Abstract
Introduction
The clinical consequences for patients with inflammatory bowel disease (IBD) who stop treatment owing to side effects have not been fully investigated.
Methods
This retrospective observational study aimed to compare patients who discontinued thiopurine treatment due to side effects with those who tolerated thiopurine treatment in the use of other IBD drugs, surgery, and fecal calprotectin values in the first 5 years after the start of thiopurine treatment.
Results
The proportion of patients with IBD who initiated thiopurine treatment at our clinic was 44% (32% ulcerative colitis and 64% Crohn's disease) and 31% (n = 94) of those patients had to stop thiopurine treatment within 5 years due to side effects. Patients who discontinued thiopurine treatment due to intolerance were significantly older (median age 33 vs. 27 years, p = 0.003), significantly more often used prednisolone (89 vs. 76%, p = 0.009), and used to a lesser extent TNF inhibitors at the start of thiopurine treatment (3% vs. 9%, p = 0.062). Budesonide treatment and non-TNF inhibitor second-line therapy were significantly more commonly used in patients who discontinued thiopurine treatment owing to side effects, but there were no statistically significant differences in the use of other treatments. The proportion of patients with a median FC >200 μg/g was significantly higher during follow-up in patients with UC who discontinued thiopurine treatment owing to side effects.
Conclusions
Patients who discontinued thiopurines owing to side effects were prescribed more budesonide and non-TNF inhibitor second-line therapy, but there were no differences in the use of TNF inhibitors, prednisolone, or surgery.
Keywords: Calprotectin, Crohn's disease, Inflammatory bowel disease, Side effects, Surgery, Thiopurines, Ulcerative colitis
Introduction
Thiopurines have been used to treat patients with inflammatory bowel disease (IBD) since the 1960s [1]. Thiopurines (azathioprine, 6-mercaptopurine, and thioguanine) are purine analogs that reduce cell proliferation and have immune-modulating properties. They were predominantly used as treatment for leukemia but have been used at lower doses for autoimmune inflammatory disorders [2]. There is evidence for effect of azathioprine and 6-mercaptopurine, respectively, in the maintenance treatment of both Crohn's disease (CD) [3, 4] and ulcerative colitis (UC) [5]. Thiopurines may also inhibit the production of autoantibodies against biologics used to treat IBD [6, 7]. Despite the introduction of novel treatments for IBD, thiopurines are commonly used as maintenance treatment in patients with IBD and approximately 40% of patients with CD and 20% of patients with UC will start thiopurine treatment within the first 5 years after diagnosis [8]. However, approximately 15–40% of patients treated with thiopurines experience side effects that lead to drug withdrawal or reduction of dosages [9]. The side effects are dose dependent (nausea, myelotoxicity, hepatotoxicity) or dose independent (malaise, fever, musculoskeletal pain, and pancreatitis) [9, 10].
The clinical consequences for patients who discontinue thiopurine treatment owing to side effects have been poorly studied. In optimal IBD care, a patient who starts treatment should be followed up on a reasonable time scale, and treatment should be modified after clinical effects and possible side effects. A patient with adverse events of thiopurine should be considered to step-up to a more effective IBD treatment. However, there is also a risk that the patient and/or doctor will struggle for a long period of time to cope with side effects and, therefore, delay treatment.
The aim of this retrospective observational study was to compare patients who discontinued thiopurine treatment due to side effects with patients who tolerated thiopurine treatment in the use of other IBD treatments, the risk of surgery, and the chance of achieving remission (an annual median fecal calprotectin [FC] level <200 μg/g). The study focused on the first 5 years after the patient started thiopurine treatment.
Methods
This was a retrospective observational study of all patients who started treatment with thiopurines for IBD at our hospital (Norrland University Hospital, Sweden). The included patients were patients with an endoscopically or imaging-verified diagnosis of IBD who were treated at the Department of Medicine at our hospital. The study was restricted to patients born in 1956 or later who were diagnosed with IBD after 2006. Patients diagnosed before 2006 were excluded because the complete data on drug prescriptions in the digital medical record system were not fully reliable until 2006. The patient had to be at least 18 years old when starting thiopurine treatment. The exclusion criteria were patients diagnosed or treated outside the catchment area.
A thorough medical chart review that focused on medical treatments, including surgery for IBD, and the results of the FC tests were collected. All patients were observed for 5 years after the first thiopurine treatment was initiated or until December 31, 2022. For each year, patients were categorized as users of a certain IBD drug if they were prescribed the drug at least once during that year. The TNF inhibitors used in these patients were adalimumab and infliximab. Second-line non-TNF inhibitor therapy was defined as treatment with vedolizumab, ustekinumab, or tofacitinib. A new IBD surgery after the start of thiopurine therapy was noted and was registered as a cumulative new surgery. The median FC level for each patient was recorded for each year. If the patient had discontinued thiopurine treatment, the reason for withdrawal was noted. Patients who stopped treatment due to intolerance (side effects) were compared to those who were tolerant to thiopurine treatment (no side effects reported) in the use of drugs for IBD, surgery, or FC levels. We also compared patients who stopped thiopurine due to intolerance within 12 months versus after 12 months. Remission was defined as a median FC <200 μg/g FC each year after starting thiopurine treatment. At our hospital, the method used for FC analysis was the CALPRO® Calprotectin ELISA Test (ALP) [11]. A subset of patients had 6-thioguanine nucleotide (6-TGN) in whole blood analyzed by liquid chromatography-mass spectrometry [12] and for those patients the median 6-TGN levels for the first 5 years were calculated.
