Abstract
Bleeding is a well-recognized side effect of anticoagulant therapy, which is used to treat venous thromboembolism (VTE) in individuals of all ages, including those of female sex, who commonly experience VTE as a complication of hormonal therapies and/or pregnancy. Heavy menstrual bleeding (HMB) is also extremely common in reproductive aged individuals of female sex. Despite these overlapping situations, relatively little attention has been paid to the impact of anticoagulant-associated HMB on treatment strategies and the patient experience. In this review, we summarize incidence and complications of HMB in anticoagulated individuals as well as management strategies for HMB in this population. We also address the patient experience, including the impact of HMB on quality of life and the impact of discontinuing hormonal therapies at the time of VTE diagnosis and anticoagulant initiation. We conclude by highlighting specific gaps related to the patient experience of anticoagulant-associated HMB in both the research and clinical settings.
Keywords: Menstruation, Menorrhagia, Venous thromboembolism, Anticoagulants, Abnormal uterine bleeding
Introduction
Venous thromboembolism (VTE) affects up to 900,000 individuals in the United States annually.[1] An estimated 46% of these individuals are female.[2] While risk of VTE increases with age, female individuals experience unique risks during their reproductive years, including use of hormonal therapies such as combined hormonal contraceptives (CHCs) and pregnancy. As a result, a disproportionate number of VTE events effecting reproductive-aged (15–45 years) individuals occur in females.[3]
In addition to experiencing increased risk of VTE compared to male counterparts, reproductive-age females experience unique risks with regard to bleeding complications, specifically menstruation and pregnancy. Menstruation is expected to occur roughly every 28 days in virtually all individuals of female sex for an average of 40 years. This equates to up to 500 individual cycles over a lifetime. Twenty to thirty percent of the general population of menstruating individuals will experience heavy menstrual bleeding (HMB) during this time period, the current definition of which is “excessive menstrual losses which interferes with the [individual’s] physical, emotional, social and material quality of life.”[4]
The combination of increased risk of VTE and the recurrent, cyclic bleeding event known as menstruation require additional consideration and attention when balancing risks and benefits of therapies for both VTE and HMB. In this review we discuss the incidence and impact of HMB in anticoagulated individuals as well as management strategies, from both the clinician perspective and the patient perspective. Accordingly, discussion will be centered around a case, highlighting patient experience in addition to data supporting diagnostic and management strategies.
Case
A 27-year-old woman reports to the emergency department with a report of bilateral, pleuritic chest pain for several hours. She also notes a two-week history of ongoing left-sided leg pain. Her past medical history is notable for mild asthma and iron deficiency. Her only current medication is Ethinyl estradiol 0.03mg/drospirenone 3mg which she takes continuously. This was initiated approximately 3 months ago for treatment of heavy menstrual bleeding resulting in iron deficiency anemia. She is mildly tachycardic with shallow respirations. Current hemoglobin is 12.1, MCV is 80fL, plasma d-dimer is >3,000ng/mL and imaging is significant for bilateral segmental and subsegmental pulmonary emboli. Her CHCs are discontinued, she is prescribed rivaroxaban for a planned 3-month course and discharged home.
Anticoagulation and HMB
As discussed above, HMB effects up to a third of the general menstruating population.[5] This clearly increases in anticoagulated individuals with an estimated prevalence of 60–70%.[6] However, estimates remain rough and highly variable due to a number of limitations in how HMB is addressed, or in many cases not addressed, in clinical studies. Virtually all registry trials of anticoagulants, including direct oral anticoagulants (DOACs) such as rivaroxaban, rely on International Society of Thrombosis and Haemostasis definitions of major bleeding (MB) and clinically relevant nonmajor bleeding (CRNMB).[7, 8] These measures are inadequate to detect HMB for two primary reasons.
The first reason MB and CRNMB criteria fail to detect HMB is that they are designed for discrete bleeding events, not recurrent ones. Many patients with HMB do develop anemia and the majority have iron deficiency with or without anemia,[9] but due to the fact that blood loss is slower, occurring over days each month, criteria such as a 2g/dL drop in hemoglobin and/or need for blood transfusion are rarely met, even though annual blood loss may very well exceed that seen in a single major bleeding event.[9]
The second limitation of MB and CRNMB criteria for detecting HMB is the fact that 1) researchers rarely ask about it specifically in study questionnaires and 2) patients/research subjects are often reticent to volunteer this information. A survey of 1000 women in the UK not on hormonal birth control or hormone replacement therapy reported that 55% had had to take time off work for menstrual symptoms, 60% of whom had a disciplinary hearing for amount of time off work and 51% of whom subsequently lost their jobs. Despite this, less than 50%, and as few as 23% women in some regions of England had talked to or were willing to talk to their general practitioner about it.[10] While more research is needed to fully understand the reasons for this, there can be little doubt that the stigma associated with menstruation, treating it as something “dirty” or shameful instead of a totally normal bodily process, contributes to this hesitancy.
