Abstract
Background
Surgical reconstruction is often necessary for severe tracheobronchial stenosis resulting from tuberculosis (TB). However, the long-term efficacy of this approach remains unclear. This study investigated the safety and long-term outcomes of surgery for severe post-TB tracheobronchial stenosis.
Methods
We conducted a retrospective study of 48 patients with severe post-TB tracheobronchial stenosis who underwent surgical reconstruction between 2015 and 2018 in a TB-endemic region. Pre- and postoperative evaluations included Karnofsky performance status, modified Medical Research Council (mMRC) dyspnea scale, spirometry, chest computed tomography (CT) scan, and bronchoscopy. The primary outcome was intervention-requiring restenosis over the long term.
Results
The mean patient age was 30.6±9.9 years, with 91.7% females. Airway fibrosis was the predominant lesion (93.8%), affecting the bronchi (93.8%) and trachea (6.2%). All the patients underwent resection and anastomosis, and 56.2% required lobectomy. Postoperative complications occurred in 13 patients (27.1%), with prolonged air leaks being the most prevalent (12.5%). All complications resolved with conservative management. Significant improvements in performance status, dyspnea, and lung function were observed postoperatively and sustained for over 5 years. Within a median follow-up of 69 months, five cases of intervention-requiring restenosis occurred within the first year. The freedom from restenosis rate was 90% from 1 year onwards.
Conclusions
Surgical reconstruction is safe and effective in treating severe post-TB tracheobronchial stenosis. Larger studies are required to validate these findings.
Keywords: Tuberculosis (TB), tracheal stenosis, bronchial stenosis, surgical plasty, reconstruction
Highlight box.
Key findings
• Surgical reconstruction is safe and effective for severe post-tuberculosis (TB) tracheobronchial stenosis.
What is known and what is new?
• Surgical reconstruction is often necessary for severe tracheobronchial stenosis resulting from TB, but its long-term efficacy is unclear.
• Surgical reconstruction has a good long-term efficacy for severe post-TB tracheobronchial stenosis.
What is the implication, and what should change now?
• Surgical reconstruction should be performed in cases of severe post-TB tracheobronchial stenosis.
Introduction
Tuberculosis (TB) remains a global public health threat, ranking as the 13th leading cause of death worldwide and the 2nd leading cause of death among human immunodeficiency virus (HIV) or acquired immunodeficiency syndrome (AIDS) patients. Approximately one-quarter of the global population carries latent TB, with 5–10% developing active disease. Each year, an estimated 10 million new cases and 1.5 million deaths occur (1).
Tracheobronchial TB, a leading cause of benign airway stenosis and affecting 10–40% of pulmonary TB patients (2-7), presents with diverse and often misleading symptoms owing to its variable location and disease stage (8-13). This challenges the timely diagnosis and risk of progression to airway narrowing, lung collapse, and TB reactivation. Despite the World Health Organization (WHO) guidelines and anti-TB therapy, no standard treatment algorithm exists for tracheobronchial stenosis. Interventions includes ablative techniques (electric or laser), balloon dilation to stenting, and tracheobronchial surgery.
Vietnam ranks among the top 30 countries with the highest TB and multidrug-resistant TB burden. In 2019, with a TB incidence rate of 176/100,000 and a mortality rate of 11.8/100,000 (14), Vietnam still lacks routine tracheobronchial TB screening. This leads to late diagnosis and ineffectiveness of conservative interventions, forcing reliance on tracheobronchial surgery for advanced stenotic cases.
Despite this reliance on surgery, long-term outcome data on its effectiveness in post-TB tracheobronchial stenosis are scarce. Our center, with its extensive experience with this procedure, aimed to address this gap through this study. We evaluated the safety and long-term effectiveness of tracheobronchial surgery in treating severe post-TB tracheobronchial stenosis, providing valuable insights for improving patient outcomes and treatment strategies. We present this article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-230/rc).
Methods
Study design and population
This retrospective study was conducted at the Department of Thoracic Surgery, Pham Ngoc Thach Hospital, a tertiary referral hospital for TB treatment in Southern Vietnam. All patients who underwent tracheobronchial surgery for post-TB tracheobronchial stenosis between January 2015 and December 2018 were included in this study. The patients were followed up until December 2022. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional ethics committee of Pham Ngoc Thach Hospital (No. 1595/PNT-HĐĐĐ, dated November 18, 2020) and individual consent for this retrospective analysis was waived.
