Abstract
Background
Sodium–Glucose Cotransporter-2 Inhibitor (SGLT2i) is a novel oral drug for treating type 2 diabetes mellitus (T2DM) with demonstrated cardiovascular benefits. Previous studies in apolipoprotein E knockout mice have shown that SGLT2i is associated with attenuated progression of atherosclerosis. However, whether this effect extends to T2DM patients with coronary atherosclerosis in real-world settings remains unknown.
Methods
In this longitudinal cohort study using coronary computed tomography angiography (CCTA), T2DM patients who underwent ≥ 2 CCTA examinations at our center between 2019 and 2022 were screened. Eligible patients had multiple study plaques, defined as non-obstructive stenosis at baseline and not intervened during serial CCTAs. Exclusion criteria included a CCTA time interval < 12 months, prior SGLT2i treatment, or initiation/discontinuation of SGLT2i during serial CCTAs. Plaque volume (PV) and percent atheroma volume (PAV) were measured for each study plaque using CCTA plaque analysis software. Patients and plaques were categorized based on SGLT2i therapy and compared using a 1:1 propensity score matching (PSM) analysis.
Results
The study included 236 patients (mean age 60.5 ± 9.5 years; 69.1% male) with 435 study plaques (diameter stenosis ≥ 50%, 31.7%). Following SGLT2i treatment for a median duration of 14.6 (interquartile range: 13.0, 20.0) months, overall, non-calcified, and low-attenuation PV and PAV were significantly decreased, while calcified PV and PAV were increased (all p < 0.001). Meanwhile, reductions in overall PV, non-calcified PV, overall PAV, and non-calcified PAV were significantly greater in SGLT2i-treated compared to non-SGLT2i-treated plaques (all p < 0.001). PSM analysis showed that SGLT2i treatment was associated with higher reductions in overall PV (− 11.77 mm3 vs. 4.33 mm3, p = 0.005), non-calcified PV (− 16.96 mm3 vs. − 1.81 mm3, p = 0.017), overall PAV (− 2.83% vs. 3.36%, p < 0.001), and non-calcified PAV (− 4.60% vs. 0.70%, p = 0.003). These findings remained consistent when assessing annual changes in overall and compositional PV and PAV. Multivariate regression models demonstrated that SGLT2i therapy was associated with attenuated progression of overall or non-calcified PV or PAV, even after adjusting for cardiovascular risk factors, medications, and baseline overall or non-calcified PV or PAV, respectively (all p < 0.05). The effect of SGLT2i on attenuating non-calcified plaque progression was consistent across subgroups (all p for interaction > 0.05).
Conclusions
In this longitudinal CCTA cohort of T2DM patients, SGLT2i therapy markedly regressed coronary overall PV and PAV, mainly result from a significant reduction in non-calcified plaque.
Graphical abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s12933-024-02368-y.
Keywords: Atherosclerotic plaque, Type 2 diabetes mellitus, Coronary computed tomography angiography, Sodium-Glucose Cotransporter-2 Inhibitor, Longitudinal study
Introduction
According to the International Diabetes Federation, approximately 537 million individuals worldwide were affected by type 2 diabetes mellitus (T2DM) in 2021, with this number expected to rise to 783 million by 2045 [1]. Patients with T2DM develop atherosclerosis at a younger age and progress more rapidly compared to those without T2DM [2], thus increasing their risk of atherosclerotic cardiovascular disease (ASCVD) [3, 4]. While improved glycemic control with traditional glucose-lowering drugs has shown benefits in patients with newly diagnosed T2DM, these strategies are less effective in T2DM patients with established ASCVD, who face an elevated risk of premature cardiovascular events [5].
Sodium-Glucose Cotransporter-2 Inhibitor (SGLT2i) is a novel oral hypoglycemic agent for the treatment of T2DM, which reduces glucose reabsorption by inhibiting SGLT2 in the renal proximal tubules [6]. The cardiovascular benefits of SGLT2i have been examined in six cardiovascular outcome trials (CVOTs) among T2DM patients [7–12]. A recent meta-analysis of these trials has demonstrated a significant reduction in the composite of cardiovascular death, myocardial infarction (MI), or stroke, particularly among patients with ASCVD [13]. Therefore, international guidelines recommend SGLT2i for patients with T2DM and ASCVD to reduce cardiovascular events, irrespective of glycosylated hemoglobin A1c levels and concomitant antidiabetic medications [14]. However, the ASCVD-based benefits of SGLT2i in T2DM patients remain unclear.
The pathological basis of ASCVD lies in the initiation and development of atherosclerotic plaque, which traditionally leads to MI or ischemic stroke. Preclinical studies have shown that SGLT2i can attenuate the progression of aortic plaque in apolipoprotein E knockout (ApoE−/−) mice [15, 16]. Additionally, SGLT2i therapy has been effective in stabilizing atherosclerotic plaque in a tandem stenosis ApoE−/− mouse model [17]. Consistent with these findings, compared to non-SGLT2i treatment, SGLT2i was associated with a 9% reduction in MI in the aforementioned meta-analysis (95% confidence intervals [CI], 0.84–0.99) [13].
Therefore, the cardiovascular outcome benefits of SGLT2i may be attributed to the attenuation of plaque progression. However, this hypothesis has not been investigated among T2DM patients with coronary atherosclerosis in real-world settings. Importantly, the modification of coronary plaque response to medication can be accurately traced using high-resolution coronary computed tomography angiography (CCTA) and artificial intelligence-aided post-processing software [18]. Hence, we performed a longitudinal CCTA cohort study among T2DM patients with coronary atherosclerosis to evaluate the effects of SGLT2i on the progression of coronary atherosclerosis.
Methods
Study design and population
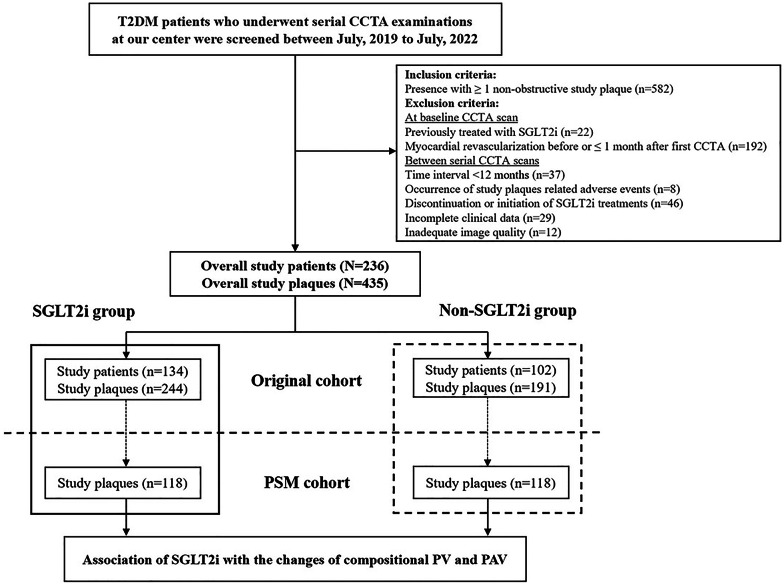
Patients diagnosed with T2DM who underwent at least two clinically indicated CCTA examinations at Beijing Anzhen Hospital, Capital Medical University, were screened between July 2019 to July 2022. Eligible T2DM patients had more than one study plaque, defined as non-obstructive plaque in one of the major coronary arteries. Exclusion criteria included: (1) Patients previously treated with SGLT2i before enrollment; (2) Patients who underwent myocardial revascularization before or within 1 month after the first CCTA; (3) Patients with a time interval between serial CCTA scans of less than 12 months(4) Patients with study plaque-related adverse events; (5) Patients who discontinued or initiated SGLT2i treatments during serial CCTA scans (6) Patients with incomplete clinical data and inadequate image quality. Study participants and plaques were categorized based on initiation of SGLT2i therapy within 1 month after the first CCTA scan. After propensity score matching (PSM), plaque progression was compared between SGLT2i and non-SGLT2i groups (Fig. 1). SGLT2i group received treatment of Dapagliflozin (5 mg daily), Empagliflozin (10 mg daily) or Canagliflozin (100 mg daily) at the first CCTA and follow-up. The study protocol was in accordance with the Declaration of Helsinki and was approved by the Medical Ethics Committee of Beijing Anzhen Hospital, with informed consent obtained from all participants.
