Abstract
Background
High myopia is one of the major causes of visual impairment and has an ever-increasing prevalence, especially in East Asia. It is characterized by excessive axial elongation, leading to various blinding complications that extend beyond mere refractive errors and persist immovably after refractive surgery, presenting substantial public health challenge.
Main text
High myopia-related complications include lens pathologies, atrophic and tractional maculopathy, choroidal neovascularization, peripheral retinal degenerations and retinal detachment, and glaucoma and heightened susceptibility to intraocular pressure (IOP) elevation. Pathological lens changes characteristic of high myopia include early cataractogenesis, overgrowth of lens, weakened zonules, and postoperative capsular contraction syndrome, possibly driven by inflammatory pathogenesis, etc. Dome-shaped macula and cilioretinal arteries are two newly identified protective factors for central vision of highly myopic patients. These patients also face risks of open-angle glaucoma and IOP spike following intraocular surgery. Morphologic alternations of optic nerve in high myopia can complicate early glaucoma detection, necessitating comprehensive examinations and close follow-up. Anatomically, thinner trabecular meshwork increases this risk; conversely lamina cribrosa defects may offer a fluid outlet, potentially mitigating the pressure. Notably, anxiety has emerged as the first recognized extra-ocular complication in high myopia, with an underlying inflammatory pathogenesis that connects visual stimulus, blood and brain.
Conclusions
High myopia induces multiple ocular and potential mental health complications, underscoring the need to develop more effective strategies to improve both physical and emotional well-being of these patients, among which anti-inflammation might possibly represent a promising new target.
Keywords: High myopia, Cataract, Myopic maculopathy, Open-angle glaucoma, Anxiety
1. Introduction
High myopia is defined by an excessive elongation of the eyeball, resulting in a refractive error exceeding −6.00 diopters or an axial length surpassing 26mm. This condition poses a significant public health challenge, particularly in East Asia where it is notably prevalent.1,2 In 2020, around 399 million people, or 5.1% of the global population, were highly myopic. Forecasts suggest this figure will rise to approximately 938 million, representing 9.8% of the world population, by the year 2050. In China, research conducted across different areas indicates a prevalence of high myopia among the elderly, ranging from 2.3% to 4.7%3,4; in stark contrast, data from a decade earlier revealed that almost 20% of the young adult demographic were suffering from high myopia.5 This disparity underscores a concerning trend, implying a potentially substantial increase in the number of individuals facing high myopia in the coming generations. The COVID-19 pandemic has exacerbated such situation; with home quarantine and school closure, the prevalence of myopia in children has reportedly doubled.6,7 Therefore, current data may significantly underestimate the prospective trajectory of high myopia.
Among highly myopic eyes, pathologic myopia can occur, which may lead to irreversible visual function decline. The definition of pathologic myopia is centered on the pathological changes in the posterior pole of the eyeball caused by excessive elongation of the axial length. According to the International Myopia Institute, pathologic myopia is defined as a subset of myopia characterized by maculopathy equal to or more serious than diffuse chorioretinal atrophy and/or the presence of a posterior staphyloma.8 Related complications may extend beyond mere refractive errors, encompassing a spectrum of ocular pathologies that can severely compromise corrected vision and overall ocular health. Cataract, myopic maculopathy, peripheral retinal degeneration and retinal detachment (RD), and open-angle glaucoma (OAG) have long been acknowledged as four major complications of high myopia, of which cataracts represent the most common one. While they can be addressed through surgical intervention, management of cataracts in patients with high myopia have considerable challenges, as they frequently experience a heightened incidence of perioperative complications, adding complexity to their treatment.9, 10, 11, 12, 13 In China, myopic macular degeneration accounted for 59.6% of bilateral visual impairment among middle-aged adults.14 Over half of eyes with high myopia demonstrate peripheral retinal lesions, and among these, 4.55% present with retinal tears, which increase the risk of RD by 12–39 times compared to non-myopic eyes.15, 16, 17, 18, 19, 20, 21 A recent Mendelian Randomization analysis has substantiated a strong bidirectional genetic causal link between myopia and OAG.22 Additionally, it is also worth noting that a latest study by our group has explored the potential link between high myopia and psychiatric disorder, implicating an inflammatory pathogenesis connecting eye, blood, and brain.23
The high incidence of high myopia underscores the urgency for effective prevention and control strategies in public health and ophthalmology. Despite the need, the progress in addressing high myopia lags behind its increasing prevalence. And attention must specially focus on managing the associated complications of high myopia. These complications will all persist even after refractive surgery, representing chronic issues emanating from pathological alternations within the aging myopic eyeball. This review aims to provide an updated synopsis of high myopia-related complications from clinical manifestations to underlying mechanisms.
2. Ocular complications of high myopia
2.1. Lens pathology
2.1.1. Early-onset cataract in high myopia
Cataractogenesis is the commonest complication of high myopia. The association between high myopia and cataract has been consistently corroborated by a multitude of epidemiological studies. Jeon et al.'s study showed that individuals with high myopia undergo cataract surgery nearly a decade earlier than their non-highly myopic counterparts.24 Furthermore, Yu et al. summarized the clinical characteristics of presenile cataract patients undergoing surgical intervention between the ages of 30 and 54, and identified high myopia as a predominant etiological factor.25 The Blue Mountains Eye Study reported a heightened incidence of specific types of cataracts in highly myopic individuals: high myopia was associated with increased incidence of nuclear cataract (odds ratio 3.01, 95% confidence intervals 1.35–6.71) and increased incidence of posterior subcapsular cataract (odds ratio 7.80, 95% confidence interval 3.51–17.35).26 Pan et al. also found that the severity of myopia was associated with an increasing trend of posterior subcapuslar cataract among Malay adults.27 Further supportive findings from Haarmann et al. also quantified the risk, indicating that high myopia conferred an increased risk of nuclear cataract with an odds ratio of 2.86 and increased risk of posterior subcapsular cataract with an odds ratio of 4.55.17 Our group further addressed the relationship between high myopia and dark nucleus cataract and reported that the odds ratio was 5.16 (95% confidence interval: 3.98–6.69).28 As for cortical cataract, no epidemiological studies reported an association.
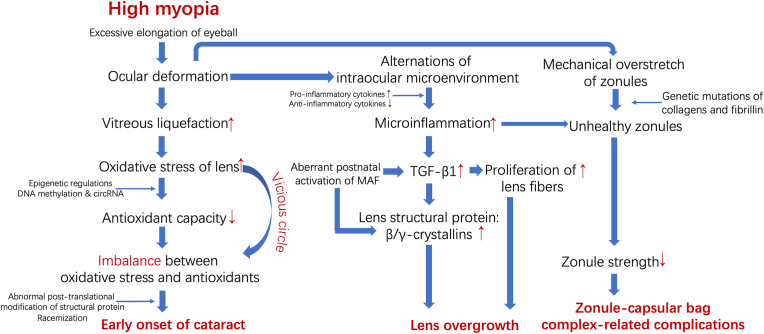
A widely accepted hypothesis postulates that the precocious onset of cataracts in high myopia is largely attributed to more pronounced vitreous liquefaction, which exposes the lens to elevated levels of oxygen from the retina, inciting imbalance between oxidative stress and antioxidants to lens and ultimately lens opacities.28,29 Contemporary research into the pathogenesis of highly myopic cataract has yielded significant insights. Given the progressive nature of high myopia in the postnatal period, our research group has focused on the role of epigenetics in cataractogenesis of high myopia and was pioneer in reporting a reduction in αA-crystallin expression in dark nuclear cataract, implicating DNA hypermethylation as the underlying mechanism.28 Subsequent investigations have identified similar epigenetic modulation of antioxidant genes (GSTP1 and TXNRD2) in lenses of high myopia, leading to aggravated oxidative stress.29 Additionally, our group has also unveiled the involvement of circular RNAs in this pathological process.30 Beyond epigenetics, Yang et al. have observed the activation of unfolded protein response in the lenses of patients with high myopia.31 Moreover, our group detected elevated level of d-Asp 58 in αA-crystallin from the lens of highly myopic cataract indicating the participation of racemization in cataractogenesis of these patients (Fig. 1).32
Fig. 1.