We included patients who (I) had confirmed pulmonary or tracheobronchial TB, diagnosed and treated with anti-TB therapy for at least three months, with a negative sputum smear for acid-fast bacilli, (II) had severe post-TB tracheobronchial stenosis demonstrated by chest computed tomography (CT) scan and flexible bronchoscopy, with a stenosis diameter exceeding 50% and a lumen size of the stenotic segment of ≤6 mm, (III) developed post-stenotic pulmonary complications such as pneumonia, consolidation, or atelectasis, and (IV) underwent tracheobronchial surgical reconstruction for the treatment of stenosis. We excluded patients with complex post-TB tracheobronchial stenosis involving multiple sites that required different interventions.
Surgical technique
All patients underwent general anesthesia with specific endotracheal tube placement based on stenosis location: tracheal intubation for tracheal stenosis and endobronchial intubation for bronchial procedures. The surgical approach and patient positioning were adapted according to the anatomy involved.
For carinal and right bronchial stenosis, the patients were placed in a 90-degree right lateral decubitus position. A 12–15 cm right posterior lateral thoracotomy in the fifth intercostal space was performed. For left bronchial stenosis, patients were placed in a 90-degree left lateral decubitus position and a 12–15 cm left posterior lateral thoracotomy at the fifth intercostal space was performed. A Carlens double-lumen endotracheal tube was used for anesthesia.
For tracheal stenosis, patients were placed in the supine position. A 4–6 cm transverse neck incision was performed at 2–3 cm above the sternal notch. Partial median sternotomy was combined for stenosis of the lower third of the trachea. Following resection of the stenotic segment, the initial endotracheal tube was replaced with a new endotracheal tube that was directly placed into the distal trachea through tracheal transection. After posterior membranous anastomosis of the trachea, the second endotracheal tube was removed and the initial tube was advanced to continue anesthesia.
In all cases, complete resection of the fibrotic lesions ensured smooth cut ends for anastomosis (Figure 1). Typically, end-to-end anastomosis of the trachea or the main bronchus was performed. In some cases, lobectomy with lobar bronchus anastomosis to the trachea, carina, or main bronchus was performed (Figure 2). We did not use any tissue to buttress the anastomosis due to no signs of active infection. Slide tracheoplasty or bronchoplasty was not performed in any patient due to the fibrotic lesions. Several tension reduction techniques were applied, including tracheal mobilization, neck flexion, hilar mobilization, and division of the inferior pulmonary ligament. For tracheal resections exceeding 3–4.5 cm, chin-to-chest fixation with 1-0 Vicryl sutures was performed for 10 days. For tracheal resections greater than 4.5 cm, hyoid bone release was added.
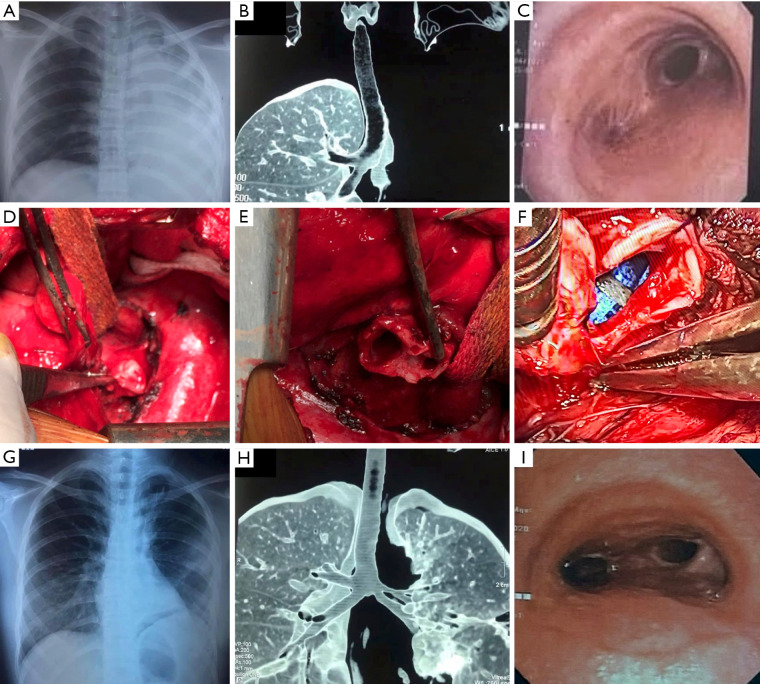
Figure 1.
Preoperative, intraoperative, and postoperative images of a case with left main bronchial stenosis. (A) Left lung collapse on preoperative chest X-ray; (B) left main bronchial occlusion on preoperative chest CT-scan with 3D reconstruction; (C) left main bronchial stenosis on preoperative bronchoscopy; (D) intraoperative image of left main bronchial occlusion; (E) anastomosis of the left main bronchus after resection of the stenotic segment; (F) anastomosis of the carina; (G) postoperative chest X-ray; (H) postoperative chest CT-scan with 3D reconstruction; (I) postoperative bronchoscopy image. CT, computed tomography.
