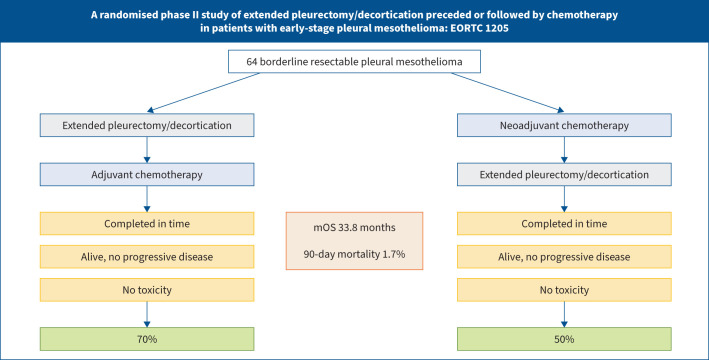
Graphical abstract
Overview of the study findings. mOS: median overall survival.
Abstract
Background
The role of surgery in pleural mesothelioma remains controversial. It may be appropriate in highly selected patients as part of a multimodality treatment including chemotherapy. Recent years have seen a shift from extrapleural pleuropneumonectomy toward extended pleurectomy/decortication. The most optimal sequence of surgery and chemotherapy remains unknown.
Methods
EORTC-1205-LCG was a multicentric, noncomparative phase 2 trial, 1:1 randomising between immediate (arm A) and deferred surgery (arm B), followed or preceded by chemotherapy. Eligible patients (Eastern Cooperative Oncology Group 0–1) had treatment-naïve, borderline resectable T1–3 N0–1 M0 mesothelioma of any histology. Primary outcome was rate of success at 20 weeks, a composite end-point including 1) successfully completing both treatments within 20 weeks; 2) being alive with no signs of progressive disease; and 3) no residual grade 3–4 toxicity. Secondary end-points were toxicity, overall survival, progression-free survival and process indicators of surgical quality.
Findings
69 patients were included in this trial. 56 (81%) patients completed three cycles of chemotherapy and 58 (84%) patients underwent surgery. Of the 64 patients in the primary analysis, 21 out of 30 patients in arm A (70.0%; 80% CI 56.8–81.0%) and 17 out of 34 patients (50.0%; 80% CI 37.8–62.2%) in arm B reached the statistical end-point for rate of success. Median progression-free survival and overall survival were 10.8 (95% CI 8.5–17.2) months and 27.1 (95% CI 22.6–64.3) months in arm A, and 8.0 (95% CI 7.2–21.9) months and 33.8 (95% CI 23.8–44.6) months in arm B. Macroscopic complete resection was obtained in 82.8% of patients. 30- and 90-day mortality were both 1.7%. No new safety signals were found, but treatment-related morbidity was high.
Interpretation
EORTC 1205 did not succeed in selecting a preferred sequence of pre- or post-operative chemotherapy. Either procedure is feasible with a low mortality, albeit consistent morbidity. A shared informed decision between surgeon and patient remains essential.
Shareable abstract
EORTC 1205 did not succeed in selecting a preferred sequence of pre- or post-operative chemotherapy. Both approaches are feasible in multimodality treatment of pleural mesothelioma. https://bit.ly/3U6jyVM
Introduction
Pleural mesothelioma is a rare tumour of the pleural cavity, in ∼80% of cases associated with prior asbestos exposure. Prognosis is poor, due to the disease being usually diagnosed in an advanced stage, with <10% of patients being alive after 5 years [1]. In fit patients with early-stage disease, surgical series and registry studies consistently report better outcomes in association with bimodality or trimodality treatments, but selection and recall bias are not unlikely [2–4].
The traditional surgical approach was extrapleural pleuropneumonectomy (EPP), in which complete macroscopic resection is attempted by removing the parietal pleura with the ipsilateral lung. The Mesothelioma and Radical Surgery (MARS) trial compared chemotherapy with or without EPP and reported a longer median survival and less serious adverse events in the no EPP cohort, but was underpowered to demonstrate a significant difference [5]. Nevertheless, most centres have replaced EPP by the lung-sparing extended (if removal of pericardium and/or diaphragm) pleurectomy/decortication (e-P/D), with similar results [6]. A detailed overview of both techniques can be found elsewhere [7]. The definition of e-P/D was based on the international consensus report on surgical techniques for pleural mesothlioma [8].
Nevertheless, the role of surgery in pleural mesothelioma remains controversial. The 2020 joint European Respiratory Society/European Society of Thoracic Surgeons/European Association for Cardio-Thoracic Surgery/European Society for Radiotherapy and Oncology guideline states that no clear recommendation can be given, but surgery may be appropriate in highly selected patients as part of a multimodality treatment including chemotherapy [9]. The exact sequence of chemotherapy and surgery is unknown, as both approaches have been used in clinical trials.
In order to further explore e-P/D as part of multimodality treatment, this technique has to be standardised across expert centres and the best sequence of surgery and chemotherapy has to be determined. These were the objectives of this phase 2 randomised trial (registered at clinicaltrials.gov, identifier number NCT02436733).
