Abstract
CD46 is a transmembrane complement regulatory protein widely expressed on nucleated human cells. Laboratory-adapted strains of measles virus (MV) bind to the extracellular domains of CD46 to enter human cells. The cytoplasmic portion of CD46 consists of a common juxtamembrane region and different distal sequences called Cyt1 and Cyt2. The biological functions of these cytoplasmic sequences are unknown. In this study, we show that expression of human CD46 with the Cyt1 cytoplasmic domain in mouse macrophages enhances production of nitric oxide (NO) in response to MV infection in the presence of gamma interferon (IFN-γ). Human CD46 does not increase the basal levels of NO production in mouse macrophages and does not augment NO production induced by double-stranded polyribonucleotides. Replacing the cytoplasmic domain of human CD46 with Cyt2 reduces MV and IFN-γ-induced NO production in mouse macrophages. Deleting the entire cytoplasmic domains of human CD46 does not prevent MV infection but markedly attenuates NO production in response to MV and IFN-γ. Mouse macrophages expressing a tailless human CD46 mutant are more susceptible to MV infection and produce 2 to 3 orders of magnitude more infectious virus than mouse macrophages expressing human CD46 with intact cytoplasmic domains. These results reveal a novel function of CD46 dependent on the cytoplasmic domains (especially Cyt1), which augments NO production in macrophages. These findings may have significant implications for roles of CD46 in innate immunity and MV pathogenesis.
Measles is a major cause of childhood morbidity and mortality (6, 53, 56). Current measles vaccines do not offer adequate protection in infants (17), hindering efforts of global control of measles. Understanding the pathogenic and attenuation mechanisms of the causative agent, measles virus (MV), can facilitate development of more effective measles vaccines or therapeutics.
A hallmark of MV infection is suppression of cellular immunity, which can lead to severe secondary infections common in fatal cases of measles (5, 10, 20). Interaction between MV and cells of the immune system plays a critical role in this process. MV infection of B and T lymphocytes in culture arrests progression through the cell cycle (49). MV-infected peripheral blood mononuclear cells suppress proliferative response of neighboring lymphocytes, apparently by producing an inhibitory factor(s) (60) or by cell contact (61). MV also induces apoptotic cell death in cultured monocytes and fibroblasts (13). In mice transplanted with human thymic tissues, MV replicates mainly in thymic epithelium yet induces apoptosis in thymocytes (3). MV infection blocks allostimulatory function of dendritic cells for activating T lymphocytes (21, 64). MV also inhibits monocytes/macrophages and dendritic cells from secreting interleukin-12 (IL-12) (16, 32), which is important for T helper-1 and natural killer cell functions (70).
Monocytes/macrophages are major in vivo targets for MV in human patients (14). Macrophages, which serve as a first line defense against microbial pathogens (15, 50), are derived from progenitor cells of the myelomonocytic lineage. Cells at different stages of differentiation along this lineage show differential susceptibility to MV. Immature human myelomonocytic cells support MV replication efficiently and produce infectious virus particles (22). By contrast, MV replication in monocytes and differentiated macrophages is highly restricted, regardless of whether those cells are stimulated with phorbol ester and calcium ionophore (72, 74). The block in MV replication in macrophages appears to be at both posttranscription and posttranslation levels, resulting in high levels of viral RNA but low levels of viral proteins and no infectious virus production (22). Depleting monocytes/macrophages from human peripheral blood mononuclear cells enhances MV replication in the remaining cells, which consist mostly of lymphocytes (72, 74). This finding suggests that monocytes/macrophages may produce suppressing factors that inhibit MV replication, rather than lack essential factors that support MV replication.
To facilitate study of the interaction between MV and macrophages, we have generated RAW264.7 mouse macrophages expressing human membrane cofactor protein (CD46), a cellular receptor for laboratory-adapted strains of MV (11, 54). RAW264.7 mouse macrophages have been used extensively for studying macrophage functions, and they can be easily transfected to generate cell lines expressing foreign DNA, a procedure lethal to most primary monocytes/macrophages and monocytic cell lines (68). Most important, RAW264.7 mouse macrophages expressing human CD46 exhibit restriction of MV replication reminiscent of differentiated human macrophages. Specifically, MV efficiently enters RAW264.7 mouse macrophages expressing human CD46 and initially synthesizes high levels of viral RNA and proteins. After day 2 of infection, viral protein synthesis and virus production are drastically suppressed just as in differentiated human macrophages (22, 35). This pattern of MV replication in RAW264.7 mouse macrophages expressing human CD46 is consistent with the hypothesis that MV provokes an antiviral response that restricts virus replication in mouse macrophages. These cells thus offer a useful model for studying the interaction between MV and macrophages.
Interestingly, MV can infect RAW264.7 cells and some other mouse macrophage lines in the absence of human CD46 to cause a prolonged noncytopathic infection with continued viral protein synthesis and virus production (19, 35). Unexpectedly, expression of human CD46 in mouse macrophages restricts rather than promotes MV replication (35). This restriction has not been seen in other rodent fibroblasts and lymphoid cells expressing human CD46 (11, 54, 77). This finding raises the intriguing possibility that CD46, while serving as a receptor for MV, can also contribute to suppression of MV replication in macrophages.
CD46 is a transmembrane complement regulatory protein widely expressed on nucleated human cells (42, 65). The extracellular portion of the molecule consists of four short consensus repeats and several serine-, threonine-, and proline-rich (STP) regions. The cytoplasmic portion consists of an invariant juxtamembrane region followed by different distal sequences (Cyt1 and Cyt2) generated by alternative mRNA splicing. The previously known function of CD46 is to bind and promote cleavage of complement components C3b or C4b, to protect autologous cells from complement lysis (42, 65). MV and C3b/C4b bind to different but partially overlapping regions mapped to the short consensus repeat domains (29, 46). The cytoplasmic domains of CD46 are unnecessary for MV infection or protection against complement (44, 73). We previously found that joining the cytoplasmic domains of CD46 to glutathionine S-transferase enables the fusion proteins to associate with kinase activity in mouse macrophage lysates (76). However, the biological functions of the CD46 cytoplasmic domains remain unknown.
In this study, we demonstrate that expression of human CD46 enhances the response of mouse macrophages to MV and gamma interferon (IFN-γ), leading to production of high levels of nitric oxide (NO), a gaseous radical with antimicrobial and immunomodulating properties. This response is dependent on the CD46 cytoplasmic domains, especially Cyt1. Mouse macrophages expressing a tailless human CD46 mutant are highly susceptible to MV infection but do not produce high levels of NO upon MV infection and IFN-γ treatment. These results provide the first evidence that CD46 may serve a novel role in macrophage host defense, and the cytoplasmic domains of CD46 may play a role in this function.
MATERIALS AND METHODS
Mouse macrophage lines.
Mouse macrophage cell line RAW264.7 (gift of Alan Aderem, University of Washington) was cultured in RPMI 1640 supplemented with 10% fetal bovine serum (FBS; GIBCO BRL). Mouse macrophage lines expressing human CD46 were generated by transfecting RAW264.7 cells with plasmids based on the pME18S vector, which contained CD46 cDNAs driven by SRα promoter (69) and the neomycin resistance gene driven by the promoter of reticuloendotheliosis virus (24). MCP-1, MCP-2, and ΔCyt0 cDNAs encode STP-C isoforms of human CD46 with Cyt1, Cyt2, and no cytoplasmic sequences, respectively (25). Control cells were transfected with the same vector without CD46 cDNA. Transfection was performed as previously described (35). Briefly, 5 × 106 RAW264.7 cells were mixed with 25 μg of plasmid DNA in 250 μl of phosphate-buffered saline for 10 min at room temperature. Electroporation was performed with a Bio-Rad Gene Pulser set at 300 V and 960 μF. The samples were immediately transferred into a 60-mm-diameter dish containing 5 ml of RPMI 1640 supplemented with 10% FBS. At 24 to 36 h after transfection, the cells were replenished with fresh medium containing 400 μg of the neomycin analog G418 (GIBCO BRL) per ml. G418-resistant colonies were propagated in the same medium and screened for high expression of CD46 by surface protein immunoprecipitation as previously described (25, 78). Mouse macrophage clones expressing comparable levels of the different forms of human CD46 were selected for further studies.
Virus infection.
