Abstract
Background and Objectives
Surgery is widely performed for refractory epilepsy in patients with Sturge-Weber syndrome (SWS), but reports on its effectiveness are limited. This study aimed to analyze seizure, motor, and cognitive outcomes of surgery in these patients and to identify factors associated with the outcomes.
Methods
This was a multicenter retrospective observational study using data from patients with SWS and refractory epilepsy who underwent epilepsy surgery between 2000 and 2020 at 16 centers throughout China. Longitudinal postoperative seizures were classified by Engel class, and Engel class I was regarded as seizure-free outcome. Functional (motor and cognitive) outcomes were evaluated using the SWS neurologic score, and improved or unchanged scores between baseline and follow-up were considered to have stable outcomes. Outcomes were analyzed using Kaplan-Meier analyses. Multivariate Cox regression was used to identify factors associated with outcomes.
Results
A total of 214 patients with a median age of 2.0 (interquartile range 1.2–4.6) years underwent surgery (focal resection, FR [n = 87]; hemisphere surgery, HS [n = 127]) and completed a median of 3.5 (1.7–5.0) years of follow-up. The overall estimated probability for being seizure-free postoperatively at 1, 2, and 5 years was 86.9% (95% CI 82.5–91.6), 81.4% (95% CI 76.1–87.1), and 70.7% (95% CI 63.3–79.0), respectively. The overall estimated probability of being motor stable at the same time post operatively was 65.4% (95% CI 58.4–71.2), 80.2% (95% CI 73.8–85.0), and 85.7% (95% CI 79.5–90.1), respectively. The overall probability for being cognition stable at 1, 2, and 5 years was 80.8% (95% CI 74.8–85.5), 85.1% (95% CI 79.3–89.2), and 89.5% (95% CI 83.8–93.2), respectively. Both FR and HS were effective at ensuring seizure control. For different HS techniques, modified hemispherotomy had comparable outcomes but improved safety compared with anatomical hemispherectomy. Regarding FR, partial resection (adjusted hazard ratio [aHR] 11.50, 95% CI 4.44–29.76), acute postoperative seizure (APOS, within 30 days of surgery; aHR 10.33, 95% CI 3.94–27.12), and generalized seizure (aHR 3.09, 95% CI 1.37–6.94) were associated with seizure persistence. For HS, seizure persistence was associated with APOS (aHR 27.61, 9.92–76.89), generalized seizure (aHR 7.95, 2.74–23.05), seizure frequency ≥30 times/month (aHR 4.76, 1.27–17.87), and surgical age ≥2 years (aHR 3.78, 1.51–9.47); motor stability was associated with severe motor defects (aHR 5.23, 2.27–12.05) and postoperative seizure-free status (aHR 3.09, 1.49–6.45); and cognition stability was associated with postoperative seizure-free status (aHR 2.84, 1.39–5.78) and surgical age <2 years (aHR 1.76, 1.13–2.75).
Discussion
FR is a valid option for refractory epilepsy in patients with SWS and has similar outcomes to those of HS, with less morbidity associated with refractory epilepsy. Early surgical treatment (under the age of 2 years) leads to better outcomes after HS, but there is insufficient evidence that surgical age affects FR outcomes. These findings warrant future prospective multicenter cohorts with international cooperation and prolonged follow-up in better exploring more precise outcomes and developing prognostic predictive models.
Classification of Evidence
This study provides Class IV evidence that in children with SWS and refractory seizures, surgical resection—focal, hemispherectomy, or modified hemispherotomy—leads to improved outcomes.
Introduction
Sturge-Weber syndrome (SWS) is a rare congenital neurocutaneous disorder1 characterized by port-wine stains and other vascular malformations, resulting in intracranial (leptomeningeal) and/or cutaneous (mainly facial) capillary malformations and glaucoma.2 Epilepsy is the most common associated neurologic deficit and occurs in more than 70% of patients with SWS.3,4 Notably, approximately 50% of patients with SWS develop refractory epilepsy, and antiseizure medications (ASMs) cannot adequately control these seizures.3,5,6 Medically intractable seizures are associated with poor motor and cognitive functions and affect patients with SWS, families, and society.7,8
Epilepsy surgery is widely performed for patients with SWS with refractory epilepsy.9,10 Although several studies have reported surgical outcomes, the low incidence of this disease has resulted in small case numbers with limited follow-up periods.9-13 Our previous single-center study described the experiences of 90 surgical patients with SWS.14 To date, this is the largest study on this topic.15 However, this study included a limited sample size of patients who underwent different surgeries, thus affecting its ability to identify factors associated with outcomes; moreover, the results might be limited regarding applicability across different centers. Due to the lack of available data, only 2-year follow-up data were reported, and factors associated with functional outcomes were not analyzed. This multicenter study was conducted to analyzed longitudinal seizure, motor, and cognitive outcomes and to identify factors associated with the outcomes.
Methods
We formed a national multicenter collaboration (SWS-Chinese Collaboration) and conducted this large-scale retrospective longitudinal study on epilepsy surgery for SWS. In this study, we analyzed seizure, motor, and cognitive outcomes at 1, 2, and 5 years and identified demographic and clinical factors associated with surgical outcomes, thus comprehensively providing novel findings regarding this rare disorder. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology checklist.
Participants
The study sample size was calculated for Cox regression16 (with a statistical power of 90%) using PASS software, version 2021, which required at least 58 valid samples per study group. Subsequently, we appealed for participating centers at national China Association Against Epilepsy (CAAE) conferences and additionally through personal contacts. A total of 36 centers from all regions of China17 with claimed possible patients with SWS from 2000 to 2020 were surveyed. Teams of senior neurologists and neurosurgeons reviewed all medical records within the study periods from these centers and received confirmation of SWS diagnoses and epilepsy surgeries9,14,18 from 16 included centers.
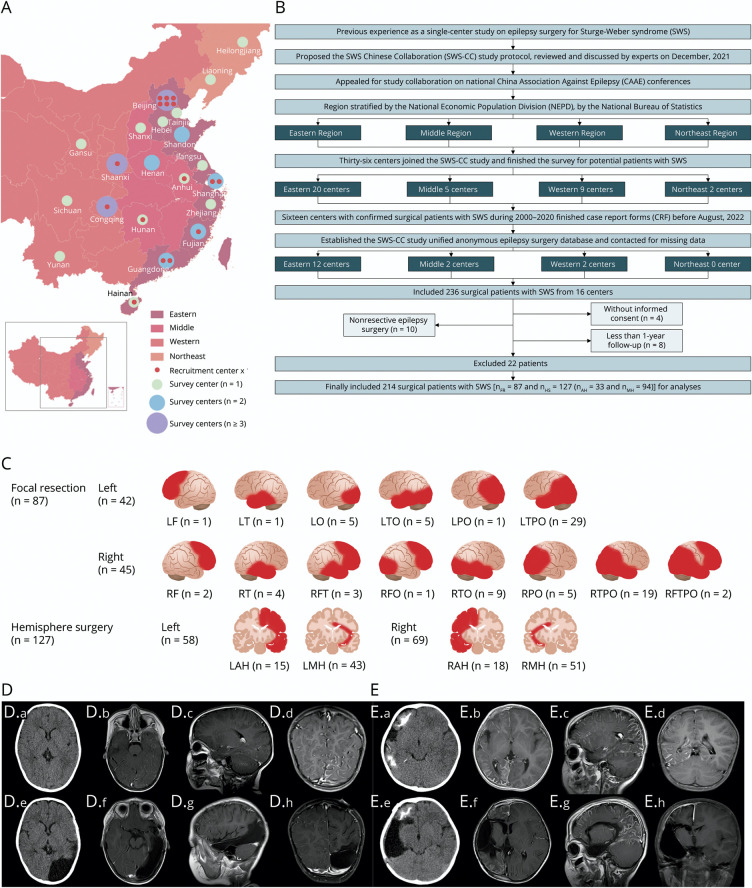
All patients underwent evaluations and surgeries at their local centers. Patients were not referred to coordinated sites. Consecutive case report forms and medical records were screened, collected, and reassessed by the study teams. A unified database was established to enter individual anonymous data. The inclusion criteria were as follows: (1) clinical diagnosis of SWS,9,14,18 with pathologic verifications, if possible; (2) resective epilepsy surgery (i.e., focal resection [FR] or hemisphere surgery [HS]; divided into 2 techniques, anatomical hemispherectomy [AH] and modified/functional hemispherotomy [MH]) from 2000 to 2020; and (3) completion of at least 1 year of follow-up. Patients who provided informed consent and who underwent only nonresective surgeries were excluded. For patients with inadequate follow-up periods or missing data, contact was prioritized for further interviews, and those who could not be contacted were excluded. The 90 patients who were previously included in our single-center study were also included.14 The participant centers, study design, and participant inclusion procedure are shown in Figure 1.
Figure 1. Graphical Representation of Study Design, Participant Inclusion Process, and Surgery Types.
(A) Survey (n = 36) and inclusion (n = 16) centers; (B) study design and participant inclusion process; (C) surgical types and areas of all included patients with SWS (N = 214); (D–E) pre- and postsurgical neuroimaging of 2 patients with SWS who underwent focal resection and hemisphere surgery, respectively. LAH = left anatomical hemispherectomies (n = 15); LF = left frontal resection (n = 1); LMH = left modified hemispherotomy (n = 43); LO = left occipital resection (n = 5); LPO = left parieto-occipital resection (n = 1); LT = left temporal resection (n = 1); LTO = left temporo-occipital resection (n = 5); LTPO = left temporo-parieto-occipital resection (n = 29); RAH = right anatomical hemispherectomy (n = 18); RF = right frontal resection (n = 2); RFO = right fronto-occipital resection (n = 1); RFT = right fronto-temporal resection (n = 3); RFTPO = right fronto-temporo-parieto-occipital resection (n = 2); RMH = right modified hemispherotomy (n = 51); RPO = right parieto-occipital resection (n = 5); RT = right temporal resection (n = 4); RTO = right temporo-occipital resection (n = 9); RTPO = right temporo-parieto-occipital resection (n = 19); SWS = Sturge-Weber syndrome. (D) Preoperative and postoperative CT and MR images of a patient with SWS who underwent focal resection (LTO) and E, Preoperative and postoperative CT and MR images of a patient with SWS who underwent hemisphere surgery (RMH). (D-i and E-i) preoperative CT (axial image); (D-ii and E-ii) preoperative MRI (enhanced T1WI, axial image); (D-iii and E-iii) preoperative MRI (enhanced T1WI, sagittal image); (D-iv and E-iv) preoperative MRI (enhanced T1WI, coronal image); (D-v and E-v) postoperative CT (axial image); (D-vi and E-vi) postoperative MRI (enhanced T1WI, axial image); (D-vii and E-vii) postoperative MRI (enhanced T1WI, sagittal image); (D-viii and E-viii) postoperative MRI (enhanced T1WI, coronal image).