IBM SPSS Statistics version 28.1.1 was used for the data analysis. The χ2 test or the Fisher's exact test were used to compare the proportions. The Mann-Whitney U test was used to compare the continuous data. Kaplan-Meier curves were used to illustrate drug survival.
Results
Thiopurine Drug Survival
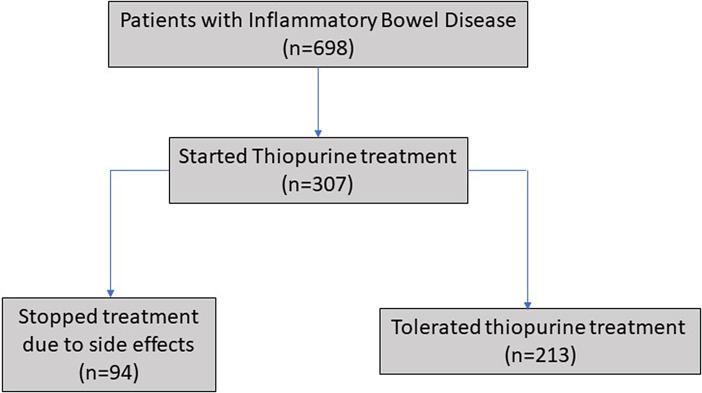
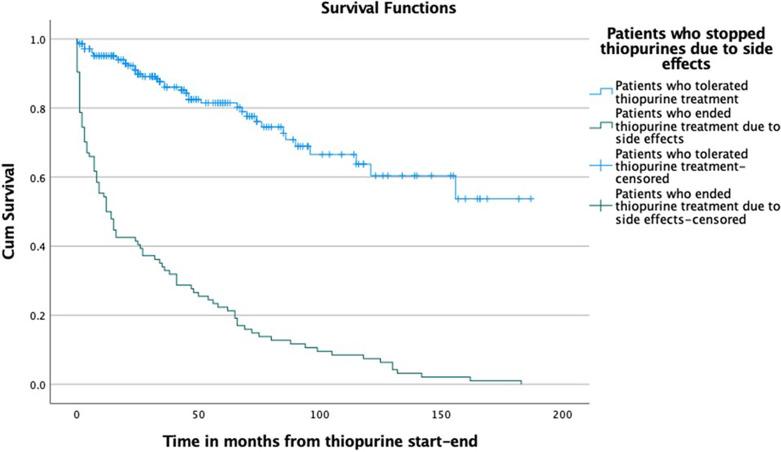
The proportion of patients with IBD who initiated thiopurine treatment at our clinic was 44% (32% UC and 64% CD), and 31% of those patients discontinued thiopurine treatment within 5 years owing to side effects (shown in Fig. 1). The thiopurine drug survival for patients who stopped thiopurines due to side effects and for patients who tolerated thiopurines is shown in Figure 2. For patients intolerant to thiopurines, the median time to treatment withdrawal was 13 months (25th–75th percentile 2–54 months) and for those who were tolerant 37 months (20–72 months).
Fig. 1.
Recruitment of the study population.
Fig. 2.
Drug survival for thiopurine treatment in patients who stopped treatment due to side effects and in patients who tolerated thiopurines.
Patients Who Stopped Thiopurine Treatment due to Side Effects
Patients who discontinued thiopurine treatment because of intolerance were significantly older, significantly more often on steroids, and used TNF inhibitors to a lesser extent at the start of thiopurine treatment (Table 1). Nausea and/or vomiting were the most common causes of withdrawal owing to side effects (Table 2). Other common causes of withdrawal were elevated liver enzyme levels and abdominal pain, whereas leukopenia was a less common cause of withdrawal (Table 2). In patients tolerant to thiopurines, the most common cause of discontinuation treatment was a request from the patient to end the treatment. A subset of patients had 6-TGN levels analyzed, but there were no differences in 6-TGN levels between the group who stopped treatment due to side effects (n = 36) and those who tolerated thiopurines (n = 88) (302 vs. 338 pmol/8 × 108; p = 0.181).
Table 1.
