Abstract
Purpose
This article reports detailed quality-of-life data including preferred and actual place of care from SCORAD, the only large prospective randomized trial in metastatic spinal cord compression (MSCC).
Methods
SCORAD compared 2 doses of radiotherapy in patients with MSCC: 8 Gy single fraction and 20 Gy in 5 fractions. In total, 686 patients were randomized, of whom 590 had Health-Related Quality of Life (HRQoL) data collected at baseline and at least 1 later time point. HRQoL was measured using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 supplemented with the QLU-C10D and data on place of care at weeks 1, 4, 8, and 12 postrandomization. Quality-of-Life Adjusted Survival was computed by multiplying Kaplan-Meier survival probabilities with the UK utility weights obtained from the QLU-C10D.
Results
Patients with a baseline physical functioning score of above 50 demonstrated a 28% reduction in the risk of death (hazard ratio [HR] = 0.72, 99% confidence interval [CI] = 0.54 to 0.95; P = .003). An increased risk of death was associated with fatigue (HR = 1.35, 99% CI = 1.03 to 1.76; P = .0040), dyspnea (HR = 1.61, 99% CI = 1.24 to 2.08; P < .001), and appetite loss (HR = 1.25, 99% CI = 0.99 to 1.59; P = .014). The preferred place of care for the majority was at home or with relatives (61%-74% across the 12 weeks) but achieved by only 53% at 8 weeks.
Conclusions
Prolonged survival in patients with MSCC was associated with better HRQoL. More than 60% of patients preferred to be cared for at home or with relatives, but only half were able to achieve this. There was no difference in HRQoL between the multifraction and single-fraction groups.
Trial registration
ISRCTN97555949 and ISRCTN97108008.
Metastatic spinal cord compression (MSCC) is a serious condition resulting from advanced cancer with poor survival (1), characterized by back pain and neurological changes, which can lead to substantial impairments in mobility and continence. The associated loss of independence with paraplegia and potential need for a catheter or loss of bowel control can be devastating, notwithstanding the need for urgent treatment and the short survival associated with this condition. Therefore, health-related quality of life (HRQoL) has great importance in this setting, but detailed quality-of-life parameters are rarely cited in published data. Neither the NICE guidelines (1) nor the Cancer Care Ontario guidelines (2) specifically recommend the use of quality-of-life assessments for evaluation or response.
HRQoL deterioration is often assumed to be associated with worse prognosis, but there is little published evidence to support this in MSCC. Previously published prognostic indices after surgery or radiotherapy for MSCC did not include any quality-of-life (3) parameters in their analysis; however, 1 retrospective study after surgery or radiotherapy for MSCC demonstrated a relationship between changes in HRQoL measured by EQ-5D and survival (4).
Disability associated with MSCC places an added burden on carers with both physical and psychological needs. Many studies (5-10) have shown that for patients with terminal cancer, their preferred place of death is at home, but for MSCC patients this may be more difficult to achieve without major input from domiciliary nursing and palliative care services.
The SCORAD trial is the largest randomized trial conducted in patients with MSCC (11). Although not reaching the noninferiority limit defined, SCORAD found similar ambulatory rates between single-fraction radiotherapy and multiple fractions delivered over 5 days. We hypothesized that receiving single-fraction radiotherapy will have no effect on HRQoL impairment at 8 weeks and over time in SCC when compared with multiple fractions. The primary research objectives of this study are to investigate the association between baseline HRQoL scores and overall survival in the SCORAD trial, analyze treatment differences in HRQoL scores at 8 weeks and over time, compare Quality-of-Life Adjusted Survival (QAS) between treatment groups, and assess potential treatment differences in the actual place of care over time.
Patients and methods
Study design and participants
SCORAD was a prospective noninferiority randomized trial comparing 2 radiotherapy fractionation schedules, a single dose of 8 Gy and 20 Gy in 5 fractions in MSCC. A total of 686 patients were randomized, comprising 341 patients in the multifraction radiotherapy group and 345 in the single-fraction radiotherapy group. Although not reaching the noninferiority limit defined, the 2 dose groups had no clinically important differences in ambulatory status, recovery, or pain control. Importantly, with a median overall survival of only 13 weeks, the 2 radiotherapy schedules had no difference in overall survival (hazard ratio [HR] = 1.02, 95% confidence interval [CI] = 0.74 to 1.41; P = .91). The conclusion was that patients can be treated with a single dose, thus avoiding unnecessary additional clinic visits. A subsequent analysis (3) examining prognostic factors for overall survival in the SCORAD trial showed evidence that overall survival was significantly worse in male patients; lung, gastrointestinal, and other types of cancer; compression at C1-T12; presence of nonskeletal metastases; and poor ambulatory status.
