Abstract
Purpose
Lipid nanoparticles (LNPs) show promise in their ability to introduce mRNA to drive protein expression in specific cell types of the mammalian eye. Here, we examined the ability of mRNA encapsulated in LNPs with two distinct formulations to drive gene expression in mouse and human retina and other ocular tissues.
Methods
We introduced mRNA-carrying LNPs into two biological systems. Intravitreal injections were tested to deliver LNPs into the mouse eye. Human retinal pigment epithelium (RPE) and retinal explants were used to assess mRNA expression in human tissue. We analyzed specificity of expression using histology, immunofluorescence, and imaging.
Results
In mice, mRNAs encoding GFP and ciliary neurotrophic factor (CNTF) were specifically expressed by Müller glia and RPE. Acute inflammatory changes measured by microglia distribution (Iba-1) or interleukin-6 (IL-6) expression were not observed 6 hours post-injection. Human RPE also expressed high levels of GFP. Human retinal explants expressed GFP in cells with apical and basal processes consistent with Müller glia and in perivascular cells consistent with macrophages.
Conclusions
We demonstrated the ability to reliably transfect subpopulations of retinal cells in mouse eye tissues in vivo and in human ocular tissues. Of significance, intravitreal injections were sufficient to transfect the RPE in mice. To our knowledge, we demonstrate delivery of mRNA using LNPs in human ocular tissues for the first time.
Translational Relevance
Ocular gene-replacement therapies using non-viral vector methods are a promising alternative to adeno-associated virus (AAV) vectors. Our studies show that mRNA LNP delivery can be used to transfect retinal cells in both mouse and human tissues without inducing significant inflammation. This methodology could be used to transfect retinal cell lines, tissue explants, mice, or potentially as gene-replacement therapy in a clinical setting in the future.
Keywords: lipid nanoparticle (LPN), mRNA, human retina, retinal pigment epithelium (RPE), mueller cell
Introduction
The retina plays an integral role in visual processing, and disorders of the retina are diverse in clinical presentation and etiology. Inherited retinal disorders (IRDs) are a heterogenous group of genetic disorders that may result in vision impairment and blindness. There are numerous types of IRDs, including retinitis pigmentosa (RP), choroideremia, Stargardt disease, cone-rod dystrophy (CRD), and Leber congenital amaurosis (LCA). To date, over 270 genes have been identified and associated with retinal disorders.1 Estimates of IRD prevalence vary but suggest that IRDs affect approximately 1 in 2000 individuals and over 5 million people worldwide, influencing quality of life and causing significant economic burden.2–4
Despite notable advances in diagnostic capabilities with next-generation sequencing (NGS) and long-read sequencing (LRS), therapeutic options for IRDs remain limited. IRDs caused by monogenic, loss-of-function mutations are particularly attractive candidates for gene replacement therapies. Ocular immune privilege has made viral-mediated gene therapy strategies feasible.5,6 Adeno-associated virus (AAV)-based gene augmentation therapies for IRDs have been most promising clinically, although lentivirus-mediated strategies have also been investigated.7,8 In 2017, the US Food and Drug Administration (FDA) approved voretigene neparvovec, an AAV2-based therapy to treat RPE65-mediated LCA and the first gene-replacement therapy for an IRD.9
There are limitations with the current viral-vector mediated therapies. Transgenes larger than 4.7 kb are challenging to deliver efficiently in AAV systems.10 There are also concerns that viral vectors can cause inappropriate and excessive immune system activation.11 Additional considerations must be made for delivery of the genetic material via intravitreal versus subretinal injections, with intravitreal injections generally considered safer because this method does not directly damage or detach the retina. However, subretinal injections provide more direct access to the subretinal space, and, therefore, the retinal pigment epithelium (RPE).12
Lipid nanoparticles (LNPs) are an alternative, non-viral based method of transfecting retinal tissue. DNA delivery using LNPs has had limited success in non-dividing cells, such as retinal cells.13 However, delivery of mRNA encapsulated in LNPs provides an alternative to AAV-based therapies that allows for delivery of larger transgenes in a system that has low immunogenicity. Two previous studies demonstrated the ability to deliver mRNA to the mouse retina with varying LNP formulations.14,15 Subretinal injections of LNPs containing mRNA transfected mouse retinal Müller glia, RPE, optic nerve head, and trabecular meshwork, in an apolipoprotein adsorption and phagocytosis-independent manner. Notably, intravitreal injections in these studies did not lead to LNP penetration and transfection of the RPE.14,15 Additionally, peptide guided LNPs have been used to deliver mRNA to photoreceptors, Müller glia, and RPE in rodents and nonhuman primate retinas.16
Due the unique combination of advancements in identifying genetic mutations that cause retinal degenerations, such as IRDs, ocular immune privilege, and the use of mRNA therapeutics, it is an opportune time to explore mRNA LNP gene augmentation therapies for the retina. In this study, we investigated the ability of specifically formulated mRNA and LNPs to transfect mouse and human retinal cells. The mRNA sequences were optimized with chemically modified uridine to limit activation of the innate immune response, as well as to maximize protein expression. LNPs were designed to bind to receptors that induce endocytosis of the mRNA; mRNA LNPs that are not released from endosomes fuse with lysosomes for degradation. One of the challenges about using LNP to deliver genomic material is the adverse immune response; the LNP formulations used in this study were shown to be cleared from the liver and spleen significantly faster and more effectively than older, legacy LNP formulations.17,18
We aimed to characterize the ability of these engineered mRNA LNPs to transfect retinal cells in both in vivo and ex vivo systems. Intravitreal injections of enhanced GFP (EGFP) mRNA LNPs into the mouse vitreous chamber resulted in EGFP expression in the Müller glia and RPE of the retina. Additionally, EGFP mRNA was expressed in human retinal and RPE cells. To our knowledge, this was the first study that demonstrated in vivo transfection of mouse RPE using intravitreal injections, which have the advantage of being less invasive and safer than subretinal injections. Furthermore, we examined markers of acute inflammation post-delivery of mRNA LNPs, showed delivery of a therapeutic agent ciliary neurotrophic factor (CNTF), and performed ex vivo transfections of human retina and RPE. The results from this study advance our understanding of potential applications for mRNA LNP technology in the rodent and human retina.
Methods
Intravitreal Injections
All protocols used in this study received approval prior to the start of experiments by the Institutional Animal Care and Use Committee of Seattle Children's Research Institute (Protocol 00050). Six- to 7-week-old male CD-1 mice (Charles River Laboratories, Wilmington, MA, USA) were injected intravitreally with mRNA LNPs. Mice were anesthetized in an isoflurane chamber. A sterile, 30G needle (BD Biosciences, Franklin Lakes, NJ, USA) was inserted at an approximately 45-degree angle along the limbus to create an initial puncture at the border of the cornea and sclera. A beveled, glass micropipette with a 5 µM tip (Clunbury Scientific, Bloomfield Hills, MI, USA) was filled with either 1X PBS, pH 7.4 or mRNA LNPs, with 0.1% Fast Green FCF dye (Sigma-Aldrich, St. Louis, MO, USA). A FemtoJet 4i electronic microinjection machine (Eppendorf, Hamburg, Germany) was used to deliver the reagents into the same initial puncture hole with the following parameters: injection pressure 290 hPa and compensation pressure 10 hPa. A maximum of 1 µL volume of mRNA LNP (1.8 mg/mL) was introduced into the vitreous cavity per injection. The amino lipid formulations used in these studies, 2T(Heptadecan-9-yl 8-((2-hydroxyethyl)(8-(nonyloxy)-8-oxooctyl)amino)octanoate) and 6T (2-(dinonylamino)-1-(4-(N-(2-(dinonylamino)ethyl)-N-nonylglycyl)piperazin-1-yl)ethan-1-one), were previously tested in rat, mouse, and nonhuman primate in vivo models and are approximately 80 to 100 nm in size.17,18
Histology of Mouse Retinas
Mouse eyes were enucleated, small holes were created in the corneas, and then they were fixed in 4% paraformaldehyde (PFA) in 1X PBS, pH 7.4 for 1 hour at room temperature while gently shaking. Eyes were rinsed in 1X PBS, then dissection was performed to remove any extraocular tissue, most of the cornea, and the lens. Eyecups were then processed and equilibrated in increasing concentrations of 5%, 15%, and 30% sucrose in 1X PBS and then in a well-mixed 1:1 solution of 30% sucrose/optimal cutting temperature compound (OCT; Sakura Finetek USA, Torrance, CA, USA) before freezing rapidly on dry ice. Frozen eyecups were sectioned to 40 µM thickness at −20°C on a Leica CM3050 S Cryostat (Leica, Wetzlar, Germany) onto TruBond 380 adhesive slides (Electron Microscopy Sciences, Hatfield, PA, USA), allowed to dry at room temperature for 1 hour, and then frozen with desiccant at −80°C.
