Abstract
Pregnant individuals with viral illness may experience significant morbidity and have higher rates of pregnancy and neonatal complications. With the growing number of viral infections and new viral pandemics, it is important to examine the effects of infection during pregnancy on both the gestational parent and the offspring. Febrile illness and inflammation during pregnancy are correlated with risk for autism, attention deficit/hyperactivity disorder, and developmental delay in the offspring in human and animal models. Historical viral epidemics had limited follow-up of the offspring of affected pregnancies. Infants exposed to seasonal influenza and the 2009 H1N1 influenza virus experienced increased risks of congenital malformations and neuropsychiatric conditions. Zika virus exposure in utero can lead to a spectrum of abnormalities, ranging from severe microcephaly to neurodevelopmental delays which may appear later in childhood and in the absence of Zika-related birth defects. Vertical infection with severe acute respiratory syndrome coronavirus-2 has occurred rarely, but there appears to be a risk for developmental delays in the infants with antenatal exposure. Determining how illness from infection during pregnancy and specific viral pathogens can affect pregnancy and neurodevelopmental outcomes of offspring can better prepare the community to care for these children as they grow.
INTRODUCTION
When a new viral infection emerges, it is important to include pregnant people and children in research since the impact of the infection may be different in these groups. In pregnancy, there are dynamic immunological adaptations to tolerate gestation of a genetically foreign fetus while also protecting the pregnant person from infection.1,2 These immunologic changes may alter susceptibility to and severity of infections, and thus the pregnant person’s altered response to infectious diseases should be considered when planning for emerging infectious disease threats.3 Pregnant individuals have had poorer outcomes from past viral epidemics and pandemics and the offspring of affected pregnancies have not always been studied longitudinally. It is known that the developing brain is vulnerable to the effects of viral exposures throughout the fetal period and in early life. Historically, pathogens known to cause direct damage to the developing fetus have been described using the “TORCH” acronym, however the list of pathogens is now too large to fit the acronym.4 In other, less well-studied infections, or even in common febrile viral illnesses, it is difficult to distinguish whether the teratogenic effect is caused by the pathogen itself, or if the febrile and inflammatory state caused by systemic illness during pregnancy may have its own detrimental effects on the intrauterine environment and therefore fetal development.5
Since the newest iteration of the coronavirus pandemic has taken hold, and millions of pregnant individuals worldwide have been infected and given birth, it remains to be seen how this virus will affect the neurodevelopmental outcomes of children. Transplacental or vertical transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has been reported, though rare.6–8 Some reports found IgM against SARS-CoV-2 in neonatal blood shortly after birth, which suggests possible congenital infection or severe placental pathology.7,9,10 However, of greater concern is the potential impact of the heightened maternal immune response to SARS-CoV-2 and the possible cytokine storm which may occur in some individuals.
There is evidence from previous epidemiologic, animal, and birth cohort studies that maternal immune activation (MIA) and the fever response itself can affect offspring neurodevelopment.4,11–19 If that is the case, then will there be differences in neurodevelopment between children born to symptomatic parents vs. asymptomatic parents following antenatal SARS-CoV-2 infection? Will the effects in offspring differ based on the degree of symptoms and gestational age at exposure to infection? A look into pregnancy outcomes in previous viral epidemics may provide insight into what is to come following the births of infants to parents infected with SARS-CoV-2 during pregnancy.
This objective of this review is to describe maternal symptoms during pregnancy from viral infections and their potential impacts on offspring, to review outcomes from historical and seasonal viral infections, and to describe some of the more recent well-known viral infections and what is known regarding impact on offspring neurodevelopment (Figure 1). In this review, we aim to examine how illness from infection during pregnancy in addition to the specific viral pathogen affects pregnancy and neurodevelopmental outcomes of the offspring.
Figure 1: Viral exposures in pregnancy impact offspring neurodevelopment.
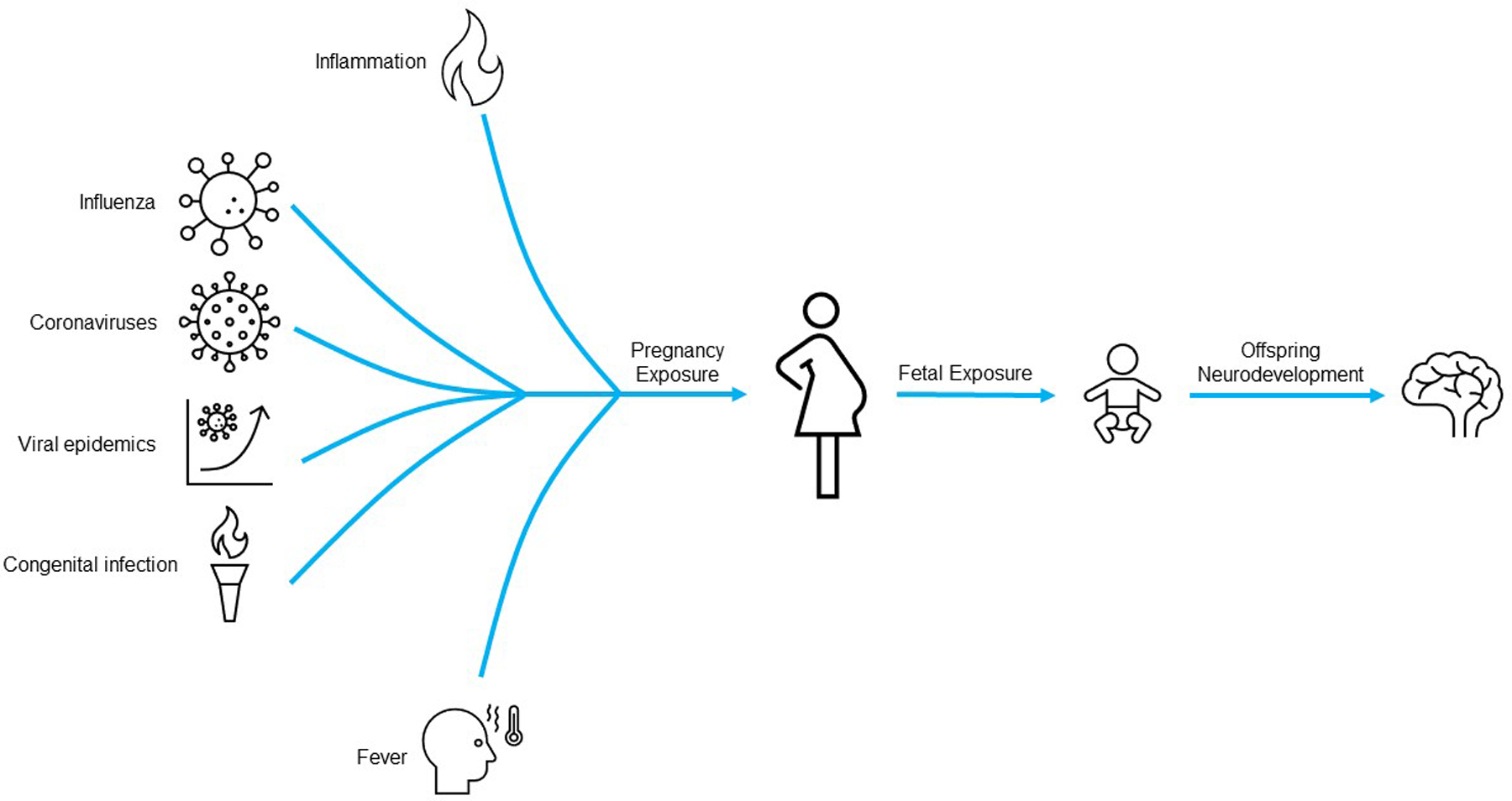
A graphical summary of how gestational exposure to multiple factors, including viral infections, fever, and immune activation in the gestational parent or the fetus may influence offspring neurodevelopment.