Schematic diagram illustrating the pathological mechanisms associated with lens pathology in individuals with high myopia. The excessive elongation of eyeballs results in alterations in the highly myopic intraocular microenvironment, marked by increased oxidative stress and microinflammation. These factors contribute to the early onset of cataract, lens overgrowth, and weakened zonular strength, mediated through processes such as epigenetic regulations, post-translational modifications, and aberrant activation of growth factor pathways.
2.1.2. Lens overgrowth in high myopia
In addition to the development of cataracts, aberrant lens growth in high myopia has also been noted.
Our group conducted a series of clinical observations and found increased risks of intraocular lens (IOL) instability post-implantation in highly myopic eyes, which can substantially impair both visual acuity and quality of patients.10,33, 34, 35 Using high-resolution magnetic resonance imaging, we firstly measured and compared different dimensions of lens in 144 emmetropic and 105 highly myopic eyes and discovered significant larger equatorial diameters and volumes of highly myopic lens,36 which helps explain the more severe IOL decentration and rotation resulting from the incompatibility between the fixed size of IOLs and the expanded lens capsular bags of highly myopic eyes.
Further mechanistic explorations uncovered the over-activation of transcriptional factor MAF after birth in the lens of highly myopic eyes, which resulted in excessive productions of lens structural protein β/γ-crystallins.36 This change is accompanied by heightened fiber compaction in the germinative zone of lens, potentially contributing to the nuclear cataract phenotype commonly observed in high myopia. Furthermore, through single-cell RNA sequencing of lens from highly myopic mice, our group depicted its transcriptome atlas and discovered the inhibition of NOTCH2 signaling in promoting lens fiber cell differentiation, thereby exacerbating lens overgowth in these individuals37(Fig. 1).
Such overgrowth, on one hand, may precipitate nuclear sclerosis in lenses of highly myopic eyes; on the other hand, necessitates meticulous attention from cataract surgeon during cataract procedures. Our group evaluated the stability of toric IOLs and multifocal IOLs with different haptic designs in highly myopic eyes and found that plate-haptic IOLs showed better rotational and centered stability and better visual quality compared with C-loop haptic IOLs.38,39 Hence, proper IOL selection is imperative to optimize surgical outcomes of high myopes.
2.1.3. Zonule-capsular bag complex-related complications in high myopia after cataract surgery
Although direct observations of lens zonules in highly myopic eyes remain unreported to date, it is almost a consensus of clinicians that lens zonules in high myopia is weaker than emmetropia, supported by several surgical complications in these eyes, such as sudden increased anterior chamber depth and floppy capsular bag.40,41 Besides, Ascaso et al. reported high myopia to be predisposing factor to zonular dehiscence and late IOL-capsular bag complex dislocation.42 Fan et al. reported an odds ratio of late IOL dislocation up to 11.268 for highly myopic eyes.43
Besides mechanical stretch from excessive axial elongation, mechanisms behind weakened zonules in high myopia are largely unknown. Some pedigree and sporadic analyses of patients with myopia, lens subluxation and systemic disorders suggested involvement of genetic mutations of collagens and fibrillin.44,45 Besides, the unique microenvironment of highly myopic eyes may also contribute to the pathologies of zonules: a proinflammatory status in the aqueous humor of high myopia featuring elevated proinflammatory MCP-1 level and decreased anti-inflammatory IL-1ra was identified by our group.46 The exacerbation of zonular laxity in the pro-inflammatory highly myopic intraocular environment somewhat resembles that observed in retinitis pigmentosa, yet the specific pathological correlates await further histopathological investigation (Fig. 1).
Inadequate capsular support due to zonular weakness might also contribute to increased capsular contraction syndrome after cataract surgery in highly myopic eyes. Capsular contraction syndrome is an exaggerated reduction in the anterior capsular opening and capsular bag diameter after lens removal. Our group reported an incidence rate of 2.1% in highly myopic patients, 14 times of the incidence among emmetropic eyes12 and explored the molecular basis of this condition and found that elevated TGF-β2 level in aqueous humor of highly myopic cataract patients may be a reasonable explanation.47,48
2.2. Myopic maculopathy
Myopic maculopathy is the most important complication of high myopia, which encompasses a spectrum of degenerative and tractional changes in macula, leading to progressive vision loss8,49 (summarized in Table 1). In 2015, Ohno-Matsui et al. firstly proposed an international photographic classification and grading system (META-PM) for myopic maculopathy based on fundus photographs, in which myopic maculopathy was categorized into "no myopic retinal degenerative lesion (Category 0)", "tessellated fundus" (Category 1), "diffuse chorioretinal atrophy" (Category 2), "patchy chorioretinal atrophy" (Category 3), and "macular atrophy" (Category 4), with additional three "plus" lesions, namely, lacquer cracks, myopic choroidal neovascularization, and Fuchs spot.50 While META-PM provided a comprehensive framework for fundus grading in myopic maculopathy, it did not include descriptions of vitreomacular interface alterations in highly myopic eyes. In response to this gap, Ruiz-Medrano et al. classified a different aspect of myopic maculopathy in 2018, based on three key factors: atrophy (A), traction (T), and neovascularization (N), incorporating characteristics observed by multimodal imaging including fundus photographs and optical coherence tomography.51 Based on ATN system, Ye et al. reported the distribution of visual impairment with myopic maculopathy across different age groups: most participants younger than 50 years had normal vision; the percentage of myopic maculopathy and related visual impairment increased nonlinearly with older age with turning point at 45 years for macular atrophic maculopathy and 50 years for macular tractional maculopathy (MTM) and myopic neovascular maculopathy.52
Table 1.
Summary of different types of myopic macular and their associated factors.
| Myopic maculopathy | Imaging tool | Grading system | Category | Pathophysiological basis | Risk factors | Protective factors |
|---|---|---|---|---|---|---|
| Atrophic | Fundus photography | META-PM/ATN |
|
Axial elongation, choroidal thinning, Bruch's membrane defects, decreased retinal perfusion | Tessellated fundus, age, worse SE, longer AXL |
Cilioretinal arteries (temporal "cake-fork", temporal "ribbon", and "multiple") |
| Tractional | OCT | ATN |
|
Worse SE, longer AXL, V-shaped LMH |
Elevated tissue inside the LMH, dome-shaped macula | |
| CNV | OCT/Angiography | ATN |
|
mCNV in the other eye, severer lacquer crack, thinner subfoveal/inferior choroidal thickness, the nasal posterior staphyloma height from the fovea |
/ |
OCT: optical coherence tomography, MH: macular hole, CNV: choroidal neovascularization, SE: spherical equivalence, AXL: axial length, LMH: lamellar macular hole.
