Abstract
Purpose of Review
Despite the availability of effective vaccines against the three primary pathogens (Streptococcus pneumoniae, Haemophilus influenzae type b, and Neisseria meningitidis) that cause bacterial meningitis, this condition remains a significant cause of morbidity, neurologic sequelae, and mortality among children and adults living in low-income and middle-income countries.
Recent Findings
Bacterial meningitis represents a significant public health challenge for national and global health systems. Since vaccine-preventable meningitis remains highly prevalent in low-income and middle-income countries, the World Health Organization (WHO) recently developed a global roadmap to defeating meningitis by 2030 and ameliorating its associated neurological sequelae.
Summary
There is a need for a global approach to surveillance and prevention of bacterial meningitis. Increasing vaccination coverage with conjugate vaccines against pneumococcus and meningococcus with optimal immunization schedules are high-value healthcare interventions. Additionally, overcoming diagnostic challenges and the early institution of empirical antibiotic therapy and, when feasible, adjunctive steroid therapy constitutes the pillars of reducing the disease burden of bacterial meningitis in resource-limited settings.
Keywords: Bacterial meningitis, Children, Adults, Haemophilus influenzae b, Streptococcus pneumoniae, Neisseria meningitidis, Low- and middle-income countries
Introduction
Bacterial meningitis (BM) is a significant global public health problem [1••, 2]. BM ranks among the top 10 causes of death in high-income countries for children younger than 14 [1••, 2]. The incidence of BM is higher in low-income and middle-income countries, causing a substantial disease burden in terms of premature death and disability [1••, 3, 4••]. In this condition, bacterial pathogens colonizing the nasopharynx or entering the bloodstream through the lower airways reach the central nervous system by crossing the blood–brain barrier, producing a life-threatening condition with case fatality varying from 20 to 50% [3, 4••, 5•]. Death occurs due to multiorgan failure resulting from bacteremia, sepsis, or brain herniation secondary to increased intracranial hypertension due to cytotoxic cerebral edema or intracranial bleeding caused by inflammatory angiitis [5•]. Subdural effusion and focal neurological deficits may occur in the short term [5•, 6]. However, lifelong disabilities manifesting as hearing loss, cognitive impairment, seizures, and hydrocephalus may appear in more than 20% of those who survive the acute process [7•, 8]. Neurological sequelae are higher in pediatric populations living in low-income and lower-middle-income settings (low-income economies are defined as those with a GNI per capita of US$1135 or less and middle-income countries those with a GNI per capita of $1136 to $4465) [2, 3, 4••, 5•, 9].
The introduction of highly effective conjugate vaccines to prevent infection and invasive disease (bacteremia and meningitis) by the three major bacterial pathogens—Streptococcus pneumoniae, Haemophilus influenzae type b, and Neisseria meningitidis—has been broadly deployed in high-income countries and have reduced the incidence of BM in these settings [10–12]. For example, in Europe, the Netherlands, and England, the introduction and broad deployment of conjugate vaccines and adjunctive therapy with dexamethasone have reduced the incidence of bacterial meningitis and improved clinical outcomes [13, 14]. However, limited financial resources and health expenditures have limited the introduction of conjugate vaccines in low-income and middle-income settings—where BM’s most significant disease burden exists [2, 3, 4••]. In response to these prevailing health inequalities, in November 2020, the World Health Assembly (WHA) approved an initiative that calls for nation-states and key international stakeholders to reduce the incidence and deaths caused by vaccine-preventable meningitis and eliminate epidemics of meningitis [2]. The World Health Organization (WHO) roadmap also focuses on reducing disability and sequelae and improving the quality of life of those affected with bacterial meningitis by 2030. There are five essential pillars to achieve these goals, including prevention and epidemic control, diagnosis and treatment, care and support of people affected with meningitis, advocacy, and disease surveillance [2, 3]. In this review, we summarize the epidemiology and burden of disease caused by BM in low-income and middle-income countries. We also present an overview of potential interventions needed to reduce the impact of BM in these settings.
Epidemiology and Burden of Disease of Bacterial Meningitis in Low-Income and Middle-Income Countries
Worldwide, most of the burden of ABM is caused by three major bacterial pathogens: Streptococcus pneumoniae, Neisseria meningitidis, and Haemophilus influenzae type b [2, 4••]. There are other important bacterial pathogens to consider in the differential diagnosis of BM according to age group [7•, 8, 9, 12]. For example, in neonates, enteric gram-negative bacilli such as Escherichia coli or Listeria monocytogenes, and Streptococcus agalactiae (group B Streptococci or GBS) are frequent pathogens causing neonatal sepsis. Listeria monocytogenes, Streptococcus pneumoniae, and H. influenzae are essential to consider among older adults [4••, 7•, 8].
