Abstract
Esophageal cancer (EC) is a malignant tumour with high morbidity and mortality rates. Recent studies have shown that much progress has been made in the research of radiotherapy in EC. This study aims to provide a comprehensive overview of the knowledge structure and research hotspots of radiotherapy in EC through bibliometrics. Publications related to radiotherapy in EC from 2014 to 2023 were searched on the web of science core collection database. VOSviewers, CiteSpace and R package ‘bibliometrix’ were used to conduct this bibliometric analysis. In total, 4258 articles from 76 countries led by China and the USA were included. The Chinese Academy of Medical Sciences—Peking Union Medical College has the highest number of publications. International Journal of Radiation Oncology Biology Physics is the most popular journal and also the most co-cited journal in this field. These publications come from 21 972 authors among which Liao Zhongxing had published the most papers and Cooper JS was co-cited most often. Neoadjuvant chemoradiotherapy and strategies based on it are the main topics in this research field. ‘IMRT’ and ‘immunotherapy’ are the primary keywords of emerging research hotspots. This is a bibliometric study that comprehensively summarizes the research trends and developments of radiotherapy in EC. This information identifies recent research frontiers and hot directions, which will provide a reference for scholars studying radiotherapy in EC.
Keywords: bibliometrics, esophageal cancer, radiotherapy, CiteSpace, VOSviewers
INTRODUCTION
Globally, oesophageal cancer (EC) is the ninth most frequently diagnosed malignancy and the sixth leading cause of cancer death [1]. The most common histologic subtypes of EC include squamous cell carcinoma followed by adenocarcinoma [2]. Oesophageal squamous cell carcinoma (ESCC) is the most prevalent histologic type in Asian countries, while oesophageal adenocarcinoma (EAC) is more common in Western countries (North America, Australia and Western Europe).
The management of EC, which is challenging and highly complex, depends on patients’ characteristics, including performance status and coexisting morbidities and tumour characteristics. Radiotherapy has been playing an indispensable role in the treatment of EC since the first linear accelerator was applied in clinical practice. In recent years, with the rapid development of biopharmaceutical technology and the updating and iteration of linear accelerators, studies in the field of radiotherapy in EC have emerged one after another.
Bibliometrics is a literature analysis method that analyzes the output and status of publications in a particular research field from a quantitative and qualitative perspective [3]. During the analysis, we can obtain detailed information about authors, keywords, journals, countries, institutions, references, etc. in the relevant research field [4]. Bibliometric tools such as CiteSpace [5], VoSviewer [6], R package ‘bibliometrix’ [7] and HistCite [8] are commonly applied to visualize the results of literature analysis, and these tools have been widely used in medical fields, such as oncology [9, 10] and orthopedics [11].
Studies of radiotherapy in EC have not emerged by means of bibliometrics. An in-depth bibliometric study of publications, countries, institutions, journals, authors and keywords is still required. This study aimed to perform a bibliometric analysis of publications of radiotherapy in EC in the recent 20 years (from 2004 to 2023) to identify current research status, and to look forward to the research trends and future development prospects in this field.
MATERIALS AND METHODS
Data source and literature search strategy
Web of Science (WoS) was selected as the primary database for this study due to its comprehensive coverage of over 12 000 academic journals and its frequent usage by researchers. When compared to other databases such as Scopus, Medline and PubMed, WoS provides the most comprehensive and reliable bibliometric analysis 6. The relevant articles in Web of Science Core Collection (WoSCC) from 1 January 2004 to 31 December 2023 were searched and exported on 20 January 2024, using all database versions. All authors agreed on the search strategy following consultations with senior literature search experts, with the following criteria: (TS = (oesophageal cancer)) AND (TS = (radiotherapy)). To facilitate further analysis of literature content, only articles and reviews written in English were included. A complete record and cited references were then extracted from relevant publications, saved in plain text format, for further research. The keyword frequency, search strategy, and the number of articles are displayed in Figure 1.
Fig. 1.
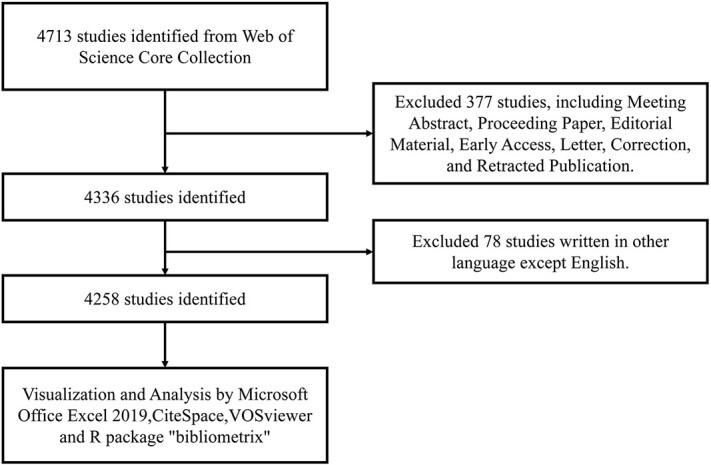
Publications screening flowchart.
Software for bibliometric analysis
This study used R version 4.3.1 [12], VOSviewer [13] and CiteSpace [14] as the software tools for performing bibliometric analysis. We calculated the frequency of collaboration between countries using the Bibliometrix R package version 4.3.1 [15]. The number of publications, citations and keyword frequency were calculated using VOSviewer. With the software’s embedded clustering algorithm, co-occurrence networks of essential keywords from scientific literature were constructed and visualized [16]. The co-authorship and co-occurrence analysis was the primary focus of this study. This tool was used to analyse country, institution and author collaborations. To identify highly cited references and keywords that experienced significant citation increases during a specific period, CiteSpace was used. Through the online bibliometrics website (https://bibliometric.com/), we were able to visualize international collaborations between countries.
RESULTS
A total of 4713 articles were retrieved, of which 455 were excluded, leaving a total of 4258 articles to be exported as plain text.
Overview of publication status
For the period from 2004 to 2023, we collected a total of 4258 related papers and review papers (Fig. 2). As shown in Figure 2, the number of articles related to EC radiotherapy shows an increasing trend, indicating that this research topic has been attracting considerable attention. In particular, the number of articles published reached 378 in 2022.
Fig. 2.
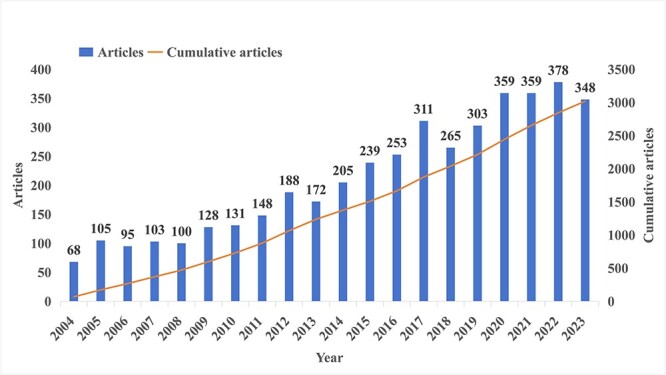
Number of publications per year and the cumulative number.
Country and institutional analysis
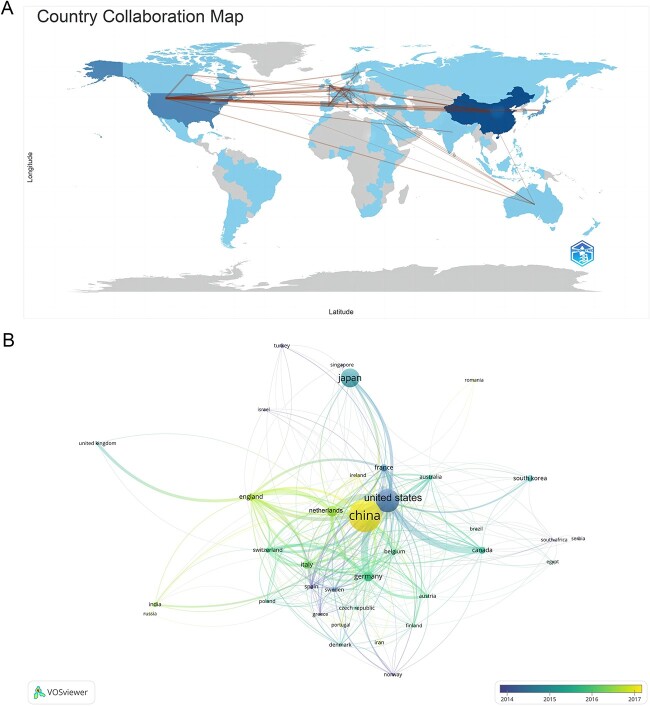
These publications came from 76 countries and 3708 institutions. The top ten countries are distributed in Europe, Asia and North America, mainly in Europe (n = 5) and Asia (n = 3). Among the countries, the country with the largest number of publications is China (n = 1750, 41.1%), followed by the USA (929, 21.8%), Japan (n = 640, 15.0%) and the Netherlands (n = 232, 5.4%) (Table 1). It is worth noting that, publications in China, the USA and Japan have been increased rapidly in recent years, especially in China (Fig. 3). Subsequently, we filtered and visualized 76 countries based on the number of publications more than or equal to 5, and constructed a collaborative network based on the number and relationship of publications in each country (Fig. 4A and B). Notably, there is a lot of active cooperation between different countries. For example, China has close cooperation with the USA, Germany, and Japan; the USA has active cooperation with China, Germany and Canada.