2.2.1. Myopic atrophic maculopathy and protective factor
Myopic atrophic maculopathy, corresponding to myopic macular degeneration (MMD) proposed by META-PM, has a prevalence of 49% in patients with high myopia according to a study involving 2963 patients across seven countries on four continents. Another analysis which included 71,052 participants from 10 countries on four continents, revealed that the overall prevalence of MMD in the general population was 1.7%.53 Foo et al. investigated predictive factors for MMD in a 12-year longitudinal study of Singapore adults and found that tessellated fundus (META-PM category 1) was a major predictor of MMD, among other factors including age, worse spherical equivalent and longer axial length.54
Axial elongation, choroidal thinning, Bruch's membrane changes, and retinal pigment epithelium (RPE) dysfunction, individually or in combination underlie the pathogenesis of MMD. Using quantitative OCT angiography, Zheng et al. demonstrated that in eyes with MMD severity of diffuse chorioretinal atrophy (META-PM category 2) or worse, retinal perfusion decreased significantly with longer axial length.55 Patchy atrophies (META-PM category 3) represent a hallmark of the more advanced stages of MMD. An observational study led by Ohno-Matsui revealed that progressive and continuous choroidal thinning played a key role in MMD progression from no maculopathy to tessellation and to diffuse atrophy, whereas regarding further progression to patchy atrophy, Bruch's membrane defects might be involved.56
Certain anatomical feature in high myopia may also protect from MMD. Cilioretinal arteries is the most common congenital anomaly of the retinal circulation, belonging to the posterior ciliary artery system, which arise from the peripapillary choroid or directly from one of the short posterior ciliary arteries. Before, cilioretinal artery sparing was mostly reported in central retinal artery occlusion or age-related macular degeneration.57,58 In 2020, our group firstly reported a photographic classification for cilioretinal arteries (temporal "cake-fork", temporal "ribbon", "multiple", and "nasal") and found that apart from the "nasal" category, the other three categories could ameliorate the macular blood supply, resulting in lower MMD grades and better visual acuity.59, 60, 61 These findings suggest the vital role of macular blood supply in protecting from MMD of highly myopic eyes.
Research into molecular basis underlying MMD has been sparse, mostly concentrating on the biomarker identification in biospecimens of highly myopic patients. Wong et al. measured the aqueous humor concentrations of several cytokines of highly myopic eyes, and found that eyes with MMD had significantly lower vascular endothelial growth factor (VEGF)-A levels and higher matrix metalloproteinase (MMP)-2 levels than control eyes. Besides, MMP-2 levels correlated positively, while VEGF-A levels correlated negatively with longer axial length. However, both the concentrations of VEGF-A and MMP-2 were not significantly associated with MMD after adjusting for axial length, which suggests that the predominant mechanism underlying the development of atrophic MMD may be axial elongation, driven in part by MMP-2 related mechanisms.62 Another pioneering investigation of tear inflammatory cytokines in eyes with MMD conducted by our group revealed that highly myopic eyes presented significantly higher levels of tear IL-6 and MCP-1 and higher IL-6 and MCP-1 tear levels were independent predictors of higher MMD category.63 Given the elevated inflammation in tear and intraocular fluids of highly myopic eyes and the persistent progressive nature of photoreceptor atrophy and RPE degeneration throughout life, chronic inflammation might explain the pathogenesis of MMD partially. Besides, Yu et al. tried to provide new molecular insights into myopic atrophic maculopathy by investigating the proteom of aqueous humor and found increased protein level of glial fibrillary acidic protein (GFAP) and complement-associated molecules including C7, C8A, C1QC, C1QA, and fibrinogen alpha chain (FGA) in aqueous humor samples of highly myopic patients with atrophic maculopathy.64
2.2.2. Myopic tractional maculopathy and protective factor
MTM is a series of pathological conditions that are attributed to tractional changes in highly myopic eyes characterized by retinoschisis, lamellar or full thickness macular hole (MH), and foveal RD. The prevalence of traction maculopathy in the general population is relatively low. Yet according to a population-based study that included 2913 myopic eyes, MTM was found in 0.9% of myopic eyes and 7.3% of highly myopic eyes.65 Li et al. found a retinoschisis prevalence of 43.5% in an elderly high myopia population, among which 37.9% had paravascular retinoschisis, 27.5% had peripapillary retinoschisis, and 23.0% had macular retinoschisis; worse spherical equivalent and longer axial length were also risk factors for MTM as they were for MMD.66
Shimada et al. investigated the natural course of MTM in 207 highly myopic eyes with a follow-up duration of two years and found that 11.6% eyes experienced progression of MTM and 3.9% even showed a decrease or complete resolution of the macular retinoschisis.67 With regard to lamellar macular hole (LMH), Hsia et al. found that elevated tissue inside the LMH could protect against further structural worsening while V-shaped LMH predicted it.68 Our group observed that epiretinal membrane progressed in over 20% of highly myopic eyes, but visual acuity remained stable, whereas significantly greater best-corrected visual acuity reduction was detected in eyes with myopic retinoschisis or MH progression.69 What's more, if preretinal traction induces inner laminar dehiscence, a full-thickness MH may develop with a macular RD in highly myopic eyes.70 In these cases, the MH is the break that led to RD, which results from over-stretch of the retinal layers and the absence of dome-shaped macula.69
Among pathological changes of posterior contour of highly myopic eyes, dome-shaped macula is a unique one, characterized by an anterior convex protrusion of the macula toward the vitreous cavity. The incidence of dome-shaped macula in highly myopic eyes ranged from 4.4% to 13.8% according to different observational studies.71, 72, 73 Previously, dome-shaped macula was considered to be associated with an increased risk of complications. According to a cross-sectional study, dome-shaped macula showed significantly more Bruch's membrane defects and a greater severity of myopic macular degeneration.74 The risk for shallow sub-foveal serous fluid and extra-foveal retinoschisis was also higher in eyes with dome-shaped macula as reported by Zhao et al.75 However, they also reported better visual acuity in patients with dome-shaped macula and the proportion of foveoschisis was much lower. Our group evaluated the visual outcomes of 891 highly myopic eyes that underwent cataract surgery and reached similar conclusion: despite the greater association of dome-shaped macula with extrafoveal retinoschisis, the associations with sight-threatening complications including foveoschisis and mCNV were lower and the presence of dome-shaped macula indicated better visual acuity after cataract surgery among these highly myopic eyes.73
Regarding the molecular mechanisms underlying MTM, using ultra-performance liquid chromatography‒mass spectrometry, Tang et al. profiled the metabolomics of vitreous humor samples collected from highly myopic patients with MH or myopic retinoschisis (using non-highly myopic MH patients as controls) and identified certain alterations in metabolites. Further bioinformatics analysis revealed that tryptophan metabolism and uric acid were closely correlated with MH or myopic retinoschisis in highly myopic patients.76
2.2.3. Myopic choroidal neovascularization
Myopic choroidal neovascularization (mCNV) is one of the most severe complications of high myopia and can rapidly progress and cause irreversible central vision loss if left untreated. The prevalence of mCNV among highly myopic eyes ranged from 5% to 11%. More importantly, 62% of these patients developed mCNV before 50 years old and a history of mCNV in one eye may increase the risk of mCNV in the fellow eye to 34.8%.77 Ikuno et al. analyzed the ocular risk factors for mCNV and found that severer lacquer crack and thinner subfoveal/inferior choroidal thickness was significantly associated with presence of mCNV and the absolute value of the nasal posterior staphyloma height from the fovea was also significantly greater in eyes with mCNV.78
mCNV had a discouraging natural course: around 90% of patients were left with a best-corrected visual acuity less than 20/200 within five years since its onset, which was associated with the mCNV-related macular atrophy. Considering the age range of patients with mCNV, the diagnosis and proper treatment to prevent permanent visual loss in these patients are of utmost importance both from clinical and socioeconomic perspectives. Intravitreal injection of anti-vascular endothelial growth factor (VEGF) agents is currently the first-line treatment for mCNV. Compared with age-related macular degeneration, mCNV needs fewer injections and most studies showed significant improvement of vision at one year after treatment. However, recurrence may occur. Victor et al. analyzed and identified requiring three or more injections for initial disease control, older age, larger mCNV, juxtafoveal CNV, destruction or absence of RPE and ellipsoid zone as risk factors for mCNV recurrence.79 Coelho et al. revealed that 27.9% of mCNV eyes had macular Bruch's membrane defects and the percentage increased to 36.8% after anti-VEGF injections during a median follow-up of 28.5 months.80 Besides, eyes with macular Bruch's membrane defects at baseline showed a poorer response to anti-VEGF therapy and worse visual outcomes.