Materials and methods
Study design
The complete study protocol has been published elsewhere [10]. In short, eligible patients with pathologically proven pleural mesothelioma (irrespective of histology), cT1–3 N0–2 M0 (stage I, II and III according to Union for International Cancer Control TNM7), who were treatment naïve (except for diagnostic thoracoscopy with recommended talc pleurodesis), had Eastern Cooperative Oncology Group performance score 0–1 and were fit for chemotherapy and surgery were enrolled in this study. Focal chest wall involvement was allowed, but widespread chest wall or mediastinal involvement (T4) was excluded, as were contralateral, supraclavicular or coeliac lymph nodes (N3). A positron emission tomography (PET) scan was mandatory to exclude extrathoracic disease. Tumour evaluation with chest/upper abdomen computed tomography (CT) was performed at baseline, between both treatment modalities, at the end of treatment and continued every 3 months to assess the secondary end-points of progression-free survival (PFS) and overall survival (OS).
Patients were randomised 1:1 between immediate surgery (arm A), followed by adjuvant chemotherapy (three cycles of cisplatin 75 mg·m−2 plus pemetrexed 500 mg·m−2 every 3 weeks) or deferred surgery (arm B), preceded by neoadjuvant chemotherapy (three cycles of the same regimen). A later amendment also allowed substituting cisplatin with carboplatin (area under the curve 5 mg·mL−1·min−1 every 3 weeks) given the growing evidence for the equivalence of carboplatin and the more convenient administration during the coronavirus disease 2019 (COVID-19) pandemic. Neoadjuvant chemotherapy had to start between 2 and 6 weeks after tumour assessment, with surgery following between 3 and 6 weeks after day 1 of the last chemotherapy cycle. Upfront surgery had to take place between 2 and 6 weeks after tumour assessment, with adjuvant chemotherapy starting between 3 and 6 weeks after resection. An overview of the patient flow and study timelines is shown in supplementary figure S1. Randomisation was performed centrally, using the European Organisation for Research and Treatment of Cancer (EORTC) randomisation system; patients were stratified according to tumour stage and institution.
The minimal surgical procedure to be performed was a parietal and visceral pleurectomy with resection of all visible tumour (P/D). If indicated, removal of pericardium, hemidiaphragm, lung parenchyma and/or chest wall was also performed (e-P/D). More details can be found in the study protocol in the supplementary material. Intraoperative conversion to pneumonectomy was allowed at surgeon's discretion. The specific technique of extended pleurectomy/decortication was described in detail in the EORTC 1205 Quality Assurance for Surgery Guidelines (supplementary material). A specific training by a senior (cardio)thoracic surgeon was required to be allowed to participate as surgical centre.
Outcome
The primary end-point was the composite end-point “rate of success at 20 weeks after treatment start in the per-protocol population”, developed by EORTC statisticians, encompassing 1) having successfully completed both treatment modalities per protocol within 20 weeks (with a margin of 2 weeks); and 2) being alive, without signs of progressive disease at week 20 (with a margin of 2 weeks); and 3) having no residual grade 3 or 4 treatment-related adverse events at week 20 (with a margin of 2 weeks).
Secondary end-points were PFS and OS from date of randomisation, all in the per-protocol population; toxicity as assessed by the Common Terminology Criteria for Adverse Events version 4.0 (analysis for toxicity was based on the safety population), post-operative mortality at 30 and 90 days, and other process indicators of surgical quality and uniformity (surgical data are descriptive with appropriate confidence intervals), all in the per-protocol population. No quality-of-life data were collected.
Statistical analysis
The trial was developed to determine if in the e-P/D setting either approach, neoadjuvant or adjuvant, is feasible to justify further investigation. Each arm has been tested individually using a single proportion test designed to reject a rate of 65% (H0) at the 10% significance level. With a statistical power of 90% to detect a true rate of 85% (H1), 32 patients had to be enrolled in each arm (64 in total), of whom ≥25 had to be successful, to declare an arm feasible. Due to an imbalance between the two arms (four patients did not start treatment in arm A), the required number of eligible patients was not reached. The decision rule was therefore replaced by an analysis based on confidence intervals: an arm will be declared feasible if the lower bound of the one-sided 90% confidence interval is greater than H0=65%. The lower 90% one-sided confidence intervals and the 80% two-sided confidence intervals have been estimated using the exact Clopper–Pearson method. In addition, the p-value of the single proportion test are provided for reference.
The analyses of the primary and secondary end-points (PFS and OS) were performed on all randomised patients who are eligible and started treatment (per-protocol population). Estimates of the median PFS and OS were obtained using the Kaplan–Meier technique. The 95% two-sided confidence interval for the median was calculated using the reflected confidence interval method. Estimates of the event-free rate at a fixed time point were obtained using the Kaplan–Meier technique and 95% two-sided confidence intervals were calculated using Greenwood's estimation of the standard deviation. Estimates of hazard ratios and their 95% two-sided confidence intervals were obtained by Cox regression using arm A as reference. Kaplan–Meier curves were drawn for both the experimental and control arms on the same plot.
A measure of association between baseline characteristics and treatment arms (table 1) has been provided through a Chi-squared test (categorical variables) and a two-sample t-test (continuous variables).
TABLE 1.