FEVER AS A MODULATOR OF NEURODEVELOPMENT
A hallmark of coronavirus disease 2019 (COVID-19) and of many other viral infections is fever. It is possible that the febrile state itself is teratogenic. Hyperthermia and fever during pregnancy have been suggested to cause a wide range of functional and neural tube defects and could act as a nidus for the development of neurological disorders including autism spectrum disorders (ASD), schizophrenia, and cerebral palsy.5,20 In their meta-analysis, Dreier et. al. noted an increased risk of adverse offspring health in association with fever during pregnancy, between 1.5- and 3.0-fold increased risk for neural tube defects, congenital heart defects, and oral clefts when pregnant individuals were febrile in the first trimester, as well as an increased risk for ASD and developmental delay in offspring.21 Zerbo et. al. confirmed these findings, with both ASD and developmental delay associated with exposure to fever during pregnancy.22 In agreement, Croen et. al. found that infection during the second trimester with accompanying fever elevated the risk of ASD twofold.23 Another study also found association between fever during pregnancy and developmental coordination disorders at age seven years.24 A large cohort study of fever in pregnancy and ASD was performed in Denmark and included 96,736 children aged 8–14 years born between 1997 and 2003 from the Danish National Birth Cohort. The group obtained the diagnoses of ASD and infantile autism from the Danish Psychiatric Central Register; 976 (1%) children from the cohort had a diagnosis of ASD. They found that mild, common infections and short febrile episodes during pregnancy were not associated with ASD/infantile autism, but that there was a two- and three-fold increased risk of infantile autism with prenatal exposure to influenza infections and prolonged episodes of fever, respectively.11,12 These child outcomes correlated with a more severe illness and increased inflammatory response in pregnancy. There is also evidence that antipyretic medications may offer a protective effect against neurodevelopmental disorders when used during a febrile illness in the gestational parent.21,22,25–27 However, some studies did not find a protective effect to antipyretic use.23
The mechanism of this disruption to offspring development is not clearly known, but several mechanisms have been proposed. When there is an infection in pregnancy, the maternal immune system is activated, and there are changing concentrations of cytokines within the fetal environment. Some pyogenic cytokines, such as interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor alpha (TNF-a) cause the hypothalamus to raise the set point of body temperature.28 This elevated body temperature is hypothesized to effect protein synthesis, folding, and enzyme production, which results in altered or dysfunctional cellular processes (i.e., proliferation, migration, differentiation, apoptosis).20,28 As a result of the fever, a heat shock response ensues.29 Heat shock proteins help stabilize these processes against thermal stress.28 However, the heat shock response is prioritized over other cellular activities, which can inhibit further protein synthesis and cell proliferation.20 If a fever occurs during certain windows of vulnerability during fetal neurodevelopment, it is thought that these processes can be disrupted.
Studies have shown that ASD may be associated with prenatal exposure to a wide range of infectious agents.11,15,22,23,30 The diversity of potential infectious triggers begs the question of whether host factors, such as the fever response or activation of the maternal immune system may have a role in the development of these neurodevelopmental consequences.31 Some studies have found higher levels of inflammatory cytokines in amniotic fluid32 or parental mid-gestational serum33 during the gestations of offspring with ASD compared to offspring without ASD. Parental immune responses to infection, including the timeline of febrile episodes relative to vulnerable periods in fetal brain development, may influence risk of ASD in offspring. Hornig et. al. performed a prospective cohort study in Norway to assess for an association between prenatal fever and ASD risk.26 They found that ASD risk is increased for those with fever in pregnancy, particularly when it occurs in the second trimester, and the risk is magnified with exposure to three or more fevers during the second trimester.26
Attention deficit/hyperactivity disorder (ADHD) has also been hypothesized to potentially be related to fever during pregnancy. Gustavson et. al. aimed to characterize the link between fever during pregnancy and ADHD diagnosis in offspring by age eight years through a prospective cohort study.34 They found that fever during pregnancy, especially during the first trimester, carried two and a half times higher odds of the diagnosis of ADHD in offspring compared to children unexposed to fever in utero, and these children also had the highest inattention symptoms.34 Another Danish cohort study found no overall association between exposure to fever or infection in pregnancy and ADHD occurrence in the offspring but did find higher rates of ADHD in the offspring when the exposure was considered during specific gestational periods. Specifically, genitourinary infections were associated with ADHD in offspring even after adjusting for fever, suggesting that the increase may be more associated with the local inflammatory response or treatment used to treat the infection rather than the symptom of fever alone.35 Others have wondered whether prenatal fever can also affect the offspring’s academic performance. Dreier et. al. found no association between prenatal fever and prenatal exposure to common infections and academic performance in second through eighth grade.36 In the absence of a viral pandemic, twenty percent of people report one or more episodes of fever during their pregnancy37, and this percentage is likely to be higher since the onset of widespread SARS-CoV-2 infections during the COVID-19 pandemic. It is possible that SARS-CoV-2 infections with symptoms (including fever) may be associated with higher rates of abnormal neurodevelopment, given the correlation between fever in pregnancy and neuropsychiatric disorders in the offspring.
INFLAMMATION DURING GESTATION
While fever and inflammation often co-occur in infections, inflammation can occur without a fever; thus, it is important to distinguish the two when considering causative agents for fetal neurodevelopmental impacts during pregnancy. It is hypothesized that pregnant individuals in their first and third trimester are in a pro-inflammatory state, and an infection could lead to even more severe inflammation when occurring during these more vulnerable periods of pregnancy.1,2 Inflammation in the gestational parent has also been associated with changes in fetal brain development.5,38,39 Cytokines, a cornerstone of inflammation, are regulators of neuronal migration, proliferation, and neuronal cellular differentiation during brain development.40 Proinflammatory cytokines, however, are known to cause neurotoxicity and may cause direct damage to oligodendrocytes and neurons through activation of microglia which can lead to subsequent neurobehavioral abnormalities from antenatal exposure to inflammation.41,42 Jones et. al. sought to measure levels of cytokines in the maternal serum for children who had ASD with and without intellectual disability and compared them to control counterparts. The study found significantly increased levels of proinflammatory cytokines in gestational parents of children with ASD with intellectual disability, but not in gestational parents of children with ASD alone or gestational parents of control children.43 These findings contrast with a study with a smaller sample size, which found that increased levels of proinflammatory cytokines in mid-gestational parental serum were associated with parents of children with ASD vs. without.33 Another study reported a possible link between immune activation in the gestational parent and offspring intelligence and executive function.44
C-reactive protein (CRP) is an acute phase reactant often associated with low-grade inflammation in both infectious and non-infectious states and is activated by IL-6.45 From the Finnish National Birth Registry, Brown et. al. studied the stored serum of the pregnant parent drawn in the first and early second trimesters from over one million births.46 They found that gestational parents with CRP levels in the highest quintile compared to the lowest quintile, had a 43% elevated risk of ASD in the offspring. This finding suggests that CRP elevation in the first or early second trimesters may be an early biomarker for ASD, and that inflammation may have a significant role in the subsequent development of ASD for some individuals.46 Inflammatory exposure of the fetal brain during gestation may thus be an important factor in affecting offspring neurodevelopmental outcomes.
PREVIOUS CORONAVIRUS EPIDEMICS AND PANDEMICS: PREGNANCY AND NEONATAL OUTCOMES
Viruses have been affecting the maternal-placental-fetal triad for centuries with impacts on pregnancy and offspring outcomes (Figure 1). During the worldwide outbreak of Severe Acute Respiratory Syndrome (SARS) in 2003, the number of cases in pregnant individuals was too small to draw definite conclusions as to whether SARS was more severe in pregnancy than otherwise, but some editorials from hospitals with high numbers of cases of pregnant SARS patients described adverse fetal effects and increased risk for critical illness in the gestational parent.47 The largest case series of pregnant people from the 2003 SARS outbreak was from Hong Kong, where twelve pregnant people with SARS were admitted to five public hospitals; three of them died, giving a case-fatality rate of 25%.48 In a case-control study conducted in the same region, pregnant patients with SARS had more severe disease and increased risk of admission to intensive care units (ICU) than nonpregnant patients.49 Live-born infants did not acquire the infection through vertical transmission, although some fetuses had severe intrauterine growth restriction and preterm neonates sometimes developed necrotizing enterocolitis.50,51
In a systematic review of studies including 79 hospitalized pregnant patients with coronavirus infections (41 with COVID-19, 12 with [Middle Eastern Respiratory Syndrome] MERS, and 26 with SARS), greater than 90% of whom also had pneumonia, preterm birth was the most common pregnancy complication.52 Preeclampsia and cesarean delivery were also more common than in the general population.52 In this study, the most common adverse neonatal outcome was fetal distress, with more than half of all newborns admitted to the neonatal intensive care unit (NICU).52 Fortunately, there were no cases of death from COVID-19 among the gestational parents in this study, however in pregnant patients with MERS and SARS infections the mortality pooled proportion in pregnant patients ranged from 25%−30%.52 Very few pregnancy-associated cases of MERS coronavirus infection have been documented (n=11), but according to Alfaraj et. al., 91% of them had adverse clinical outcomes. Six pregnant patients required intensive care and three died.53 The infant death rate was 27%, and the maternal death rate was 27%, similar to the overall case fatality rate of MERS in the general population.53 Overall, there is strong epidemiologic evidence that pregnant patients are at higher risk of severe illness and mortality from viral infections, most notably during pandemics including influenza (discussed later in this review).54–56 High symptomatology in gestational parents has potential risk to offspring neurodevelopment as described, however, conclusions have not been made in studies of prior coronavirus epidemics and pandemics.