Upregulated VEGF in response to hypoxia has long been acknowledged as the crucial stimulor and the theoretical basis for clinical application of anti-VEGF injections in treating CNV, both in mCNV and age-related macular degeneration (AMD). Molecular explorations into CNV are mostly focused on regulations of VEGF, yet studies particularly on mCNV were very limited possibly due to limitations in establishing proper animal models. Liu et al. proposed another miRNA-21-hypoxia inducible factor (HIF)-1α-VEGF axis in highly myopic guinea pig with 532-nm laser induced CNV.81 Considering the different response to anti-VEGF injections between highly myopic patients and AMD patients, further in-depth mechanistic investigations of mCNV are warranted.
2.3. Peripheral retinal lesions and retinal detachment and efficient screening
Individuals with high myopia are susceptible to peripheral retinal degenerations due to excessive elongation of the eyeball which causes overstretching of outer wall of eye, earlier vitreous liquefaction, and degenerative changes in retina and choroid. Peripheral retinal lesions were reported to be present in over half of the highly myopic eyes.21 Vision-threatening peripheral retinal complications include lattice degeneration and retinal tears which might lead to RD.
Chen et al. found that white without pressure and lattice degeneration were present in 57.2% and 16.9% of high myopes between 19 and 25 years old, ranking the most common two peripheral retinal degeneration and the prevalence of peripheral retinal hole/tear/detachment was 4.8%.82 Another hospital-based survey conducted in North India found a 20% prevalence of lattice degeneration in highly myopic participants aged between 10 and 40 years and 4.4% had retinal break/detachment, both mostly seen in the temporal quadrant of the fundus.83 Significant positive association between the prevalence of peripheral retinal lesions with age and axial length was also reported.
Rhegmatogenous retinal detachment (RRD) is the most serious complication resulting from peripheral retinal degenerations that may cause blindness in high myopia. In myopic eyes, vitreous liquefaction occurs at an earlier age, resulting in earlier posterior vitreous detachment than emmetropic eyes and within highly myopic population this trend is more obvious with higher degree of myopia, which is coupled with increased peripheral retinal thinning and pathologic lesions. Mitry et al. found a strong association between myopia and early onset RDs with myopia diagnosed in over 80% of phakic RDs in patients aged under 50.84
Ocular surgery such as cataract surgery might further add to this risk. In the general population, the long-term postoperative incidence of RD was less than 1% whereas in highly myopic eyes, the number increased to 1.74%–9.21%.12,85,86 Morano et al. conducted a retrospective cohort study including 1983712 patients who underwent cataract surgery and found that increased odds of RRD within one year after cataract surgery were observed for operated eyes with lattice degeneration (10.53) and that lattice degeneration conferred the highest odds of 43.86 for retinal tears.87 Hence, detecting retinal tears before cataract surgery is essential. Our group reported that combined application of B-scan ultrasound and eye-steering ultrawide-field fundus imaging presented a quick screen and satisfactory performance, reaching the sensitivity of 95.69% and specificity of 99.88%, both of which were comparable to indirect ophthalmoscopy regardless of the number, type and location of retinal tears.18
2.4. Open-angle glaucoma and related abnormalities
Individuals with high myopia are at a higher risk of developing open-angle glaucoma and optic disc abnormalities such as optic disc tilt and peripapillary atrophy. The elongated posterior pole of the eyeball in high myopia contributes to structural changes in the optic nerve head and increases the susceptibility to OAG. With development of imaging techniques, angle structure changes in high myopes have also been unveiled in predisposing eyes to potential OAG and postoperative IOP elevation.
2.4.1. Open-angle glaucoma
OAG is a chronic progressive optic neuropathy which can deteriorate beyond patients' awareness. The relationship between high myopia and OAG has long been a hot topic with various studies indicating their relationship. According to a meta-analysis conducted by Ha et al., the odds ratios of the association between high myopia and OAG doubled that between low myopia and OAG.88 Wang et al. recently reported the 10-year incidence of OAG in a Chinese adult cohort and found the highest incidence of 13.3% in high myopia group whereas in emmetropia group the incidence dropped to 2.1%.89
Even with an understanding of the relationship between OAG and high myopia, the actual clinical practice reveals that due to the diverse variations in the optic disc, the presence of posterior scleral staphyloma, and peripapillary atrophy, OAG exhibits a highly concealed nature, making it extremely susceptible to being overlooked. The early and rapid identification of normal-tension glaucoma is even more challenging. Bowd et al. developed a multimodal deep learning classifier for OAG diagnosis in highly myopic eyes integrating texture-based en face image, retinal nerve fiber layer (RNFL) thickness map image and confocal scanning laser ophthalmoscope image and reached a diagnostic accuracy up to 0.89.90 Using multiple mendelian randomization models and multivariate genomic structural modeling, Chong et al. discovered a strong bidirectional genetic causal link between myopia and OAG which was mainly mediated by intraocular pressure (IOP), suggesting that even without a clear clinical diagnosis, myopic eyes could benefit from the IOP-lowering treatment for glaucoma.22 In daily clinical setting, visual field examinations are also cruial in helping prompt diagnosis for glaucoma suspect.
Chen et al. analyzed the schlemm's canal and trabecular meshwork morphology using OCT to characterize the underlying pathophysiological mechanisms involved in high myopia-related OAG and for the first time found a larger Schlemm's canal diameter and area, as well as decreased trabecular meshwork thickness in high myopia.91 Bu et al. delved into the pathological mechanism of high myopia-related OAG by establishing a guinea pig model of ocular hypertension with high myopia and in 2024 published the first molecular findings underlying this condition: inhibition of YAP-TGF-β axis could ameliorate high myopia-related ocular hypertension by targeting mechanics-induced trabecular meshwork dysfunction.
2.4.2. IOP spike in high myopia after intraocular surgery
High myopia has been reported to be a risk factor for early IOP spike after multiple intraocular surgeries, including phacoemulsification, vitrectomy with silicone oil injection.92, 93, 94 Our group reported that 28% of highly myopic patients exhibited an early IOP spike at one day after phacoemulsification, compared to 10% of the emmetropic control group. Moreover, the mean IOP values for highly myopic patients during postoperative follow-ups remained higher than those of the emmetropic patients. Notably, a portion of the highly myopic cohort eventually developed OAG.95
Follow-up investigations by our group further uncovered anatomical features underlying these phenomena: a smaller vertical diameter of Schlemm canal and a thinner trabecular meshwork were the two risk factors for early IOP spike after cataract surgery in highly myopic eye.96 Moreover, the presence of lamina cribrosa defects was a protective factor for early IOP spikes, possibly due to a valve-like "back door" effect for IOP regulation in these eyes: lamina cribrosa defects might be responsible for the fluid outflow and thus resulted in decreased IOP elevation after surgery.97
Of note, corticosteroid eyedrops are routinely prescribed as postoperative medication following nearly all intraocular surgeries to mitigate undesirable inflammatory reactions. However, in the case of highly myopic patients, the administration of these medications necessitates heightened caution, accompanied by meticulous monitoring of IOP.98 Achieving an equilibrium between alleviating inflammation and maintaining IOP within acceptable limits is imperative.
3. The brain complication of high myopia
The conception of high myopia as a condition confined solely to the eye has been long-standing, due in part to the ocular immune privilege status. Nevertheless, latest accumulating evidence has begun to an extra-ocular component of the disease.