Immunofluorescence of Mouse Retinas
Slides were washed once with 1X PBS, then incubated with blocking buffer (0.1% Triton X-100, 0.02% sodium dodecyl sulfate, and 1% bovine serum albumin in 1X PBS) for 30 minutes. The slides were incubated with DAPI (Invitrogen Cat# D1306) and primary antibodies for 4 hours: chicken anti-GFP (1:750, Thermo Fisher Scientific Cat# A10262, RRID:AB_2534023), rabbit anti-SOX9 (1:500, Millipore Cat# AB5535, RRID:AB_2239761), rabbit anti-RPE65 (1:500, Abcam Cat# ab231782), rabbit anti-CNTF (1:500, Proteintech Cat# 27342-1-AP, RRID:AB_2880848), rabbit anti-Iba1 (1:500, FUJIFILM Wako Shibayagi Cat# 019-19741, RRID:AB_839504), rabbit anti-interleukin-6 (1:500, Abcam Cat# ab7737, RRID:AB_306031), and Isolectin GS-IB4 from Griffonia simplicifolia, Alexa Fluor 647 Conjugate (1:500, Invitrogen Cat# I32450). Slides were washed with 1X PBS 3 times and incubated in fluorescently coupled Alexa Fluor secondary antibodies (Invitrogen) for 2 hours. Slides were washed 3 times in 1X PBS and mounted with Prolong Gold Antifade Mountant (Invitrogen #P36931). For flat mount immunostaining, mouse retinas were incubated in primary and secondary antibodies for 24 hours.
LNP Treatment of Cultured Human Retinal Pigment Epithelium
Developing human eye tissue was received from the University of Washington Birth Defects Research Laboratory (BDRL) with ethics board approval and maternal written consent was acquired before specimen collection in compliance with federal and state regulations. RPE from 115 day developing eyes was isolated according to established protocol19 and cultured in 5% v/v FBS in RPE MEM-alpha media with Rock inhibitor (10 µM) for 1 week and switched to 1% v/v FBS RPE media.20 The cells were cultured to confluence and passaged 4 times before plating to 8-chamber slides (Thermo Fisher Scientific Cat#177445) coated with Matrigel (Thermo Fisher Scientific Cat# CB-40230 or Corning Cat# 356230) at a cell density of 200,000 cells/well. These were grown for 5 weeks before starting the treatment. EGFP mRNA in 2T LNP was added at a ratio of either 10 µL to 190 µL 1% RPE culture media (0.1 mg/mL), 25 µL LNP to 175 µL media (0.25 mg/mL), or 50 µL LNP to 150 µL media (0.5 mg/mL). For controls, we used media only or 10 µL diluent or 50 µL diluent to media in the same ratio as LNP. Each control and LNP treatment were done in duplicate. The treatment was left on cells for 2 hours at 37°C and then removed and replaced with media only overnight at 37°C. The cells were then washed with 1X DPBS, fixed in 4% PFA with 0.01% Triton-X for 10 minutes, switched to 4% PFA with sucrose for 5 to 10 minutes, and then washed twice with 1X DPBS before immunofluorescence staining.
Immunofluorescence of Cultured Human RPE
The fixed RPE cells were incubated in blocking buffer (0.1% Triton X-100 with 0.02% SDS and 1% BSA in 1X PBS) for 1 hour at room temperature with mild shaking. Primary antibodies used were the following: anti-GFP (1:100, ThermoFisher Cat# A10262, RRID:AB_2534023) with anti-ZO-1 (1:100, Abcam Cat#ab221547, RRID:AB_2892660), and DAPI (1:500 of a 1:1000 solution, Invitrogen Cat# D1302). The cells were incubated in primary antibody solution with gentle shaking at 4°C, and then removed the next day with 3 washes of 1X PBS. Cells were incubated in secondary antibodies for 1 hour, which consisted of anti-chicken IgY (1:500, ThermoFisher Cat#A11039, RRID:AB_2534096) and anti-rabbit IgG (1:500, ThermoFisher Cat# A32795, RRID:AB_2762835).
LNP Treatment, Histology, and Immunofluorescence of Adult Human Globes
Postmortem adult human globes from a de-identified donor were obtained from Lions VisionGift (Portland, OR, USA). The tissue was provided with the donor's de-identified medical records including the following information: time and cause of death, postmortem interval prior to cryopreservation of tissue, age, perceived race, and sex. The donor did not have a medical history of ophthalmological conditions or interventions. Two globes from one donor were used for these experiments. Tissue biopsy punches (4 mm) were used to collect cross-sections of the retina, RPE, and sclera. Tissue explants were taken of the fovea/macula and at the optic nerve head, as well as approximately 15 additional punches of the peripheral retina. Explanted retina/RPE punches were incubated for 4 hours at 37°C in DMEM with diluent, EGFP mRNA encapsulated in 2TLNP, or EGFP mRNA encapsulated in 6T LNP to a final concentration of 0.1 mg/mL. Then, the DMEM/LNP mixture was removed and replaced with fresh DMEM for an additional 20 hours.
Tissue explants were washed 3 times with 1X PBS, then fixed with 4% PFA in 1X PBS for 1 hour at room temperature while gently shaking. Explants were placed in blocking solution (0.1% Triton X-100, 0.02% sodium dodecyl sulfate, and 1% bovine serum albumin in 1X PBS) for 1 hour. Retinal explants were incubated with DAPI (Invitrogen Cat# D1306) and primary antibodies for 3 days: chicken anti-GFP (1:750, Thermo Fisher Scientific Cat# A10262, RRID:AB_2534023), and rabbit anti-GFAP (1:500, Abcam Cat# ab7260, RRID:AB_305808). Slides were washed with 1X PBS 3 times and incubated in fluorescently coupled Alexa Fluor secondary antibodies (Invitrogen) for 3 hours. Tissue punches were washed 3 times in 1X PBS and mounted with Prolong Gold Antifade Mountant (Invitrogen #P36931).
Therapeutic Targets Review
We utilized RetNet to identify genes and loci implicated in IRDs.1 For our literature review of potential mRNA LNP therapeutic targets, we then examined gene expression in the peripheral retina from Supplementary Table S2 of a published single-cell human retina and organoid database.21 This atlas lists the level of gene expression (normalized to 10,000 transcript counts per cell type) in various retinal cell types. We cross referenced the two databases to rank disease genes by level of expression in Müller cells (MC1, MC2, and MC3) and RPE. Genes included are above gene expression level of 1. Supplemental information about diseases and affected proteins was drawn from the RetNet database. Additional information regarding gene reference and protein coding region size were drawn from the Ensembl genome browser.22 Candidate targets were filtered to remove artifacts of rod RNA contamination by removing those candidates whose expression was two times higher in rods than Müller glia or RPE.
