11 cases with COVID-19 pneumonia).
In our study, 73% of patients received any antimicrobial prophylaxis in contrast to the lower rate in previous studies (31%–49%) (1, 18). There is no clear explanation for this difference; the proportion of neutropenic and AML patients included were not significantly different. Decision on prophylaxis administration remained at the physician’s discretion and perhaps was also the result of environmental factors and the COVID-19 pandemic. It may be hypothesized that the treating physicians could have tried to prevent the occurrence of infection by any means to keep the patient alive and safe off the hospital, which, especially at the beginning of the pandemic, could become the site of the SARS-CoV-2 spread.
In our work, infectious complications were observed more commonly in patients taking prophylaxis (44% vs. 22%), especially during the first cycle. Nonetheless, in multivariate analysis, having received antibacterial prophylaxis did not retain a significant impact on infection occurrence probably due to the imbalance between the groups, i.e., with prophylaxis and without prophylaxis. Patients with prophylaxis belonged to the potentially higher-risk groups with more AML patients, with more severe/profound neutropenia and with azacitidine used as a subsequent antineoplastic therapy.
Antibiotic prophylaxis was not routinely used in available randomized clinical trials of MDS/AML patients treated with hypomethylating agents; therefore, it was not possible to assess its impact on infection risk (11). Data on antimicrobial prophylaxis derived from retrospective studies reported divergent results. Some of them demonstrated reduced bacterial infection incidence in patients receiving antimicrobial prophylaxis especially in neutropenic patients (neutrophil count < 0.5 × 109/L), while others did not show any impact (1, 13, 18–21).
Our study has an advantage over other analyses in terms of its prospective design. Nevertheless, it is also not randomized and does not control for intervariabiliity between groups in terms of population and intervention choice.
Antibacterial prophylaxis is routinely used in AML patients treated with intensive chemotherapy. Fluoroquinolone prophylaxis is associated with a lower rate of bloodstream infections and episodes of fever during neutropenia, although recently its positive effect on reducing mortality has been contested (22). Different AML and MDS biology and intensive chemotherapy (IC) and HMA mechanisms of action do not allow translating conclusions regarding prophylaxis in IC-treated patients into HMA management. MDS patients are characterized by prolonged neutropenia and delayed response to HMA; thus, long-term antibiotic treatment may induce resistance, adverse events, and the development of MDR bacterial gut colonization. There are reports demonstrating that prolonged quinolone administration significantly increases antimicrobial resistance, especially among Gram-negative bacteria, not only to quinolones but also to piperacilin/tazobactam and carbapenems (23, 24).
As mentioned earlier, our study has some limitations including the heterogeneous population of patients enrolled in regard to the underlying diagnosis (AML, CMML, and MDS), azacitidine used in different lines of therapy, and non-randomized, diverse antimicrobial prophylactic management. Another limitation is that, since 2021, the venetoclax/azacitidine (Ven/AZA) combination therapy has been the standard of care for unfit AML patients and the number of azacitidine monotherapy-treated AML patients in routine practice is limited nowadays. However, our study has been designed and initiated in 2018 before Ven/AZA was licensed in AML. We decided against the co-option of Ven/AZA-treated patients from 2021 because it would harm the group coherence and may further complicate the interpretation of the results.
On the other hand, this is one of the largest studies focusing on infection in azacitidine treated patients reflecting real-life of prospectively included and in detail clinically and microbiologically described MDS/AML/CMML population.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by Bioethics Committee, Medical University Warsaw. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
Author contributions
KM: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. KL: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. ESi: Conceptualization, Data curation, Formal analysis, Methodology, Project administration, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. JD-S: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing. PB: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Writing – original draft, Writing – review & editing. OS: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. AG: Data curation, Investigation, Methodology, Writing – original draft, Writing – review & editing. MO-S: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. AO: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing. ZW: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. JŚ: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. ESu: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. AB: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. RM: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. KB: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. GB: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing.