Basal characteristics
| Patients who ended thiopurine treatment due to side effects (n = 94) | Patients who tolerated thiopurine treatment (n = 213) | p value | |
|---|---|---|---|
| Female gender, % (n) | 47.9 (45) | 38.5 (82) | 0.124 |
| Median age at diagnosis (25th–75th percentile), years | 32 (22–42) | 25 (20–34) | 0.002 |
| Median age at start of thiopurine treatment (25th–75th percentile), years | 33 (24–45) | 27 (21–36) | 0.003 |
| UC, % (n) | 41.5 (39) | 47.9 (102) | 0.300 |
| UC Montreal classification, % (n) | |||
| A1 | 10.3 (4) | 7.8 (8) | 0.032 |
| A2 | 56.4 (22) | 77.5 (79) | |
| A3 | 33.3 (13) | 14.7 (15) | |
| E1 | 5.1 (2) | 4.9 (5) | 0.712 |
| E2 | 43.6 (17) | 36.3 (37) | |
| E3 | 51.3 (20) | 58.6 (60) | |
| CD Montreal classification, % (n) | |||
| A1 | 5.5 (2) | 11.7 (13) | 0.429 |
| A2 | 72.7 (40) | 66.7 (74) | |
| A3 | 21.8 (12) | 21.6 (24) | |
| L1 | 41.8 (23) | 28.8 (32) | 0.212 |
| L2 | 43.6 (24) | 49.5 (55) | |
| L3 | 14.5 (8) | 21.6 (24) | |
| B1 | 61.8 (34) | 64.0 (71) | 0.420 |
| B2 | 21.8 (12) | 14.4 (16) | |
| B3 | 16.4 (9) | 21.6 (24) | |
| Bp | 12.7 (7) | 20.7 (23) | 0.284 |
| Drugs at the time of start of thiopurine treatment, % (n) | |||
| Five ASA | 54.3 (51) | 55.3 (118) | 0.853 |
| Prednisolone | 89.4 (84) | 76.5 (163) | 0.009 |
| TNF inhibitors | 3.2 (3) | 9.4 (20) | 0.062 |
| Second-line non-TNF inhibitor therapya | 1.1 (1) | 1.9 (4) | >0.999 |
| A history of surgery for IBD at the time of start of thiopurine treatment, % (n) | 8.5 (8) | 8.5 (18) | >0.999 |
| Median time between diagnosis to start of thiopurine treatment (25th–75th percentile), months | 6 (1–29) | 7 (2–27) | 0.385 |
| Median time from start of thiopurine treatment to withdrawal (25th–75th percentile), months | 13 (2–54) | 37 (20–72) | <0.001 |
| Received a second attempt of thiopurine treatment, % (n) | 59.6 (56) | 2.3 (5) | <0.001 |
| Received a third attempt of thiopurine treatment, % (n) | 8.5 (8) | 0.5 (1) | <0.001 |
| Tested for TPMT genotype, % (n) | 52.1 (49) | 54.0 (115) | 0.763 |
| TPMT genotype 1/3A | 2.1 (2) | 4.2 (9) | 0.654 |
aSecond-line non-TNF inhibitor therapy = vedoluzimab, ustekinumab, or tofacitinib.
Table 2.
Causes for withdrawal of thiopurine treatment
| Cause of withdrawal | Patients who ended thiopurine treatment due to side effects (n = 94), % (n) | Patients who ended treatment within 12 months due to side effects (n = 47), % (n) |
|---|---|---|
| Nausea/vomiting | 42.5 (40) | 38.3 (18) |
| Elevated liver enzymes | 22.3 (21) | 25.5 (12) |
| Abdominal pain | 21.3 (20) | 25.5 (12) |
| Fatigue/malaise | 16.0 (15) | 14.9 (7) |
| Fever or increased inflammatory parameters | 10.6 (10) | 19.1 (9) |
| Muscle/joint pain | 8.5 (8) | 10.6 (5) |
| Pancreatitis | 7.4 (7) | 10.6 (5) |
| Leukopenia | 6.4 (6) | 6.4 (3) |
| Infections | 3.2 (3) | 2.1 (1) |
| Itching/rash | 3.2 (3) | 2.1 (1) |
| Angioedema | 2.1 (2) | 2.1 (1) |
| Anemia | 2.1 (2) | 2.1 (1) |
| Headache | 2.1 (2) | 0 (0) |
| Loss of appetite | 2.1 (2) | 2.1 (1) |
| Tremor | 1.1 (1) | 0 (0) |
| Alopecia | 1.1 (1) | 2.1 (1) |
| Not specified cause | 3.2 (3) | 4.2 (2) |
| Patients who tolerated thiopurine treatment (n = 41), % (n) | Patients who tolerated thiopurine treatment but ended treatment within 12 months (n = 10), % (n) | |
|---|---|---|
| Patient request | 43.9 (18) | 60.0 (6) |
| Lack of effect | 26.8 (11) | 20.0 (2) |
| Long-term remission | 22.0 (9) | 10.0 (1) |
| Pregnancy | 7.3 (3) | 0 (0) |
| Cancer | 2.4 (1) | 0 (0) |
| Other | 2.4 (1) | 10.0 (1) |
Some patients have reported more than one side effect.