BM occurs in all settings. However, like many other infectious diseases, socio-economic factors influence BM’s incidence and clinical outcomes [1••, 2]. Recent estimates demonstrate that the incidence of BM varies from 0.9 per 100,000 individuals per year in high-income countries to 80 per 100,000 individuals per year in low-income settings [4••]. Most of the disease burden is concentrated in resource-constrained countries, including parts of sub-Saharan Africa, South Asia, Central Asia, East Asia and the Pacific, and Latin America (Fig. 1) [1••, 2, 3, 4••]. Approximately 500 million people worldwide are carriers of N. meningitidis in their nasopharynx, but only some develop meningitis or disseminated meningococcal infection [15]. Worldwide, the burden of BM is most significant in the meningitis belt of sub-Saharan Africa, which extends from Senegal in West Africa to Ethiopia and Somalia in the Horn of Africa [9, 12]. In this African region, epidemics of Neisseria meningitidis serogroups A, W, X, and C are highly prevalent [5•]. In the twentieth century, N. meningitidis serogroup A was responsible for up to 80% of cases of meningococcal meningitis, where significant epidemics of meningococcal meningitis were described to occur every 5 to 12 years, with attack rates sometimes reaching more than 1000 cases per 100,000 population [9]. A strong seasonal pattern of meningococcal meningitis coincides with the dry season from January to April. It is believed that the dust particles carried by the dry winds of the Harmattan in Western Africa irritate the nasopharyngeal mucous membranes, facilitating the entry of N. meningitidis in populations with a high carriage rate of meningococcus and other pathogens [12]. However, since 2000 and the deployment of the serogroup A vaccine, other serogroups W, X, and C, and Streptococcus pneumoniae are now responsible for most cases, leading to new epidemic waves with high lethality rates [9, 12]. More importantly, many meningitis epidemics have been described outside the meningitis belt, reaching central parts of the continent. Since 2014, significant outbreaks of BM have been reported in many settings, including Chile, Nigeria, Chile, Niger, Fiji, and Kyrgyzstan [2].
Fig. 1.
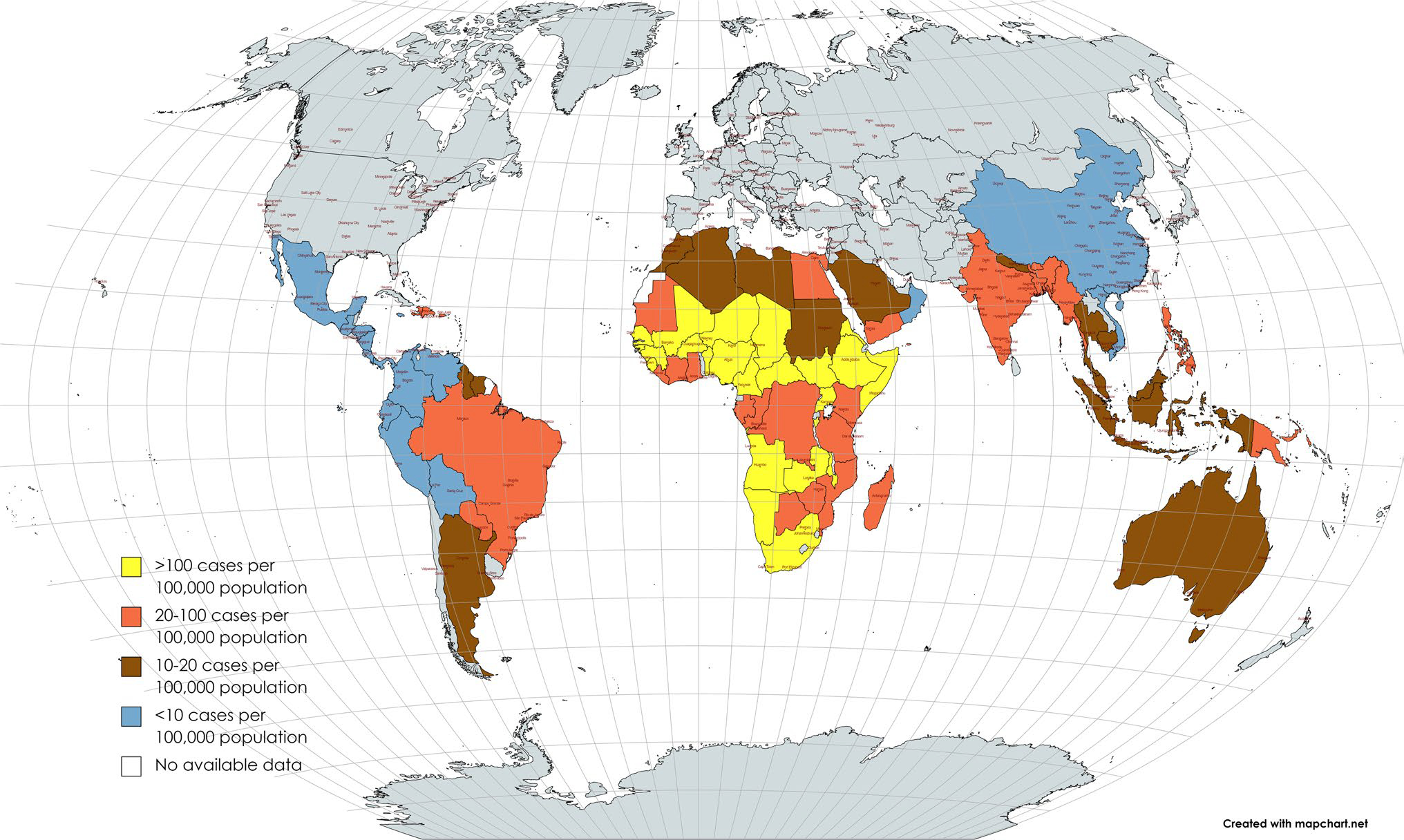
Incidence rates of bacterial meningitis in the tropics and subtropics *Figure adapted and modified from 2. Source: WHO GIS Centre for Health. **Most low-income and lower-middle-income countries are geographically located in the tropics (between the Tropic of Capricorn [23°27′ degrees south of the equator] and the Tropic of Cancer [23°27’ degrees north of the equator]) and the subtropics
