Neonatal alloimmune thrombocytopenia—caused by movement of maternal alloantibodies directed against fetal platelet antigens across the placenta—is the most common cause of severe neonatal thrombocytopenia.1 These alloantibodies cause thrombocytopenia, which may result in intracranial haemorrhage.2,3 Most cases arise unexpectedly, and prompt diagnosis and treatment are essential to reduce the chances of death and disability caused by the haemorrhaging.4,5 Neonatal alloimmune thrombocytopenia should be strongly suspected in cases of neonatal thrombocytopenia with no evidence of sepsis, systemic disease, or skeletal anomalies associated with thrombocytopenia, and where maternal idiopathic thrombocytopenic pupura has been excluded. We describe two cases with typical clinical presentations in which standard methods for detection of maternal antibodies gave negative results, and we show that empirical treatment with human platelet antigen 1a negative platelets yielded successful outcomes in both cases.
Case 1
A two day old girl presented with several left sided and secondary generalised tonic-clonic seizures. She had been delivered normally at full term, after an uneventful gestation. Neurological examination between seizures had normal results, and otherwise she was completely well.
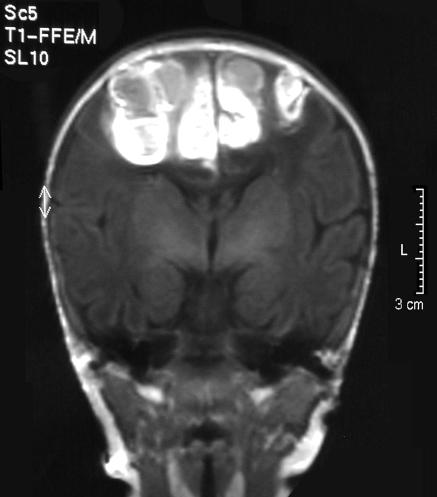
Her platelet count was 5×109/l, haemoglobin concentration was 158 g/l, and white cell count was 16.7×109/l. A magnetic resonance scan showed fresh extensive haemorrhage in the interhemispheric fissure and sulci (fig 1). The brain and ventricles were normal. Viral swabs and culture showed no infectious cause for the thrombocytopenia. The baby's mother had a normal platelet count.
Fig 1.
Magnetic resonance image showing fresh extensive haemorrhage in the interhemispheric fissure and sulci (case 1)
On the basis of these findings, we suspected neonatal alloimmune thrombocytopenia. Maternal antibodies specific to the platelets were not detected by the indirect platelet immunofluorescence test,6 monoclonal antigen immobilisation of platelet antigen assay,7 or a GTI PAK-12 antigen capture enzyme linked immunosorbent assay (ELISA). Maternal plasma and paternal platelets did not cross match.
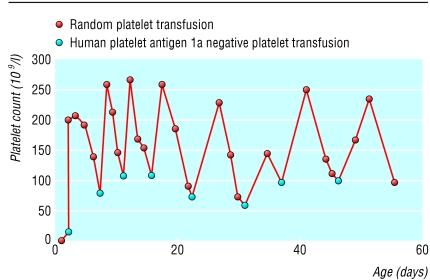
We gave the baby phenobarbital, a random platelet transfusion, and intravenous immunoglobulin (400 mg/kg). We saw no increase in platelets and tried giving human platelet antigen 1a negative platelets the next day, and the platelet count increased (fig 2). Eight more transfusions of these antigen 1a negative platelets were given, approximately weekly, until the platelet count remained greater than 100×109/l without transfusion. We stopped giving the baby phenobarbital. Subsequently, the baby developed normally and had no neurodevelopmental problems at 18 months.
Fig 2.
Response to platelet transfusions (case 1)
Investigation for human platelet antigen 1a confirmed that the mother was negative and the father was positive. We did another monoclonal antigen immobilisation of platelet antigen assay with an increased serum to cell ratio (Porcelijn et al, 3rd European conference on platelet and granulocyte immunobiology, Cambridge, 1994), and antihuman platelet antigen 1a antibody was detectable using this technique.