Outcomes
HRQoL was measured using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire–Core 30 (QLQ-C30) (12-15), completed by patients at baseline and at weeks 1, 4, 8, and 12 postrandomization. To allow patients some leeway for questionnaire completion, the HRQoL evaluations at the week 8 time point included assessments made from day 49 to 62 postrandomization. Similar assessments were conducted at weeks 1, 4, and 12, corresponding to days 7-13, 21-34, and 70-97 postrandomization, respectively.
The 15 primary scales of QLQ-C30 were supplemented with the QLU-C10D, a preference-based measure derived from 10 of the 15 dimensions of QLQ-C30: Physical Functioning, Role Functioning, Social Functioning, Emotional Functioning, Pain, Fatigue, Nausea and Vomiting, Insomnia, Appetite Loss, and Diarrhoea. QLU-C10D was used to produce UK-validated utility weights.
Overall survival was measured as the time from randomization until death from any cause; otherwise, patients were censored at the date last seen alive. The QAS was computed by multiplying Kaplan-Meier survival probabilities with the UK utility weights obtained from the QLU-C10D. The outcomes gathered on preferred and actual places of care were classified into distinct categories: home or with relatives; hospital care; living in care home, hospice, or other locations; and instances where data were not available or missing.
Data analysis
The association between baseline HRQoL scores and overall survival was assessed using univariable and multivariable Cox regression, adjusted for treatment, primary tumor, ambulatory status, sex, spinal cord compression (SCC) site, and nonskeletal metastases. HRQoL scores were modeled continuously and categorically (≤50 vs >50 for QLQ-C30; ≤0.5 vs >0.5 for QLU-C10D). Scores range from 0 to 100 for all QoL endpoints. For the global health and functional scales, 0 indicates poor health and 100 is good health. For the symptom scale, 0 indicates no symptoms and 100 is a high level of symptoms. For QLU-C10D, 0 represents death and 1 represents perfect health (16). For the global health and functional scales and QLU-C10D utility score, a positive difference indicates that single-fraction therapy was better, and a negative difference indicates that multifraction therapy was better. For the symptoms scale, a negative difference indicates that single-fraction therapy was better, and a positive difference indicates that multifraction therapy was better. Hazard ratios (HRs) compared high scores with low scores, with adjustments made for multiple testing using 99% confidence intervals (CIs) and associated P values.
An analysis of covariance examined differences in mean HRQoL scores between arms at 8 weeks. Estimates were mean differences (single fraction minus multifraction) adjusted for baseline scores and covariates selected on the basis of findings from our previous analysis of prognostic factors (3) (primary tumor, ambulatory status, sex, location of SCC site, and nonskeletal metastases). Positive differences indicated that single fraction was better for global, functional scales and QLU-C10D; negative differences indicated that multifraction was better for symptoms. Ninety-nine percent confidence intervals were used, and statistical significance was set at 1% to account for multiple testing.
A mixed model evaluated treatment impact on HRQoL scores over time. The model included treatment, time, primary tumor, ambulatory status, sex, SCC site, and nonskeletal metastases as fixed effects. Repeated HRQoL measures used a random intercept and slope model with an unstructured variance–covariance matrix. Treatment effects (single fraction minus multifraction) were presented along with 99% confidence interval, and significance was set at 1%. Estimated HRQoL means over time were graphically presented by treatment. The interpretation of QoL differences between treatment groups was based on guidelines for trivial, small, medium, and large QoL effects (17).
For QAS analysis, a 12-week timeframe postrandomization was chosen, in line with the set protocol timepoint for discontinuing HRQoL measurements. Average QLU-C10D utility weights were estimated via a random intercept and time-slope model. The resulting estimated utility weights were combined with survival rates, estimated via the Kaplan-Meier method, to compute the 12-week QAS for each treatment arm, which was then defined as the area under the survival-utility curve. Bootstrap-based 95% confidence interval and P values for QAS treatment differences were presented.
Preferred and actual place of care were summarized visually using bar graphs of percentages of patients alive at 1, 4, 8, and 12 weeks. Fisher exact tests assessed the association between treatment and actual place of care at each timepoint set with 1% statistical significance.
Reporting followed CONSORT PRO guidelines (18). Analysis used STATA 17.0.
Results
Participant flow and HRQoL assessment compliance
Of the 686 patients, 684 patients had HRQoL data at any timepoint and 590 patients had a baseline value plus at least 1 later value. The demographics and disease features were evenly distributed between the 2 treatment groups and among patients with HRQoL assessment at any timepoint and during follow-up (see details in Supplementary Table 1, available online). HRQoL completion rates were similar between the trial arms (Table 1), with a total of 1090 assessments in the multifraction group and 1066 in the single-fraction group. Nearly all patients—98% in both groups—underwent HRQoL assessment at baseline (day 0). Completion rates decreased over time but remained similar between groups: 75% (multifraction) and 71% (single fraction) beyond week 1 (day 7), 69% and 70% beyond week 4 (day 28), 69% and 67% beyond week 8 (day 56), and 44% and 52% beyond week 12 (day 84).