Microscopy, Image Analysis, and Quantification
Images of retinal cryosections were obtained using sequential scanning protocols on a Leica TCS SP5 confocal microscope. Images were acquired using comparable acquisition settings per treatment group and adjusted to have minimal saturation of signal. Analysis and acquisition were not masked except for preliminary analyses and microglial counts. Maximum intensity projections (MIPs) and scale bars were created using ImageJ software (National Institutes of Health [NIH], Bethesda, MD, USA). Quantification of EGFP expression in RPE cells was performed using ImageJ software and corrected total cell fluorescence calculations were used as previously reported to adjust for area and background (https://theolb.readthedocs.io/en/latest/imaging/measuring-cell-fluorescence-using-imagej.html). For total fluorescence, the IntDen was measured across the whole image and corrected for area and background. For segmented analysis, single-channel images of ZO-1 (tight junction marker used to denote edges of RPE cells in these analyses) and EGFP were converted to gray scale first and then threshold adjusted (Otsu) to match signal intensity. The threshold image was converted to binary, and a ZO-1 mask was created. Then, the ZO-1 binary mask was subtracted from the EGFP binary file. The regions of interest (ROIs) were measured by adjusting the range of particle size to capture EGFP. Areas of no signal were used for generating the background with the manual draw tool and measured for area, minimum/maximum, and integrated density, similarly to the EGFP positive regions. Corrected total cell fluorescence was calculated with the following formula CTCF = Integrated Density - (Area of selected cell * Mean Fluorescence of background), using three areas of background. Images and quantifications were saved for statistical analysis.
Statistical Analysis
Statistical tests were performed using GraphPad Prism version 9 (GraphPad Software, San Diego, CA, USA). Nested t-tests were performed to compare all conditions, and significance was shown for EGFP expression in RPE cells treated with EGFP mRNA LNP versus media only control.
Results
Transfection of CD-1 Mouse Eyes With mRNA LNPs and Analysis of Cellular Expression
To assess the ability of mRNA encapsulated in LNPs to transfect retinal cells in an in vivo system, mRNA LNPs were delivered to CD-1 mice, chosen for the similarity of mouse retinal layers to the human retina. The basic experimental set-up was the following: intravitreal injection of mRNA LNPs were performed (one per eye) on anesthetized adult, CD-1 mice, animals were then allowed to recover for various time points between 48 hours and 2 weeks before harvest and subsequent analysis using immunofluorescence and confocal imaging (Fig. 1). First, to determine the ability to successfully deliver mRNA LNPs to the mouse eye, EGFP mRNA encapsulated in LNPs of two lipid formulations (2T and 6T) was injected intravitreally. Cellular uptake of the 2T formulation is low-density lipoprotein receptor (LDLR) mediated, whereas the uptake of 6T is LDLR-independent. Eyes were harvested at various time points to assess duration of the EGFP expression. Confocal microscopy images of retinal cross-sections were analyzed for EGFP expression. EGFP is expressed in multiple cell types in the retina at 48 hours (Fig. 2A), 72 hours (Fig. 2B), 96 hours (Fig. 2C), 1 week (Fig. 2D), and 2 weeks (Fig. 2E) post-injection with EGP mRNA for both 2T and 6T lipid formulations. Variable patterns of transfection were observed and are likely due to positional effects of injection. EGFP signal is specific to LNP GFP 2T and 6T retinas and was not observed in retinas injected with LNP cre-recombinase, one times PBS, or in uninjected retinas (Supplementary Fig. S1). Of note, expression of LNP delivered GFP does not cover the entire retina, which may result from the limited volume introduced (<1 uL). Additionally, outside of the retina, there was EGFP expression in the cornea, iris, and iridocorneal angle 48 hours post-injection with EGFP mRNA LNP, 2T formulation (Supplementary Fig. S2).
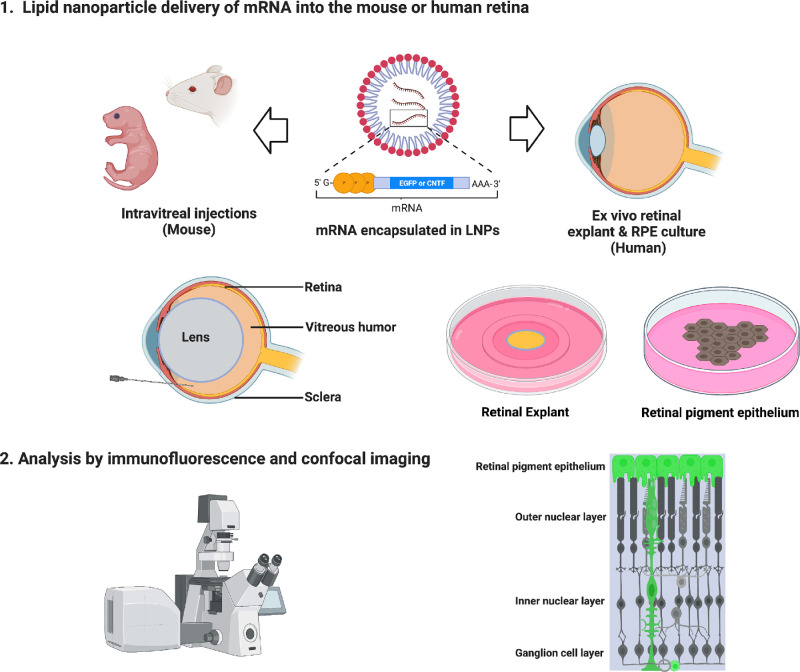
Figure 1.
Schematic of experimental workflow to deliver mRNA encapsulated in LNPs. Adult and neonatal CD-1 mice were used for in vivo experiments. Intravitreal injections of mRNA LNPs were performed to deliver the reagents to the vitreous chamber. Ex vivo experiments were performed using human retinal explants or human RPE cultures. After histological processing, analysis was completed using immunofluorescence and confocal imaging to determine transfection cell type expression and duration.
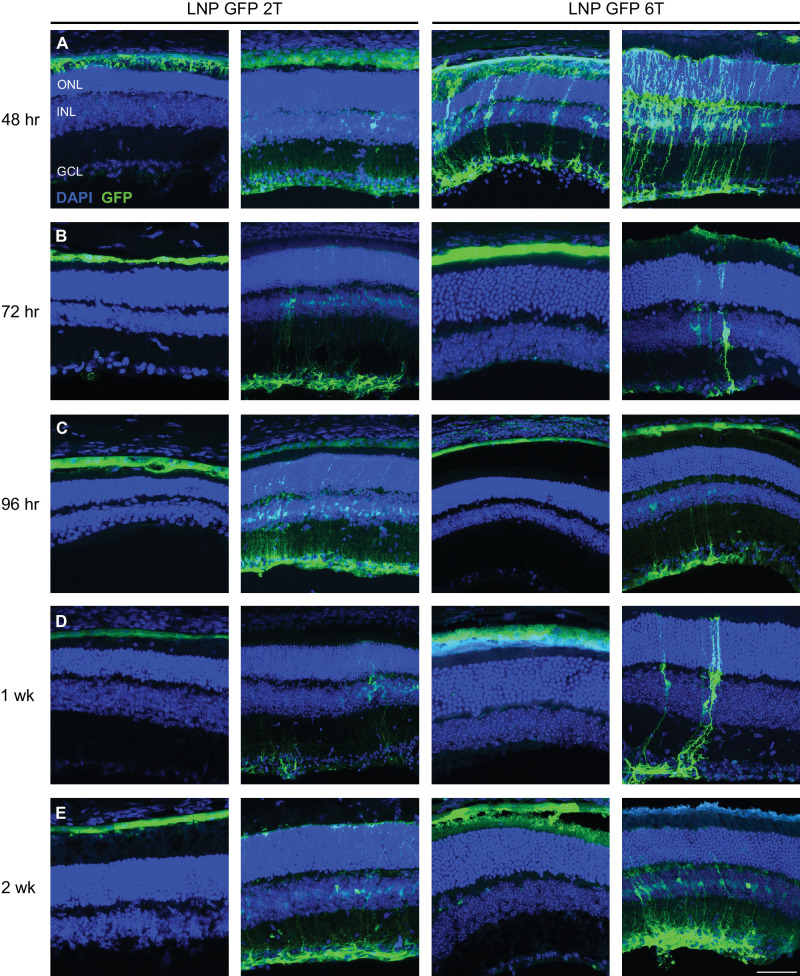
Figure 2.
