Abstract
Background:
Dopaminergic neurons in the ventral tegmental area (VTA) are crucially involved in regulating arousal, making them a potential target for reversing general anesthesia. Electrical deep brain stimulation (DBS) of the VTA restores consciousness in animals anesthetized with drugs that primarily enhance GABAA receptors. However, it is unknown if VTA DBS restores consciousness in animals anesthetized with drugs that target other receptors.
Objective:
To evaluate the efficacy of VTA DBS in restoring consciousness after exposure to four anesthetics with distinct receptor targets.
Methods:
Sixteen adult Sprague-Dawley rats (8 female, 8 male) with bipolar electrodes implanted in the VTA were exposed to dexmedetomidine, fentanyl, ketamine, or sevoflurane to produce loss of righting, a proxy for unconsciousness. After receiving the dopamine D1 receptor antagonist, SCH-23390, or saline (vehicle), DBS was initiated at 30 μA and increased by 10 μA until reaching a maximum of 100 μA. The current that evoked behavioral arousal and restored righting was recorded for each anesthetic and compared across drug (saline/SCH-23390) condition. Electroencephalogram, heart rate and pulse oximetry were recorded continuously.
Results:
VTA DBS restored righting after sevoflurane, dexmedetomidine, and fentanyl-induced unconsciousness, but not ketamine-induced unconsciousness. D1 receptor antagonism diminished the efficacy of VTA stimulation following sevoflurane and fentanyl, but not dexmedetomidine.
Conclusions:
Electrical DBS of the VTA restores consciousness in animals anesthetized with mechanistically distinct drugs, excluding ketamine. The involvement of the D1 receptor in mediating this effect is anesthetic-specific.
Keywords: Ventral tegmental area, Dexmedetomidine, Fentanyl, Ketamine, General anesthesia, Dopamine D1 receptor
1. Introduction
General anesthesia is a drug-induced state of unconsciousness that makes it possible to conduct more than 200 million major surgical procedures around the world each year [1,2]. While general anesthesia can be induced by a variety of pharmacologically diverse agents, there are currently no drugs available to reverse this state in humans. Instead, clinicians rely on the pharmacokinetics of drug clearance to restore consciousness in anesthetized patients. Consequently, problems related to recovery from anesthesia such as delayed emergence, delirium, and cognitive dysfunction may occur. Delayed emergence can result in longer stays in the operating room and worsen patient outcomes [3]. Identifying anesthetic reversal agents is challenging because anesthetics have multiple receptor targets and produce their effects through mechanistically distinct actions [4]. Identifying common mechanisms capable of restoring consciousness following exposure to diverse anesthetics is therefore of great clinical interest.
The past decade has observed significant progress towards mapping brain loci and circuits associated with the process of anesthetic emergence. Notably, dopaminergic circuits originating in the ventral tegmental area (VTA), a midbrain structure known for its role in reward signaling, have garnered significant attention for their ability to induce or accelerate emergence from general anesthesia in rodent models [5–8]. Early investigations demonstrated that rats anesthetized with isoflurane or propofol could be rapidly returned to a conscious state by electrically stimulating the VTA [9]. Since then, multiple studies have shown that dopaminergic neurons projecting from the VTA onto dopamine D1 receptor (D1R)-expressing downstream targets, such as in the nucleus accumbens and prefrontal cortex, facilitate anesthetic emergence [10–14]. Thus far, investigations into anesthetic reversal with activation of VTA dopaminergic neurotransmission have focused on a subset of general anesthetics that primarily function by enhancing GABAA receptor activity – namely, isoflurane [9,12,15–17], sevoflurane [10,14,18,19], and propofol [9,13,19,20]. However, as anesthesia is maintained with pharmacologically distinct agents, including opioids, sedatives, and dissociative drugs, it is crucial to determine whether VTA activation promotes conscious recovery within the broader spectrum of anesthetic agents.
Here we assessed the efficacy of VTA DBS in rats after inducing unconsciousness with four drugs known to act via distinct receptor mechanisms: sevoflurane, dexmedetomidine (α2 adrenergic receptor agonist), fentanyl (μ-opioid receptor agonist), and ketamine (NMDA receptor antagonist). Further, we hypothesized that DBS-induced restoration of consciousness is mediated by dopamine via D1R-mediated signaling, and therefore will be attenuated by pretreatment with the D1R antagonist, SCH-23390. These studies may identify the VTA as a promising target for restoring consciousness across a broad range of anesthetics used clinically.
2. Material and methods
2.1. Animals
Adult male and female Sprague Dawley rats (Charles River Laboratories, Wilmington, MA, United States) aged 2–6 months (weight: 280g–400g at time of surgery) were housed with ad libitum access to water and chow on a 12-h light/dark cycle. Animals had chronic femoral venous catheters and one-channel magnetic Vascular Access Buttons (Instech Laboratories, Plymouth Meeting, PA) pre-implanted by Charles River Laboratories. Experiments were conducted during the day.
Previous work using electrical DBS to reverse isoflurane-induced unconsciousness identified a statistically significant effect using 5 animals per group [9]. Because the present investigation was designed to assess drugs not used previously and to detect potential sex differences, our target sample size was 16 animals (8 males and 8 females). Previous studies have identified significant sex differences in sensitivity to ketamine and dexmedetomidine in rodents with this sample size [21]. Power calculations were done using G*Power (Aichach, Germany) v3.1. With 16 animals, our study could capture differences in righting probability with effect sizes as small as w = 0.7 with a power of 0.8. Three animals with electrodes inadvertently placed outside the VTA were excluded from the analysis and replaced.
All animal procedures were approved by the Massachusetts General Hospital Institutional Animal Care and Use Committee and in accordance with the NIH Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize animal suffering. Reporting of the animal research in this study complies with the ARRIVE guidelines [22].
2.2. Electrode implantation surgery
General anesthesia was maintained at 1.5–2% isoflurane (Henry Schein, Melville, NY) delivered at 1L/min in 100 % oxygen for the duration of the surgery. Surgical procedures were as described previously [9,23]. In brief, rats were mounted on a stereotaxic apparatus (Model 960, Kopf Instruments, Tujunga, CA) and a microdrill (Patterson Dental Supply, Wilmington, MA) was used to perform craniotomies for the VTA DBS and electroencephalogram (EEG) electrode placements. A two-channel bipolar platinum-tungsten electrode (Plastics One, Wall-ingford, CT) was implanted in the left VTA using the following co-ordinates with reference to bregma: 4.80 mm anterior-posterior (AP), −0.90 mm medial-lateral (ML), and −8.35 mm dorsal-ventral (DV). The electrode was coated with the lipophilic long-chain dialkylcarbocyanine tracer DiI ((1,1′-dioctadecyl-3,3,3′,3′-tetramethylindo-carbocyanine perchlorate) to histologically confirm placement within the VTA.
Additional craniotomies were made for extradural frontal (+3.50 AP, −1.50 ML), parietal (−1.50AP, ±1.50ML), and cerebellar (−11.50AP, −1.50ML) EEG wires and ground screw (−10.00AP, +2.5 ML). Between six to eight anchor screws were affixed across the skull to secure the Electrode Interface Board (EIB-16, NeuraLynx, Bozeman, MT). Two electromyogram (EMG) wires were placed in the left and right nuchal muscles. EEG and EMG wires were affixed to the EIB-16 using large gold pins and secured to the skull with C&B Metabond (Parkell, Edgewood, NY) and dental acrylic. Animals received three doses of 4 mg/kg keto-profen subcutaneously at 24-h intervals. A minimum recovery period of at least 7 days was provided following implantation surgery, and the rats were individually housed following surgery for the duration of the study to avoid damaging the implants.
2.3. EEG recordings
Recordings took place in a custom-built acrylic chamber (30.48cm × 30.48cm × 30.48 cm) with separate ports for recording cables, gas inflow, gas scavenging, and monitoring of anesthetic agents. Recordings began one to 5 min prior to drug exposure and ended after animals regained righting. Signals were continuously recorded with an Omniplex D Neural Data Acquisition System (Plexon, Dallas, TX). Analog signals were amplified with a 1x gain 16-channel headstage (HST/32V-G1 LN, Plexon), digitized with a sampling rate of 40 kHz with Plexon Mini-DigiAmp, and digitally filtered (Bessel, 4 poles, 200Hz cut off) and down-sampled to 1 kHz in OmniPlex Server.