WHO coordinates the Global Invasive Bacterial Vaccine-Preventable Diseases Surveillance Network (IB-VPD) to support conjugate vaccines against S. pneumoniae, N. meningitidis, and H. influenzae introduction decisions and implementation [16••, 17••, 18••, 19]. In this network, sentinel hospitals in highly affected countries report cases of children under 5 years of age hospitalized for suspected bacterial meningitis. In 2019, this network included 123 laboratories that tested and validated specimens, reporting confirmatory testing results and strain characterization by polymerase chain reaction. From 2014 through 2019, more than 137,000 cases were reported to WHO by 58 countries [2, 16••]. As part of this surveillance network, most countries that reported cases of BM were from the African region, and most episodes were caused by S. pneumoniae (61%), H. influenzae b (18%), and N. meningitidis (21.4%). Of all cases of BM, 11% died, and S. pneumoniae caused the most. Importantly, data from this network demonstrated a strong correlation between the introduction of pneumococcal conjugate vaccine and a reduction in the proportion of cases of BM caused by pneumococcal serotypes contained in the vaccine [16••, 17••]. Nonetheless, infection caused by S. pneumoniae remained Africa’s most common cause of pediatric bacterial meningitis [19].
Before the widespread use of conjugate vaccines against H. influenzae b, this organism was the most common cause of severe invasive infections in children, causing approximately 300,000 to 500,000 deaths yearly [2, 3, 15]. Fortunately, cases of H. influenzae b have declined substantially in countries that have introduced the H. influenzae b conjugate vaccine [10]. Nonetheless, reports from the IV-BPD WHO surveillance network have also shown that the incidence of Hib meningitis cases has declined after introducing the Hib vaccine in many settings, but mostly in high-income countries [16••]. However, H. influenzae remains a leading cause of bacterial meningitis in low- and middle-income countries, highlighting the need to improve vaccination coverage against this pathogen through routine immunization activities conducted by national immunization programs and supplementary immunization activities (catch-up strategies) [20–25].