Table 1.
Top 10 countries and institutions on research of radiotherapy in oesophageal cancer
| Rank | Country | Counts | Institutions | Counts |
|---|---|---|---|---|
| 1 | China (Asia) | 1750 (41.1%) | Chinese Academy of Medical Sciences—Peking Union Medical College (China) | 440 (10.3%) |
| 2 | The USA (North America) | 929 (21.8%) | The University of Texas MD Anderson Cancer Centre (the USA)a | 435 (10.2%) |
| 3 | Japan (Asia) | 640 (15.0%) | The University of Texas System (the USA)a | 351 (8.2%) |
| 4 | Netherlands (Europe) | 232 (5.4%) | Shandong First Medical University and Shandong Academy of Medical Sciences (China) | 253 (5.9%) |
| 5 | Germany (Europe) | 198 (4.7%) | Fudan University (China) | 186 (4.4%) |
| 6 | Italy (Europe) | 132 (3.1%) | Cancer Institute and Hospital—CAMS (China) | 182 (4.3%) |
| 7 | The UK (Europe) | 119(2.8%) | Sun Yat Sen University (China) | 179 (4.2%) |
| 8 | France (Europe) | 113 (2.7%) | National Cancer Centre—Japan (Japan) | 150 (3.5%) |
| 9 | Canada (North America) | 108 (2.5%) | University of Amsterdam (the Netherlands) | 141 (3.3%) |
| 10 | Korea (Asia) | 91 (2.1%) | Shandong University (China) | 133 (3.1%) |
aMD Anderson Cancer Centre and the University of Texas System were counted separately.
Fig. 3.
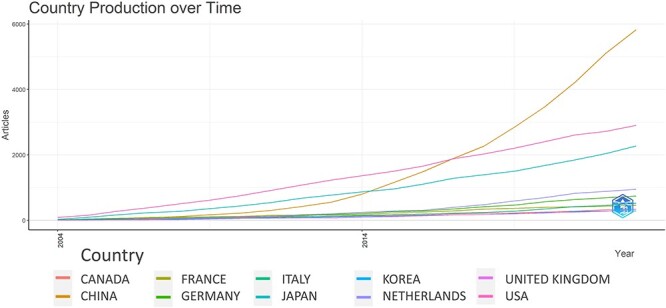
Country production over time (Top 10).
Fig. 4.
The geographical distribution (A) and visualization of countries or regions (B) on research of radiotherapy in oesophageal cancer.
The top 10 institutions are located in four countries, with three-fifths of them located in China. The first three institutions that published the most relevant papers are: Chinese Academy of Medical Sciences—Peking Union Medical College (n = 440, 10.3%), the University of Texas MD Anderson Cancer Centre (n = 435, 10.2%), and the University of Texas System (n = 351, 8.2%) (Table 1) (MD Anderson Cancer Centre and the University of Texas System were counted separately.)
Subsequently, we selected 83 institutions based on the minimum number of publications equal to 20 for visualization and constructed a collaborative network based on the number and relationship of publications of each institution (Fig. 5). As shown in Figure 5, the cooperation between the University of Texas MD Anderson Cancer Centre, Sun Yat Sen University and Shandong University is very close, and there is active cooperation between Shandong University and Nanjing Medical University.
Fig. 5.
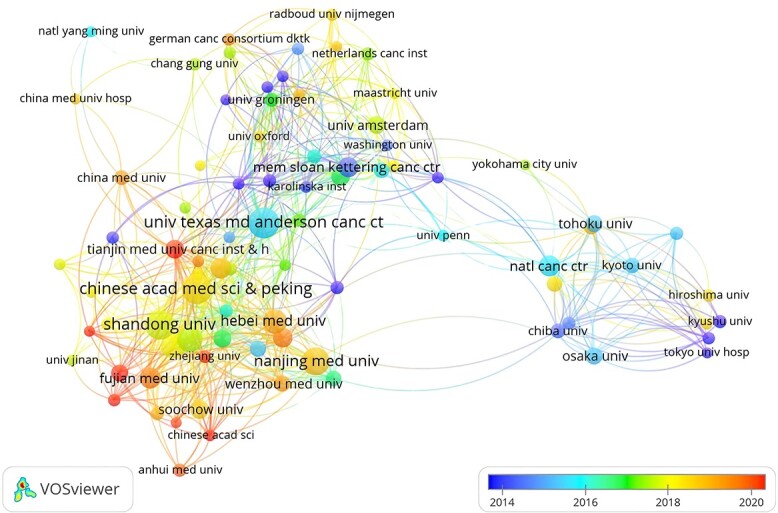
The visualization of institutions on research of radiotherapy in oesophageal cancer.
Journals and co-cited journals
Publications related to radiotherapy in EC were published in 620 journals. As shown in Table 2, International Journal of Radiation Oncology Biology Physics published most papers (n = 242, 5.7%), followed by Radiotherapy and Oncology 171 (4.0%), Diseases of the Oesophagus 163 (3.8%) and Frontiers in Oncology 145 (3.4%). Among the top 10 journals, the journal with the highest impact factor is International Journal of Radiation Oncology Biology Physics (IF = 7.0), followed by Radiotherapy and Oncology (IF = 5.7). Subsequently, we screened 46 journals based on the minimum number of relevant publications equal to 20 and mapped the journal network (Fig. 6A). Figure 6A shows that International Journal of Radiation Oncology Biology Physics has active citation relationships with Frontiers in Oncology, Radiation Oncology, Radiotherapy and Oncology, and Diseases of the Oesophagus, etc.
Table 2.
Top 10 journals for research of radiotherapy in oesophageal cancer
| Rank | Journal | Counts | Country | IF | JCR-c |
|---|---|---|---|---|---|
| 1 | International Journal of Radiation Oncology Biology Physics | 242 (5.7%) | The USA | 7.0 | Q1 |
| 2 | Radiotherapy and Oncology | 171 (4.0%) | The Netherlands | 5.7 | Q1 |
| 3 | Diseases of the Oesophagus | 163 (3.8%) | Australia | 2.6 | Q4 |
| 4 | Frontiers in Oncology | 145 (3.4%) | Switzerland | 4.7 | Q2 |
| 5 | Radiation Oncology | 142 (3.3%) | UK | 3.6 | Q2 |
| 6 | Oesophagus | 72(1.7%) | Japan | 2.4 | Q3 |
| 7 | Anticancer Research | 65(1.5%) | Greece | 2.0 | Q4 |
| 8 | Cancers | 64(1.5%) | Switzerland | 5.2 | Q1 |
| 9 | Annals of Surgical Oncology | 63(1.5%) | The USA | 3.7 | Q1/Q2 |
| 10 | Medicine | 60(1.4%) | The USA | 1.6 | Q3 |
Fig. 6.
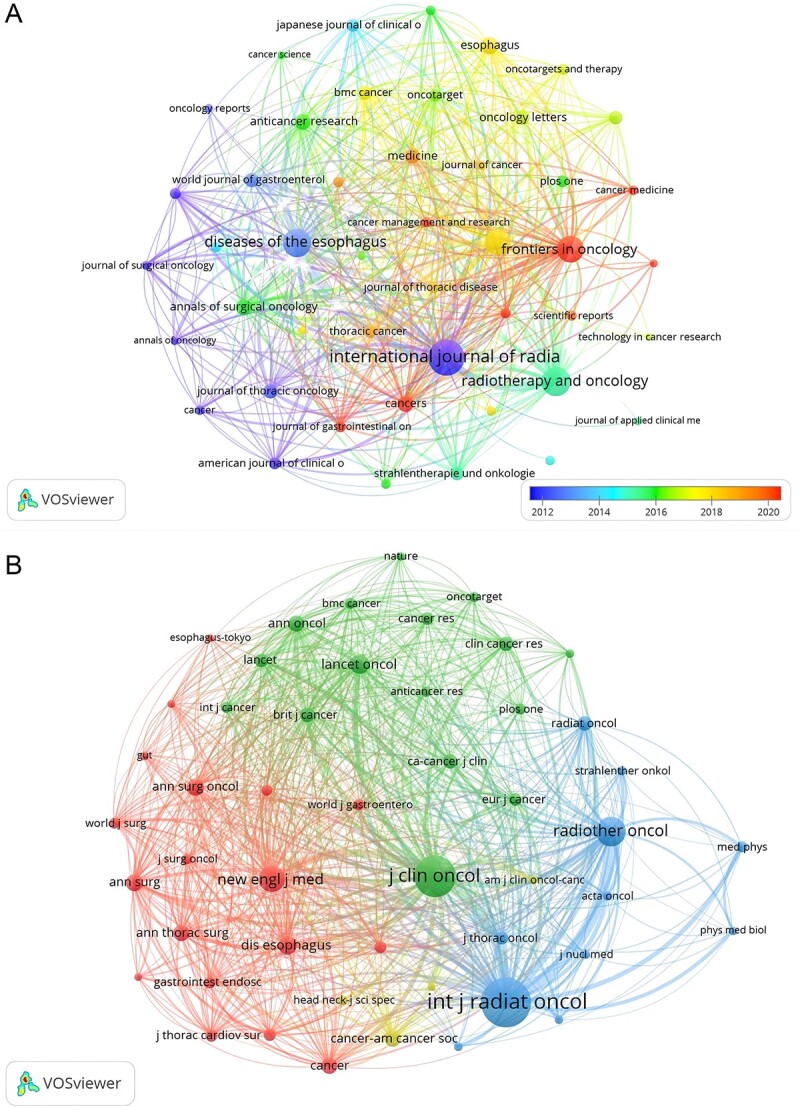
The visualization of journals (A) and co-cited journals (B) on research of radiotherapy in oesophageal cancer.