Funding Statement
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- 1. Bainschab A, Quehenberger F, Greinix HT, Krause R, Wölfler A, Sill H, et al. Infections in patients with acute myeloid leukemia treated with low-intensity therapeutic regimens: risk factors and efficacy of antibiotic prophylaxis. Leuk Res. (2016) 42:47–51. doi: 10.1016/j.leukres.2016.01.014 [DOI] [PubMed] [Google Scholar]
- 2. Mądry K, Lis K, Biecek P, Młynarczyk M, Rytel J, Górka M, et al. Predictive model for infection risk in myelodysplastic syndromes, acute myeloid leukemia, and chronic myelomonocytic leukemia patients treated with azacitidine; azacitidine infection risk model: the polish adult leukemia group study. Clin Lymphoma Myeloma Leuk. (2019) 19(5):264–74.e4. doi: 10.1016/j.clml.2019.01.002 [DOI] [PubMed] [Google Scholar]
- 3. Taplitz RA, Kennedy EB, Bow EJ, Crews J, Gleason C, Hawley DK, et al. JOURNAL OF CLINICAL ONCOLOGY antimicrobial prophylaxis for adult patients with cancer-related immunosuppression: ASCO and IDSA clinical practice guideline update. J Clin Oncol. (2018) 36(30):3043–54. doi: 10.1200/JCO.18.00374 [DOI] [PubMed] [Google Scholar]
- 4. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. Revised. 4th ed. Geneva, Switzerland: WHO Press World Health Organization; (2017). [Google Scholar]
- 5. Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. (1997) 89:2079–88. doi: 10.1182/blood.v89.6.2079 [DOI] [PubMed] [Google Scholar]
- 6. Greenberg PL, Tuechler H. Revised international prognostic scoring system for MDS. Blood. (2012) 120:2454–65. doi: 10.1182/blood-2012-03-420489.The [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. della Porta MG, Malcovati L, Strupp C, Ambaglio I, Kuendgen A, Zipperer E, et al. Risk stratification based on both disease status and extra-hematologic comorbidities in patients with myelodysplastic syndrome. Haematologica. (2011) 96(3):441–9. doi: 10.3324/haematol.2010.033506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Fenaux P, Mufti GJ, Hellstrom-Lindberg E, Santini V, Finelli C, Giagounidis A, et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study Correspondence. Lancet Oncol. (2009) 10:223–32. doi: 10.1016/S1470-2045(09)70003-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Dombret H, Seymour JF, Butrym A, et al. International phase 3 study of azacitidine vs conventional care regimens in older patients with newly diagnosed AML with >30% blasts. Blood. (2015) 126:291–9. doi: 10.1182/blood-2015-01-621664 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Kasprzak A, Andresen J, Nachtkamp K, Kündgen A, Schulz F, Strupp C, et al. Infectious complications in patients with myelodysplastic syndromes: A report from the düsseldorf MDS registry. Cancers (Basel). (2024) 16(4):808. doi: 10.3390/cancers16040808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Shargian-Alon L, Gurion R, Raanani P, Yahav D, Gafter-Gvili A. Hypomethylating agents-associated infections—Systematic review and meta-analysis of randomized controlled trials. Clin Lymphoma Myeloma Leuk. (2018) 18:603–610.e1. doi: 10.1016/j.clml.2018.05.017 [DOI] [PubMed] [Google Scholar]
- 12. Merkel D, Filanovsky K, Gafter-gvili A, Vidal L, Aviv A, Gatt ME, et al. Predicting infections in high-risk patients with myelodysplastic syndrome/acute myeloid leukemia treated with azacitidine : a retrospective multicenter study. Am J Hematol. (2013) 88(2):130–4. doi: 10.1002/joh.23368 [DOI] [PubMed] [Google Scholar]