Diagnostic and Treatment Challenges Associated with Suspected Cases of BM in Low-And-Middle-Income Countries
The initial clinical workup and prompt initiation of empiric antimicrobial therapy for a child, adolescent, or adult with suspected BM depends on early recognition of clinical signs of meningitis and obtaining appropriate diagnostic evaluation [8]. However, there are significant limitations in diagnosing BM in resource-limited settings [4••]. The classical triad of fever, stiff neck, and a change in mental status occurs in less than 50% of cases [4••, 5•]. The most common presenting symptoms include headache, fever, stiff neck, nausea, and altered mental status (median Glasgow Coma Scale of 11) [4••]. The diagnostic accuracy of classic meningeal signs in adults with suspected bacterial meningitis is suboptimal. The inaccuracy of clinical symptoms of meningitis syndrome highlights the need for an ancillary laboratory workup to aid in the diagnostic confirmation. Once a case of BM is suspected, the standard of care is obtaining blood cultures; cranial computed tomography (CT scan), when indicated; and lumbar puncture to get cerebrospinal fluid (CSF) for gram stain examination, and culture, cell counts, and biochemical assessment are considered the standard of care. The culture of CSF is positive in more than 85% of cases; therefore, it is regarded as the most critical test to obtain when feasible. Gram staining of the CSF is a quicker test with high specificity. Bacterial antigen testing CSF can assist in the diagnosis, but it does not replace the role of gram staining or culture. Studies conducted in low-income countries with a high burden of N. meningitidis meningococcal antigens in CSF have shown high sensitivity and negative predictive value [4••]. Despite all these recommendations, the challenges imposed by performing lumbar punctures to obtain CSF and the limited laboratory capacity at the subnational and district level of many low-income settings, patients are treated with empiric antimicrobial regimens without laboratory confirmation.
While there is WHO guidance on managing BM in the pediatric age, no WHO guidelines exist for treating adults with BM [2]. Optimally, the management algorithms for BM in all age groups include initiating empirical antimicrobial therapy and dexamethasone [10–12]. The choice of empiric antibiotic therapy is influenced by the epidemiology of the most frequently encountered bacterial pathogens (Tables 1 and 2) [4••, 7•, 8]. Dexamethasone reduces CSF inflammation and can improve BM clinical outcomes [2, 4••, 7•, 8]. Using steroids in the pediatric age groups has reduced long-term sequelae (i.e., hearing loss) [5•, 6, 8]. In adults, there is also evidence that dexamethasone improves clinical outcomes in adult populations [4••, 7•, 8].
Table 1.
Differential diagnosis of suspected bacterial meningitis in low-and middle-income countries
| Condition | Etiologic agent | Diagnostic considerations in CSF |
|---|---|---|
|
| ||
| Viral encephalitis | Herpesviruses: HSV-1, HSV-2, VZV, HHV6 Mosquito-borne Flavivirus: West Nile, Virus, Zika, Dengue, Murray Vile, St. Louis Tick-borne Flavivirus: Powassan, Tick-borne virus encephalitis Togavirus: Western Equine, Eastern Equine, and Venezuelan Equine |
Moderate lymphocytic pleocytosis with elevated red blood cells and high protein level Elevated red blood cells in cases of herpes encephalitis |
| Viral meningitis | Non-polio enteroviruses (e.g., EV 71, EV D68) Arboviral (West Nile, Virus, Zika, Dengue, Chikungunya) Toscana virusesa Herpesviruses: HSV-1, HSV-2, VZV, EBV, HHV6, CMV HIV Mumps virus LCMV |
Non-polio enteroviruses are frequent during the summer and fall seasons Mild to moderate degree of lymphocytic pleocytosis CSF glucose: In most cases, the glucose level is average. However, it may be low in cases of mumps Acute HIV infection produces a mononucleosis like illness associated with meningitis LCMV is associated with substantial leukocyte elevation in CSF and hypoglycorrachia |
| Tuberculous meningitis | Mycobacterium tuberculosis complex | Moderate to substantial lymphocytic pleocytosis and elevated protein levels Frequent cause of angiitis of Circle of Willis. It is important to distinguish TB meningitis from subarachnoid neurocysticercosis in settings with high rates of cysticercosis |
| Fungal meningitis |
Cryptococcus neoformans
Cryptococcus gattii Coccidioides immitis Fusarium solani c Histoplasma capsulatum |
Moderate to substantial lymphocytic pleocytosis and hypoglycorrachia (mimicking tuberculous meningitis or neurosarcoidosis) |
| Parasitic meningitis (meningoencephalitis) |
Gnathostoma spirigerum
Angiostrongyloides cantonensis Baylisascaris procionis (exposure to infected feces of racoons) Trypanosoma cruzi d Trypanosoma brucei rhodesiense Trypanosoma brucei gambiense Toxoplasma gondii |