Studies which do address HMB are most often observational, without the benefit of randomization or, in many cases, prospective data collection. Post-hoc analyses of registry trials, while offering improved oversight and comparators, are limited by criteria which grossly underestimate occurrence of HMB. Despite this recognized limitation, there is no true “gold standard” measure of HMB for studies that do address it. The most objective approach, the alkaline hematin method, can accurately quantify menstrual blood loss (MBL) but is prohibitively demanding and costly as it requires collection and processing of all menstrual hygiene products for an entire cycle.[11] Other methods, such as the Pictorial Blood Loss Assessment Chart (PBAC) are easier to administer but not universally used or adaptable to methods other than standard, disposable pads and tampons.[12] Ideally, pads and tampons should also be consistent (requiring the study to provide them) for a fully accurate comparison. For this reason, a variety of other subjective methods are often used, with inconsistent findings. While the clinical definition of HMB, based upon the perspective of the patient, has come a long way, research definitions lag behind.
A number of available reviews have summarized and attempted to combine the available data on rates of HMB with various anticoagulants. Prevalence estimates are highly variable but most available data strongly suggest an increased rate of HMB with rivaroxaban (a near two-fold increase compared to low molecular weight heparin (LMWH)/warfarin), and perhaps a decreased rate with dabigatran (slightly more than half the rate compared to LMWH/warfarin) with apixaban appearing roughly equivalent to warfarin for this outcome.[6, 13–16] The increased rate with rivaroxaban is presumed to be due to higher circulating doses of anticoagulant in order to facilitate daily vs. twice daily dosing. Based on previous estimates with warfarin it seems very likely that more than three-quarters of menstruating individuals treated with rivaroxaban, such as our case study, are likely to develop HMB while on anticoagulation.[6] This risk is higher if the patient has a history of HMB,[15] as our patient does, and must be balanced against the convenience of daily vs. twice daily dosing of anticoagulation, which can have a meaningful impact on adherence.[17]
Impact of HMB
As previously mentioned, a definition of HMB based solely on quality of life, which conveniently bypasses the need for painstaking counting and measuring of menstrual products, has come into favor in clinical practice. Most importantly, this addresses the fact that HMB can severely, negatively affect quality of life. Patients with HMB and related health complications including iron deficiency experience fatigue, missed school/work, isolation and generally negatively impacted health-related quality of life.[18] A survey study of 2000 women in the United Kingdom revealed that 69% of individuals with symptoms from menstruation reported it had a negative impact at work. Fifty-three percent reported they were unable to go to work at some point and 4% missed work on a monthly basis. Six percent report that their menstrual symptoms had an impact leading to formal action and 12% reported menstrual symptoms had a negative impact on career progression.[19] Additionally, menstrual hygiene products are expensive and period poverty, defined as lack of access to adequate menstrual hygiene products, education, and sanitation facilities, is a common problem internationally and is only worsened by HMB.[20, 21] Other aspects of quality of life which may be adversely impacted by HMB are listed in Figure 1.
Figure 1:

impacts of heavy menstrual bleeding (HMB) on physical, social, emotional and material domains of quality of life.