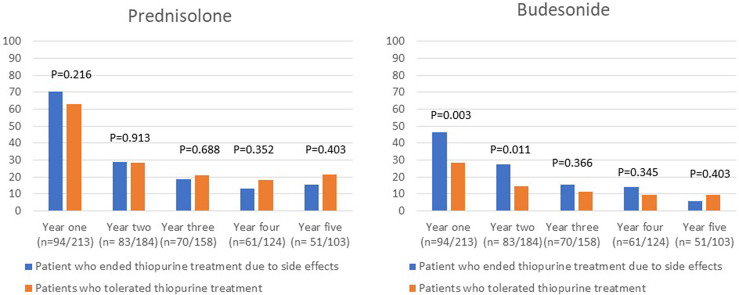
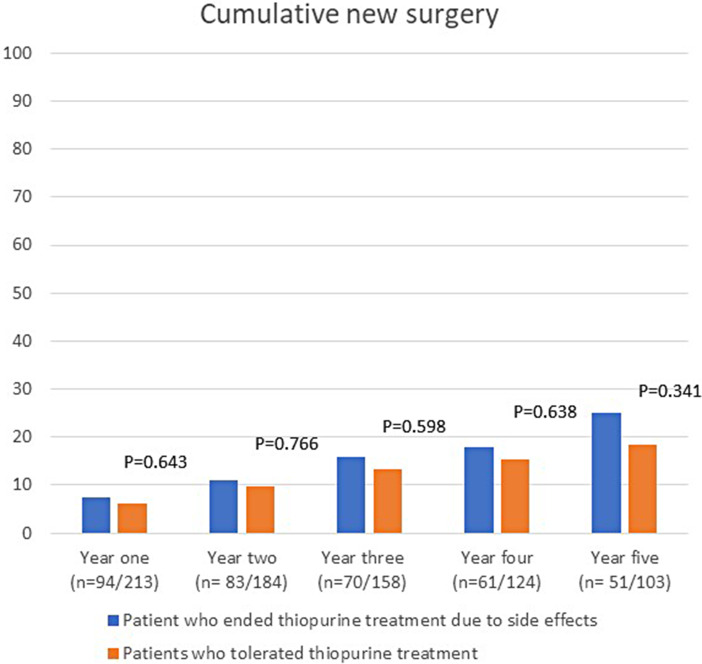
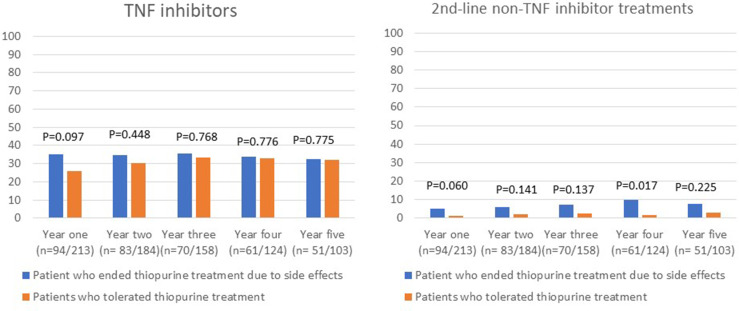
There were no statistically significant differences in the proportion of patients who used prednisolone, TNF inhibitors, or had surgery in the first 5 years after initiating thiopurine treatment between the group of patients who discontinued treatment because of intolerance and the group of patients who were tolerant to thiopurine treatment (shown in Fig. 3–5). Budesonide treatment was significantly more commonly used in the first 2 years in the patients who stopped thiopurine treatment owing to side effects (shown in Fig. 3). Second-line non-TNF inhibitor treatment was also more commonly used among patients who discontinued thiopurines owing to intolerance and was significantly more commonly used 4 years after initiating thiopurine treatment (shown in Fig. 4). When analyzing patients with UC and CD separately, there were no statistically significant differences in the use of drugs for IBD between patients who stopped thiopurines due to side effects and those who tolerated thiopurines.
Fig. 3.
Use of prednisolone and budesonide treatment the first 5 years after start of thiopurine treatment.
Fig. 5.
Cumulative new surgery for IBD the first 5 years after start of thiopurine treatment.
Fig. 4.
Use of TNF inhibitors and second-line non-TNF inhibitor treatment the first 5 years after start of thiopurine treatment.