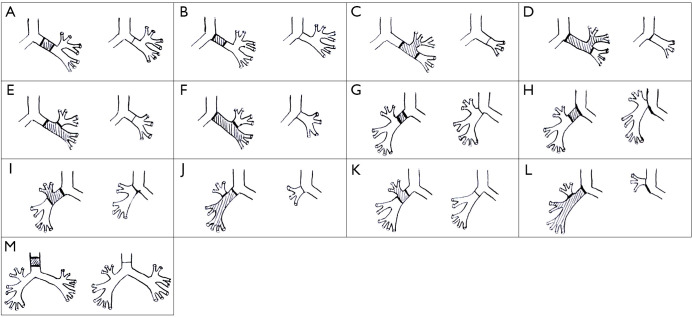
Figure 2.
Types of anastomosis. (A) End-to-end anastomosis of the left main bronchus; (B) anastomosis of the left main bronchus to the carina; (C) anastomosis of the left lower lobe bronchus to the left main bronchus; (D) anastomosis of the left lower lobe bronchus to the carina; (E) anastomosis of the left upper lobe bronchus to the left main bronchus; (F) anastomosis of the left upper lobe bronchus to the carina; (G) end-to-end anastomosis of the right main bronchus; (H) anastomosis of the right main bronchus to the trachea; (I) anastomosis of the right intermediate bronchus to the carina; (J) anastomosis of the right upper lobe bronchus to the right main bronchus; (K) anastomosis of the right intermediate bronchus to the right main bronchus; (L) anastomosis of the right upper lobe bronchus to the trachea; (M) end-to-end anastomosis of the trachea.
Anastomosis was performed using 3-0 PDS sutures. We first performed two interrupted sutures to mark and avoid twisting. Continuous sutures were placed in the posterior half. Interrupted or continuous sutures were used for the anterior half depending on the tension of the anastomosis. Anastomotic leaks were checked before the final closure. A neck wound drain and chest tube were placed.
Preoperative and postoperative management
Before surgery, patients were evaluated for their history of TB and anti-TB treatment, symptoms and signs, Karnofsky performance status, modified Medical Research Council (mMRC) dyspnea scale, and spirometry. Diagnostic imaging included chest radiography, chest CT with 3D reconstruction of the tracheobronchial tree, and bronchoscopy. All patients underwent a pre-surgical evaluation by interventionists, but the lesions were not amenable to treatment with endoscopic techniques. Tracheobronchial stenosis was classified into four grades, which is in line with the Myer-Cotton grading system used for subglottic stenosis: mild (stenosis 0–50%), moderate (stenosis 51–70%), severe (stenosis 71–99%), and occlusion (stenosis 100%).
After surgery, the patients were monitored clinically and underwent chest radiography and bronchoscopy. The neck drain was removed when the drainage volume was ≤10 mL in 24 hours and there was no air leak. The chest tube was removed when the drainage volume was ≤100 mL in 24 hours and there was no air leak. Patients were discharged when they were able to perform daily activities, the drains had been removed, the chest radiography showed lung expansion, and bronchoscopy showed no anastomotic stenosis of >50%.
After discharge, the patients were followed up at 1, 3, 6, and 12 months, and annually thereafter. At each follow-up visit, they underwent a physical examination, Karnofsky performance status assessment, mMRC dyspnea scale assessment, chest radiography, and evaluation of complications. We performed bronchoscopies at scheduled intervals (1 day, 1 month, and 1 year after surgery) to monitor patients’ recovery. Additionally, bronchoscopy was performed more frequently if any abnormalities, such as anastomotic stenosis or dehiscence, were identified during previous examinations. A chest CT scan with 3D reconstruction of the tracheobronchial tree and spirometry were performed 1 year after surgery for monitoring lung function improvement.
Outcomes
Safety was assessed based on intraoperative and postoperative complications within 30 days of the surgery. Effectiveness was assessed by the improvement of the Karnofsky performance status, mMRC dyspnea scale, spirometry parameters, and the occurrence of anastomotic stenosis or recurrent tracheobronchial stenosis requiring interventions such as balloon dilation, stenting, or reoperation. Recurrent anastomotic stenosis or tracheobronchial stenosis was diagnosed using flexible bronchoscopy and chest CT scan with 3D reconstruction of the tracheobronchial tree. Reintervention for recurrent stenosis was indicated when the stenosis was >50% or caused complications such as atelectasis.
Statistical analysis
Continuous variables were summarized as mean (standard deviation) or median (interquartile range), and categorical variables were summarized as frequencies (percentages). The long-term outcome (recurrent stenosis requiring intervention) was analyzed using the Kaplan-Meier method and was shown with a Kaplan-Meier curve. The paired t-test, Wilcoxon signed-rank test, and McNemar’s test were used to compare respiratory function parameters and airflow obstruction before and after surgery. Univariable Cox proportional hazards models were used to explore the factors associated with long-term recurrent stenosis. All tests were two-sided, and a P value of less than 0.05 was considered statistically significant. Statistical analyses were performed using R statistical software version 4.1.2.
