The treatment of childhood cancer has been increasingly successful over the past 30 years. Most paediatric cancers are now curable with multiagent chemotherapy in combination with surgery and radiotherapy. The overall survival five years after diagnosis is now 70% for all paediatric malignancies. The incidence is low (1200-1300 children affected each year in Britain), but with the sustained improvement in survival the number of long term survivors is increasing—about 850 additional survivors of childhood cancer each year. With this improved survival, it is important to increase our knowledge of any long term costs in the form of physical and psychosocial adverse health outcomes.
This review looks at the evidence relating to long term clinical follow up after childhood cancer and considers ways to develop such follow up for the future. An awareness of the possible long term complications is important not only for optimising health care for the current survivors but also for modifying future treatment protocols to avoid therapies that are associated with unacceptable morbidity or mortality.
We have summarised the evidence on selected long term complications; this evidence is inevitably based on retrospective studies. In the final section we discuss the development of a strategy for the clinical follow up of long term survivors.
Summary points
Long term follow up strategies are needed because of increasing numbers of survivors of childhood cancers
Models for follow up need to be developed and formally evaluated
Increasing numbers of survivors may have medical problems that will require ongoing specialist follow up
The role, training programmes, and career structure of the late effects nurse practitioner needs to be developed
The primary care physician may have an important role in long term follow up
There is a need for prospective evaluation of new treatments and randomised studies of clinical interventions to resolve substantial uncertainties for this increasing population of long term survivors
Second primary tumours
Within 25 years of diagnosis of childhood cancer in Britain about 4% of survivors develop a second primary cancer—about six times the expected number of cancers.1 This excess risk among survivors is attributable to the carcinogenic effects of treatments for the original childhood cancer as well as to genetic predisposition (figure). This can be illustrated by second primary bone cancer, which affects about 1% of survivors within 20 years of diagnosis of the original childhood cancer.2 Bone cancers, mostly osteosarcomas, are the most common solid second cancers observed after heritable retinoblastoma and after all types of childhood cancer other than retinoblastoma.2 About 7% of survivors of retinoblastoma and 0.5% of survivors of other childhood cancers are affected by bone cancer within 20 years of diagnosis of the original cancer, corresponding to about 380 times and 25 times the expected number of bone cancers in the two groups.2 The large excess after heritable retinoblastoma is attributable to the carcinogenic influence of both constitutional mutations in the RB gene and exposure of bone to radiotherapy and alkylating agents; the excess risk after all childhood cancers other than retinoblastoma is also related to the carcinogenic effects of radiotherapy and alkylating agents.2,3 There is probably some element of genetic predisposition, which would include, for example, constitutional mutations of the p53 gene.4
Second primary leukaemia is diagnosed in about 0.2% of survivors of childhood cancer in Britain within six years of diagnosis of the original cancer—about eight times the expected number of leukaemias.5 It has been established that increased cumulative exposure to alkylating agentsor epipodophyllotoxins increases the risk of subsequent leukaemia.5,6 In addition, other topoisomerase II inhibitors, including the anthracyclines, seem to be leukaemogenic.
Two important caveats limit our current understanding of the risks of second primary cancers. Firstly, there is still considerable uncertainty about the long term risks of the adult carcinomas observed most commonly in the general population, including carcinomas of the lung, large intestine, and breast. Secondly, current knowledge of the longer term risks of second cancers is based on treatments used many years ago, and there will be an inevitable delay before we can assess the longer term consequences of current treatments with confidence.
Cardiovascular disease
Cardiovascular disease can occur as a consequence of cancer treatment and contributes appreciably to the late morbidity and mortality of survivors.7 Most cardiovascular damage is the result of a direct effect by radiation and chemotherapeutic agents (particularly anthracyclines), but injury to other organs can contribute indirectly.