Patient and tumour characteristics in the per-protocol population
| Arm A immediate surgery | Arm B deferred surgery | p-value | Arm A+arm B overall population | |
|---|---|---|---|---|
| Patients | 30 | 34 | 64 | |
| Sex | 0.567 | |||
| Male | 22 (73.3) | 27 (79.4) | 49 (76.6) | |
| Female | 8 (26.7) | 7 (20.6) | 15 (23.4) | |
| Age years | 66.0 (49.0–76.0) | 67.0 (41.0–79.0) | 0.514 | 66.0 (41.0–79.0) |
| ECOG-PS | 0.975 | |||
| 0 | 14 (46.7) | 16 (47.1) | 30 (46.9) | |
| 1 | 16 (53.3) | 18 (52.9) | 34 (53.1) | |
| Histological subtype | 0.558 | |||
| Epithelioid | 26 (86.7) | 29 (85.3) | 55 (85.9) | |
| Biphasic | 3 (10.0) | 4 (11.8) | 7 (10.9) | |
| Sarcomatoid | 1 (3.3) | 0 (0.0) | 1 (1.6) | |
| Missing | 0 (0.0) | 1 (2.9) | 1 (1.6) | |
| Clinical tumour stage (TNM7) | 0.336 | |||
| Stage I | 13 (43.3) | 18 (52.9) | 31 (48.4) | |
| Stage II | 9 (30.0) | 5 (14.7) | 14 (21.9) | |
| Stage III | 8 (26.7) | 11 (32.4) | 19 (29.7) |
Data are presented as n, n (%) or median (range), unless otherwise stated. ECOG-PS: Eastern Cooperative Oncology Group performance score; TNM: tumour node metastasis.
Results
Between October 2016 and November 2021, 69 patients were enrolled in five participating tertiary-care hospitals from three countries (Belgium, the Netherlands, Egypt). Due to the COVID-19 pandemic, accrual was temporarily halted. 34 patients were randomised to arm A and 35 to arm B. Five patients were excluded from the per-protocol analysis, thus leaving 64 patients in the final analysis, of whom 30 were in arm A and 34 in arm B (figure 1). Reasons for exclusion were progressive disease before start of study intervention (n=1), wrong diagnosis (n=1), withdrawal of consent (n=1), no surgery performed due to COVID-19 restrictions (n=1) in arm A and incorrect staging (n=1) in arm B. Patient and tumour characteristics are reported in table 1 (per-protocol population) and supplementary table S1 (intention-to-treat population). Both treatment arms were well-balanced.
FIGURE 1.
Overview of the consort flow chart. COVID-19: coronavirus disease 2019; PET: positron emission tomography; CT: computed tomography.
Seven (10%) patients in arm A received no adjuvant chemotherapy. In arm B, all patients received at least two cycles of chemotherapy. Of the 62 patients who underwent chemotherapy, 56 (81%) completed the full three cycles. Details are reported in table 2. Cisplatin was more often used in the neoadjuvant setting (74% versus 44% of patients), whereas carboplatin was more often used in adjuvant setting (27% versus 14% of patients). Seven patients switched from cisplatin to carboplatin during treatment. Neoadjuvant chemotherapy was more often successfully completed (91% received the full three cycles) compared to adjuvant chemotherapy (71% received the full three cycles).
TABLE 2.
Overview of systemic treatments
| Arm A immediate surgery | Arm B deferred surgery | Arm A+arm B overall population | |
|---|---|---|---|
| Patients | 34 | 35 | 69 |
| Did not receive chemotherapy | 7 (20.6) | 0 (0.0) | 7 (10.1) |
| Completed at least one cycle of chemotherapy | 27 (79.4) | 35 (100.0) | 62 (89.9) |
| Completed at least two cycles of chemotherapy | 25 (73.5) | 35 (100.0) | 60 (86.9) |
| Completed all three cycles of chemotherapy | 24 (70.6) | 32 (91.4) | 56 (81.2) |
| Type of platinum | |||
| Cisplatin | 15 (44.1) | 26 (74.3) | 41 (59.4) |
| Carboplatin | 9 (26.5) | 5 (14.3) | 14 (20.3) |
| Both | 3 (8.8) | 4 (11.4) | 7 (10.1) |
| Pemetrexed median relative dose-intensity % | 95.8 | 97.9 | 97.9 |
| Cisplatin median relative dose-intensity % | 95.6 | 97.5 | 96.7 |
Data are presented as n or n (%), unless otherwise stated. Dose-intensity for carboplatin was not collected, as it was only allowed after an amendment to the protocol.
Detailed results for the primary end-point are reported in table 3. 21 out of 30 patients in arm A and 17 out of 34 patients in arm B fulfilled all three required criteria in order to be successful, which gives a success rate of 70.0% in arm A (lower 90% CI 56.8–100.0, p=0.358), and 50.0% in arm B (lower 90% CI 37.8–100.0, p=0.976). Therefore, neither of the arms could be considered successful. The difference between arm A and B is mainly a consequence of the higher number of patients with progressive disease at week 18–22 in arm B (41% compared to 17%). As this difference could be inferred to post-operative sequelae in resorption, we checked their evolution 3 months later. All patients with progressive disease at week 18–22 were further progressing either locally or distantly 3 months later. Results for criteria 1 and 3 were similar in both arms.
TABLE 3.