Results
Fifty patients with post-TB tracheobronchial stenosis underwent tracheobronchial surgical plasty at our hospital between January 2015 and December 2018. We excluded two patients with complex stenosis (long segmental tracheal stenosis with main bronchial stenosis) who were treated with tracheal stenting combined with main bronchial surgical plasty. Finally, 48 patients were included in the study and followed-up until 2022. Surgery was indicated for these patients because endotracheal interventions were failed (2 patients) or lesions were deemed too complex for interventional bronchoscopy (46 patients).
The mean age was 30.6±9.9 years. Females were predominant (91.7%). Most patients had a history of TB (38 patients, 79.2%), and the remaining 10 patients (20.8%) had active TB. The median time from TB diagnosis to tracheobronchial stenosis diagnosis was 12 months (interquartile range, 7–60 months). Most of the patients (89.6%) had only local symptoms. The general condition of the patients was reduced, with 4 patients (8.3%) having moderate dyspnea and most (91.6%) having a Karnofsky score of 70–80. Among 45 patients with preoperative spirometry, there were 31 cases (68.9%) with restrictive lung disease, all of which were mild or moderate, and 12 patients (26.7%) with obstructive lung disease. Three cases were severe, five cases were moderate, and four cases were mild. All baseline characteristics of patients before surgery are shown in Table 1.
Table 1. Patient characteristics.
| Characteristics | All patients (N=48) |
|---|---|
| Age (years) | 30.6±9.9 [14–60] |
| Female | 44 (91.7) |
| TB status | |
| Pulmonary TB | 45 (93.8) |
| Bronchial TB | 3 (6.2) |
| TB activity | |
| Active TB | 10 (20.8) |
| Past TB | 38 (79.2) |
| Time from TB diagnosis to tracheobronchial stenosis (months) | 12 [7–60] |
| Symptoms | |
| Asymptomatic | 1 (2.1) |
| Local symptoms only | 43 (89.6) |
| Local and systemic symptoms | 4 (8.3) |
| Severity of dyspnea | |
| None | 28 (58.3) |
| Mild | 16 (33.3) |
| Moderate | 4 (8.3) |
| Karnofsky performance status score | |
| 90 | 2 (4.2) |
| 80 | 16 (33.3) |
| 70 | 28 (58.3) |
| 60 | 2 (4.2) |
| FVC (%)* | 61.4±12.8 [43–91] |
| FEV1 (%)* | 54.4±11.5 [15–75] |
| FEV1/FVC (%)* | 77.4±14.8 [31–98] |
| FEF 25–75 (%)* | 42.8±14.0 [7–72] |
| PEF (%)* | 56.8±14.8 [18–80] |
| Restrictive lung disease* | 31 (68.9) |
| Severity of restrictive disease (N=31) | |
| Mild | 10 (32.3) |
| Moderate | 21 (67.7) |
| Obstructive lung disease* | 12 (26.7) |
| Severity of obstructive disease (N=12) | |
| Mild | 4 (33.3) |
| Moderate | 5 (41.7) |
| Severe | 3 (25.0) |
| Length of follow-up (months) | 69 [61–74] |
Statistics are presented as mean ± standard deviation [range], median [interquartile range], and n (%). *, N=45. TB, tuberculosis; FVC, forced vital capacity; FEV1, forced expiratory volume in 1 second; PEF, peak expiratory flow; FEF 25–75, forced expiratory flow between 25% and 75% of vital capacity.
Chest radiography showed airway stenosis in only 2 cases (4.2%), but atelectasis in 34 cases (70.8%), pulmonary consolidation in 13 cases (27.1%), and linear fibrosis in 7 cases (14.6%). CT showed similar cases of atelectasis, pulmonary consolidation, and linear fibrosis; however, CT detected 100% of cases with airway stenosis. CT and bronchoscopy determined the location of stenosis was similar, with 3 cases (6.2%) of tracheal stenosis, 36 cases (75%) of left main bronchial stenosis, and 9 cases (18.8%) of right main bronchial stenosis. However, the location of the lesion on the bronchus differed between the CT, bronchoscopy, and intraoperative findings. The results of the CT were similar to those of surgery, with most cases having lesions involving the entire bronchus. Bronchoscopy revealed mostly proximal bronchial stenosis. This may be because the bronchoscope could not pass through the stenotic site. Most lesions were airway fibrosis (93.8%). The degree of stenosis was quite similar between CT and bronchoscopy, with most cases being severe stenosis or occlusion. Full detailed characteristics of the lesions are shown in Table 2.