Edmonston strain MV stocks were propagated in African green monkey kidney (CV-1) cells. Mouse macrophages were detached with 1 mM EDTA and incubated in suspension with MV at a multiplicity of infection (MOI) of 1 or 2 at room temperature for 1 h in a small volume of RPMI 1640 medium. After adsorption, the unattached virus was removed by washing with serum-free medium, and cells were resuspended in fresh medium containing 10% FBS and plated into 35-mm-diameter culture dishes at a density of 2 × 106 cells per dish. One day (24 h) postinfection, cells were replenished with medium without or with different concentrations of recombinant mouse IFN-γ (Pharmingen), as indicated in the figure legends. Culture medium was collected on day 2 or 3 of infection (48 or 72 h postinfection) for NO assay.
Poly(I-C) treatment.
Mouse macrophages were treated with different concentrations of poly(I-C) (Pharmacia Biotech) in RPMI 1640 medium without or with 500 U mouse IFN-γ per ml. One or two days (24 or 48 h) after treatment, culture medium was collected for NO assay.
Protein analysis.
One day postinfection, MV-infected and uninfected mouse macrophage lines were labeled for 1 h with 30 μCi of [35S]methionine (Du Pont NEN Research Products) per culture. Cells were lysed in radioimmunoprecipitation buffer (10 mM Tris-HCl [pH 7.5], 150 mM NaCl, 1% Triton X-100, 1% deoxycholate, 0.1% sodium dodecyl sulfate [SDS]). Viral proteins were immunoprecipitated with the MV-specific antiserum GM (23) and analyzed by SDS-polyacrylamide gel electrophoresis (SDS-PAGE). Surface and intracellular CD46 expression was determined by immunoprecipitating the cell surface or intracellular proteins with monoclonal antibody (MAb) M177 followed by immunoblotting with the same MAb (25, 78).
NO2− determination.
NO production was examined by determining the concentrations of stable end product nitrite (NO2−) in the culture medium by a colorimetric method (7). Triplicate samples containing equal volumes (100 μl) of cell-free culture medium and Greiss reagent (1% sulfanilamide, 0.1% N-1-naphthylenediamine, 5% H3PO4; Sigma) were mixed in a 96-well plate at room temperature for 15 min. Optical density was measured with a microplate reader (EL808; BioTek Instrument, Inc.) at 550 nm. A standard curve was generated for each experiment using known concentrations of sodium nitrite as references.
RESULTS
Mouse macrophages expressing human CD46 produce NO in response to MV infection and IFN-γ.
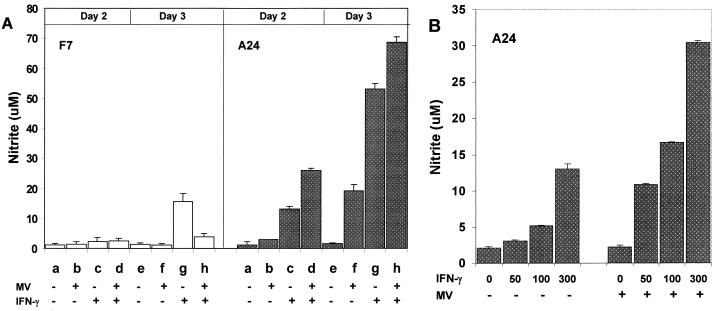
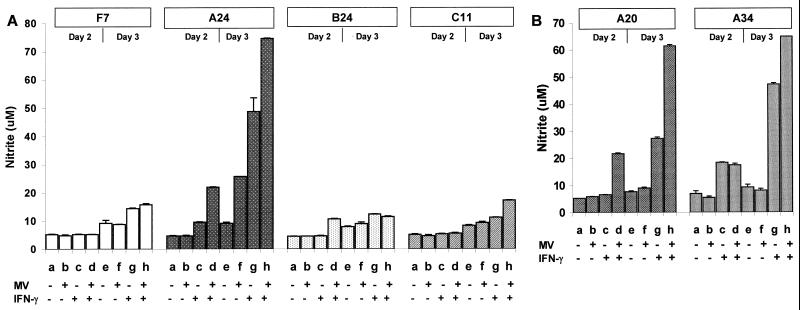
We first compared NO production from mouse macrophages expressing human CD46 with the Cyt1 cytoplasmic domain (A24 line) or transfected with vector alone (F7 line) after MV infection (Edmonston strain at an MOI of 1) or IFN-γ treatment, by measuring accumulation of stable product NO2− in the culture medium. The CD46-negative F7 mouse macrophages did not produce significant levels of NO in response to MV (Fig. 1A, F7, lanes b and f). These cells produced moderate levels of NO in response to 500 U of IFN-γ per ml (Fig. 1A, F7, lanes c and g). MV infection did not further increase NO production from F7 mouse macrophages treated with IFN-γ (Fig. 1A, F7, lanes d and h). Expression of human CD46 did not increase the basal levels of NO production in the A24 mouse macrophages in the absence of stimuli (Fig. 1A, A24, lanes a and e). Upon infection by MV, A24 mouse macrophages expressing human CD46 produced increasing levels of NO (Fig. 1, A24, lanes b and f). In the absence of IFN-γ, the absolute levels of NO induced by MV in CD46-expressing mouse macrophages were quite variable (see below). Treatment with IFN-γ alone induced high levels of NO in A24 cells (Fig. 1, A24, lanes c and g). Most important, MV infection consistently enhanced the NO levels induced by IFN-γ in A24 mouse macrophages on both days 2 and 3 after infection (Fig. 1, A24, lanes d and h). These results, which have been reproduced in five separate experiments, show that MV augments NO production in mouse macrophages expressing human CD46 with the Cyt1 cytoplasmic domain in the presence of IFN-γ.
FIG. 1.
NO production from mouse macrophages in response to MV infection and IFN-γ. (A) Mouse macrophages expressing CD46 with the Cyt1 cytoplasmic domain (A24) or CD46-negative mouse macrophages (F7) were mock infected or infected with Edmonston strain MV at an MOI of 1. One day after infection, the cells were replenished with fresh medium without or with mouse IFN-γ (500 U/ml). On day 2 or 3 after infection, the culture medium was collected and assayed in triplicate for NO2− accumulation. Open and stippled columns represent mean values of NO2− concentrations in the F7 and A24 cultures, respectively. (B) A24 mouse macrophages were infected with MV at an MOI of 0.1 and replenished with medium without or with 50 to 300 U of mouse IFN-γ per ml. Concentrations of NO2− in the culture medium were determined from triplicate samples on day 3 after infection. Bars atop the columns show standard deviation values.
The absolute levels of NO induced by MV in A24 mouse macrophages varied with the dosage of input virus. At a low MOI (0.1), MV did not induce significant levels of NO in the absence of IFN-γ (Fig. 1B, lane 0). However, MV synergized with IFN-γ to enhance NO production in A24 mouse macrophages over a wide range of IFN-γ and virus dosages. For instance, MV infection at an MOI of 0.1 significantly enhanced NO production from A24 mouse macrophages treated with 50 to 300 U of IFN-γ per ml compared to IFN-γ treatment alone (Fig. 1B, lanes 50 to 300). Thus, MV augments NO response in mouse macrophages expressing human CD46 over broad IFN-γ concentrations consistent with the dosages needed to stimulate primary human or mouse monocytes/macrophages in previous studies (32, 36). To maximally differentiate the NO response between CD46-positive and CD46-negative mouse macrophages, we chose the highest concentration (300 or 500 U/ml) of IFN-γ for the subsequent experiments.
Expression of human CD46 does not affect poly(I-C)-induced NO production in mouse macrophages.