Table 1.
HRQoL assessment compliance between treatment groups
| Timing and number of QoL assessments | Multifraction radiotherapy group | Single fraction radiotherapy group |
|---|---|---|
| n = 341 | n = 345 | |
| Total QoL assessments | 1090 | 1066 |
| No. of patients by number of QoL assessments | ||
| 0 | 0 (0%) | 2 (1%) |
| 1 | 36 (11%) | 47 (14%) |
| 2 | 89 (26%) | 100 (29%) |
| 3 | 78 (23%) | 59 (17%) |
| 4 | 48 (14%) | 43 (12%) |
| 5 | 90 (26%) | 94 (27%) |
| Median (range) | 3 (1-5) | 3 (1-5) |
| No. of patients assessed for HRQoLf | ||
| Baselinea | 333/341 (98%)f | 338/345 (98%) |
| Beyond week 1b | 245/325 (75%) | 241/340 (71%) |
| Beyond week 4c | 195/281 (69%) | 188/269 (70%) |
| Beyond week 8d | 145/211 (69%) | 142/212 (67%) |
| Beyond week 12e | 71/162 (44%) | 80/155 (52%) |
QOL forms completed on day 0. HRQoL = health-related quality of life.
QOL forms completed beyond day 7.
QOL forms completed beyond day 28.
QOL forms completed beyond day 56.
QOL forms completed beyond day 84.
Shows the actual number of completed QOL forms divided by the expected number, which includes only those who were alive at that specific timepoint.
Association between baseline HRQoL and overall survival
Table 2 shows the results from the Cox regression analyses of the association between baseline HRQoL and overall survival. After adjustment for baseline factors, patients with a baseline physical functioning score of lower than 50 demonstrated a 28% reduction in the risk of death compared with those scoring 50 or higher (HR = 0.72, 99% CI = 0.54 to 0.95; P = .003). Similar findings were observed for social functioning and QLU-C10D utility score scales. For global health status, no significant association was observed with overall survival in the multivariate analysis (HR = 0.87, 99% CI = 0.63 to 1.20; P = .260). Among the symptom scales, higher levels of fatigue and dyspnea were associated with increased risk of death (HR for fatigue = 1.35, 99% CI = 1.03 to 1.76; P = .004; HR for dyspnea = 1.61, 99% CI = 1.24 to 2.08; P < .001). Similar results were found when baseline HRQoL scores were included in the model as a continuous variable (see Supplementary Table 2, available online).
Table 2.
Cox regression for the association between baseline HRQoL scores (categorical) and overall survival
| QoL scorea |
Overall survival univariate Cox modelb |
Overall survival multivariate Cox modelc |
||||
|---|---|---|---|---|---|---|
| ≤50 score N (%)d | >50 score N (%) | HR (99% CI) >50 score vs ≤50 score | P | HR (99% CI) >50 score vs ≤50 score | P | |
| Global Health Status | ||||||
| Global Health Status | 544 (79%) | 123 (18%) | 0.67 (0.49 to 0.91) | .001 | 0.87 (0.63 to 1.20) | .26 |
| Functional scales | ||||||
| Physical Functioning | 477 (70%) | 194 (28%) | 0.58 (0.45 to 0.75) | <.001 | 0.72 (0.54 to 0.95) | .003 |
| Role Functioning | 571 (83%) | 100 (15%) | 0.69 (0.50 to 0.96) | .003 | 0.89 (0.63 to 1.25) | .37 |
| Emotional Functioning | 241 (35%) | 428 (62%) | 0.87 (0.68 to 1.10) | .12 | 1.06 (0.83 to 1.35) | .53 |
| Cognitive Functioning | 231 (34%) | 438 (64%) | 0.69 (0.54 to 0.87) | <.001 | 0.83 (0.65 to 1.06) | .06 |
| Social Functioning | 456 (66%) | 213 (31%) | 0.68 (0.53 to 0.88) | <.001 | 0.78 (0.60 to 1.00) | .01 |
| Symptom scales | ||||||
| Fatigue | 202 (29%) | 466 (68%) | 1.68 (1.30 to 2.17) | <.001 | 1.35 (1.03 to 1.76) | .004 |
| Nausea and Vomiting | 607 (88%) | 63 (9%) | 1.27 (0.87 to 1.87) | .11 | 1.20 (0.81 to 1.78) | .23 |
| Pain | 175 (26%) | 496 (72%) | 1.29 (0.99 to 1.68) | .02 | 1.17 (0.89 to 1.53) | .14 |