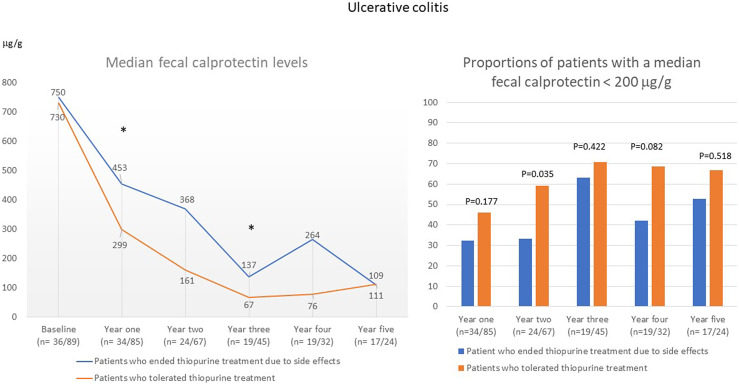
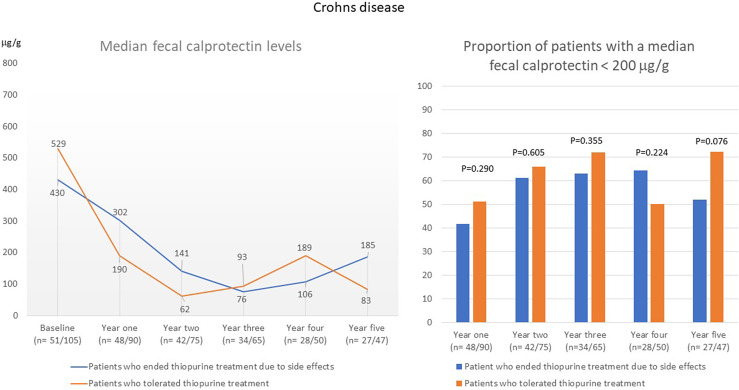
In patients with UC who discontinued thiopurine treatment because of intolerance, the median FC value was significantly higher in the first year (453 μg/g vs. 299 μg/g; p = 0.046) and in the third year (137 μg/g vs. 67 μg/g; p = 0.049) than in patients who were tolerant to thiopurines (shown in Fig. 6). The proportion of patients with a median FC of <200 μg/g was significantly lower in the group that discontinued thiopurine treatment during the second year after starting treatment (shown in Fig. 6). In patients with CD, there were no differences in median FC or in the proportion of patients with a median FC <200 μg/g between patients who discontinued thiopurine treatment due to intolerance and those who were tolerant to thiopurines (shown in Fig. 7).
Fig. 6.
Annual median FC level (µg/g) and the proportion of patients with an annual median FC <200 μg/g in patients with UC the first 5 years after start of thiopurine treatment.
Fig. 7.
Annual median FC level (µg/g) and the proportion of patients with an annual median FC <200 μg/g in patients with CD the first 5 years after start of thiopurine treatment.
Patients Who Stopped Thiopurine Treatment within 12 Months due to Side Effects
Tables 3 and 4 present data on patients with IBD who stopped thiopurines due to side effects within 12 months (n = 47) compared to those who stopped after 12 months (n = 47). Fever and increased inflammatory markers were more commonly reported among patients who stopped thiopurines within 12 months (Table 2). Patients who stopped within 12 months were significantly less commonly prescribed a second thiopurine but had a significantly higher risk for new surgery in the first year compared to patients who stopped treatment due to side effects after 12 months (Table 4). There were no differences between the groups in the use of prednisolone, TNF-inhibitor, or second-line non-TNF inhibitor therapy, but patients who stopped within 12 months more often used budesonide in the second year of follow-up. There were no differences in the proportion of patients who had a median FC <200 μg/g at follow-up between patients who stopped thiopurines due to side effects within 12 months and patients who stopped after 12 months.
Table 3.
Patients who stopped thiopurines due to side effects within 1 year compared to patients who stopped thiopurines due to side effects after 1 year of treatment
| Patients who ended thiopurine treatment due to side effects within 1 year (n = 47) | Patients who ended thiopurine treatment due to side effects after 1 year (n = 47) | p value | |
|---|---|---|---|
| Female gender, % (n) | 46.8 (22) | 48.9 (23) | 0.124 |
| Median age at diagnosis (25th–75th percentile), years | 35 (24–45) | 28 (22–37) | 0.184 |
| Median age at start of thiopurine treatment (25th–75th percentile), years | 36 (25–47) | 30 (22–40) | 0.125 |
| UC, % (n) | 49 (23) | 34 (16) | 0.143 |
| CD, % (n) | 51 (24) | 56 (31) | |
| Drugs at the time of start of thiopurine treatment, % (n) | |||
| Five ASA | 55 (26) | 53 (25) | 0.836 |
| Prednisolone | 94 (44) | 85 (40) | 0.316 |
| TNF inhibitors | 0 (0) | 6 (3) | 0.242 |
| Second-line non-TNF inhibitor therapya | 0 (0) | 2 (1) | >0.999 |
| A history of surgery for IBD at the time of start of thiopurine treatment, % (n) | 13 (6) | 4 (2) | 0.267 |
| Median time between diagnosis to start of thiopurine treatment (25th–75th percentile), months | 11 (1–34) | 4 (2–24) | 0.635 |
| Median time from start of thiopurine treatment to withdrawal (25th–75th percentile), months | 2 (1–7) | 54 (29–84) | <0.001 |
| Received a second attempt of thiopurine treatment, % (n) | 32 (15) | 87 (41) | <0.001 |
| Received a third attempt of thiopurine treatment, % (n) | 6 (3) | 11 (5) | 0.714 |
| Tested for TPMT genotype, % (n) | 53 (25) | 51 (24) | 0.836 |
| TPMT genotype 1/3A | 4 (2) | 0 (0) | 0.495 |
aSecond-line non-TNF inhibitor therapy = vedolizumab, ustekinumab, or tofacitinib.