Individuals with HMB frequently find themselves having to change a menstrual hygiene product more often than every 2 hours,[9] which can be incredibly disruptive to school, work and social activities. Individuals in professions which do not allow frequent bathroom breaks, such as teachers and nurses, may struggle to attend work at all during their periods. “Normal” menses should last approximately 2–7 days but individuals with HMB frequently have periods lasting 7–10 days or even longer, equating to ≥25% of days each cycle. As referenced above, a survey study of menstruating individuals found that, despite some reporting even losing their jobs due to missing work for menstruation related symptoms, fewer than half of women felt comfortable talking to their doctor about their periods.[10] This serves to emphasize the heavy impact of shame and stigma on individuals suffering from HMB and other menstruation related symptoms. In addition to frequent changes of protection and prolonged days of bleeding, a defining feature of HMB is iron deficiency,[9] which can be associated with a wide variety of symptoms, including fatigue, brain fog, restless legs, dizziness, hair loss, muscle weakness/soreness and pica, all of which can be profoundly disturbing to quality of life.[22–24]
Anecdotally, in this author’s clinical practice and experience working with patients and advocates, many menstruating individuals feel unprepared for the increase in menstrual bleeding they experience upon initiating an anticoagulant. They report that their healthcare providers rarely, if ever, discuss the potential impact of an anticoagulant on their periods and many find the increase in bleeding with their first period to be frightening, adding, in some cases, to the already difficult experience of a VTE diagnosis.[25]
In addition to this dramatic impact on quality of life, available data demonstrate an increased risk, as high as five-fold, of recurrent VTE in individuals with vs. without HMB on rivaroxaban.[13] While the precise mechanism of this increase remains unproven, decreased adherence to anticoagulation must be considered as a possible cause, particularly given the combination of stigma around and potentially devastating impact of HMB for patients who may not feel they have other good options.
Case continued
As could be anticipated, our patient experienced withdrawal bleeding shortly after discontinuing CHCs, this was compounded by her rivaroxaban use and, despite using maximally absorptive products, she found herself having to change protection every hour for the first two days. Even doubling up on protection (wearing a pad and tampon together) only allowed her to go 3–4 hours between changes. As a result, and due to the fact that her work as a teacher offered infrequent bathroom breaks, she found herself having to miss multiple days of work.
At her three-month follow-up to determine final duration of anticoagulation she was found to have iron deficiency anemia, with a hemoglobin of 9.8g/dL, an MCV of 73.2fL and a ferritin of 7ng/mL. She reported fatigue, decreased exercise tolerance, hair loss and symptoms of depression.
Impact of discontinuing hormonal therapy
As our case demonstrates, due to the fact that estrogen-containing therapies such as CHCs are a common provoking factor for VTE in reproductive age females, such therapies are often discontinued upon diagnosis. The impact of such a decision, while rarely considered by providers, can be enormous for the patient.