INFLUENZA: SEASONAL AND PANDEMIC
Pregnant patients are at elevated risk for severe illness and complications from influenza during seasonal influenza3,54–57 and pandemics.56,58–61 Although proper control groups were not used for comparison, mortality rates among pregnant patients in the influenza pandemics of 1918 and 1957 appeared to be abnormally high.59,61 Among 1,350 reported cases of influenza among pregnant individuals in Maryland, USA during the pandemic of 1918, the proportion of deaths was reportedly 27%, with a rate of 45% deaths among 86 pregnant individuals in Chicago, USA.59 In Minnesota, USA during the 1957 pandemic, influenza was the leading cause of death in pregnant individuals, accounting for nearly 20% of deaths in pregnant individuals during the pandemic period. Half of the individuals of childbearing age who died from influenza in Minnesota during this period were pregnant.61 A systematic review examining the effects of pandemic influenza A (H1N1 2009), found H1N1 caused significantly more disease of higher severity in pregnant patients compared with age-matched nonpregnant controls, including pneumonia and subsequent acute respiratory distress syndrome (ARDS) requiring mechanical ventilation.58 Pregnancy was associated with increased risk of hospital and ICU admission, and of death.62 Preterm and emergency cesarean-section deliveries were frequently reported.63 In a United Kingdom cohort study, neonatal mortality rate was higher in infants born to infected patients (39 per 1,000 total births) than in infants of uninfected patients (7 per 1,000 total births). This could be explained by the increase in rates of stillbirth and preterm births.64 In terms of neurodevelopmental outcomes, there has been limited long-term follow-up of children born to parents who had H1N1 2009 infections. In one study, early prenatal exposure to pandemic H1N1 influenza in gestational weeks 0–8 was modestly associated with delayed development and higher temperamental scores compared to non-exposed infants at age 6 months. Similar effects were not noted in infants exposed after the 9th gestational week.65
Even in non-pandemic influenza, pregnant individuals are at higher risk of adverse effects compared to non-pregnant individuals of similar age. In a study of over 4,000 patients of childbearing age during nineteen influenza seasons, pregnant patients were compared to postpartum patients (a group considered to be most similar to pregnant patients in demographics and health status) and were found to be significantly more likely to be hospitalized for a cardiopulmonary event during influenza season.57
In addition, some studies suggest an increased risk for other adverse outcomes among infants born to gestational parents with influenza infections during pregnancy, including neural tube defects, cleft lip and palate, cardiovascular malformations, and neuropsychiatric conditions identified during childhood.27,38,66 A population-based cohort study reported almost a twofold increased risk of ASD after self-reported influenza infection during pregnancy.12 Deykin and MacMahon determined that the offspring of nearly 8% of pregnant patients who had influenza or exposure to influenza during their pregnancy later developed ASD.67 Animal and birth cohort studies have linked development of schizophrenia in offspring to influenza infection of the pregnant parent during pregnancy.17–19 However, Zerbo et. al. found that influenza infection was not associated with increased risk of ASD or developmental disorders, but fever was associated, as described above.22 Other studies have reported that influenza infection at any time during pregnancy was not associated with increased risk of development of ASD or developmental delay in offspring.68,69 It seems that a diverse selection of respiratory viruses, including seasonal and pandemic influenza viruses, have the potential to contribute to poorer pregnancy outcomes and may alter the gestational environment.
ANIMAL MODELS
Animal studies have served as helpful models for maternal immune activation (MIA), especially the polyinosinic:polycytidylic acid (poly(I:C)) model, which uses a synthetic analog of double stranded RNA to mimic a viral infection in the host.70 In a study using this model, pregnant mice were injected with poly(I:C), producing offspring that exhibit autistic features of increased repetitive and stereotyped behaviors and deficient social and communitive behaviors.71 This concurs with another study using the poly(I:C) model which noted highly abnormal behavior including deficits in prepulse inhibition in the acoustic startle response of animals exposed to early inflammation.72 Other studies have shown abnormalities in communication, altered social behavior, repetitive behaviors, impaired learning and memory in mice exposed to inflammation during gestation.73 The extent and type of abnormality has been associated with the timing and severity of inflammatory response during pregnancy.41
The inflammatory cytokine IL-6 has been identified as a key intermediary in the neurobehavioral development of offspring following maternal inflammation. Smith et. al. reported that a single maternal injection of IL-6 during mid-gestation of mouse pregnancy causes altered behavior in offspring resembling ASD and schizophrenia, such as decreased prepulse inhibition and latent inhibition defects.74 However, these behavior changes were prevented with the administration of anti-IL-6 antibody in the poly(I:C) model and were not seen with maternal immune activation in IL-6 knockout mice.74 A recent prospective longitudinal study found that maternal IL-6 was inversely associated with human offspring cognitive development at 12 months of age.75 This study noted changes in fronto-limbic white matter (important structure in socioemotional and cognitive development) detected on brain magnetic resonance imaging scans that were associated with maternal IL-6 concentrations during pregnancy.75 Another study noted sustained alterations in patterns of microglial process motility and behavioral deficits in the brains of embryonic mice in the setting of MIA induced in the early second trimester.76 Other studies found that interleukin-17a and Th17 cells are critical drivers in inducing ASD-like phenotypes in offspring of rodent mothers subjected to immune activation during pregnancy.39,77 Pregnant rhesus monkeys given poly(I:C) produced offspring with abnormal repetitive behaviors, altered communication, and atypical social interactions, all behaviors resembling ASD and schizophrenia.78 These findings strengthen the concern that MIA can adversely affect the intrauterine environment, which may in turn have neurodevelopmental consequences on offspring.1,2,5
Similar behavioral changes have been seen in animal models with influenza infection during pregnancy. Prenatal influenza infection in the mouse has been associated with histopathologic changes in the brain79 and behavioral alterations in offspring resembling ASD and schizophrenia.17 Respiratory infection of pregnant mice with human influenza virus yields offspring that exhibit highly abnormal behavior, including deficits in pre-pulse inhibition in the acoustic startle response, deficiency in exploratory behavior, and deficiency in social interaction.72 Although influenza viral RNA has not been found in the fetal brain in mice, these changes suggest that fetal effects could be secondary to MIA, rather than the result of a direct viral effect.16 These findings have also been seen in the rhesus monkey. Short and Lubach noted significant reductions in gray matter throughout most of the cortex and decreased white matter in the parietal cortex in the offspring of rhesus mothers inoculated with moderately virulent H3N2 influenza in the early third trimester; these changes were still present at the monkey-equivalent of early childhood, so it is suspected that these changes were permanent.80 Despite these brain structural differences, early behavioral functioning and stress response were similar to rhesus control subjects. One of the few differences the group noted was that the flu-exposed offspring were quicker to develop autonomy from the mother by four months of age, which is an effect that can be seen in other psychologically stressed pregnancies.80
Critical cellular pathways and neuronal function/connectivity are altered when exposed to MIA in mice, including derangements in neurotransmitter systems. Glutamatergic, GABAergic, dopaminergic, serotonergic, and cholinergic systems seem to be particularly vulnerable to MIA.81 Expression of genes coding for proteins involved in these systems are affected by epigenetic changes such as DNA methylation, which have been associated with MIA.82–85 In particular, inflammation can NMDA receptor signaling and the developmental excitatory-inhibitory switch in GABAergic pathways.81,83,86 Previously discussed animal model studies indicate a number of ways in which exposure to inflammation during the fetal period can affect various processes in the developing brain, and lead to behavioral changes resembling neuropsychiatric disorders in offspring.