Intravitreal injections of LNP-packaged EGFP mRNA transfects retinal cells in the mouse. Two amino lipid formulations, 2T and 6T, were compared for their ability to transfect retinal cells at various time points. Confocal images of retinal cross-sections with DAPI and anti-GFP immunostaining are shown. EGFP is expressed in similar cell populations at (A) 48 hours, (B) 72 hours, (C) 96 hours, (D) 1 week, and (E) 2 weeks for both 2T and 6T LNPs. Images shown are representative of at least three retinas (scale bar = 50 µm).
Next, to identify the specific retinal cell types transfected by EGFP mRNA LNPs, immunofluorescence using anti-GFP and co-staining with antibody markers of Müller glia (anti-SOX9) and RPE (anti-RPE65) was used in visualizing retinal cross-sections. Nuclei of Müller cells are stained with anti-SOX9, which overlapped with nuclei transfected with EGFP for both 2T and 6T at 48 hours post-injection (Figs. 3A, 3C). Similarly, RPE cells are stained with anti-RPE65, which overlapped with EGFP in the RPE of the retina for both lipid formulations (Figs. 3B, 3D). Thus, EGFP was expressed in Müller glia and RPE cells in the mouse retina. Interestingly, the transfection pattern in neonatal CD-1 mice was partially different than in adult mice. EGFP mRNA 2TLNPs were injected intravitreally to P0 CD-1 pups, and retinas were harvested 24 hours after injection. Retinal cross-sections show EGFP expression in perivascular cells near endothelial cells (co-stained with anti-Isolectin GS-B4) and RPE cells (Supplementary Fig. S3).
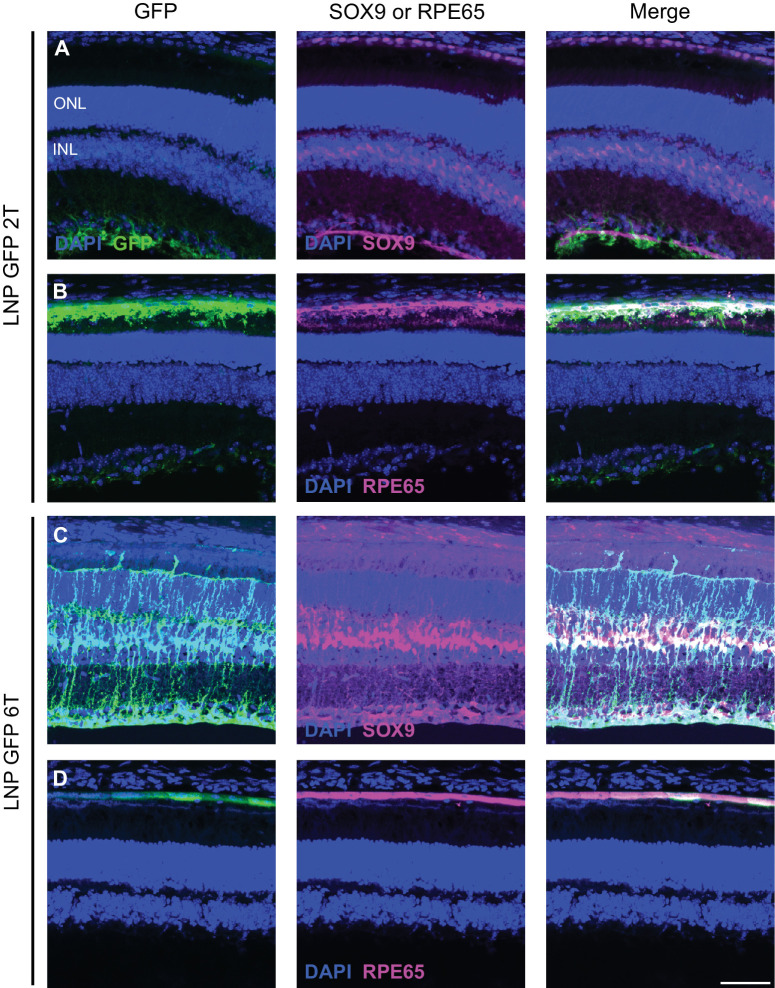
Figure 3.
LNP delivery of EGFP mRNA transfects Müller glia and RPE cells in the mouse retina. Retinal cross-sections were co-stained with DAPI, anti-GFP, and either anti-SOX9 or anti-RPE65 immunofluorescence 48 hours after intravitreal injections of EGFP mRNA encapsulated in 2Tor 6T LNPs. (A) For the 2T formulation, cells labelled with anti-GFP colocalize with nuclei labelled with anti-SOX9 in the inner nuclear layer, indicating EGFP expression in the Müller glia. (B) Cells that express EGFP also express RPE65, indicating EGFP expression in the RPE. Similarly, for 6T, there is co-expression of EGFP and (C) SOX9 and (D) RPE65. Images shown are representative of at least three retinas (scale bar = 50 µm).
Because Müller glia and RPE cells can be transfected with EGFP mRNA LNPs, delivery of a potential retinal therapeutic agent, CNTF, was assessed next. EGFP mRNA 2T LNPs were delivered either alone via intravitreal injection or in conjunction with CNTF mRNA 2T LNPs in adult CD-1 mice. When EGFP mRNA is delivered alone, there was EGFP transfection of Müller glia, and CNTF was diffusely present in the retina, with slightly higher expression in the ganglion cell layer (GCL; Fig. 4A). Injection of EGFP and CNTF mRNA resulted in expression of EGFP in the Müller glia and increased expression of CNTF in the Müller glia that was absent in the EGFP mRNA alone condition (Fig. 4B). This indicates that CNTF mRNA was successfully transfected into mouse retinal cells.
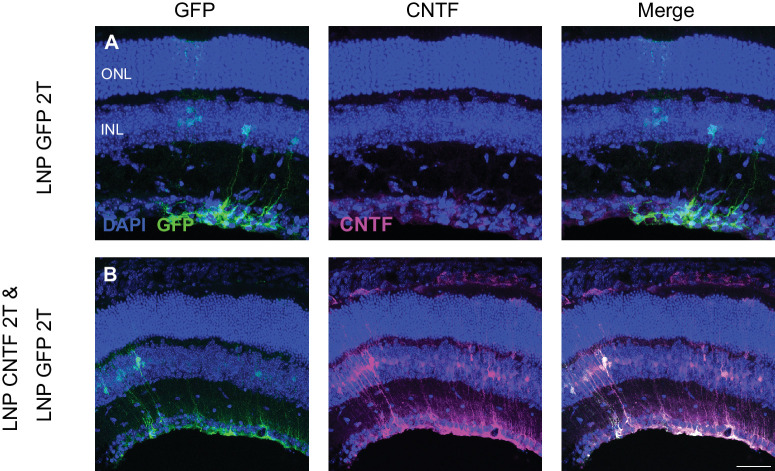
Figure 4.