| Dyspnea | 481 (70%) | 188 (27%) | 1.81 (1.41 to 2.33) | <.001 | 1.61 (1.24 to 2.08) | <.001 |
| Insomnia | 359 (52%) | 309 (45%) | 1.31 (1.04 to 1.64) | .003 | 1.07 (0.85 to 1.36) | .45 |
| Appetite Loss | 356 (52%) | 313 (46%) | 1.44 (1.15 to 1.81) | <.001 | 1.25 (0.99 to 1.59) | .01 |
| Constipation | 332 (48%) | 333 (49%) | 1.23 (0.98 to 1.54) | .02 | 1.01 (0.79 to 1.28) | .94 |
| Diarrhoea | 602 (88%) | 59 (9%) | 1.27 (0.86 to 1.87) | .12 | 1.18 (0.79 to 1.76) | .28 |
| Financial Difficulties | 548 (80%) | 119 (17%) | 0.96 (0.71 to 1.29) | .70 | 0.81 (0.59 to 1.10) | .08 |
| QLU-C10D (0-1 scale) | ||||||
| Utility score | 497 (72%) | 131 (19%) | 0.51 (0.37 to 0.69) | P < .01 | 0.67 (0.48 to 0.93) | .002 |
Scores range from 0 to 100 for all endpoints with exception of QLU-C10D. For the global health and functional scales (including satisfaction with health care and sexual functioning), 0 indicates poor health and 100 is good health. For all symptom scales, 0 indicates no symptoms and 100 is high level of symptoms. For QLU-C10D, 0 represents death and 1 represents perfect health. CI = confidence interval; HR = hazard ratio; HRQoL = health-related quality of life.
Low-score QoL response values vs high-score QoL response values.
Adjusted for treatment and baseline covariates: primary tumor, ambulatory status, sex, location of spinal cord compression site, and nonskeletal metastases.
Missing data mean that percentages do not add up to 100%.
Treatment difference in HRQoL at 8 weeks and over time
After adjusting for baseline HRQoL scores and other relevant clinical variables, the analysis of covariance identified no evidence of treatment differences at 8 weeks in patient-reported symptoms at the 1% significance level. (Table 3). At the 8-week point, patients receiving single-fraction treatment had an average of 9.5 score points higher on the insomnia symptom scale (treatment difference = 9.5, 99% CI = -0.56 to +19.57; P = .02) compared with those receiving multiple fractions. No significant treatment differences at the 1% or 5% level were observed in functioning scales, global health status, or the QLU-C10D scale.
Table 3.
Treatment difference in HRQoL at 8 weeks, adjusting for quality of life at baseline (analysis of covariance)
| QoL responseb | Multifraction |
Single fraction |
Model adjusting for baseline quality of life and baseline characteristicsa |
|||
|---|---|---|---|---|---|---|
| Baseline, mean (SD) | Week 8, mean (SD) | Baseline, mean (SD) | Week 8, mean (SD) | Treatment mean differencec in quality of life at 8 weeksd (99% CI) | P e | |
| Global Health Status | ||||||
| Global Health Status | 35.99 (22.99) | 43.73 (23.36) | 36.45 (25.55) | 42.73 (25.33) | −1.76 (−9.66 to +6.13) | .56 |
| Functional scales | ||||||
| Physical functioning | 37.32 (31.87) | 35.35 (31.30) | 45.17 (28.61) | 37.09 (29.92) | −2.99 (−11.86 to +5.88) | .38 |
| Role functioning | 29.69 (32.12) | 28.77 (31.99) | 28.93 (29.49) | 29.64 (31.67) | −0.51 (−10.76 to +9.74) | .90 |
| Emotional functioning | 66.48 (27.25) | 72.62 (25.57) | 64.99 (26.69) | 69.10 (27.29) | −2.87 (−10.92 to +5.17) | .35 |
| Cognitive functioning | 72.27 (26.24) | 75.56 (25.66) | 69.89 (25.39) | 73.19 (27.39) | −1.02 (−9.18 to +7.14) | .75 |
| Social functioning | 47.62 (34.53) | 39.60 (35.74) | 44.89 (32.38) | 41.60 (33.63) | 2.44 (−8.03 to +12.90) | .55 |
| Symptom scales | ||||||
| Fatigue | 56.92 (25.55) | 56.82 (26.12) | 60.89 (24.46) | 54.75 (29.11) | −2.56 (−11.20 to +6.08) | .44 |