FETAL BRAIN DYSGENESIS
Within the maternal-placental-fetal triad, interactions between genetic and epigenetic factors can damage sensitive parts of the developing brain that contain early neuronal and glial cell lineages. Some populations of progenitor cells seem to be affected by MIA depending on the timing of exposure. Microglial motility, migration, and function have been noted as abnormal when exposed to inflammation during gestation, with earlier exposure in the first half of pregnancy leading to abnormal differentiation pathways that adversely affect signaling, synapse formation, and neuronal connectivity in the second half of pregnancy.76,87,88 Imbalances in cell positioning and connectivity affect the development and interactions between various types of neurons and glial cells, which in turn disrupts their proper differentiation. Interneurons participate in signaling in very early fetal brain development, and with injury, these excitatory and inhibitory properties can malfunction and affect mature brain function which are later expressed as a range of neurodevelopmental disorders including intellectual disability, ASD, and epilepsy.89,90
During the second half of pregnancy, abnormal migration, dendritic arborization, synaptogenesis, and myelination contribute to early brain dysgenesis that originated during the first half of pregnancy.91 Para et al. describes two forms of fetal inflammatory response syndrome (FIRS) that may differentiate earlier vs. later effects on the brain based on distinct pathologic processes.92 FIRS Type I has been described with intra-amniotic infections from ascending lower genital tract infections and FIRS type II has been associated with chronic inflammatory lesions of the placenta.93–95 In a study using RNA sequencing of human cord blood samples from preterm infants diagnosed with FIRS type I or FIRS type II, FIRS Type I was found to be characterized by an upregulation of host immune responses, including neutrophil and monocyte functions, together with a proinflammatory cytokine storm and a downregulation of T cell processes, indicating involvement of the innate limb of immunity consistent with host defense. In contrast, FIRS type II comprised a mild chronic inflammatory response involving perturbation of HLA transcripts, suggestive of fetal semiallograft rejection.92 During critical periods of brain development during the fetal period, exposure to MIA or FIRS may disrupt populations of progenitor cells, ultimately leading to compounding effects on cell communication, differentiation, myelination, and connectivity. These changes could lead to visible changes in offspring behavior.
COVID-19 (SARS-CoV-2) PANDEMIC
Between January 20, 2020 and July 25, 2022, there were 225,656 cases of COVID-19 in pregnant individuals reported to the U.S. Centers for Disease Control and Prevention (CDC) and 306 deaths.96 Based on data from previous pandemics, pregnant individuals are at higher risk of acquiring infection and dying compared to nonpregnant counterparts.59,61,97 Owing to differences in physiology during pregnancy, pregnancy represents a vulnerable state that may be associated with worse outcomes in COVID-19.98,99 Pregnant and recently pregnant patients with COVID-19 admitted to hospitals for any reason were more likely to be admitted to the ICU or need invasive ventilation than non-pregnant patients of a similar age group, and risks were even higher in pregnant Hispanic and non-Hispanic Black patients.100–103 In analysis of COVID-19 surveillance data from January to June 2020, the CDC found that more than 2,500 (31.5%) pregnant people with COVID-19 required hospitalization, much higher than non-pregnant cohorts (5.8%).100,103 A systematic review and meta-analysis demonstrated that a high proportion of pregnant people with COVID-19 had preterm birth (22%) and cesarean delivery (48%).99,100,104,105 Their babies are more likely to be admitted to the NICU.98 SARS-CoV-2 can cause placentitis from direct infection of the placenta and can lead to stillbirth and neonatal death due to poor perfusion and placental insufficiency.106 The Delta variant was associated with more severe outcomes than pre-Delta for pregnancy: five times higher risk of admission to ICU, 76% higher risk of invasive ventilation or needing extracorporeal membrane oxygenation (ECMO), 1.3 times higher risk of death than that of nonpregnant people.107 The Omicron variant spread rapidly but appeared to cause less severe disease than the Delta variant.108 A study in Scotland found that 77.4% of nearly 5,000 COVID-19 cases, 90.9% of COVID-associated hospital admissions, and 98% of COVID-associated critical care admissions as well as all infant deaths occurred in pregnant people who were unvaccinated at the time of COVID-19 diagnosis (compared to fully vaccinated pregnant people). Vaccine uptake had been poor among pregnant people in Scotland, with only 32.3% of pregnant people giving birth in October 2021 having had two doses of vaccine compared to 77.4% in all people of childbearing age.109 In both variants, most early neonatal SARS-CoV-2 infections occurred among unvaccinated pregnant patients with non-severe COVID-19.110 Pregnant individuals are a particularly vulnerable group with regard to COVID-19 severity, and the risks of poor neonatal outcomes are elevated with severe COVID-19. Some of the differences in neurodevelopment we may see in offspring exposed to SARS-CoV-2 may be related to the adverse effects of severe illness including need for maternal intensive care, preterm delivery, cesarean delivery, and need for higher levels of neonatal care.
COVID-19 AND NEURODEVELOPMENT: CURRENT KNOWLEDGE
The COVID-19 pandemic sparked significant interest in potential adverse effects of viral-triggered hyperinflammatory states on pregnancy and offspring. It has been suspected that vagal nerve suppression of proinflammatory cytokine release in the cholinergic anti-inflammatory pathway (CAP) can play a role in the severity of SARS-CoV-2 infection. It is possible that disruption of either the maternal or fetal CAP could exacerbate the severity of gestational hyperinflammation and lead to increased risk for neurodevelopmental disorders in the offspring.111 Another possible mechanism that could potentially contribute to increased risk of neurodevelopmental disorders is alteration in DNA methylation patterns. Viral infection during pregnancy is thought to affect neurodevelopment through epigenetic mechanisms, and a recent study out of Victoria, Australia has found changes in DNA methylation in genes involved in pathways that play a role in neurodevelopment in human infants exposed to SARS-CoV-2 infection during gestation.85 SARS-CoV-2 is known to infect the placenta and can lead to preeclampsia, preterm birth, and intrauterine growth restriction, which on their own can lead to adverse neurodevelopmental outcomes.112 Disruption of the maternal-fetal interface could lead to more inflammatory cytokines crossing the placenta into the newborn, causing developmental delay.112,113 Fetal brain injury may also be possible but has been infrequently reported. A case report from Germany reported a pregnant individual with only mild symptoms of COVID-19 in the second trimester who presented in preterm labor with elevated serum inflammatory markers. The infant was delivered prematurely and had bilateral intracranial bleeds which the group suspects were caused by a post-COVID systemic inflammatory response produced coagulopathy affecting fetal circulation.114
Because of the rather small collective level of knowledge and heterogeneity of COVID-19 infections, such as strain variability, symptoms, gestational age, vaccination status, and co-morbidities of the gestational parent, it is difficult to define a congenital viral syndrome for SARS-CoV-2. Clinical and epidemiologic data show a myriad of possible neurodevelopmental disabilities following prenatal viral infection. It is critical to understand these possible outcomes, especially in a disease as prevalent as COVID-19. There is evidence that antenatal and early neonatal exposure to SARS-CoV-2 with symptomatic infection of the gestational parent is associated with lower scores on developmental screening tools in the first six months of age.115 Edlow et. al. found that infants prenatally exposed to SARS-CoV-2 were more likely to be diagnosed with a neurodevelopmental disorder than those unexposed, even after accounting for preterm births.116
Other studies have not been able to verify this link, and instead associate being born during a pandemic (without exposure to prenatal SARS-CoV-2 infection) as the risk factor for developing a neurodevelopmental disorder.117,118 A study from Kuwait found no increase in the rate of developmental delay in infants exposed to SARS-CoV-2 infection during gestation compared to a historical control of infants born before the COVID-19 pandemic.119 Longitudinal studies of child neurodevelopmental outcomes are needed for infants born with antenatal exposure to SARS-CoV-2, as changes in neurodevelopment may not appear until later childhood or adolescence.120
It cannot be understated that being pregnant and becoming a new parent can be stressful experiences – to do this during a global pandemic with social isolation is undoubtedly more stressful, even if one does not become ill.121,122 Critical clinical protocols were put in place to protect and care for newborns early in the pandemic, but these efforts had a negative effect on mental health in pregnancy.123 On top of the isolation and fear of COVID-19 unknowns, the stress of becoming significantly ill with a virus can impact the developing fetus. Pregnant patients during the pandemic had higher levels of stress, anxiety, and depression compared to pre-pandemic pregnant controls.124 In a quantitative fetal brain magnetic resonance imaging study, fetal brain volumes were decreased and fetal cortical gyration was delayed in pregnancies during the COVID-19 pandemic compared to pre-pandemic pregnancies.124 It is known that postnatal bonding has important implications for infant brain development, and the consequences of maternal mental health conditions on offspring may not be evident for years to come.121 Thus, neurodevelopmental effects on offspring of the pandemic may relate to a number of factors, including viral exposure in utero, changes in gestational environment, maternal mental health both prenatally and postnatally, and underlying structural brain differences.
