Abstract
Background and purpose:
Alzheimer's disease (AD) is a common neurodegenerative disease and the fifth leading cause of death among the elderly. The development of drugs for AD treatment is based on inhibiting cholinesterase (ChE) activity and inhibiting amyloid-beta peptide and tau protein aggregations. Many in vitro findings have demonstrated that thiazole-and thiazolidine-based compounds have a good inhibitory effect on ChE and other elements involved in the AD pathogenicity cascade.
Experimental approach:
In the present review, we collected available documents to verify whether these synthetic compounds can be a step forward in developing new medications for AD. A systematic literature search was performed in major electronic databases in April 2021. Twenty-eight relevant in vitro and in vivo studies were found and used for data extraction.
Findings/Results:
Findings demonstrated that thiazole-and thiazolidine-based compounds could ameliorate AD's pathologic condition by affecting various targets, including inhibition of ChE activity, amyloid-beta, and tau aggregation in addition to cyclin-dependent kinase 5/p25, beta-secretase-1, cyclooxygenase, and glycogen synthase kinase-3β.
Conclusion and implications:
Due to multitarget effects at micromolar concentration, this review demonstrated that these synthetic compounds could be considered promising candidates for developing anti-Alzheimer drugs.
Keywords: Alzheimer's disease, Amyloid beta, Cholinesterase, Glycogen synthase kinase, Thiazolidine, Thiazole.
INTRODUCTION
Alzheimer's disease (AD) is the most common neurodegenerative disease in which neurons' structural and functional features are lost. This disease has been the fifth leading cause of death among people over 65 (1,2). The lifetime risk of AD is 15% for people over 65 and 40% for people over 80. The prevalence of AD and other types of dementia is higher in older people, and epidemiological data show that the incidence of Alzheimer's is higher in women than in men (3). It is a progressive disease whose symptoms gradually begin and become severe. The disease affects various brain functions, and minor memory problem is usually the first sign of the disease (4,5). As the disease progresses, memory problems worsen, and other symptoms such as misperception, confusion, impaired decision-making and planning, and speech and language problems appear. The course of the disease includes gradual impairment of memory, judgment and language skills, and behavioral changes. Microscopic biopsy indicates cortical atrophy with enlarged ventricles in the brain. These clinical manifestations reflect neural degeneration in the cerebral cortex, especially in the temporoparietal cortex and hippocampus (6).
The pathogenicity of Alzheimer's and many other neurodegenerative diseases are common, including incorrect folding and accumulation of proteins (accumulation of amyloid beta (Aβ) and formation of amyloid plaques), oxidative stress, and formation of free radicals, imbalance of metals homeostatic system, and excessive phosphorylation of tau proteins. In addition, genetics and the environment are determinant factors in the pathogenesis of this disease (7,8). In Aβ, globular proteins (amyloid bodies) are formed outside the neurons in some areas of the brain, while fibrous protein structures are included in the cell body of the neurons (9). Aβ is a peptide of 36-43 amino acids and a major component of amyloid plaques in the brains of Alzheimer's patients. These peptides are derived from the amyloid precursor protein. They are expressed in the cells of the nervous system, which have many functions, including cell-to-cell attachment, cell contact, and formation of extracellular matrix and cytoskeleton. Aging plaques are composed of protein strands called amyloid bodies and some other proteins called apolipoprotein E, synuclein, and alpha-antichymotrypsin (10). The formation of these plaques seems to be one of the main causes of Alzheimer's since they break the connection between nerve cells and eventually cause the death of nerve cells and the destruction of brain tissue.
Pathological abnormalities of AD include depositing two filamentous proteins of Aβ and tau proteins. Aβ bound to apolipoprotein E is located outside neurons, while tau proteins are derived from microtubules inside neurons (11). AD affects the primary cholinergic neurons, so the treatment should be based on the drugs that inhibit the reduction of acetylcholine at the synapses. AD is typically treated symptomatically, and most of the drugs approved for treating Alzheimer's modulate neurotransmitters such as acetylcholine and glutamate. Disease-modifying therapies delay the onset of the disease and slow its progression. Standard drug treatments for AD include acetylcholinesterase (AChE) inhibitors and N-methyl-D-aspartate antagonists (12,13).
Due to the role of AChE and carbonic anhydrase in treating AD, finding new compounds with inhibitory effects on these enzymes can be a step toward developing new anti-Alzheimer. There is accumulating evidence that carbonic anhydrase increases in the AD brain. The collected evidence shows that the design of hybrid molecules that act specifically on enzymes and different protein targets involved in the pathogenicity of AD, with good effectiveness and at sub-toxic doses, can be an effective approach for treating the treatment of neurodegenerative diseases such as AD. Thiazolidine and thiazole derivatives have demonstrated strong inhibitory effects against the enzymes and protein targets of the AD pathogenicity cascade and inhibited the aggregation of Aβ and tau (Scheme 1). In the present study, we aimed to systematically review the available literature on the effectiveness of thiazole and thiazolidine derivatives in treating AD.
Scheme 1.
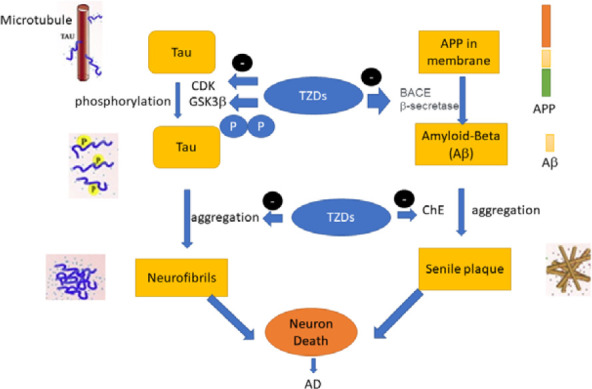
Enzymes and targets of AD pathogenesis. AD, Alzheimer disease; CDK, cyclin dependent kinase; GSK glycogen synthase kinase; ChE, cholinesterase; BACE, β-site amyloid precursor protein cleaving enzyme; APP, amyloid precursor protein; Aβ, amyloid-beta; TZD, thiazolidines.
METHODS
Study search and protocol
A systematic literature search was performed on Web of Science, PubMed, Scopus, Science Direct, Embase, and Ovid in April 2021. No time or language limit was used in the search strategy. The search query used for this purpose includes Thiazolidine “OR thiazole OR thiazol” and “Alzheimer's disease OR Alzheimer's OR Alzheimer” in PubMed. In Scopus, “Thiazolidine and its similar terms” were searched in the title, abstract, and keywords, and then “Alzheimer's with all equivalents” were searched within the results. A hand search into Google Scholar and references of the included studies further improved the search. All procedures, including database search, article selection, and data synthesis, were performed independently by two research team members. Still, disagreements between the authors were resolved in each step before proceeding to the next stage. PRISMA Checklist 2009, a good protocol for reporting systematic reviews, was used for the study design and article selection process (14).