Overview of results of the rate of success (primary end-point), overall survival and progression-free survival analysis (secondary end-points)
| Arm A immediate surgery | Arm B deferred surgery | Arm A+arm B overall population | |
|---|---|---|---|
| Patients | 30 | 34 | 64 |
| Successfully completed both treatment modalities per protocol within 20 weeks # | 24 (80) | 27 (79) | 51 (80) |
| Alive, without signs of progressive disease at week 20 # | 25 (83) | 20 (59) | 45 (70) |
| No residual grade 3 or 4 treatment-related adverse events at week 20 # | 29 (97) | 34 (100) | 63 (98) |
| Patients fulfilling all the above criteria (i.e. treatment success) | 21 | 17 | 38 |
| Rate of success (two-sided 80% CI) % | 70 (56.8–81.0) | 50 (37.8–62.2) | |
| One-sided p-value | 0.358 | 0.976 | |
| Progression-free survival months | 10.8 (8.5–17.2) | 8.0 (7.2–21.9) | 10.3 (8.0–17.0) |
| Progression-free survival % | |||
| At 1 year | 44.9 (26.6–61.6) | 42.5 (25.6–58.4) | 43.6 (31.1–55.4) |
| At 2 years | 22.8 (9.5–39.5) | 29.8 (15.3–45.8) | 26.7 (16.3–38.2) |
| Hazard ratio (95% CI) | Reference | 1.08 (0.61–1.92) | |
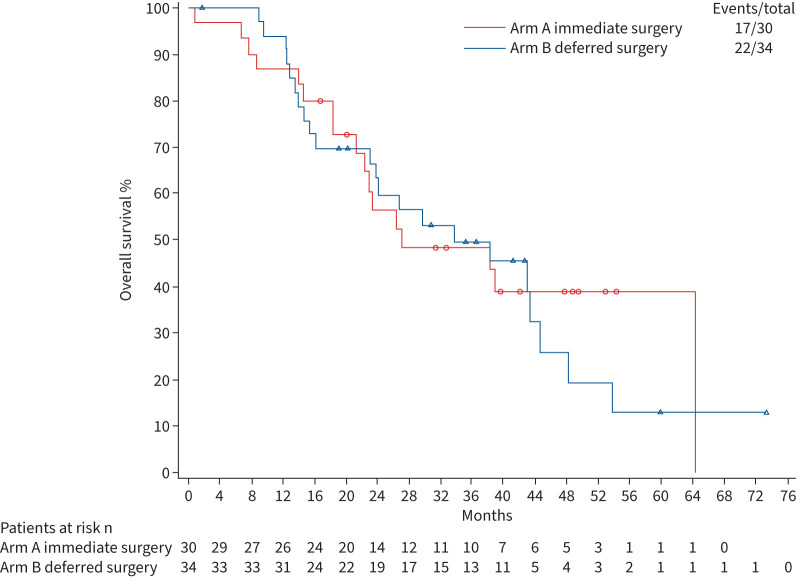
| Overall survival months | 27.1 (22.6–64.3) | 33.8 (23.8–44.6) | 33.8 (23.8–44.6) |
| Overall survival % | |||
| At 1 year | 86.7 (68.3–94.8) | 93.9 (77.9–98.4) | 90.5 (80.1–95.6) |
| At 2 years | 56.6 (36.0–72.8) | 63.1 (44.1–77.1) | 60.3 (46.8–71.5) |
| Hazard ratio (95% CI) | Reference | 1.22 (0.64–2.34) |
Data are presented as n, n (%) or median (95% CI), unless otherwise stated. #: 20 weeks with a margin of 2 weeks allowed (18–22 weeks).
There was no statistically significant difference in OS (hazard ratio (HR) 1.22, 95% CI 0.64–2.34) (table 3), and the median was 27.1 (95% CI 22.6–64.3) months for arm A and 33.8 (95% CI 23.8–44.6) months for arm B. 2-year OS rates was 56.6% and 63.1%, respectively. OS curves for both treatment arms are depicted in figure 2. OS curves per histology can be found in figure 3. Median PFS was 10.8 (95% CI 8.5–17.2) months in arm A and 8.0 (95% CI 7.2–21.9) months in arm B; again, the difference was not statistically significant (HR 1.08, 95% CI 0.61–1.92). 2-year PFS was 22.8% for arm A, and 29.8% for arm B (supplementary figure S2).
FIGURE 2.
Kaplan–Meier analysis of overall survival in both treatment arms.
FIGURE 3.
Kaplan–Meier analysis of overall survival in both treatment arms per histology. NE: not evaluable.
An overview of surgical quality indicators is given in table 4. 58 patients underwent surgery, with a median duration of the procedure of almost 6 h. Pleurectomy and decortication was performed in all patients; the majority also required additional resections of the hemidiaphragm (81.0%), pericardium (55.2%), lung parenchyma (44.8%), chest wall (12.1%) and other structures (15.5%). In one patient (arm B), conversion to extrapleural pleuropneumonectomy was required. Macroscopic complete resection (R1) was reached in 82.8% of patients. Neoadjuvant chemotherapy did not influence the number of complete resections. More technical difficulties were reported in arm B (deferred surgery) compared to arm A (immediate surgery): 28.6% versus 13.3%. There was no difference in the median duration of stay (p=0.38).
TABLE 4.