CNTF mRNA in 2T LNP transfects mouse Müller glia cells. Intravitreal injections of EGFP or both EGFP and CNTF mRNA were done. (A) Control EGFP injections show EGFP expression in the Müller glia and basal CNTF expression in the GCL based on immunofluorescence staining. (B) When EGFP and CNTF mRNA were co-delivered, EGFP mRNA expression is in the Müller glia. CNTF expression is increased and in Müller glia cells as well. There is only partial overlap of EGFP and CNTF expression, indicating that there are populations of Müller glia cells that express either EGFP only, CNTF only, or both. Images shown are representative of at least three retinas (scale bar = 50 µm).
The Effect of mRNA LNP Delivery on Inflammation in the Mouse Retina
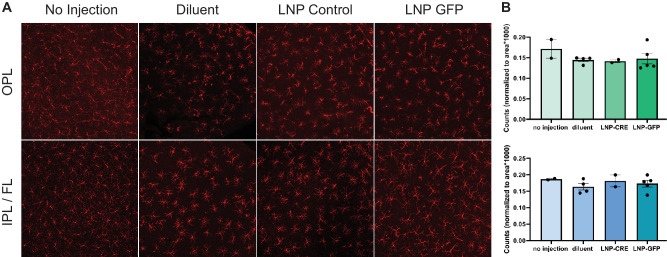
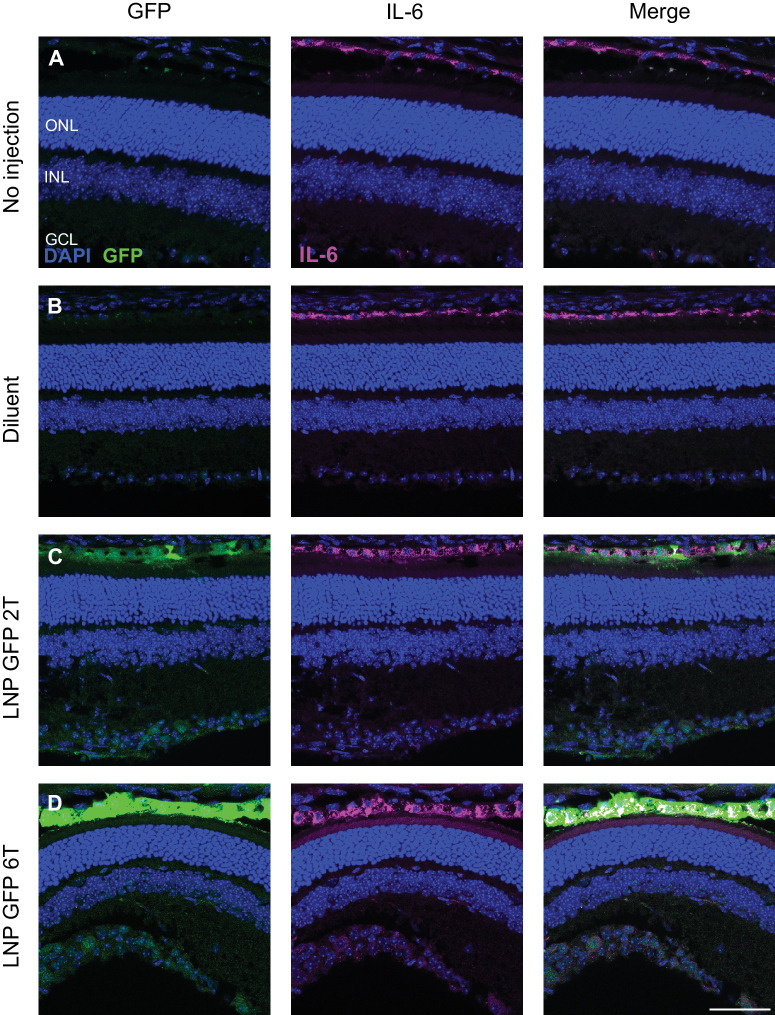
In vivo inflammation in response to intravitreal injections of mRNA LNPs was investigated in adult CD-1 mice. Observation of mice post-injection yielded no external signs of inflammation (i.e. no conjunctival redness or discharge). A more detailed analysis was performed to compare the numbers and behavior of retinal microglia (Iba1+ cells) and levels of cytokine interleukin-6 (IL-6) expression. Four conditions were compared for microglia: (1) no injection, (2) injection with 1X PBS diluent, (3) injection with a control 6T LNP with mRNA encoding the non-fluorescent protein cre-recombinase, or (4) injection with EGFP mRNA 6T LNP. Eyes were harvested 6 hours post-injection, and the distribution of microglia in the retinal layers was similar between all four conditions (Fig. 5). Four conditions were compared for the level of IL-6 expression: (1) no injection, (2) injection with 1X PBS diluent, (3) injection with EGFP mRNA 2T LNP, or (4) injection with EGFP mRNA 6T LNP. Eyes were harvested 6 hours post-injection, and the distribution of IL-6 expression was determined to be comparable across conditions by immunofluorescence (Fig. 6).
Figure 5.
Distribution of microglia in LNP EGFP injected and control retinas. (A) Microglial cells were visualized using anti-Iba1 immunostaining of flatmounted, uninjected mouse retinas and retinas injected with LNP GFP 6T, LNP CRE-recombinase (Control), or 1xPBS (Diluent) with Fast Green dye. Images represent maximum intensity projections of volumes acquired in the outer plexiform layer (OPL) or the inner plexiform layer and fiber layer (IPL/FL). (B) Quantification of Iba1+ microglial cells in the OPL or IPL/FL normalized to area (n > 2 retinas per condition). Each point on the bar graph represents a single retina where the number of microglia in two independent mid-peripheral regions were quantified and normalized by area. Error bars represent standard deviation.
Figure 6.
Intravitreal injections of EGFP mRNA LNPs does not affect the localization of cytokine IL-6. Expression of interleukin-6 in the RPE was similar comparing 4 conditions: (A) no injection, (B) PBS controls, (C) EGFP 2T LNP, or (D) EGFP 6T LNP at 6 hours post-intravitreal injection in mouse retinas. Images shown are representative of at least three retinas (scale bar = 50 µm).
Potential Therapeutic Targets to Treat Retinal Disorders Using mRNA LNPs
Based on these preliminary results of the ability to deliver mRNA and increase protein expression in Müller glia and RPE cells specifically, there is the opportunity to further investigate therapeutic agents for delivery using mRNA LNP technology in future studies. A literature review of protein-coding genes that are affected in Müller glia or RPE in inherited visual disorders found on RetNet are presented in Supplementary Tables S1 and S2. For example, replacement therapy of RLBP1 targeting Müller glia to treat recessive retinitis pigmentosa or TIMP3 in RPE cells to treat Sorsby fundus dystrophy could be explored further in future studies.