Inclusion criteria
The review articles, editorials, case reports, and conference papers were excluded at the first step of document evaluation. Next, irrelevant articles were excluded from further evaluation. Also, computational modeling and docking studies were disqualified from further assessment. They were only included if in vivo assessment or cell culture experiments were performed alongside the docking studies. Furthermore, documents on thiazole and thiazolidine derivatives performed on imaging probes of Alzheimer's were also excluded.
Data synthesis and the variables
All required information including the author's name and the conjugated parts of thiazole and thiazolidine derivatives were extracted and summarized for data extraction. In addition, the main target and inhibitory values were recorded and described qualitatively.
RESULTS
After a literature search and considering the defined inclusion criteria, 7481 articles were collected from the database search, of which 2328 were from PubMed, 3699 were in Scopus, and 1413 were in other databases. By limiting the records to the described inclusion criteria, 34 articles were achieved. After removing irrelevant documents, 28 articles were selected for data extraction. The selection process of articles is demonstrated in Fig. 1.
Fig. 1.
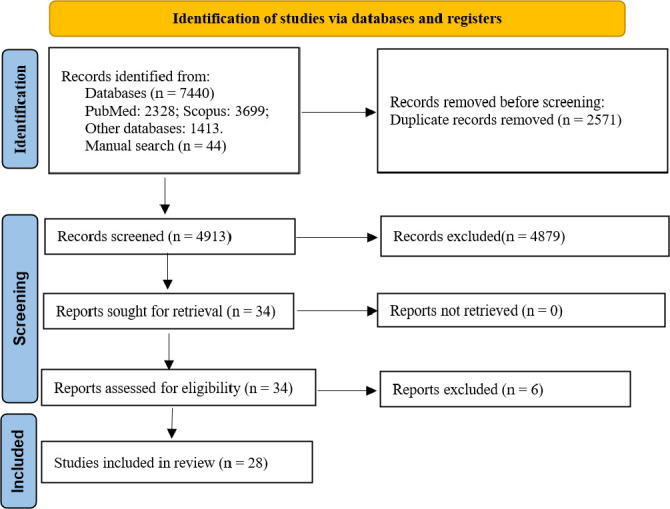
Selection of articles based on inclusion criteria.
Study findings
Inhibition of tauopathy, Aβ aggregation, and ChE inhibition were the most important aims of the included studies. However, the designed molecules could also target other enzymes and natural elements that were involved in the pathogenesis of Alzheimer's. Below, these effects are described in detail. Also, the produced therapeutic agents with possible anti-Alzheimer's activity and the most important inhibitory targets with the reported inhibitory values are summarized in Table 1.
Table 1.
Detailed knowledge of the included studies.
| Conjugate part | Main target | Other targets | Animal/cell type | Inhibition value (IC50) | Reference |
|---|---|---|---|---|---|
| Thiazolidin-4-ones | AChE | - | Rat | AChE = 3.11 μM | (15) |
|
| |||||
| Aminothiazole B-ring | Aβ42 | - | SHSY5Y | > 1 μM | (16) |
|
| |||||
| L1 [4-(benzo[d]thiazol-2-yl) -2-((4,7-dimethyl-1,4,7- triazonan-1-yl) methyl)-6- methoxyphenol] | Aβ | p-tau | Transgenic 5xFAD mice | 40-50% reduction of Aβ and p-tau | (17) |
|
| |||||
| Thiazolidin-4-one | AChE | - | Wistar rats | 4.46 μM | (18) |
|
| |||||
| Benzylidene-thiazolidine- 2,4-diones | IL- 1β | ER-β, NLRP3 inflammasome | J774A.1 cells | 77% Inhibition of IL-1 β release at 10 μM | (19) |
|
| |||||
| 6,6-Dimethyl-3-(2- hydroxyethyl) thio-1- (thiazol-2-yl)-6,7-dihydro-2- benzothiophene-4(5H)-one | α5 GABAA receptors | Aβ | Mice, HEK293 cells | GABA = 10 μM | (20) |
|
| |||||
| 1 -(2-Bromobenzyl)-4-((4- oxo-4-((4-phenylthiazol-2-yl) amino) butanamido) methyl) pyridin-1-ium bromide | ChE | Aβ | PC12 cells | AChE = 21.49 μM BuChE = 35.29 μM 42.66% Aβ inhibition at 10 μM | (21) |
|
| |||||
| Thiazolopyridyl tetrahydroacridine | GSK-3β, ChE | Tau protein | Mice | AChE = 1.2 nM, BChE = 149.8 nM, hGSK-3β = 22.2 nM | (22) |
|
| |||||
| 4-hydroxy-4-phenyl-3- (pyridin-4-ylmethyl) thiazolidine-2-thione | GSK-3 | - | RAW 264.7 mouse macrophage, human embryonic kidney (HEK293), THP-1 human monocytic cells | GSK-3α = 22 nM GSK-3β = 11 nM | (23) |
|
| |||||
| Benzylpyridinium-based benzothiazole | ChE | Aβ | PC12 and HepG2 cells | AChE = 14 nM BuChE = 182 nM 44.9% Aβ inhibition at 50 μM | (24) |
|
| |||||
| Methyl(4 phenylbutyl) amine | ChE | Histamine H3 receptor | Mice | AChE = 13.96 μM BuChE = 14.62 μM H3 antagonist activity (pA2 = 8.27) | (25) |
|
| |||||
| 5-(Difluoromethyl)-2- (ethylamino)-3a,6,7,7a- tetrahydro-5H-pyrano [3,2- d]thiazole-6,7-diol | OGA | Tau protein | Monkey, dog, rat | 10 mg/kg, orally | (26) |
|
| |||||
| Quinoxaline-bisthiazole | β-secretase | Edema and inflammation | Rat | β-secretase = 3 μM, 69% inhibition of edema | (27) |
|
| |||||
| Tacrine -2- phenylbenzothiazole | AChE | Aβ | SH-SY5Y cells | AChE = 0.27 μM 44.6% Aβ inhibition | (28) |
|
| |||||
| 1-(3-Chloro-4- hydroxyphenyl)-3-(6- ethoxybenzo[d]thiazol-2-yl) urea | 17β-HSD10 | - | HEK293 mts 17β- HSD10 cells | ≥ 77% inhibition at 25 μM | (29) |
|
| |||||
| 2,4-Thiazolidinedione | GSK-3β | AcPHF6 | SH-SY5Y | GSK-3β = 0.89 μM, AcPHF6 aggregation is 80% at 10 μM | (30) |
|
| |||||
| Thiazolyl-thiadiazines | β-secretase | Aβ | Rat | β-secretase = 9 μM 63% edema inhibition | (31) |
|
| |||||
| 2-substituted-thio-N-(4- substituted-thiazol/ 1H- imidazol-2-yl) acetamide | β-secretase | Effect on blood-brain barrier | HEK293 cells | β-secretase = 4.6 μM, High BBB permeability and low toxicity | (32) |
|
| |||||
| Donepezil and diarylthiazole | ChE | Aβ1-42, p- Tau, cleaved caspase 3, cleaved PARP | SH-SY5Y cells, mice | AChE = 0.30 μM BuChE = 1.84 μM | (33) |
|
| |||||
| 4-methyl-5-(2-(nitrooxy) ethyl) thiazol-3-ium chloride | Aβ | Tau protein | Mice | >60% Aβ inhibition (by 1 mg/kg, i.p. + 20 mg/kg/day orally for 12 weeks) | (34) |
|
| |||||
| Thiazole acetamide | ChE | β-secretase, Aβ | HeLa cells | AChE = 3.14 μM, BuChE = 2.94 μM, Aβ = 10.6 μM β-secretase= 13.2 μM | (35) |
|
| |||||