As shown in Table 3, among the top 10 co-cited journals, three journals were cited more than 5000 times, and International Journal of Radiation Oncology Biology Physics (Co-citation = 13 015) was the most cited journal, followed by Journal of Clinical Oncology (co-citation = 9674), Radiotherapy and Oncology (co-citation = 5436). In addition, the impact factor of New England Journal of Medicine is the highest (IF = 158.5), followed by Lancet Oncology (IF = 51.1). Journals with the minimum co-citation equal to 500 were filtered to map the co-citation network (Fig. 6B). As shown in Figure 6B, International Journal of Radiation Oncology Biology Physics has positive co-citation relationships with Journal of Clinical Oncology, Radiotherapy and Oncology, New England Journal of Medicine, and Lancet Oncology, etc.
Table 3.
Top 10 co-cited journals for research of radiotherapy in esophageal cancer
| Rank | Journal | Co-citation | Country | IF | JCR-c |
|---|---|---|---|---|---|
| 1 | International Journal of Radiation Oncology Biology Physics | 13 015 | The USA | 7.0 | Q1 |
| 2 | Journal of Clinical Oncology | 9674 | The USA | 45.3 | Q1 |
| 3 | Radiotherapy and Oncology | 5436 | The Netherlands | 5.7 | Q1 |
| 4 | New England Journal of Medicine | 4259 | The USA | 158.5 | Q1 |
| 5 | Lancet Oncology | 2670 | The USA | 51.1 | Q1 |
| 6 | Diseases of the Oesophagus | 2384 | Australia | 2.6 | Q4 |
| 7 | Annals of Surgery | 2056 | The USA | 9.0 | Q1 |
| 8 | Annals of Surgical Oncology | 1962 | The USA | 3.7 | Q1 |
| 9 | Cancer | 1928 | The USA | 6.2 | Q1 |
| 10 | Annals of Oncology | 1890 | The UK | 50.5 | Q1 |
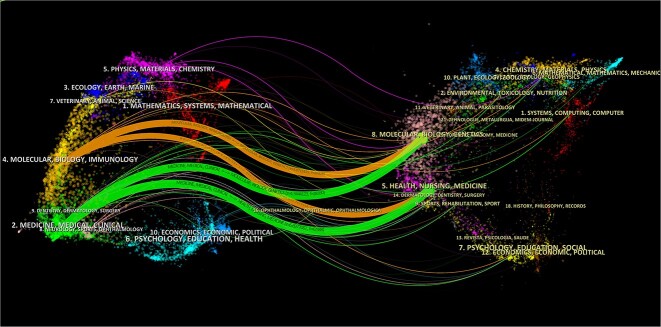
The dual-map overlay of journals shows the citation relationships between journals and co-cited journals, with clusters of citing journals on the left and clusters of cited journals on the right [17]. As shown in Figure 7, the orange path and green path are the main citation paths, which represents the research published in Molecular/Biology/Genetics journals and Health/Nursing/Medicine journals are mainly cited by literature in Molecular/Biology/Immunology journals and Medicine/Medical/Clinical journals.
Fig. 7.
The dual-map overlay of journals on research of radiotherapy in oesophageal cancer. Notes: the citing journals are on the left, the cited journals are on the right, and the coloured path represents a citation relationship.
Authors and co-cited authors
A total of 21 972 authors participated in research of radiotherapy in EC. Among the top 10 authors, six authors published more than or equal to 40 papers (Table 4). We build a collaborative network based on authors whose number of published papers is more than or equal to 15 (Fig. 8A). Liao Zhongxing, Yu Jinming and Lin Steven H have the largest nodes because they publish the most related publications. Besides, we observed close collaboration among multiple authors. For example, Xiao Zefen has close cooperation with Wang Xin, Sun Xincheng and Zhang Wencheng; Lin SH has active cooperation with Wang Xin, Xi Mian and Zhang Wencheng, etc.
Table 4.
Top 10 authors and co-cited authors on research of radiotherapy in oesophageal cancer
| Rank | Authors | Count | Co-cited Authors | Citations |
|---|---|---|---|---|
| 1 | Liao ZX | 57 | Cooper JS | 734 |
| 2 | Yu JM | 48 | Minsky BD | 724 |
| 3 | Lin SH | 47 | Van Hagen P | 700 |
| 4 | Xiao ZF | 41 | Stahl M | 631 |
| 5 | Komaki R | 41 | Herskovic A | 563 |
| 6 | Zhang WC | 40 | Jemal A | 514 |
| 7 | Sun XC | 35 | Ajanj JA | 470 |
| 8 | Wang X | 33 | Mariette C | 411 |
| 9 | Zhao KL | 32 | Rice TW | 408 |
| 10 | Jingu KC | 32 | Burmeister BH | 402 |
Fig. 8.
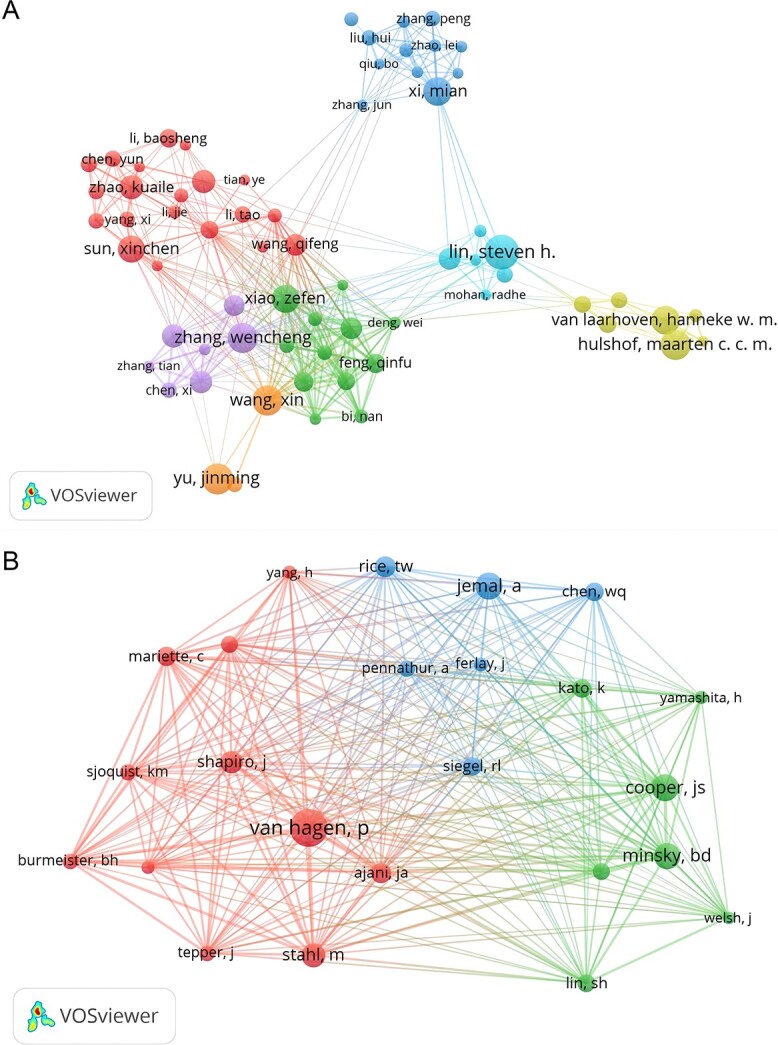
The visualization of authors (A) and co-cited authors (B) on research of radiotherapy in oesophageal cancer.
Among the 45 498 co-cited authors, six authors were co-cited more than 500 times (Table 4). The most co-cited author is Cooper JS (n = 734), followed by Minsky BD (n = 724), Van Hagen P (n = 700), Stahl M (n = 631) and Herskovic A (n = 563). Authors with minimum co-citations equal to 200 were filtered to map co-citation network graphs (Fig. 8B). As shown in Figure 8B, there are also active collaborations among different co-cited authors, such as Van Hagen P, Cooper JS and Minsky BD.