Assessments and Epilepsy Surgeries
Preoperative demographic characteristics, seizure records, neuroimaging, EEG, and functional assessments and conditions were collected. Two evaluation teams with experience in related fields independently performed the data extraction, assessments, and secondary reviews for each record. The teams did not know the surgical procedure and were only responsible for either presurgical or postsurgical records for reducing observer bias.
Seizure type, status epilepticus (SE), and seizure clustering (SC) were classified according to the 2017 International League Against Epilepsy (ILAE) classification,19 the 2015 ILAE definition,20 and findings of a previous study,21 respectively. For this surgical cohort, all patients with SC were confirmed to have refractory seizures, thus excluding seizure-free patients following SC. Available neuroimaging and EEG findings were re-evaluated. The concordance of EEG and neuroimaging was defined as the localization of the same brain region or hemisphere. Because various scales have been used to assess neurologic functions, the widely used SWS neurologic score was developed by Kelley et al.,22 involving the hemiparesis (scored 0–4), cognition (scored 0–5), and seizure (scored 0–4) domains, which were retrospectively assigned for unification. Higher scores indicated more severe symptoms (worse functions). Assessments were performed twice by 2 independent experienced teams after carefully reviewing the original assessments and clinical condition of each patient, and their inconsistencies were resolved by joint discussions.
Each center has its own surgical criteria for SWS. However, our interviews suggested that the following surgical indications and principles in surgical type selection applied across these centers were similar to our previous study.14 The surgical indications required to performing epilepsy surgery for patients with SWS who had functional defects and refractory epilepsy for at least 6 months (patients with early refractory SE may have earlier surgeries to avoid catastrophic epilepsy) or performing surgery for patients without functional defects who failed in a prolonged 24-month ASM trial.9,11,14 In addition, surgical decisions should balance potential benefits and risks, and the wishes of patients and their guardians should be considered.9,11,14 In guiding selection of surgical types (FR or HS), the epileptogenic zone was mainly determined according to neuroimaging (CT and MRI) findings and semiology, while video-EEG and other techniques, such as PET, could also help in localization and lateralization.23,24 Epilepsy surgery was performed to resect the epileptogenic zone, which mainly contained the vascular malformations and adjacent calcified zones, which were ultimately determined by multidisciplinary discussions. For lesions involving only focal areas or several brain lobes, FR was performed, while unilateral hemisphere involvement was treated with HS. In this cohort, no patients with focal lesions underwent surgery with HS or vice versa. Patients with bilateral involvement did not undergo resective epilepsy surgery. The use of these principles in epilepsy surgery for patients with SWS also reached a consensus at CAAE conferences. We confirmed that all the included patients met these indications.9,11,14
The 2 surgical types FR and HS were regarded as the study groups. The 2 techniques, AH and MH, were regarded as subgroups of HS. Generally, FR was performed as a lesion or lobe(s) resection, while AH removed the whole affected hemisphere, and MH was modified from AH through removal and disconnection steps. The detailed typical procedures can be found in previous studies by the study team.11 In this study, among 87 FRs, 42 were performed on the left side, mainly including 29 temporo-parieto-occipital resections, 5 temporo-occipital resections, 5 occipital resections, and others, while the other 45 were performed on the right side, mainly including 19 temporo-parieto-occipital resections, 9 temporo-occipital resections, 5 parieto-occipital resections, and others. Among the 127 HSs, 58 were performed on the left side, including 43 MHs and 15 AHs, and 69 were performed on the right side, including 51 MHs and 18 AHs. The surgical types, surgical areas, and presurgical and postsurgical neuroimaging results are shown in Figure 1. Surgical duration, blood loss and transfusion, postoperative hospital stay, complications and treatment, and acute postoperative seizures (APOSs; within 30 days after surgery, ILAE definition25) were recorded. For patients who underwent FR, specialists in neurosurgery and neuroimaging from the study team rereviewed all the preoperative, intraoperative, and postoperative records and postoperative images to determine whether lesions (vascular malformations, adjacent calcifications, or other identified epileptogenic zones) were fully resected.
Surgical Outcomes
The included centers were required to provide seizure outcome information regarding Engel class,25 ASM use, functional assessments, and conditions at 6 months and at each year of follow-up. Seizure outcomes were categorized as seizure-free (i.e., Engel class I) or seizure persistence (i.e., Engel classes II–IV). Compared with presurgical ASMs, ASM reduction was defined as dose reduction or partial discontinuation, and ASM discontinuation was considered discontinuation of all ASMs used (completely seizure-free without ASMs). Patients with improved (decreased) or unchanged (unchanged) scores in the hemiparesis and cognition domains of the SWS neurologic scores between baseline and follow-up were considered to have motor stable and cognition stable outcomes.
Statistical Analysis
The study protocol and statistical analysis plan are available in eSAP 1. The statistical data (number [percentage], median [interquartile range, IQR], or mean [SD]) and tests were determined based on the variable type, distribution (Shapiro-Wilk or Kolmogorov-Smirnov test), and variance (Levene test). Differences were tested with the 2 independent sample t test, Mann-Whitney U test, paired sample t test, Wilcoxon signed-rank test, Pearson χ2 test, or Fisher exact test, as appropriate. Correlations were tested by the Spearman correlation analysis.
Due to the wide range of follow-up periods (1–10), time-to-event and Kaplan-Meier analyses were used to estimate the probability of remaining seizure-free and the cumulative probabilities of recovering to motor stable and cognition stable status at 1, 2, and 5 years. Kaplan-Meier curves were plotted with log-rank tests for comparisons. Cox proportional hazards regression was applied to identify factors associated with the outcomes. The Youden index was used to stratify continuous variables (e.g., age) into categorical variables. Potential factors with p < 0.10 in univariable analyses were subsequently entered into multivariable analyses (in a backward fashion) for adjustment effects. The adjusted hazard ratio (aHR) and its 95% CI were reported for the independent factors.
All the statistical tests were 2-tailed, and the significance level was set as α = 0.05. A p value <0.05 was considered to indicate statistical significance. Unless otherwise noted post hoc, all analyses were planned. There were no incomplete values of major variables in this study. Statistical analyses were performed using R software, version 4.2.2, and SPSS software, version 27.
Standard Protocol Approvals, Registrations, and Patient Consents
The SanBo Brain Hospital of Capital Medical University ethical standards committee approved this study (SBNK-YJ-2022-026-01). All included patients or their guardians prospectively provided informed consent for anonymous use of their clinical data for scientific research. This study was conducted in accordance with the tenets of the 1964 Declaration of Helsinki and its later amendments.
Data Availability
Data that support the findings of this study are included in the article/supplementary material.
Results
Baseline Characteristics
The baseline characteristics are summarized in Table 1. A total of 236 surgical patients were screened, and 22 patients were excluded due to a lack of informed consent (n = 4), nonresective epilepsy surgery (n = 10, all underwent only vagus nerve stimulation), or inadequate follow-up data (n = 8; 6 FRs and 2 AHs; none had seizures at the 6-month follow-up but could not be contacted further). eTable 1 summarizes the region divisions, provinces, survey and inclusion centers, and number of patients included from each center. No surgical or late mortality was reported.
Table 1.
Baseline Characteristics and Groups of the Included Patients With SWS (n = 214)
| Characteristic | Total (N = 214) | Study groups | HS subgroups | ||||
| FR (n = 87) | HS (n = 127) | p Value | AH (n = 33) | MH (n = 94) | p Value | ||
| Sex, n (%) | |||||||
| Male | 112 (52.3) | 49 (56.3) | 63 (49.6) | 0.33 | 18 (54.5) | 45 (47.9) | 0.51 |
| Female | 102 (47.7) | 38 (43.7) | 64 (50.4) | 15 (45.5) | 49 (52.1) | ||
| Seizure type, n (%)a | |||||||
| Focal-onset seizures | 189 (88.3) | 71 (81.6) | 118 (92.9) | 0.06 | 31 (93.9) | 87 (92.6) | 0.98 |
| Motor | 143 (66.8) | 57 (65.5) | 86 (67.7) | 23 (69.7) | 63 (67.0) | ||
| Nonmotor | 13 (6.1) | 4 (4.6) | 9 (7.1) | 3 (9.1) | 6 (6.4) | ||
| FBTCS | 45 (21.0) | 13 (14.9) | 32 (25.2) | 8 (24.2) | 24 (25.5) | ||
| Generalized-onset seizures, n (%) | 46 (21.5) | 29 (33.3) | 17 (13.4) | 4 (12.1) | 13 (13.8) | ||
| Motor | 41 (19.2) | 24 (27.6) | 17 (13.4) | 4 (12.1) | 13 (13.8) | ||
| Nonmotor | 10 (4.7) | 5 (5.7) | 5 (3.9) | 1 (3.0) | 4 (4.3) | ||
| Facial capillary malformations, n (%) | |||||||
| Presence | 191 (89.3) | 70 (80.5) | 121 (95.3) | <0.001b | 33 (100.0) | 88 (93.6) | 0.31 |
| Absence | 23 (10.7) | 17 (19.5) | 6 (4.7) | 0 (0.0) | 6 (6.4) | ||
| Glaucoma, n (%) | |||||||
| Presence | 84 (39.3) | 41 (47.1) | 43 (33.9) | 0.15 | 19 (57.6) | 24 (25.5) | 0.003b |
| Absence | 101 (47.2) | 36 (41.4) | 65 (51.2) | 12 (36.4) | 53 (56.4) | ||
| Unknown | 29 (13.6) | 10 (11.5) | 19 (15.0) | 2 (6.1) | 17 (18.1) | ||
| Motor defects, n (%) | |||||||
| Presence | 130 (60.7) | 9 (10.3) | 121 (95.3) | <0.001b | 31 (93.9) | 90 (95.7) | >0.999 |
| Absence | 84 (39.3) | 78 (89.7) | 6 (4.7) | 2 (6.1) | 4 (4.3) | ||
| Cognitive impairment, n (%) | |||||||
| Presence | 146 (68.2) | 30 (34.5) | 116 (91.3) | <0.001b | 28 (84.8) | 88 (93.6) | 0.24 |
| Absence | 68 (31.8) | 57 (65.5) | 11 (8.7) | 5 (15.2) | 6 (6.4) | ||
| Seizure onset, y, median (IQR) | 0.4 (0.3–0.9) | 0.7 (0.3–2.0) | 0.3 (0.2–0.6) | <0.001b | 0.3 (0.2–0.8) | 0.3 (0.2–0.5) | 0.90 |
| Seizure duration, y, median (IQR) | 1.5 (0.9–3.7) | 3.2 (1.3–10.0) | 1.3 (0.8–2.0) | <0.001b | 1.3 (0.9–2.0) | 1.2 (0.7–2.0) | 0.42 |
| Age at surgery, y, median (IQR) | 2.0 (1.2–4.6) | 4.6 (1.9–13.0) | 1.6 (1.0–2.8) | <0.001b | 1.7 (1.2–2.6) | 1.6 (1.0–2.8) | 0.51 |
| Follow-up, y, median (IQR) | 3.5 (1.7–5.0) | 4.0 (1.5–6.0) | 3.0 (1.7–5.0) | 0.09 | 4.5 (3.2–6.5) | 2.6 (1.3–4.0) | <0.001b |
Abbreviations: AH = anatomical hemispherectomy; FBTCS = focal to bilateral tonic-clonic seizure; FR = focal resection; HS = hemisphere surgery; IQR = interquartile range; MH = modified hemispherotomy; SWS = Sturge-Weber syndrome.