Eosinophilic meningitis defined as having more than 10 eosinophils/mm and/or more than 10% of CSF leukocytes being eosinophils |
| Spirochetal meningitis |
Borrerlia burgdorferi Treponema pallidum Leptospira spp. |
Mononuclear pleocytosis > 70% in cases of Lyme disease. Many patients have a history of erythema migrans Neurosyphilis is associated with mild protein elevation and mild mononuclear pleocytosis |
| Drug-induced meningitis | NSAIDS Trimethoprim/sulfamethoxazole Intravenous immune globulin (IVIG)b |
Mimics viral meningitis but occasionally, it may be associated with purulent CSF |
| Autoimmune and paraneoplastic encephalitis | Limbic encephalitis (small cell lung cancer or testicular tumors) Brainstem encephalitis Myelitis Encephalomyelitis |
Anti-NMDA receptor antibodies Anti-MA2 (Anti-TA) antibodies Anti-HU |
| Neoplastic invasion of the meninges | Large cell-lymphomas Acute leukemia Solid organ neoplasms (breast, melanoma, lung, and gastrointestinal malignancies) |
Most cases of neoplastic invasion of the meninges cause mild, to moderate lymphocytic pleocytosis with hypoglycorrachia Eosinophilia in CSF is associated with Hodgkin’s disease |
Human African trypanosomiasis (HAT) or sleeping sickness is caused by either T. brucei rhodesiense or T. brucei gambiense. HAT is endemic in 36 countries in Africa. Infection with T. brucei rhodesiense progresses rapidly within weeks, while gambiense HAT is usually slowly progressive
NSAIDS non-steroidal anti-inflammatory drugs
Toscana virus is transmitted by sandflies in the Mediterranean during the summer and fall seasons
These are the most common drugs associated with meningitis. However, there are reports of many other drugs causing meningeal inflammation
Outbreak associated with epidural anesthesia for cosmetic procedures in the USA-Mexico border
Chagas disease or American trypanosomiasis may cause meningoencephalitis in patients with acute infection or from reactivation of latent infection in patients with advanced immunosuppression caused by HIV/AIDS
Table 2.
Empirical antimicrobial therapy for suspected bacterial meningitis by age group
| Age-group | Most frequently isolated bacterial pathogens | Empiric antimicrobial therapy* |
|---|---|---|
|
| ||
| Neonates |
Listeria monocytogenes Escherichia coli (or other enteric-gram negative bacilli) Streptococcus agalactiae |
Ampicillin plus Cefotaxime or Gentamicin |
| 1–24 months |
Streptococcus agalactiae Escherichia coli Streptococcus pneumoniae Neisseria meningitidis** Haemophilus influenzae b** |
Vancomycin + cefotaxime or ceftriaxone |
| 2–50 years |
Streptococcus agalactiae Escherichia coli Streptococcus pneumoniae Haemophilus influenzae b** |
Vancomycin + cefotaxime or ceftriaxone + ampicillin*** |
| 50 years and older |
Streptococcus pneumoniae
Neisseria meningitidis Listeria monocytogenes |
Vancomycin + ceftriaxone or cefotaxime + ampicillin*** |
The intravenous route is the preferred mode of administration of antimicrobials
Ceftriaxone is preferred over chloramphenicol for treating H. influenza b meningitis, given the increasing antimicrobial resistance of this pathogen to chloramphenicol. The administration of one or two doses of long-acting chloramphenicol is recommended during outbreaks of N. meningitidis; alternatively, intramuscular administration of ceftriaxone is considered an alternative
Trimethoprim/sulfamethoxazole is recommended for cases of severe penicillin-allergy
It is also important to note that in many settings, the emergence of antimicrobial resistance, particularly of ceftriaxone-resistant S. pneumoniae (defined as minimum inhibitory concentration ≥ 2 mg/L), requires the concomitant use of intravenous vancomycin to ensure efective treatment of the infection [4••, 8]. There is evidence of increasing antimicrobial resistance of bacterial pathogens isolated in low-income settings with a high prevalence of BM. For example, a recent study from Hawassa, Ethiopia, showed rising rates of antimicrobial resistance to penicillin, ceftriaxone, and chloramphenicol among isolates of S. pneumoniae and N. meningitidis obtained from patients hospitalized with BM [26]. Rising antimicrobial resistance to chloramphenicol among H. influenzae and N. meningitidis isolates has limited the clinical use of this antimicrobial in many low-income settings [27]. Increasing antimicrobial resistance patterns in these settings highlight the need for paying further attention to preventive strategies, such as increasing immunization coverage against the three significant pathogens causing BM [26–28]. Indeed, national healthcare plans must include vaccines that prevent BM as a strategic priority to counteract the growing concern of antimicrobial resistance [10].