Fig. 1.
Flowchart of study process. CCTA coronary computed tomographic angiography, PSM propensity score matching, PV plaque volume, PAV percent atheroma volume, SGLT2i Sodium-Glucose Cotransporter-2 Inhibitor, T2DM type 2 diabetes mellitus
Data collection and follow-up
Demographic information, risk factors, medical histories, laboratory results, CCTA scan parameters, and medications were retrieved from electronic medical records. Adverse events and medications during longitudinal CCTA scans were monitored through outpatient or telephone interviews. Study plaque-related adverse events were defined as cardiac death, MI or revascularization during series CCTA follow-ups attributed to the study plaques.
CCTA acquisition
All CCTA examinations were conducted using a 256-slice CT scanner (Revolution CT, GE Healthcare, USA) following the guidelines of the Society of Cardiovascular Computed Tomography [19]. A bolus of 40–60 mL of contrast media (Ultravist, Bayer) at a concentration of 370 mg iodine/mL was injected into the antecubital vein at a rate of 4–5 mL/s, followed by 30 mL of saline. CT scan parameters included a reconstructed layer thickness of 0.625 mm, a gantry rotation time of 0.28 s, as well as a tube voltage of 100 or 120 kV. The tube current was automatically adjusted using Smart-mA technology. Identical acquisition parameters were maintained for each patient at both baseline and follow-up CCTA scans.
CCTA quantitative analysis
Anonymous CCTA datasets were transferred to an offline workstation for image quantitative analysis using semi-automated coronary plaque analysis software (Circle Cardiovascular Imaging, Canada, Version 5.13) with manual correction. Computed tomography-derived fractional flow reserve related to study plaque was calculated using an artificial intelligence-based automated analysis software (Shukun Technology, Beijing) [20]. CCTA images were analyzed by independent level-III experts who were blinded to the presence or absence of SGLT2i therapy and the order of serial CCTA scans.
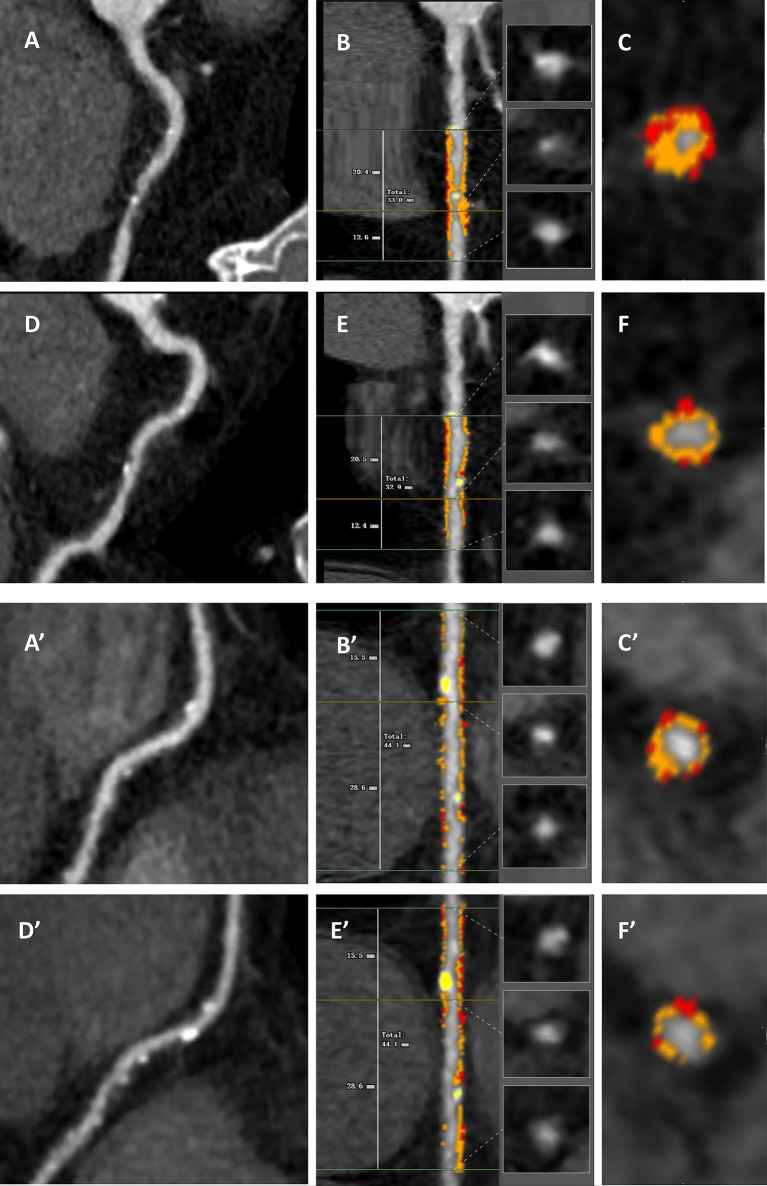
Segments of major coronary arteries with a diameter ≥ 2 mm were evaluated based on a modified 17-segment model. The presence of atherosclerotic plaque was defined as any tissue exceeding 1 mm3 within or close to the lumen, which could be discriminated from surrounding structures and identified in more than 2 consecutive planes. Study plaques were confined to those with a diameter stenosis < 70% (i.e., non-obstructive) and not intervened. The 3D quantitative parameters included overall and compositional plaque volume (PV) and percent atheroma volume (PAV). Plaque composition was analyzed using the following Hounsfield unit (HU) thresholds: calcified (≥ 350 HU), non-calcified (< 350 HU), and low-attenuation (< 30 HU) [21–23]. For the longitudinal analysis of changes in PV and PAV between serial CCTA scans, coronary plaques were co-registered using consistent landmarks (e.g., distance from the ostium or the branch vessels). Representative serial cases of T2DM patients treated with or without SGLT2i were shown in Fig. 2.
Fig. 2.
Representative patients treated with or without SGLT2i underwent longitudinal assessment of compositional plaque volume. A–F A 72 years old male with hypertension and hyperlipidemia (BMI 24.5 kg/m2, LDL-C 1.35 mmol/L, FPG 5.75 mmol/L) treated with SGLT2i, moderate intensity statin and ezetimibe. His baseline overall PV in the proximal right coronary artery (A) was 277.60 mm3 with non-calcified PV 276.06 mm3, low-attenuated PV 51.63 mm3 and calcified PV 1.54 mm3 (B, C). After 12.3 months, the overall PV in the proximal right coronary artery (D) decreased to 234.64 mm3 with non-calcified PV 232.86 mm3, low-attenuated PV 39.84 mm3 and calcified PV 1.78 mm3 (E, F). A’–F’ A 67 years old male with hypertension and hyperlipidemia (BMI 26.1 kg/m2, LDL-C 1.40 mmol/L, FPG 6.23 mmol/L) treated with moderate intensity statin and ezetimibe but without SGLT2i. His baseline overall PV in the middle right coronary artery (A’) was 185.27 mm3 with non-calcified PV 181.21 mm3, low-attenuated PV 34.78 mm3 and calcified PV 4.06 mm3 (B’, C’). After 15.7 months, the overall PV in the middle right coronary artery (D’) was 185.65 mm3 with non-calcified PV 178.21 mm3, low-attenuated PV 49.26 mm3 and calcified PV 7.44 mm3 (E’, F’). The orange, yellow and red overlays represent non-calcified, calcified and low-attenuated PV, espectively
To assess inter- and intra-observer variabilities, a second level-III reader re-analyzed 8 randomly selected plaques, and the original level-III reader re-analyzed 12 randomly selected plaques 3 months after the first analysis.
Statistical analysis
Data were presented for the SGLT2i and non-SGLT2i groups on a per-patient level and a per-plaque level, respectively. Continuous variables were expressed as the mean ± standard deviation (SD) or medians with interquartile ranges (IQR), and compared between groups using the paired or unpaired Student’s t-test or Mann–Whitney U-test. Categorical variables were presented as frequencies (percentages) and compared between groups using the chi-square test or Fisher’s exact test.