Case 2
During the mother's next pregnancy, neonatal alloimmune thrombocytopenia again affected the fetus, with antihuman platelet antigen 1a antibodies detectable with only the modified assay technique. This baby was managed by fetal blood sampling and intrauterine transfusions, followed by five postnatal transfusions of platelets negative to human platelet antigen 1a; treatment was successful. The platelet count of the baby was stable without transfusions five weeks after birth.
Case 3
A newborn boy delivered at full term by ventouse extraction had a widespread petechial rash and a cephalhaematoma. He was otherwise well. His platelet count was 13×109/l, haemoglobin concentration 175 g/l, and white cell count 24.2×109/l. We found no obvious cause, either clinically or on laboratory testing, for thrombocytopenia. The mother's platelet count was normal. Ultrasound examination showed no evidence of an intracranial haemorrhage. Neonatal alloimmune thrombocytopenia was suspected and we gave the baby one unit of platelets negative for human platelet antigen 1a, which resulted in a platelet count of 103×109/l after transfusion. We did not detect antihuman platelet antigen 1a antibodies in the maternal plasma.
One week later, the baby bled from his umbilical stump. The platelet count was 20×109/l. The mother was human platelet antigen 1a negative and the father human platelet antigen 1a positive: the most likely diagnosis was neonatal alloimmune thrombocytopenia. We gave the baby two units of platelets negative for human platelet antigen 1a resulting in a platelet count of 112×109/l after transfusion. Further bleeding 11 days later was associated with a platelet count of 18×109/l. He received another unit of human platelet antigen 1a negative platelets, achieving a platelet count of 163×109/l. Further transfusions were unnecessary. Four weeks after the birth of the baby, we detected antihuman platelet antigen 1a antibodies in the mother's plasma by a standard monoclonal antigen immobilisation assay for platelet antigen.
Discussion
These cases emphasise the need for prompt treatment of neonatal alloimmune thrombocytopenia. The condition should be suspected in any baby presenting with thrombocytopenia or haemorrhage in the absence of systemic disease. The National Blood Service provides platelets negative for human platelet antigen 1a and 5b for the immediate treatment of neonates with thrombocytopenia suspected of having or known to have alloimmunity.
Neonatal alloimmune thrombocytopenia is the commonest cause of severe thrombocytopenia in the first few days of life,8 resulting in 400-600 cases in the United Kingdom each year. Delays in treating the condition are mainly because of delays in diagnosis,9 which can lead to death or lasting disability in an affected baby.
The pathogenesis of neonatal alloimmune thrombocytopenia is similar to that of haemolytic disease of a newborn baby. The mother is negative for an allotype of a platelet antigen that the fetus has inherited from the father. The parents are incompatible for human platelet antigens 1a or 5b in 90% of cases.2 Placental transfer of IgG alloantibodies may result in considerable fetal thrombocytopenia as early as 16 weeks' gestation.4 Although 2.5% of white people are negative for human platelet antigen 1a, the incidence of 1a negativity in white populations is about 1 in 1500 live births.2,10 Alloimmunisation to human platelet antigen 1a is strongly associated with HLADRw52a (HLADR3*0101),11 which is carried by a third of people in the United Kingdom. However, alloimmunisation does not always result in neonatal thrombocytopenia—the cause may be other, as yet unidentified, factors.
Diagnostic tests for neonatal alloimmune thrombocytopenia usually include detection of maternal antibody together with finding out whether the mother and the father have the respective platelet antigens. Maternal platelet antibodies are usually detectable against paternal platelets. However, in up to 30% of cases no antibody is found.12 Occasionally, increasing the volumes of maternal serum in the monoclonal antigen immobilisation of platelet antigen assay or acid elution of the antibody may detect antibodies.13 Moreover, unlike haemolytic disease of newborn babies, antibody titres and isotypes are not associated with severity of disease.14
In these cases, maternal platelet specific antibodies were not detectable at presentation, but were later found, justifying the clinical decision to treat empirically with antigen negative platelets. In most cases of neonatal alloimmune thrombocytopenia, platelet numbers should be normal at age 2 weeks.
Neonatal alloimmune thrombocytopenia may occur without detectable maternal alloantibodies and demands prompt treatment
We thank Robin Roberts-Gant for help preparing the figures.