| N'-(4-(2,4-dichlorophenyl) thiazol-2-yl)-2-(2-oxobenzo[d] thiazol-3(2H)-yl) acetohydrazide | ChE | - | NIH/3T3 cells | AChE = 25.5 μM BuChE = 80 μM | (36) |
|
| |||||
| N-(4-(Benzo[d]thiazol-2- ylmethyl)phenyl)-6-(1,2,3,4- tetrahydroacridin -9-ylamino) hexanamide | AChE | Aβ | SHSY-5Y cells | AChE = 0.57 μM, 61.3% Aβ inhibition | (37) |
|
| |||||
| Benzo[d]thiazol-2- ylimino)methyl)-4-nitrophenol + CuCl2 | superoxide dismutase SOD | Aβ | PC12 cells | Without CuCl2 = 1.3 μM With CuCl2 = 0.19 μM | (38) |
|
| |||||
| Phenylthiazole-tacrine | ChE | Aβ | Cortical neurons | AChE = 5.78 μM, BuChE = 5.75 μM, Inhibition of Ca2+ overload and Aβ (66%) at 20 μM | (39) |
|
| |||||
| 2-aminothiazoles | Tau protein | - | M17-TAU-P301L cells | Tau = 14 nW | (40) |
|
| |||||
| N-[3-({2-[(2E)-2- Benzylidenehydrazino]-2- oxoethyl }sulfanyl)-5-({2-[(2-chloroacetyl)amino] -1,3-thiazol-4-yl}methyl)-4H-1,2,4-triazol-4-yl] - 4-chlorobenzamide | CDK5/p25 | - | Vero-C-1008 cells | cdk5 = 42 nM | (41,42) |
|
| |||||
| Phenylthiazolyl-hydrazides | Tau protein | LDH | N2A cells | Tau = 7.7 μM, 47.4% LDH inhibition | (43) |
AChE, Acetylcholinesterase; Aβ, amyloid-β; IL, interleukin; BACE-1, β-secretase; GSK-3β, glycogen synthase kinase 3 beta; AcPHF6, tau derived hexapeptide; ER-β, estrogen-receptor β; GABA, gamma-aminobutyric acid type A; BuChE, butyrylcholinesterase; PARP, poly (ADP-ribose) polymerase; 17β-HSD10, 17β-hydroxysteroid dehydrogenase type 10; CDK5, cyclin-dependent kinase; hCAs, human carbonic anhydrases; OGA, O- GlcNAcase; SOD, superoxide dismutase; LDH, lactate dehydrogenas; SH-SY5Y, human neuroblastoma cell line.
ChE inhibition
ChE is the most important enzyme involved directly in the pathogenesis of Alzheimer's. Inhibition of ChE is the main goal of designing new therapeutics for treating neurodegenerative diseases. Findings of enzyme kinetics demonstrated that AChE inhibition followed a mixed inhibition mechanism through selective and non-specific inhibition of all isoforms of ChE (21). The compounds containing benzyl piperidine-linked diaryl thiazoles as the core also showed significant anti-ChE activity with IC50 values of 0.30 μM and 1.84 μM for AChE and butyrylcholinesterase (BuChE), respectively. It also showed substantial neuroprotection, as evidenced by decreasing Aβ 1 -42-induced toxicity and tauopathy, which may be attributed to their reported antioxidant activities. Due to their good oral absorption and toleration of up to 2000 mg/kg oral administration, without considerable toxic effects in several animal studies, benzyl piperidine-linked diaryl thiazoles were suggested as a potent anti-Alzheimer drug (33). Thiazolidin-4-one was also reported as a potent AChE inhibitor with an IC50 value of 4.46 μM in the hippocampus of rats (18). Similarly, a compound containing bromobenzyl-phenylthiazol-amino-pyridinium bromide and phenyl thiazole-tacrine hybrids demonstrated strong AChE and BuChE inhibition at 21.49 and 35.29, respectively, in addition to more than 40-60% Aβ inhibition at 10 to 20 μM (21,39).
Aβ and tau proteins
Aβ peptide is a proteolytic fragment of the transmembrane amyloid precursor protein and the main pathological element of Alzheimer's disease found as the amyloid plaques in the brains of these patients. On the other hand, tau is a brain-specific and microtubule-associated protein in neurons, which is regulated by phosphorylation. Accumulations of pathological tau protein in synapses, along with the aggregation of Aβ into amyloid plaques, is the main pathological hallmark in AD, which leads to the damage and destruction of synapses resulting in impairment of memory and cognition (44,45). In vitro, findings demonstrated that most thiazole derivatives, such as aminothiazole complexes and N-benzyl pyridinium-based benzoheterocycles, can inhibit Aβ at micromolar and nanomolar concentrations (16,24,40). Consistent animal studies have also shown that alkyl-thiazolium chloride and 4-(benzo[d]thiazol-2-yl)-2-((4,7-dimethyl-1,4,7-triazonan-1-yl)methyl)-6-methoxyphenyl can be considered a novel multifunctional drug in ameliorating oxidative damage and inhibition of Aβ-aggregation, attenuating amyloid burden and neuroinflammation (17,34).
Glycogen synthase kinase
Glycogen synthase kinase-3 (GSK-3) is a serine/threonine protein kinase, which is typically involved in regulating hormonal control of glucose homeostasis and neuroendocrine control of energy metabolism. It is also responsible for the phosphorylation of tau protein and Aβ, which are the first appearances of Alzheimer's pathology (46). Findings demonstrated that thiazolidinedione derivatives displayed promising inhibitory effects on GSK-3β and tau aggregation at an IC50 value of 0.89 μM. Moreover, they demonstrated parallel artificial membrane permeability assay-blood brain barrier permeability and cellular safety profile on the human neuroblastoma cell line (SH-SY5Y) (30). Some pyridine derivatives of thiazolidine were shown highly potent and selective inhibitory effects on GSK-3, indicating that inhibitors of GSK-3 can serve as a major target for developing potential anti-Alzheimer's therapeutics (21). New classes of drugs containing thiazol in complex with acridine and tacrine in their structure have also shown ChE and GSK-3 dual inhibitory effects, which is a key step in designing multi-target drugs to treat Alzheimer's disease (22).