| Nausea and vomiting | 14.99 (20.63) | 11.90 (17.91) | 17.34 (22.83) | 10.69 (18.61) | −1.42 (−7.43 to +4.59) | .54 |
| Pain | 68.35 (29.70) | 40.90 (33.67) | 72.13 (29.46) | 43.15 (31.81) | 1.21 (−9.69 to +12.12) | .77 |
| Dyspnea | 19.61 (27.24) | 28.77 (31.23) | 25.00 (31.41) | 34.02 (33.91) | 5.69 (−4.69 to +16.06) | .16 |
| Insomnia | 43.70 (35.45) | 30.20 (27.33) | 45.43 (37.38) | 38.48 (33.88) | 9.50 (−0.56 to +19.57) | .02 |
| Appetite loss | 33.61 (37.71) | 27.40 (31.32) | 44.17 (36.59) | 31.85 (34.53) | 3.10 (−8.09 to +14.30) | .47 |
| Constipation | 47.58 (36.96) | 28.21 (32.64) | 49.46 (35.18) | 24.06 (31.76) | −3.48 (−13.96 to +6.99) | .39 |
| Diarrhea | 12.07 (25.02) | 9.97 (21.11) | 9.56 (20.77) | 15.03 (26.80) | 6.00 (−2.24 to +14.23) | .06 |
| Financial difficulties | 19.05 (32.64) | 24.79 (34.51) | 21.41 (31.69) | 21.51 (31.00) | −2.61 (−12.77 to +7.55) | .51 |
| QLU-C10D (0-1 scale) | ||||||
| Utility score | 0.40 (0.22) | 0.47 (0.24) | 0.37 (0.21) | 0.45 (0.24) | −0.01 (−0.09 to +0.07) | .67 |
Model adjusted for baseline characteristics: primary tumor, ambulatory status, sex, location of spinal cord compression site, and nonskeletal METS. CI = confidence interval; HRQoL = health-related quality of life.
Scores range from 0 to 100 for all endpoints. For the global health and functional scales, 0 indicates poor health and 100 is good health. For the symptom scale, 0 indicates no symptoms and 100 is high level of symptoms. For QLU-C10D, 0 represents death and 1 represents perfect health.
Treatment mean difference refers to single-fraction radiotherapy arm minus multifraction radiotherapy arm.
For the global health and functional scales and QLU-C10D utility score, a positive difference indicates that single-fraction therapy was better, and a negative difference indicates that multifraction therapy was better. For the symptoms scale, a negative difference indicates that single-fraction therapy was better, and a positive difference indicates that multifraction therapy was better.
P values (2-sided) are unadjusted for multiple comparisons, so 99% CIs are shown.
The results of the mixed-model analysis of HRQoL are shown in Supplementary Table 3 (available online), which accounts for patient-specific longitudinal HRQoL measures over all timepoints. This approach did not show evidence of a treatment effect on the average HRQoL over time at the 1% and 5% significance levels for any of the HRQoL measures, including global health status, health utility score (QLU-C10D), and physical functioning (Figure 1, Supplementary Table 3, available online). The treatment difference for global health status adjusted for baseline factors was −0.88 (99% CI = −4.43 to 2.67, P = .52) and for physical functioning was 0.13 (99% CI = −3.85 to 4.11, P = .93). All the differences between the trial arms were below the clinically meaningful effect (17), indicating that the radiotherapy regimens had a similar impact on patients’ HRQoL.
Figure 1.
Average scores for (A) global quality of life and (B) health utility score (QLU-C10D) and (C) physical functioning.
Quality-of-life adjusted survival
Quality-adjusted survival over a 12-week postrandomization period was similar between the 2 treatment groups. Comparing the 2 groups, the single-fraction group had an average QAS of 3.39 weeks (95% CI = 3.11 to 3.67), and the multifraction group had an average QAS of 3.65 weeks (95% CI = 3.36 to 3.94). This observed difference of 1.99 days was not statistically significant (difference = −0.26 weeks, 95% CI = −0.66 to 0.14; P = .201).