Refer to eTable 3 for more details on the baseline characteristics (e.g., seizure types, antiepileptic drugs used, and MRI and EEG findings).
A patient might have multiple seizure types.
p < 0.01 (Mann-Whitney U test or Pearson χ2 test, as appropriate).
All 214 included patients (102 female patients, 47.7%) had confirmed SWS diagnoses.23 Eighty-seven (40.7%) of these patients underwent FR, while the other 127 (59.3%) underwent HS (33 AHs and 94 MHs). The median seizure onset age was 0.4 (IQR 0.3–0.9) years. Most patients (189, 88.3%) had focal seizures, while 21.5% (46) had generalized seizures. A median of 2 (IQR 2–3) types of ASM were observed before epilepsy surgery, and oxcarbazepine (98, 45.8%) and levetiracetam (93, 43.8%) were the 2 most commonly used ASMs. Disease manifestations such as facial capillary malformations, glaucoma, motor defects, and cognitive impairment were observed in 191 (89.3%), 84 (39.3%), 130 (60.7%), and 146 (68.2%) patients, respectively. After a median of 1.5 (0.9–3.7, range 0.1–29.0) years of seizure duration, patients underwent surgery at an age of 2.0 (1.2–4.6, range 0.2–31.0) years, with 3.5 (1.7–5.0, range 1.0–10.0) years of follow-up.
The group that had FR had lower proportions of facial capillary malformations, motor defects, and cognitive impairment; older seizure onset age; longer seizure duration; and older surgical age than the group that had HS. Subgroup comparisons revealed that more individuals in the AH group had glaucoma than those in the MH group, and the follow-up period was longer.
Surgical Procedures
The details of the surgical procedures are summarized in Table 2. Overall, the median surgical duration was 388.0 (300.0–450.0) minutes, blood loss was 500.0 (295.0–632.5) mL, blood transfusion volume was 570.0 (300.0–752.5) mL, and postoperative hospital length of stay was 14.0 (10.0–19.0) days. Subgroup analyses suggested that patients with MH had a shorter surgical duration, less blood loss, a lower requirement for transfusion, and shorter postoperative hospital stays than patients with AH.
Table 2.
Surgical Procedure; Postoperative Seizure, Motor, and Cognitive Outcomes Estimated as the 1-, 2-, and 5-Year Probabilities and at the Last Follow-Up; and Postoperative Complications of the Included Patients With SWS (n = 214)
| Subject | Total (N = 214) | Study group | HS subgroup | |||
| FR (n = 87) | HS (n = 127) | AH (n = 33) | MH (n = 94) | p Value | ||
| Surgical procedure | ||||||
| Side of surgery, n (%) | ||||||
| Left | 100 (46.7) | 42 (48.3) | 58 (45.7) | 15 (25.9) | 43 (74.1) | 0.98 |
| Right | 114 (53.3) | 45 (51.7) | 69 (54.3) | 18 (26.1) | 51 (73.9) | |
| Duration of surgery, min, median (IQR) | 388.0 (300.0–450.0) | 360.0 (280.0–420.0) | 419.0 (310.0–460.2) | 450.0 (414.0–525.0) | 384.0 (270.0–445.3) | <0.001c |
| Blood loss, mL, median (IQR) | 500.0 (295.0–632.5) | 500.0 (260.0–600.0) | 500.0 (300.0–700.0) | 600.0 (375.0–800.0) | 400.0 (237.5–600.8) | 0.003c |
| Blood transfusion, mL, median (IQR) | 570.0 (300.0–752.5) | 525.0 (200.0–700.0) | 600.0 (350.0–840.0) | 825.0 (600.0–1,050.0) | 525.0 (300.0–725.0) | <0.001c |
| Postoperative length of hospital stay, d, median (IQR) | 14.0 (10.0–19.0) | 13.0 (9.0–19.0) | 15.0 (11.0–19.0) | 19.0 (14.5–23.5) | 13.5 (11.0–18.0) | <0.001c |
| Seizure outcomes | ||||||
| Estimated seizure-free probability, % (95% CI)a | ||||||
| 1 y | 86.9 (82.5–91.6) | 83.9 (76.5–92.0) | 89.0 (83.7–94.6) | 93.9 (86.1–100.0) | 87.2 (80.7–94.2) | 0.44 |
| 2 y | 81.4 (76.1–87.1) | 77.8 (69.1–87.6) | 83.9 (77.4–90.9) | 87.5 (76.7–99.8) | 82.7 (75.0–91.2) | |
| 5 y | 70.7 (63.3–79.0) | 61.7 (50.3–75.8) | 78.9 (70.9–87.8) | 83.8 (71.7–97.9) | 76.2 (65.3–88.9) | |
| Last follow-up, n (%) | ||||||
| Seizure-free (Engel class I) | 166 (77.6) | 61 (70.1) | 105 (82.7) | 28 (84.8) | 77 (81.9) | 0.46 |
| Engel class II | 33 (15.4) | 19 (21.8) | 14 (11.0) | 2 (6.1) | 12 (12.8) | |
| Engel class III | 14 (6.5) | 6 (6.9) | 8 (6.3) | 3 (9.1) | 5 (5.3) | |
| Engel class IV | 1 (0.5) | 1 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| ASM withdrawal | ||||||
| ASM reduction, n (%) | 140 (65.4) | 56 (64.4) | 84 (66.1) | 22 (66.7) | 62 (66.0) | 0.94 |
| Time to reduction, mo, median (IQR) | 16.0 (12.0–24.0) | 24.0 (12.0–24.0) | 14.5 (12.0–24.0) | 24.0 (12.0–24.0) | 12.0 (12.0–24.0) | 0.19 |
| ASM discontinuation, n (%) | 98 (45.8) | 39 (44.8) | 59 (46.5) | 17 (51.5) | 42 (44.7) | 0.50 |
| Time to discontinuation, mo, median (IQR) | 24.0 (24.0–29.3) | 24.0 (24.0–27.0) | 24.0 (24.0–36.0) | 24.0 (24.0–42.0) | 24.0 (24.0–28.5) | 0.49 |
| Motor outcomes | ||||||
| Estimated motor-stable probability, % (95% CI)b | ||||||
| 1 y | 65.4 (58.4–71.2) | 85.1 (75.3–90.9) | 52.0 (42.4–59.9) | 48.5 (28.3–63.0) | 53.2 (41.9–62.3) | 0.37 |
| 2 y | 80.2 (73.8–85.0) | 92.5 (84.1–96.5) | 71.7 (62.3–78.8) | 67.8 (46.7–80.5) | 73.1 (61.8–81.1) | |
| 5 y | 85.7 (79.5–90.1) | 94.4 (85.6–97.8) | 79.8 (70.4–86.2) | 74.2 (53.5–85.7) | 82.3 (70.4–89.4) | |
| Last follow-up, n (%) | ||||||
| Motor-stable | 176 (82.2) | 81 (93.1) | 95 (74.8) | 24 (72.7) | 71 (75.5) | 0.77 |
| Improvement | 70 (32.7) | 8 (9.2) | 62 (48.8) | 17 (51.5) | 45 (47.9) | |
| No change | 106 (49.5) | 73 (83.9) | 33 (26.0) | 7 (21.2) | 26 (27.7) | |
| Worsening | 38 (17.8) | 6 (6.9) | 32 (25.2) | 9 (27.3) | 23 (24.5) | |
| Cognitive outcomes | ||||||
| Estimated cognition-stable probability, % (95% CI)b | ||||||
| 1 y | 80.8 (74.8–85.5) | 88.5 (79.4–93.6) | 75.6 (66.9–82.0) | 66.7 (46.0–79.4) | 78.7 (68.6–85.6) | 0.45 |
| 2 y | 85.1 (79.3–89.2) | 89.9 (80.9–94.7) | 81.7 (73.4–87.4) | 76.7 (56.2–87.6) | 83.4 (73.7–89.5) | |
| 5 y | 89.5 (83.8–93.2) | 92.0 (82.5–96.3) | 87.5 (79.3–92.5) | 86.7 (67.2–94.6) | 86.9 (76.7–92.6) | |
| Last follow-up, n (%) | ||||||
| Cognition stable | 187 (87.4) | 79 (90.8) | 108 (85.0) | 28 (84.8) | 80 (85.1) | 0.999 |
| Improvement | 91 (42.5) | 14 (16.1) | 77 (60.6) | 20 (60.6) | 57 (60.6) | |
| No change | 96 (44.9) | 65 (74.7) | 31 (24.4) | 8 (24.2) | 23 (24.5) | |
| Worsening | 27 (12.6) | 8 (9.2) | 19 (15.0) | 5 (15.2) | 14 (14.9) | |
| Postoperative complications, n (%) | ||||||
| Presence | 25 (11.7) | 13 (14.9) | 12 (9.4) | 6 (18.2) | 6 (6.4) | 0.10 |
| Absence | 189 (88.3) | 74 (85.1) | 115 (90.6) | 27 (81.8) | 88 (93.6) | |
Abbreviations: AH = anatomical hemispherectomy; ASM = antiseizure medication; FR = focal resection; HS = hemisphere surgery; MH = modified hemispherotomy; SWS = Sturge-Weber syndrome.