2.4. VTA DBS protocol and return of righting assessment
After drug-induced loss of righting, DBS was performed using a Multichannel Systems stimulus generator (STG4004; ALA Scientific, Farmingdale, NY). Stimulation was initiated using a 100Hz square wave with a current intensity of 30 μA for 30s, followed by a 30s rest period [9]. If righting did not occur, the current was increased by 10 μA increments, until a maximum of 100 μA was reached. If the animal righted during the stimulation, the current was turned off immediately and 30 s later, the experimenter returned the animal to a supine position and the final righting time was recorded. At least 72 h of rest were provided between experiments.
2.5. Initial testing of VTA DBS efficacy during continuous sevoflurane anesthesia
An initial test was conducted one week after surgery to assess behavioral responsiveness to VTA DBS during sevoflurane anesthesia. The minimum dose of sevoflurane required to maintain loss of righting was adapted based on previous methods [24]. Rats were placed in an anesthetizing chamber, and the inhaled concentration of sevoflurane was initially fixed to 3 % for 20 min to allow the brain to reach a steady-state. The concentration of sevoflurane in the chamber was monitored continuously by a Datex Ohmeda S/5 anesthesia monitor, and normothermia was maintained with a heating pad. Sevoflurane was then reduced to 1.2 % for 20 min. If the rat righted within that 20-min period, sevoflurane was increased to 3 % until loss of righting was re-established, and the concentration was then brought down to 1.4 % for 20 min. The process was repeated with sevoflurane increasing by increments of 0.2 % until loss of righting was maintained for 20 min, and then the DBS protocol was initiated. Only rats that righted during VTA DBS were retained for the study. Three rats were excluded due to lack of response to stimulation, and 8 females and 8 males were included in the final analysis. The final two sevoflurane tests were conducted at the end of the study to confirm the continued functionality of the VTA DBS electrode (Fig. 1).
Fig. 1. Study design.
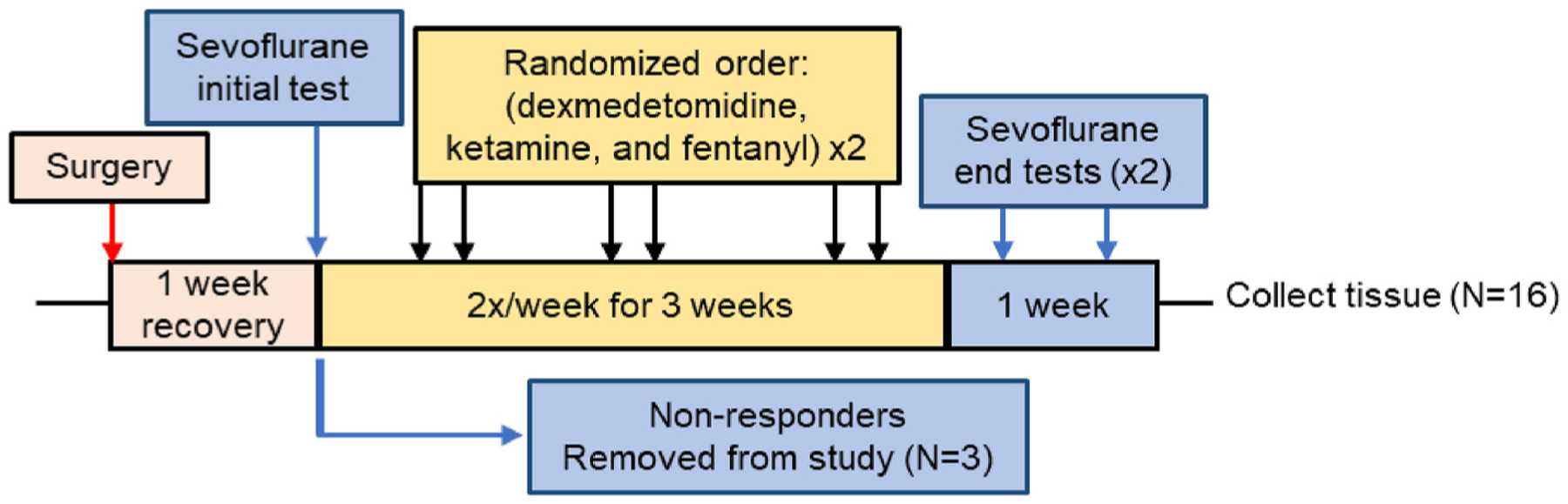
One week following electrode implantation surgery, all animals underwent DBS during continuous sevoflurane anesthesia (N = 19). Animals that did not right under sevoflurane (“Non-responders,” N = 3) were removed from the study and brains collected. The remaining 16 animals that righted with DBS during sevoflurane anesthesia were then tested with dexmedetomidine, ketamine, and fentanyl in a blocked randomized order with either saline or SCH-23390. Rats underwent DBS a maximum of twice per week, with 72 h between tests. After receiving each intravenous anesthetic twice, rats were tested under sevoflurane twice with either saline or SCH-23390 in a randomized order. Finally, rats were euthanized, and tissues collected.
2.6. Intravenous anesthetic testing
Intravenous anesthetics were administered through an implanted femoral vein catheter. If the catheter lost patency, a temporary intravenous catheter was placed in the lateral tail vein under brief isoflurane anesthesia at least 1 h prior to testing, as previously described [21]. Doses of dexmedetomidine (50 μg/kg over 10 min), ketamine (50 mg/kg over 10 min), and fentanyl (55 μg/kg over 15 min) were based on those previously used to induce a protracted state of unconsciousness in rats [21,25]. Animals were exposed to all three anesthetics twice (for a total of six testing sessions) and the order was randomized by Latin Square design. Immediately following anesthetic delivery, the rats also received either 0.5 mg/kg i.v. of the D1R-antagonist SCH-23390 (R(+)-7-Chlor-o-8-hydroxy-3-methyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-benzazepine hydrochloride, Sigma Aldrich, St. Louis, MO) or saline (vehicle). Each anesthetic agent produces distinct behavioral characteristics prior to loss of righting, making it impossible to blind the experimenter to the identity of the anesthetic. However, the experimenter was blind to the saline or SCH-23390 drug condition for all experiments. Unblinding occurred only after all data was collected for the study.
2.7. Arousal scoring
To capture additional behavioral correlates of arousal during VTA DBS, two evaluators blind to the saline/SCH-23390 condition reviewed all video recordings to assess when the first head, forelimb, hindlimb, and tail movements were observed. Movement was defined as any observable change in the position of the specified body part during the period when DBS was ongoing.
2.8. Histology
After all DBS experiments were completed, animals were deeply anesthetized with isoflurane and perfused with phosphate-buffered saline followed by a 10 % buffered formalin solution. Brains were removed and postfixed in formalin overnight. The brains were sectioned (50 μm) using a VT1000 S vibratome (Leica Microsystems, Buffalo Grove, IL). Nuclei were counterstained with 4’,6-diamidino-2-phenylindole (DAPI), and imaged using an AxioImager fluorescent microscope (Zeiss, Ober-kochen, Germany). Stained sections were compared with a rat brain atlas, and the brain region at the deepest point of the electrode was identified for each animal. We used the definition of the VTA provided by Paxinos and Watson, which includes the paranigral, para-interfascicular, and parabrachial pigmented nuclei, as well as the rostral part of the VTA, and extends from Bregma −4.56 to −6.84 mm. Of the three rats excluded from the study due to lack of response to DBS, all were found to have DBS electrodes outside the VTA region.
2.9. EEG analysis
Power spectra of frontal EEG recordings were generated using MATLAB 8.4 (MathWorks) and the Chronux software package (Cold Spring Harbor Laboratory). Spectrograms were computed using a 5s window stepped by 2.5s and constructed from 0 to 40Hz using three tapers and a bandwidth of 1Hz [16]. Spectrograms included for quantification are from 4 to 6 animals per anesthetic condition that maintained functional EEGs throughout the experiment. For each drug tested, group power spectral densities (PSDs) were calculated from pooled EEG data from each condition (SCH-23390 and saline). Fifteen-second epochs were used to generate PSDs. Three epochs were chosen: 1) prior to anesthetic delivery, 2) 1 min prior to DBS (i.e. following anesthetic and saline/SCH-23390 delivery), and 3) during DBS at the current intensity that elicited a righting response, or at 100 μA if no righting occurred. Differences in median power between the three epochs within each condition were compared at each frequency band, defined as follows: delta (1–4Hz), theta (4–8Hz), alpha (8–12Hz), beta (12–35Hz), and low gamma (35–40Hz).