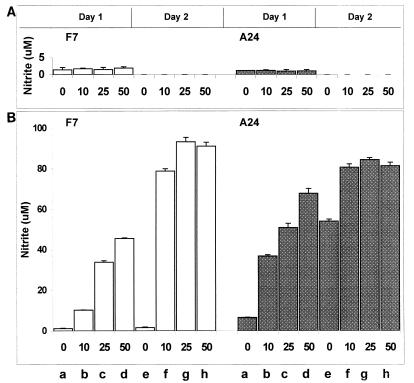
A hallmark of viral infection is production of double-stranded RNA, which can induce NO production in both human and mouse macrophages (38, 67). To see whether CD46 influenced NO response to double-stranded RNA, we treated A24 and F7 mouse macrophages with different concentrations of poly(I-C) in the presence or absence of IFN-γ. Without IFN-γ, all concentrations of poly(I-C) tested (up to 50 μg/ml) did not induce significant levels of NO in these mouse macrophages, regardless of CD46 expression (Fig. 2A). Confirming the results in Fig. 1, A24 CD46-positive mouse macrophages responded to IFN-γ alone more strongly than F7 CD46-negative mouse macrophages, producing higher levels of NO especially on day 2 after IFN-γ treatment (Fig. 2B; compare lanes e for A24 and F7). Adding IFN-γ together with increasing concentrations of poly(I-C) further enhanced NO production in a dosage-dependent manner in both F7 and A24 mouse macrophages (Fig. 2B). One day after poly(I-C) and IFN-γ treatment, A24 mouse macrophages produced higher levels of NO than F7 mouse macrophages (Fig. 2B; compare lanes b to d for F7 and A24). On day two, however, F7 and A24 cells produced comparable levels of NO (Fig. 2B, F7 and A24, lanes f to h).
FIG. 2.
NO production from mouse macrophages in response to poly(I-C) and IFN-γ. Mouse macrophages expressing CD46 with the Cyt1 cytoplasmic domain (A24) or CD46-negative mouse macrophages (F7) were treated with poly(I-C) (0 to 50 μg/ml), without (A) or with (B) mouse IFN-γ (500 U/ml). One or two days after treatment, culture medium was collected and assayed for NO2− accumulation. These cells did not produce detectable levels of NO on day 2 in the absence of IFN-γ. Open and stippled columns represent mean values of triplicate NO2− determinations. Bars atop the columns show standard deviation values.
These results show that both F7 and A24 lines of mouse macrophages can produce NO in response to double-stranded polyribonucleotides in the presence of IFN-γ. A24 mouse macrophages may respond more quickly to these stimuli, but there is no gross defect in NO synthesis in F7 mouse macrophages that may explain the low NO response upon MV infection.
Removing CD46 cytoplasmic domains facilitates MV replication and development of cytopathic effects in mouse macrophages.
A possible explanation for high NO production by A24 mouse macrophages upon MV infection is that CD46 may facilitate virus entry to produce higher levels of viral RNA. If this is true, changing or removing the cytoplasmic domains of CD46, which are not required for MV-induced membrane fusion or virus entry (25, 73), should not prevent NO production from mouse macrophages. To test this possibility, we examined mouse macrophages stably expressing three forms of human CD46 with identical extracellular domains and different cytoplasmic domains. A24 and B24 mouse macrophage lines expressed STP-C human CD46 isoforms with Cyt1 and Cyt2 cytoplasmic domains, respectively (25, 29). C11 mouse macrophage line expressed the ΔCyt0 CD46 mutant with the same extracellular domains but lacking the entire cytoplasmic region (25).
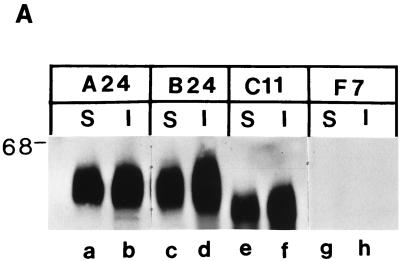
We first analyzed expression of the different CD46 isoforms and tailless mutant by surface and intracellular protein immunoprecipitation (25). The A24 and B24 lines of mouse macrophages expressed comparable levels of human CD46 with the Cyt1 and Cyt2 cytoplasmic domains, respectively, both on the cell surface and intracellularly (Fig. 3A, lanes a to d). The C11 line of mouse macrophages produced comparable levels of the tailless ΔCyt0 CD46 mutant, which appeared as a slightly smaller protein on cell surface and intracellular fractions (Fig. 3A, lanes e and f). This finding confirms that deletion of the CD46 cytoplasmic domains does not prevent transport of the mutant protein to cell surface (25).
FIG. 3.
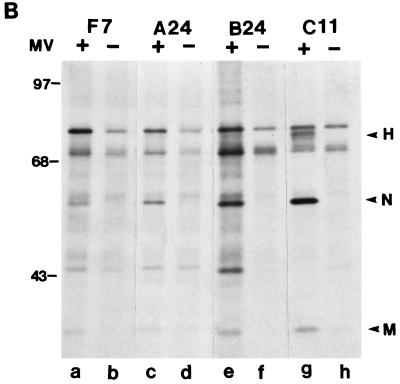
CD46 and MV protein expression in mouse macrophages. (A) Cell surface (S) and intracellular (I) proteins were immunoprecipitated with MAb M177 against human CD46 from mouse macrophages expressing CD46 with Cyt1 (A24), Cyt2 (B24), or ΔCyt0 mutant (C11) or transfected with vector alone (F7). The immunoprecipitated proteins were analyzed by Western blotting using the same MAb as described elsewhere (25). (B) F7, A24, B24, and C11 mouse macrophages were infected with Edmonston strain MV (MOI of 2). One day after infection, cells were labeled with [35S]methionine for 1 h. The cell lysates were immunoprecipitated with antiserum GM against MV proteins and analyzed by SDS-PAGE. Sizes are indicated in kilodaltons.
We infected these mouse macrophage lines with MV (Edmonston strain) and examined viral protein synthesis by [35S]methionine labeling and immunoprecipitation with an MV-specific antiserum 1 day after infection. MV-infected F7 CD46-negative mouse macrophages synthesized low levels of virus-specific proteins, including nucleocapsid (N) and matrix (M) proteins that were barely discernible from the background proteins in uninfected cells (Fig. 3B, lanes a and b, respectively). MV-infected A24 and B24 mouse macrophages expressing human CD46 with Cyt1 and Cyt2 domains, respectively, synthesized similar virus-specific proteins (Fig. 3B, lanes c and e). Interestingly, MV-infected C11 mouse macrophages expressing tailless human CD46 produced higher levels of viral proteins, including species that comigrated with N, M, and hemagglutinin (H) proteins (Fig. 3B, lane g). These results confirm that MV can infect all four mouse macrophage lines, and the C11 mouse macrophages appear to support MV infection and viral protein synthesis more efficiently than the other lines.