CONCLUSION AND FUTURE DIRECTIONS
Pregnancy has always been a vulnerable state, as highlighted by the poorer outcomes experienced by this population in the current COVID-19 pandemic as well as during prior viral epidemics and seasonal virus infections over the last century. The emergence of Zika virus (ZIKV) as a viral teratogen in 2015 emphasized the need to consider viral epidemics in pregnancy. When infected pregnant individuals, with only mild or no symptoms, began to give birth, the devastating effects of congenital Zika infection became glaringly clear.125,126 Infants were born with eye abnormalities and hearing loss, seizures, hypertonia, contractures, decreased brain tissue with subcortical calcifications, and severe microcephaly with partial skull collapse.127–130 However, it is important to note that not every Zika-exposed child had birth defects and severe symptoms, and that many children appeared normal at birth and only displayed differences in the development as they grew older. Studies have shown below average scores on developmental testing or development of abnormalities in hearing or vision in children exposed to ZIKV in utero.131–136 These findings stress the importance of continued follow-up of exposed infants and children, even when their initial evaluations do not suggest neurologic changes. In fact, the CDC recommends long-term follow-up of all infants exposed to ZIKV in utero, not just those with manifestations of congenital Zika syndrome at birth.130,134 Once effects of SARS-CoV-2 infection in pregnancy are known, it is likely that offspring from these pregnancies will need similar longitudinal neurodevelopmental follow-up.
The “exposome”, a term coined in 2005 by Wild, is an evolving concept of the totality of nonheritable factors affecting the genome, and the neural exposome would encompass those factors affecting the genome responsible for neurologic processes and plasticity.137 It includes endogenous factors (genes/epigenetics, pre-existing conditions, metabolism, microbiome, xenobiotics), exogenous factors (environmental toxicants, light and noise, climate, ecosystems, economics/education) and behavioral factors (psychosocial effects, drugs/alcohol/tobacco, lifestyle, sleep, stress).138 Exposure to inflammation in utero would be included in the exposome. Since the exposome includes all lifetime exposures, it seems that individuals are at the greatest risk for damage if insults occur during particularly critical periods of development (such as the embryonic/fetal/neonatal period).88,139 Developmental origins and life-course theories stress the relevance of fetal and neonatal development when considering the diagnosis and treatment of adult-onset cerebrovascular, cognitive, and neurodegenerative disorders.88,140 Disruption of brain development in the fetal or neonatal period often precedes childhood to adulthood disease expression by months to years.139 With an event as widespread as COVID-19, it is important to establish rigorous studies and follow offspring outcomes, so we may have an opportunity to identify the delayed onset of diseases that may be associated with SARS-CoV-2 exposure in utero.
Fetal exposure to fever, inflammation, and immune activation in the pregnant parent during gestation at certain developmental windows are likely very important ways in which viral infection in pregnancy impacts offspring neurodevelopment. In the absence of direct teratogenic effects from the virus, this exposure risk seems true regardless of the identity of the viral pathogen. For SARS-CoV-2, the scientific community is still relatively early in its study of the effects on pregnancy, birth outcomes, and infant and child neurodevelopment. However, compared to past viral epidemics and pandemics, more studies seem to be underway that will address the vulnerable populations of pregnant individuals and children and answer important questions on the risk to offspring neurodevelopment.
Future research should aim to provide serial longitudinal data linking viral infections in pregnancy to long-term outcomes in offspring. It is likely that many viral exposures in pregnancy affect offspring in similar ways and so lessons learned from one antenatal viral exposure may be applicable to other viral exposures in pregnancy or the next worldwide pandemic. Further delineation of effects on neurodevelopment, such as strain variability, symptomatology, fevers, antipyretic or anti-inflammatory use, and effects of vaccination in preventing developmental delay in children are needed. Characterization of developmental delays in children associated with congenital or early-in-life exposure to viral infections can help inform recommendations for early interventions, therapies, and treatments to help improve developmental trajectories. Clinicians caring for young children should inquire about exposure to infection during pregnancy and when present, screen for signs of developmental delay or behavioral disorders. Viral infections in pregnancy will continue, therefore it is critical that we understand the mechanisms by which they can impact offspring neurodevelopment so we can develop ways to protect and better support the developing brain.
Impact.
Viral infections have impacted pregnant people and their offspring throughout history.
Antenatal exposure to maternal fever and inflammation may increase risk of developmental and neurobehavioral disorders in infants and children.
The recent SARS-CoV-2 pandemic stresses the importance of longitudinal studies to follow pregnancies and offspring neurodevelopment.
Funding:
Dr. Mulkey receives research support from the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health (grant number R01HD102445 [PI: SBM] and R01HD107140 [Co-I: SBM]) for studies on Zika virus and SARS-CoV-2, respectively. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Disclosure Statement:
Dr. Sarah Mulkey receives grant funding from the National Institutes of Health as described in funding. Dr. Mulkey and Ms. Emma Yates do not have conflicts of interest to disclose.
Footnotes
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
REFERENCES
- 1.Liu H et al. Why are pregnant women susceptible to COVID-19? An immunological viewpoint. J. Reprod. Immunol 139, 103122 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mor G, Aldo P & Alvero AB The unique immunological and microbial aspects of pregnancy. Nat. Rev. Immunol 17, 469–482 (2017). [DOI] [PubMed] [Google Scholar]
- 3.Jamieson DJ, Theiler RN & Rasmussen SA Emerging Infections and Pregnancy. Emerg Infect 12111638–1643 12, (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fortin O & Mulkey SB Neurodevelopmental outcomes in congenital and perinatal infections. Curr. Opin. Infect. Dis 36, (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shuid AN et al. Association between Viral Infections and Risk of Autistic Disorder: An Overview. Int. J. Environ. Res. Public. Health 18, (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hosier H et al. SARS-CoV-2 infection of the placenta. J. Clin. Invest 130, 4947–4953 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dong L et al. Possible Vertical Transmission of SARS-CoV-2 From an Infected Mother to Her Newborn. JAMA 323, 1846–1848 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pulinx B et al. Vertical transmission of SARS-CoV-2 infection and preterm birth. Eur. J. Clin. Microbiol. Infect. Dis 39, 2441–2445 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zeng H et al. Antibodies in Infants Born to Mothers With COVID-19 Pneumonia. JAMA 323, 1848–1849 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zaigham M et al. Clinical-pathological features in placentas of pregnancies with SARS-CoV-2 infection and adverse outcome: case series with and without congenital transmission. BJOG Int. J. Obstet. Gynaecol 129, 1361–1374 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Atladóttir HÓ et al. Maternal Infection Requiring Hospitalization During Pregnancy and Autism Spectrum Disorders. J. Autism Dev. Disord 40, 1423–1430 (2010). [DOI] [PubMed] [Google Scholar]