β-secretase inhibitors
Beta-secretase, known as beta-site amyloid precursor protein cleaving enzyme-1 (BACE-1), is an enzyme found mainly in neurons and essential for the cleavage of Aβ precursor protein and subsequent generation of Aβ. Inhibitors of β-secretase can be of great importance in treating AD since they inhibit BACE-1 and, therefore, Aβ aggregation (47). Studies on cell models showed that thiazol-imidazole-acetamide derivatives with IC50 value of less than 5 μM and good pharmacological features such as high blood-brain barrier permeability and low cytotoxicity could serve as potent molecules with promising anti-Alzheimer's activity (32,35). On the other hand, thiazolyl-thiadiazole compounds were found as potent multitarget-directed β-secretase inhibitors with anti-inflammatory properties in the sub-micromolar range (31).
Cyclin-dependent kinase inhibitors
Cyclin-dependent kinase 5 (CDK5) is a multifaceted proline-directed serine/threonine kinase. The active form (in complex with p35 and p39) plays a critical role in the central nervous system (CNS) protecting neuronal structure and regulation of neurotransmitter release. On the other hand, this kinase's hyperactivity is associated with AD development; hence, drugs with inhibitory effects on CDK5 may be useful in developing anti-Alzheimer's therapeutics (48). In vitro studies demonstrated that the clubbed triazolyl thiazole, such as benzylidene hydrazino-thiazol-chlorobenzamide, may have CDK5/p25 inhibitory activity at sub-micromolar concentration (42).
Other targets
Thiazole and thiazolidine-based complexes were shown promising anti-Alzheimer's effects by affecting various targets. Amongst the tested compounds, some thiazolidine hybrid complexes demonstrated inflammasome inhibitory effects with 77% suppression of interleukin-1β release at 10 μM (21). Benzo-thiazol derivatives in complex with metals (CuCl2) also demonstrated multifunctional activity not only by inhibiting Aβ self-aggregation but also through reducing intracellular reactive oxygen species decreasing activity via increasing superoxide dismutase, which makes them potent amyloid inhibitors and anti-Alzheimer's agent (38). Conversely, animal studies and in vitro findings on HEK293 stable cell lines indicated that gamma-aminobutyric acid type A receptors (GABA-A) are widely distributed in neuronal networks. Therefore, inhibitors of the α5 subunit of GABA-A, such as n-alkyl-thiazol-benzothiophene derivatives, cannot be considered a suitable therapeutic target for the treatment of AD (20). Furthermore, findings of animal trials on rats, dogs, and monkeys demonstrated that difluoro methyl methylamino tetrahydro-pyranothiazole diol, as a strong and highly selective inhibitor of O-GlcNAcase with satisfactory CNS penetration capability, is a promising compound with anti-Alzheimer properties that was succeeded to human trial (26).
DISCUSSION
AD is the most common neurodegenerative disorder, which demands finding a way to prevent it and effective therapy. The development of therapeutics for AD is based on the amyloid cascade hypothesis (vaccines, β-and γ-secretase inhibitors) or targeting tau and neurofibrillary tangle formation, neuroinflammation, etc. ChE, BACE-1, amyloid-β 1-42, γ and β-secretase, phosphodiesterase type IV inhibitors are important targets for developing the anti-Alzheimer. Among these, the γ-and β-secretase inhibitors can be clustered in several heterocyclic classes (imidazole, thiazoles, indoles, benzaldehydes, pyrimidine, etc. (49). Thiazole derivatives are an important class of heterocyclic molecules with versatile pharmacological activities. Some thiazole analogs are fungicides, cardiotonic, anti-arrhythmic, and antitumor. Many thiazoles are fibrinogen receptor antagonists with antithrombotic activity, bacterial DNA gyrase B inhibitors, and lipo-oxygenase inhibitors. Aminothiazoles are known to be ligands of estrogen receptors as well as a novel class of adenosine receptor antagonists (50). Many studies have been performed on the therapeutic potentials of thiazole-and thiazolidine-based compounds in treating neurological diseases. The present review demonstrated that thiazoles and thiazolidines may be considered multi-target therapeutic agents with promising anti-Alzheimer properties. Amine-containing thiazole derivatives such as N-(2,3-dimethyl phenyl)thiazol-2-amine also showed significant inhibitory effect against ChE with reported IC50 values of 9 nM and 0.646 μM for AChE and BuChE, respectively (51).
Similarly, N-(5-isopropyl-thiazol-2-yl) isobutyrate, a potent CDK5 and CDK2/cyclin E inhibitor, could inhibit CDK5 and CDK2 at 320 nM and improve the potency of CDK5 by 60 folds (52). Moreover, preliminary evidence demonstrated that the complexation of hydroxyphenyl with benzothiazole leads to a group of multifunctional molecules with both amyloid binding and integrated metal chelating properties, which can be considered candidates for targeting Alzheimer's amyloidogenesis (53). Similarly, thiazolidinedione derivatives were shown to inhibit GSK-3β at nanomolar concentration and the aggregation of tau-derived hexapeptide AcPHF6 (80% inhibition) at 10 μM, indicating that these multi-target compounds might serve as a novel class of drugs by targeting independent but closely related targets of AD pathogenesis (30). Multifunctional benzothiazole-containing molecules in complexes with metals such as Cu2+ have been demonstrated to effectively interact with the metal binding sites on Aβ peptide and inhibit amyloid and tau aggregation in addition to dissolving the formed fibrils and thereby ameliorating Aβ-mediated cellular toxicity (38). In vitro, findings revealed that most of the thiazole and thiazolidine-based compounds, including aminothiazole, benzothiazole, and hybrid complexes such as tacrine-thiazoles had potent anti-ChE activity comparable to or even higher than donepezil as a reference drug. Without significant cytotoxicity, these compounds could inhibit Aβ self-aggregation or AChE-induced Aβ aggregation (24). Animal studies have shown that most of these compounds exhibited good oral absorption and are well-tolerated in rodents without noticeable toxic effects (33). These findings indicated that some of these thiazole and thiazolidine-based derivatives might also be useful in alleviating learning and memory deficits in an animal model of dementia, amnesia, and many other diseases of the CNS (54,55).
Molecular docking studies verified that thiazolidine-4-ones could efficiently fit into the enzyme and inhibit all isoforms of AChE (15). Thiazolocoumarinyl Schiff-base derivatives also revealed good binding affinities with all ChE isoforms but indicated superior interaction patterns with BuChE compared to AChE (56). According to the results of molecular docking evaluation, benzyl pyridinium-based benzothiazole could also effectively occupy the active site of AChE and BuChE at the vicinity of the catalytic triad, demonstrating as an effective inhibitor of ChE activity (24). Similarly, computation studies showed that thiazolyl-thiadiazole could perform as multitarget-directed ligands with multitargeted non-selectively interaction with the catalytic domain of BACE-1, cyclooxygenase 1 (COX-1) and COX-2 enzymes (31). The computational simulation results also revealed an efficient binding of thiazole-pyridiniums to both catalytic active and peripheral anionic sites resulting in dual inhibition of AChE and Aβ aggregation, making these compounds novel anti-Alzheimer agents (21).