Overview of results of the surgical quality analysis
| Arm A immediate surgery | Arm B deferred surgery | Arm A+arm B overall population | |
|---|---|---|---|
| Operated patients | 30 | 28 | 58 |
| Duration of surgery min | 372.0 (210.0–586.0) | 339.5 (250.0–457.0) | 353.0 (210.0–586.0) |
| Duration of stay days | 14.0 (6.0–37.0) | 13.0 (7.0–30.0) | 13.5 (6.0–37.0) |
| Intraoperative blood loss mL | 1700.0 (0.0–5275.0) | 1475.0 (0.0–4720.0) | 1500.0 (0.0–5275.0) |
| Macroscopic resection | |||
| Complete | 26 (86.7) | 22 (78.6) | 48 (82.8) |
| Incomplete (R2) | 4 (13.3) | 6 (21.4) | 10 (17.2) |
| Patients with complications related to surgery | 23 (76.7) | 26 (92.9) | 49 (84.5) |
| Resection of hemidiaphragm | |||
| No | 4 (13.3) | 7 (25.0) | 11 (19.0) |
| Yes | 26 (86.7) | 21 (75.0) | 47 (81.0) |
| Resection of pericardium | |||
| No | 14 (46.7) | 12 (42.9) | 26 (44.8) |
| Yes | 16 (53.3) | 16 (57.1) | 32 (55.2) |
| Resection extending to ribs/thoracic wall | |||
| No | 26 (86.7) | 25 (89.3) | 51 (87.9) |
| Yes | 4 (18.3) | 3 (10.7) | 7 (12.1) |
| Resection of any lung parenchyma | |||
| No | 17 (56.7) | 15 (53.6) | 32 (55.2) |
| Yes | 13 (43.3) | 13 (46.6) | 26 (44.8) |
| Resection extending to other structures | |||
| No | 25 (83.3) | 24 (85.7) | 49 (84.5) |
| Yes | 5 (16.7) | 4 (14.3) | 9 (15.5) |
| Mortality at 30 days | 1 (3.3) | 0 (0) | 1 (1.7) |
| Mortality at 90 days | 1 (3.3) | 0 (0) | 1 (1.7) |
Data are presented as n, median (range) or n (%).
Complication rate was high: 84.5% of patients suffered at least one complication from surgery. Nevertheless, 30- and 90-day mortality was low: 1.7% (one patient died from myocardial infarction 2 days after surgery in arm A). An overview of post-operative serious adverse events can be found as supplementary table S3. Median duration of hospital stay was 14 days, similar in both arms. Serious adverse events were present in 13 patients in arm A, and eight in arm B. Persistent adverse events of grade ≥3 at week 18–22 were found in three patients, all in arm A (pain, worsening dyspnoea, pleural haemorrhage), all of grade 3.
The treatment was well tolerated, with 80% completing both treatment modalities in time and 98% with no persistent toxicity of grade 3–4. Reasons for discontinuation of the trial were progressive disease (n=2), complications of chemotherapy (n=2) or surgery (n=2), patient (3) or investigator (n=6) decision. An overview of toxicity is given in table 5. A more detailed overview can be found in supplementary table S2. Most grade 3–4 toxicity was the expected haematological toxicity of chemotherapy. No other safety signals were found.
TABLE 5.
Overview of adverse events reported in the safety population
| Arm A immediate surgery | Arm B deferred surgery | |||
|---|---|---|---|---|
| Overall (≥1) | Grade 3–4 | Overall (≥1) | Grade 3–4 | |
| Patients | 30 | 35 | ||
| Haematological toxicity | ||||
| Anaemia | 25 (83.3) | 7 (23.3) | 27 (77.1) | 10 (28.6) |
| Leukopenia | 11 (36.7) | 2 (6.7) | 15 (42.8) | 1 (2.8) |
| Neutropenia | 7 (23.3) | 3 (10.0) | 16 (45.7) | 2 (5.7) |
| Lymphopenia | 16 (53.3) | 5 (16.6) | 11 (31.4) | 3 (8.6) |
| Thrombocytopenia | 5 (16.7) | 0 (0) | 8 (22.8) | 0 (0) |
| Gastrointestinal and hepatobiliary disorders | 20 (66.7) | 2 (6.7) | 29 (82.9) | 2 (5.7) |
| Infections | 6 (20.0) | 4 (13.3) | 6 (17.1) | 2 (5.7) |
| Cardiac disorders | 4 (13.3) | 1 (3.3)# | 3 (8.6) | 1 (2.9) |
| Respiratory and thoracic disorders | 19 (63.3) | 2 (6.7) | 20 (57.1) | 2 (5.7) |
| General disorders and administration site | 19 (63.3) | 1 (3.3) | 23 (65.7) | 0 (0) |
| Eye and ear disorders | 8 (26.6) | 0 (0) | 6 (17.1) | 0 (0) |
| Intraoperative haemorrhage | 0 (0) | 0 (0) | 1 (2.8) | 1 (2.8) |
| Metabolism and electrolyte disorders | 4 (13.3) | 1 (3.3) | 11 (31.4) | 1 (2.8) |
| Musculoskeletal and connective tissue disorders | 10 (33.3) | 0 (0) | 6 (17.1) | 0 (0) |
| Skin and subcutaneous tissue disorders | 4 (13.3) | 0 (0) | 9 (25.7) | 0 (0) |
| Vascular disorders | 9 (30.0) | 3 (10.0) | 11 (31.4) | 1 (2.9) |
| Nervous system disorders | 28 (26.7) | 0 (0) | 15 (42.9) | 2 (5.7) |
| Renal and urinary disorders | 1 (3.3) | 0 (0) | 6 (17.1) | 0 (0) |
Data are presented as n or n (%). Patients may have multiple toxicities. #: one case of grade 5 toxicity (acute myocardial infarction).
Discussion
The EORTC 1205 trial primarily addressed the optimal sequence of any perioperative chemotherapy in patients with early-stage pleural mesothelioma, considered borderline resectable by a panel of multidisciplinary experts, given the fact that no internationally accepted guidelines exist to determine resectability in pleural mesothelioma. The respective multidisciplinary tumour boards carefully assessed resectability based on the inclusion criteria and PET and CT imaging. Tumours were considered borderline resectable if R1 resection appeared feasible. Nevertheless, in our experience most operated cases turn out to be pT3 tumours due to invasion of mediastinal or pericardial fatty tissue.