Studies of mRNA LNP Delivery and Transfection in Human Fetal RPE and Adult PostMortem Retina
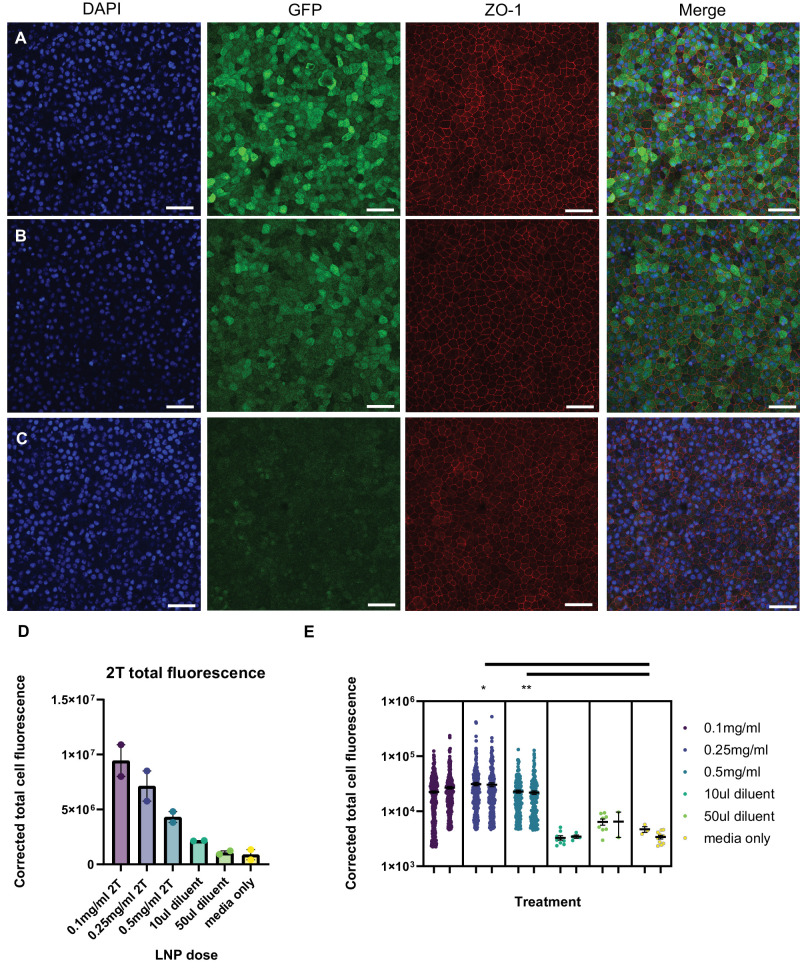
Additional studies were conducted to determine transfection efficacy and cell-type specificity of mRNA LNPs in human retinas: human fetal RPE (fRPE) tissue and adult human retina. Human fRPE cells were dissected, cultured for 6 weeks, then treated with 0.1 mg/mL, 0.25 mg/mL, or 0.5 mg/mL EGFP mRNA (2T LNP) for 2 hours, then washed with fresh media and incubated for 16 to 24 hours before confocal imaging. RPE cells that were treated with mRNA LNPs had higher expression of EGFP than those treated with diluent only (Figs. 7A–C). The corrected total cell fluorescence (CTCF) is shown in two ways: first, total fluorescence for the whole image, and second, segmented for fluorescence per cell to demonstrate the range in EGFP expression in different fRPE cells (Figs. 7D, 7E). There is a significant difference between the cells treated with 0.25 and 0.5 mg/mL doses compared to media only control (see Fig. 7E). The pattern of cytoplasmic EGFP expression is similar in fRPE cells treated with 0.1 mg/mL and 0.25 mg/mL mRNA 6T formulation of LNP (Supplementary Figs. S4A, S4B). There may be translocation of EGFP from the cytoplasm to the nucleus, as shown by the overlap of EGFP signal with DAPI; this is most prominent in the 0.5 mg/mL treatment group (Supplementary Fig. S4C).
Figure 7.
LNP delivery of EGFP mRNA transfects human fRPE in vitro. Human fRPE cells were treated with EGFP mRNA encapsulated in 2T LNPs for 2 hours prior to an overnight washout, followed by fixation and immunofluorescence for GFP and ZO-1. Cells were treated with (A) 0.1 mg/mL EGFP mRNA 2T-LNP, (B) 0.5 mg/mL mRNA 2T-LNP, or (C) diluent only (same volume as (A) Images shown are representative of two biological replicates of each treatment (scale bar = 50 µm). (D) Total GFP signal was quantified for fRPE treated with 0.1, 0.25, or 0.5 mg/mL LNP GFP and volume matched diluent or media controls (n = 2 biological replicates per condition). (E) Distribution of segmented GFP signal per cell for each of two biological replicates per treatment (* P < 0.05, ** P < 0.01; nested t-test).
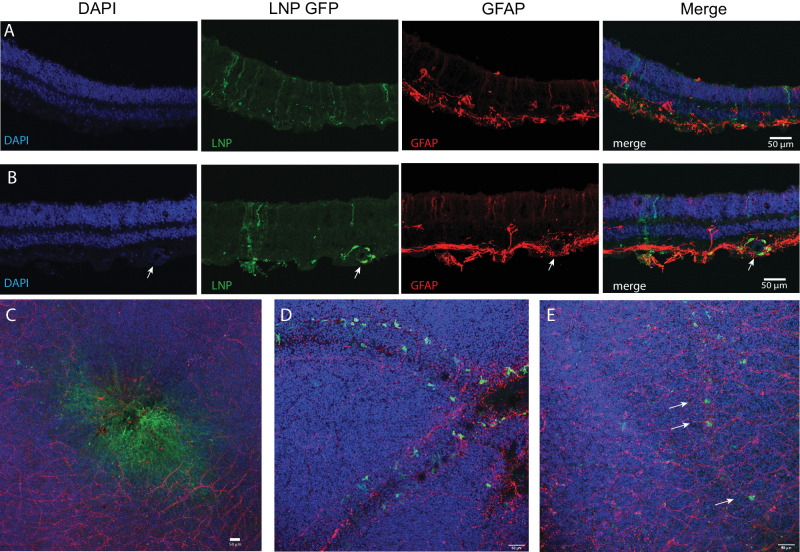
Retinal explants from postmortem adult globes were incubated with 0.1 mg/mL EGFP mRNA (2T or 6T LNP formulations) or diluent control for 4 hours, then placed in media before fixing, processing, and antibody staining for confocal imaging cross-sections and flat mounts. Human retinal cross-sections or flat mounts were stained with DAPI, anti-GFP, or anti-glial fibrillary acidic protein (GFAP). Retinal tissue incubated with EGFP mRNA in 6T LNPs express GFP in retinal cells that extend processes to the outer nuclear layer (ONL) and GCL (Fig. 8A), a similar cellular morphology to the Müller glia that express GFP in the CD-1 mouse retina experiments. Cells incubated with EGFP mRNA 6T LNPs also expressed GFP in the fovea (Fig. 8C) and in perivascular cells near blood vessels (Fig. 8E), as indicated with arrows. Human retinal explants treated with EGFP mRNA LNPs 2T yielded similar results: GFP was expressed in cells that extend apical and basal processes and surrounding blood vessels (Fig. 8B) and near blood vessels in the retina near the RPE/choroid (Fig. 8D).
Figure 8.
EGFP mRNA encapsulated in LNPs transfects adult human retina explants. Adult human post-mortem eye globes were treated with EGFP mRNA LNPs for 4 hours. (A) Confocal images of human retina cross-section treated with EGFP mRNA encapsulated in 6T LNPs and then stained with DAPI, anti-GFP, and anti-GFAP. GFP is expressed in cells with apical and basal processes. (B) Cross-section of human retina treated with EGFP mRNA encapsulated in 2T LNPs. GFP is expressed in perivascular cells surrounding a blood vessel, indicated by the arrow, and in cells with apical and basal processes. (C) Flat mount of parafoveal retina treated with EGFP mRNA in 6T LNPs. GFP is expressed in cells in the fovea. (D) Flat mount of retina with RPE and choroid treated with EGFP mRNA in 2T. Perivascular cells express GFP. (E) Flat mount of retina treated with 6T. Arrows indicate GFP positive cells along a small blood vessel. Images shown are representative of retinas and RPE from two globes from a single donor (scale bars = 50 µm).