1-, 2-, and 5-year seizure-free probabilities, motor-stable probabilities, and cognition-stable probabilities were estimated by Kaplan-Meier analysis.
Postoperative neurologic function subcategory motor function and cognitive function were evaluated by the SWS neurologic score by Kelley et al. with the hemiparesis domain and cognition domain of the scale, respectively.
p < 0.01 (Mann-Whitney U test, log-rank test, or Pearson χ2 test, as appropriate).
Surgical Outcomes
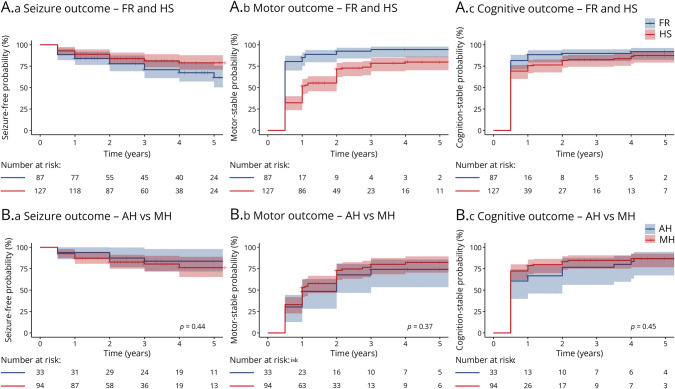
Surgical outcomes are summarized in Table 2, and Table 3 summarizes the original functional scales. A total of 214 (100%), 157 (73.4%), and 65 (30.4%) patients completed the 1, 2, and 5 years of follow-up, respectively. The estimated postoperative seizure-free probabilities were 86.9% (95% CI 82.5–91.6), 81.4% (76.1–87.1), and 70.7% (63.3–79.0) at 1, 2, and 5 years, respectively. At the last follow-up, 77.6% (166), 15.4% (33), 6.5% (14), and 0.5% (1) of patients were classified under Engel class I (seizure-free), class II (rare seizures), class III (worthwhile improvement), and class IV (no appreciable change), respectively. Furthermore, 65.4% (140) and 45.8% (98) of patients achieved ASM reduction and discontinuation, with median durations to reduction and discontinuation of 16.0 (12.0–24.0) and 24.0 (24.0–29.3) months, respectively. No significant differences were found among the different subgroups.
Table 3.
Original Scales for the Baseline and Postoperative Motor and Cognitive Function Assessments at the Last Follow-Up in Patients With SWS (n = 214)
| Scale/applicability | Focal resection (n = 87) | Hemisphere surgery (n = 127) | ||||||||
| Baseline | Postoperative | p Value | Baseline | Postoperative | p Value | |||||
| Number, n (%) | Score, mean (SD) | Number, n (%) | Score, mean (SD) | Number, n (%) | Score, mean (SD) | Number, n (%) | Score, mean (SD) | |||
| WPPSI-IV/2 y 6 mo to 6 y 11 mo | 9 (10.3) | 87.6 (5.9) | 3 (3.4) | 87.7 (10.0) | 0.98 | 14 (11.0) | 79.4 (10.2) | 4 (3.1) | 81.0 (17.6) | 0.81 |
| WISC-IV/6 y to 16 y | 23 (26.4) | 88.0 (9.3) | 37 (42.5) | 92.7 (8.3) | 0.048a | 5 (3.9) | 68.6 (13.6) | 42 (33.1) | 84.0 (11.5) | 0.008b |
| WAIS-IV/more than 16 y | 15 (17.2) | 79.7 (14.2) | 24 (27.6) | 85.8 (12.4) | 0.17 | 4 (3.1) | 50.8 (5.1) | 4 (3.1) | 65.5 (9.3) | 0.03a |
| DDST-II/1 mo to 6 y, n (%) | ||||||||||
| Normal | 25 (28.7) | 21 (84.0) | 10 (11.5) | 9 (90.0) | >0.999 | 61 (48.0) | 3 (4.9) | 30 (23.6) | 11 (36.7) | <0.001b |
| Suspect | 1 (4.0) | 1 (10.0) | 8 (13.1) | 7 (23.3) | ||||||
| Abnormal | 3 (12.0) | 50 (82.0) | 12 (40.0) | |||||||
| GMDS-III/0 to 6–8 y | 4 (4.6) | 83.4 (14.4) | 4 (4.6) | 94.3 (5.7) | 0.21 | 9 (7.1) | 65.5 (6.0) | 9 (7.1) | 85.3 (12.8) | <0.001b |
| BST/3 y to 12 y | 2 (2.3) | 91.0 (1.4) | 1 (1.1) | 95.0 (0.0) | 0.26 | 0 (0.0) | NA | 0 (0.0) | NA | NA |
Abbreviations: BST = the Binet-Simon Test; DDST-II = the Denver Developmental Screening Test, Second Edition, resulting in 3 interpretations, Normal, suspect, and abnormal; FSIQ = Full-Scale IQ; Griffiths III = the Griffiths Scales of Child Development, Third Edition, scored as the General Developmental Quotient (BST, scored as the IQ); SWS = Sturge-Weber syndrome; WAIS-IV = the Wechsler Adult Intelligence Scale—Fourth Edition, scored as the FSIQ; WISC-IV = the Wechsler Intelligence Scale for Children—Fourth Edition, scored as the FSIQ; WPPS-IV = the Wechsler Preschool and Primary Scale of Intelligence—Fourth Edition, scored as the FSIQ.
p < 0.05 (2 independent sample t test).
p < 0.01 (2 independent sample t test or Pearson χ2 test, as appropriate).
The estimated postoperative motor stable probabilities were 65.4% (58.4–71.2), 80.2% (73.8–85.0), and 85.7% (79.5–90.1) at 1, 2, and 5 years, respectively. The cognition stable probabilities at 1, 2, and 5 years were 80.8% (74.8–85.5), 85.1% (79.3–89.2), and 89.5% (83.8–93.2), respectively. At the last follow-up, 32.7% (70), 49.5% (106), and 17.8% (38) and 42.5% (91), 44.9% (96), and 12.6% (27) of patients had improved, unchanged, and worsened motor and cognitive outcomes, respectively. Patients who underwent FR, as assessed by the Wechsler Intelligence Scale for Children—Fourth Edition (WISC-IV), experienced considerable improvements, while patients who accepted HS, as assessed by the WISC-IV, the Wechsler Adult Intelligence Scale, the Denver Developmental Screening Test, and the Griffiths Scales of Child Development, also experienced considerable improvements. No significant difference was found between AH and MH. Figure 2 shows the Kaplan-Meier curves for the surgical outcomes, and eTable 2 summarizes and eFigure 1 shows the outcomes at different ages. In addition, older surgical age was positively correlated with worse preoperative motor (ρ = 0.37) and cognitive scores (ρ = 0.31) and worse postoperative cognitive scores (ρ = 0.31) in the group that had FR. Older age at surgery was positively correlated with worse preoperative motor scores (ρ = 0.28) and worse postoperative seizure (ρ = 0.18), motor (ρ = 0.22), and cognitive scores (ρ = 0.32) in the group that had HS (eFigure 2).
Figure 2. Estimated Seizure-Free, Motor-Stable, and Cognition-Stable Probabilities for the Study Groups and Subgroups.
Study groups: FR, n = 87; HS, n = 127; subgroups: AH, n = 33 vs MH, n = 94. All the statistical comparisons were performed by log-rank tests. AH = anatomical hemispherectomy; FR = focal resection; HS = hemisphere surgery; MH = modified hemispherotomy.
Complications were reported in 25 (11.7%) patients, including 5 with intracranial infections (3 FRs and 2 MHs), 6 with infected stitches (5 FRs and 1 MHs), 3 with central hyperthermia (2 AHs and 1 MHs), 3 with hydrocephalus (2 FRs and 1 AH), 2 with cerebral infarctions (MH), 2 with transient aphasias (FR), 1 with postoperative hemorrhage (AH), 1 with CSF fistula (AH), and 2 with deep venous thrombosis (1 FR and 1 AH). The proportions of complications in patients with FR, HS, AH, and MH were 14.9% (13), 9.4% (12), 18.2% (6), and 6.4% (6), respectively. These complications were treated by debridement, antibiotics, hypothermia, drainage, infarction resections, dehydrations, hematoma evacuation, fistula repairs, and anticoagulants. All these patients recovered well without sequelae.
Factors Associated With Surgical Outcomes
The results of the univariate analyses are summarized in eTable 3. After adjustment for multivariate effects (Table 4), partial resection (aHR 11.50, 95% CI 4.44–29.76), APOS (aHR 10.33, 3.94–27.12), and generalized seizures (aHR 3.09, 1.37–6.94) were associated with seizure-persistent status for FR. APOS (aHR 27.61, 9.92–76.89), generalized seizures (aHR 7.95, 2.74–23.05), seizure frequency ≥30 times per month (aHR 4.76, 1.27–17.87), and surgical age ≥2 years (aHR 3.78, 1.51–9.47) were associated with seizure-persistent status for HS. Severe (motor score ≥3, moderate-to-severe impaired fine and gross motor function) preoperative motor defects (aHR 5.23, 2.27–12.05) and postoperative seizure-free status (aHR 3.09, 1.49–6.45) were associated with motor stable status for HS. Postoperative seizure-free status (aHR 2.84, 1.39–5.78) and surgical age <2 years (aHR 1.76, 1.13–2.75) were associated with cognition stable status for HS. Figure 3 shows the analyses of potential factors associated with the outcomes.