Dai et al. profiled the serum metabolomics of individuals with high myopia and identified that eight metabolites were found to be closely correlated with high myopia.99 Metabolic pathway analyses of these metabolites of high myopia involved abnormal phospholipid, diacylglycerol, amino acid, and vitamin metabolism, which were closely correlated with oxidative stress and inflammation.
The clinical profile of highly myopic patients often includes characteristics such as anxious behavior, an obsessive attention to every detail mentioned by the doctor, and an excessive concern about potential risks. Our group's recent investigation has, for the first time, provided evidence supporting this phenomenon and has substantiated the concept of a brain complication associated with high myopia, termed "high myopia-related anxiety."
We first observed increased levels of anxiety among highly myopic patients and a simultaneous elevated systemic inflammation levels manifested as increased CCL2 and monocyte levels in these patients. Mechanistic explorations using GFP-positive bone marrow chimeric mice and parabiotic mouse model unveiled that highly myopic visual stimulation led to elevated CCL2 expression in eyes, which in turn exacerbated monocyte/macrophage infiltration into both the eyes and the brain. This process disrupted the integrity of the blood-ocular and blood-brain barriers, ultimately culminating in anxiety among highly myopic individuals.23
These findings highlight the inflammatory component of the highly myopic condition and suggest that inflammation may play a significant role in various other high myopia-related complications. Importantly, these results underscore the necessity for healthcare professionals to address not only the physical but also the emotional needs of highly myopic patients.
4. Conclusions
In the context of an escalating myopia pandemic, though significant efforts have been made to control its incidence, we are still faced with a large population of patients who are already highly myopic and affected by its complications. The traditional view of high myopia as merely an extreme refractive error has evolved, giving way to increasing recognition that it is its complications-including a myriad of oculopathies and emotional disorders-which are expected to escalate in both prevalence and severity with aging, that deserve more attention. This review summarized recent updates in clinical manifestations and underlying mechanism of high myopia-related complications and has highlighted the potential role of inflammation being the underlying pathogenesis, suggesting a paradigm shift in future investigations toward anti-inflammatory strategies to unlock innovative approaches for the prevention and management of high myopia-related complications.
Study approval
Not applicable.
Author contributions
Conception and design of study: XJZ, and YL; Manuscript preparation YD, JQM, WWH, JQ, and XJZ; Manuscript revision YD, XJZ, and YL. All authors reviewed and approved the final version of the manuscript.
Funding
This article was supported by research grants from the National Natural Science Foundation of China (82122017, 82271069, 81870642, 82371040, 81970780, 82201161, 81470613 and 81670835), Science and Technology Innovation Action Plan of Shanghai Science and Technology Commission (23Y11909800 and 21S31904900), Outstanding Youth Medical Talents of Shanghai “Rising Stars of Medical Talents” Youth Development Program, Clinical Research Plan of Shanghai Shenkang Hospital Development Center (SHDC12020111), Shanghai Municipal Key Clinical Specialty Program (shslczdzk01901).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
Thanks to all the peer reviewers and editors for their opinions and suggestions.
References
- 1.Holden B.A., Fricke T.R., Wilson D.A., et al. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology. 2016;123(5):1036–1042. doi: 10.1016/j.ophtha.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 2.Chen S., Liu X., Sha X., Yang X., Yu X. Relationship between axial length and spherical equivalent refraction in Chinese children. Adv Ophthalmol Pract Res. 2021;1(2) doi: 10.1016/j.aopr.2021.100010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xu C., Pan C., Zhao C., et al. Prevalence and risk factors for myopia in older adult east Chinese population. BMC Ophthalmol. 2017;17(1):191. doi: 10.1186/s12886-017-0574-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cheng F., Shan L., Song W., et al. Prevalence and risk factor for refractive error in rural Chinese adults in Kailu, Inner Mongolia. Ophthalmic Physiol Opt. 2021;41(1):13–20. doi: 10.1111/opo.12745. [DOI] [PubMed] [Google Scholar]
- 5.Sun J., Zhou J., Zhao P., et al. High prevalence of myopia and high myopia in 5060 Chinese university students in Shanghai. Invest Ophthalmol Vis Sci. 2012;53(12):7504–7509. doi: 10.1167/iovs.11-8343. [DOI] [PubMed] [Google Scholar]
- 6.Zhang X.J., Zhang Y., Kam K.W., et al. Prevalence of myopia in children before, during, and after COVID-19 restrictions in Hong Kong. JAMA Netw Open. 2023;6(3) doi: 10.1001/jamanetworkopen.2023.4080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Huang S., Shen F., Zhou F., et al. Myopia in elementary school students in Eastern China during the COVID-19 pandemic. Front Public Health. 2023;11 doi: 10.3389/fpubh.2023.1167379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ohno-Matsui K., Wu P.C., Yamashiro K., et al. IMI pathologic myopia. Invest Ophthalmol Vis Sci. 2021;62(5):5. doi: 10.1167/iovs.62.5.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.He W., Cheng K., Zhao L., et al. Long-term outcomes of posterior capsular opacification in highly myopic eyes and its influencing factors. Ophthalmol Ther. 2023;12(4):1881–1891. doi: 10.1007/s40123-023-00711-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhu X., He W., Zhang Y., Chen M., Du Y., Lu Y. Inferior decentration of multifocal intraocular lenses in myopic eyes. Am J Ophthalmol. 2018;188:1–8. doi: 10.1016/j.ajo.2018.01.007. [DOI] [PubMed] [Google Scholar]
- 11.Zhu X., He W., Sun X., Dai J., Lu Y. Fixation stability and refractive error after cataract surgery in highly myopic eyes. Am J Ophthalmol. 2016;169:89–94. doi: 10.1016/j.ajo.2016.06.022. [DOI] [PubMed] [Google Scholar]
- 12.Yao Y., Lu Q., Wei L., Cheng K., Lu Y., Zhu X. Efficacy and complications of cataract surgery in high myopia. J Cataract Refract Surg. 2021;47(11):1473–1480. doi: 10.1097/j.jcrs.0000000000000664. [DOI] [PubMed] [Google Scholar]
- 13.He W., Yao Y., Zhang K., et al. Clinical characteristics and early visual outcomes of highly myopic cataract eyes: the Shanghai high myopia study. Front Med. 2021;8 doi: 10.3389/fmed.2021.671521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tang Y., Wang X., Wang J., et al. Prevalence and causes of visual impairment in a Chinese adult population: the Taizhou eye study. Ophthalmology. 2015;122(7):1480–1488. doi: 10.1016/j.ophtha.2015.03.022. [DOI] [PubMed] [Google Scholar]
- 15.Yang D., Li M., Wei R., Xu Y., Shang J., Zhou X. Optomap ultrawide field imaging for detecting peripheral retinal lesions in 1725 high myopic eyes before implantable collamer lens surgery. Clin Exp Ophthalmol. 2020;48(7):895–902. doi: 10.1111/ceo.13809. [DOI] [PubMed] [Google Scholar]