Co-cited references
There are 74 964 co-cited references on research of radiotherapy in EC since 2004. In top 10 co-cited references (Table 5), all references were co-cited at least 280 times, and a reference was co-cited more than 700 times. We selected references with co-citation more than or equal to 100 for the construction of the co-citation network map (Fig. 9). According to Figure 9, ‘Van Hagen P, 2012, New Engl J Med, V366, P2074’ shows active co-cited relationships with ‘Cooper JS, 1999, JAMA-J Am Med Assoc, V281, P1623’ and ‘Minsky BD, 2002, J Clin Oncol, V20, P1167’ shows active co-cited relationships with ‘Jemal A, 2011, CA-Cancer J Clin, V61, P134’, etc.
Table 5.
Top 10 co-cited references on research of radiotherapy in oesophageal cancer
| Rank | Co-cited reference | Citations |
|---|---|---|
| 1 | Cooper JS, 1999, JAMA-J Am Med Assoc, V281, P1623 | 713 |
| 2 | Van Hagen P, 2012, New Engl J Med, V366, P2074 | 676 |
| 3 | Minsky BD, 2002, J Clin Oncol, V20, P1167 | 643 |
| 4 | Herskovic A, 1992, New Engl J Med, V326, P1593 | 530 |
| 5 | TepperJ, 2008, J Clin Oncol, V26, P1086 | 364 |
| 6 | Jemal A, 2011, CA-Cancer J Clin, V61, P134 | 357 |
| 7 | Walsh TN, 1996, New Engl J Med, V335, P462 | 313 |
| 8 | Stahl M, 2005, J Clin Oncol, V23, P2310 | 312 |
| 9 | Shapiro J, 2015, Lancet Oncol, V16, P1090 | 308 |
| 10 | Urba SG, 2001, J Clin Oncol, V19, P305 | 282 |
Fig. 9.
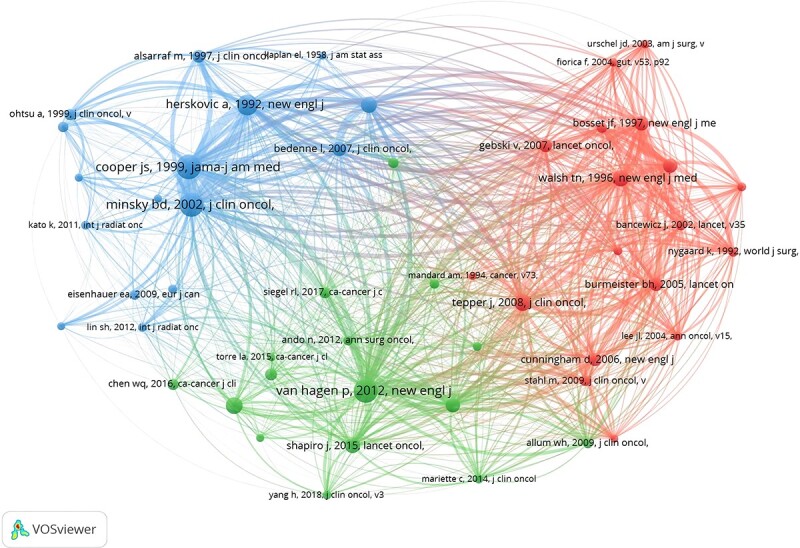
The visualization of co-cited references on research of radiotherapy in oesophageal cancer.
Reference with citation bursts
Reference with citation bursts refers to those references that are frequently cited by scholars in a certain field over a period of time. In our study, 10 references with strong citation bursts were identified by CiteSpace (Fig. 10). As shown in Figure 10, every bar represents a year, and the red bar represents strong citation burstiness. Citation bursts for references appeared as early as 2006 and as late as 2023. The reference with the strongest citation burst (strength = 115.51) was titled ‘Preoperative for Esophageal or Junctional Cancer’, authored by van Hagen P et al. with citation bursts from 2013 to 2017. The reference with the second strongest citation burst (strength = 69.31) was titled ‘Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial’, authored by Shapiro J et al. with citation bursts from 2016 to 2020. Overall, the bursts strength of these 10 references ranged from 37.92 to 115.51, and endurance strength was from 2 to 4 years. Table 6 summarizes the main research contents of the 10 references in the order of the literature in Figure 10.
Fig. 10.
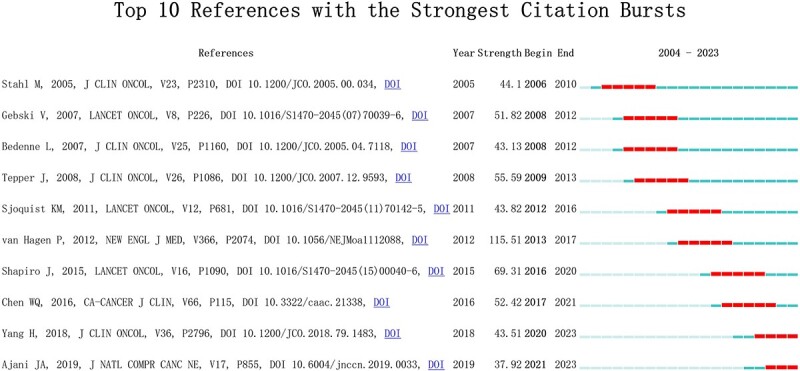
Top 10 references with strong citation bursts. A prominent bar indicates high citations in that year.
Table 6.
The main research contents of the 10 references with strong citations bursts
| Rank | Strength | Main research content |
|---|---|---|
| 1 | 115.51 | Preoperative chemoradiotherapy improved survival among patients with potentially curable oesophageal or oesophagogastric-junction cancer. |
| 2 | 69.31 | Preoperative chemoradiotherapy improved survival among patients with potentially curable oesophageal or oesophagogastric-junction cancer. |
| 3 | 55.59 | Chemoradiotherapy followed by surgery represented a long-term survival advantage in the treatment of oesophageal cancer. |
| 4 | 52.42 | Cancer Statistics in China, 2015. |
| 5 | 51.82 | Preoperative chemoradiotherapy improved survival in patients with adenocarcinoma of the oesophagus. |
| 6 | 44.10 | Adding surgery to chemoradiotherapy improves local tumour control but does not improve survival in patients with locally advanced oesophageal squamous carcinoma. |
| 7 | 43.82 | Neoadjuvant chemoradiotherapy or chemotherapy improves survival in patients with resectable oesophageal cancer compared with surgery alone. |
| 8 | 43.51 | Neoadjuvant chemoradiotherapy plus surgery improves survival over surgery alone among patients with locally advanced oesophageal squamous cell carcinoma. |
| 9 | 43.13 | There is no benefit of adding surgery after chemoradiotherapy compared with continuing additional chemoradiotherapy in patients with locally advanced thoracic oesophageal cancer who respond to chemoradiation. |
| 10 | 37.92 | Oesophageal and oesophagogastric Junction Cancers, Version 2.2019, NCCN Clinical Practice Guidelines in Oncology. |
Hotspots and frontiers
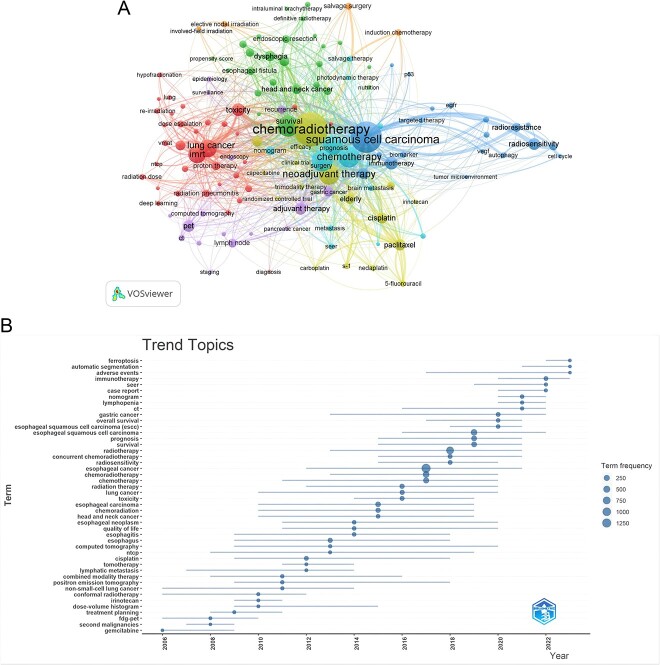
Through the co-occurrence analysis of keywords, we could quickly capture research hotspots in a certain field. Table 7 shows the top 20 high-frequency keywords in research of radiotherapy in EC. Among these keywords, chemoradiotherapy, squamous cell carcinoma, survival, neoadjuvant therapy, IMRT and toxicity appeared more than 100 times, which represented the main research directions of radiotherapy in EC. We filtered keywords with the number of occurrences more than or equal to 10 (‘radiotherapy’ and ‘oesophageal cancer’ excluded) and performed cluster analysis through VOSviewer (Fig. 11A). The thicker the lines between the nodes, the stronger the connection between the keywords. As shown in Figure 11A, we obtained seven clusters in total, representing different research directions. The keywords in blue clusters consists of squamous cell carcinoma, immunotherapy, radiosensitivity, radioresistance, epidermal growth factor receptor (EGFR), vascular endothelial growth factor (VEGF), cell cycle, biomarker, etc. The keywords in pale blue clusters consists of chemotherapy, surgery, prognosis, brain metastasis, nomogram, etc. The keywords in green clusters consist of survival, dysphagia, risk factor, palliation, endoscopic resection, oesophageal fistula, etc. The keywords in red clusters consist of intensity-modulated radiotherapy (IMRT), lung cancer, toxicity, radiation dose, esophagitis, etc. The keywords in yellow clusters consist of chemoradiotherapy, neoadjuvant therapy, meta-analysis, elderly, cisplatin, paclitaxel, nedaplatin, 5-fluorouracil, s-1, etc. The keywords in orange clusters consist of involved-field irradiation, elective nodal irradiation, salvage surgery, induction chemotherapy, etc. The keywords in purple clusters consist of adjuvant therapy, positron emission tomograpy (PET), gastric cancer, lymph node, recurrence, etc.