Table 4.
Univariate and Multivariate Analyses of Factors Associated With Postoperative Seizure, Motor, and Cognitive Outcomes in Patients With SWS (n = 214)
| Factors | Univariate analysis | Multivariate analysis | ||
| HR (95% CI) | p Value | Adjusted HR (95% CI) | p Value | |
| Focal resection (n = 87) | ||||
| Seizure outcomes/seizure persistence | ||||
| Age at surgery | ||||
| <6 y | Reference | Reference | ||
| ≥6 y | 2.27 (1.03–5.00) | 0.04c | 1.62 (0.67–3.92) | 0.29 |
| Seizure type | ||||
| Focal seizures only | Reference | Reference | ||
| Generalized seizure history | 2.34 (1.08–5.05) | 0.03c | 3.09 (1.37–6.94) | 0.006d |
| Full resection | ||||
| Yes | Reference | Reference | ||
| No | 8.00 (3.54–18.09) | <0.001d | 11.50 (4.44–29.76) | <0.001d |
| Acute postoperative seizuresa | ||||
| Yes | 6.17 (2.81–13.57) | <0.001d | 10.33 (3.94–27.12) | <0.001d |
| No | Reference | Reference | ||
| Cognitive outcomes/cognition stability | ||||
| Seizure-free status | ||||
| Yes | 1.76 (1.02–3.05) | 0.04c | NA | |
| No | Reference | NA | ||
| Hemisphere surgery (n = 127) | ||||
| Seizure outcomes/seizure persistence | ||||
| Age at surgery | ||||
| <2 y | Reference | Reference | ||
| ≥2 y | 3.26 (1.33–8.00) | 0.01c | 3.78 (1.51–9.47) | 0.005d |
| Seizure type | ||||
| Focal seizures only | Reference | Reference | ||
| Generalized seizure history | 3.94 (1.58–9.81) | 0.003d | 7.95 (2.74–23.05) | <0.001d |
| Seizure frequency | ||||
| <30 times per month | Reference | Reference | ||
| ≥30 times per month | 5.61 (1.66–19.02) | 0.006d | 4.76 (1.27–17.87) | 0.02c |
| Acute postoperative seizures | ||||
| Yes | 21.77 (8.86–53.47) | <0.001d | 27.61 (9.92–76.89) | <0.001d |
| No | Reference | Reference | ||
| Motor outcomes/motor stability | ||||
| Preoperative motor functionb | ||||
| Hemiparesis domain score <3 | Reference | Reference | ||
| Hemiparesis domain score ≥3 | 5.47 (2.38–12.58) | <0.001d | 5.23 (2.27–12.05) | <0.001d |
| Seizure-free status | ||||
| Yes | 3.30 (1.59–6.86) | 0.001d | 3.09 (1.49–6.45) | 0.003d |
| No | Reference | Reference | ||
| Cognitive outcomes/cognition stability | ||||
| Age at surgery | ||||
| <2 y | 2.13 (1.37–3.30) | <0.001d | 1.76 (1.13–2.75) | 0.01c |
| ≥2 y | Reference | Reference | ||
| Seizure-free status | ||||
| Yes | 3.40 (1.70–6.81) | <0.001d | 2.84 (1.39–5.78) | 0.004d |
| No | Reference | Reference | ||
Abbreviations: HR = hazard ratio; SWS = Sturge-Weber syndrome.
Statistical analyses were performed by univariable and multivariable Cox regression; potential factors with p values <0.10 in univariable analyses were identified and subjected to multivariable Cox regression (see eTable 3) for full univariable analysis of each potential associated factor.
Acute postoperative seizures indicate seizures that occurred within 30 days after surgery.
Preoperative motor function was evaluated by the SWS neurologic score by Kelley et al. as the hemiparesis domain; a hemiparesis domain score ≥3 indicates moderate-to-severe impaired fine and gross motor functions.
p < 0.05 (univariable Cox regression or multivariable Cox regression, as appropriate).
p < 0.01 (univariable Cox regression or multivariable Cox regression, as appropriate).
Figure 3. Kaplan-Meier Curves of All Significant Factors Associated With Seizure-Free, Motor-Stable, and Cognition-Stable Statuses in the Multivariable Analyses in the FR Group (A) and in the HS Group (B and C).
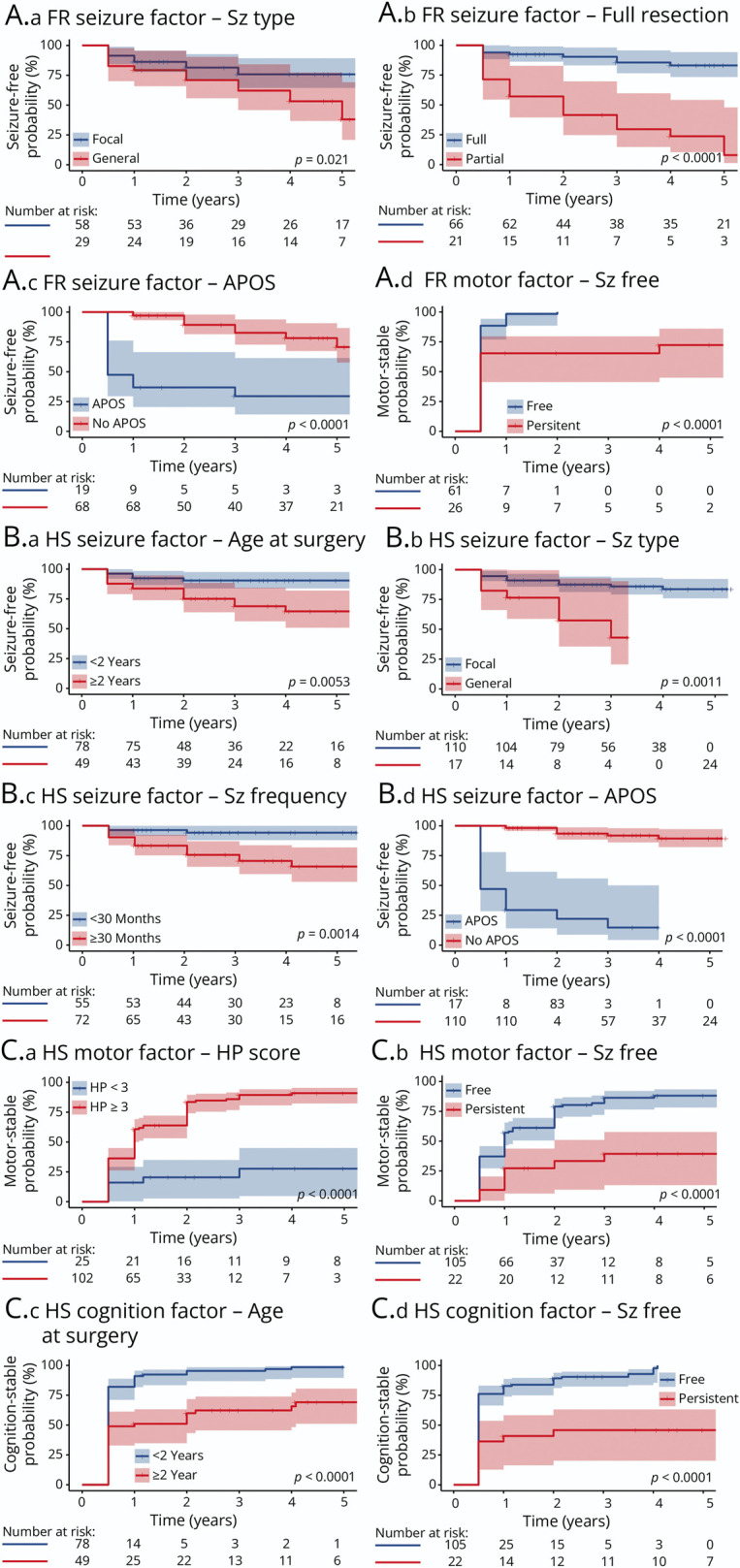
All the statistical comparisons were performed by log-rank tests; for A-iv, because seizure-free status was the only factor in the univariate analysis for cognitive stability in the group that had FR, it was not included in the multivariate analysis. APOS = acute postoperative seizure; FR = focal resection; HP = hemiparesis; HS = hemisphere surgery; Sz = seizure. All the statistical comparisons were performed by log-rank tests.
Classification of Evidence
This study provides Class IV evidence that in children with Sturge-Weber syndrome and refractory seizures, surgical resection—focal, hemispherectomy or modified hemispherotomy—leads to improved outcomes.
Discussion
To date, this study is the first and largest multicenter study on SWS surgeries.9,15 Early studies of FR for SWS revealed seizure-free survival rates of 50%–66.7% in the short term (<5 years)10,11 and 50.0%–58.3% in the long term (≥5 years).6,10,11 A previous meta-analysis reported a pooled seizure-free rate of 69.2%.15 This study revealed similar seizure-free probabilities to our previous single-center experience (79.5% for 2 years of follow-up),14 but the rates observed were slightly greater than those of earlier studies,6,10,11 potentially due to the increased proportions of full resection in this study.11,23 Some studies have suggested that FR leads to worse seizure outcomes than HS and results in only limited seizure-free periods,9,26 while other studies and current findings have indicated that FR could also result in similar long-term seizure control as HS.15,27 In addition, FR has the advantages of improved functional protection and decreased surgical risk.6,10,11,15 These findings support that FR is a valid and low-risk treatment to achieve long-term seizure control in patients with SWS with focal lesions.