To balance the differences in baseline characteristics between groups, a pre-specified 1:1 nearest-neighbor PSM analysis was performed on a per-plaque level using the following baseline variables: unbalanced variables between groups or variables might interfere with the clinical allocation of SGLT2i (blood pressure, fasting plasma glucose [FPG], stroke, CCTA time interval and medication), or well-known accelerators of atherosclerotic plaque (age, sex, blood pressure, FPG, high-sensitivity C-reactive protein [CRP], low-density lipoprotein cholesterol [LDL-C]). To account for variations in body habitus and CCTA time interval among study plaques, PAV (percentage of PV divided by vessel volume at each plaque) and annual changes of PV and PAV (changes of PV and PAV divided by CCTA interval years) were calculated, respectively. To further determine the association of SGLT2i with progression of compositional PV and PAV, multivariate regression analysis was performed. In the linear regression analysis, compositional PV and PAV were modeled as continual dependent variables, and results were shown as β and 95%CI. Meanwhile, in the logistic regression analysis, compositional PV and PAV were modeled as categorized dependent variables, and results were shown as odds ratios (ORs) and 95%CI. Independent variables with a p-value < 0.1 in the univariate analysis and known accelerators of atherosclerosis were included in the multivariate regression analysis.
Subgroup analysis was carried out to confirm whether the association of SGLT2i with PV or PAV progression was consistent across all pre-specified subgroups. Statistical analyses were conducted using SPSS 25.0 (IBM Corporation, IL, USA) as well as R Programming Language 4.2.2 (Vienna, Austria). Statistical significance was determined by p < 0.05 (two-tailed).
Results
Characteristics of study patients and plaques
In our initial analysis, 236 patients with T2DM were included, with a mean age of 60.5 ± 9.5 years, and 163 (69.1%) of them were male (Table 1). Among these patients, 68.2% had established ASCVD, while hypertension (73.7%), dyslipidemia (86.0%), and poor glucose control (median FPG of 7.5 mmol/L) were prevalent. Notably, patients treated with SGLT2i were more prone to have higher FPG levels and diastolic blood pressure (both p < 0.05) (Table 1). A total of 435 study plaques were included, with 31.7% exhibiting diameter stenosis ≥ 50%, and 25.1% having computed tomography-derived fractional flow reserve ≤ 0.80 (Table S1).
Table 1.
Baseline characteristics of the overall study patients
| Total n = 236 |
SGLT2i n = 134 |
Non-SGLT2i n = 102 |
p value | |
|---|---|---|---|---|
| Clinical characteristics | ||||
| Age, years | 60.5 ± 9.5 | 60.1 ± 10.3 | 61.1 ± 8.4 | 0.402 |
| Male, n (%) | 163 (69.1) | 95 (70.9) | 68 (66.7) | 0.486 |
| BMI, kg/m2 | 26.0 (24.1, 28.7) | 25.7 (24.0, 28.1) | 26.2 (24.2, 28.8) | 0.462 |
| SBP, mmHg | 131.5 (120.0, 140.0) | 131.7 (118.0, 140.0) | 131.5 (122.8, 141.0) | 0.365 |
| DBP, mmHg | 76.0 (68.0, 84.0) | 77.5 (69.0, 85.0) | 74.0 (65.0, 81.3) | 0.033 |
| Risk factors, n (%) | ||||
| Hypertension | 174 (73.7) | 97 (72.4) | 77 (75.5) | 0.592 |
| Dyslipidemia | 203 (86.0) | 116 (86.6) | 87 (85.3) | 0.780 |
| Current smoking | 73 (30.9) | 45 (33.6) | 28 (27.5) | 0.313 |
| Medical histories, n (%) | ||||
| Myocardial infarction | 26 (11.0) | 13 (9.7) | 13 (12.7) | 0.459 |
| Myocardial revascularization | 52 (22.0) | 32 (23.9) | 20 (19.6) | 0.433 |
| Stroke | 33 (14.0) | 16 (11.9) | 17 (16.7) | 0.300 |
| Laboratory results | ||||
| FPG, mmol/L | 7.5 (6.3, 9.2) | 7.7 (6.6, 9.7) | 7.1 (5.6, 8.6) | 0.009 |
| Creatinine, mmol/L | 72.7 (63.5, 86.7) | 72.9 (64.5, 87.0) | 72.6 (62.6, 86.0) | 0.771 |
| eGFR, mL/min/1.73 m2 | 91.7 (78.9, 101.0) | 91.4 (80.9, 101.5) | 92.0 (76.8, 100.3) | 0.799 |
| TG, mmol/L | 1.48 (1.09, 2.09) | 1.48 (1.17, 2.10) | 1.50 (1.06, 2.08) | 0.421 |
| TC, mmol/L | 3.98 (3.41, 4.68) | 3.93 (3.44, 4.68) | 4.00 (3.38, 4.90) | 0.646 |
| HDL-C, mmol/L | 1.01 (0.86, 1.19) | 1.01 (0.86, 1.18) | 1.02 (0.87, 1.24) | 0.535 |
| LDL-C, mmol/L | 2.15 (1.67, 2.77) | 2.14 (1.68, 2.76) | 2.21 (1.66, 2.78) | 0.758 |
| hs-CRP, mg/L | 1.15 (0.60, 2.51) | 1.21 (0.59, 3.21) | 1.15 (0.60, 2.32) | 0.850 |
| Serial CCTAs | ||||
| Time interval, month | 14.6 (13.0, 20.0) | 14.1 (12.9, 17.4) | 15.9 (13.2, 23.0) | 0.030 |
| Baseline tube voltage, n (%) | 0.840 | |||
| 100 kV | 159 (67.4) | 91 (67.9) | 68 (66.7) | |
| 120 kV | 77 (32.6) | 43 (32.1) | 34 (33.3) | |
| Number of study plaque, per Patient | 435 (1.84) | 244 (1.82) | 191 (1.87) | - |
| Medications, n (%) | ||||
| Metformin | 95 (40.3) | 47 (35.1) | 48 (47.1) | 0.063 |
| Incretins | 49 (20.8) | 31 (23.1) | 18 (17.6) | 0.303 |
| Insulin | 48 (20.3) | 26 (19.4) | 22 (21.6) | 0.682 |
| Statins | 236 (100) | 134 (100) | 102 (100) | 1.000 |
| Ezetimibe | 61 (25.8) | 37 (27.6) | 24 (23.5) | 0.478 |
BMI body mass index, CCTA coronary computed tomographic angiography, DBP diastolic blood pressure, eGFR estimated glomerular filtration rate, FPG fasting plasma glucose, HDL-C High density lipoprotein cholesterol, hs-CRP hypersensitive C-reactive protein, LM Left main artery, LAD left anterior descending artery, LCX left circumflex coronary artery, LDL-C low density lipoprotein cholesterol, RCA right coronary artery, SGLT2i Sodium-Glucose Cotransporter-2 Inhibitor, SBP systolic blood pressure, TG Triglyceride, TC Total cholesterol
Temporal changes of overall and compositional PV and PAV
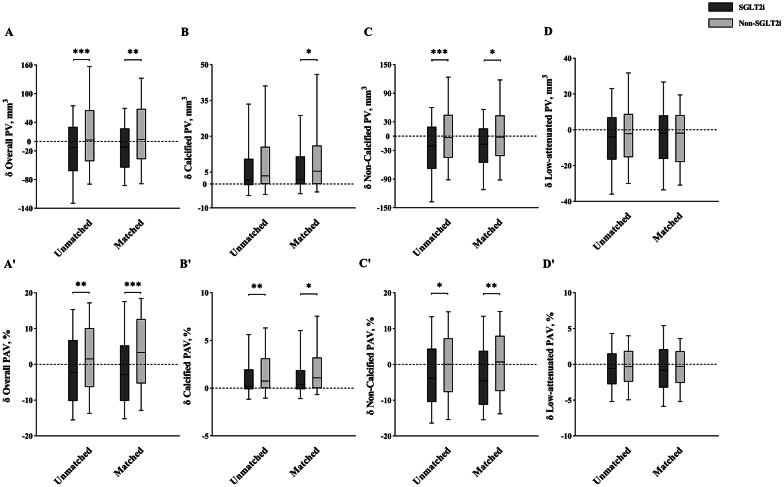
In our original analysis, after treatment with SGLT2i for a median duration of 14.6 (IQR: 13.0, 20.0) months, overall, non-calcified, and low-attenuation PV and PAV were significantly reduced, while calcified PV and PAV increased (all p < 0.001). However, these favorable plaque remodeling outcomes from SGLT2i did not translate into reductions in plaque-related anatomic and hemodynamic stenosis (all p > 0.05) (Table 2). Importantly, decreased overall PV (− 12.71 mm3 vs. 2.92 mm3, p < 0.001), non-calcified PV (− 20.01 mm3 vs. − 2.78 mm3, p < 0.001), overall PAV (− 2.25% vs. 1.52%, p = 0.003), and non-calcified PAV (− 3.88% vs. 0.04%, p = 0.010) were notably higher in SGLT2i-treated than non-SGLT2i-treated plaques (Table 2; Fig. 3).