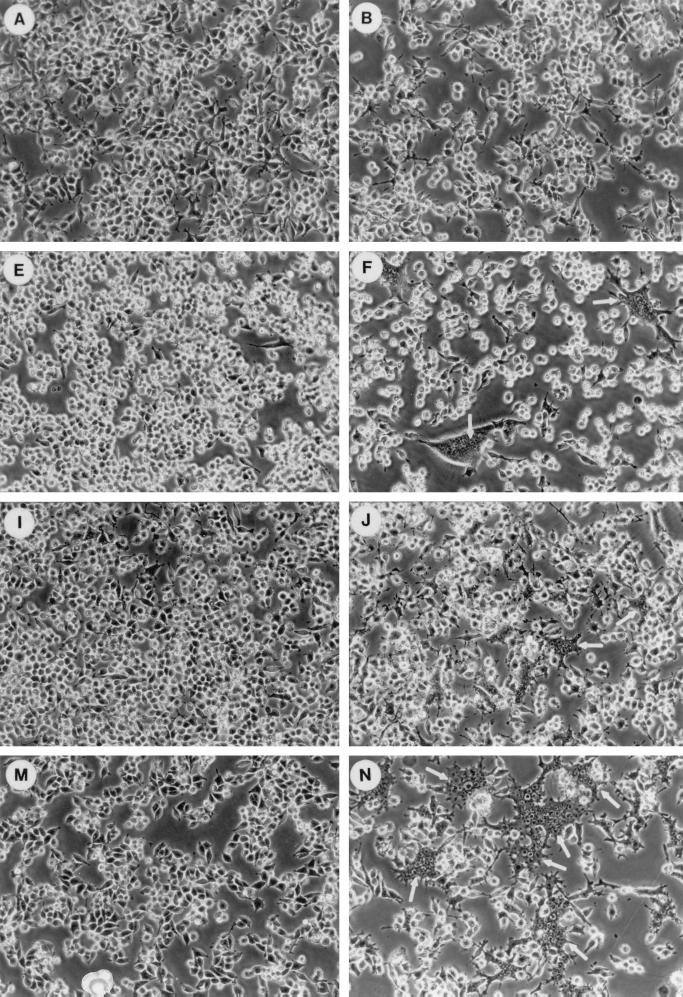
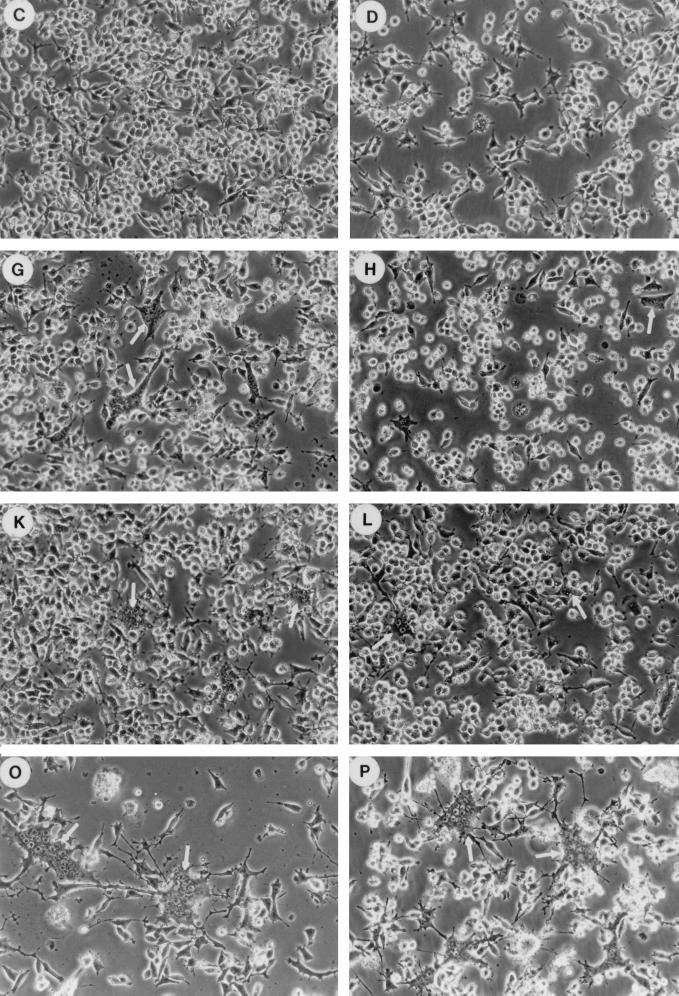
MV infection in CD46-negative mouse macrophages does not cause cytopathic effects (19, 35). Thus, MV-infected F7 macrophages were morphologically similar to uninfected macrophages on days 2 and 4 of infection (Fig. 4A to C). MV-infected A24 and B24 mouse macrophages expressing human CD46 with the Cyt1 or Cyt2 domain formed multinucleated syncytia on day 2 of infection (Fig. 4F and J). These syncytia remained localized and did not expand even after 4 days of infection (Fig. 4G and K). In contrast, C11 mouse macrophages formed very extensive syncytia on day 2 of MV infection (Fig. 4N). These syncytia expanded progressively and decimated most of the cells in the C11 culture by day 4 of infection (Fig. 4O).
FIG. 4.
Effects of CD46 cytoplasmic domains on MV infection in mouse macrophages. Mouse macrophages transfected with vector alone (F7; A to D) or expressing human CD46 with Cyt1 (A24; E to H), Cyt2 (B24; I to L), or ΔCyt0 mutant (C11; M to P) were mock infected (A, E, I, and M) or infected with MV (MOI of 1) in the absence (B, C, F, G, J, K, N, and O) or presence (D, H, L, and P) of 500 U of mouse IFN-γ per ml. Cells were examined by phase-contrast microscopy on day 2 (A, B, D, E, F, H, I, J, L, M, N, and P) or (C, G, K, and O) of infection.
Treating MV-infected F7 mouse macrophages with IFN-γ had little effects on day 2 of infection (Fig. 4D). IFN-γ treatment reduced syncytium formation in MV-infected A24 and B24 mouse macrophages on day 2 of infection (compare Fig. 4H and L to Fig. 4F and J). However, IFN-γ treatment failed to prevent the development of extensive syncytia in MV-infected C11 mouse macrophages on the same day (compare Fig. 4P and N).
Differentiated human macrophages or mouse macrophages expressing human CD46 typically do not support productive MV replication (22, 26, 35). In this experiment, F7 mouse macrophages that lacked human CD46 produced 30 to 35 PFU of infectious virus per ml on days 2 and 3 after infection (Table 1). As we reported earlier (35), A24 and B24 mouse macrophages expressing human CD46 produced even less infectious virus than the CD46-negative F7 line (Table 1). At any time after infection, the A24 culture produced no more than 5 PFU of MV per ml and the B24 culture produced no more than 15 PFU of MV per ml (Table 1), even though these cultures clearly synthesized viral proteins (Fig. 3B). Interestingly, MV-infected C11 mouse macrophages expressing the tailless CD46 mutant produced up to 1.2 × 104 PFU of infectious virus per ml on day 2 after infection (Table 1), 2 to 3 orders of magnitude more than CD46-negative F7 mouse macrophages or A24 and B24 lines expressing full-length human CD46. These results suggest that the cytoplasmic domains of human CD46 negatively affect MV replication and development of cytopathic effects in mouse macrophages.
TABLE 1.
Infectious MV production from mouse macrophages expressing human CD46
| Macrophage line | Virus titer (PFU/ml)
|
||
|---|---|---|---|
| Day 1 p.i.a | Day 2 p.i. | Day 3 p.i. | |
| F7 (CD46−) | 5 | 35 | 30 |
| A24 (Cyt1) | <5 | <5 | <5 |
| B24 (Cyt2) | 15 | 5 | <5 |
| C11 (ΔCyt0) | 300 | 1.2 × 104 | 310 |
p.i., postinfection.
CD46 cytoplasmic domains are important for augmenting NO production in mouse macrophages.
We further examined NO production in these mouse macrophage lines in response to MV infection and IFN-γ. Confirming our observations shown in Fig. 1 and 2, A24 mouse macrophages expressing human CD46 with the Cyt1 cytoplasmic domain produced higher levels of NO in response to IFN-γ than F7 CD46-negative mouse macrophages (Fig. 5A, A24 and F7, lanes c and g). MV infection further enhanced IFN-γ-induced NO production in A24 mouse macrophages 2 or 3 days after infection but not in F7 mouse macrophages (Fig. 5A, A24 and F7, lanes d and h). Interestingly, deleting the cytoplasmic domains from CD46 (C11 line) reduced NO production in response to IFN-γ and markedly attenuated synergistic augmentation of NO production by MV together with IFN-γ (Fig. 5A, C11, lanes c, d, g, and h). Even though the C11 cultures were clearly infected by MV and actively synthesizing viral proteins (Fig. 4N; Fig. 3B, lane g), these cells produced the same low levels of NO as CD46-negative F7 macrophages (Fig. 5A; compare F7 and C11). B24 mouse macrophages expressing human CD46 with the Cyt2 cytoplasmic domain also produced low levels of NO in response to MV infection and IFN-γ (Fig. 5A, B24, lanes b to d and f to h). This experiment has been repeated four times with similar results.
FIG. 5.
Effects of CD46 cytoplasmic domains on NO production from mouse macrophages in response to MV infection and IFN-γ. (A) Mouse macrophages transfected with vector alone (F7) or expressing CD46 with Cyt1 (A24), Cyt2 (B24), or ΔCyt0 (C11) were infected with Edmonston strain MV (MOI of 1). One day after infection, cells were replenished with fresh medium without or with 500 U of IFN-γ per ml. On day 2 or 3 of infection, culture medium was collected and assayed in triplicate for NO2− accumulation. Open, stippled, and striped columns represent mean values of NO2− concentrations. Bars atop the columns show standard deviation values. (B) Two additional clones of mouse macrophages expressing CD46 with Cyt1 were analyzed as for panel A.
To rule out the possibility that the A24 line mouse macrophages is an exception, we tested two other independent clones of mouse macrophages (A20 and A34) expressing human CD46 with the Cyt1 cytoplasmic domain. In the absence of IFN-γ, the A20 and A34 mouse macrophage lines produced low levels of NO upon MV infection (Fig. 5B, lanes b and f). However, the A20 and A34 lines produced high levels of NO in response to IFN-γ alone, and MV infection further enhanced IFN-γ-induced NO production just as in the A24 line (Fig. 5B, A20 and A34, lanes c, d, g, and h). We also tested two independent clones of mouse macrophages expressing human CD46 with the Cyt2 domain. Both of these clones produced lower levels of NO than the A24 line, although the absolute levels of NO varied (data not shown). We further tested an additional clone of mouse macrophages expressing the tailless ΔCyt0 mutant. These cells supported efficient MV infection without producing high levels of NO, just like the C11 line (data not shown). Finally, two other clones of CD46-negative mouse macrophages responded to MV and IFN-γ similarly to the F7 line, showing no augmenting effects on NO production (data not shown). Thus, MV synergizes with IFN-γ to enhance NO production in mouse macrophages expressing human CD46 with the Cyt1 cytoplasmic domain.