- 16.Lam D.S., Fan D.S., Chan W.M., et al. Prevalence and characteristics of peripheral retinal degeneration in Chinese adults with high myopia: a cross-sectional prevalence survey. Optom Vis Sci. 2005;82(4):235–238. doi: 10.1097/01.opx.0000159359.49457.b4. [DOI] [PubMed] [Google Scholar]
- 17.Haarman A.E.G., Enthoven C.A., Tideman J.W.L., Tedja M.S., Verhoeven V.J.M., Klaver C.C.W. The complications of myopia: a review and meta-analysis. Invest Ophthalmol Vis Sci. 2020;61(4):49. doi: 10.1167/iovs.61.4.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Meng J., Cheng K., Huang Z., et al. Combined application of B-scan ultrasonography and eye-steering ultrawide field imaging to improve the detection of retinal tears before cataract surgery. Retina. 2024 doi: 10.1097/iae.0000000000004040. [DOI] [PubMed] [Google Scholar]
- 19.Ludwig C.A., Vail D., Al-Moujahed A., et al. Epidemiology of rhegmatogenous retinal detachment in commercially insured myopes in the United States. Sci Rep. 2023;13(1):9430. doi: 10.1038/s41598-023-35520-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang Y.H., Huang C., Tseng Y.L., Zhong J., Li X.M. Refractive error and eye health: an umbrella review of meta-analyses. Front Med. 2021;8 doi: 10.3389/fmed.2021.759767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lai T.Y., Fan D.S., Lai W.W., Lam D.S. Peripheral and posterior pole retinal lesions in association with high myopia: a cross-sectional community-based study in Hong Kong. Eye (Lond) 2008;22(2):209–213. doi: 10.1038/sj.eye.6702573. [DOI] [PubMed] [Google Scholar]
- 22.Chong R.S., Li H., Cheong A.J.Y., et al. Mendelian randomization implicates bidirectional association between myopia and primary open-angle glaucoma or intraocular pressure. Ophthalmology. 2023;130(4):394–403. doi: 10.1016/j.ophtha.2022.11.030. [DOI] [PubMed] [Google Scholar]
- 23.Zhu X., Meng J., Han C., et al. CCL2-mediated inflammatory pathogenesis underlies high myopia-related anxiety. Cell Discov. 2023;9(1):94. doi: 10.1038/s41421-023-00588-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jeon S., Kim H.S. Clinical characteristics and outcomes of cataract surgery in highly myopic Koreans. Korean J Ophthalmol. 2011;25(2):84–89. doi: 10.3341/kjo.2011.25.2.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yu H.J., Kuo M.T., Wu P.C. Clinical characteristics of presenile cataract: change over 10 Years in Southern Taiwan. BioMed Res Int. 2021;2021 doi: 10.1155/2021/9385293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kanthan G.L., Mitchell P., Rochtchina E., Cumming R.G., Wang J.J. Myopia and the long-term incidence of cataract and cataract surgery: the Blue Mountains Eye Study. Clin Exp Ophthalmol. 2014;42(4):347–353. doi: 10.1111/ceo.12206. [DOI] [PubMed] [Google Scholar]
- 27.Pan C.W., Boey P.Y., Cheng C.Y., et al. Myopia, axial length, and age-related cataract: the Singapore Malay eye study. Invest Ophthalmol Vis Sci. 2013;54(7):4498–4502. doi: 10.1167/iovs.13-12271. [DOI] [PubMed] [Google Scholar]
- 28.Zhu X.J., Zhou P., Zhang K.K., Yang J., Luo Y., Lu Y. Epigenetic regulation of αA-crystallin in high myopia-induced dark nuclear cataract. PLoS One. 2013;8(12) doi: 10.1371/journal.pone.0081900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhu X., Li D., Du Y., He W., Lu Y. DNA hypermethylation-mediated downregulation of antioxidant genes contributes to the early onset of cataracts in highly myopic eyes. Redox Biol. 2018;19:179–189. doi: 10.1016/j.redox.2018.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ma S., Zhu X., Li D., et al. The differential expression of circular RNAs and the role of circAFF1 in lens epithelial cells of high-myopic cataract. J Clin Med. 2023;12(3) doi: 10.3390/jcm12030813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang J., Zhou S., Gu J., Guo M., Xia H., Liu Y. UPR activation and the down-regulation of α-crystallin in human high myopia-related cataract lens epithelium. PLoS One. 2015;10(9) doi: 10.1371/journal.pone.0137582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhu X.J., Zhang K.K., He W.W., Du Y., Hooi M., Lu Y. Racemization at the Asp 58 residue in αA-crystallin from the lens of high myopic cataract patients. J Cell Mol Med. 2018;22(2):1118–1126. doi: 10.1111/jcmm.13363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen Y., Meng J., Cheng K., et al. Influence of IOL weight on long-term IOL stability in highly myopic eyes. Front Med. 2022;9 doi: 10.3389/fmed.2022.835475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.He W., Qiu X., Zhang S., et al. Comparison of long-term decentration and tilt in two types of multifocal intraocular lenses with OPD-Scan III aberrometer. Eye (Lond) 2018;32(7):1237–1243. doi: 10.1038/s41433-018-0068-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yao Y., Meng J., He W., et al. Associations between anterior segment parameters and rotational stability of a plate-haptic toric intraocular lens. J Cataract Refract Surg. 2021;47(11):1436–1440. doi: 10.1097/j.jcrs.0000000000000653. [DOI] [PubMed] [Google Scholar]
- 36.Zhu X., Du Y., Li D., et al. Aberrant TGF-β1 signaling activation by MAF underlies pathological lens growth in high myopia. Nat Commun. 2021;12(1):2102. doi: 10.1038/s41467-021-22041-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yao Y., Wei L., Chen Z., et al. Single-cell RNA sequencing: inhibited Notch2 signalling underlying the increased lens fibre cells differentiation in high myopia. Cell Prolif. 2023;56(8) doi: 10.1111/cpr.13412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Meng J., He W., Rong X., Miao A., Lu Y., Zhu X. Decentration and tilt of plate-haptic multifocal intraocular lenses in myopic eyes. Eye Vis (Lond) 2020;7:17. doi: 10.1186/s40662-020-00186-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhu X., Meng J., He W., Rong X., Lu Y. Comparison of the rotational stability between plate-haptic toric and C-loop haptic toric IOLs in myopic eyes. J Cataract Refract Surg. 2020;46(10):1353–1359. doi: 10.1097/j.jcrs.0000000000000259. [DOI] [PubMed] [Google Scholar]
- 40.Wang D., Shi J., Guan W., et al. Fluid supplementation through weakened zonules via side-port incision to maintain intraocular pressure in high myopic eyes. Ophthalmol Ther. 2023;12(6):3323–3336. doi: 10.1007/s40123-023-00814-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pan Y., Liu Z., Zhang H. Research progress of lens zonules. Adv Ophthalmol Pract Res. 2023;3(2):80–85. doi: 10.1016/j.aopr.2023.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ascaso F.J., Huerva V., Grzybowski A. Epidemiology, etiology, and prevention of late IOL-capsular bag complex dislocation: review of the literature. J Ophthalmol. 2015;2015 doi: 10.1155/2015/805706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fan Q., Han X., Luo J., et al. Risk factors of intraocular lens dislocation following routine cataract surgery: a case-control study. Clin Exp Optom. 2021;104(4):510–517. doi: 10.1080/08164622.2021.1878829. [DOI] [PubMed] [Google Scholar]
