Abstract
Classic psychedelics and MDMA have a colorful history of recreational use, and both have recently been re-evaluated as tools for the treatment of psychiatric disorders. Several studies have been carried out to assess potential long-term effects of a regular use on cognition, delivering distinct results for psychedelics and MDMA. However, to date knowledge is scarce on cognitive performance during acute effects of those substances. In this systematic review and meta-analysis, we investigate how cognitive functioning is affected by psychedelics and MDMA during the acute drug effects and the sub-acute (“afterglow”) window. Our quantitative analyses suggest that acute cognitive performance is differentially affected by psychedelics when compared to MDMA: psychedelics impair attention and executive function, whereas MDMA primarily affects memory, leaving executive functions and attention unaffected. Our qualitative analyses reveal that executive functioning and creativity may be increased during a window of at least 24 h after the acute effects of psychedelics have subsided, whereas no such results have been observed for MDMA. Our findings may contribute to inform recommendations on harm reduction for recreational settings and to help fostering differential approaches for the use of psychedelics and MDMA within a therapeutic framework.
Keywords: Cognition, Psychedelic, MDMA, Acute, Subacute, Afterglow
Subject terms: Human behaviour, Cognitive neuroscience, Medical research
Introduction
Classic psychedelics (“psychedelics”), such as lysergic acid diethylamide (LSD) or psilocybin, can facilitate a variety of psychological effects, including perceptual distortions, cognitive restructuring, mood changes, changes in meaning of percepts, and dissolution of bodily boundaries1. These effects are believed to be mainly induced via agonism at the serotonin (5HT) 2a receptor2. In addition to the specifics of acute effects, psychedelics are unique regarding their ability to exert psychological effects that by far outlast their acute physiological presence3–6. Temporal dynamics of psychedelics can therefore be differentiated into acute (psychedelic states), sub-acute, and long-term effects5–7.
Notably, even regular high-frequent use of psychedelics has not been associated with persisting, long-term impairment of cognitive functioning8,9. In contrast, research on how psychedelics affect cognition during the acute and subacute period of drug effects has not yet revealed a clear pattern10. Nevertheless, knowledge on the acute effects of psychedelics on cognition appears to be essential for the estimation of the risk profile in recreational use, and subsequent recommendations for harm reduction11,12. Moreover, knowledge on acute and subacute cognitive effects is also important regarding potential therapeutic applications of psychedelics in the treatment of psychological conditions like depression or anxiety within a psychotherapeutic framework. For instance, there is preliminary evidence that the quality and intensity of acute subjective experiences during psychedelic states might predict long-term treatment outcomes13–15. Moreover, with regard to the subacute (“afterglow”) period, an increased effectivity for therapeutic interventions has anecdotally been reported16–18.
Even if the entactogen 3,4-methylenedioxymethamphetamine (MDMA) does not belong to the group of classic psychedelics, it has sometimes also been referred to as a “psychedelic” in the context of substance-assisted therapy19,20. Despite serotonergic neurotransmission being a shared pathway of action, psychedelics and MDMA differ in their pharmacodynamic2,21,22, toxicological23–25, and phenomenological19,26 properties. Additionally, psychedelics and MDMA also diverge in their effects beyond the stage of acute intoxication, including sub-acute and long-term effects. Psychedelics, in particular, have been shown to induce sustained sub-acute effects7 not currently detected in MDMA use. Furthermore, psychedelic use appears to be mostly non-toxic in terms of neuropsychological functioning8,9, while research in MDMA users often suggest persistent cognitive deficits, particularly in memory functioning27–31. However, these MDMA-related findings remain disputed, with some authors arguing that reported differences are more likely to stem from pre-morbid differences rather than substance-induced cognitive dysfunction32,33, and may be less pronounced than initially reported33,34. Nevertheless, the literature suggests that the repeated use of psychedelics and MDMA is related to distinct neuropsychological outcomes.
To our knowledge to date there is no data investigating the relationship between the acute cognitive effects of those substances and the longitudinal differences between psychedelics and MDMA. Meanwhile, comprehensive understanding of the acute cognitive effects of psychedelics and MDMA appears to be crucial for various areas of research and for the evaluation of acute harms associated with those substances. Considering the increasing recreational use of psychedelics and MDMA35–37, providing accurate harm reduction resources and information about potential acute effects becomes highly relevant. The cognitive effects of psychoactive substances, especially those consumed recreationally without medical supervision, play an important role in the determination of their safety profiles. In comparison, GABAergic substances like alcohol and benzodiazepines lead to reduced reaction times38–40 and vigilance41,42 while also increasing risk-taking behaviour43, and potentially inducing delusions of sobriety44. On the other hand, stimulant drugs, e.g. caffeine or amphetamine, may increase attentional performance45,46 or induce hypervigilance47, while similarly inducing a tendency to overestimate capabilities under the influence48,49. Thus, the acute cognitive effects of psychoactive substances may have consequences for issues such as road safety, performance enhancement or reduction, or social interactions. To properly evaluate the effects of psychedelics and MDMA in these domains, a comprehensive assessment of their acute cognitive effects is needed.
In addition to prevention and harm reduction, the acute and subacute cognitive effects of psychedelics and MDMA may also play a role in their use as therapeutic agents. Given that higher levels of cognitive functioning are related to better outcomes and treatment success in traditional psychotherapy50–54, it is reasonable to assume it plays an important role in the context of psychedelic-assisted therapy (PAT) as well. Moreover, while not part of the commonly-applied PAT framework, psychedelics have been used therapeutically in the form of microdoses (1/10th of a common dose, e.g., 10µg of LSD)55,56. In this context, psychedelics stemming from the black market are often used by patients for self-medication of attention deficit hyperactivity disorder57 (ADHD). This type of use, along with harm reduction aspects, considerations regarding PAP and other instances of psychedelic and MDMA use outside of trial contexts (e.g., in recreational or retreat settings), highlights the need for an up-to-date analysis of acute cognitive effects.
To provide this analysis we conducted a systematic review and meta-analysis of the acute cognitive effects of psychedelics and MDMA. Our study goes beyond previous research by aiming to include analyses of the acute cognitive effects of microdosing, the cognitive post-acute effects of psychedelics, and indirect comparisons of the acute effects of psychedelics and MDMA.
Method
Search strategy
We performed electronic searches on PubMed, Web of Science, and Embase, from the respective database inception to July 10th, 2023, following PRISMA guidelines. In addition, we searched the clinical trial registries ClinicalTrials.gov and EudraCT. The search in PubMed, Web of Science, and Embase utilized an algorithm that combined terms related to psychedelics or MDMA with terms associated with cognitive testing in an iterative manner (see Supplementary Table S1). In the trial registries, individual names of psychedelics and MDMA were used for the search. References were retrieved through electronic searches and by manual searches of the reference lists of selected articles. Articles published in English, German, French, Serbian or Spanish were included. Detailed in- and exclusion criteria are described in the Supplementary Method Section.
Data extraction
Two authors (LAB, TGR) independently screened all search results, with input from a third author (TM) in cases of disagreement regarding inclusion or exclusion. Additionally, the two raters classified all studies into acute or sub-acute assessments.
Study quality
We assessed study quality and risk of bias using the Cochrane risk of bias (RoB) rating scales58 for parallel-arm trials and cross-over trials. For sequentially designed trials, we used the rating tool from the National Heart, Lung, and Blood Institute (NHLBI)59,60. LAB and TGR independently rated study quality, and a consensus was reached through discussion for each parameter.
Meta-analysis
We included studies that reported results as mean with standard deviations into the meta-analyses and performed analyses if at least three viable studies per domain were included. As many studies reported multiple tests per domain we performed a three-level meta-analysis61 on the standardized mean difference, incorporating between-study as well as within-study heterogeneity into the model. We modeled the effect sizes as nested within their respective studies by assigning a nested random effect to the grouping variable “study”. Thus, the random intercept was allowed to vary across different studies. Test statistics and confidence intervals for the fixed effects used a t-distribution. The test of moderators checks the assumption that , which is a test for an overall effect of the variable “drugType” (MDMA vs. psychedelics). To compare both drug types we defined a contrast, testing if (Wald-Type Test).
Separate analyses were conducted for studies with sub-acute assessments (testing done up to 24h after drug administration) and microdosing approaches. Heterogeneity was measured using the I2 statistic. For studies reporting multiple dosages and/or times of administration, data for the highest dosage and closest time to peak drug concentration (tmax; based on comparison values from62–64) were selected. We planned six sensitivity analyses: (1) restricting included studies to accuracy-based test or (2) speed-based test), (3) excluding the study with the highest weight, (4) using measurements closest to drug administration instead of closest to tmax in cases of multiple measurements, (5) restricting to the same psychedelic, (6) excluding studies with a high risk of bias rating. Statistical analyses were carried out using R version 4.3.265 and the following R packages: dmetar v. 0.1.066, metafor v. 4.4.067. All used packages are cited in the Supplementary Materials.
Results
Study selection
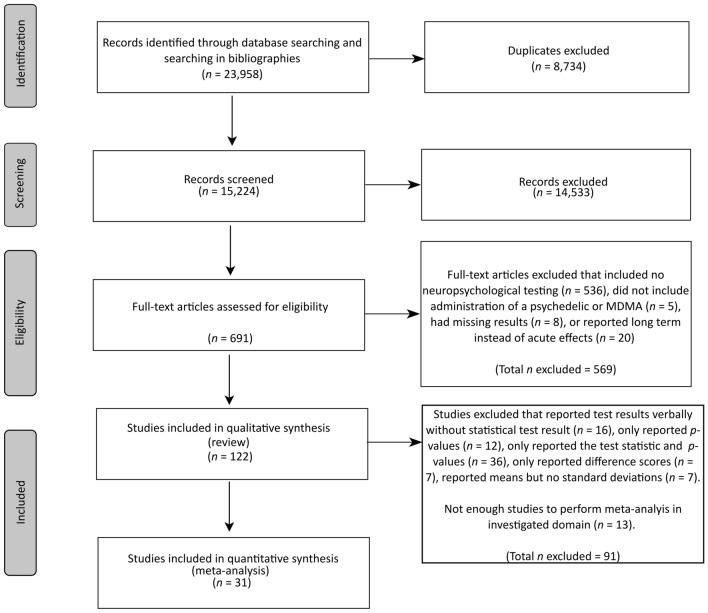
Excluding duplicates, our search yielded 15,224 records, out of which 691 were selected for full-text screening. Following full-text screening, 122 records were included in the systematic review, and 31 records were viable for quantitative analysis. There was high inter-rater agreement (Fleiss’ kappa k = 0.98; 95%CI [0.97–0.99]), with only ten cases of 691 screened full-texts involving disagreements. Two of these disagreements were related to the type of investigated substances, in which cases the third reviewer (TM) provided expertise. The other eight disagreements were due to the unclear nature of the included tests and were concluded based on literature research and expertise by reviewer TGM. Supplementary Tables S2–9 provide an overview of the included studies. Specifically, 68 studies investigated the cognitive effects of classic full-dose psychedelics (Supplementary Tables S2 and S6), 34 studies focused on MDMA (Supplementary Tables S3 and S7), nine studies explored microdosing (Supplementary Tables S4 and S8), and nine studies assessed cognition within 24 h after acute dosing (Supplementary Tables S5 and S9). Figure 1 (and Supplementary Table S11) displays details of the different phases of the search.
Figure 1.
PRISMA flowchart.