Supportive care for people who survive an episode of BM is limited in low- and middle-income countries. Many people will experience a range of sequelae, and aftercare is expensive, and most families cannot afford neurological rehabilitation pay for anti-seizure medications [2]. Many suffer long-enduring sequelae such as hearing and vision loss, adding significant disability [2]. Therefore, in addition to preventing and treating BM, there is a need for further attention to strengthen early recognition and financial support for the management of sequelae from meningitis in healthcare and community settings [2, 3].
Achieving More Significant Health Equity by Addressing Gaps in Bacterial Meningitis in Low-and-Middle-Income Countries
Given that financial resources in healthcare are limited in settings with the highest disease burden, one central consideration is that decision-makers in low- and middle-income countries must consider investing in cost-saving interventions that produce the highest health gains in their national strategic health plans [29]. In this context, primary prevention of BM is the most important intervention to reduce morbidity, mortality, and the long-term and often disabling sequelae caused by BM in low-income and middle-income settings. Enhanced efforts to introduce and increase vaccination coverage of conjugate vaccines at a national and subnational level must occur for countries with BM’s highest disease burden [10, 15]. This is not an easy task, particularly after the public health deficits exacerbated by the COVID-19 pandemic. Ensuring support to reduce the prices of vaccines and increase sustainable access largely depends on the support of international organizations, including GAVI (Global Alliance Vaccine Initiative), WHO, and UNICEF [2, 3]. Critical activities of the WHO roadmap to control BM by 2020 rely on implementing locally appropriate immunization strategies tailored to achieve high vaccination coverage against N. meningitidis, S. pneumoniae, and H. influenzae. Other essential strategies to consider in preventing infections of bacterial pathogens that can lead to BM include screening against Streptococcus agalactiae during obstetric evaluations in resource-limited settings. This strategy may reduce the number of cases of neonatal sepsis caused by this pathogen. Additionally, chemoprophylaxis is generally used for close contact with cases of meningococcal meningitis. Still, the impact of antimicrobial chemoprophylaxis with rifampin, ciprofloxacin, or ceftriaxone in the context of large epidemics of meningococcal meningitis is controversial and not routinely recommended.
To achieve a meaningful impact in reducing the impact of BM in resource-limited settings, disease surveillance is an essential tool to guide public health decisions. Thus, countries with high incidences of BM must continue adopting, integrating, or implementing minimum national and subnational standards for surveillance of the primary meningitis pathogens, focusing on laboratory capacity and data management systems [2, 10]. Additionally, building support of international laboratory networks for diagnostic capacity testing, including molecular characterization, and monitoring the emergence of antimicrobial resistance are critical elements of an effective surveillance system for reducing the disease burden of BM.
Conclusions
Bacterial meningitis remains a significant cause of morbidity and mortality in all age groups in middle and low-income countries. In these settings, BM accounts for most of the estimated 5.6 million disability-adjusted life years attributed to BM globally. As part of the WHO effort to defeat meningitis by 2030, national immunization programs need to emphasize the widespread introduction and increased coverage of conjugate vaccines in resource-limited settings where the disease burden is most significant. Along with poverty alleviation and social improvement programs, increasing vaccination coverage against the three major pathogens that cause BM will have essential reductions in the incidence of BM, preventing the often-devastating long-term sequelae and further antimicrobial resistance. In the meantime, efforts targeting the optimization of diagnosis and earlier institution of empiric antimicrobial therapy and steroids in low-income settings remain crucial.
Footnotes
Competing Interests The authors declare no competing interests.
Human and Animal Rights and Informed Consent This article does not contain any studies with human or animal subjects performed by any of the authors.
References
Papers of particular interest, published recently, have been highlighted as:
• Of importance
•• Of major importance
- 1.••. Zunt ZJR Kassebaum NJ Blake N Glennie L Wright C Nichols E et al. 2019. GBD 2016 Neurology Collaborators Global, regional, and national burden of neurological disorders, a systematic analysis for the Global Burden of Disease Study 2016 Lancet Neurol. 18 5 459 480. 10.1016/S1474-4422(18)30499-X. This study assessed the global burden of bacterial meningitis.
- 2.World Health Organization. Defeating meningitis by 2020: a global road map. 2021. https://www.who.int/publications/i/item/9789240026407. Accessed 29 Sept 2023
- 3.Greenwood B, Sow S, Preziosi MP. Defeating meningitis by 2030 - an ambitious target. Trans R Soc Trop Med Hyg. 2021;115(10):1099–101. 10.1093/trstmh/trab133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.••. Progress Hasbun R. and challenges in bacterial meningitis: a review. JAMA. 2022;328(21):2147–54. 10.1001/jama.2022.20521. A comprehensive review of salient epidemiology and clinical aspects of BM in adults.