Table 2.
Baseline and follow-up CCTA findings on a per-plaque level
| Unmatched | Matched | |||||
|---|---|---|---|---|---|---|
| SGLT2i | Non-SGLT2i | p value | SGLT2i | Non-SGLT2i | p value | |
| (n = 244) | (n = 191) | (n = 118) | (n = 118) | |||
| Plaque volume, mm3 | ||||||
| Overall | ||||||
| Baseline | 221.69 (146.11, 328.02) | 221.73 (154.99, 308.26) | 0.958 | 220.50 (144.55, 292.13) | 217.36 (161.95, 300.27) | 0.894 |
| Follow-up | 215.06 (136.64, 301.24) | 230.67(154.69, 335.58) | 0.310 | 210.56 (138.72, 276.58) | 218.49(156.28, 317.15) | 0.129 |
| Change from baseline | − 12.71 (− 62.16, 30.40) | 2.92 (− 41.45, 65.18) | < 0.001 | − 11.77 (− 54.58, 27.03) | 4.33 (− 37.06, 68.07) | 0.005 |
| p value | < 0.001 | 0.073 | 0.018 | 0.040 | ||
| Calcified | ||||||
| Baseline | 5.94 (0.47, 28.16) | 11.62 (1.94, 39.26) | 0.011 | 6.02 (0.52, 21.71) | 13.23 (2.26, 39.84) | 0.013 |
| Follow-up | 9.98 (1.11, 34.36) | 18.31 (3.30, 51.62) | 0.006 | 11.75 (1.66, 24.85) | 19.30 (3.72, 52.98) | 0.011 |
| Change from baseline | 1.75 (− 0.45, 10.58) | 3.45 (0.00, 15.65) | 0.055 | 1.96 (− 0.11, 11.54) | 5.38 (0.00, 16.15) | 0.033 |
| p value | < 0.001 | < 0.001 | < 0.001 | < 0.001 | ||
| Non-calcified | ||||||
| Baseline | 202.92 (136.54, 280.27) | 194.31 (128.35, 281.72) | 0.533 | 198.17 (141.49, 263.09) | 188.92 (127.93, 256.09) | 0.447 |
| Follow-up | 177.26 (119.71, 264.13) | 186.56 (127.68, 278.02) | 0.082 | 175.84 (120.85, 242.35) | 178.57 (126.01, 261.16) | 0.610 |
| Change from baseline | − 20.01 (− 68.31, 19.68) | − 2.78 (− 45.05, 44.60) | < 0.001 | − 16.96 (− 55.51, 16.04) | − 1.81 (− 41.26, 43.27) | 0.017 |
| p value | <0.001 | 0.893 | 0.001 | 0.636 | ||
| Low-attenuated | ||||||
| Baseline | 29.86 (19.26, 51.73) | 30.56 (19.32, 50.68) | 0.832 | 27.51 (18.82, 48.47) | 30.49 (19.43, 49.80) | 0.739 |
| Follow-up | 26.76 (16.67, 42.79) | 27.21 (17.27, 48.03) | 0.602 | 27.04 (17.26, 38.89) | 25.21 (17.77, 41.33) | 0.757 |
| Change from baseline | − 4.05 (− 16.66, 6.99) | − 2.14 (− 15.27, 8.92) | 0.188 | − 1.92 (− 16.33, 8.04) | − 1.82 (− 17.91, 8.18) | 0.977 |
| p value | <0.001 | 0.154 | 0.162 | 0.168 | ||
| Percent atheroma volume, % | ||||||
| Overall | ||||||
| Baseline | 43.68 (35.71, 53.12) | 40.20 (32.08, 50.25) | 0.030 | 44.13 (37.14, 52.84) | 40.01 (32.01, 52.57) | 0.086 |
| Follow-up | 42.25 (34.64, 51.40) | 42.98 (34.98, 52.04) | 0.782 | 43.47 (35.25, 50.96) | 44.21 (36.53, 54.27) | 0.202 |
| Change from baseline | − 2.25 (− 10.23, 6.77) | 1.52 (− 6.31, 10.15) | 0.003 | − 2.83 (− 10.17, 5.27) | 3.36 (− 5.31, 12.65) | < 0.001 |
| p value | 0.047 | 0.028 | 0.043 | 0.003 | ||
| Calcified | ||||||
| Baseline | 1.10 (0.09, 4.51) | 2.15 (0.37, 6.15) | 0.010 | 1.17 (0.09, 3.80) | 2.15 (0.47, 6.58) | 0.020 |
| Follow-up | 1.96 (0.19, 6.52) | 3.57 (0.70, 8.09) | 0.008 | 2.25 (0.36, 5.14) | 3.62 (0.81, 9.46) | 0.018 |
| Change from baseline | 0.21 (− 0.11, 1.96) | 0.74 (0.00, 3.12) | 0.003 | 0.41 (− 0.10, 1.88) | 1.08 (0.00, 3.20) | 0.018 |
| p value | < 0.001 | < 0.001 | < 0.001 | < 0.001 | ||
| Non-calcified | ||||||
| Baseline | 39.06 (30.55, 49.92) | 36.18 (27.23, 46.87) | 0.025 | 39.97 (33.02, 48.86) | 35.81 (25.61, 47.63) | 0.024 |
| Follow-up | 37.75 (27.58, 47.56) | 36.09 (27.85, 45.41) | 0.546 | 38.31 (27.65, 47.30) | 37.16 (28.79, 47.04) | 0.973 |
| Change from baseline | − 3.88 (− 10.48, 4.38) | 0.04 (− 7.73, 7.28) | 0.010 | − 4.60, (− 11.24, 3.78) | 0.70 (− 7.42, 7.98) | 0.003 |
| p value | < 0.001 | 0.987 | 0.002 | 0.345 | ||
| Low-attenuated | ||||||
| Baseline | 5.93 (3.93, 9.17) | 5.54 (3.56, 8.92) | 0.355 | 5.89 (3.94, 9.51) | 5.98 (3.60, 9.52) | 0.613 |
| Follow-up | 5.36 (3.41, 8.80) | 5.66 (3.22, 8.70) | 0.937 | 5.40 (3.44, 9.30) | 5.94 (3.29, 9.00) | 0.829 |
| Change from baseline | − 0.60 (− 2.77, 1.50) | − 0.33 (− 2.43, 1.86) | 0.373 | − 0.81 (− 3.24, 2.11) | − 0.30 (− 2.58, 1.82) | 0.467 |
| p value | 0.009 | 0.252 | 0.050 | 0.271 | ||
| Diameter stenosis, % | ||||||
| Baseline | 41.8 (28.1, 55.3) | 37.9 (23.1, 52.4) | 0.041 | 41.6 (26.0, 54.6) | 38.9 (25.4, 54.7) | 0.716 |
| Follow-up | 39.2 (26.0, 51.6) | 36.2 (25.5, 53.0) | 0.501 | 40.0 (27.8, 53.6) | 37.2 (27.1, 55.1) | 0.526 |
| Changes from baseline | − 1.5 (− 12.3, 9.0) | 0.2 (− 11.6, 10.5) | 0.330 | 0.8 (− 11.8, 9.1) | 0.9 (− 10.6, 10.7) | 0.870 |
| p value | 0.121 | 0.839 | 0.972 | 0.950 | ||
| FFR-CT | ||||||
| Baseline | 0.89 (0.81, 0.95) | 0.88 (0.80, 0.93) | 0.493 | 0.89 (0.82, 0.94) | 0.87 (0.79, 0.93) | 0.651 |
| Follow-up | 0.89 (0.81, 0.94) | 0.89 (0.80, 0.94) | 0.918 | 0.88 (0.81, 0.94) | 0.87 (0.80, 0.93) | 0.950 |
| Changes from baseline | 0.00 (− 0.04, 0.03) | 0.00 (− 0.03, 0.03) | 0.738 | 0.00 (− 0.04, 0.03) | 0.00 (− 0.03, 0.03) | 0.837 |
| p value | 0.419 | 0.779 | 0.459 | 0.590 | ||
FFR-CT, computed tomography-derived fractional flow reserve, Other abbreviations shown in Table 1
Fig. 3.