- 12.Atladóttir HÓ, Henriksen TB, Schendel DE & Parner ET Autism After Infection, Febrile Episodes, and Antibiotic Use During Pregnancy: An Exploratory Study. Pediatrics 130, e1447–e1454 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bohmwald K, Andrade CA & Kalergis AM Contribution of Pro-Inflammatory Molecules Induced by Respiratory Virus Infections to Neurological Disorders. Pharmaceuticals 14, (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zimmer A, Youngblood A, Adnane A, Miller BJ & Goldsmith DR Prenatal exposure to viral infection and neuropsychiatric disorders in offspring: A review of the literature and recommendations for the COVID-19 pandemic. Brain. Behav. Immun 91, 756–770 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zerbo O et al. Maternal Infection During Pregnancy and Autism Spectrum Disorders. J. Autism Dev. Disord 45, 4015–4025 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shi L, Tu N & Patterson PH Maternal influenza infection is likely to alter fetal brain development indirectly: the virus is not detected in the fetus. Int. J. Dev. Neurosci 23, 299–305 (2005). [DOI] [PubMed] [Google Scholar]
- 17.Patterson PH Immune involvement in schizophrenia and autism: Etiology, pathology and animal models. Behav. Brain Res 204, 313–321 (2009). [DOI] [PubMed] [Google Scholar]
- 18.Mednick SA, Machon RA, Huttunen MO & Bonett D Adult Schizophrenia Following Prenatal Exposure to an Influenza Epidemic. Arch. Gen. Psychiatry 45, 189–192 (1988). [DOI] [PubMed] [Google Scholar]
- 19.Brown AS et al. Serologic evidence for prenatal influenza in the etiology of schizophrenia. Abstr. IXth Int. Congr. Schizophr. Res 60, 34 (2003). [Google Scholar]
- 20.Edwards MJ, Saunders RD & Shiota K Effects of heat on embryos and foetuses. Int. J. Hyperthermia 19, 295–324 (2003). [DOI] [PubMed] [Google Scholar]
- 21.Dreier JW, Andersen A-MN & Berg-Beckhoff G Systematic Review and Meta-analyses: Fever in Pregnancy and Health Impacts in the Offspring. Pediatrics 133, e674–e688 (2014). [DOI] [PubMed] [Google Scholar]
- 22.Zerbo O et al. Is Maternal Influenza or Fever During Pregnancy Associated with Autism or Developmental Delays? Results from the CHARGE (CHildhood Autism Risks from Genetics and Environment) Study. J. Autism Dev. Disord 43, 25–33 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Croen LA et al. Infection and Fever in Pregnancy and Autism Spectrum Disorders: Findings from the Study to Explore Early Development. Autism Res. 12, 1551–1561 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Holst C, Jørgensen SE, Wohlfahrt J, Nybo Andersen A-M & Melbye M Fever during pregnancy and motor development in children: a study within the Danish National Birth Cohort. Dev. Med. Child Neurol 57, 725–732 (2015). [DOI] [PubMed] [Google Scholar]
- 25.Currenti SA Understanding and Determining the Etiology of Autism. Cell. Mol. Neurobiol 30, 161–171 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hornig M et al. Prenatal fever and autism risk. Mol. Psychiatry 23, 759–766 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ács N, Bánhidy F, Puhó E & Czeizel AE Maternal influenza during pregnancy and risk of congenital abnormalities in offspring. Birt. Defects Res. A. Clin. Mol. Teratol 73, 989–996 (2005). [DOI] [PubMed] [Google Scholar]
- 28.Martin RP & Dombrowski SC Prenatal Exposures: Psychological and Educational Consequences for Children. xiii, 284 (Springer Science + Business Media, New York, NY, US, 2008). [Google Scholar]
- 29.Graham JM Jr., Edwards MJ & Edwards MJ Teratogen update: Gestational effects of maternal hyperthermia due to febrile illnesses and resultant patterns of defects in humans. Teratology 58, 209–221 (1998). [DOI] [PubMed] [Google Scholar]
- 30.Fang S-Y, Wang S, Huang N, Yeh H-H & Chen C-Y Prenatal Infection and Autism Spectrum Disorders in Childhood: A Population-Based Case-Control Study in Taiwan. Paediatr. Perinat. Epidemiol 29, 307–316 (2015). [DOI] [PubMed] [Google Scholar]
- 31.Patterson PH Maternal infection and immune involvement in autism. Trends Mol. Med 17, 389–394 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Abdallah MW et al. Amniotic fluid inflammatory cytokines: Potential markers of immunologic dysfunction in autism spectrum disorders. World J. Biol. Psychiatry 14, 528–538 (2013). [DOI] [PubMed] [Google Scholar]
- 33.Goines PE et al. Increased midgestational IFN-γ, IL-4 and IL-5 in women bearing a child with autism: A case-control study. Mol. Autism 2, 13 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gustavson K et al. Maternal fever during pregnancy and offspring attention deficit hyperactivity disorder. Sci. Rep 9, 9519 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Werenberg Dreier J et al. Fever and infections in pregnancy and risk of attention deficit/hyperactivity disorder in the offspring. J. Child Psychol. Psychiatry 57, 540–548 (2016). [DOI] [PubMed] [Google Scholar]
- 36.Dreier JW, Berg-Beckhoff G, Andersen PK & Andersen A-MN Prenatal Exposure to Fever and Infections and Academic Performance: A Multilevel Analysis. Am. J. Epidemiol 186, 29–37 (2017). [DOI] [PubMed] [Google Scholar]
- 37.Collier SA, Rasmussen SA, Feldkamp ML & Honein MA Prevalence of self-reported infection during pregnancy among control mothers in the National Birth Defects Prevention Study. Birt. Defects Res. A. Clin. Mol. Teratol 66, 193–201 (2009). [DOI] [PubMed] [Google Scholar]
- 38.Cordeiro CN, Tsimis M & Burd I Infections and Brain Development. Obstet. Gynecol. Surv 70, (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Estes ML & McAllister AK Maternal TH17 cells take a toll on baby’s brain. Science 351, 919–920 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Deverman BE & Patterson PH Cytokines and CNS Development. Neuron 64, 61–78 (2009). [DOI] [PubMed] [Google Scholar]
- 41.Meyer U et al. The Time of Prenatal Immune Challenge Determines the Specificity of Inflammation-Mediated Brain and Behavioral Pathology. J. Neurosci 26, 4752–4762 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Meyer U, Yee BK & Feldon J The Neurodevelopmental Impact of Prenatal Infections at Different Times of Pregnancy: The Earlier the Worse? The Neuroscientist 13, 241–256 (2007). [DOI] [PubMed] [Google Scholar]
- 43.Jones KL et al. Autism with intellectual disability is associated with increased levels of maternal cytokines and chemokines during gestation. Mol. Psychiatry 22, 273–279 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dozmorov MG et al. Associations between maternal cytokine levels during gestation and measures of child cognitive abilities and executive functioning. Brain. Behav. Immun 70, 390–397 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gabay C & Kushner I Acute-Phase Proteins and Other Systemic Responses to Inflammation. N. Engl. J. Med 340, 448–454 (1999). [DOI] [PubMed] [Google Scholar]
- 46.Brown AS et al. Elevated maternal C-reactive protein and autism in a national birth cohort. Mol. Psychiatry 19, 259–264 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wong SF, Chow KM & de Swiet M Severe Acute Respiratory Syndrome and pregnancy. BJOG Int. J. Obstet. Gynaecol 110, 641–642 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wong SF et al. Pregnancy and perinatal outcomes of women with severe acute respiratory syndrome. Am. J. Obstet. Gynecol 191, 292–297 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lam CM et al. A case-controlled study comparing clinical course and outcomes of pregnant and non-pregnant women with severe acute respiratory syndrome. BJOG Int. J. Obstet. Gynaecol 111, 771–774 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ng PC, Leung CW, Chiu WK, Wong SF & Hon EKL SARS in Newborns and Children. Biol. Neonate 85, 293–298 (2004). [DOI] [PubMed] [Google Scholar]
- 51.Li AM & Ng PC Severe acute respiratory syndrome (SARS) in neonates and children. Arch. Dis. Child. - Fetal Neonatal Ed 90, F461 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Di Mascio D et al. Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis. Am. J. Obstet. Gynecol. MFM 2, (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Alfaraj SH, Al-Tawfiq JA & Memish ZA Middle East Respiratory Syndrome Coronavirus (MERS-CoV) infection during pregnancy: Report of two cases & review of the literature. J. Microbiol. Immunol. Infect 52, 501–503 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Silasi M et al. Viral Infections During Pregnancy. Am. J. Reprod. Immunol 73, 199–213 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kwon J-Y, Romero R & Mor G New Insights into the Relationship between Viral Infection and Pregnancy Complications. Am. J. Reprod. Immunol 71, 387–390 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kourtis AP, Read JS & Jamieson DJ Pregnancy and Infection. N. Engl. J. Med 370, 2211–2218 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Neuzil KM, Reed GW, Mitchel EF, Simonsen L & Griffin MR Impact of Influenza on Acute Cardiopulmonary Hospitalizations in Pregnant Women. Am. J. Epidemiol 148, 1094–1102 (1998). [DOI] [PubMed] [Google Scholar]