2.10. Statistical analysis
All statistical analyses were conducted using RStudio (Ri386 version 4.1.3). We assessed the probability of righting or movement under saline or SCH-23390 for each anesthetic using the R package ‘coxme’ for mixed factor survival analysis [26]. Pre-treatment condition (saline or SCH-23390) was set as a fixed effect and individual rat identity as a random intercept. The model was fit by maximum likelihood method. Integrated log likelihood chi squared values are reported for each model. For sevoflurane experiments, where three tests were compared, log likelihood estimates for all contrasts are reported with Bonferroni post-hoc correction.
Final return of righting latency following dexmedetomidine, fentanyl, and ketamine was compared between saline- and SCH-23390-treated rats using R studio package ‘lmer’ employing the Satterthwaite approximation for degrees of freedom and fit by restricted maximum likelihood method [27]. Treatment condition and sex were set as fixed effects and animal as a random intercept.
Mean spectral power at each defined frequency band was compared across three temporal epochs (pre-anesthetic, 1 min pre-DBS, and during DBS at the maximum current) and between saline- and SCH-23390-treated conditions using the R studio package ‘lmer’. Treatment condition, epoch, and band frequency were set as fixed effects and animal as a random intercept. When a significant interaction was found, pairwise comparisons were assessed using Bonferroni correction.
3. Results
3.1. VTA DBS during continuous sevoflurane anesthesia elicits arousal and restores righting that is attenuated by D1R antagonism
Histological analysis revealed VTA electrode placement in 16 of 19 rats (Fig. 2A). As shown in Fig. 2B, three rats that did not exhibit arousal or return of righting with DBS during continuous sevoflurane anesthesia were found to have electrodes inadvertently placed outside the VTA (ventral posteromedial thalamus, parafascicular thalamus, and substantia nigra). Data from these animals, which were removed from all further experiments, demonstrate that arousal and righting in response to DBS is location-dependent. In all 16 rats with VTA electrodes, stimulation during the first sevoflurane exposure elicited arousal and restored righting (Fig. 2C). The median current that induced righting during sevoflurane anesthesia was 35 μA.
Fig. 2. VTA DBS restores righting during continuous sevoflurane anesthesia.
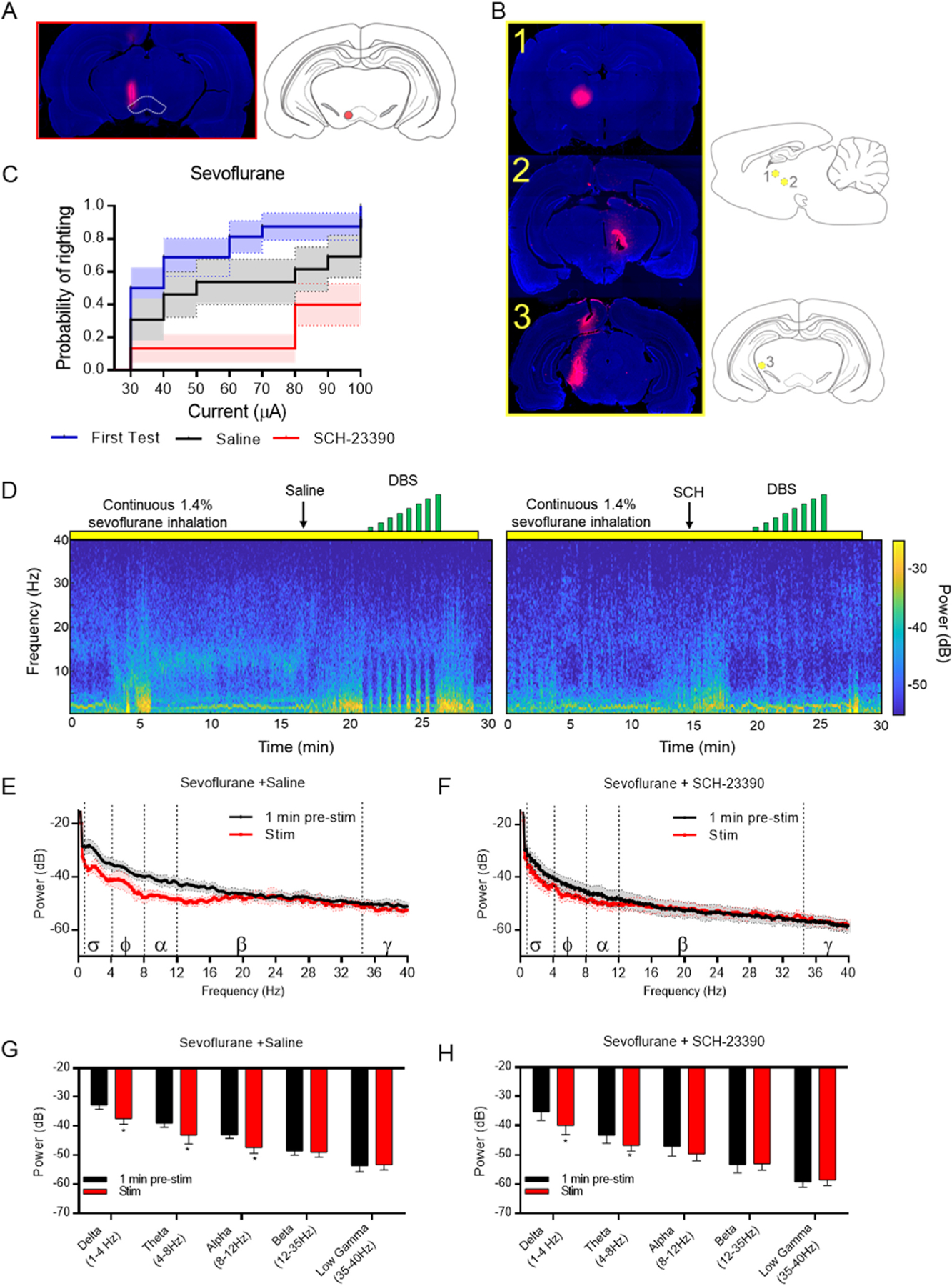
A) Left: Representative histology of the electrode DiI track in a rat that completed the study with the VTA outlined in white. Right: Deepest region with DiI staining depicted in red. B) Left: Histology from the three rats that did not respond to DBS during sevoflurane anesthesia. DiI tracks confirm that electrode placement was outside the VTA. Right: Depiction of the deepest region with DiI from each of the three rats removed from the study shown in yellow. C) Probability of righting during VTA DBS at indicated currents during continuous sevoflurane anesthesia. Survival curve depicts the first sevoflurane test (blue), and the last two sevoflurane tests following saline (black) or SCH-23390 pre-treatment (red) (N = 16). D) Representative spectrograms from the same rat under continuous sevoflurane anesthesia, pretreated with either saline (left) or SCH-23390 (right) followed by DBS (indicated by green bars). E) Power spectral density (PSD) plots comparing 1 min prior to DBS (black) and during DBS at the maximum current delivered (red) after saline pre-treatment. Mean ± SEM, N = 4. F) PSD plots comparing 1 min prior to DBS (black) and during DBS at the maximum current delivered (red) after SCH-23390 pre-treatment. Mean ± SEM, N = 5. G-H) Median spectral power (and 95%CI) at indicated frequency bands during continuous sevoflurane anesthesia after pretreatment with (G) saline or (H) SCH-23390. * = p < 0.05, ** = p < 0.01.