Patient preferences and actual place of care
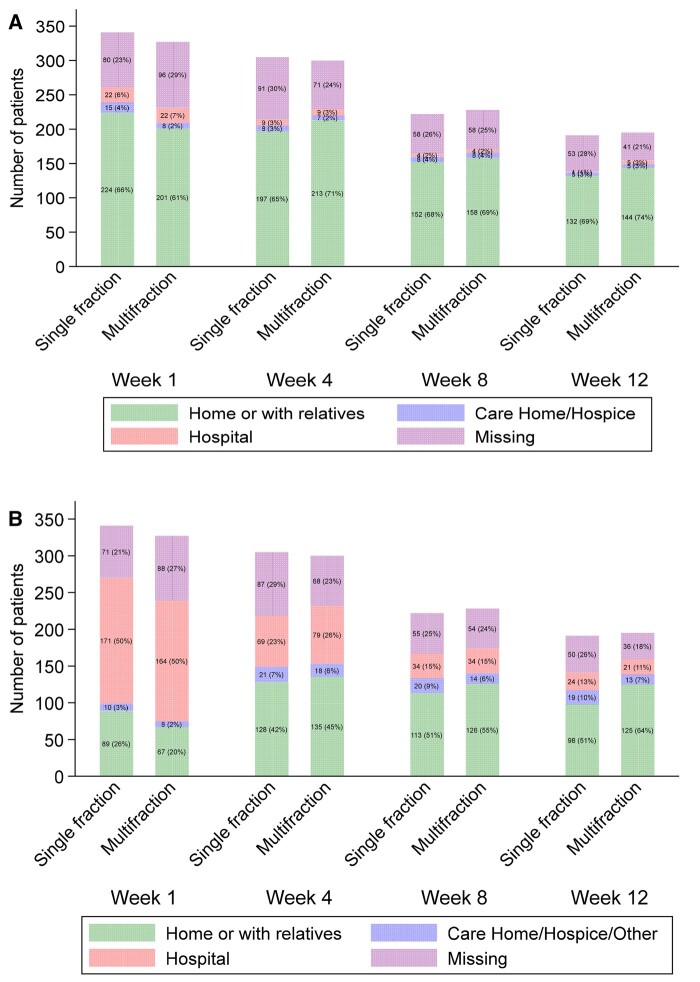
The majority of patients (61%-74%) expressed a consistent preference to stay at home or with family across weeks 1, 4, 8, and 12 of the study period (Figure 2, A). However, less than 5% of patients indicated a preference to reside in a care home or hospice over time. Despite these preferences, the percentage of patients actually receiving care at home or with relatives at weeks 1, 4, 8, and 12 ranged from 26% to 51% in single fraction and 20% to 64% multifraction, respectively (Figure 2, B). Over time, there was a decline in the percentage of patients in each treatment group who reported hospitalization as their actual place of care. At week 1, half of the patients were hospitalized in each group. This percentage decreased to 13% and 11% for the single-fraction and multifraction groups, respectively, by week 12. Importantly, there was no significant association between the treatment group and the actual place of care across all weeks (P values: week 1 = .170, week 4 = .338, week 8 = .643, week 12 = .078).
Figure 2.
Preferred place of care (A) and actual place of care (B) for 668 patients (341 single fraction, 327 multifraction).
Discussion
Overall, the HRQoL analysis from SCORAD confirms that there is no statistical or clinically meaningful difference in the average HRQoL over time in patients treated with radiotherapy between a single dose of 8 Gy and 20 Gy in 5 fractions for metastatic spinal canal compression. Average scores for global health status and physical functioning scales remained poor throughout the 12 weeks posttreatment, showing no significant signs of improvement over this period. This finding is consistent with the primary endpoint of the study, which showed that there was little or no change in ambulatory status at 8 weeks in 76% (261/342) of patients, with 62% (213/342) maintaining good ambulatory function (Grade 1-2) (19). Our data suggest that the main role of radiotherapy in this setting is to maintain functioning and highlight the need for early diagnosis before major neurological deficits develop.
A systematic review to assess the impact of spinal cord compression on HRQoL and understand the factors contributing to it (12) found only 6 nonrandomized studies that addressed this specific question, and, notably, the most recent among them was published in 2013. Two of these used semistructured interviews in a small subgroup of patients, the other 4 using validated tools: SF36, Schedule for the Evaluation of QoL (SIEQoL Dw), and FACT-G. Overall, these studies reported “good quality of life” at 1 month but recognized that there was selection for the less frail patients with better prognosis to complete the questionnaires or interviews. A considerable strength of the SCORAD study is the availability of baseline HRQoL data for 98% of all patients, and notably high compliance rates in HRQoL assessment at 4 and 8 weeks (70% or 383/550, and 68% or 287/423, respectively).
It is generally accepted that MSCC is related to worse HRQoL as well as poor survival, but the relationship has not been quantified reliably until our findings here. Patients predominantly scored 50 or lower for global health, physical, role, and social functioning (79%, 70%, 83%, and 66%, respectively). In contrast, for cognitive and emotional functioning 62% and 64% of patients, respectively, achieved scores above 50. This is in keeping with one study identified in the systematic review (20), which found that more than 50% of patients did not exhibit anxiety or depression “shortly after diagnosis.” It is postulated that this may be because the implications of the diagnosis have yet to be realized at that early point or unrealistic expectations with regard to outcome have been given. This may also be an effect of the use of high-dose steroids at this point in the management of MSCC. In terms of symptom scores, pain and fatigue are more prominent than other symptoms, which is consistent with the findings of the systematic review (19). This review also highlighted the significant issue of loss of bowel control. However, in SCORAD, only 9% of patients had a score for diarrhea above 50, potentially reflecting a different population with earlier diagnosis.