Quality rating
All 83 studies that were assessed using the Cochrane RoB tool, including both parallel-arm as well as cross-over studies, were rated as having a high risk of bias. Out of the 37 studies assessed with the NHLBI instrument, 34 were considered to be of poor quality, and three were rated as fair quality (see Supplementary Tables S2–5). The high risk of bias in the included studies is mainly due to the unblinding effects of psychedelics and MDMA68. If proper blinding would have been accomplished, five sequential studies would have been rated fair, while 41 placebo-controlled studies would have been rated as having some concern and 5 studies as having a low risk of bias. For the purpose of the respective sensitivity analyses, we excluded the blinding criterion from the quality rating, allowing to control for differences in other sources of bias risk between studies.
Qualitative analysis
Here, we present the results regarding the influences of psychedelics and MDMA on memory performance, executive functioning, and attention. Additional findings on other cognitive abilities, such as visuospatial skills, intelligence, motor skills, and language functions, are provided in the supplement.
Full-dose, acute assessment of psychedelics
Out of the 67 studies investigating the acute effects of full-dose psychedelics, 55 studies (n = 1062) specifically assessed memory, executive function, or attention. Supplementary Table S2 provides an overview of study design, participants, and tests administered. Cognitive tests were conducted between 1069 and 36070,71 min after drug administration, with four studies70–73 involving repeated assessments. LSD dosages ranged from 40 µg74 to 2 µg/kg75, psilocybin dosages ranged from 4576 to ~ 430 µg/kg71, DMT dosages ranged from 0.1769 to 0.32 mg/kg77,78, one study administered 100 ml of ayahuasca79, and ten studies involved multiple dosages70,71,73,74,76–78,80–82.
Memory
A total of 21 studies (n = 553) investigated memory performance under the influence of ayahuasca79, DOM83,84, LSA72, LSD74,75,82,85–92, 2C-B93 and psilocybin71–73,93–96, utilizing 22 distinct memory tests. The majority of studies (1 LSA, 3 psilocybin, 6 LSD, 1 DOM)72–74,82,84,85,88–90,95,96 did not show any acute effects of psychedelics on memory across fifteen tasks. On the other hand, eleven studies (4 psilocybin, 1 2C-B, 6 LSD, 1 ayahuasca)71,72,75,79,82,86,87,91–94 demonstrated impaired memory performance across twelve tasks, while only one study (DOM)83 using one task showed improved memory performance.
Executive functioning
Twenty-four studies (n = 503) applied 27 distinct tasks to assess the acute effects of Ayahuasca79,97,98, 2C-B93, LSD80,81,87–89,99–107, mescaline108 and psilocybin71,93,109–112 on executive functioning. Sixteen of the included studies (1 2C-B, 6 psilocybin, 10 LSD)71,81,87,88,93,99–104,107,109–112 reported impaired executive functions across 19 tasks, while twelve studies (2 psilocybin, 3 ayahuasca, 7 LSD)71,79,80,87,89,97–99,101,105,106,111 reported no effects across 15 tasks. Additionally, five studies (2 ayahuasca, 2 LSD, 1 mescaline)79,97,99,101,108 showed improved performance across six tests.
Attention
Twenty-three studies (n = 291) investigated the acute effects of DMT69,77,78,113, 2C-B93, mescaline114, LSD74,80,81,90,115–118, and psilocybin70,76,93,95,109,119–123, utilizing 18 distinct attention tasks. Among these studies, fifteen (2 DMT, 1 2C-B, 10 psilocybin, 3 LSD)69,70,76,77,93,95,109,116–123 reported impaired attentional performance in 17 tasks, while nine studies (2 DMT, 6 LSD, 1 mescaline)74,78,80,81,90,113–116 reported no effect across eleven tasks.
Full-dose, acute assessment of MDMA
Among the 34 studies on MDMA, 33 studies (n = 716) examined its effects of MDMA on memory, executive functioning, and attention, as summarized in Supplementary Table S3. Cognitive tests were administered between 15 min124 and 11 h125,126 min after drug administration, with three studies124–126 involving repeated assessments. Dosages used ranged from 25 mg127 to 1.7 mg/kg128,129, with five studies involving multiple dosages125–127,130,131.
Memory
Twenty-two studies (n = 454)124–127,131–147 on MDMA examined its effect on memory using 25 different tests. Out of these studies, sixteen124,126,131–140,142,143,146–148 concluded that MDMA acutely impairs memory across 23 tasks, while twelve studies124,127,132–134,137,140–142,144,145,149 showed no effect in 13 tasks. In addition, one study133 reported improved performance in one measure.
Executive functioning
Thirteen studies (n = 240)125,127,129,130,135,137,140,141,144,150–153 investigated the effects of acute MDMA exposure on executive functioning, utilizing 14 different tasks. The majority of these studies125,127,129,130,135,137,144,150–153 showed no significant effect of MDMA on fourteen tasks, while four studies130,140,141,152 reported impaired performance across four tasks.
Attention
Thirteen studies (n = 270)21,124,125,127,128,133,135,141,144,147,153–155 assessed the effects of MDMA on attention using 18 different tests. The majority of these studies21,124,125,127,128,133,141,144,147,153–155 found no effect of MDMA on attentional performance across 17 measures, while four studies125,135,141,153 reported impaired attention in four tasks.
Microdose, acute assessment of psychedelics
Eight studies (n = 209) conducted investigations on the cognitive effects of psychedelic microdosing156–163 on memory, executive functioning, and attention, as detailed in Supplementary Table S4. The studies utilized LSD157–159,161–163, psilocybin-containing truffles156, and psilocybin-containing mushrooms160.
Memory
Four of these studies (n = 124)157,158,161,162 examined the effects of microdosed LSD on memory using five tests, and none of them found any significant effect.
Executive functioning
Five studies (n = 161) evaluated the effects of microdoses of LSD157,161,163 and psilocybin156,160 on executive functioning using nine different tasks. Among them, two studies160,163 reported impaired performance in two tasks, one study156 reported improved performance in two tasks, while the majority of studies157,160,161,163 found no significant difference across eleven tasks.
Attention
Four of the reviewed studies (n = 106) assessed attentional performance under the influence of a LSD158,159,163 or psilocybin160 microdoses using six different tasks. Three of these studies158,160,163 found no significant effect on attentional performance across five tasks, while one study159 reports reduced attention in one task.
Full-dose, sub-acute assessment of psychedelics
Eight studies (n = 282)4,99,164–169 had their participants perform cognitive tasks of memory, executive functioning, or attention on the day after consumption of a psychedelic substance (Supplementary Table S5).
Memory
Three studies (n = 46)166,167,170 reported sub-acute effects of LSD on five different memory tasks. Two of these studies166,170 showed increased memory performance across three tasks, and two studies 167,170 showed no effect in two task.
Executive functioning
Six studies (n = 288) evaluated sub-acute executive functioning using five different tasks in psilocybin165, ayahuasca4,164,168, 5-Meo-DMT167,169, and LSD170. Out of these studies, two164,170 showed impaired performance in two tasks, while four studies4,165,168,169 reported improvement across four measures, and three studies4,167,170 found no effect in four tasks.
Attention
Two studies (n = 46)167,170 investigated sub-acute effects of LSD using two tasks and observed no significant difference in performance compared to placebo.
Full-dose, sub-acute assessment of MDMA
Two studies (n = 27)144,171 assessed the subacute cognitive effects of MDMA and found no effect on three memory tasks, three executive functioning tasks and one attention task (see Supplementary Table S5).
Quantitative analysis
Memory
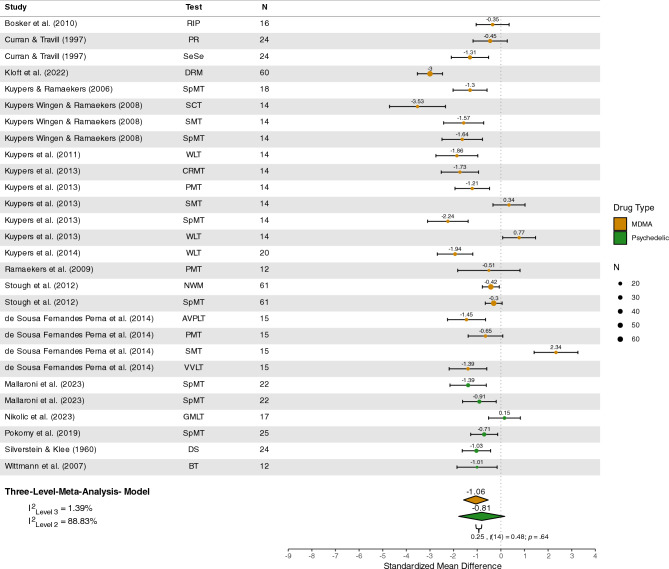
Sixteen studies73,75,87,93,96,126,127,132,133,136,138–141,143,148 were included in the quantitative analysis of memory tasks, with a total of n = 368 participants. Memory was not significantly impaired under psychedelics (Z = − 0.81 [95%CI − 1.78; 0.17], p = 0.10) but under MDMA (Z = − 1.06 [95% CI − 1.58; − 0.54], p < 0.001), see Figure 2. Furthermore, no difference in memory performance was detected between these substances (Z = 0.25 [95% CI − 0.86; 1.35] p = 0.64). The estimated variance components were τ2Level3 = 0.02 and τ2Level2 = 1.09. Therefore I2Level3 = 1.39% of total variation could be attributed to between-study, and I2Level2 = 88.83% to within-study heterogeneity.
Figure 2.
Forest plot of psychedelic and MDMA studies involving memory tasks. MDMA, 3,4-methylenedioxymethamphetamine; RIP, rapid information processing; PR, prose recall; SeSe, serial sevens; VVLT, visual verbal learning task; PMT, prospective memory task; SMT, Sternberg memory task; AVPLT, abstract visual pattern learning task; DRM, Deese/Roediger-McDermott word list; SpMT, spatial memory task; SCT, star counting task; WLT, word learning task; CRMT, continuous recognition memory task; NWM, numeric working span; GMLT, Groton maze learning task; SpWM, spatial working memory task; DS, digit span; BT, block tapping.
Executive functioning
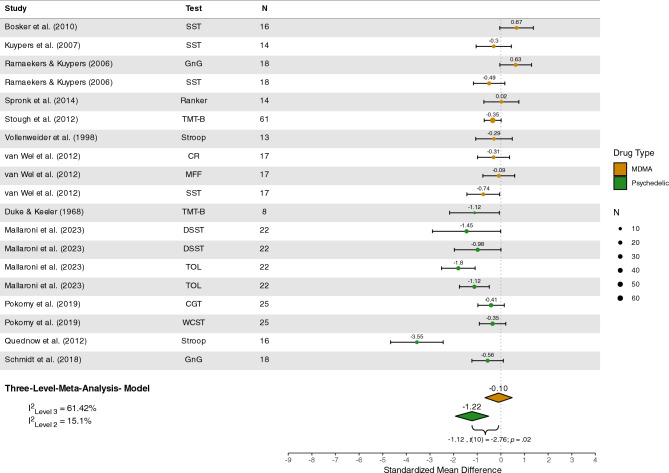
Twelve studies87,93,100,109,110,125,127,129,130,141,151,152 with a total of n = 242 participants were included in the quantitative analysis of executive functioning. Meta-analyses revealed significant impairment under psychedelics (Z = − 1.22 [95%CI − 1.92; − 0.52], p = 0.003), but not under MDMA (Z = − 0.10 [95% CI − 0.67; 0.48], p = 0.72), with a significant group difference observed (Z = − 1.12 [95% CI − 2.03; − 0.22], p = 0.02), see Figure 3. The estimated variance components were τ2Level3 = 0.31 and τ2Level2 = 0.08 Therefore
Figure 3.