Discontinuation of CHCs almost universally results in withdrawal bleeding, something which individuals who use such therapies continuously or regularly and are subsequently accustomed to regular or no menstrual bleeding, are rarely counseled to expect. As discussed above, this can have a dramatic, negative impact on quality of life. As CHCs are a highly effective treatment for HMB, discontinuation can be expected to result in increased menstrual blood loss in many, something which may be compounded by initiation of anticoagulation therapy.
Additionally, CHCs, while associated primarily with pregnancy prevention in most providers’ minds, are effective therapies for a multitude of symptoms and conditions, including but not limited to dysmenorrhea, endometriosis, premenstrual dysphoric disorder, acne and hirsutism (Figure 2). Use of CHCs can also reduce future incidence of ectopic pregnancy, benign breast disease, ovarian cysts, ovarian cancer, endometrial cancer and colorectal cancer.
Figure 2:
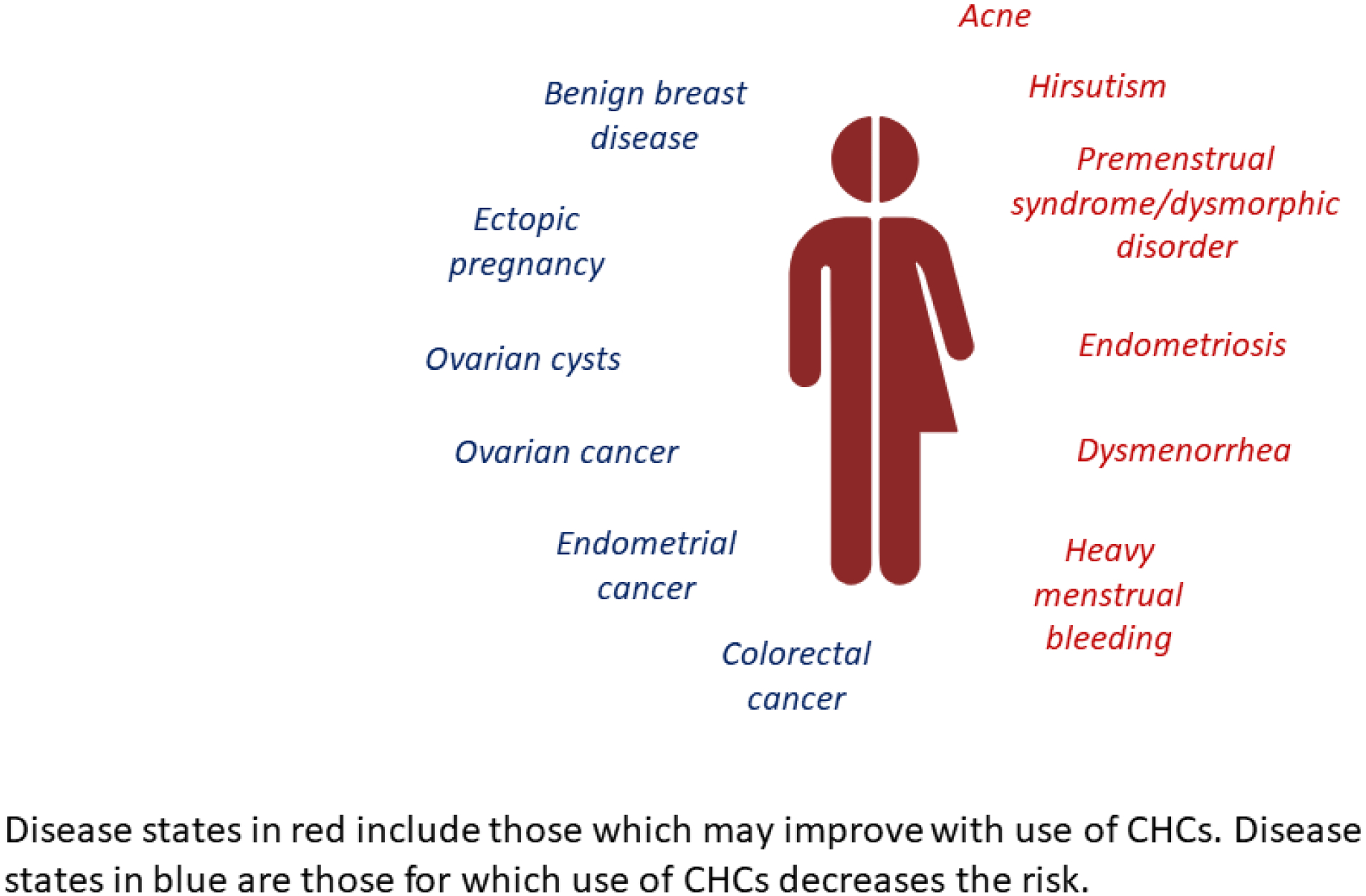
non-contraceptive benefits of combined hormonal contraceptives (CHCs).
The importance of effective contraception is paramount, particularly in patients experiencing or at risk for VTE. Experts commonly recommend that individuals wait until at least 3 months after a VTE event to become pregnant, similar to recommendations around delaying elective surgeries. Furthermore, many anticoagulants can lead to adverse pregnancy outcomes. Warfarin has known teratogenic effects and while data on risk of direct oral anticoagulants in pregnancy are not available, use is not recommended in pregnancy.[26] Patients who subsequently suffer from pregnancy complications such as spontaneous abortion and/or ectopic pregnancy may suffer from increased risk of bleeding complications due to anticoagulant use.
Management of HMB
Strategies of HMB management are largely the same, regardless of whether or not the HMB is associated with anticoagulation. CHCs, along with tranexamic acid, are first-line therapies for HMB in the general population.[27] CHCs can be used to regulate, increase or decrease frequency of menstrual bleeding, reduce menstrual blood loss or even induce amenorrhea. Additionally, and as outlined in Figure 2, they can be used to reduce and/or ameliorate other symptoms associated with menstruation. While the increased risk of VTE with CHCs is generally considered to outweigh the benefits in individuals with a personal history of VTE, not on anticoagulation, post-hoc data of individuals involved in registry trials of apixaban and rivaroxaban suggest that hormonal therapies, including CHCs, can be safely used in individuals on therapeutic anticoagulation.[14] This is of particular importance in disease states such as endometriosis, which may only be responsive to estrogen-based therapies, and the risk and impact of anticoagulation on quality of life may be outweighed by benefits of disease management. However, it may be relevant even in individuals who require long term anticoagulation and desire one or more of the benefits of CHCs outlined in Figure 2. Individualized and informed risk/benefit analysis should be offered to all patients.
Given the inevitability of withdrawal bleeding and high likelihood of return of or development of new HMB, many patients may prefer to continue on CHCs while anticoagulated, if given the option. Occasionally, patients on long term anticoagulation may even need to start a new therapy with CHCs, particularly in circumstances such as the development of endometriosis, for which CHCs are a major component of therapy. It is, however, important to note that the increased risk of VTE continues for 2 to 4 weeks past discontinuation of estrogen containing therapies,[28] therefore transition to an alternative method of menstrual management should be initiated a least a month before discontinuation of anticoagulation.