Sustained drug release and increasing the circulation time of drugs is a novel approach to enhance the efficiency of therapy and increase the therapeutic index, which can be achieved by modification in nanostructures and conjugation with nanoparticles (57). Modifying these potent ChE inhibitors in nanostructure may help develop new medications to effectively treat neurodegenerative disorders (41,57,58). Conjugation of the synthesized compounds with antimicrobial peptides may also enhance treatment efficacy since antimicrobial peptides have cell-penetrating potential, antioxidant properties, and inhibitory effects on various enzymes, including ChE (59,60). Findings revealed that hybrid complexes of thiazole and thiazolidine with other biologically active compounds could enhance their therapeutic values. In this regard, the inhibitory activity of thiazolidin-4-ones containing the propylpiperidine core against ChE was significantly improved, making them potential candidates for future Alzheimer's therapy. Complexation of macromolecules such as a peptide, acridine, and tacrine produces hybrid supermolecules, which may improve the binding efficiency of the compounds to a variety of target enzymes, leading to enhanced biological efficiency of thiazole and thiazolidine (61,62,63). Tacrine-based hybrids of thiazole can also be considered effective multi-targeted agents against AD (28,37,64). Findings have also demonstrated that multipotent phenyl thiazole-tacrine hybrids can inhibit ChE activity and Ca2+ overload with 66% inhibition of Aβ aggregation at mi cromolar concentration (39). These hybrid complexes are emerging AD therapeutics, which represent a worthwhile value in developing new multi-target-directed anti-Alzheimer's drugs. However, further, in vivo and human studies are required in terms of evaluation of their ChE inhibitory capacity and inhibition of Aβ aggregation to warrant the effectiveness of these hybrid compounds.
CONCLUSION
Our results strongly support that these multi-target molecules may provide new insights into developing potent anti-Alzheimer's therapies by inhibiting ChE activity, Aβ, and tau aggregation and effectively facilitating cellular toxicity and neuroinflammation. Some of these compounds, in addition to anti-amyloid, neuroprotective, and anti-amnesic properties, demonstrated good inhibitory effects on other enzymes involved in the pathogenesis of AD, including CDK5/p25, BACE-1, COX, and GSK-3β. Overall, these molecules can be considered potent AD therapeutics, which can attenuate the formation of amyloid plaques and tau aggregation and thus can be proposed as promising agents for further research to develop anti-Alzheimer's drugs.
Conflict of interest statement
All authors declared no conflict of interest in this study.
Authors' contribution
M. Nejabat, F. Hadizadeh, and K. Abnous contributed to the conceptualization and supervision of the study; Z. Abdollahi contributed to data curation, preparation and writing of the original draft of the article. The finalized manuscript was approved by all authors.
Acknowledgments
This work was financially supported by Vice-Presidency for research at Mashhad University of Medical Sciences through Grant No. 981617.
REFERENCES
- 1.Alzheimer's association report. 2021 Alzheimer's disease facts and figures. Alzheimers Dement. 2021;17(3):327–406. doi: 10.1002/alz.12328. DOI: 10.1002/alz.12328. [DOI] [PubMed] [Google Scholar]
- 2.Hassanzadeh A, Yegdaneh A, Rabbani M. Effects of hydroalcoholic, methanolic, and hexane extracts of brown algae Sargassum angustifolium on scopolamine-induced memory impairment and learning deficit in rodents. Res Pharm Sci. 2023;18(3):292–302. doi: 10.4103/1735-5362.371585. DOI: 10.4103/1735-5362.371585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stix G. Alzheimer's: forestalling the darkness. Sci Am. 2010;302(6):50–57. doi: 10.1038/scientificamerican0610-50. DOI: 10.1038/scientificamerican0610-50. [DOI] [PubMed] [Google Scholar]
- 4.Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology. 2013;80(19):1778–1183. doi: 10.1212/WNL.0b013e31828726f5. DOI: 10.1212/WNL.0b013e31828726f5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Teymuori M, Yegdaneh A, Rabbani M. Effects of Piper nigrum fruit and Cinnamum zeylanicum bark alcoholic extracts, alone and in combination, on scopolamine-induced memory impairment in mice. Res Pharm Sci. 2021;16(5):474–481. doi: 10.4103/1735-5362.323914. DOI: 10.4103/1735-5362.323914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Whitwell JL, Jack CR, Przybelski SA, Parisi JE, Senjem ML, Boeve BF, et al. Temporoparietal atrophy: a marker of AD pathology independent of clinical diagnosis. Neurobiol Aging. 2011;32(9):1531–1541. doi: 10.1016/j.neurobiolaging.2009.10.012. DOI: 10.1016/j.neurobiolaging.2009.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lin L, Zheng LJ, Zhang LJ. Neuroinflammation, gut microbiome, and Alzheimer's disease. Mol Neurobiol. 2018;55(11):8243–8250. doi: 10.1007/s12035-018-0983-2. DOI: 10.1007/s12035-018-0983-2. [DOI] [PubMed] [Google Scholar]