Table 7.
Top 20 keywords on research of radiotherapy in oesophageal cancer
| Rank | Keywords | Counts | Rank | Keywords | Counts |
|---|---|---|---|---|---|
| 1 | Oesophageal cancer | 1673 | 11 | Lung cancer | 191 |
| 2 | Radiation therapy | 960 | 12 | Toxicity | 104 |
| 3 | Chemoradiotherapy | 737 | 13 | Immunotherapy | 98 |
| 4 | Squamous carcinoma | 622 | 14 | Adjuvant therapy | 89 |
| 5 | Chemotherapy | 277 | 15 | PET | 88 |
| 6 | Survival | 277 | 16 | Radiosensitivity | 88 |
| 7 | Surgery | 275 | 17 | Paclitaxel | 82 |
| 8 | Neoadjuvant therapy | 262 | 18 | Cisplatin | 74 |
| 9 | Prognosis | 239 | 19 | Adenocarcinoma | 73 |
| 10 | IMRT | 193 | 20 | Elderly | 66 |
Fig. 11.
Keyword cluster analysis (A) and trend topic analysis (B).
The trend topic analysis of the keywords (Fig. 11B) showed that from 2010 to 2013, the research in this period mainly focused on target volume, complete response, irinotecan, tomotherapy, conformal therapy, dose-volumes. Since 2013, scholars have begun to actively explore the comprehensive treatment strategy of EC, and the main keywords are trimodality therapy, dysphagia, chemoradiotherapy, prognosis, squamous cell carcinoma, overall survival and neoadjuvant chemoradiotherapy. Besides, immunotherapy has appeared frequently in the past three years (2020–2022), so it is very likely to represent the current research hotspots of radiotherapy in EC.
DISCUSSION
General information
From 2004 to 2013, research in this field was still in its infancy, with annual publication of less than 200 papers. From 2014 to 2023, the number of publications began to increase stably. However, in 2017, the number of publications had rapidly increased to 311, probability because Pabolizumab and Navulizumab were approved as indications for the treatment of advanced EC in the USA and Japan in 2017, respectively, marking the beginning of an era of immunotherapy for EC.
China, the USA and Japan are major countries conducting research of radiotherapy in EC, and the most publications were produced in China 1750 (41.1%). Six of the top ten research institutions are located in China, followed by the USA. The number of publications indicated that Chinese scholars are more concerned about the radiotherapy of EC, probably related to the high incidence rate of EC in China. We noticed the close cooperation among four countries: China has close cooperation with the USA, Germany and Japan; the USA has active cooperation with China, Germany and Canada.
When it comes to research institutions, there is a good cooperative relationship between some of them, such as the University of Texas MD Anderson Cancer Centre, Sun Yat Sen University and Shandong University, and there is active cooperation between Shandong University and Nanjing Medical University. Therefore, we strongly recommend research institutions in various countries carry out extensive cooperation and communication to jointly promote the development of radiotherapy in EC.
Most of the research of radiotherapy in EC was published in International Journal of Radiation Oncology Biology Physics (IF = 7.0, Q1), indicating it is currently the most popular journal in this research field. Regarding the co-cited journals, we could find most of them are high-impact Q1 journals. Obviously, these journals are high-quality international journals, providing support for the study of radiotherapy in EC.
From the perspective of the author, Liao Zhongxing, Yu Jinming, Lin Steven H, Xiao Zefen, Komaki R and Zhang Wencheng published more than 40 papers per capita. Professor Lin Steven H published the most articles with 57 papers.
Lin Steven H and his group have been devoted to both clinical studies and physical technology related to proton beam therapy (PBT), and conducted a series of clinical trials. Lin published the first report of the prospective study evaluating normal tissue toxicity from concurrent chemotherapy (CChT) and PBT (CChT/PBT) with 62 EC patients between 2006 and 2010 and concluded that this modality was associated with a few severe toxicities, but the pathologic response and clinical outcomes were encouraging [18].
Lin reviewed contemporary data from 580 EC patients treated with nCRT at three academic institutions from 2007 to 2013 and revealed that advanced radiation therapy technologies (IMRT and PBT) were associated with significantly reduced rate of postoperative complications and length of in-hospital stay compared to 3D conformal radiation therapy (3D-CRT), with PBT displaying the greatest benefit in a number of clinical endpoints [19].
Lin summarized [20] that the dosimetric advantage of IMRT over three-dimensional conformal radiotherapy can translate into improved clinical outcomes, and volumetric modulated arc therapy can produce equivalent or superior dosimetric quality with significantly higher treatment efficiency over IMRT in EC. Furthermore, compared with photon therapy, proton therapy has the potential to achieve further clinical improvement due to their physical properties.
In order to reveal whether dosimetric advantages of PBT translate to improved clinical outcomes compared with IMRT, Lin conducted a phase IIB randomized controlled clinical trials [21] with 107 patients analyzed from April 2012 to March 2019. The results showed that for locally advanced EC, PBT reduced the risk and severity of adverse events (AEs) compared with IMRT while maintaining similar progression-free survival (PFS).
Knowledge base
A co-cited reference refers to a reference that is cited together by multiple other publications, so that co-cited references could be considered as the research basis in a field [14].
In terms of co-cited authors, Cooper JS (n = 734) is the most frequently cited author, followed by Minsky BD (n = 724) and Van Hagen P (n = 700).
Cooper JS and his group conducted a chemoradiotherapy of locally advanced EC long-term follow-up of a prospective randomized trial (RTOG 85–01). The randomized controlled trial was conducted from 1985 to 1990 with a follow-up of at least 5 years, followed by a prospective cohort study conducted between May 1990 and April 1991. The results showed that combined therapy increases the survival of patients who have squamous cell or adenocarcinoma of the oesophagus, T1–3 N0–1 M0, compared with RT alone. As a large-scale randomized controlled clinical study, the RTOG 85–01 study [22] has laid the foundation for synchronous chemoradiotherapy in the treatment of non-surgical locally advanced EC.
Minsky BD and his group conducted a phase III trial [23] of combined-modality therapy for EC (RTOG 94–05) to compare the local/regional control, survival, and toxicity of combined-modality therapy using high-dose (64.8 Gy) vs standard-dose (50.4 Gy) radiation therapy for the treatment of patients with EC. The RTOG 94–05 study revealed that the higher radiation dose did not increase survival or local/regional control, but did increase toxic side effects. Based on this trial, the standard radiation dose for patients treated with concurrent chemoradiotherapy for EC has been recommended at 50.4 Gy and has been used to this day. However, researchers in different countries have never stopped discussing radiation dose, which is one of the reasons why this study has been cited repeatedly over the past 20 years.
From March 2004 to 2008, Van Hagen P and his group conducted a randomized controlled clinical trials (CROSS) [24] to compare chemoradiotherapy followed by surgery with surgery alone in this patient population. They revealed that preoperative chemoradiotherapy improved survival among patients with potentially curable oesophageal or esophagogastric-junction cancer. Obviously, the achievements of Van Hagen P laid a theoretical and experimental foundation for exploring preoperative chemoradiotherapy of EC. The CROSS study is the only RCT study in the field of EC and esophagogastric junction cancer, which is also the longest follow-up report, up to 10 years, among all large studies of neoadjuvant radiotherapy and chemotherapy for EC. This large 10-year follow-up report further confirmed and highlighted the survival advantages of radiotherapy and chemotherapy neoadjuvant over surgery alone.
Hotspots and frontiers
References with citation bursts represent emerging topics within a particular research field, as these references have been frequently cited by researchers in recent years [25]. According to the main research contents of references with strong citations bursts (Table 6), we can find that neoadjuvant chemoradiotherapy and strategies based on it are the current major topics in the research of radiotherapy in EC. In addition to references with citation bursts, keywords can also help us quickly capture the distribution and evolution of hotspots in the research field of radiotherapy in EC. Excluding keywords such as radiotherapy, and oesophageal cancer, Table 7 mainly includes the following keywords: chemoradiotherapy, squamous cell carcinoma, surgery, neoadjuvant therapy, IMRT, immunotherapy, adjuvant therapy, positron emission tomography (PET), radiosensitivity, adenocarcinoma and elderly. According to keyword clustering analysis and trend topic analysis (Fig. 11), we concluded that the research of radiotherapy in EC mainly focuses on the following aspects:
Squamous cell carcinoma
In 2020, the majority of new cases worldwide (85%) were squamous cell carcinoma (SCC), with the remaining cases being adenocarcinoma (AC) (14%). Both types exhibit distinct geographical patterns [26]. SCC incidence rates were highest in the regions of Eastern Africa and Eastern and South-Central Asia, which is likely due to exposure to specific carcinogens such as tobacco smoking, hot beverage consumption and indoor air pollution. In contrast, AC has been found to be dominant in several populations, particularly in males from North America, Northern and Western Europe, and Oceania who have experienced a decline in smoking prevalence (a risk factor for SCC) [27]. Additionally, changes in the prevalence of AC risk factors, such as gastroesophageal reflux disease (GORD), Barrett’s oesophagus, abdominal obesity and a decrease in Helicobacter pylori infection, have contributed to this trend [28].