- 44.Zhou Y., Guo D., Cao Q., Zhang X., Jin G., Zheng D. Genotype variant screening and phenotypic analysis of FBN1 in Chinese patients with isolated ectopia lentis. Mol Med Rep. 2021;23(4) doi: 10.3892/mmr.2021.11914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Meredith S.P., Richards A.J., Bearcroft P., Pouson A.V., Snead M.P. Significant ocular findings are a feature of heritable bone dysplasias resulting from defects in type II collagen. Br J Ophthalmol. 2007;91(9):1148–1151. doi: 10.1136/bjo.2006.112482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhu X., Zhang K., He W., et al. Proinflammatory status in the aqueous humor of high myopic cataract eyes. Exp Eye Res. 2016;142:13–18. doi: 10.1016/j.exer.2015.03.017. [DOI] [PubMed] [Google Scholar]
- 47.Zhang K., Zhu X., Chen M., et al. Elevated transforming growth factor-β2 in the aqueous humor: a possible explanation for high rate of capsular contraction syndrome in high myopia. J Ophthalmol. 2016;2016 doi: 10.1155/2016/5438676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhu X.J., Chen M.J., Zhang K.K., Yang J., Lu Y. Elevated TGF-β2 level in aqueous humor of cataract patients with high myopia: potential risk factor for capsule contraction syndrome. J Cataract Refract Surg. 2016;42(2):232–238. doi: 10.1016/j.jcrs.2015.09.027. [DOI] [PubMed] [Google Scholar]
- 49.Sun C.B., You Y.S., Liu Z., et al. Myopic macular retinoschisis in teenagers: clinical characteristics and spectral domain optical coherence tomography findings. Sci Rep. 2016;6 doi: 10.1038/srep27952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ohno-Matsui K., Kawasaki R., Jonas J.B., et al. International photographic classification and grading system for myopic maculopathy. Am J Ophthalmol. 2015;159(5):877. doi: 10.1016/j.ajo.2015.01.022. 83.e7. [DOI] [PubMed] [Google Scholar]
- 51.Ruiz-Medrano J., Montero J.A., Flores-Moreno I., Arias L., García-Layana A., Ruiz-Moreno J.M. Myopic maculopathy: current status and proposal for a new classification and grading system (ATN) Prog Retin Eye Res. 2019;69:80–115. doi: 10.1016/j.preteyeres.2018.10.005. [DOI] [PubMed] [Google Scholar]
- 52.Ye L., Chen Q., Hu G., et al. Distribution and association of visual impairment with myopic maculopathy across age groups among highly myopic eyes - based on the new classification system (ATN) Acta Ophthalmol. 2022;100(4):e957–e967. doi: 10.1111/aos.15020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Shi H., Guo N., Zhao Z., He X., Li J., Duan J. Global prevalence of myopic macular degeneration in general population and patients with high myopia: a systematic review and meta-analysis. Eur J Ophthalmol. 2023 doi: 10.1177/11206721231185816. [DOI] [PubMed] [Google Scholar]
- 54.Foo L.L., Xu L., Sabanayagam C., et al. Predictors of myopic macular degeneration in a 12-year longitudinal study of Singapore adults with myopia. Br J Ophthalmol. 2023;107(9):1363–1368. doi: 10.1136/bjophthalmol-2021-321046. [DOI] [PubMed] [Google Scholar]
- 55.Zheng F., Chua J., Ke M., et al. Quantitative OCT angiography of the retinal microvasculature and choriocapillaris in highly myopic eyes with myopic macular degeneration. Br J Ophthalmol. 2022;106(5):681–688. doi: 10.1136/bjophthalmol-2020-317632. [DOI] [PubMed] [Google Scholar]
- 56.Yokoi T., Jonas J.B., Shimada N., et al. Peripapillary diffuse chorioretinal atrophy in children as a sign of eventual pathologic myopia in adults. Ophthalmology. 2016;123(8):1783–1787. doi: 10.1016/j.ophtha.2016.04.029. [DOI] [PubMed] [Google Scholar]
- 57.Kim Y.H., Park K.H., Woo S.J. Clinical manifestations and visual prognosis of cilioretinal artery sparing central retinal artery occlusion. Korean J Ophthalmol. 2020;34(1):27–34. doi: 10.3341/kjo.2019.0099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Snyder K., Yazdanyar A., Mahajan A., Yiu G. Association between the cilioretinal artery and choroidal neovascularization in age-related macular degeneration: a secondary analysis from the age-related eye disease study. JAMA Ophthalmol. 2018;136(9):1008–1014. doi: 10.1001/jamaophthalmol.2018.2650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhu X., Meng J., Wei L., Zhang K., He W., Lu Y. Cilioretinal arteries and macular vasculature in highly myopic eyes: an OCT angiography-based study. Ophthalmol Retina. 2020;4(10):965–972. doi: 10.1016/j.oret.2020.05.014. [DOI] [PubMed] [Google Scholar]
- 60.Wei L., Meng J., Cheng K., et al. Contrast sensitivity function: a more sensitive index for assessing protective effects of the cilioretinal artery on macular function in high myopia. Invest Ophthalmol Vis Sci. 2022;63(13):25. doi: 10.1167/iovs.63.13.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Meng J., Wei L., Zhang K., He W., Lu Y., Zhu X. Cilioretinal arteries in highly myopic eyes: a photographic classification system and its association with myopic macular degeneration. Front Med. 2020;7 doi: 10.3389/fmed.2020.595544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wong C.W., Yanagi Y., Tsai A.S.H., et al. Correlation of axial length and myopic macular degeneration to levels of molecular factors in the aqueous. Sci Rep. 2019;9(1) doi: 10.1038/s41598-019-52156-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Guo D., Qi J., Du Y., et al. Tear inflammatory cytokines as potential biomarkers for myopic macular degeneration. Exp Eye Res. 2023;235 doi: 10.1016/j.exer.2023.109648. [DOI] [PubMed] [Google Scholar]
- 64.Yu H., Zhong Z., Zhao Y., et al. Insights into myopic choroidal neovascularization based on quantitative proteomics analysis of the aqueous humor. BMC Genom. 2023;24(1):767. doi: 10.1186/s12864-023-09761-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Matsumura S., Sabanayagam C., Wong C.W., et al. Characteristics of myopic traction maculopathy in myopic Singaporean adults. Br J Ophthalmol. 2021;105(4):531–537. doi: 10.1136/bjophthalmol-2020-316182. [DOI] [PubMed] [Google Scholar]
- 66.Li M., Yu J., Chen Q., et al. Clinical characteristics and risk factors of myopic retinoschisis in an elderly high myopia population. Acta Ophthalmol. 2023;101(2):e167–e176. doi: 10.1111/aos.15234. [DOI] [PubMed] [Google Scholar]
- 67.Shimada N., Tanaka Y., Tokoro T., Ohno-Matsui K. Natural course of myopic traction maculopathy and factors associated with progression or resolution. Am J Ophthalmol. 2013;156(5):948–957.e1. doi: 10.1016/j.ajo.2013.06.031. [DOI] [PubMed] [Google Scholar]
- 68.Hsia Y., Ho T.C., Yang C.H., Hsieh Y.T., Lai T.T., Yang C.M. Clinical characteristics and long-term evolution of lamellar macular hole in high myopia. PLoS One. 2020;15(5) doi: 10.1371/journal.pone.0232852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Meng J., Chen Y., Cheng K., et al. LONG-TERM progression pattern of myopic tractional maculopathy: outcomes and risk factors. Retina. 2023;43(7):1189–1197. doi: 10.1097/iae.0000000000003791. [DOI] [PubMed] [Google Scholar]
- 70.De Giacinto C., Pastore M.R., Cirigliano G., Tognetto D. Macular hole in myopic eyes: a Narrative review of the current surgical techniques. J Ophthalmol. 2019;2019 doi: 10.1155/2019/3230695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ohsugi H., Ikuno Y., Oshima K., Yamauchi T., Tabuchi H. Morphologic characteristics of macular complications of a dome-shaped macula determined by swept-source optical coherence tomography. Am J Ophthalmol. 2014;158(1):162. doi: 10.1016/j.ajo.2014.02.054. 70.e1. [DOI] [PubMed] [Google Scholar]