Table 2. Lesion characteristics.
| Characteristics | X-ray findings (N=48) |
CT-scan findings (N=48) | Bronchoscopy findings (N=48) | Intraoperative findings (N=48) |
|---|---|---|---|---|
| Airway stenosis | 2 (4.2) | 48 (100.0) | – | – |
| Collapse | 34 (70.8) | 39 (81.3) | – | – |
| Consolidation | 13 (27.1) | 16 (33.3) | – | – |
| Linear fibrosis | 7 (14.6) | 7 (14.6) | – | – |
| Location of stenosis | ||||
| Trachea | – | 3 (6.2) | 3 (6.2) | – |
| Left main bronchus | – | 36 (75.0) | 36 (75.0) | – |
| Right main bronchus | – | 9 (18.8) | 9 (18.8) | – |
| Location of the lesion in the bronchus (N=45) | ||||
| Proximal bronchus | – | 4 (8.9) | 31 (68.9) | 1 (2.2) |
| Middle bronchus | – | 9 (20.0) | 9 (20.0) | 11 (24.4) |
| Distal bronchus | – | 5 (11.1) | 5 (11.1) | 2 (4.4) |
| Entire bronchus | – | 27 (60.0) | 0 (0.0) | 31 (68.9) |
| Type of lesion | ||||
| Airway fibrosis | – | – | 45 (93.8) | – |
| Bronchomalacia | – | – | 2 (4.2) | – |
| Airway distortion | – | – | 1 (2.1) | – |
| Length of the stenotic segment (mm) | – | 25.8±8.5 | – | 27.5±7.5 |
| Lumen diameter of the stenotic segment (mm) | – | 2.3±2.1 | 2.5±2.1 | – |
| Classification of stenosis* | ||||
| Moderate stenosis (51–70%) | – | 6 (12.5) | 8 (17.8) | – |
| Severe stenosis (71–99%) | – | 23 (47.9) | 22 (48.9) | – |
| Occlusion (100%) | – | 19 (39.6) | 15 (33.3) | – |
Statistics are presented as mean ± standard deviation and n (%). *N=45 for bronchoscopy (in 3 patients the lesion could not be evaluated by bronchoscopy). CT, computed tomography.
All patients underwent resection of the stenotic segment and anastomosis, with 27 patients (56.2%) undergoing lobectomy. Pathological findings showed several features suggestive of TB, including Langhans giant cells (45.8%) and caseous necrosis (14.6%). The most common anastomosis technique was lobar bronchus-to-carina anastomosis (33.3%), followed by lobar bronchus-to-main bronchus anastomosis (20.8%), end-to-end main bronchus anastomosis (16.7%), and main bronchus to carina anastomosis (16.7%). Three patients with tracheal stenosis underwent end-to-end anastomosis of the trachea. Five patients (10.4%) required tracheal mobilization, and 45 patients (93.8%) required hilum mobilization. The median surgical duration was 160 minutes, and the median blood loss was 100 mL. Thirteen patients (27.1%) had complications, the most common of which was prolonged air leakage (6 cases, 12.5%). Other complications, including atelectasis (4.2%), hoarseness (4.2%), and cardiovascular complications (4.2%), were less common. No anastomotic leak or dehiscence was observed. All patients with complications were treated conservatively and recovered within one month. Details of surgical techniques and complications are in Table 3.
Table 3. Operative characteristics and complications.
| Characteristics | All patients (N=48) |
|---|---|
| Surgical approach | |
| Resection and anastomosis | 21 (43.8) |
| Resection and anastomosis plus lobectomy | 27 (56.2) |
| Method of anastomosis | |
| End-to-end anastomosis of trachea | 3 (6.2) |
| End-to-end anastomosis of main bronchus | 8 (16.7) |
| Anastomosis of main bronchus to carina | 8 (16.7) |
| Anastomosis of main bronchus to trachea | 2 (4.2) |
| Anastomosis of lobar bronchus to main bronchus | 10 (20.8) |
| Anastomosis of lobar bronchus to carina | 16 (33.3) |
| Anastomosis of lobar bronchus to trachea | 1 (2.1) |
| Mobilization of the trachea | 5 (10.4) |
| Mobilization of the hilum | 45 (93.8) |
| Operating time (min) | 160 [144–180] |
| Blood loss (mL) | 100 [100–200] |
| Duration of chest drainage (days) | 4 [3–6] |
| Postoperative hospital length of stay (days) | 10 [8–13] |
| Pathological finding | |
| Granulomatous inflammation tissue | 33 (68.8) |
| Calcification | 25 (52.1) |
| Unspecified necrosis | 25 (52.1) |
| Langhans giant cells | 22 (45.8) |
| Fibrous tissue | 16 (33.3) |
| Caseous necrosis | 7 (14.6) |
| Operative complication | 13 (27.1) |
| Prolonged air leak | 6 (12.5) |
| Atelectasis | 2 (4.2) |
| Hoarseness | 2 (4.2) |
| Cardiovascular complications | 2 (4.2) |
| Pneumonia | 1 (2.1) |
| Hemoptysis | 1 (2.1) |
Statistics are presented as median [interquartile range] and n (%).