Chemotherapy
By far the most important cardiotoxic agents are the anthracyclines, which cause focal myocyte death with replacement fibrosis.7 The damage seems to be progressive.8 The risk factors have been well elucidated; cumulative dose is the major factor, accompanied by young age, length of follow up, and sex (female more than male).9 The incidence of death and symptomatic heart disease varies between 1% and 10%.9–11 Various studies using detailed techniques, not used in routine practice, to study subclinical cardiac dysfunction reported prevalence rates between 23% and 85%.9 The lower prevalence rate occurred at cumulative doses of 90-300 mg/m2. The full implications for future cardiac disease are unknown.
Radiation
Radiation damage to the heart and large vessels is mediated through ischaemia. Vascular damage causes atherosclerosis, as seen in early onset coronary artery disease in survivors of Hodgkin's disease.12 Patients receiving total body irradiation for bone marrow transplant conditioning must also be considered at risk. Radiation damage adds to the effect of anthracycline cardiotoxicity.
Fertility
The impact of combination cytotoxic chemotherapy on gonadal function depends on the sex and age of the child undergoing treatment and the nature and dosage of the drugs used. Drugs known to damage the gonads include procarbazine, cytosine arabinoside, and the alkylating agents, particularly cyclophosphamide, chlorambucil, mustine, melphalan, busulphan, and the nitrosoureas. Both the testes and the ovaries are vulnerable to radiation damage.13
Chemotherapy
The current management of acute lymphoblastic leukaemia in children in the United Kingdom includes cyclophosphamide. Although the long term fertility for this group of patients is not known, the available evidence suggests that the total dose of cyclophosphamide (2-3 g/m2) is unlikely to cause sterility.14,15
Treatment for Hodgkin's disease with “ChlVPP” (chlorambucil, vinblastine, procarbazine, prednisolone) is known to damage the gonads, particularly in males, and the agents implicated are chlorambucil and procarbazine. In a recent long term follow up study, 89% of the boys treated before puberty had severe damage to the germinal epithelium; recovery of spermatogenesis is unlikely. Around half of girls treated for Hodgkin's disease prepubertally with six or more courses of ChlVPP had raised plasma gonadotrophin concentrations, but longer follow up is needed to determine whether these women recover function or go on to have a premature menopause.16 “ABVD” (adriamycin, bleomycin, vinblastine, dacarbazine), which does not contain alkylating agents or procarbazine, is considerably less gonadotoxic. Current regimens with hybrid protocols are likely to preserve fertility in women and in approximately half of men.
Radiotherapy
The total dose and fractionation schedule of testicular irradiation determines the degree of damage. The risk of Leydig cell damage associated with radiation is directly related to the dose delivered and inversely related to the age at treatment. Doses above 20 Gy cause Leydig cell failure in most prepubertal boys, but doses above 30 Gy are required in adolescent boys and young adults.17 Permanent azoospermia is likely in most patients receiving more than 4 Gy.
Abdominal, pelvic, and total body irradiation can all result in ovarian damage.18 The human oocyte is sensitive to radiation, and the risk of ovarian failure increases with dose of radiotherapy.19 If there is evidence of ovarian failure then sex steroid replacement therapy is needed from puberty through to at least the fifth decade for bone mineralisation and cardiovascular protection.
Uterine radiation in childhood increases the incidence of nulliparity, fetal loss, and small for dates infants. The mechanism underlying this finding remains unclear, but both a reduction in the elasticity of the uterine musculature and uterine vascular damage have been suggested. Patients should be counselled accordingly and their obstetricians forewarned.20
Education, psychosocial, and quality of life issues
Early work detailing the psychosocial consequences of childhood cancer found that survivors experienced a range of educational, behavioural, and social problems. These findings have not always been substantiated in later work. In particular, the extent of problems experienced seems to be moderated by the disease and its treatment, as well as by demographic and family variables.21,22
Children miss substantial amounts of schooling during treatment, and this has consequences for both academic achievement and social relationships. In the days when children with acute lymphoblastic leukaemia were treated by chemotherapy and radiotherapy to the central nervous system, fairly consistent evidence showed that academic achievement and learning were compromised. Children under 5 years at diagnosis are particularly vulnerable. Although some children seem to show a general decline in intellectual function, others show deficits in specific skills, including attention, concentration, and mathematical reasoning.23 Some studies have found that it is the combination of radiotherapy and intrathecal methotrexate that limits children's intellectual functioning.24 Children with a central nervous system tumour treated with high doses of cranial irradiation are at considerable risk in terms of compromised educational outcomes, with some work indicating that approximately 50% have special educational needs.