Determining the optimal dose of radical radiotherapy for EC has been a controversial issue. The standard dose in Europe and the USA is between 50 and 50.4 Gy, while in China and Japan, it is typically between 60 and 70 Gy due to differences in histological type. However, the RTOG 94–05 study revealed that higher radiation dose (64.8 Gy) did not increase survival or local/regional control compared to the standard dose (50.4 Gy), but did increase toxic side effects. The recommended standard radiation dose for patients undergoing concurrent chemoradiotherapy for EC is 50.4 Gy, based on this trial. The ARTDECO phase 3 randomized study investigated the use of the simultaneous integrated boost (SIB) technique for inoperable EC. The study compared 50.4 Gy in 28 fractions to the primary tumour and adjacent lymphatics with a boost to the primary tumour to 61.6 Gy [29]. However, the study did not show a significant improvement in locoregional PFS for either SCC or AC, and there was no overall survival (OS) benefit. It is important to note that although current evidence does not demonstrate the benefit of dose escalation, most of the randomized trials did not separately analyse the outcomes based on different histology types.
A meta-analysis was conducted to compare the OS outcomes of patients with different histology types who were treated with 60 Gy or more and those treated with less than 60 Gy. The best OS was observed in SCC treated with a high dose, followed by AC with a low dose, SCC with a low dose, and the worst OS was observed in AC treated with a high dose. This study proposes four subgroups that could inform the design of clinical trials. Further large clinical trials are needed to address this controversy [30].
Neoadjuvant therapy
The purpose of adjuvant chemotherapy is to control and eliminate potential micrometastases in the body. Adjuvant radiotherapy is designed to control non-R0 resected local lesions and suspicious scattered tumour cells. A combination of both aims is to improve the long-term survival rate in patients with locally advanced EC after surgery. Neoadjuvant therapy for EC has gradually become an important exploration direction. It has been previously reported that the effects of neoadjuvant radiotherapy and chemotherapy are better than adjuvant radiotherapy and chemotherapy [31].
In 2007, Gebski et al. [32] included eight randomized controlled trials of neoadjuvant radiotherapy and conducted a meta-analysis. The analysis showed that preoperative chemoradiation could reduce the risk of death in patients with EC (HR = 0.81, P = 0.002), reducing the relative risk of death by 19% and increasing the 2-year survival rate of patients with EC by 13%. Since then, neoadjuvant chemoradiotherapy has become the standard model for multidisciplinary treatment of EC.
In 2010, Vallböhmer et al. [33] retrospectively analyzed 229 cases of EC after neoadjuvant therapy, including 118 ADC, 118 SCC (284 neoadjuvant chemoradiation, 15 neoadjuvant chemotherapy). The 5-year survival rate was 55%, disease-specific 5-year survival rate was 68%, recurrence rate was 3.4% (n = 70), local recurrence rate was 3.3% and distant recurrence rate was 20.1%. Cox regression analysis determined that age was the only independent predictor of survival, and gender, histology, type of oesophagectomy, type of neoadjuvant therapy and number of resected lymph nodes had no effect on prognosis. It can be seen that the response rate after neoadjuvant chemoradiotherapy, that is, the effect of clinical imaging on the atrophy of the lesion is a good indication for the follow-up of surgery.
The epoch-making randomized controlled multicentre trial of neoadjuvant chemoradiation for EC was reported in the 2008 CROSS study [34] whereby neoadjuvant chemoradiotherapy in patients with potentially curable EC was compared with surgery alone, with 175 patients in each group. The 2012 CROSS study [24] reported preliminary results: of the 161 patients who underwent chemoradiotherapy, 47 (29%) had pCR. The median OS of the chemoradiotherapy-operative group was 49.4 months, compared with 24.0 months in the surgical group. The OS rate of the chemoradiotherapy-operative group was significantly improved (HR = 0.657, P = 0.003). In the 2015, CROSS study [29] after a median of 45 months of follow-up, a total of 366 patients were analyzed (178 in the neoadjuvant chemoradiation group and 188 in the surgery alone group). The median survival of the neoadjuvant chemoradiation group and the surgery alone group was 48.6 months and 24.0 months, respectively (P = 0.003). The median survival of patients with neoadjuvant chemoradiotherapy plus surgery in the AC group was 43.2 months, compared with 27.1 months in the surgery alone group (P = 0.038). Long-term follow-up in the CROSS study confirmed a significant benefit in the OS of neoadjuvant chemoradiotherapy in patients with resectable oesophageal or oesophagogastric junctional cancer.
IMRT
In recent decades, multiple sophisticated radiotherapy approaches have emerged, such as 3D-CRT, IMRT, image-guided radiotherapy, tomotherapy, intensity-modulated arc therapy and volumetric modulated arc therapy. These techniques aim to enhance target area conformity and radiation dosage while simultaneously lowering toxicity levels in normal organs [35–37].
Based on an inverse planning algorithm and complex beam arrangements, IMRT can improve dose conformity and spare OARs more effectively than 3D-CRT. Overall, the majority of dosimetric studies have consistently shown the advantages of IMRT in enhancing planning target volume (PTV) coverage, homogeneity and conformity in EC [38–44]. Nevertheless, the benefits of IMRT in OARs sparing can vary depending on the tumour location.
Kole et al. [40] examined 19 patients who received treatment with IMRT and compared the 3-field and 4-field 3D-CRT plans on the same patients. Based on their findings, they concluded that IMRT significantly decreased the dose to the heart compared to 3D-CRT. The study demonstrated a noteworthy decrease in mean dose (22.9 vs 28.2 Gy) and V30 (24.8% vs 61.0%; P < 0.05) as well as a significant improvement in the target conformity via IMRT, as indicated by the conformality index (the ratio of total volume receiving 95% of prescription dose to the planning target volume receiving 95% of prescription dose). The mean conformality index decreased from 1.56 to 1.30 with the implementation of IMRT. However, a study [45] conducted on 45 patients with EC who underwent 3D-CRT or IMRT treatment showed that patients receiving IMRT had higher rates of radiation pneumonitis, which was associated with increased V20 and V30 values.
The target range choice of involved-field irradiation (IFI) or elective lymph node drainage area prophylactic irradiation (ENI) is a controversial issue in the field of oesophageal tumour radiotherapy, especially in the era of high-precision irradiation. When ENI was selected as the target field for patients undergoing radical synchronous radiotherapy, the incidence of serious adverse reactions was reported to be up to 50% or more [23, 46]. In the study by Lyu et al. [47], 228 patients with stages II and III thoracic SCC received concurrent chemoradiation. The results showed that the ENI and IFI groups had similar rates of overall failure, local failure and lymph node failure both in-field and out-of-field. Zhang et al. [48] similarly found no statistically significant difference in the OS and PFS of the patients in the EN and IFI groups. Meta-analyses have consistently concluded that IFI does not compromise local control or survival rates, while mitigating the adverse effects of radiotherapy compared to ENI [49, 50].
Immunotherapy
The ATTRACTION-1 study was the first clinical trial to investigate the effectiveness of a PD-1 monoclonal antibody in treating advanced oesophageal squamous cell cancer using immunotherapy as a third-line treatment [51]. Since then, studies on the application of immunotherapy to various stages of EC have continued to emerge.
In the PALACE-1 study, the pCR rate was as high as 55.6% after combining pembrolizumab with the same neoadjuvant radiotherapy regimen (41.4 Gy), which was superior to the previous CROSS study (29%) and NEOCRTEC5010 study (43.2%) [52]. In this study, a radiotherapy dose of 41.4 Gy did not increase the incidence of postoperative complications but increased pulmonary toxicity. When multidrug combination therapy is performed, the toxicity of chemotherapeutic agents, immune checkpoint inhibitors and radiotherapy may be superimposed. Therefore, 41.4 Gy may be a safer radiotherapy dose when combined with immunotherapy for EC. It is necessary to obtain long-term results to confirm whether the benefits of combined treatment outweigh the risks.
A phase 3 trial (CheckMate 577) demonstrated that nivolumab, a checkpoint inhibitor, as adjuvant therapy in patients with oesophageal or gastroesophageal junction cancer after neoadjuvant chemoradiotherapy, was associated with significantly longer disease-free survival (22.4 months vs 11.0 months) in those who received adjuvant nivolumab compared to those who received placebo [53].