Contributors: All authors prepared this manuscript. JH and DJR managed the clinical care of case 1 and sibling; MFM gave clinical advice in case one sibling; JB and RR obtained clinical data on the sibling; HY managed the clinical care of case 2 and RR obtained data. DA and MFM were responsible for laboratory testing. DJR edited the paper and is guarantor.
Funding: DJR is a Howard Hughes international research scholar.
Competing interests: None declared.
References
- 1.Moulinier J. Alloimmunisation maternelle antiplaquettaire duzo. Proceedings of the 6th congress of the European Society of Hematology. Paris: European Society of Hematology, 1953: 817-20.
- 2.Mueller-Eckhardt C, Kiefel V, Grubert A, Kroll H, Weisheit M, Schmidt S, et al. 348 cases of fetal alloimmune thrombocytopenia. Lancet 1989;1: 363-6. [DOI] [PubMed] [Google Scholar]
- 3.Zalneraitis EL, Young TSK, Krishnamoorthy KS. Intracranial haemorrhage in utero as a complication of isoimmune thrombocytopenia. J Pediatr 1979;95: 611-4. [DOI] [PubMed] [Google Scholar]
- 4.Blanchette VS, Johnson J, Rand M. The management of alloimmune neonatal thrombocytopenia. Bailliere's Clin Haematol 2000;13: 365-90. [DOI] [PubMed] [Google Scholar]
- 5.Ouwehand W, Smith G, Ranasinghe E. Management of severe alloimmune thrombocytopenia in the newborn. Arch Dis Child Fetal Neonatal Ed 2000;82: 173-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Von dem Borne AEGK, Verheught FWA, Oosterhof F, von Riesz E, Brutel de la Riviere A, Engelfriet CP. A simple immunofluorescence test for the detection of platelet antibodies. Br J Haematol 1978;39: 195-207. [DOI] [PubMed] [Google Scholar]
- 7.Kiefel V, Santoso S, Weisheit M, Mueller-Eckhardt C. Monoclonal antibody-specific immobilisation of platelet antigens (MAIPA): a new tool for the identification of platelet-reactive antibodies. Blood 1987;70: 1722-6. [PubMed] [Google Scholar]
- 8.Burrows RF, Kelton MD. Fetal thrombocytopenia and its relation to maternal thrombocytopenia. N Engl J Med 1993;329: 1463-6. [DOI] [PubMed] [Google Scholar]
- 9.Murphy MF, Verjee S, Greaves M. Inadequacies in the postnatal management of fetomaternal alloimmune thrombocytopenia. Br J Haematol 1999;105: 123-6. [PubMed] [Google Scholar]
- 10.Williamson LM, Hackett G, Rennie J, Palmer C, Maciver C, Hadfield R, et al. The natural history of fetomaternal alloimmunisation to the platelet-specific antigen HPA-1a as determined by antenatal screening. Blood 1998;92: 2280-7. [PubMed] [Google Scholar]
- 11.Valentin N, Vergracht A, Bignon JD, Cheneau ML, Blanchard D, Kaplan C, et al. HLA-DRw52a is involved in alloimmunization against PL-A1 antigen. Human Immunol 1990;27: 73-9. [DOI] [PubMed] [Google Scholar]
- 12.Kaplan C, Daffos F, Forestier F, Morel MC, Chesnel N, Tchernia G. Current trends in neonatal alloimmune thrombocytopenia: diagnosis and therapy. In: Kaplan-Gouet C, Schlegel N, Salmon Ch, McGregor J, eds. Platelet immunology: fundamental and clinical aspects. Paris: Colloque INSERM/John Libbey Eurotext, 1991: 267-78.
- 13.Marzusch K, Fischer Dietl J, Wiest E, Volklein K, Schnaid M. Difficulties in the antenatal assessment of neonatal alloimmune thrombocytopenia. Acta Obstet Gynecol Scand 1993;72: 583-5. [DOI] [PubMed] [Google Scholar]
- 14.Prouix C, Filion M, Goldman M, Bradley A, Devine D, Decary F, Chartrand P. Analysis of immunoglobulin class, IgG subclass and titre of HPA-1a antibodies in alloimmunised mothers giving birth to babies with or without neonatal alloimmune thrombocytopenia. Br J Haematol 1994;87: 813-7. [DOI] [PubMed] [Google Scholar]