Measurement of social function is more complex than measurement of academic function, and perhaps for this reason there have been fewer attempts to describe social functioning among survivors. Among children of school age, there is some evidence that survivors of a central nervous system tumour are less popular with other children.25,26 Given their physical limitations, many survivors are in need of appropriate and sensitive counselling to enable them to choose and succeed in appropriate employment.27
The literature points to considerable variation in quality of life among survivors. In part this reflects different methods used. Larger scale population studies have found few differences between survivors and healthy peers. Small scale, more detailed assessments often reveal important compromises in mental health.
Growth, bone mineral density, and body composition
Cranial radiotherapy as part of the treatment for childhood cancer may adversely affect growth. The mechanism by which intensive chemotherapy contributes to growth impairment is not well understood.28 29 Children who have been treated with low dose cranial radiotherapy are at risk of precocious puberty and growth hormone insufficiency. Children treated with higher doses are at risk of an evolving endocrinopathy, with growth hormone insufficiency developing early and, in some children, gonadotrophin, thyroid, or cortisol deficiency developing later.30 Total body or spinal radiation may compromise spinal growth through a direct effect on the epiphyses. Most children treated with high dose cranial radiotherapy for a brain tumour will need treatment with growth hormone; the growth response is attenuated in those children who have received additional spinal radiotherapy—the younger the child at treatment the greater the effect.31 Thyroid dysfunction after total body and cranio-spinal irradiation is well recognised. There is also an increased risk of malignancy of the thyroid when this organ has been included in the radiation field.
It is becoming clear that treatment for childhood cancer may result in reduced bone mineral density.32 The reduction in bone mineral density would be expected to predict an increased fracture rate, but this has not yet been demonstrated.
Evidence is mounting to show that children treated for lymphoblastic leukaemia become obese during treatment and remain obese at final height.28 Most studies have shown that both sexes are equally affected, whereas it is known that impaired growth and precocious puberty following childhood acute lymphoblastic leukaemia are more common in girls.28–30 There are many potential contributing factors, including growth hormone insufficiency secondary to cranial irradiation, steroids, and chemotherapy; and, most recently, reduced physical activity has been noted, the reasons for which are not clear.
Long term clinical follow up
It is clear from the preceding sections that the evidence base to guide the establishment of a structure for long term clinical follow up is incomplete. The prevailing wisdom among many paediatric oncologists and haematologists is that all survivors of childhood cancer should be followed up for life. This has important resource implications, and the evidence for such practice is not clear. There is undoubtedly a need to ascertain the risks and causes of late effects of treatment, with the hope that successive generations of patients will benefit from protocol modifications resulting from a better understanding of the clinical course of the disease and its treatment. The British Childhood Cancer Survivor Study has been developed to obtain estimates of the risks of particular adverse health outcomes occurring among survivors and their offspring and to investigate the relation of such risks to different types of treatment received for childhood cancer. Such national, population based studies will provide a basis for the further development of strategies for long term clinical follow up. In addition, clinical research will be needed to investigate outstanding questions requiring direct contact with patients.
There is a group of survivors for whom the benefit of clinical follow up is not established and for whom postal or telephone follow up may be all that is needed. For patients who have been treated with surgery alone or low risk chemotherapy, postal or telephone follow up every one or two years is likely to be sufficient (level 1 in table). Other groups—for example, patients who have received a bone marrow transplant or radiotherapy—are likely to benefit from continued clinical follow up (level 3 in table). This leaves most patients being treated according to current treatment protocols, who may benefit from clinical follow up led by a nurse or primary care doctor every one or two years once they have reached five years after treatment (level 2 in table).