Scientific arguments for either a neoadjuvant or an adjuvant approach are both sound and stem from more than a decade ago, when e-P/D overtook EPP as the preferred surgical modality.
The trial did not succeed in its primary end-point, as neither approach had a sufficiently high success rate, which was considered a clinically relevant and validated composite end-point for treatment adherence, efficacy and late toxicity. Neither did any approach differ in the time-to-event end-points of progression-free and overall survival. This finding should not be a surprise, as in early-stage nonsmall cell lung cancer (NSCLC), no significant difference in outcome was observed between both approaches in pooled data from a large-scale systematic review [11].
Our second goal addresses the quality and outcome of the surgical procedure across different institutes. Although e-P/D was associated with a high incidence of overall complications (>75%), and serious adverse events occurred in 30.4% of the patients, 90-day mortality was low (1.7%) and from the surgical point of view, a neoadjuvant chemotherapy approach showed no major differences compared to an adjuvant one. A difference in duration of hospital stay (which was reported in a systematic review [12]) was not present in this study.
Hence, physicians involved in the expert care of patients with pleural mesothelioma can still proceed either way, as the neoadjuvant and adjuvant chemotherapy approaches appear to be equivalent. Patient preference, operational and logistical considerations such as waiting lists and availability of operating room time, are to be included in this decision. Furthermore, survival outcome data look favourable, compared to recently reported single-centre series [13].
The strength of this trial lies in its strict use of the pragmatic clinical end-point of success rate, which was developed in an earlier trial. Nevertheless, a composite end-point introduces challenges in interpreting the overall treatment success. The exclusion of four patients from arm A for not starting treatment caused an imbalance between both arms and led to the study becoming underpowered. This may have biased results, but evidently this reflects the challenge of conducting a clinical trial with multiple treatment modalities.
In the EORTC 08031 trial evaluating trimodal therapy including EPP, a success rate of 42% was observed [14], comparable to the 50% success rate of the neoadjuvant approach in EORTC 1205. When diving deeper into the data, one wonders whether the observed difference in progression rate at 20 weeks in favour of the patients in the adjuvant chemotherapy arm are due to ongoing post-operative sequelae in the patients treated with neoadjuvant chemotherapy incorrectly qualified as progression. We have reanalysed the imaging data of these “pseudoprogressing” patients at their next follow-up CT scan (3 months later); unfortunately, all had unmistakably progressive disease, either locally or locoregionally at that time point.
Another strength of the trial lies in its staging and decision-making: PET-CT scan and multidisciplinary team decision were mandatory for eligibility. Surgery was performed in centres of excellence and all involved surgeons were trained in the e-P/D procedure. Key moments of the operation were recorded and centrally reviewed together with surgical technical details.
This brings us to the issue whether our patients have anyhow benefited from a resection, as recent and older randomised evidence show the contrary. Whereas MARS1 reported a possible detrimental effect of EPP compared to chemotherapy [5], the recently released MARS2 data show no difference in survival between patients treated with chemotherapy with or without e-P/D (HR 0.48, 95% CI 0.18–1.29; p=0.15) [15]. These results cast a doubt on the role of any surgery in pleural mesothelioma. Although intertrial comparisons are likely biased and the issue of resection was not the goal of the EORTC 1205 trial, we notice that median overall survival in MARS2 for resected patients was <24 months, comparing to >33 months in the neoadjuvant chemotherapy arm of in our trial. Similarly, operative mortality at 90 days in MARS2 was 8.9%, compared to 1.7% in our trial. These results suggest a difference in patient selection, casting doubt on the generalisability of the MARS2 results to a more conservatively selected patient population. Further evidence in favour of surgery is given by a 2008 meta-analysis by Flores et al. [16], reporting an operative mortality of 4.7% in 278 e-P/Ds across three American hospitals.
We vouch for the role of e-P/D in selected patients with borderline resectable pleural mesothelioma, provided it is carried out by an expert multidisciplinary team in a high-volume centre of mesothelioma care. The outcome for nonepithelioid histology appeared to be worse in the eight patients included in this study. Whether nonepithelioid patients may benefit less from surgery is unclear; this difference may be due to the poorer response to chemotherapy and could possibly be mended by the addition of immunotherapy.
Lastly, the issue of the optimal sequence of chemotherapy and surgery may be shifting towards the neoadjuvant approach. In NSCLC, perioperative checkpoint inhibitors plus chemotherapy have made their entrance in the clinic [17] and are even becoming standard of care in stage II and III NSCLC [18]. In unresectable pleural mesothelioma, dual immunotherapy has become the standard of care in first-line systemic treatment [19]. Phase 3 trials combining immunotherapy and chemotherapy in the unresectable setting are ongoing [20–22]. A combination of chemotherapy and immune checkpoint inhibition sounds attractive for the future multimodality management of borderline resectable mesothelioma.
Another approach to improve local control to be further investigated is adding intensity-modulated radiotherapy to the pleura, which has been shown to be feasible in two phase 2 trials [23, 24] and is currently being investigated in a phase 3 trial [25]. During surgery, applying hyperthermic intrathoracic chemotherapy may also improve local control, but this modality has not yet been evaluated in a randomised trial, and its early results should be confirmed in controlled studies [26, 27].