Temporal changes in compositional PV and PAV. *p < 0.05, **p < 0.01, ***p < 0.001. PV, plaque volume, PAV, percent atheroma volume
The inter-observer variability of overall PV and PAV was 0.95 and 0.94, respectively, with corresponding intra-observer variability being 0.96 for both.
In the PSM analysis, baseline clinical and plaque characteristics were well-balanced between SGLT2i and non-SGLT2i groups (Table 2, Table S1). Similarly, compared to non-SGLT2i-treated plaques, those treated with SGLT2i showed a greater decrease in overall PV (− 11.77 mm3 vs. 4.33 mm3, p = 0.005), non-calcified PV (− 16.96 mm3 vs. − 1.81 mm3, p = 0.017), overall PAV (− 2.83% vs. 3.36%, p < 0.001), and non-calcified PAV (− 4.60% vs. 0.70%, p = 0.003), along with a lower increase in calcified PV (1.96 mm3 vs. 5.38 mm3, p = 0.033) and calcified PAV (0.41% vs. 1.08%, p = 0.018) (Table 2; Fig. 3).
Annual changes of overall and compositional PV and PAV
When adjusting for differences in CCTA time interval, patients treated with SGLT2i consistently exhibited a higher decrease in overall PV (− 13.13 mm3/year vs. 2.28 mm3/year, p < 0.001), non-calcified PV (− 19.05 mm3/year vs. − 1.13 mm3/year, p < 0.001), overall PAV (− 2.38%/year vs. 1.26%/year, p = 0.002), and non-calcified PAV (− 3.47%/year vs. 0.02%/year, p = 0.002), while showing a lower increase in calcified PV (1.38 mm3/year vs. 2.47 mm3/year, p = 0.055) and calcified PAV (0.19%/year vs. 0.65%/year, p = 0.050). Importantly, these effects of SGLT2i on compositional PV and PAV remained unchanged in the PSM analysis (Table S2, Figure S1).
Associations of SGLT2i with progression of overall and compositional PV and PAV
In a multivariate linear regression model, SGLT2i was negatively associated with overall (β = − 42.19, 95% CI − 63.13 to − 21.24; p < 0.001) and non-calcified (β = − 37.82, 95% CI − 57.85 to − 17.78; p < 0.001) PV progression, independent of age, sex, time interval, body mass index, diastolic blood pressure, current smoker status, LDL-C, high-sensitivity CRP, FPG, Metformin usage, Incretins, and baseline overall or non-calcified PV. When progression of overall or non-calcified PV was modeled as a binary variable, the negative effect of SGLT2i on the improvement of overall or non-calcified PV persisted (all p < 0.01). Additionally, SGLT2i improved the progression of overall and non-calcified PAV, whether PAV was modeled as a continuous or binary variable (all p < 0.05) (Table 3).
Table 3.
Associations of SGLT2i with progression of compositional PV and PAV
| Adjusted variables* | Continuous plaque progression | Binary plaque progression | |||
|---|---|---|---|---|---|
| β (95%CI) | p value | OR (95%CI) | p value | ||
| Compositional PV | |||||
| Overall | + Baseline overall PV | − 42.19 (− 63.13, − 21.24) | < 0.001 | 0.47 (0.31, 0.72) | 0.001 |
| Calcified | + Baseline calcified PV | − 2.62 (− 8.28, 3.04) | 0.364 | 0.66 (0.43, 1.03) | 0.067 |
| Non-calcified | + Baseline non-calcified PV | − 37.82 (− 57.85, − 17.78) | < 0.001 | 0.51 (0.33, 0.78) | 0.002 |
| Low-attenuated | + Baseline low-attenuated PV | − 5.39 (− 12.17, 1.40) | 0.119 | 0.89 (0.58, 1.35) | 0.572 |
| Compositional PAV | |||||
| Overall | + Baseline overall PAV | − 3.19 (− 5.37, − 1.01) | 0.004 | 0.45 (0.28, 0.72) | 0.001 |
| Calcified | + Baseline calcified PAV | − 0.60 (− 1.33, 0.13) | 0.107 | 0.55 (0.36, 0.86) | 0.008 |
| Non-calcified | + Baseline non-calcified PAV | − 2.57 (− 4.74, − 0.40) | 0.020 | 0.51 (0.32, 0.80) | 0.004 |
| Low-attenuated | + Baseline low-attenuated PAV | − 0.26 (− 1.01, 0.48) | 0.488 | 0.82 (0.53, 1.26) | 0.358 |
Variables in the basic model consisted of age, sex, time interval, BMI, DBP, current smoker, LDL-C, hs-CRP, FPG, Metformin, Incretins and SGLT2i
*Multivariate regression model adjusted for basic model + baseline compositional PV or PAV
Subgroup analysis
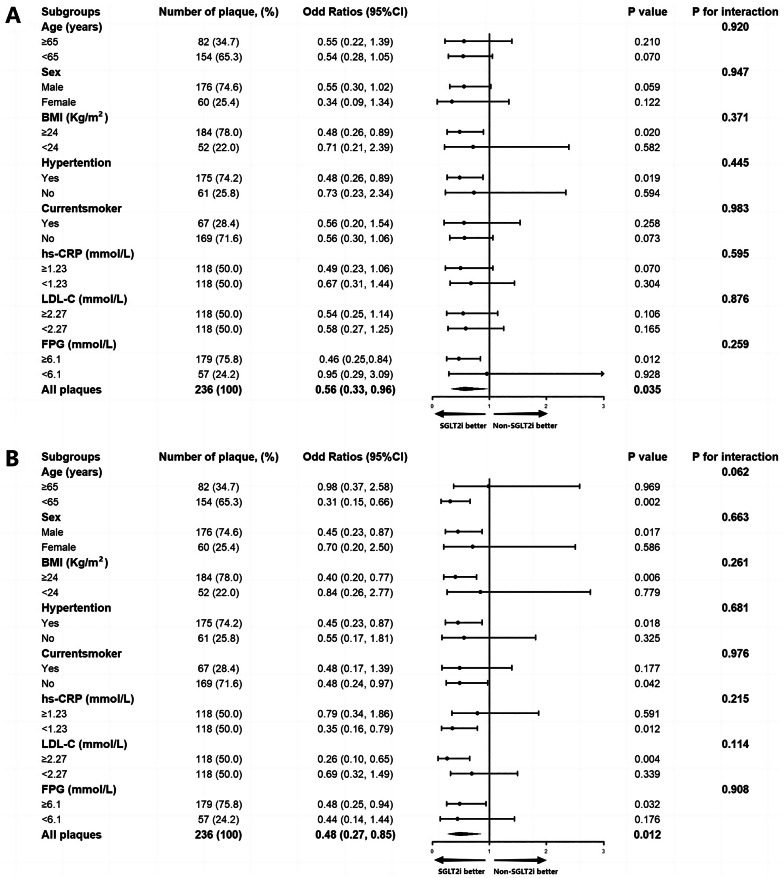
To further elucidate the effect of SGLT2i on the progression of overall and non-calcified plaque, subgroup analysis of the PSM cohort was conducted based on age (< 65 or ≥ 65 years), sex (male or female), body mass index (< 24 or ≥ 24 kg/m2), hypertension (with or without), current smoker (yes or no), high-sensitivity CRP level (< 1.23 or ≥ 1.23 mmol/L), LDL-C (< 2.27 or ≥ 2.27 mmol/L), and FPG (< 6.1 or ≥ 6.1 mmol/L). After adjusting for baseline overall PV or PAV, no significant interactions between SGLT2i and overall PV or PAV were observed among subgroups, except for those with age < 65 favoring SGLT2i therapy (Figure S2). However, no significant interactions were detected among all subgroups between SGLT2i and non-calcified PV or PAV (all p for interaction > 0.05) (Fig. 4).
Fig. 4.