Table 4.
Use of drugs and cumulative surgery in patients who stopped thiopurines due to side effects within 1 year compared to patients who stopped thiopurines due to side effects after 1 year of treatment
| Patients who ended thiopurine treatment due to side effects within 1 year (n = 47) | Patients who ended thiopurine treatment due to side effects after 1 year (n = 47) | p value | |
|---|---|---|---|
| Prednisolone, % (n) | |||
| Year one (n = 94) | 66 (31) | 74 (35) | 0.367 |
| Year two (n = 83) | 36 (13) | 23 (11) | 0.206 |
| Year three (n = 70) | 18 (5) | 19 (8) | >0.999 |
| Year four (n = 62) | 17 (4) | 11 (4) | 0.700 |
| Year five (n = 52) | 22 (4) | 12 (4) | 0.430 |
| Budesonide, % (n) | |||
| Year one (n = 94) | 43 (20) | 50 (23) | 0.471 |
| Year two (n = 83) | 39 (14) | 19 (9) | 0.046 |
| Year three (n = 70) | 11 (5) | 19 (8) | 0.511 |
| Year four (n = 62) | 12 (6) | 16 (6) | >0.999 |
| Year five (n = 52) | 5 (1) | 6 (2) | >0.999 |
| TNF inhibitors, % (n) | |||
| Year one (n = 94) | 38 (18) | 32 (15) | 0.517 |
| Year two (n = 83) | 36 (13) | 34 (16) | 0.845 |
| Year three (n = 70) | 33 (9) | 37 (16) | 0.802 |
| Year four (n = 62) | 36 (9) | 32 (12) | 0.791 |
| Year five (n = 52) | 32 (6) | 33 (11) | >0.999 |
| Second-line non-TNF inhibitor therapya, % (n) | |||
| Year one (n = 94) | 6 (3) | 4 (2) | >0.999 |
| Year two (n = 83) | 8 (3) | 4 (2) | 0.648 |
| Year three (n = 70) | 11 (3) | 5 (2) | 0.367 |
| Year four (n = 62) | 16 (4) | 5 (2) | 0.210 |
| Year five (n = 52) | 11 (2) | 6 (2) | 0.617 |
| Cumulative new surgery, % (n) | |||
| Year one (n = 94) | 15 (7) | 0 (0) | 0.006 |
| Year two (n = 83) | 17 (6) | 6 (3) | 0.161 |
| Year three (n = 70) | 27 (7) | 9 (4) | 0.087 |
| Year four (n = 62) | 25 (6) | 13 (5) | 0.313 |
| Year five (n = 52) | 39 (7) | 18 (6) | 0.108 |
aSecond-line non-TNF inhibitor therapy = vedolizumab, ustekinumab, or tofacitinib.
Discussion
To the best of our knowledge, this is the first study that aimed to explore the impact on a patient with IBD who had to stop thiopurine treatment due to side effects with regard to disease activity and treatment at follow-up in real-life clinical practice. At our clinic, treatment discontinuation due to side effects was common and affected approximately 1 out of 3 patients in the first 5 years after the start of treatment.
In patients with UC who stopped thiopurine treatment due to side effects, there was a lower proportion of patients who reached a median FC level <200 μg/g in the second year after starting treatment. The increased inflammatory activity observed in patients with UC in our study who stopped thiopurine treatment may reflect an insufficient response by the doctor in the need to optimize treatment. Overall, we found no pronounced differences in the use of other IBD treatments in the first 5 years after the start of thiopurine treatment between patients who stopped treatment due to side effects and those who tolerated thiopurines. When including all patients with IBD, only budesonide and second-line non-TNF inhibitor treatment were slightly more commonly prescribed among patients who had to stop thiopurine treatment owing to side effects, but there were no significant differences when analyzing patients with UC and CD separately. Regarding the use of TNF inhibitors, there was a higher proportion of patients who received TNF inhibitors in the first year among patients with IBD who stopped treatment due to side effects, but the difference was not statistically significant.
The use of novel therapies (vedolizumab, ustekinumab, or tofacitinib) was low in our study. It can be explained that most data were registered in a time period when these treatments were not available, and when introduced, there could be a restricted use for economic reasons (more expensive treatments). In addition, the age of the patients might have influenced the choice of therapy in patients with IBD in our study. Patients who stopped treatment owing to side effects in our study were significantly older than those who tolerated thiopurines and perhaps the doctors at our clinic were more restricted to step-up treatment due to fear of infections and cancer in elderly associated with immunosuppressive drugs [13–15].