Table 4 shows that the patients’ condition improved significantly after surgery and was maintained for over 5 years. From 6 months onwards, no patients had dyspnea. Respiratory function also improved significantly at 1 year after surgery (Table 5). The proportion of patients with restrictive lung disease and obstructive lung disease also decreased significantly, and there were no patients with severe obstructive lung disease after surgery.
Table 4. Clinical parameters during the follow-up period.
| Parameter | Pre-surgery (N=48) | 1 month (N=48) | 3 months (N=48) | 6 months (N=48) | 1 year (N=48) | 2 years (N=48) | 3 years (N=48) | 4 years (N=48) | 5 years (N=44) | 6 years (N=30) | 7 years (N=9) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Karnofsky performance status score | |||||||||||
| 100 | 0 (0.0) | 12 (25.0) | 26 (54.2) | 36 (75.0) | 40 (83.3) | 42 (87.5) | 42 (87.5) | 46 (95.8) | 44 (100.0) | 30 (100.0) | 9 (100.0) |
| 90 | 2 (4.2) | 29 (60.4) | 18 (37.5) | 10 (20.8) | 7 (14.6) | 5 (10.4) | 6 (12.5) | 2 (4.2) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 80 | 16 (33.3) | 6 (12.5) | 3 (6.3) | 2 (4.2) | 1 (2.1) | 1 (2.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 70 | 28 (58.3) | 1 (2.1) | 1 (2.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 60 | 2 (4.2) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Severity of dyspnea | |||||||||||
| None | 28 (58.3) | 46 (95.8) | 47 (97.9) | 48 (100.0) | 48 (100.0) | 48 (100.0) | 48 (100.0) | 48 (100.0) | 44 (100.0) | 30 (100.0) | 9 (100.0) |
| Mild | 16 (33.3) | 2 (4.2) | 1 (2.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Moderate | 4 (8.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
Statistics are presented as n (%).
Table 5. Spirometry parameters before surgery and 1 year after surgery.
| Parameter | Pre-surgery (N=45) | Post-surgery (N=48) | P value |
|---|---|---|---|
| FVC (%) | 61.4±12.8 | 80.0±10.9 | <0.001 |
| FEV1 (%) | 54.4±11.5 | 73.4±12.1 | <0.001 |
| FEV1/FVC (%) | 77.4±14.8 | 78.4±7.9 | 0.98 |
| FEF 25–75 (%) | 42.8±14.0 | 57.0±18.1 | <0.001 |
| PEF (%) | 56.8±14.8 | 69.1±17.5 | <0.001 |
| Restrictive lung disease | 31 (68.9) | 18 (37.5) | 0.008 |
| Severity of restrictive disease | 31 | 18 | 0.01 |
| Mild | 10 (32.3) | 16 (88.9) | |
| Moderate | 21 (67.7) | 2 (11.1) | |
| Obstructive lung disease | 12 (26.7) | 5 (10.4) | 0.03 |
| Severity of obstructive disease | 12 | 5 | 0.76 |
| Mild | 4 (33.3) | 4 (80.0) | |
| Moderate | 5 (41.7) | 1 (20.0) | |
| Severe | 3 (25.0) | 0 (0.0) |
Statistics are presented as mean ± standard deviation and n (%). FVC, forced vital capacity; FEV1, forced expiratory volume in 1 second; FEF 25–75, forced expiratory flow between 25% and 75% of vital capacity; PEF, peak expiratory flow.
During the follow-up period, five patients with recurrent stenosis required treatment within the first year after surgery. Of these, three patients were successfully treated with balloon dilation. After reintervention, the patients only had mild stenosis with no subsequent pulmonary complications. The remaining two patients required pneumonectomy and were stable afterwards. The Kaplan-Meier estimate of freedom from recurrent stenosis requiring treatment from one year onwards was 90% (95% confidence interval: 81–99%). We did not find any statistically significant factors associated with recurrent stenosis that required treatment (Table S1).
Discussion
This study demonstrated the safety and effectiveness of tracheobronchial surgery for post-TB tracheobronchial stenosis. While an operative complication rate of 27.1% was observed, all complications resolved with medical management. The 5-year rate of freedom from restenosis was 90%. The performance status, dyspnea severity, and spirometry parameters showed significant improvements postoperatively.