Subgroup analysis for the effect of SGLT2i on the progression of non-calcified PV and PAV. Subgroup analysis of the propensity score matching cohort for the effect of SGLT2i on the progression of non-calcified plaque volume (A) and percent atheroma volume (B). The black vertical solid line represents the OR value of 1. The subgroup analysis was adjusted for baseline non-calcified plaque volume or percent atheroma volume. PV plaque volume, PAV percent atheroma volume
Discussion
The main findings of this longitudinal CCTA cohort study were as follows: (1) SGLT2i therapy significantly reduced overall PV, primarily driven by a marked reduction of non-calcified PV, after following up for 14.6 months in median. (2) These findings were consistent in the PSM analysis between the SGLT2i and non-SGLT2i groups, in a sensitivity analysis with calculation of PAV and annualized change in PV, and in a multivariate model with adjustment of baseline PV or PAV. (3) Moreover, SGLT2i-treated plaques showed less increase in calcified PV and PAV compared to non-SGLT2i-treated plaques. (4) The effects of SGLT2i on optimal plaque remodeling were independent of age, sex, multiple cardiovascular risk factors, and co-existing medications. (5) However, the benefits of SGLT2i on plaque modification did not result in reductions of plaque-related anatomic and hemodynamic stenosis.
Modification of atherosclerotic plaque
Growing evidence suggests that adverse cardiac events in patients with ASCVD are closely related to the atherosclerotic plaque itself rather than plaque-related anatomic and hemodynamic stenosis, known as the plaque hypothesis [24]. Therefore, stabilizing and regressing atherosclerotic plaque are primary objectives in ASCVD management. Previous trials have shown promising results in delaying plaque development using statins, evolocumab, low-dose colchicine, and icosapent ethyl [25–28]. However, the effect of SGLT2i on plaque progression among uncontrolled T2DM patients with ASCVD, where diabetes accelerates plaque progression, particularly in the non-calcified component, remains unclear [29].
SGLT2i regresses atherosclerosis
A multicenter, randomized, open-label trial (Using Tofogliflozin for Possible better Intervention against Atherosclerosis for type 2 diabetes patients [UTOPIA]trial) evaluated the effect of tofogliflozin on preventing the development of carotid atherosclerosis among Japanese T2DM patients. Results indicated a significant reduction in carotid intima-media thickness (IMT) measured by ultrasonography at 2.2 and 4.3-year follow-ups, respectively. However, this reduction was not significantly different from conventional therapy [30, 31]. In contrast, our study comprehensively evaluated coronary plaque characteristics using high-resolution CCTA, which was more repeatable and less operator-dependent than ultrasonography. During a median follow-up of 14.6 months, we observed that traditional antidiabetic and antilipidemic therapies were not associated with decreased PV, whereas SGLT2i significantly reduced PV, particularly in SGLT2i-treated plaques, compared to non-SGLT2i-treated plaques. The differential effects of SGLT2i on plaque regression between our and UTOPIA trials may be attributed to the patient population, with our study comprising two-thirds of patients with established ASCVD. Additionally, CCTA provides more accurate and repeatable measurements of plaque features compared to ultrasonography. Importantly, we have systematically controlled the process of serial CCTA acquisition, and inter- and intra-observer variabilities of CCTA quantitative analysis were optimal.
SGLT2i modifies plaque composition
In a sub-analysis of the UTOPIA trial, neither tofogliflozin nor conventional medications reduced the ultrasonic gray-scale median of carotid atherosclerosis, which comprises lipids, inflammatory infiltrations, and/or hemorrhages [32]. However, our study demonstrated a marked decrease in overall PV, predominantly driven by non-calcified PV, a component associated with adverse cardiovascular events [33]. Recent data from the Scottish Computed Tomography of the Heart (SCOT-HEART) trial highlighted the prognostic significance of low-attenuation non-calcified PV [34]. Although the reduction of low-attenuation PV by SGLT2i was not statistically significant, it might be masked by moderate-intensity statin therapy, which was prescribed to each patient of the current study. In line with this hypothesis, calcified PV consistently increased independently of SGLT2i treatment [25].
To further investigate the effect of SGLT2i on plaque composition and vulnerability, intracoronary optical coherence tomography, with image resolution of ~ 10 μm, was used. Sardu et al. investigated 369 T2DM patients with multivessel non-obstructive coronary stenosis and found that SGLT2i treatment was associated with a lower burden of lipid and macrophage-rich plaque and a thicker minimal fibrous cap at a 1-year follow-up [35]. Similarly, among 109 T2DM patients with co-existing acute coronary syndrome, Kurozumi et al. demonstrated that 6-month treatment with SGLT2i significantly improved the thickness of fibrous cap and reduced the total lipid arc [36]. These findings suggest that SGLT2i may decrease the vulnerable components of plaque and enhance plaque stabilization.
Insights from pre-clinical studies
In the nature history of atherosclerotic plaque progression, monocyte-macrophage axis and relevant inflammatory pathways play a vital role. Dapagliflozin and Ipragliflozin were found to suppress macrophage polarization and macrophage foam cell formation in streptozotocin-induced diabetic ApoE−/− mice [37]. In ApoE−/− mice fed a western diet, Empagliflozin or Canagliflozin reduced aortic arch plaque and levels of pro-inflammatory cytokines, such as tumor necrosis factor-α, interleukin-6, monocyte chemoattractant protein-1, and vascular cell adhesion molecule-1 in the circulation or plaque [15, 16]. Empagliflozin reduced CD68+ macrophages and lipid content, while increased collagen content in the atherosclerotic plaque of diabetic mice [38]. Similarly, Dapagliflozin treatment in a dedicated vulnerable plaque model of ApoE−/− mice induced collagen accumulation and fibrosis, increased cap-to-plaque height ratio, and elevated nicotinamide adenine dinucleotide oxidase 4 expression, indicating improved plaque stability [17].
Meanwhile, SGLT2i also reduced atheroma burden and lipid accumulation accompanied by suppression of Toll-like receptor 4/nuclear factor-kappa B signaling pathway, and their downstream inflammatory effectors in normoglycemic rabbit model [39]. Moreover, SGLT2i was also proved to reduce leukocyte adhesion and endothelial dysfunction that may contribute to plaque regression [38, 40].
Besides the benefits of SGLT2i on non-calcified plaque composition, it might reduce vascular calcification. Canagliflozin or Empagliflozin treatment prevented aortic calcification in mice through downregulating the expression of nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3 signaling pathway [41, 42]. In addition, Dapagliflozin reduced vascular calcification through blocking endoplasmic reticulum stress-dependent thioredoxin domain containing 5 upregulation and promoting subsequent runt-related transcription factor-2 proteasomal degradation [43].
Study limitations
There were certain limitations in this study. Firstly, as a single-center study with a relatively small sample size, its generalizability may be limited. However, the study might provide valuable insights into the real-world use of SGLT2i among high-risk patients with T2DM in a tertiary hospital setting in China. Secondly, although we have performed thoughtful analysis to robustly ascertain the main findings, unadjusted variables may still exist, potentially affecting the link between SGLT2i and plaque regression (e.g., blood glucose control, other diabetes medications). Meanwhile, the retrospective nature of the study precludes establishing causality between SGLT2i and plaque regression, necessitating confirmation through dedicated prospective studies. Therefore, while the study generates hypotheses, caution should be exercised in generalizing the findings to other racial or ethnic populations. Thirdly, serial follow-up lab indices (e.g., hemoglobin A1c) or blood samples were lacking, precluding us to further robust and investigate mechanism of SGLT2i treatment effects. Lastly, a more comprehensive CCTA assessment of coronary plaque using advanced post-processing techniques is warranted, encompassing high-risk features, pericoronary inflammation, and biomechanical characteristics.
Conclusions
In this longitudinal CCTA cohort study involving T2DM patients, SGLT2i therapy significantly regressed coronary overall PV and PAV, primarily driven by a marked reduction in the non-calcified plaque component. These findings offer insights into the potential mechanisms underlying the observed cardioprotective effects of SGLT2i in previous CVOTs.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
None.
Funding
This study was supported by the grant from National Key Research and Development Program of China (2022YFC3602500).