Together, these results suggest that the extracellular domains of CD46 are sufficient for MV entry and replication in mouse macrophages, but the cytoplasmic domains of CD46, particularly Cyt1, can modulate NO production and may negatively modulate MV replication in mouse macrophages.
DISCUSSION
MV infection of mouse macrophages expressing human CD46 exhibits characteristics reminiscent of those in differentiated human macrophages. In macrophages of both species, MV produces high levels of viral RNA, low levels of viral proteins, and virtually no infectious virus (22, 26, 35). Expression of human CD46 in mouse macrophages appears to exacerbate rather than alleviate the restriction of viral protein synthesis and virus production (35). We hypothesize that human CD46, while serving as a receptor for MV, may also enhance antimicrobial functions that restrict MV replication in macrophages. The present study shows that expression of human CD46 in mouse macrophages enhances NO production in response to MV infection and IFN-γ. This finding may have significant implications for possible roles of CD46 in innate immune response and MV pathogenesis.
CD46 was originally identified as a human cell receptor for laboratory-adapted MV Edmonston strain and Edmonston-like Halle strain (11, 54). Subsequent studies show that many wild-type MV strains do not interact with CD46 (4, 27, 39, 62, 63) but may utilize an unidentified receptor expressed on human and primate lymphoid cells commonly used for isolating MV from clinical specimens (4, 27, 34). After some wild-type MV is propagated in cultured primate fibroblasts, the virus exhibits increased hemadsorption activity which correlates with mutations in the viral H protein (66). Site-specific mutagenization studies show that one or two amino acid changes (e.g., Asn-481 to Tyr) in the wild-type MV H protein can allow it to bind CD46 (4, 27, 39). This raises a puzzling question: Why doesn’t wild-type MV spontaneously acquire these mutations to use ubiquitous CD46 as a receptor in human hosts? One possibility is that strong positive selection favors MV that utilizes the unidentified receptor. However, the change of Asn-481 to Tyr does not prevent H protein from binding to the putative receptor on primate lymphoid cells (27). Thus, negative selection may prevent wild-type MV from accumulating mutations that broaden the receptor-binding specificity to interact with CD46 in vivo. Our data suggest that interaction with CD46 may provoke antimicrobial responses including NO production at least in mouse macrophages.
Our present findings may explain the difficulty of using transgenic rodent species expressing human CD46 for studying MV infection. Except under special conditions described below, transgenic mice and rats expressing human CD46 generally do not support complete MV replication (8, 26, 55). Notably, B and T lymphocytes and lung and kidney cells from transgenic mice expressing human CD46 are susceptible to MV infection. By contrast, both peritoneal and bone marrow-derived macrophages do not support MV replication, due to a block at a posttranscription level (26). There have been notable reports of successful MV infection in transgenic mice expressing human CD46. In one study, a human CD46 gene was placed under control of a neuron-specific promoter, so that human CD46 expression was localized in the central nervous system (CNS) (57). After intracerebral inoculation, Edmonston strain MV caused a severe CNS infection in these mice (57). In another study, human CD46 was expressed in genetically modified mice that lacked type I IFN (IFN-α/β) receptor. In those animals, Edmonston strain MV caused widespread systemic infection after intranasal inoculation (52). These findings suggest that immunocompetent mice expressing human CD46 ubiquitously are resistant to MV infection. However, expression of human CD46 in an immunologically privileged site such as the CNS or in mice defective of type I IFN response facilitates MV replication in vivo. These observations are consistent with the hypothesis that ubiquitous expression of human CD46 in immunocompetent mice enhances antiviral response that restricts MV replication, especially in macrophages (35, 75).
The mechanisms by which CD46 augments NO production in mouse macrophages are unknown. Since poly(I-C) enhances IFN-γ-induced NO production in mouse macrophages (Fig. 2), we initially suspected that CD46 might simply increase intracellular viral RNA by facilitating virus entry. Several lines of evidence have made this interpretation unlikely. First, deleting the CD46 cytoplasmic domains does not prevent MV from infecting mouse macrophages (Fig. 3B; Fig. 4N) yet markedly attenuates the NO response (Fig. 5A). This finding indicates that MV infection alone is insufficient to enhance NO production from mouse macrophages without the CD46 cytoplasmic domains. Second, CD46 with the Cyt1 domain augments NO production more strongly than that with the Cyt2 domain (Fig. 5), even though both CD46 isoforms can serve as receptors for MV in mouse macrophages (Fig. 3B; Fig. 4F and J). Third, expression of human CD46 in mouse macrophages enhances IFN-γ-induced NO production in the absence of MV infection (Fig. 1, 2, and 5), indicating that the NO-augmenting effect of CD46 is not strictly dependent on viral RNA. CD46 normally binds complement components C3b or C4b to protect autologous cells from complement lysis (42, 65). C3b and its cleavage product iC3b are the respective ligands for complement receptors CR1 and CR3, which can activate phagocytic cell functions including calcium signaling, phagocytosis, and respiratory burst (12, 48, 79, 80). It is conceivable that in addition to the known complement regulatory function, CD46 may serve an additional role in macrophage activation by augmenting antimicrobial responses including NO production against complement-opsonized microbes.
Indeed, there is increasing evidence that CD46 may transmit signals to modulate different cell functions. In human monocytes/macrophages, MV infection or cross-linking of CD46 leads to suppression of IL-12 production (32). In human astrocytoma cells, MV infection or cross-linking of CD46 induces IL-6 (18). In human B cells, MV infection or cross-linking of CD46 synergizes with IL-4 to enhance immunoglobulin E class switching (28). CD46 is also a human cell receptor for adhesion by Neisseria gonorrhoeae (31). Binding of N. gonorrhoeae pili to human epithelial cells triggers increase in cytosolic calcium (30). The present study suggests that human CD46 can augment NO production in mouse macrophages in the presence of IFN-γ. The requirement of IFN-γ for maximal NO production may be biologically relevant. NO is toxic, and its production needs to be tightly regulated to prevent accidental damage to host cells. The synergistic effect of MV and IFN-γ may reflect a safety mechanism that ensure that macrophages release high levels of NO only when these cells are stimulated by both microbial products and activated T lymphocytes. Similarly, MV- or CD46 cross-linking-induced immunoglobulin E class switching in B cells also requires cooperation with a cytokine, IL-4 (28). Thus, CD46 appears to serve as a regulatory signaling molecule that modulates diverse cellular functions, and interaction between MV and CD46 may affect these functions. It will be of great interest to see whether the different CD46 cytoplasmic domains modulate different cellular functions.
The antiviral mechanisms of NO are not well understood. Previous studies show that NO inhibits a wide range of DNA and RNA viruses, but the degrees of inhibition vary (58). NO inhibits replication or pathogenic effects of large DNA viruses, including ectromelia virus, vaccinia virus, herpes simplex virus type 1, and Epstein-Barr virus (33, 47). Some RNA viruses, including vesicular stomatitis virus, Japanese encephalitis virus, and rhinovirus, are also sensitive to NO (7, 41, 59). Other RNA viruses, such as Sindbis virus and tick-borne encephalitis virus, are less sensitive to NO in vitro, but NO apparently influences replication or pathogenesis of these viruses in vivo (36, 71). MV replication is restricted in both A24 and B24 mouse macrophages (35), yet B24 macrophages do not produce high levels of NO (Fig. 5A). Therefore, NO alone cannot completely explain the restriction of MV replication in mouse macrophages. Since MV protein synthesis is markedly inhibited in mouse macrophages expressing human CD46 after day 2 of infection (35), these cells may produce type I IFN known to inhibit MV protein synthesis (40). Experiments are in progress to determine the nature of the antiviral factor(s) inhibiting MV replication in mouse macrophages expressing human CD46.