HS is an established treatment for unilateral SWS.9,10,12,28 This study revealed similar seizure-free probabilities to our previous single-center study (87.0% seizure-free at 2 years)14 and other short-term (73.7%–100% seizure-free, <5 years)12,13,28 and long-term (72.0%–100% seizure-free; ≥5 years)9-11,28 studies on HS for SWS. In addition, current and previous studies9-13,28 have observed only a few complications after HS and have not reported severe sequelae. These results provide further evidence for the long-term effectiveness and safety of HS for unilateral SWS. However, vascular malformations in SWS complicate interoperative management.9 To make surgery safer and less invasive, AH is gradually being replaced by MH to limit the resection volume, blood loss, and craniotomy size.29 In addition, although we did not find bilaterally involved patients on preoperative or postoperative MRI in this study, underdiagnoses during evaluation (especially on MRI of patients younger than 1 year)9,23 might introduce bilateral patients during surgery, and the range of craniotomy limits intraoperative discoveries, which might contribute to postsurgical seizure persistence.
This study revealed several factors associated with good and poor seizure outcomes after surgery. Full resection was considered the most powerful factor for FR. Previous studies have also suggested that complete resection results in a significantly greater seizure-free rate than partial resection.10,11,14 Partial resections are usually performed when the eloquent cortex is involved to protect function,10,11 while residual seizures still have a negative influence on the neurologic functions and quality of life of patients.30 Thus, careful preoperative localization of the epileptogenic zone and functional areas should be performed, and full resection should be balanced to achieve seizure-free status with functional protection to determine optimal resection areas. Preoperative neuroimaging may not fully reveal SWS involvement,9,23 and careful intraoperative adjustment and postoperative review are needed. APOS seems to be a common early indicator of poor surgical outcomes in patients with epileptic diseases,31-34 which might indicate insufficient resection or that critical seizure networks were inadequately interrupted.31 Previous SWS studies did not consider this factor, but this study indicated that APOS should also be considered for FR and HS. Several studies on epileptic diseases other than SWS have suggested that generalized seizures might be related to seizure persistence after FR or HS,35-37 which was also observed in this study. It has been assumed that this persistence might be a sign of seizure activity spreading to other areas or to the contralateral brain.35-37 Previous SWS studies evaluated only small samples of patients with generalized seizures, potentially limiting their ability to identify this factor.6,9-14,28 Notably, Kossoff et al.21 revealed that SC followed by seizure-free status did not increase the risk of functional disability. Patients with this pattern should be treated carefully for seizure refractoriness. Younger surgical age and infrequent seizures were found to be positive factors for HS in a previous single-center study14 and were verified in this study, suggesting the potential positive benefits of early surgery for these patients. For FR, younger surgical age (<2 years) was a positive factor for seizure-free status in a previous single-center study.14 In this study, surgical age ≥6 years was a significant factor for seizure persistence in FR according to univariate analyses but not according to multivariate analyses. The increased cutoff age in this study might be related to the inclusion of more patients with older surgical ages at FR. In addition, post hoc analyses revealed that surgical age ≥6 years was associated with a lower full resection rate (p < 0.001), suggesting that surgical age might be an indirect factor through complete resection. With plasticity reduction, more caution in the resection area may be taken in older patients, resulting in a lower full resection rate.10,11 Thus, there is insufficient evidence that surgical age directly influences seizure outcomes after FR.
During the follow-up, more patients gradually demonstrated stable function, and those patients with repeated outcomes also exhibited a tendency toward improvement, suggesting a rehabilitation procedure. Other SWS studies on FR6,10,11 and HS9-13 have also suggested that epilepsy surgeries could result in either improved or unchanged functional outcomes. Functional reorganization is usually observed in the epilepsy-injured brain.38 Thus, the removed epileptogenic zone might already have a functional compensatory area. Furthermore, plasticity in the developing brain contributes to future improvements without seizures.39,40 In reserving functions, HS is preferred for patients who already have contralateral hemiparesis at baseline, and essential functions are supported bilaterally or contralateral to the resected hemisphere.11
Several factors associated with functional outcomes were also revealed. Because most FR patients had stable postoperative functions, only seizure-free status was found to be associated with motor stability in the univariate analysis. Postoperative seizure-free status was also found to be a multivariable factor associated with motor and cognitive stability in patients with HS. These findings further highlight the importance of achieving seizure-free status, which is consistent with the findings of previous studies11,30 and further verifies the current understanding that the main purpose of epilepsy surgery is to stop uncontrollable seizures and prevent motor or cognitive function from further worsening due to seizures.14 Increased surgical age was another independent factor associated with cognitive stability in HS patients. Patients with early HS have been shown to have better functional outcomes in current and many other studies,10,11,13 and longer seizure duration is associated with severe preoperative functional decline.7,14 Thus, early HS surgery could be considered to achieve better functions. Severe baseline motor defects were also associated with motor stability in the group that had HS, which further suggested that patients with severe motor defects were more likely to obtain functional benefits. Several studies have suggested that brain calcification observed by neuroimaging is an indicator of tissue damage from brain injury in patients with SWS,41 which might be associated with earlier seizure onset, longer seizure duration, and more severe symptoms, resulting in poorer neurocognitive outcomes.2,14,41,42 Neuroimaging and EEG features can be varied and informative in patients with SWS.2,23,43 However, this study did not find a clear association between these parameters and surgical outcomes. Further research should explore new technologies, such as advanced MRI sequences, nuclear medicine imaging, and quantitative EEG, to better explore their value as diagnostic tools, monitoring methods, and factors associated with outcomes.23,44,45
The optimal surgical age for SWS has been debated. Some have advocated early surgery to attain better seizure and functional outcomes,10,11,13 whereas others have advised that older surgical age might result in better seizure outcomes12 or that age does not influence outcomes.11 However, most of these studies involved mixed analyses of different surgery types, which is understandable due to the limited sample sizes. The results of this study suggest that the optimal surgical age should be evaluated individually for each surgery type. First, patients who underwent FR were significantly older at surgery and demonstrated better baseline functions than patients who underwent HS. Second, for HS, younger surgical age (<2 years) was associated with better seizure and cognitive outcomes, while for FR, surgical age might not be directly associated with surgical outcomes. Thus, early surgery is a better choice for HS. FR may be delayed for patients with mild functional deficits to confirm their seizure refractoriness after adequate ASM trials,14,46 and the surgical decision should balance the potential benefits and risks. In addition, the increased risk associated with surgeries at early ages (especially <1 year) and low body weights (especially <11 kg) should be balanced,47 and factors other than age should be considered.
Furthermore, although most of the included patients were children, some (15/214, 7.0%) patients underwent epilepsy surgery in adulthood (aged >18 years), which has been less commonly previously reported.9,11,12 The adult patients achieved a lower seizure-free rate (53.3%, p = 0.047) and comparable motor stable and cognition stable rates (93.3% and 93.3%, respectively) with those of pediatric patients. The lower seizure-free rate might be related to greater concern about decreased neurologic functional plasticity during adulthood, thus restricting the resection area. In addition, previous48,49 and current studies have suggested that adult patients with SWS are more likely to have more severe neurologic deterioration. These findings also support that early surgical treatment should be considered when surgical eligibility is met.
First, the retrospective observational nature of this study may have introduced selection bias and imbalanced control factors. Several measures were applied to control for potential biases, which can be found in the protocol. Second, although we tried our best to recruit patients nationwide, most patients traveled to more experienced eastern centers for surgeries, which led to only limited included samples in other areas; moreover, some centers with small sample sizes might have limited experience and underreported their results (including failures), which might have introduced bias. In addition, this study could not capture all eligible nationwide patients or data, which might have affected the final results. Third, because the sample size was limited, we included only 5 years of follow-up to avoid exaggeration. Data from an extended 10-year follow-up can be found in eFigure 3. Future prospective multicenter cohorts with international cooperation and prolonged follow-up could be helpful for better exploring more precise outcomes and developing prognostic predictive models, which could constitute a future research direction. Fourth, because surgical patients might have had more severe baseline symptoms,14 their outcomes were not compared with those of nonsurgical patients. A future prospective study (especially random controlled trials) with comparable nonsurgical control groups would be better in exploring and interpreting outcomes of epilepsy surgeries. Finally, because of inadequate baseline comparability, the FR and HS outcomes were not directly statistically compared; further propensity score–matched studies are needed.
The FR and HS epilepsy surgeries were effective and safe for refractory epilepsy in patients with SWS after a long-term follow-up. FR is a valid option and achieves similar results as HS with lower morbidity. Among the different HS techniques, MH had nonsignificant outcomes but improved safety when compared with AH. Several factors associated with outcomes were identified that can help clinical practice. Early surgical treatment (under the age of 2 years if possible) may provide better seizure and functional outcomes after HS, but there is insufficient evidence that surgical age affects FR outcomes. The optimal surgical age should be evaluated independently for different surgeries and conditions.
Acknowledgment
The authors thank the China Association Against Epilepsy (CAAE), especially the CAAE Youth Committee (CYC), for their great support in establishing the study collaboration. The authors thank all the patients and their families for participating in the SWS-CC study.