A preferred alternative for patients with history of VTE planning to discontinue anticoagulation is the levonorgestrel intrauterine device (LNG-IUD), which has dual benefit for HMB management and highly effective contraception. In addition to reducing overall menstrual blood loss, the LNG-IUD is associated with an amenorrhea rate of 20% within the first year, increasing to ~40% between years 3–8, and a <1% contraceptive failure rate.[29, 30] Establishing with a practitioner who is able to insert IUDs may take some time, however, and continuing CHCs (so long as anticoagulation is also continued) until placement is a very reasonable approach. The etonogestrel implant is another long acting, reversible contraceptive (LARC) method which can reduce overall menstrual blood loss, but can commonly be associated with irregular and intermenstrual bleeding which some patients find troublesome.[31]
Depomedroxyprogesterone acetate (DMPA) injections boast the highest rate of amenorrhea (>55%) but have a number of side effects, including being the only progesterone-only option which is associated with increased risk of VTE and therefore is not recommended in this population outside of ongoing therapeutic anticoagulation.[32] Progesterone only pills can also be used but may also result in irregular bleeding and are less effective as contraception.
Antifibrinolytics, such as tranexamic acid, are considered first line therapy for HMB outside the setting of acute thrombosis.[27] While many express theoretical concerns about the risk of new thrombosis, this theory has been disproven by large volumes of data from a variety of randomized controlled trials, including in high risk populations such as trauma victims and postpartum patients, demonstrating no increased risk of VTE.[33] It is important to note, however, that fibrinolysis is a key process to VTE resolution and therefore initiation of antifibrinolytics is best delayed at least until initial symptoms improve and likely for as long as one to three months. Data on the use of antifibrinolytics in anticoagulated patients are lacking, however tranexamic acid was one of the treatment strategies employed for HMB associated with anti-Xa use in the MEDEA study, data from which are not yet available at the time of this review.[34]
In addition to the above-mentioned methods, which may be useful in patients with HMB due to any cause, in individuals on anticoagulation, consideration may be given to switching from an anticoagulant with a higher rate of HMB, such as rivaroxaban, to one with a slightly lower rate of HMB, such as apixaban. However, this approach has not been studied and in patients such as the one described here, who already suffered from HMB prior to anticoagulant initiation, this is unlikely to be adequate as a sole approach. Alternative modifications, such as interrupting or reducing the dose of anticoagulant on days of bleeding, have not been studied for safety and, given the aforementioned data regarding increased rates of VTE in individuals with HMB while on anticoagulation, may pose a serious risk.[35]
Importantly, individuals who develop new HMB outside the setting of a recent initiation of anticoagulation should be evaluated by a gynecologist for potential structural causes. Menstruating individuals with heavy bleeding, and even those without, would ideally be screened for iron deficiency at the time of anticoagulation initiation. Iron supplementation can be a key component of the management of HMB and may, theoretically, even impact heaviness of bleeding itself due to impact on platelet function.[36]
Case concluded
Our patient’s HMB is initially treated with oral progesterone, until she is able to establish with a gynecologist who evaluates her for potential structural causes of HMB and places a levonorgestrel IUD for long-term management. She also receives iron supplementation in the form of intravenous iron dextran. Three months later she is evaluated by her primary care physician, as she is no longer on anticoagulation, and reports much decreased menstrual bleeding as well as decreased fatigue and increased exercise tolerance.
Conclusion
While the initial focus of VTE therapy is, appropriately, prevention of progression and recurrence, it is essential also to consider the impact of treatment on uterine bleeding, particularly in menstruating individuals. This impact has been grossly underestimated thus far largely due to inadequate and inconsistent use of appropriate outcome measures in prospective studies. However, it is clear that HMB is very common among menstruating individuals on anticoagulation, that rates are increased with some anticoagulants, such as rivaroxaban, compared to others, and that HMB can have an enormous impact on patient quality of life.