- 72.Baca-Moreno J., Henaine-Berra A., Osorio-Landa H.K., Berrones-Medina D., García-Aguirre G. Prevalence of dome-shaped macula and tilted disc syndrome in high myopia. Ophthalmic Surg Lasers Imaging Retina. 2023;54(10):568–572. doi: 10.3928/23258160-20230823-02. [DOI] [PubMed] [Google Scholar]
- 73.Zhu X., He W., Zhang S., Rong X., Fan Q., Lu Y. Dome-shaped macula: a potential protective factor for visual acuity after cataract surgery in patients with high myopia. Br J Ophthalmol. 2019;103(11):1566–1570. doi: 10.1136/bjophthalmol-2018-313279. [DOI] [PubMed] [Google Scholar]
- 74.García-Zamora M., Flores-Moreno I., Ruiz-Medrano J., et al. Atrophic, tractional, and neovascular grading system in a dome-shaped macula and ridge-shaped macula highly myopic cohort. Ophthalmologica. 2023;246(2):107–112. doi: 10.1159/000528993. [DOI] [PubMed] [Google Scholar]
- 75.Zhao X., Ding X., Lyu C., et al. Observational study of clinical characteristics of dome-shaped macula in Chinese Han with high myopia at Zhongshan Ophthalmic Centre. BMJ Open. 2018;8(12) doi: 10.1136/bmjopen-2018-021887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tang Y.P., Zhang X.B., Hu Z.X., et al. Vitreous metabolomic signatures of pathological myopia with complications. Eye (Lond) 2023;37(14):2987–2993. doi: 10.1038/s41433-023-02457-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Silva R. Myopic maculopathy: a review. Ophthalmologica. 2012;228(4):197–213. doi: 10.1159/000339893. [DOI] [PubMed] [Google Scholar]
- 78.Ikuno Y., Jo Y., Hamasaki T., Tano Y. Ocular risk factors for choroidal neovascularization in pathologic myopia. Invest Ophthalmol Vis Sci. 2010;51(7):3721–3725. doi: 10.1167/iovs.09-3493. [DOI] [PubMed] [Google Scholar]
- 79.Victor A.A., Andayani G., Djatikusumo A., et al. Recurrence risk of myopic choroidal neovascularisation: a systematic review of current study. BMJ Open Ophthalmol. 2023;8(1) doi: 10.1136/bmjophth-2023-001396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Coelho J., Ferreira A., Abreu A.C., et al. Choroidal neovascularization secondary to pathological myopia-macular Bruch membrane defects as prognostic factor to anti-VEGF treatment. Graefes Arch Clin Exp Ophthalmol. 2021;259(9):2679–2686. doi: 10.1007/s00417-021-05142-y. [DOI] [PubMed] [Google Scholar]
- 81.Liu L., Zhu D., Ding W., Zhang T., Ma X., Zou J. MiRNA-21-HIF-1α-VEGF Axis is associated with myopic choroidal neovascularization in Guinea pigs. Ophthalmic Res. 2022;65(5):493–505. doi: 10.1159/000522511. [DOI] [PubMed] [Google Scholar]
- 82.Chen D.Z., Koh V., Tan M., et al. Peripheral retinal changes in highly myopic young Asian eyes. Acta Ophthalmol. 2018;96(7):e846–e851. doi: 10.1111/aos.13752. [DOI] [PubMed] [Google Scholar]
- 83.Khatwani N., Makhija S., Ahuja A. Clinical profile and distribution of peripheral retinal changes in myopic population in a hospital-based study in North India. Indian J Ophthalmol. 2022;70(4):1280–1285. doi: 10.4103/ijo.IJO_946_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mitry D., Charteris D.G., Yorston D., et al. The epidemiology and socioeconomic associations of retinal detachment in Scotland: a two-year prospective population-based study. Invest Ophthalmol Vis Sci. 2010;51(10):4963–4968. doi: 10.1167/iovs.10-5400. [DOI] [PubMed] [Google Scholar]
- 85.Daien V., Le Pape A., Heve D., Carriere I., Villain M. Incidence, risk factors, and impact of age on retinal detachment after cataract surgery in France: a National population study. Ophthalmology. 2015;122(11):2179–2185. doi: 10.1016/j.ophtha.2015.07.014. [DOI] [PubMed] [Google Scholar]
- 86.Neuhann I.M., Neuhann T.F., Heimann H., Schmickler S., Gerl R.H., Foerster M.H. Retinal detachment after phacoemulsification in high myopia: analysis of 2356 cases. J Cataract Refract Surg. 2008;34(10):1644–1657. doi: 10.1016/j.jcrs.2008.06.022. [DOI] [PubMed] [Google Scholar]
- 87.Morano M.J., Khan M.A., Zhang Q., et al. Incidence and risk factors for retinal detachment and retinal tear after cataract surgery: IRIS® registry (intelligent research in sight) analysis. Ophthalmol Sci. 2023;3(4) doi: 10.1016/j.xops.2023.100314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ha A., Kim C.Y., Shim S.R., Chang I.B., Kim Y.K. Degree of myopia and glaucoma risk: a dose-response meta-analysis. Am J Ophthalmol. 2022;236:107–119. doi: 10.1016/j.ajo.2021.10.007. [DOI] [PubMed] [Google Scholar]
- 89.Wang Y.X., Yang H., Wei C.C., Xu L., Wei W.B., Jonas J.B. High myopia as risk factor for the 10-year incidence of open-angle glaucoma in the Beijing Eye Study. Br J Ophthalmol. 2023;107(7):935–940. doi: 10.1136/bjophthalmol-2021-320644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Bowd C., Belghith A., Rezapour J., et al. Multimodal deep learning classifier for primary open angle glaucoma diagnosis using wide-field optic nerve head cube scans in eyes with and without high myopia. J Glaucoma. 2023;32(10):841–847. doi: 10.1097/ijg.0000000000002267. [DOI] [PubMed] [Google Scholar]
- 91.Chen Z., Song Y., Li M., et al. Schlemm's canal and trabecular meshwork morphology in high myopia. Ophthalmic Physiol Opt. 2018;38(3):266–272. doi: 10.1111/opo.12451. [DOI] [PubMed] [Google Scholar]
- 92.Fan F., Luo Y., Lu Y., Liu X. Reasons for early ocular hypertension after uneventful cataract surgery. Eur J Ophthalmol. 2014;24(5):712–717. doi: 10.5301/ejo.5000441. [DOI] [PubMed] [Google Scholar]
- 93.Fan W., Zhang C., Ge L., et al. Prediction model for elevated intraocular pressure risk after silicone oil filling based on clinical features. Front Med. 2023;10 doi: 10.3389/fmed.2023.1340198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jabbour E., Azar G., Antoun J., Kourie H.R., Abdelmassih Y., Jalkh A. Incidence and risk factors of ocular hypertension following pars Plana vitrectomy and silicone oil injection. Ophthalmologica. 2018;240(3):129–134. doi: 10.1159/000489792. [DOI] [PubMed] [Google Scholar]
- 95.Zhu X., Qi J., He W., et al. Early transient intraocular pressure spike after cataract surgery in highly myopic cataract eyes and associated risk factors. Br J Ophthalmol. 2020;104(8):1137–1141. doi: 10.1136/bjophthalmol-2019-315117. [DOI] [PubMed] [Google Scholar]
- 96.Qi J., He W., Lu Q., Zhang K., Lu Y., Zhu X. Schlemm canal and trabecular meshwork features in highly myopic eyes with early intraocular pressure elevation after cataract surgery. Am J Ophthalmol. 2020;216:193–200. doi: 10.1016/j.ajo.2020.02.005. [DOI] [PubMed] [Google Scholar]
- 97.He W., Wei L., Liu S., et al. Role of optic nerve head characteristics in predicting intraocular pressure spikes after cataract surgery in highly myopic eyes. Ophthalmol Ther. 2023;12(4):2023–2033. doi: 10.1007/s40123-023-00714-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Choi W., Kim J.D., Bae H.W., Kim C.Y., Seong G.J., Kim M. Axial length as a risk factor for Steroid-induced ocular hypertension. Yonsei Med J. 2022;63(9):850–855. doi: 10.3349/ymj.2022.63.9.850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Dai L., Yang W., Qin X., et al. Serum metabolomics profiling and potential biomarkers of myopia using LC-QTOF/MS. Exp Eye Res. 2019;186 doi: 10.1016/j.exer.2019.107737. [DOI] [PubMed] [Google Scholar]