- 8.Mirzaee M, Semnani S, Roshandel G, Nejabat M, HesariH Z, Joshaghani H. Strontium and antimony serum levels in healthy individuals living in high-and low-risk areas of esophageal cancer. J Clin Lab Anal. 2020;34(7):1–8. doi: 10.1002/jcla.23269. e23269. DOI: 10.1002/jcla.23269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Engel M, Do-Ha D, MunozL SS, Ooi L. Common pitfalls of stem cell differentiation: a guide to improving protocols for neurodegenerative disease models and research. Cell Mol Life Sci. 2016;73(19):3693–3709. doi: 10.1007/s00018-016-2265-3. DOI: 10.1007/s00018-016-2265-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jones S V, Kounatidis I. Nuclear factor-kappa B and Alzheimer disease, unifying genetic and environmental risk factors from cell to humans. Front Immunol. 2017;8(1805):1–9. doi: 10.3389/fimmu.2017.01805. DOI: 10.3389/fimmu.2017.01805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Serrano-Pozo A, Frosch MP, Masliah E, Hyman BT. Neuropathological alterations in Alzheimer disease. Cold Spring Harb Perspect Med. 2011;1(1):1–24. doi: 10.1101/cshperspect.a006189. a006189. DOI: 10.1101/cshperspect.a006189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shega JW, Ellner L, Lau DT, Maxwell L. Cholinesterase inhibitor and N-methyl-D-aspartic acid receptor antagonist use in older adults with end-stage dementia: a survey of hospice medical directors. J Palliat Med. 2009;12(9):779–783. doi: 10.1089/jpm.2009.0059. DOI: 10.1089/jpm.2009.0059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pashaei H, Rouhani A, Nejabat M, Hadizadeh F, Mirzaei S, Nadri H, et al. Synthesis and molecular dynamic simulation studies of novel N-(1-benzylpiperidin-4-yl) quinoline-4-carboxamides as potential acetylcholinesterase inhibitors. J Mol Struct. 2021;1244(130919):1–10. DOI: 10.1016/j.molstruc.2021.130919. [Google Scholar]
- 14.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Getzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–e34. doi: 10.1016/j.jclinepi.2009.06.006. DOI: 10.1016/j.jclinepi.2009.06.006. [DOI] [PubMed] [Google Scholar]
- 15.da Silva DS, Soares MSP, Martini F, Pesarico AP, de Mattos BDS, de Souza AA, et al. In vitro effects of 2-{4-[methylthio(methylsulfonyl)]phenyl}-3-substitutedthiazolidin-4-ones on the acetyl-cholinesterase activity in rat brain and lymphocytes: isoform selectivity, kinetic analysis, and molecular docking. Neurochem Res. 2020;45(2):241–253. doi: 10.1007/s11064-019-02929-8. DOI: 10.1007/s11064-019-02929-8. [DOI] [PubMed] [Google Scholar]
- 16.Rynearson KD, Buckle RN, Herr RJ, Mayhew NJ, Chen X, Paquette WD, et al. Design and synthesis of novel methoxypyridine-derived gamma-secretase modulators. Bioorg Med Chem. 2020;28(22):1–24. doi: 10.1016/j.bmc.2020.115734. 115734. DOI: 10.1016/j.bmc.2020.115734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rynearson KD, Buckle RN, Herr RJ, Mayhew NJ, Chen X, Paquette WD, et al. Design and synthesis of novel methoxypyridine-derived gamma-secretase modulators. Bioorg Med Chem. 2020;28(22):1–24. doi: 10.1016/j.bmc.2020.115734. 115734. DOI: 10.1016/j.bmc.2020.115734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.das Neves AM, Berwaldt GA, Avila CT, Goulart TB, Moreira BC, Ferreira TP, et al. Synthesis of thiazolidin-4-ones and thiazinan-4-ones from 1-(2-aminoethyl)pyrrolidine as acetylcholinesterase inhibitors. J Enzyme Inhib Med Chem. 2020;35(1):31–41. doi: 10.1080/14756366.2019.1680659. DOI: 10.1080/14756366.2019.1680659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Abdullaha M, Ali M, Kour D, Kumar A, Bharate SB. Discovery of benzo[cd]indol-2-one and benzylidene-thiazolidine-2,4-dione as new classes of NLRP3 inflammasome inhibitors via ER-beta structure based virtual screening. Bioorg Chem. 2020;95(103500):1–22. doi: 10.1016/j.bioorg.2019.103500. DOI: 10.1016/j.bioorg.2019.103500. [DOI] [PubMed] [Google Scholar]
- 20.Petrache AL, Khan AA, Nicholson MW, Monaco A, Kuta-Siejkowska M, Haider S, et al. Selective modulation of alpha5 GABAA receptors exacerbates aberrant inhibition at key hippocampal neuronal circuits in APP mouse model of Alzheimer's disease. Front Cell Neurosci. 2020;14(568194):1–18. doi: 10.3389/fncel.2020.568194. DOI: 10.3389/fncel.2020.568194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ghotbi G, Mahdavi M, Najafi Z, Moghadam FH, Hamzeh-Mivehroud M, Davaran S, et al. Design, synthesis, biological evaluation, and docking study of novel dual-acting thiazole-pyridiniums inhibiting acetylcholinesterase and β-amyloid aggregation for Alzheimer's disease. Bioorg Chem. 2020;103(104186):1–15. doi: 10.1016/j.bioorg.2020.104186. DOI: 10.1016/j.bioorg.2020.104186. [DOI] [PubMed] [Google Scholar]
- 22.Jiang X, Zhou J, Wang Y, Chen L, Duan Y, Huang J, et al. Rational design and biological evaluation of a new class of thiazolopyridyl tetrahydroacridines as cholinesterase and GSK-3 dual inhibitors for Alzheimer's disease. Eur J Med Chem. 2020;207(112751):1–16. doi: 10.1016/j.ejmech.2020.112751. DOI: 10.1016/j.ejmech.2020.112751. [DOI] [PubMed] [Google Scholar]
- 23.Noori MS, Bhatt PM, Courreges MC, Ghazanfari D, Cuckler C, Orac CM, et al. Identification of a novel selective and potent inhibitor of glycogen synthase kinase-3. Am J Physiol Cell Physiol. 2019;317(6):C1289–C1303. doi: 10.1152/ajpcell.00061.2019. DOI: 10.1152/ajpcell.00061.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Salehi N, Mirjalili BBF, Nadri H, Abdolahi Z, Forootanfar H, Samzadeh-Kermani A, et al. Synthesis and biological evaluation of new N-benzylpyridinium-based benzoheterocycles as potential anti-Alzheimer's agents. Bioorg Chem. 2019;83:559–568. doi: 10.1016/j.bioorg.2018.11.010. DOI: 10.1016/j.bioorg.2018.11.010. [DOI] [PubMed] [Google Scholar]
- 25.Jonczyk J, Lodarski K, Staszewski M, Godyn J, Zareba P, Soukup O, et al. Search for multifunctional agents against Alzheimer's disease among non-imidazole histamine H3 receptor ligands. In vitro and in vivo pharmacological evaluation and computational studies of piperazine derivatives. Bioorg Chem. 2019;90(103084):1–39. doi: 10.1016/j.bioorg.2019.103084. DOI: 10.1016/j.bioorg.2019.103084. [DOI] [PubMed] [Google Scholar]
- 26.Selnick HG, Hess JF, Tang C, Liu K, Schachter JB, Ballard JE, et al. Discovery of MK-8719, a potent O-GlcNAcase inhibitor as a potential treatment for tauopathies. J Med Chem. 2019;62(22):10062–10097. doi: 10.1021/acs.jmedchem.9b01090. DOI: 10.1021/acsjmedchem.9b01090. [DOI] [PubMed] [Google Scholar]
- 27.Sagar SR, Singh DP, Das RD, Panchal NB, Sudarsanam V, Nivsarkar M, et al. Pharmacological investigation of quinoxaline-bisthiazoles as multitarget-directed ligands for the treatment of Alzheimer's disease. Bioorg Chem. 2019;89(102992):1–43. doi: 10.1016/j.bioorg.2019.102992. DOI: 10.1016/j.bioorg.2019.102992. [DOI] [PubMed] [Google Scholar]