Quality-of-life adjusted survival is an important endpoint reflecting the proportion of remaining survival that a patient will, on average, experience good HRQoL as defined by the health utility weighting derived specifically for a UK population. In this study, the largest utility decrements were associated with physical functioning and pain, which are clearly major components reducing the health utility weighting for patients with MSCC. No difference is seen between the 2 dose levels overall, and the levels of good quality survival remain fairly constant with only a small reduction with time distinct from actual survival, which falls rapidly across the 12-week period. This means that longer survivors will benefit most from the impact of treatment upon HRQoL with at 12 weeks around one-half of patients alive having good HRQoL compared with less than 40% at baseline.
The strength of this study is that it uses prospective data from a randomized trial. Compliance with relatively complex assessments such as HRQoL in the palliative setting when, despite entry criteria stating a minimum of 3 months survival, there is a substantial reduction in the trial population as a result of progressive metastatic disease, is challenging. We achieved a high rate of baseline assessments, which minimized the impact of this, but nonetheless the results at 8 weeks are derived from only two-thirds of the population.
More than 60% of patients preferred home care at diagnosis, although this should be interpreted in the light of the demographics of the population, which were predominantly male and aged older than 70 years. In fact, only half actually achieved this, which may reflect medical complications requiring inpatient care or limited community support for relatively high dependency patients. This finding has important implications for health-care provision in the management of MSCC.
In conclusion, prolonged survival in patients with MSCC was associated with better HRQoL (physical, social functioning, fatigue, dyspnea, and appetite loss). Across all timepoints, there were no clinically relevant differences in any HRQoL domain between the multifraction and single-fraction group. There were minimal differences in the major clinical endpoints ambulatory status and overall survival between treatment groups (19), and taken together with our results on HRQoL, the SCORAD trial provides evidence that patients with MSCC need only one dose of radiotherapy, avoiding the extra clinic visits associated with multifraction therapy when they have only a few months left to live. An important implication for the care of these patients is the observation that pain, fatigue, and physical functioning are prominent contributors to quality of life in this population. Early involvement of palliative care services skilled in pain control, management of fatigue, and physical rehabilitation could make a substantial impact and should be made available to all patients presenting with MSCC.
Supplementary Material
Acknowledgments
SCORAD investigators included K. Hopkins, V. Misra, T. Holt, R. McMenemin, F. McKinna, M. Krishnaswamy, A. Bates, N. O’Rourke, J. Lester, T. Sevitt, D. Roos, G. Brown, S. Shibu Thomas, S. Forsyth.
The trial was sponsored by University College London (Trial Sponsor No.: UCL/09/0199) and coordinated centrally by Cancer Research UK and UCL Cancer Trials Centre (UCL CTC).
The International Standard Randomised Controlled Trial Number is ISRCTN97108008.
Protocol available at: http://www.ctc.ucl.ac.uk/
The funder played no role in the analysis of data or in writing this article.
Contributor Information
Peter J Hoskin, Mount Vernon Cancer Centre, Northwood, UK; Division of Cancer Sciences, University of Manchester, Manchester, UK.
Krystyna Reczko, CRUK & UCL Cancer Trials Centre, London, UK.
Memuna Rashid, CRUK & UCL Cancer Trials Centre, London, UK.
Allan Hackshaw, CRUK & UCL Cancer Trials Centre, London, UK.
Andre Lopes, CRUK & UCL Cancer Trials Centre, London, UK.
the SCORAD Investigators:
K Hopkins, V Misra, T Holt, R McMenemin, F McKinna, M Krishnaswamy, A Bates, N O’Rourke, J Lester, T Sevitt, D Roos, G Brown, S Shibu Thomas, and S Forsyth
Data availability
Data sharing is subject to restrictions imposed by the ethical approval for this study and national data protection regulations. Requests can be made directly to the corresponding author.
Author contributions
Peter J. Hoskin, MD (Conceptualization; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing), Krystyna Reczko, BSc (Data curation; Project administration; Supervision; Validation; Writing—review & editing), Memuna Rashid, BSc (Data curation; Formal analysis; Methodology; Validation; Writing—original draft; Writing—review & editing), Allan Hackshaw, MSc (Conceptualization; Data curation; Formal analysis; Funding acquisition; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing), Lopes Andre, MSc (Data curation; Formal analysis; Investigation; Methodology; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing).
Funding
This work was supported by CRUK Project Grants C2422/7932 and C2422/A11408; Cancer Council Queensland and UK National Institute of Health Research. PJ Hoskin is supported by NIHR Manchester Biomedical Research Centre (NIHR203308).
Conflicts of interest
The authors confirm that they have no conflicts of interest.