Glossary
- AH
anatomical hemispherectomy
- aHR
adjusted hazard ratio
- APOS
acute postoperative seizure
- ASM
antiseizure medication
- CAAE
China Association Against Epilepsy
- FR
focal resection
- HS
hemisphere surgery
- ILAE
International League Against Epilepsy
- IQR
interquartile range
- MH
modified hemispherotomy
- SC
seizure clustering
- SE
status epilepticus
- SWS
Sturge-Weber syndrome
- WISC-IV
Wechsler Intelligence Scale for Children—Fourth Edition
Appendix 1. Authors
| Name | Location | Contribution |
| Shu Wang, MD | Department of Neurosurgery, SanBo Brain Hospital, and Department of Neurosurgery, Beijing Tiantan Hospital, Capital Medical University, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; study concept or design; and analysis or interpretation of data |
| Qing-Zhu Liu, MD | Pediatric Epilepsy Center, Peking University First Hospital, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Rui Zhao, MD | Department of Neurosurgery, Children's Hospital of Fudan University, Shanghai, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Xuan Zhai, MD | Department of Neurosurgery, Children's Hospital of Chongqing Medical University, National Clinical Research Center for Child Health and Disorders, Ministry of Education Key Laboratory of Child Development and Disorders, Chongqing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Kai Zhang, MD | Department of Neurosurgery, Beijing Tiantan Hospital, Capital Medical University, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Lixin Cai, MD | Pediatric Epilepsy Center, Peking University First Hospital, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Shaochun Li, MD | Epilepsy Center, Guangdong Sanjiu Brain Hospital, Guangzhou, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Zhiquan Yang, MD | Department of Neurosurgery, Xiangya Hospital, Central South University, Changsha, Hunan, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Yongzhi Shan, MD | Department of Neurosurgery, Xuanwu Hospital, Capital Medical University, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Kangping Ma, MD | Department of Neurosurgery, Capital Institute of Pediatrics, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Yunlin Li, MD | Department of Neurosurgery, Capital Institute of Pediatrics, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Jie Hu, MD | Department of Neurosurgery, Huashan Hospital, Fudan University, Shanghai, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Lisen Sui, MD | Department of Epilepsy Center, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangdong, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Hongwei Cheng, MD | Department of Neurosurgery, The First Affiliated Hospital of Anhui Medical University, Hefei, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Xiaoli Li, MD | Department of Neurology, Affiliated ZhongDa Hospital, Southeast University, Nanjing, Jiangsu, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Jianyun Su, MD | Department of Neurology, Affiliated Children's Hospital of Xi'an Jiaotong University, Shaanxi, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Meng Zhao, MD | Department of Neurosurgery, Henan Sanbo Brain Hospital, Zhengzhou, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Xiongfei Wang, MD | Department of Neurosurgery, SanBo Brain Hospital, Capital Medical University, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Jian Zhou, MD | Department of Neurosurgery, SanBo Brain Hospital, Capital Medical University, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data |
| Mengyang Wang, MD | Department of Neurology, SanBo Brain Hospital, Capital Medical University, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; and study concept or design |
| Tianfu Li, MD | Department of Neurology, SanBo Brain Hospital, Capital Medical University, Beijing, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; and study concept or design |
| Jianguo Zhang, MD | Department of Neurosurgery, Beijing Tiantan Hospital, Capital Medical University; Department of Functional Neurosurgery, Beijing Neurosurgical Institute, Capital Medical University; Beijing Key Laboratory of Neurostimulation, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; and study concept or design |
| Shuli Liang, MD | Functional Neurosurgery Department, Beijing Children's Hospital, Capital Medical University, National Center for Children's Health, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; study concept or design; and analysis or interpretation of data |
| Guoming Luan, MD, PhD | Department of Neurosurgery, SanBo Brain Hospital, Capital Medical University; Beijing Key Laboratory of Epilepsy; Center of Epilepsy, Beijing Institute of Brain Disorders, Collaborative Innovation Center for Brain Disorders, Capital Medical University, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis or interpretation of data; and additional contributions: supervision, project administration, and funding acquisition |
| Yuguang Guan, MD, PhD | Department of Neurosurgery, SanBo Brain Hospital, Capital Medical University; Beijing Key Laboratory of Epilepsy; Center of Epilepsy, Beijing Institute of Brain Disorders, Collaborative Innovation Center for Brain Disorders, Capital Medical University, China | Drafting/revision of the article for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis or interpretation of data; and additional contributions: supervision, project administration, and funding acquisition |
Appendix 2. Coinvestigators
| Name | Affiliation | Role | Contribution |
| Zhiqiang Cui, MD | Department of Neurosurgery, The First Medical Center of PLA General Hospital, Beijing, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Qiang Guo, MD | Department of Neurosurgery, Guangdong Sanjiu Brain Hospital, Guangzhou, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Changzheng Dong, MD | Department of Neurosurgery, Hebei General Hospital, Shijiazhuang, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Weidong Yang, MD | Department of Neurosurgery, Tianjin Medical University General Hospital, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Shangchen Xu, MD | Department of Neurosurgery, Shandong Provincial Hospital Affiliated with Shandong First Medical University, Jinan, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Minzhong Wang, MD | Department of Neurology, Shandong Provincial Hospital Affiliated with Shandong First Medical University, Jinan, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Weiguo Li, MD | Department of Neurosurgery, Qilu Hospital of Shandong University, Jinan, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Junming Zhu, MD | Department of Neurosurgery, The Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Jinfeng Zhang, MD | Department of Neurosurgery, Fujian Sanbo Funeng Brain Hospital, Fuzhou, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Jixue Yang, MD | Department of Neurosurgery, The Third Affiliated Hospital of Zhengzhou University, Henan, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Zhenyang Liu, MD | Department of Neurosurgery, Shanxi Bethune Hospital Shanxi Academy of Medical Sciences, Taiyuan, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Changqing Liu, MD | Department of Neurosurgery, Chongqing Sanbo Jiangling Hospital, Chongqing Sanbo Changan Hospital, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Hua Zhang, MD | Department of Neurosurgery, The First Affiliated Hospital of Xi'an Jiaotong University, Shaanxi, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Zhiqiang Yan, MD | Department of Neurosurgery, Xijing Hospital, Fourth Military Medical University, Xi'an, Shaanxi, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Heng Zhang, MD | Department of Neurosurgery, West China Hospital, Sichuan University, Chengdu, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Xinding Zhang, MD | Department of Neurosurgery, Lanzhou University Second Hospital, Gansu, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Jie Ren, MD | Department of Neurosurgery, Kunming Sanbo Brain Hospital, Yunnan, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Jiahang Sun, MD | Department of Neurosurgery, The Second Affiliated Hospital, Harbin Medical University, Heilongjiang, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Shaoyi Li, MD | Department of Neurosurgery, Shengjing Hospital of China Medical University, Shenyang, Liaoning | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Yuan Zhang, MD | Neonatal Center, Beijing Children's Hospital, Capital Medical University, National Center for Children's Health, China | Site Investigator | Investigation of potential patients; coordinated medical records for site |
| Yali Liu, PhD | Center for Clinical Epidemiology and Evidence-based Medicine, Beijing Children's Hospital, Capital Medical University, National Center for Children's Health, China | Site Investigator | Data curation, biostatistical review |
Study Funding
This work was supported by the National Key Research and Development Program of China (2021YFC2401203 and 2018YFC2000700).
Disclosure
The authors report no relevant disclosures. Go to Neurology.org/N for full disclosures.
References
- 1.Shirley MD, Tang H, Gallione CJ, et al. Sturge-Weber syndrome and port-wine stains caused by somatic mutation in GNAQ. N Engl J Med. 2013;368(21):1971-1979. doi: 10.1056/NEJMoa1213507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.De la Torre AJ, Luat AF, Juhasz C, et al. A multidisciplinary consensus for clinical care and research needs for Sturge-Weber syndrome. Pediatr Neurol. 2018;84:11-20. doi: 10.1016/j.pediatrneurol.2018.04.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pascual-Castroviejo I, Pascual-Pascual SI, Velazquez-Fragua R, Viano J. Sturge-Weber syndrome: study of 55 patients. Can J Neurol Sci. 2008;35(3):301-307. doi: 10.1017/s0317167100008878 [DOI] [PubMed] [Google Scholar]
- 4.Jagtap S, Srinivas G, Harsha KJ, Radhakrishnan N, Radhakrishnan A. Sturge-Weber syndrome: clinical spectrum, disease course, and outcome of 30 patients. J Child Neurol. 2013;28(6):725-731. doi: 10.1177/0883073812451326 [DOI] [PubMed] [Google Scholar]
- 5.Sudarsanam A, Ardern-Holmes SL. Sturge-Weber syndrome: from the past to the present. Eur J Paediatr Neurol. 2014;18(3):257-266. doi: 10.1016/j.ejpn.2013.10.003 [DOI] [PubMed] [Google Scholar]
- 6.Maton B, Krsek P, Jayakar P, et al. Medically intractable epilepsy in Sturge-Weber syndrome is associated with cortical malformation: implications for surgical therapy. Epilepsia. 2010;51(2):257-267. doi: 10.1111/j.1528-1167.2009.02304.x [DOI] [PubMed] [Google Scholar]