The NEXUS study examined the effectiveness and safety of tirilizumab in combination with CRT for unresectable ESCC in a single-arm, phase II trial. The results revealed a remarkably high pCR rate of 61.5% and major pathological response (MPR) of 76.9%, along with a favourable safety profile [54]. However, additional phase III studies are necessary to validate the efficacy of this option. Several phase III randomized controlled studies (RCTs) of immuno-combination definitive chemoradiotherapy (dCRT) are currently underway or have been completed, including the RATIONALE 311 study, the KEYNOTE-975 study, the KUNLUN study, SKYSCRAPER-07 study and NCT04426955. The doses of radiation used in each study ranged from 50 to 64 Gy. It is anticipated that further positive results will provide a high-level, evidence-based rationale for the future application of this combination therapy.
CONCLUSIONS
Radiotherapy plays a vital role in treating EC. The growing number of publications indicates that radiotherapy research in EC is highly valued worldwide. China and the USA are at the forefront, but collaboration between institutions and countries needs further strengthening. While the advancement of radiotherapy technology and clinical trials on neoadjuvant chemoradiotherapy in EC hold promise for improving OS, exploring radiotherapy as combined with immunotherapy may enhance our grasp of EC and facilitate the screening of genetic beneficiaries. Notably, aside from clinical research, attention should also be devoted to basic research. It is a crucial means to uncover the pathogenesis and developmental patterns of EC, forming a cornerstone for successful clinical transformation.
ACKNOWLEDGEMENTS
All authors reviewed the manuscript and approved the submitted version.
Contributor Information
Yue Feng, Department of Radiotherapy, Shuguang Hospital Affiliated to Shanghai University of Chinese Traditional Medicine, 528 Zhangheng Road, Pudong New Area, Shanghai 200135, China.
Minghe Lv, Department of Radiotherapy, Shuguang Hospital Affiliated to Shanghai University of Chinese Traditional Medicine, 528 Zhangheng Road, Pudong New Area, Shanghai 200135, China.
Su Zeng, Department of Radiotherapy, Shuguang Hospital Affiliated to Shanghai University of Chinese Traditional Medicine, 528 Zhangheng Road, Pudong New Area, Shanghai 200135, China.
Hongwei Zeng, Department of Radiotherapy, Shuguang Hospital Affiliated to Shanghai University of Chinese Traditional Medicine, 528 Zhangheng Road, Pudong New Area, Shanghai 200135, China.
Jingping Yu, Department of Radiotherapy, Shuguang Hospital Affiliated to Shanghai University of Chinese Traditional Medicine, 528 Zhangheng Road, Pudong New Area, Shanghai 200135, China.
CONFLICT OF INTEREST
All authors declared no conflict of interest.
FUNDING
This work was supported by the Scientific Research Project of Shanghai Municipal Health Commission (grant number: 202 340 160), the Science and Technology Development Project of Shanghai University of Traditional Chinese Medicine (grant number: 23KFL105), the Integrated Chinese and Western Medicine Project of Shuguang Hospital affiliated with Shanghai University of Traditional Chinese Medicine (grant number: SGZXY-202201).
AUTHOR CONTRIBUTIONS
J.Y. designed the experiments; Y.F. wrote the manuscript; M.Lv, S.Z. and H.Z. did the manuscript editing.
PRESENTATION AT A CONFERENCE
None.
CLINICAL TRIAL REGISTRATION NUMBER
Not required.
References
- 1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209–49. 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2. DiSiena M, Perelman A, Birk J, Rezaizadeh H. Esophageal cancer: an updated review. South Med J 2021;114:161–8. 10.14423/SMJ.0000000000001226. [DOI] [PubMed] [Google Scholar]
- 3. Wang B, Xing D, Zhu Y, et al. The state of exosomes research: a global visualized analysis. Biomed Res Int 2019;2019:1. Published 2019 Apr 3–10. 10.1155/2019/1495130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ke L, Lu C, Shen R, et al. Knowledge mapping of drug-induced liver injury: a scientometric investigation (2010-2019). Front Pharmacol 2020;11:842. Published 2020 Jun 5. 10.3389/fphar.2020.00842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Synnestvedt MB, Chen C, Holmes JH. CiteSpace II: visualization and knowledge discovery in bibliographic databases. AMIA Annu Symp Proc 2005;2005:724–8. [PMC free article] [PubMed] [Google Scholar]
- 6. Yeung AWK, Tzvetkov NT, Balacheva AA, et al. Lignans: quantitative analysis of the research literature. Front Pharmacol 2020;11:37. Published 7 Feb 2020. 10.3389/fphar.2020.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Li C, Ojeda-Thies C, Renz N, et al. The global state of clinical research and trends in periprosthetic joint infection: a bibliometric analysis. Int J Infect Dis 2020;96:696–709. 10.1016/j.ijid.2020.05.014. [DOI] [PubMed] [Google Scholar]
- 8. Lu C, Liu M, Shang W, et al. Knowledge mapping of Angelica sinensis (Oliv.) Diels (Danggui) research: a scientometric study. Front Pharmacol 2020;11:294. Published 2020 Mar 13. 10.3389/fphar.2020.00294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Shi S, Gao Y, Liu M, et al. Top 100 most-cited articles on exosomes in the field of cancer: a bibliometric analysis and evidence mapping. Clin Exp Med 2021;21:181–94. 10.1007/s10238-020-00624-5. [DOI] [PubMed] [Google Scholar]
- 10. Teles RHG, Yano RS, Villarinho NJ, et al. Advances in breast cancer management and extracellular vesicle research, a bibliometric analysis. Curr Oncol 2021;28:4504–20. Published 8 November 2021. 10.3390/curroncol28060382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Wu H, Cheng K, Tong L, et al. Knowledge structure and emerging trends on osteonecrosis of the femoral head: a bibliometric and visualized study. J Orthop Surg Res 2022;17:194. 10.1186/s13018-022-03068-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ihaka R, Gentleman R. R: a language for data analysis and graphics. J Comput Graph Stat 1996;5:299–314. 10.1080/10618600.1996.10474713. [DOI] [Google Scholar]
- 13. van EckNJ, Waltman L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010;84:523–38. 10.1007/s11192-009-0146-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Chen C. CiteSpace II: detecting and visualizing emerging trends and transient patterns in scientific literature. J Am Soc Inf Sci Technol 2006;57:359–77. 10.1002/asi.20317. [DOI] [Google Scholar]
- 15. Aria M, Cuccurullo C. Bibliometrix: an R-tool for comprehensive science mapping analysis. J Inf 2017;11:959–75. 10.1016/j.joi.2017.08.007. [DOI] [Google Scholar]
- 16. Jiang ST, Liu YG, Zhang L, et al. Immune-related adverse events: a bibliometric analysis. Front Immunol 2022;13:1096806. Published 15 December 2022. 10.3389/fimmu.2022.1096806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Chen C. Science mapping: a systematic review of the literature. J Data Inf Sci 2017;2:1–40. 10.1515/jdis-2017-0006. [DOI] [Google Scholar]
- 18. Lin SH, Komaki R, Liao Z, et al. Proton beam therapy and concurrent chemotherapy for esophageal cancer. Int J Radiat Oncol Biol Phys 2012;83:e345–51. 10.1016/j.ijrobp.2012.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Lin SH, Merrell KW, Shen J, et al. Multi-institutional analysis of radiation modality use and postoperative outcomes of neoadjuvant chemoradiation for esophageal cancer. Radiother Oncol 2017;123:376–81. 10.1016/j.radonc.2017.04.013. [DOI] [PubMed] [Google Scholar]
- 20. Xi M, Lin SH. Recent advances in intensity modulated radiotherapy and proton therapy for esophageal cancer. Expert Rev Anticancer Ther 2017;17:635–46. 10.1080/14737140.2017.1331130. [DOI] [PubMed] [Google Scholar]
- 21. Lin SH, Hobbs BP, Verma V, et al. Randomized phase IIB trial of proton beam therapy versus intensity-modulated radiation therapy for locally advanced Esophageal cancer. J Clin Oncol 2020;38:1569–79. 10.1200/JCO.19.02503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Cooper JS, Guo MD, Herskovic A, et al. Chemoradiotherapy of locally advanced esophageal cancer: long-term follow-up of a prospective randomized trial (RTOG 85-01). Radiation Therapy Oncology Group JAMA 1999;281:1623–7. [DOI] [PubMed] [Google Scholar]
- 23. Minsky BD, Pajak TF, Ginsberg RJ, et al. INT 0123 (radiation therapy oncology group 94-05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol 2002;20:1167–74. 10.1200/JCO.2002.20.5.1167. [DOI] [PubMed] [Google Scholar]