Our study population’s characteristics align with previously reported findings (15-21): a predominance of young females and a higher prevalence of left bronchial involvement with TB. The higher frequency of tracheobronchial stenosis in young females with post-TB remains unclear. However, one hypothesis suggests that sociocultural expectations discouraging women from expectorating phlegm may contribute to prolonged exposure to TB bacilli in the bronchi. As for the greater prevalence of left bronchial involvement, anatomic compression by the aortic arch and the higher incidence of TB in the left-sided mediastinal and hilar lymph nodes are proposed contributing factors. A unique characteristic of our study population was the predominance of airway chronic fibrotic lesions with very narrow lumen diameter. This type of lesion limits the feasibility of airway interventions, potentially making surgery the only definitive treatment option. Additionally, surgery offers the advantage of addressing concomitant lung injuries through lobectomy, which was performed in 56.2% of our cases.
After resecting the airway lesions, the anastomosis technique is crucial to ensure that the anastomosis is not under tension and has an adequate blood supply. We used 3-0 PDS sutures with continuous suturing in the posterior half of the anastomosis. For the anterior half of the anastomosis, we used either a continuous or an interrupted suture technique. In all cases, the anastomosis was not under tension, and the technique was easy to perform. Similarly, a previous study showed that continuous suturing provided sufficient blood supply to the bronchial anastomosis for healing unless the bronchus was excessively mobilized (22).
Anastomotic stricture is a concerning issue that requires monitoring after tracheobronchial reconstruction, particularly for post-TB lesions. The causes can be poor suturing techniques, uneven diameters of the two ends, or dense suturing. Anastomotic inflammation can be caused by sutures and other stimuli, leading to granulation tissue hyperplasia or scarring. In this study, there were five cases of anastomotic stricture of more than 50% causing atelectasis that required intervention, all of which occurred within the first year. Of these, three cases were successfully dilated, and two cases could not be further reconstructed and required pneumonectomy. According to some reports, bronchoscopy can be considered to remove granulation tissue and treat anastomotic strictures with balloon dilation or stenting. However, all these methods have low efficacy. In contrast, incomplete resection of fibrotic lesions and inadequate use of anti-TB drugs after surgery can also lead to late restenosis (23,24). This study did not find any statistically significant factors associated with recurrent stenosis requiring intervention, which may be due to the small sample size and small number of events.
The main limitations of this study are the relatively small sample size and absence of a control group, attributable to the rarity of the disease, which restricts the generalizability of our findings. However, consistent treatment by an experienced team of physicians and the extended follow-up period lend strength to our conclusions regarding the safety and effectiveness of surgical reconstruction for post-TB tracheobronchial stenosis.
Conclusions
Our study demonstrates that tracheobronchial surgery is a safe and effective treatment for severe post-TB tracheobronchial stenosis. Both anastomosis technique and thorough patient follow-up play crucial roles in the success of surgical interventions. The potential for restenosis highlights the importance of careful vigilance and early management. To validate our findings further, future studies with larger sample sizes are warranted.
Supplementary
The article’s supplementary files as
Acknowledgments
Funding: None.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional ethics committee of Pham Ngoc Thach Hospital (No. 1595/PNT-HĐĐĐ, dated November 18, 2020) and individual consent for this retrospective analysis was waived.
Footnotes
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-230/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-230/dss
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-230/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-230/coif). The authors have no conflicts of interest to declare.
References
- 1.World Health Organization. Tuberculosis. 2024. Available online: https://www.who.int/news-room/fact-sheets/detail/tuberculosis, accessed 8 January 2024.