Opportunities for improvement in the care of these individuals abound in both clinical and research settings. In the research setting, standardization and consistent use of clinically meaningful measures of HMB and other forms of abnormal bleeding must be routinely used in studies of medications which may increase HMB. In the clinical setting there are many points of contact at which to improve care for these patients. Firstly, an up-front discussion about the increased risk of HMB on anticoagulation, taking into consideration the individual’s own menstrual history, the risks associated with the anticoagulant of choice and potential discontinuation of any therapies which may have impacted menstrual bleeding, either as a primary goal or side effect is essential. Individuals who do experience VTE in the setting of exogenous estrogen, such as CHCs, must be counseled on the risks and benefits of continuing or discontinuing these therapies and should be included in shared decision making around how to manage these important therapies. Finally, at each point of follow-up, both volume and impact of menstrual losses must be considered, extending beyond evaluation for anemia and iron deficiency to include impact on quality of life and daily functioning.
In conclusion, HMB and other forms of abnormal bleeding are far more common and impactful in patients on anticoagulant therapy than previously recognized. Stigma can be a major barrier to full and open discussion about risks and concerns for both patient and provider. Risks associated with missing and failing to manage this treatment complication range from inconvenience to missed work or school, lost jobs and even increased rates of recurrent VTE. Clinicians and patients will both benefit from an increased, open, nonjudgmental communication about risks associated with anticoagulants and treatment strategies for HMB. Questions about uterine bleeding must be included in follow-up assessments for all patients at risk, particularly as patients may be less likely to report these symptoms than other, less stigmatized ones such as epistaxis or bruising. A variety of management strategies are available and appropriate management can provide great satisfaction to both patient and provider and must be an essential component of the care of anticoagulated patients.
Table 1:
Opportunities for Intervention to Reduce Impact of AUB in Anticoagulated Individuals
| At VTE Diagnosis |
| Assess for history of HMB |
| Assess for and treat iron deficiency |
| Defer discontinuation of CHCs until an alternative strategy for HMB management and/or contraception can be implemented (so long as patient is on anticoagulation) |
Discuss anticipated impact of anticoagulation on uterine bleeding
|
| On Follow-up |
| Assess for and treat HMB, if indicated, at every visit |
| Assess for iron deficiency, treat and reassess regularly if identified |
| Discuss importance of and options for safe and effective contraception to prevent undesired pregnancy, with potential dual benefit of menstrual management |
Conflicts of Interest
BSB receives research funding from the Office of Research on Women’s Health of the National Institutes for Health and the Foundation for Women and Girls with Blood Disorders.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Prevention CfDC. Data and Statistics on Venous Thromboembolism.
- 2.Beckman MG, Hooper WC, Critchley SE, Ortel TL. Venous Thromboembolism: A Public Health Concern. American Journal of Preventive Medicine. 2010; 38: S495–S501. 10.1016/j.amepre.2009.12.017. [DOI] [PubMed] [Google Scholar]
- 3.Arnesen CAL, Veres K, Horváth-Puhó E, Hansen JB, Sørensen HT, Brækkan SK. Estimated lifetime risk of venous thromboembolism in men and women in a Danish nationwide cohort: impact of competing risk of death. Eur J Epidemiol. 2022; 37: 195–203. 10.1007/s10654-021-00813-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Excellence NIfHaC. Heavy menstrual bleeding: assessment and management. 2021. [PubMed]
- 5.Karlsson TS, Marions LB, Edlund MG. Heavy menstrual bleeding significantly affects quality of life. Acta obstetricia et gynecologica Scandinavica. 2014; 93: 52–7. 10.1111/aogs.12292. [DOI] [PubMed] [Google Scholar]