In addition to antimicrobial activities, NO has potent immunomodulating properties (45). For example, NO production is largely responsible for the suppressor effects of macrophages on B- and T-lymphocyte functions (1, 37, 51). NO induced by macrophages and dendritic cells can induce apoptosis in these cells as well as bystanders (2, 9, 43). These effects of NO mimic many of the immunomodulating effects associated with MV infection (see the introduction). This raises the interesting question whether NO contributes to immunosuppression and apoptosis of immune cells associated with MV infection.
In summary, the data presented here demonstrate that human CD46 enhances NO production in mouse macrophages in response to MV infection and IFN-γ, and the Cyt1 cytoplasmic domain of CD46 is important for this function. Further studies of this phenomenon may provide significant insight into mechanisms of MV pathogenesis and attenuation and into possible roles of complement regulatory proteins in innate immunity in general.
ACKNOWLEDGMENTS
We thank Alan Aderem for providing the RAW264.7 cells and for useful discussions.
This work was supported by Public Health Service grant AI41667 from the National Institutes of Health.
REFERENCES
- 1.Albina J E, Abate J A, Henry W L., Jr Nitric oxide production is required for murine resident peritoneal macrophages to suppress mitogen-stimulated T cell proliferation. Role of IFN-γ in the induction of the nitric oxide-synthesizing pathway. J Immunol. 1991;147:144–148. [PubMed] [Google Scholar]
- 2.Albina J E, Cui S, Mateo R B, Reichner J S. Nitric oxide-mediated apoptosis in murine peritoneal macrophages. J Immunol. 1993;150:5080–5085. [PubMed] [Google Scholar]
- 3.Auwaerter P G, Kaneshima H, McCune J M, Wiegand G, Griffin D E. Measles virus infection of thymic epithelium in the SCID-hu mouse leads to thymocyte apoptosis. J Virol. 1996;70:3734–3740. doi: 10.1128/jvi.70.6.3734-3740.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bartz R, Brinckmann U, Dunster L M, Rima B, ter Meulen V, Schneider-Schaulies J. Mapping amino acids of the measles virus hemagglutinin responsible for receptor (CD46) downregulation. Virology. 1996;224:334–337. doi: 10.1006/viro.1996.0538. [DOI] [PubMed] [Google Scholar]
- 5.Beckford A P, Kaschula R O C, Stephen C. Factors associated with fatal cases of measles: a retrospective autopsy study. S Afr Med J. 1985;68:858–863. [PubMed] [Google Scholar]
- 6.Bellini W J, Rota J S, Rota P A. Virology of measles virus. J Infect Dis. 1994;170:S15–S23. doi: 10.1093/infdis/170.supplement_1.s15. [DOI] [PubMed] [Google Scholar]
- 7.Bi Z, Reiss C S. Inhibition of vesicular stomatitis virus infection by nitric oxide. J Virol. 1995;69:2208–2213. doi: 10.1128/jvi.69.4.2208-2213.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blixenkrone-Moller M, Bernard A, Bencsik A, Sixt N, Diamond L E, Logan J S, Wild T F. Role of CD46 in measles virus infection in CD46 transgenic mice. Virology. 1998;249:238–248. doi: 10.1006/viro.1998.9301. [DOI] [PubMed] [Google Scholar]
- 9.Bonham C A, Lu L, Li Y, Hoffman R A, Simmons R L, Thomson A W. Nitric oxide production by mouse bone marrow-derived dendritic cells: implications for the regulation of allogeneic T cell responses. Transplantation. 1996;62:1871–1877. doi: 10.1097/00007890-199612270-00033. [DOI] [PubMed] [Google Scholar]
- 10.Borrow P, Oldstone M B A. Measles virus-mononuclear cell interactions. Curr Top Microbiol Immunol. 1995;191:85–100. doi: 10.1007/978-3-642-78621-1_6. [DOI] [PubMed] [Google Scholar]
- 11.Dorig R E, Marcil A, Chopra A, Richardson C D. The human CD46 molecule is a receptor for measles virus (Edmonston strain) Cell. 1993;75:295–305. doi: 10.1016/0092-8674(93)80071-l. [DOI] [PubMed] [Google Scholar]
- 12.Ernst J D, Rosales J L, Zimmerli S. Calcium signaling initiated by CR1(CD35) crosslinking is mediated by phagocyte Fcγ receptors in cis. Biochem Biophys Res Commun. 1995;209:1032–1038. doi: 10.1006/bbrc.1995.1601. [DOI] [PubMed] [Google Scholar]
- 13.Esolen L M, Park S W, Hardwick J M, Griffin D E. Apoptosis as a cause of death in measles virus-infected cells. J Virol. 1995;69:3955–3958. doi: 10.1128/jvi.69.6.3955-3958.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Esolen L M, Ward B J, Moench T R, Griffin D E. Infection of monocytes during measles. J Infect Dis. 1993;168:47–52. doi: 10.1093/infdis/168.1.47. [DOI] [PubMed] [Google Scholar]
- 15.Fearon D T, Locksley R M. The instructive role of innate immunity in the acquired immune response. Science. 1996;272:50–54. doi: 10.1126/science.272.5258.50. [DOI] [PubMed] [Google Scholar]
- 16.Fugier-Vivier I, Servet-Delprat C, Rivailler P, Rissoan M-C, Liu Y-J, Rabourdin-Combe C. Measles virus suppresses cell-mediated immunity by interfering with the survival and functions of dendritic and T cells. J Exp Med. 1997;186:813–823. doi: 10.1084/jem.186.6.813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gellin B G, Katz S L. Measles: state of the art and future directions. J Infect Dis. 1994;170:S3–S14. doi: 10.1093/infdis/170.supplement_1.s3. [DOI] [PubMed] [Google Scholar]
- 18.Ghali M, Schneider-Schaulies J. Receptor (CD46)- and replication-mediated interleukin-6 induction by measles virus in human astrocytoma cells. J Neurovirol. 1998;4:521–530. doi: 10.3109/13550289809113496. [DOI] [PubMed] [Google Scholar]
- 19.Goldman M B, Buckthal D J, Picciotto S, O’Bryan T A, Goldman J N. Measles virus persistence in an immortalized murine macrophage cell line. Virology. 1995;207:12–22. doi: 10.1006/viro.1995.1047. [DOI] [PubMed] [Google Scholar]
- 20.Griffin D E, Ward B J, Esolen L M. Pathogenesis of measles virus infection: a hypothesis for altered immune responses. J Infect Dis. 1994;170:S24–S31. doi: 10.1093/infdis/170.supplement_1.s24. [DOI] [PubMed] [Google Scholar]
- 21.Grosjean I, Caux C, Bella C, Berger I, Wild F, Banchereau J, Kaiserlian D. Measles virus infects human dendritic cells and blocks their allostimulatory properties for CD4+ T cells. J Exp Med. 1997;186:801–812. doi: 10.1084/jem.186.6.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Helin E, Salmi A A, Vanharanta R, Vainionpaa R. Measles virus replication in cells of myelomonocytic lineage is dependent on cellular differentiation stage. Virology. 1999;253:35–42. doi: 10.1006/viro.1998.9460. [DOI] [PubMed] [Google Scholar]
- 23.Hirano A. Subacute sclerosing panencephalitis virus dominantly interferes with replication of wild-type measles virus in a mixed infection: implication for viral persistence. J Virol. 1992;66:1891–1898. doi: 10.1128/jvi.66.4.1891-1898.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hirano A, Wong T C. Functional interaction between transcription elements in the long terminal repeat of reticuloendotheliosis virus: cooperative DNA-binding of promoter- and enhancer-specific factors. Mol Cell Biol. 1988;8:5232–5244. doi: 10.1128/mcb.8.12.5232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hirano A, Yant S, Iwata K, Korte-Sarfaty J, Okabe M, Seya T, Nagasawa S, Wong T C. Human cell receptor CD46 is downregulated through recognition of a membrane-proximal region of the cytoplasmic domain in persistent measles virus infection. J Virol. 1996;70:6929–6936. doi: 10.1128/jvi.70.10.6929-6936.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Horvat B, Rivailler P, Varior-Krishnan G, Cardoso A, Gerlier D, Rabourdin-Combe C. Transgenic mice expressing human measles virus (MV) receptor CD46 provide cells exhibiting different permissivities to MV infection. J Virol. 1996;70:6673–6681. doi: 10.1128/jvi.70.10.6673-6681.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hsu E C, Sarangi F, Iorio C, Sidhu M S, Udem S A, Dillehay D L, Xu W, Rota P A, Bellini W J, Richardson C D. A single amino acid change in the hemagglutinin protein of measles virus determines its ability to bind CD46 and reveals another receptor on marmoset B cells. J Virol. 1998;72:2905–2916. doi: 10.1128/jvi.72.4.2905-2916.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Imani F, Proud D, Griffin D E. Measles virus infection synergizes with IL-4 in IgE class switching. J Immunol. 1999;162:1597–1602. [PubMed] [Google Scholar]