We believe there is a role for a key worker, who may be a primary care or hospital doctor or a specialist nurse, to be assigned to each patient once treatment has been completed and the risk of relapse is low. Who is most appropriate will depend on local resources and the level of follow up needed. We would encourage the development of the late effects nurse practitioner, but the training programme and career structure remain to be addressed. Therapy based guidelines for follow up are available and provide a clinical framework for the development of long term follow up.33
Conclusions
An increasing literature base is becoming available to underpin decisions about the clinical follow up of long term survivors of childhood cancer; this is inevitably based on retrospective studies, and prospective evaluation of new treatments is needed. Information to guide and inform the follow up of survivors of childhood cancer will come from national population based cohort studies, large multicentre clinical studies, and randomised clinical trials designed to evaluate both survival and long term toxicities associated with different treatment strategies. As knowledge accumulates, the level of clinical surveillance should more closely match clinical need.
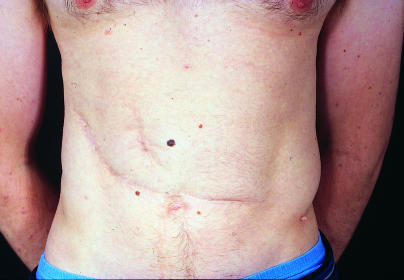
Figure.
This patient had surgery and abdominal radiotherapy for Wilms' tumour when he was 2½ years old. He developed a second primary tumour (malignant melanoma) within the radiation field in his early 20s
Table.
Possible levels of follow up more than five years from completion of treatment
| Level | Treatment | Method of follow up | Frequency | Examples of tumours |
|---|---|---|---|---|
| 1 | Surgery alone | Post or telephone | 1-2 years | Wilms' tumour stage I or II |
| Low risk chemotherapy | Langerhans cell histiocytosis (single system disease) | |||
| Germ cell tumours (surgery only) | ||||
| 2 | Chemotherapy | Led by nurse or primary care doctor | 1-2 years | Most patients (eg acute lymphoblastic leukaemia in first remission) |
| Low dose cranial irradiation (<24 Gy) | ||||
| 3 | Radiotherapy, except low dose cranial irradiation | Medically supervised late effects clinic | Annual | Brain tumours |
| After bone marrow transplant | ||||
| Megatherapy | Patients with stage IV tumours (any tumour type) |
Acknowledgments
We acknowledge valuable contributions from members of the UKCCSG Late Effects Committee. The paper has been approved by the UKCCSG Manuscripts Committee.
Footnotes
Competing interests: None declared.
References
- 1.Hawkins MM, Draper GJ, Kingston JE. Incidence of second primary tumours among childhood cancer survivors. Br J Cancer. 1987;56:339–347. doi: 10.1038/bjc.1987.200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hawkins MM, Wilson LM, Burton HS, Potok MH, Winter DL, Marsden HB, et al. Radiotherapy, alkylating agents, and the risk of bone cancer after childhood cancer. J Natl Cancer Inst. 1996;88:270–278. doi: 10.1093/jnci/88.5.270. [DOI] [PubMed] [Google Scholar]
- 3.Tucker MA, D'Angio GJ, Boice JD, Jr, Strong LC, Li FP, Stovall M, et al. Bone sarcomas linked to radiotherapy and chemotherapy in children. N Engl J Med. 1987;317:588–593. doi: 10.1056/NEJM198709033171002. [DOI] [PubMed] [Google Scholar]
- 4.Neugut AI, Meadows AT, Robinson E. Multiple primary cancers. Baltimore: Lippincott, Williams, and Wilkins; 1999. [Google Scholar]