- 6.De Crem N, Peerlinck K, Vanassche T, Vanheule K, Debaveye B, Middeldorp S, Verhamme P, Peetermans M. Abnormal uterine bleeding in VTE patients treated with rivaroxaban compared to vitamin K antagonists. Thrombosis Research. 2015; 136: 749–53. 10.1016/j.thromres.2015.07.030. [DOI] [PubMed] [Google Scholar]
- 7.Kaatz S, Ahmad D, Spyropoulos AC, Schulman S, Subcommittee on Control of A. Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. Journal of thrombosis and haemostasis : JTH. 2015; 13: 2119–26. [DOI] [PubMed] [Google Scholar]
- 8.Schulman S, Kearon C, Subcommittee on Control of Anticoagulation of the S, Standardization Committee of the International Society on T, Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. Journal of thrombosis and haemostasis : JTH. 2005; 3: 692–4. 10.1111/j.1538-7836.2005.01204.x. [DOI] [PubMed] [Google Scholar]
- 9.Warner PE, Critchley HO, Lumsden MA, Campbell-Brown M, Douglas A, Murray GD. Menorrhagia I: measured blood loss, clinical features, and outcome in women with heavy periods: a survey with follow-up data. American journal of obstetrics and gynecology. 2004; 190: 1216–23. 10.1016/j.ajog.2003.11.015. [DOI] [PubMed] [Google Scholar]
- 10.Braithwaite J Period Survey Results: Workplace Stigma and Most Common Symptoms. ForthWithLife, 2022. [Google Scholar]
- 11.Magnay JL, O’Brien S, Gerlinger C, Seitz C. A systematic review of methods to measure menstrual blood loss. BMC Womens Health. 2018; 18: 142. 10.1186/s12905-018-0627-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zakherah MS, Sayed GH, El-Nashar SA, Shaaban MM. Pictorial blood loss assessment chart in the evaluation of heavy menstrual bleeding: diagnostic accuracy compared to alkaline hematin. Gynecol Obstet Invest. 2011; 71: 281–4. 10.1159/000320336. [DOI] [PubMed] [Google Scholar]
- 13.Bryk AH, Piróg M, Plens K, Undas A. Heavy menstrual bleeding in women treated with rivaroxaban and vitamin K antagonists and the risk of recurrent venous thromboembolism. Vascular Pharmacology. 2016; 87: 242–7. 10.1016/j.vph.2016.11.003. [DOI] [PubMed] [Google Scholar]
- 14.Martinelli I, Lensing AWA, Middeldorp S, Levi M, Beyer-Westendorf J, van Bellen B, Bounameaux H, Brighton TA, Cohen AT, Trajanovic M, Gebel M, Lam P, Wells PS, Prins MH. Recurrent venous thromboembolism and abnormal uterine bleeding with anticoagulant and hormone therapy use. Blood. 2016; 127: 1417–25. 10.1182/blood-2015-08-665927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Samuelson Bannow BT, Chi V, Sochacki P, McCarty OJT, Baldwin MK, Edelman AB. Heavy menstrual bleeding in women on oral anticoagulants. Thrombosis Research. 2021; 197: 114–9. 10.1016/j.thromres.2020.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huisman MV, Ferreira M, Feuring M, Fraessdorf M, Klok FA. Less abnormal uterine bleeding with dabigatran than warfarin in women treated for acute venous thromboembolism. Journal of thrombosis and haemostasis : JTH. 2018; 16: 1775–8. 10.1111/jth.14226. [DOI] [PubMed] [Google Scholar]
- 17.Iskedjian M, Einarson TR, MacKeigan LD, Shear N, Addis A, Mittmann N, Ilersich AL. Relationship between daily dose frequency and adherence to antihypertensive pharmacotherapy: evidence from a meta-analysis. Clin Ther. 2002; 24: 302–16. 10.1016/s0149-2918(02)85026-3. [DOI] [PubMed] [Google Scholar]
- 18.Fernandez-Jimenez MC, Moreno G, Wright I, Shih PC, Vaquero MP, Remacha AF. Iron Deficiency in Menstruating Adult Women: Much More than Anemia. Womens Health Rep (New Rochelle). 2020; 1: 26–35. 10.1089/whr.2019.0011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Development CIoPa. Menstruation and Support at Work. 2023. [Google Scholar]
- 20.Jaafar H, Ismail SY, Azzeri A. Period Poverty: A Neglected Public Health Issue. Korean J Fam Med. 2023; 44: 183–8. 10.4082/kjfm.22.0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dutton B, Kai J. Women’s experiences of heavy menstrual bleeding and medical treatment: a qualitative study in primary care. Br J Gen Pract. 2023; 73: e294–e301. 10.3399/bjgp.2022.0460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Beatrix J, Piales C, Berland P, Marchiset E, Gerbaud L, Ruivard M. Non-anemic iron deficiency: correlations between symptoms and iron status parameters. Eur J Clin Nutr. 2022; 76: 835–40. 10.1038/s41430-021-01047-5. [DOI] [PubMed] [Google Scholar]