- 2.Shahzad T, Irfan M. Endobronchial tuberculosis-a review. J Thorac Dis 2016;8:3797-802. 10.21037/jtd.2016.12.73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chung HS, Lee JH. Bronchoscopic assessment of the evolution of endobronchial tuberculosis. Chest 2000;117:385-92. 10.1378/chest.117.2.385 [DOI] [PubMed] [Google Scholar]
- 4.Um SW, Yoon YS, Lee SM, et al. Predictors of persistent airway stenosis in patients with endobronchial tuberculosis. Int J Tuberc Lung Dis 2008;12:57-62. [PubMed] [Google Scholar]
- 5.Ozkaya S, Bilgin S, Findik S, et al. Endobronchial tuberculosis: histopathological subsets and microbiological results. Multidiscip Respir Med 2012;7:34. 10.1186/2049-6958-7-34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Su Z, Cheng Y, Wu Z, et al. Incidence and Predictors of Tracheobronchial Tuberculosis in Pulmonary Tuberculosis: A Multicentre, Large-Scale and Prospective Study in Southern China. Respiration 2019;97:153-9. 10.1159/000492335 [DOI] [PubMed] [Google Scholar]
- 7.Wei J, Qin S, Li W, et al. Analysis of clinical characteristics of 617 patients with benign airway stenosis. Front Med (Lausanne) 2023;10:1202309. 10.3389/fmed.2023.1202309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yaguchi D, Kimura H, Inoue N, et al. Tuberculous bronchial stenosis treated with balloon dilatation. QJM 2019;112:539-40. 10.1093/qjmed/hcy307 [DOI] [PubMed] [Google Scholar]
- 9.Xiong XF, Xu L, Fan LL, et al. Long-term follow-up of self-expandable metallic stents in benign tracheobronchial stenosis: a retrospective study. BMC Pulm Med 2019;19:33. 10.1186/s12890-019-0793-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Siow WT, Lee P. Tracheobronchial tuberculosis: a clinical review. J Thorac Dis 2017;9:E71-7. 10.21037/jtd.2017.01.49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nguyen Ho L, Tran Van N, Le Thuong V, et al. Hilar asymmetry in endobronchial tuberculosis patients: An often-overlooked clue. Int J Infect Dis 2019;80:80-3. 10.1016/j.ijid.2019.01.001 [DOI] [PubMed] [Google Scholar]
- 12.Moon SM, Lee WY, Shin B. Clinical characteristics and drug resistance profile of patients with endobronchial tuberculosis in South Korea: single-center experience. Ann Palliat Med 2023;12:487-95. 10.21037/apm-22-1218 [DOI] [PubMed] [Google Scholar]
- 13.Zhuang R, Yang M, Xu L, et al. Characteristics analysis of 157 cases of central airway stenosis due to tracheobronchial tuberculosis: A descriptive study. Front Public Health 2023;11:1115177. 10.3389/fpubh.2023.1115177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.World Health Organization. Tuberculosis in Viet Nam. 2024. Available online: https://www.who.int/vietnam/health-topics/tuberculosis, accessed 8 January 2024.
- 15.Cho YC, Kim JH, Park JH, et al. Tuberculous Tracheobronchial Strictures Treated with Balloon Dilation: A Single-Center Experience in 113 Patients during a 17-year Period. Radiology 2015;277:286-93. 10.1148/radiol.2015141534 [DOI] [PubMed] [Google Scholar]
- 16.Lee JH, Park SS, Lee DH, et al. Endobronchial tuberculosis. Clinical and bronchoscopic features in 121 cases. Chest 1992;102:990-4. 10.1378/chest.102.4.990 [DOI] [PubMed] [Google Scholar]
- 17.Lee KCH, Tan S, Goh JK, et al. Long-term outcomes of tracheobronchial stenosis due to tuberculosis (TSTB) in symptomatic patients: airway intervention vs. conservative management. J Thorac Dis 2020;12:3640-50. 10.21037/JTD-20-670 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lei Y, Tian-Hui Z, Ming H, et al. Analysis of the surgical treatment of endobronchial tuberculosis (EBTB). Surg Today 2014;44:1434-7. 10.1007/s00595-014-0865-9 [DOI] [PubMed] [Google Scholar]
- 19.Pathak V, Shepherd RW, Shojaee S. Tracheobronchial tuberculosis. J Thorac Dis 2016;8:3818-25. 10.21037/jtd.2016.12.75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ryu YJ, Kim H, Yu CM, et al. Use of silicone stents for the management of post-tuberculosis tracheobronchial stenosis. Eur Respir J 2006;28:1029-35. 10.1183/09031936.00020906 [DOI] [PubMed] [Google Scholar]
- 21.Vinh VH, Khoi NV, Quang NVD, et al. Surgical repair for post-tuberculosis tracheobronchial stenosis. Asian Cardiovasc Thorac Ann 2021;29:26-32. 10.1177/0218492320963972 [DOI] [PubMed] [Google Scholar]
- 22.Kutlu CA, Goldstraw P. Tracheobronchial sleeve resection with the use of a continuous anastomosis: results of one hundred consecutive cases. J Thorac Cardiovasc Surg 1999;117:1112-7. 10.1016/S0022-5223(99)70247-3 [DOI] [PubMed] [Google Scholar]
- 23.Li Z, Mao G, Gui Q, et al. Bronchoplasty for treating the whole lung atelectasis caused by endobronchial tuberculosis in main bronchus. J Thorac Dis 2018;10:4000-5. 10.21037/jtd.2018.06.126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tsukioka T, Takahama M, Nakajima R, et al. Surgical reconstruction for tuberculous airway stenosis: management for patients with concomitant tracheal malacia. Gen Thorac Cardiovasc Surg 2015;63:379-85. 10.1007/s11748-015-0536-9 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The article’s supplementary files as