Conclusion
EORTC 1205 did not succeed in selecting a preferred sequence of pre- or post-operative chemotherapy in borderline resectable patients with pleural mesothelioma. Either procedure is feasible with a low mortality, albeit consistent morbidity. A shared informed decision between surgeon and patient remains essential. Given the complexity of surgery, systemic treatment and radiotherapy, borderline resectable mesothelioma should be treated by a multidisciplinary team in an expert centre.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material ERJ-02114-2023.Supplement (340.4KB, pdf)
Surgical guidelines ERJ-02114-2023.Guidelines (2.3MB, pdf)
Shareable PDF
Acknowledgements
This work was supported by the Lung Cancer Group of the European Organisation for Research and Treatment of Cancer (EORTC), a grant from Kom op tegen Kanker (Stand up to Cancer), and the Flemish Cancer Society from Belgium. The investigators wish to thank Eli Lilly and Company Limited for providing pemetrexed (Alimta) to all Egyptian patients, as well as Loic Lang-Lazdunski (CHU Lausanne, Switzerland) for expert advice, all participating patients and the following investigators: Annelies Janssens and Reinier Wener (University Hospital Antwerp), Karim Vermaelen (University Hospital Ghent) and Rabab Gaafar† (National Cancer Institute Cairo).
Footnotes
This clinical trial was prospectively registered at clinicaltrials.gov with identifier number NCT02436733.
This article has an editorial commentary: https://doi.org/10.1183/13993003.00896-2024
Ethics approval: The trial was approved by the ethics committees of all participating centres.
Conflict of interest: The authors have no potential conflicts of interest to disclose.
Support statement: This work was funded by a grant from Stand up against Cancer (Belgium). Eli Lilly and Company Limited provided pemetrexed for the Egyptian patients but was not involved in the trial. Funding information for this article has been deposited with the Crossref Funder Registry.
Data availability
Individual participant data (after de-identification) collected during the trial, study protocol, statistical analysis plan and informed consent form will be made available for other researchers after a motivated request to and after approval from the EORTC.
References
- 1.Milano MT, Zhang H. Malignant pleural mesothelioma: a population-based study of survival. J Thorac Oncol 2010; 5: 1841–1848. doi: 10.1097/JTO.0b013e3181f1cf2b [DOI] [PubMed] [Google Scholar]
- 2.Raskin J, Van Schil PE, Meerbeeck JPV. Surgical series in mesothelioma: navigating between biases. Transl Lung Cancer Res 2023; 12: 184–186. doi: 10.21037/tlcr-23-34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Saddoughi SA, Abdelsattar ZM, Blackmon SH. National trends in the epidemiology of malignant pleural mesothelioma: a National Cancer Data Base study. Ann Thorac Surg 2018; 105: 432–437. doi: 10.1016/j.athoracsur.2017.09.036 [DOI] [PubMed] [Google Scholar]
- 4.Nelson DB, Rice DC, Niu J, et al. Predictors of trimodality therapy and trends in therapy for malignant pleural mesothelioma. Eur J Cardiothorac Surg 2018; 53: 960–966. doi: 10.1093/ejcts/ezx427 [DOI] [PubMed] [Google Scholar]
- 5.Treasure T, Lang-Lazdunski L, Waller D, et al. Extra-pleural pneumonectomy versus no extra-pleural pneumonectomy for patients with malignant pleural mesothelioma: clinical outcomes of the Mesothelioma and Radical Surgery (MARS) randomised feasibility study. Lancet Oncol 2011; 12: 763–772. doi: 10.1016/S1470-2045(11)70149-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cao C, Tian D, Park J, et al. A systematic review and meta-analysis of surgical treatments for malignant pleural mesothelioma. Lung Cancer 2014; 83: 240–245. doi: 10.1016/j.lungcan.2013.11.026 [DOI] [PubMed] [Google Scholar]
- 7.Wolf AS, Daniel J, Sugarbaker DJ. Surgical techniques for multimodality treatment of malignant pleural mesothelioma: extrapleural pneumonectomy and pleurectomy/decortication. Semin Thorac Cardiovasc Surg 2009; 21: 132–148. doi: 10.1053/j.semtcvs.2009.07.007 [DOI] [PubMed] [Google Scholar]
- 8.Rice D, Rusch V, Pass H, et al. Recommendations for uniform definitions of surgical techniques for malignant pleural mesothelioma: a consensus report of the International Association for the Study of Lung Cancer International Staging Committee and the International Mesothelioma Interest Group. J Thorac Oncol 2011; 6: 1304–1312. doi: 10.1097/JTO.0b013e3182208e3f [DOI] [PubMed] [Google Scholar]
- 9.Opitz I, Scherpereel A, Berghmans T, et al. ERS/ESTS/EACTS/ESTRO guidelines for the management of malignant pleural mesothelioma. Eur J Cardiothorac Surg 2020; 58: 1–24. doi: 10.1093/ejcts/ezaa158 [DOI] [PubMed] [Google Scholar]
- 10.Raskin J, Surmont V, Cornelissen R, et al. A randomized phase II study of pleurectomy/decortication preceded or followed by (neo-)adjuvant chemotherapy in patients with early stage malignant pleural mesothelioma (EORTC 1205). Transl Lung Cancer Res 2018; 7: 593–598. doi: 10.21037/tlcr.2018.05.07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lim E, Harris G, Patel A, et al. Preoperative versus postoperative chemotherapy in patients with resectable non-small cell lung cancer: systematic review and indirect comparison meta-analysis of randomized trials. J Thorac Oncol 2009; 4: 1380–1388. doi: 10.1097/JTO.0b013e3181b9ecca [DOI] [PubMed] [Google Scholar]