Discussion
Gene therapies are a promising treatment for monogenic retinal disorders. IRDs can lead to retinal degeneration and eventual vision impairment or loss, which has a significant impact on quality of life. Due to the lack of the retina's ability to regenerate, earlier intervention and treatment is important to prevent irreversible damage. Currently, most potential treatments being investigated utilize AAVs or lentiviral vectors to deliver gene amplification. The only FDA-approved gene therapy for a visual disorder is voretigene neparvovec, an AAV2-based RPE65 replacement therapy for LCA.9 Although extremely promising, viral vectors have limited packaging capacity and have raised concerns about immunogenicity and genotoxicity, with limited ability to deliver larger transgenes or perform repeated injections.23 In this study, we tested a non-viral method of gene augmentation therapy in retinas: mRNA encapsulated in LNPs. We successfully delivered EGFP mRNA LNPs to mouse retinas in vivo using intravitreal injections and demonstrated the ability to transfect primarily RPE and Müller glia cells. The kinetics of EGFP expression was rapid, seen as early as 6 hours and lasting at least 2 weeks. EGFP was expressed in retinal cells with similar tropism and kinetics when using LNPs that were either ApoE- and LDLR-dependent (2T) or LDLR-independent (6T). Furthermore, mRNA LNPs were able to transfect mouse Müller glia cells with a potential therapeutic agent, CNTF, demonstrating the ability to deliver a protein besides EGFP.
LNP-encapsulated mRNA delivery has been studied in various organ systems, including in the retina, liver, kidneys, cerebral cortex, and red blood cells.14,15,24–29 Clinically, the first use of mRNA LNP technology were 2 mRNA severe acute respiratory syndrome-coronavirus 2 (SARS-CoV-2) vaccinations developed during the coronavirus disease 2019 (COVID-19() pandemic.30,31 In the retina specifically, previous studies examined the delivery of mCherry and luciferase mRNA LNP to the mouse retina, with expression in RPE and Müller glia cells over 4 to 96 hours post-subretinal injection.14,15 Our studies showed transfection in similar cell types in the mouse: RPE, Müller glia, Müller cell endfeet, and trabecular meshwork. There is variability in EGFP distribution in the retina, even at given time points. Future studies optimizing the injection method and volume could help standardize patterns and completeness of transduction. Notably, we demonstrated the ability to transfect retinal cells for a longer duration, at least 2 weeks, and additionally were able to transfect the RPE using intravitreal injections, as opposed to the more invasive subretinal injections. The amino lipid component of the LNP (LNP 2T or 6T) plays a role in cellular uptake of the mRNA LNP, and, thus, can affect how bioavailable the mRNA is for transfection. Further studies are required to determine what role the other LNP components (or mix of components), phospholipid, cholesterol, and pegylated lipid (PEG-lipid), play in the duration of EGFP expression delivered by these LNPs.
Our ability to target the RPE with intravitreal injections is valuable; subretinal injections are often preferred for targeting the subretinal space but come with significant risks and would not be preferred for repeated injections: retinal detachment, vitreous hemorrhage, and choroidal neovascularization.32 There are numerous RPE-specific deficiencies, such as TIMP3 in Sorsby fundus dystrophy or BEST1 that causes macular dystrophy that could benefit from gene replacement therapy targeting the RPE, such as the mRNA LNPs in this study. Future studies should additionally take into consideration whether the LNP formulation or placement of the injection cause temporary vision perturbation.
Our studies additionally reviewed the activation of the immune system to the mRNA LNP injections. We chose two immune markers to examine: IL-6, a cytokine that is upregulated and plays a key role during acute inflammation, and Iba1, a marker of microglia, immune surveillance cells in the retina.33,34 Our results at a 6-hour time point showed no difference in IL-6 levels or Iba1 distribution between the control groups with no injection and PBS only intravitreal injection compared with EGFP 6T or 2T LNP intravitreal injections. These preliminary immune studies suggest that there is no significant immune system activation or reaction to the mRNA LNPs in the mouse eyes 6 hours post-treatment. Further studies including more cytokines and components of the complement system could be done to examine acute and chronic innate and adaptive system involvement in response to mRNA LNP therapy.
To our knowledge, our studies are the first to effectively transfect human retina and RPE ex vivo using LNP-encapsulated mRNA. The ability to transfect human retinal and RPE cells expands our potential applications, both clinically and experimentally. LNPs could be used for targeted gene replacement therapy in the human RPE or retina. The safety profile of mRNA LNPs is more ideal than viral vectors because they are less immunogenic, are cleared from the body, and are not able to incorporate into the genome.17,35 There is overlap between the cell-type specificity of mRNA LNPs in the mouse and human retina: in both systems, RPE are transfected and cells that have processes that extend apically and basally express GFP, characteristic of Müller glia. Perivascular cells in the retina, possibly macrophages, express GFP in both adult postmortem globes and neonatal mouse retinas. Importantly, we also showed the ability to transfect cells in the human fovea, which is a structure absent in the mouse retina. Overall, these results in human retinal and RPE tissue demonstrate that LNPs can be used to reliably deliver mRNA to both mouse and human retinas.
Additionally, mRNA LNPs are an intriguing option for experimental applications, like for transient transfection of various cell lines. Human RPE and induced pluripotent- or embryonic stem cell-derived RPE are difficult to transfect using traditional plasmid DNA methods, such as FuGENE, at least partially due to low cell viability during these protocols.36–40 The mRNA LNPs used in these studies transfected cells with over 50% efficiency and caused minimal cell death. Therefore, this is a promising alternative for reliable and effective transfection of RPE and possibly other difficult to transfect cell lines.
LNP-encapsulated mRNA technology is exciting for a variety of future experimental and therapeutic applications. The ability to modify both mRNA and LNP to have less immunogenic effects, combined with its inability to incorporate into the genome and cause mutations, offers a safer alternative to viral vector-mediated gene therapies. Repeated intravitreal injections can be considered if longer durations of expression are needed for specific clinical applications. The ability to target the RPE with intravitreal injections makes this a unique opportunity to use gene therapy in the subretinal space without the need for subretinal injections. Additionally, the Müller glia are involved in retinal regeneration pathways, and therefore, mRNA LNPs in this study that target these cells could be used to deliver factors that could regenerate the retina.41,42 LNP formulations can also be adjusted to target different cell types in the retina or to potentially increase gene expression duration. Exciting experimental applications can be pursued as well, such as delivering Cas9 for CRISPR/Cas9 gene editing or Cre recombinase using mRNA LNPs in vitro or in vivo. These future studies will further our understanding of the effectiveness and safety of LNP-encapsulated mRNA in the eye and advance the field of retinal gene therapy.
Supplementary Material
Acknowledgments
The authors thank LuLu Callies and Stella Xu for their assistance with intravitreal injections and members of the Cherry Lab for their feedback during manuscript preparation.
Funded in part through a Sponsored Research Agreement (SRA) grant from Moderna, Inc. to T.J.C. and through discretionary funds provided to TJC by Seattle Children's Research Institute (SCRI). Funding was provided by NIH under NICHD Grant R24HD000836 to I.A.G.
Author Contributions: Experiments were designed, executed, and analyzed by C.Z.C., G.L.S., A.L.E., and T.J.C. with input from A.F. and P.G.V.M. on LNP and mRNA design and I.A.G. and BDRL on human tissue experiments. IAG, BDRL (University of Washington, Seattle, WA, USA), and Lions VisionGift (Portland, OR, USA) collected human samples. C.Z.C., A.L.E., and T.J.C. wrote the manuscript with revisions and feedback from all authors.
Disclosure: C.Z. Chambers, None; G.L. Soo, None; A.L. Engel, None; I.A. Glass, None; A. Frassetto, Moderna, Inc. (E, F, I); P.G.V. Martini, Moderna, Inc. (E, F, I); T.J. Cherry, None
References
- 1. Retinal Information Network (RetNet). Available at: https://web.sph.uth.edu/RetNet/. 2022.