Elderly have previously been reported to be less tolerant to thiopurines. In a Spanish registry study [16], patients starting thiopurines when over 60 years of age reported more adverse events in comparison to patients 18–50 years old (43.4 vs. 29.7%; p < 0.001) and also more commonly were excluded due to infections and malignancies (40.4 vs. 28.3%; p < 0.001). Adverse events in the elderly also significantly more often led to the withdrawal of thiopurine treatment. Our study only included persons younger than 60 years when starting treatment, but, also in that age span, side effects that led to withdrawal of treatment were more commonly reported by those of higher age. Consistently, in a Canadian study, being >40 years of age when starting thiopurine treatment was a risk factor for discontinuation of treatment due to side effects [17]. In addition to intolerance in the elderly to thiopurines, female gender has been associated with a higher risk of side effects with thiopurines [17, 18]. In our study, there was a higher proportion of women in the group who stopped treatment because of side effects, but the difference was not significant.
Budesonide treatment was significantly more commonly used in the first 2 years in the patients who stopped thiopurines owing to side effects. Budesonide is effective in inducing remission in UC [19] and CD [20] but is not proved to be effective for the maintenance of remission in CD [20] and no study has tested budesonide for maintenance treatment in UC. The higher use of budesonide treatment in patients who stopped thiopurines due to side effects in our study could reflect an increased relapse rate of IBD flares, but budesonide could also have been incorrectly prescribed for maintenance treatment instead of biologics or other IBD therapies.
The cumulative risk of surgery did not differ between patients who stopped thiopurines due to side effects and those who tolerated thiopurines. However, patients with IBD who stopped thiopurine treatment due to side effects within 12 months were significantly at a higher risk for surgery in the first year compared to patients who stopped treatment due to side effects after 12 months. The threshold for surgery in the patients who stopped thiopurines early might have been lower due to increased age, lack of response, or intolerance to previous medical treatments. Interestingly, only 1 out of 3 of the patients who stopped thiopurine within 12 months due to side effects were prescribed a second thiopurine. A possible explanation for not testing a second thiopurine could be the characteristics of side effects reported for the first thiopurine. For example, fever, increased inflammatory markers, and pancreatitis were more commonly reported by the patients who stopped thiopurines early.
Patients who later stopped thiopurine treatment because of side effects more often received prednisolone treatment at baseline. This is a new finding that may have several explanations. For example, there could be a synergistic effect of the side effects of steroids and thiopurines, or steroid treatment may reflect a more severe IBD activity in a patient, leading to increased gastrointestinal symptoms and/or vulnerability to the sensation of side effects. During the follow-up period, there were no differences in the use of prednisolone between patients who stopped thiopurines and those who tolerated thiopurines.
The present study has several limitations. First, this is a single-center study with relatively few patients which make comparisons between subgroups of patients unreliable. Second, this study is based on retrospective clinical data. Third, we have no data on clinical effects except for FC values measured in routine clinical practice and FC was not analyzed in all patients. We also lack data on how the initiation of thiopurine therapy was performed for each patient. The strength of the study is that we study the intention to treat with thiopurines in real clinical practice and that we included all patients with IBD who started thiopurine treatment in our clinic.
In conclusion, in clinical practice, patients who discontinued thiopurines owing to side effects did not generally differ in the use of other treatments for IBD in the first 5 years after initiating thiopurine treatment, except for the use of budesonide and second-line non-TNF inhibitor treatment, which was increased compared to patients who tolerated thiopurines. Patients with UC who stopped treatment due to side effects had higher FC levels at follow-up than those who tolerated thiopurines.
Acknowledgment
We thank the Medical Department at Norrland University Hospital for providing access to data from medical records.
Statement of Ethics
The study protocol was reviewed by the Swedish Ethical Review Authority (Dnr 2022-060609-01) and judged as a study with no significant ethical considerations. Written informed consent from the parent/legal guardian of participants was not required for this retrospective study in accordance with local/national guidelines. The study was performed in accordance with Swedish law for Patient Safety and the directions of the Swedish Data Protection Authority.
Conflict of Interest Statement
The authors have nothing to declare.
Funding Sources
This work was supported by the Region of Västerbotten, Sweden.
Author Contributions
P.K. planned the study. H.G.S. collected all data from medical journals. H.G.S. and P.K. performed all the analyses and interpreted the data. All the authors have read and approved the final manuscript.
Funding Statement
This work was supported by the Region of Västerbotten, Sweden.
Data Availability Statement
Data are available from the corresponding author on reasonable request. Data are not publicly available due to risk of compromising secrecy.