- 28.Rajeshwari R, Chand K, Candeias E, Cardoso SM, Chaves S, Santos MA. New multitarget hybrids bearing tacrine and phenylbenzothiazole motifs as potential drug candidates for Alzheimer's disease. Molecules. 2019;24(3):1–15. doi: 10.3390/molecules24030587. 587. DOI: 10.3390/molecules24030587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Aitken L, Benek O, McKelvie BE, Hughes RE, Hroch L, Schmidt M, et al. Novel benzothiazole-based ureas as 17beta-HSD10 inhibitors, a potential Alzheimer's disease treatment. Molecules. 2019;24(15):1–25. doi: 10.3390/molecules24152757. 2757. DOI: 10.3390/molecules24152757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gandini A, Bartolini M, Tedesco D, Martinez-Gonzalez L, Roca C, Campillo NE, et al. Tau-centric multitarget approach for Alzheimer's disease: development of first-in-class dual glycogen synthase kinase 3beta and tau-aggregation inhibitors. J Med Chem. 2018;61(17):7640–7656. doi: 10.1021/acs.jmedchem.8b00610. DOI: 10.1021/acsjmedchem.8b00610. [DOI] [PubMed] [Google Scholar]
- 31.Sagar SR, Singh DP, Panchal NB, Das RD, Pandya DH, Sudarsanam V, et al. Thiazolyl-thiadiazines as beta site amyloid precursor protein cleaving enzyme-1 (BACE-1) inhibitors and anti-inflammatory agents: multitarget-directed ligands for the efficient management of Alzheimer's Disease. ACS Chem Neurosci. 2018;9(7):1663–1679. doi: 10.1021/acschemneuro.8b00063. DOI: 10.1021/acschemneuro.8b00063. [DOI] [PubMed] [Google Scholar]
- 32.Yan G, Hao L, Niu Y, Huang W, Wang W, Xu F, et al. 2-Substituted-thio-N-(4-substituted-thiazol/1H-imidazol-2-yl)acetamides as BACE1 inhibitors: synthesis, biological evaluation and docking studies. Eur J Med Chem. 2017;137:462–475. doi: 10.1016/j.ejmech.2017.06.020. DOI: 10.1016/j.ejmech.2017.06.020. [DOI] [PubMed] [Google Scholar]
- 33.Shidore M, Machhi J, Shingala K, Murumkar P, Sharma MK, Agrawal N, et al. Benzylpiperidine-linked diarylthiazoles as potential anti-Alzheimer's agents: synthesis and biological evaluation. J Med Chem. 2016;59(12):5823–5846. doi: 10.1021/acs.jmedchem.6b00426. DOI: 10.1021/acs.jmedchem.6b00426. [DOI] [PubMed] [Google Scholar]
- 34.Luo J, Lee SH, VandeVrede L, Qin Z, Ben Aissa M, Larson J, et al. A multifunctional therapeutic approach to disease modification in multiple familial mouse models and a novel sporadic model of Alzheimer's disease. Mol Neurodegener. 2016;11(35):1–14. doi: 10.1186/s13024-016-0103-6. DOI: 10.1186/s13024-016-0103-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sun ZQ, Tu LX, Zhuo FJ, Liu SX. Design and discovery of novel thiazole acetamide derivatives as anticholinesterase agent for possible role in the management of Alzheimer's. Bioorg Med Chem Lett. 2016;26(3):747–750. doi: 10.1016/j.bmcl.2016.01.001. DOI: 10.1016/j.bmcl.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 36.Turan-Zitouni G, Ozdemir A, Kaplancikli ZA, Altintop MD, Temel HE, Ciftci GA. Synthesis and biological evaluation of some thiazole derivatives as new cholinesterase inhibitors. J Enzyme Inhib Med Chem. 2013;28(3):509–514. doi: 10.3109/14756366.2011.653355. DOI: 10.3109/14756366.2011.653355. [DOI] [PubMed] [Google Scholar]
- 37.Keri RS, Quintanova C, Marques SM, Esteves AR, Cardoso SM, Santos MA. Design, synthesis and neuroprotective evaluation of novel tacrine-benzothiazole hybrids as multi-targeted compounds against Alzheimer's disease. Bioorg Med Chem. 2013;21(15):4559–4569. doi: 10.1016/j.bmc.2013.05.028. DOI: 10.1016/j.bmc.2013.05.028. [DOI] [PubMed] [Google Scholar]
- 38.Geng J, Li M, Wu L, Ren J, Qu X. Liberation of copper from amyloid plaques: making a risk factor useful for Alzheimer's disease treatment. J Med Chem. 2012;55(21):9146–9155. doi: 10.1021/jm3003813. DOI: 10.1021/jm3003813. [DOI] [PubMed] [Google Scholar]
- 39.Wang Y, Wang F, Yu JP, Jiang FC, Guan XL, Wang CM, et al. Novel multipotent phenylthiazole-tacrine hybrids for the inhibition of cholinesterase activity, beta-amyloid aggregation and Ca2+ overload. Bioorg Med Chem. 2012;20(21):6513–6522. doi: 10.1016/j.bmc.2012.08.040. DOI: 10.1016/j.bmc.2012.08.040. [DOI] [PubMed] [Google Scholar]
- 40.Lagoja I, Pannecouque C, Griffioen G, Wera S, Rojasdelaparra VM, Van Aerschot A. Substituted 2-aminothiazoles are exceptional inhibitors of neuronal degeneration in tau-driven models of Alzheimer's disease. Eur J Pharm Sci. 2011;43(5):386–392. doi: 10.1016/j.ejps.2011.05.014. DOI: 10.1016/j.ejps.2011.05.014. [DOI] [PubMed] [Google Scholar]
- 41.Zare-Zardini H, Ferdowsian F, Soltaninejad H, Ghorani Azam A, Soleymani S, Zare-Shehneh M, et al. Application of nanotechnology in biomedicine: a major focus on cancer therapy. J Nano Res. 2015;35:55–66. DOI: 10.4028/www.scientific. net/JNanoR. 35.55. [Google Scholar]
- 42.Shiradkar MR, Akula KC, Dasari V, Baru V, Chiningiri B, Gandhi S, et al. Clubbed thiazoles by MAOS: a novel approach to cyclin-dependent kinase 5/p25 inhibitors as a potential treatment for Alzheimer's disease. Bioorg Med Chem. 2007;15(7):2601–2610. doi: 10.1016/j.bmc.2007.01.043. DOI: 10.1016/j.bmc.2007.01.043. [DOI] [PubMed] [Google Scholar]
- 43.Pickhardt M, Larbig G, Khlistunova I, Coksezen A, Meyer B, Mandelkow EM, et al. Phenylthiazolyl-hydrazide and its derivatives are potent inhibitors of tau aggregation and toxicity in vitro and in cells. Biochemistry. 2007;46(35):10016–10023. doi: 10.1021/bi700878g. DOI: 10.1021/bi700878g. [DOI] [PubMed] [Google Scholar]