Forest plot of psychedelic and MDMA studies involving executive functioning tasks. MDMA, 3,4-methylenedioxymethamphetamine; SST, stop-signal task; DT, discounting task; GnG, go/no-go task; TMT-B, trail-making task trial B; MFF, matching familiar figures; CR, cue-reversal task; TOL, Tower of London; DSST, digit-symbol substitution; WCST, Wisconsin card sorting task; CGT, Cambridge gambling task.
I2Level3 = 61.42% of total variation could be attributed to between-study, and I2Level2 = 15.10% to within-study heterogeneity.
Attention
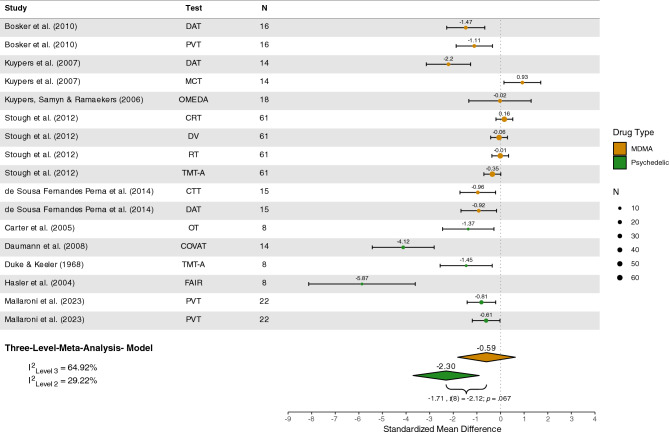
Ten studies69,76,93,95,109,125,127,133,141,155 encompassing n = 184 participants were included in the quantitative analysis of attention. Significant impairment was detected under psychedelics (Z = − 2.30 [95%CI − 3.71; − 0.90], p = 0.005) but not MDMA (Z = − 0.59 [95%CI − 1.82; 0.63], p = 0.30), see Figure 4. However, subgroup analyses indicated no significant difference in attentional performance under psychedelics compared to MDMA (Z = − 1.71 [95%CI − 3.57; 0.15], p = 0.07). The estimated variance components were τ2Level3 = 1.05 and τ2Level2 = 0.47. Therefore I2Level3 = 64.92% of total variation could be attributed to between-study, and I2Level2 = 29.22% to within-study heterogeneity.
Figure 4.
Forest plot of psychedelic and MDMA studies involving attention tasks. MDMA, 3,4-methylenedioxymethamphetamine; DAT, divided attention task; PVT, psychomotor vigilance task; CTT, critical tracking task; OMEDA, object movement under divided attention; MCT, Macworth clock task; RT, simple reaction time; DV, digit vigilance; CRT, choice reaction time; TMT-A, trail-making task trial A; OT, object tracking; COVAT, covert orienting of attention test; FAIR, Frankfurt attention inventory.
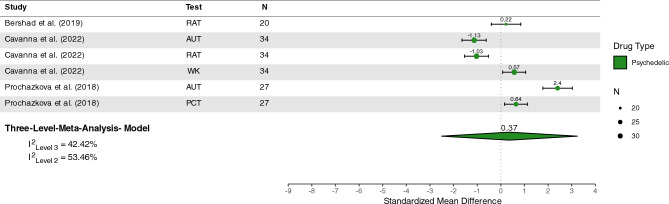
Microdosing
Only three microdosing could be included for quantitative analysis and these studies were exclusively related to the domain of creativity. Thus, no quantitative analysis of microdosing effects on other cognitive domains could be performed. Three studies156,157,160, involving n = 81 participants, were included in a quantitative analysis of creativity tasks under microdose conditions. The overall effect estimate revealed no significant effect of psychedelic microdoses on creativity tasks (Z = 0.37 [95%CI − 2.51; 3.24], p = 0.64), see Figure 5. The estimated variance components were τ2Level3 = 0.76 and τ2Level2 = 0.96 Therefore
Figure 5.
Plot of microdosing studies involving creativity tasks. LSD, lysergic acid diethylamide; RAT, remote association task; AUT, alternative uses task; WK, Wallach-Kogan Test; PCT, picture concept task.
I2Level3 = 42.42% of total variation could be attributed to between-study, and I2Level2 = 54.46% to within-study heterogeneity.
Sensitivity analyses
An overview of all completed sensitivity analyses can be found in Supplementary Table S10.
When limiting studies to speed-based tasks the significant effects of MDMA memory disappeared (Z = 1.43 [95%CI − 12.87; 10.01], p = 0.36). Similarly, the effect of psychedelics on executive functioning is non-significant in speed-based tasks (Z = − 1.54 [95%CI − 3.63; 0.55], p = 0.10) and when limiting the study selection to LSD (Z = − 0.43 [95%CI − 2.63; 1.78], p = 0.25) or psilocybin (Z = − 2.11 [95%CI − 5.12; 0.91], p = 0.09) alone. Regarding attention, the significant effect of psychedelics only persists when excluding the study with highest weight (Z = − 2.92 [95%CI − 4.65; − 1.18], p = 0.006), but not for any other sensitivity analysis, see Supplementary Table S10.
Discussion
In our analysis of the acute and subacute effects of classic psychedelics and MDMA on cognition, we report the following findings: acute effects of classic psychedelics are consistently associated with reduced cognitive performance across multiple domains. Our qualitative and quantitative results indicate that the most pronounced effects are observed in the domain of attention, whereas memory and executive functioning remain less intensively affected. In contrast, MDMA primarily impacts acute memory performance. Notably, the included studies were highly heterogeneous in terms of the tests applied, reported outcomes of each test, drug dosages, and time between drug and test administration, which hinders accurate comparisons across studies. Our results provide important information in the context of psychedelic harm reduction, for instance, when it comes to choosing a safe setting for recreational use, considering to drive a car or to elsewise participate in road traffic. In addition, impaired memory functions under MDMA might potentially reduce certainty in users about the amount of consumed MDMA which could lead to dangerous redosing and overdosing172.
Our results also provide some insight when it comes to fostering therapeutic interventions for the framework of psychedelic-assisted therapy (PAT). Specifically, during the acute experience, applying cognitive interventions might be challenging, if patients are unable to properly follow the therapist’s guidance. In contrast, with psychedelics specifically, there might be a place for increased therapeutic support during the days after psychedelic dosing sessions, since cognitive performance is potentially increased during that subacute (‘afterglow’) window. For MDMA, on the other hand, the effects on memory performance might actually be beneficial in the context of Posttraumatic Stress Disorder (PTSD)-specific therapy that involves reconsolidation of traumatic memories under the influence of MDMA173. The reduced ability to encode, especially emotional149, information might reduce reconsolidation of traumatic memories and promote extinction, thus promoting a reduction of PTSD symptoms174. In fact, there is already a line of research focused on producing psychopharmacological agents that can modify memory to support PTSD therapy175. Thus, our results support the notion that psychedelic and MDMA-assisted therapy are two distinct forms of therapies that are applicable to different disorders and different treatment approaches, as reflected in their distinct cognitive effects facilitating distinct therapeutic interventions. In addition to the consistent evidence that altered reconsolidation of memories is a main mechanism of MDMA-assisted therapy173,176, the re-experiencing of memories might be relevant in therapy with classic psychedelics as well177,178.
The disparity between psychedelics and MDMA with regard to acute cognitive effects may be linked to differences in acute subjective effects. In short, while psychedelic experiences might be characterized by strong perceptual distortions26,76,179 and potentially distracting experiences such as reduced self-other boundaries180, MDMA experiences are often marked by an pronounced emotional pattern of effects26,181. The emotional effects of MDMA may impair the ability to retrieve state-incongruent memories (e.g., retrieving neutral stimuli such as word lists, in a highly aroused positive emotional state)149,182,183 and as such lead to the observed memory deficits, while the effects of psychedelics might provide sensory distraction that reduces acute attention. Notably, under microdosing conditions which lack the perceptual alterations of psychedelics184, those effects on cognition disappear156,159,160,163. While this may suggest that the nature of the psychedelic alterations might be inherently distracting, this could also be the result of dose-dependent effects, as studies with multiple dosages of psilocybin showed dose-dependent effects on cognitive performance71,73,73,76.
While it is unclear if the observed difference is due to these factors, there is strong evidence indicating a reduction in attention under psychedelics which aligns with the recently proposed model of cognitive functioning under psychedelics by Sayalı and Barrett185. This model posits that psychedelics induce a transient increase of cognitive flexibility while simultaneously impairing attentional capacities. As we found preliminary evidence that attention was significantly reduced when focusing on speed-based tasks but not accuracy-based tasks, our findings support the notion that psychedelics specifically reduce sustained attention or vigilance but not cognitive control (as is needed for accuracy-based tasks). Additional confirmatory evidence is provided by research regarding creative performance under the influence of psychedelics99,111,186 and animal research187,188 showing an increased ability to switch action patterns under psilocybin.
Furthermore, this model of increased cognitive flexibility aligns with results regarding neuropsychological consequences of long-term psychedelic use. For instance, there is no evidence for lasting cognitive deficits8,9, but some preliminary evidence indicating potential sustained cognitive benefits beyond the acute psychedelic effects. Our qualitative review indicated that executive functioning and creativity may be increased within a window of 24 h after a using a psychedelic4,165,168,169. Furthermore, studies have shown improvements in executive functioning one week after psilocybin administration189, and there is as some evidence of improved executive performance in individuals who regularly use ayahuasca190,191. Finally alterations in neuronal networks of executive control192, salience and default mode networks193 have been reported, as well as increased neuroplasticity for up to one week after administration194. As for MDMA, only few studies have investigated the sub-acute effects, and the evidence so far indicates no impairments or improvements on cognition144,171. Even though, animal research indicates that MDMA might enhance critical periods of social learning in the days after administration195,196, there is also evidence linking MDMA-induced memory deficits to alterations in serotonergic functioning197–199, which is in line with findings indicating deteriorations in the serotonergic system after repeated MDMA use200. Neurobiological long-term consequences of repeated use might also account for the proposed long-term cognitive deficits associated with MDMA use27. However, while neuronal adaptive processes that occur as a result of MDMA use are consistently reported, the significance and relevance of these changes for cognitive alterations remain a topic of ongoing debate34.
Limitations
First, the included studies reported results from various cognitive tests and employed heterogeneous research methods, including different dosages or times of drug administration. This heterogeneity limits the robustness of the presented results and reduces the certainty of our conclusion. However, we addressed this limitation by selecting the highest dosage and dosage closest to suspected peak effects for the meta-analysis and by combining multiple cognitive tests to load on the same factor, facilitating quantitative analysis (as is commonly done in other fields201). This enables us to draw conclusions despite methodological variety, although these conclusions should be considered as preliminary. Future studies should take care to report common protocols to allow for analysis of different phases of cognitive processing under psychedelics (e.g.183).
Secondly, the chosen methods did not allow for more fine-grained analyses of dose-dependent or time-dependent effects of psychedelics or MDMA on cognition. Future studies should aim to assess the effects of a broader range of psychedelic dosages (from micro- to macro-dosing) and across multiple timepoints.
Thirdly, we did not analyze the contextual setting of the psychedelic/MDMA session or the test administration. Since psychedelic effects are known to be highly influenced by context202, variations in the settings of the experience itself may have impacted the effects on cognition.