References
- 1. Brooke BN, Hoffmann DC, Swarbrick ET. Azathioprine for Crohn’s disease. Lancet. 1969;2(7621):612–4. [DOI] [PubMed] [Google Scholar]
- 2. Sahasranaman S, Howard D, Roy S. Clinical pharmacology and pharmacogenetics of thiopurines. Eur J Clin Pharmacol. 2008;64(8):753–67. [DOI] [PubMed] [Google Scholar]
- 3. Chande N, Patton PH, Tsoulis DJ, Thomas BS, MacDonald JK. Azathioprine or 6-mercaptopurine for maintenance of remission in Crohn’s disease. Cochrane Database Syst Rev. 2015;2015(10):CD000067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Gjuladin-Hellon T, Iheozor-Ejiofor Z, Gordon M, Akobeng AK. Azathioprine and 6-mercaptopurine for maintenance of surgically-induced remission in Crohn’s disease. Cochrane Database Syst Rev. 2019;8(8):CD010233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Timmer A, Patton PH, Chande N, McDonald JW, MacDonald JK. Azathioprine and 6-mercaptopurine for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2016;2016(5):CD000478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ben-Horin S, Waterman M, Kopylov U, Yavzori M, Picard O, Fudim E, et al. Addition of an immunomodulator to infliximab therapy eliminates antidrug antibodies in serum and restores clinical response of patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013;11(4):444–7. [DOI] [PubMed] [Google Scholar]
- 7. Ungar B, Kopylov U, Engel T, Yavzori M, Fudim E, Picard O, et al. Addition of an immunomodulator can reverse antibody formation and loss of response in patients treated with adalimumab. Aliment Pharmacol Ther. 2017;45(2):276–82. [DOI] [PubMed] [Google Scholar]
- 8. Kirchgesner J, Lemaitre M, Rudnichi A, Racine A, Zureik M, Carbonnel F, et al. Therapeutic management of inflammatory bowel disease in real-life practice in the current era of anti-TNF agents: analysis of the French administrative health databases 2009-2014. Aliment Pharmacol Ther. 2017;45(1):37–49. [DOI] [PubMed] [Google Scholar]
- 9. Pudipeddi A, Kariyawasam V, Haifer C, Baraty B, Paramsothy S, Leong RW. Safety of drugs used for the treatment of Crohn's disease. Expert Opin Drug Saf. 2019;18(5):357–67. [DOI] [PubMed] [Google Scholar]
- 10. Singh A, Mahajan R, Kedia S, Dutta AK, Anand A, Bernstein CN, et al. Use of thiopurines in inflammatory bowel disease: an update. Intest Res. 2022;20(1):11–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tibble J, Sigthorsson G, Foster R, Sherwood R, Fagerhol M, Bjarnason I. Faecal calprotectin and faecal occult blood tests in the diagnosis of colorectal carcinoma and adenoma. Gut. 2001;49(3):402–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kirchherr H, Shipkova M, von Ahsen N. Improved method for therapeutic drug monitoring of 6-thioguanine nucleotides and 6-methylmercaptopurine in whole-blood by LC/MSMS using isotope-labeled internal standards. Ther Drug Monit. 2013;35(3):313–21. [DOI] [PubMed] [Google Scholar]
- 13. Cottone M, Kohn A, Daperno M, Armuzzi A, Guidi L, D'Inca R, et al. Advanced age is an independent risk factor for severe infections and mortality in patients given anti-tumor necrosis factor therapy for inflammatory bowel disease. Clin Gastroenterol Hepatol. 2011;9(1):30–5. [DOI] [PubMed] [Google Scholar]
- 14. D'Arcy ME, Beachler DC, Pfeiffer RM, Curtis JR, Mariette X, Seror R, et al. Tumor necrosis factor inhibitors and the risk of cancer among older Americans with rheumatoid arthritis. Cancer Epidemiol Biomarkers Prev. 2021;30(11):2059–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ytterberg SR, Bhatt DL, Connell CA. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. Reply. N Engl J Med. 2022;386(18):1768. [DOI] [PubMed] [Google Scholar]
- 16. Calafat M, Manosa M, Canete F, Ricart E, Iglesias E, Calvo M, et al. Increased risk of thiopurine-related adverse events in elderly patients with IBD. Aliment Pharmacol Ther. 2019;50(7):780–8. [DOI] [PubMed] [Google Scholar]
- 17. Moran GW, Dubeau MF, Kaplan GG, Yang H, Eksteen B, Ghosh S, et al. Clinical predictors of thiopurine-related adverse events in Crohn’s disease. World J Gastroenterol. 2015;21(25):7795–804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Chaparro M, Ordas I, Cabre E, Garcia-Sanchez V, Bastida G, Penalva M, et al. Safety of thiopurine therapy in inflammatory bowel disease: long-term follow-up study of 3,931 patients. Inflamm Bowel Dis. 2013;19(7):1404–10. [DOI] [PubMed] [Google Scholar]
- 19. Barberio B, Marsilio I, Buda A, Bertin L, Semprucci G, Zanini A, et al. Efficacy and safety of oral beclomethasone dipropionate and budesonide MMX versus 5-aminosalicylates or placebo in ulcerative colitis: a systematic review and meta-analysis. Therap Adv Gastroenterol. 2023;16:17562848231188549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kuenzig ME, Rezaie A, Kaplan GG, Otley AR, Steinhart AH, Griffiths AM, et al. Budesonide for the induction and maintenance of remission in Crohn’s disease: systematic review and meta-analysis for the cochrane collaboration. J Can Assoc Gastroenterol. 2018;1(4):159–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are available from the corresponding author on reasonable request. Data are not publicly available due to risk of compromising secrecy.