- 23.Pasricha SR, Tye-Din J, Muckenthaler MU, Swinkels DW. Iron deficiency. Lancet. 2021; 397: 233–48. 10.1016/S0140-6736(20)32594-0. [DOI] [PubMed] [Google Scholar]
- 24.Zia A, Stanek J, Christian-Rancy M, Savelli S, O’Brien SH. Iron deficiency and fatigue among adolescents with bleeding disorders. Am J Hematol. 2022; 97: 60–7. 10.1002/ajh.26389. [DOI] [PubMed] [Google Scholar]
- 25.Rosovsky RRW, Klok F. The CLUES Study: A Critical Look at Understanding the Emotional Suffering of Blood Clot Survivors. ISTH 2022 Congress. London, 2022. [Google Scholar]
- 26.Bates SM, Rajasekhar A, Middeldorp S, McLintock C, Rodger MA, James AH, Vazquez SR, Greer IA, Riva JJ, Bhatt M, Schwab N, Barrett D, LaHaye A, Rochwerg B. American Society of Hematology 2018 guidelines for management of venous thromboembolism: venous thromboembolism in the context of pregnancy. Blood Adv. 2018; 2: 3317–59. 10.1182/bloodadvances.2018024802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tsakiridis I, Giouleka S, Koutsouki G, Kostakis N, Kalogiannidis I, Kourtis A, Athanasiadis A, Goulis DG, Dagklis T. Investigation and management of abnormal uterine bleeding in reproductive-aged women: a descriptive review of national and international recommendations. The European Journal of Contraception & Reproductive Health Care. 2022; 27: 504–17. 10.1080/13625187.2022.2112169. [DOI] [PubMed] [Google Scholar]
- 28.Hugon-Rodin J, Fontana P, Poncet A, Streuli I, Casini A, Blondon M. Longitudinal profile of estrogen-related thrombotic biomarkers after cessation of combined hormonal contraceptives. Blood. 2024; 143: 70–8. 10.1182/blood.2023021717. [DOI] [PubMed] [Google Scholar]
- 29.Creinin MD, Schreiber CA, Turok DK, Cwiak C, Chen BA, Olariu AI. Levonorgestrel 52 mg intrauterine system efficacy and safety through 8 years of use. American journal of obstetrics and gynecology. 2022; 227: 871.e1–.e7. 10.1016/j.ajog.2022.05.022. [DOI] [PubMed] [Google Scholar]
- 30.Sergison JE, Maldonado LY, Gao X, Hubacher D. Levonorgestrel intrauterine system associated amenorrhea: a systematic review and metaanalysis. American journal of obstetrics and gynecology. 2019; 220: 440–8.e8. 10.1016/j.ajog.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Niu X, Luo Q, Wang C, Zhu L, Huang L. Effects of Etonogestrel implants on pelvic pain and menstrual flow in women suffering from adenomyosis or endometriosis: Results from a prospective, observational study. Medicine (Baltimore). 2021; 100: e24597. 10.1097/md.0000000000024597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tepper NK, Whiteman MK, Marchbanks PA, James AH, Curtis KM. Progestin-only contraception and thromboembolism: A systematic review. Contraception. 2016; 94: 678–700. 10.1016/j.contraception.2016.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Taeuber I, Weibel S, Herrmann E, Neef V, Schlesinger T, Kranke P, Messroghli L, Zacharowski K, Choorapoikayil S, Meybohm P. Association of Intravenous Tranexamic Acid With Thromboembolic Events and Mortality: A Systematic Review, Meta-analysis, and Meta-regression. JAMA Surgery. 2021; 156: e210884–e. 10.1001/jamasurg.2021.0884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hamulyák EN, Wiegers HMG, Scheres LJJ, Hutten BA, de Lange ME, Timmermans A, Westerweel PE, Nijziel MR, Kruip M, Ten Wolde M, Ypma PF, Klok FA, Nieuwenhuizen L, van Wissen S, Hovens MMC, Faber LM, Kamphuisen PW, Büller HR, Middeldorp S. Heavy menstrual bleeding on direct factor Xa inhibitors: Rationale and design of the MEDEA study. Res Pract Thromb Haemost. 2021; 5: 223–30. 10.1002/rth2.12471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bryk AH, Pirog M, Plens K, Undas A. Heavy menstrual bleeding in women treated with rivaroxaban and vitamin K antagonists and the risk of recurrent venous thromboembolism. Vascul Pharmacol. 2016; 87: 242–7. 10.1016/j.vph.2016.11.003. [DOI] [PubMed] [Google Scholar]
- 36.Elstrott BK, Lakshmanan HHS, Melrose AR, Jordan KR, Martens KL, Yang CJ, Peterson DF, McMurry HS, Lavasseur C, Lo JO, Olson SR, DeLoughery TG, Aslan JE, Shatzel JJ. Platelet reactivity and platelet count in women with iron deficiency treated with intravenous iron. Res Pract Thromb Haemost. 2022; 6: e12692. 10.1002/rth2.12692. [DOI] [PMC free article] [PubMed] [Google Scholar]