- 13. Trubiano JA, Dickinson M, Thursky KA, Spelman T, Seymour JF, Slavin MA, et al. Incidence, etiology and timing of infections following azacitidine therapy for myelodysplastic syndromes. Leuk Lymphoma. (2017) 58(10):2379–86. doi: 10.1080/10428194.2017.1295141 [DOI] [PubMed] [Google Scholar]
- 14. Averbuch D, Orasch C, Cordonnier C, Livermore DM, Mikulska M, Viscoli C, et al. European guidelines for empirical antibacterial therapy for febrile neutropenic patients in the era of growing resistance: summary of the 2011 4th European Conference on Infections in Leukemia Diana. Haematologica. (2013) 98(12):1826–35. doi: 10.3324/haematol.2013.091025.DE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Sugiura S, Ito T, Koyama N, Sasaki N, Ikai H, Imanaka Y. Asymptomatic C-reactive protein elevation in neutropenic children. Pediatr Int. (2017) 59:23–8. doi: 10.1111/ped.13077 [DOI] [PubMed] [Google Scholar]
- 16. Dou L, Shi M, Song J, Niu X, Niu J, Wei S, et al. The prognostic significance of C-reactive protein to albumin ratio in newly diagnosed acute myeloid leukaemia patients. Cancer Manag Res. (2022) 14:303–16. doi: 10.2147/CMAR.S343580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Schuck A, Goette MC, Neukirchen J, Kuendgen A, Gattermann N, Schroeder T, et al. A retrospective study evaluating the impact of infectious complications during azacitidine treatment. Ann Hematol. (2017) 96(7):1097–104. doi: 10.1007/s00277-017-3001-2 [DOI] [PubMed] [Google Scholar]
- 18. Vilorio-Marqués L, Castañón Fernández C, Mora E, Gutiérrez L, Rey Bua B, Jiménez Lorenzo MJ, et al. Relevance of infections on the outcomes of patients with myelodysplastic syndromes, chronic myelomonocytic leukemia, and acute myeloid leukemia treated with hypomethylating agents: a cohort study from the GESMD. Ther Adv Hematol. (2022) 13:20406207221127547. doi: 10.1177/20406207221127547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Lorenzana N, Avila LF, Alonso S, Colado E, Bernal T. The impact of antimicrobial prophylaxis in morbidity and infections during azacitidine treatment. Ann Hematol. (2017) 96:1833–40. doi: 10.1007/s00277-017-3091-x [DOI] [PubMed] [Google Scholar]
- 20. Lee JH, Lee KH, Lee JH, Kim D-Y, Kim S-H, Lim S-N, et al. Decreased incidence of febrile episodes with antibiotic prophylaxis in the treatment of decitabine for myelodysplastic syndrome. Leuk Res. (2011) 35(4):499–503. doi: 10.1016/j.leukres.2010.07.006 [DOI] [PubMed] [Google Scholar]
- 21. Ofran Y, Filanovsky K, Gafter-Gvili A, Vidal L, Aviv A, Gatt ME, et al. Higher infection rate after 7- compared with 5-day cycle of azacitidine in patients with higher-risk myelodysplastic syndrome. Clin Lymphoma Myeloma Leuk. (2015) 15(6):e95–9. doi: 10.1016/j.clml.2015.02.030 [DOI] [PubMed] [Google Scholar]
- 22. Mikulska M, Averbuch D, Tissot F, Cordonnier C, Akova M, Calandra T, et al. Fluoroquinolone prophylaxis in haematological cancer patients with neutropenia: ECIL critical appraisal of previous guidelines. J Infection. (2018) 76(1):20–37. doi: 10.1016/j.jinf.2017.10.009 [DOI] [PubMed] [Google Scholar]
- 23. Clerici D, Galli L, Greco R, Lugli AP, Erbella F, Ripa M, et al. Levofloxacin prophylaxis vs no prophylaxis in patients with neutropenia within an endemic country for carbapenem-resistant GNB. Blood Adv. (2023) 7(9):1621–34. doi: 10.1182/bloodadvances.2022008226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. De Rosa FG, Motta I, Audisio E, et al. Epidemiology of Bloodstream Infections in Patients with Acute Myeloid Leukemia Undergoing Levofloxacin Prophylaxis (2013). Available online at: http://www.biomedcentral.com/1471–2334/13/563. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.