References
- 1. Metastatic Spinal Cord Compression in Adults: Risk Assessment, Diagnosis and Management Clinical Guideline [CG75]. https://www.nice.org.uk/guidance/cg75/chapter/Introduction. Accessed February 3, 2024. [PubMed]
- 2. Loblaw DA, Mitera G, Ford M, Laperriere N. A 2011 updated systematic review and clinical practice guideline for the management of malignant extradural spinal cord compression. Int J Radiation Oncol Biol Phys. 2012;84(2):312-317. [DOI] [PubMed] [Google Scholar]
- 3. Hoskin PJ, Hopkins K, Misra V, et al. Prognostic factors for survival and ambulatory status at 8 weeks with metastatic spinal cord compression in the SCORAD randomised trial. Radiother Oncol. 2022;173:77-83. [DOI] [PubMed] [Google Scholar]
- 4. Morgen SS, Engelholm SA, Larsen CF, Søgaard R, Dahl B. Health-related quality of life in patients with metastatic spinal cord compression. Orthopaed Surg. 2016;8(3):309-315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Higginson IJ, Daveson BA, Morrison RS, et al. BuildCARE. Social and clinical determinants of preferences and their achievement at the end of life: prospective cohort study of older adults receiving palliative care in three countries. BMC Geriatr. 2017;17(1):271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Valentino TCDO, Paiva CE, de Oliveira MA, et al. Preference and actual place-of-death in advanced cancer: prospective longitudinal study. BMJ Support Palliat Care. 2023;spcare-2023-004299. doi: 10.1136/spcare-2023-004299 [DOI] [PubMed] [Google Scholar]
- 7. Gomes B, Calanzani N, Gysels M, et al. Heterogeneity and changes in preferences for dying at home: a systematic review. BMC Palliat Care. 2013;12:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Nilsson J, Blomberg C, Holgersson G, et al. End-of-life care: where do cancer patients want to die? A systematic review. Asia Pac J Clin Oncol. 2017;13(6):356-364. [DOI] [PubMed] [Google Scholar]
- 9. Vidal M, Rodriguez-Nunez A, Hui D, et al. Place-of-death preferences among patients with cancer and family caregivers in inpatient and outpatient palliative care. BMJ Support Palliat Care. 2020;12(e4):e501-e504. [DOI] [PubMed] [Google Scholar]
- 10. Fereidouni A, Rassouli M, Salesi M, et al. Preferred place of death in adult cancer patients: a systematic review and metaanalysis. Front Psychol. 2021;12:704590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hoskin PJ, Hopkins K, Misra V, et al. Effect of single-fraction vs multifraction radiotherapy on ambulatory status among patients with spinal canal compression from metastatic cancer: the SCORAD randomized clinical trial. JAMA. 2019;322(21):2084-2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Fayers P, Aaronson N, Bjordal K, Groenvold M, Curran D, Bottomley A. EORTC QLQ–C30 Scoring Manual. 3rd ed. Brussels: European Organisation for Research and Treatment of Cancer; 2001. [Google Scholar]
- 13. Fayers PM, , AaronsonNK, , BjordalK, et al, on behalf of the EORTC Quality of Life Group. The EORTC QLQ-C30 Scoring Manual 3rd ed. Brussels: European Organisation for Research and Treatment of Cancer; 2001. [Google Scholar]
- 14. https://www.eortc.org/app/uploads/sites/2/2018/08/Specimen-QLQ-C30-English.pdf. Accessed February 9, 2024.
- 15. Gamper E, Jansen F, Buettner M, et al, on behalf of the EORTC Quality of Life Group. EORTC Quality of Life Utility-Core 10 Dimensions (QLU-C10D) - User Manual. Brussels: European Organisation for Research and Treatment of Cancerh; 2023.
- 16. Norman R, Mercieca-Bebber R, Rowen D, et al. UK utility weights for the EORTC QLU-C10D. Health Econ. 2019;28(12):1385-1401. [DOI] [PubMed] [Google Scholar]
- 17. Cocks K, King MT, Velikova G, St-James MM, Fayers PM, Brown JM. Evidence-based guidelines for determination of sample size and interpretation of the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30. J Clin Oncol. 2011;29(1):89-96. [DOI] [PubMed] [Google Scholar]
- 18. Calvert M, Blazeby J, Altman DG, et al. CONSORT PRO Group. Reporting of patient-reported outcomes in randomized trials. The CONSORT PRO extension. JAMA. 2013;309(8):814-822. [DOI] [PubMed] [Google Scholar]
- 19. Harris M. Quality of life in patients with malignant spinal cord compression: a review of evidence-based literature. Int J Palliat Nurs. 2016;22(1):37-43. [DOI] [PubMed] [Google Scholar]
- 20. Levack P, Graham J, Kidd J. Listen to the patient: quality of life of patients with recently diagnosed malignant cord compression in relation to their disability. Palliat Med. 2004;18(7):594-601. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sharing is subject to restrictions imposed by the ethical approval for this study and national data protection regulations. Requests can be made directly to the corresponding author.