- 58.Jamieson DJ et al. H1N1 2009 influenza virus infection during pregnancy in the USA. The Lancet 374, 451–458 (2009). [DOI] [PubMed] [Google Scholar]
- 59.Harris JW Influenza Occurring in pregnant women: a statistical study of thirteen hundred and fifty cases. J. Am. Med. Assoc 72, 978–980 (1919). [Google Scholar]
- 60.Nuzum JW, Pilot I, Stangl FH, Bonar BE Pandemic influenza and pneumonia in a large civil hospital. J. Am. Med. Assoc 71, 1562–1565 (1918). [PubMed] [Google Scholar]
- 61.Freeman DW & Barno A Deaths from Asian influenza associated with pregnancy. Am. J. Obstet. Gynecol 78, 1172–1175 (1959). [DOI] [PubMed] [Google Scholar]
- 62.Louie JK, Acosta M, Jamieson DJ, Honein MA & Group, C. P. (H1N1) W. Severe 2009 H1N1 Influenza in Pregnant and Postpartum Women in California. N. Engl. J. Med 362, 27–35 (2010). [DOI] [PubMed] [Google Scholar]
- 63.Mosby LG, Rasmussen SA & Jamieson DJ 2009 pandemic influenza A (H1N1) in pregnancy: a systematic review of the literature. Am. J. Obstet. Gynecol 205, 10–18 (2011). [DOI] [PubMed] [Google Scholar]
- 64.Pierce M, Kurinczuk JJ, Spark P, Brocklehurst P & Knight M Perinatal outcomes after maternal 2009/H1N1 infection: national cohort study. BMJ 342, d3214 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Borren I et al. Early prenatal exposure to pandemic influenza A (H1N1) infection and child psychomotor development at 6 months – A population-based cohort study. Early Hum. Dev 122, 1–7 (2018). [DOI] [PubMed] [Google Scholar]
- 66.Rasmussen SA, Jamieson DJ & Uyeki TM Effects of influenza on pregnant women and infants. Am. J. Obstet. Gynecol 207, S3–S8 (2012). [DOI] [PubMed] [Google Scholar]
- 67.Deykin EY, Macmahon B Viral exposure and autism. Am. J. Epidemiol 109, 628–638 (1979). [DOI] [PubMed] [Google Scholar]
- 68.Zerbo O et al. Association Between Influenza Infection and Vaccination During Pregnancy and Risk of Autism Spectrum Disorder. JAMA Pediatr. 171, e163609–e163609 (2017). [DOI] [PubMed] [Google Scholar]
- 69.Dassa D, Takei N, Sham PC & Murray RM No association between prenatal exposure to influenza and autism. Acta Psychiatr. Scand 92, 145–149 (1995). [DOI] [PubMed] [Google Scholar]
- 70.Mueller FS et al. Influence of poly(I:C) variability on thermoregulation, immune responses and pregnancy outcomes in mouse models of maternal immune activation. Brain. Behav. Immun 80, 406–418 (2019). [DOI] [PubMed] [Google Scholar]
- 71.Malkova NV, Yu CZ, Hsiao EY, Moore MJ & Patterson PH Maternal immune activation yields offspring displaying mouse versions of the three core symptoms of autism. Brain. Behav. Immun 26, 607–616 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Shi L, Fatemi SH, Sidwell RW & Patterson PH Maternal Influenza Infection Causes Marked Behavioral and Pharmacological Changes in the Offspring. J. Neurosci 23, 297 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Dunaevsky A & Bergdolt L Brain changes in a maternal Immune activation model of neurodevelopmental brain disorders. Prog Neurobiol 175, 1–19 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Smith SEP, Li J, Garbett K, Mirnics K & Patterson PH Maternal Immune Activation Alters Fetal Brain Development through Interleukin-6. J. Neurosci 27, 10695 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Rasmussen JM et al. Maternal Interleukin-6 concentration during pregnancy is associated with variation in frontolimbic white matter and cognitive development in early life. NeuroImage 185, 825–835 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ozaki K et al. Maternal immune activation induces sustained changes in fetal microglia motility. Sci. Rep 10, 21378 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Choi GB et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 351, 933–939 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Machado CJ, Whitaker AM, Smith SEP, Patterson PH & Bauman MD Maternal Immune Activation in Nonhuman Primates Alters Social Attention in Juvenile Offspring. Biol. Psychiatry 77, 823–832 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Fatemi SH et al. Prenatal Viral Infection Leads to Pyramidal Cell Atrophy and Macrocephaly in Adulthood: Implications for Genesis of Autism and Schizophrenia. Cell. Mol. Neurobiol 22, 25–33 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Short SJ et al. Maternal Influenza Infection During Pregnancy Impacts Postnatal Brain Development in the Rhesus Monkey. Biol. Psychiatry 67, 965–973 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Corradini I et al. Maternal Immune Activation Delays Excitatory-to-Inhibitory Gamma-Aminobutyric Acid Switch in Offspring. Biol. Psychiatry 83, 680–691 (2018). [DOI] [PubMed] [Google Scholar]
- 82.Basil P et al. Prenatal maternal immune activation causes epigenetic differences in adolescent mouse brain. Transl. Psychiatry 4, e434–e434 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Richetto J et al. Genome-wide DNA Methylation Changes in a Mouse Model of Infection-Mediated Neurodevelopmental Disorders. Genet. Epigenetic Risks Autism Spectr. Disord 81, 265–276 (2017). [DOI] [PubMed] [Google Scholar]
- 84.Shrestha S & Offer SM Epigenetic regulations of GABAergic neurotransmission: relevance for neurological disorders and epigenetic therapy. Med. Epigenetics 4, 1–19 (2016). [Google Scholar]
- 85.Hill RA et al. Maternal SARS-CoV-2 exposure alters infant DNA methylation. Brain Behav. Immun. - Health 27, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Fujita Y, Ishima T & Hashimoto K Supplementation with D-serine prevents the onset of cognitive deficits in adult offspring after maternal immune activation. Sci. Rep 6, 37261 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zhang J, Jing Y, Zhang H, Bilkey DK & Liu P Maternal immune activation altered microglial immunoreactivity in the brain of postnatal day 2 rat offspring. Synapse 73, e22072 (2019). [DOI] [PubMed] [Google Scholar]
- 88.Scher MS “The First Thousand Days” Define a Fetal/Neonatal Neurology Program. Front. Pediatr 9, 683138 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Huntley MA et al. Genome-Wide Analysis of Differential Gene Expression and Splicing in Excitatory Neurons and Interneuron Subtypes. J. Neurosci 40, 958–973 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Katsarou A, Moshé SL & Galanopoulou AS Interneuronopathies and their role in early life epilepsies and neurodevelopmental disorders. Epilepsia Open 2, 284–306 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Molnár Z et al. New insights into the development of the human cerebral cortex. J. Anat 235, 432–451 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Para R et al. The Distinct Immune Nature of the Fetal Inflammatory Response Syndrome Type I and Type II. ImmunoHorizons 5, 735–751 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jung E et al. The fetal inflammatory response syndrome: the origins of a concept, pathophysiology, diagnosis, and obstetrical implications. Semin. Fetal. Neonatal Med 25, 101146 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kim CJ et al. Acute chorioamnionitis and funisitis: definition, pathologic features, and clinical significance. Am. J. Obstet. Gynecol 213, S29–S52 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Lee J et al. Characterization of the Fetal Blood Transcriptome and Proteome in Maternal Anti‐Fetal Rejection: Evidence of a Distinct and Novel Type of Human Fetal Systemic Inflammatory Response. Am. J. Reprod. Immunol 70, 265–284 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.COVID Data Tracker. Centers for Disease Control and Prevention https://covid.cdc.gov/covid-data-tracker (2020).