Fourth, blinding participants and researchers in studies involving the acute administration of psychoactive substances is a well-known challenge68. As indicated in the risk of bias assessment, most of the included studies did not achieve full blinding, which may have introduced strong expectancy effects, reducing the validity of the reported results. However, unlike in clinical studies, it remains unclear what participants' expectations are in cognitive assessments under the influence of psychoactive substances and how these expectations might influence actual test performance.
Fifth, we were not able to incorporate analyses taking into account previous drug exposure of participants. While most studies reported if their participants had ever used the investigated substance, only a fraction reported use frequency in a fashion that would allow for co-variate analysis. This lack of data could be an additional factor contributing to the high heterogeneity found in our results, especially considering the chronic tolerance reported by MDMA users27.
Sixth, while we reviewed a large number of studies in a qualitative manner only few studies could be included for quantitative analysis. Thus, our quantitative results are not representative of the literature at large but are based on a few select studies. Future studies in this field should take care to report as much of the raw data as possible to support more overarching analysis efforts like ours.
Finally, all included studies were conducted with healthy volunteers and did not include patient populations. As the effects of psychotropic medications sometimes can vary between the healthy and patient populations203, the same may be true for psychedelics and MDMA. Further studies are needed to investigate the cognitive effects of psychedelics and MDMA within the framework of psychedelic-assisted therapy.
Conclusion
This study is the first to contain a meta-analytic assessment of the acute cognitive effects of classic psychedelics, including a comparison with the effects of MDMA. Additionally, this is the largest synthesis of neuropsychological data for psychedelics as well as MDMA so far conducted. We report robust evidence that psychedelics acutely reduce attention, and preliminary evidence that psychedelics may also have acute detrimental effects on memory and executive functioning. In contrast, MDMA appears to have the strongest impact on memory performance, whereas attention and executive functions are less intensively affected in the acute phases. Conversely, during the sub-acute (‘afterglow’) period when acute effects have worn off, psychedelics potentially exert beneficial effects on executive functioning, while such effects have not been found for MDMA. Our findings add neuropsychological evidence for fostering distinct therapeutic approaches for psychedelics when compared to MDMA-assisted therapy. Given deteriorations of attention under psychedelics, psychotherapeutic interventions might be less fruitful during acute effects of the substance when compared to the subacute “afterglow” period. In contrast, given the specifics of acute effects of MDMA on cognition, psychotherapeutic techniques may be more fruitful during the acute experience when compared to psychedelics, whereas therapy during the subacute window might be somewhat less effective. In sum, there is a need for more research regarding cognitive underpinnings of classic psychedelics and MDMA with respect to prevention and for their use as tools for psychotherapy.
Supplementary Information
Acknowledgements
We thank all authors who shared their data upon request. Open access funding provided by the Open Access Publishing Fund of Philipps-Universität Marburg.
Author contributions
LAB, TGR, and TM conceptualized the study. LAB, and TGR acquired and curated the data. LAB, TGR and TM performed the qualitative assessment. NJH analyzed the quantitative data and created the corresponding figures. LAB, TGR and TM collaborated on the original draft. RK participated in interpretation of the data and provided research resources. EAEA assisted in creating the figures and participated in in interpretation of the data, while she and all other authors participated in writing and reviewing the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Data availability
The datasets used and analyzed during the current study are available in the GitHub repository: https://github.com/nickhaf/MDMA_Psychedelics_meta.
Code availability
The code for the analysis and the corresponding data are available in the GitHub repository: https://github.com/nickhaf/MDMA_Psychedelics_meta.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Lukas A. Basedow and Tomislav Majić.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-65391-9.
References
- 1.Preller KH, Vollenweider FX. Phenomenology, structure, and dynamic of psychedelic states. Curr. Top. Behav. Neurosci. 2018;36:221–256. doi: 10.1007/7854_2016_459. [DOI] [PubMed] [Google Scholar]
- 2.Vollenweider FX, Preller KH. Psychedelic drugs: Neurobiology and potential for treatment of psychiatric disorders. Nat. Rev. Neurosci. 2020;21:611–624. doi: 10.1038/s41583-020-0367-2. [DOI] [PubMed] [Google Scholar]
- 3.Barrett FS, Doss MK, Sepeda ND, Pekar JJ, Griffiths RR. Emotions and brain function are altered up to one month after a single high dose of psilocybin. Sci. Rep. 2020;10:2214. doi: 10.1038/s41598-020-59282-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Murphy-Beiner A, Soar K. Ayahuasca’s ‘afterglow’: Improved mindfulness and cognitive flexibility in ayahuasca drinkers. Psychopharmacology. 2020;237:1161–1169. doi: 10.1007/s00213-019-05445-3. [DOI] [PubMed] [Google Scholar]
- 5.Studerus E, Kometer M, Hasler F, Vollenweider FX. Acute, subacute and long-term subjective effects of psilocybin in healthy humans: A pooled analysis of experimental studies. J. Psychopharmacol. Oxf. Engl. 2011;25:1434–1452. doi: 10.1177/0269881110382466. [DOI] [PubMed] [Google Scholar]
- 6.Evens R, Schmidt ME, Majić T, Schmidt TT. The psychedelic afterglow phenomenon: A systematic review of subacute effects of classic serotonergic psychedelics. Ther. Adv. Psychopharmacol. 2023;13:20451253231172256. doi: 10.1177/20451253231172254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Majić T, Schmidt TT, Gallinat J. Peak experiences and the afterglow phenomenon: When and how do therapeutic effects of hallucinogens depend on psychedelic experiences? J. Psychopharmacol. Oxf. Engl. 2015;29:241–253. doi: 10.1177/0269881114568040. [DOI] [PubMed] [Google Scholar]
- 8.Basedow LA, Riemer TG, Reiche S, Kreutz R, Majić T. Neuropsychological functioning in users of serotonergic psychedelics—A systematic review and meta-analysis. Front. Pharmacol. 2021;12:739966. doi: 10.3389/fphar.2021.739966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Halpern JH, Pope HG. Do hallucinogens cause residual neuropsychological toxicity? Drug Alcohol Depend. 1999;53:247–256. doi: 10.1016/S0376-8716(98)00129-X. [DOI] [PubMed] [Google Scholar]
- 10.Bălăeţ M. Psychedelic cognition—The unreached frontier of psychedelic science. Front. Neurosci. 2022;16:832375. doi: 10.3389/fnins.2022.832375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bender D, Hellerstein DJ. Assessing the risk–benefit profile of classical psychedelics: A clinical review of second-wave psychedelic research. Psychopharmacology. 2022;239:1907–1932. doi: 10.1007/s00213-021-06049-6. [DOI] [PubMed] [Google Scholar]
- 12.Johnstad PG. Who is the typical psychedelics user? Methodological challenges for research in psychedelics use and its consequences. Nord. Alkohol-Nark. NAT. 2021;38:35–49. doi: 10.1177/1455072520963787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kangaslampi S. Association between mystical-type experiences under psychedelics and improvements in well-being or mental health—A comprehensive review of the evidence. J. Psychedelic Stud. 2023;7:18–28. doi: 10.1556/2054.2023.00243. [DOI] [Google Scholar]
- 14.Yaden DB, Griffiths RR. The subjective effects of psychedelics are necessary for their enduring therapeutic effects. ACS Pharmacol. Transl. Sci. 2021;4:568–572. doi: 10.1021/acsptsci.0c00194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ko K, Knight G, Rucker JJ, Cleare AJ. Psychedelics, mystical experience, and therapeutic efficacy: A systematic review. Front. Psychiatry. 2022;13:917199. doi: 10.3389/fpsyt.2022.917199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pahnke WN, Kurland AA, Unger S, Savage C, Grof S. The experimental use of psychedelic (LSD) psychotherapy. JAMA. 1970;212:1856–1863. doi: 10.1001/jama.1970.03170240060010. [DOI] [PubMed] [Google Scholar]
- 17.Gorman I, Nielson EM, Molinar A, Cassidy K, Sabbagh J. Psychedelic harm reduction and integration: A transtheoretical model for clinical practice. Front. Psychol. 2021;12:645246. doi: 10.3389/fpsyg.2021.645246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Anderson BT, et al. Psilocybin-assisted group therapy for demoralized older long-term AIDS survivor men: An open-label safety and feasibility pilot study. eClinicalMedicine. 2020;27:100538. doi: 10.1016/j.eclinm.2020.100538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Breeksema JJ, et al. Adverse events in clinical treatments with serotonergic psychedelics and MDMA: A mixed-methods systematic review. J. Psychopharmacol. 2022;36:1100–1117. doi: 10.1177/02698811221116926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chi T, Gold JA. A review of emerging therapeutic potential of psychedelic drugs in the treatment of psychiatric illnesses. J. Neurol. Sci. 2020;411:116715. doi: 10.1016/j.jns.2020.116715. [DOI] [PubMed] [Google Scholar]
- 21.Hysek CM, et al. Duloxetine inhibits effects of MDMA (‘ecstasy’) in vitro and in humans in a randomized placebo-controlled laboratory study. PLoS ONE. 2012;7:e36476. doi: 10.1371/journal.pone.0036476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liechti ME, Saur MR, Gamma A, Hell D, Vollenweider FX. Psychological and physiological effects of MDMA (‘Ecstasy’) after pretreatment with the 5-HT(2) antagonist ketanserin in healthy humans. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2000;23:396–404. doi: 10.1016/S0893-133X(00)00126-3. [DOI] [PubMed] [Google Scholar]
- 23.Schlag AK, Aday J, Salam I, Neill JC, Nutt DJ. Adverse effects of psychedelics: From anecdotes and misinformation to systematic science. J. Psychopharmacol. 2022;36:258–272. doi: 10.1177/02698811211069100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Henríquez-Hernández LA, Rojas-Hernández J, Quintana-Hernández DJ, Borkel LF. Hofmann vs. paracelsus: Do psychedelics defy the basics of toxicology?—A systematic review of the main ergolamines, simple tryptamines, and phenylethylamines. Toxics. 2023;11:148. doi: 10.3390/toxics11020148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Steele TD, McCANN UD, Ricaurte GA. 3,4-Methylenedioxymethamphetamine (MDMA, “Ecstasy”): Pharmacology and toxicology in animals and humans. Addiction. 1994;89:539–551. doi: 10.1111/j.1360-0443.1994.tb03330.x. [DOI] [PubMed] [Google Scholar]
- 26.Holze F, et al. Distinct acute effects of LSD, MDMA, and d-amphetamine in healthy subjects. Neuropsychopharmacology. 2020;45:462–471. doi: 10.1038/s41386-019-0569-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Parrott AC. Human psychobiology of MDMA or ‘Ecstasy’: An overview of 25 years of empirical research. Hum. Psychopharmacol. Clin. Exp. 2013;28:289–307. doi: 10.1002/hup.2318. [DOI] [PubMed] [Google Scholar]
- 28.Gouzoulis-Mayfrank E, Daumann J. Neurotoxicity of methylenedioxyamphetamines (MDMA; ecstasy) in humans: How strong is the evidence for persistent brain damage? Addict. Abingdon Engl. 2006;101:348–361. doi: 10.1111/j.1360-0443.2006.01314.x. [DOI] [PubMed] [Google Scholar]
- 29.Zakzanis KK, Campbell Z, Jovanovski D. The neuropsychology of ecstasy (MDMA) use: A quantitative review. Hum. Psychopharmacol. Clin. Exp. 2007;22:427–435. doi: 10.1002/hup.873. [DOI] [PubMed] [Google Scholar]
- 30.Kalechstein AD, De La Garza R, Mahoney JJ, Fantegrossi WE, Newton TF. MDMA use and neurocognition: A meta-analytic review. Psychopharmacology. 2007;189:531–537. doi: 10.1007/s00213-006-0601-2. [DOI] [PubMed] [Google Scholar]
- 31.Nulsen CE, Fox AM, Hammond GR. Differential effects of ecstasy on short-term and working memory: A meta-analysis. Neuropsychol. Rev. 2010;20:21–32. doi: 10.1007/s11065-009-9124-z. [DOI] [PubMed] [Google Scholar]
- 32.Halpern JH, et al. Reply to Parrott (2011), Fisk et al. (2011) and Rodgers et al. (2011) Addiction. 2011;106:1270–1372. doi: 10.1111/j.1360-0443.2011.03481.x. [DOI] [Google Scholar]
- 33.Doblin R, et al. A reconsideration and response to Parrott AC (2013) “Human psychobiology of MDMA or ‘Ecstasy’: An overview of 25 years of empirical research”. Hum. Psychopharmacol. Clin. Exp. 2014;29:105–108. doi: 10.1002/hup.2389. [DOI] [PubMed] [Google Scholar]
- 34.Montgomery C, Roberts CA. Neurological and cognitive alterations induced by MDMA in humans. Exp. Neurol. 2022;347:113888. doi: 10.1016/j.expneurol.2021.113888. [DOI] [PubMed] [Google Scholar]
- 35.Chan G, et al. An age-period-cohort analysis of trends in psychedelic and ecstasy use in the Australian population. Addict. Behav. 2022;127:107216. doi: 10.1016/j.addbeh.2021.107216. [DOI] [PubMed] [Google Scholar]
- 36.Winstock, A. R. et al. Global Drug Survey (GDS) 2020 Psychedelics Key Findings Report.https://www.globaldrugsurvey.com/ (2021).