- 5.•. Zainel A, Mitchell H, Sadarangani M. Bacterial meningitis in children: neurological complications, associated risk factors, and prevention. Microorganisms. 2021;9(3):535. 10.3390/microorganisms9030535. A descriptive review of neurological complications associated to BM in children.
- 6.Lovera D, Amarilla S, Araya S, Galeano F, González N, Martínez de Cuellar C, Apodaca S, Arbo A. Risk factors for death and severe neurological sequelae in childhood bacterial meningitis. Pediatr Emerg Care. 2022. 38(12):637–643. 10.1097/PEC.0000000000002651 [DOI] [PubMed] [Google Scholar]
- 7.•. van de Beek D, de Gans J, Tunkel AR, Wijdicks EF. Community acquired bacterial meningitis in adults. N Engl J Med. 2006;354(1):44–53. 10.1056/NEJMra052116. A comprehensive review of the literature on bacterial meningitis.
- 8.Tunkel AR, Hartman BJ, Kaplan SL, Kaufman BA, Roos KL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39:1267–84. [DOI] [PubMed] [Google Scholar]
- 9.Barichello T, Rocha Catalão CH, Rohlwink UK, van der Kuip M, Zaharie D, Solomons RS, van Toorn R. Tutu van Furth M, Hasbun R, Iovino F, Namale VS. Bacterial meningitis in Africa Front Neurol. 2023;14(14): 822575. 10.3389/fneur.2023.822575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McIntyre PB, O’Brien KL, Greenwood B, van de Beek D. Effect of vaccines on bacterial meningitis worldwide. Lancet. 2012;380(9854):1703–11. 10.1016/S0140-6736(12)61187-8. [DOI] [PubMed] [Google Scholar]
- 11.Runde TJ, Anjum F, Hafner JW. Bacterial meningitis. 2023. Aug 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023. [PubMed] [Google Scholar]
- 12.Mazamay S, Guégan JF., Diallo N. Bompangue D, Bokabo E, Muyembe JJ, et al. An overview of bacterial meningitis epidemics in Africa from 1928 to 2018 with a focus on epidemics outside the belt. BMC Infect Dis. 21, 1027 (2021). 10.1186/s12879- [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Koelman DLH, Brouwer MC, Ter Horst L, Bijlsma MW, van der Ende A, van de Beek D. Pneumococcal meningitis in adults: a prospective nationwide cohort study over a 20-year period. Clin Infect Dis. 2022;74(4):657–67. 10.1093/cid/ciab477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Martin NG, Sadarangani M, Pollard AJ, Goldacre MJ. Hospital admission rates for meningitis and septicaemia caused by Haemophilus influenzae, Neisseria meningitidis, and Streptococcus pneumoniae in children in England over five decades: a population-based observational study. Lancet Infect Dis. 2014;14(5):397–405. 10.1016/S1473-3099(14)70027-1. [DOI] [PubMed] [Google Scholar]
- 15.Franco-Paredes C, Lammoglia L, Hernández I, Santos-Preciado JI. Epidemiology and outcomes of bacterial meningitis in Mexican children: 10-year experience (1993–2003). Int J Infect Dis. 2008;12(4):380–6. 10.1016/j.ijid.2007.09.012. [DOI] [PubMed] [Google Scholar]
- 16.••. Nakamura T, Cohen AL, Schwartz S, Mwenda JM, Weldegebriel G, Biey JNM, et al. The global landscape of pediatric bacterial meningitis data reported to the World Health Organization-coordinated Invasive Bacterial Vaccine-Preventable Disease Surveillance network, 2014–2019. J Infect Dis. 2021;224(12 Suppl 2):S161–73. 10.1093/infdis/jiab217. The IB-VPD surveillance network by WHO demonstrated that bacterial meningitis continues to cause many deaths in sub-Saharan Africa, and most deaths are caused by Streptococcus pneumoniae.