- 97.Rasmussen SA et al. Pandemic Influenza and Pregnant Women: Summary of a Meeting of Experts. Am. J. Public Health 99, S248–S254 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Narang K et al. SARS-CoV-2 Infection and COVID-19 During Pregnancy: A Multidisciplinary Review. Mayo Clin. Proc 95, 1750–1765 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Villar J et al. Maternal and Neonatal Morbidity and Mortality Among Pregnant Women With and Without COVID-19 Infection: The INTERCOVID Multinational Cohort Study. JAMA Pediatr. 175, 817–826 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kumar R et al. SARS-CoV-2 infection during pregnancy and pregnancy-related conditions: Concerns, challenges, management and mitigation strategies–a narrative review. J. Infect. Public Health 14, 863–875 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zambrano LD et al. Morbidity and Mortality Weekly Report Update: Characteristics of Symptomatic Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status. Cent. Dis. Control MMWR Morb. Mortal. Wkly. Rep 1641–1647 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Allotey J et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. BMJ 370, m3320 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ellington S et al. Characteristics of Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status — United States, January 22–June 7, 2020. MMWR Morb. Mortal. Wkly. Rep 69, 769–775 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Piekos SN et al. The effect of maternal SARS-CoV-2 infection timing on birth outcomes: a retrospective multicentre cohort study. Lancet Digit. Health 4, e95–e104 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Khalil A et al. SARS-CoV-2 infection in pregnancy: A systematic review and meta-analysis of clinical features and pregnancy outcomes. EClinicalMedicine 25, 100446 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Schwartz DA, Mulkey SB & Roberts DJ SARS-CoV-2 placentitis, stillbirth, and maternal COVID-19 vaccination: clinical–pathologic correlations. Am. J. Obstet. Gynecol 228, 261–269 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Strid P et al. Coronavirus Disease 2019 (COVID-19) Severity Among Women of Reproductive Age With Symptomatic Laboratory-Confirmed Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection by Pregnancy Status—United States, 1 January 2020–25 December 2021. Clin. Infect. Dis 75, S317–S325 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Iuliano AD et al. Trends in Disease Severity and Health Care Utilization During the Early Omicron Variant Period Compared with Previous SARS-CoV-2 High Transmission Periods - United States, December 2020-January 2022. Cent. Dis. Control MMWR Morb. Mortal. Wkly. Rep 71, 146–152 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Stock SJ et al. SARS-CoV-2 infection and COVID-19 vaccination rates in pregnant women in Scotland. Nat. Med 28, 504–512 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Adhikari EH et al. COVID-19 Cases and Disease Severity in Pregnancy and Neonatal Positivity Associated With Delta (B.1.617.2) and Omicron (B.1.1.529) Variant Predominance. JAMA 327, 1500–1502 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Reyes-Lagos JJ et al. A Translational Perspective of Maternal Immune Activation by SARS-CoV-2 on the Potential Prenatal Origin of Neurodevelopmental Disorders: The Role of the Cholinergic Anti-inflammatory Pathway. Front. Psychol 12, (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Dubey H, Sharma RK, Krishnan S & Knickmeyer R SARS-CoV-2 (COVID-19) as a possible risk factor for neurodevelopmental disorders. Front. Neurosci 16, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Granja MG et al. SARS-CoV-2 Infection in Pregnant Women: Neuroimmune-Endocrine Changes at the Maternal-Fetal Interface. Neuroimmunomodulation 28, 1–21 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Engert V et al. Severe Brain Damage in a Moderate Preterm Infant as Complication of Post-COVID-19 Response during Pregnancy. Neonatology 118, 505–508 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mulkey SB et al. Neurodevelopment in infants with antenatal or early neonatal exposure to SARS-CoV-2. Early Hum. Dev 175, 105694 (2022). [DOI] [PubMed] [Google Scholar]
- 116.Edlow AG, Castro VM, Shook LL, Kaimal AJ & Perlis RH Neurodevelopmental Outcomes at 1 Year in Infants of Mothers Who Tested Positive for SARS-CoV-2 During Pregnancy. JAMA Netw. Open 5, e2215787–e2215787 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Hessami K et al. COVID-19 Pandemic and Infant Neurodevelopmental Impairment: A Systematic Review and Meta-analysis. JAMA Netw. Open 5, e2238941–e2238941 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Shuffrey LC et al. Association of Birth During the COVID-19 Pandemic With Neurodevelopmental Status at 6 Months in Infants With and Without In Utero Exposure to Maternal SARS-CoV-2 Infection. JAMA Pediatr. 176, e215563–e215563 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Ayed M et al. Neurodevelopmental outcomes of infants born to mothers with SARS-CoV-2 infections during pregnancy: a national prospective study in Kuwait. BMC Pediatr. 22, 319 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Lavallée A & Dumitriu D Low Risk of Neurodevelopmental Impairment in the COVID-19 Generation Should Not Make Researchers Complacent. JAMA Netw. Open 5, e2238958–e2238958 (2022). [DOI] [PubMed] [Google Scholar]
- 121.Firestein MR, Dumitriu D, Marsh R & Monk C Maternal Mental Health and Infant Development During the COVID-19 Pandemic. JAMA Psychiatry 79, 1040–1045 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mulkey SB Lessons learned in caring for newborns from one viral outbreak to the next. Pediatr. Res (2023) doi: 10.1038/s41390-023-02937-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Celik IH, Ozkaya Parlakay A & Canpolat FE Management of neonates with maternal prenatal coronavirus infection and influencing factors. Pediatr. Res (2023) doi: 10.1038/s41390-023-02855-0. [DOI] [PubMed] [Google Scholar]
- 124.Lu Y-C et al. Maternal psychological distress during the COVID-19 pandemic and structural changes of the human fetal brain. Commun. Med 2, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Halai U-A et al. Maternal Zika Virus Disease Severity, Virus Load, Prior Dengue Antibodies, and Their Relationship to Birth Outcomes. Clin. Infect. Dis 65, 877–883 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Honein MA et al. Birth Defects Among Fetuses and Infants of US Women With Evidence of Possible Zika Virus Infection During Pregnancy. JAMA 317, 59–68 (2017). [DOI] [PubMed] [Google Scholar]
- 127.Muller WJ & Mulkey SB Lessons about early neurodevelopment in children exposed to ZIKV in utero. Nat. Med 25, 1192–1193 (2019). [DOI] [PubMed] [Google Scholar]
- 128.Moore CA et al. Characterizing the Pattern of Anomalies in Congenital Zika Syndrome for Pediatric Clinicians. JAMA Pediatr. 171, 288–295 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.França GVA et al. Congenital Zika virus syndrome in Brazil: a case series of the first 1501 livebirths with complete investigation. The Lancet 388, 891–897 (2016). [DOI] [PubMed] [Google Scholar]
- 130.Adebanjo T et al. Update: Interim Guidance for the Diagnosis, Evaluation, and Management of Infants with Possible Congenital Zika Virus Syndrome - United States, October 2017. Cent. Dis. Control MMWR Morb. Mortal. Wkly. Rep 66, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Mulkey SB et al. Neurodevelopmental Abnormalities in Children With In Utero Zika Virus Exposure Without Congenital Zika Syndrome. JAMA Pediatr. 174, 269–276 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Marbán-Castro E et al. Zika virus infection in pregnant travellers and impact on childhood neurodevelopment in the first two years of life: A prospective observational study. Travel Med. Infect. Dis 40, 101985 (2021). [DOI] [PubMed] [Google Scholar]
- 133.Grant R et al. In utero Zika virus exposure and neurodevelopment at 24 months in toddlers normocephalic at birth: a cohort study. BMC Med. 19, 12 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Rice ME et al. Vital Signs: Zika-Associated Birth Defects and Neurodevelopmental Abnormalities Possibly Associated with Congenital Zika Virus Infection — U.S. Territories and Freely Associated States, 2018. Cent. Dis. Control MMWR Morb. Mortal. Wkly. Rep (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Hoen B et al. Pregnancy Outcomes after ZIKV Infection in French Territories in the Americas. N. Engl. J. Med 378, 985–994 (2018). [DOI] [PubMed] [Google Scholar]
- 136.Nielsen-Saines K et al. Delayed childhood neurodevelopment and neurosensory alterations in the second year of life in a prospective cohort of ZIKV-exposed children. Nat. Med 25, 1213–1217 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Wild CP Complementing the Genome with an “Exposome”: The Outstanding Challenge of Environmental Exposure Measurement in Molecular Epidemiology. Cancer Epidemiol. Biomarkers Prev 14, 1847–1850 (2005). [DOI] [PubMed] [Google Scholar]
- 138.Tamiz AP, Koroshetz WJ, Dhruv NT & Jett DA A focus on the neural exposome. Neuron 110, 1286–1289 (2022). [DOI] [PubMed] [Google Scholar]
- 139.Scher MS The neural exposome influences the preterm fetal-to-neonatal connectome. Pediatr. Res 95, 9–11 (2023). [DOI] [PubMed] [Google Scholar]
- 140.Scher MS Chapter 1 - Fetal neurology: Principles and practice with a life-course perspective. in Handbook of Clinical Neurology (eds. de Vries LS & Glass HC) vol. 162 1–29 (Elsevier, 2019). [DOI] [PubMed] [Google Scholar]