Abbreviations
- ASCVD
Atherosclerotic cardiovascular disease
- ApoE−/−
Apolipoprotein E knockout
- CVOT
Cardiovascular outcome trials
- CI
Confidence intervals
- CCTA
Coronary computed tomography angiography
- HU
Hounsfield unit
- IQR
Interquartile ranges
- IMT
Intima-media thickness
- ORs
Odd ratios
- MI
Myocardial infarction
- PV
Plaque volume
- PAV
Percent atheroma volume
- PSM
Propensity score matching
- SGLT2i
Sodium–Glucose Cotransporter-2 Inhibitor
- T2DM
Type 2 diabetes mellitus
- UTOPIA trial
Using Tofogliflozin for Possible better Intervention against Atherosclerosis for type 2 diabetes patients trial
Author contributions
THZ, XLG, TLC, HKZ, XMZ and YX contributed to data collection and data analysis; THZ, YD and DMS contributed to manuscript writing; LX, YD and YJZ made contributions to study design and intellectual direction.All the authors have read and approved the final manuscript for publication.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
The study protocol was in accordance with the Declaration of Helsinki and was approved by the Medical Ethics Committee of Beijing Anzhen Hospital. Informed consent was obtained from all study participants.
Consent for publication
If the manuscript is accepted, we approve it for publication in Cardiovascular Diabetology.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yu Du, Email: duyupla0604@outlook.com.
Lei Xu, Email: leixu2001@hotmail.com.
Yujie Zhou, Email: azzyj12@163.com.
References
- 1.Magliano DJ, Boyko EJDA. IDF Diabetes Atlas. Brussels: International Diabetes Federation; 2021. [Google Scholar]
- 2.Kalofoutis C, Piperi C, Kalofoutis A, Harris F, Phoenix D, Singh J. Type II diabetes mellitus and cardiovascular risk factors: current therapeutic approaches. Exp Clin Cardiol. 2007;12(1):17–28. [PMC free article] [PubMed] [Google Scholar]
- 3.Carracher AM, Marathe PH, Close KL. International diabetes federation 2017. J Diabetes. 2018;10(5):353–6. 10.1111/1753-0407.12644 [DOI] [PubMed] [Google Scholar]
- 4.Chawla A, Chawla R, Jaggi S. Microvasular and macrovascular complications in diabetes mellitus: distinct or continuum? Indian J Endocrinol Metab. 2016;20(4):546–51. 10.4103/2230-8210.183480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Holman RR, Sourij H, Califf RM. Cardiovascular outcome trials of glucose-lowering drugs or strategies in type 2 diabetes. Lancet. 2014;383(9933):2008–17. 10.1016/S0140-6736(14)60794-7 [DOI] [PubMed] [Google Scholar]
- 6.Thrasher J. Pharmacologic management of type 2 diabetes mellitus: available therapies. Am J Cardiol. 2017;120(1S):S4–16. 10.1016/j.amjcard.2017.05.009 [DOI] [PubMed] [Google Scholar]
- 7.Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, Mattheus M, Devins T, Johansen OE, Woerle HJ, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. New Engl J Med. 2015;373(22):2117–28. 10.1056/NEJMoa1504720 [DOI] [PubMed] [Google Scholar]
- 8.Bhatt DL, Szarek M, Pitt B, Cannon CP, Leiter LA, McGuire DK, Lewis JB, Riddle MC, Inzucchi SE, Kosiborod MN, et al. Sotagliflozin in patients with diabetes and chronic kidney disease. New Engl J Med. 2021;384(2):129–39. 10.1056/NEJMoa2030186 [DOI] [PubMed] [Google Scholar]
- 9.Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR. Canagliflozin and cardiovascular and renal events in type 2 diabetes. New Engl J Med. 2017;377(7):644–57. 10.1056/NEJMoa1611925 [DOI] [PubMed] [Google Scholar]
- 10.Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. New Engl J Med. 2019;380(4):347–57. 10.1056/NEJMoa1812389 [DOI] [PubMed] [Google Scholar]
- 11.Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink H, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull S, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. New Engl J Med. 2019;380(24):2295–306. 10.1056/NEJMoa1811744 [DOI] [PubMed] [Google Scholar]
- 12.Cannon CP, Pratley R, Dagogo-Jack S, Mancuso J, Huyck S, Masiukiewicz U, Charbonnel B, Frederich R, Gallo S, Cosentino F, et al. Cardiovascular outcomes with Ertugliflozin in Type 2 diabetes. New Engl J Med. 2020;383(15):1425–35. 10.1056/NEJMoa2004967 [DOI] [PubMed] [Google Scholar]
- 13.McGuire DK, Shih WJ, Cosentino F, Charbonnel B, Cherney D, Dagogo-Jack S, Pratley R, Greenberg M, Wang S, Huyck S, et al. Association of SGLT2 inhibitors with cardiovascular and kidney outcomes in patients with type 2 diabetes: a Meta-analysis. JAMA Cardiol. 2021;6(2):148–58. 10.1001/jamacardio.2020.4511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marx N, Federici M, Schutt K, Muller-Wieland D, Ajjan RA, Antunes MJ, Christodorescu RM, Crawford C, Di Angelantonio E, Eliasson B, et al. 2023 ESC guidelines for the management of cardiovascular disease in patients with diabetes. Eur Heart J. 2023;44(39):4043–140. 10.1093/eurheartj/ehad192 [DOI] [PubMed] [Google Scholar]
- 15.Han JH, Oh TJ, Lee G, Maeng HJ, Lee DH, Kim KM, Choi SH, Jang HC, Lee HS, Park KS, et al. The beneficial effects of empagliflozin, an SGLT2 inhibitor, on atherosclerosis in ApoE (−/−) mice fed a western diet. Diabetologia. 2017;60(2):364–76. 10.1007/s00125-016-4158-2 [DOI] [PubMed] [Google Scholar]
- 16.Nasiri-Ansari N, Dimitriadis GK, Agrogiannis G, Perrea D, Kostakis ID, Kaltsas G, Papavassiliou AG, Randeva HS, Kassi E. Canagliflozin attenuates the progression of atherosclerosis and inflammation process in APOE knockout mice. Cardiovasc Diabetol. 2018;17(1):106. 10.1186/s12933-018-0749-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen YC, Jandeleit-Dahm K, Peter K. Sodium–glucose co-transporter 2 (SGLT2) inhibitor dapagliflozin stabilizes diabetes-induced atherosclerotic plaque instability. J Am Heart Assoc. 2022;11(1):e022761. 10.1161/JAHA.121.022761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van Rosendael SE, van den Hoogen IJ, Lin FY, Andreini D, Al-Mallah MH, Budoff MJ, Cademartiri F, Chinnaiyan K, Choi JH, Conte E, et al. Clinical and coronary plaque predictors of atherosclerotic nonresponse to statin therapy. Jacc-Cardiovasc Imag. 2023;16(4):495–504. 10.1016/j.jcmg.2022.10.017 [DOI] [PubMed] [Google Scholar]
- 19.Abbara S, Blanke P, Maroules CD, Cheezum M, Choi AD, Han BK, Marwan M, Naoum C, Norgaard BL, Rubinshtein R, et al. SCCT guidelines for the performance and acquisition of coronary computed tomographic angiography: a report of the society of Cardiovascular computed Tomography Guidelines Committee: endorsed by the North American Society for Cardiovascular Imaging (NASCI). J Cardiovasc Comput. 2016;10(6):435–49. [DOI] [PubMed] [Google Scholar]
- 20.Guo B, Jiang M, Guo X, Tang C, Zhong J, Lu M, Liu C, Zhang X, Qiao H, Zhou F et al. Diagnostic and prognostic performance of artificial intelligence-based fully-automated on-site CT-FFR in patients with CAD. Sci Bull 2024;69:1472 [DOI] [PubMed]
- 21.Shaw LJ, Blankstein R, Bax JJ, Ferencik M, Bittencourt MS, Min JK, Berman DS, Leipsic J, Villines TC, Dey D, et al. Society of cardiovascular computed tomography/North American Society of cardiovascular imaging—expert consensus document on coronary CT imaging of atherosclerotic plaque. J Cardiovasc Comput. 2021;15(2):93–109. [DOI] [PubMed] [Google Scholar]
- 22.Dey D, Schepis T, Marwan M, Slomka PJ, Berman DS, Achenbach S. Automated three-dimensional quantification of noncalcified coronary plaque from coronary CT angiography: comparison with intravascular US. Radiology. 2010;257(2):516–22. 10.1148/radiol.10100681 [DOI] [PubMed] [Google Scholar]