- 37.Uosukainen H, Tacke U, Winstock A. Self-reported prevalence of dependence of MDMA compared to cocaine, mephedrone and ketamine among a sample of recreational poly-drug users. Int. J. Drug Policy. 2015;26:78–83. doi: 10.1016/j.drugpo.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 38.Leung SY. Benzodiazepines, opioids and driving: An overview of the experimental research. Drug Alcohol Rev. 2011;30:281–286. doi: 10.1111/j.1465-3362.2011.00311.x. [DOI] [PubMed] [Google Scholar]
- 39.Tzambazis K, Stough C. Alcohol impairs speed of information processing and simple and choice reaction time and differentially impairs higher-order cognitive abilities. Alcohol Alcohol. 2000;35:197–201. doi: 10.1093/alcalc/35.2.197. [DOI] [PubMed] [Google Scholar]
- 40.Maylor EA, Rabbitt PMA. Alcohol, reaction time and memory: A meta-analysis. Br. J. Psychol. 1993;84:301–317. doi: 10.1111/j.2044-8295.1993.tb02485.x. [DOI] [PubMed] [Google Scholar]
- 41.Koelega HS. Alcohol and vigilance performance: A review. Psychopharmacology. 1995;118:233–249. doi: 10.1007/BF02245951. [DOI] [PubMed] [Google Scholar]
- 42.Koelega HS. Benzodiazepines and vigilance performance: A review. Psychopharmacology. 1989;98:145–156. doi: 10.1007/BF00444684. [DOI] [PubMed] [Google Scholar]
- 43.Fromme K, Katz E, D’Amico E. Effects of alcohol intoxication on the perceived consequences of risk taking. Exp. Clin. Psychopharmacol. 1997;5:14–23. doi: 10.1037/1064-1297.5.1.14. [DOI] [PubMed] [Google Scholar]
- 44.Finnigan F, Hammersley R, Cooper T. An examination of next-day hangover effects after a 100 mg/100 ml dose of alcohol in heavy social drinkers. Addiction. 1998;93:1829–1838. doi: 10.1046/j.1360-0443.1998.931218298.x. [DOI] [PubMed] [Google Scholar]
- 45.Riccio CA, Waldrop JJM, Reynolds CR, Lowe P. Effects of stimulants on the continuous performance test (CPT) J. Neuropsychiatry Clin. Neurosci. 2001;13:326–335. doi: 10.1176/jnp.13.3.326. [DOI] [PubMed] [Google Scholar]
- 46.MacQueen DA, et al. Amphetamine improves mouse and human attention in the 5-choice continuous performance test. Neuropharmacology. 2018;138:87–96. doi: 10.1016/j.neuropharm.2018.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Koelega HS. Stimulant drugs and vigilance performance: A review. Psychopharmacology. 1993;111:1–16. doi: 10.1007/BF02257400. [DOI] [PubMed] [Google Scholar]
- 48.Grant JD, et al. Subjective reactions to cocaine and marijuana are associated with abuse and dependence. Addict. Behav. 2005;30:1574–1586. doi: 10.1016/j.addbeh.2005.02.007. [DOI] [PubMed] [Google Scholar]
- 49.Penning R, Veldstra JL, Daamen AP, Olivier B, Verster JC. Drugs of abuse, driving and traffic safety. Curr. Drug Abuse Rev. 2010;3:23–32. doi: 10.2174/1874473711003010023. [DOI] [PubMed] [Google Scholar]
- 50.Kashyap H, Mehta UM, Reddy RP, Bharath RD. Role of cognitive control in psychotherapy: An integrated review. Indian J. Psychol. Med. 2022 doi: 10.1177/02537176221128611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nijdam MJ, de Vries G-J, Gersons BPR, Olff M. Response to psychotherapy for posttraumatic stress disorder: The role of pretreatment verbal memory performance. J. Clin. Psychiatry. 2015;76:18178. doi: 10.4088/JCP.14m09438. [DOI] [PubMed] [Google Scholar]
- 52.Scott JC, et al. Verbal memory functioning moderates psychotherapy treatment response for PTSD-Related nightmares. Behav. Res. Ther. 2017;91:24–32. doi: 10.1016/j.brat.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 53.Carreira K, et al. A controlled evaluation of monthly maintenance interpersonal psychotherapy in late-life depression with varying levels of cognitive function. Int. J. Geriatr. Psychiatry. 2008;23:1110–1113. doi: 10.1002/gps.2031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Johnco C, Wuthrich VM, Rapee RM. The influence of cognitive flexibility on treatment outcome and cognitive restructuring skill acquisition during cognitive behavioural treatment for anxiety and depression in older adults: Results of a pilot study. Behav. Res. Ther. 2014;57:55–64. doi: 10.1016/j.brat.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 55.Hutten NRPW, Mason NL, Dolder PC, Kuypers KPC. Self-rated effectiveness of microdosing with psychedelics for mental and physical health problems among microdosers. Front. Psychiatry. 2019;10:672. doi: 10.3389/fpsyt.2019.00672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hutten NRPW, Mason NL, Dolder PC, Kuypers KPC. Motives and side-effects of microdosing with psychedelics among users. Int. J. Neuropsychopharmacol. 2019;22:426–434. doi: 10.1093/ijnp/pyz029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Haijen ECHM, Hurks PPM, Kuypers KPC. Microdosing with psychedelics to self-medicate for ADHD symptoms in adults: A prospective naturalistic study. Neurosci. Appl. 2022;1:101012. doi: 10.1016/j.nsa.2022.101012. [DOI] [Google Scholar]
- 58.Higgins JPT, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.NHLBI. Assessing Cardiovascular Risk: Systematic Evidence Review from the Risk Assessment Work Group. 139 https://www.nhlbi.nih.gov/health-topics/assessing-cardiovascular-risk (2013).
- 60.Goff DC, et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk. J. Am. Coll. Cardiol. 2014;63:2935–2959. doi: 10.1016/j.jacc.2013.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Harrer M, Cuijpers P, Furukawa T, Ebert D. Doing Meta-Analysis With R: A Hands-On Guide. Chapman & Hall/CRC Press; 2021. [Google Scholar]
- 62.de la Torre R, et al. Pharmacology of MDMA in humans. Ann. N. Y. Acad. Sci. 2000;914:225–237. doi: 10.1111/j.1749-6632.2000.tb05199.x. [DOI] [PubMed] [Google Scholar]
- 63.Passie T, Seifert J, Schneider U, Emrich HM. The pharmacology of psilocybin. Addict. Biol. 2002;7:357–364. doi: 10.1080/1355621021000005937. [DOI] [PubMed] [Google Scholar]
- 64.Dolder PC, Schmid Y, Haschke M, Rentsch KM, Liechti ME. Pharmacokinetics and concentration-effect relationship of oral LSD in humans. Int. J. Neuropsychopharmacol. 2015;19:pyv072. doi: 10.1093/ijnp/pyv072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.R Core Team. R version 4.3.2. (2023).
- 66.Harrer, M., Cuijpers, P., Furukawa, T. & Ebert, D. D. Dmetar: Companion R Package For The Guide ‘Doing Meta-Analysis in R’. (2019).
- 67.Viechtbauer, W. metafor: Meta-Analysis Package for R. (2023).