- 29.Iwata K, Seya T, Yanagi Y, Pesando J M, Johnson P M, Okabe M, Ueda S, Ariga H, Nagasawa S. Diversity of sites for measles virus binding and for inactivation of complement C3b and C4b on membrane cofactor protein CD46. J Biol Chem. 1995;270:1–5. doi: 10.1074/jbc.270.25.15148. [DOI] [PubMed] [Google Scholar]
- 30.Kallstrom H, Islam M S, Berggren P-O, Jonsson A-B. Cell signaling by the type IV pili of pathogenic Neisseria. J Biol Chem. 1998;273:21777–21782. doi: 10.1074/jbc.273.34.21777. [DOI] [PubMed] [Google Scholar]
- 31.Kallstrom H, Liszewski M K, Atkinson J P, Jonsson A B. Membrane cofactor protein (MCP or CD46) is a cellular pilus receptor for pathogenic Neisseria. Mol Microbiol. 1997;25:639–647. doi: 10.1046/j.1365-2958.1997.4841857.x. [DOI] [PubMed] [Google Scholar]
- 32.Karp C L, Wysocka M, Wahl L M, Ahearn J M, Cuomo P J, Sherry B, Trinchieri G, Griffin D E. Mechanism of suppression of cell-mediated immunity by measles virus. Science. 1996;273:228–231. doi: 10.1126/science.273.5272.228. [DOI] [PubMed] [Google Scholar]
- 33.Karupiah G, Xie Q-W, Buller R M L, Nathan C, Duarte C, MacMicking J D. Inhibition of viral replication by interferon-γ-induced nitric oxide synthase. Science. 1993;261:1445–1448. doi: 10.1126/science.7690156. [DOI] [PubMed] [Google Scholar]
- 34.Kobune F, Sakata H, Sugiura A. Marmoset lymphoblastoid cells as a sensitive host for isolation of measles virus. J Virol. 1990;64:700–705. doi: 10.1128/jvi.64.2.700-705.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Korte-Sarfaty J, Pham V D, Yant S, Hirano A, Wong T C. Expression of human complement regulatory protein CD46 restricts measles virus replication in mouse macrophages. Biochem Biophys Res Commun. 1998;249:432–437. doi: 10.1006/bbrc.1998.9173. [DOI] [PubMed] [Google Scholar]
- 36.Kreil T R, Eibl M M. Nitric oxide and viral infection: no antiviral activity against a flavivirus in vitro and evidence for contribution to pathogenesis in experimental infection in vivo. Virology. 1996;219:304–306. doi: 10.1006/viro.1996.0252. [DOI] [PubMed] [Google Scholar]
- 37.Krenger W, Falzarano G, Delmonte J, Jr, Snyder K M, Byon J C, Ferrara J L. Interferon-gamma suppresses T-cell proliferation to mitogen via the nitric oxide pathway during experimental acute graft-versus-host disease. Blood. 1996;88:1113–1121. [PubMed] [Google Scholar]
- 38.Laszlo D J, Henson P M, Remigio L K, Weinstein L, Sable C, Noble P W, Riches D W H. Development of functional diversity in mouse macrophages: mutual exclusion of two phenotypic states. Am J Pathol. 1993;143:587–597. [PMC free article] [PubMed] [Google Scholar]
- 39.Lecouturier V, Fayolle J, Caballero M, Carabana J, Celma M L, Fernandez-Munoz R, Wild T F, Buckland R. Identification of two amino acids in the hemagglutinin glycoprotein of measles virus (MV) that govern hemadsorption, HeLa cell fusion, and CD46 downregulation: phenotypic markers that differentiate vaccine and wild-type MV strains. J Virol. 1996;70:4200–4204. doi: 10.1128/jvi.70.7.4200-4204.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Leopardi R, Hyypia T, Vainionpaa R. Effect of interferon-α on measles virus replication in human peripheral blood mononuclear cells. APMIS. 1992;100:125–131. doi: 10.1111/j.1699-0463.1992.tb00850.x. [DOI] [PubMed] [Google Scholar]
- 41.Lin Y-L, Huang Y-L, Ma S-H, Yeh C-T, Chiou S-Y, Chen L-K, Liao C-L. Inhibition of Japanese encephalitis virus infection by nitric oxide: antiviral effect of nitric oxide on RNA virus replication. J Virol. 1997;71:5227–5235. doi: 10.1128/jvi.71.7.5227-5235.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liszewski K M, Post T W, Atkinson J P. Membrane cofactor protein (MCP or CD46): newest member of the regulators of complement activation gene cluster. Annu Rev Immunol. 1991;9:431–455. doi: 10.1146/annurev.iy.09.040191.002243. [DOI] [PubMed] [Google Scholar]
- 43.Lu L, Bonham C A, Chambers F G, Watkins S C, Hoffman R A, Simmons R L, Thomson A W. Induction of nitric oxide synthase in mouse dendritic cells by IFN-gamma, endotoxin, and interaction with allogeneic T cells: nitric oxide production is associated with dendritic cell apoptosis. J Immunol. 1996;157:3577–3586. [PubMed] [Google Scholar]
- 44.Lublin D M, Coyne K E. Phospholipid-anchored and transmembrane versions of either decay-accelerating factor or membrane cofactor protein show equal efficiency in protection from complement-mediated cell damage. J Exp Med. 1991;174:35–44. doi: 10.1084/jem.174.1.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.MacMicking J, Xie Q-W, Nathan C. Nitric oxide and macrophage function. Annu Rev Immunol. 1997;15:323–350. doi: 10.1146/annurev.immunol.15.1.323. [DOI] [PubMed] [Google Scholar]
- 46.Manchester M, Valsamakis A, Kaufman R, Liszewski M K, Alvarez J, Atkinson J P, Lublin D M, Oldstone M B A. Measles virus and C3 binding sites are distinct on membrane cofactor protein (CD46) Proc Natl Acad Sci USA. 1995;92:2303–2307. doi: 10.1073/pnas.92.6.2303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mannick J B, Asano K, Izumi K, Kieff E, Stamler J S. Nitric oxide produced by human B lymphocytes inhibits apoptosis and Epstein-Barr virus replication. Cell. 1994;79:1137–1146. doi: 10.1016/0092-8674(94)90005-1. [DOI] [PubMed] [Google Scholar]
- 48.Matsuno R, Aramaki Y, Arima H, Adachi Y, Ohno N, Yadomae T, Tsuchiya S. Contribution of CR3 to nitric oxide production from macrophages stimulated with high dose of LPS. Biochem Biophys Res Commun. 1998;244:115–119. doi: 10.1006/bbrc.1998.8231. [DOI] [PubMed] [Google Scholar]
- 49.McChesney M B, Oldstone M B. Virus-induced immunosuppression: infections with measles virus and human immunodeficiency virus. Adv Immunol. 1989;45:335–380. doi: 10.1016/s0065-2776(08)60696-3. [DOI] [PubMed] [Google Scholar]
- 50.Medzhitov R, Janeway C A. Innate immunity: impact on the adaptive immune response. Curr Opin Immunol. 1997;9:4–9. doi: 10.1016/s0952-7915(97)80152-5. [DOI] [PubMed] [Google Scholar]
- 51.Mills C D. Molecular basis of “suppressor” macrophages. Arginine metabolism via the nitric oxide synthase pathway. J Immunol. 1991;146:2719–2723. [PubMed] [Google Scholar]
- 52.Mrkic B, Pavlovic J, Rulicke T, Volpe P, Buchholz C J, Hourcade D, Atkinson J P, Aguzzi A, Cattaneo R. Measles virus spread and pathogenesis in genetically modified mice. J Virol. 1998;72:7420–7427. doi: 10.1128/jvi.72.9.7420-7427.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Murray C J, Lopez A D. Mortality by cause for eight regions of the world: global burden of disease study. Lancet. 1997;349:1269–1276. doi: 10.1016/S0140-6736(96)07493-4. [DOI] [PubMed] [Google Scholar]