To confirm that electrodes remained functional throughout the study, two final tests were conducted under sevoflurane anesthesia following either intravenous saline or SCH-23390 (0.5 mg/kg), a selective D1R antagonist. The probability of righting during each of the three sevoflurane tests was compared using a mixed cox regression model. The likelihood of righting during the first sevoflurane test and during the final test with saline treatment was not significantly different (HR = 1.879, 95%CI[0.88,4.00], p = 0.3034) (Fig. 2C). No significant sex differences were observed (Supplemental Figs. 1A–B). These data demonstrate that repeated testing with different anesthetic agents did not attenuate the efficacy of DBS to induce righting over time. However, movement of the head, forelimbs, hindlimbs, or tail during DBS did significantly differ between the first and final test with saline, with greater current needed before the first movements were observed in the final test (Supplemental Figs. 2A–D). During the final tests, saline-treated rats were significantly more likely to right during VTA DBS than SCH-23390-treated rats (HR = 4.486, 95%CI[1.57,12.83], p = 0.0154) (Fig. 2C). These data suggest that VTA DBS is in part mediated by the release of dopamine and its subsequent activation of D1Rs on downstream targets.
To assess whether the righting differences observed between saline- and SCH-23390-treated rats produced differences in frontal EEG spectra (depicted in Figs. 2D), 30s epochs from 1 min prior to DBS were compared to a 30s epoch during DBS at the maximum current applied. In both saline- and SCH-23390-treated rats, only modest changes in frontal PSDs were observed during DBS (Fig. 2E–H). VTA DBS during continuous sevoflurane anesthesia following saline treatment significantly reduced delta (t(27) = 2.947, p = 00.0066), theta (t(27) = 2.574, p = 0.0159), alpha (t(27) = 2.721, p = 0.0113), and power. VTA DBS during continuous sevoflurane anesthesia following SCH-23390 treatment significantly reduced delta power (t(36) = 3.228, p = 0.0023) and theta power (t(36) = 2.438, p = 00.0188).
3.2. VTA DBS following dexmedetomidine-induced unconsciousness elicits arousal and restores righting independent of D1R antagonism
Dexmedetomidine (50 μg/kg over 10 min) caused loss of righting in all animals. After either saline or SCH-23390, VTA DBS induced righting in 12/16 rats (Fig. 3A) and produced comparable movement in the head, limbs, and tail (Supplemental Fig. S2 E–H). The median current to induce righting was 55 μA in both saline- and SCH-23390-treated rats. This was not statistically different. There was no effect of sex on probability of righting for either treatment condition (Supplemental Figs. 1B–C).
Fig. 3. VTA DBS restores righting following dexmedetomidine-induced unconsciousness, independent of D1R antagonism.
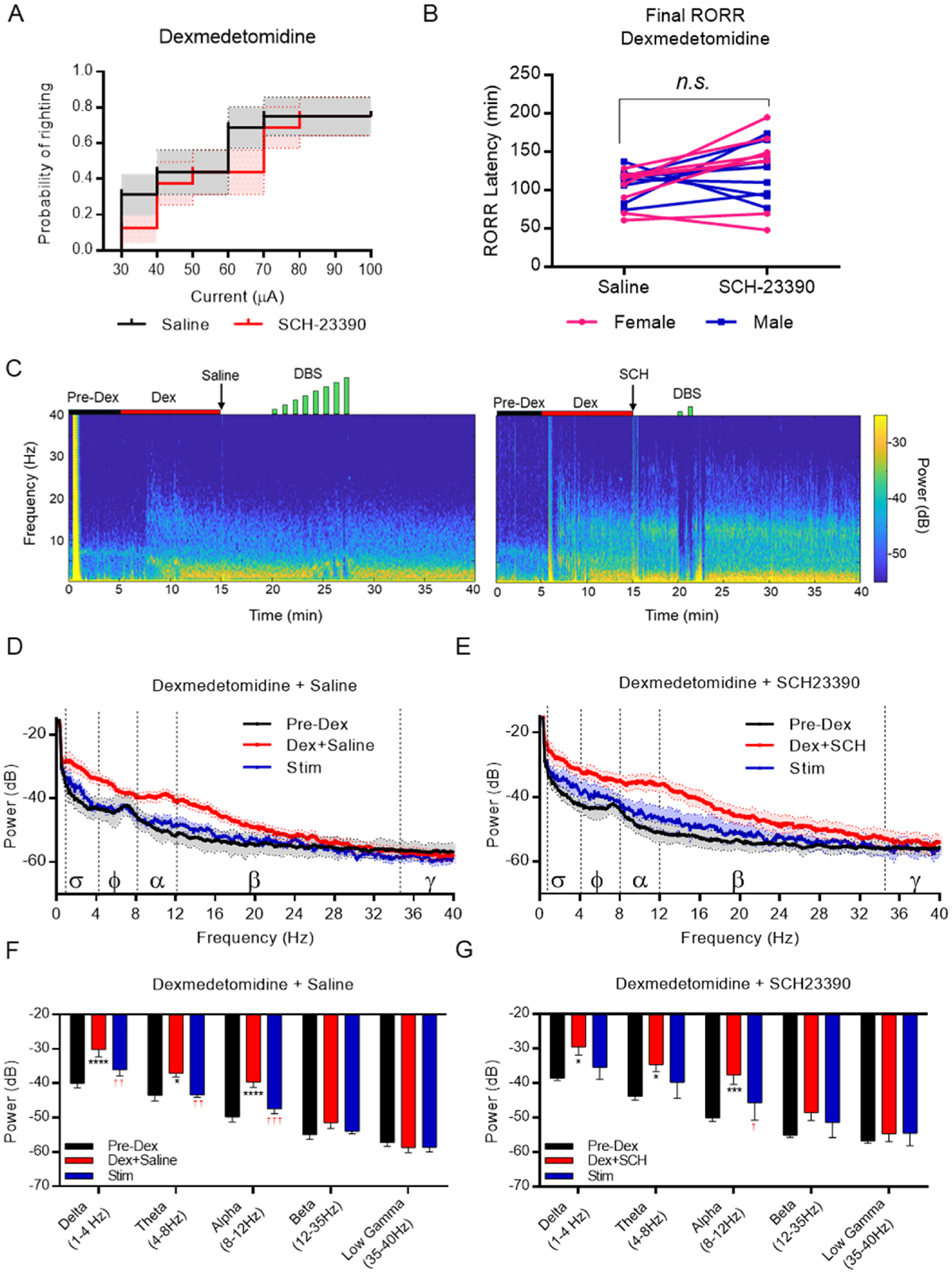
A) Probability of righting following dexmedetomidine-induced unconsciousness in rats pre-treated with either saline of SCH-23390 (N = 16). B) Final latency to return of righting following dexmedetomidine-induced unconsciousness in rats pretreated with either saline or SCH-23390 (N = 16). C) Representative spectrograms from a rat treated with dexmedetomidine and either saline (left) or SCH-23390 (right) followed by DBS (in green). D) PSD plots comparing epochs from the pre-dexmedetomidine awake state (black), following dexmedetomidine delivery and saline treatment (red), and the maximum current delivered (blue). Mean ± SEM, N = 6. E) PSD plots comparing epochs from the pre-dexmedetomidine awake state (black), following dexmedetomidine delivery and SCH-23390 treatment (red), and the maximum current delivered (blue). Mean ± SEM, N = 6. F-G) Median spectral power (and 95%CI) at indicated frequency bands of the same epochs in D-E in (F) saline-treated and (G) SCH-23390-treated rats. * = p < 0.05, ** = p < 0.01 versus awake epoch. † = p < 00.05, †† = p < 00.01 versus Dex+drug epoch. N = 6.
In rats that righted with DBS, discontinuing stimulation resulted in loss of righting being re-established in 30 s for all animals. The final latency for return of righting showed a modest increase following SCH-23390 that was not statistically significant (Ftrt(1,15.09) = 2.3695, p = 0.1426) (Fig. 3B).
We next assessed frontal EEG (representative traces shown in Fig. 3C) power spectra at three 30-s epochs: before dexmedetomidine (Pre-Dex), following dexmedetomidine and drug treatment (Dex+saline or Dex+SCH), and during VTA electrical stimulation (Stim) (Fig. 3D–G). The mixed model revealed a significant interaction between epoch and band frequency (Fepoch×band(8,142) = 2.8947, p = 0.0051), but no main effect or interaction of drug treatment. Bonferroni-corrected pairwise comparisons revealed that dexmedetomidine significantly increased power in the delta, alpha, and theta bands compared with baseline (Fig. 3F). VTA stimulation significantly reduced the dexmedetomidine-induced increase in delta, theta, and alpha power (Fig. 3F). In the SCH-23390 condition, VTA stimulation significantly reduced the Dex+SCH-induced increase in alpha (Fig. 3G).