- 68.Aday JS, et al. Great Expectations: Recommendations for improving the methodological rigor of psychedelic clinical trials. Psychopharmacology. 2022 doi: 10.1007/s00213-022-06123-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Daumann J, et al. Pharmacological modulation of the neural basis underlying inhibition of return (IOR) in the human 5-HT2A agonist and NMDA antagonist model of psychosis. Psychopharmacology. 2008;200:573–583. doi: 10.1007/s00213-008-1237-1. [DOI] [PubMed] [Google Scholar]
- 70.Vollenweider FX, Csomor PA, Knappe B, Geyer MA, Quednow BB. The effects of the preferential 5-HT2A agonist psilocybin on prepulse inhibition of startle in healthy human volunteers depend on interstimulus interval. Neuropsychopharmacology. 2007;32:1876–1887. doi: 10.1038/sj.npp.1301324. [DOI] [PubMed] [Google Scholar]
- 71.Barrett FS, Carbonaro TM, Hurwitz E, Johnson MW, Griffiths RR. Double-blind comparison of the two hallucinogens psilocybin and dextromethorphan: Effects on cognition. Psychopharmacology. 2018;235:2915–2927. doi: 10.1007/s00213-018-4981-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Heim E, Heimann H, Lukács G. Die psychische Wirkung der mexikanischen Droge “Ololiuqui“ am Menschen. Psychopharmacologia. 1968;13:35–48. doi: 10.1007/BF00401617. [DOI] [PubMed] [Google Scholar]
- 73.Wittmann M, et al. Effects of psilocybin on time perception and temporal control of behaviour in humans. J. Psychopharmacol. 2007;21:50–64. doi: 10.1177/0269881106065859. [DOI] [PubMed] [Google Scholar]
- 74.Sloane B, Doust JWL. Psychophysiological investigations in experimental psychoses: Results of the exhibition of d-lysergic acid diethylamide to psychiatric patients. J. Ment. Sci. 1954;100:129–144. doi: 10.1192/bjp.100.418.129. [DOI] [PubMed] [Google Scholar]
- 75.Silverstein AB, Klee GD. The effect of lysergic acid diethylamide on digit span. J. Clin. Exp. Psychopathol. Q. Rev. Psychiatry Neurol. 1960;21:11–14. [PubMed] [Google Scholar]
- 76.Hasler F, Grimberg U, Benz MA, Huber T, Vollenweider FX. Acute psychological and physiological effects of psilocybin in healthy humans: A double-blind, placebo-controlled dose?effect study. Psychopharmacology. 2004;172:145–156. doi: 10.1007/s00213-003-1640-6. [DOI] [PubMed] [Google Scholar]
- 77.Gouzoulis-Mayfrank E, et al. Inhibition of return in the human 5HT2A agonist and NMDA antagonist model of psychosis. Neuropsychopharmacology. 2006;31:431–441. doi: 10.1038/sj.npp.1300882. [DOI] [PubMed] [Google Scholar]
- 78.Heekeren K, et al. Mismatch negativity generation in the human 5HT2A agonist and NMDA antagonist model of psychosis. Psychopharmacology. 2008;199:77–88. doi: 10.1007/s00213-008-1129-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bouso JC, Fábregas JM, Antonijoan RM, Rodríguez-Fornells A, Riba J. Acute effects of ayahuasca on neuropsychological performance: Differences in executive function between experienced and occasional users. Psychopharmacology. 2013;230:415–424. doi: 10.1007/s00213-013-3167-9. [DOI] [PubMed] [Google Scholar]
- 80.Primac DW. Effects of centrally acting drugs on two tests of brain damage. Arch. Neurol. Psychiatry. 1957;77:328. doi: 10.1001/archneurpsyc.1957.02330330114018. [DOI] [PubMed] [Google Scholar]
- 81.Kornetsky C. Comparison of psychological effects of certain centrally acting drugs in man. Arch. Neurol. Psychiatry. 1957;77:318. doi: 10.1001/archneurpsyc.1957.02330330104016. [DOI] [PubMed] [Google Scholar]
- 82.Jarvik ME, Abramson HA, Hirsch MW. Lysergic acid diethylamide (LSD-25): VI. Effect upon recall and recognition of various stimuli. J. Psychol. 1955;39:443–454. doi: 10.1080/00223980.1955.9916194. [DOI] [Google Scholar]
- 83.Snyder SH, Faillace LA, Weingartner H. DOM (STP), a new hallucinogenic drug, and DOET: Effects in normal subjects. Am. J. Psychiatry. 1968;125:113–120. doi: 10.1176/ajp.125.3.357. [DOI] [PubMed] [Google Scholar]
- 84.Weingartner H, Snyder SH, Faillace LA. DOM (STP), a new hallucinogenic drug: Specific perceptual changes. J. Clin. Pharmacol. New Drugs. 1971;11:103–111. doi: 10.1177/009127007101100205. [DOI] [PubMed] [Google Scholar]
- 85.Aronson H, Watermann CE, Klee GD. The effect of d-lysergic acid diethylamide (LSD-25) on learning and retention. J. Clin. Exp. Psychopathol. Q. Rev. Psychiatry Neurol. 1962;23:17–23. [PubMed] [Google Scholar]
- 86.Silverstein AB. Effects of lysergic acid diethylamide (LSD-25) on intellectual functions. Arch. Neurol. Psychiatry. 1958;80:477. doi: 10.1001/archneurpsyc.1958.02340100077018. [DOI] [PubMed] [Google Scholar]
- 87.Pokorny T, Duerler P, Seifritz E, Vollenweider FX, Preller KH. LSD acutely impairs working memory, executive functions, and cognitive flexibility, but not risk-based decision-making. Psychol. Med. 2019 doi: 10.1017/S0033291719002393. [DOI] [PubMed] [Google Scholar]
- 88.Goldberger L. Cognitive test performance under LSD-25, placebo and isolation. J. Nerv. Ment. Dis. 1966;142:4–9. doi: 10.1097/00005053-196601000-00002. [DOI] [PubMed] [Google Scholar]
- 89.Lienert GA. Mental age regression induced by lysergic acid diethylamide. J. Psychol. 1966;63:3–11. doi: 10.1080/00223980.1966.10544806. [DOI] [PubMed] [Google Scholar]
- 90.Safer J. The effect of LSD on sleep-deprived men. Psychopharmacologia. 1970;17:414–424. doi: 10.1007/BF00403812. [DOI] [PubMed] [Google Scholar]
- 91.Brengelmann JC. Effects of LSD-25 on tests of personality. J. Ment. Sci. 1958;104:1226–1236. doi: 10.1192/bjp.104.437.1226. [DOI] [PubMed] [Google Scholar]
- 92.Barendregt JT. Performance on some objective tests under LSD-25. In: Jores A, Freyberger H, Stokvis B, editors. Advances in Psychosomatic Medicine. S. Karger AG; 1959. pp. 217–219. [DOI] [PubMed] [Google Scholar]
- 93.Mallaroni P, et al. Assessment of the acute effects of 2C-B vs. psilocybin on subjective experience, mood, and cognition. Clin. Pharmacol. Ther. 2023 doi: 10.1002/cpt.2958. [DOI] [PubMed] [Google Scholar]
- 94.Vollenweider FX, Vollenweider-Scherpenhuyzen MFI, Bäbler A, Vogel H, Hell D. Psilocybin induces schizophrenia-like psychosis in humans via a serotonin-2 agonist action. NeuroReport. 1998;9:3897–3902. doi: 10.1097/00001756-199812010-00024. [DOI] [PubMed] [Google Scholar]
- 95.Carter OL, et al. Using psilocybin to investigate the relationship between attention, working memory, and the serotonin 1A and 2A receptors. J. Cogn. Neurosci. 2005;17:1497–1508. doi: 10.1162/089892905774597191. [DOI] [PubMed] [Google Scholar]
- 96.Nikolič M, et al. Psilocybin intoxication did not affect daytime or sleep-related declarative memory consolidation in a small sample exploratory analysis. Eur. Neuropsychopharmacol. 2023;74:78–88. doi: 10.1016/j.euroneuro.2023.04.019. [DOI] [PubMed] [Google Scholar]
- 97.Kuypers KPC, et al. Ayahuasca enhances creative divergent thinking while decreasing conventional convergent thinking. Psychopharmacology. 2016;233:3395–3403. doi: 10.1007/s00213-016-4377-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ramaekers J, et al. Altered state of consciousness and mental imagery as a function of N,N-dimethyltryptamine concentration in ritualistic ayahuasca users. J. Cogn. Neurosci. 2023 doi: 10.1162/jocn_a_02003. [DOI] [PubMed] [Google Scholar]
- 99.Wießner I, et al. LSD and creativity: Increased novelty and symbolic thinking, decreased utility and convergent thinking. J. Psychopharmacol. 2022;36:348–359. doi: 10.1177/02698811211069113. [DOI] [PubMed] [Google Scholar]
- 100.Schmidt A, et al. Acute LSD effects on response inhibition neural networks. Psychol. Med. 2018;48:1464–1473. doi: 10.1017/S0033291717002914. [DOI] [PubMed] [Google Scholar]
- 101.Zegans LS. The effects of LSD-25 on creativity and tolerance to regression. Arch. Gen. Psychiatry. 1967;16:740. doi: 10.1001/archpsyc.1967.01730240096014. [DOI] [PubMed] [Google Scholar]
- 102.Resnick O, Krus DM, Raskin M. LSD-25 action in normal subjects treated with a monoamine oxidase inhibitor. Life Sci. 1964;3:1207–1214. doi: 10.1016/0024-3205(64)90037-2. [DOI] [PubMed] [Google Scholar]
- 103.Resnick O, Krus DM, Raskin M. Accentuation of the psychological effects of LSD-25 in normal subjects treated with reserpine. Life Sci. 1965;4:1433–1437. doi: 10.1016/0024-3205(65)90022-6. [DOI] [PubMed] [Google Scholar]
- 104.Ostfeld AM. Effects of LSD 25 and JB 318 on tests of visual and perceptual functions in man. Fed. Proc. 1961;20:876–884. [PubMed] [Google Scholar]
- 105.Kanen JW, et al. Effect of lysergic acid diethylamide (LSD) on reinforcement learning in humans. Psychol. Med. 2022 doi: 10.1017/S0033291722002963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Cohen BD. Comparison of phencyclidine hydrochloride (sernyl) with other drugs: Simulation of schizophrenic performance with phencyclidine hydrochloride (Sernyl), lysergic acid diethylamide (LSD-25), and amobarbital (Amytal) sodium; II. Symbolic and sequential thinking. Arch. Gen. Psychiatry. 1962;6:395. doi: 10.1001/archpsyc.1962.01710230063007. [DOI] [PubMed] [Google Scholar]
- 107.Wapner S, Krus DM. Effects of lysergic acid diethylamide, and differences between normals and schizophrenics on the Stroop Color-Word Test. J. Neuropsychiatry. 1960;2:76–81. [PubMed] [Google Scholar]
- 108.Harman WW, Mckim RH, Mogar RE, Fadiman J. Psychedelic agents in creative problem-solving: A pilot. Psychol. Rep. 1966;19:211–227. doi: 10.2466/pr0.1966.19.1.211. [DOI] [PubMed] [Google Scholar]
- 109.Duke RB, Keeler MH. The effects of Psilocybin, dextro-amphetamine and placebo on performance of the trail making test. J. Clin. Psychol. 1968;24:316–317. doi: 10.1002/1097-4679(196807)24:3<316::AID-JCLP2270240310>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 110.Quednow BB, Kometer M, Geyer MA, Vollenweider FX. Psilocybin-induced deficits in automatic and controlled inhibition are attenuated by ketanserin in healthy human volunteers. Neuropsychopharmacology. 2012;37:630–640. doi: 10.1038/npp.2011.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Mason NL, et al. Spontaneous and deliberate creative cognition during and after psilocybin exposure. Transl. Psychiatry. 2021;11:1–13. doi: 10.1038/s41398-021-01335-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kometer M, et al. Psilocybin biases facial recognition, goal-directed behavior, and mood state toward positive relative to negative emotions through different serotonergic subreceptors. Biol. Psychiatry. 2012;72:898–906. doi: 10.1016/j.biopsych.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 113.Heekeren K, et al. Prepulse inhibition of the startle reflex and its attentional modulation in the human S-ketamine and N, N-dimethyltryptamine (DMT) models of psychosis. J. Psychopharmacol. 2007;21:312–320. doi: 10.1177/0269881107077734. [DOI] [PubMed] [Google Scholar]
- 114.Dittrich A. Alteration of behavioural changes induced by 3,4,5-trimethoxyphenylethylamine (mescaline) by pretreatment with 2,4,5-trimethoxyphenylethylamine: A self-experiment. Psychopharmacologia. 1971;21:229–237. doi: 10.1007/BF00403861. [DOI] [PubMed] [Google Scholar]
- 115.Rosenbaum G. Comparison of sernyl with other drugs: Simulation of schizophrenic performance with sernyl, LSD-25, and amobarbital (amytal) sodium; I. Attention, motor function, and proprioception. AMA Arch. Gen. Psychiatry. 1959;1:651. doi: 10.1001/archpsyc.1959.03590060113013. [DOI] [PubMed] [Google Scholar]
- 116.Abramson HA, Jarvik ME, Hirsch MW. Lysergic acid diethylamide (Lsd-25): X. Effect on reaction time to auditory and visual stimuli. J. Psychol. 1955;40:39–52. doi: 10.1080/00223980.1955.9712962. [DOI] [Google Scholar]