- 23.Sheahan M, Ma X, Paik D, Obuchowski NA, St PS, Newman WR, Rae G, Perlman ES, Rosol M, Keith JJ, et al. Atherosclerotic plaque tissue: noninvasive quantitative assessment of characteristics with Software-aided measurements from conventional CT angiography. Radiology. 2018;286(2):622–31. 10.1148/radiol.2017170127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stone PH, Libby P, Boden WE. Fundamental pathobiology of coronary atherosclerosis and clinical implications for chronic ischemic heart Disease Management-the Plaque hypothesis: a narrative review. JAMA Cardiol. 2023;8(2):192–201. 10.1001/jamacardio.2022.3926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van Rosendael AR, van den Hoogen IJ, Gianni U, Ma X, Tantawy SW, Bax AM, Lu Y, Andreini D, Al-Mallah MH, Budoff MJ, et al. Association of Statin Treatment with Progression of Coronary atherosclerotic plaque composition. JAMA Cardiol. 2021;6(11):1257–66. 10.1001/jamacardio.2021.3055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hirai K, Imamura S, Hirai A, Ookawara S, Morishita Y. Effect of Evolocumab on vulnerable coronary plaques: a serial coronary computed Tomography Angiography Study. J Clin Med 2020, 9(10):3338 [DOI] [PMC free article] [PubMed]
- 27.Vaidya K, Arnott C, Martinez GJ, Ng B, McCormack S, Sullivan DR, Celermajer DS, Patel S. Colchicine therapy and plaque stabilization in patients with acute coronary syndrome: a CT coronary angiography study. JACC-Cardiovasc Imag. 2018;11(2 Pt 2):305–16. 10.1016/j.jcmg.2017.08.013 [DOI] [PubMed] [Google Scholar]
- 28.Budoff MJ, Bhatt DL, Kinninger A, Lakshmanan S, Muhlestein JB, Le VT, May HT, Shaikh K, Shekar C, Roy SK, et al. Effect of icosapent ethyl on progression of coronary atherosclerosis in patients with elevated triglycerides on statin therapy: final results of the EVAPORATE trial. Eur Heart J. 2020;41(40):3925–32. 10.1093/eurheartj/ehaa652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Won KB, Lee SE, Lee BK, Park HB, Heo R, Rizvi A, Lin FY, Kumar A, Hadamitzky M, Kim YJ, et al. Longitudinal assessment of coronary plaque volume change related to glycemic status using serial coronary computed tomography angiography: a PARADIGM (progression of AtheRosclerotic PlAque DetermIned by Computed TomoGraphic Angiography Imaging) substudy. J Cardiovasc Comput. 2019;13(2):142–7. [DOI] [PubMed] [Google Scholar]
- 30.Katakami N, Mita T, Yoshii H, Shiraiwa T, Yasuda T, Okada Y, Torimoto K, Umayahara Y, Kaneto H, Osonoi T, et al. Tofogliflozin does not delay progression of carotid atherosclerosis in patients with type 2 diabetes: a prospective, randomized, open-label, parallel-group comparative study. Cardiovasc Diabetol. 2020;19(1):110. 10.1186/s12933-020-01079-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Katakami N, Mita T, Yoshii H, Shiraiwa T, Yasuda T, Okada Y, Kurozumi A, Hatazaki M, Kaneto H, Osonoi T, et al. Tofogliflozin long-term effects on atherosclerosis progression and major clinical parameters in patients with type 2 diabetes mellitus lacking a history of cardiovascular disease: a 2-year extension study of the UTOPIA trial. Cardiovasc Diabetol. 2023;22(1):143. 10.1186/s12933-023-01879-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Katakami N, Mita T, Maeda N, Sato Y, Watada H, Shimomura I. Evaluation of the effect of tofogliflozin on the tissue characteristics of the carotid wall-a sub-analysis of the UTOPIA trial. Cardiovasc Diabetol. 2022;21(1):19. 10.1186/s12933-022-01451-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Thomsen C, Abdulla J. Characteristics of high-risk coronary plaques identified by computed tomographic angiography and associated prognosis: a systematic review and meta-analysis. Eur Heart J-CARD Img. 2016;17(2):120–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Williams MC, Kwiecinski J, Doris M, McElhinney P, D’Souza MS, Cadet S, Adamson PD, Moss AJ, Alam S, Hunter A, et al. Low-attenuation Noncalcified Plaque on Coronary computed Tomography Angiography predicts myocardial infarction: results from the Multicenter SCOT-HEART Trial (Scottish Computed Tomography of the HEART). Circulation. 2020;141(18):1452–62. 10.1161/CIRCULATIONAHA.119.044720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sardu C, Trotta MC, Sasso FC, Sacra C, Carpinella G, Mauro C, Minicucci F, Calabro P, D’ AM, D’ AF, et al. SGLT2-inhibitors effects on the coronary fibrous cap thickness and MACEs in diabetic patients with inducible myocardial ischemia and multi vessels non-obstructive coronary artery stenosis. Cardiovasc Diabetol. 2023;22(1):80. 10.1186/s12933-023-01814-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kurozumi A, Shishido K, Yamashita T, Sato D, Uchida S, Koyama E, Tamaki Y, Hayashi T, Miyashita H, Yokoyama H, et al. Sodium–glucose Cotransporter-2 inhibitors stabilize coronary plaques in acute coronary syndrome with diabetes mellitus. Am J Cardiol. 2024;214:47–54. 10.1016/j.amjcard.2023.12.056 [DOI] [PubMed] [Google Scholar]
- 37.Terasaki M, Hiromura M, Mori Y, Kohashi K, Nagashima M, Kushima H, Watanabe T, Hirano T. Amelioration of hyperglycemia with a sodium–glucose cotransporter 2 inhibitor prevents macrophage-driven atherosclerosis through macrophage foam cell formation suppression in type 1 and type 2 diabetic mice. PLoS ONE. 2015;10(11):e0143396. 10.1371/journal.pone.0143396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pennig J, Scherrer P, Gissler MC, Anto-Michel N, Hoppe N, Funer L, Hardtner C, Stachon P, Wolf D, Hilgendorf I, et al. Glucose lowering by SGLT2-inhibitor empagliflozin accelerates atherosclerosis regression in hyperglycemic STZ-diabetic mice. Sci Rep-UK. 2019;9(1):17937. 10.1038/s41598-019-54224-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lee SG, Lee SJ, Lee JJ, Kim JS, Lee OH, Kim CK, Kim D, Lee YH, Oh J, Park S, et al. Anti-inflammatory effect for atherosclerosis progression by sodium–glucose cotransporter 2 (SGLT-2) inhibitor in a normoglycemic rabbit model. Korean Circ J. 2020;50(5):443–57. 10.4070/kcj.2019.0296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ganbaatar B, Fukuda D, Shinohara M, Yagi S, Kusunose K, Yamada H, Soeki T, Hirata KI, Sata M. Empagliflozin ameliorates endothelial dysfunction and suppresses atherogenesis in diabetic apolipoprotein E-deficient mice. Eur J Pharmacol. 2020;875:173040. 10.1016/j.ejphar.2020.173040 [DOI] [PubMed] [Google Scholar]
- 41.Chen A, Lan Z, Li L, Xie L, Liu X, Yang X, Wang S, Liang Q, Dong Q, Feng L, et al. Sodium–glucose cotransporter 2 inhibitor canagliflozin alleviates vascular calcification through suppression of nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3 inflammasome. Cardiovasc Res. 2023;119(13):2368–81. 10.1093/cvr/cvad119 [DOI] [PubMed] [Google Scholar]
- 42.Li XX, Chen ZD, Sun XJ, Yang YQ, Jin H, Liu NF. Empagliflozin ameliorates vascular calcification in diabetic mice through inhibiting Bhlhe40-dependent NLRP3 inflammasome activation. Acta Pharmacol Sin. 2024;45(4):751–64. 10.1038/s41401-023-01217-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wu S, Luo X, Chen Y, Wang Z, Liu X, Sun N, Zhao J, Luo W, Zhang J, Tong X, et al. Sodium–glucose cotransporter 2 inhibitors attenuate vascular calcification by suppressing endoplasmic reticulum protein thioredoxin domain containing 5 dependent osteogenic reprogramming. Redox Biol. 2024;73:103183. 10.1016/j.redox.2024.103183 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.