3.3. VTA DBS following fentanyl-induced unconsciousness elicits arousal and restores righting that is attenuated by D1 antagonism
Fentanyl (55 μg/kg over 15 min) caused loss of righting in all animals. With saline pre-treatment, VTA DBS induced movement in the head, limbs, and tail (Supplemental Figs. 2I–L) and restored righting in 15/16 rats with a median current of 35 μA (Fig. 4A). When rats were pretreated with SCH-23390, VTA DBS restored righting in 12/16 rats with a median current of 65 μA. Mixed effects Cox regression revealed that SCH-23390 significantly reduced the likelihood of righting with VTA DBS (HR = 0.3506, 95%CI[0.18,0.70], p = 0.0150). There was no effect of sex on probability of righting in either treatment condition (Supplemental Figs. 1E–F).
Fig. 4. VTA DBS restores righting following fentanyl-induced unconsciousness, which is attenuated by D1R antagonism.
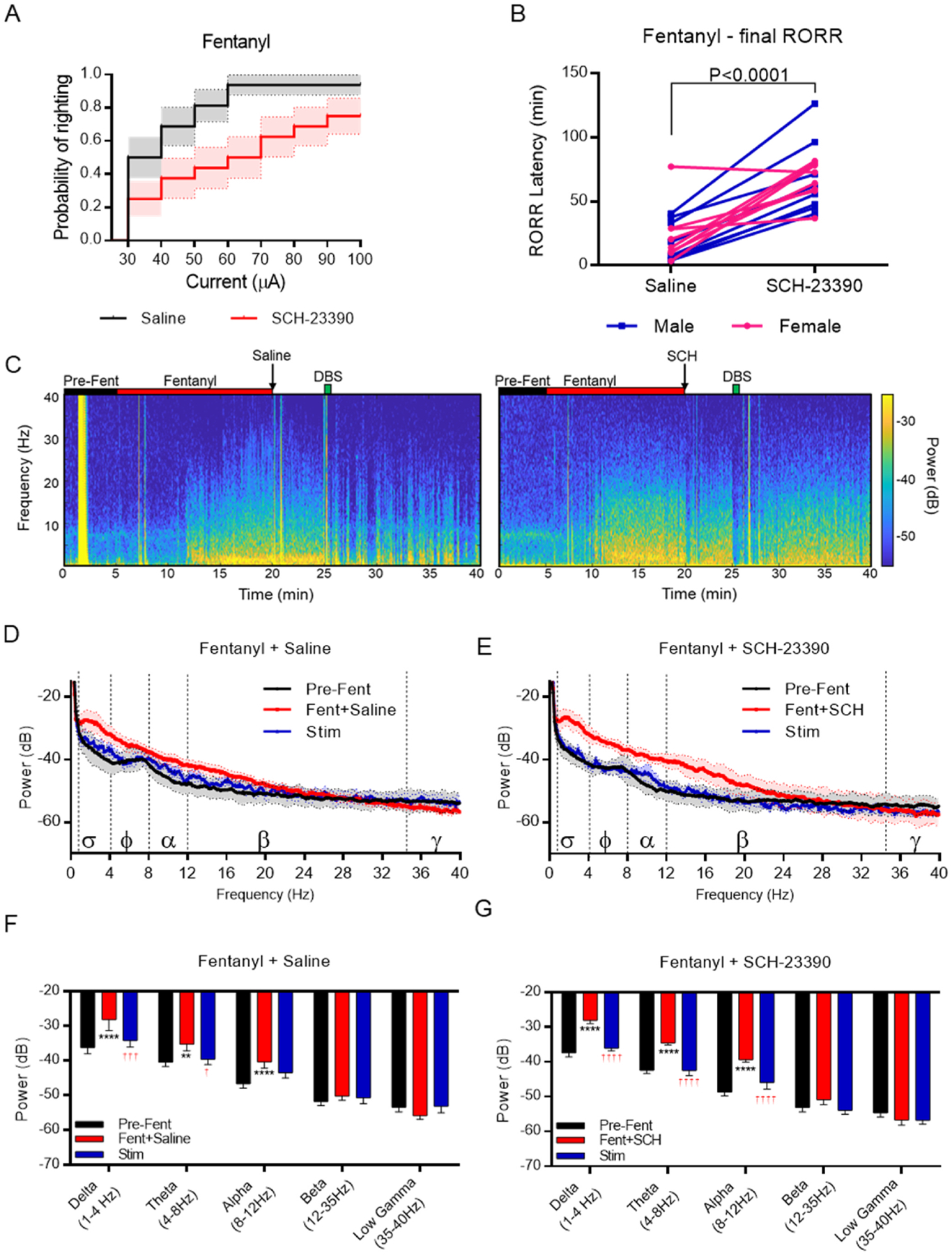
A) Probability of righting following fentanyl-induced unconsciousness in rats treated with either saline or SCH-23390 (N = 16). B) Final latency to return of righting following fentanyl-induced unconsciousness in rats pretreated with either saline or SCH-23390 (N = 16). C) Representative spectrograms from a rat treated with fentanyl and either saline (left) or SCH-23390 (right) followed by DBS (in green). D) PSD plots comparing epochs from the pre-fentanyl awake state (black), following fentanyl delivery and saline treatment (red), and during the maximum current delivered (blue). Mean ± SEM, N = 5. E) PSD plots comparing epochs from the pre-fentanyl awake state (black), following fentanyl delivery and SCH-23390 treatment (red), and the maximum current delivered (blue). Mean ± SEM, N = 5. F-G) Median spectral power (and 95% CI) at indicated frequency bands of the same epochs in D-E in (F) saline-treated and (G) SCH-23390-treated rats. * = p < 0.05, *** = p < 0.001, **** = p < 0.0001 versus awake epoch. † = p < 00.05, ††† = p < 00.001, †††† = p < 00.0001 versus Fentanyl+drug epoch. N = 5.
Shortly after discontinuing VTA DBS, all animals could be returned to a supine position. A mixed effects model comparing sex and drug treatment on final latency for return of righting revealed a significant main effect of drug treatment (Ftrt(1,16.032) = 30.8947, p < 0.0001), but not sex (Fsex(1,14.025) = 0.4095, p = 0.5325) (Fig. 4B). Pretreatment with SCH-23390 increased final righting time by 42.2 min (95%CI[24.6,59.7]) in fentanyl-treated rats.
Frontal EEG spectra revealed a significant increase in power in the delta, theta, and alpha bands following fentanyl (Fig. 4D and E). Three-way ANOVA comparing epoch, frequency band, and drug condition revealed a significant interaction of band and epoch (Fband×epoch(8,145) = 10.04, p < 0.0001) but not a three-way interaction. In both saline-and SCH-23390-treated rats, VTA DBS significantly reversed fentanyl-induced changes in spectral power in the low frequency bands (Fig. 4F and G).
3.4. VTA DBS following ketamine-induced unconsciousness does not restore righting
Ketamine (50 mg/kg over 10 min) caused loss of righting in all animals. Regardless of drug condition, VTA DBS did not restore righting in 15/16 rats, with only one rat per drug condition responding at any current (Fig. 5A). In both instances of righting, animals could be returned to a supine position 30 s after turning off the stimulation. Despite not producing a robust righting response, VTA DBS did produce observable movements in the head, limbs, and tail in ketamine-treated animals (Supplemental Figs. 2M–P). Interestingly, SCH-23390 decreased VTA DBS-induced head and forelimb movement (Supplemental Figs. 2M–N).
Fig. 5. VTA DBS does not elicit arousal or restore righting following ketamine-induced unconsciousness.