- 44.Gulisano W, Maugeri D, Baltrons MA, Fa M, Amato A, Palmeri A, et al. Role of amyloid-beta and tau proteins in Alzheimer's disease: confuting the amyloid cascade. J Alzheimers Dis. 2018;64(s1):S611–S631. doi: 10.3233/JAD-179935. DOI: 10.3233/JAD-179935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pickett EK, Herrmann AG, McQueen J, Abt K, Dando O, Tulloch J, et al. Amyloid beta and tau cooperate to cause reversible behavioral and transcriptional deficits in a model of Alzheimer's disease. Cell Rep. 2019;29(11):3592–3604.e5. doi: 10.1016/j.celrep.2019.11.044. DOI: 10.1016/j.celrep.2019.11.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chen J, Fan A, Li S, Xiao Y, Fu Y, Chen JS, et al. APP mediates tau uptake and its overexpression leads to the exacerbated tau pathology. Cell Mol Life Sci. 2023;80(5):123. doi: 10.1007/s00018-023-04774-z. DOI: 10.1007/s00018-023-04774-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cole SL, Vassar R. The Alzheimer's disease beta-secretase enzyme, BACE1. Mol Neurodegener. 2007;2(22):1–25. doi: 10.1186/1750-1326-2-22. DOI: 10.1186/1750-1326-2-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chico LK, Van Eldik LJ, Watterson DM. Targeting protein kinases in central nervous system disorders. Nat Rev Drug Discov. 2009;8(11):892–909. doi: 10.1038/nrd2999. DOI: 10.1038/nrd2999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Masand N, Gupta SP, Khosa RL, Patil VM. Heterocyclic secretase inhibitors for the treatment of Alzheimer's disease: an overview. Cent Nerv Syst Agents Med Chem. 2017;17(1):3–25. DOI: 10.2174/1570159X13666151029105752. [PubMed] [Google Scholar]
- 50.Sharma PC, Sinhmar A, Sharma A, Rajak H, Pathak DP. Medicinal significance of benzothiazole scaffold: an insight view. J Enzyme Inhib Med Chem. 2012;28(2):240–266. doi: 10.3109/14756366.2012.720572. DOI: 10.3109/14756366.2012.720572. [DOI] [PubMed] [Google Scholar]
- 51.Iqbal J, al-Rashida M, Babar A, Hameed A, Khan MS, Munawar MA, et al. Cholinesterase inhibitory activities of N-phenylthiazol-2-amine derivatives and their molecular docking studies. Med Chem. 2015;11(5):489–496. doi: 10.2174/1573406411666141230104536. DOI: 10.2174/1573406411666141230104536. [DOI] [PubMed] [Google Scholar]
- 52.Tang W, Lin C, Yu Q, Zhang D, Liu Y, Zhang L, et al. novel medicinal chemistry strategies targeting CDK5 for Drug Discovery. J Med Chem. 2023;66(11):7140–7161. doi: 10.1021/acs.jmedchem.3c00566. DOI: 10.1021/acsjmedchem.3c00566. [DOI] [PubMed] [Google Scholar]
- 53.Rodriguez-Rodriguez C, de Groot SN, Rimola A, Alvarez-Larena A, Lloveras V, Vidal-Gancedo J, et al. Design, selection, and characterization of thioflavin-based intercalation compounds with metal chelating properties for application in Alzheimer's disease. J Am Chem Soc. 2009;131(4):1436–1451. doi: 10.1021/ja806062g. DOI: 10.1021/ja806062g. [DOI] [PubMed] [Google Scholar]
- 54.Ahmadi A, Roghani M, Noori S, Nahri-Niknafs B. Substituted aminobenzothiazole derivatives of tacrine: synthesis and study on learning and memory impairment in scopolamine-induced model of amnesia in rat. Mini Rev Med Chem. 2019;19(1):72–78. doi: 10.2174/1389557518666180716122608. DOI: 10.2174/1389557518666180716122608. [DOI] [PubMed] [Google Scholar]
- 55.Demirayak Ş, Şahin Z, Ertaş M, F Bülbül E, Bender C, Biltekin S N, et al. Novel thiazole-piperazine derivatives as potential cholinesterase inhibitors. J Heterocycl Chem. 2019;56(12):3370–3386. DOI: 10.1002/jhet.3734. [Google Scholar]
- 56.Raza R, Saeed A, Arif M, Mahmood S, Muddassar M, Raza A, et al. Synthesis and biological evaluation of 3-thiazolocoumarinyl Schiff-base derivatives as cholinesterase inhibitors. Chem Biol Drug Des. 2012;80(4):605–615. doi: 10.1111/j.1747-0285.2012.01435.x. DOI: 10.1111/j.1747-0285.2012.01435.x. [DOI] [PubMed] [Google Scholar]
- 57.Ghafoorianfar S, Ghorani-Azam A, Mohajeri SA, Farzin D. Efficiency of nanoparticles for treatment of ocular infections: systematic literature review. J Drug Deliv Sci Technol. 2020;57(101765):1–22. DOI: 10.1016/jjddst.2020.101765. [Google Scholar]
- 58.Mohseni S, Aghayan M, Ghorani-Azam A, Behdani M, Asoodeh A. Evaluation of antibacterial properties of barium zirconate titanate (BZT) nanoparticle. Braz J Microbiol. 2014;45(4):1393–1399. doi: 10.1590/s1517-83822014000400033. DOI: 10.1590/s1517-83822014000400033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sanchis I, Spinelli R, Aschemacher N, Humpola MV, Siano A. Acetylcholinesterase inhibitory activity of a naturally occurring peptide isolated from Boana pulchella (Anura: Hylidae) and its analogs. Amino Acids. 2020;52(3):387–396. doi: 10.1007/s00726-019-02815-1. DOI: 10.1007/s00726-019-02815-1. [DOI] [PubMed] [Google Scholar]
- 60.Riahi-Zanjani B, Balali-Mood M, Es'haghi Z, Asoodeh A, Ghorani-Azam A. Molecular modeling and experimental study of a new peptide-based microextraction fiber for preconcentrating morphine in urine samples. J Mol Model. 2019;25(3):1–12. doi: 10.1007/s00894-019-3925-7. 25. DOI: 10.1007/s00894-019-3925-7. [DOI] [PubMed] [Google Scholar]
- 61.Asoodeh A, Sepahi S, Ghorani-Azam A. Purification and modeling amphipathic alpha helical antimicrobial peptides from skin secretions of Euphlyctis cyanophlyctis. Chem Biol Drug Des. 2014;83(4):411–417. doi: 10.1111/cbdd.12256. DOI: 10.1111/cbdd. 12256. [DOI] [PubMed] [Google Scholar]
- 62.Ghorani-Azam A, Balali-Mood M, Aryan E, Karimi G, Riahi-Zanjani B. Effect of amino acid substitution on biological activity of cyanophlyctin-p and brevinin-2R. J Mol Struct. 2018;1158:14–18. DOI: 10.1016/j.molstruc.2018.01.015. [Google Scholar]
- 63.Baig MH, Ahmad K, Rabbani G, Choi I. Use of peptides for the management of Alzheimer's disease: diagnosis and inhibition. Front Aging Neurosci. 2018;10(21):1–6. doi: 10.3389/fnagi.2018.00021. DOI: 10.3389/fnagi.2018.00021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nepovimova E, Svobodova L, Dolezal R, Hepnarova V, Junova L, Jun D, et al. Tacrine-benzothiazoles: novel class of potential multitarget anti-Alzheimers drugs dealing with cholinergic, amyloid and mitochondrial systems. Bioorg Chem. 2021;107(104596):1–19. doi: 10.1016/j.bioorg.2020.104596. DOI: 10.1016/j.bioorg.2020.104596. [DOI] [PubMed] [Google Scholar]