- 54.Naniche D, Varior-Krishnan G, Cervoni F, Wild T F, Rossi B, Rabourdin-Combe C, Gerlier D. Human membrane cofactor protein (CD46) acts as a cellular receptor for measles virus. J Virol. 1993;67:6025–6032. doi: 10.1128/jvi.67.10.6025-6032.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Niewiesk S, Schneider-Schaulies J, Ohnimus H, Jassoy C, Schneider-Schaulies S, Diamond L, Logan J S, ter Meulen V. CD46 expression does not overcome the intracellular block of measles virus replication in transgenic rats. J Virol. 1997;71:7969–7973. doi: 10.1128/jvi.71.10.7969-7973.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Norrby E, Oxman M N. Measles virus. In: Fields B N, Knipe D M, editors. Virology. New York, N.Y: Raven Press; 1990. pp. 1013–1044. [Google Scholar]
- 57.Rall G F, Manchester M, Daniels L R, Callahan E M, Belman A R, Oldstone M B. A transgenic mouse model for measles virus infection of the brain. Proc Natl Acad Sci USA. 1997;94:4659–4663. doi: 10.1073/pnas.94.9.4659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Reiss C S, Komatsu T. Does nitric oxide play a critical role in viral infections? J Virol. 1998;72:4547–4551. doi: 10.1128/jvi.72.6.4547-4551.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sanders S P, Sierkierski E S, Porter J D, Richards S M, Proud D. Nitric oxide inhibits rhinovirus-induced cytokine production and viral replication in a human respiratory epithelial cell line. J Virol. 1998;72:934–942. doi: 10.1128/jvi.72.2.934-942.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sanchez-Lanier M, Guerin P, McLaren L C, Bankhurst A D. Measles virus-induced suppression of lymphocyte proliferation. Cell Immunol. 1988;116:367–381. doi: 10.1016/0008-8749(88)90238-9. [DOI] [PubMed] [Google Scholar]
- 61.Schlender J, Schnorr J J, Spielhoffer P, Cathomen T, Cattaneo R, Billeter M A, ter Meulen V, Schneider-Schaulies S. Interaction of measles virus glycoproteins with the surface of uninfected peripheral blood lymphocytes induces immunosuppression in vitro. Proc Natl Acad Sci USA. 1996;93:13194–13199. doi: 10.1073/pnas.93.23.13194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Schneider-Schaulies J, Dunster L M, Kobune F, Rima B, ter Meulen V. Differential down regulation of CD46 by measles virus strains. J Virol. 1995;69:7257–7259. doi: 10.1128/jvi.69.11.7257-7259.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schneider-Schaulies J, Schnorr J-J, Brinckmann U, Dunster L M, Baczko K, Liebert U G, Schneider-Schaulies S, ter Meulen V. Receptor usage and differential downregulation of CD46 by measles virus wild type and vaccine strains. Proc Natl Acad Sci USA. 1995;92:3943–3947. doi: 10.1073/pnas.92.9.3943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Schnorr J J, Xanthakos S, Keikavoussi P, Kampgen E, ter Meulen V, Schneider-Schaulies S. Induction of maturation of human blood dendritic cell precursors by measles virus is associated with immunosuppression. Proc Natl Acad Sci USA. 1997;94:5326–5331. doi: 10.1073/pnas.94.10.5326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Seya T, Nomura M, Murakami Y, Begum N A, Matsumoto M, Nagasawa S. CD46 (membrane cofactor protein of complement, measles virus receptor): structural and functional divergence among species. Int J Mol Med. 1998;1:809–816. doi: 10.3892/ijmm.1.5.809. . (Review.) [DOI] [PubMed] [Google Scholar]
- 66.Shibahara K, Hotta H, Katayama Y, Homma M. Increased binding activity of measles virus to monkey red blood cells after long-term passages in Vero cell cultures. J Gen Virol. 1994;75:3511–3516. doi: 10.1099/0022-1317-75-12-3511. [DOI] [PubMed] [Google Scholar]
- 67.Snell J C, Chernyshev O, Gilbert D L, Colton C A. Polyribonucleotides induce nitric oxide production by human monocyte-derived macrophages. J Leukoc Biol. 1997;62:369–373. doi: 10.1002/jlb.62.3.369. [DOI] [PubMed] [Google Scholar]
- 68.Stacey K J, Ross I L, Hume D A. Electroporation and DNA-dependent cell death in murine macrophages. Immunol Cell Biol. 1993;71:75–85. doi: 10.1038/icb.1993.8. [DOI] [PubMed] [Google Scholar]
- 69.Takebe Y, Seiki M, Fujisawa J-I, Hoy P, Yokota K, Arai K-I, Yoshida M, Arai N. SRα promoter: an efficient and versatile mammalian cDNA expression system composed of the simian virus 40 early promoter and R-U5 segment of human T-cell leukemia virus type 1 long terminal repeat. Mol Cell Biol. 1988;8:466–472. doi: 10.1128/mcb.8.1.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Trinchieri G. Interleukin 12: a proinflammatory cytokine with immunoregulatory functions that bridges innate resistance and antigen-specific adaptive immunity. Annu Rev Immunol. 1995;13:251–276. doi: 10.1146/annurev.iy.13.040195.001343. [DOI] [PubMed] [Google Scholar]
- 71.Tucker P C, Griffin D E, Choi S, Bul N, Wesselingh S. Inhibition of nitric oxide synthase increases the mortality in Sindbis virus encephalitis. J Virol. 1996;70:3972–3977. doi: 10.1128/jvi.70.6.3972-3977.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Vainionpaa R, Hyypia T, Akerman K E. Early signal transduction in measles virus-infected lymphocytes is unaltered, but second messengers activate virus replication. J Virol. 1991;65:6743–6748. doi: 10.1128/jvi.65.12.6743-6748.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Varior-Krishnan G, Trescol-Biemont M C, Niniche D, Rabourdin-Combe C, Gerlier D. Glycosyl-phosphatidylinositol-anchored and transmembrane forms of CD46 display similar measles virus receptor properties: virus binding, fusion, and replication; down regulation by hemagglutinin; and virus uptake and endocytosis for antigen presentation by major histocompatibility complex class II molecules. J Virol. 1994;68:7891–7899. doi: 10.1128/jvi.68.12.7891-7899.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Vydelingum S, Suryanarayana K, Marusyk R G, Salmi A A. Replication of measles virus in human monocytes and T cells. Can J Microbiol. 1995;41:620–623. doi: 10.1139/m95-082. [DOI] [PubMed] [Google Scholar]
- 75.Wong, T. C., and A. Hirano. Role of CD46 in measles virus infection: friend or foe? Curr. Top. Virol., in press.
- 76.Wong T C, Yant S, Harder B J, Korte-Sarfaty J, Hirano A. The cytoplasmic domains of complement regulatory protein CD46 interact with multiple kinases in macrophages. J Leukoc Biol. 1997;62:892–900. doi: 10.1002/jlb.62.6.892. [DOI] [PubMed] [Google Scholar]
- 77.Yanagi Y, Hu H L, Seya T, Yoshikura H. Measles virus infects mouse fibroblast cell lines but its multiplication is severely restricted in the absence of CD46. Arch Virol. 1994;138:39–53. doi: 10.1007/BF01310037. [DOI] [PubMed] [Google Scholar]
- 78.Yant S, Hirano A, Wong T C. Identification of a cytoplasmic Tyr-X-X-Leu motif essential for down regulation of human cell receptor CD46 in persistent measles virus infection. J Virol. 1997;71:766–770. doi: 10.1128/jvi.71.1.766-770.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zarewych D M, Kindzelskii A L, Todd R F, Petty H R. LPS induces CD14 association with complement receptor type 3, which is reversed by neutrophil adhesion. J Immunol. 1996;156:430–433. [PubMed] [Google Scholar]
- 80.Zhou M-J, Brown E J. CR3 (Mac-1, αMβ2, CD11b/CD18) and FcγRIII cooperate in generation of a neutrophil respiratory burst: requirement for FcγRII and tyrosine phosphorylation. J Cell Biol. 1994;125:1407–1416. doi: 10.1083/jcb.125.6.1407. [DOI] [PMC free article] [PubMed] [Google Scholar]