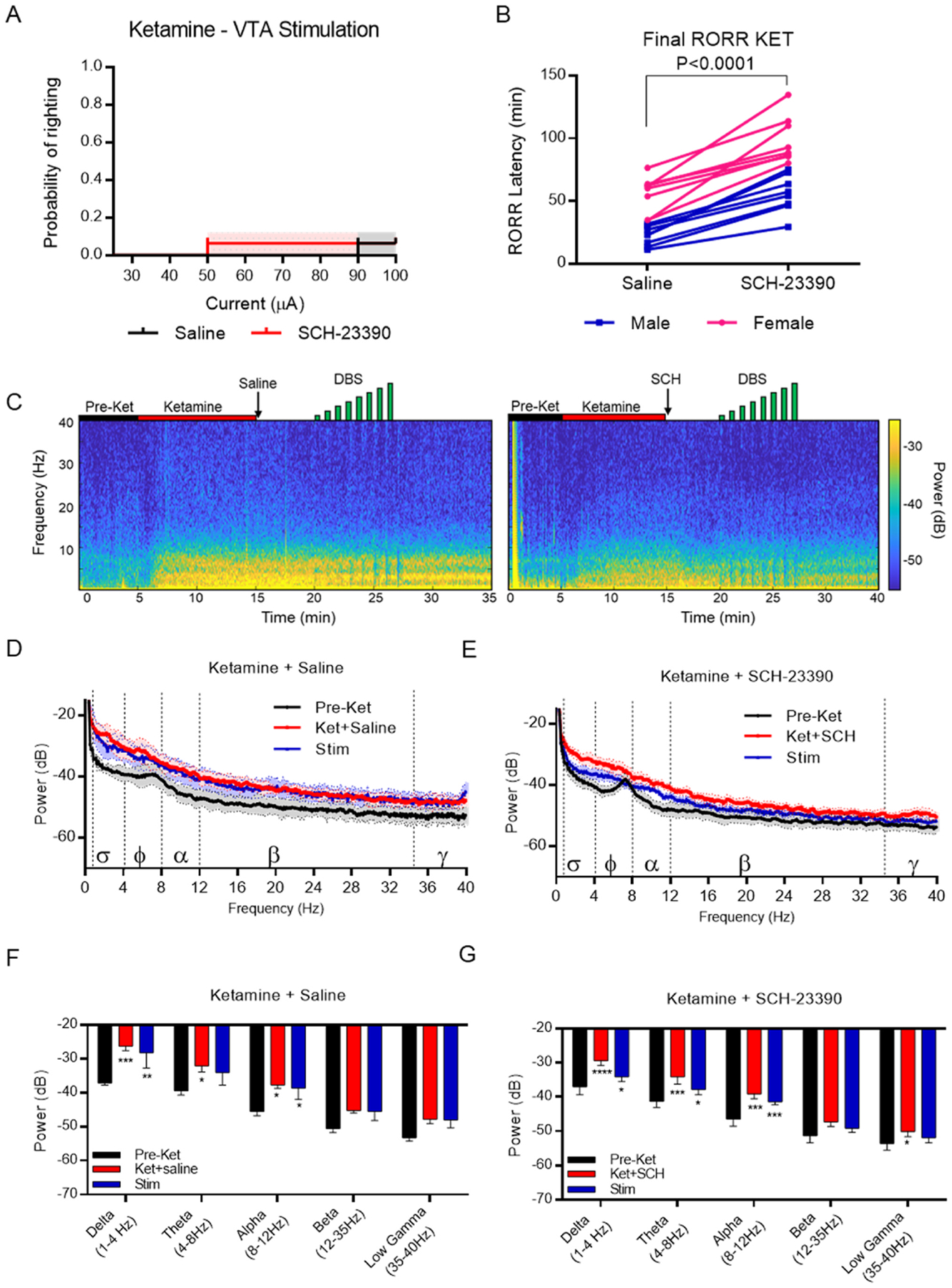
A) Probability of righting following ketamine-induced unconsciousness in rats treated with either saline of SCH-23390 (N = 16). B) Final latency to return of righting following ketamine-induced unconsciousness in rats pretreated with either saline or SCH-23390 (N = 16). C) Sample spectrograms from the same rat treated with ketamine and either saline (left) or SCH-23390 (right) followed by DBS (in green). D) PSD plots comparing epochs from the pre-ketamine awake state (black), following ketamine delivery and saline treatment (red), and during the maximum current delivered (blue). Mean ± SEM, N = 5. E) PSD plots comparing epochs from the pre-ketamine awake state (black), following ketamine delivery and SCH-23390 treatment (red), and the maximum current delivered (blue). Mean ± SEM, N = 5. F-G) Mean spectral power at indicated frequency bands of the same epochs in D-E in (F) saline-treated and (G) SCH-23390-treated rats. * = p < 0.05, ** = p < 0.01, *** = p < 0.001, **** = p < 0.0001 versus awake epoch. N = 5.
Final recovery of righting was significantly affected by both sex (Fsex(1,14.1) = 46.93, p < 0.0001) and drug treatment (Ftrt(1,15.4) = 49.74, p < 0.0001) (Fig. 5B). Regardless of drug treatment, female rats took 34.2 min (95%CI[18.9,49.5]) longer to right than males; across both sexes, SCH-23390 treatment delayed righting by an average of 43.1 min (95%CI[28.8,57.4]). There was no interaction between sex and drug treatment.
Representative spectrograms computed from frontal EEG recordings after ketamine are shown (Fig. 5C). Ketamine produced a protracted change in the frontal EEG signature which was not restored with VTA DBS (Fig. 5D and E). The mixed model revealed a significant main effect of epoch (Fepoch(2,115) = 29.30, p < 0.0001) on spectral power. Simple effects with Bonferroni correction identified that ketamine significantly increases power in the delta, theta, and alpha bands compared with the awake state in both saline- and SCH-23390-treated rats (Fig. 5F and G). VTA DBS did not reverse the ketamine-induced increase in lower frequency spectral power for either drug condition (Fig. 5F and G). Moreover, direct comparisons across anesthetic conditions reveal that EEG spectral power with VTA DBS during ketamine-induced unconsciousness remains significantly elevated compared with VTA DBS during dexmedetomidine- and sevoflurane-induced unconsciousness (Supplemental Figs. 3A–B). These data support the notion that ketamine anesthesia is uniquely resistant to the arousal-promoting effects of VTA DBS in rats.
4. Discussion
Here we demonstrate that VTA DBS reverses sevoflurane-, dexmedetomidine-, and fentanyl-induced unconsciousness, but not ketamine-induced unconsciousness, in both male and female rats. Interestingly, the probability of righting with VTA DBS is reduced by treating rats with the D1R antagonist, SCH-23390, following sevoflurane and fentanyl, but not dexmedetomidine. SCH-23390 treatment also delayed final righting latency following fentanyl and ketamine, but not dexmedetomidine. Finally, frontal EEG recordings revealed that VTA DBS generates broadband decreases in power for frequencies <20Hz in rats rendered unconscious with sevoflurane, dexmedetomidine, and fentanyl, but not ketamine.
Previous work demonstrated that VTA DBS induces emergence from isoflurane and propofol anesthesia in rats [9]. Importantly, when the electrode was placed in the substantia nigra, a dopaminergic nucleus lateral to the VTA, anesthetized animals did not exhibit signs of behavioral arousal during DBS. In this study, we found that in three rats with electrodes implanted outside the VTA, DBS with a maximum current of 100 μA produced no righting response during continuous sevoflurane anesthesia. Taken together, these data confirm that arousal and recovery of righting observed with DBS is region-specific.
As a selective α2-adrenergic receptor agonist, dexmedetomidine’s sedating effects have been primarily attributed to inhibition of arousal-promoting noradrenergic signaling by the locus coeruleus [28,29]. The locus coeruleus and VTA have extensive reciprocal projections [30–32]. Chemogenetic inhibition of locus coeruleus neurons attenuates phasic VTA firing [33], and chemical lesions of the locus coeruleus attenuate release of dopamine in the nucleus accumbens [34]. Similarly, both intravenous dexmedetomidine and local injections of dexmedetomidine into the locus coeruleus in rats have been shown to decrease dopamine levels in the nucleus accumbens in a dose-dependent manner [35,36]. Although SCH-23390 potently inhibits the arousal response and restoration of righting induced by d-amphetamine in rats rendered unconscious with dexmedetomidine [21], in this study we unexpectedly found no effect of SCH-23390 on VTA DBS-induced righting nor movement following dexmedetomidine. This suggests that d-amphetamine’s actions primarily involve a D1R-dependent mechanism, whereas VTA DBS promotes arousal via additional mechanisms that do not involve D1 receptors.