- 17.••.Kourna Hama M, Khan D, Laouali B, Okoi C, Yam A, Haladou M, Worwui A, Ndow PS, Nse Obama R, Mwenda JM, Biey J, Ntsama B, Kwambana-Adams BA, Antonio M. Pediatric bacterial meningitis surveillance in Niger: increased importance of Neisseria meningitidis serogroup C, and a decrease in Streptococcus pneumoniae following 13-valent pneumococcal conjugate vaccine introductioN. Clin Infect Dis. 2019;69(Suppl 2):S133–9. 10.1093/cid/ciz598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.••.Mandomando I, Mwenda JM, Nakamura T, de Gouveia L, von Gottberg A, Kwambana-Adams BA, et al. Evaluation of laboratories supporting Invasive Bacterial Vaccine-Preventable Disease (IB-VPD) Surveillance in the World Health Organization African region, through the performance of coordinated external quality assessment. Trop Med Infect Dis. 2023;8(8):413. 413. 10.3390/tropicalmed8080413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kwambana-Adams BA, Liu J, Okoi C, Mwenda JM, Mohammed NI, Tsolenyanu E, Renner LA, Ansong D, Tagbo BN, Bashir MF, Hama MK, Sonko MA, Gratz J, Worwui A, Ndow P, Cohen AL, Serhan F, Mihigo R, Antonio M, Houpt E; Paediatric Bacterial Meningitis Surveillance Network in West Africa. Etiology of Pediatric Meningitis in West Africa Using Molecular Methods in the Era of Conjugate Vaccines against Pneumococcus, Meningococcus, and Haemophilus influenzae Type b. Am J Trop Med Hyg. 2020. 103(2):696–703. 10.4269/ajtmh.19-0566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Parellada CI, Abreu AJL, Birck MG, Dias CZ, Moreira TDNF, Julian GS, Batista PM, Orengo JC, Bierrenbach AL. Trends in pneumococcal and bacterial meningitis in Brazil from 2007 to 2019. Vaccines (Basel). 2023;11(8): 1279. 10.3390/vaccines11081279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Truong KH, Do VC, Pham NNM, Ho TV, Phan TTQ, Hoang TA, et al. Childhood bacterial meningitis surveillance in Southern Vietnam: trends and vaccination implications from 2012 to 2021. Open Forum Infect Dis. 2023;10(7): 2ad229. 10.1093/ofd/ofad229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bui HT, Hoang VH, Ngo TV, Bui HV. Etiology and clinical features of bacterial meningitis in adults at National Hospital for Tropical Diseases during 2015–2018. Jpn J Infect Dis. 2023;76(2):101–5. 10.7883/yoken.JJID.2021.789. [DOI] [PubMed] [Google Scholar]
- 23.Franklin K, Kwambana-Adams B, Lessa FC, Soeters HM, Cooper L, Coldiron ME, Mwenda J, Antonio M, Nakamura T, Novak R, Cohen AL. Pneumococcal meningitis outbreaks in Africa, 2000–2018: systematic literature review and meningitis surveillance database analyses. J Infect Dis. 2021;224(12 Suppl 2):S174–83. 10.1093/infdis/jiab105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mazamay S, Broutin H, Bompangue D, Muyembe JJ, Guégan JF. The environmental drivers of bacterial meningitis epidemics in the Democratic Republic of Congo, central Africa. PLoS Negl Trop Dis. 2020;14(10): e0008634. e0008634. 10.1371/journal.pntd.0008634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Obiero CW, Mturi N, Mwarumba S, Ngari M, Newton C, Boele van Hensbroek M, Berkley JA. Clinical features to distinguish meningitis among young infants at a rural Kenyan hospital. Arch Dis Child. 2021. 106(2):130–136 10.1136/archdischild-2020-318913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Garlicki AM, Jawień M, Pancewicz SA, Moniuszko-Malinowska A. Bacterial meningitis in adults - principles of diagnosis and antibiotic treatment. Przegl Epidemiol. 2023;77(1):3–22. 10.32394/pe.77.01. [DOI] [PubMed] [Google Scholar]
- 27.Assegu Fenta D, Lemma K, Tadele H, Tadesse BT, Derese B. Antimicrobial sensitivity profile and bacterial isolates among suspected pyogenic meningitis patients attending at Hawassa University Hospital: Cross-sectional study. BMC Microbiol. 2020;20(1): 125. 10.1186/s12866-020-01808-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duke T, Michael A, Mokela D, Wal T, Reeder J. Chloramphenicol or ceftriaxone, or both, as treatment for meningitis in developing countries? Arch Dis Child. 2003;88(6):536–9. 10.1136/adc.88.6.536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Leech AA, Kim DD, Cohen JT, Neumann PJ. Are low and middle-income countries prioritising high-value healthcare interventions? BMJ Glob Health. 2020;5(2): e001850. 10.1136/bmjgh-2019-001850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sibomana O, Hakayuwa CM. The meningitis outbreak returns to Niger: concern, efforts, challenges, and recommendations. Immun Inflamm Dis. 2023;11(7):953. 10.1002/iid3.953. [DOI] [PMC free article] [PubMed] [Google Scholar]