- 5.Hawkins MM, Wilson LM, Stovall MA, Marsden HB, Potok MH, Kingston JE, et al. Epipodyophyllotoxins, alkylating agents, and radiation and risk of secondary leukaemia after childhood cancer. BMJ. 1992;304:951–958. doi: 10.1136/bmj.304.6832.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tucker MA, Meadows AT, Boice JD, Jr, Stovall M, Oberlin O, Stone BJ, et al. Leukaemia after therapy with alkylating agents for childhood cancer. J Natl Cancer Inst. 1987;78:459–464. [PubMed] [Google Scholar]
- 7.Truesdell S, Schwartz CL, Clark E, Constine LS. Cardiovascular effects of cancer. In: Schwartz CL, Hobbie WL, Constine LS, Ruccione KS, editors. Survivors of childhood cancer. St Louis: Mosby; 1994. [Google Scholar]
- 8.Goorin AM, Chauvenet AR, Perez-Atayde AR, Cruz J, McKane R, Lipshultz SE. Initial congestive heart six to ten years after doxorubicin chemotherapy for childhood cancer. J Pediatr. 1990;116:144–147. doi: 10.1016/s0022-3476(05)81668-3. [DOI] [PubMed] [Google Scholar]
- 9.Nysom K, Colan SD, Lipshultz SE. Late cardiotoxicity following anthracycline therapy for childhood cancer. Prog Pediatr Cardiol. 1998;8:121–138. [Google Scholar]
- 10.Sorenson K, Levitt G, Sebag-Montefiore D, Bull C, Sullivan I. Cardiac function in Wilms' tumour survivors. J Clin Oncol. 1995;13:1546–1556. doi: 10.1200/JCO.1995.13.7.1546. [DOI] [PubMed] [Google Scholar]
- 11.Sorensen K, Levitt G, Bull C, Chessells J, Sullivan I. Anthracycline dose in childhood acute lymphoblastic leukaemia: issues of early survival versus late cardiotoxicity. J Clin Oncol. 1997;15:61–68. doi: 10.1200/JCO.1997.15.1.61. [DOI] [PubMed] [Google Scholar]
- 12.Hancock SL, Tucker MA, Hoppe RT. Factors affecting late mortality from heart disease after treatment of Hodgkin's disease. JAMA. 1993;270:1949–1955. [PubMed] [Google Scholar]
- 13.Waring AB, Wallace WHB. Subfertility following treatment of childhood cancer. Hosp Med. 2000;61:550–557. doi: 10.12968/hosp.2000.61.8.1398. [DOI] [PubMed] [Google Scholar]
- 14.Wallace WH, Shalet SM, Tetlow LJ, Morris-Jones PH. Ovarian function following the treatment of childhood acute lymphoblastic leukaemia. Med Pediatr Oncol. 1993;21:333–339. doi: 10.1002/mpo.2950210505. [DOI] [PubMed] [Google Scholar]
- 15.Quigley C, Cowell C, Jimenez M, Burger H, Kirk J, Bergin M, et al. Normal or early development of puberty despite gonadal damage in children treated for acute lymphoblastic leukemia. N Engl J Med. 1989;321:143–151. doi: 10.1056/NEJM198907203210303. [DOI] [PubMed] [Google Scholar]
- 16.Mackie EJ, Radford M, Shalet SM. Gonadal function following chemotherapy for childhood Hodgkin's disease. Med Pediatr Oncol. 1996;27:74–78. doi: 10.1002/(SICI)1096-911X(199608)27:2<74::AID-MPO2>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 17.Shalet SM, Horner A, Ahmed SR, Morris-Jones PH. Leydig cell damage after testicular irradiation for lymphoblastic leukaemia. Med Pediatr Oncol. 1985;13:65–68. doi: 10.1002/mpo.2950130204. [DOI] [PubMed] [Google Scholar]
- 18.Sanders JE, Hawley J, Levy W, Gooley T, Buckner CD, Deeg HJ, et al. Pregnancies following high-dose cyclophosphamide with or without high-dose busulfan or total-body irradiation and bone marrow transplantation. Blood. 1996;87:3045–3052. [PubMed] [Google Scholar]
- 19.Wallace WH, Shalet SM, Hendry JH, Morris-Jones PH, Gattamaneni HR. Ovarian failure following abdominal irradiation in childhood: the radiosensitivity of the human oocyte. Br J Radiol. 1989;62:995–998. doi: 10.1259/0007-1285-62-743-995. [DOI] [PubMed] [Google Scholar]