Approximately 50 % of VTA projections are non-dopaminergic [37]; though a portion of these neurons include inhibitory GABAergic neurons, glutamatergic efferents to the nucleus accumbens and prefrontal cortex account for 10 % and 40 % of non-dopaminergic VTA projections, respectively [38]. In retrograde labelling studies, only 1 % of projections from the VTA to the locus coeruleus were shown to be dopaminergic [32]; hence, arousal and righting induced by VTA DBS in dexmedetomidine-treated animals may be driven by disinhibiting the locus coeruleus via a dopamine-independent pathway. More recently, glutamatergic neurons from the VTA projecting to the lateral septum have been shown to promote emergence from isoflurane anesthesia [39]. As electrical stimulation is not cell-type specific, future investigations using more selective techniques, such as optogenetics, may shed light on the involvement of different cell types in regulating arousal.
In contrast to dexmedetomidine, VTA DBS-induced arousal and righting following fentanyl appears to be partially mediated by dopaminergic signaling. While most animals recovered righting with VTA DBS in the presence of SCH-23390, significantly higher currents were required compared with saline-treated animals. Additionally, SCH-23390 significantly delayed final return of righting following fentanyl. These results are consistent with previous work showing that d-amphetamine accelerates recovery from fentanyl-induced unconsciousness [25]. In addition to being a μ-opioid receptor agonist, radi-oligand binding assays have identified that fentanyl, but not morphine, is an antagonist of the D1R and the vesicular transporter for monoamine storage, VMAT2 [40]. Although in vitro and in vivo rodent studies have shown that opioids disinhibit VTA neurons [41–43] and increase extracellular dopamine concentrations in the nucleus accumbens [44], we hypothesize that the high doses of fentanyl used in the present study were sufficient to inhibit D1R, leading to unconsciousness. This would explain why VTA DBS caused reversal of these fentanyl-induced effects. The present findings potentially identify the VTA as a novel target for reversal of fentanyl-induced unconsciousness.
VTA DBS did not restore righting in rats anesthetized with ketamine, though bodily movements associated with arousal were detected. Meta-analyses of rodent data show that acute, sub-anesthetic doses of ketamine enhance firing in the VTA and increase dopamine levels in the cortex, striatum, and nucleus accumbens [45]. One mechanism posited for these findings is through NMDA receptor blockade on GABAergic VTA interneurons. By inhibiting GABAergic interneurons, ketamine produces disinhibition of VTA glutamatergic neurons, leading to enhanced excitatory activity and dopamine release [45]. Therefore, ketamine-induced unconsciousness likely involves a mechanism that persists in the presence of elevated dopaminergic activity. Indeed, ketamine paradoxically activates several arousal-promoting nuclei, including noradrenergic neurons in the locus coeruleus and cholinergic neurons in the basal forebrain [46], while concurrently producing unconsciousness at high doses. How unconsciousness is maintained in the presence of elevated arousal-promoting neurotransmitters remains un-clear. However, we found that SCH-23390 treatment inhibited VTA DBS-induced head and forelimb movements and delayed righting in ketamine-treated animals, suggesting D1R antagonism increases sensitivity to ketamine-induced unconsciousness.
Finally, we also observed a significant impact of sex on latency to return of righting following ketamine, with females taking significantly longer to recover than males. This finding is consistent with previous work using equivalent doses of ketamine in rats [21]. Sex differences in anesthetic sensitivity are under active investigation in the field. Particularly, the contribution of gonadal hormones to anesthetic sensitivity is being extensively characterized in both preclinical [47–50] and clinical contexts [51,52]. Rodent studies have now identified testosterone as a key modulator of anesthetic sensitivity [48]. In the present study, however, sex did not impact probability of righting during VTA DBS for any drug condition tested.
This investigation has several limitations to consider. First, VTA DBS does not exclusively activate VTA neurons. While we observed that rodents with misplaced electrodes did not respond to electric stimulation, the involvement of neighboring projections cannot be fully excluded. Ultimately, the findings of this study identify the VTA as a region of interest to explore with more selective techniques. Additionally, VTA DBS may produce more profound changes in EEG activity than observed in this study. When DBS resulted in rapid recovery of the righting reflex, often within seconds of turning on the current, the stimulation was terminated, potentially before significant changes could be detected in cortical EEG readings. More profound and persistent neurophysiological alterations might be observed by extending the duration of VTA stimulation. Finally, the behavioral measures reported – righting and arousal scoring – correlate with level of consciousness (i.e., arousal). These measures do not capture the content of consciousness [53–55], nor do they provide insights into cognitive recovery with stimulation. Whether the arousal induced by VTA DBS facilitates cognitive recovery will be a clinically important question to address in future studies.
5. Conclusions
VTA DBS restores arousal and righting in rats rendered unconscious with sevoflurane, dexmedetomidine, and fentanyl, but not ketamine. VTA DBS-induced righting is significantly inhibited by D1R antagonism following sevoflurane, modestly inhibited following fentanyl, and not affected following dexmedetomidine. These results suggest that while VTA DBS elicits a robust behavioral arousal response following the administration of mechanistically distinct anesthetics, these effects may not be driven by the same neural circuits.
Supplementary Material
Funding
This work was supported by the National Institutes of Health [R01-GM126155], Bethesda, Maryland and the Department of Anesthesia, Critical Care and Pain Medicine at Massachusetts General Hospital.
Declaration of competing interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:
Ken Solt reports financial support was provided by National Institutes of Health and the Department of Anesthesia, Critical Care and Pain Medicine at Massachusetts General Hospital. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Abbreviations:
- VTA
ventral tegmental area
- EEG
electroencephalography
- D1R
dopamine D1 receptor
- DBS
deep brain stimulation
Footnotes
CRediT authorship contribution statement
Kathleen F. Vincent: Writing – original draft, Project administration, Methodology, Investigation, Formal analysis, Data curation. Edlyn R. Zhang: Writing – review & editing, Writing – original draft, Project administration, Methodology, Investigation, Formal analysis, Data curation. Angel J. Cho: Investigation, Data curation. Risako Kato-Miyabe: Methodology, Investigation, Data curation. Olivia G. Mallari: Investigation, Data curation. Olivia A. Moody: Methodology, Investigation, Data curation. David P. Obert: Data curation, Formal analysis, Investigation, Writing – review & editing. Gwi H. Park: Data curation, Investigation, Methodology. Ken Solt: Writing – review & editing, Writing – original draft, Supervision, Resources, Project administration, Methodology, Investigation, Funding acquisition, Conceptualization.
Appendix A. Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.brs.2024.05.012.