- 12.Verna V, Ahern CA, Berlind CG, et al. Treatment of malignant pleural mesothelioma with chemotherapy preceding versus after surgical resection. J Thorac Cardiovasc Surg 2019; 157: 758–766. doi: 10.1016/j.jtcvs.2018.10.039 [DOI] [PubMed] [Google Scholar]
- 13.Klotz LV, Hoffmann H, Shah R, et al. Multimodal therapy of epithelioid pleural mesothelioma: improved survival by changing the surgical treatment approach. Transl Lung Cancer Res 2022; 11: 2230–2242. doi: 10.21037/tlcr-22-199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Van Schil PE, Baas P, Gaafar R, et al. Trimodality therapy for malignant pleural mesothelioma: results from an EORTC phase II multicentre trial. Eur Respir J 2010; 36: 1362–1369. doi: 10.1183/09031936.00039510 [DOI] [PubMed] [Google Scholar]
- 15.Lim E, Waller D, Lau K, et al. MARS2: a multicentre randomised trial comparing (extended) pleurectomy decortication versus no (extended) pleurectomy decortication for patients with malignant pleural mesothelioma. J Thorac Oncol 2023; 18: Suppl., PL03.10. doi: 10.1016/j.jtho.2023.09.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Flores RM, Pass HI, Seshan VE, et al. Extrapleural pneumonectomy versus pleurectomy/decortication in the surgical management of malignant pleural mesothelioma: results in 663 patients. J Thorac Cardiovasc Surg 2008; 135: 620–626. doi: 10.1016/j.jtcvs.2007.10.054 [DOI] [PubMed] [Google Scholar]
- 17.Forde PM, Spicer J, Lu S, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med 2022; 386: 1973–1985. doi: 10.1056/NEJMoa2202170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lovly CM. Perioperative immunotherapy – a KEY toward improved outcomes for early-stage lung cancer? N Engl J Med 2023; 389: 560–561. doi: 10.1056/NEJMe2305762 [DOI] [PubMed] [Google Scholar]
- 19.Baas P, Scherpereel A, Nowak AK, et al. First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 2021; 397: 375–386. doi: 10.1016/S0140-6736(20)32714-8 [DOI] [PubMed] [Google Scholar]
- 20.Kok PS, Forde PM, Hughes B, et al. Protocol of DREAM3R: durvalumab with chemotherapy as first-line treatment in advanced pleural mesothelioma – a phase 3 randomised trial. BMJ Open 2022; 12: e057663. doi: 10.1136/bmjopen-2021-057663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chu QS, Piccirillo MC, Greiller L, et al. IND227 phase III (P3) study of cisplatin/pemetrexed (CP) with or without pembrolizumab (pembro) in patients (pts) with malignant pleural mesothelioma (PM): a CCTG, NCIN, and IFCT trial. J Clin Oncol 2023; 41: LBA8505. DOI: 10.1200/JCO.2023.41.17_suppl.LBA8505 [DOI] [Google Scholar]
- 22.BEAT-meso: Bevacizumab and Atezolizumab in Malignant Pleural Mesothelioma (BEAT-meso). NCT03762018. https://clinicaltrials.gov/study/NCT03762018.
- 23.Rimner A, Zauderer MG, Gomez DR, et al. Phase II study of hemithoracic intensity-modulated pleural radiation therapy (IMPRINT) as part of lung-sparing multimodality therapy in patients with malignant pleural mesothelioma. J Clin Oncol 2016; 34: 2761–2768. doi: 10.1200/JCO.2016.67.2675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Arrieta O, Ruiz F, Blake-Cerda M, et al. Locoregional control and toxicity after pleurectomy/decortication and intensity-modulated pleural radiation therapy in patients with malignant pleural mesothelioma. Thorac Cancer 2020; 11: 3448–3455. doi: 10.1111/1759-7714.13668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chemotherapy Followed by Surgery and Neoadjuvant Hemothoracic Intensity Modified Radiation Therapy (IMRT) for Patients with Malignant Pleural Mesothelioma. NCT02613312. https://clinicaltrials.gov/study/NCT02613312.
- 26.Dawson AG, Kutywayo K, Mohammed SB, et al. Cytoreductive surgery with hyperthermic intrathoracic chemotherapy for malignant pleural mesothelioma: a systematic review. Thorax 2023; 78: 409–417. doi: 10.1136/thoraxjnl-2021-218214 [DOI] [PubMed] [Google Scholar]
- 27.Migliore M, Fiore M, Filippini T, et al. Comparison of video-assisted pleurectomy/decortication surgery plus hyperthermic intrathoracic chemotherapy with VATS talc pleurodesis for the treatment of malignant pleural mesothelioma: a pilot study. Heliyon 2023; 9: e16685. doi: 10.1016/j.heliyon.2023.e16685 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material ERJ-02114-2023.Supplement (340.4KB, pdf)
Surgical guidelines ERJ-02114-2023.Guidelines (2.3MB, pdf)
This one-page PDF can be shared freely online.
Shareable PDF ERJ-02114-2023.Shareable (609.5KB, pdf)
Data Availability Statement
Individual participant data (after de-identification) collected during the trial, study protocol, statistical analysis plan and informed consent form will be made available for other researchers after a motivated request to and after approval from the EORTC.