- 117.Netz B, Jonsson C-O, Bergqvist S. Effects of lysergic acid diethylamide (LSD-25) on normal subjects in a schizophrenia-discriminating test battery. Scand. J. Psychol. 1963;4:143–148. doi: 10.1111/j.1467-9450.1963.tb01321.x. [DOI] [Google Scholar]
- 118.Sjoerdsma A, Kornetsky C, Evarts E. Lysergic acid diethylamide in patients withk excess serotonin. Arch. Neurol. Psychiatry. 1956;75:488–492. doi: 10.1001/archneurpsyc.1956.02330230038003. [DOI] [PubMed] [Google Scholar]
- 119.Gouzoulis-Mayfrank E, et al. Effects of the hallucinogen psilocybin on covert orienting of visual attention in humans. Neuropsychobiology. 2002;45:205–212. doi: 10.1159/000063672. [DOI] [PubMed] [Google Scholar]
- 120.Umbricht D, et al. Effects of the 5-HT2A agonist psilocybin on mismatch negativity generation and AX-continuous performance task: Implications for the neuropharmacology of cognitive deficits in schizophrenia. Neuropsychopharmacology. 2003;28:170–181. doi: 10.1038/sj.npp.1300005. [DOI] [PubMed] [Google Scholar]
- 121.Thatcher K, Wiederholt WC, Fischer R. An electroencephalographic analysis of personality-dependent performance under psilocybin. Agents Actions. 1971;2:21–26. doi: 10.1007/BF01965376. [DOI] [PubMed] [Google Scholar]
- 122.Spitzer M, et al. Increased activation of indirect semantic associations under psilocybin. Biol. Psychiatry. 1996;39:1055–1057. doi: 10.1016/0006-3223(95)00418-1. [DOI] [PubMed] [Google Scholar]
- 123.Rynearson RR, Wilson MR, Bickford RG. Psilocybin-induced changes in psychologic function, electroencephalogram, and light-evoked potentials in human subjects. Mayo Clin. Proc. 1968;43:191–204. [PubMed] [Google Scholar]
- 124.Dumont G, et al. Acute psychomotor effects of MDMA and ethanol (co-) administration over time in healthy volunteers. J. Psychopharmacol. 2010;24:155–164. doi: 10.1177/0269881108099214. [DOI] [PubMed] [Google Scholar]
- 125.Kuypers KPC, Wingen M, Samyn N, Limbert N, Ramaekers JG. Acute effects of nocturnal doses of MDMA on measures of impulsivity and psychomotor performance throughout the night. Psychopharmacology. 2007;192:111–119. doi: 10.1007/s00213-006-0679-6. [DOI] [PubMed] [Google Scholar]
- 126.Kuypers KPC, Wingen M, Ramaekers JG. Memory and mood during the night and in the morning after repeated evening doses of MDMA. J. Psychopharmacol. 2008 doi: 10.1177/0269881107083990. [DOI] [PubMed] [Google Scholar]
- 127.Bosker WM, Kuypers KPC, Conen S, Ramaekers JG. Dose-related effects of MDMA on psychomotor function and mood before, during, and after a night of sleep loss. Psychopharmacology. 2010;209:69–76. doi: 10.1007/s00213-009-1767-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Gamma A. 3,4-Methylenedioxymethamphetamine (MDMA) modulates cortical and limbic brain activity as measured by [H215O]-PET in healthy humans. Neuropsychopharmacology. 2000;23:388–395. doi: 10.1016/S0893-133X(00)00130-5. [DOI] [PubMed] [Google Scholar]
- 129.Vollenweider FX. Psychological and cardiovascular effects and short-term sequelae of MDMA (“Ecstasy”) in MDMA-naïve healthy volunteers. Neuropsychopharmacology. 1998;19:11. doi: 10.1038/sj.npp.1395197. [DOI] [PubMed] [Google Scholar]
- 130.Ramaekers JG, Kuypers KPC. Acute effects of 3,4-methylenedioxymethamphetamine (MDMA) on behavioral measures of impulsivity: Alone and in combination with alcohol. Neuropsychopharmacology. 2006;31:1048–1055. doi: 10.1038/sj.npp.1300894. [DOI] [PubMed] [Google Scholar]
- 131.Parrott AC, Lasky J. Ecstasy (MDMA) effects upon mood and cognition: Before, during and after a Saturday night dance. Psychopharmacology. 1998;139:261–268. doi: 10.1007/s002130050714. [DOI] [PubMed] [Google Scholar]
- 132.Curran HV, Travill RA. Mood and cognitive effects of ± 3,4-methylenedioxymethamphetamine (MDMA, ‘ecstasy’): Week-end ‘high’ followed by mid-week low. Addiction. 1997;92:821–831. [PubMed] [Google Scholar]
- 133.de Sousa Fernandes Perna EB, et al. Memory and mood during MDMA intoxication, with and without memantine pretreatment. Neuropharmacology. 2014;87:198–205. doi: 10.1016/j.neuropharm.2014.03.008. [DOI] [PubMed] [Google Scholar]
- 134.Downing J. The psychological and physiological effects of MDMA on normal volunteers. J. Psychoactive Drugs. 1986;18:335–340. doi: 10.1080/02791072.1986.10472366. [DOI] [PubMed] [Google Scholar]
- 135.Dumont GJH, et al. Acute neuropsychological effects of MDMA and ethanol (co-)administration in healthy volunteers. Psychopharmacology. 2008;197:465–474. doi: 10.1007/s00213-007-1056-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kloft L, Otgaar H, Blokland A, Toennes SW, Ramaekers JG. Remembering Molly: Immediate and delayed false memory formation after acute MDMA exposure. Eur. Neuropsychopharmacol. 2022;57:59–68. doi: 10.1016/j.euroneuro.2022.01.005. [DOI] [PubMed] [Google Scholar]
- 137.Kuypers KPC, Ramaekers JG. Transient memory impairment after acute dose of 75mg 3.4-methylene-dioxymethamphetamine. J. Psychopharmacol. Oxf. Engl. 2005;19:633–639. doi: 10.1177/0269881105056670. [DOI] [PubMed] [Google Scholar]
- 138.Kuypers KP, Wingen M, Heinecke A, Formisano E, Ramaekers JG. MDMA intoxication and verbal memory performance: A placebo-controlled pharmaco-MRI study. J. Psychopharmacol. 2011;25:1053–1061. doi: 10.1177/0269881111405361. [DOI] [PubMed] [Google Scholar]
- 139.Kuypers KPC, et al. No evidence that MDMA-induced enhancement of emotional empathy is related to peripheral oxytocin levels or 5-HT1a receptor activation. PLoS ONE. 2014;9:e100719. doi: 10.1371/journal.pone.0100719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kuypers K, Torre R, Farre M, Pujadas M, Ramaekers J. Inhibition of MDMA-induced increase in cortisol does not prevent acute impairment of verbal memory. Br. J. Pharmacol. 2013;168:607–617. doi: 10.1111/j.1476-5381.2012.02196.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Stough C, et al. The acute effects of 3,4-methylenedioxymethamphetamine and d-methamphetamine on human cognitive functioning. Psychopharmacology. 2012;220:799–807. doi: 10.1007/s00213-011-2532-9. [DOI] [PubMed] [Google Scholar]
- 142.van Wel JHP, et al. Blockade of 5-HT2 receptor selectively prevents MDMA-induced verbal memory impairment. Neuropsychopharmacology. 2011;36:1932–1939. doi: 10.1038/npp.2011.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Ramaekers JG, Kuypers KPC, Wingen M, Heinecke A, Formisano E. Involvement of inferior parietal lobules in prospective memory impairment during acute MDMA (Ecstasy) intoxication: An event-related fMRI study. Neuropsychopharmacology. 2009;34:1641–1648. doi: 10.1038/npp.2008.219. [DOI] [PubMed] [Google Scholar]
- 144.Kirkpatrick MG, et al. A direct comparison of the behavioral and physiological effects of methamphetamine and 3,4-methylenedioxymethamphetamine (MDMA) in humans. Psychopharmacology. 2012;219:109–122. doi: 10.1007/s00213-011-2383-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Hoshi R, Pratt H, Mehta S, Bond AJ, Curran HV. An investigation into the sub-acute effects of ecstasy on aggressive interpretative bias and aggressive mood—are there gender differences? J. Psychopharmacol. 2006;20:291–301. doi: 10.1177/0269881106060505. [DOI] [PubMed] [Google Scholar]
- 146.Haijen E, et al. Peripheral endocannabinoid concentrations are not associated with verbal memory impairment during MDMA intoxication. Psychopharmacology. 2018;235:709–717. doi: 10.1007/s00213-017-4787-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Hasler F, Studerus E, Lindner K, Ludewig S, Vollenweider F. Investigation of serotonin-1A receptor function in the human psychopharmacology of MDMA. J. Psychopharmacol. 2009;23:923–935. doi: 10.1177/0269881108094650. [DOI] [PubMed] [Google Scholar]
- 148.Kuypers KPC, Ramaekers JG. Acute dose of MDMA (75 mg) impairs spatial memory for location but leaves contextual processing of visuospatial information unaffected. Psychopharmacology. 2006;189:557–563. doi: 10.1007/s00213-006-0321-7. [DOI] [PubMed] [Google Scholar]
- 149.Doss MK, Weafer J, Gallo DA, de Wit H. MDMA impairs both the encoding and retrieval of emotional recollections. Neuropsychopharmacology. 2018;43:791–800. doi: 10.1038/npp.2017.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Schmidt A, et al. Comparative effects of methylphenidate, modafinil, and MDMA on response inhibition neural networks in healthy subjects. Int. J. Neuropsychopharmacol. 2017;20:712–720. doi: 10.1093/ijnp/pyx037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Spronk DB, Dumont GJH, Verkes RJ, De Bruijn ERA. The acute effects of MDMA and ethanol administration on electrophysiological correlates of performance monitoring in healthy volunteers. Psychopharmacology. 2014;231:2877–2888. doi: 10.1007/s00213-014-3456-y. [DOI] [PubMed] [Google Scholar]
- 152.van Wel JHP, et al. Effects of acute MDMA intoxication on mood and impulsivity: Role of the 5-HT2 and 5-HT1 receptors. PLoS ONE. 2012;7:e40187. doi: 10.1371/journal.pone.0040187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Lamers CTJ, et al. Dissociable effects of a single dose of ecstasy (MDMA) on psychomotor skills and attentional performance. J. Psychopharmacol. 2003;17:379–387. doi: 10.1177/0269881103174015. [DOI] [PubMed] [Google Scholar]
- 154.Dumont G, et al. Acute psychomotor, memory and subjective effects of MDMA and THC co-administration over time in healthy volunteers. J. Psychopharmacol. 2011;25:478–489. doi: 10.1177/0269881110376687. [DOI] [PubMed] [Google Scholar]
- 155.Kuypers KPC, Samyn N, Ramaekers JG. MDMA and alcohol effects, combined and alone, on objective and subjective measures of actual driving performance and psychomotor function. Psychopharmacology. 2006;187:467–475. doi: 10.1007/s00213-006-0434-z. [DOI] [PubMed] [Google Scholar]
- 156.Prochazkova L, et al. Exploring the effect of microdosing psychedelics on creativity in an open-label natural setting. Psychopharmacology. 2018;235:3401–3413. doi: 10.1007/s00213-018-5049-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Bershad AK, Schepers ST, Bremmer MP, Lee R, de Wit H. Acute subjective and behavioral effects of microdoses of lysergic acid diethylamide in healthy human volunteers. Biol. Psychiatry. 2019;86:792–800. doi: 10.1016/j.biopsych.2019.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Family N, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of low dose lysergic acid diethylamide (LSD) in healthy older volunteers. Psychopharmacology. 2019 doi: 10.1007/s00213-019-05417-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Yanakieva S, et al. The effects of microdose LSD on time perception: A randomised, double-blind, placebo-controlled trial. Psychopharmacology. 2019;236:1159–1170. doi: 10.1007/s00213-018-5119-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Cavanna F, et al. Microdosing with psilocybin mushrooms: A double-blind placebo-controlled study. Transl. Psychiatry. 2022;12:307. doi: 10.1038/s41398-022-02039-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Wit H, Molla HM, Bershad A, Bremmer M, Lee R. Repeated low doses of LSD in healthy adults: A placebo-controlled, dose–response study. Addict. Biol. 2022;27:e13143. doi: 10.1111/adb.13143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Fallon, S. J. No Evidence That LSD Microdosing Affects Recall or the Balance between Distracter Resistance and Updating. 10.1101/2021.12.02.470935 (2021).