References
- [1].Brown EN, Lydic R, Schiff ND. General anesthesia, sleep, and coma. N Engl J Med 2010;363(27):2638–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Weiser TG, et al. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet 2008;372(9633):139–44. [DOI] [PubMed] [Google Scholar]
- [3].Cascella M, Bimonte S, Di Napoli R. Delayed emergence from anesthesia: what we know and how we act. Local Reg Anesth 2020;13:195–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Forman SA, Chin VA. General anesthetics and molecular mechanisms of unconsciousness. Int Anesthesiol Clin 2008;46(3):43–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Wang J, et al. Dopaminergic System in promoting recovery from general anesthesia. Brain Sci 2023;13(4). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Heshmati M, Bruchas MR. Historical and modern evidence for the role of reward circuitry in emergence. Anesthesiology 2022;136(6):997–1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Moody OA, et al. The neural circuits underlying general anesthesia and sleep. Anesth Analg 2021;132(5):1254–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Vincent KF, Solt K. Modulating anesthetic emergence with pathway-selective dopamine signaling. Curr Opin Anaesthesiol 2023;36(5):468–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Solt K, et al. Electrical stimulation of the ventral tegmental area induces reanimation from general anesthesia. Anesthesiology 2014;121(2):311–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Bao WW, et al. Nucleus accumbens neurons expressing dopamine D1 receptors modulate states of consciousness in sevoflurane anesthesia. Curr Biol 2021;31(9): 1893–1902 e5. [DOI] [PubMed] [Google Scholar]
- [11].Gui H, et al. Dopaminergic projections from the ventral tegmental area to the nucleus accumbens modulate sevoflurane anesthesia in mice. Front Cell Neurosci 2021;15:671473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Yang B, et al. Activation of dopamine signals in the olfactory tubercle facilitates emergence from isoflurane anesthesia in mice. Neurochem Res 2021;46(6): 1487–501. [DOI] [PubMed] [Google Scholar]
- [13].Zhang Y, et al. Dopamine D1 receptor in the nucleus accumbens modulates the emergence from propofol anesthesia in rat. Neurochem Res 2021;46(6):1435–46. [DOI] [PubMed] [Google Scholar]
- [14].Song Y, et al. Dopaminergic neurons in the ventral tegmental-prelimbic pathway promote the emergence of rats from sevoflurane anesthesia. Neurosci Bull 2022;38 (4):417–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Kenny JD, et al. Physostigmine and methylphenidate induce distinct arousal states during isoflurane general anesthesia in rats. Anesth Analg 2016;123(5):1210–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Taylor NE, et al. Optogenetic activation of dopamine neurons in the ventral tegmental area induces reanimation from general anesthesia. Proc Natl Acad Sci U S A 2016;113(45):12826–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Zhang Y, et al. Dopamine D1 receptor in the NAc shell is involved in delayed emergence from isoflurane anesthesia in aged mice. Brain Behav 2021;11(1): e01913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Wang H, et al. Restoring VTA DA neurons excitability accelerates emergence from sevoflurane general anesthesia of anxiety state. Biochem Biophys Res Commun 2021;565:21–8. [DOI] [PubMed] [Google Scholar]
- [19].Kenny JD, et al. Dextroamphetamine (but not atomoxetine) induces reanimation from general anesthesia: implications for the roles of dopamine and norepinephrine in active emergence. PLoS One 2015;10(7):e0131914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Guo J, et al. Dopamine transporter in the ventral tegmental area modulates recovery from propofol anesthesia in rats. J Chem Neuroanat 2022;121:102083. [DOI] [PubMed] [Google Scholar]
- [21].Kato R, et al. D-amphetamine rapidly reverses dexmedetomidine-induced unconsciousness in rats. Front Pharmacol 2021;12:668285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Kilkenny C, et al. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. Vet Clin Pathol 2012;41(1):27–31. [DOI] [PubMed] [Google Scholar]
- [23].Guidera JA, et al. Sevoflurane induces coherent slow-delta oscillations in rats. Front Neural Circ 2017;11:36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Solt K, et al. Methylphenidate actively induces emergence from general anesthesia. Anesthesiology 2011;115(4):791–803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Moody OA, et al. D-amphetamine accelerates recovery of consciousness and respiratory drive after high-dose fentanyl in rats. Front Pharmacol 2020;11: 585356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Therneau TM. Mixed effects cox models. 2022-10-03 18:38:46 UTC; 2.2–18.1, https://cran.r-project.org/web/packages/coxme/coxme.pdf; 2022.
- [27].Bates D, et al. Fitting linear mixed-effects models using {lme4}. J Stat Software 2015;67(1):1–48. [Google Scholar]
- [28].Scholz J, Tonner PH. Alpha2-adrenoceptor agonists in anaesthesia: a new paradigm. Curr Opin Anaesthesiol 2000;13(4):437–42. [DOI] [PubMed] [Google Scholar]
- [29].Chiu TH, et al. Action of dexmedetomidine on rat locus coeruleus neurones: intracellular recording in vitro. Eur J Pharmacol 1995;285(3):261–8. [DOI] [PubMed] [Google Scholar]
- [30].Beckstead RM, Domesick VB, Nauta WJ. Efferent connections of the substantia nigra and ventral tegmental area in the rat. Brain Res 1979;175(2):191–217. [DOI] [PubMed] [Google Scholar]
- [31].Simon H, et al. Anatomical relationships between the ventral mesencephalic tegmentum–a 10 region and the locus coeruleus as demonstrated by anterograde and retrograde tracing techniques. J Neural Transm 1979;44(1–2):77–86. [DOI] [PubMed] [Google Scholar]
- [32].Swanson LW. The projections of the ventral tegmental area and adjacent regions: a combined fluorescent retrograde tracer and immunofluorescence study in the rat. Brain Res Bull 1982;9(1–6):321–53. [DOI] [PubMed] [Google Scholar]
- [33].Fois GR, et al. Locus coeruleus activation during environmental novelty gates cocaine-induced long-term hyperactivity of dopamine neurons. iScience 2022;25 (4):104154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Lategan AJ, Marien MR, Colpaert FC. Effects of locus coeruleus lesions on the release of endogenous dopamine in the rat nucleus accumbens and caudate nucleus as determined by intracerebral microdialysis. Brain Res 1990;523(1):134–8. [DOI] [PubMed] [Google Scholar]
- [35].Whittington RA, Virag L. Dexmedetomidine-induced decreases in accumbal dopamine in the rat are partly mediated via the locus coeruleus. Anesth Analg 2006;102(2):448–55. [DOI] [PubMed] [Google Scholar]
- [36].Whittington RA, et al. Dexmedetomidine decreases extracellular dopamine concentrations in the rat nucleus accumbens. Brain Res 2001;919(1):132–8. [DOI] [PubMed] [Google Scholar]
- [37].Breton JM, et al. Relative contributions and mapping of ventral tegmental area dopamine and GABA neurons by projection target in the rat. J Comp Neurol 2019; 527(5):916–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Yamaguchi T, et al. Mesocorticolimbic glutamatergic pathway. J Neurosci 2011;31 (23):8476–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Zhang S, et al. Ventral tegmental area glutamatergic neurons facilitated emergence from isoflurane anesthesia involves excitation of lateral septum gamma-aminobutyric acid-ergic neurons in mice. Anesth Analg 2023. [DOI] [PubMed] [Google Scholar]
- [40].Torralva R, et al. Fentanyl but not morphine interacts with nonopioid recombinant human neurotransmitter receptors and transporters. J Pharmacol Exp Therapeut 2020;374(3):376–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Lesscher HM, et al. ERK1/2 activation in rat ventral tegmental area by the mu-opioid agonist fentanyl: an in vitro study. Neuroscience 2003;116(1):139–44. [DOI] [PubMed] [Google Scholar]
- [42].Matsui A, et al. Separate GABA afferents to dopamine neurons mediate acute action of opioids, development of tolerance, and expression of withdrawal. Neuron 2014; 82(6):1346–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Galaj E, et al. Dissecting the role of GABA neurons in the VTA versus SNr in opioid reward. J Neurosci 2020;40(46):8853–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Yoshida Y, et al. Fentanyl increases dopamine release in rat nucleus accumbens: involvement of mesolimbic mu- and delta-2-opioid receptors. Neuroscience 1999; 92(4):1357–65. [DOI] [PubMed] [Google Scholar]
- [45].Radford CKD, et al. Effects of subanesthetic intravenous ketamine infusion on corticosterone and brain-derived neurotrophic factor in the plasma of male sprague-dawley rats. AANA J (Am Assoc Nurse Anesth) 2018;86(5):393–400. [PubMed] [Google Scholar]
- [46].Lu J, et al. Role of endogenous sleep-wake and analgesic systems in anesthesia. J Comp Neurol 2008;508(4):648–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Zhang Y, et al. Estrogen receptor-A in medial preoptic area contributes to sex difference of mice in response to sevoflurane anesthesia. Neurosci Bull 2022;38(7): 703–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Wasilczuk AZ, et al. Hormonal basis of sex differences in anesthetic sensitivity. Proc Natl Acad Sci U S A 2024;121(3):e2312913120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Vincent KF, et al. Oestrous cycle affects emergence from anaesthesia with dexmedetomidine, but not propofol, isoflurane, or sevoflurane, in female rats. Br J Anaesth 2023;131(1):67–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Moody OA, Vincent KF, Solt K. Sex, drugs, and anaesthesia research. Br J Anaesth 2021;127(3):340–3. [DOI] [PubMed] [Google Scholar]
- [51].Zhou X, Wang T, Huang S. Effects of the menstrual cycle on bispectral index and anesthetic requirement in patients with preoperative intravenous dexmedetomidine following propofol induction. Int J Clin Exp Med 2014;7(12): 5663–8. [PMC free article] [PubMed] [Google Scholar]
- [52].Han C, et al. Influence of different menstrual phases on the Bispectral Index during dexmedetomidine sedation. Kaohsiung J Med Sci 2016;32(11):586–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Mashour GA. Consciousness. Anesth Analg 2022;134(6):1118–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Bachmann T, Hudetz AG. It is time to combine the two main traditions in the research on the neural correlates of consciousness: C = L × D. Front Psychol 2014;5: 940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Laureys S The neural correlate of (un)awareness: lessons from the vegetative state. Trends Cognit Sci 2005;9(12):556–9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