- 20.Critchley HO, Wallace WH, Shalet SM, Mamtora H, Higginson J, Anderson DC. Abdominal irradiation in childhood; the potential for pregnancy. Br J Obstet Gynaecol. 1992;99:392–394. doi: 10.1111/j.1471-0528.1992.tb13755.x. [DOI] [PubMed] [Google Scholar]
- 21.Eiser C. Practitioner review: Long term consequences of childhood cancer. J Child Psychol Psychiatry. 1998;39:621–633. [PubMed] [Google Scholar]
- 22.Eiser C, Vance YH, Hill JJ. Examining the psychological consequences of surviving childhood cancer: the systematic review as a research method in pediatric psychology. J Pediatr Psychol. 2000;25:449–460. doi: 10.1093/jpepsy/25.6.449. [DOI] [PubMed] [Google Scholar]
- 23.Mulhern RK. Neuropsychological late effects. In: Bearison A, Mulhern R, editors. Pediatric psychooncology. New York: Oxford University Press; 1994. [Google Scholar]
- 24.Hill JM, Kornblith AB, Jones D, Freeman A, Holland JF, Glicksman AS, et al. A comparative study of the long term psychosocial functioning of childhood acute lymphoblastic leukemia survivors treated by intrathecal methotrexate with or without cranial irradiation. Cancer. 1998;82:208–218. [PubMed] [Google Scholar]
- 25.Noll RB, Bulowski WM, Davies WH, Koontz K, Kulkarni R. Adjustment in the peer system of adolescents with cancer: a two-year study. J Pediatr Psychol. 1993;18:351–364. doi: 10.1093/jpepsy/18.3.351. [DOI] [PubMed] [Google Scholar]
- 26.Noll RB, MacLean WE, Whitt JK, Kaleita TA, Stehbens JA, Waskerwitz MJ, et al. Behavioral adjustment and social functioning of long-term survivors of childhood leukemia: parent and teacher reports. J Pediatr Psychol. 1997;22:827–842. doi: 10.1093/jpepsy/22.6.827. [DOI] [PubMed] [Google Scholar]
- 27.Eiser C, Cool P, Grimer RJ, Carter SR, Cotter IM, Ellis AJ, et al. Quality of life following treatment for a malignant primary bone tumour around the knee. Sarcoma. 1997;1:39–45. doi: 10.1080/13577149778461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Davies HA, Didcock E, Didi M, Ogilvy-Stuart A, Wales JK, Shalet SM. Growth, puberty and obesity after treatment for leukaemia. Acta Paediatr Suppl. 1995;411:45–50. doi: 10.1111/j.1651-2227.1995.tb13862.x. [DOI] [PubMed] [Google Scholar]
- 29.Sklar C, Mertens A, Walter A, Mitchell D, O'Leary M, Hutchinson R, et al. Final height after treatment for acute lymphoblastic leukaemia: comparison of no cranial irradiation, 1800 cGy and 2400 cGy cranial irradiation. J Pediatr. 1993;123:59–64. doi: 10.1016/s0022-3476(05)81537-9. [DOI] [PubMed] [Google Scholar]
- 30.Shalet SM, Clayton PE, Price DA. Growth and pituitary function in children treated for brain tumours or acute lymphoblastic leukaemia. Horm Res. 1988;50:53–61. doi: 10.1159/000181029. [DOI] [PubMed] [Google Scholar]
- 31.Sulmont V, Brauner R, Fontura M, Rappaport R. Response to growth hormone treatment and final height after cranial or craniospinal irradiation. Acta Paediatr Scand. 1990;79:542–549. doi: 10.1111/j.1651-2227.1990.tb11509.x. [DOI] [PubMed] [Google Scholar]
- 32.Nysom K, Holm K, Michaelsen KF, Hertz H, Muller J, Molgaard C. Bone mass after treatment for acute lymphoblastic leukemia in childhood. J Clin Oncol. 1998;16:3752–3760. doi: 10.1200/JCO.1998.16.12.3752. [DOI] [PubMed] [Google Scholar]
- 33.Kissen GKN, Wallace WHB. Therapy based guidelines for long-term follow up of children treated for cancer. Published by Pharmacia on behalf of the Late Effects Group of the UKCCSG, 1995.