- 7.Bosnyák E, Behen ME, Guy WC, Asano E, Chugani HT, Juhász C. Predictors of cognitive functions in children with Sturge-Weber syndrome: a longitudinal study. Pediatr Neurol. 2016;61:38-45. doi: 10.1016/j.pediatrneurol.2016.05.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Luat AF, Behen ME, Chugani HT, Juhasz C. Cognitive and motor outcomes in children with unilateral Sturge-Weber syndrome: effect of age at seizure onset and side of brain involvement. Epilepsy Behav. 2018;80:202-207. doi: 10.1016/j.yebeh.2018.01.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bianchi F, Auricchio AM, Battaglia DI, Chieffo DRP, Massimi L. Sturge-Weber syndrome: an update on the relevant issues for neurosurgeons. Childs Nerv Syst. 2020;36(10):2553-2570. doi: 10.1007/s00381-020-04695-3 [DOI] [PubMed] [Google Scholar]
- 10.Arzimanoglou AA, Andermann F, Aicardi J, et al. Sturge-Weber syndrome: indications and results of surgery in 20 patients. Neurology. 2000;55(10):1472-1479. doi: 10.1212/wnl.55.10.1472 [DOI] [PubMed] [Google Scholar]
- 11.Bourgeois M, Crimmins DW, de Oliveira RS, et al. Surgical treatment of epilepsy in Sturge-Weber syndrome in children. J Neurosurg. 2007;106(1 suppl):20-28. doi: 10.3171/ped.2007.106.1.20 [DOI] [PubMed] [Google Scholar]
- 12.Kossoff EH, Buck C, Freeman JM. Outcomes of 32 hemispherectomies for Sturge-Weber syndrome worldwide. Neurology. 2002;59(11):1735-1738. doi: 10.1212/01.wnl.0000035639.54567.5c [DOI] [PubMed] [Google Scholar]
- 13.Steinbok P, Gan PY, Connolly MB, et al. Epilepsy surgery in the first 3 years of life: a Canadian survey. Epilepsia. 2009;50(6):1442-1449. doi: 10.1111/j.1528-1167.2008.01992.x [DOI] [PubMed] [Google Scholar]
- 14.Wang S, Pan J, Zhao M, et al. Characteristics, surgical outcomes, and influential factors of epilepsy in Sturge-Weber syndrome. Brain. 2022;145(10):3431-3443. doi: 10.1093/brain/awab470 [DOI] [PubMed] [Google Scholar]
- 15.Frank NA, Greuter L, Dill PE, Guzman R, Soleman J. Focal lesionectomy as surgical treatment of epilepsy in patients with Sturge-Weber syndrome: a case-based systematic review and meta-analysis. Neurosurg Focus. 2022;52(5):E4. doi: 10.3171/2022.2.FOCUS21788 [DOI] [PubMed] [Google Scholar]
- 16.Hsieh FY, Lavori PW. Sample-size calculations for the Cox proportional hazards regression model with nonbinary covariates. Control Clin Trials. 2000;21(6):552-560. doi: 10.1016/s0197-2456(00)00104-5 [DOI] [PubMed] [Google Scholar]
- 17.Wang S, Zhang Y, Guan Y, et al. A nationwide evaluation of the prevalence of and risk factors associated with anxiety, depression and insomnia symptoms during the return-to-work period of coronavirus disease 2019 in China. Soc Psychiatry Psychiatr Epidemiol. 2021;56(12):2275-2286. doi: 10.1007/s00127-021-02046-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Luat AF, Juhasz C, Loeb JA, et al. Neurological complications of Sturge-Weber syndrome: current status and unmet needs. Pediatr Neurol. 2019;98:31-38. doi: 10.1016/j.pediatrneurol.2019.05.013 [DOI] [PubMed] [Google Scholar]
- 19.Fisher RS, Cross JH, D'Souza C, et al. Instruction manual for the ILAE 2017 operational classification of seizure types. Epilepsia. 2017;58(4):531-542. doi: 10.1111/epi.13671 [DOI] [PubMed] [Google Scholar]
- 20.Trinka E, Cock H, Hesdorffer D, et al. A definition and classification of status epilepticus: report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia. 2015;56(10):1515-1523. doi: 10.1111/epi.13121 [DOI] [PubMed] [Google Scholar]
- 21.Kossoff EH, Ferenc L, Comi AM. An infantile-onset, severe, yet sporadic seizure pattern is common in Sturge-Weber syndrome. Epilepsia. 2009;50(9):2154-2157. doi: 10.1111/j.1528-1167.2009.02072.x [DOI] [PubMed] [Google Scholar]
- 22.Kelley TM, Hatfield LA, Lin DD, Comi AM. Quantitative analysis of cerebral cortical atrophy and correlation with clinical severity in unilateral Sturge-Weber syndrome. J Child Neurol. 2005;20(11):867-870. doi: 10.1177/08830738050200110201 [DOI] [PubMed] [Google Scholar]
- 23.Pinto AL, Chen L, Friedman R, et al. Sturge-Weber syndrome: brain magnetic resonance imaging and neuropathology findings. Pediatr Neurol. 2016;58:25-30. doi: 10.1016/j.pediatrneurol.2015.11.005 [DOI] [PubMed] [Google Scholar]
- 24.Smegal LF, Sebold AJ, Hammill AM, et al. Multicenter research data of epilepsy management in patients with Sturge-Weber syndrome. Pediatr Neurol. 2021;119:3-10. doi: 10.1016/j.pediatrneurol.2021.02.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wieser HG, Blume WT, Fish D, et al. ILAE Commission Report. Proposal for a new classification of outcome with respect to epileptic seizures following epilepsy surgery. Epilepsia. 2001;42(2):282-286. doi: 10.1046/j.1528-1157.2001.35100.x [DOI] [PubMed] [Google Scholar]
- 26.Comi AM. Advances in Sturge-Weber syndrome. Curr Opin Neurol. 2006;19(2):124-128. doi: 10.1097/01.wco.0000218226.27937.57 [DOI] [PubMed] [Google Scholar]
- 27.Di Rocco C, Tamburrini G. Sturge-Weber syndrome. Childs Nerv Syst. 2006;22(8):909-921. doi: 10.1007/s00381-006-0143-2 [DOI] [PubMed] [Google Scholar]
- 28.Basheer SN, Connolly MB, Lautzenhiser A, Sherman EM, Hendson G, Steinbok P. Hemispheric surgery in children with refractory epilepsy: seizure outcome, complications, and adaptive function. Epilepsia. 2007;48(1):133-140. doi: 10.1111/j.1528-1167.2006.00909.x [DOI] [PubMed] [Google Scholar]
- 29.Young CC, Williams JR, Feroze AH, et al. Pediatric functional hemispherectomy: operative techniques and complication avoidance. Neurosurg Focus. 2020;48(4):E9. doi: 10.3171/2020.1.FOCUS19889 [DOI] [PubMed] [Google Scholar]
- 30.Maragkos GA, Geropoulos G, Kechagias K, Ziogas IA, Mylonas KS. Quality of life after epilepsy surgery in children: a systematic review and meta-analysis. Neurosurgery. 2019;85(6):741-749. doi: 10.1093/neuros/nyy471 [DOI] [PubMed] [Google Scholar]
- 31.Giridharan N, Horn PS, Greiner HM, Holland KD, Mangano FT, Arya R. Acute postoperative seizures as predictors of seizure outcomes after epilepsy surgery. Epilepsy Res. 2016;127:119-125. doi: 10.1016/j.eplepsyres.2016.08.026 [DOI] [PubMed] [Google Scholar]
- 32.Jayalakshmi S, Vooturi S, Vadapalli R, Madigubba S, Panigrahi M. Predictors of surgical outcome in focal cortical dysplasia and its subtypes. J Neurosurg. 2022;136(2):512-522. doi: 10.3171/2020.12.JNS203385 [DOI] [PubMed] [Google Scholar]
- 33.Kogias E, Bast T, Schubert-Bast S, et al. Multilobar epilepsy surgery in childhood and adolescence: predictors of long-term seizure freedom. Neurosurgery. 2020;88(1):174-182. doi: 10.1093/neuros/nyaa368 [DOI] [PubMed] [Google Scholar]
- 34.McGovern RA, N V Moosa A, Jehi L, et al. Hemispherectomy in adults and adolescents: seizure and functional outcomes in 47 patients. Epilepsia. 2019;60(12):2416-2427. doi: 10.1111/epi.16378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Elsharkawy AE, El-Ghandour NM, Oppel F, et al. Long-term outcome of lesional posterior cortical epilepsy surgery in adults. J Neurol Neurosurg Psychiatry. 2009;80(7):773-780. doi: 10.1136/jnnp.2008.164145 [DOI] [PubMed] [Google Scholar]
- 36.Ramesha KN, Mooney T, Sarma PS, Radhakrishnan K. Long-term seizure outcome and its predictors in patients with recurrent seizures during the first year aftertemporal lobe resective epilepsy surgery. Epilepsia. 2011;52(5):917-924. doi: 10.1111/j.1528-1167.2010.02891.x [DOI] [PubMed] [Google Scholar]
- 37.Hu WH, Zhang C, Zhang K, Shao XQ, Zhang JG. Hemispheric surgery for refractory epilepsy: a systematic review and meta-analysis with emphasis on seizure predictors and outcomes. J Neurosurg. 2016;124(4):952-961. doi: 10.3171/2015.4.JNS14438 [DOI] [PubMed] [Google Scholar]
- 38.Goldmann RE, Golby AJ. Atypical language representation in epilepsy: implications for injury-induced reorganization of brain function. Epilepsy Behav. 2005;6(4):473-487. doi: 10.1016/j.yebeh.2005.03.012 [DOI] [PubMed] [Google Scholar]
- 39.Wiebe S, Berg AT. Big epilepsy surgery for little people: what's the full story on hemispherectomy? Neurology. 2013;80(3):232-233. doi: 10.1212/WNL.0b013e31827dec32 [DOI] [PubMed] [Google Scholar]
- 40.Van Schooneveld MM, Braun KP. Cognitive outcome after epilepsy surgery in children. Brain Dev. 2013;35(8):721-729. doi: 10.1016/j.braindev.2013.01.011 [DOI] [PubMed] [Google Scholar]
- 41.Pilli VK, Behen ME, Hu J, et al. Clinical and metabolic correlates of cerebral calcifications in Sturge-Weber syndrome. Dev Med Child Neurol. 2017;59(9):952-958. doi: 10.1111/dmcn.13433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wu J, Tarabishy B, Hu J, et al. Cortical calcification in Sturge-Weber syndrome on MRI-SWI: relation to brain perfusion status and seizure severity. J Magn Reson Imaging. 2011;34(4):791-798. doi: 10.1002/jmri.22687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kossoff EH, Bachur CD, Quain AM, Ewen JB, Comi AM. EEG evolution in Sturge-Weber syndrome. Epilepsy Res. 2014;108(4):816-819. doi: 10.1016/j.eplepsyres.2014.01.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hatfield LA, Crone NE, Kossoff EH, et al. Quantitative EEG asymmetry correlates with clinical severity in unilateral Sturge-Weber syndrome. Epilepsia. 2007;48(1):191-195. doi: 10.1111/j.1528-1167.2006.00630.x [DOI] [PubMed] [Google Scholar]
- 45.Juhasz C, Hu J, Xuan Y, Chugani HT. Imaging increased glutamate in children with Sturge-Weber syndrome: association with epilepsy severity. Epilepsy Res. 2016;122:66-72. doi: 10.1016/j.eplepsyres.2016.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia. 2010;51(6):1069-1077. doi: 10.1111/j.1528-1167.2009.02397.x [DOI] [PubMed] [Google Scholar]
- 47.Vadera S, Griffith SD, Rosenbaum BP, et al. National trends and in-hospital complication rates in more than 1600 hemispherectomies from 1988 to 2010: a nationwide inpatient sample study. Neurosurgery. 2015;77(2):185-191; discussion 191. doi: 10.1227/NEU.0000000000000815 [DOI] [PubMed] [Google Scholar]
- 48.Aupy J, Bonnet C, Arnould JS, Fernandez P, Marchal C, Zanotti-Fregonara P. Focal inhibitory seizure with prolonged deficit in adult Sturge-Weber syndrome. Epileptic Disord. 2015;17(3):328-331. doi: 10.1684/epd.2015.0755 [DOI] [PubMed] [Google Scholar]
- 49.Sujansky E, Conradi S. Outcome of Sturge-Weber syndrome in 52 adults. Am J Med Genet. 1995;57(1):35-45. doi: 10.1002/ajmg.1320570110 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data that support the findings of this study are included in the article/supplementary material.