- 163.Hutten NRPW, et al. Mood and cognition after administration of low LSD doses in healthy volunteers: A placebo controlled dose-effect finding study. Eur. Neuropsychopharmacol. 2020;41:81–91. doi: 10.1016/j.euroneuro.2020.10.002. [DOI] [PubMed] [Google Scholar]
- 164.Kiraga MK, et al. Persisting effects of ayahuasca on empathy, creative thinking, decentering, personality, and well-being. Front. Pharmacol. 2021;12:721537. doi: 10.3389/fphar.2021.721537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Mason NL, Mischler E, Uthaug MV, Kuypers KPC. Sub-acute effects of psilocybin on empathy, creative thinking, and subjective well-being. J. Psychoactive Drugs. 2019;51:123–134. doi: 10.1080/02791072.2019.1580804. [DOI] [PubMed] [Google Scholar]
- 166.Ornelas IM, et al. Nootropic effects of LSD: Behavioral, molecular and computational evidence. Exp. Neurol. 2022;356:114148. doi: 10.1016/j.expneurol.2022.114148. [DOI] [PubMed] [Google Scholar]
- 167.Reckweg J, et al. A phase 1, dose-ranging study to assess safety and psychoactive effects of a vaporized 5-methoxy-N,N-dimethyltryptamine formulation (GH001) in healthy volunteers. Front. Pharmacol. 2021;12:760671. doi: 10.3389/fphar.2021.760671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Uthaug MV, et al. Sub-acute and long-term effects of ayahuasca on affect and cognitive thinking style and their association with ego dissolution. Psychopharmacology. 2018;235:2979–2989. doi: 10.1007/s00213-018-4988-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Uthaug MV, et al. A single inhalation of vapor from dried toad secretion containing 5-methoxy-N, N-dimethyltryptamine (5-MeO-DMT) in a naturalistic setting is related to sustained enhancement of satisfaction with life, mindfulness-related capacities, and a decrement of psychopathological symptoms. Psychopharmacology. 2019;236:2653–2666. doi: 10.1007/s00213-019-05236-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Wießner I, et al. LSD, afterglow and hangover: Increased episodic memory and verbal fluency, decreased cognitive flexibility. Eur. Neuropsychopharmacol. 2022;58:7–19. doi: 10.1016/j.euroneuro.2022.01.114. [DOI] [PubMed] [Google Scholar]
- 171.Pirona A, Morgan M. An investigation of the subacute effects of ecstasy on neuropsychological performance, sleep and mood in regular ecstasy users. J. Psychopharmacol. 2010;24:175–185. doi: 10.1177/0269881109102780. [DOI] [PubMed] [Google Scholar]
- 172.Palamar JJ. There’s something about Molly: The underresearched yet popular powder form of ecstasy in the United States. Subst. Abuse. 2017;38:15–17. doi: 10.1080/08897077.2016.1267070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Feduccia AA, Mithoefer MC. MDMA-assisted psychotherapy for PTSD: Are memory reconsolidation and fear extinction underlying mechanisms? Prog. Neuropsychopharmacol. Biol. Psychiatry. 2018;84:221–228. doi: 10.1016/j.pnpbp.2018.03.003. [DOI] [PubMed] [Google Scholar]
- 174.Kida S. Reconsolidation/destabilization, extinction and forgetting of fear memory as therapeutic targets for PTSD. Psychopharmacology. 2019;236:49–57. doi: 10.1007/s00213-018-5086-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Meister L, et al. Pharmacological memory modulation to augment trauma-focused psychotherapy for PTSD: A systematic review of randomised controlled trials. Transl. Psychiatry. 2023;13:1–9. doi: 10.1038/s41398-023-02495-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Luoma JB, Shahar B, Kati Lear M, Pilecki B, Wagner A. Potential processes of change in MDMA-Assisted therapy for social anxiety disorder: Enhanced memory reconsolidation, self-transcendence, and therapeutic relationships. Hum. Psychopharmacol. Clin. Exp. 2022;37:e2824. doi: 10.1002/hup.2824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Weiss B, Wingert A, Erritzoe D, Campbell WK. Prevalence and therapeutic impact of adverse life event reexperiencing under ceremonial ayahuasca. Sci. Rep. 2023;13:9438. doi: 10.1038/s41598-023-36184-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Healy CJ. The acute effects of classic psychedelics on memory in humans. Psychopharmacology. 2021;238:639–653. doi: 10.1007/s00213-020-05756-w. [DOI] [PubMed] [Google Scholar]
- 179.Holze F, et al. Direct comparison of the acute effects of lysergic acid diethylamide and psilocybin in a double-blind placebo-controlled study in healthy subjects. Neuropsychopharmacology. 2022;47:1180–1187. doi: 10.1038/s41386-022-01297-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Mason NL, et al. Me, myself, bye: Regional alterations in glutamate and the experience of ego dissolution with psilocybin. Neuropsychopharmacology. 2020 doi: 10.1038/s41386-020-0718-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Baylen CA, Rosenberg H. A review of the acute subjective effects of MDMA/ecstasy. Addiction. 2006;101:933–947. doi: 10.1111/j.1360-0443.2006.01423.x. [DOI] [PubMed] [Google Scholar]
- 182.Levine LJ, Pizarro DA. Emotion and memory research: A grumpy overview. Soc. Cogn. 2004;22:530–554. doi: 10.1521/soco.22.5.530.50767. [DOI] [Google Scholar]
- 183.Doss MK, de Wit H, Gallo DA. The acute effects of psychoactive drugs on emotional episodic memory encoding, consolidation, and retrieval: A comprehensive review. Neurosci. Biobehav. Rev. 2023;150:105188. doi: 10.1016/j.neubiorev.2023.105188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Fadiman J. The Psychedelic Explorer’s Guide: Safe, Therapeutic, and Sacred Journeys. Park Street Press; 2011. [DOI] [PubMed] [Google Scholar]
- 185.Sayalı C, Barrett FS. The costs and benefits of psychedelics on cognition and mood. Neuron. 2023;111:614–630. doi: 10.1016/j.neuron.2022.12.031. [DOI] [PubMed] [Google Scholar]
- 186.Baggott, M. J. Psychedelics and Creativity: A Review of the Quantitative Literature. https://peerj.com/preprints/1202 (2015) 10.7287/peerj.preprints.1202v1.
- 187.Torrado Pacheco A, Olson RJ, Garza G, Moghaddam B. Acute psilocybin enhances cognitive flexibility in rats. Neuropsychopharmacology. 2023 doi: 10.1038/s41386-023-01545-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Hibicke M, Kramer HM, Nichols CD. A single administration of psilocybin persistently rescues cognitive deficits caused by adolescent chronic restraint stress without long-term changes in synaptic protein gene expression in a rat experimental system with translational relevance to depression. Psychedelic Med. 2023;1:54–67. doi: 10.1089/psymed.2022.0012. [DOI] [Google Scholar]
- 189.Doss MK, et al. Psilocybin therapy increases cognitive and neural flexibility in patients with major depressive disorder. Transl. Psychiatry. 2021;11:574. doi: 10.1038/s41398-021-01706-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Bouso JC, et al. Personality, psychopathology, life attitudes and neuropsychological performance among ritual users of ayahuasca: A longitudinal study. PLoS ONE. 2012;7:e42421. doi: 10.1371/journal.pone.0042421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Bouso JC, et al. Long-term use of psychedelic drugs is associated with differences in brain structure and personality in humans. Eur. Neuropsychopharmacol. 2015;25:483–492. doi: 10.1016/j.euroneuro.2015.01.008. [DOI] [PubMed] [Google Scholar]
- 192.McCulloch DE-W, et al. Lasting effects of a single psilocybin dose on resting-state functional connectivity in healthy individuals. J. Psychopharmacol. 2022;36:74–84. doi: 10.1177/02698811211026454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Pasquini L, Palhano-Fontes F, Araujo DB. Subacute effects of the psychedelic ayahuasca on the salience and default mode networks. J. Psychopharmacol. 2020;34:623–635. doi: 10.1177/0269881120909409. [DOI] [PubMed] [Google Scholar]
- 194.Calder AE, Hasler G. Towards an understanding of psychedelic-induced neuroplasticity. Neuropsychopharmacology. 2023;48:104–112. doi: 10.1038/s41386-022-01389-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Nardou R, et al. Oxytocin-dependent reopening of a social reward learning critical period with MDMA. Nature. 2019;569:116–120. doi: 10.1038/s41586-019-1075-9. [DOI] [PubMed] [Google Scholar]
- 196.Nardou R, et al. Psychedelics reopen the social reward learning critical period. Nature. 2023;618:790–798. doi: 10.1038/s41586-023-06204-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Reneman L, et al. The acute and chronic effects of MDMA (“Ecstasy”) on cortical 5-HT2A receptors in rat and human brain. Neuropsychopharmacology. 2002;26:387–396. doi: 10.1016/S0893-133X(01)00366-9. [DOI] [PubMed] [Google Scholar]
- 198.Verkes RJ, et al. Cognitive performance and serotonergic function in users of ecstasy. Psychopharmacology. 2001;153:196–202. doi: 10.1007/s002130000563. [DOI] [PubMed] [Google Scholar]
- 199.Luciana M, Collins PF, Depue RA. Opposing roles for dopamine and serotonin in the modulation of human spatial working memory functions. Cereb. Cortex. 1998;8:218–226. doi: 10.1093/cercor/8.3.218. [DOI] [PubMed] [Google Scholar]
- 200.Roberts CA, Jones A, Montgomery C. Meta-analysis of molecular imaging of serotonin transporters in ecstasy/polydrug users. Neurosci. Biobehav. Rev. 2016;63:158–167. doi: 10.1016/j.neubiorev.2016.02.003. [DOI] [PubMed] [Google Scholar]
- 201.Cuzen NL, Koopowitz S-M, Ferrett HL, Stein DJ, Yurgelun-Todd D. Methamphetamine and cannabis abuse in adolescence: A quasi-experimental study on specific and long-term neurocognitive effects. BMJ Open. 2015;5:e005833. doi: 10.1136/bmjopen-2014-005833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Hartogsohn I. Constructing drug effects: A history of set and setting. Drug Sci. Policy Law. 2017;3:2050324516683325. doi: 10.1177/2050324516683325. [DOI] [Google Scholar]
- 203.Zohar J, Mueller EA, Insel TR, Zohar-Kadouch RC, Murphy DL. Serotonergic responsivity in obsessive-compulsive disorder: Comparison of patients and healthy controls. Arch. Gen. Psychiatry. 1987;44:946–951. doi: 10.1001/archpsyc.1987.01800230026006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and analyzed during the current study are available in the GitHub repository: https://github.com/nickhaf/MDMA_Psychedelics_meta.
The code for the analysis and the corresponding data are available in the GitHub repository: https://github.com/nickhaf/MDMA_Psychedelics_meta.