- 24. Van Hagen P, Hulshof MC, van LanschotJJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 2012;366:2074–84. 10.1056/NEJMoa1112088. [DOI] [PubMed] [Google Scholar]
- 25. Miao Y, Zhang Y, Yin L. Trends in hepatocellular carcinoma research from 2008 to 2017: a bibliometric analysis. PeerJ 2018;6:e5477. Published 15 August 2018. 10.7717/peerj.5477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Morgan E, Soerjomataram I, Rumgay H, et al. The global landscape of esophageal squamous cell carcinoma and esophageal adenocarcinoma incidence and mortality in 2020 and projections to 2040: new estimates from GLOBOCAN 2020. Gastroenterology 2022;163:649–658.e2. 10.1053/j.gastro.2022.05.054. [DOI] [PubMed] [Google Scholar]
- 27. Arnold M, Laversanne M, Brown LM, et al. Predicting the future burden of Esophageal cancer by histological subtype: international trends in incidence up to 2030. Am J Gastroenterol 2017;112:1247–55. 10.1038/ajg.2017.155. [DOI] [PubMed] [Google Scholar]
- 28. Rumgay H, Arnold M, Laversanne M, et al. International trends in Esophageal squamous cell carcinoma and adenocarcinoma incidence. Am J Gastroenterol 2021;116:1072–6. 10.14309/ajg.0000000000001121. [DOI] [PubMed] [Google Scholar]
- 29. Shapiro J, van LanschotJJB, Hulshof MCCM, et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol 2015;16:1090–8. 10.1016/S1470-2045(15)00040-6. [DOI] [PubMed] [Google Scholar]
- 30. Liou Y, Lan TL, Lan CC. A meta-analysis and review of radiation dose escalation in definitive radiation therapy between squamous cell carcinoma and adenocarcinoma of esophageal cancer. Cancers (Basel) 2024;16:658. 10.3390/cancers16030658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Fujita H, Sueyoshi S, Tanaka T, et al. Prospective non-randomized trial comparing esophagectomy-followed-by-chemoradiotherapy versus chemoradiotherapy-followed-by- esophagectomy for T4 esophageal cancers. J Surg Oncol 2005;90:209–19. 10.1002/jso.20259. [DOI] [PubMed] [Google Scholar]
- 32. Gebski V, Burmeister B, Smithers BM, et al. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta-analysis. Lancet Oncol 2007;8:226–34. 10.1016/S1470-2045(07)70039-6. [DOI] [PubMed] [Google Scholar]
- 33. Vallböhmer D, Hölscher AH, DeMeester S, et al. A multicenter study of survival after neoadjuvant radiotherapy/chemotherapy and esophagectomy for ypT0N0M0R0 esophageal cancer. Ann Surg 2010;252:744–9. 10.1097/SLA.0b013e3181fb8dde. [DOI] [PubMed] [Google Scholar]
- 34. van HeijlM, van LanschotJJ, Koppert LB, et al. Neoadjuvant chemoradiation followed by surgery versus surgery alone for patients with adenocarcinoma or squamous cell carcinoma of the esophagus (CROSS). BMC Surg 2008;8:21. Published 26 November 2008. 10.1186/1471-2482-8-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Hu X, He W, Wen S, et al. Is IMRT superior or inferior to 3DCRT in radiotherapy for NSCLC? A meta-analysis. PLoS One 2016;11:e0151988. Published 21 April 2016. 10.1371/journal.pone.0151988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Ling TC, Slater JM, Nookala P, et al. Analysis of intensity-modulated radiation therapy (IMRT), proton and 3D conformal radiotherapy (3D-CRT) for reducing perioperative cardiopulmonary complications in Esophageal cancer patients. Cancers (Basel) 2014;6:2356–68. Published 5 December 2014. 10.3390/cancers6042356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ghosh S, Kapoor R, Gupta R, et al. An evaluation of three dimensional conformal radiation therapy versus intensity modulated radiation therapy in radical chemoradiation of esophageal cancer: a dosimetric study. Clin Cancer Invest J 2012;1:65–70. 10.4103/2278-0513.99565. [DOI] [Google Scholar]
- 38. Chandra A, Guerrero TM, Liu HH, et al. Feasibility of using intensity-modulated radiotherapy to improve lung sparing in treatment planning for distal esophageal cancer. Radiother Oncol 2005;77:247–53. 10.1016/j.radonc.2005.10.017. [DOI] [PubMed] [Google Scholar]
- 39. Nutting CM, Bedford JL, Cosgrove VP, et al. A comparison of conformal and intensity-modulated techniques for oesophageal radiotherapy. Radiother Oncol 2001;61:157–63. 10.1016/S0167-8140(01)00438-8. [DOI] [PubMed] [Google Scholar]
- 40. Kole TP, Aghayere O, Kwah J, et al. Comparison of heart and coronary artery doses associated with intensity-modulated radiotherapy versus three-dimensional conformal radiotherapy for distal esophageal cancer. Int J Radiat Oncol Biol Phys 2012;83:1580–6. 10.1016/j.ijrobp.2011.10.053. [DOI] [PubMed] [Google Scholar]
- 41. Wu VW, Sham JS, Kwong DL. Inverse planning in three-dimensional conformal and intensity-modulated radiotherapy of mid-thoracic oesophageal cancer. Br J Radiol 2004;77:568–72. 10.1259/bjr/19972578. [DOI] [PubMed] [Google Scholar]
- 42. Fakhrian K, Oechsner M, Kampfer S, et al. Advanced techniques in neoadjuvant radiotherapy allow dose escalation without increased dose to the organs at risk: planning study in esophageal carcinoma. Strahlenther Onkol 2013;189:293–300. 10.1007/s00066-012-0297-7. [DOI] [PubMed] [Google Scholar]
- 43. Zhang WZ, Chen JZ, Li DR, et al. Simultaneous modulated accelerated radiation therapy for esophageal cancer: a feasibility study. World J Gastroenterol 2014;20:13973–80. 10.3748/wjg.v20.i38.13973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Wu Z, Xie C, Hu M, et al. Dosimetric benefits of IMRT and VMAT in the treatment of middle thoracic esophageal cancer: is the conformal radiotherapy still an alternative option? J Appl Clin Med Phys 2014;15:93–101. Published 8 May 2014. 10.1120/jacmp.v15i3.4641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Kumar G, Rawat S, Puri A, et al. Analysis of dose-volume parameters predicting radiation pneumonitis in patients with esophageal cancer treated with 3D-conformal radiation therapy or IMRT. Jpn J Radiol 2012;30:18–24. 10.1007/s11604-011-0002-2. [DOI] [PubMed] [Google Scholar]
- 46. Ishikura S, Nihei K, Ohtsu A, et al. Long-term toxicity after definitive chemoradiotherapy for squamous cell carcinoma of the thoracic esophagus. J Clin Oncol 2003;21:2697–702. 10.1200/JCO.2003.03.055. [DOI] [PubMed] [Google Scholar]
- 47. Lyu J, Abulimiti Y, Li T, et al. Involved field irradiation (IFI) versus elective nodal irradiation (ENI) in combination with concurrent chemotherapy for esophageal thoracic squamous cell cancer: a prospective, randomized, multicenter, controlled study. Chin J Radiat Oncol 2018;27:245–9. [Google Scholar]
- 48. Zhang J, Li M, Zhang K, et al. Concurrent chemoradiation of different doses (50.4 Gy vs. 59.4 Gy) and different target field (ENI vs. IFI) for locally advanced esophageal squamous cell carcinoma:results from a randomized, multicenter phase III clinical trial. Int J Radiat Oncol 2022;114:S15. 10.1016/j.ijrobp.2022.07.357. [DOI] [Google Scholar]
- 49. Wang H, Song C, Zhao X, et al. The role of involved field irradiation versus elective nodal irradiation in definitive radiotherapy or chemoradiotherapy for esophageal cancer- a systematic review and meta-analysis. Front Oncol 2022;12:1034656. Published 2 November 2022. 10.3389/fonc.2022.1034656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Cheng YJ, Jing SW, Zhu LL, et al. Comparison of elective nodal irradiation and involved-field irradiation in esophageal squamous cell carcinoma: a meta-analysis. J Radiat Res 2018;59:604–15. 10.1093/jrr/rry055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Kudo T, Hamamoto Y, Kato K, et al. Nivolumab treatment for oesophageal squamous-cell carcinoma: an open-label, multicentre, phase 2 trial. Lancet Oncol 2017;18:631–9. 10.1016/S1470-2045(17)30181-X. [DOI] [PubMed] [Google Scholar]
- 52. Li C, Zhao S, Zheng Y, et al. Preoperative pembrolizumab combined with chemoradiotherapy for oesophageal squamous cell carcinoma (PALACE-1). Eur J Cancer 2021;144:232–41. 10.1016/j.ejca.2020.11.039. [DOI] [PubMed] [Google Scholar]
- 53. Kelly RJ, Ajani JA, Kuzdzal J, et al. Adjuvant Nivolumab in resected Esophageal or gastroesophageal junction cancer. [published correction appears in N Engl J Med. 2023 Feb 16;388(7):672]. N Engl J Med 2021;384:1191–203. 10.1056/NEJMoa2032125. [DOI] [PubMed] [Google Scholar]
- 54. Xin W, Chen X, Kang X, et al. Neoadjuvant multimodality RX including immunotherapy for highly selective unresectable locally advanced esophageal squamous cell carcinoma (NEXUS): a prospective, single-arm, phase II trial[EB/OL]. ESMO IO. Abstract 2022;92. [Google Scholar]