- 2. Hanany M, Rivolta C, Sharon D, Daiger SP. Worldwide carrier frequency and genetic prevalence of autosomal recessive inherited retinal diseases, doi: 10.1073/pnas.1913179117/-/DCSupplemental. [DOI] [PMC free article] [PubMed]
- 3. Gong J, Cheung S, Fasso-Opie A, et al.. The impact of inherited retinal diseases in the US and Canada from a cost-of-illness perspective. Clin Ophthalmol. 2021; 15: 2855–2866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Fenner BJ, Tan TE, Barathi AV, et al.. Gene-based therapeutics for inherited retinal diseases. Front Genet. 2022; 12: 1–17. Preprint at 10.3389/fgene.2021.794805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zhou R, Caspi RR. Ocular immune privilege. F1000 Biol Rep. 2010; 2: 1–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Taylor AW. Ocular immune privilege. Eye. 2009; 23: 1885–1889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hashimoto T, Gibbs D, Lillo C, et al.. Lentiviral gene replacement therapy of retinas in a mouse model for Usher syndrome type 1B. Gene Ther. 2007; 14: 584–594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Do Rhee K, Nusinowitz S, Chao K, Yu F, Bok D, Yang XJ. CNTF-mediated protection of photoreceptors requires initial activation of the cytokine receptor gp130 in Müller glial cells. Proc Natl Acad Sci USA. 2013; 110: E4520–E4529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Russell S, Bennett J, Wellman JA, et al.. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet. 2017; 390: 849–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Colella P, Ronzitti G, Mingozzi F. Emerging issues in AAV-mediated in vivo gene therapy. Mol Ther Methods Clin Dev. 2018; 8: 87–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Mingozzi F, High KA. Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood. 2013; 122: 23–36. Preprint at 10.1182/blood-2013-01-306647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ross M, Ofri R. The future of retinal gene therapy: evolving from subretinal to intravitreal vector delivery. Neural Regen Res. 2021; 16: 1751–1759. Preprint at 10.4103/1673-5374.306063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zou S, Scarfo K, Nantz MH, Hecker JG. Lipid-mediated delivery of RNA is more efficient than delivery of DNA in non-dividing cells. Int J Pharm. 2010; 389: 232–243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Patel S, Ryals RC, Weller KK, Pennesi ME, Sahay G. Lipid nanoparticles for delivery of messenger RNA to the back of the eye. J Control Release. 2019; 303: 91–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ryals RC, et al.. The effects of PEGylation on LNP based mRNA delivery to the eye. PLoS One. 2020; 15: 1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Herrera-Barrera M, Ryals RC, Gautam M, et al.. Peptide-guided lipid nanoparticles deliver mRNA to the neural retina of rodents and nonhuman primates. Sci Adv. 2023; 9(2): eadd4623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Sabnis S, Kumarasinghe ES, Salerno T, et al.. A novel amino lipid series for mRNA delivery: improved endosomal escape and sustained pharmacology and safety in non-human primates. Mol Ther. 2018; 26: 1509–1519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Jiang L, Berraondo P, Jericó D, et al.. Systemic messenger RNA as an etiological treatment for acute intermittent porphyria. Nat Med. 2018; 24: 1899–1909. [DOI] [PubMed] [Google Scholar]
- 19. Sonoda S, Spee C, Barron E, Ryan SJ, Kannan R, Hinton DR. A protocol for the culture and differentiation of highly polarized human retinal pigment epithelial cells. Nat Protoc. 2009; 4: 662–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Du J, Yanagida A, Knight K, et al.. Reductive carboxylation is a major metabolic pathway in the retinal pigment epithelium. Proc Natl Acad Sci USA. 2016; 113: 14710–14715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Cowan CS, Renner M, De Gennaro M, et al.. Cell types of the human retina and its organoids at single-cell resolution. Cell. 2020; 182: 1623–1640.e34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ensembl. Available at: https://uswest.ensembl.org/index.html. 2022.
- 23. High KA, Roncarolo MG. Gene therapy. N Engl J Med. 2019; 381: 455–464. [DOI] [PubMed] [Google Scholar]
- 24. An D, Schneller JL, Frassetto A, et al.. Systemic messenger RNA therapy as a treatment for methylmalonic acidemia. Cell Rep. 2017; 21: 3548–3558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Chen CY, Tran DM, Cavedon A, et al.. Treatment of hemophilia A using factor VIII messenger RNA lipid nanoparticles. Mol Ther Nucleic Acids. 2020; 20: 534–544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lechner SM, Abbad L, Boedec E, et al.. IgA1 protease treatment reverses mesangial deposits and hematuria in a model of IgA nephropathy. J Am Soc Nephrol. 2016; 27: 2622–2629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Khoja S, Lliu XB, Truong B, et al.. Intermittent lipid nanoparticle mRNA administration prevents cortical dysmyelination associated with arginase deficiency. Mol Ther Nucleic Acids. 2022; 28: 859–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Balakrishnan B, An D, Nguyen V, DeAntonis C, Martini PGV, Lai K. Novel mRNA-based therapy reduces toxic galactose metabolites and overcomes galactose sensitivity in a mouse model of classic galactosemia. Mol Ther. 2020; 28: 304–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Truong B, Allegri G, Liu XB, et al.. Lipid nanoparticle-targeted mRNA therapy as a treatment for the inherited metabolic liver disorder arginase deficiency. Proc Natl Acad Sci USA. 2019; 116: 21150–21159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Baden LR, El Sahly HM, Essink B, et al.. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021; 384: 403–416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Polack FP, Thomas SJ, Kitchin N, et al.. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020; 383: 2603–2615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Peng Y, Tang L, Zhou Y. Subretinal injection: a review on the novel route of therapeutic delivery for vitreoretinal diseases. Ophthalmic Res. 2017; 58: 217–226. Preprint at 10.1159/000479157. [DOI] [PubMed] [Google Scholar]
- 33. Mesquida M, Leszczynska A, Llorenç V, Adán A. Interleukin-6 blockade in ocular inflammatory diseases. Clin Exp Immunol. 2014; 176: 301–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Li L, Eter N, Heiduschka P. The microglia in healthy and diseased retina. Exp Eye Res. 2015; 136: 116–130. [DOI] [PubMed] [Google Scholar]
- 35. Doktorovová S, Kovačević AB, Garcia ML, Souto EB. Preclinical safety of solid lipid nanoparticles and nanostructured lipid carriers: current evidence from in vitro and in vivo evaluation. Eur J Pharm Biopharm. 2016; 108: 235–252. [DOI] [PubMed] [Google Scholar]
- 36. Sunshine JC, Sunshine SB, Bhutto I, Handa JT, Green JJ. Poly(β-amino ester)-nanoparticle mediated transfection of retinal pigment epithelial cells in vitro and in vivo. PLoS One. 2012; 7: e37543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. del Pozo-Rodríguez A, Delgado D, Solinís MA, Gascón AR, Pedraz JL. Solid lipid nanoparticles for retinal gene therapy: transfection and intracellular trafficking in RPE cells. Int J Pharm. 2008; 360: 177–183. [DOI] [PubMed] [Google Scholar]
- 38. Hansson ML, Albert S, González Somermeyer L, et al.. Efficient delivery and functional expression of transfected modified mRNA in human embryonic stem cell-derived retinal pigmented epithelial cells. J Biol Chem. 2015; 290: 5661–5672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Trigueros S, Domènech EB, Toulis V, Marfany G. In vitro gene delivery in retinal pigment epithelium cells by plasmid DNA-wrapped gold nanoparticles. Genes (Basel). 2019; 10: 289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Li HL, Zheng XZ, Wang HP, Li F, Wu Y, Du LF. Ultrasound-targeted microbubble destruction enhances AAV-mediated gene transfection in human RPE cells in vitro and rat retina in vivo. Gene Ther. 2009; 16: 1146–1153. [DOI] [PubMed] [Google Scholar]
- 41. Goldman D. Müller glial cell reprogramming and retina regeneration. Nat Rev Neurosci. 2014; 15: 431–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ahmad I, del Debbio CB, Das AV, Parameswaran S. Müller glia: a promising target for therapeutic regeneration. Invest Ophthalmol Vis Sci. 2011; 52: 5758–5764. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.